Published online Oct 27, 2013. doi: 10.4254/wjh.v5.i10.589
Revised: September 7, 2013
Accepted: October 16, 2013
Published online: October 27, 2013
Processing time: 84 Days and 2.4 Hours
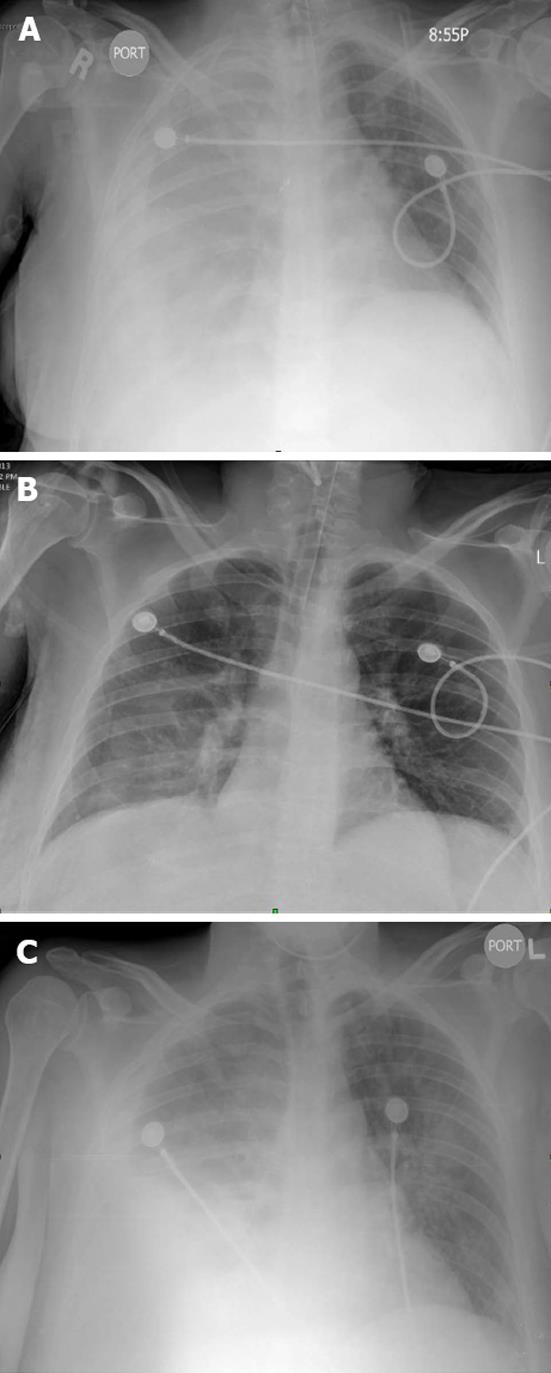
We report this case of a 43-year-old woman with hepatitis-C cirrhosis who presented with a large right sided pleural effusion complicated by hypoxic respiratory failure and altered mentation necessitating dependence on mechanical ventilation. The pleural effusion spontaneously resolved upon initiation of mechanical positive pressure ventilation and recurred almost immediately after weaning the patient off the ventilator. The pre-ventilation, ventilation and post-ventilation chest X-ray films in chronological order present a striking visual demonstration of fluid dynamics and pathophysiology of hepatic hydrothorax, thereby obviating the need for a dedicated diagnostic test. We also report this case to highlight the treatment strategies for this often intractable complication.
Core tip: We aim to publish this fascinating presentation of a case of hepatic hydrothorax that clearly provides an insight into pathophysiology of its development. We also presented its natural course and management. Such awareness will prompt institution of timely therapy and prevent long term sequelae of this potentially fatal condition.
- Citation: Gaduputi V, Tariq H, Kanneganti K. A fascinating presentation of hepatic hydrothorax. World J Hepatol 2013; 5(10): 589-591
- URL: https://www.wjgnet.com/1948-5182/full/v5/i10/589.htm
- DOI: https://dx.doi.org/10.4254/wjh.v5.i10.589
Hepatic hydrothorax is an uncommon manifestation of portal hypertension from cirrhosis which results in extracellular fluid accumulation. Hepatic hydrothorax is defined as a transudative pleural effusion in cirrhotic patients without underlying pulmonary or cardiac disease and is often > 500 mL in volume[1,2]. It is seen in approximately 5%-12% of all patients with cirrhosis[1,3,4]. Accumulation of fluid within the pleural cavity has greater clinical ramifications when compared to ascites, as even lesser volumes (1-2 L) can cause significant dyspnea and hypoxia. Therefore, prompt drainage of pleural fluid along with implementation of salt restriction and initiation of diuresis is often required.
A 43-year-old Hispanic woman who was born and raised in the United States, presented to the hospital with acutely altered mental status. The patient was known to have liver cirrhosis (Child-Pugh Class: C) from Hepatitis-C infection. A provisional diagnosis of hepatic encephalopathy was made and the patient was admitted to the critical care unit. Physical examination at the time of admission revealed a tachypneic and hypoxic disoriented woman. Other pertinent positive findings of physical examination included icteric sclera, decreased breath sounds over the right lung fields and bilateral pitting type of pedal edema. The patient was intubated and placed on mechanical ventilation. A chest X-ray taken immediately after intubation showed complete opacification of right hemithorax from a massive pleural effusion (Figure 1A).
Initial set of laboratory studies showed normocytic, normochromic anemia (Hemoglobin of 9.6 g/dL), severe thrombocytopenia (39 K/μL) , abnormal coagulation parameters (PT of 20.6 s and aPTT of 62.2 s) , abnormal liver enzymes (alanine transaminase of 41 IU/L, aspartate transaminase of 98 IU/L, elevated alkaline phosphatase of 193 IU/L) with jaundice (elevated total and direct bilirubin levels of 3.8 and 2.8 mg/dL respectively), profound serum hypoalbuminemia (2.0 g/dL). Serum ammonia level was elevated at 245 μmol/L. Echocardiogram revealed concentric left ventricular hypertrophy with normal ejection fraction of 83%. Thoracentesis was performed, with pleural fluid revealing a total protein of 1.0 gm/dL and a Serum-Pleural fluid Albumin Gradient of > 1.1, alluding to its transudative properties.
Hospital course of the patient was marked by treatment for hepatic encephalopathy with Lactulose and Rifaximin; work-up for a possible underlying precipitating factor. Septic work up including blood and urine cultures were non-contributory. The patient was not started on intravenous diuretics, in-spite of hypervolemia, in view of progressively worsening renal function (serum creatinine had trended up from 1.1 to 1.9 mg/dL) within 24 h of admission. Patient was initiated on intravenous Albumin infusions at a dose of 1 gm/kg. body weight/d. Renal function improved with these colloid infusions thus making Hepatorenal Syndrome unlikely and pre-renal azotemia secondary to third-spacing, more likely. Chest X-ray taken a day after the intubation revealed near-complete resolution of the right sided pleural effusion (Figure 1B). Patient was continued on mechanical ventilation for another day before being extubated upon improvement in her respiratory status. The patient was noted to have progressive improvement in her mentation and stable respiratory function over the next few days. On day-6 of hospitalization, patient was again found to be disoriented and hypoxic with a chest X-ray showing recurrence of the right sided pleural effusion (Figure 1C). The patient was reintubated and placed on mechanical ventilation.
Patient was considered for a tracheostomy in view of prolonged and recurrent intubation. However she could not undergo the procedure in view of her unstable medical condition, severe thrombocytopenia and progressively worsening coagulopathy. Patient was subsequently transferred to hospice care unit for palliative extubation.
This patient presented with multiple symptoms that are prototypical of hepatic hydrothorax secondary to decompensated liver cirrhosis, including dyspnea, hypoxia, laterality of pleural effusion (right sided in up to 65%-87% of cases)[4], transudative nature of pleural effusion and intractable nature of the pleural effusion. Even as hepatic hydrothorax is extensively reported in literature and could be found in almost 5%-10% of all patients with cirrhosis, the peculiar and strikingly labile presentation of hepatic hydrothorax in our patient, is noteworthy.
Several mechanisms have been postulated to explain the development of hepatic hydrothorax including decreased colloid osmotic pressure from hypoalbuminemia[5], increased azygos system pressure leading to leakage of plasma into the pleural space[5,6] and transdiaphragmatic migration of peritoneal fluid into the pleural space via lymphatic channels[7]. However, the most widely accepted theory is of the leakage of ascitic fluid via diaphragmatic defects[8,9]. The development of hepatic hydrothorax parallels other forms of fluid accumulation in cirrhosis. The basic pathophysiology of accumulation of fluid within the third space in cirrhosis, involves development of portal hypertension[10]; splanchnic vasodilation with subsequent activation of RAAS (renin-angiotensin-aldosterone system), activation of sympathetic nervous system and augmentation of Vasopressin release[11-13].
The dynamics of third space fluid are governed by the pressures of the cavities in which it exists. The negative intrathoracic pressure during inspiration aids in migration of fluid from peritoneal to the pleural space[3]. If the volume of this fluid exceeds the absorptive capacity of the pleural membranes, hepatic hydrothorax ensues. Our patient clearly exhibited this remarkable physiology of fluid dynamics while on and off mechanical ventilation. Patient had severe intrinsic liver disease resulting in formation of ascites and subsequently hepatic hydrothorax by mechanisms aforementioned. Mechanical ventilation imparted positive pressure that is transmitted to the intrapleural space. This positive pressure pushed the fluid back into the now-less pressurized peritoneal cavity, explaining the complete resolution of the right sided pleural effusion, post-intubation. After weaning off the positive pressure ventilation, the negative intrapleural pressure was quickly reestablished to aid patient inspire. This collaterally led to transmigration of fluid from ascitic cavity into the pleural space, thereby leading to reestablishment of hepatic hydrothorax.
The corner stone of management of hepatic hydrothorax is evaluation for liver transplantation[14]. However, in patients such as ours, who are acutely symptomatic and are not candidates for liver transplantation, the goal is to provide symptomatic relief. These palliative measures include large volume therapeutic thoracentesis[15] without the placement of an indwelling chest tube[16,17], salt restriction and diuresis[1,3]. Patients with hydrothorax refractory to these conservative measures might benefit from TIPS (Transjugular Intrahepatic Portosystemic Shunt) placement. However the success rate of TIPS is not encouraging in patients with advanced liver disease (Child Pugh Class C) such as seen in our patient[18]. The patient was not an ideal candidate for TIPS placement in view of her hepatic encephalopathy as well.
This case was unique, as it clearly demonstrates the fluid movements mirroring the pressure gradients between two different third space compartments, without the use of nuclear isotope studies involving the intraperitoneal administration of 99mTc-sulphur colloid or 99mTc-human serum albumin. This case highlights the challenges posed by refractory hepatic hydrothorax and often poor outcomes in presence of underlying advanced liver disease.
P- Reviewer Tanaka T S- Editor Zhai HH L- Editor A E- Editor Yan JL
| 1. | Cardenas A, Kelleher T, Chopra S. Review article: hepatic hydrothorax. Aliment Pharmacol Ther. 2004;20:271-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 100] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 2. | Kinasewitz GT, Keddissi JI. Hepatic hydrothorax. Curr Opin Pulm Med. 2003;9:261-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 42] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 3. | Strauss RM, Boyer TD. Hepatic hydrothorax. Semin Liver Dis. 1997;17:227-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 131] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 4. | Garcia N, Mihas AA. Hepatic hydrothorax: pathophysiology, diagnosis, and management. J Clin Gastroenterol. 2004;38:52-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 5. | MORROW CS, KANTOR M, ARMEN RN. Hepatic hydrothorax. Ann Intern Med. 1958;49:193-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 89] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Islam N, Ali S, Kabir H. Hepatic hydrothorax. Br J Dis Chest. 1965;59:222-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Roussos A, Philippou N, Mantzaris GJ, Gourgouliannis KI. Hepatic hydrothorax: pathophysiology diagnosis and management. J Gastroenterol Hepatol. 2007;22:1388-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 9. | Lieberman FL, Hidemura R, Peters RL, Reynolds TB. Pathogenesis and treatment of hydrothorax complicating cirrhosis with ascites. Ann Intern Med. 1966;64:341-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 150] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Ginès P, Fernández-Esparrach G, Arroyo V, Rodés J. Pathogenesis of ascites in cirrhosis. Semin Liver Dis. 1997;17:175-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 69] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Asbert M, Ginès A, Ginès P, Jiménez W, Clària J, Saló J, Arroyo V, Rivera F, Rodés J. Circulating levels of endothelin in cirrhosis. Gastroenterology. 1993;104:1485-1491. [PubMed] |
| 12. | Henriksen JH, Bendtsen F, Gerbes AL, Christensen NJ, Ring-Larsen H, Sørensen TI. Estimated central blood volume in cirrhosis: relationship to sympathetic nervous activity, beta-adrenergic blockade and atrial natriuretic factor. Hepatology. 1992;16:1163-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Arroyo V, Bosch J, Gaya-Beltrán J, Kravetz D, Estrada L, Rivera F, Rodés J. Plasma renin activity and urinary sodium excretion as prognostic indicators in nonazotemic cirrhosis with ascites. Ann Intern Med. 1981;94:198-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 82] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 14. | Xiol X, Tremosa G, Castellote J, Gornals J, Lama C, Lopez C, Figueras J. Liver transplantation in patients with hepatic hydrothorax. Transpl Int. 2005;18:672-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 63] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 15. | Lazaridis KN, Frank JW, Krowka MJ, Kamath PS. Hepatic hydrothorax: pathogenesis, diagnosis, and management. Am J Med. 1999;107:262-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 114] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 16. | Orman ES, Lok AS. Outcomes of patients with chest tube insertion for hepatic hydrothorax. Hepatol Int. 2009;3:582-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 17. | Runyon BA, Greenblatt M, Ming RH. Hepatic hydrothorax is a relative contraindication to chest tube insertion. Am J Gastroenterol. 1986;81:566-567. [PubMed] |
| 18. | Wilputte JY, Goffette P, Zech F, Godoy-Gepert A, Geubel A. The outcome after transjugular intrahepatic portosystemic shunt (TIPS) for hepatic hydrothorax is closely related to liver dysfunction: a long-term study in 28 patients. Acta Gastroenterol Belg. 2007;70:6-10. [PubMed] |