Published online Jun 15, 2021. doi: 10.4251/wjgo.v13.i6.462
- This article has been corrected.
- See: World J Gastrointest Oncol. May 15, 2022; 14(5): 1065-1066
Peer-review started: February 11, 2021
First decision: March 29, 2021
Revised: April 4, 2021
Accepted: April 20, 2021
Article in press: April 20, 2021
Published online: June 15, 2021
Processing time: 116 Days and 3 Hours
Gastrointestinal stromal tumors can occur in any part of the gastrointestinal tract, but gastric stromal tumors (GSTs) are the most common. All GSTs have the potential to become malignant, and these can be divided into four different grades by risk from low to high: Very low risk, low risk, medium risk, and high risk. Current guidelines all recommend early complete excision of GSTs larger than 2 cm in diameter. However, it is not clear whether small GSTs (sGSTs, i.e., those smaller than 2 cm in diameter) should be treated as early as possible. The National Comprehensive Cancer Network recommends that endoscopic ultrasonography-guided (EUS-guided) fine-needle aspiration biopsy and imaging (computed tomography or magnetic-resonance imaging) be used to assess cancer risk for sGSTs detected by gastroscopy to determine treatment. When EUS indicates a higher risk of tumor, surgical resection is recommended. There are some questions on whether sGSTs also require early treatment. Many studies have shown that endoscopic treatment of GSTs with diameters of 2-5 cm is very effective. We here address whether endoscopic therapy is also suitable for sGSTs. In this paper, we try to explain three questions: (1) Does sGST require treatment? (2) Is digestive endoscopy a safe and effective means of treating sGST? and (3) When sGSTs are at different sites and depths, which endoscopic treatment method is more suitable?
Core Tip: Gastric stromal tumors (GSTs) are all malignant, but generally, the smaller the diameter, the more likely the tumor is inert. However, GSTs smaller than 2 cm in diameter are also at risk of growing and becoming more malignant. Endoscopic treatment of GSTs smaller than 5 cm in diameter is comparable to surgical treatment. Early endoscopic resection is safe and effective when there are high risk factors for GSTs smaller than 2 cm in diameter or the patients cannot be followed regularly, and different endoscopic treatment methods can be selected according to the tumor site and depth.
- Citation: Chen ZM, Peng MS, Wang LS, Xu ZL. Efficacy and safety of endoscopic resection in treatment of small gastric stromal tumors: A state-of-the-art review. World J Gastrointest Oncol 2021; 13(6): 462-471
- URL: https://www.wjgnet.com/1948-5204/full/v13/i6/462.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v13.i6.462
Gastrointestinal stromal tumors (GISTs) originate from the Cajal stromal cells of the gastrointestinal tract[1]. These are the most common tumors arising from the mesenchymal tissue of the digestive tract and can occur in any part of the tract. Studies have shown that the most common GISTs are gastric stromal tumors (GSTs; 55.6%), followed by GISTs originating in the small intestine (31.8%), colon (6%), in other sites or with multiple occurrences (5.5%), and esophagus (only 0.7%)[2]. In China and some other developed Asian countries, the incidence of GIST is 16-22 per million[3,4]. With the advancement and popularization of high-resolution endoscopy, the detection rate of small GST (sGSTs) with diameters of < 2 cm and without obvious symptoms has been significantly improved[5].
Gastroscopy can find submucosal tumors (SMTs) of the stomach. Common gastric SMTs include GSTs, leiomyomas, lipomas, neuroendocrine tumors, granulosa cell tumors, and (relatively rarely) Schwann cell tumors[6]. However, gastroscopy cannot determine whether the lesion might be a GST, nor distinguish it from an extracavitary compressing lesion. At present, it is recognized that endoscopic ultrasonography (EUS) is the most accurate imaging method for evaluating gastric SMTs. As it can distinguish various types of SMTs of the stomach, it plays a very important role in determining tumor location and choosing treatment method[7-9]. Studies have found that EUS is better than computed tomography (CT) or magnetic-resonance imaging (MRI) for SMTs < 2 cm[10,11]. The general manifestation of GSTs under EUS is as follows: The tumor originates from the muscularis propria, except for a small part that originates from the muscularis mucosa. Small tumors often have uniform hypoechoic structures with clear boundaries, while large tumors can show irregular boundaries as well as uniform or uneven internal echo[12,13]. However, gastroscopic and EUS images of sGSTs and leiomyomas are very similar, and pathological immunohistochemical examinations are required to distinguish the two[14].
GISTs tend to be potentially malignant, but there is no absolute distinction between benign and malignant GISTs. According to degree of risk from low to high, GISTs are divided into four grades: Extremely low risk, low risk, intermediate risk, and high risk[15]. The progression of GSTs is currently uncertain; 10%-30% of cases are highly malignant[16]. Studies have found that the larger the diameter of the GST and the greater the mitotic count, the higher the metastasis rate[17]. For GSTs with a diameter > 2 cm, guidelines suggest that the tumors be completely removed early on, resection margin histology should be negative, and tumor rupture should be avoided during surgery. However, how to treat sGST remains controversial at home and abroad[18-20].
Studies have shown[21,22] that sGSTs generally have very low proliferation activity; the tumor cells have inert biological characteristics, especially in micro-GSTs (diameter < 1 cm), which have even lower proliferation activity. A small-sample study showed that the metastatic rate of sGSTs is negligible regardless of mitotic count[18]. However, some studies have shown that sGSTs can also pose a high risk of malignant transformation[23-26], and GIST cells can continue to proliferate, increasing the diameter of the lesion. One study showed that after an average follow-up period of 17.3 mo for sGSTs, the diameter of the lesion increased in 13.0% of patients[27]. A large-sample study of surveillance, epidemiology and end results data found that about 11.4% of sGISTs were accompanied by local progression or even distant metastasis when first diagnosed[28]. Some studies have also found that sGSTs were diagnosed as highly malignant tumors or even with distant metastasis after resection[29-31].
