Published online Feb 27, 2015. doi: 10.4254/wjh.v7.i2.127
Peer-review started: September 2, 2014
First decision: October 14, 2014
Revised: October 28, 2014
Accepted: November 17, 2014
Article in press: November 19, 2014
Published online: February 27, 2015
Processing time: 169 Days and 23.3 Hours
Portal hypertensive enteropathy (PHE) is a condition that describes the pathologic changes and mucosal abnormalities observed in the small intestine of patients with portal hypertension. This entity is being increasingly recognized and better understood over the past decade due to increased accessibility of the small intestine made possible by the introduction of video capsule endoscopy and deep enteroscopy. Though challenged by its diverse endoscopic appearance, multiple scoring systems have been proposed to classify the endoscopic presentation and grade its severity. Endoscopic findings can be broadly categorized into vascular and non-vascular lesions with many subtypes of both categories. Clinical manifestations of PHE can range from asymptomatic incidental findings to fatal gastrointestinal hemorrhage. Classic endoscopic findings in the setting of portal hypertension may lead to a prompt diagnosis. Occasionally histopathology and cross sectional imaging like computed tomography or magnetic resonance imaging may be helpful in establishing a diagnosis. Management of overt bleeding requires multidisciplinary approach involving hepatologists, endoscopists, surgeons, and interventional radiologists. Adequate resuscitation, reduction of portal pressure, and endoscopic therapeutic intervention remain the main principles of the initial treatment. This article reviews the existing evidence on PHE with emphasis on its classification, diagnosis, clinical manifestations, endoscopic appearance, pathological findings, and clinical management. A new schematic management of ectopic variceal bleed is also proposed.
Core tip: Portal hypertensive enteropathy (PHE) is an under recognized complication of portal hypertension. It can present with a broad spectrum of clinical manifestations and endoscopic findings, making its diagnosis challenging. Video capsule endoscopy and deep enteroscopy are diagnostic tools of choice. PHE should be considered in patients with portal hypertension who present with occult or overt gastrointestinal bleeding, especially when portal hypertensive gastropathy and advanced cirrhosis are also present. Adequate resuscitation, reduction of portal pressure, and endoscopic therapeutic intervention remain the mainstay of initial treatment though definitive management may require a multidisciplinary approach involving hepatologists, endoscopists, surgeons, and interventional radiologists.
- Citation: Mekaroonkamol P, Cohen R, Chawla S. Portal hypertensive enteropathy. World J Hepatol 2015; 7(2): 127-138
- URL: https://www.wjgnet.com/1948-5182/full/v7/i2/127.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i2.127
Portal hypertension is defined as increased pressure in portal circulation, as estimated with the measurement of hepatic venous pressure gradient (HVPG)[1] which is the difference between wedge hepatic venous pressure (hepatic sinusoidal pressure) and free hepatic venous pressure. HVPG of more than 6 mmHg is considered abnormal[1,2]. Clinically significant portal hypertension occurs when HVPG is more than 10 mmHg and it is considered severe portal hypertension when HVPG is more than 12 mmHg[3,4]. Cirrhosis with consequent increased hepatic vascular resistance and portal venous inflow remains the most common etiology of portal hypertension, though it can also occasionally be seen due to other pre and post hepatic conditions such as congestive heart failure, Budd-Chiari syndrome, portal vein thrombosis, etc. The major clinical manifestations of portal hypertension were first recognized in the late nineteenth century and portal hypertension was described as a clinical syndrome of ascites, splenomegaly, and esophageal hemorrhage by Gilbert and Carnot in 1902[5]. Rupture and consequent hemorrhage from esophageal varices remains the most lethal complication of portal hypertension and therefore guidelines now call for periodic endoscopy for surveillance of these varices. Upper and lower endoscopy have led to recognition of other mucosal changes of gastrointestinal tract which previously had been less well recognized.
Microcirculatory changes of gastric mucosa, as a result of portal hypertension was first described as congestive gastropathy in 1985 by McCormack et al[6]. The characteristic findings of mosaic-pattern mucosa (irregular, cleft-bordered polygonal reticulated area, or snake skin-like pattern), small flat red-point lesions, elevated large cherry red spots, and black-brown spots have been variously termed as inflammatory gastritis, mucosal vasculopathy, portal hypertensive mucosa, and portal hypertensive gastropathy[6-10]. Subsequent studies suggested that these changes may be found in other areas of gastrointestinal tract as well[11-14]. The term, portal hypertensive colopathy, was used to describe similar abnormalities found in the colon of patients with portal hypertension. Endoscopic findings of portal hypertensive colopathy include diffuse erythematous and edematous mucosa, inflammatory (colitis-like) lesions, angiodysplasia-like lesions, and ectopic or anorectal varices[15-24].
Once the gastric and colonic mucosal changes associated with portal hypertension were recognized, jejunal and ileal mucosal changes were also noted. Occasional reports suggested small bowel bleeding in cirrhotic patients which was presumed to be a consequence of portal hypertension. The introduction of video capsule endoscopy and deep enteroscopy has increased our ability to evaluate the small bowel completely with good quality imaging and has led to better recognition and understanding of these portal hypertensive changes in the small bowel. This article reviews the existing evidence on portal hypertension-related changes in the small intestine, also known as portal hypertensive enteropathy (PHE) with emphasis on its classification, clinical manifestations, endoscopic appearance, and clinical management.
PHE previously termed portal hypertensive intestinal vasculopathy[25], includes pathologic changes and mucosal abnormalities observed in the small intestine of patients with portal hypertension[26]. Like other portal hypertensive related changes, PHE has also been reported in non-cirrhotic etiologies of portal hypertension[27]. The definition and diagnosis of PHE have evolved over the past decade due to increased accessibility of the small intestine. Introduction of video capsule endoscopy (VCE) and deep enteroscopy has shed more light into this area of the gastrointestinal tract and redefined the disease. Multiple scoring systems have been proposed to classify PHE and to grade its severity[28-30].
De Palma et al[29] were among the first groups to study small intestinal changes in cirrhotic patients with portal hypertension using VCE[29]. They found that portal hypertensive gastropathy and portal hypertensive colopathy are significantly associated with PHE and suggested these changes across different parts of the gastrointestinal tract could be the regional manifestations of the same process in portal hypertension, rather than distinct entities. Other parameters that were associated with PHE in their study were grade 2+ or larger esophageal varices and Child-Pugh class C cirrhosis. They classified endoscopic findings of PHE into two categories: mucosal inflammatory-like abnormalities (edema, erythema, granularity, friability) and vascular lesions (cherry red spots, telangiectasias, or angiodysplasia-like lesions, varices)[29].
Abdelaal et al[28] classified PHE lesions into 4 subtypes: inflammatory-like lesions, red spots, angioectasia, and small bowel varices. They created a scoring system based on VCE findings, giving each type one point with an additional point for multiple (more than two) lesions. Using this PHE score, they confirmed the findings by De Palma et al[29] by redemonstrating the association between PHE and portal hypertensive gastropathy, large esophageal varices, Child-Pugh class C cirrhosis, and a history of prior endoscopic variceal injection sclerotherapy or banding ligation in a non-randomized, case-controlled, prospective study[28].
Kodama et al[30] proposed a PHE scoring system based on double balloon enteroscopy findings. They classified PHE lesions into 2 categories: villous abnormalities and vascular lesions. They further subclassified each category into 3 subtypes: edema, atrophy and reddening for villous abnormalities, and angiodysplasia-like lesions, dilated/proliferated vessels, and varices for vascular lesions. A single point is given for each type, resulting in a scoring system with a maximum of 6 points. This scoring system, however, was associated with only the presence of ascites, making its clinical significance unclear[30].
In summary, PHE lesions can be described based on VCE findings or optical endoscopic findings. They can be categorized into subtypes of vascular and non-vascular lesions. Despite multiple proposed scoring systems as described in Table 1, presently there is insufficient data to standardize or validate these systems. Besides ectopic varices and bleeding lesions, the clinical significance of other subtypes of mucosal changes remains unclear.
| De Palma et al[29] | Abdelaal et al[28] | Kodama et al[30] | |
| Classification | Inflammatory lesions | Inflammatory-like lesions | Villous abnormalities |
| Vascular lesions | Red spots | Edema | |
| Angioectasia | Atrophy | ||
| Small bowel varices | Erythema | ||
| Vascular lesions | |||
| angiodysplasia-like lesions | |||
| dilated/proliferated vessels | |||
| varices | |||
| Scoring system | None | One point for each type of lesion. An additional point for > 2 lesions | One point for each type of lesion |
| Clinical significance | Associated with PHG, PHC, large esophageal varices and Child-Pugh class C cirrhosis | Associated with PHG, large esophageal varices, Child-Pugh class C cirrhosis, and a history of EVL | Associated with ascites |
There is great heterogeneity among reported prevalence of PHE ranging from 15% up to 82% in cirrhotic patients[5,27,29,31]. We believe that this heterogeneity is a result of a wide spectrum of clinical severity of portal hypertension in cirrhotic patients, together with increase in reported prevalence due to recent advances in small bowel imaging in the past decade. The prevalence of PHE ranges only 15%-25% in studies where traditional endoscopic modalities like duodenoscopy, push enteroscopy or colonoscopy with ileal intubation were used[14,20] but is much higher (40%-82%) in other studies when VCE was used[5,26,29,32-35].
Prevalence of each type of endoscopic finding also varies among studies. Red spots (22.2%-62.2%) and angiodysplasia-like lesions (24.3%-55.7%) seem to be more common than inflammatory-like lesions (5.6%-13%) or varices (8.1%-38.9%). Mixed lesions, especially multiple vascular lesions (varice, angiodysplasia-like, and red spots) can be seen in up to 22.3%[26,29,36]. Small bowel varices account for 12%-35% of all ectopic varices[37,38]. Portal hypertensive polypoid enteropathy is a rarer manifestation of PHE and polypoid lesions are less common in the small intestine (0.3%) compared to polypoid gastropathy (0.6%)[39].
Actively bleeding lesions are not uncommon and can be seen in up to 17.8% of all patients with PHE, suggesting their clinical significance as a possible culprit source of obscure overt or obscure occult gastrointestinal bleeding. The bleeding lesions are commonly from angiodysplasia-like lesions and varices, though occasionally can be associated with polypoid enteropathy as well[26,29].
PHE should be suspected when there is gastrointestinal bleeding or anemia not otherwise explained by more common etiologies, along with signs of portal hypertension such as ascites, splenomegaly, thrombocytopenia, or hepatic venous pressure gradient more than 8 mmHg[39,40]. PHE can present as anemia, melena, hematochezia, hematemesis or may be asymptomatic. Fatal and life threatening ectopic variceal hemorrhage in small intestine has also been reported[26,38-41]. Small intestinal variceal rupture can present with a classic triad of hematochezia (without hematemesis), portal hypertension, and previous intra-abdominal surgery[38,42,43]. The most common indication for diagnostic work up is occult gastrointestinal bleeding.
Certain clinical parameters have been shown to associate with PHE and can serve as clues to search for the disease. These parameters include large esophageal varices, history of endoscopic variceal injection sclerotherapy, history of endoscopic variceal ligation, portal hypertensive colopathy, portal hypertensive gastropathy, low hemoglobin, thrombocytopenia, splenomegaly, multiple signs of portal hypertension seen on CT scan, and Child-Pugh class C[26,28,29,36,44,45]. However, not all of these associations were seen in every study. Only advanced cirrhosis (Child-Pugh class C) and the presence of portal hypertensive gastropathy were consistently associated with PHE in most studies[28,29,36,44]. PHE has also been reported in non-cirrhotic etiologies of portal hypertension like polycystic liver disease, portal vein thrombosis, and Budd-Chiari syndrome[27,46-48]. PHE is usually diagnosed by a VCE or a deep enteroscopy[35].
Endoscopically, PHE can be associated with a wide range of mucosal changes including the mucosal edema, congested rounded blunt villi giving a classic “herring-roe” appearance, loss of vascularization, friability, hyperrhemia, flat red spots, angiodysplasia-like lesions, pigmented black-brown spots, mucosal granularity, ulcers, reticulated mosaic-like pattern mucosa, protruding red bumps, inflammatory polyps, and varices[11,26,39,44,47,49,50]. Recognizing these findings through an optical endoscope or capsule endoscopy can lead to a prompt diagnosis and avoid further unnecessary and potentially harmful intervention. As mentioned above, these endoscopic findings can be classified into vascular and non-vascular lesions. They can be further categorized as inflammatory-like lesions, red spots, angioectasia, and small bowel varices[28]. Special attention should be paid to vascular lesions such as red spots, angioectasia-like lesions, and varices, which are more likely to cause clinically significant bleeding and are amenable to endoscopic intervention[28].
