Published online Sep 28, 2015. doi: 10.4254/wjh.v7.i21.2352
Peer-review started: April 22, 2015
First decision: June 2, 2015
Revised: June 22, 2015
Accepted: August 30, 2015
Article in press: September 7, 2015
Published online: September 28, 2015
Processing time: 154 Days and 20.8 Hours
Liver cirrhosis, a devastating liver fibrosis caused by hepatitis/inflammation or tumors, is a major comorbid factor in known surgery fields, such as cardiovascular and abdominal surgeries. It is important to review possible comorbid results in neurosurgical procedures in cirrhotic patients. In the reviewed literature, Child-Pugh and model for end-stage liver disease scores are commonly used in the assessment of surgical risks for cirrhotic patients undergoing abdominal, cardiovascular or neurosurgical procedures. The major categories of neurosurgery are traumatic brain injury (TBI), spontaneous intracranial hemorrhage (SICH), brain tumors, and spinal instrumentation procedures. TBI was reported with surgical mortality as high as 34.5% and a complication rate of 87.2%. For SICH, mortality ranged from 22.7% to 47.0%, while complications were reported to be 43.2%. Less is discussed in brain tumor patients; still the postoperative hemorrhage rate approached 26.7%. In spinal fusion instrumentation procedures, the complication rate was as high as 41.0%. Preoperative assessment and correction could possibly decrease complications such as hemorrhage, wound infection and other cirrhosis-related complications (renal, pulmonary, ascites and encephalopathy). In this study, we reviewed the neurosurgical-related literature with regard to liver cirrhosis as a prognostic factor influencing neurosurgical outcomes.
Core tip: Liver cirrhosis is a major comorbid factor for surgical patients, including neurosurgery. We reviewed published articles in the field of neurosurgical procedures. For the high incidence of morbidity/mortality rate, in cirrhotic patients, procedures should be carefully assessed and managed aggressively toward the coagulopathy and nutritional status.
- Citation: Chen CC, Huang YC, Yeh CN. Neurosurgical procedures in patients with liver cirrhosis: A review. World J Hepatol 2015; 7(21): 2352-2357
- URL: https://www.wjgnet.com/1948-5182/full/v7/i21/2352.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i21.2352
Liver cirrhosis has been identified as a risk factor for increased morbidity and mortality in surgical patients[1-9], trauma patients[3-5,9], and patients undergoing emergent major surgery such as laparotomy, cardiac, and thoracic surgery[1,5,9-12]. A search of the literature found overall mortality rates as high as 45%-50% for patients with liver cirrhosis undergoing emergent nonhepatic surgery[8,13-17] and 40% for trauma surgery[3-5]. Furthermore, liver cirrhosis was also associated with increased postoperative morbidity and longer hospitalization in patients undergoing elective nonhepatic surgery[13-15,18]. Liver cirrhosis, and its associated bleeding tendency, is also a challenge for neurosurgeons in brain and spine surgery. For example, spontaneous intracranial hemorrhage (SICH)[19,20] and traumatic head injury[21,22] in cirrhotic patients have been reported in the literature; however, such reports have been sporadic, and relatively little has been published regarding cirrhotic patients who undergo neurosurgical procedures.
In our previous study[21], we found the overall complication rate for brain surgery with liver cirrhosis was 52.1% and the mortality rate was 24.3%. These high rates emphasize the importance of reviewing the recommendations for patients harboring this devastating systemic disease, including for patients needing neurosurgical procedures. The following study reviewed the risks and outcomes accompanying neurosurgical procedures in patients with liver cirrhosis.
For patients with traumatic brain injury (TBI), liver cirrhosis is a significant risk factor for postoperative complications. Over a 5-year-period, according to the National Trauma Databank in America[22], cirrhotic patients with TBI significantly experienced more ventilator days compared with noncirrhotic patients (2.9 ± 6.4 d vs 2.0 ± 6.4 d, P < 0.001). In addition, overall mortality was almost two-fold higher for cirrhotic compared with noncirrhotic TBI patients (34.0% vs 18.1%, OR = 2.34, 95%CI: 1.05-5.20, P = 0.035).
In our previous study[21], we found the complication, rebleeding and mortality rates reached 84.4%, 68.8%, and 37.5%, respectively, in acute TBI patients. According to the Child-Pugh score system (Child), the complication rate increased in a step-wise fashion from 38.7% to 60.0% and then 84.2%, the rebleeding rate from 29.3% to 48.0% and then 63.2%, and the mortality rate from 5.3% to 38.0% and then 63.2% for Child A, B and C patients, respectively. Child classification was significantly associated with higher risk of complication [Child B vs A with odds ratio: 2.84 (95%CI: 1.28-6.29), and Child C vs A: 5.39 (95%CI: 1.32-22.02)] and risk of mortality [Child C vs A: 30.43 (95%CI: 7.71-120.02), and Child B vs A: 10.88 (95%CI: 3.42-34.63)].
Notably, in patients undergoing minimally invasive burr holes for chronic subdural hemorrhage, the rebleeding/recurrent rate was 66.7% (10/15 patients) and mortality rate was 33.3% (5/15 patients). Uncontrolled encephalopathy, varices bleeding or associated pneumonia, sepsis and multiple organ failure were hypothesized to be causes of mortality.
Liver cirrhosis is a well-known risk factor for SICH[19,20,23], despite the relatively rare occurrence of SICH in cirrhotic patients (incidence ranged from 0.7% to 0.8%)[19,20]. However, SICH is life-threatening and is sometimes overlooked due to the similarities of neurologic deficits caused by hepatic encephalopathy; most of the cirrhotic patients who developed SICH were sent to the emergency room for resuscitation and then admitted to the neurosurgery wards for surgical intervention or to the neurology wards for medical treatment if no surgical indication.
Surgical procedures to decrease intracranial pressure in SICH cases, such as craniotomy (hematoma removal) or ventricular drainage (cerebrospinal fluid diversion), were performed only in life-threatening instances. As shown in our previous study, the surgical complication rate was 43.2%, the rebleeding rate was 36.4%, and the mortality rate was 22.7%[21].
Thus, some cirrhotic patients with SICH who underwent medical treatment experienced poor neurologic outcomes even after surgery, including brainstem failure due to delayed medical intervention or severe coagulopathy. The overall mortality rate of cirrhotic patients with SICH, with or without surgery, has been reported as high as 47.0%[20]. Outcomes were associated with the size of the hematoma (P < 0.005) and with the initial Glasgow Coma Scale score (P < 0.05) and the Child-Pugh classification (P = 0.05)[20].
There is little literature discussing the outcome for cirrhotic patients with brain tumors who underwent craniotomy. Jiang et al[24] reported a median survival of 3 (range 2.2-3.8) mo for 41 cases even with successful intracranial excision of brain metastasis from hepatocellular carcinoma (HCC). We reported 15 cases of brain tumor (meningioma, glioma, pituitary adenoma, lymphoma, and metastasis from breast cancer and HCC) patients who received neurosurgical procedures (craniotomy/biopsy)[21]. Four of these patients (26.7%) had immediate postoperative intracranial hemorrhage.
Despite the lack of clear evidence-based indications nor contraindications for cirrhotic patients with brain tumors, we presumed that the preoperative assessment of neurological deficits, Karnofsky performance scale, and corrections of pre-existing coagulopathy may be crucial for the patients’ outcome.
Spine surgery is a common procedure in some neurosurgery centers (up to 70% of daily practice). Most spine surgeries are elective and less invasive, involving a small amount of intraoperative blood loss. Instrumentations for spinal stability have been widely used in patients with degenerative lumbar and cervical spine diseases to enhance stability and improve clinical results. However, instrumentation inevitably encounters more extensive wound exposure, bony destruction, and intraoperative blood loss. Published literature has shown that the complication rate of lumbar fusion instrumentation ranged from 3% to 15%[25,26]. In a retrospective study of lumbar instrumentation by Liao et al[27], patients in the cirrhotic group had significantly more blood loss (P = 0.049) and significantly longer hospitalization (P = 0.023). Complication rate was also significantly higher in the cirrhotic patient group than in the control group (41% vs 10%, P = 0.007). Patients with Child Class B score had a significantly higher incidence of complications than those with Child Class A (86% vs 27%, P = 0.006). Subanalysis in patients with Child Class A showed that those with a Child score of 6 also had higher complication rates than those with a score of 5 (P = 0.001).
Child score, hypoalbuminemia, ascites, and increased blood loss were identified risk factors for complications, and deteriorated hepatic encephalopathy would contribute to unsatisfactory outcomes. Whenever elective instrumented lumbar surgeries are being considered for cirrhotic patients, preoperative correction could reduce or avoid these postoperative complications.
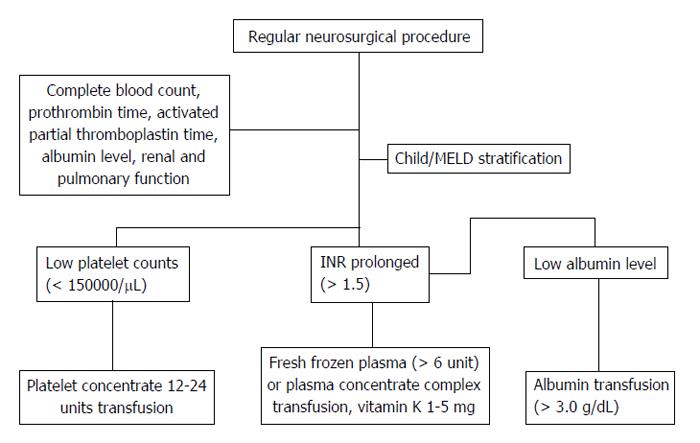
For patients with cirrhotic liver, Child-Pugh[28] and model for end-stage liver disease (MELD)[29] scores are both relied on for the prognosis prediction. The Child system contains serum bilirubin/albumin level, prothrombin time, ascites, and hepatic encephalopathy, while MELD includes serum bilirubin, creatinine, and etiology. Concerns for surgical procedures in cirrhotic patients mostly focused on the impact on coagulopathy, including clotting and the fibrinolytic system. We proposed an algorithm to assess the risk for complications and management of cirrhotic patients receiving regular neurosurgical procedures (Figure 1).
Vitamin K is essential for factor II, VII, IX and X and protein C, S and Z[30]. Liver cirrhosis is a common etiology for vitamin K deficiency due to poor nutrition or cholestasis. In a meta-analysis, intravenous vitamin K supplement from 1 to 5 mg may reverse the excess anticoagulants[31]. Thus, in spontaneous intracranial hemorrhage patients with anticoagulation medications, Degos et al[32] suggested 5-10 mg of vitamin K if international normalized ratio > 1.5 preoperatively.
Fresh frozen plasma transfusion could correct almost all coagulation factors. Youssef et al[33] suggested higher units (> 6 units) of flash frozen plasma is considered to reverse the prolonged prothrombin time. A concentrated blood product, prothrombin complex concentrate[34], is shown in a clinical trial (phase IIIb study) for its superiority and safety in patients with coagulopathy[34,35].
For thrombocytopenia, evidence has shown that platelet counts should be > 50000/μL before implementation of an invasive procedure[36]. However, for patients taking antiplatelet medication who have normal platelet counts, the evidence for platelet transfusion is not as clear. Evidence from a Stage IIb indicates that transfusion with concentrated platelets would be beneficial for patients taking antiplatelet medication[32].
In addition, serum albumin level is possibly related to surgical outcome in patients with liver cirrhosis. In a retrospective study, corrections of pre- and postoperative serum albumin levels in cirrhotic patients who underwent microsurgery for free-flap reconstruction were shown to decrease the intensive care unit stay and complications[37].
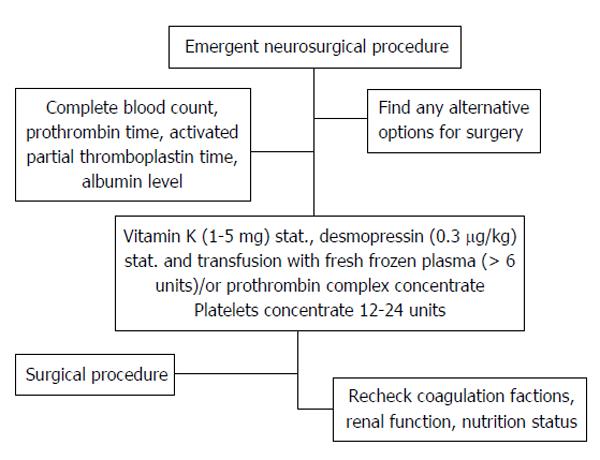
Because there was no consensus regarding a standard procedure for emergent neurosurgical interventions, we proposed an algorithm (Figure 2). Emergent, mostly life-threatening, procedures mandate that surgeons make fast and effective decisions. Prospective studies are urgently needed to demonstrate the most essential components to prevent intraoperative bleeding, wound infection, and other associated complications, such as pneumonia, consciousness disturbance, or hepatorenal syndrome.
Liver cirrhosis is a diffuse process characterized by fibrosis and conversion of normal liver architecture into structurally abnormal nodules[38]. It was a major chronic devastating disease with high mortality rate (up to 72.7 deaths/10000 population) distributed most frequently in central Asia and the sub-Saharan region[39]. Studies have shown significantly high morbidity/mortality rates in cirrhotic patients undergoing major surgery. Neurosurgical procedures, which have not been researched extensively, have been performed conservatively on cirrhotic patients. However, the need for neurosurgery often precludes the potential cirrhotic complications.
In this review, we discussed the outcome from different etiologies, including TBI, SICH, brain tumor, and spine procedures (Table 1). Preoperative assessment and correction of coagulopathy and nutritional status may be important, despite lack of evidence. Still, the high morbidity/mortality rate in this devastating underlying disease may demand the need to develop evidence-based studies to elucidate the timing and parameters for appropriate correction leading to better surgical outcome in neurosurgical procedures.
| Neurosurgical type/ref. | Patient (n) | Overall morbidity | Child A | Child B | Child C | Overall mortality | Child A | Child B | Child C |
| TBI | |||||||||
| Lustenberger et al[22] | 47 | NA | NA | NA | NA | 34% | NA | NA | NA |
| Chen et al[21] | 32 | 84.4% | 38.7% | 60.0% | 84.2% | 37.5% | 5.3% | 38.0% | 63.2% |
| SICH | |||||||||
| Chen et al[21] | 44 | 43.2% | NA | NA | NA | 22.7% | NA | NA | NA |
| Huang et al[20] | 36 | NA | NA | NA | NA | 47% | 11.1% | 27.7% | 8.3% |
| Brain tumor | |||||||||
| Chen et al[21] | 15 | 26.7% | NA | NA | NA | NA | NA | NA | NA |
| Spine surgery | |||||||||
| Liao et al[27] | 29 | 41.0% | 27.0% | 86.0% | NA | NA | NA | NA | NA |
We acknowledge Department of Neurosurgery and General Surgery, Chang Gung Memorial Hospital, Linkou, Taiwan.
P- Reviewer: Barone DG, Tai DI S- Editor: Ma YJ L- Editor: A E- Editor: Liu SQ
non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
| 1. | An Y, Xiao YB, Zhong QJ. Open-heart surgery in patients with liver cirrhosis: indications, risk factors, and clinical outcomes. Eur Surg Res. 2007;39:67-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Chen CC, Hsu PW, Erich Wu TW, Lee ST, Chang CN, Wei KC, Chuang CC, Wu CT, Lui TN, Hsu YH. Stereotactic brain biopsy: Single center retrospective analysis of complications. Clin Neurol Neurosurg. 2009;111:835-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 3. | Christmas AB, Wilson AK, Franklin GA, Miller FB, Richardson JD, Rodriguez JL. Cirrhosis and trauma: a deadly duo. Am Surg. 2005;71:996-1000. [PubMed] |
| 4. | Dangleben DA, Jazaeri O, Wasser T, Cipolle M, Pasquale M. Impact of cirrhosis on outcomes in trauma. J Am Coll Surg. 2006;203:908-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 5. | Demetriades D, Constantinou C, Salim A, Velmahos G, Rhee P, Chan L. Liver cirrhosis in patients undergoing laparotomy for trauma: effect on outcomes. J Am Coll Surg. 2004;199:538-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 53] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 6. | Doberneck RC, Sterling WA, Allison DC. Morbidity and mortality after operation in nonbleeding cirrhotic patients. Am J Surg. 1983;146:306-309. [PubMed] |
| 7. | Fang JF, Chen RJ, Lin BC, Hsu YB, Kao JL, Chen MF. Liver cirrhosis: an unfavorable factor for nonoperative management of blunt splenic injury. J Trauma. 2003;54:1131-1136; discussion 1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Garrison RN, Cryer HM, Howard DA, Polk HC. Clarification of risk factors for abdominal operations in patients with hepatic cirrhosis. Ann Surg. 1984;199:648-655. [PubMed] |
| 9. | Georgiou C, Inaba K, Teixeira PG, Hadjizacharia P, Chan LS, Brown C, Salim A, Rhee P, Demetriades D. Cirrhosis and trauma are a lethal combination. World J Surg. 2009;33:1087-1092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Talving P, Lustenberger T, Okoye OT, Lam L, Smith JA, Inaba K, Mohseni S, Chan L, Demetriades D. The impact of liver cirrhosis on outcomes in trauma patients: a prospective study. J Trauma Acute Care Surg. 2013;75:699-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Thielmann M, Mechmet A, Neuhäuser M, Wendt D, Tossios P, Canbay A, Massoudy P, Jakob H. Risk prediction and outcomes in patients with liver cirrhosis undergoing open-heart surgery. Eur J Cardiothorac Surg. 2010;38:592-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 12. | Tinkoff G, Rhodes M, Diamond D, Lucke J. Cirrhosis in the trauma victim. Effect on mortality rates. Ann Surg. 1990;211:172-177. [PubMed] |
| 13. | Csikesz NG, Nguyen LN, Tseng JF, Shah SA. Nationwide volume and mortality after elective surgery in cirrhotic patients. J Am Coll Surg. 2009;208:96-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 124] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 14. | Eker HH, van Ramshorst GH, de Goede B, Tilanus HW, Metselaar HJ, de Man RA, Lange JF, Kazemier G. A prospective study on elective umbilical hernia repair in patients with liver cirrhosis and ascites. Surgery. 2011;150:542-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Gray SH, Vick CC, Graham LA, Finan KR, Neumayer LA, Hawn MT. Umbilical herniorrhapy in cirrhosis: improved outcomes with elective repair. J Gastrointest Surg. 2008;12:675-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Mansour A, Watson W, Shayani V, Pickleman J. Abdominal operations in patients with cirrhosis: still a major surgical challenge. Surgery. 1997;122:730-375; discussion 735-736. [PubMed] |
| 17. | Sirinek KR, Burk RR, Brown M, Levine BA. Improving survival in patients with cirrhosis undergoing major abdominal operations. Arch Surg. 1987;122:271-273. [PubMed] |
| 18. | Marrocco-Trischitta MM, Kahlberg A, Astore D, Tshiombo G, Mascia D, Chiesa R. Outcome in cirrhotic patients after elective surgical repair of infrarenal aortic aneurysm. J Vasc Surg. 2011;53:906-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Grønbaek H, Johnsen SP, Jepsen P, Gislum M, Vilstrup H, Tage-Jensen U, Sørensen HT. Liver cirrhosis, other liver diseases, and risk of hospitalisation for intracerebral haemorrhage: a Danish population-based case-control study. BMC Gastroenterol. 2008;8:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 20. | Huang HH, Lin HH, Shih YL, Chen PJ, Chang WK, Chu HC, Chao YC, Hsieh TY. Spontaneous intracranial hemorrhage in cirrhotic patients. Clin Neurol Neurosurg. 2008;110:253-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Chen CC, Hsu PW, Lee ST, Chang CN, Wei KC, Wu CT, Hsu YH, Lin TK, Lee SC, Huang YC. Brain surgery in patients with liver cirrhosis. J Neurosurg. 2012;117:348-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Lustenberger T, Talving P, Lam L, Inaba K, Branco BC, Plurad D, Demetriades D. Liver cirrhosis and traumatic brain injury: a fatal combination based on National Trauma Databank analysis. Am Surg. 2011;77:311-314. [PubMed] |
| 23. | Boudouresques G, Hauw JJ, Meininger V, Escourolle R, Pertuiset B, Buge A, Lhermitte F, Castaigne P. Hepatic cirrhosis and intracranial hemorrhage: significance of the association in 53 pathological cases. Ann Neurol. 1980;8:204-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 24. | Jiang XB, Ke C, Zhang GH, Zhang XH, Sai K, Chen ZP, Mou YG. Brain metastases from hepatocellular carcinoma: clinical features and prognostic factors. BMC Cancer. 2012;12:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 25. | Bjarke Christensen F, Stender Hansen E, Laursen M, Thomsen K, Bünger CE. Long-term functional outcome of pedicle screw instrumentation as a support for posterolateral spinal fusion: randomized clinical study with a 5-year follow-up. Spine (Phila Pa 1976). 2002;27:1269-1277. [PubMed] |
| 26. | Kim KT, Lee SH, Lee YH, Bae SC, Suk KS. Clinical outcomes of 3 fusion methods through the posterior approach in the lumbar spine. Spine (Phila Pa 1976). 2006;31:1351-137; discussion 1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 142] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 27. | Liao JC, Chen WJ, Chen LH, Niu CC, Fu TS, Lai PL, Tsai TT. Complications associated with instrumented lumbar surgery in patients with liver cirrhosis: a matched cohort analysis. Spine J. 2013;13:908-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 28. | Child CG. Surgery and portal hypertension. 1964;50-64. |
| 29. | Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, ter Borg PC. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 2000;31:864-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1967] [Cited by in RCA: 2069] [Article Influence: 82.8] [Reference Citation Analysis (0)] |
| 30. | Mann KG. Biochemistry and physiology of blood coagulation. Thromb Haemost. 1999;82:165-174. [PubMed] |
| 31. | Dezee KJ, Shimeall WT, Douglas KM, Shumway NM, O’malley PG. Treatment of excessive anticoagulation with phytonadione (vitamin K): a meta-analysis. Arch Intern Med. 2006;166:391-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 32. | Degos V, Westbroek EM, Lawton MT, Hemphill JC, del Zoppo GJ, Young WL. Perioperative management of coagulation in nontraumatic intracerebral hemorrhage. Anesthesiology. 2013;119:218-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 33. | Youssef WI, Salazar F, Dasarathy S, Beddow T, Mullen KD. Role of fresh frozen plasma infusion in correction of coagulopathy of chronic liver disease: a dual phase study. Am J Gastroenterol. 2003;98:1391-1394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 121] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 34. | Sarode R, Milling TJ, Refaai MA, Mangione A, Schneider A, Durn BL, Goldstein JN. Efficacy and safety of a 4-factor prothrombin complex concentrate in patients on vitamin K antagonists presenting with major bleeding: a randomized, plasma-controlled, phase IIIb study. Circulation. 2013;128:1234-1243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 586] [Cited by in RCA: 613] [Article Influence: 51.1] [Reference Citation Analysis (0)] |
| 35. | Dickneite G, Pragst I. Prothrombin complex concentrate vs fresh frozen plasma for reversal of dilutional coagulopathy in a porcine trauma model. Br J Anaesth. 2009;102:345-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 36. | Tosetto A, Balduini CL, Cattaneo M, De Candia E, Mariani G, Molinari AC, Rossi E, Siragusa S; Italian Society for Haemostasis and Thrombosis. Management of bleeding and of invasive procedures in patients with platelet disorders and/or thrombocytopenia: Guidelines of the Italian Society for Haemostasis and Thrombosis (SISET). Thromb Res. 2009;124:e13-e18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 37. | Kao HK, Chen WF, Chen CH, Shyu VB, Cheng MH, Chang KP. The roles of albumin levels in head and neck cancer patients with liver cirrhosis undergoing tumor ablation and microsurgical free tissue transfer. PLoS One. 2012;7:e52678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 38. | Anthony PP, Ishak KG, Nayak NC, Poulsen HE, Scheuer PJ, Sobin LH. The morphology of cirrhosis: definition, nomenclature, and classification. Bull World Health Organ. 1977;55:521-540. [PubMed] |
| 39. | Mokdad AA, Lopez AD, Shahraz S, Lozano R, Mokdad AH, Stanaway J, Murray CJ, Naghavi M. Liver cirrhosis mortality in 187 countries between 1980 and 2010: a systematic analysis. BMC Med. 2014;12:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 720] [Cited by in RCA: 749] [Article Influence: 68.1] [Reference Citation Analysis (0)] |