The United States National Comprehensive Cancer Network issued the latest soft-tissue sarcoma guidelines on May 28, 2020[32]. For sGST found by gastroscopy, EUS-guided fine-needle aspiration biopsy (FNAB) and imaging examination (CT or MRI) are recommended to assess the risk posed by the tumor and then determine treatment options. EUS manifestations suggesting a higher risk[33] are irregular edges, cystic change, ulcer formation, hyperechoic foci, and heterogeneity. The National Comprehensive Cancer Network guidelines recommend that patients with such EUS manifestations be treated by surgical resection; if there are no such manifestations, regular EUS or imaging follow-up can be considered, but no specific follow-up surveillance is suggested[32]. Lachter et al[34] retrospectively analyzed 70 cases of GST monitored by EUS and found that sGSTs with diameters of > 17 mm grew easily. Fang et al[35] conducted EUS follow-up with a median time of 24.0 mo and found that sGSTs with diameters > 14 mm were prone to tumor enlargement accompanied by clinical symptoms. Gao et al[26] conducted a retrospective analysis of 69 cases of sGST and found that tumors < 9.5 mm in diameter could be evaluated every 2-3 years, but those ≥ 9.5 mm in diameter should be surveyed every 6-12 mo.
Studies have shown that due to sample size limitations, the diagnosis rate of EUS-FNAB for sGSTs is 71%[36]. Therefore, only pathological evaluation of postoperative specimens can determine the malignant potential of sGSTs. Gastroscopy, EUS, and other examinations required for sGST review are all invasive procedures, and long-term, high-frequency follow-up also imposes heavy economic and psychological burdens on patients. Patients who know there is a lesion with malignant potential in their bodies and who do not undergo treatment are prone to anxiety, irritability, and other negative emotions, which seriously affect their quality of life. By communicating with sGST patients, researchers have found that most of such patient are strongly willing to undergo surgical or endoscopic treatment[37].
GST is almost completely tolerant to traditional radiotherapy and chemotherapy. The main avenue of metastasis is the blood; lymph node metastasis is rare. Therefore, lymph node dissection is generally unnecessary. Surgical treatment should completely remove the tumor and preserve gastric function as much as possible. Extensive gastrectomy cannot improve survival rate; the biological characteristics of GST greatly play to the advantages of minimally invasive endoscopic surgery[38,39].
Digestive endoscopic technology has rapidly developed and been widely popularized. Gastroscopy has significant efficacy in the treatment of GSTs. Digestive endoscopy for the treatment of GSTs with a diameter < 5 cm and no metastasis has the same efficacy as traditional surgery and laparoscopic surgery, with less trauma, shorter operation time, fewer complications, lower treatment costs, and faster recovery[40]. A recent meta-analysis of 12 studies including a total of 1292 patients with sGSTs[41] compared efficacy and safety between endoscopic resection and laparoscopic resection. The results showed that endoscopic operation time was shorter than that of laparoscopic resection; there were no significant differences between the two in intraoperative bleeding, postoperative hospital stay, postoperative exhaust time, or postoperative complication rate; and patients treated by endoscopy were able to resume eating earlier. The expert consensus issued by China in 2018 recommended the following[6]: (1) Lesions with no metastasis or with extremely low risk of metastasis; (2) Possibility of complete resection by endoscopic techniques; and (3) Low residual and recurrence risks are suitable for endoscopic resection. During endoscopic resection, the principle of tumor-free treatment should be followed: The tumor should be completely removed, and the tumor capsule should be intact during resection.
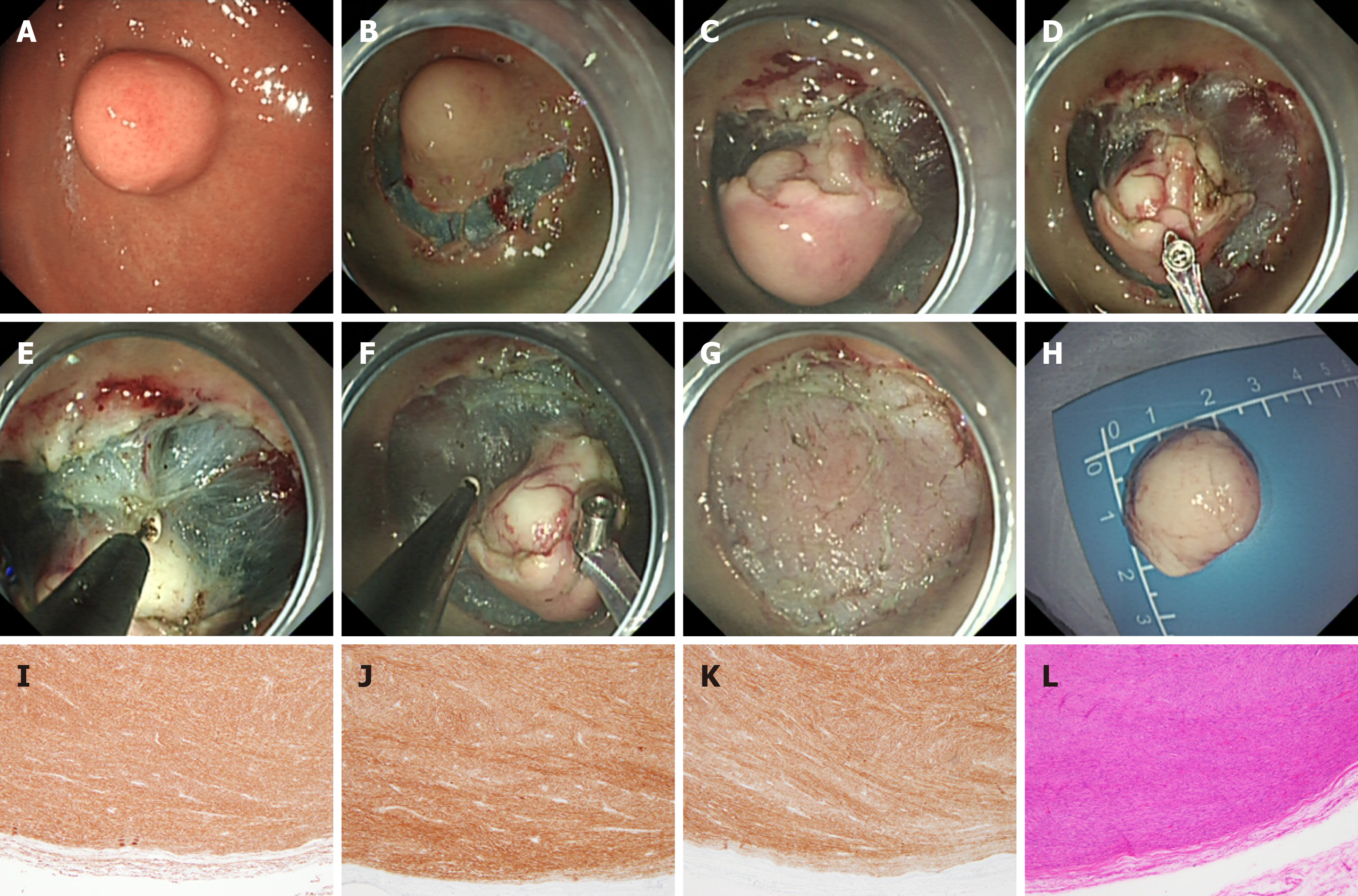
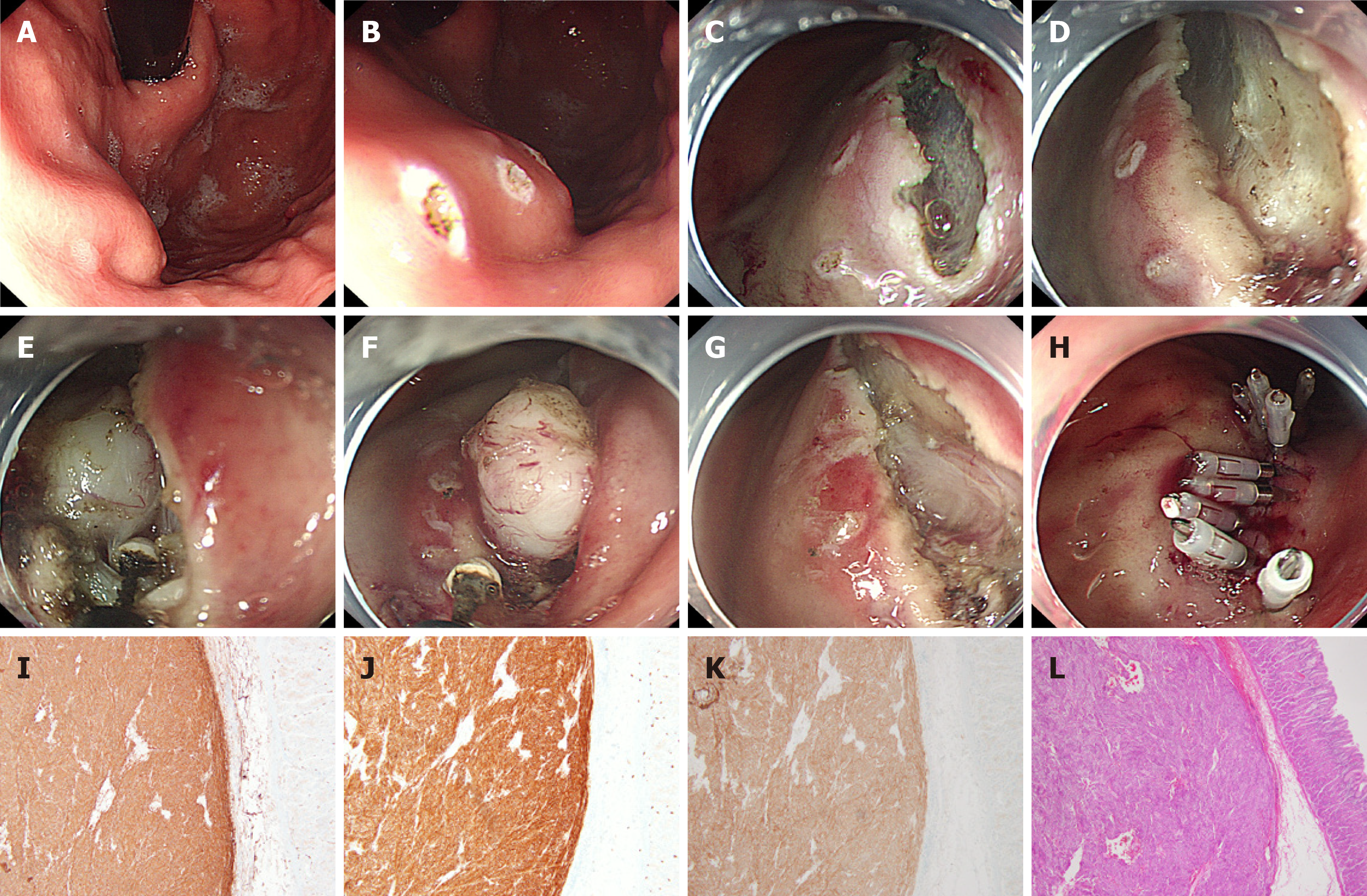
At present, commonly used methods for gastroscopic treatment of sGST include endoscopic submucosal dissection (ESD), endoscopic submucosal excavation (ESE), endoscopic full-thickness resection (EFTR), and submucosal-tunneling endoscopic resection (STER). ESD (Figure 1) is often used to treat GSTs derived from the superficial layer of the muscle propria. A retrospective analysis of 168 patients[42] showed an overall resection rate of ESD in GST of 100%; only two (1.2%) patients had delayed bleeding, and no local recurrence or distant metastasis of the tumor was observed during a follow-up period of 6-67 mo (median, 25 mo). A study comparing ESD and laparoscopic treatment of sGSTs[43] found that ESD could significantly reduce operation time, blood loss, and patient hospital stay and that the two groups did not significantly differ in recurrence rate or survival time after tumor resection. It is reported in the literature[44,45] that the main complications of ESD treatment of GSTs are perforation (0%-8.2%) and bleeding (0%-15.6%). In most patients, bleeding and perforation complications can be controlled through endoscopic treatment. ESE (Figure 2) is a variation of ESD; the main difference is that ESE can excavate sGSTs from the deep layer of the muscularis propria. Jeong et al[46] reported for the first time that ESE used to treat GSTs derived from the muscularis propria has a high complete resection rate and an acceptable complication rate. The complete resection rate of ESE is reported to be 90%-100%; the main complication is perforation, with an incidence rate of 0%-20%, and most cases can be treated under endoscopy[47-50]. Studies have also pointed out that ESE resection of tumors originating from the submucosal muscularis propria would result in incomplete resection of the tumor capsule, leading to residual tumor cells, and that excessive excavation can lead to perforation[5,51]. Studies have also revealed that the complete resection rate in endoscopic treatment of sGSTs (concomitant stromal tumors) is low, especially of micro-GSTs (< 1 cm). Compared with bigger tumors, it is more difficult to remove normal tissues surrounding the capsules of small tumors and to dissect out the tumors themselves[46]. ESD and ESE resection of fundic sGST entails use of the U-type reverse endoscope, which is difficult to operate and can damage tumor capsules during removal of tumors, affecting resection integrity. Even worse, abundant blood vessels around the tumors must be cut off, which can easily lead to intraoperative bleeding, thereby impairing physicians’ vision and operational accuracy, prolonging the operation, and increasing the risk of perforation[52]. ESE is suggested to be suitable for the treatment of sGSTs derived from the muscularis propria and growing into the gastric cavity.
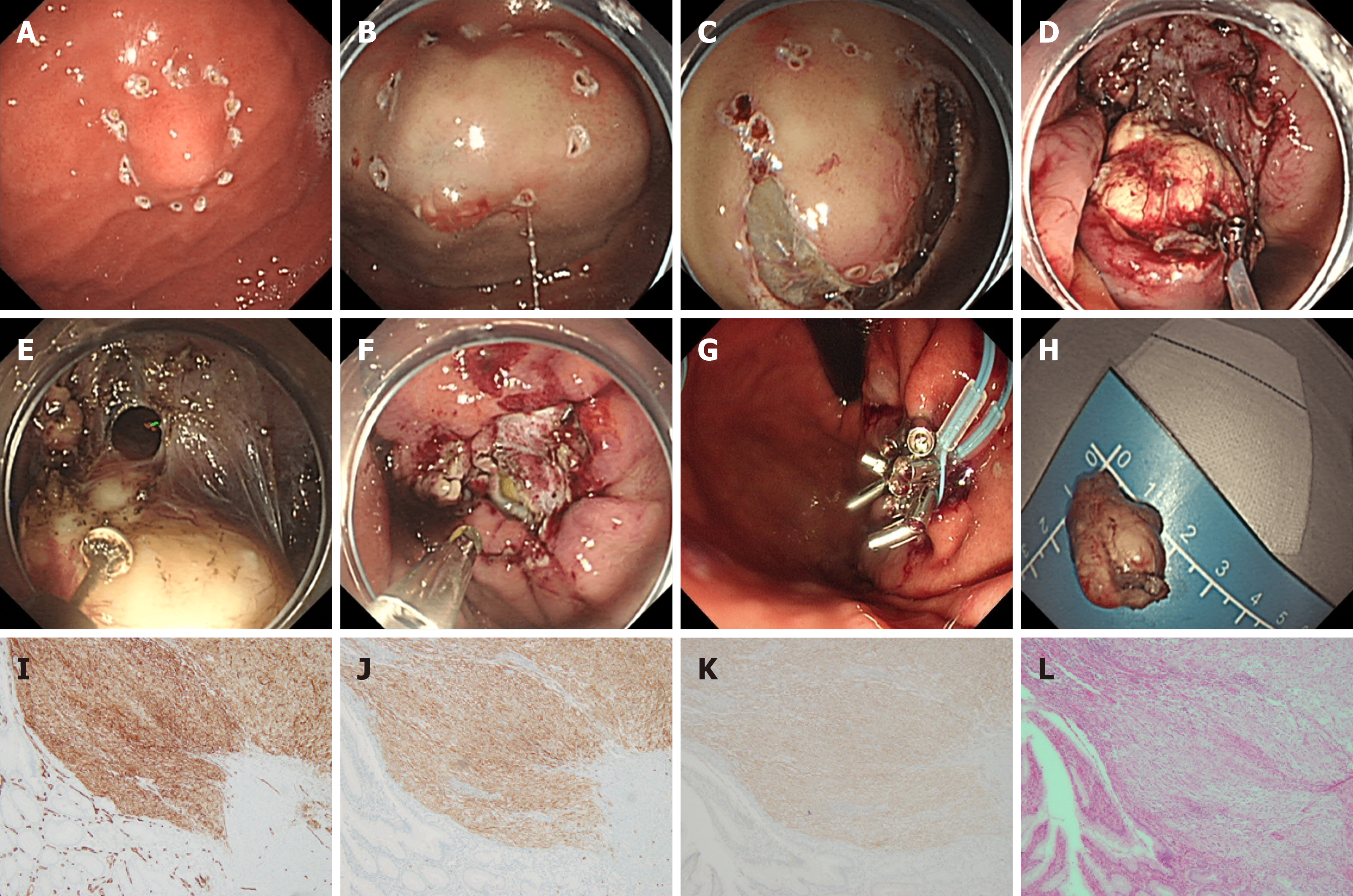
EFTR (Figure 3) can be classified as a special form of ESD/ESE with active rather than passive perforation; the gastric wall is closed after it undergoes a full-thickness resection. Therefore, perforation in EFTR is not considered a complication. EFTR can be used to treat sGSTs with intracavitary and extracavitary growth. Wang et al[53] were the first to perform EFTR in order to treat GST, removing 66 such tumors with diameters of < 3.5 cm. The complete resection rate was 100%. There were five cases of intraoperative bleeding, in all of which bleeding was successfully stopped and the perforation completely closed under endoscopy. Studies have reported that the complete resection rate in EFTR treatment can be as high as 87.5%-100.0%, and the complication rate is low. There are a few reports of abdominal infection after EFTR, which improved after treatment[54,55]. Another study[56] showed that EFTR is equivalent to laparoscopic surgery in the safety and efficacy of resectioning smaller-diameter GSTs, but EFTR can significantly reduce operation time, intraoperative blood loss, and patient hospital stay. It has been reported that the incidence of electrocoagulation syndrome is 3.1% after EFTR; although in such cases the syndrome is less severe than bleeding and perforation, it has a high incidence in the stomach and its symptoms can be similar to those of ordinary postoperative perforation[37]. However, active EFTR perforation to completely remove the tumor is relatively traumatic, and the endoscopist must have a high level of skill to close the perforation fistula; therefore, the procedure is recommended to be completed by a physician experienced in endoscopic treatment. When perforation occurs during endoscopic treatment, the use of metal clips alone or combined with nylon string (the purse string suture) has a good preventive effect against complications[57].
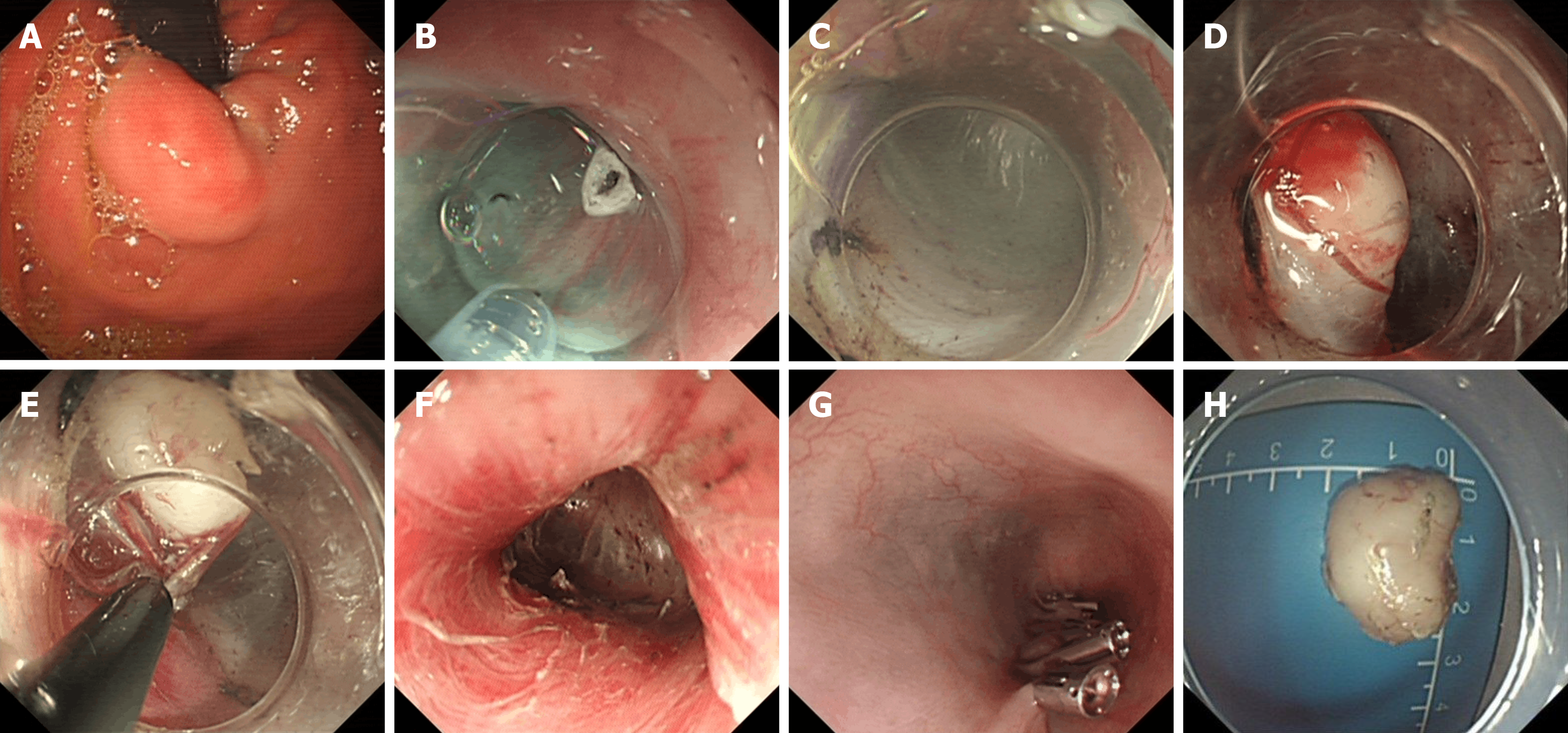
STER (Figure 4) is suitable for the cardia where a tunnel is easy to build, or sGSTs at the fundus of the stomach near the cardia. When sGST is not close to the cardia, it is difficult to establish a tunnel, and therefore the STER technique is not applicable. STER has many advantages, including maintenance of mucosal integrity, small wounds, fast healing, clear operating vision, and reduced risk of pleural and abdominal infections[58]. A clinical study involving 290 cases showed that the overall incidence of complications was high [23.4% (68/290)], but only a small percentage thereof (10.0%) required therapeutic intervention[59]. A study involving 430 cases[60] showed that the complete resection rate of STER was 98.1%, the rate of postoperative gas-related complications was 21.5%, that of inflammation-related complications was 8.4%, and that of delayed bleeding was 2.2%. There were no cases of death or recurrence of tumors related to STER.
At present, there is no consensus on whether sGSTs need treatment. However, if EUS examination determines that the sGST has high-risk manifestations, resection is recommended. Even EUS-FNAB examination cannot completely determine the risk grade of sGST, which tends to be potentially malignant. Although the tumor cells have inert biological characteristics, patients who live with tumors and need long-term endoscopic follow-up have heavy psychological and economic burdens to bear. The efficacy of digestive endoscopy in the treatment of sGSTs is equivalent to that of surgery, with no effect on gastric function, less trauma, lower treatment cost, and shorter hospital stay. For sGST patients with high-risk manifestations or those who cannot tolerate endoscopic follow-up but who actively demand treatment, endoscopic sGST resection by physicians experienced in endoscopic treatment is effective and safe.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Yang CW S-Editor: Zhang H L-Editor: Wang TQ P-Editor: Li JH
| 1. | Kwon JG, Hwang SJ, Hennig GW, Bayguinov Y, McCann C, Chen H, Rossi F, Besmer P, Sanders KM, Ward SM. Changes in the structure and function of ICC networks in ICC hyperplasia and gastrointestinal stromal tumors. Gastroenterology. 2009;136:630-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 2. | Søreide K, Sandvik OM, Søreide JA, Giljaca V, Jureckova A, Bulusu VR. Global epidemiology of gastrointestinal stromal tumours (GIST): A systematic review of population-based cohort studies. Cancer Epidemiol. 2016;40:39-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 579] [Cited by in RCA: 520] [Article Influence: 57.8] [Reference Citation Analysis (1)] |
| 3. | Cho MY, Sohn JH, Kim JM, Kim KM, Park YS, Kim WH, Jung JS, Jung ES, Jin SY, Kang DY, Park JB, Park HS, Choi YD, Sung SH, Kim YB, Kim H, Bae YK, Kang M, Chang HJ, Chae YS, Lee HE, Park DY, Lee YS, Kang YK, Kim HK, Chang HK, Hong SW, Choi YH, Shin O, Gu M, Kim YW, Kim GI, Chang SJ. Current trends in the epidemiological and pathological characteristics of gastrointestinal stromal tumors in Korea, 2003-2004. J Korean Med Sci. 2010;25:853-862. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Chan KH, Chan CW, Chow WH, Kwan WK, Kong CK, Mak KF, Leung MY, Lau LK. Gastrointestinal stromal tumors in a cohort of Chinese patients in Hong Kong. World J Gastroenterol. 2006;12:2223-2228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 50] [Cited by in RCA: 57] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Nishida T, Goto O, Raut CP, Yahagi N. Diagnostic and treatment strategy for small gastrointestinal stromal tumors. Cancer. 2016;122:3110-3118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 110] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 6. | Gastrointestinal Surgery Group; Chinese Medical Association Gastroenterology Endoscopy Branch, Chinese Medical Association Endoscopy Specialized Committee, Chinese Medical Association Gastrointestinal Endoscopy Branch, Chinese Medical Association Surgery Branch. Chinese expert consensus on endoscopic diagnosis and treatment of digestive tract submucosal oncology (2018 edition). Zhonghua Xiaohua Neijing Zazhi. 2018;35:536-546. |
| 7. | Landi B, Palazzo L. The role of endosonography in submucosal tumours. Best Pract Res Clin Gastroenterol. 2009;23:679-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Polkowski M, Butruk E. Submucosal lesions. Gastrointest Endosc Clin N Am. 2005;15:33-54, viii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 52] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 9. | Hwang JH, Saunders MD, Rulyak SJ, Shaw S, Nietsch H, Kimmey MB. A prospective study comparing endoscopy and EUS in the evaluation of GI subepithelial masses. Gastrointest Endosc. 2005;62:202-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 144] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 10. | Okten RS, Kacar S, Kucukay F, Sasmaz N, Cumhur T. Gastric subepithelial masses: evaluation of multidetector CT (multiplanar reconstruction and virtual gastroscopy) versus endoscopic ultrasonography. Abdom Imaging. 2012;37:519-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Brand B, Oesterhelweg L, Binmoeller KF, Sriram PV, Bohnacker S, Seewald S, De Weerth A, Soehendra N. Impact of endoscopic ultrasound for evaluation of submucosal lesions in gastrointestinal tract. Dig Liver Dis. 2002;34:290-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 12. | Chen TH, Hsu CM, Chu YY, Wu CH, Chen TC, Hsu JT, Yeh TS, Lin CJ, Chiu CT. Association of endoscopic ultrasonographic parameters and gastrointestinal stromal tumors (GISTs): can endoscopic ultrasonography be used to screen gastric GISTs for potential malignancy? Scand J Gastroenterol. 2016;51:374-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 13. | Jeon SW, Park YD, Chung YJ, Cho CM, Tak WY, Kweon YO, Kim SK, Choi YH. Gastrointestinal stromal tumors of the stomach: endosonographic differentiation in relation to histological risk. J Gastroenterol Hepatol. 2007;22:2069-2075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Humphris JL, Jones DB. Subepithelial mass lesions in the upper gastrointestinal tract. J Gastroenterol Hepatol. 2008;23:556-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 699] [Cited by in RCA: 865] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 16. | Chun SY, Kim KO, Park DS, Lee IJ, Park JW, Moon SH, Baek IH, Kim JH, Park CK, Kwon MJ. Endoscopic submucosal dissection as a treatment for gastric subepithelial tumors that originate from the muscularis propria layer: a preliminary analysis of appropriate indications. Surg Endosc. 2013;27:3271-3279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 75] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 17. | Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006;23:70-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1244] [Cited by in RCA: 1304] [Article Influence: 72.4] [Reference Citation Analysis (33)] |
| 18. | von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Kane JM, Keedy V, Kim E, Koon H, Mayerson J, McCarter M, McGarry SV, Meyer C, Morris ZS, O'Donnell RJ, Pappo AS, Paz IB, Petersen IA, Pfeifer JD, Riedel RF, Ruo B, Schuetze S, Tap WD, Wayne JD, Bergman MA, Scavone JL. Soft Tissue Sarcoma, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:536-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 460] [Article Influence: 76.7] [Reference Citation Analysis (1)] |
| 19. | Li J, Ye Y, Wang J, Zhang B, Qin S, Shi Y, He Y, Liang X, Liu X, Zhou Y, Wu X, Zhang X, Wang M, Gao Z, Lin T, Cao H, Shen L; Chinese Society Of Clinical Oncology Csco Expert Committee On Gastrointestinal Stromal Tumor. Chinese consensus guidelines for diagnosis and management of gastrointestinal stromal tumor. Chin J Cancer Res. 2017;29:281-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 139] [Article Influence: 17.4] [Reference Citation Analysis (1)] |
| 20. | Casali PG, Abecassis N, Aro HT, Bauer S, Biagini R, Bielack S, Bonvalot S, Boukovinas I, Bovee JVMG, Brodowicz T, Broto JM, Buonadonna A, De Álava E, Dei Tos AP, Del Muro XG, Dileo P, Eriksson M, Fedenko A, Ferraresi V, Ferrari A, Ferrari S, Frezza AM, Gasperoni S, Gelderblom H, Gil T, Grignani G, Gronchi A, Haas RL, Hassan B, Hohenberger P, Issels R, Joensuu H, Jones RL, Judson I, Jutte P, Kaal S, Kasper B, Kopeckova K, Krákorová DA, Le Cesne A, Lugowska I, Merimsky O, Montemurro M, Pantaleo MA, Piana R, Picci P, Piperno-Neumann S, Pousa AL, Reichardt P, Robinson MH, Rutkowski P, Safwat AA, Schöffski P, Sleijfer S, Stacchiotti S, Sundby Hall K, Unk M, Van Coevorden F, van der Graaf WTA, Whelan J, Wardelmann E, Zaikova O, Blay JY; ESMO Guidelines Committee and EURACAN. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv68-iv78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 289] [Article Influence: 41.3] [Reference Citation Analysis (1)] |
| 21. | Rossi S, Gasparotto D, Toffolatti L, Pastrello C, Gallina G, Marzotto A, Sartor C, Barbareschi M, Cantaloni C, Messerini L, Bearzi I, Arrigoni G, Mazzoleni G, Fletcher JA, Casali PG, Talamini R, Maestro R, Dei Tos AP. Molecular and clinicopathologic characterization of gastrointestinal stromal tumors (GISTs) of small size. Am J Surg Pathol. 2010;34:1480-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 96] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 22. | Agaimy A, Wünsch PH, Hofstaedter F, Blaszyk H, Rümmele P, Gaumann A, Dietmaier W, Hartmann A. Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol. 2007;31:113-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 236] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 23. | Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, Miettinen M, O'Leary TJ, Remotti H, Rubin BP, Shmookler B, Sobin LH, Weiss SW. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10:81-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 275] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 24. | Gill KR, Camellini L, Conigliaro R, Sassatelli R, Azzolini F, Messerotti A, Woodward TA, Wallace MB, Jamil LH, Raimondo M. The natural history of upper gastrointestinal subepithelial tumors: a multicenter endoscopic ultrasound survey. J Clin Gastroenterol. 2009;43:723-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 25. | Kim MY, Jung HY, Choi KD, Song HJ, Lee JH, Kim DH, Choi KS, Lee GH, Kim JH. Natural history of asymptomatic small gastric subepithelial tumors. J Clin Gastroenterol. 2011;45:330-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 26. | Gao Z, Wang C, Xue Q, Wang J, Shen Z, Jiang K, Shen K, Liang B, Yang X, Xie Q, Wang S, Ye Y. The cut-off value of tumor size and appropriate timing of follow-up for management of minimal EUS-suspected gastric gastrointestinal stromal tumors. BMC Gastroenterol. 2017;17:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 27. | Lok KH, Lai L, Yiu HL, Szeto ML, Leung SK. Endosonographic surveillance of small gastrointestinal tumors originating from muscularis propria. J Gastrointestin Liver Dis. 2009;18:177-180. [PubMed] |
| 28. | Coe TM, Fero KE, Fanta PT, Mallory RJ, Tang CM, Murphy JD, Sicklick JK. Population-Based Epidemiology and Mortality of Small Malignant Gastrointestinal Stromal Tumors in the USA. J Gastrointest Surg. 2016;20:1132-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 29. | Bara T, Bancu S, Bara T Jr, Mureşan M, Bancu L, Azamfirei L, Podeanu D, Mureşan S. [Gastric stromal tumor with liver and subcutaneus metastasis. Case report]. Chirurgia (Bucur). 2009;104:621-624. [PubMed] |
| 30. | Suzuki K, Yasuda T, Nagao K, Hori T, Watanabe K, Kanamori M, Kimura T. Metastasis of gastrointestinal stromal tumor to skeletal muscle: a case report. J Med Case Rep. 2014;8:256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 31. | Sahin E, Yetişyiğit T, Oznur M, Elboğa U. Gastric gastrointestinal stromal tumor with bone metastases - case report and review of the literature. Klin Onkol. 2014;27:56-59. [PubMed] |
| 32. | National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Soft Tissue Sarcoma, Version 2. 2020. [cited 28 May 2020]. Available from: https://www.nccn.org/professionals/physician_gls/pdf/sarcoma_blocks.pdf. |
| 33. | ASGE Standards of Practice Committee, Evans JA, Chandrasekhara V, Chathadi KV, Decker GA, Early DS, Fisher DA, Foley K, Hwang JH, Jue TL, Lightdale JR, Pasha SF, Sharaf R, Shergill AK, Cash BD, DeWitt JM. The role of endoscopy in the management of premalignant and malignant conditions of the stomach. Gastrointest Endosc. 2015;82:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 207] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 34. | Lachter J, Bishara N, Rahimi E, Shiller M, Cohen H, Reshef R. EUS clarifies the natural history and ideal management of GISTs. Hepatogastroenterology. 2008;55:1653-1656. [PubMed] |
| 35. | Fang YJ, Cheng TY, Sun MS, Yang CS, Chen JH, Liao WC, Wang HP. Suggested cutoff tumor size for management of small EUS-suspected gastric gastrointestinal stromal tumors. J Formos Med Assoc. 2012;111:88-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 36. | Akahoshi K, Sumida Y, Matsui N, Oya M, Akinaga R, Kubokawa M, Motomura Y, Honda K, Watanabe M, Nagaie T. Preoperative diagnosis of gastrointestinal stromal tumor by endoscopic ultrasound-guided fine needle aspiration. World J Gastroenterol. 2007;13:2077-2082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 159] [Cited by in RCA: 184] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 37. | Li B, Qi ZP, Zhou PH, Yao LQ, Xu MD, Ren Z, Shi Q, Chen T, Chai SL, Zhong YS. The value of endoscopic full-thickness resection for small and micro-gastrointestinal stromal tumors in the fundus of the stomach. Zhongguo Shiyong Waike Zazhi. 2017;37:1281-1285. [DOI] [Full Text] |
| 38. | Hiki N, Nunobe S, Ohashi M. [Laparoscopy and endoscopy cooperative surgery (LECS) for gastric submucosal tumor]. Nihon Geka Gakkai Zasshi. 2014;115:102-104. [PubMed] |
| 39. | Matsuhashi N, Osada S, Yamaguchi K, Okumura N, Tanaka Y, Imai H, Sasaki Y, Nonaka K, Takahashi T, Futamura M, Yoshida K. Long-term outcomes of treatment of gastric gastrointestinal stromal tumor by laparoscopic surgery: review of the literature and our experience. Hepatogastroenterology. 2013;60:2011-2015. [PubMed] |
| 40. | Zhang Q, Gao LQ, Han ZL, Li XF, Wang LH, Liu SD. Effectiveness and safety of endoscopic resection for gastric GISTs: a systematic review. Minim Invasive Ther Allied Technol. 2018;27:127-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 41. | Wang C, Gao Z, Shen K, Cao J, Shen Z, Jiang K, Wang S, Ye Y. Safety and efficiency of endoscopic resection versus laparoscopic resection in gastric gastrointestinal stromal tumours: A systematic review and meta-analysis. Eur J Surg Oncol. 2020;46:667-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 42. | An W, Sun PB, Gao J, Jiang F, Liu F, Chen J, Wang D, Li ZS, Shi XG. Endoscopic submucosal dissection for gastric gastrointestinal stromal tumors: a retrospective cohort study. Surg Endosc. 2017;31:4522-4531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 43. | Meng Y, Li W, Han L, Zhang Q, Gong W, Cai J, Li A, Yan Q, Lai Q, Yu J, Bai L, Liu S, Li Y. Long-term outcomes of endoscopic submucosal dissection versus laparoscopic resection for gastric stromal tumors less than 2 cm. J Gastroenterol Hepatol. 2017;32:1693-1697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 44. | Oda I, Suzuki H, Nonaka S, Yoshinaga S. Complications of gastric endoscopic submucosal dissection. Dig Endosc. 2013;25 Suppl 1:71-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 163] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 45. | Saito I, Tsuji Y, Sakaguchi Y, Niimi K, Ono S, Kodashima S, Yamamichi N, Fujishiro M, Koike K. Complications related to gastric endoscopic submucosal dissection and their managements. Clin Endosc. 2014;47:398-403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 46. | Jeong ID, Jung SW, Bang SJ, Shin JW, Park NH, Kim DH. Endoscopic enucleation for gastric subepithelial tumors originating in the muscularis propria layer. Surg Endosc. 2011;25:468-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 47. | Kim HH, Kim GH, Kim JH, Choi MG, Song GA, Kim SE. The efficacy of endoscopic submucosal dissection of type I gastric carcinoid tumors compared with conventional endoscopic mucosal resection. Gastroenterol Res Pract. 2014;2014:253860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 48. | Chen T, Yao LQ, Xu MD, Zhang YQ, Chen WF, Shi Q, Cai SL, Chen YY, Xie YH, Ji Y, Chen SY, Zhou PH, Zhong YS. Efficacy and Safety of Endoscopic Submucosal Dissection for Colorectal Carcinoids. Clin Gastroenterol Hepatol. 2016;14:575-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 49. | Qi ZP, Shi Q, Liu JZ, Yao LQ, Xu MD, Cai SL, Li B, Take I, Zhang YQ, Chen WF, Zhong YS, Zhou PH. Efficacy and safety of endoscopic submucosal dissection for submucosal tumors of the colon and rectum. Gastrointest Endosc 2018; 87: 540-548. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 50. | Wang S, Shen L. Efficacy of Endoscopic Submucosal Excavation for Gastrointestinal Stromal Tumors in the Cardia. Surg Laparosc Endosc Percutan Tech. 2016;26:493-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 51. | Park CH, Kim EH, Jung DH, Chung H, Park JC, Shin SK, Lee YC, Kim H, Lee SK. Impact of periodic endoscopy on incidentally diagnosed gastric gastrointestinal stromal tumors: findings in surgically resected and confirmed lesions. Ann Surg Oncol. 2015;22:2933-2939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 52. | Yu FB, Xiong HY. Value inquiry of combination of endoscopic resection and endoscopic band ligation closure in treatment of gastric submucosal tumors originated from the muscular is propria layer. Zhongguo Neijing Zazhi. 2012;18:121-124. |
| 53. | Wang L, Ren W, Fan CQ, Li YH, Zhang X, Yu J, Zhao GC, Zhao XY. Full-thickness endoscopic resection of nonintracavitary gastric stromal tumors: a novel approach. Surg Endosc. 2011;25:641-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 54. | Zhou PH, Yao LQ, Qin XY, Cai MY, Xu MD, Zhong YS, Chen WF, Zhang YQ, Qin WZ, Hu JW, Liu JZ. Endoscopic full-thickness resection without laparoscopic assistance for gastric submucosal tumors originated from the muscularis propria. Surg Endosc. 2011;25:2926-2931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 245] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 55. | Wang H, Feng X, Ye S, Wang J, Liang J, Mai S, Lai M, Feng H, Wang G, Zhou Y. A comparison of the efficacy and safety of endoscopic full-thickness resection and laparoscopic-assisted surgery for small gastrointestinal stromal tumors. Surg Endosc. 2016;30:3357-3361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 56. | Schmidt A, Damm M, Caca K. Endoscopic full-thickness resection using a novel over-the-scope device. Gastroenterology 2014; 147: 740-742. e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 57. | Shi Q, Chen T, Zhong YS, Zhou PH, Ren Z, Xu MD, Yao LQ. Complete closure of large gastric defects after endoscopic full-thickness resection, using endoloop and metallic clip interrupted suture. Endoscopy. 2013;45:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 109] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 58. | Lu J, Lu X, Jiao T, Zheng M. Endoscopic management of upper gastrointestinal submucosal tumors arising from muscularis propria. J Clin Gastroenterol. 2014;48:667-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 59. | Chen T, Zhang C, Yao LQ, Zhou PH, Zhong YS, Zhang YQ, Chen WF, Li QL, Cai MY, Chu Y, Xu MD. Management of the complications of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors. Endoscopy. 2016;48:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 60. | Song S, Wang X, Zhang S, Li Y, Zhang X, Chu X. Efficacy and complications of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors and exploration for influencing factors. Z Gastroenterol. 2018;56:365-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |