Published online Apr 10, 2016. doi: 10.4253/wjge.v8.i7.330
Peer-review started: November 10, 2015
First decision: December 18, 2015
Revised: January 1, 2016
Accepted: January 29, 2016
Article in press: January 31, 2016
Published online: April 10, 2016
Processing time: 147 Days and 15.8 Hours
AIM: To investigate factors related to recurrence following en bloc resection using endoscopic submucosal dissection (ESD) in patients with early gastric cancer (EGC).
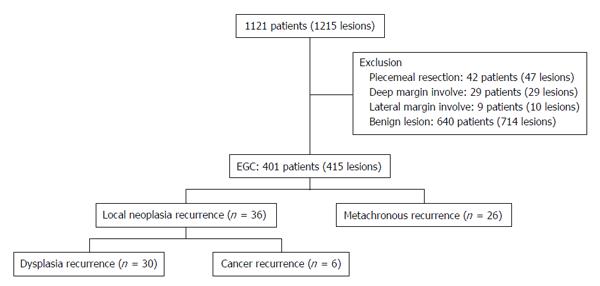
METHODS: A total of 1121 patients (1215 lesions) who had undergone ESD for gastric neoplasia between April 2003 and May 2010 were retrospectively reviewed. Data from 401 patients (415 lesions) were analyzed, following the exclusion of those who underwent piecemeal resection, with deep resection margin invasion or lateral margin infiltration, and diagnosed with benign lesions.
RESULTS: Local recurrence after en bloc ESD was found in 36 cases (8.7%). Unclear resection margins, long procedure times, and narrow safety margins were identified as risk factors for recurrence. Lesions located in the upper third of the stomach showed a higher rate of recurrence than those located in the lower third of the stomach (OR = 2.9, P = 0.03). The probability of no recurrence for up to 24 mo was 79.9% in those with a safety resection margin ≤ 1 mm and 89.5% in those with a margin > 1 mm (log-rank test, P = 0.03).
CONCLUSION: Even in cases in which en bloc ESD is performed for EGC, local recurrence still occurs. To reduce local recurrences, more careful assessment will be needed prior to the implementation of ESD in cases in which the tumor is located in the upper third of the stomach. In addition, clear identification of tumor boundaries as well as the securing of sufficient safety resection margins will be important.
Core tip: Unclear resection margins, long procedure times, and narrow safety margins were identified as risk factors for recurrence following en bloc endoscopic submucosal dissection (ESD) for early gastric cancer. Lesions located in the upper third of the stomach demonstrated more recurrences than those located in the lower third of the stomach. To reduce local recurrences, more careful assessment will be needed prior to the implementation of ESD in cases in which the tumor is located in the upper third of the stomach. In addition, clear identification of tumor boundaries as well as the securing of sufficient safety resection margins will be important.
- Citation: Lee JY, Cho KB, Kim ES, Park KS, Lee YJ, Lee YS, Jang BK, Chung WJ, Hwang JS. Risk factors for local recurrence after en bloc endoscopic submucosal dissection for early gastric cancer. World J Gastrointest Endosc 2016; 8(7): 330-337
- URL: https://www.wjgnet.com/1948-5190/full/v8/i7/330.htm
- DOI: https://dx.doi.org/10.4253/wjge.v8.i7.330
As regular national gastric cancer screening via endoscopy is being implemented in South Korea with an increased interest in health, findings of early gastric cancer (EGC) and precancerous lesions are increasing rapidly[1,2]. In addition, due to advances in the development of endoscopy-related tools and equipment and improvements in the procedural skills of doctors, performing endoscopic treatment for EGC is getting easier[3]. As a result, existing endoscopic mucosal resection (EMR) has led to significant progress in endoscopic submucosal dissection (ESD), in terms of resection techniques, and regardless of the size of the lesions, en bloc resection has become possible[4].
The classic EMR method is a simple procedure, but it has limitations in that the ratio of en bloc resection to complete resection decreases depending on the size of the lesion[5,6]. In the contrast, the ESD method is a relatively complex procedure with a high level of difficulty, but it has a higher rate of en bloc resection than the EMR method, with the capacity to perform accurate post-resection pathological assessment, and it has recently become widely available as a treatment for EGC[5-8]. In endoscopic resection, accomplishing reconstruction of dissected tissues when the resection is performed in a piecemeal fashion and determining whether complete resection of the lesion has been achieved is difficult, and this results in higher rates of local recurrence. Therefore, en bloc resection is being suggested as the standard method of ESD as it increases the accuracy of pathological assessment of complete resection and lowers the rate of local recurrence[9]. Incomplete resection procedures have been identified as an independent factor that increases the risk of local recurrence[10], but although en bloc resection has been practiced, there have been very few studies on the risk factors associated with local recurrence after en bloc resection. To that end, the aim of the current study was to investigate factors related to local recurrence in patients with EGC who underwent en bloc resection via ESD.
The medical records of 1121 patients (1215 lesions) who had undergone ESD for the treatment of gastric neoplasia between April 2003 and May 2010 at Keimyung University Dongsan Hospital (Daegu, South Korea) were retrospectively reviewed. Because we aimed to evaluate the risk factors for local recurrence after en bloc resection only and to analyze the risk factors depending on the safety resection margin, patients who underwent partial resection, with deep resection margin invasion or lateral margin infiltration, and diagnosed with benign lesions were excluded. Finally, data from 401 patients (415 lesions) were analyzed (Figure 1). Written informed consent was obtained from all patients. This study was approved by the Institutional Review Board of the Keimyung University Dongsan Medical Center, South Korea (DSMC 2015-10-047).
The ESD procedure was performed following a standard method. First the boundaries of the lesions were clarified using a solution of indigo carmine diluted to 10 times its volume, and the margins were marked with a 5 mm space from the boundaries of the lesions using an argon plasma laser connected to an ERBE VIO 300D electrosurgical unit (ERBE United States, Marietta, GA, United States). For submucosal injection, a solution was used consisting of hypertonic saline solution 100 mL, 1:1000 epinephrine 1 mL, and indigo carmine 1 mL. The incision knife was connected to the ERBE VIO 300D electrosurgical unit, a flex knife (Olympus, Tokyo, Japan) was used in mucosal incision, and the IT-2 knife (Olympus, Tokyo, Japan) was used for most submucosal dissection, but in some cases, a hook knife (Olympus, Tokyo, Japan) was used as well. Most procedures were carried out in Endocut I mode (Effect 2), and in some portions containing blood vessels, forced coagulation mode (Effect 1) was utilized.
For histopathological examination, resected specimens were sectioned perpendicularly at 2-mm intervals. The EGC location was classified into upper third, middle third, and lower third according to the location of the center point. The gross type of EGC was classified into type I (protruded type), type II (superficial type), and type III (excavated type) in accordance with the classification methods of the Japan Gastroenterological Endoscopy Society, and type II was subdivided again into type IIa (superficial elevated), type IIb (flat type), and type IIc (superficial depressed type)[11]. In cases in which various shapes were mixed in one lesion, it was recorded as the mixed type. Based on the histological findings, tissues of the lesion were classified into differentiated type adenocarcinoma (well or moderately differentiated adenocarcinoma) and undifferentiated type adenocarcinoma (poorly differentiated or signet ring cell adenocarcinoma). Tumor involvement in the lateral and deep margins, lymphatic and vascular involvement, and the presence of submucosal invasion was assessed. In cases of submucosal infiltration, invasion depth was measured and quantified.
The following clinical variables were investigated: Patient age, sex, gross tumor type, en bloc resection rate, location, size, histology, procedure time, safety margin, local neoplasia recurrence rate, and local cancer recurrence rate.
En bloc resection was defined as a resection in a single piece, whereas piecemeal resection was conducted in multiple pieces. Complete resection was defined as complete reconstruction of the lesion with negative deep and lateral margins with no lymphovascular involvement. The sizes of lesions were categorized into less than 20 mm, 21-30 mm, 31-40 mm, and over 40 mm. When malignant cells were found from the resection site within 3 mo after endoscopic removal of gastric carcinoma, the case was defined as incomplete resection, and when malignant cells or dysplastic cells (low grade, high grade) were found from the resection site during follow-up examinations after 3 mo, the case was defined as local recurrence of neoplasia. When only malignant cells were found from the resection site, the case was defined as local cancer recurrence. In addition, when neoplasia (dysplasia or malignant) was found from a site other than the resection site during follow-up observation, the case was defined as metachronous recurrence. Procedure time was defined as the time from the start of marking to complete removal of the tumor. Safety margins were defined as the distance between the lesion and the edges of the cuts around the resected specimen.
Patients were followed up with endoscopic examinations and biopsy at 3, 6, 12 and 24 mo after ESD. To detect local recurrence or metachronous cancer, biopsy was performed at the treatment-related scar in the case of any suspicious abnormalities. The cumulative neoplasia recurrence-free rate was estimated.
SPSS software version 18.0 for Windows (SPSS, Inc., Chicago, IL, United States) was used for statistical analysis. For comparison of continuous variables between two groups, the independent samples t-test was used, while for comparison of frequency variables, the χ2 test was used through cross analysis. Continuous variables were presented as means ± SD, and count variables were presented in the forms of frequency and percentage. Multivariate analysis was performed using binary logistic regression methods. Cumulative recurrence rates and recurrence times were calculated by the Kaplan-Meier method, and they were compared with each other using a log-rank test. A P value less than 0.05 was considered statistically significant. The statistical methods of this study were reviewed by Lee YJ and Lee YS.
The mean age of patients was 64.2 ± 9.8 years and 291 (70.1) patients were men. For the gross type of tumor, 146 (35.2%) cases were type IIa and this was the most frequent type. Regarding the location of lesions, 271 (65.0%) patients had lesions in the lower third of the stomach, representing the highest frequency, followed by 129 (30.9%) patients with lesions in the mid-third of the stomach, and 15 (3.6%) patients with lesions in the upper third of stomach. Regarding the size of tumors removed by ESD, tumors ≤ 20 mm were found in 110 (28.0%) cases, tumors 21-30 mm were found in 77 (18.8%) cases, tumors 31-40 mm were found in 122 (29.4%) cases, and tumors over 40 mm were found in 100 (24.1%) cases. Histologically, well differentiated adenocarcinoma and moderately differentiated adenocarcinoma were observed in 195 (47.0%) and 180 (43.4%) cases, respectively, constituting ≥ 90%. The mean follow-up period for these patients was 19.7 mo (Table 1).
| No. of lesions | |
| n = 415 | |
| Age, yr (mean ± SD) | 64.2 ± 9.8 |
| Sex, n (%) | |
| Male | 291 (70.1) |
| Female | 124 (29.9) |
| Gross type of tumor, n (%) | |
| Protruded (I) | 29 (7.0) |
| Superficial elevated (IIa) | 146 (35.2) |
| Flat (IIb) | 76 (18.3) |
| Superficial depressed (IIc) | 134 (32.3) |
| Excavated (III) | 2 (0.5) |
| Mixed | 28 (6.7) |
| En bloc resection, n (%) | 415 (100) |
| Piecemeal resection, n (%) | 0 (0) |
| Tumor location, n (%) | |
| Upper | 15 (3.6) |
| Mid | 129 (30.9) |
| Lower | 271 (65.0) |
| Tumor size, n (%) | |
| ≤ 20 mm | 116 (28.0) |
| 21-30 mm | 77 (18.8) |
| 31-40 mm | 122 (29.4) |
| > 40 mm | 100 (24.1) |
| Histology, n (%) | |
| Well differentiated | 195 (47.0) |
| Moderate differentiated | 180 (43.4) |
| Poorly differentiated | 30 (7.2) |
| Signet ring cell | 10 (2.4) |
| Follow-up period, mo (mean ± SD) | 19.7 ± 17.5 |
Local neoplasia recurrence was observed in 36 (8.7%) cases, but there was no significant difference in age at the time of diagnosis, sex, tumor size, location, or degree of differentiation when compared to the non-recurrence group (Table 2). However, there were many recurrences in cases in which tumors had ill-defined margins (33.3% vs 17.4%, P = 0.02), long procedure times (63.5 min vs 48.8 min, P = 0.02), and narrow safety resection margins (3.1 mm vs 4.2 mm, P = 0.03) (Table 2). The performance of multivariate analysis revealed that ill-defined tumor margin was the element factor that related to local neoplasia recurrence (P = 0.03) (Table 2).
| Recurrence | No recurrence | Univariate | Multivariate | |
| n = 36 | n = 379 | P value | P value | |
| Age, yr (mean ± SD) | 66.7 ± 9.0 | 64.0 ± 9.9 | 0.11 | |
| Male/female | 25/11 | 266/113 | 0.93 | |
| Tumor margin, n (%) | 0.02 | 0.03 | ||
| Well-defined | 24 (66.7) | 313 (82.6) | ||
| Ill-defined | 12 (33.3) | 66 (17.4) | ||
| Tumor size, n (%) | 0.62 | |||
| ≤ 20 mm | 9 (25.0) | 107 (28.2) | ||
| 21-30 mm | 6 (16.7) | 71 (18.7) | ||
| 31-40 mm | 14 (38.9) | 108 (28.5) | ||
| > 40 mm | 7 (19.4) | 93 (24.5) | ||
| Tumor location, n (%) | 0.05 | |||
| Upper | 3 (8.3) | 12 (3.2) | ||
| Mid | 14 (38.9) | 115 (30.3) | ||
| Lower | 19 (52.8) | 252 (66.5) | ||
| Histology, n (%) | 0.7 | |||
| Well differentiated | 17 (47.2) | 178 (47.0) | ||
| Moderate differentiated | 13 (36.1) | 167 (44.1) | ||
| Poorly differentiated | 6 (16.7) | 24 (6.3) | ||
| Signet ring cell | 0 (0.0) | 10 (2.6) | ||
| Procedure time, min (mean ± SD) | 63.5 ± 56.9 | 48.8 ± 34.3 | 0.02 | 0.06 |
| Safety margin, mm (mean ± SD) | 3.1 ± 2.1 | 4.2 ± 2.9 | 0.03 | 0.05 |
When 1 mm was used as the reference value, 63 (15.2%) cases were found to have safety resection margins ≤ 1 mm. There was no difference in age at the time of diagnosis, sex, tumor size, location, or degree of differentiation between the two groups. Nevertheless, the group with safety resection margins ≤ 1 mm was found to have more lesions located in the upper third and mid-third of the stomach (P < 0.0001) and had longer operation times (P = 0.04) (Table 3). Multivariate analysis revealed that the patients with lesions located in the upper third of the stomach demonstrated more recurrences than those with lesions located in the lower third of the stomach (OR = 2.900, 95%CI: 1.110-7.579, P = 0.03) (Table 4). Designating 1 mm as the safety resection margin, there was no difference in recurrence of neoplasia, but there was more frequent recurrence of cancer (P = 0.006) (Table 5).
| Safety margin ≤1 mm | Safety margin > 1 mm | P value | |
| n = 63 | n = 352 | ||
| Age, yr (mean ± SD) | 65.9 ± 11.1 | 63.9 ± 9.5 | 0.14 |
| Male/female | 38/25 | 253/99 | 0.12 |
| Tumor margin, n (%) | 0.52 | ||
| Well-defined | 53 (84.1) | 284 (80.7) | |
| Ill-defined | 10 (15.9) | 68 (19.3) | |
| Tumor size, n (%) | 0.55 | ||
| ≤ 20 mm | 21 (33.3) | 95 (27.0) | |
| 21-30 mm | 12 (19.0) | 65 (18.5) | |
| 31-40 mm | 14 (22.2) | 108 (30.7) | |
| > 40 mm | 16 (25.4) | 84 (23.9) | |
| Tumor location, n (%) | < 0.0001 | ||
| Upper | 7 (11.1) | 8 (2.3) | |
| Mid | 31 (49.2) | 98 (27.8) | |
| Lower | 25 (39.7) | 246 (69.9) | |
| Histology, n (%) | 0.85 | ||
| Well differentiated | 32 (50.8) | 163 (46.3) | |
| Moderate differentiated | 24 (38.1) | 156 (44.3) | |
| Poorly differentiated | 3 (4.8) | 27 (7.7) | |
| Signet ring cell | 4 (6.3) | 6 (1.7) | |
| Procedure time, min (mean ± SD) | 58.9 ± 43.8 | 48.5 ± 35.4 | 0.04 |
| Multivariate analysis | ||
| Odds ratio (95%CI) | P value | |
| Location | ||
| Upper | 2.90 (1.11-7.58) | 0.03 |
| Mid | 1.10 (0.48-2.55) | 0.82 |
| Lower | 1 (ref) | |
| Ill-defined margin | 2.32 (1.00-4.96) | 0.03 |
| Safety margin ≤1 mm | Safety margin > 1 mm | P value | |
| n = 63 | n = 352 | ||
| Neoplasia recurrence | 9 (14.3) | 27 (7.7) | 0.09 |
| Cancer recurrence | 4 (6.3) | 2 (0.6) | 0.006 |
During the entire follow-up observation period, 6 cases (6/415, 1.4%) were observed of the recurrence of malignancy at the same site, and 26 cases (26/415, 6.3%) were observed of metachronous gastric carcinoma (Figure 1). In addition, the probability of no recurrence for up to 24 mo was 79.9% in those with safety resection margin ≤ 1 mm and 89.5% in those with margins that exceeded 1 mm, indicating that the local recurrence of neoplasia was observed more frequently in those with safety resection margins ≤ 1 mm, and the difference between the two groups was significant (P = 0.03) (Figure 2).
In cases of lesions larger than 20 mm, ESD offers far superior en bloc resection rates and very low local recurrence rates when compared with EMR[12]. In general, the results of ESD for lesions larger than 20 mm have demonstrated an en bloc resection rate of over 90% with little local recurrence, while EMR has demonstrated very low en bloc resection rates of about 60% in cases of lesions sized about 10 mm and 14%-40% for lesions sized about 20-30 mm, and the local recurrence rate is about 10%[13,14]. Regarding the en bloc resection rate, following the resection, determining complete resection with histological accuracy and thereby significantly reducing the occurrence of any situations that require unnecessary additional treatment, re-treatment, or surgical treatment due to local recurrence is possible. Due to these advantages, ESD is being used as a major treatment method for EGC.
The current study investigated the factors related to recurrence in patients with EGC who had undergone en bloc resection using ESD. Even in cases in which en bloc resection was performed, local recurrence of neoplasia was observed in 36 patients (8.7%). When a comparison was performed between the recurrence group and the non-recurrence group, the identified risk factors for recurrence included unclear resection margins, long procedure times, and narrow safety margins, whereas among the factors related to sufficient safety resection margins, it was found that the location of the tumor was an important factor. In particular, tumor location in the upper third of stomach was identified as having the greatest association with recurrence.
The visual tumor boundaries and safety resection margins of tumors had been identified as the risk factors for local recurrence. The introduction of ESD has increased the rates of en bloc resection and complete resection, but incomplete resection, in which resection margins are found to be positive in the post-ESD pathological testing, remains problematic. This results in cases in which the degree of horizontal invasion at the lesion is not assessed accurately and there is a failure to secure sufficient safety resection margins prior to performing the procedure[15-17]. In the current study as well, the group with visually unclear tumor margins showed a higher rate of post-ESD recurrences (33.3% vs 17.4%, P = 0.03), and more incidences of recurrent tumors were found among those with safety resection margins ≤ 1 mm. Thus, good visual observation of the boundaries of lesions and the securing of sufficient safety resection margins before performing the procedures would be helpful in reducing local recurrence. However, since it is better to attempt minimal incision in order to minimize the procedure time and complications, as possible, accurate diagnosis is required before performing ESD. There have been reports suggesting that in cases in which the boundaries of the tumor are unclear, a preoperative biopsy on the ambient area of the lesion could be useful[18,19], and the horizontal degree of invasion of the tumor could be assessed via chromoendoscopy[20] or narrow-band imaging magnifying endoscopy[21].
The most important factor that has effects on local recurrence following the implementation of EMR or ESD is whether complete resection is performed. Ono et al[22] reported that the rate of local recurrence was 2% in cases of complete resection, while in contrast, recurrence was found in 18% of 85 patients either who had incomplete resection or in whom it was impossible to make assessments. Isomoto et al[23] also reported that while only 0.2% of patients who underwent complete resection had experienced local recurrence, 10.3% of patients who had incomplete resection had been found to have local recurrence, indicating that the complete resection group had a statistically significant lower rate of local recurrence in comparison to the incomplete resection group. Takenaka et al[6] presented a study on factors affecting local recurrence following ESD. They reported no cases of local recurrence among lesions that had been completely resected, but patients who underwent incomplete resection had local recurrences. Statistical analysis had confirmed that incomplete resection and local recurrence had a very high level of correlation. The authors analyzed the factors that cause incomplete resection and identified tumor size ≥ 30 mm, tumor location in the mid-third or upper third, and any ulcer or ulcerative scar on the lesion as the risk factors that can cause incomplete resection. Imagawa et al[24] also reported that tumor location (upper third, 74% vs mid-third, 77% vs lower third, 91%, P < 0.05) and tumor size (> 20 mm, 59% vs < 20 mm, 89%, P < 0.0001) were important elements of complete resection. In our study, it was confirmed that the more lesions were located in the upper third, the more frequent local recurrences were. However, according to the results of our study, tumor size was identified as having no significant correlation with recurrence, and it was considered that the procedures were implemented after securing sufficient safety resection margins considering the risk of recurrence as the tumor sizes increased. The underlying causes of more frequent local recurrences when lesions are located in the upper third of the stomach are, first, when the tumor is located nearer to the upper third, the endoscopic approach becomes difficult, resulting in difficult setting of accurate boundaries; second, this region has unclear boundaries of the mucosa in many cases; and third, this area has a larger distribution of blood vessels than any other site, which causes frequent bleeding during the procedure[25]. The use of side-view endoscopes or multi-bending endoscopes can offer easy access to these sites, which is very helpful in performing the procedures[26].
A molecular pathological epidemiology approach, which analyzes tumor molecular pathology of resected tumors, can predict recurrence after ESD. Semba et al[27] reported that EGC demonstrating intestinal claudin-positive phenotype has a high risk of synchronous and metachronous gastric neoplasia. Hasuo et al[28] investigated the correlation between microsatellite instability (MSI) status and the incidence of metachronous recurrence after initial ESD. They demonstrated that patients with the MSI-type tumors showed a high incidence of metachronous recurrence within a 3-year observation period after initial ESD. These molecular approaches are expected to be of value for decisions regarding therapy and surveillance after ESD.
The advantage of the current study is that it was conducted in patients who underwent en bloc resection only, and those patients with deep and lateral resection margin invasion were excluded, so that we could analyze the risk factors depending on the safety resection margins. However, the study also has limitations in that the follow-up periods were different, as it was a retrospective study, and there were differences in the number of biopsies during the follow-up endoscopy.
In conclusion, even in cases in which en bloc resection using ESD is performed for EGC, local recurrence occurs. In terms of risk factors related to local recurrence, tumor location and the visual boundaries of the tumor are important. In order to reduce post-ESD local recurrences, more careful assessment will be needed prior to the implementation of ESD in cases in which the tumor is located in the upper third of the stomach. In addition, clear identification of tumor boundaries as well as the securing of sufficient safety resection margins will be important.
En bloc resection is suggested as the standard method of endoscopic submucosal dissection (ESD) as it increases the accuracy of pathological assessment of complete resection and lowers the ratio of local recurrence. However, although en bloc resection has been practiced, there are few studies regarding the risk factors associated with local recurrence after en bloc resection.
The authors aimed to investigate factors related to recurrence in patients who had undergone en bloc resection using ESD for early gastric cancer (EGC).
Unclear resection margins, long procedure times, and narrow safety margins were identified as risk factors for recurrence lesions located in the upper third of the stomach demonstrated more recurrences than those located in the lower third of the stomach.
Even in cases in which en bloc resection for ESD is performed, local recurrence occurs. Regarding risk factors related to local recurrence, tumor location and the visual boundaries of the tumor are important. In order to reduce post-ESD local recurrences, more careful assessment will be needed prior to the implementation of ESD in cases in which the tumor is located in the upper third of the stomach. In addition, clear identification of tumor boundaries as well as the securing of sufficient safety resection margins will be important as well.
EGC is defined as malignant tumor confined to the mucosa or the submucosa regardless of lymph node metastases. ESD is an endoscopic technique for the treatment of early gastrointestinal neoplasms allowing direct dissection of the submucosal layer of the lesion with en bloc resection.
This is a large retrospective study on risk factor for local recurrence after ESD of early gastric cancer. The topic is important and interesting.
P- Reviewer: Bordas JM, Ogino S, Sperti C S- Editor: Qi Y L- Editor: A E- Editor: Liu SQ
| 1. | Kim SG. Endoscopic Resection of Early Gastric Cancer. Korean J Gastroenterol. 2009;54:77. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 2. | Nam SY, Choi IJ, Park KW, Kim CG, Lee JY, Kook MC, Lee JS, Park SR, Lee JH, Ryu KW. Effect of repeated endoscopic screening on the incidence and treatment of gastric cancer in health screenees. Eur J Gastroenterol Hepatol. 2009;21:855-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 3. | Choi KD. Endoscopic resection of early gastric cancer. Korean J Med. 2011;81:40-46. |
| 4. | Ono H. Endoscopic submucosal dissection for early gastric cancer. Chin J Dig Dis. 2005;6:119-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Oka S, Tanaka S, Kaneko I, Mouri R, Hirata M, Kawamura T, Yoshihara M, Chayama K. Advantage of endoscopic submucosal dissection compared with EMR for early gastric cancer. Gastrointest Endosc. 2006;64:877-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 526] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 6. | Takenaka R, Kawahara Y, Okada H, Hori K, Inoue M, Kawano S, Tanioka D, Tsuzuki T, Yagi S, Kato J. Risk factors associated with local recurrence of early gastric cancers after endoscopic submucosal dissection. Gastrointest Endosc. 2008;68:887-894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 112] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 7. | Watanabe K, Ogata S, Kawazoe S, Watanabe K, Koyama T, Kajiwara T, Shimoda Y, Takase Y, Irie K, Mizuguchi M. Clinical outcomes of EMR for gastric tumors: historical pilot evaluation between endoscopic submucosal dissection and conventional mucosal resection. Gastrointest Endosc. 2006;63:776-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 165] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 8. | Nakamoto S, Sakai Y, Kasanuki J, Kondo F, Ooka Y, Kato K, Arai M, Suzuki T, Matsumura T, Bekku D. Indications for the use of endoscopic mucosal resection for early gastric cancer in Japan: a comparative study with endoscopic submucosal dissection. Endoscopy. 2009;41:746-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 109] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 9. | Jang JS, Lee EJ, Lee SW, Lee JH, Roh MH, Han SY, Choi SR, Jeong JS. Endoscopic submucosal dissection for early gastric cancer and gastric adenoma. Korean J Gastroenterol. 2007;49:356-363. |
| 10. | Park JC, Lee SK, Seo JH, Kim YJ, Chung H, Shin SK, Lee YC. Predictive factors for local recurrence after endoscopic resection for early gastric cancer: long-term clinical outcome in a single-center experience. Surg Endosc. 2010;24:2842-2849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 11. | Japanese Gastric Cancer Association. Japanese Classification of Gastric Carcinoma - 2nd English Edition. Gastric Cancer. 1998;1:10-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Ono H. Early gastric cancer: diagnosis, pathology, treatment techniques and treatment outcomes. Eur J Gastroenterol Hepatol. 2006;18:863-866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 13. | Miyata M, Yokoyama Y, Okoyama N, Joh T, Seno K, Sasaki M, Ohara H, Nomura T, Kasugai K, Itoh M. What are the appropriate indications for endoscopic mucosal resection for early gastric cancer? Analysis of 256 endoscopically resected lesions. Endoscopy. 2000;32:773-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 53] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Noda M, Kodama T, Atsumi M, Nakajima M, Sawai N, Kashima K, Pignatelli M. Possibilities and limitations of endoscopic resection for early gastric cancer. Endoscopy. 1997;29:361-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 15. | Tanabe H, Iwashita A, Haraoka S. Pathological evaluation concerning curability of endoscopic submucosal dissection of early gastric cancer including lesions with obscure margins. Stom Intest. 2006;41:53-66. |
| 16. | Mishima T, Miyake N, Chonan A. Incompletely resected case under the extended indication of endoscopic submucosal dissection for early gastric cancer. Stomach Intestine. 2008;43:33-43. |
| 17. | Nagahama T, Suketo S, Yorioka M. Current status of endoscopic submucosal dissection in early stomach cancer. Stomach Intestine. 2008;43:33-43. |
| 18. | Kakushima N, Ono H, Tanaka M, Takizawa K, Yamaguchi Y, Matsubayashi H. Factors related to lateral margin positivity for cancer in gastric specimens of endoscopic submucosal dissection. Dig Endosc. 2011;23:227-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Kang EJ, Cho JY, Lee TH, Jin SY, Cho WY, Bok JH, Kim HG, Kim JO, Lee JS, Lee IH. Frozen Section Biopsy to Evaluation of Obscure Lateral Resection Margins during Gastric Endoscopic Submucosal Dissection for Early Gastric Cancer. J Gastric Cancer. 2011;11:155-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 20. | Ida K, Hashimoto Y, Takeda S. Endoscopic diagnosis of gastric cancer with dye scattering. Am J Gastroenterol. 1975;63:316-320. |
| 21. | Nagahama T, Yao K, Maki S, Yasaka M, Takaki Y, Matsui T, Tanabe H, Iwashita A, Ota A. Usefulness of magnifying endoscopy with narrow-band imaging for determining the horizontal extent of early gastric cancer when there is an unclear margin by chromoendoscopy (with video). Gastrointest Endosc. 2011;74:1259-1267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 145] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 22. | Ono H, Kondo H, Gotoda T, Shirao K, Yamaguchi H, Saito D, Hosokawa K, Shimoda T, Yoshida S. Endoscopic mucosal resection for treatment of early gastric cancer. Gut. 2001;48:225-229. [PubMed] |
| 23. | Isomoto H, Shikuwa S, Yamaguchi N, Fukuda E, Ikeda K, Nishiyama H, Ohnita K, Mizuta Y, Shiozawa J, Kohno S. Endoscopic submucosal dissection for early gastric cancer: a large-scale feasibility study. Gut. 2009;58:331-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 470] [Cited by in RCA: 519] [Article Influence: 32.4] [Reference Citation Analysis (1)] |
| 24. | Imagawa A, Okada H, Kawahara Y, Takenaka R, Kato J, Kawamoto H, Fujiki S, Takata R, Yoshino T, Shiratori Y. Endoscopic submucosal dissection for early gastric cancer: results and degrees of technical difficulty as well as success. Endoscopy. 2006;38:987-990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Tsuji Y, Ohata K, Ito T, Chiba H, Ohya T, Gunji T, Matsuhashi N. Risk factors for bleeding after endoscopic submucosal dissection for gastric lesions. World J Gastroenterol. 2010;16:2913-2917. [PubMed] |
| 26. | Isshi K, Tajiri H, Fujisaki J, Mochizuki K, Matsuda K, Nakamura Y, Saito N, Narimiya N. The effectiveness of a new multibending scope for endoscopic mucosal resection. Endoscopy. 2004;36:294-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 27. | Semba S, Hasuo T, Satake S, Nakayama F, Yokozaki H. Prognostic significance of intestinal claudins in high-risk synchronous and metachronous multiple gastric epithelial neoplasias after initial endoscopic submucosal dissection. Pathol Int. 2008;58:371-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 28. | Hasuo T, Semba S, Li D, Omori Y, Shirasaka D, Aoyama N, Yokozaki H. Assessment of microsatellite instability status for the prediction of metachronous recurrence after initial endoscopic submucosal dissection for early gastric cancer. Br J Cancer. 2007;96:89-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |