Published online Feb 15, 2019. doi: 10.4251/wjgo.v11.i2.102
Peer-review started: August 30, 2018
First decision: October 26, 2018
Revised: December 6, 2018
Accepted: January 10, 2019
Article in press: January 10, 2019
Published online: February 15, 2019
Processing time: 170 Days and 1.3 Hours
Identification of germ-line mutations in pancreatic ductal adenocarcinoma (PDAC) could impact on patient/family.
To assess the referral pathways for genetic consultations in PDAC.
Electronic records of PDAC patients were reviewed retrospectively. Patients eligible for genetic consultation referral were identified following the European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC) criteria.
Four-hundred patients were eligible. Of 113 patients (28.3%) meeting EUROPAC criteria, 8.8% were referred for genetic opinion. Germ-line mutations were identified in 0.75% of the whole population.
Earlier referrals and increased awareness may be able to overcome the low rate of successful genetic appointments.
Core tip: Electronic records of consecutive patients diagnosed with pancreatic ductal adenocarcinoma (PDAC) were reviewed retrospectively. The European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC) criteria were employed to identify patients eligible for genetic consultation referral. Out of 400 eligible patients, 113 (28.3% of the whole population) met referral criteria, only 10 (8.8%) were referred for genetic opinion. There was a low referral rate even for patients fulfilling EUROPAC criteria and a significant number of patients did not attend the consultation due to deteriorating performance status. Earlier referral, and increased awareness may optimise genetic services referral for patients with PDAC.
- Citation: Fulton AJ, Lamarca A, Nuttall C, McCallum L, Pihlak R, O’Reilly D, Lalloo F, McNamara MG, Hubner RA, Clancy T, Valle JW. Identification of patients with pancreatic adenocarcinoma due to inheritable mutation: Challenges of daily clinical practice. World J Gastrointest Oncol 2019; 11(2): 102-116
- URL: https://www.wjgnet.com/1948-5204/full/v11/i2/102.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v11.i2.102
The most common form of pancreatic malignancy is pancreatic ductal adenocarcinoma (PDAC), with a 1 in 100 lifetime risk within the general population (up to age 75)[1]. The diagnosis of PDAC is associated with poor survival; 1-year and 5-year survival rates are 20% and 3%, respectively[2], despite surgical intervention and the implementation of chemotherapeutic guidelines outlined by the international experts groups such as the European Society for Medical Oncology (ESMO)[3]. Currently, curative resection and adjuvant chemotherapy with gemcitabine and capecitabine[4,5] or modified-FOLFIRINOX (combined therapy of oxaliplatin, irinotecan, and 5-fluorouracil (5-FU)[6]) is considered standard of care treatment for resectable disease stages; however risk of relapse remains high (66%-92% develop recurrent disease within 2 years)[3]. In the palliative setting, gemcitabine-based treatments or FOLFIRINOX are considered standard options of treatment[3].
Although most cases of PDAC are sporadic (related to non-heritable modifiable risk factors such as smoking, alcohol and pancreatitis[3,7]), a small percentage of cases will arise in the context of a hereditary aberration[8-11], such as germ-line Breast cancer gene (BRCA1/2) mutation (reported to be present in around 1% to 4.6% of patients with pancreatic cancer[3,12,13]; higher in other series[14]). The prevalence of BRCA1/2 mutations in the general population is approximately 1 in 400, two thirds of which are BRCA2[15]. Therefore, BRCA2 mutation is the commonest inherited predisposition for the development of PDAC[16]. A number of other germ-line mutations such as PALB2, CDKN2A, ATM, p53 and mismatch repair genes (MLH1, MSH2, MSH6) are known to also predispose an individual to develop PDAC[17,18]. Germline mutations in these genes are relatively rare[19]; however, when present, they commonly have high penetrance. For example, a germline mutation in CDKN2A confers a 38-fold increased risk of developing PDAC compared to the general population[20,21].
Detection of patients harbouring a germ-line mutation predisposing them to PDAC is initiated by establishing the patients’ prior personal history and family history of malignancy, which triggers a referral to genetic services[22]. Approximately 10%-20% of patients diagnosed with PDAC report a prior personal history of cancer or family history of cancer[11,13], requiring a genetic consultation. Previous studies have shown that there is a significant association between presence of germ-line mutation and personal/family history of breast (10.7% of patient with personal/family history of breast cancer were found to have a germ-line mutation vs 2.1% of patient without personal/family history of breast cancer who were identified to have a germ-line aberration; P-value < 0.001) and colorectal (11.1% vs 2.8%; P-value 0.002) cancer[11]. A different study identified an even higher percentage of patients with germ-line mutations (15.1%)[23]. Therefore, when this “selected” population is analysed, around 10%-15% of patients (i.e., 1%-3% of the whole PDAC population)[11,12] are expected to harbour a germ-line mutation which explains the increased predisposition to PDAC. Unfortunately, identification of this patient population remains challenging for clinicians in daily clinical practice and having reliable criteria should help capturing all patients who require referral for genetic opinion.
The European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC) published research guidelines for the identification of patients with a potential germ-line mutation who should be referred for genetic consultation (Table 1)[24]. The EUROPAC guidelines have thus far not been implemented in the genetic service diagnostic setting; however they have been agreed at an international level in the research setting. A number of alternative referral guidelines exist even though they have not been extensively evaluated in clinical trial settings. Inter-guideline variation is significant and produces heterogeneity in referral practice. The National Comprehensive Cancer Network (NCCN) guidelines recommend BRCA testing for selected PDAC patients[25]. These include PDAC patients with a first, second or third degree relative with ovarian carcinoma; breast cancer diagnosed under 50 years old; two relatives with breast, pancreatic or prostate cancer at any age or any patient with PDAC and Ashkenazi Jewish ancestry. Additionally, guidelines have also been published by the American College of Gastroenterology (ACG)[26]. Recently, the National Institute for Health and Care Excellence (NICE) guidelines (United Kingdom) have also been updated and provide guidance on this issue[27].
| EUROPAC criteria | |
| Criterion 1 | ≥ 2 first-degree relatives with pancreatic cancer |
| Criterion 2 | ≥ 3 relatives with pancreatic cancer |
| Criterion 3 | Possible associated cancer syndrome (defined as sub-criteria below) in addition to the case of pancreatic cancer being studied |
| Criterion 3.a: BRCA1/2 | Personal/family history (≥ 1 first/second-degree relatives) of breast/ovarian cancer |
| Criterion 3.b: Familial Atypical Multiple Mole Melanoma (FAMMM) syndrome | Personal/family history of melanoma in ≥ 1 first/second degree relative AND a high total body naevi count (often > 50) |
| Criterion 3.c: Lynch syndrome | Personal/family history (≥ 1 first/second-degree relatives) of a Lynch syndrome-associated cancer (such as colorectal, endometrial, small bowel, renal) |
| Criterion 3.d: Peutz-Jeghers syndrome | Oral/mucous membrane pigmentation +/- a personal/family history (≥ 1 first/second-degree relatives) of gastrointestinal cancers in first/second degree relatives |
An alternative approach would be to test for germ-line mutations in “non-selected” populations (i.e., when patients are analysed regardless of personal or family history of cancer). When such strategy is pursued, rate of patients identified to have a germ-line aberration may increase up to 3.9%-4.6% out of the whole PDAC population[13,28,29]. Despite this, current clinical practice still relies on analysis of “selected” patients, since this is considered a more cost/effective approach. Genetic testing is only advocated in ”non-selected” patients if a mutation rate of 10% or greater exists[30]. Thus, “non-selected” approach in PDAC remains investigational only. In addition, higher mutation rates have also been detected in “selected” and “non-selected” groups in the research setting when broader gene panels testing have been implemented[31-33], however the clinical utility of such mutations is still unclear.
This study aimed to explore the rate of appropriate referrals for a genetic consultation in patients diagnosed with PDAC and the outcome of such referrals in current daily oncology clinical practice. We aimed to identify challenges and potential solutions.
The study set out to review 400 consecutive patients diagnosed with PDAC. Eligible patients were those diagnosed with PDAC who were seen at The Christie NHS Foundation Trust (Manchester, United Kingdom) between September 2012 and December 2015. Patients with pancreatic malignancies other than PDAC (e.g., neuroendocrine tumours) or with other malignancies arising from the hepato-pancreato-biliary tract were excluded. This study was approved by The Christie NHS Foundation Trust Audit Committee (CE15/1575).
Patients were identified through electronic records. Electronic case notes were reviewed retrospectively for data including demographics, diagnosis and treatment. Comorbidities were collected following the Adult Co-morbidity Evaluation (ACE-27) criteria[34]. The focus was on reviewing patient’s previous personal malignancy history and family history in order to assess the presence of risk factors[3,7] for PDAC (including personal and family history of cancer) and the percentage of patients meeting criteria for referral for genetic counselling (applying the EUROPAC criteria[35], Table 1). In addition, outcome from such referrals and details regarding other known risk factors, such as smoking or diabetes were collected. Excess alcohol intake was defined as patients having a notation of “excess alcohol intake” in their electronic notes, or having consumed greater than the recommended amount of units in the 2010-2015 United Kingdom guidelines[36]. It was aimed to establish the proportion of patients fulfilling the EUROPAC criteria that were referred to the regional clinical genetics service, and subsequent testing if deemed appropriate. Information on whether the patient had been referred to the genetic service and the outcome of such referrals was retrieved.
The primary objectives of this study were to assess the appropriateness of referral to genetic services in patients diagnosed with PDAC, and to assess the current referral practice for identification of potential areas of improvement. The primary end-point was the percentage of patients meeting the EUROPAC criteria referred for genetic counselling. Secondary objectives included the frequency of genetic aberration identified and the characteristics of such populations, together with the frequency of any other modifiable risk factors.
Although a formal sample size calculation was not performed for this study, and based on the fact that an infrequent event was been explored (this study was targeting “selected” population, thus 10%-20% of patients would be expected to meet referral criteria; but only around 1% out of the whole population would be expected to be identified a germ-line mutation), a target sample of 400 patients was pre-specified in order to secure enough patient representation. Statistical analysis was carried out using GraphPad Prism and Stata v.12 packages. Frequency tables for each of the variables were created. Continuous variables were analysed by calculating median and range/95% confidence intervals (95%CI). The characteristics of the population of patients meeting the EUROPAC criteria were compared to the cohort of patients who did not meet such criteria: univariate analysis (χ2 or t-test as appropriate according to the type of variable) and multivariable analysis (logistic regression) were performed; variables with P-value < 0.05 in the univariate analysis were included in the multivariable analysis.
In order to identify 400 eligible patients for our analysis, a total of 408 patients were screened; the commonest reason for exclusion was the presence of histological entity other than adenocarcinoma (Figure 1).
Of the 400 patients in the study, 206 (51.5%) patients were male and 194 (48.5%) were female, with a median age of 67.7 years (range 29.9-94.2). Out of the 400 patients, 338 (84.5%) were referred for consideration of systemic chemotherapy with palliative intent: 230 (57.5% of the 400) went on to receive palliative chemotherapy, of which 94 (40.9%) received gemcitabine single agent (Table 2).
| Parameter | Frequency | Relative percentage (%) | |
| Patient demographic | |||
| Gender | Male | 206 | 51.5 |
| Female | 194 | 48.5 | |
| Age | Median | 67.7 yr | |
| Range | 29.9-94.2 | ||
| Comorbidity grade (according to Adult Comorbidity Evaluation-27)[34] | None | 149 | 37.32 |
| Mild | 164 | 412 | |
| Moderate | 60 | 152 | |
| Severe | 27 | 6.82 | |
| Pathological confirmation of malignancy | Yes | 355 | 88.82 |
| No1 | 45 | 11.32 | |
| Stage | Localised (I-II) | 4 | 1.0 |
| Locally advanced (III) | 202 | 50.5 | |
| Metastatic (IV) | 194 | 48.5 | |
| Eastern Cooperative Oncology Group Performance Status at time of PDAC diagnosis | 0 | 43 | 10.82 |
| 1 | 198 | 49.52 | |
| 2 | 98 | 24.52 | |
| 3 | 58 | 14.52 | |
| 4 | 3 | 0.82 | |
| Treatment characteristics | |||
| Treatment intent at time of referral3 | Curative | 62 | 15.5 |
| Palliative | 338 | 84.5 | |
| Curative management | |||
| Patient received curative surgery? | Yes | 65 | 16.3 |
| Patient received adjuvant chemotherapy after curative surgery | Yes | 42 | 65.6 |
| No | 22 | 34.4 | |
| Unknown | 1 | n/a | |
| Type of adjuvant chemotherapy | 5-fluorouracil | 1 | 2.42 |
| Capecitabine | 18 | 42.92 | |
| Gemcitabine | 23 | 54.82 | |
| Palliative management | |||
| Patient received palliative chemotherapy | Yes | 230 | 57.5 |
| No | 170 | 42.5 | |
| Type of palliative chemotherapy | FOLFIRINOX | 37 | 16.1 |
| Gemcitabine/Capecitabine | 60 | 26.1 | |
| Gemcitabine/Cisplatin | 5 | 2.2 | |
| Gemcitabine NabPaclitaxel | 27 | 11.7 | |
| Gemcitabine monotherapy | 94 | 40.9 | |
| Other4 | 7 | 3.0 | |
A total of 233 patients (74.9%) had a history of alcohol consumption, with 33 of these (14.2%) having a documented history of “excess” alcohol consumption. One-hundred and eighty-seven patients (55.8%) had a personal history of smoking, and 71 patients (21.2%) were active smokers. Additionally, 103 patients had a previous diagnosis of type 2 diabetes mellitus (25.9%). Other risk factors are summarised in Table 3.
| Non-heritable risk factors | Frequency | Relative percentage (%) | |
| Smoking | |||
| Smoker (active or ex-smoker) | Yes | 187 | 55.8 |
| No | 148 | 44.2 | |
| Unknown | 65 | n/a | |
| Active smoker | Yes | 71 | 21.2 |
| No | 264 | 78.8 | |
| Unknown | 65 | n/a | |
| Alcohol | |||
| Alcohol consumption | Yes | 233 | 74.9 |
| No | 78 | 25.1 | |
| Unknown | 89 | n/a | |
| “Excess” consumption | Yes | 33 | 14.2 |
| No | 200 | 85.8 | |
| Pancreatitis | |||
| Previous pancreatitis | Yes | 11 | 2.8 |
| No | 386 | 97.2 | |
| Unknown | 3 | n/a | |
| Diagnosed > 2 yr before PDAC | Yes | 6 | 54.6 |
| No | 5 | 45.5 | |
| IPMN | |||
| Past medical history of IPMN | Yes | 3 | 0.8 |
| No | 394 | 99.2 | |
| Unknown | 3 | n/a | |
| Diabetes | |||
| Type 2 diabetes mellitus | Yes | 103 | 25.9 |
| No | 295 | 74.1 | |
| Unknown | 2 | n/a | |
| Diagnosed > 2 yr before PDAC | Yes | 39 | 57.4 |
| No | 29 | 42.7 | |
| Unknown | 35 | n/a | |
| Type 1 diabetes mellitus | Yes | 5 | 1.3 |
| No | 393 | 98.7 | |
| Unknown | 2 | n/a | |
Out of the whole population of patients included, 202 patients (50.5%) had a family history of any cancer and 113 (28.3% of patients from the whole population; 33.8% of patients from those 334 who had information regarding family history available) had a prior history of cancer or family history of cancer meeting the EUROPAC referral criteria (Table 4). Of the 113 patients meeting the EUROPAC criteria (as defined in Table 1), 60 patients met criteria 3.a for BRCA 1/2 testing (53.10%) followed by criteria 3.c (29 patients; 25.7%), criteria 3.d (12 patients; 10.6%), criteria 1 (10 patients; 8.8%) and criteria 3.b (2 patients; 1.8%). Family history of cancer was not recorded in the patient’s case notes for 66 of the 400 patients (16.5%).
| Risk Factor | Frequency | Relative percentage (%) | |
| Any personal1/family history of cancer | Yes | 202 | 50.5 |
| No | 132 | 33.0 | |
| Unknown | 66 | 16.5 | |
| Any personal1 or family history of cancer meeting EUROPAC[24] criteria | Yes | 113 | 28.3 |
| No | 221 | 55.2 | |
| Unknown | 66 | 16.5 | |
| Criteria met | Criterion 1 (≥ 2 first-degree relatives with pancreatic cancer) | 10 | 8.8 |
| Criterion 3.a (Personal/family history (≥ 1 first/second-degree relatives) of breast/ovarian cancer) | 60 | 53.1 | |
| Criterion 3.b [Personal/family history of melanoma in ≥ 1 first/second degree relative AND a high total body naevi count (often > 50)] | 2 | 1.8 | |
| Criterion 3.c [Personal/family history (≥ 1 first/second-degree relatives) of a Lynch syndrome-associated cancer (such as colorectal, endometrial, small bowel, renal)] | 29 | 25.7 | |
| Criterion 3.d (Oral/mucous membrane pigmentation +/- a personal/family history (≥ 1 first/second-degree relatives) of gastrointestinal cancers in first/second degree relatives) | 12 | 10.6 | |
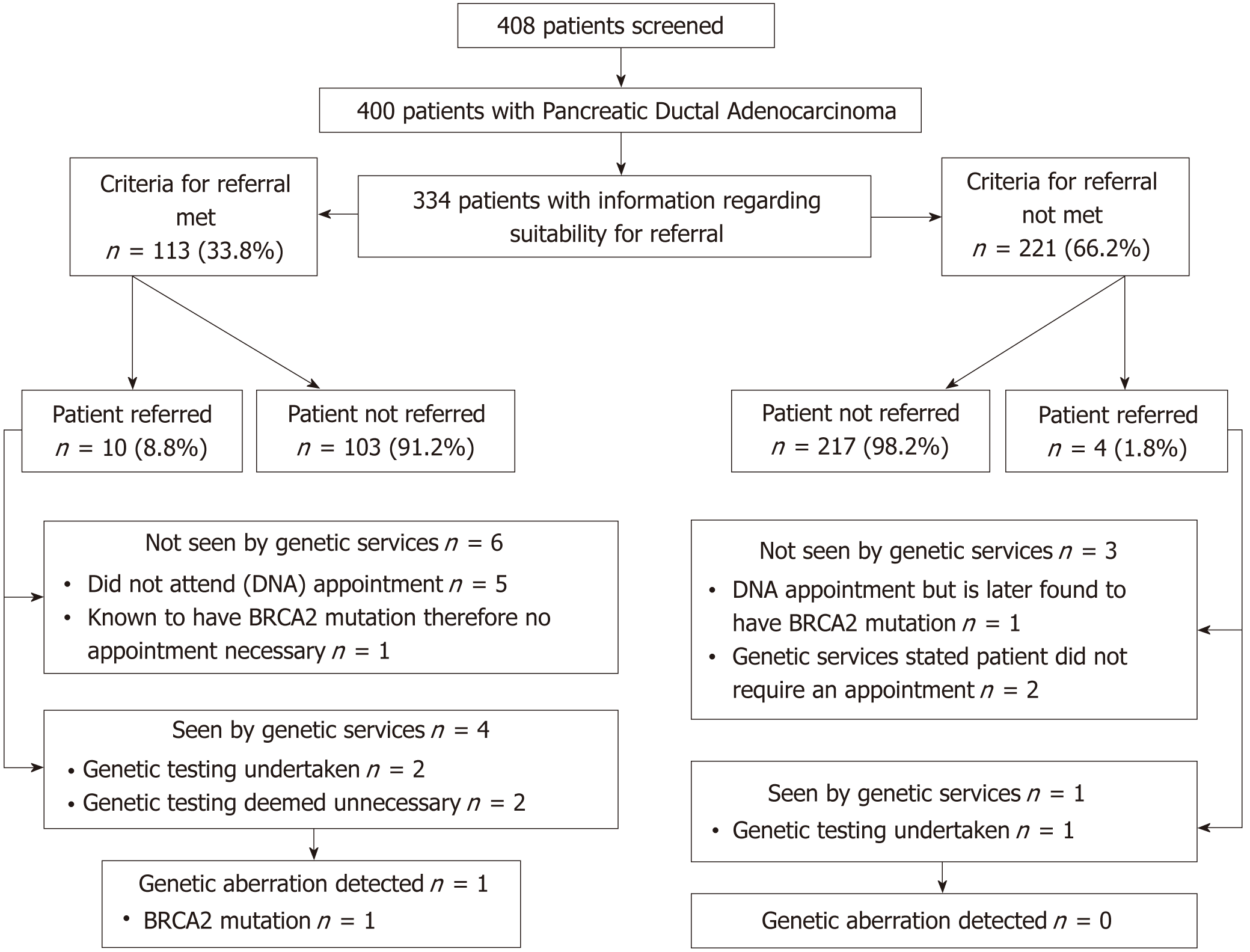
Only 10 of the 113 patients (8.8%) meeting the referral criteria were referred to the regional genetic service. Moreover, only 4 of these 10 patients referred (40%) were ultimately seen by the genetics team, with one patient identified as having a BRCA2 mutation identified through this pathway (Figure 2).
A total of 103 patients met the EUROPAC criteria but were not referred. Most of these patients (97 out of 103) attended for palliative chemotherapy and 59 started chemotherapy. Performance status for this subpopulation of patients was poor: ECOG PS2 (22.3%) and PS3 (13.6%). Fifty-three of the 103 patients had a history of smoking, whilst 71 (68.9%) had a history of alcohol consumption.
Characteristics of those patients who did and did not meet the EUROPAC criteria were compared (Tables 5 and 6) using univariate analysis. The following characteristics were statistically significant predictors of fulfilling the EUROPAC criteria in the univariate analysis and were therefore included in the multivariable logistic regression: patient gender (P-value 0.0041), comorbidity scale (P-value 0.040); malignancy stage (P-value 0.015), treatment intent (P-value 0.022); alcohol intake (P-value 0.025) and family history of any malignancy (P-value < 0.0001). The presence of previous alcohol consumption (Odds Ratio (OR) 2.4 (95%CI 1.1-5.1); P-value 0.022) and presence of any prior malignancy or family history of malignancy (OR 25.3 (95%CI 8.8-72.6); P-value < 0.001) were independent factors for identification of patients likely to meet EUROPAC criteria in the multivariable analysis (Table 7).
| Patients who did not meet criteria for referral (n = 221) | Patients who did meet criteria for referral (n = 113) | Univariate analysis | ||||
| Frequency | Relative percentage | Frequency | Relative percentage | P value | ||
| Demographic characteristics | ||||||
| Gender | Male | 120 | 54.3 | 48 | 42.5 | 0.0041 |
| Female | 101 | 45.7 | 65 | 57.5 | ||
| Age (yr) | Mean (95%CI) | 66.3 (64.9-67.7) | 67.2 (65.4-68.9) | 0.4726 | ||
| Comorbidity grade (ACE-27)[34] | None | 75 | 33.9 | 45 | 39.8 | 0.040 |
| Mild | 97 | 43.9 | 39 | 34.5 | ||
| Moderate | 28 | 12.7 | 24 | 21.2 | ||
| Severe | 21 | 9.5 | 4 | 4.4 | ||
| Pathological confirmation of malignancy | Yes | 194 | 87.8 | 102 | 90.3 | 0.499 |
| No | 27 | 12.2 | 11 | 9.7 | ||
| Stage | Localised | 4 | 1.8 | 0 | 0 | 0.015 |
| Locally advanced | 120 | 54.3 | 46 | 40.7 | ||
| Metastatic | 97 | 43.9 | 67 | 59.3 | ||
| ECOG performance status | 0 | 21 | 9.45 | 14 | 12.4 | 0.534 |
| 1 | 107 | 48.4 | 58 | 51.3 | ||
| 2 | 60 | 27.2 | 25 | 22.1 | ||
| 3 | 32 | 14.5 | 14 | 12.4 | ||
| 4 | 1 | 0.5 | 2 | 1.8 | ||
| Treatment characteristics | ||||||
| Treatment intent | Curative | 38 | 17.2 | 9 | 7.9 | 0.022 |
| Palliative | 183 | 82.8 | 104 | 92.0 | ||
| Curative surgery | Yes | 38 | 17.2 | 13 | 11.5 | 0.171 |
| No | 183 | 82.8 | 100 | 88.5 | ||
| Adjuvant chemotherapy | Yes | 23 | 10.4 | 9 | 7.9 | 0.465 |
| No | 197 | 89.6 | 104 | 92.0 | ||
| Palliative chemotherapy | Yes | 125 | 56.6 | 65 | 57.5 | 0.867 |
| No | 96 | 43.4 | 48 | 42.5 | ||
| Patients who did not meet criteria for referral (n = 194) | Patients who did meet criteria for referral (n = 98) | Univariate analysis | ||||
| Frequency | Relative percentage | Frequency | Relative percentage | P value | ||
| Personal past medical history | ||||||
| Smoker (active or ex-smoker) | Yes | 108 | 55.7 | 55 | 56.1 | 0.941 |
| No | 86 | 44.3 | 43 | 43.9 | ||
| Active smoker | Yes | 45 | 23.2 | 16 | 16.3 | 0.173 |
| No | 149 | 76.8 | 82 | 83.7 | ||
| Alcohol consumption | Yes | 131 | 70.8 | 75 | 83.3 | 0.025 |
| No | 54 | 29.2 | 15 | 16.7 | ||
| “Excess” alcohol consumption | Yes | 24 | 12.9 | 5 | 5.6 | 0.060 |
| No | 161 | 87.0 | 85 | 94.4 | ||
| Previous pancreatitis | Yes | 7 | 3.2 | 1 | 0.9 | 0.195 |
| No | 213 | 96.8 | 112 | 99.1 | ||
| Pancreatitis diagnosed > 2 yr before PDAC | Yes | 5 | 2.3 | 0 | 0 | 0.3493 |
| No | 2159 | 97.7 | 113 | 100 | ||
| Past medical history of IPMN | Yes | 0 | 0 | 0 | 0 | n/a |
| No | 220 | 100 | 113 | 100 | ||
| Type 2 diabetes mellitus | Yes | 61 | 27.7 | 27 | 23.9 | 0.453 |
| No | 156 | 72.3 | 86 | 76.1 | ||
| Type 2 diabetes mellitus diagnosed > 2 yr before PDAC | Yes | 22 | 11.0 | 11 | 10.6 | 0.910 |
| No | 178 | 89.0 | 93 | 89.4 | ||
| Type 1 diabetes mellitus | Yes | 2 | 0.9 | 3 | 2.7 | 0.215 |
| No | 218 | 99.1 | 110 | 97.4 | ||
| Family past medical history | ||||||
| Any family history of cancer | Yes | 93 | 42.1 | 109 | 96.5 | < 0.0001 |
| No | 128 | 57.9 | 4 | 3.5 | ||
| Characteristic | Multivariable analysis (logistic regression) | |
| OR(95%CI) | P value | |
| Gender: Female (Ref) vs male | 0.7 (0.4-1.2) | 0.211 |
| Comorbidity scale: None/mild (Ref) vs moderate/severe | 1.1 (0.5-2.2) | 0.787 |
| Stage: Localised/locally advanced (Ref) vs metastatic | 1.5 (0.8-2.9) | 0.207 |
| Treatment intent: Curative (Ref) vs metastatic | 2.0 (0.8-5.4) | 0.158 |
| History alcohol consumption: No (Ref) vs yes | 2.4 (1.1-5.1) | 0.022 |
| Family history of any malignancy: No (Ref) vs yes | 25.3 (8.8-72.6) | < 0.001 |
In total, 14 of the 400 patients (3.5%) were referred to the Regional Genetics Service (10 of whom met EUROPAC criteria for referral; 4 were referred despite EUROPAC criteria not being met but suspected to be at high-risk) (Figure 2). Of the 14 patients referred, 5 patients (35.7%) were seen, 3 of whom underwent screening for mutations in BRCA1/2. The remaining 9 patients who were referred to the genetics service were not seen by the genetics team (64.3%) due to the following reasons: patients did not attend the appointment (6 patients), referral criteria not met (2 patients) and patient already known to have a pathogenic mutation in BRCA2 and genetic consultation and follow-up was already in place (1 patient). One of the patients was referred despite not meeting EUROPAC criteria due to young age at diagnosis (29 years old); although the patient did not attend the scheduled genetic counselling appointment, she was later identified to have a BRCA2 mutation (as part of the screening process for an ongoing clinical trial).
In total, 3 patients in the whole patient population (0.75%) were found to harbour a germ-line mutation (all were BRCA2 mutations). One patient had a history of smoking and two had a history of alcohol consumption. No other modifiable risk factors were met for these patients. All three patients found to have a BRCA2 mutation had a family history of any malignancy, although only two met EUROPAC criteria. The patient who didn’t meet the EUROPAC criteria had a maternal grandmother with metastatic liver cancer from an unknown primary and was referred on an age at diagnosis basis (patient age 29 years at diagnosis). Both patients meeting the EUROPAC criteria fulfilled criterion 3.a (as defined in Table 1) [one patient’s mother and maternal grandmother had breast cancer, the second patient was noted to have a strong family history of breast and ovarian cancer on the maternal side].
This study identified multiple challenges for adequate genetic referral and genetic counselling for patients diagnosed with PDAC. Our results highlighted the necessity to improve referral practice to the Regional Genetic Services in patients with PDAC when the EUROPAC criteria are implemented. The low rate of patients actually referred to genetic services when meeting referral criteria, along with the high percentage of referred patients who were too unwell to attend by the time of the genetics appointment, are areas for improvement. Adjustments in the referral criteria (i.e., inclusion of patients diagnosed with PDAC below the age of 40 years old) and increased awareness within clinicians are potential solutions to this problem.
Assessment of prior personal and family history of malignancy are the cornerstones for identification of adequate referrals for genetic consultation in patients diagnosed with PDAC (so called “selected” population). Based on previous research, around 10-20% of all patients diagnosed with PDAC would meet criteria for referral for genetic testing on the basis of multiple cases of PDAC within a family[12] or due to presence of other personal/family history of cancer[11,13]. Interestingly, our study identified that almost 30% of the whole population met any of the EUROPAC criteria for referral for genetic assessment. This percentage is slightly higher to previously reported and may require adequate resources[11,13]. In the study by Holter and colleagues, out of the 306 patients explored 52 patients (16.9%) had previous personal history of cancer, and 59 (19.3%) and 37 (12.1%) patients had family history of breast/ovarian or pancreatic cancer, respectively[13]. Similar rates were seen in the study by Grant et al, in which 12.4%, 13.9% and 19.7% of all 290 patients analysed had family history of colorectal, pancreas and breast cancer, respectively[11]. In our study, most of the patients who met the EUROPAC criteria (as defined in Table 1) did so due to the presence of a first degree relative with breast or ovarian cancer (Criterion 3.a) or due to family history of colorectal cancer (Criterion 3.c), which is likely to be related to the high incidence of these malignancies[16,37]. In contrast, only 8.8% of patients were suitable for referral based on suspected familial PDAC (EUROPAC criterion 1 and 2). EUROPAC criteria may therefore need some refinement: (1) further details may be required for identification of patients/family members with family history of colorectal likely to have Lynch syndrome, and (2) similarly, a potential upper age threshold of breast cancer diagnosis may be of interest to prevent referral of sporadic mutation cases.
Overall, around 1% of patients from the whole series had a germ-line mutation identified, in keeping with expected results for population of patients tested based on clinical criteria such as EUROPAC[12] (so called “selected” population). It is worth mentioning that our study identified a patient with BRCA2 germ-line mutation who was diagnosed at a young age and who was tested due to this reason (as per local criteria), even in the absence of meeting EUROPAC criteria for referral. Based on this, it could be considered for EUROPAC criteria to expand to include patients diagnosed with PDAC at a young age, although the exact age cut-off cannot be determined from our study.
Although the rate of patients expected to harbour germ-line aberrations is low, the clinical implications of carrying such a mutation, for both patients and families, are significant. Firstly, identification of such germ-line aberrations would allow for these patients and their families to undergo appropriate genetic counselling, which could include screening for other relevant malignancies (i.e., breast cancer). Families at risk of developing PDAC could also benefit from an effective screening programme, even though such screening has yet to be defined for PDAC[38]. Detection of pancreatic cancer via screening would identify a greater proportion of patients with earlier stage tumours, which has been shown to have a better overall survival[38]. Secondly, for patients harbouring a germ-line BRCA mutation, approaches using Poly (ADP-ribose) polymerase (PARP) inhibitors[39] or DNA-damage agents such as platinum compounds are considered to be more efficacious[40]. PARP inhibitors are already in use for the treatment of ovarian tumours harbouring a BRCA mutation, demonstrating prolonged progression-free survival in these patients[41,42]. In PDAC, a phase II clinical trial using olaparib, a PARP inhibitor, in PDAC patient with either germ-line BRCA1 or BRCA2 mutated PDAC achieved a response rate of 21.7%[43] even after progression on an average of two prior lines of treatment[43]. Additionally, a phase II clinical trial assessing the utility of the PARP inhibitor veliparib in previously treated BRCA or PALB2- mutated PDAC showed single-agent activity of veliparib in the PDAC cohort[44]. These encouraging results have promoted the evaluation of PARP inhibitors earlier in the treatment pathway of patients with advanced PDAC (e.g., the POLO clinical trial exploring the benefit of maintenance olaparib compared to placebo following induction platinum-based chemotherapy; http://www.clinicaltrials.gov: NCT02184195). Whilst results regarding the role of PARP inhibitors in patient carriers of germ-line BRCA mutations diagnosed with PDAC are awaited, the hypothesis that platinum-based chemotherapy achieves better outcomes (response rate or progression-free survival) in this population of patients[45] remains strong; although clinical data are mostly limited to retrospective series[46,47].
Even though a significant number of patients met criteria for referral for genetic consultation, less than 10% of them were actually referred. Patients with any family history of cancer and/or alcohol consumption were the ones with increased risk of meeting EUROPAC criteria, thus the ones that may require in depth consideration. This may help clinicians to focus on a specific population of interest and increase the appropriate referral rate to genetic services.
This study identified a significant number of patients who had absent documentation of any family history of cancer from the medical notes which may be reflection of lack of awareness within clinicians. In addition, a significant number of patients were referred but not seen due to poor performance status. Earlier referral in the patient pathway (for example at the first point of contact, e.g., with surgical/gastroenterology teams) may avoid this from happening. In addition, these patients could be offered the opportunity to provide DNA for storage and for future analysis as appropriate (i.e., to provide relatives with genetic information should the patient become too unwell to attend genetic services).
Regarding non-inheritable risk factors, 25% of patients in this cohort who had type 2 diabetes mellitus had their diabetes diagnosed within 2 years of their PDAC diagnosis. This is in keeping with research demonstrating a significantly higher prevalence of PDAC in patients with new onset type-2 diabetes[48]. The most commonly noted modifiable risk factors in this study were, as expected, cigarette smoking[49] and alcohol consumption[50]. The reduction of exposure to these risk factors would hopefully lead to a reduction in PDAC incidence, along with the incidence of many other malignancies.
The inherent limitations of retrospective studies apply to this study. For example, the number of patients with missing information for many of the risk factors explored in this study is likely related to a reporting bias. In order to reduce any selection bias, this was a consecutive series of patients and the demographic characteristics identified in this study are in keeping with national averages[51] and the distribution of chemotherapy regimen usage was also reflective of current evidence for the treatment of PDAC. Prospective studies are required to further explore the potential role of extending the current EUROPAC criteria. Comparison of referral cohorts across multiple centres when various referral guidelines are applied and any bearing this has on germ-line mutation identification would provide quantitative comparisons between guidelines. This is one potential avenue of pursuit which was not addressed in this study. The multitude of other referral guidelines could also have been implemented in this study (e.g., NICE, NCCN, ACG and local guidelines)[25-27] in order to develop a comprehensive review of referral guidelines, which in turn would enable comparison of positive and negative aspects of different guidelines, in order to produce or implement the most efficacious one. Testing for other germline mutations which predispose individuals to developing PDAC would also be another area of improvement for this study. Identifying such mutations would have enabled potential genetic counselling for family members. This study was a single centre experience, however it was in a high throughput tertiary hospital. Further analysis of referral to genetic services for potential germline mutations would benefit from the inclusion of multiple centres using numerous referral criteria.
In summary, identification of germ-line mutations, such as BRCA2, may have important implications for patients (with respect to choice of treatment, often in the clinical trial setting) and their families. This study identified that an increased awareness among pancreatic cancer clinicians and earlier referral in the patient pathway may ensure that patients fulfilling the EUROPAC criteria are assessed by expert clinical genetics teams. Moreover, the EUROPAC referral criteria may need to be broadened to include extremely young patients regardless of other criteria being met before implementation into the clinical setting.
Approximately 10% of patients diagnosed with pancreatic ductal adenocarcinoma (PDAC) report a significant family history of cancer, requiring genetic consultation; 10% of those referred are expected to have a germ-line predisposition (i.e., 1% of the whole PDAC population).
Referrals for genetic consultations for patients diagnosed with pancreatic ductal adenocarcinoma are many times overlooked, probably due to a lack of awareness.
To understand current referral pathway for genetic consultation and areas for potential improvement.
In this study, electronic records of consecutive patients diagnosed with PDAC were reviewed retrospectively. The European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC) criteria were employed to identify patients eligible for genetic consultation referral.
Of 400 patients eligible, 113 patients (28.3% of the whole population) met referral criteria, only 10 (8.8%) were referred for genetic opinion. Germ-line mutations (BRCA2) were identified in three patients (0.75% of the whole population); one patient was tested due to young age at presentation (not conforming to EUROPAC criteria).
There was a low referral rate even for patients fulfilling EUROPAC criteria. A significant number of patients did not attend the genetic consultation due to deteriorating performance status.
Earlier referral, increased awareness of genetic services/testing amongst clinicians, together with the use of appropriate referral criteria may be required to optimise genetic services referral for patients with PDAC.
Manuscript source: Invited manuscript
Specialty type: Oncology
Country of origin: United Kingdom
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Yip D S- Editor: Ma YJ L- Editor: A E- Editor: Bian YN
| 1. | Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18694] [Cited by in RCA: 21371] [Article Influence: 2137.1] [Reference Citation Analysis (3)] |
| 2. | Cancer Research UK. Children's cancers incidence statistics. Available from: URL: http://www.cancerresearchuk.org/health-professional/cancer-statistics/childrens-cancers/incidence#heading-Three. |
| 3. | Ducreux M, Cuhna AS, Caramella C, Hollebecque A, Burtin P, Goéré D, Seufferlein T, Haustermans K, Van Laethem JL, Conroy T, Arnold D; ESMO Guidelines Committee. Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2015;26 Suppl 5:v56-v68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 905] [Cited by in RCA: 930] [Article Influence: 93.0] [Reference Citation Analysis (0)] |
| 4. | Neoptolemos JP, Palmer D, Ghaneh P, Valle JW, Cunningham D, Wadsley J, Meyer T, Anthoney A, Glimelius B, Falk S, Segersvard R, Izbicki JR, Middleton GW, Ross PJ, Wasan H, Mcdonald A, Crosby TDL, Psarelli EE, Hammel P, Buchler MW. ESPAC-4: A multicenter, international, open-label randomized controlled phase III trial of adjuvant combination chemotherapy of gemcitabine (GEM) and capecitabine (CAP) versus monotherapy gemcitabine in patients with resected pancreatic ductal adenocarcinoma. J Clin Oncol. 2017;34. [RCA] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 5. | Neoptolemos JP, Palmer DH, Ghaneh P, Psarelli EE, Valle JW, Halloran CM, Faluyi O, O'Reilly DA, Cunningham D, Wadsley J, Darby S, Meyer T, Gillmore R, Anthoney A, Lind P, Glimelius B, Falk S, Izbicki JR, Middleton GW, Cummins S, Ross PJ, Wasan H, McDonald A, Crosby T, Ma YT, Patel K, Sherriff D, Soomal R, Borg D, Sothi S, Hammel P, Hackert T, Jackson R, Büchler MW; European Study Group for Pancreatic Cancer. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. Lancet. 2017;389:1011-1024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1327] [Cited by in RCA: 1394] [Article Influence: 174.3] [Reference Citation Analysis (0)] |
| 6. | Conroy T, Hammel P, Hebbar M, Abdelghani MB, Wei AC, Raoul JL, Chone L, Francois E, Artru P, Biagi JJ, Lecomte T, Assenat E, Faroux R, Ychou M, Volet J, Sauvanet A, Jouffroy-Zeller C, RAT P, Castan F, Bachet JB. Unicancer GI PRODIGE 24/CCTG PA.6 trial: A multicenter international randomized phase III trial of adjuvant mFOLFIRINOX versus gemcitabine (gem) in patients with resected pancreatic ductal adenocarcinomas. J Clin Oncol. 2018;36:suppl; abstr LBA4001. [RCA] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 7. | Maisonneuve P, Lowenfels AB. Risk factors for pancreatic cancer: a summary review of meta-analytical studies. Int J Epidemiol. 2015;44:186-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 291] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 8. | Connor AA, Gallinger S. Hereditary Pancreatic Cancer Syndromes. Surg Oncol Clin N Am. 2015;24:733-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 9. | Blanco A, de la Hoya M, Osorio A, Diez O, Miramar MD, Infante M, Martinez-Bouzas C, Torres A, Lasa A, Llort G, Brunet J, Graña B, Perez Segura P, Garcia MJ, Gutiérrez-Enríquez S, Carracedo Á, Tejada MI, Velasco EA, Calvo MT, Balmaña J, Benitez J, Caldés T, Vega A. Analysis of PALB2 gene in BRCA1/BRCA2 negative Spanish hereditary breast/ovarian cancer families with pancreatic cancer cases. PLoS One. 2013;8:e67538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 10. | Tischkowitz M, Xia B. PALB2/FANCN: recombining cancer and Fanconi anemia. Cancer Res. 2010;70:7353-7359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 147] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 11. | Grant RC, Selander I, Connor AA, Selvarajah S, Borgida A, Briollais L, Petersen GM, Lerner-Ellis J, Holter S, Gallinger S. Prevalence of germline mutations in cancer predisposition genes in patients with pancreatic cancer. Gastroenterology. 2015;148:556-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 237] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 12. | Stoffel EM. Screening in GI Cancers: The Role of Genetics. J Clin Oncol. 2015;33:1721-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Holter S, Borgida A, Dodd A, Grant R, Semotiuk K, Hedley D, Dhani N, Narod S, Akbari M, Moore M, Gallinger S. Germline BRCA Mutations in a Large Clinic-Based Cohort of Patients With Pancreatic Adenocarcinoma. J Clin Oncol. 2015;33:3124-3129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 304] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 14. | Lucas AL, Frado LE, Hwang C, Kumar S, Khanna LG, Levinson EJ, Chabot JA, Chung WK, Frucht H. BRCA1 and BRCA2 germline mutations are frequently demonstrated in both high-risk pancreatic cancer screening and pancreatic cancer cohorts. Cancer. 2014;120:1960-1967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 15. | Prevalence and penetrance of BRCA1 and BRCA2 mutations in a population-based series of breast cancer cases. Anglian Breast Cancer Study Group. Br J Cancer. 2000;83:1301-1308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 459] [Cited by in RCA: 487] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 16. | Couch FJ, Johnson MR, Rabe KG, Brune K, de Andrade M, Goggins M, Rothenmund H, Gallinger S, Klein A, Petersen GM, Hruban RH. The prevalence of BRCA2 mutations in familial pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2007;16:342-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 207] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 17. | Pihlak R, Valle JW, McNamara MG. Germline mutations in pancreatic cancer and potential new therapeutic options. Oncotarget. 2017;8:73240-73257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 18. | Rustgi AK. Familial pancreatic cancer: genetic advances. Genes Dev. 2014;28:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 19. | Klein AP. Genetic susceptibility to pancreatic cancer. Mol Carcinog. 2012;51:14-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 165] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 20. | Borg A, Sandberg T, Nilsson K, Johannsson O, Klinker M, Måsbäck A, Westerdahl J, Olsson H, Ingvar C. High frequency of multiple melanomas and breast and pancreas carcinomas in CDKN2A mutation-positive melanoma families. J Natl Cancer Inst. 2000;92:1260-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 233] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 21. | Rutter JL, Bromley CM, Goldstein AM, Elder DE, Holly EA, Guerry D 4th, Hartge P, Struewing JP, Hogg D, Halpern A, Sagebiel RW, Tucker MA. Heterogeneity of risk for melanoma and pancreatic and digestive malignancies: a melanoma case-control study. Cancer. 2004;101:2809-2816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Permuth-Wey J, Egan KM. Family history is a significant risk factor for pancreatic cancer: results from a systematic review and meta-analysis. Fam Cancer. 2009;8:109-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 147] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 23. | Salo-Mullen EE, O'Reilly EM, Kelsen DP, Ashraf AM, Lowery MA, Yu KH, Reidy DL, Epstein AS, Lincoln A, Saldia A, Jacobs LM, Rau-Murthy R, Zhang L, Kurtz RC, Saltz L, Offit K, Robson ME, Stadler ZK. Identification of germline genetic mutations in patients with pancreatic cancer. Cancer. 2015;121:4382-4388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 160] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 24. | The European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC). Familial Pancreatic Cancer Entry Criteria 2017. Available from: URL: https://www.lctu.org.uk/LCTU_NET/Frontend/Default.aspx?Data=W1tRMjl1ZEdWdWRFbEVdXVtNakE0XVtbYkc5allXeGxdXVtPUT09XQ%3d%3d. |
| 25. | Daly MB, Pilarski R, Berry M, Buys SS, Farmer M, Friedman S, Garber JE, Kauff ND, Khan S, Klein C, Kohlmann W, Kurian A, Litton JK, Madlensky L, Merajver SD, Offit K, Pal T, Reiser G, Shannon KM, Swisher E, Vinayak S, Voian NC, Weitzel JN, Wick MJ, Wiesner GL, Dwyer M, Darlow S. NCCN Guidelines Insights: Genetic/Familial High-Risk Assessment: Breast and Ovarian, Version 2.2017. J Natl Compr Canc Netw. 2017;15:9-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 362] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 26. | Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW; American College of Gastroenterology. ACG clinical guideline: Genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol. 2015;110:223-62; quiz 263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 957] [Cited by in RCA: 1090] [Article Influence: 109.0] [Reference Citation Analysis (0)] |
| 27. | National Guideline Alliance (UK). Pancreatic cancer in adults: diagnosis and management, 2018. Available from: URL: http://europepmc.org/books/NBK487233;jsessionid=37B8932982158043DBFA70D1C93721D7. |
| 28. | Shindo K, Yu J, Suenaga M, Fesharakizadeh S, Cho C, Macgregor-Das A, Siddiqui A, Witmer PD, Tamura K, Song TJ, Navarro Almario JA, Brant A, Borges M, Ford M, Barkley T, He J, Weiss MJ, Wolfgang CL, Roberts NJ, Hruban RH, Klein AP, Goggins M. Deleterious Germline Mutations in Patients With Apparently Sporadic Pancreatic Adenocarcinoma. J Clin Oncol. 2017;35:3382-3390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 328] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 29. | Hu C, Hart SN, Polley EC, Gnanaolivu R, Shimelis H, Lee KY, Lilyquist J, Na J, Moore R, Antwi SO, Bamlet WR, Chaffee KG, DiCarlo J, Wu Z, Samara R, Kasi PM, McWilliams RR, Petersen GM, Couch FJ. Association Between Inherited Germline Mutations in Cancer Predisposition Genes and Risk of Pancreatic Cancer. JAMA. 2018;319:2401-2409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 400] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 30. | Robson ME, Bradbury AR, Arun B, Domchek SM, Ford JM, Hampel HL, Lipkin SM, Syngal S, Wollins DS, Lindor NM. American Society of Clinical Oncology Policy Statement Update: Genetic and Genomic Testing for Cancer Susceptibility. J Clin Oncol. 2015;33:3660-3667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 438] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 31. | Domchek SM, Bradbury A, Garber JE, Offit K, Robson ME. Multiplex genetic testing for cancer susceptibility: out on the high wire without a net? J Clin Oncol. 2013;31:1267-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 32. | Roberts NJ, Norris AL, Petersen GM, Bondy ML, Brand R, Gallinger S, Kurtz RC, Olson SH, Rustgi AK, Schwartz AG, Stoffel E, Syngal S, Zogopoulos G, Ali SZ, Axilbund J, Chaffee KG, Chen YC, Cote ML, Childs EJ, Douville C, Goes FS, Herman JM, Iacobuzio-Donahue C, Kramer M, Makohon-Moore A, McCombie RW, McMahon KW, Niknafs N, Parla J, Pirooznia M, Potash JB, Rhim AD, Smith AL, Wang Y, Wolfgang CL, Wood LD, Zandi PP, Goggins M, Karchin R, Eshleman JR, Papadopoulos N, Kinzler KW, Vogelstein B, Hruban RH, Klein AP. Whole Genome Sequencing Defines the Genetic Heterogeneity of Familial Pancreatic Cancer. Cancer Discov. 2016;6:166-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 278] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 33. | Shah PD, Nathanson KL. Application of Panel-Based Tests for Inherited Risk of Cancer. Annu Rev Genomics Hum Genet. 2017;18:201-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Piccirillo JF, Tierney RM, Costas I, Grove L, Spitznagel EL. Prognostic importance of comorbidity in a hospital-based cancer registry. JAMA. 2004;291:2441-2447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 889] [Cited by in RCA: 945] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 35. | The European Registry of Hereditary Pancreatitis and Familial Pancreatic Cancer (EUROPAC). Familial Pancreatic Cancer Entry Criteria. Available from: URL: https://www.lctu.org.uk/LCTU_NET/Frontend/?Data=W1tiRzlqWVd4bF1dW09RPT1d. |
| 36. | Department of Health. 2010 to 2015 government policy: harmful drinking. Available from: URL: https://www.gov.uk/government/publications/2010-to-2015-government-policy-harmful-drinking/2010-to-2015-government-policy-harmful-drinking. |
| 37. | Cancer Research UK. Bowel cancer statistics 2017. Available from: URL: http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/bowel-cancer#heading-Zero. |
| 38. | Poruk KE, Firpo MA, Adler DG, Mulvihill SJ. Screening for pancreatic cancer: why, how, and who? Ann Surg. 2013;257:17-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 185] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 39. | Fong PC, Boss DS, Yap TA, Tutt A, Wu P, Mergui-Roelvink M, Mortimer P, Swaisland H, Lau A, O'Connor MJ, Ashworth A, Carmichael J, Kaye SB, Schellens JH, de Bono JS. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med. 2009;361:123-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3070] [Cited by in RCA: 2864] [Article Influence: 179.0] [Reference Citation Analysis (0)] |
| 40. | Lee JM, Ledermann JA, Kohn EC. PARP Inhibitors for BRCA1/2 mutation-associated and BRCA-like malignancies. Ann Oncol. 2014;25:32-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 268] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 41. | Ledermann J, Harter P, Gourley C, Friedlander M, Vergote I, Rustin G, Scott CL, Meier W, Shapira-Frommer R, Safra T, Matei D, Fielding A, Spencer S, Dougherty B, Orr M, Hodgson D, Barrett JC, Matulonis U. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer: a preplanned retrospective analysis of outcomes by BRCA status in a randomised phase 2 trial. Lancet Oncol. 2014;15:852-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 984] [Cited by in RCA: 1121] [Article Influence: 101.9] [Reference Citation Analysis (0)] |
| 42. | Mirza MR, Monk BJ, Herrstedt J, Oza AM, Mahner S, Redondo A, Fabbro M, Ledermann JA, Lorusso D, Vergote I, Ben-Baruch NE, Marth C, Mądry R, Christensen RD, Berek JS, Dørum A, Tinker AV, du Bois A, González-Martín A, Follana P, Benigno B, Rosenberg P, Gilbert L, Rimel BJ, Buscema J, Balser JP, Agarwal S, Matulonis UA; ENGOT-OV16/NOVA Investigators. Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. N Engl J Med. 2016;375:2154-2164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1483] [Cited by in RCA: 1858] [Article Influence: 206.4] [Reference Citation Analysis (0)] |
| 43. | Kaufman B, Shapira-Frommer R, Schmutzler RK, Audeh MW, Friedlander M, Balmaña J, Mitchell G, Fried G, Stemmer SM, Hubert A, Rosengarten O, Steiner M, Loman N, Bowen K, Fielding A, Domchek SM. Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation. J Clin Oncol. 2015;33:244-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1153] [Cited by in RCA: 1328] [Article Influence: 120.7] [Reference Citation Analysis (0)] |
| 44. | Lowery MA, Kelsen DP, Capanu M, Smith SC, Lee JW, Stadler ZK, Moore MJ, Kindler HL, Golan T, Segal A, Maynard H, Hollywood E, Moynahan M, Salo-Mullen EE, Do RKG, Chen AP, Yu KH, Tang LH, O'Reilly EM. Phase II trial of veliparib in patients with previously treated BRCA-mutated pancreas ductal adenocarcinoma. Eur J Cancer. 2018;89:19-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 125] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 45. | Lohse I, Borgida A, Cao P, Cheung M, Pintilie M, Bianco T, Holter S, Ibrahimov E, Kumareswaran R, Bristow RG, Tsao MS, Gallinger S, Hedley DW. BRCA1 and BRCA2 mutations sensitize to chemotherapy in patient-derived pancreatic cancer xenografts. Br J Cancer. 2015;113:425-432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 46. | Kibble A, Al-Shamahi A, Kuennemann K, Marqués F, Tremosa L, Cole P. Highlights from the 52nd Annual Meeting of the American Society of Clinical Oncology (ASCO) (June 3-7, 2016 - Chicago, Illinois, USA). Drugs Today (Barc). 2016;52:407-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 47. | Golan T, Kanji ZS, Epelbaum R, Devaud N, Dagan E, Holter S, Aderka D, Paluch-Shimon S, Kaufman B, Gershoni-Baruch R, Hedley D, Moore MJ, Friedman E, Gallinger S. Overall survival and clinical characteristics of pancreatic cancer in BRCA mutation carriers. Br J Cancer. 2014;111:1132-1138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 335] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 48. | Illés D, Terzin V, Holzinger G, Kosár K, Róka R, Zsóri G, Ábrahám G, Czakó L. New-onset type 2 diabetes mellitus--A high-risk group suitable for the screening of pancreatic cancer? Pancreatology. 2016;16:266-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 49. | Yuan C, Morales-Oyarvide V, Babic A, Clish CB, Kraft P, Bao Y, Qian ZR, Rubinson DA, Ng K, Giovannucci EL, Ogino S, Stampfer MJ, Gaziano JM, Sesso HD, Cochrane BB, Manson JE, Fuchs CS, Wolpin BM. Cigarette Smoking and Pancreatic Cancer Survival. J Clin Oncol. 2017;35:1822-1828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 79] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 50. | Becker AE, Hernandez YG, Frucht H, Lucas AL. Pancreatic ductal adenocarcinoma: risk factors, screening, and early detection. World J Gastroenterol. 2014;20:11182-11198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 200] [Cited by in RCA: 230] [Article Influence: 20.9] [Reference Citation Analysis (5)] |
| 51. | Cancer Research UK. Pancreatic cancer incidence statistics. Available from: URL: http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/pancreatic-cancer/incidence#heading-One.. |