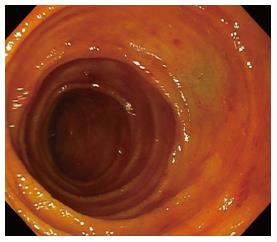
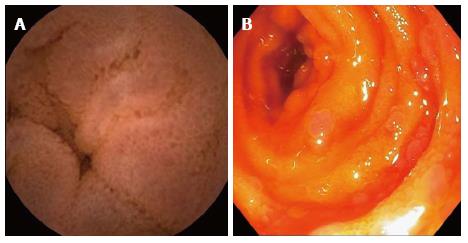
Red spots are small, symmetrical, uniformly erythematous, vascular areas on intestinal mucosa as shown in Figure 1. The lesions are usually flat. They are a very common manifestation of PHE and have been reported in up to 55% of cirrhotic patients with portal hypertension in one series[28].
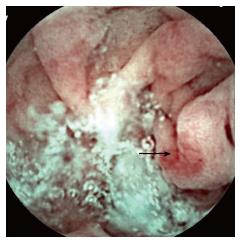
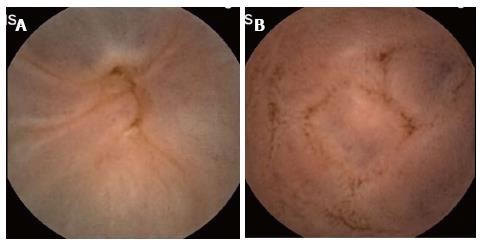
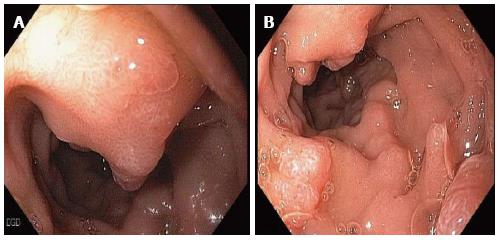
Angioectasia are aberrant submucosal vascular lesions, characterized as small red patches with arborizing ectatic vessels as shown in Figure 2. Variceal lesions in the small bowel are described as tortuously enlarged veins that usually have serpiginous or nodular shape with overlying mosaic-like shining mucosa with bluish discoloration as shown in Figure 3A and B[26,27]. Endoscopic characters of these lesions should be promptly recognized to avoid potentially disastrous diagnostic biopsy attempts.
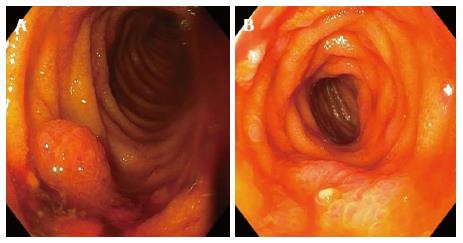
Duodenal varices are most commonly found in the duodenal bulb and the second portion of duodenum[51-53]. Jejunal and ileal varices are more commonly seen in patients with history of abdominal surgery due to post surgical portosystemic collaterals related to bowel anastomosis and are more difficult to diagnose due to their deep location in the intestinal tract[38,42,43]. Small intestinal varices usually originate from portal venous trunk or superior mesenteric vein[48,54,55]. Polypoid enteropathy is a rare manifestation of PHE and can present in any part of the small intestine. Polypoid lesions can have varied endoscopic manifestations. They can be single or multiple, sessile or pedunculated, small or large and may occasionally mimic adenomatous polyps as shown in Figure 4A and B. They usually arise in the background of inflamed mucosa with classic mosaic or herring-roe pattern and have been associated with occult or overt gastrointestinal bleeding[39,49,50,56].
Other inflammatory lesions include mucosal edema, granularity, patchy erythematous mucosa, and herring roe appearance (rounded blunted villi on the background of congested mucosa with granularity) as shown in Figures 5 and 6. Clinical significance of these lesions is unclear but it is believed that they are less likely to cause overt gastrointestinal bleeding[26,28,44].
Some of these endoscopic findings are non-specific and can pose a diagnostic dilemma especially in a patient whose portal hypertensive status is unknown. Differential diagnosis includes inflammatory bowel disease, celiac disease, arteriovenous malformations, and familial adenomatous polyposis. Biopsy of non-vascular lesions can be performed with caution to confirm the diagnosis.
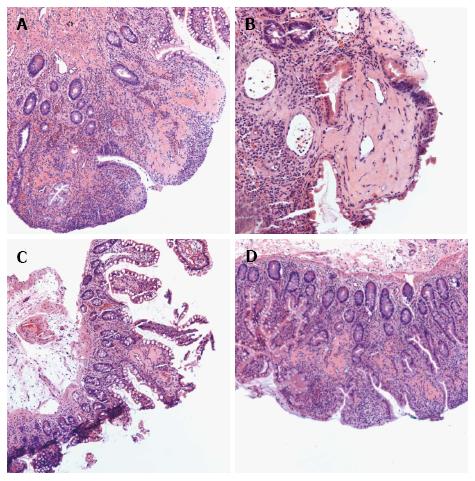
In portal hypertension when splanchnic blood flow cannot effectively return to the systemic circulation, consequently splanchnic vasodilation ensues and mucosa of small intestine becomes congested. Therefore, histopathologic changes of PHE show evidence of congested mucosa and vascular ectasia. These histologic findings include capillary dilation in the lamina propria (mean vascular diameter of 380 micrometers), increased capillary wall thickness, fibromuscular proliferation, a decreased villous/crypt ratio, neovascularization, vascular ectasia, vessels containing fibrin thrombi, inflammatory lymphoplasmatocytic cells infiltration in the lamina propria, reactional nucleocytoplasmic atypia in the epithelial cells, and crenulated aspect of the glands[13,14,39,57-59]. However, these histopathological changes are non-specific and can be seen in patients without portal hypertension and in patients with normal endoscopic findings[12-14]. Therefore, the diagnosis of PHE should not be made on histopathology alone, but rather in conjunction with other clinical and endoscopic characteristics.
Jeon et al[26] has evaluated the use of computerized tomography (CT) scan findings of portal hypertension such as esophageal varices, gastric varices, peri umbilical varices, portal hypertensive colopathy, portal hypertensive gastropathy, portal hypertensive cholecystopathy, splenomegaly, and ascites as the radiologic predictors of PHE. They have created a scoring system giving each CT finding one point with a maximum score of 6 points. A CT score of more than 3 was found to be significantly associated with PHE[26]. Cross sectional imaging by computed tomography angiography and magnetic resonance angiography can also aid in evaluation of vascular origin of the ectopic varices[55,60].
Abdelaal et al[28] have explored the use of a transient elastrography (FibroScan®), a novel non-invasive ultrasound-based technology using pulse-echo ultrasound signals to measure liver stiffness as a surrogate marker of severity of portal hypertension in cirrhotic patients[4,61]. They found that a high transient elastography score had a linear relation with a high PHE score (r2 = 0.314, P-value 0.004), suggesting that higher severity of PHE may be associated with higher degree of liver disease and portal hypertension. Mean liver stiffness measurement in PHE group was 29 kPa, which is much higher than the portal hypertensive cut-off value of 13.6 kPa[4,28]. They concluded that transient elastography may be a new non-invasive method for detecting the presence and severity of PHE in cirrhotic patients[28].
MANAGEMENT
VCE is the preferred initial diagnostic modality in evaluating the small bowel due to its non-invasive nature[5,27,33,35]. It also serves as a road map for subsequent interventions. A deep enteroscopy is warranted when a therapeutically amenable lesion is found on VCE in patients with obscure gastrointestinal bleeding[27]. Different endoscopic findings may require different therapeutic interventions.
Due to its rarity and insufficient evidence, there are no standardized therapeutic guidelines for symptomatic PHE[40]. Clinical significance and the need for any intervention for inflammatory lesions and red spots remain unclear. Argon plasma coagulation is generally used for angioectasia[27] while multiple approaches have been used for small bowel varices and polypoid enteropathy[39,55,56,62,63].
Treatment of portal hypertensive polypoid enteropathy depends on number of polyps and endoscopic accessibility. A polypectomy can be safely performed if the polyp is accessible and amenable for endoscopic removal. Endoclip can be used at the stalk to achieve complete hemostasis[56]. Argon plasma coagulation can be used on the inflamed surface of bleeding polyp to achieve hemostasis but recurrent bleeding has been reported[39]. Non-selective beta blocker, transjugular intrahepatic portosystemic shunt (TIPS), surgical small bowel resection, and liver transplantation have all been reported anecdotally to be successful treatment for portal hypertensive polypoid enteropathy[39,56].
Bleeding small bowel varices occurs in 0.4% of patients with portal hypertension and account for up to 5% of all variceal bleeding. Similar to esophagogastric variceal hemorrhage, it usually presents as massive life-threatening hemorrhage with mortality rate as high as 40%[38,40,51,54]. Available therapeutic options are endoscopic treatment, surgical interventions, and interventional radiological approaches. Similar to polypoidal lesions, management of small bowel varices depends largely on endoscopic accessibility, patient’s surgical risk, available therapy and local expertise.
Adequate resuscitation with intravenous fluids, blood product transfusion, close monitoring, and airway protection remain the main principles of initial management. The benefit of medical management has not been extensively studied in ectopic varices or other manifestations of PHE, but given its established role in esophageal and gastric variceal management[64], it is reasonable to consider the use of vasoactive agents such as octreotide and non-selective beta blockers to reduce the splanchnic and portal pressure in both primary and secondary prophylaxis of ectopic varices[40].
Despite the technical challenges, endoscopic band ligation and endoscopic variceal obturation with tissue glue monomer such as N-butyl-2-cyanoacrylate are endoscopic interventions of choice for hemostasis in ectopic variceal rupture. However, the achieved hemostasis is temporary and re-bleeding is a major concern[52,53]. Endoscopic variceal obturation has shown better success and lower re-bleeding rate compared to endoscopic variceal banding[38,65-68]. Endoscopic sclerotherapy and band ligation are not recommended for large varices, especially the varices with larger diameter than the endoscope itself. This is because incomplete banding can lead to mucosal defect in the remaining varix causing recurrent hemorrhage while excessive dilution of injected sclerosant in large varices decreases the success rate of the sclerotherapy[40,63]. Limited evidence suggests supplemental use of argon plasma coagulation after variceal band ligation as a successful intervention in prevention of rebleeding in esophageal and ileal varices and can be considered in endoscopic management of ectopic varices as well[69-71].
Interventional radiology approaches are effective modalities especially for large varices, lesions that are not endoscopically accessible, and for patients with poor overall general condition who are poor surgical candidates[38,63,72]. These interventions include TIPS, balloon-occluded retrograde transvenous obliteration (B-RTO), and percutaneous coil embolization[38,40,55,62].
Even though TIPS is relatively safe procedure, widely available, and with good success rate, it has limitations in patients with hepatic encephalopathy, high MELD score, severely decompensated cirrhosis, and severe hepatic atrophy[73,74]. It, however, remains an effective intervention to control the variceal bleeding and prevent recurrence, especially in those who have failed endoscopic and medical management. The American Association for the Study of Liver Diseases (AASLD) has recommended TIPS as the preferred approach for the prevention of rebleeding of ectopic varices (including intestinal, stomal, and anorectal varices)[75].
Percutaneous transhepatic and transjugular coil embolization are other radiological interventions that may offer a focal occlusion of the feeding vein of the varices. They can be safely performed even in a large varix, and have good short term results. However, as the portal system is not decompressed, they carry a very high recurrent bleeding rate[76-81]. A retrospective study by Macedo et al[76] showed that even though an immediate bleeding control can be achieved in 75% of patients receiving coil embolization, the rebleeding rate was 67% with a mean bleeding-free interval of 7.8 mo. This result was similar to previous studies that demonstrated a rebleeding rate as high as 55% at 6 mo and 92% at 4 years[76,82]. A combined approach of TIPS and percutaneous embolization in the setting of persistent bleeding after TIPS is preferred[76].
B-RTO is a non-surgical therapy that can occlude not only the varices, but also the feeding afferent and efferent vessel. It is particularly useful in patients who bleed at lower portal pressure, have non-patent portal vein, or are not candidates for TIPS. The success of B-RTO in treating small bowel ectopic varices has been reported, especially in the Japanese literature[83-86]. However, B-RTO can cause significant elevation of porto-systemic pressure gradient and subsequent variceal formation has also been reported[55,87].
Surgical approaches, such as portosystemic surgical shunt, segmental small bowel resection and surgical variceal ligation, can be performed but are usually reserved for patients who are refractory to other therapy[38,41,88,89]. Surgical shunt such as distal splenorenal shunt has equal efficacy as TIPS with no difference in survival or hepatic encephalopathy rate but is less cost-effective approach compared to TIPS[75]. Other surgical approaches such as duodenotomy with simple oversewing of the varix and duodenal dearterialization with stapling have been reported[90,91].
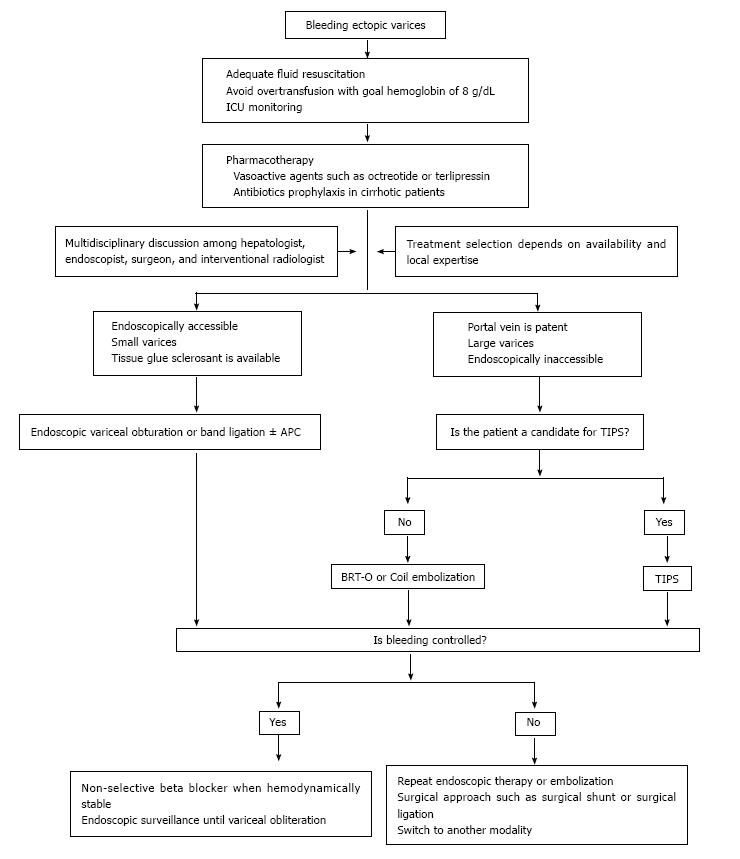
Similar to what Helmy et al[40] have proposed, management of bleeding ectopic varices should focus on adequate initial resuscitation and use a multimodality approach depending on availability and local expertise of each institution. We suggest an algorithm for management of bleeding ectopic varices in Figure 7. As mentioned above, we also recommend individually tailored therapies for non variceal bleeding depending on the clinical situation and local expertise.
PHE-associated small bowel mucosal changes are increasingly being recognized due to introduction of VCE and deep enteroscopy of VCE and deep enteroscopy which enables a more thorough small bowel evaluation than previously possible. Endoscopically, PHE can have myriad presentations ranging from mild mucosal inflammatory changes to angioectasias, inflammatory appearing polyps, and occasionally as large ectopic varices. Due to the non-specific endoscopic and histopathological findings, a high index of suspicion is required to recognize and accurately diagnose this condition. VCE and/or a deep enteroscopy are the current preferred modalities for establishing the diagnosis. PHE has been known to cause significant life-threatening overt gastrointestinal bleeding or be a source of occult gastrointestinal blood loss. However, due to low prevalence and lack of large studies, its prognostic value and clinical significance on morbidity and mortality remain unclear. Management of PHE has not yet been standardized and should be individualized based on acuity and severity of the hemorrhage, endoscopic accessibility of the lesion, surgical risk of the patient, patency of portal vein, available therapy and expertise of each institution.
The authors would like to acknowledge Yue Xue MD, Department of Pathology, Emory School of Medicine for providing the picture of histopathological changes in PHE (Figure 8).
P- Reviewer: Neuberger J S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Burroughs AK, Groszmann R, Bosch J, Grace N, Garcia-Tsao G, Patch D, Garcia-Pagan JC, Dagher L. Assessment of therapeutic benefit of antiviral therapy in chronic hepatitis C: is hepatic venous pressure gradient a better end point? Gut. 2002;50:425-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 79] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 2. | Sanyal AJ, Bosch J, Blei A, Arroyo V. Portal hypertension and its complications. Gastroenterology. 2008;134:1715-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 240] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 3. | Ripoll C, Groszmann R, Garcia-Tsao G, Grace N, Burroughs A, Planas R, Escorsell A, Garcia-Pagan JC, Makuch R, Patch D. Hepatic venous pressure gradient predicts clinical decompensation in patients with compensated cirrhosis. Gastroenterology. 2007;133:481-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 747] [Cited by in RCA: 811] [Article Influence: 45.1] [Reference Citation Analysis (1)] |
| 4. | Vizzutti F, Arena U, Romanelli RG, Rega L, Foschi M, Colagrande S, Petrarca A, Moscarella S, Belli G, Zignego AL. Liver stiffness measurement predicts severe portal hypertension in patients with HCV-related cirrhosis. Hepatology. 2007;45:1290-1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 527] [Cited by in RCA: 527] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 5. | Goulas S, Triantafyllidou K, Karagiannis S, Nicolaou P, Galanis P, Vafiadis I, Tzivras M, Mavrogiannis C. Capsule endoscopy in the investigation of patients with portal hypertension and anemia. Can J Gastroenterol. 2008;22:469-474. [PubMed] |
| 6. | McCormack TT, Sims J, Eyre-Brook I, Kennedy H, Goepel J, Johnson AG, Triger DR. Gastric lesions in portal hypertension: inflammatory gastritis or congestive gastropathy? Gut. 1985;26:1226-1232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 390] [Cited by in RCA: 380] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 7. | Tarnawski AS, Sarfeh IJ, Stachura J, Hajduczek A, Bui HX, Dabros W, Gergely H. Microvascular abnormalities of the portal hypertensive gastric mucosa. Hepatology. 1988;8:1488-1494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 74] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Sarfeh IJ, Tarnawski A. Gastric mucosal vasculopathy in portal hypertension. Gastroenterology. 1987;93:1129-1131. [PubMed] |
| 9. | Carpinelli L, Primignani M, Preatoni P, Angeli P, Battaglia G, Beretta L, Bortoli A, Capria A, Cestari R, Cosentino F. Portal hypertensive gastropathy: reproducibility of a classification, prevalence of elementary lesions, sensitivity and specificity in the diagnosis of cirrhosis of the liver. A NIEC multicentre study. New Italian Endoscopic Club. Ital J Gastroenterol Hepatol. 1997;29:533-540. [PubMed] |
| 10. | Primignani M, Carpinelli L, Preatoni P, Battaglia G, Carta A, Prada A, Cestari R, Angeli P, Gatta A, Rossi A. Natural history of portal hypertensive gastropathy in patients with liver cirrhosis. The New Italian Endoscopic Club for the study and treatment of esophageal varices (NIEC). Gastroenterology. 2000;119:181-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 157] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 11. | Thiruvengadam R, Gostout CJ. Congestive gastroenteropathy--an extension of nonvariceal upper gastrointestinal bleeding in portal hypertension. Gastrointest Endosc. 1989;35:504-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Nagral AS, Joshi AS, Bhatia SJ, Abraham P, Mistry FP, Vora IM. Congestive jejunopathy in portal hypertension. Gut. 1993;34:694-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 13. | Misra V, Misra SP, Dwivedi M, Gupta SC. Histomorphometric study of portal hypertensive enteropathy. Am J Clin Pathol. 1997;108:652-657. [PubMed] |
| 14. | Desai N, Desai D, Pethe V, Deodhar KP, Sawant P, Nanivadekar S. Portal hypertensive jejunopathy: a case control study. Indian J Gastroenterol. 2004;23:99-101. [PubMed] |
| 15. | Kozarek RA, Botoman VA, Bredfeldt JE, Roach JM, Patterson DJ, Ball TJ. Portal colopathy: prospective study of colonoscopy in patients with portal hypertension. Gastroenterology. 1991;101:1192-1197. [PubMed] |
| 16. | Hosking SW, Smart HL, Johnson AG, Triger DR. Anorectal varices, haemorrhoids, and portal hypertension. Lancet. 1989;1:349-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 129] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 17. | Ganguly S, Sarin SK, Bhatia V, Lahoti D. The prevalence and spectrum of colonic lesions in patients with cirrhotic and noncirrhotic portal hypertension. Hepatology. 1995;21:1226-1231. [PubMed] |
| 18. | Misra SP, Dwivedi M, Misra V. Prevalence and factors influencing hemorrhoids, anorectal varices, and colopathy in patients with portal hypertension. Endoscopy. 1996;28:340-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 69] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 19. | Misra SP, Misra V, Dwivedi M. Effect of esophageal variceal sclerotherapy on hemorrhoids, anorectal varices and portal colopathy. Endoscopy. 1999;31:741-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 20. | Misra SP, Dwivedi M, Misra V, Gupta M. Ileal varices and portal hypertensive ileopathy in patients with cirrhosis and portal hypertension. Gastrointest Endosc. 2004;60:778-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 21. | Ito K, Shiraki K, Sakai T, Yoshimura H, Nakano T. Portal hypertensive colopathy in patients with liver cirrhosis. World J Gastroenterol. 2005;11:3127-3130. [PubMed] |
| 22. | Ghoshal UC, Biswas PK, Roy G, Pal BB, Dhar K, Banerjee PK. Colonic mucosal changes in portal hypertension. Trop Gastroenterol. 2001;22:25-27. [PubMed] |
| 23. | Eleftheriadis E, Kotzampassi K, Karkavelas G, Tzioufa V, Papadimitriou K, Aletras H. Portal Hypertensive Colopathy-Endoscopic, Hernodynamic and Morphometric Study. Digestive Endoscopy. 1993;5:224-230. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 24. | Tam TN, NG WW, Lee SD. Colonic mucosal changes in patients with liver cirrhosis. Gastrointest Endosc. 1995;42:408-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Viggiano TR, Gostout CJ. Portal hypertensive intestinal vasculopathy: a review of the clinical, endoscopic, and histopathologic features. Am J Gastroenterol. 1992;87:944-954. [PubMed] |
| 26. | Jeon SR, Kim JO, Kim JB, Ye BD, Chang DK, Shim KN, Cheung DY, Kim JS, Choi MG, Song HJ. Portal hypertensive enteropathy diagnosed by capsule endoscopy in cirrhotic patients: a nationwide multicenter study. Dig Dis Sci. 2014;59:1036-1041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 27. | Figueiredo P, Almeida N, Lérias C, Lopes S, Gouveia H, Leitão MC, Freitas D. Effect of portal hypertension in the small bowel: an endoscopic approach. Dig Dis Sci. 2008;53:2144-2150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 28. | Abdelaal UM, Morita E, Nouda S, Kuramoto T, Miyaji K, Fukui H, Tsuda Y, Fukuda A, Murano M, Tokioka S. Evaluation of portal hypertensive enteropathy by scoring with capsule endoscopy: is transient elastography of clinical impact? J Clin Biochem Nutr. 2010;47:37-44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | De Palma GD, Rega M, Masone S, Persico F, Siciliano S, Patrone F, Matantuono L, Persico G. Mucosal abnormalities of the small bowel in patients with cirrhosis and portal hypertension: a capsule endoscopy study. Gastrointest Endosc. 2005;62:529-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 133] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 30. | Kodama M, Uto H, Numata M, Hori T, Murayama T, Sasaki F, Tsubouchi N, Ido A, Shimoda K, Tsubouchi H. Endoscopic characterization of the small bowel in patients with portal hypertension evaluated by double balloon endoscopy. J Gastroenterol. 2008;43:589-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 31. | Akyuz F, Pinarbasi B, Ermis F, Uyanikoglu A, Demir K, Ozdil S, Besisik F, Kaymakoglu S, Boztas G, Mungan Z. Is portal hypertensive enteropathy an important additional cause of blood loss in portal hypertensive patients? Scand J Gastroenterol. 2010;45:1497-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 32. | Leighton JA, Triester SL, Sharma VK. Capsule endoscopy: a meta-analysis for use with obscure gastrointestinal bleeding and Crohn’s disease. Gastrointest Endosc Clin N Am. 2006;16:229-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 33. | Triester SL, Leighton JA, Leontiadis GI, Fleischer DE, Hara AK, Heigh RI, Shiff AD, Sharma VK. A meta-analysis of the yield of capsule endoscopy compared to other diagnostic modalities in patients with obscure gastrointestinal bleeding. Am J Gastroenterol. 2005;100:2407-2418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 419] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 34. | Tang SJ, Zanati S, Dubcenco E, Cirocco M, Christodoulou D, Kandel G, Haber GB, Kortan P, Marcon NE. Diagnosis of small-bowel varices by capsule endoscopy. Gastrointest Endosc. 2004;60:129-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 35. | Rondonotti E, Villa F, Dell’ Era A, Tontini GE, de Franchis R. Capsule endoscopy in portal hypertension. Clin Liver Dis. 2010;14:209-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 36. | Rondonotti E, Villa F, Signorelli C, de Franchis R. Portal hypertensive enteropathy. Gastrointest Endosc Clin N Am. 2006;16:277-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 37. | Lebrec D, Benhamou JP. Ectopic varices in portal hypertension. Clin Gastroenterol. 1985;14:105-121. [PubMed] |
| 38. | Sato T, Akaike J, Toyota J, Karino Y, Ohmura T. Clinicopathological features and treatment of ectopic varices with portal hypertension. Int J Hepatol. 2011;2011:960720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (35)] |
| 39. | Lemmers A, Evrard S, Demetter P, Verset G, Gossum AV, Adler M, Devière J, Moine OL. Gastrointestinal polypoid lesions: a poorly known endoscopic feature of portal hypertension. United European Gastroenterol J. 2014;2:189-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 40. | Helmy A, Al Kahtani K, Al Fadda M. Updates in the pathogenesis, diagnosis and management of ectopic varices. Hepatol Int. 2008;2:322-334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 41. | Kastanakis M, Anyfantakis D, Katsougris N, Bobolakis E. Massive gastrointestinal bleeding due to isolated jejunal varices in a patient without portal hypertension. Int J Surg Case Rep. 2013;4:439-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 42. | Cappell MS, Price JB. Characterization of the syndrome of small and large intestinal variceal bleeding. Dig Dis Sci. 1987;32:422-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 35] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | Yuki N, Kubo M, Noro Y, Kasahara A, Hayashi N, Fusamoto H, Ito T, Kamada T. Jejunal varices as a cause of massive gastrointestinal bleeding. Am J Gastroenterol. 1992;87:514-517. [PubMed] |
| 44. | Higaki N, Matsui H, Imaoka H, Ikeda Y, Murakami H, Hiasa Y, Matsuura B, Onji M. Characteristic endoscopic features of portal hypertensive enteropathy. J Gastroenterol. 2008;43:327-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 45. | Repici A, Pennazio M, Ottobrelli A, Barbon V, De Angelis C, De Lio A. Endoscopic capsule in cirrhotic patients with portal hypertension: spectrum and prevalence of small bowel lesions. Endoscopy. 2005;37:A72. |
| 46. | Gentilucci UV, Gallo P, Perrone G, Del Vescovo R, Galati G, Spataro S, Mazzarelli C, Pellicelli A, Afeltra A, Picardi A. Non-cirrhotic portal hypertension with large regenerative nodules: a diagnostic challenge. World J Gastroenterol. 2011;17:2580-2584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 47. | Tang SJ, Jensen DM, Gralnek IM, Roth BE. Portal hypertensive enteropathy in a patient with polycystic liver disease: a unique endoscopic finding. Gastrointest Endosc. 2002;56:924-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 48. | Amin R, Alexis R, Korzis J. Fatal ruptured duodenal varix: a case report and review of literature. Am J Gastroenterol. 1985;80:13-18. [PubMed] |
| 49. | Sawada K, Ohtake T, Ueno N, Ishikawa C, Abe M, Miyoshi S, Suzuki Y, Tokusashi Y, Fujiya M, Kohgo Y. Multiple portal hypertensive polyps of the jejunum accompanied by anemia of unknown origin. Gastrointest Endosc. 2011;73:179-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 50. | Oluyemi A, Amole A. Portal hypertensive duodenopathy manifesting as “kissing” duodenal ulcers in a nigerian with alcoholic cirrhosis: a case report and brief review of the literature. Case Rep Med. 2012;2012:618729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 51. | Jonnalagadda SS, Quiason S, Smith OJ. Successful therapy of bleeding duodenal varices by TIPS after failure of sclerotherapy. Am J Gastroenterol. 1998;93:272-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 52. | Shiraishi M, Hiroyasu S, Higa T, Oshiro S, Muto Y. Successful management of ruptured duodenal varices by means of endoscopic variceal ligation: report of a case. Gastrointest Endosc. 1999;49:255-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 53. | Yoshida Y, Imai Y, Nishikawa M, Nakatukasa M, Kurokawa M, Shibata K, Shimomukai H, Shimano T, Tokunaga K, Yonezawa T. Successful endoscopic injection sclerotherapy with N-butyl-2-cyanoacrylate following the recurrence of bleeding soon after endoscopic ligation for ruptured duodenal varices. Am J Gastroenterol. 1997;92:1227-1229. [PubMed] |
| 54. | Khouqeer F, Morrow C, Jordan P. Duodenal varices as a cause of massive upper gastrointestinal bleeding. Surgery. 1987;102:548-552. [PubMed] |
| 55. | Koo SM, Jeong SW, Jang JY, Lee TH, Jeon SR, Kim HG, Kim JO, Kim YJ. Jejunal variceal bleeding successfully treated with percutaneous coil embolization. J Korean Med Sci. 2012;27:321-324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 56. | Zeitoun JD, Chryssostalis A, Terris B, Prat F, Gaudric M, Chaussade S. Portal hypertensive duodenal polyp: a case report. World J Gastroenterol. 2007;13:1451-1452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 24] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 57. | El-Khayat HR, El Khattib A, Nosseir M, Ezz MA, El-Amin H, Fouad YM. Portal hypertensive enteropathy before and after variceal obliteration: an endoscopic, histopathologic and immunohistochemical study. J Gastrointestin Liver Dis. 2010;19:175-179. [PubMed] |
| 58. | Tang S. Portal Hypertensive Gastropathy, Enteropathy, and Colopathy. VJGIEN. 2013;1:269-271. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 59. | Shudo R, Yazaki Y, Sakurai S, Uenishi H, Yamada H, Sugawara K. Duodenal erosions, a common and distinctive feature of portal hypertensive duodenopathy. Am J Gastroenterol. 2002;97:867-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 48] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 60. | Lim LG, Lee YM, Tan L, Chang S, Lim SG. Percutaneous paraumbilical embolization as an unconventional and successful treatment for bleeding jejunal varices. World J Gastroenterol. 2009;15:3823-3826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 28] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 61. | Sandrin L, Fourquet B, Hasquenoph JM, Yon S, Fournier C, Mal F, Christidis C, Ziol M, Poulet B, Kazemi F. Transient elastography: a new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med Biol. 2003;29:1705-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1967] [Cited by in RCA: 1936] [Article Influence: 88.0] [Reference Citation Analysis (0)] |
| 62. | Haskal ZJ, Scott M, Rubin RA, Cope C. Intestinal varices: treatment with the transjugular intrahepatic portosystemic shunt. Radiology. 1994;191:183-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 73] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 63. | Norton ID, Andrews JC, Kamath PS. Management of ectopic varices. Hepatology. 1998;28:1154-1158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 254] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 64. | Garcia-Tsao G, Sanyal AJ, Grace ND, Carey W. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology. 2007;46:922-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1229] [Cited by in RCA: 1210] [Article Influence: 67.2] [Reference Citation Analysis (0)] |
| 65. | Brechmann T, Schmiegel W, Nicolas V, Reiser M. Gastrointestinal bleeding 30 years after a complicated cholecystectomy. World J Gastroenterol. 2010;16:4747-4750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 66. | Albert J. Endoscopic Therapy of Variceal Bleeding from the Small Bowel. Video. VJGIEN. 2013;1:226-227. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 67. | Gertsch P, Blumgart LH. Cure of a bleeding duodenal varix by sclerotherapy. Br J Surg. 1988;75:717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 68. | Barbish AW, Ehrinpreis MN. Successful endoscopic injection sclerotherapy of a bleeding duodenal varix. Am J Gastroenterol. 1993;88:90-92. [PubMed] |
| 69. | Cipolletta L, Bianco MA, Rotondano G, Piscopo R, Prisco A, Garofano ML. Prospective comparison of argon plasma coagulator and heater probe in the endoscopic treatment of major peptic ulcer bleeding. Gastrointest Endosc. 1998;48:191-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 90] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Furukawa K, Aoyagi Y, Harada T, Enomoto H. The usefulness of prevention consolidation therapy of esophageal varices using an argon plasma coagulation technique. Hepatol Res. 2002;23:220-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 71. | Nakamura S, Mitsunaga A, Murata Y, Suzuki S, Hayashi N. Endoscopic induction of mucosal fibrosis by argon plasma coagulation (APC) for esophageal varices: A prospective randomized trial of ligation plus APC vs. ligation alone. Endoscopy. 2001;33:210-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 43] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 72. | Guth E, Katz MD, Hanks SE, Teitelbaum GP, Ralls P, Korula J. Recurrent bleeding from ileal varices treated by transjugular intrahepatic portosystemic shunt: value of Doppler ultrasonography in diagnosis and follow-up. J Ultrasound Med. 1996;15:67-69. [PubMed] |
| 73. | Vidal V, Joly L, Perreault P, Bouchard L, Lafortune M, Pomier-Layrargues G. Usefulness of transjugular intrahepatic portosystemic shunt in the management of bleeding ectopic varices in cirrhotic patients. Cardiovasc Intervent Radiol. 2006;29:216-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 93] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 74. | Vangeli M, Patch D, Terreni N, Tibballs J, Watkinson A, Davies N, Burroughs AK. Bleeding ectopic varices--treatment with transjugular intrahepatic porto-systemic shunt (TIPS) and embolisation. J Hepatol. 2004;41:560-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 150] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 75. | Boyer TD, Haskal ZJ. The Role of Transjugular Intrahepatic Portosystemic Shunt (TIPS) in the Management of Portal Hypertension: update 2009. Hepatology. 2010;51:306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 390] [Cited by in RCA: 405] [Article Influence: 27.0] [Reference Citation Analysis (1)] |
| 76. | Macedo TA, Andrews JC, Kamath PS. Ectopic varices in the gastrointestinal tract: short- and long-term outcomes of percutaneous therapy. Cardiovasc Intervent Radiol. 2005;28:178-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 77. | Ozaki CK, Hansen M, Kadir S. Transhepatic embolization of superior mesenteric varices in portal hypertension. Surgery. 1989;105:446-448. [PubMed] |
| 78. | Samaraweera RN, Feldman L, Widrich WC, Waltman A, Steinberg F, Greenfield A, Srinivasan M, Robbins AH, Johnson WC. Stomal varices: percutaneous transhepatic embolization. Radiology. 1989;170:779-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 42] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 79. | Smith-Laing G, Scott J, Long RG, Dick R, Sherlock S. Role of percutaneous transhepatic obliteration of varices in the management of hemorrhage from gastroesophageal varices. Gastroenterology. 1981;80:1031-1036. [PubMed] |
| 80. | Toumeh KK, Girardot JD, Choo IW, Andrews JC, Cho KJ. Percutaneous transhepatic embolization as treatment for bleeding ileostomy varices. Cardiovasc Intervent Radiol. 1995;18:179-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 81. | Viamonte M, Pereiras R, Russell E, Le Page J, Hutson D. Transhepatic obliteration of gastroesophageal varices: results in acute and nonacute bleeders. AJR Am J Roentgenol. 1977;129:237-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 82. | L’Herminé C, Chastanet P, Delemazure O, Bonnière PL, Durieu JP, Paris JC. Percutaneous transhepatic embolization of gastroesophageal varices: results in 400 patients. AJR Am J Roentgenol. 1989;152:755-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 42] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 83. | Haruta I, Isobe Y, Ueno E, Toda J, Mitsunaga A, Noguchi S, Kimura T, Shimizu K, Yamauchi K, Hayashi N. Balloon-occluded retrograde transvenous obliteration (BRTO), a promising nonsurgical therapy for ectopic varices: a case report of successful treatment of duodenal varices by BRTO. Am J Gastroenterol. 1996;91:2594-2597. [PubMed] |
| 84. | Sonomura T, Horihata K, Yamahara K, Dozaiku T, Toyonaga T, Hiroka T, Sato M. Ruptured duodenal varices successfully treated with balloon-occluded retrograde transvenous obliteration: usefulness of microcatheters. AJR Am J Roentgenol. 2003;181:725-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 38] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 85. | Akazawa Y, Murata I, Yamao T, Yamakawa M, Kawano Y, Nomura N, Isomoto H, Mizuta Y, Murase K, Kohno S. Successful management of bleeding duodenal varices by endoscopic variceal ligation and balloon-occluded retrograde transvenous obliteration. Gastrointest Endosc. 2003;58:794-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 86. | Ohta M, Yasumori K, Saku M, Saitsu H, Muranaka T, Yoshida K. Successful treatment of bleeding duodenal varices by balloon-occluded retrograde transvenous obliteration: a transjugular venous approach. Surgery. 1999;126:581-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 87. | Tanihata H, Minamiguchi H, Sato M, Kawai N, Sonomura T, Takasaka I, Nakai M, Sahara S, Nakata K, Shirai S. Changes in portal systemic pressure gradient after balloon-occluded retrograde transvenous obliteration of gastric varices and aggravation of esophageal varices. Cardiovasc Intervent Radiol. 2009;32:1209-1216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 88. | Yoshida H, Mamada Y, Taniai N, Yoshioka M, Hirakata A, Kawano Y, Mizuguchi Y, Shimizu T, Ueda J, Uchida E. Treatment modalities for bleeding esophagogastric varices. J Nippon Med Sch. 2012;79:19-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 89. | Ueda J, Yoshida H, Mamada Y, Taniai N, Mizuguchi Y, Shimizu T, Matsumoto S, Kakinuma D, Ishikawa Y, Kanda T. Successful emergency enterectomy for bleeding ileal varices in a patient with liver cirrhosis. J Nippon Med Sch. 2006;73:221-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 90. | Hidajat N, Stobbe H, Hosten N, Schroeder RJ, Fauth M, Vogl T, Felix R. Transjugular intrahepatic portosystemic shunt and transjugular embolization of bleeding rectal varices in portal hypertension. AJR Am J Roentgenol. 2002;178:362-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 91. | McAlister VC, Al-Saleh NA. Duodenal dearterialization and stapling for severe hemorrhage from duodenal varices with portal vein thrombosis. Am J Surg. 2005;189:49-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |