Published online Jul 18, 2015. doi: 10.4254/wjh.v7.i14.1856
Peer-review started: October 20, 2014
First decision: December 17, 2014
Revised: January 22, 2015
Accepted: July 7, 2015
Article in press: July 8, 2015
Published online: July 18, 2015
Processing time: 290 Days and 15.9 Hours
Liver transplantation is the optimal treatment for many patients with advanced liver disease, including decompensated cirrhosis, hepatocellular carcinoma and acute liver failure. Organ shortage is the main determinant of death on the waiting list and hence living donor liver transplantation (LDLT) assumes importance. Biliary complications are the most common post operative morbidity after LDLT and occur due to anatomical and technical reasons. They include biliary leaks, strictures and cast formation and occur in the recipient as well as the donor. The types of biliary complications after LDLT along with their etiology, presenting features, diagnosis and endoscopic and surgical management are discussed.
Core tip: Living donor liver transplantation (LDLT) is associated with increased risk of post transplant biliary complications in recipients and donors, namely bile leaks and biliary strictures. Large bile leaks present early after LDLT and are treated with endoscopic stenting. Ischemic injury to cholangiocytes is the main cause of stricture formation. These may present early or late and are managed with endoscopic dilation followed by stent placement. Occasionally, surgical repair may be required. Cast formation may complicate biliary strictures, requiring endocopic extraction and frequent replacement of stents with cleaning of biliary sludge and debris.
- Citation: Simoes P, Kesar V, Ahmad J. Spectrum of biliary complications following live donor liver transplantation. World J Hepatol 2015; 7(14): 1856-1865
- URL: https://www.wjgnet.com/1948-5182/full/v7/i14/1856.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i14.1856
Liver transplantation (LT) is the optimal treatment for many patients with advanced liver disease, including decompensated cirrhosis, hepatocellular carcinoma and acute liver failure. The vast majority of LT involves the use of organs from deceased donors but despite strategies to increase the supply of deceased donors, organ shortage continues to be the main determinant of death on the waiting list[1]. Due to the lack of organs and also cultural and societal beliefs against the use of deceased donors, living donor LT (LDLT) with split liver grafts was developed in the late 1980s[2,3]. LDLT has potential benefits over deceased donor LT (DDLT) including lower overall costs with elective transplantation, better graft viability and reduced cold ischemia time, and theoretical immunological advantages suggested by the lower incidence of steroid resistant rejection[4-6]. Recipient survival is higher in LDLT but this has to be tempered against the risk of donor complications. Recipient morbidity in LDLT is primarily related to the risk of biliary complications which are twice as common as seen with DDLT[7]. Hospitalization rates and duration of hospital stay post LDLT are also significantly higher than after DDLT even in experienced centers and this is primarily attributed to the higher incidence of biliary complications[8].
The incidence of biliary complications after orthotopic liver transplantation varies between 11%-35%[7,9], with a decreasing trend in recent years. These include strictures, leaks, casts, sludge, stones and Sphincter of Oddi dysfunction of which strictures, bile leaks and cast formation are the commonest, affecting patient and graft survival as well as re transplantation rates. Biliary complications occur because of several anatomical and technical reasons and the management depends on a multi-disciplinary approach involving surgery, hepatology and radiology.
It is important to have an understanding of hepatic vascular anatomy as it explains the high incidence of biliary complications after LT. The liver parenchyma has a dual blood supply via the hepatic artery and portal vein, but the biliary system is only supplied arterially. The biliary epithelium is more liable to ischemic injury than hepatocytes. While bile ducts are relatively more tolerant than hepatocytes to anoxic injury, they are more susceptible to reoxygenation/reperfusion injury[10]. This in part explains the biliary complication rate as does the higher incidence of ischemic cholangiopathy in donation after cardiac death (DCD) organs compared to donation after brain death organs[11,12].
The biliary tree is divided into 3 segments: the hilar segment consisting of the right and left hepatic ducts, the supra-duodenal segment consisting of the common hepatic duct (CHD) and the upper common bile duct (CBD) and the retro-pancreatic segment consisting of the lower CBD. The supra-duodenal duct receives its blood supply in the form of a plexus of many small arteries, mainly the 3 o’clock artery and the 9 o’clock artery running along the lateral borders of the duct arising from the retro-portal, retro-duodenal artery, gastro-duodenal artery, right branch of the hepatic artery, and/or cystic artery. Around 60% of the arterial supply runs superiorly, mainly from the gastro-duodenal artery, around 40% runs inferiorly from the common hepatic artery with a tiny fraction coming off the main trunk of the middle hepatic artery[13,14] (Figure 1).
The hilar and intrahepatic ducts are supplied by the peri-biliary vascular plexus, a network of capillaries arising from the terminal arterial branches of the right and left hepatic artery which also connects with the peri-ductal plexus supplying the supra-duodenal bile duct. A communicating arcade of blood vessels connecting the right and left arterial system of the liver is located within the hilar plate originating from the segment 4 artery and the right branch of the middle hepatic artery. This communicating arcade is spared during LDLT to provide adequate blood supply to the donor duct[15].
Most of the arterial supply of the middle portion of the CBD comes from the retro-duodenal and retro-portal arteries below, and less comes from the right hepatic artery above. During surgery, when these are dissected the middle part of the CBD is prone to ischemic injury. The nature of the arterial supply is the basis for why ischemia chiefly affects the middle third of the CBD, followed by the hepatic duct confluence, with intrahepatic involvement being the least common. Segment 4 and the central portion of the left hepatic duct are often supplied by the right arterial system which is generally transected while performing a right hepatectomy compromising the blood supply to the donor biliary system contributing to donor morbidity[11,13,14].
Generally the stump of the donor bile duct is divided away from the confluence of bile ducts to avoid a stricture of the bile duct remaining in the donor liver resulting in a higher incidence of multiple ducts in the right liver graft[14,16]. Two or more ductal anastomoses has been shown to be a risk factor for developing biliary complications[17,18]. However, studies published[19] since 2008 have shown a considerable drop in overall incidence of biliary complications in recipients owing to more experience and better technique of the biliary anastomosis.
Biliary complications occurring after LDLT are classified according to the Clavien system described below[20,21] (Table 1).
| Grade 1 | Any alteration from the ideal postoperative course, with complete recovery or which can be easily controlled and which fulfills the following general characteristics: |
| (1) Not life threatening | |
| (2) Not requiring use of drugs other than immunosuppressants, analgesics, antipyretics, anti-inflammatory agents, antiemetic, drugs required for urinary retention or lower urinary tract infection, arterial hypertension, hyperlipidemia or transient hyperglycemia | |
| (3) Requiring only therapeutic procedures that can be performed at the bedside | |
| (4) Postoperative bleeding requiring ≤ 3 units of blood transfusion | |
| (5) Never associated with a prolongation of ICU stay or total hospital stay to more than twice the median stay for the procedure in the population of the study | |
| Grade 2 | Any complication that is potentially life threatening or results in ICU stay > 5 d, hospital stay > 4 wk for the recipient, but which does not result in residual disability or persistent disease |
| Grade 3 | Any complication with residual or lasting functional disability or development of malignant disease |
| Grade 4 | Complications that lead to re transplantation (grade 4a) or death (grade 4b) |
The incidence of biliary complications after LDLT is very variable but can be divided into two main categories: bile leaks and strictures of the biliary tree (Table 2). The type of graft used in LDLT affects the complication rate, depending on whether the right or the left lobe is used. To try and ensure adequate graft function and prevent small for size syndrome, the graft size required is dependent on the weight of the recipient (typically at least 0.8%-1% of the recipient weight). Hence, in adult to adult liver transplantation the larger right lobe is almost always used. This typically increases the complication rate but the management strategies remain similar.
| Ref. | Year | Country | Grafts (n) | Biliary complications (%) | |||
| Right | Left | Leaks | Strictures | Overall rate | |||
| Ghobrial et al[22] | 2001 | United States | 20 | 25 | - | - | |
| Gondolesi et al[23] | 2004 | United States | 96 | 0 | 21.9 | 22.9 | 40.6 |
| Liu et al[24] | 2004 | China | 41 | 0 | 7.3 | 24.3 | 24.3 |
| Giacomoni et al[25] | 2006 | Italy | 23 | 0 | 21.7 | 21.7 | 34.8 |
| Soejima et al[26] | 2006 | Japan | 50 | 132 | 11.5 | 25.3 | 36.8 |
| Shah et al[27] | 2007 | Canada | 128 | 0 | 14.8 | 17.1 | 26.0 |
| Mita et al[28] | 2008 | Japan | 5 | 226 | - | 9.5 | - |
| Freise et al[7] | 2008 | United States (A2ALL) | 384 | 0 | 27.2 | 18 | 35.5 |
| Marubashi et al[29] | 2009 | Japan | 57 | 26 | 1.2 | 7.2 | 8.4 |
| Lin et al[30] | 2009 | China | - | - | - | - | 8.9 |
| Wadhawan et al[31] | 2010 | India | 338 | 0 | 8.8 | 10.3 | 19 |
| Kim et al[32] | 2010 | South Korea | 22 | 0 | 0 | 9.1 | 9.1 |
| Soin et al[14] | 2010 | India | 218 | 26 | 2 | 3.7 | 5 |
Bile leaks are a common biliary complication after LDLT compared to DDLT. In the United States, the multicenter A2ALL study reported two thirds of biliary complications after LDLT were due to bile leaks compared to less than a third after DDLT. Studies have reported a 6%-27% overall incidence of bile leaks after LDLT[31,33-35]. Most of these bile leaks were Clavien grade 2 or 3 complications resulting in prolonged hospital stay or permanent disability while a few resulted in graft failure, re transplantation and occasionally death of the recipient, though grade 4 complications in LDLT were less common than in DDLT. Anastomoses involving three or more donor bile ducts were associated with an increased risk while hepatitis C virus cirrhosis as the indication for LT and greater surgical expertise were associated with a lower risk for developing bile leaks[7].
There are two main types of bile leak after LDLT-anastomotic leaks, and cut surface leaks[36]. Anastomotic leaks are the more common type and occur more frequently with Roux-en-Y anastomoses than with duct to duct anastomoses[23]. Cut surface bile leaks usually originate from small bile ducts in the caudate lobe that are transected during surgery and are usually detected incidentally at reoperation[37].
Bile leaks have been shown to decrease overall survival and graft survival post transplant[23,38] and are also a significant risk for subsequent development of a stricture[23,27,31,38].
Bile leaks usually present within 2 mo after LDLT, with most presenting 2-14 d post transplant[7,23,39]. They may present as bilious ascites, biloma or persistent post-operative bile drainage or can be incidentally discovered during computed tomography or endoscopic retrograde cholangiopancreatography (ERCP) performed for other reasons[27].
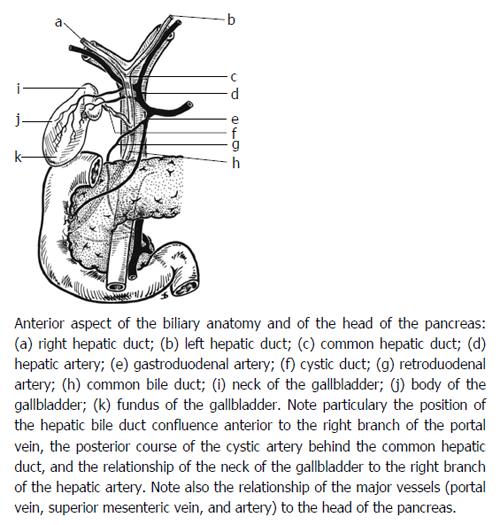
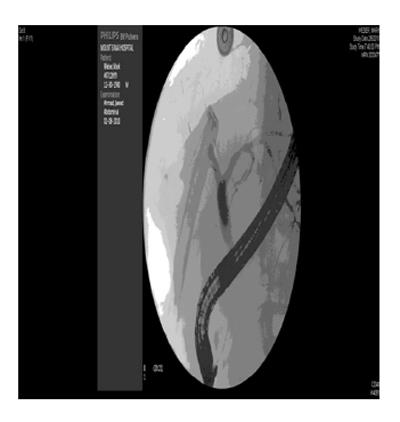
Treatment is often not required for small leaks as they usually heal spontaneously. Larger leaks can be managed with endoscopic treatment by transpapillary stenting (with or without sphincterotomy) which typically seals the leak. Endoscopic methods have shown excellent success in the management of bile leaks, with a reported resolution rate of 82%-92%[31,34]. Percutaneous drainage and stenting by endoscopic retrograde cholangiography (ERC) may be performed simultaneously if there is a significant collection. If copious biliary drainage persists or if there is simultaneous stricture development, surgical reconstruction or conversion of the anastomoses may be performed[27,34]. Most bile leaks in recipients of LDLT resolve within 3 mo after presentation, with a median time to resolution of 1 mo[38]. Figure 2 demonstrates a leak at the anastomosis in a right lobe recipient which was successfully treated with stent placement.
Biliary strictures are also common after LDLT. The A2ALL study reported the incidence of biliary strictures to be 18%-21%[7] with other studies reporting an incidence of 13%[31]. Most biliary strictures described in these studies were Clavien grade 2 or 3 complications. Biliary strictures are of 2 types - anastomotic strictures (AS) and non-AS (NAS).
Anastomotic strictures: Anastomotic strictures occur at the site of duct to duct anastomosis and are typically isolated and shorter in length.
The development of AS is associated with multiple operative factors such as biliary ischemia, cold ischemia time, type of anastomosis (duct to duct vs hepatico-jejunostomy), single vs double duct anastomosis, surgical expertise, prior bile leak and donor factors such as age, gender, weight, blood type and liver steatosis. In DDLT, transplantation in the post MELD era and the use of DCD organs also appears to influence AS formation[18,31,40-44].
The incidence of AS is reported to be around 8%-31% after LDLT[23,24,34], with a cumulative incidence of 6.6%, 10.6% and 12.3% after 1, 5 and 10 years respectively after DDLT[41].
Anastomotic strictures may present either early or late post-transplant. The median time to presentation reported varies between 2.5-9 mo post-transplant[23], with most presenting within 6 mo[18].
The most common presentation is an asymptomatic patient with elevated cholestatic liver enzymes. Abdominal pain, jaundice, fever, increased liver enzymes and recurrent cholangitis may also be presenting features and if present, warrant further investigation for an AS.
If an AS is suspected, liver ultrasound with Doppler imaging or computed tomography angiography to rule out hepatic artery thrombosis should be performed. Ultrasound alone has poor sensitivity for detecting a stricture and is generally followed by magnetic resonance cholangiopancreatography which is a non-invasive diagnostic test with 94.9% sensitivity and 88.9% specificity. The gold standard for diagnosing biliary strictures remains ERCP.
Serial endoscopic balloon dilatation with stenting is the main treatment for an AS. Balloon dilatation followed by plastic stent placement has shown better results than stenting alone. In general, stents are changed every few months, and if the stricture is adequately treated, they are removed between 3-12 mo[45]. Verdonk et al[41] showed that 75% of AS could be successfully stented by ERCP, with a median of 3 ERCP sessions for diagnosis and successful treatment of the stricture. They also showed a higher number of ERCP sessions and greater number of stents were required to treat strictures presenting after 6 mo compared with those presenting earlier. The success of endoscopic treatment varies between 53%-88% depending on center experience[23,31,34,46] and is preferred as the initial method of treatment. In cases where endoscopic management has failed, percutaneous trans hepatic biliary dilatation and stenting of strictures may be attempted, however the success rate of this method is lower and has a higher complication rate[23,41].
Surgical management may be attempted if both endoscopic and percutaneous treatment fails, especially if there are concomitant bile leaks. This may involve surgical repair or revision of the anastomosis from a duct to duct anastomosis to hepaticojejunostomy[47].
The most common complications of endoscopic, percutaneous and surgical methods of treating biliary strictures are recurrent cholangitis, post procedural bleeding, post ERCP pancreatitis, peritonitis and rarely death[45,48].
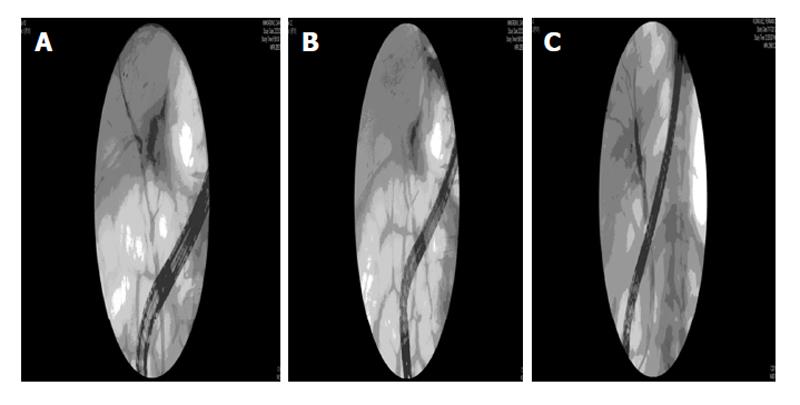
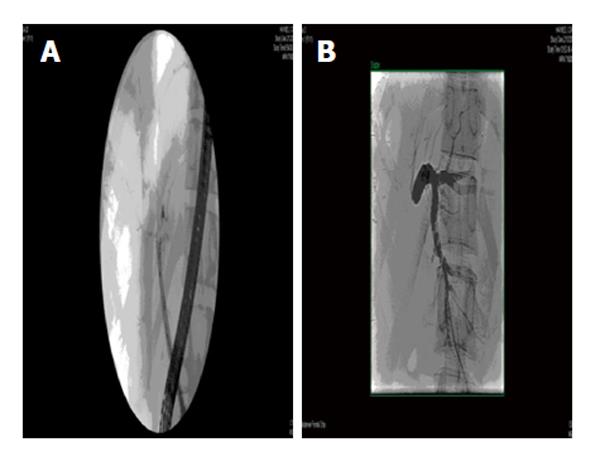
Figure 3 illustrates a typical anastomotic stricture which was treated with multiple dilations and stent placement.
Non anastomotic strictures: Non anastomotic strictures usually occur in the hilar region but may occur diffusely in the recipient biliary tract. They are thought to be related to ischemic and /or immune injury to the biliary mucosa during LT.
A number of operative factors such as total ischemia time, hepatic artery thrombosis, total operative time, type of bile duct anastomosis, and recipient factors such as pre transplant liver disease especially primary sclerosing cholangitis (PSC), bile salt composition and chronic ductopenic rejection as well as donor factors like ABO incompatibility, cytomegalovirus (CMV) infection, donor and recipient gender matching and miscellaneous factors like preservation techniques have all been variably associated with development of NAS[49-51].
Studies by Moench et al[52] and Buis et al[53] attempted to classify NAS into those caused by macro-angiopathy (hepatic artery thrombosis), micro-angiopathy (prolonged ischemia times and preservation injury) and immunological causes (ABO incompatibility, CMV infection, autoimmune hepatitis, or PSC and rejection).
In DDLT early NAS are found more often at the bifurcation of the CHD common hepatic duct or around the CBD common bile duct while late NAS are more often peripherally located within the liver[54].
Hepatic artery thrombosis and prolonged ischemia times both result in ischemic injury to the biliary endothelium, which heals by fibrosis and stricture formation. Previous studies have shown that the biliary epithelium is exquisitely sensitive to ischemia[10]. During LT, the blood supply to the bile ducts via the pancreatic head and gastro duodenal artery is interrupted, making the bile ducts solely dependent on the hepatic artery for perfusion, and thus more susceptible to ischemic injury. In LDLT cold ischemia time is short so interruption to hepatic artery flow is the main concern. Immunologically mediated injury of the biliary epithelium may be from direct cytokine mediated activation of inflammatory cells and thus more often affects the peripheral bile ducts.
The reported incidence of NAS varies between 9%-32%[49,55]. Guichelaar et al[49] found the mean duration to presentation varies between 23.6 ± 34.2 wk to after LDLT. Other studies have described the presentation being between 3.3-5.9 mo, with a median of 4.1 mo[55,56].
Non-anastomotic strictures present in the same way as AS with elevated cholestatic liver enzymes, abdominal pain, pruritus or cholangitis. Biliary ductal dilatation may also be seen incidentally on imaging. Studies have suggested a variation in the time to presentation with NAS secondary to ischemic causes presenting before 1 year and NAS secondary to immunological causes presenting after 1 year[55].
Initial evaluation may include liver United States with Doppler examination of the vasculature. However, this method has only 33%-66% sensitivity and may not be suitable for detecting biliary complications in liver transplant recipients. Magnetic resonance cholangiogram has good sensitivity and specificity and is the best initial non-invasive diagnostic test. Endoscopic retrograde cholangiogram allows simultaneous diagnosis and intervention and is less invasive than percutaneous cholangiography[57]. Rarely, a liver biopsy may be required to differentiate rejection or CMV infection in a patient with elevated cholestatic enzymes and concern for biliary obstruction due to NAS. The ERCP findings in NAS include pre-stenotic dilatation and mucosal narrowing. Non-anastomotic strictures tend to be multiple and longer than AS and less amenable to endoscopic treatment.
Endoscopic management for NAS includes balloon dilatation of all accessible strictures with plastic stent placement and replacement every few months and cleaning out of biliary sludge and casts that may be present in the damaged duct. The success of endoscopic management of NAS in LDLT has been disappointing, with a success rate of 25%-30%[34,57-59] which is below that for NAS seen with DDLT or for AS in LDLT. Percutaneous interventions have a success rate of 40%-85%, but are more invasive and associated with hemorrhagic complications and bile leaks. Non-anastomotic strictures require regular surveillance. Long term outcomes of NAS include recurrent cholangitis, development of biliary cirrhosis and decreased graft survival. Endoscopic and percutaneous methods are often only a temporary solution and re-transplantation has to be considered[27,49,56].
Biliary complications after LDLT are less likely to respond to endoscopic therapy than in DDLT, so preventive strategies to avoid these are important[60]. In right lobe LDLT, high hilar dissection to create a short donor stump and a long recipient stump and ductoplasty to ensure adequate vascularization of the duct ends and intraoperative cholangiogram to early identify biliary leaks are being examined as strategies to reduce the incidence of both AS and NAS[27,47,61]. However, leaving too long a common hepatic duct remnant in the recipient also poses a risk, as some part of this may develop ischemia and later develop a stricture[62].
Other techniques like side to side duct anastomosis and use of interrupted vs uninterrupted biliary sutures have shown minimal benefit[63,64]. Generally, good perfusion of the biliary end and avoidance of vascular injury is the best way to prevent biliary complications[57,59]
Another potential complication of NAS is the formation of casts that deposit in the biliary tree, typically in the setting of ischemic injury. There are 2 main types, composed of either collagen from sloughed off necrotic biliary epithelium or precipitated bile with high bilirubin content. The second type are more frequently seen with biliary strictures[31,65,66] and lead to obstruction and an increased incidence of cholangitis.
Biliary casts develop in 4%-18% of LDLT recipients and are associated with an increased morbidity in the recipient[67-69]. Ischemic events, hepatic artery thrombosis and the presence of biliary strictures are all independently associated with the development of biliary casts[62]. Recurrent cholangitis, prolonged cold ischemia time and acute cellular rejection have also been hypothesized as risk factors.
Biliary cast syndrome presents within a year of transplant, usually within 16 wk, though some delayed cases have been described[67,70]. Elevated cholestatic liver enzymes or incidental bile duct dilatation with echogenic material filling the bile duct may be seen on ultrasound. However, ultrasound has low sensitivity and biliary casts can only reliably be detected by ERC or percutaneous transhepatic cholangiography (PTC), where they may appear as irregular filling defects within the biliary tree[65].
Endoscopic or percutaneous removal of casts with the use of basket or balloon devices, irrigation and hydraulic or mechanical lithotripsy is the typical management strategy. The success of endoscopic and percutaneous methods is reported at 25%-70%[67,70].
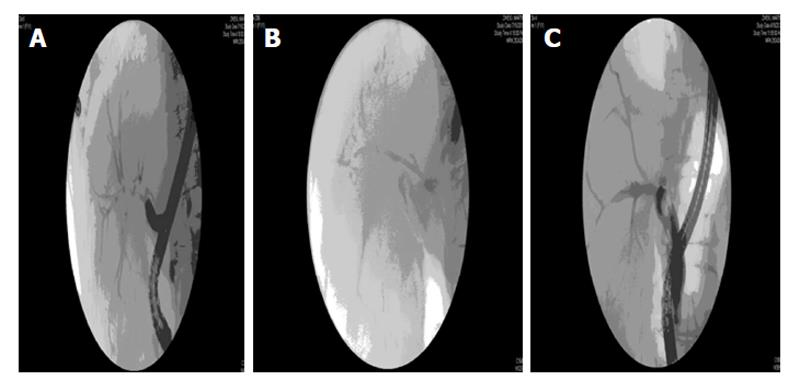
A complex NAS is shown in Figure 4 with biliary cast formation. Several ERCPs and dilation, cast extraction and stenting were required over several years with preservation of graft function.
Adult to adult LDLT also carries some risks to the donor beyond the typical complications associated with abdominal surgery. Various studies (Table 3) have reported a 6%-18% incidence of donor biliary complications[39,71-73]. Most of these complications were classified as Clavien grade 3 or 4.
| Ref. | n | Graft type | Leak (%) | Stricture (%) | Overall rate (%) |
| Iida et al[76] | 500 | Right | 10.6 | 1.6 | 12.2 |
| 762 | Left | 4.7 | 0.3 | 4.9 | |
| El-Meteini et al[77] | 207 | Right | 22 | 1.6 | 13.04 |
| Taketomi et al[78] | 69 | Right | - | - | 10.1 |
| 137 | Left | - | - | 2.9 | |
| Lo et al[79] | 561 | Right | 6.1 | 1.1 | 7.1 |
| 939 | Left | ||||
| Shio et al[75] | 434 | Right | 9.9 | 2.1 | 11.1 |
| 297 | Left | 1.7 | 1 | 2.4 | |
| Ghobrial et al[72] | 393 | Right | 9 | 0.5 | 9.6 |
| Ozgor et al[74] | 500 | Right | - | - | 10.8 |
| Left | |||||
| European Liver | 276 | Right | 5 | 3 | 8 |
| Transplant Registry[80] | Left |
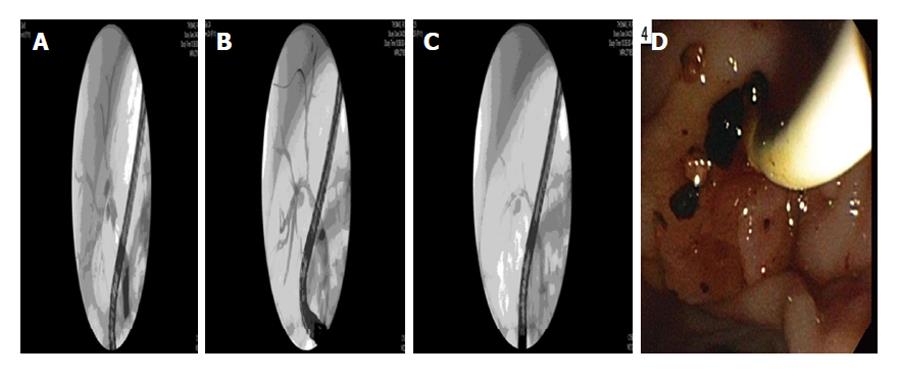
In contrast to the recipient, bile leaks and biliary fistulas are more common in the donor than strictures. The A2ALL study in the United States reported on almost 400 patients who donated the right lobe and found an incidence of 9% of bile leak or biloma, with a 0.5%-1.5% incidence of post-operative biliary strictures since no biliary anastomosis is required in the donor[72,74]. Due to larger graft size, complications are most after right lobe donation and least with left lateral grafts[74-76]. Factors associated with developing bile leaks include elevated pre-operative alkaline phosphatase levels to > 86 IU/L and requiring a blood transfusion during surgery but center experience was not a factor in donor biliary complications[72]. Figure 5 demonstrates a bile leak from the right common hepatic duct stump a few days after right lobe donation.
Donor biliary complications generally present within 2 wk of surgery. Bile leaks can be noted from bilious drain output or present with pain or suspicion for an intra-abdominal collection. Imaging can also be helpful. As in the recipient, strictures present with elevated cholestatic liver enzymes or jaundice.
Management of bile leaks and strictures is similar to the recipient with ERCP and stent placement the mainstay. Almost 80% of leaks were successfully treated by ERCP or percutaneous drainage, though a few required surgical revision or repair[75]. Strictures can be more difficult to manage after right lobe donation as they form as the liver regenerates and wire access to the remaining left lobe biliary tree can be very difficult either endoscopically or percutaneously. Surgical revision is then required. Figure 6 shows a stricture that developed at the takeoff of the left common hepatic duct a few weeks after right lobe donation which could not be treated at ERCP or PTC. The patient was asymptomatic but presented with rising cholestatic enzymes and was successfully treated with biliary bypass surgery.
The development of LDLT with split liver grafts has allowed for elective liver transplants with shortened wait times. It offers several advantages over DDLT but carries an increased risk of biliary complications, mainly bile leaks and strictures. These present within a few weeks to months post transplant. They are usually managed endoscopically, with stenting for bile leaks and dilatation followed by stenting for strictures. Occasionally, endoscopic methods fail and surgical repair or even re transplantation may be required. Strategies to avoid vascular injury and ischemia of the biliary tree are important in preventing these complications.
P- Reviewer: Celikbilek M, Peltec A, Rodriguez-Castro KI S- Editor: Tian YL L- Editor: A E- Editor: Liu SQ
| 1. | Song AT, Avelino-Silva VI, Pecora RA, Pugliese V, D’Albuquerque LA, Abdala E. Liver transplantation: fifty years of experience. World J Gastroenterol. 2014;20:5363-5374. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 68] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 3. | Pichlmayr R, Ringe B, Gubernatis G, Hauss J, Bunzendahl H. [Transplantation of a donor liver to 2 recipients (splitting transplantation)--a new method in the further development of segmental liver transplantation]. Langenbecks Arch Chir. 1988;373:127-130. [PubMed] |
| 4. | Alonso EM, Piper JB, Echols G, Thistlethwaite JR, Whitington PF. Allograft rejection in pediatric recipients of living related liver transplants. Hepatology. 1996;23:40-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 56] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 5. | Doyle MB, Maynard E, Lin Y, Vachharajani N, Shenoy S, Anderson C, Earl M, Lowell JA, Chapman WC. Outcomes with split liver transplantation are equivalent to those with whole organ transplantation. J Am Coll Surg. 2013;217:102-112; discussion 113-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 6. | Maluf DG, Stravitz RT, Cotterell AH, Posner MP, Nakatsuka M, Sterling RK, Luketic VA, Shiffman ML, Ham JM, Marcos A. Adult living donor versus deceased donor liver transplantation: a 6-year single center experience. Am J Transplant. 2005;5:149-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 125] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 7. | Freise CE, Gillespie BW, Koffron AJ, Lok AS, Pruett TL, Emond JC, Fair JH, Fisher RA, Olthoff KM, Trotter JF. Recipient morbidity after living and deceased donor liver transplantation: findings from the A2ALL Retrospective Cohort Study. Am J Transplant. 2008;8:2569-2579. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 236] [Cited by in RCA: 213] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 8. | Merion RM, Shearon TH, Berg CL, Everhart JE, Abecassis MM, Shaked A, Fisher RA, Trotter JF, Brown RS, Terrault NA. Hospitalization rates before and after adult-to-adult living donor or deceased donor liver transplantation. Ann Surg. 2010;251:542-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 9. | Vagefi PA, Parekh J, Ascher NL, Roberts JP, Freise CE. Outcomes with split liver transplantation in 106 recipients: the University of California, San Francisco, experience from 1993 to 2010. Arch Surg. 2011;146:1052-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 10. | Noack K, Bronk SF, Kato A, Gores GJ. The greater vulnerability of bile duct cells to reoxygenation injury than to anoxia. Implications for the pathogenesis of biliary strictures after liver transplantation. Transplantation. 1993;56:495-500. [PubMed] |
| 11. | Deltenre P, Valla DC. Ischemic cholangiopathy. Semin Liver Dis. 2008;28:235-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 78] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 12. | Chan EY, Olson LC, Kisthard JA, Perkins JD, Bakthavatsalam R, Halldorson JB, Reyes JD, Larson AM, Levy AE. Ischemic cholangiopathy following liver transplantation from donation after cardiac death donors. Liver Transpl. 2008;14:604-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 176] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 13. | Castaing D. Surgical anatomy of the biliary tract. HPB (Oxford). 2008;10:72-76. [RCA] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Soin AS, Kumaran V, Rastogi AN, Mohanka R, Mehta N, Saigal S, Saraf N, Mohan N, Nundy S. Evolution of a reliable biliary reconstructive technique in 400 consecutive living donor liver transplants. J Am Coll Surg. 2010;211:24-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Gunji H, Cho A, Tohma T, Okazumi S, Makino H, Shuto K, Mochizuki R, Matsubara K, Hayano K, Mori C. The blood supply of the hilar bile duct and its relationship to the communicating arcade located between the right and left hepatic arteries. Am J Surg. 2006;192:276-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 62] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Soejima Y, Fukuhara T, Morita K, Yoshizumi T, Ikegami T, Yamashita Y, Sugimachi K, Taketomi A, Maehara Y. A simple hilar dissection technique preserving maximum blood supply to the bile duct in living donor liver transplantation. Transplantation. 2008;86:1468-1469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Wang SF, Huang ZY, Chen XP. Biliary complications after living donor liver transplantation. Liver Transpl. 2011;17:1127-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 89] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 18. | Seehofer D, Eurich D, Veltzke-Schlieker W, Neuhaus P. Biliary complications after liver transplantation: old problems and new challenges. Am J Transplant. 2013;13:253-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 215] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 19. | Blumgart LH, Hann LE. Blumgart’s Surgery of the Liver, Pancreas and Biliary Tract. 5th ed. 2012;31-57.e1. [DOI] [Full Text] |
| 20. | Clavien PA, Camargo CA, Croxford R, Langer B, Levy GA, Greig PD. Definition and classification of negative outcomes in solid organ transplantation. Application in liver transplantation. Ann Surg. 1994;220:109-120. [PubMed] |
| 21. | Broelsch CE, Frilling A, Testa G, Malago M. Living donor liver transplantation in adults. Eur J Gastroenterol Hepatol. 2003;15:3-6. [PubMed] |
| 22. | Ghobrial RM, Saab S, Lassman C, Lu DS, Raman S, Limanond P, Kunder G, Marks K, Amersi F, Anselmo D. Donor and recipient outcomes in right lobe adult living donor liver transplantation. Liver Transpl. 2002;8:901-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 112] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 23. | Gondolesi GE, Varotti G, Florman SS, Muñoz L, Fishbein TM, Emre SH, Schwartz ME, Miller C. Biliary complications in 96 consecutive right lobe living donor transplant recipients. Transplantation. 2004;77:1842-1848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 184] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 24. | Liu CL, Lo CM, Chan SC, Fan ST. Safety of duct-to-duct biliary reconstruction in right-lobe live-donor liver transplantation without biliary drainage. Transplantation. 2004;77:726-732. [PubMed] |
| 25. | Giacomoni A, Lauterio A, Slim AO, Vanzulli A, Calcagno A, Mangoni I, Belli LS, De Gasperi A, De Carlis L. Biliary complications after living donor adult liver transplantation. Transpl Int. 2006;19:466-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Soejima Y, Taketomi A, Yoshizumi T, Uchiyama H, Harada N, Ijichi H, Yonemura Y, Ikeda T, Shimada M, Maehara Y. Biliary strictures in living donor liver transplantation: incidence, management, and technical evolution. Liver Transpl. 2006;12:979-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 97] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 27. | Shah SA, Grant DR, McGilvray ID, Greig PD, Selzner M, Lilly LB, Girgrah N, Levy GA, Cattral MS. Biliary strictures in 130 consecutive right lobe living donor liver transplant recipients: results of a Western center. Am J Transplant. 2007;7:161-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 156] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 28. | Mita A, Hashikura Y, Masuda Y, Ohno Y, Urata K, Nakazawa Y, Ikegami T, Terada M, Yamamoto H, Miyagawa S. Nonsurgical policy for treatment of bilioenteric anastomotic stricture after living donor liver transplantation. Transpl Int. 2008;21:320-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Marubashi S, Dono K, Nagano H, Kobayashi S, Takeda Y, Umeshita K, Monden M, Doki Y, Mori M. Biliary reconstruction in living donor liver transplantation: technical invention and risk factor analysis for anastomotic stricture. Transplantation. 2009;88:1123-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 30. | Lin TS, Concejero AM, Chen CL, Chiang YC, Wang CC, Wang SH, Liu YW, Yang CH, Yong CC, Jawan B. Routine microsurgical biliary reconstruction decreases early anastomotic complications in living donor liver transplantation. Liver Transpl. 2009;15:1766-1775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 31. | Wadhawan M, Kumar A, Gupta S, Goyal N, Shandil R, Taneja S, Sibal A. Post-transplant biliary complications: an analysis from a predominantly living donor liver transplant center. J Gastroenterol Hepatol. 2013;28:1056-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | Kim SH, Lee KW, Kim YK, Cho SY, Han SS, Park SJ. Tailored telescopic reconstruction of the bile duct in living donor liver transplantation. Liver Transpl. 2010;16:1069-1074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 33. | Scanga AE, Kowdley KV. Management of biliary complications following orthotopic liver transplantation. Curr Gastroenterol Rep. 2007;9:31-38. [PubMed] |
| 34. | Yazumi S, Yoshimoto T, Hisatsune H, Hasegawa K, Kida M, Tada S, Uenoyama Y, Yamauchi J, Shio S, Kasahara M. Endoscopic treatment of biliary complications after right-lobe living-donor liver transplantation with duct-to-duct biliary anastomosis. J Hepatobiliary Pancreat Surg. 2006;13:502-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 35. | Testa G, Malagó M, Valentín-Gamazo C, Lindell G, Broelsch CE. Biliary anastomosis in living related liver transplantation using the right liver lobe: techniques and complications. Liver Transpl. 2000;6:710-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 140] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 36. | Gunawansa N, McCall JL, Holden A, Plank L, Munn SR. Biliary complications following orthotopic liver transplantation: a 10-year audit. HPB (Oxford). 2011;13:391-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 37. | Jassem W, Heaton ND, Rela M. Reducing bile leak following segmental liver transplantation: understanding biliary anatomy of the caudate lobe. Am J Transplant. 2008;8:271-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 38. | Zimmerman MA, Baker T, Goodrich NP, Freise C, Hong JC, Kumer S, Abt P, Cotterell AH, Samstein B, Everhart JE. Development, management, and resolution of biliary complications after living and deceased donor liver transplantation: a report from the adult-to-adult living donor liver transplantation cohort study consortium. Liver Transpl. 2013;19:259-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 90] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 39. | Azoulay D, Bhangui P, Andreani P, Salloum C, Karam V, Hoti E, Pascal G, Adam R, Samuel D, Ichai P. Short- and long-term donor morbidity in right lobe living donor liver transplantation: 91 consecutive cases in a European Center. Am J Transplant. 2011;11:101-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 40. | Park JB, Kwon CH, Choi GS, Chun JM, Jung GO, Kim SJ, Joh JW, Lee SK. Prolonged cold ischemic time is a risk factor for biliary strictures in duct-to-duct biliary reconstruction in living donor liver transplantation. Transplantation. 2008;86:1536-1542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 67] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 41. | Verdonk RC, Buis CI, Porte RJ, van der Jagt EJ, Limburg AJ, van den Berg AP, Slooff MJ, Peeters PM, de Jong KP, Kleibeuker JH. Anastomotic biliary strictures after liver transplantation: causes and consequences. Liver Transpl. 2006;12:726-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 235] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 42. | Foley DP, Fernandez LA, Leverson G, Anderson M, Mezrich J, Sollinger HW, D’Alessandro A. Biliary complications after liver transplantation from donation after cardiac death donors: an analysis of risk factors and long-term outcomes from a single center. Ann Surg. 2011;253:817-825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 337] [Cited by in RCA: 308] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 43. | Sundaram V, Jones DT, Shah NH, de Vera ME, Fontes P, Marsh JW, Humar A, Ahmad J. Posttransplant biliary complications in the pre- and post-model for end-stage liver disease era. Liver Transpl. 2011;17:428-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 44. | Baccarani U, Isola M, Adani GL, Avellini C, Lorenzin D, Rossetto A, Currò G, Comuzzi C, Toniutto P, Risaliti A. Steatosis of the hepatic graft as a risk factor for post-transplant biliary complications. Clin Transplant. 2010;24:631-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 45. | Schwartz DA, Petersen BT, Poterucha JJ, Gostout CJ. Endoscopic therapy of anastomotic bile duct strictures occurring after liver transplantation. Gastrointest Endosc. 2000;51:169-174. [PubMed] |
| 46. | Ribeiro JB, Martins Fde S, Garcia JH, Cunha AC, Pinto RA, Satacaso MV, Prado-Júnior FP, Pessoa RR. Endoscopic management of biliary complications after liver transplantation. Arq Bras Cir Dig. 2012;25:269-272. [PubMed] |
| 47. | Kasahara M, Egawa H, Takada Y, Oike F, Sakamoto S, Kiuchi T, Yazumi S, Shibata T, Tanaka K. Biliary reconstruction in right lobe living-donor liver transplantation: Comparison of different techniques in 321 recipients. Ann Surg. 2006;243:559-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 186] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 48. | Poley JW, Lekkerkerker MN, Metselaar HJ, Kuipers EJ, Bruno MJ. Clinical outcome of progressive stenting in patients with anastomotic strictures after orthotopic liver transplantation. Endoscopy. 2013;45:567-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 49. | Guichelaar MM, Benson JT, Malinchoc M, Krom RA, Wiesner RH, Charlton MR. Risk factors for and clinical course of non-anastomotic biliary strictures after liver transplantation. Am J Transplant. 2003;3:885-890. [PubMed] |
| 50. | Sanchez-Urdazpal L, Gores GJ, Ward EM, Maus TP, Wahlstrom HE, Moore SB, Wiesner RH, Krom RA. Ischemic-type biliary complications after orthotopic liver transplantation. Hepatology. 1992;16:49-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 291] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 51. | Howell JA, Gow PJ, Angus PW, Jones RM, Wang BZ, Bailey M, Fink MA. Early-onset versus late-onset nonanastomotic biliary strictures post liver transplantation: risk factors reflect different pathogenesis. Transpl Int. 2012;25:765-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 52. | Moench C, Moench K, Lohse AW, Thies J, Otto G. Prevention of ischemic-type biliary lesions by arterial back-table pressure perfusion. Liver Transpl. 2003;9:285-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 104] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 53. | Buis CI, Verdonk RC, Van der Jagt EJ, van der Hilst CS, Slooff MJ, Haagsma EB, Porte RJ. Nonanastomotic biliary strictures after liver transplantation, part 1: Radiological features and risk factors for early vs. late presentation. Liver Transpl. 2007;13:708-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 160] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 54. | Verdonk RC, Buis CI, Porte RJ, Haagsma EB. Biliary complications after liver transplantation: a review. Scand J Gastroenterol Suppl. 2006;89-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 133] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 55. | Verdonk RC, Buis CI, van der Jagt EJ, Gouw AS, Limburg AJ, Slooff MJ, Kleibeuker JH, Porte RJ, Haagsma EB. Nonanastomotic biliary strictures after liver transplantation, part 2: Management, outcome, and risk factors for disease progression. Liver Transpl. 2007;13:725-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 56. | Graziadei IW, Schwaighofer H, Koch R, Nachbaur K, Koenigsrainer A, Margreiter R, Vogel W. Long-term outcome of endoscopic treatment of biliary strictures after liver transplantation. Liver Transpl. 2006;12:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 157] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 57. | Sharma S, Gurakar A, Jabbour N. Biliary strictures following liver transplantation: past, present and preventive strategies. Liver Transpl. 2008;14:759-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 275] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 58. | Yazumi S, Chiba T. Biliary complications after a right-lobe living donor liver transplantation. J Gastroenterol. 2005;40:861-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 59. | Tsujino T, Sugawara Y, Omata M. Management of biliary strictures after living donor liver transplantation. Gastrointest Endosc. 2009;70:599-600; author reply 600-601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 60. | Buxbaum JL, Biggins SW, Bagatelos KC, Ostroff JW. Predictors of endoscopic treatment outcomes in the management of biliary problems after liver transplantation at a high-volume academic center. Gastrointest Endosc. 2011;73:37-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 61. | Ishiko T, Egawa H, Kasahara M, Nakamura T, Oike F, Kaihara S, Kiuchi T, Uemoto S, Inomata Y, Tanaka K. Duct-to-duct biliary reconstruction in living donor liver transplantation utilizing right lobe graft. Ann Surg. 2002;236:235-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 62. | Chok KS, Lo CM. Prevention and management of biliary anastomotic stricture in right-lobe living-donor liver transplantation. J Gastroenterol Hepatol. 2014;29:1756-1763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 63. | Davidson BR, Rai R, Kurzawinski TR, Selves L, Farouk M, Dooley JS, Burroughs AK, Rolles K. Prospective randomized trial of end-to-end versus side-to-side biliary reconstruction after orthotopic liver transplantation. Br J Surg. 1999;86:447-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 64. | Castaldo ET, Pinson CW, Feurer ID, Wright JK, Gorden DL, Kelly BS, Chari RS. Continuous versus interrupted suture for end-to-end biliary anastomosis during liver transplantation gives equal results. Liver Transpl. 2007;13:234-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 65. | Starzl TE, Putnam CW, Hansbrough JF, Porter KA, Reid HA. Biliary complications after liver transplantation: with special reference to the biliary cast syndrome and techniques of secondary duct repair. Surgery. 1977;81:212-221. [PubMed] |
| 66. | Yang YL, Zhang C, Lin MJ, Shi LJ, Zhang HW, Li JY, Yu Q. Biliary casts after liver transplantation: morphology and biochemical analysis. World J Gastroenterol. 2013;19:7772-7777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 67. | Shah JN, Haigh WG, Lee SP, Lucey MR, Brensinger CM, Kochman ML, Long WB, Olthoff K, Shaked A, Ginsberg GG. Biliary casts after orthotopic liver transplantation: clinical factors, treatment, biochemical analysis. Am J Gastroenterol. 2003;98:1861-1867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 68. | O’Connor HJ, Vickers CR, Buckels JA, McMaster P, Neuberger JM, West RJ, Elias E. Role of endoscopic retrograde cholangiopancreatography after orthotopic liver transplantation. Gut. 1991;32:419-423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 69. | Spier BJ, Pfau PR, Lorenze KR, Knechtle SJ, Said A. Risk factors and outcomes in post-liver transplantation bile duct stones and casts: A case-control study. Liver Transpl. 2008;14:1461-1465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 70. | Pfau PR, Kochman ML, Lewis JD, Long WB, Lucey MR, Olthoff K, Shaked A, Ginsberg GG. Endoscopic management of postoperative biliary complications in orthotopic liver transplantation. Gastrointest Endosc. 2000;52:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 178] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 71. | Berg CL, Gillespie BW, Merion RM, Brown RS, Abecassis MM, Trotter JF, Fisher RA, Freise CE, Ghobrial RM, Shaked A. Improvement in survival associated with adult-to-adult living donor liver transplantation. Gastroenterology. 2007;133:1806-1813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 155] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 72. | Ghobrial RM, Freise CE, Trotter JF, Tong L, Ojo AO, Fair JH, Fisher RA, Emond JC, Koffron AJ, Pruett TL. Donor morbidity after living donation for liver transplantation. Gastroenterology. 2008;135:468-476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 347] [Cited by in RCA: 326] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 73. | Beavers KL, Sandler RS, Shrestha R. Donor morbidity associated with right lobectomy for living donor liver transplantation to adult recipients: a systematic review. Liver Transpl. 2002;8:110-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 169] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 74. | Ozgor D, Dirican A, Ates M, Gönültas F, Ara C, Yilmaz S. Donor complications among 500 living donor liver transplantations at a single center. Transplant Proc. 2012;44:1604-1607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 75. | Shio S, Yazumi S, Ogawa K, Hasegawa K, Tsuji Y, Kida M, Yamauchi J, Ida H, Tada S, Uemoto S. Biliary complications in donors for living donor liver transplantation. Am J Gastroenterol. 2008;103:1393-1398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 76. | Iida T, Ogura Y, Oike F, Hatano E, Kaido T, Egawa H, Takada Y, Uemoto S. Surgery-related morbidity in living donors for liver transplantation. Transplantation. 2010;89:1276-1282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 103] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 77. | El-Meteini M, Hamza A, Abdalaal A, Fathy M, Bahaa M, Mukhtar A, Abouelfetouh F, Mostafa I, Shaker M, Abdelwahab S. Biliary complications including single-donor mortality: experience of 207 adult-to-adult living donor liver transplantations with right liver grafts. HPB (Oxford). 2010;12:109-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 78. | Taketomi A, Kayashima H, Soejima Y, Yoshizumi T, Uchiyama H, Ikegami T, Yamashita Y, Harada N, Shimada M, Maehara Y. Donor risk in adult-to-adult living donor liver transplantation: impact of left lobe graft. Transplantation. 2009;87:445-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 92] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 79. | Lo CM. Complications and long-term outcome of living liver donors: a survey of 1,508 cases in five Asian centers. Transplantation. 2003;75:S12-S15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 207] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 80. | European Liver Transplant Registry. [Accessed 2015 Jan 8]. Available from: http://www.eltr.org/. |