Published online Mar 16, 2022. doi: 10.4253/wjge.v14.i3.113
Peer-review started: June 30, 2021
First decision: October 18, 2021
Revised: November 1, 2021
Accepted: February 15, 2022
Article in press: February 15, 2022
Published online: March 16, 2022
Processing time: 258 Days and 21.2 Hours
Due to the advent of the screening programs for colorectal cancer and the era of quality assurance colonoscopy the number the polyps that can be considered difficult, including large (> 20 mm) laterally spreading tumors (LSTs), has increased in the last decade. All LSTs should be assessed carefully, looking for suspicious areas of submucosal invasion (SMI), such as nodules or depressed areas, describing the morphology according to the Paris classification, the pit pattern, and vascular pattern. The simplest, most appropriate and safest endoscopic treatment with curative intent should be selected. For LST-granular homogeneous type, piecemeal endoscopic mucosal resection should be the first option due to its biological low risk of SMI. LST-nongranular pseudodepressed type has an increased risk of SMI, and en bloc resection should be mandatory. Underwater endoscopic mucosal resection is useful in situations where submucosal injection alters the operative field, e.g., for the resection of scar lesions, with no lifting, adjacent tattoo, incomplete resection attempts, lesions into a colonic diverticulum, in ileocecal valve and lesions with intra-appendicular involvement. Endoscopic full thickness resection is very useful for the treatment of difficult to resect lesions of less than 20 up to 25 mm. Among the indications, we highlight the treatment of polyps with suspected malignancy because the acquired tissue allows an exact histologic risk stratification to assign patients individually to the best treatment and avoid surgery for low-risk lesions. Endoscopic submucosal dissection is the only endoscopic procedure that allows completes en bloc resection regardless of the size of the lesion. It should therefore be indicated in the treatment of lesions with risk of SMI.
Core Tip: The number of detected large laterally spreading tumors has increased in the last decade. Herein, we review the current landscape of different endoscopic techniques that allow us to resect difficult laterally spreading tumors. We also describe strategies in problematic situations such as scarred lesions or difficult areas and how to treat adverse events related to colonoscopy.
- Citation: Castillo-Regalado E, Uchima H. Endoscopic management of difficult laterally spreading tumors in colorectum. World J Gastrointest Endosc 2022; 14(3): 113-128
- URL: https://www.wjgnet.com/1948-5190/full/v14/i3/113.htm
- DOI: https://dx.doi.org/10.4253/wjge.v14.i3.113
Therapeutic endoscopy is nowadays a well consolidated area in the gastroenterology field, covering techniques such as gastroscopy, colonoscopy, enteroscopy, endoscopic retrograde cholangiopancreatography and therapeutic endoscopic ultrasound. In the last decade, techniques for resection of early gastrointestinal neoplasia have become widespread worldwide and gaining popularity among young endoscopists with special interests in learning endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD). The main societies have published their statements[1-4].
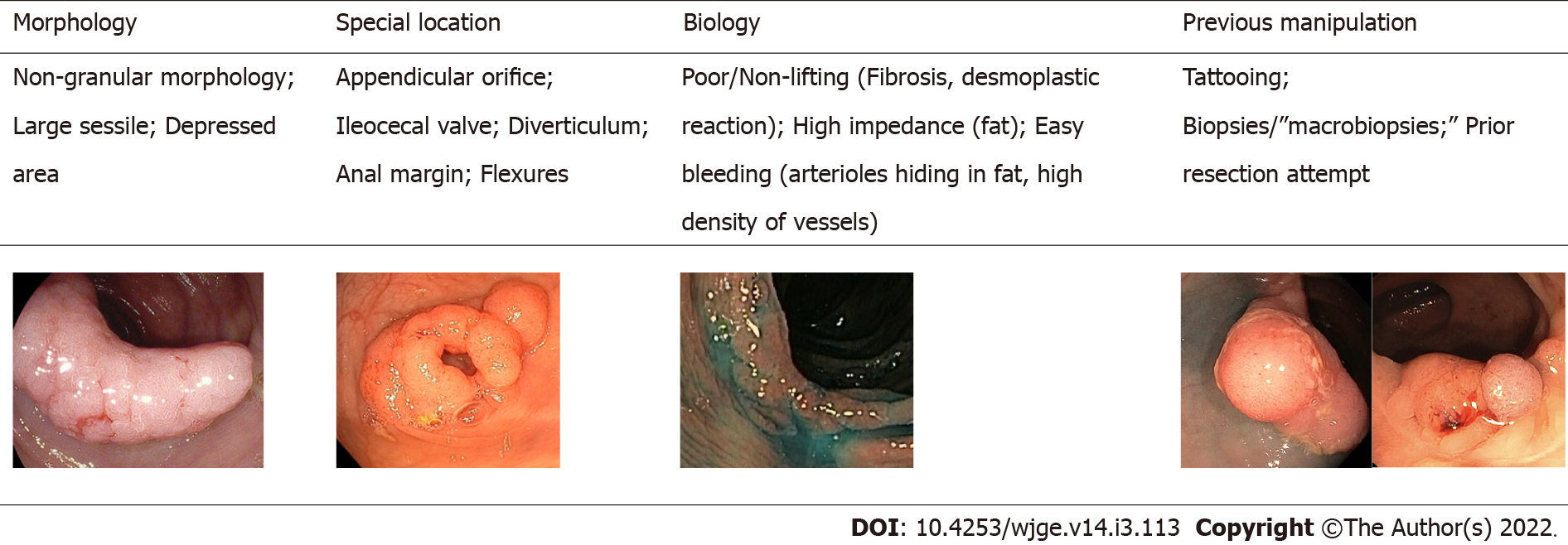
On the other hand, with the advent of the screening programs for colorectal cancer and adopted in Europe, Australia, Asia and North America and the era of quality assurance and high-definition colonoscopy, the number of advanced mucosal neoplasia and early cancer in the colon, including the polyps that can be considered difficult, has increased in the last decade[5]. The definition of a difficult polyp is not well established. These polyps are typically defined by their size (generally considered as those greater than or equal to 20 mm), morphology, location, biology and previous manipulation (Figure 1).
Thus, the endoscopist should have the skills to detect and characterize all types of colorectal lesions and should be able to predict their risk of deep submucosal invasion (SMI) with high accuracy and proceed to endoscopic resection if it is indicated. The optical diagnosis with image-enhanced endoscopy is the key and mandatory first step before management of a colorectal polyp. First, morphology should be assessed and described according to the Paris Classification, including surface [granular or non-granular in cases of laterally spreading tumors (LSTs) or presence of ulcerations] and looking for demarcated areas (nodules, depressions or marked erythema). Then, virtual chromoendoscopy with blue light technology should be applied to investigate the surface and microvascular patterns. There are different classifications that help predict the risk of deep SMI, like Narrow Band Imaging (NBI) International Colorectal Endoscopic classification that does not need optical zoom or Japan NBI Expert Team (JNET) classification that uses optical zoom. The subclass JNET3 includes deep submucosal invasive lesions; JNET2a includes mostly intraepithelial lesions (e.g., low-grade dysplasia), while that of JNET2b could be found in intramucosal lesions and lesions with SMI. In those cases, pit pattern evaluation with chromoendoscopy and optical zoom using crystal violet or indigo carmine should be recommended, especially in the demarcated areas that may have a higher risk of SMI[6].
The endoscopic treatment of colorectal lesions should be reserved to all early neoplastic lesions with low risk of SMI and thus ideally no risk of lymph node metastasis. If the lesion is considered to have risk of lymph node metastasis, surgery should be considered as a first option.
There is strong evidence now to recommend the EMR as the first-line therapy for non-invasive lesions. It has good results and lesser mortality compared to surgery, and the patients could be discharged the same day (even elderly patients or patients with a severe comorbidity)[7,8].
Herein, we review the techniques for endoscopic resection of the LST, including complex lesions.
The term LST, initially reported by Kudo et al[9], refers to flat lesions larger than 10 mm that grow laterally along the colonic wall, being classified as granular (LST-G) and non-granular (LST-NG).
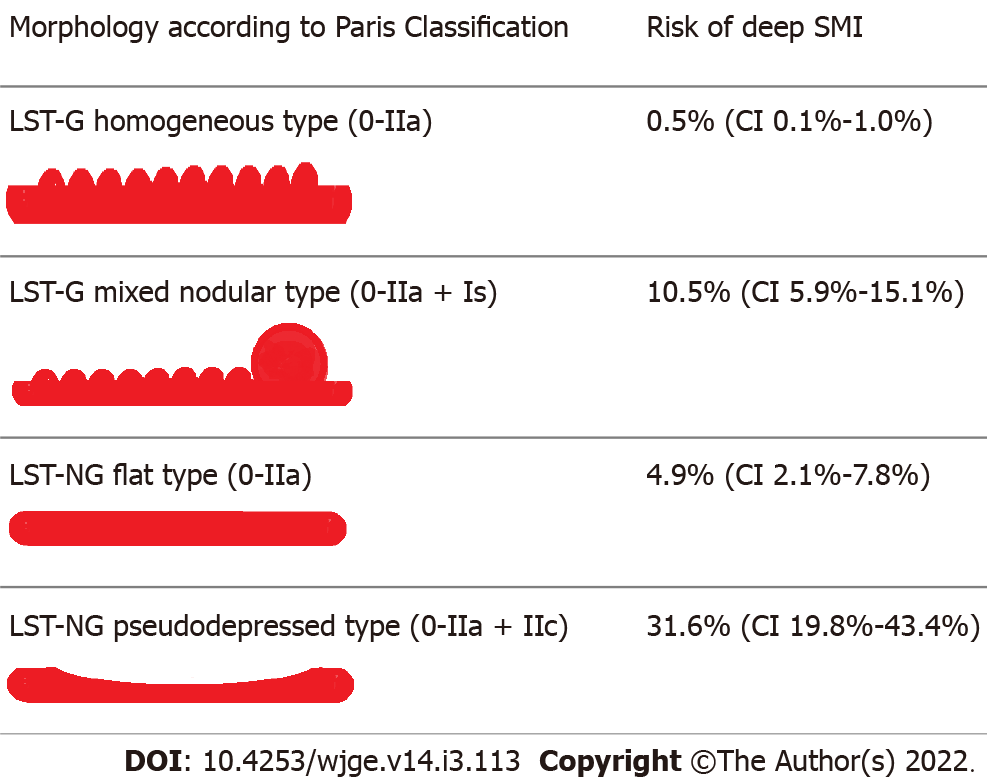
The LST-G can be classified as LST-G homogeneous type (Paris Classification 0-IIa) if they show a granular homogeneous surface (usually < 3 mm) or as LST-G nodular mixed type (Paris Classification 0-II + Is) if they have one or more sessile nodules, with those greater than 10 mm carrying an increasing risk of SMI. The LST-NG can be classified as LST-NG flat type (Paris Classification 0-IIa) or LST-NG pseudodepressed type (Paris Classification 0-IIa + IIc)[10].
Their characteristic growth (lateral rather than vertical) appear to be caused by adequate co-expression of β-catenin and E-cadherin in the basolateral membrane, type IV collagen along the basement membrane and expression of atypical protein kinase λ/ι (an essential cell polarity regulator) like normal colonic mucosa[11].
They also seem to overexpress lipocalin-2 and metallopeptidase-9 in a correlated manner to advanced stages (worse pathology grading), being both suggested as potential serum biomarkers for LST progression[12].
The types of LST have a different biology. For example, the LST-G type express CpG island methylator phenotype-high involving more than two loci and has a high prevalence of K-ras mutations (especially in the right colon), whereas the LST-NG type have less K-ras mutations and are CpG island methylator phenotype-low[13,14]. New non-invasive diagnostic biomarkers are being explored with the microbiome signature being one of them.
Clinically, the LST-NG type tend to be more aggressive with a higher incidence of advanced carcinoma, especially the pseudodepressed type, with incidences of 19.8%-43.4%. On the other hand, LST-G type tend to have less submucosal carcinoma, being rare in the LST-G homogeneous type (0.5%; 95%CI: 0.1%-1.0%) irrespective of the size of the lesion (Figure 2)[15].
Location is variable. Granular type is more often localized in the cecum and rectum and non-granular in the right colon[16].
For large LST-G homogeneous type, piecemeal EMR should be the first option irrespective of the size of the lesion most of the time due to its biological low risk of SMI. For LST-G nodular mixed type careful inspection of the surface and vascular patterns (specially in nodules > 10 mm) should be done to rule out signs of deep SMI prior to treatment.
For LST-NG type, en bloc resection should be considered as the first option in all cases due to its higher risk of SMI (especially for the pseudodepressed type). Thus, ESD or surgical treatment should be decided according to local expertise in case the lesion is too big for en bloc EMR. Endoscopic full thickness resection (EFTR) may be an alternative if the lesion is suitable.
In some cases, LST-NG flat type might be resected in piecemeal if the surface and vascular patterns show no signs of SMI. These considerations are summarized in Table 1.
| LST suitable for piecemeal EMR | Comments | LST not suitable for piecemeal EMR | Comments |
| LST-G homogeneous type | Very low risk for deep SMI, independent of size of the lesion | LST-NG pseudodepressed type | En bloc resection |
| LST-G mixed nodular type with no signs of SMI | Consider en bloc resection first. If not, careful inspection of surface/pit pattern and vascular pattern specially in the larger nodules (≥ 10 mm), resect the nodular area apart (e.g., JNET2a) | LST-G mixed nodular or NG flat with risk of SMI | En bloc resection (e.g., JNET2b, pit pattern V) |
| LST-NG flat with no demarcated area and no signs of SMI | Consider en bloc resection first. If not, careful inspection of surface/pit pattern and vascular pattern (e.g., JNET2a) |
Knowing the basic principles of diathermy is mandatory for endoscopists. Knowledge on the management of electrosurgery may be able to improve procedural outcomes and safety for our patients[17].
Electrosurgery uses radiofrequency electricity to generate heat in the tissue itself rather than applying heat from an outside source. The snares and most endoscopic knives commonly used in the west are monopolar [the electricity flows from the active electrode (snare) to the neutral electrode placed in the patient skin]. Fortunately, the electrosurgical units use high frequency alternating current (300 kHz to 5 MHz) to avoid neuromuscular stimulation. Thus, the risk of complications is mainly related to the amount of heat produced.
Power is the amount of energy consumed per unit time, and it is measured in watts. The energy dissipated as heat when the electric current (amperes) passes through the resistance (ohms) of the tissue held by the snare is measured in joules. There are two main clinical effects when the electric current is applied to the tissue by the snare: boiling (cells burst resulting in cutting tissue) and coagulation. If there is more current per unit of area (current density), then more heat is produced; therefore, the smaller the area of tissue trapped into the snare, a lesser amount of power is needed to heat the tissue.
Electrosurgical cutting is produced when a continuous alternating current with more than 200 voltage peaks is applied to the tissue, raising very rapidly the intracellular fluid temperature and boiling the cells (so they burst) with steam formation. Electrosurgical coagulation is produced if the tissue is heated slowly by an intermittent electric current. The temperature rises within cells, the cells shrink, and the cellular proteins coagulate, turning the tissue white (like the effect of heating the albumin of an egg). However, if current application to the tissue continues, then it produces carbon and smoke. This thermal damage may obscure the specimen margins on pathological evaluation.
If the current used has less than 200 voltage peaks, then the effect would be a superficial “pure coagulation” (e.g., SOFT COAG mode in ICC 200 and VIO 300D; ERBE, Tüebingen, Germany). If the current used has more than 200 voltage peaks and is activated 10% or less of the time (of the duty cycle, the fraction of time current flows each second that the activating pedal is depressed), then it would produce a deep coagulation (FORCED COAG mode of ERBE has 4% duty cycle). Even the “purest” cutting current can have some coagulation effect in the tissue around the cutting area where there is not enough heating to boil the cells but to dehydrate and coagulate proteins. Thus, the more cutting or coagulating effect would depend on the duty cycle. The more time energy is delivered by pushing the pedal, the greater the heat is produced and the chances of having a thermal-related complication, such as deep muscle layer injury or perforation.
To perform an EMR, alternating cutting and coagulating output is very useful (e.g., in the ENDOCUT mode of ERBE that alternates cutting current with SOFT COAG). For ERBE VIO 300, it would be recommended to use ENDO CUT Q effect 3 (cut duration 1, cut interval 6) for cutting and SOFT COAG Effect 4 (max. watts 80) for snare tip soft coagulation. The power settings (if they are not self-regulated by the electrosurgical units) should be adapted to the instrument used according to the manufacturer’s recommendation, and it is recommended to use the lowest power that will allow the resection[18].
Once we have set the mode and power, we can control by closing the snare on the area of tissue to resect (smaller area, less current needed for tissue cell burst) and the time we deliver that power to the tissue by pressing the pedal. The timing of the pedal is also very important during ESD.
Nowadays, endoscopes with optical narrow band technology using “blue light” to display the mucosa and vessels in high contrast, such as NBI (Olympus, Tokyo, Japan), Blue Light Imaging (Fujifilm, Tokyo, Japan) or i-scan Optical Enhancement (Pentax, Tokyo, Japan), should be used for endoscopic assessment of the lesion prior to resection, especially if there is optical magnification, to rule out signs of SMI[19].
Olympus has recently incorporated new postprocessing functions in the EVIS X1 system that includes extended depth of field and texture and color enhancement imaging that improves the visibility using white light endoscopy and red dichromatic imaging that enhances the visibility of deep blood vessels and bleeding. These functions could help diagnose and manage complications.
To facilitate resection for polyps in the rectosigmoid area and proximal colon, a gastroscope and a pediatric colonoscope or a short colonoscope may be used, respectively[20]. New colonoscopes like the RetroView™ EC34-i10T, PCF-H190TL/I EVIS EXERA III (Olympus) and Eluxeo EC-740TM/TL [Treier Endoscopie (part of the Duomed Group), Beromünster, Switzerland] provide excellent maneuverability due to a smaller bending radius of the distal tip, and 210° deflection is ideal for the detection and treatment of hard-to-reach lesions.
CO2 insufflation is highly recommended for therapeutic colonoscopy. It reduces pain after EMR of LSTs, which might be a cause of admission, especially in patients with a long duration of polypectomy[21].
A solution mixed with a blue dye is commonly used. The submucosal solution could be a crystalloid like normal saline solution or a colloid solution like glycerol or a succinylated gelatin. The inexpensive succinylated gelatin (gelafusine, gelafundin) was shown to be superior to saline solution requiring fewer injections, resections and an overall reduced EMR time[22]. A meta-analysis showed that use of viscous solutions during EMR leads to higher rates of en bloc resection and lower rates of residual lesions compared with normal saline solution especially with colonic polyp greater than 2 cm[23]. Nonetheless, research to determine the ideal submucosal injection is still ongoing.
Eleview® (Cosmo Pharmaceuticals, Dublin, Ireland), ORISE™ gel (Boston Scientific, Marlborough, MA, United States) and LiftUp® (Endotherapuetics, Australia) are synthetic solutions that were specifically designed to provide a submucosal cushion of optimal height and duration[24,25]. When compared to normal saline solution, Eleview® has demonstrated better cushion-forming ability and a duration of lift of up to 45 min. A double-blind randomized controlled trial comparing Eleview® with saline showed that the mean injected volume was significantly lower, and there was a trend towards shorter procedure and a lower number of resection pieces with this new solution. Despite all these advantages, larger, multicenter, prospective controlled trials are required to compare performance of Eleview®, ORISE™ gel and LiftUp® to other available viscous submucosal solutions for EMR and ESD.
An inert dye such as indigo carmine (or alternatively methylene blue) is added to stain the submucosal layer blue and facilitate the delineation of the lesion margins. The authors do not use adrenaline for submucosal injection, but diluted adrenaline (1/100000-1/300000) could be added according to the preferences of the endoscopist[26].
The distal cap attachment may contribute to stabilize the tip of the scope, improve visualization of the operative field and facilitate resecting lesions in difficult locations[27]. They are especially useful to create tension of submucosal fibers during ESD. Conic shaped short ST hood may be useful for non-lifting and other complex lesions when access to submucosal space could be difficult.
Deep sedation is preferred by the authors for EMR or underwater EMR (UEMR). Prophylactic antibiotics should be considered in cases of EMR or ESD of LST in the distal rectum (as drainage bypasses the liver) especially when a large resection defect (> 4 cm) is expected[28]. Consider buscopan or glucagon to reduce bowel peristalsis during the procedure.
The choice of a specific snare may rely on size and morphology of the lesion, its location, the endoscopist technique and preference or what type of snare is familiar. There are some snares that combine different sizes and shapes, but no clear benefit of one shape over the other has been demonstrated[2]. In cases of cold EMR, a dedicated cold snare is recommended. For hot EMR and UEMR, the authors’ preference is a rounded stiff snare 15 mm for most cases.
Like the choice of snare, it may depend on the lesion and the endoscopist preference. There are many types of ESD knives, but it is highly recommended to have water-jet or water injection capability to save time during dissection.
The most important step is to provide a good endoscopic diagnosis of the lesion, to be sure that the endoscopic resection would have a curative intention. The only way the endoscopic resection will be curative is if all the neoplastic cells are within the lesion we resect, even if they are malignant cells. But if there is a distant spread of the neoplastic tissue (e.g., lymphatics), then the treatment will not be curative. By endoscopic inspection we can predict the risk of deep SMI, telling us that there could be a risk of lymph node metastases. That is why during preoperative evaluation the endoscopist should rule out signs of deep SMI.
The endoscopist should use the best scope (better if there is magnification or dual focus with optical narrow band “blue light” technology), use Paris classification to describe the morphology of the lesion and assess demarcated areas of risk of SMI, such as the nodular and depressed areas. This assessment should focus on pit pattern and vascular pattern.
The JNET Classification was proposed in 2016 according to NBI magnifying endoscopy[6]. It consists of the following four categories, combining vessel and surface patterns: Type 1, the hyperplastic polyp or sessile serrated adenoma/polyp with “invisible” vessel pattern with regular dark or white spots similar to surrounding normal mucosa; Type 2A, the adenoma with low grade dysplasia, with regular vessels (in caliber and distribution) and surface pattern (corresponding to pit pattern III or IV); Type 2B, the adenoma with high grade dysplasia, or sometimes shallow submucosal cancer, with moderately distorted vessels and irregular or obscure surface pattern (corresponding to pit pattern Vi); and Type 3, an invasive cancer with amorphous areas with markedly distorted vessels or avascular areas.
However, in a retrospective study from prospectively collected records (n = 1402 lesions), Type 2B presented low sensitivity (42%) even among expert Japanese endoscopists. Therefore, some authors have suggested that Type 2B requires further investigation using pit pattern diagnosis to differentiate the Vi (irregular; superficial SMI) and Vn (non-structural; deep SMI)[29].
If there is a high suspicion of deep SMI, the patient should undergo a surgical procedure or an endoscopic technique for en bloc resection. It is also very important to delimitate the margins of the lesion, especially if it is a serrated adenoma.
In the LST-G homogeneous type (Paris 0-IIa) of any size, the risk of deep SMI is very low, which makes EMR almost always suitable[2-4,15].
“Classic” EMR is based on inject and resect technique (Table 2). It may be helpful for en bloc resection of lesions up to 2 cm and for piecemeal resection in bigger LSTs. For piecemeal resection 10 mm to 15 mm snares are usually recommended. For cold EMR, a specific cold snare is recommended. For a successful piecemeal EMR the resection should be performed in a systematic manner, sequentially from the first point of resection or entry in the submucosal plane, including 2-3 mm of apparently normal mucosa at the borders and including the edge of the advancing mucosal defect to avoid islands and bridges of neoplastic tissue.
| Steps for endoscopic resection | |
| (1) Endoscopic evaluation | Using Paris classification, pit pattern and vascular pattern to characterize the lesions and define the risk of deep SMI |
| (2) Strategy | Decide en bloc vs piecemeal resection according to risk of SMI. Consider patient position and gravity |
| (3) EMR technique | |
| Injection | Needle tangential to the plane. Inject whilst “stabbing” the mucosa helps accurately find the SM plane. Use a dynamic injection technique |
| Resection | Put the area to resect ideally between 5-6 o’clock (with colonoscope); accommodate the snare over the lesion and push “down,” aspirate to decrease tension and maximize tissue capture; close the snare tightly; check for mobility and degree of closure of the snare handle (usually < 1 cm distance between thumb and fingers), be sure there is no muscle trapped, otherwise release the tissue (in case of doubt, open and close the snare to “drop out” possible muscular entrapment); press the pedal to resect |
| Wash and check mucosal defect | Check the mucosal defect produced to rule out signs of muscle layer damage or perforation |
| Hemostasis | If there is mild intraprocedural bleeding, try first snare tip soft coagulation. If necessary, coagulating forceps or clips can be helpful |
| Systematic inject and resect | Continue resection injecting when necessary to maintain submucosal cushion. Resect 2-3 mm of normal mucosa to ensure margins. Try not to leave islands or bridges between resections |
| (4) UEMR technique | |
| Water filling | Aspirate all the gas and fill the lumen of the working space with water or saline (turning off insufflation may help) to create a gravity-free environment |
| Resection | Put the area to resect ideally between 5-6 o’clock (with colonoscope); accommodate the snare over the lesion “torque and crimp” and push “down” to get the floating lesion inside the snare; aspirate and irrigate more water to help the capture of the tissue; close the snare tightly and separate the tissue from the wall. Press the pedal to resect. Underwater, higher outputs might be needed for resection/coagulation due to the heat sink effect |
| Wash and check mucosal defect | Check the mucosal defect produced to rule out signs of muscle layer damage or perforation. As no dye is used to stain the submucosa, the operator should become familiarized with the aspect of the “transparent” fibers |
| Hemostasis | In cases of jet bleeding gas insufflation might be needed to find the bleeding point |
| Systematic gas aspiration water irrigation and resection | Continue resection aspirating gas or irrigating water when necessary. Resect 2-3 mm of normal mucosa to ensure margins. Try not to leave islands or bridges between resections |
| (5) Final inspection | Check the scar to rule out residual neoplastic tissue or signs of deep injury. In cases of piecemeal resection, thermal ablation with the tip of the snare (Soft COAG 80 W) to coagulate the mucosal borders of the scar reduces risk of recurrence |
| (6) Specimen retrieval and assessment | Consider using a net for retrieval. Big nodules should be sent separately if it was piecemeal resection |
The final mucosal defect should be checked for signs of injury or residual tissue. It is useful to use a topical submucosal chromoendoscopy with indigo carmine to rule out deep injury. It can be injected or sprayed superficially over the defect with the needle catheter close. The submucosa would pick up the blue color. If there is muscle layer exposed, then it would remain unstained[4,30].
After finishing piecemeal EMR, snare tip coagulation of the normal appearing margins and mucosal defect using SOFT COAG 80W is beneficial as it can reduce 4-fold the rate of residual or recurrent adenoma[4,30,31] even after en bloc EMR.
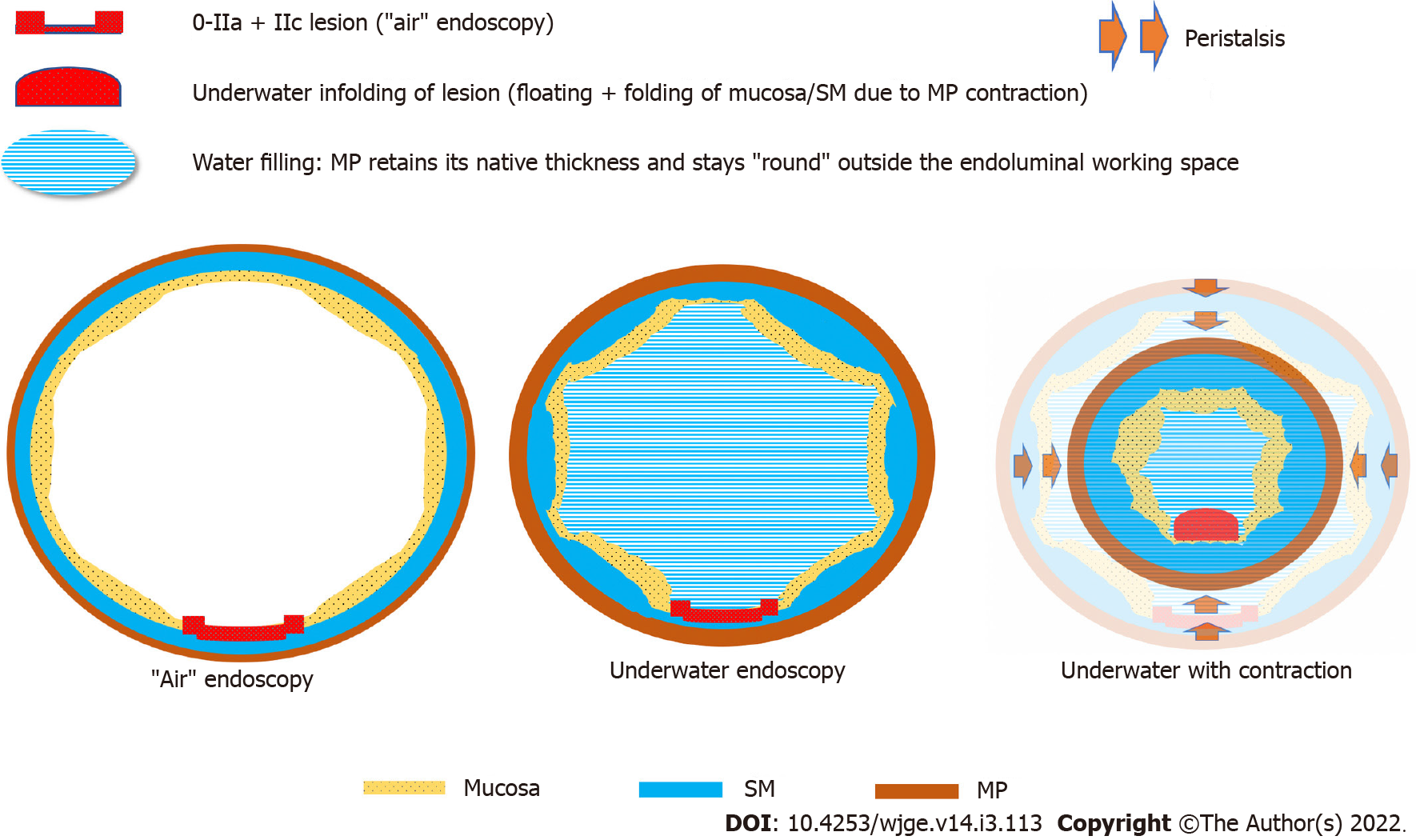
UEMR, described by Nett et al[32] in 2012, has been shown to enable safe resection of LST. UEMR is performed by aspirating all the gas from the colonic lumen and instilling water or saline to fill the cavity. The colonic lesion “floats” in a lumen filled with fluid, and the muscularis propria retains a circular configuration and does not follow involutions of the mucosa and submucosa even during peristaltic contractions (Figure 3), making it easier to snare the lesion[33] (Table 2).
In recent years, meta-analysis has supported that UEMR resection achieves a higher en bloc resection rate and less post-endoscopic resection recurrence compared to conventional EMR, especially when polyps greater than or equal to 20 mm are resected. In contrast, no significant differences were detected with respect to the occurrence of adverse events[34,35].
In daily clinical practice, UEMR is very useful due to its effectiveness, safety and easy learning. This technique can be used for the resection of scar lesions with no lifting, adjacent tattoo, incomplete resection attempts, lesions into a colonic diverticulum, in the ileocecal valve with ileal component and lesions with intra-appendicular involvement[36].
UEMR may also be useful for en bloc resection of pseudodepressed less than or equal to 2 cm LST-NG in which en bloc resection is mandatory due to the high risk of SMI[33].
Another advantage of UEMR is that it is a “reversible” technique. In the case that en bloc resection of a high-risk lesion does not seem feasible, all the water can be aspirated, and the technique can be changed either to ESD or EFTR.
EFTR is an emerging technique for removal of complex colorectal lesions. Since the introduction of the full thickness resection device (FTRD; Ovesco Endoscopy AG, Tübingen, Germany) in Germany in 2014[37] several studies have reported encouraging results on the short-term safety and efficacy of EFTR[38,39].
To perform an EFTR, the lateral margins of the lesion are first marked with the probe that is part of the set or by other means (e.g., snare tip coagulation or argon plasma). Thereafter, the colonoscope is retracted, and the FTRD is mounted and advanced to the target lesion. The lesion is then pulled into the resection cap with the grasping forceps. After deployment of the clip, the snare is closed, and the tissue is cut. To avoid unintended incorporation of organs next to the colonic wall, traction of the target lesion without suction is recommended, and when necessary, suction should be performed very gently and with caution. After resection, the specimen is recovered, and inspection of the resection site to check for the correct position of the over-the-scope clip is mandatory. For colonic lesions, prOVECAP (Ovesco Endoscopy, Tübingen, Germany), a cap similar in size to the FTRD cap, can be mounted on the instrument tip to evaluate accessibility to the target lesion. The keys to technical success are the right size of the lesion, performing correct traction and coordinated teamwork[40].
General indications for EFTR are residual adenoma after endoscopic resection, non-lifting sign adenoma, histological R1 resection (deep and lateral positive margins at histology), suspected T1 carcinoma, adenomas at difficult anatomic locations (appendiceal orifice, diverticulum, folds) and subepithelial lesions[38,39].
Among the indications for EFTR, we highlight the treatment of polyps with suspected malignancy due to its clinical impact because in most cases the acquired tissue allows an exact histologic risk stratification to assign patients individually to the best treatment and avoid surgery for low-risk lesions. In a retrospective multicenter study that included 64 patients with incomplete resection of malignant polyps, the performance of EFTR obviated the need for surgery in most of these patients (84%) by classifying them as low risk and therefore may be the method of choice for this indication[41].
A recent meta-analysis including nine studies conducted in European countries with 469 Lesions showed a pooled rate of technical success, full thickness resection and R0 resection of 94.0% (95%CI: 89.8%-97.3%), 89.5% (95%CI: 83.9%-94.2%) and 84.9% (95%CI: 75.1%-92.8%), respectively; a pooled estimate of bleeding, perforation and post-polypectomy syndrome of 2.2% (95%CI: 0.4%-4.9%), 0.19% (95%CI: 0.00-1.25%) and 2.3% (95%CI: 0.1%-6.3%), respectively and pooled rates of residual/recurrent adenoma and surgery for any reason of 8.5% (95%CI: 4.1%-14.0%) and 6.3% (2.4%-11.7%), respectively. These results show that EFTR with an FTRD system is efficient and safe for treating non-lifting and invasive colorectal lesions with conventional EMR and ESD criteria[42]. Nonetheless, future studies are needed to investigate the role of EFTR in large colorectal lesions and specify its indications.
ESD was first described in Japan for the treatment of early gastric cancer and adopted for the treatment of colonic lesions. It is the only procedure that allows complete en bloc resection regardless of the size of the lesion.
It is a technically demanding procedure, requires a long learning curve and requires a longer procedure time than EMR[43]. Adverse events are more common for ESD than for EMR, with published perforation rates of about 5%[44]. Nevertheless, the safety profile is adequate because almost all ESD complications can be managed endoscopically, and the risk of surgery related to post-ESD complications (2%) is low[45].
It basically consists of entering the submucosal space, which is a virtual space that we will create with a solution injected into the submucosa. The classic technique includes marking the lesion to be resected and injecting a lifting agent into the submucosa at its periphery. Using the endoscopic knife, the mucosa is incised circumferentially, and the lesion is separated from the muscularis propria. Additional submucosal injections are performed as necessary to lift the central portion of the lesion to allow for complete resection. Other strategies for ESD have been described, such as pocket-creation method or tunnel[46]. Traction is recommended for colonic lesions, e.g., using rubber band-clip technique because it can significantly decrease the procedure time, increase the en bloc resection rate and the R0 resection rate[47].
There are several tips thoroughly commented on elsewhere in the literature[48].
If there is no complication during the procedure and there are no special risk factors, then the patient could be discharge within 1-3 h after EMR/UEMR or ESD of small lesions, or 24 h or less after EFTR. If there are symptoms, risk factors for complications or special situations (very large lesion), then a longer period of observation might be consider. If there is any sign of complication (pain with abdominal distension, vomiting, rectal bleeding, fever) perform a blood test and or computed tomography scan according to the clinical suspicion and act according to the results. If perforation with peritonitis is suspected, then surgery should be evaluated[49].
It is very important to differentiate post-polypectomy syndrome, a benign complication with a good prognosis in most cases that can be treated medically[50], secondary to excess coagulation and thermal injury of the colonic wall in which computed tomography scan may show a severe mural thickening with stratified enhancement pattern with surrounding infiltration but no air[51]. It is extremely important to recognize deep mural injury (DMI) signs such as the target sign during or immediately after finishing the EMR using the Sydney Classification of DMI (Table 3)[52].
| Sydney Classification of deep mural injury | |
| Type 0 | Normal defect. Blue mat appearance of obliquely oriented intersecting submucosal connective tissue fibers (with a blue dye such as indigo carmine or methylene blue) |
| Type 1 | MP visible but no mechanical injury (“Whale” sign) |
| Type 2 | Focal loss of the submucosal plane raising concern for MP injury or rendering the MP defect uninterpretable |
| Type 3 | MP injured, specimen target sign or defect mirror target sign identified |
| Type 4 | Actual hole within a white cautery ring, no observed contamination |
| Type 5 | Actual hole within a white cautery ring, observed contamination |
The right colon (and cecum) is the thinner part of colon and might be more prone to complications such as perforation, but in one study it seemed that the transverse colon might have more incidence of DMI. The transverse colon is highly mobile, and it has a long mesentery. It is possible that the muscular propria could be more mobile and be trapped easily without “feeling” that we snare the muscular layer.
If there are signs of DMI, then an endoscopic treatment could be offered according to the experience of the endoscopist by using through-the-scope clips for iatrogenic perforations less than 1 cm and the use of the over-the-scope clip could be considered for defects 1-2 cm[53]. For larger iatrogenic perforations, endoscopic treatments with endoscopic suturing or a polyloop and clips method using a double-channel or single-channel endoscope have been described[54,55].
Prophylactic clipping of muscular injury (target signs) might protect against delayed clinical perforation. If the perforation had leakage of colonic fluid, then a surgical approach might be a better option.
Bleeding is a frequent complication of EMR and ESD. Intraprocedural bleeding (IPB) is relatively common, being most of the time an auto limited event from cutting small capillary vessels or vessels that may require coagulation. The IPB rate in the literature is over 10%. In an observational multicenter study that analyzed data from EMR of sessile colorectal polyps greater than or equal to 20 mm in size (mean size: 35.5 mm) of 1172 patients, IPB was observed in 133 (11.3%)[56].
The small bleeding during procedure could be minimized by adding diluted adrenaline to the submucosal injection solution and could be treated with coagulating current using the tip of the snare
IPB that requires endoscopic treatment is associated with a longer procedure time, higher risk of clinically significant post procedural bleeding and recurrence at first surveillance after piecemeal EMR[56].
Post procedural bleeding is also relatively frequent. In a prospective study involving 1039 patients after EMR, 6% had a clinically significant delayed post-polypectomy bleeding, 21% of them (13 patients) being unstable and 26% (16 patients) requiring blood transfusion. Most of the patients (55%) were managed conservatively, 44% underwent colonoscopy, and 1 patient required primary embolization and surgery[57].
To control the active bleeding after EMR or ESD, mechanical therapy (e.g., through-the-scope/cap-mounted clips) and/or contact thermal coagulation are helpful. In cases of inadequate or failed hemostasis with ongoing bleeding, hemostatic topical agents can be used as a secondary treatment option[58].
The risk factors for clinically significant delayed post procedural bleeding include lesions larger than 3 or 4 cm, located in the proximal colon, elderly patients, patients with major comorbidities, taking antiplatelets and absence of use of epinephrine. Two scores have been published to predict the risk of delayed bleeding in two different populations, with similar results summarized in Table 4[59,60].
| Age ≥ 75-yr-old | Lesion ≥ 40 mm | ASA III-IV | Location proximal to transverse colon | Aspirin | Clips | |
| Yes | 1 | 1 | 1 | 3 | 2 | 0 |
| No | 0 | 0 | 0 | 0 | 0 | 2 |
| Risk of bleeding after EMR | ||||||
| Low risk 0.6% (0.2%-1.8%) | 0-3 points | |||||
| Medium risk 5.5% (3.8%-7.9%) | 4-7 points | |||||
| Elevated risk 40% (21.8%-61.1%) | 8-10 points | |||||
Prophylactic endoscopic coagulation with a coagulating forceps (with low-power coagulation) does not seem to significantly decrease the incidence of clinically significant post-EMR bleeding. Nonetheless, a recent meta-analysis has shown benefit when clipping polyps measuring greater than or equal to 20 mm, especially in the proximal colon[61].
In recent years, coverage agents have been developed to cover large mucosal defects that appear to be effective in the prevention of late complications, but randomized controlled trials and head-to-head comparative studies of shielding products are still needed[62].
Recurrence or residual neoplastic tissue after EMR can be easily solved endoscopically in most of cases during surveillance since treatment after first revision is usually successful.
Early recurrence of large conventional adenomas seems to be around 16% at first surveillance colonoscopy (SC), with a cumulative recurrence around 20% after second SC 1 year after and around 28% after 2 years. Large sessile serrated adenomas/polyp recurrence seems to be lower, at about 7% from 12 mo onwards[7].
First SC at 3-6 mo after piecemeal EMR of polyps greater than or equal to 20 mm is recommended for scar assessment and the intervals to the next colonoscopy at 1 year and then 3 years[4,30]. It has been published that after EMR of lesions smaller than 4 cm without significant intraprocedural bleeding (not requiring endoscopic treatment) and with low-grade dysplasia, the first SC can be safely scheduled at 18 mo. The Sydney EMR recurrence tool (Table 5) was developed to help predict the risk of recurrence after piecemeal EMR, with a 92% negative predictive value for recurrence at first SC, for Sydney EMR recurrence tool 0 lesions[63]. It is also very important to treat other synchronic lesions, clear the rest of the colon or rule out a serrated polyposis in cases of resection of large serrated lesions.
| Risk factor | Score |
| LST size ≥ 40 mm | 2 |
| IPB requiring endoscopic control | 1 |
| High-grade dysplasia | 1 |
| Total | 4 |
| Cumulative incidence of EDR% (standard error) | |
| SERT = 0 | 9.8% (2.2); 6 mo FU |
| 11.6% (2.5); 18 mo FU | |
| SERT = 1-4 | 23.0% (2.5); 6 mo FU |
| 36.3% (3.2); 18 mo FU | |
It is very important to carefully inspect the scar. The scar might be identified as a pale area with disruption of vascular pattern or fold convergence. All the edges and center of the scar should be interrogated, looking for a transition point where a non-neoplastic pit or vascular pattern turns into a neoplastic pattern (Kudo pit pattern, NBI International Colorectal Endoscopic and JNET classification) and being aware of post-EMR scar clip artifact using a high-definition endoscope with optical narrow band technology[64].
In surveillance cases with local recurrence, endoscopic resection with repeat EMR, snare or avulsion method can be performed, and ablation of the perimeter of the post-treatment site may be considered. If there is a retained clip in the scar, the procedure should be the same. In case there is a suspicious area of residual polyp, the retained clip should not prevent endoscopic resection of the residual tissue[4,30].
The actual problems of EMR are the treatment of fibrotic tissues or non-lifting tissues as well as difficult areas for endoscopic resection.
Peri/intra-appendicular orifice lesions
In this scenario, EMR is a technical challenge because of difficult endoscopic access due to the narrow lumen of the appendix and thin colonic wall at the base of the cecum, which means a high risk of perforation. Nonetheless in expert hands, it is a safe and effective treatment, but if more than 50% of the circumference of the appendicular orifice (AO) is involved, then surgery should be considered[65]. As it is a narrow area, injection must be small to avoid narrowing the working field, and use of mini snares is helpful.
UEMR has been shown to enable safe resection of AO lesions, especially those limited to the rim. In a series of 27 consecutive patients with AO adenomas (median size 15 mm, range 8-50 mm), 89% successful resection was achieved, with 59% of lesions being resected en bloc. Post-polypectomy syndrome occurred in 7% of cases. No other complications occurred, and over a median follow-up of 29 wk only 10% of patients (n = 2) had residual adenoma present[66].
With underwater submersion, the appendix can partially evert into the cecal lumen, and the colonic lesion “floats” in a lumen filled with water. This allows endoscopic resection without previous submucosal injection, which makes lesions that affect the AO more accessible to endoscopic resection. To maximize tissue capture, contraction of the muscularis propria followed by the torque-and-crimp technique can be expected with the open loop[32]. In cases of residual tissue deep in the AO, a combination of air suction and more water infusion can help to evert residual tissue, making it accessible for snare resection[36].
ESD for lesions located in close proximity to the AO remains a challenging technique. In a retrospective study that included 76 lesions, en bloc resection was achieved in 72 (94.7%) and median tumor size was 36 mm (10-110 mm). One patient experienced intraoperative perforation, was treated by clip closure, later developed appendicitis and underwent emergency ileocecal surgical resection; another patient experienced postoperative appendicitis and recovered with antibiotic treatment. Despite the challenges of working in the region of the cecum and AO, this study demonstrates that ESD performed by skilled and experienced endoscopists can be a safe and effective technique[67].
EFTR is another endoscopic treatment option. In a multicenter study in Germany that included 50 lesions, with mean size of 18 mm, EFTR was technically successful in 48 (96%), and R0 resection was achieved in 32 patients (64%). Post interventional appendicitis occurred in 7 patients (14%) during follow-up, and conservative treatment was sufficient in half of the cases[68]. The authors believe that the EFTR of appendicular lesions is a promising modality in a certain group of patients, but further studies are required to prospectively evaluate the feasibility and safety of this technique.
A new injection and a mini/small snare should be tried. If it is not possible to snare, then sometimes the suction pseudopolyp technique or precutting with the tip of the snare around the non-lifting area may help. Otherwise, cold avulsion with forceps and snare tip soft coagulation/ablation of the scar area seems to be helpful in small areas of benign residual tissue. In this situation, UEMR and band ligation with or without resection can also be performed.
If it is not possible to resect with the inject and resect technique, then the non-lifting part of the lesion could be resected by cold avulsion (forceps), pre-cutting EMR[69], UEMR, ESD, EFTR[42] or surgery (the latter especially if there are suspicious areas of SMI). The same recommendation would apply to fibrotic lesions secondary to tattoo, multiple biopsies, the biology of the lesion or SMI, showing non-lifting sign, “jet sign” or canyoning. The authors find UEMR especially useful in this situation for benign lesions. As it is a “reversible” technique, if it is not suitable, then another technique like ESD or EFTR could be performed during the same session. If there is suspicion of malignancy, then surgery or EFTR might be preferable.
It is very important to define the borders of the lesion and if the ileum is involved, then sometimes a cap is helpful[27]. In cases of classic EMR, the amount of submucosal injection should be small if there is a flat lesion over the ileocecal valve to avoid excessive tension in the submucosal cushion since it is very easy that the snare slips while closing in this situation. A mini snare may be helpful when the ileum is involved. It is a safe procedure, and stenosis after EMR seems to be rare. Although it is complex, successful EMR seems to be greater than 90% in experienced hands. Extensive involvement of the terminal ileum or both ileocecal valve lips are associated with EMR failure[70]. UEMR is a good option, and the one preferred by the authors at this location.
Because of the innervation in distal rectum, the use of long-acting local anesthetic (ropivacaine or bupivacaine) in the submucosal injectate (avoiding intravascular injection and requiring cardiac monitoring) for submucosal injection around the anorectal region and prophylactic antibiotics should be considered[28]. The use of a gastroscope for increased mobility and retroflexion may be helpful. It is safe to perform the endoscopic resection over the dentate line and hemorrhoidal columns. When performing ESD at this location, the operator should be aware that there could be muscular fibers on the submucosal layer on this location (it is the exception in the gastrointestinal tract).
It is a subjective term, which covers different situations, such as scope instability. Working using retroversion (easier with a gastroscope or a pediatric colonoscope) might stabilize the endoscope facilitating the resection sometimes. In the proximal colon, a distal attachment such as Endocuff or using a balloon enteroscope or a double balloon platform (Dilumen, Lumendi, Westport, Conn, United States) might help to stabilize the scope.
There are different endoscopic techniques for the resection of complex colorectal LST that the therapeutic colonoscopist should be aware of. EMR (inject and resect) is useful for most colorectal benign lesions. UEMR is a very useful technique since it avoids the need for submucosal injection. It might be a very good alternative in non-lifting lesions or in difficult locations like ileocecal valve, AO, narrow sigmoid or peridiverticular area where there is a narrow space where injection could make the access more difficult. ESD is the only technique that allows en bloc resection regardless of the size of the lesion, being especially useful for large LSTs that harbor risk for SMI, for example large LST with big nodules in the rectosigmoid area. EFTR on the other hand is the technique that allows the deepest margins and because of that might be the best choice for endoscopic resection of less than 2.5 cm suspected malignant LST.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: Asociación Española de Gastroenterología, 1646.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Fiori E S-Editor: Zhang H L-Editor: A P-Editor: Zhang H
| 1. | Fernández-Esparrach G, Calderón Á, De-la-Peña J, Díaz-Tasende JB, Esteban JM, Gimeno-García AZ, Herreros-de-Tejada A, Martínez-Ares D, Nicolás-Pérez D, Nogales Ó, Ono A, Orive-Calzada A, Parra-Blanco A, Rodríguez-Muñoz S, Sánchez-Hernández E, Sánchez-Yague A, Vázquez-Sequeiros E, Vila J, López-Rosés L; Sociedad Española de Endoscopia Digestiva (SEED). Endoscopic submucosal dissection. Sociedad Española de Endoscopia Digestiva (SEED) clinical guideline. Rev Esp Enferm Dig. 2014;106:120-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Ferlitsch M, Moss A, Hassan C, Bhandari P, Dumonceau JM, Paspatis G, Jover R, Langner C, Bronzwaer M, Nalankilli K, Fockens P, Hazzan R, Gralnek IM, Gschwantler M, Waldmann E, Jeschek P, Penz D, Heresbach D, Moons L, Lemmers A, Paraskeva K, Pohl J, Ponchon T, Regula J, Repici A, Rutter MD, Burgess NG, Bourke MJ. Colorectal polypectomy and endoscopic mucosal resection (EMR): European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2017;49:270-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 559] [Cited by in RCA: 766] [Article Influence: 95.8] [Reference Citation Analysis (0)] |
| 3. | Tanaka S, Kashida H, Saito Y, Yahagi N, Yamano H, Saito S, Hisabe T, Yao T, Watanabe M, Yoshida M, Saitoh Y, Tsuruta O, Sugihara KI, Igarashi M, Toyonaga T, Ajioka Y, Kusunoki M, Koike K, Fujimoto K, Tajiri H. Japan Gastroenterological Endoscopy Society guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection. Dig Endosc. 2020;32:219-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 272] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 4. | Kaltenbach T, Anderson JC, Burke CA, Dominitz JA, Gupta S, Lieberman D, Robertson DJ, Shaukat A, Syngal S, Rex DK. Endoscopic Removal of Colorectal Lesions-Recommendations by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2020;91:486-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 93] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 5. | Zauber AG, Winawer SJ, O'Brien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, Shi W, Bond JH, Schapiro M, Panish JF, Stewart ET, Waye JD. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1952] [Cited by in RCA: 2286] [Article Influence: 175.8] [Reference Citation Analysis (2)] |
| 6. | Sano Y, Tanaka S, Kudo SE, Saito S, Matsuda T, Wada Y, Fujii T, Ikematsu H, Uraoka T, Kobayashi N, Nakamura H, Hotta K, Horimatsu T, Sakamoto N, Fu KI, Tsuruta O, Kawano H, Kashida H, Takeuchi Y, Machida H, Kusaka T, Yoshida N, Hirata I, Terai T, Yamano HO, Kaneko K, Nakajima T, Sakamoto T, Yamaguchi Y, Tamai N, Nakano N, Hayashi N, Oka S, Iwatate M, Ishikawa H, Murakami Y, Yoshida S, Saito Y. Narrow-band imaging (NBI) magnifying endoscopic classification of colorectal tumors proposed by the Japan NBI Expert Team. Dig Endosc. 2016;28:526-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 455] [Cited by in RCA: 402] [Article Influence: 44.7] [Reference Citation Analysis (1)] |
| 7. | Pellise M, Burgess NG, Tutticci N, Hourigan LF, Zanati SA, Brown GJ, Singh R, Williams SJ, Raftopoulos SC, Ormonde D, Moss A, Byth K, P'Ng H, Mahajan H, McLeod D, Bourke MJ. Endoscopic mucosal resection for large serrated lesions in comparison with adenomas: a prospective multicentre study of 2000 lesions. Gut. 2017;66:644-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 92] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 8. | Bronsgeest K, Huisman JF, Langers A, Boonstra JJ, Schenk BE, de Vos Tot Nederveen Cappel WH, Vasen HFA, Hardwick JCH. Safety of endoscopic mucosal resection (EMR) of large non-pedunculated colorectal adenomas in the elderly. Int J Colorectal Dis. 2017;32:1711-1717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Kudo S, Kashida H, Nakajima T, Tamura S, Nakajo K. Endoscopic diagnosis and treatment of early colorectal cancer. World J Surg. 1997;21:694-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 165] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 10. | Kudo Se, Lambert R, Allen JI, Fujii H, Fujii T, Kashida H, Matsuda T, Mori M, Saito H, Shimoda T, Tanaka S, Watanabe H, Sung JJ, Feld AD, Inadomi JM, O'Brien MJ, Lieberman DA, Ransohoff DF, Soetikno RM, Triadafilopoulos G, Zauber A, Teixeira CR, Rey JF, Jaramillo E, Rubio CA, Van Gossum A, Jung M, Vieth M, Jass JR, Hurlstone PD. Nonpolypoid neoplastic lesions of the colorectal mucosa. Gastrointest Endosc. 2008;68:S3-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 364] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 11. | Ichikawa Y, Nagashima Y, Morioka K, Akimoto K, Kojima Y, Ishikawa T, Goto A, Kobayashi N, Watanabe K, Ota M, Fujii S, Kawamata M, Takagawa R, Kunizaki C, Takahashi H, Nakajima A, Maeda S, Shimada H, Inayama Y, Ohno S, Endo I. Colorectal laterally spreading tumors show characteristic expression of cell polarity factors, including atypical protein kinase C λ/ι, E-cadherin, β-catenin and basement membrane component. Oncol Lett. 2014;8:977-984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Wang X, Li A, Guo Y, Wang Y, Zhao X, Xiang L, Han Z, Li Y, Xu W, Zhuang K, Yan Q, Zhong J, Xiong J, Liu S. iTRAQ-Based Proteomics Screen identifies LIPOCALIN-2 (LCN-2) as a potential biomarker for colonic lateral-spreading tumors. Sci Rep. 2016;6:28600. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Hiraoka S, Kato J, Tatsukawa M, Harada K, Fujita H, Morikawa T, Shiraha H, Shiratori Y. Laterally spreading type of colorectal adenoma exhibits a unique methylation phenotype and K-ras mutations. Gastroenterology. 2006;131:379-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 14. | Voorham QJ, Rondagh EJ, Knol DL, van Engeland M, Carvalho B, Meijer GA, Sanduleanu S. Tracking the molecular features of nonpolypoid colorectal neoplasms: a systematic review and meta-analysis. Am J Gastroenterol. 2013;108:1042-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 15. | Bogie RMM, Veldman MHJ, Snijders LARS, Winkens B, Kaltenbach T, Masclee AAM, Matsuda T, Rondagh EJA, Soetikno R, Tanaka S, Chiu HM, Sanduleanu-Dascalescu S. Endoscopic subtypes of colorectal laterally spreading tumors (LSTs) and the risk of submucosal invasion: a meta-analysis. Endoscopy. 2018;50:263-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 16. | Kim KO, Jang BI, Jang WJ, Lee SH. Laterally spreading tumors of the colorectum: clinicopathologic features and malignant potential by macroscopic morphology. Int J Colorectal Dis. 2013;28:1661-1666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Marín-Gabriel JC, Romito R, Guarner-Argente C, Santiago-García J, Rodríguez-Sánchez J, Toyonaga T. Use of electrosurgical units in the endoscopic resection of gastrointestinal tumors. Gastroenterol Hepatol. 2019;42:512-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 18. | Morris ML, Tucker RD, Baron TH, Song LM. Electrosurgery in gastrointestinal endoscopy: principles to practice. Am J Gastroenterol. 2009;104:1563-1574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Kodashima S, Fujishiro M. Novel image-enhanced endoscopy with i-scan technology. World J Gastroenterol. 2010;16:1043-1049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 130] [Cited by in RCA: 151] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 20. | Voudoukis E, Tribonias G, Tavernaraki A, Theodoropoulou A, Vardas E, Paraskeva K, Chlouverakis G, Paspatis GA. Use of a double-channel gastroscope reduces procedural time in large left-sided colonic endoscopic mucosal resections. Clin Endosc. 2015;48:136-141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Kim SY, Chung JW, Kim JH, Kim YJ, Kim KO, Kwon KA, Park DK. Carbon dioxide insufflation during endoscopic resection of large colorectal polyps can reduce post-procedure abdominal pain: A prospective, double-blind, randomized controlled trial. United European Gastroenterol J. 2018;6:1089-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Moss A, Bourke MJ, Metz AJ. A randomized, double-blind trial of succinylated gelatin submucosal injection for endoscopic resection of large sessile polyps of the colon. Am J Gastroenterol. 2010;105:2375-2382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 23. | Yandrapu H, Desai M, Siddique S, Vennalganti P, Vennalaganti S, Parasa S, Rai T, Kanakadandi V, Bansal A, Titi M, Repici A, Bechtold ML, Sharma P, Choudhary A. Normal saline solution versus other viscous solutions for submucosal injection during endoscopic mucosal resection: a systematic review and meta-analysis. Gastrointest Endosc. 2017;85:693-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 24. | Lisotti A, Marocchi G, Calì A, Fusaroli P. Endoscopic mucosal resection of large colonic laterally spreading tumors using a dedicated viscous solution for submucosal injection (ORISE gel): a short case series (with video). Eur J Gastroenterol Hepatol. 2021;33:650-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 25. | Wedi E, Koehler P, Hochberger J, Maiss J, Milenovic S, Gromski M, Ho N, Gabor C, Baulain U, Ellenrieder V, Jung C. Endoscopic submucosal dissection with a novel high viscosity injection solution (LiftUp) in an ex vivo model: a prospective randomized study. Endosc Int Open. 2019;7:E641-E646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Rivero-sanchez L, Ortiz O, Pellise M. Chromoendoscopy Techniques in Imaging of Colorectal Polyps and Cancer : Overview and Practical Applications for Detection and Characterization. Tech Innov Gastrointest Endosc. 2021;23:30-41. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 27. | Lew D, Kashani A, Lo SK, Jamil LH. Efficacy and safety of cap-assisted endoscopic mucosal resection of ileocecal valve polyps. Endosc Int Open. 2020;8:E241-E246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 28. | La Regina D, Mongelli F, Fasoli A, Lollo G, Ceppi M, Saporito A, Garofalo F, Di Giuseppe M, Ferrario di Tor Vajana A. Clinical Adverse Events after Endoscopic Resection for Colorectal Lesions: A Meta-Analysis on the Antibiotic Prophylaxis. Dig Dis. 2020;38:15-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 29. | Kobayashi S, Yamada M, Takamaru H, Sakamoto T, Matsuda T, Sekine S, Igarashi Y, Saito Y. Diagnostic yield of the Japan NBI Expert Team (JNET) classification for endoscopic diagnosis of superficial colorectal neoplasms in a large-scale clinical practice database. United European Gastroenterol J. 2019;7:914-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 30. | Hassan C, Antonelli G, Dumonceau JM, Regula J, Bretthauer M, Chaussade S, Dekker E, Ferlitsch M, Gimeno-Garcia A, Jover R, Kalager M, Pellisé M, Pox C, Ricciardiello L, Rutter M, Helsingen LM, Bleijenberg A, Senore C, van Hooft JE, Dinis-Ribeiro M, Quintero E. Post-polypectomy colonoscopy surveillance: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2020. Endoscopy. 2020;52:687-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 314] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 31. | Klein A, Tate DJ, Jayasekeran V, Hourigan L, Singh R, Brown G, Bahin FF, Burgess N, Williams SJ, Lee E, Sidhu M, Byth K, Bourke MJ. Thermal Ablation of Mucosal Defect Margins Reduces Adenoma Recurrence After Colonic Endoscopic Mucosal Resection. Gastroenterology. 2019;156:604-613.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 189] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 32. | Nett A, Binmoeller K. Underwater Endoscopic Mucosal Resection. Gastrointest Endosc Clin N Am. 2019;29:659-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 33. | Uchima H, Colán-Hernández J, Binmoeller KF. Peristaltic contractions help snaring during underwater endoscopic mucosal resection of colonic non-granular pseudodepressed laterally spreading tumor. Dig Endosc. 2021;33:e74-e76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (2)] |
| 34. | Ni DQ, Lu YP, Liu XQ, Gao LY, Huang X. Underwater vs conventional endoscopic mucosal resection in treatment of colorectal polyps: A meta-analysis. World J Clin Cases. 2020;8:4826-4837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Tziatzios G, Gkolfakis P, Triantafyllou K, Fuccio L, Facciorusso A, Papanikolaou IS, Antonelli G, Nagl S, Ebigbo A, Probst A, Hassan C, Messmann H. Higher rate of en bloc resection with underwater than conventional endoscopic mucosal resection: A meta-analysis. Dig Liver Dis. 2021;53:958-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 36. | Uchima H, Colan-Hernandez J, Caballero N, Marín I, Calafat M, Luna D, Moreno V. Underwater endoscopic mucosal resection of an adenomatous lesion with deep extension into the appendiceal orifice. Endoscopy. 2021;53:334-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 37. | Schmidt A, Damm M, Caca K. Endoscopic full-thickness resection using a novel over-the-scope device. Gastroenterology. 2014;147:740-742.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 38. | Aepli P, Criblez D, Baumeler S, Borovicka J, Frei R. Endoscopic full thickness resection (EFTR) of colorectal neoplasms with the Full Thickness Resection Device (FTRD): Clinical experience from two tertiary referral centers in Switzerland. United European Gastroenterol J. 2018;6:463-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 64] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 39. | Andrisani G, Soriani P, Manno M, Pizzicannella M, Pugliese F, Mutignani M, Naspetti R, Petruzziello L, Iacopini F, Grossi C, Lagoussis P, Vavassori S, Coppola F, La Terra A, Ghersi S, Cecinato P, De Nucci G, Salerno R, Pandolfi M, Costamagna G, Di Matteo FM. Colo-rectal endoscopic full-thickness resection (EFTR) with the over-the-scope device (FTRD®): A multicenter Italian experience. Dig Liver Dis. 2019;51:375-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 40. | Hageman L, Strebus J, van der Spek BW. Endoscopische 'full-thickness'-resectie van colorectale poliepen [Endoscopic full-thickness resection of colorectal polyps]. Ned Tijdschr Geneeskd. 2015;160:A9903. [PubMed] |
| 41. | Kuellmer A, Mueller J, Caca K, Aepli P, Albers D, Schumacher B, Glitsch A, Schäfer C, Wallstabe I, Hofmann C, Erhardt A, Meier B, Bettinger D, Thimme R, Schmidt A; FTRD study group. Endoscopic full-thickness resection for early colorectal cancer. Gastrointest Endosc. 2019;89:1180-1189.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 42. | Li P, Ma B, Gong S, Zhang X, Li W. Efficacy and safety of endoscopic full-thickness resection in the colon and rectum using an over-the-scope device: a meta-analysis. Surg Endosc. 2021;35:249-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 43. | Arezzo A, Passera R, Marchese N, Galloro G, Manta R, Cirocchi R. Systematic review and meta-analysis of endoscopic submucosal dissection vs endoscopic mucosal resection for colorectal lesions. United European Gastroenterol J. 2016;4:18-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 108] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 44. | De Ceglie A, Hassan C, Mangiavillano B, Matsuda T, Saito Y, Ridola L, Bhandari P, Boeri F, Conio M. Endoscopic mucosal resection and endoscopic submucosal dissection for colorectal lesions: A systematic review. Crit Rev Oncol Hematol. 2016;104:138-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 132] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 45. | Thorlacius H, Rönnow CF, Toth E. European experience of colorectal endoscopic submucosal dissection: a systematic review of clinical efficacy and safety. Acta Oncol. 2019;58:S10-S14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 46. | Hayashi Y, Miura Y, Yamamoto H. Pocket-creation method for the safe, reliable, and efficient endoscopic submucosal dissection of colorectal lateral spreading tumors. Dig Endosc. 2015;27:534-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 47. | Tsuji K, Yoshida N, Nakanishi H, Takemura K, Yamada S, Doyama H. Recent traction methods for endoscopic submucosal dissection. World J Gastroenterol. 2016;22:5917-5926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 50] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 48. | Saito Y, Abe S, Inoue H, Tajiri H. How to Perform a High-Quality Endoscopic Submucosal Dissection. Gastroenterology. 2021;161:405-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 49. | Wagner KT, Fung E. Polypectomy Techniques. Surg Clin North Am. 2020;100:1049-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 50. | Kim SY, Kim HS, Park HJ. Adverse events related to colonoscopy: Global trends and future challenges. World J Gastroenterol. 2019;25:190-204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 91] [Cited by in RCA: 168] [Article Influence: 28.0] [Reference Citation Analysis (8)] |
| 51. | Shin YJ, Kim YH, Lee KH, Lee YJ, Park JH. CT findings of post-polypectomy coagulation syndrome and colonic perforation in patients who underwent colonoscopic polypectomy. Clin Radiol. 2016;71:1030-1036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 52. | Burgess NG, Bassan MS, McLeod D, Williams SJ, Byth K, Bourke MJ. Deep mural injury and perforation after colonic endoscopic mucosal resection: a new classification and analysis of risk factors. Gut. 2017;66:1779-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 137] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 53. | Paspatis GA, Arvanitakis M, Dumonceau JM, Barthet M, Saunders B, Turino SY, Dhillon A, Fragaki M, Gonzalez JM, Repici A, van Wanrooij RLJ, van Hooft JE. Diagnosis and management of iatrogenic endoscopic perforations: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement - Update 2020. Endoscopy. 2020;52:792-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 54. | Ryu JY, Park BK, Kim WS, Kim K, Lee JY, Kim Y, Park JY, Kim BJ, Kim JW, Choi CH. Endoscopic closure of iatrogenic colon perforation using dual-channel endoscope with an endoloop and clips: methods and feasibility data (with videos). Surg Endosc. 2019;33:1342-1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Castillo-Regalado E, Huertas C, Torrealba L, Hombrados M, Figa M, Busquets D, Uchima H. Endoscopic full-thickness resection in the rectum closed with PolyLoop-and-clips method using single-channel endoscope. Endoscopy. 2022;54:E24-E25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (2)] |
| 56. | Burgess NG, Metz AJ, Williams SJ, Singh R, Tam W, Hourigan LF, Zanati SA, Brown GJ, Sonson R, Bourke MJ. Risk factors for intraprocedural and clinically significant delayed bleeding after wide-field endoscopic mucosal resection of large colonic lesions. Clin Gastroenterol Hepatol. 2014;12:651-61.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 200] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 57. | Burgess NG, Williams SJ, Hourigan LF, Brown GJ, Zanati SA, Singh R, Tam W, Butt J, Byth K, Bourke MJ. A management algorithm based on delayed bleeding after wide-field endoscopic mucosal resection of large colonic lesions. Clin Gastroenterol Hepatol. 2014;12:1525-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 58. | Triantafyllou K, Gkolfakis P, Gralnek IM, Oakland K, Manes G, Radaelli F, Awadie H, Camus Duboc M, Christodoulou D, Fedorov E, Guy RJ, Hollenbach M, Ibrahim M, Neeman Z, Regge D, Rodriguez de Santiago E, Tham TC, Thelin-Schmidt P, van Hooft JE. Diagnosis and management of acute lower gastrointestinal bleeding: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2021;53:850-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 112] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 59. | Albéniz E, Fraile M, Ibáñez B, Alonso-Aguirre P, Martínez-Ares D, Soto S, Gargallo CJ, Ramos Zabala F, Álvarez MA, Rodríguez-Sánchez J, Múgica F, Nogales Ó, Herreros de Tejada A, Redondo E, Pin N, León-Brito H, Pardeiro R, López-Roses L, Rodríguez-Téllez M, Jiménez A, Martínez-Alcalá F, García O, de la Peña J, Ono A, Alberca de Las Parras F, Pellisé M, Rivero L, Saperas E, Pérez-Roldán F, Pueyo Royo A, Eguaras Ros J, Zúñiga Ripa A, Concepción-Martín M, Huelin-Álvarez P, Colán-Hernández J, Cubiella J, Remedios D, Bessa I Caserras X, López-Viedma B, Cobian J, González-Haba M, Santiago J, Martínez-Cara JG, Valdivielso E, Guarner-Argente C; Endoscopic Mucosal Resection Endoscopic Spanish Society Group. A Scoring System to Determine Risk of Delayed Bleeding After Endoscopic Mucosal Resection of Large Colorectal Lesions. Clin Gastroenterol Hepatol. 2016;14:1140-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 74] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 60. | Bahin FF, Rasouli KN, Byth K, Hourigan LF, Singh R, Brown GJ, Zanati SA, Moss A, Raftopoulos S, Williams SJ, Bourke MJ. Prediction of Clinically Significant Bleeding Following Wide-Field Endoscopic Resection of Large Sessile and Laterally Spreading Colorectal Lesions: A Clinical Risk Score. Am J Gastroenterol. 2016;111:1115-1122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (1)] |
| 61. | Bishay K, Meng ZW, Frehlich L, James MT, Kaplan GG, Bourke MJ, Hilsden RJ, Heitman SJ, Forbes N. Prophylactic clipping to prevent delayed colonic post-polypectomy bleeding: meta-analysis of randomized and observational studies. Surg Endosc. 2022;36:1251-1262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 62. | Lorenzo-Zúñiga V, Bustamante-Balén M, Pons-Beltrán V. Prevention of late complications with coverage agents in endoscopic resection of colorectal lesions: Current landscape in gastrointestinal endoscopy. World J Gastroenterol. 2021;27:1563-1568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 63. | Tate DJ, Desomer L, Klein A, Brown G, Hourigan LF, Lee EY, Moss A, Ormonde D, Raftopoulos S, Singh R, Williams SJ, Zanati S, Byth K, Bourke MJ. Adenoma recurrence after piecemeal colonic EMR is predictable: the Sydney EMR recurrence tool. Gastrointest Endosc. 2017;85:647-656.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 111] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 64. | Kandel P, Brand EC, Pelt J, Ball CT, Chen WC, Bouras EP, Gomez V, Raimondo M, Woodward TA, Wallace MB; EMR SCAR Group. Endoscopic scar assessment after colorectal endoscopic mucosal resection scars: when is biopsy necessary (EMR Scar Assessment Project for Endoscope (ESCAPE) trial). Gut. 2019;68:1633-1641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 65. | Tate DJ, Desomer L, Awadie H, Goodrick K, Hourigan L, Singh R, Williams SJ, Bourke MJ. EMR of laterally spreading lesions around or involving the appendiceal orifice: technique, risk factors for failure, and outcomes of a tertiary referral cohort (with video). Gastrointest Endosc. 2018;87:1279-1288.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 66. | Binmoeller KF, Hamerski CM, Shah JN, Bhat YM, Kane SD. Underwater EMR of adenomas of the appendiceal orifice (with video). Gastrointest Endosc. 2016;83:638-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 67. | Jacob H, Toyonaga T, Ohara Y, Tsubouchi E, Takihara H, Baba S, Yoshizaki T, Kawara F, Tanaka S, Ishida T, Hoshi N, Morita Y, Umegaki E, Azuma T. Endoscopic submucosal dissection of cecal lesions in proximity to the appendiceal orifice. Endoscopy. 2016;48:829-836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 68. | Schmidbaur S, Wannhoff A, Walter B, Meier B, Schäfer C, Meining A, Caca K. Risk of appendicitis after endoscopic full-thickness resection of lesions involving the appendiceal orifice: a retrospective analysis. Endoscopy. 2021;53:424-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 69. | Yoshida N, Inoue K, Dohi O, Yasuda R, Hirose R, Naito Y, Murakami T, Ogiso K, Inada Y, Inagaki Y, Morinaga Y, Kishimoto M, Itoh Y. Efficacy of precutting endoscopic mucosal resection with full or partial circumferential incision using a snare tip for difficult colorectal lesions. Endoscopy. 2019;51:871-876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 70. | Ponugoti PL, Broadley HM, Garcia J, Rex DK. Endoscopic management of large ileocecal valve lesions over an 18-year interval. Endosc Int Open. 2019;7:E1646-E1651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |