Published online Jan 16, 2014. doi: 10.4253/wjge.v6.i1.27
Revised: November 27, 2013
Accepted: December 13, 2013
Published online: January 16, 2014
Processing time: 127 Days and 20 Hours
Behcet’s disease (BD) is a rare and life-long disorder characterized by inflammation of blood vessels throughout the body. BD was originally described in 1937 as a syndrome involving oral and genital ulceration in addition to ocular inflammation. Intestinal BD refers to colonic ulcerative lesions documented by objective measures in patients with BD. Many studies have shown that over 40% of BD patients have gastrointestinal complaints. Symptoms include abdominal pain, diarrhea, nausea, anorexia and abdominal distension. Although gastrointestinal symptoms are common, the demonstration of gastrointestinal ulcers is rare. This so-called intestinal BD accounts for approximately 1% of cases. There is no specific test for BD, and the diagnosis is based on clinical criteria. The manifestations of intestinal BD are similar to those of other colitis conditions such as Crohn’s disease or intestinal tuberculosis, thus, it is challenging for gastroenterologists to accurately diagnose intestinal BD in patients with ileo-colonic ulcers. However, giant ulcers distributed in the esophagus and ileocecal junction with gastrointestinal hemorrhage are rare in intestinal BD. Here, we present a case of untypical intestinal BD. The patient had recurrent aphthous ulceration of the oral mucosa, and esophageal and ileo-colonic ulceration, but no typical extra-intestinal symptoms. During examination, the patient had massive acute lower gastrointestinal bleeding. The patient underwent ileostomy after an emergency right hemicolectomy and partial ileectomy, and was subsequently diagnosed with incomplete-type intestinal BD by pathology. The literature on the evaluation and management of this condition is reviewed.
Core tip: We present a patient with fever, abdominal pain and skip ulcers accompanied by lower gastrointestinal hemorrhage. Although the patient had undergone a number of examinations, no diagnosis was made. The patient underwent emergency surgery due to unmanageable lower gastrointestinal hemorrhage. Pathology of the resected bowel containing ulcer lesions indicated ectasia and blood vessel hyperplasia. The patient was diagnosed with incomplete-type intestinal Behcet’s disease (BD). BD can influence any region of the gastrointestinal tract. It is more difficult to diagnose when intestinal BD is accompanied by multiple ulcers in various positions throughout the entire digestive tract.
- Citation: Wang ZK, Shi H, Wang SD, Liu J, Zhu WM, Yang MF, Liu C, Lu H, Wang FY. Confusing untypical intestinal Behcet’s disease: Skip ulcers with severe lower gastrointestinal hemorrhage. World J Gastrointest Endosc 2014; 6(1): 27-31
- URL: https://www.wjgnet.com/1948-5190/full/v6/i1/27.htm
- DOI: https://dx.doi.org/10.4253/wjge.v6.i1.27
Intestinal Behcet’s disease (BD) refers to colonic ulcerative lesions documented by objective measures in patients with BD. Bechguard first described gastrointestinal involvement in 1940[1]. Oshima et al[2] reported that over 40% of BD patients had gastrointestinal complaints. Symptoms included abdominal pain, diarrhea, nausea, anorexia and abdominal distension[2]. Although gastrointestinal symptoms are common, the demonstration of gastrointestinal ulcers is rare. This so-called intestinal BD accounts for approximately 1% of cases[3,4].
The manifestations of intestinal BD are similar to other colitis conditions such as Crohn’s disease or intestinal tuberculosis, therefore, it is challenging for gastroenterologists to accurately diagnose intestinal BD in patients with ileo-colonic ulcers. It is more difficult to diagnose when intestinal BD is accompanied by multiple ulcers in various positions throughout the entire digestive tract. Furthermore, giant skip ulcers and gastrointestinal hemorrhage are rare in intestinal BD. Here, we present a case of untypical intestinal BD. The patient had giant ulcers distributed in the esophagus and ileocecal junction accompanied by lower gastrointestinal hemorrhage, but no typical extra-intestinal symptoms.
A 47-year-old male presented to our hospital in October 2010 due to abdominal pain, fever and diarrhea. His abdominal pain was located in the epigastric region and lower right quadrant with no radiation. The pain was crampy and intermittent throughout the day. He could not recall what made the pain better or worse, but the symptoms had been present for approximately one year. In addition to fever and diarrhea the patient also experienced headache. His maximum temperature was 41 °C. Endoscopy showed multiple giant ulcers in the esophagus and ileocolonic region. His clinical diagnosis was documented as Crohn’s disease, and 5-aminosalicylate (mesalazine) 4.0 mg and prednisone 40 mg were administered orally. The above-mentioned symptoms gradually improved. However, his temperature rose when the dose of prednisone was tapered. The patient was referred to our hospital for further treatment.
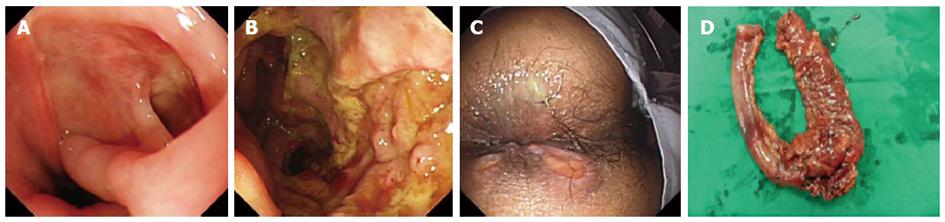
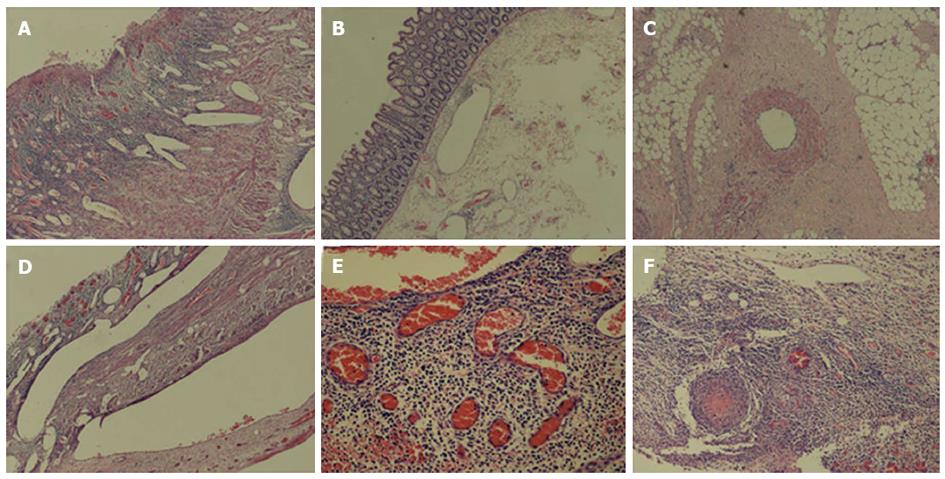
The patient’s past medical history consisted of recurrent oral aphthous ulcerations, folliculitis and facial acne-like lesions from 2009. He also had a history of chronic headaches. He denied ever using alcohol, tobacco products or illicit drugs. His family history was only significant for peptic ulcer disease and diabetes mellitus. On physical examination, his body temperature was 39.5 °C, heart rate was 102 bpm and arterial blood pressure was 126/72 mmHg. There was some aphthous ulceration on the oral mucous membrane and multiple acne-like lesions on both cheeks and the neck. His abdominal pain was located in the epigastric region and lower right quadrant without rebound tenderness. On examination of the crissum, no ulceration was observed. The results of clinical laboratory tests and examinations are shown in Table 1. Based on these results it was hoped to discriminate intestinal BD from Crohn’s disease. The patient was treated with oral prednisone 40 mg/d and a proton pump inhibitor. During further examination, the patient had massive acute lower gastrointestinal bleeding. Bleeding was located in the ileocecal region by emergent colonoscopy. A few oval ulcers were found around the crissum (Figure 1C). We were unable to achieve hemostasis by medical treatment. A surgical consult was obtained, and the patient underwent an emergency right hemicolectomy and partial ileectomy with ileostomy. During surgery, we observed that the wall of the cecum was thick and the lumen between the ileum and colon was filled with blood. Macroscopic examination of the resected material showed occasional discoloration in the serosa, mucosal edema, an ulcer (4 cm × 4 cm) and occasional necrosis in a segment 32 cm in length involving the ileocecal region (Figure 1D). On microscopic examination of the ulcer involving the serosa, there was mixed-type purulent cell infiltration rich in neutrophils, congestion and capillary proliferation. There was considerable thickening of some arterioles and venules, lymphocyte infiltration in and around the vessel wall, thrombus and recanalization in some vessels at the base of the ulcer (Figure 2). Treatment with oral prednisone 40 mg/d and thalidomide 300 mg/d was started after dermal sutures were removed. The abdominal symptoms and crissum ulcers gradually improved, and he was discharged 2 mo after admission.
| Clinical examinations | Results |
| Routine blood examination | White blood cells 14400/μL, red blood cells 408 × 104/μL, hemoglobin 11.6 g/dL, hematocrit 35.7%, platelets 220 × 104/μL, C-reactive protein 38.9 mg/dL, blood sedimentation 58 mm/h |
| Routine stool examination | White blood cells, 20-30/HP; red blood cells, filled visual fields |
| Blood biochemistry | Total protein, 8.6 g/dL; AST, 36 IU/L; ALT, 32 IU/L; LDH, 171 IU/L; and total bilirubin, 0.2 mg/dL |
| Bacteriologic culture of blood, urine, and stool | Negative |
| Serum antinuclear antibody and antituberculosis antibody | Negative |
| PPD skin test and T-spot test | Negative |
| Pathergy test | Positive |
| Gastroscopy | Giant ulceration in the inferior extremity of the esophagus (Figure 1A) |
| Colonoscopy | A large ulcer in the ileo-cecal junction (Figure 1B) |
| Pathological examination of the endoscopic biopsy specimen | Nonspecific ulceration |
| Abdominal CT imaging | Thickening of the intestinal canal of the ascending colon and ileocecal region |
| Whole gastrointestinal barium meal examination | Inflammatory changes in the ascending colon and ileocecal region |
BD is a rare and life-long disorder characterized by inflammation of blood vessels throughout the body[5]. BD was originally described in 1937 as a syndrome involving oral and genital ulceration in addition to ocular inflammation[6]. Since then, BD has been recognized and many other manifestations have been added to the original triad. The etiology of BD is unknown. To date, research has revealed that infectious, autoimmune and genetic mechanisms may cause this disease[7]. BD mostly affects children and young adults between the second and fourth decades of life[8]. Those affected before the age of 25 years (early onset) and males have been shown to have more severe disease symptoms[9].
Intestinal BD occurs most frequently along the ancient Silk Road which extends from the Far East to the Mediterranean basin. The prevalence varies widely among geographic locations. In Japan, the prevalence rate is 10 in 100000, in Saudi Arabia it is 20 in 100000, and in Northern Europe and in the United States it is only 0.3 per 100000. The male-to-female ratio also varies by geographic location. Men predominate in Egypt, Turkey, Israel, and Iran, whereas women predominate in Europe, the United States, and Japan. The age of onset can range from infancy to the 70s, although the highest frequency occurs in the third and fourth decades. Involvement of the gastrointestinal tract is variable in different populations, being more common in Japan (50%-60%) and less common in the Mediterranean basin, including Turkey (0%-5%)[1]. Although the reasons for this peculiar geographic distribution of intestinal BD are unknown, it may provide clues for the elucidation of putative etiological agents or genetic factors that might be associated with intestinal BD.
BD can influence any region of the gastrointestinal tract. The mouth is the most common gastrointestinal site affected by BD followed by the ileocecal region[10]. Intestinal lesions are located on the antimesenteric side. Gastrointestinal symptoms related to BD include abdominal pain, nausea and vomiting. Some rare symptoms are present in emergency conditions, such as intestinal perforation or bleeding[11,12].
There is no specific test for BD, and the diagnosis is based on clinical criteria. In Japan, diagnostic criteria for BD have been established by the BD Research Committee (Table 2 and Table 3)[13,14]. Based on these criteria, the present case was a suspected type of BD. The patient was subsequently confirmed to have incomplete-type intestinal BD by pathology. It can be difficult to diagnose untypical intestinal BD. Moreover, intestinal BD manifests mainly in the terminal ileum, and esophageal lesions are rare. The patient had esophageal and terminal ileum ulcers accompanied by recurrent oral aphthous ulcerations, similar to Crohn’s disease. Inflammatory bowel diseases should be kept in mind in the differential diagnosis of intestinal BD. Although International Study Group criteria for BD accurately distinguish between BD and Crohn’s disease[15], there are some common features. Similar to Crohn’s disease, BD manifests as discrete intestinal ulcers and discontinuous bowel involvement. Both of these diseases share extra-intestinal manifestations, such as arthritis and uveitis. Rectal sparing is common in both diseases. Intestinal lesions in Crohn’s disease tend to be longitudinal ulcers with a cobblestone appearance, while those in BD are round and oval “punched-out” ulcers. Moreover, epithelioid granuloma is one of the pathological characteristics of Crohn’s disease, whereas it is uncommon in intestinal BD. Another feature of Behcet’s colitis is lymphocyte venulitis, which is a type of vasculitis. Despite these differences, it can be difficult to differentiate between these two diseases.
| Maior |
| Recurrent aphthous ulceration of the oral mucous membrane |
| Skin lesion |
| Erythema nodosum |
| Subcutaneous thrombophlebitis |
| Folliculitis, acne-like lesion |
| Cutaneous hyperirritability |
| Eye lesion |
| Iridocyclitis |
| Chorioretinitis, retinouveitis |
| Definite history of chorioretinitis of retinouveitis |
| Genital ulcer |
| Minor |
| Arthritis without deformity and ankylosis |
| Gastrointestinal lesion characterized by ileocecal ulcers |
| Epididymitis |
| Vascular lesion |
| Central nervous system symptoms |
| Diagnosis |
| Complete type:4 major features |
| Incomplete type: |
| 3 major features |
| Major + 2 minor features |
| Typical ocular symptom + 1 major or 2 minor features |
| Suspected type: |
| 2 major features |
| 1 major + 2 minor |
| Diagnosis of intestinal Behcet’s disease can be made if |
| There is a typical oval-shaped large ulcer in the terminal ileum or |
| There are ulcerations or inflammation in the small or large intestine; |
| And clinical findings meet the diagnostic criteria of Behcet’s disease |
Ten percent of patients with BD accompanied by intestinal involvement require surgical treatment. The complications most frequently requiring surgery are perforation and bleeding. The recurrence rate after surgery has been reported to be 40%-87.5% and frequently appears at the anastomosis site. If suitable medical treatment is given after surgery, this condition can be improved. Thalidomide is a synthetic glutamic acid derivative first introduced in 1956 in Germany as an over-the-counter medication. The Food and Drug Administration approved its use in the treatment of erythema nodosum leprosum. Furthermore, it was shown to be effective in unresponsive dermatological conditions such as actinic prurigo, adult Langerhans cell histiocytosis, aphthous stomatitis, Behçet’s syndrome and others. Zhang et al[16] reported a 29-year-old patient with a five-year history of BD who was administered prednisone and thalidomide. The patient was well with blood sedimentation and C-reactive protein in the normal range. Sayarlioglu et al[17] reported a patient with intestinal BD and recurrent perforating intestinal ulcers under immunosuppressive treatment with methylprednisolone and cyclophosphamide. The patient’s symptoms did not disappear until she was treated with thalidomide[17]. These reports suggest the beneficial effects of thalidomide in BD. Direskeneli et al[18] revealed that thalidomide decreased TNF-alpha receptor levels, CD8/CD11b+ T cells and natural killer cells during early treatment and increased CD4+CD45RO+ memory T and gammadelta+ T cells during longer treatment in patients with BD. Therefore, thalidomide, in small doses, was thought to be safe and effective in the treatment of intestinal BD, was not addictive and did not have acute side-effects such as motor impairment.
In conclusion, practitioners should be aware of intestinal BD which accompanies intestinal ulcers and could lead to perforation or hemorrhage. Urgent surgical resection is mandatory in the case of hemorrhea without effective medical treatment, and medical treatment is required after surgery.
The patient had fever and abdominal pain, accompanied with skip ulcers with lower gastrointestinal severe hemorrhage.
The case should be diagnosed as untypical Intestinal behcet’s disease (BD).
The case should be difference from Crohn’s disease and gastrointestinal tuberculosis.
Serum antinuclear antibody, antituberculosis antibody, PPD cutantest and T-spot test were all negative, but pathergy test was positive.
Endoscopy displayed giant ulceration in the inferior extremity of esophagus and the ileocecal junction, abdominal computed tomography imaging indicated the intestinal canal of ascending colon and ileocecal region was thicken, and whole gastrointestinal barium meal examination presented the inflammatory change of ascending colon and ileocecal region.
Pathological examination of the endoscopic biopsy specimen indicated nonspecific ulcer, but the ulcer of ileocecal lesion resected by surgical encroached whole range with ectasia and hyperplasia blood vessels.
The patient underwent an emergency right hemicolectomy and partial ileectomy with ileostomy because of unmanageable lower gastrointestinal severe hemorrhage, and continued the treatment of prednisone and thalidomide.
BD can influence any level of the gastrointestinal tract. The mouth is the most common gastrointestinal sites affected by BD. Next site is the ileocecal region. Intestinal lesions are located on the antimesenteric side. Gastrointestinal symptoms related to BD are abdominal pain, nausea and vomiting. Some rare symptoms present in emergency conditions, such as intestinal perforation or bleeding. The manifestations of intestinal BD similar to other colitis such as Crohn’s disease or intestinal tuberculosis, therefore it is still challenging for gastroenterologist to accurately diagnose intestinal BD among the patients with ileo-colonic ulcers. Meanwhile it was more difficult to diagnose when intestinal BD accompanied with multiple ulcers in different positions of whole digestive tract. Furthermore skip giant ulcers and gastrointestinal hemorrhage are rare in intestinal BD.
Clinical practitioners should be aware of intestinal BD which accompanies intestinal ulcers since the case probably could lead to perforation or hemorrhage. Urgent surgical resection is mandatory in case of hemorrhea without efficient medical treatment. And the medicinal treatments are still needed after surgery.
The authors have presented a rare disorder of BD. The case reported here is interesting.
P- Reviewers: Kouraklis G, Kate V, Mezalek ZT S- Editor: Song XX L- Editor: A E- Editor: Zhang DN
| 1. | Altintaş E, Senli MS, Polat A, Sezgin O. A case of Behçet's disease presenting with massive lower gastrointestinal bleeding. Turk J Gastroenterol. 2009;20:57-61. |
| 2. | Oshima Y, Shimizu T, Yokohari R, Matsumoto T, Kano K, Kagami T, Nagaya H. Clinical Studies on Behçet’s Syndrome. Ann Rheum Dis. 1963;22:36-45. |
| 3. | Kasahara Y, Tanaka S, Nishino M, Umemura H, Shiraha S, Kuyama T. Intestinal involvement in Behçet’s disease: review of 136 surgical cases in the Japanese literature. Dis Colon Rectum. 1981;24:103-106. |
| 4. | Masugi J, Matsui T, Fujimori T, Maeda S. A case of Behçet’s disease with multiple longitudinal ulcers all over the colon. Am J Gastroenterol. 1994;89:778-780. |
| 5. | Davatchi F, Shahram F, Chams-Davatchi C, Shams H, Nadji A, Akhlaghi M, Faezi T, Sadeghi Abdollahi B. How to deal with Behcet’s disease in daily practice. Int J Rheum Dis. 2010;13:105-116. |
| 6. | Deuter CM, Kötter I, Wallace GR, Murray PI, Stübiger N, Zierhut M. Behçet’s disease: ocular effects and treatment. Prog Retin Eye Res. 2008;27:111-136. |
| 7. | Ghate JV, Jorizzo JL. Behçet’s disease and complex aphthosis. J Am Acad Dermatol. 1999;40:1-18; quiz 19-20. |
| 8. | Koné-Paut I, Yurdakul S, Bahabri SA, Shafae N, Ozen S, Ozdogan H, Bernard JL. Clinical features of Behçet’s disease in children: an international collaborative study of 86 cases. J Pediatr. 1998;132:721-725. |
| 9. | Yazici H, Tüzün Y, Pazarli H, Yurdakul S, Ozyazgan Y, Ozdoğan H, Serdaroğlu S, Ersanli M, Ulkü BY, Müftüoğlu AU. Influence of age of onset and patient's sex on the prevalence and severity of manifestations of Behçet's syndrome. Ann Rheum Dis. 1984;43:783-789. |
| 10. | Choi IJ, Kim JS, Cha SD, Jung HC, Park JG, Song IS, Kim CY. Long-term clinical course and prognostic factors in intestinal Behçet’s disease. Dis Colon Rectum. 2000;43:692-700. |
| 11. | Ketch LL, Buerk CA, Liechty D. Surgical implications of Behçet’s disease. Arch Surg. 1980;115:759-760. |
| 12. | Ebert EC. Gastrointestinal manifestations of Behçet’s disease. Dig Dis Sci. 2009;54:201-207. |
| 13. | Mizushima Y. [Revised diagnostic criteria for Behçet’s disease in 1987]. Ryumachi. 1988;28:66-70. |
| 14. | Kobayashi K, Ueno F, Bito S, Iwao Y, Fukushima T, Hiwatashi N, Igarashi M, Iizuka BE, Matsuda T, Matsui T. Development of consensus statements for the diagnosis and management of intestinal Behçet’s disease using a modified Delphi approach. J Gastroenterol. 2007;42:737-745. |
| 15. | Tunç R, Uluhan A, Melikoğlu M, Ozyazgan Y, Ozdoğan H, Yazici H. A reassessment of the International Study Group criteria for the diagnosis (classification) of Behçet's syndrome. Clin Exp Rheumatol. 2001;19:S45-S47. |
| 16. | Zhang Z, Jian X, Liu H, Zhang W, Zhou Q. Recurrent aortic aneurysm due to Behcet’s disease: a case report from China. Ann Thorac Cardiovasc Surg. 2013;19:173-175. |
| 17. | Sayarlioglu M, Kotan MC, Topcu N, Bayram I, Arslanturk H, Gul A. Treatment of recurrent perforating intestinal ulcers with thalidomide in Behçet’s disease. Ann Pharmacother. 2004;38:808-811. |
| 18. | Direskeneli H, Ergun T, Yavuz S, Hamuryudan V, Eksioglu-Demiralp E. Thalidomide has both anti-inflammatory and regulatory effects in Behcet’s disease. Clin Rheumatol. 2008;27:373-375. |