Published online Feb 16, 2013. doi: 10.4253/wjge.v5.i2.74
Revised: November 9, 2012
Accepted: November 24, 2012
Published online: February 16, 2013
Processing time: 124 Days and 3.7 Hours
Pneumatosis of the gastrointestinal tract is a rare condition characterized by the presence of air filled cavities in the gastrointestinal tract wall. Its occurrence has been described throughout the gastrointestinal tract from the esophagus to the rectum, however it is most commonly reported in the small intestine. Despite multiple case reports in literature, its pathogenesis still remains unclear. Pneumatosis may be idiopathic or associated with a variety of disorders namely peptic ulcer disease, jejunoileal bypass, intestinal obstruction and non-gastrointestinal disorders like asthma, chronic obstructive pulmonary disease, systemic lupus erythematosus, infectious enteritis, etc. We here present a rare case of pneumatosis of the esophagus diagnosed incidentally at an esophagogastroduodenoscopy (EGD). A 78-year-old asymptomatic woman underwent EGD and colonoscopy at our hospital for evaluation of anemia. Few months prior to EGD, she had undergone excision of laryngocele at our hospital. EGD revealed extensive submucosal blebs distributed throughout the esophagus, otherwise unremarkable stomach and duodenum. Colonoscopy showed a tubular adenomatous polyp. Since our patient was asymptomatic she did not require any surgical intervention. Management of pneumatosis depends on the underlying cause.
- Citation: Chelimilla H, Makker JS, Dev A. Incidental finding of esophageal pneumatosis. World J Gastrointest Endosc 2013; 5(2): 74-78
- URL: https://www.wjgnet.com/1948-5190/full/v5/i2/74.htm
- DOI: https://dx.doi.org/10.4253/wjge.v5.i2.74
Pneumatosis of the gastrointestinal tract is well known in medical literature. It is characterized by the presence of air filled spaces in the submucosa and subserosa.
It can appear in different locations in the gastrointestinal tract from the esophagus to the rectum, but more commonly the small bowel is involved[1-4]. Pneumatosis of the gastrointestinal tract can be symptomatic or completely asymptomatic, where it is detected incidentally during endoscopy or autopsy. Clinical manifestations are variable and depend on the underlying clinical condition and the site of gastrointestinal tract involved. Commonly reported gastrointestinal symptoms include retrosternal chest pain, abdominal pain and gastrointestinal bleed. Initial evaluation of pneumatosis should be focussed on ruling out acute abdomen or sepsis and identification of the underlying pathology.
Pneumatosis involving the esophagus is a relatively rare condition. Ever since the first description of pneumatosis in medical literature about three centuries ago, only eight case reports of esophageal pneumatosis have been reported so far. We here report the ninth case of esophageal pneumatosis found incidentally on an esophagogastroduodenoscopy (EGD).
A 78-year-old African American woman was seen in our gastroenterology clinic for evaluation of anemia.
Five months before evaluation in the gastroenterology clinic, she was admitted to our hospital with symptoms of right-sided neck pain, dysphagia and sore throat of three days duration. She denied any shortness of breath, drooling of saliva, cough or chest pain. She reported no weight loss, fever or sick contacts.
Her medical history included diabetes mellitus, hypertension, sickle cell trait and chronic hepatitis C. She had no known allergies. She had smoked 1 pack per day for the past 50 years and had quit a year ago. She denied any alcohol or substance abuse.
She denied having any surgery in the past and her family history was unremarkable.
Initial vital signs and laboratory findings have been summarized in Tables 1 and 2 respectively. General physical examination showed a firm, nontender, non-mobile mass on the right side of the neck. Thyroid gland was normal on examination as was the remainder of the examination.
| Vital signs | |
| Temperature | 98.2°F |
| Blood pressure | 208/103 mmHg |
| Pulse rate | 108 beats per minute |
| Respiratory rate | 20 breaths per minute |
| Oxygen saturation | 95% (breathing ambient air) |
| Laboratory values | |
| Hematocrit | 38.90% |
| White blood cell count | 7800/mm3 |
| (77% neutrophils and 13% lymphocytes) | |
| Platelet count | 229 000/μL |
| Prothrombin time | 11.8 s |
| Activated partial thromboplastin time | 26.4 s |
| Serum sodium | 137 mEq/L |
| Serum potassium | 3.6 mEq/L |
| Serum blood urea nitrogen | 13 mg/dL |
| Serum creatinine | 1.5 mg/dL |
| Serum total protein | 6.9 g/dL |
| Serum albumin | 3.3 g/dL |
| Serum alanine aminotransferase | 13 U/L |
| Serum aspartate transaminase | 25 U/L |
| Serum total bilirubin and direct bilirubin | 1.2 mg/dL, 0.7 mg/dL |
| Serum creatine kinase | 225 U/L |
| Hepatitis panel | Hepatitis C antibody positive |
| Hepatitis B antigen negative |
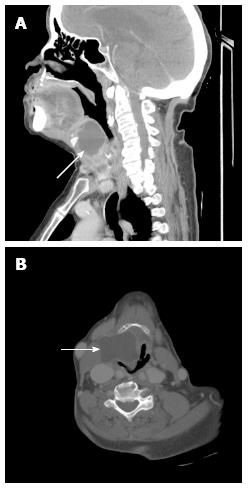
Chest radiograph was normal. Computed tomography (CT) of neck soft tissue was obtained after intravenous contrast which revealed uniformly hypodense, loculated collection with well demarcated thin enhancing margin measuring 3.8 cm × 2.5 cm × 4.2 cm bordering the right oropharynx and extended from the palatine tonsils to the tip of the esophagus (Figure 1). Displacement of the upper air passage towards the left side was also noted. Patient was evaluated by ear nose and throat surgeon and was taken to operating room for excision of a possible infected laryngocele through a laryngotomy approach. Intraoperatively, approximately 5 cm × 7 cm cystic mass involving the larynx extending from the hyoid bone to the thyroid cartilage on the right side was noticed. The cyst extended through the thyrohyoid area into the larynx and communicated with the laryngeal vestibule. There was significant edema and fullness of the right aryepiglottic fold and supraglottic larynx, which obstructed view of the glottic airway. The patient required fiberoptic intubation for achieving general anesthesia.
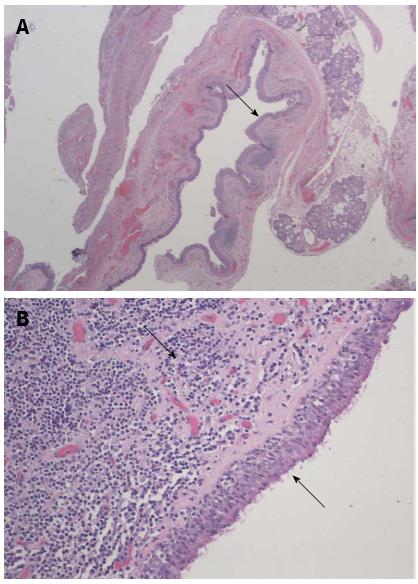
After excision, a membranous saccular tissue measuring 4 cm × 2.8 cm × 0.3 cm was submitted for a pathological diagnosis, which later revealed respiratory epithelium-lined membranous fibrous tissue consistent with laryngocele (Figure 2). Pathological examination of the purulent fluid submitted with the laryngocele revealed acute inflammatory cell infiltrate comprising polymorphs, macrophages and cellular debris, and these features were suggestive of an abscess.
After the procedure, patient remained intubated with respiration supported on positive pressure mechanical ventilation for 11 d. Postoperative course was otherwise uneventful and patient was discharged home after a hospital stay of 2 wk.
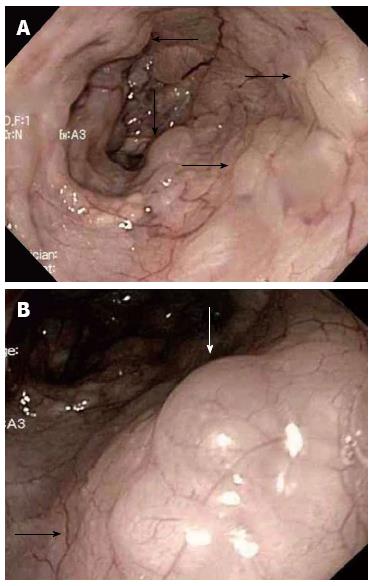
Four months after discharge from the hospital, she was asymptomatic and seen in the clinic for evaluation of anemia. Upper gastrointestinal endoscopy was done which incidentally showed extensive submucosal blebs of the esophagus without any involvement of the stomach (Figure 3). There was gastric erythema with biopsy showing Helicobacter pylori gastritis. Colonoscopy was also performed at the same time, which revealed a tubular adenomatous polyp but otherwise unremarkable.
Pneumatosis of the gastrointestinal tract was first reported in a cadaver in the year 1730 by Thorpe[5] and Du vernoi[6]. Later in the year 1876, pneumatosis intestinalis was reported in humans by Bang[7]. Since then numerous cases of pneumatosis of the gastrointestinal tract have been described. In 1979, Jamart reported a review of 919 cases of pneumatosis intestinalis and found that pneumatosis of small bowel was predominantly subserous and that of colon was submucosal in location[8]. Pneumatosis can involve any part of the gastrointestinal tract from the esophagus to the rectum, but has been reported more commonly in small bowel than large bowel. Cases of gastric pneumatosis have also been published but esophageal pneumatosis has been a rare occurrence.
On review of the available medical literature, we found eight cases (Table 3) of esophageal pneumatosis reported till date. Interestingly, most of these cases including our case as well were reported in females except for two cases. Vanasin et al[9], described the first case of esophageal pneumatosis in a 62 year old woman related to esophageal stricture. Katz et al[10] reported three cases of esophageal pneumatosis related to fiberesophagoscopy. Fifth case was described by McKelvie et al[11] in a 76-year-old man with infectious gastritis. Sixth case in a 74-year-old woman post duodeno-pancreatectomy was published in 1998 by Tixedor et al[12]. Mclaughlin et al[13] reported the seventh case of esophageal pneumatosis in a 77-year-old man who had underwent resection of cholangiocarcinoma. Yahng et al[14] reported the eighth case in a 62-year-old woman who developed esophageal pneumatosis post chemotherapy with neutropenia.
| Ref. | Age | Sex | Etiology | Treatment | Follow up |
| Vanasin et al[9] | 62 | F | Esophageal stricture | None | Post mortem diagnosis. |
| Died of aspiration pneumonia | |||||
| Katz et al[10] | 85 | F | Post esophagoscopy | Surgery | Died in 6 wk |
| Katz et al[10] | 46 | F | Post esophagoscopy | Surgery | Survived |
| Katz et al[10] | 39 | F | Post esophagoscopy | Antibiotics | Survived |
| McKelvie et al[11] | 76 | M | Infection | None | Post mortem diagnosis |
| Tixedor et al[12] | 74 | F | Post pancreato-duodenectomy | Nasogastric aspiration | Survived |
| Mclaughlin et al[13] | 77 | M | Gastric dilatation | 100% oxygen | Survived |
| Yahng et al[14] | 62 | F | Chemotherapy and neutropenia | Observation | Died in 5 wk due to ARDS |
Pneumatosis may be idiopathic or may occur secondary to underlying pathological process. These secondary forms of pneumatosis account for about 85% of total cases[15,16]. Pneumatosis intestinalis has been described in various medical conditions including necrotizing enterocolitis, pyloric stenosis, peptic ulcer disease, jejunoileal bypass, and intestinal obstruction. Besides being described in gastrointestinal disorders, it has also been reported in non-gastrointestinal disorders like asthma, chronic obstructive pulmonary disease, celiac disease, systemic lupus erythematosus, infectious enteritis, acquired immunodeficiency syndrome, primary immunodeficiency, leukemia, organ transplantation and bone marrow transplantation[3,17].
Despite several cases reported in literature, pathogenesis of pneumatosis remains unclear. Two important theories proposed among several other proposed are mechanical and bacterial theory. According to mechanical theory, air present in the gastrointestinal tract lumen dissects into the walls of the gastrointestinal tract through a mucosal tear. Bacterial theory proposes that the air entering the walls of bowel is produced by gas forming bacteria in the bowel.
In our patient, we hypothesize that resection of the laryngocele followed by positive pressure ventilation may have predisposed to the formation of the pneumatosis, and hence supporting mechanical theory. Other possible etiology in our patient could be the underlying chronic bronchitis secondary to long standing smoking.
Typically it is an asymptomatic condition as in our patient. However may have a broad spectrum of presentation from an incidental finding to retrosternal chest pain, abdominal pain or septic shock. Symptoms may vary depending upon site of gastrointestinal tract involved.
Work up includes radiographs of the chest and the abdomen. CT, magnetic resonance imaging and barium studies[3] can also be used, but CT has better sensitivity to detect intramural air[18,19].
Management largely depends on the underlying etiology. Various treatment modalities have been utilized in the past including oxygen therapy and antibiotic use. Normobaric oxygen therapy, after its first experimental use in 1935, was used successfully for treatment of pneumatosis by Forgacs et al[20]. Later in 1978, the use of hyperbaric oxygen was also shown to be beneficial by Masterson et al[21]. Antibiotic therapy with Metronidazole has also been reported to be efficacious in literature supporting the bacterial theory of genesis[22].
Algorithm for surgical management of pneumatosis has been suggested by Greenstein et al[23] in cases of obstruction, elevated white cell count, advanced age and portal venous gas. Endoscopic modalities of therapy with cyst puncture and sclerotherapy have also been described[24]. But with numerous case reports of success with conservative therapy, treatment of pneumatosis essentially remains non-surgical unless acute abdomen is suspected.
P- Reviewer Donati F S- Editor Gou SX L- Editor A E- Editor Zhang DN
| 1. | Boerner RM, Fried DB, Warshauer DM, Isaacs K. Pneumatosis intestinalis. Two case reports and a retrospective review of the literature from 1985 to 1995. Dig Dis Sci. 1996;41:2272-2285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 75] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Gagliardi G, Thompson IW, Hershman MJ, Forbes A, Hawley PR, Talbot IC. Pneumatosis coli: a proposed pathogenesis based on study of 25 cases and review of the literature. Int J Colorectal Dis. 1996;11:111-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 79] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 3. | Heng Y, Schuffler MD, Haggitt RC, Rohrmann CA. Pneumatosis intestinalis: a review. Am J Gastroenterol. 1995;90:1747-1758. [PubMed] |
| 4. | Rennenberg RJ, Koek GH, Van Hootegem P, Stockbrügger RW. Pneumatosis cystoides intestinalis, four cases of a rare disease. Neth J Med. 2002;60:22-25. [PubMed] |
| 5. | Thorpe GJ. Pneumatosis cystoides intestinalis associated with abdominal carcinomatosis. Postgrad Med J. 1965;41:41-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 6. | DuVernoi JG. Anatomische Beobachtungen der Unter der Aussern und Innern Haut der Gedarme Eingeschlossenen Luft. Phys Med Abhandl Acad Wissenschin Petersb. 1783;2:182. |
| 7. | Bang BLF. Luftholdige Kyster I Vaggen af ileum og I Nydannot Bindevav pa Sammes Serosa. Nord med Ark. 1876;18:1. |
| 8. | Jamart J. Pneumatosis cystoides intestinalis. A statistical study of 919 cases. Acta Hepatogastroenterol (Stuttg). 1979;26:419-422. [PubMed] |
| 9. | Vanasin B, Wright JR, Schuster MM. Pneumatosis cystoides esophagi. Case report supporting theory of submucosal spread. JAMA. 1971;217:76-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 10. | Katz D, Cano R, Antonelle M. Benign air dissection of the esophagus and stomach at fiberesophagoscopy. Gastrointest Endosc. 1972;19:72-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 11. | McKelvie PA, Fink MA. A fatal case of emphysematous gastritis and esophagitis. Pathology. 1994;26:490-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Tixedor N, Taourel P, Adell JF, Bruel JM. Extensive esophageal pneumatosis after acute dilatation of the stomach. AJR Am J Roentgenol. 1998;171:272-273. [PubMed] |
| 13. | Mclaughlin SA, Nguyen JH. Conservative management of nongangrenous esophageal and gastric pneumatosis. Am Surg. 2007;73:862-864. [PubMed] |
| 14. | Yahng SA, Kim HJ. Fortuitous detection of esophageal pneumatosis in a neutropenic patient. Korean J Hematol. 2011;46:151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 15. | Knechtle SJ, Davidoff AM, Rice RP. Pneumatosis intestinalis. Surgical management and clinical outcome. Ann Surg. 1990;212:160-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 154] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 16. | Koss LG. Abdominal gas cysts (pneumatosis cystoides intestinorum hominis); an analysis with a report of a case and a critical review of the literature. AMA Arch Pathol. 1952;53:523-549. [PubMed] |
| 17. | Hepgur M, Ahluwalia MS, Anne N, Thomas J, Liu H, Schiff MD, Loud PA, Hahn TE, Bullard Dunn KM, McCarthy PL. Medical management of pneumatosis intestinalis in patients undergoing allogeneic blood and marrow transplantation. Bone Marrow Transplant. 2011;46:876-879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | Rha SE, Ha HK, Lee SH, Kim JH, Kim JK, Kim JH, Kim PN, Lee MG, Auh YH. CT and MR imaging findings of bowel ischemia from various primary causes. Radiographics. 2000;20:29-42. [PubMed] |
| 19. | Rabushka LS, Kuhlman JE. Pneumatosis intestinalis. Appearance on MR examination. Clin Imaging. 1994;18:258-261. [PubMed] |
| 20. | Forgacs P, Wright PH, Wyatt AP. Treatment of intestinal gas cysts by oxygen breathing. Lancet. 1973;1:579-582. [PubMed] |
| 21. | Masterson JS, Fratkin LB, Osler TR, Trapp WG. Treatment of pneumatosis cystoides intestinalis with hyperbaric oxygen. Ann Surg. 1978;187:245-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 22. | Tak PP, Van Duinen CM, Bun P, Eulderink F, Kreuning J, Gooszen HG, Lamers CB. Pneumatosis cystoides intestinalis in intestinal pseudoobstruction. Resolution after therapy with metronidazole. Dig Dis Sci. 1992;37:949-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 23. | Greenstein AJ, Nguyen SQ, Berlin A, Corona J, Lee J, Wong E, Factor SH, Divino CM. Pneumatosis intestinalis in adults: management, surgical indications, and risk factors for mortality. J Gastrointest Surg. 2007;11:1268-1274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 24. | Johansson K, Lindström E. Treatment of obstructive pneumatosis coli with endoscopic sclerotherapy: report of a case. Dis Colon Rectum. 1991;34:94-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |