Published online Aug 27, 2023. doi: 10.4254/wjh.v15.i8.939
Peer-review started: May 15, 2023
First decision: July 10, 2023
Revised: July 23, 2023
Accepted: August 11, 2023
Article in press: August 11, 2023
Published online: August 27, 2023
Processing time: 99 Days and 7.5 Hours
Primary sclerosing cholangitis (PSC) is a chronic and progressive immune-mediated cholangiopathy causing biliary tree inflammation and scarring, leading to liver cirrhosis and end-stage liver disease. Diagnosis of PSC is challenging due to its nonspecific symptoms and overlap with other liver diseases. Despite the rising incidence of PSC, there is no proven medical therapy that can alter the natural history of the disease. While liver transplantation (LT) is the most effective approach for managing advanced liver disease caused by PSC, post-transplanta
Core Tip: Despite recent advancements in the field of hepatology, therapeutic options for the medical management of primary sclerosing cholangitis (PSC) are limited. Liver transplantation (LT) remains the primary treatment for patients with end stage liver disease (ESLD) secondary to PSC. Both deceased donor liver transplant and living donor liver transplant have demonstrated successful outcomes in patients with ESLD. Psychosocial patient factors also play a significant role in the outcome LT. Addressing ethical issues is crucial to ensure healthcare equity. Recent developments in digital technology and stem cell therapy suggest a promising future for LT in PSC.
- Citation: Shah YR, Nombera-Aznaran N, Guevara-Lazo D, Calderon-Martinez E, Tiwari A, Kanumilli S, Shah P, Pinnam BSM, Ali H, Dahiya DS. Liver transplant in primary sclerosing cholangitis: Current trends and future directions. World J Hepatol 2023; 15(8): 939-953
- URL: https://www.wjgnet.com/1948-5182/full/v15/i8/939.htm
- DOI: https://dx.doi.org/10.4254/wjh.v15.i8.939
Primary sclerosing cholangitis (PSC) is a chronic immune-mediated cholangiopathy characterized by inflammation and scarring of the biliary tree, affecting both intrahepatic and extrahepatic bile ducts. Its etiology remains idiopathic and it presents a wide spectrum of symptoms and complications. PSC is a rare condition with a global incidence rate ranging from 0 to 1.58 cases per 100000 per year, and a prevalence bracket of 0 to 31.7 cases per 100000 persons[1]. Recent research northern Europe has shown an increasing frequency of both new cases and overall instances of PSC[1]. As compared to the adult population, the incidence and prevalence of PSC is lower in the pediatric population at 0.2 and 1.5 per 100000 children[2].
Currently, there is no proven no medical therapy to treat effectively or alter the natural history of PSC[3]. As a result, the prognosis of this condition is poor, and it is strongly associated with an elevated risk of developing liver cirrhosis and end-stage liver disease, often necessitating liver transplantation (LT)[4]. The development and progression of PSC involve a combination of genetic susceptibility and environmental factors, although the contribution of genetic factors remains limited[5]. On the other hand, environmental factors, particularly the gut microbiota, have gained increasing attention in PSC development[6]. Additionally, approximately 70% of PSC patients have concomitant inflammatory bowel disease (IBD), which serves as a strong risk factor for colon, bile duct, and gallbladder cancers[7]. The co-occurrence of IBD and PSC is evident, with 2%-7.5% of IBD patients developing PSC[4].
Advancements in noninvasive imaging techniques, such as magnetic resonance cholangiopancreatography (MRCP), have improved the understanding and diagnosis of PSC, including its relationship to LT. PSC accounts for approximately 10% of all liver transplants performed annually[4-8]. However, post-transplant recurrence of PSC has been reported, highlighting the need for a better understanding of its underlying pathogenesis and the development of more comprehensive therapeutic strategies[9]. Despite these challenges, long-term outcomes following transplantation are encouraging, with a 5-year survival rate of 89% and favorable graft survival rates[10].
PSC is a poorly understood domain and has been presented with a huge void in terms of concrete solutions that are yet to be fulfilled. In this review, we discuss the current understanding of PSC’s pathogenesis, clinical presentation, management options, the scope of LT, and associated challenges.
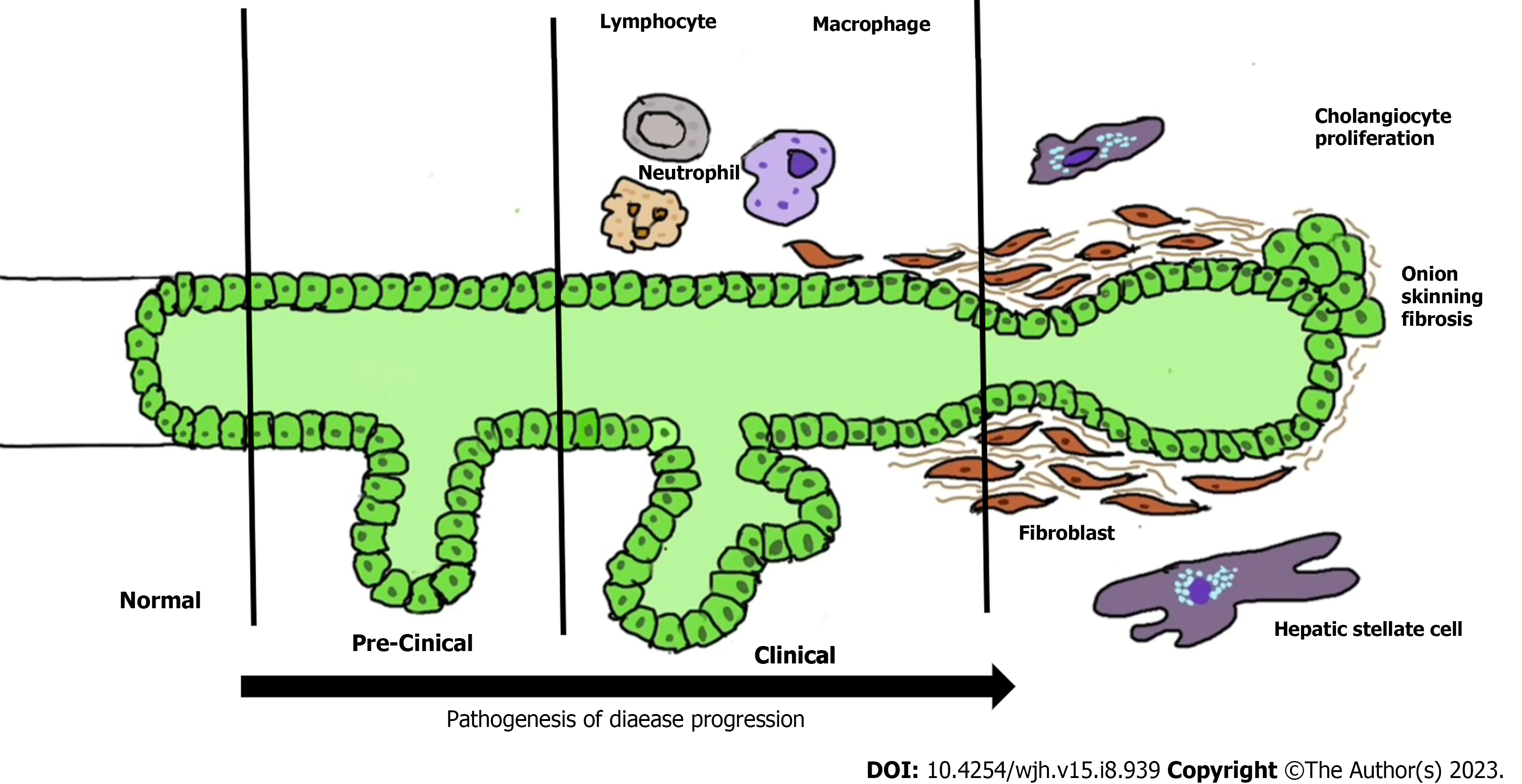
Inflammation and fibrosis of the bile ducts are two primary processes in the pathogenesis of PSC. However, the mechanism of inflammation and fibrosis in PSC are not fully understood. It is believed that various factors such as Ischemic, traumatic, infectious, autoimmune, or toxic injuries cause damage to cells, leading to the release of “danger-associated molecular patterns (DAMPs)”. These DAMPs activate the innate immune system through “pattern recognition receptors”[11]. Chronic inflammatory response mediated by DAMPs and the recruitment and activation of innate or adaptive immune cells play a critical role in initiating and perpetuating the activation of profibrogenic cells into myofibroblasts through the release of cytokines, chemokines, and reactive oxygen species (ROS). ROS and oxidative stress can induce hepatocyte injury, cell death, and parenchymal cell proliferation, along with altered remodeling and increased expression of tissue inhibitors of metalloproteinases[12]. Additionally, certain cytokines produced by damaged cells, such as interleukin (IL)-1a, IL-33, and others, directly or indirectly promote the development of a Th2 immune response, which is believed to promote fibrosis. Th2 immune response is recognized to have profibrotic properties through the release of IL-4, IL-5, IL-10, and IL-13[11,13]. However, the exact mechanisms and interactions between these processes are still being investigated in the context of PSC pathogenesis.
Cholangiocytes can be activated by various insults such as infections, cholestasis, etc., leading to increased proliferation along with pro-fibrotic and pro-inflammatory secretions through pleiotropic autocrine and paracrine mechanisms[14]. Persistent biliary cell damage causes an inflammatory reaction that leads to a pathological reparative reaction with excessive deposition of scar tissue around the injured ducts. The biliary epithelium is exposed to cytokines and chemokines secreted by innate and adaptive immune cells in response to DAMPs. If biliary homeostasis is not restored, there will be a maladaptive chronic inflammatory response stimulating the deposition of connective tissue (Figure 1)[14].
Genetic factors play a significant role in PSC pathogenesis. Studies have demonstrated an increased risk of PSC among first-degree relatives of patients with the disease[15]. Genome-wide association studies have identified over 20 susceptibility genes for PSC, with the human leukocyte antigen (HLA) complex on chromosome six showing the strongest association[16-20]. Patients with PSC exhibit chromosomal instability and immunosenescence, as evidenced by higher rates of short telomere length and telomere aggregates compared to patients with IBD[21,22]. It is important to note that genetic findings explain less than 10% of the disease liability, while environmental factors account for over 50% of it[22].
The microbiome has also been implicated in PSC pathogenesis, with bacteria potentially triggering an aberrant immune response and perpetuating inflammation[23]. Studies have shown an enrichment of Barnesiallaceae and Blautia families and Barnesiellacaeae genus in PSC patients. Microbiome shifts associated with PSC are observed in Clostridiales and Bacteroidales orders, with more than 80% of shifts occurring within the former order. However, the causal relationship between these shifts remain unclear due to limited sample size[24]. Some environmental triggers have been investigated, indicating a higher prevalence of PSC in rural areas and a possible connection to agricultural activities, pesticides, or fertilizers[25]. Close contact with dogs or cats has also been identified as a potential trigger, suggesting the pathogenic role of an unidentified agent such as a toxin or microbiome[26]. Additionally, coffee consumption and smoking have been suggested as protective factors against PSC[27].
PSC is characterized by bile duct injury and fibrosis, leading to a variety of symptoms and signs. It is commonly associated with IBD[12]. It typically affects individuals between the ages of 30 and 40, with a higher prevalence in men[28]. PSC patients are identified during general health examinations or the investigation for another disease, and about 50% patients are asymptomatic[29]. When symptoms occur, the most common are pruritus, fatigue, and right upper quadrant pain; less frequent symptoms are weight loss and fever[29,30]. Physical exam often reveals jaundice, hepatomegaly, splenomegaly, and excoriation marks[29,31]. PSC is a progressive cholestatic liver disease associated with complications such as bacterial cholangitis, dominant strictures, gallbladder polyps, adenocarcinoma, and cholangiocarcinoma (CCA)[29,30,32]. Disease progression may differ in children due to absence of other risk factors like alcohol abuse or polypharmacy that can lead to faster progression of the liver disease[33].
Diagnosis of PSC relies on the presence of cholestasis markers [alkaline phosphatase (ALP) and gamma-glutamyl (GGT) transferase], characteristic bile duct changes on imaging and the exclusion of secondary causes[29,20]. The elevation of serum ALP is the commonest marker[29,32]. ALP is not reliable in children as it can be elevated due to high bone turnover. So, GGT transferase is more commonly used as a diagnostic marker in the pediatric population[34]. However, the transient blockage of the strictured bile ducts can create fluctuations in ALP and bilirubin levels. The total serum bilirubin level is usually normal, but an increase or fluctuations in bilirubin levels indicate the presence of dominant strictures or advanced liver disease[32,35]. The dominant strictures are present in around 45% of adult patients at the diagnosis of PSC as compared to < 5% in the pediatric population[34]. Additionally, serum aminotransferases are elevated 2-3 times the upper limit of normal[29]. In cases of a high level of serum aminotransferases, autoimmune hepatitis should be ruled out[36].
PSC is commonly associated with an underlying IBD, with ulcerative colitis (UC) being the most prevalent. Both PSC and UC have an autoimmune component, which is reflected in the presence of autoantibodies. The most frequently reported autoantibodies in PSC and UC are perinuclear antineutrophil cytoplasmic antibodies, found in 26%-94% of PSC cases and 50%-70% of UC cases[37,38]. Additional autoantibodies reported in PSC include antinuclear antibodies (present in 8%-77% of patients) and smooth muscle antibodies (present in 0%-83% of patients)[32,38]. However, it's important to highlight that these autoantibodies lack specificity and are not necessary for a diagnosis of PSC.
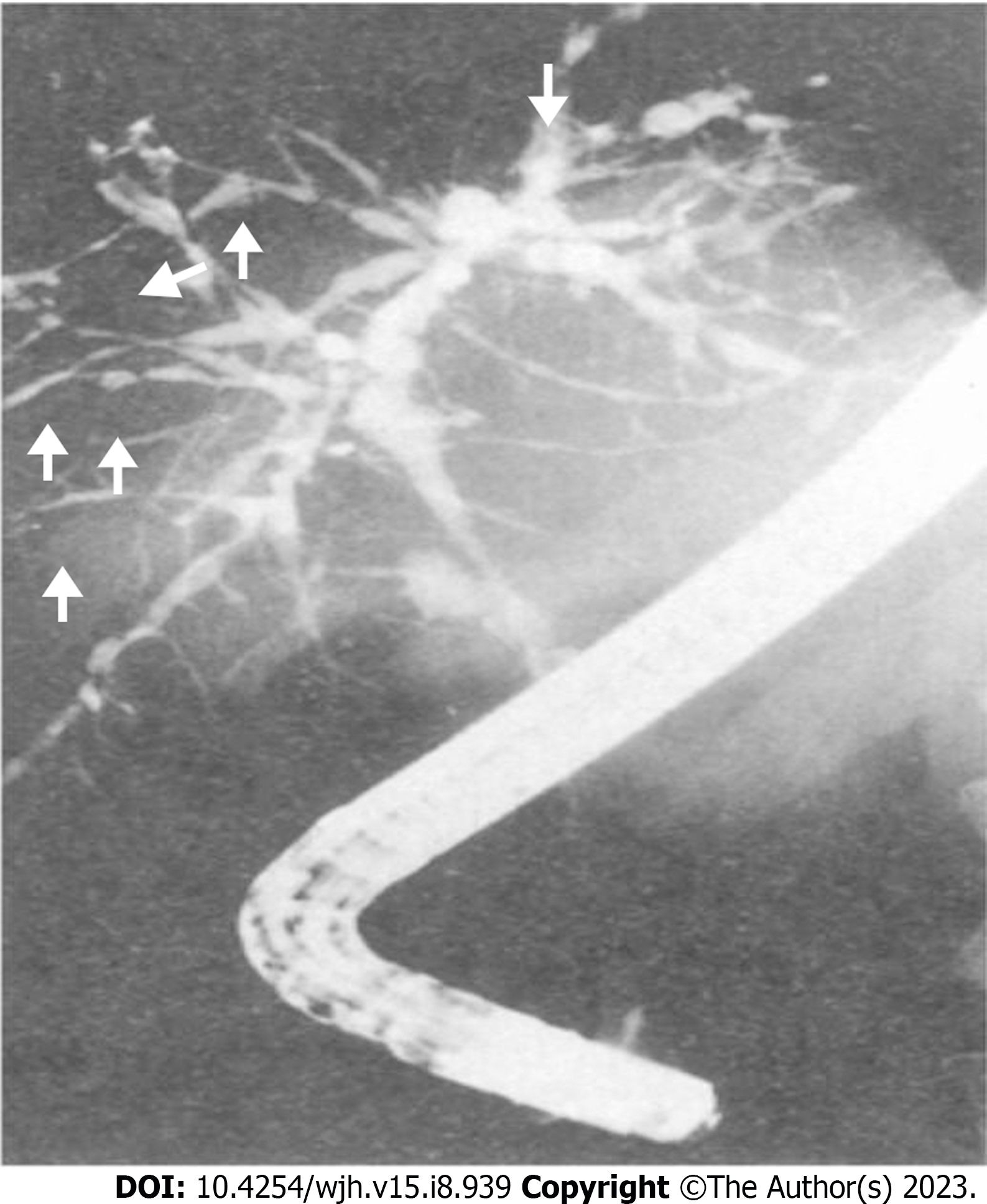
Imaging techniques such as abdominal X-ray and ultrasound can reveal abnormal bile ducts and exclude gallstones. However, these techniques are unable to provide a clear view of intrahepatic biliary ducts. Additionally, sclerosis does not dilate the ducts enough to be seen on imaging, resulting in suboptimal assessment in suspected cases of PSC[39]. For this reason, cholangiography assessment is essential for the diagnosis of PSC, as the morphological features of PSC mainly involve biliary ductal changes, while liver parenchymal changes develop later[39]. The common imaging findings in PSC seen on MRCP or endoscopic retrograde cholangiopancreatography (ERCP) include intrahepatic and extrahepatic bile duct strictures, which alternate with normal or dilated bile ducts showing a beading appearance (Figure 2)[39-41]. MRCP is preferred as an initial non-invasive imaging method, while ERCP is reserved for therapeutic interventions[30,32,42,43]. The sensitivity and specificity for ERCP in the diagnosis of PSC are 89% and 80% respectively[44]. MRCP has shown a sensitivity of 86% and specificity of 94% for diagnosis of PSC, with superior cost-effectiveness compared to ERCP[45,46].
PSC is staged using a four-stage system first developed by Ludwig et al[47] in 1978, which is shown in Table 1. Several good prognostic factors have been identified, including young age, female sex, small duct phenotype, and the presence of Crohn’s disease[48]. In early disease, the Mayo PSC risk score can be useful in predicting short-term survival, but it cannot predict the need for LT[49,50]. However, a meta-analysis has shown that the United Kingdom-PSC score and the PSC risk estimate tool are better at predicting long-term risk[51-53]. The components of each prognostic score are listed in Table 2[49,52-57].
| Primary sclerosing cholangitis stages | Histological finding | |
| Stage I | Portal stage | Presence of portal hepatitis and edema confined to the portal triads with mononuclear infiltration |
| Stage II | Periportal stage | The inflammation progresses to the periportal space causing periductal fibrosis with dilation of the portal triads. There is absence of bridging necrosis or septal fibrosis |
| Stage III | Septal stage | Characterized by the presence of fibrous septae and/or bridging fibrosis |
| Stage IV | Cirrhosis | Established cirrhosis with the presence of fibrous septa and nodular regeneration |
| Clinical scores | Components |
| Mayo risk score | Age, bilirubin, histological stage, hemoglobin and presence of inflammatory bowel disease |
| Revised Mayo risk score | Age, bilirubin, albumin, aspartate aminotransferase and variceal bleeding |
| Amsterdam–Oxford model | Primary sclerosis cholangitis (PSC) subtype, age at PSC diagnosis, albumin, alkaline phosphatase, aspartate aminotransferase, bilirubin and platelets |
| Short-term United Kingdom-pSC risk score | Bilirubin, albumin, hemoglobin, and platelets count at diagnosis |
| Long-term United Kingdom-pSC risk score | Age at diagnosis, bilirubin at the second year, alkaline phosphatase at the second year, albumin at the second year, platelets at the second year, presence of extrahepatic biliary disease at diagnosis, and variceal hemorrhage by the second year |
| Primary sclerosing cholangitis risk estimate tool | Bilirubin, albumin, alkaline phosphatase, platelets, aspartate transaminases, hemoglobin, sodium, PSC duration and age |
| Model for end stage liver disease | Dialysis at least twice in the past week, creatinine, bilirubin, international normalized ratio and sodium |
| Child-Pugh score | Bilirubin, albumin, international normalized ratio, ascites and encephalopathy |
The management of PSC focuses on slowing the disease progression and managing its complications. However, there is no definitive treatment to halt the disease process. LT is a viable option for advanced cases and has shown favorable outcomes.
Ursodeoxycholic acid (UDCA), a hydrophilic bile acid, is commonly used in the treatment of cholestatic liver diseases and is extensively studied in PSC[58]. Its mechanisms of action include protecting cholangiocytes against cytotoxic hydrophobic bile acids in early stages, stimulating hepatobiliary secretion in more advanced stages, and protection of hepatocytes against bile acid-induced apoptosis in all stages[59,60]. UDCA has been shown to improve liver function tests, its impact on survival rates, prevention of CCA, or clinical symptoms is inconclusive[61-63]. However, other data has shown that meaningful reductions in ALP levels have been associated with better outcomes in PSC[64-66]. In addition, withdrawing UDCA may be associated with increase in fatigue, pruritus, liver biochemistries, and Mayo PSC risk score[58,67]. American Association for the Study of Liver Diseases (AASLD) updated their guidelines on PSC management in 2022, to suggest a dose of 13-23 mg/kg/d of UDCA, with continued use if there is a reduction or normalization of ALP levels and/or improvement of symptoms after 12 mo of treatment[68].
Immunosuppressive therapies, including glucocorticoids, cyclosporine, tacrolimus, methotrexate, and mycophenolate mofetil, have been explored for PSC treatment. However, a systematic review concluded that these agents, either as monotherapy or in combination do not reduce the risk of mortality or LT, and monotherapy may increase adverse effects[69]. Recent findings from a meta-analysis suggest that immune-modulating therapy may benefit patients with high baseline levels of ALP (> 420 U/L) and aspartate transaminase (> 80 U/L)[70]. Nevertheless, immunosuppressive agents should be reserved for patients with overlap syndromes such as autoimmune hepatitis-PSC or IgG4-associated cholangitis[68]. Ongoing clinical trials are investigating potential treatments options, such as cilofexor (a nonsteroidal farnesoid X receptor agonist), and 24-nor UDCA (a derivative of UDCA), which show promising results[71,72].
Medical management of PSC has several limitations, despite various drugs being investigated, a recent meta-analysis concluded that there is currently insufficient evidence to show differences in effectiveness measures, such as mortality, health-related quality of life, cirrhosis, or LT between any active pharmacological intervention and no intervention[73]. The high risk of bias in most assessed trials further underscores the need for well-designed randomized controlled trials with adequate follow-up in order to improve pharmacological management of patients with PSC. An overview of various clinical trials and meta-analysis assessing the efficacy and adverse effects of medications used in management of PSC is described in Table 3[61-63,69,70,72,74-76].
| Ref. | Year | Type | Objective | Results | |||||||
| Death | Symptoms (fatigue, pruritus) | Liver transplantation | Histological improvement | Marker values (bilirubin, GGT, ALP, ALT or AST) | Cholangiographic changes | Cholangiocarcinoma | Adverse events | ||||
| Ursodeoxycholic acid | |||||||||||
| Shi et al[74] | 2009 | Meta-analysis of RCT (8 RCT, 465 patients) | Evaluate the effect and safety of UDCA in PSC | No significant effect | No significant effect | No significant effect | Significant difference | No significant effect | No significant effect on improvement | No significant difference on incidence | No significant difference on incidence |
| Othman et al[61] | 2012 | Meta-analysis of RCT (7 RCT, 553 patients) | Investigate the efficacy of UDCA in PSC | No significant effect | No significant effect | No significant effect | No significant effect | Significantly decrease ALP, GGT, bilirubin, ALT or AST | No significant effect on improvement | No significant difference on incidence | No significant difference on incidence |
| Poropat et a[62] | 2011 | Meta-analysis of RCT (8 RCT, 592 patients) | Assess the beneficial and harmful effects of BA for patients with PSC | No significant effect | No significant effect | No significant effect | No significant effect | Significantly decrease ALP, GGT, bilirubin or AST. Not significant effect on albumin | No significant effect on improvement | No significant difference on incidence | No significant difference on incidence |
| Triantos et al[63] | 2011 | Meta-analysis of RCT (8 RCT, 567 patients) | Evaluate if UDCA is useful for PSC | No significant effect | No significant effect | No significant effect | No significant effect | Not reported | Not reported | No significant difference on incidence | Not reported |
| Immunosuppressive therapies: glucocorticoids, cyclosporine, tacrolimus, methotrexate and mycophenolate mofetil | |||||||||||
| Peng et al[69] | 2017 | Meta-analysis of RCT (7 RCT, 266 patients) | Evaluate the safety and efficiency of IA for the treatment of PSC | No significant effect | Not reported | No significant effect | Not reported | No significant improvement on liver biochemistry except AST | Not reported | Not reported | Significant increase on incidence |
| Liu et al[70] | 2022 | Meta-analysis (7 RCT and 14 observational, 737 patients) | Assess the efficacy and adverse effects of immunomodulators in adult patients with PSC | Not reported | Not reported | Not reported | Not reported | Significantly decrease ALP. Not significant effect on bilirubin and AST | Not reported | Not reported | 16.1% of patients had severe AEs1 |
| Antibiotics | |||||||||||
| Shah et al[75] | 2019 | Meta-analysis of clinical trials (3 RCT and 2 open labeled trials, 124 patients) | Assess the effect of antibiotic therapy (vancomycin, metronidazole, rifaximin and minocycline) in PSC with or without inflammatory bowel disease | Not reported | Not reported | Not reported | Not reported | Significant reduction in ALP and bilirubin | Not reported | Not reported | 8.9 % of patients had severe AEs1 |
| Probiotics | |||||||||||
| Vleggaar et al[76] | 2008 | RCT that included 14 patients | Assess potential beneficial effects of probiotics in PSC | Not reported | No significant effect | Not reported | Not reported | No significant effect on bilirubin, ALP, GGT, AST, ALT, prothrombin, albumin or bile salts | Not reported | Not reported | Not reported |
| Newer drugs | |||||||||||
| Fickert et al[72] | 2017 | RCT that included 161 patients | Evaluate the safety and efficacy of three doses of oral nor UDCA compared with placebo in patients with PSC | Not reported | No significant effect | Not reported | Not reported | Significantly decrease ALP, GGT, ALT or AST | Not reported | Not reported | 14 patients had severe AE1 |
While most cases of PSC are characterized by multifocal bile duct strictures, a few have a localized high-grade stricture (dominant stricture) superimposed on diffuse disease that can cause jaundice or cholangitis[77]. Furthermore, CCA may appear as a dominant stricture[78,79]. Hence, brush cytology of the biliary tree, endobiliary biopsy, and fluorescence in situ hybridization should be performed to assess it[68]. AASLD recommends ERCP for the evaluation of relevant strictures as well as new-onset or worsening pruritus, unexplained weight loss, worsening serum liver test abnormalities, rising serum cancer antigen 19-9, recurrent bacterial cholangitis, or progressive bile duct dilation[68]. However, it is important to consider that PSC patients undergoing ERCP have an increased risk of bacterial cholangitis and pancreatitis, so antimicrobial prophylaxis should be administered before the procedure[80-84].
Indications for liver transplant in PSC: LT is performed in patients with PSC when medical therapy has reached its limits[85]. PSC is a hepatic condition with a variable clinical course. LT becomes necessary when the patient develops end-stage liver disease and complications related to portal hypertension, such as ascites, hepatic encephalopathy, variceal hemorrhage, or spontaneous bacterial peritonitis[51,85]. In 2006, United Network Of Organ Sharing reported that 6650 patients received liver transplants, while 17221 were on the waiting list[86]. To address the insufficient number of deceased donors and long wait times, living donor liver transplant (LDLT) emerged as an alternative with favorable outcomes for acute and chronic liver diseases, provided appropriate selection criteria[87,88].
In the United States, Model For End Stage Liver Disease (MELD) score is used by Organ Procurement and Transportation in Network (OPTN) to prioritize liver transplant recipients. LT is considered when MELD score is ≥ 15, indicating hepatocellular dysfunction[85]. The MELD score incorporates the patient’s serum bilirubin level, international normalized ratio, and serum creatinine level[89]. MELD exceptions for LT are granted to patients with at least two admissions within a 1-year period for acute cholangitis with a documented bloodstream in infection or with evidence of sepsis requiring vasopressors for hemodynamic instability, as well as those with a diagnosis of CCA[90]. The inclusion and exclusion criteria for LT in patients with CCA are detailed in Table 4[68,91]. The LT process involves a multidisciplinary team with different roles, as outlined in Table 5[92].
| Inclusion criteria | Exclusion criteria |
| Evidence of positive tumor cells or cells strongly suspicious for CCA on biopsy | Evidence of extra hepatic disease or lymph node enlargement |
| Malignant appearing stricture on radiograph and 1 of the following criteria (a or b or c) | Previous malignancy excluding skin or cervical cancer within 5 yr before diagnosis of cholangiocarcinoma |
| a. Ca 19-19 > 100 U/mL in the absence of acute bacterial cholangitis | History of abdominal radiotherapy |
| b. Polysomy on fluorescence in-situ hybridization | Uncontrolled infection before treatment |
| c. Hilar mass < 3 cm in radial diameter on cross-sectional imaging | Prior attempt of surgical tumors reaction and subsequent violation of tumor plane |
| - | Any medical condition precluding transplantation |
| - | Any transperitoneal biopsy including percutaneous and/or endoscopic ultrasonographic-guided fine needle aspiration |
| Role | Description |
| Transplant hepatologist | A medical doctor who specializes in liver disease |
| Liver transplant surgeon | Evaluates the patient and determines whether a liver transplant is the best option by considering surgical contraindications |
| Transplant nurse coordinator | Serves as the primary contact for the patient throughout the transplant process, ensures that testing is up-to-date, and provides education on the transplant process |
| Transplant social worker | Focuses on the psychological and social aspects of end-stage liver disease and provides mental health support as needed |
| Transplant nutritionist | Assesses the patient's nutritional status, including weight patterns and dietary intake, and makes recommendations for an optimal diet |
| Financial coordinator | Reviews the patient's medical insurance coverage and assists with obtaining adequate coverage for the transplant |
| Transplant pharmacist | Reviews the patient's medication list for any contraindications before the transplant and provides education on new medications after the transplant |
Psychosocial evaluation in liver transplant candidates: Patients with PSC may experience anxiety, depression, substance use disorder, or other psychological symptoms due to the chronic and potentially progressive nature of the disease[93]. 35%-65% of transplant candidates meet criteria for an internalizing disorder as a result of waiting and anticipating surgery[94]. Chronic illness can impact the quality of life, especially in case of conditions like PSC where patients may have unpredictable flares of symptoms. The psychological evaluation can also help in identifying coping strategies that patients can use to manage their symptoms and improve their overall well-being[93]. Various psychological instruments used for psychosocial evaluation in transplant candidates are listed in Table 6[93].
| Instrument name | Description | Scoring/rating | Reliability | Validity |
| Beck depression inventory | Self-report measure of depressive symptoms | 21-item scale, higher scores indicate more severe depressive symptoms | High test-retest reliability, internal consistency, and concurrent validity | Established validity in measuring depressive symptoms in various populations |
| Hamilton depression rating scale | Clinician-rated scale to assess severity of depressive symptoms | 17-item scale, higher scores indicate more severe depressive symptoms | High inter-rater reliability, internal consistency, and concurrent validity | Established validity in measuring depressive symptoms in various populations |
| General health questionnaire | Self-report measure of general mental health | 12-item or 28-item scale, higher scores indicate poorer mental health | High internal consistency, test-retest reliability, and concurrent validity | Widely used in assessing mental health in general populations |
| Primary care evaluation of mental disorders-patient health questionnaire | Self-report measure of common mental disorders | 9-item scale, higher scores indicate greater severity of mental disorder symptoms | High sensitivity and specificity, test-retest reliability, and convergent validity | Widely used in primary care settings to screen for mental disorders |
| Transplant evaluation rating scale | Clinician-rated scale to assess psychosocial functioning in transplant recipients | 10 aspects of psychosocial functioning rated on a 5-point scale, higher scores indicate better adjustment | Good inter-rater reliability and validity in liver transplant recipients | Specific to evaluating psychosocial functioning in transplant recipients |
| Psychosocial assessment of candidates for transplantation | Clinician-rated scale to assess psychosocial acceptability of transplant candidates | 8 subscales rated on a 5-point scale, with initial and final overall ratings | Established reliability and validity in evaluating psychosocial acceptability of transplant candidates | Widely used in evaluating transplant candidate suitability |
| Stanford integrated psychosocial assessment for transplantation | Clinician-rated scale to assess psychosocial functioning in transplant candidates | Comprehensive assessment covering multiple domains of psychosocial functioning | Limited data on reliability and validity, but shows promise in transplant candidate evaluation | Developed specifically for evaluating psychosocial functioning in transplant candidates |
Overall, the medical and psychological evaluation in PSC plays a crucial role in assessing the severity of the disease and identifying any associated conditions, as well as addressing psychological factors that may affect the patient's quality of life[93].
Ethical considerations in liver transplant candidates: Organ transplantation raises significant ethical considerations, making it one of the most controversial disciplines in medicine[95]. Key ethical concerns related to organ retrieval include accurately diagnosing brain death, respecting the patient's known wishes regarding organ donation, and upholding the principle of altruism in living organ donation[96]. When it comes to living organ donors, ensuring their understanding of the surgery’s risks, benefits and potential complications is crucial, especially during the informed consent process. Comprehensive discussions on short-term and long-term outcomes should take place at this stage[97]. There is notable regional variability in the application and acceptance of MELD exceptions for LT. A study revealed that, despite OPTN’s clinical criteria, nearly 80% of exception applications for PSC and cholangitis were approved by regional review boards regardless of the indication[98]. This highlights the need for a standardized national review board to ensure equitable access to LT for patients with PSC and bacterial cholangitis[98].
Sex-based disparities in organ transplantation are also a concern. The MELD score, which relies on creatinine levels, underestimates true renal function in females due to lower muscle mass. Additionally, men face an increased risk of hepatocellular carcinoma, which can lead to MELD exceptions[95]. A analysis of waitlisted candidates from the scientific registry for transplant recipients showed that Hispanics with MELD scores < 20 had an 8% lower deceased donor liver transplant (DDLT) rate compared to Whites[99]. Asian patients with MELD score < 15, on the other hand, had a 24% higher DDLT rate compared to Whites, but this rate dropped by 46% for Asian patients with MELD scores between 30-40 compared to Whites[99]. As the field of LT continues to evolve, addressing ethical concerns requires filling knowledge gaps with robust and carefully gathered data that go beyond the informed consent of donors[95].
Outcomes of liver transplant in patients with PSC: The one-year and five-year survival rates were better in patients with LT for impaired quality of life (97.4% and 94.9%) as compared to patients with LT for end-stage liver disease (91.4% and 88.6% respectively) based on a retrospective study on 74 patients with LT[100]. The one-year and five-year survival rates for patients with suspicion of neoplasia prior to the LT were 95.8% and 74.1% respectively[100]. A larger study of 6071 patients had similar outcomes with patient survival rate of 89.7%, 79.8%, 70.7%, 58.3%, 43.8% and 20.4% respectively at 1, 5, 10, 15, 20, and 30 years respectively[101]. Based on a study of 6911 LT patients from the OPTN database, the unadjusted survival rate was significantly higher among the LDLT group as compared to the DDLT group[102]. The most common factors associated with death after LT were infections, malignancies, cardiovascular diseases, graft failure (GF) due to rejection, and hepatic artery thrombosis (HAT)[101,103,104].
Patients with LT for PSC have an acute cellular rejection (ACR) rate of 20-40%, requiring additional immunosuppression[105]. ACR does not affect long-term graft or survival outcomes in patients with LT, as opposed to patients with renal transplant[105]. A retrospective study of patients with a diagnosis of PSC (24 patients) and PSC-autoimmune hepatitis overlap (2 patients) without evidence of CCA at the time of LDLT showed allograft rejection successfully managed by immunosuppression in 11.5% patients, postoperative bile leak in 7.6% patients managed conservatively, and biliary stricture in 11.5% patients with successful ERCP and biliary stent placement[103]. Biliary strictures and bile leaks are other common complications after LT with an incidence of 5%-15% in patients who received DDLT and 28%-32% in recipients of right lobe LDLT[106]. The mean time interval for presentation of biliary strictures after LT is 5-8 mo[106]. Biliary strictures are classified into anastomotic variant and non-anastomotic (NAS) variant. NAS variants may be caused due to HAT and non-HAT etiologies like chronic ductopenic rejection, ABO incompatibility, PSC causing recurrent or ischemic strictures, age of the donors, duration of use of vasopressors, prolonged cold and warm ischemia times, preservative injury, and donation after cardiac deaths[106]. NAS variants related to ischemia usually present within one year and those related to immunological factors present after one year of LT[107].
A study based on the review of 22 publications with a total of 1399 patients who underwent LT for PSC showed that the recurrence rate of PSC was around 18.5%, ranging from 5.7%-59.1%[108]. Another study with a patient population of 230 had a recurrence rate of 23.5% with a median of 4.6 years after LT[108]. Some of the most common factors related to an increased risk of recurrence of PSC are presence of HLA-DRB1*08 in the donor or recipient, absence of donor HLA DR52, older and younger recipients, male recipients, development of UC after LT, requirement of a longer duration of maintenance therapy with steroids (> 3 mo), steroid resistant ACR, and the presence of CCA or concurrent infection with cytomegalovirus in the donor[108]. Due to a high recurrence rate of PSC in patients and a 4-fold increase in the risk of GF or mortality within 5 years of LT; liver re-transplant (ReLT) is considered to extend survival[109].
Quality of life and psychological outcomes after liver transplant: The health-related quality of life (HRQOL) and employment after LT depends on the etiology of the ESLD. In a cross-sectional study of 356 patients post LT, the return to employment rates within six months were highest amongst patients with PSC (2.4 times) and alcoholic cirrhosis (2.5 times) as compared to patients with primary biliary cirrhosis. However, post LT HRQOL was comparable among different ESLD etiologies[110]. Early retirement was also significantly higher, reaching 83% in patients with PSC[110]. Most commonly reported symptoms of physical distress after LT were fatigue, muscle weakness, increased appetite, headache, backache, and bruising which were higher in females over one year as compared to men[111]. The most commonly reported symptoms of psychological distress at one year were sleeplessness and mood swings, followed by nervousness, depression, and difficulty concentrating[111]. Recipients of LT rated their overall health as 7.17 ± 2.22 out of a possible score of 10 based on a questionnaire adapted from Karnofsky functional performance scale, medical outcomes study short form (SF-36), and psychosocial adjustment to illness scale, with 10 being the best outcome[112]. The greatest benefit reported post LT was “being alive”. The worst factor reported about being a LT recipient was dependence on medications and the cost of insurance and medications[112].
Future directions in management of PSC: Recent advancement in digital technology have opened up new possibilities of enhancing the understanding of liver anatomy and vascular structures through the creation of three-dimensional (3D) liver models using data from computed tomography and magnetic resonance imaging scans[113]. PSC often involves the development of strictures and narrowing of the bile ducts, making LT surgery more challenging[114]. The emerging technique of 3D liver transplant offers surgeons assistance in planning surgical procedures, including precise identification of blood vessels and bile ducts, improving the accuracy and efficiency of transplant surgeries[115,116].
Stem cell therapy and gene therapy represent two emerging treatments with potential for managing PSC. Stem cell therapy involves using stem cells to repair and regenerate damaged liver tissue[117]. Various types of stem cells , such as mesenchymal stem cells, induced pluripotent stem cells, and embryonic stem cells, have been investigated for their potential in treating liver diseases, including PSC. Studies indicate that stem cell therapy may reduce inflammation and promote tissue regeneration in PSC cases[117,118]. Gene therapy, on the other hand, utilizes genes to modify the expression of specific proteins involved in the development and progression of the disease. One potential target for gene therapy in PSC is the nuclear factor kappa B (NF-κB) pathway, which plays a role in liver inflammation regulation. Inhibiting the NF-κB pathway has shown promise in reducing inflammation and fibrosis in PSC[119]. Despite the potential benefits of stem cell and gene therapies in PSC, further research is needed to determine their safety and efficacy. Ongoing clinical trials are evaluating the use of these therapies in PSC treatment, and their outcomes will determine their future role in managing the disease[117].
LT plays a very crucial role in the management of patients with PSC due to limited options and studies on outcomes with medical management. This review offers a summary of clinical features, diagnosis, medical management and a detailed discussion on the indications, clinical and psychosocial outcomes, ethical dilemmas, and future aspects in the field of liver transplant for management of PSC. The review also highlights the important aspect of pReLT psychosocial evaluation, as well as psychosocial outcomes post-transplant, which plays a pivotal role in preventing mental health crises in the patients. Significant efforts need to be directed towards addressing the ethical issues in liver transplant for equity of the care. Patients with PSC will also greatly benefit from more advances in the medical management of PSC.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: American College of Gastroenterology; American Gastroenterological Association; American Society for Gastrointestinal Endoscopy.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Gad EH, Egypt; Mogahed EA, Egypt S-Editor: Qu XL L-Editor: A P-Editor: Cai YX
| 1. | Mehta TI, Weissman S, Fung BM, Sotiriadis J, Lindor KD, Tabibian JH. Global incidence, prevalence and features of primary sclerosing cholangitis: A systematic review and meta-analysis. Liver Int. 2021;41:2418-2426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (38)] |
| 2. | Cotter JM, Mack CL. Primary sclerosing cholangitis: Unique aspects of disease in children. Clin Liver Dis (Hoboken). 2017;10:120-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (35)] |
| 3. | Eaton JE, Talwalkar JA, Lazaridis KN, Gores GJ, Lindor KD. Pathogenesis of primary sclerosing cholangitis and advances in diagnosis and management. Gastroenterology. 2013;145:521-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 271] [Cited by in RCA: 295] [Article Influence: 24.6] [Reference Citation Analysis (36)] |
| 4. | Karlsen TH, Folseraas T, Thorburn D, Vesterhus M. Primary sclerosing cholangitis - a comprehensive review. J Hepatol. 2017;67:1298-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 373] [Cited by in RCA: 568] [Article Influence: 71.0] [Reference Citation Analysis (35)] |
| 5. | Lazaridis KN, LaRusso NF. The Cholangiopathies. Mayo Clin Proc. 2015;90:791-800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 156] [Article Influence: 15.6] [Reference Citation Analysis (37)] |
| 6. | Qin N, Yang F, Li A, Prifti E, Chen Y, Shao L, Guo J, Le Chatelier E, Yao J, Wu L, Zhou J, Ni S, Liu L, Pons N, Batto JM, Kennedy SP, Leonard P, Yuan C, Ding W, Hu X, Zheng B, Qian G, Xu W, Ehrlich SD, Zheng S, Li L. Alterations of the human gut microbiome in liver cirrhosis. Nature. 2014;513:59-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1230] [Cited by in RCA: 1538] [Article Influence: 139.8] [Reference Citation Analysis (40)] |
| 7. | Jussila A, Virta LJ, Pukkala E, Färkkilä MA. Malignancies in patients with inflammatory bowel disease: a nationwide register study in Finland. Scand J Gastroenterol. 2013;48:1405-1413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (39)] |
| 8. | Staufer K, Kivaranovic D, Rasoul-Rockenschaub S, Soliman T, Trauner M, Berlakovich G. Waitlist mortality and post-transplant survival in patients with cholestatic liver disease - Impact of changes in allocation policy. HPB (Oxford). 2018;20:916-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (16)] |
| 9. | Vera A, Moledina S, Gunson B, Hubscher S, Mirza D, Olliff S, Neuberger J. Risk factors for recurrence of primary sclerosing cholangitis of liver allograft. Lancet. 2002;360:1943-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 138] [Article Influence: 6.0] [Reference Citation Analysis (36)] |
| 10. | Kashyap R, Mantry P, Sharma R, Maloo MK, Safadjou S, Qi Y, Jain A, Maliakkal B, Ryan C, Orloff M. Comparative analysis of outcomes in living and deceased donor liver transplants for primary sclerosing cholangitis. J Gastrointest Surg. 2009;13:1480-1486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 39] [Article Influence: 2.4] [Reference Citation Analysis (40)] |
| 11. | Mack M. Inflammation and fibrosis. Matrix Biol. 2018;68-69:106-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 365] [Article Influence: 52.1] [Reference Citation Analysis (0)] |
| 12. | Parola M, Pinzani M. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues. Mol Aspects Med. 2019;65:37-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 785] [Article Influence: 112.1] [Reference Citation Analysis (0)] |
| 13. | Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012;18:1028-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2470] [Cited by in RCA: 2640] [Article Influence: 203.1] [Reference Citation Analysis (0)] |
| 14. | Banales JM, Huebert RC, Karlsen T, Strazzabosco M, LaRusso NF, Gores GJ. Cholangiocyte pathobiology. Nat Rev Gastroenterol Hepatol. 2019;16:269-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 347] [Article Influence: 57.8] [Reference Citation Analysis (1)] |
| 15. | Bergquist A, Montgomery SM, Bahmanyar S, Olsson R, Danielsson A, Lindgren S, Prytz H, Hultcrantz R, Lööf LA, Sandberg-Gertzén H, Almer S, Askling J, Ehlin A, Ekbom A. Increased risk of primary sclerosing cholangitis and ulcerative colitis in first-degree relatives of patients with primary sclerosing cholangitis. Clin Gastroenterol Hepatol. 2008;6:939-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 126] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 16. | Paziewska A, Habior A, Rogowska A, Zych W, Goryca K, Karczmarski J, Dabrowska M, Ambrozkiewicz F, Walewska-Zielecka B, Krawczyk M, Cichoz-Lach H, Milkiewicz P, Kowalik A, Mucha K, Raczynska J, Musialik J, Boryczka G, Wasilewicz M, Ciecko-Michalska I, Ferenc M, Janiak M, Kanikowska A, Stankiewicz R, Hartleb M, Mach T, Grzymislawski M, Raszeja-Wyszomirska J, Wunsch E, Bobinski T, Mikula M, Ostrowski J. A novel approach to genome-wide association analysis identifies genetic associations with primary biliary cholangitis and primary sclerosing cholangitis in Polish patients. BMC Med Genomics. 2017;10:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Liu JZ, Hov JR, Folseraas T, Ellinghaus E, Rushbrook SM, Doncheva NT, Andreassen OA, Weersma RK, Weismüller TJ, Eksteen B, Invernizzi P, Hirschfield GM, Gotthardt DN, Pares A, Ellinghaus D, Shah T, Juran BD, Milkiewicz P, Rust C, Schramm C, Müller T, Srivastava B, Dalekos G, Nöthen MM, Herms S, Winkelmann J, Mitrovic M, Braun F, Ponsioen CY, Croucher PJ, Sterneck M, Teufel A, Mason AL, Saarela J, Leppa V, Dorfman R, Alvaro D, Floreani A, Onengut-Gumuscu S, Rich SS, Thompson WK, Schork AJ, Næss S, Thomsen I, Mayr G, König IR, Hveem K, Cleynen I, Gutierrez-Achury J, Ricaño-Ponce I, van Heel D, Björnsson E, Sandford RN, Durie PR, Melum E, Vatn MH, Silverberg MS, Duerr RH, Padyukov L, Brand S, Sans M, Annese V, Achkar JP, Boberg KM, Marschall HU, Chazouillères O, Bowlus CL, Wijmenga C, Schrumpf E, Vermeire S, Albrecht M; UK-PSCSC Consortium, Rioux JD, Alexander G, Bergquist A, Cho J, Schreiber S, Manns MP, Färkkilä M, Dale AM, Chapman RW, Lazaridis KN; International PSC Study Group, Franke A, Anderson CA, Karlsen TH; International IBD Genetics Consortium. Dense genotyping of immune-related disease regions identifies nine new risk loci for primary sclerosing cholangitis. Nat Genet. 2013;45:670-675. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 335] [Cited by in RCA: 302] [Article Influence: 25.2] [Reference Citation Analysis (0)] |
| 18. | Karlsen TH, Franke A, Melum E, Kaser A, Hov JR, Balschun T, Lie BA, Bergquist A, Schramm C, Weismüller TJ, Gotthardt D, Rust C, Philipp EE, Fritz T, Henckaerts L, Weersma RK, Stokkers P, Ponsioen CY, Wijmenga C, Sterneck M, Nothnagel M, Hampe J, Teufel A, Runz H, Rosenstiel P, Stiehl A, Vermeire S, Beuers U, Manns MP, Schrumpf E, Boberg KM, Schreiber S. Genome-wide association analysis in primary sclerosing cholangitis. Gastroenterology. 2010;138:1102-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 246] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 19. | Zhang CC, Voitl R, Hippchen T, Weiss KH, Sauer P, Rupp C. Evaluation of two functional CD24 polymorphisms in primary sclerosing cholangitis. Scand J Gastroenterol. 2020;55:581-587. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 20. | Alberts R, de Vries EMG, Goode EC, Jiang X, Sampaziotis F, Rombouts K, Böttcher K, Folseraas T, Weismüller TJ, Mason AL, Wang W, Alexander G, Alvaro D, Bergquist A, Björkström NK, Beuers U, Björnsson E, Boberg KM, Bowlus CL, Bragazzi MC, Carbone M, Chazouillères O, Cheung A, Dalekos G, Eaton J, Eksteen B, Ellinghaus D, Färkkilä M, Festen EAM, Floreani A, Franceschet I, Gotthardt DN, Hirschfield GM, Hoek BV, Holm K, Hohenester S, Hov JR, Imhann F, Invernizzi P, Juran BD, Lenzen H, Lieb W, Liu JZ, Marschall HU, Marzioni M, Melum E, Milkiewicz P, Müller T, Pares A, Rupp C, Rust C, Sandford RN, Schramm C, Schreiber S, Schrumpf E, Silverberg MS, Srivastava B, Sterneck M, Teufel A, Vallier L, Verheij J, Vila AV, Vries B, Zachou K; International PSC Study Group, The UK PSC Consortium, Chapman RW, Manns MP, Pinzani M, Rushbrook SM, Lazaridis KN, Franke A, Anderson CA, Karlsen TH, Ponsioen CY, Weersma RK. Genetic association analysis identifies variants associated with disease progression in primary sclerosing cholangitis. Gut. 2018;67:1517-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 21. | Laish I, Katz H, Stein A, Liberman M, Naftali T, Kitay-Cohen Y, Biron-Shental T, Konikoff FM, Amiel A. Telomere dysfunction in peripheral blood lymphocytes from patients with primary sclerosing cholangitis and inflammatory bowel disease. Dig Liver Dis. 2015;47:790-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Jiang X, Karlsen TH. Genetics of primary sclerosing cholangitis and pathophysiological implications. Nat Rev Gastroenterol Hepatol. 2017;14:279-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 103] [Article Influence: 12.9] [Reference Citation Analysis (1)] |
| 23. | Allegretti JR, Kassam Z, Carrellas M, Mullish BH, Marchesi JR, Pechlivanis A, Smith M, Gerardin Y, Timberlake S, Pratt DS, Korzenik JR. Fecal Microbiota Transplantation in Patients With Primary Sclerosing Cholangitis: A Pilot Clinical Trial. Am J Gastroenterol. 2019;114:1071-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 169] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 24. | Torres J, Bao X, Goel A, Colombel JF, Pekow J, Jabri B, Williams KM, Castillo A, Odin JA, Meckel K, Fasihuddin F, Peter I, Itzkowitz S, Hu J. The features of mucosa-associated microbiota in primary sclerosing cholangitis. Aliment Pharmacol Ther. 2016;43:790-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 119] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 25. | Dyson JK, Blain A, Foster Shirley MD, Hudson M, Rushton S, Jeffreys Jones DE. Geo-epidemiology and environmental co-variate mapping of primary biliary cholangitis and primary sclerosing cholangitis. JHEP Rep. 2021;3:100202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 26. | Tenca A, Färkkilä M, Jalanko H, Vapalahti K, Arola J, Jaakkola T, Penagini R, Vapalahti O, Kolho KL. Environmental Risk Factors of Pediatric-Onset Primary Sclerosing Cholangitis and Autoimmune Hepatitis. J Pediatr Gastroenterol Nutr. 2016;62:437-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 27. | Andersen IM, Tengesdal G, Lie BA, Boberg KM, Karlsen TH, Hov JR. Effects of coffee consumption, smoking, and hormones on risk for primary sclerosing cholangitis. Clin Gastroenterol Hepatol. 2014;12:1019-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 28. | Singh S, Talwalkar JA. Primary sclerosing cholangitis: diagnosis, prognosis, and management. Clin Gastroenterol Hepatol. 2013;11:898-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 29. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of cholestatic liver diseases. J Hepatol. 2009;51:237-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1382] [Cited by in RCA: 1204] [Article Influence: 75.3] [Reference Citation Analysis (1)] |
| 30. | Lindor KD, Kowdley KV, Harrison ME; American College of Gastroenterology. ACG Clinical Guideline: Primary Sclerosing Cholangitis. Am J Gastroenterol. 2015;110:646-59; quiz 660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 338] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 31. | Chapman R, Fevery J, Kalloo A, Nagorney DM, Boberg KM, Shneider B, Gores GJ; American Association for the Study of Liver Diseases. Diagnosis and management of primary sclerosing cholangitis. Hepatology. 2010;51:660-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 835] [Article Influence: 55.7] [Reference Citation Analysis (0)] |
| 32. | Rabiee A, Silveira MG. Primary sclerosing cholangitis. Transl Gastroenterol Hepatol. 2021;6:29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 33. | Peeling Back The Layers of Pediatric Primary Sclerosing Cholangitis AASLD. 2022. Available from: https://www.aasld.org/Liver-fellow-network/core-series/clinical-pearls/peeling-back-layers-pediatric-primary-sclerosing. |
| 34. | Deneau MR, Mack C, Perito ER, Ricciuto A, Valentino PL, Amin M, Amir AZ, Aumar M, Auth M, Broderick A, DiGuglielmo M, Draijer LG, Tavares Fagundes ED, El-Matary W, Ferrari F, Furuya KN, Gupta N, Hochberg JT, Homan M, Horslen S, Iorio R, Jensen MK, Jonas MM, Kamath BM, Kerkar N, Kim KM, Kolho KL, Koot BGP, Laborda TJ, Lee CK, Loomes KM, Martinez M, Miethke A, Miloh T, Mogul D, Mohammad S, Mohan P, Moroz S, Ovchinsky N, Palle S, Papadopoulou A, Rao G, Rodrigues Ferreira A, Sathya P, Schwarz KB, Shah U, Shteyer E, Singh R, Smolka V, Soufi N, Tanaka A, Varier R, Vitola B, Woynarowski M, Zerofsky M, Zizzo A, Guthery SL. The Sclerosing Cholangitis Outcomes in Pediatrics (SCOPE) Index: A Prognostic Tool for Children. Hepatology. 2021;73:1074-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 35. | Boberg KM, Chapman RW, Hirschfield GM, Lohse AW, Manns MP, Schrumpf E; International Autoimmune Hepatitis Group. Overlap syndromes: the International Autoimmune Hepatitis Group (IAIHG) position statement on a controversial issue. J Hepatol. 2011;54:374-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 347] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 36. | Dyson JK, Beuers U, Jones DEJ, Lohse AW, Hudson M. Primary sclerosing cholangitis. Lancet. 2018;391:2547-2559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 287] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 37. | Conrad K, Roggenbuck D, Laass MW. Diagnosis and classification of ulcerative colitis. Autoimmun Rev. 2014;13:463-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 255] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 38. | Hov JR, Boberg KM, Karlsen TH. Autoantibodies in primary sclerosing cholangitis. World J Gastroenterol. 2008;14:3781-3791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 109] [Cited by in RCA: 117] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 39. | Khoshpouri P, Habibabadi RR, Hazhirkarzar B, Ameli S, Ghadimi M, Ghasabeh MA, Menias CO, Kim A, Li Z, Kamel IR. Imaging Features of Primary Sclerosing Cholangitis: From Diagnosis to Liver Transplant Follow-up. Radiographics. 2019;39:1938-1964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 40. | Ito K, Mitchell DG, Outwater EK, Blasbalg R. Primary sclerosing cholangitis: MR imaging features. AJR Am J Roentgenol. 1999;172:1527-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 64] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 41. | Prokopič M, Beuers U. Management of primary sclerosing cholangitis and its complications: an algorithmic approach. Hepatol Int. 2021;15:6-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 42. | Schramm C, Eaton J, Ringe KI, Venkatesh S, Yamamura J; MRI working group of the IPSCSG. Recommendations on the use of magnetic resonance imaging in PSC-A position statement from the International PSC Study Group. Hepatology. 2017;66:1675-1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 43. | Angulo P, Pearce DH, Johnson CD, Henry JJ, LaRusso NF, Petersen BT, Lindor KD. Magnetic resonance cholangiography in patients with biliary disease: its role in primary sclerosing cholangitis. J Hepatol. 2000;33:520-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 40] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 44. | Berstad AE, Aabakken L, Smith HJ, Aasen S, Boberg KM, Schrumpf E. Diagnostic accuracy of magnetic resonance and endoscopic retrograde cholangiography in primary sclerosing cholangitis. Clin Gastroenterol Hepatol. 2006;4:514-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 100] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 45. | Dave M, Elmunzer BJ, Dwamena BA, Higgins PD. Primary sclerosing cholangitis: meta-analysis of diagnostic performance of MR cholangiopancreatography. Radiology. 2010;256:387-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 156] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 46. | Talwalkar JA, Angulo P, Johnson CD, Petersen BT, Lindor KD. Cost-minimization analysis of MRC versus ERCP for the diagnosis of primary sclerosing cholangitis. Hepatology. 2004;40:39-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 64] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 47. | Ludwig J, Dickson ER, McDonald GS. Staging of chronic nonsuppurative destructive cholangitis (syndrome of primary biliary cirrhosis). Virchows Arch A Pathol Anat Histol. 1978;379:103-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 586] [Cited by in RCA: 528] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 48. | Weismüller TJ, Trivedi PJ, Bergquist A, Imam M, Lenzen H, Ponsioen CY, Holm K, Gotthardt D, Färkkilä MA, Marschall HU, Thorburn D, Weersma RK, Fevery J, Mueller T, Chazouillères O, Schulze K, Lazaridis KN, Almer S, Pereira SP, Levy C, Mason A, Naess S, Bowlus CL, Floreani A, Halilbasic E, Yimam KK, Milkiewicz P, Beuers U, Huynh DK, Pares A, Manser CN, Dalekos GN, Eksteen B, Invernizzi P, Berg CP, Kirchner GI, Sarrazin C, Zimmer V, Fabris L, Braun F, Marzioni M, Juran BD, Said K, Rupp C, Jokelainen K, Benito de Valle M, Saffioti F, Cheung A, Trauner M, Schramm C, Chapman RW, Karlsen TH, Schrumpf E, Strassburg CP, Manns MP, Lindor KD, Hirschfield GM, Hansen BE, Boberg KM; International PSC Study Group. Patient Age, Sex, and Inflammatory Bowel Disease Phenotype Associate With Course of Primary Sclerosing Cholangitis. Gastroenterology. 2017;152:1975-1984.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 364] [Article Influence: 45.5] [Reference Citation Analysis (1)] |
| 49. | Kim WR, Therneau TM, Wiesner RH, Poterucha JJ, Benson JT, Malinchoc M, LaRusso NF, Lindor KD, Dickson ER. A revised natural history model for primary sclerosing cholangitis. Mayo Clin Proc. 2000;75:688-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 50. | Ponsioen CY, Vrouenraets SM, Prawirodirdjo W, Rajaram R, Rauws EA, Mulder CJ, Reitsma JB, Heisterkamp SH, Tytgat GN. Natural history of primary sclerosing cholangitis and prognostic value of cholangiography in a Dutch population. Gut. 2002;51:562-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 198] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 51. | Schmeltzer PA, Russo MW. Systematic Review of Prognostic Models Compared to the Mayo Risk Score for Primary Sclerosing Cholangitis. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 52. | Goode EC, Clark AB, Mells GF, Srivastava B, Spiess K, Gelson WTH, Trivedi PJ, Lynch KD, Castren E, Vesterhus MN, Karlsen TH, Ji SG, Anderson CA, Thorburn D, Hudson M, Heneghan MA, Aldersley MA, Bathgate A, Sandford RN, Alexander GJ, Chapman RW, Walmsley M; UK-PSC Consortium, Hirschfield GM, Rushbrook SM. Factors Associated With Outcomes of Patients With Primary Sclerosing Cholangitis and Development and Validation of a Risk Scoring System. Hepatology. 2019;69:2120-2135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 67] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 53. | Eaton JE, Vesterhus M, McCauley BM, Atkinson EJ, Schlicht EM, Juran BD, Gossard AA, LaRusso NF, Gores GJ, Karlsen TH, Lazaridis KN. Primary Sclerosing Cholangitis Risk Estimate Tool (PREsTo) Predicts Outcomes of the Disease: A Derivation and Validation Study Using Machine Learning. Hepatology. 2020;71:214-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 54. | Wiesner RH, Grambsch PM, Dickson ER, Ludwig J, MacCarty RL, Hunter EB, Fleming TR, Fisher LD, Beaver SJ, LaRusso NF. Primary sclerosing cholangitis: natural history, prognostic factors and survival analysis. Hepatology. 1989;10:430-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 426] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 55. | de Vries EM, Wang J, Williamson KD, Leeflang MM, Boonstra K, Weersma RK, Beuers U, Chapman RW, Geskus RB, Ponsioen CY. A novel prognostic model for transplant-free survival in primary sclerosing cholangitis. Gut. 2018;67:1864-1869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 56. | Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D'Amico G, Dickson ER, Kim WR. A model to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3462] [Cited by in RCA: 3678] [Article Influence: 153.3] [Reference Citation Analysis (0)] |
| 57. | Child CG, Turcotte J. Surgery and portal hypertension. Clinics in Gastroenterology. 1979;8:525-541. [DOI] [Full Text] |
| 58. | Wunsch E, Trottier J, Milkiewicz M, Raszeja-Wyszomirska J, Hirschfield GM, Barbier O, Milkiewicz P. Prospective evaluation of ursodeoxycholic acid withdrawal in patients with primary sclerosing cholangitis. Hepatology. 2014;60:931-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 94] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 59. | Paumgartner G, Beuers U. Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited. Hepatology. 2002;36:525-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 477] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 60. | Beuers U. Drug insight: Mechanisms and sites of action of ursodeoxycholic acid in cholestasis. Nat Clin Pract Gastroenterol Hepatol. 2006;3:318-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 271] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 61. | Othman MO, Dunkelberg J, Roy PK. Urosdeoxycholic acid in primary sclerosing cholangitis: a meta-analysis and systematic review. Arab J Gastroenterol. 2012;13:103-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 62. | Poropat G, Giljaca V, Stimac D, Gluud C. Bile acids for primary sclerosing cholangitis. Cochrane Database Syst Rev. 2011;2011:CD003626. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 63. | Triantos CK, Koukias NM, Nikolopoulou VN, Burroughs AK. Meta-analysis: ursodeoxycholic acid for primary sclerosing cholangitis. Aliment Pharmacol Ther. 2011;34:901-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 64. | Hilscher M, Enders FB, Carey EJ, Lindor KD, Tabibian JH. Alkaline phosphatase normalization is a biomarker of improved survival in primary sclerosing cholangitis. Ann Hepatol. 2016;15:246-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 65. | Al Mamari S, Djordjevic J, Halliday JS, Chapman RW. Improvement of serum alkaline phosphatase to <1.5 upper limit of normal predicts better outcome and reduced risk of cholangiocarcinoma in primary sclerosing cholangitis. J Hepatol. 2013;58:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 146] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 66. | Stanich PP, Björnsson E, Gossard AA, Enders F, Jorgensen R, Lindor KD. Alkaline phosphatase normalization is associated with better prognosis in primary sclerosing cholangitis. Dig Liver Dis. 2011;43:309-313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 120] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 67. | Black DD, Mack C, Kerkar N, Miloh T, Sundaram SS, Anand R, Gupta A, Alonso E, Arnon R, Bulut P, Karpen S, Lin CH, Rosenthal P, Ryan M, Squires RH, Valentino P, Elsea SH, Shneider BL. A Prospective Trial of Withdrawal and Reinstitution of Ursodeoxycholic Acid in Pediatric Primary Sclerosing Cholangitis. Hepatol Commun. 2019;3:1482-1495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 68. | Bowlus CL, Arrivé L, Bergquist A, Deneau M, Forman L, Ilyas SI, Lunsford KE, Martinez M, Sapisochin G, Shroff R, Tabibian JH, Assis DN. AASLD practice guidance on primary sclerosing cholangitis and cholangiocarcinoma. Hepatology. 2023;77:659-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 155] [Article Influence: 77.5] [Reference Citation Analysis (0)] |
| 69. | Peng X, Luo X, Hou JY, Wu SY, Li LZ, Zheng MH, Wang LY. Immunosuppressive Agents for the Treatment of Primary Sclerosing Cholangitis: A Systematic Review and Meta-Analysis. Dig Dis. 2017;35:478-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 70. | Liu X, Wang H, Liu X, Bridle K, Crawford D, Liang X. Efficacy and safety of immune-modulating therapy for primary sclerosing cholangitis: A systematic review and meta-analysis. Pharmacol Ther. 2022;237:108163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 71. | Trauner M, Gulamhusein A, Hameed B, Caldwell S, Shiffman ML, Landis C, Eksteen B, Agarwal K, Muir A, Rushbrook S, Lu X, Xu J, Chuang JC, Billin AN, Li G, Chung C, Subramanian GM, Myers RP, Bowlus CL, Kowdley KV. The Nonsteroidal Farnesoid X Receptor Agonist Cilofexor (GS-9674) Improves Markers of Cholestasis and Liver Injury in Patients With Primary Sclerosing Cholangitis. Hepatology. 2019;70:788-801. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 217] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 72. | Fickert P, Hirschfield GM, Denk G, Marschall HU, Altorjay I, Färkkilä M, Schramm C, Spengler U, Chapman R, Bergquist A, Schrumpf E, Nevens F, Trivedi P, Reiter FP, Tornai I, Halilbasic E, Greinwald R, Pröls M, Manns MP, Trauner M; European PSC norUDCA Study Group. norUrsodeoxycholic acid improves cholestasis in primary sclerosing cholangitis. J Hepatol. 2017;67:549-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 203] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 73. | Saffioti F, Gurusamy KS, Hawkins N, Toon CD, Tsochatzis E, Davidson BR, Thorburn D. Pharmacological interventions for primary sclerosing cholangitis: an attempted network meta-analysis. Cochrane Database Syst Rev. 2017;3:CD011343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 74. | Shi J, Li Z, Zeng X, Lin Y, Xie WF. Ursodeoxycholic acid in primary sclerosing cholangitis: meta-analysis of randomized controlled trials. Hepatol Res. 2009;39:865-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 75. | Shah A, Crawford D, Burger D, Martin N, Walker M, Talley NJ, Tallis C, Jones M, Stuart K, Keely S, Lewindon P, Macdonald GA, Morrison M, Holtmann GJ. Effects of Antibiotic Therapy in Primary Sclerosing Cholangitis with and without Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Semin Liver Dis. 2019;39:432-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 76. | Vleggaar FP, Monkelbaan JF, van Erpecum KJ. Probiotics in primary sclerosing cholangitis: a randomized placebo-controlled crossover pilot study. Eur J Gastroenterol Hepatol. 2008;20:688-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 80] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 77. | May GR, Bender CE, LaRusso NF, Wiesner RH. Nonoperative dilatation of dominant strictures in primary sclerosing cholangitis. AJR Am J Roentgenol. 1985;145:1061-1064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 54] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 78. | Stiehl A, Rudolph G, Klöters-Plachky P, Sauer P, Walker S. Development of dominant bile duct stenoses in patients with primary sclerosing cholangitis treated with ursodeoxycholic acid: outcome after endoscopic treatment. J Hepatol. 2002;36:151-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 180] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 79. | Björnsson E, Lindqvist-Ottosson J, Asztely M, Olsson R. Dominant strictures in patients with primary sclerosing cholangitis. Am J Gastroenterol. 2004;99:502-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 122] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 80. | Bangarulingam SY, Gossard AA, Petersen BT, Ott BJ, Lindor KD. Complications of endoscopic retrograde cholangiopancreatography in primary sclerosing cholangitis. Am J Gastroenterol. 2009;104:855-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 98] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 81. | Navaneethan U, Jegadeesan R, Nayak S, Lourdusamy V, Sanaka MR, Vargo JJ, Parsi MA. ERCP-related adverse events in patients with primary sclerosing cholangitis. Gastrointest Endosc. 2015;81:410-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 82. | Ismail S, Kylänpää L, Mustonen H, Halttunen J, Lindström O, Jokelainen K, Udd M, Färkkilä M. Risk factors for complications of ERCP in primary sclerosing cholangitis. Endoscopy. 2012;44:1133-1138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 83. | von Seth E, Arnelo U, Enochsson L, Bergquist A. Primary sclerosing cholangitis increases the risk for pancreatitis after endoscopic retrograde cholangiopancreatography. Liver Int. 2015;35:254-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 84. | Gustafsson A, Enochsson L, Tingstedt B, Olsson G. Antibiotic prophylaxis and its effect on postprocedural adverse events in endoscopic retrograde cholangiopancreatography for primary sclerosing cholangitis. JGH Open. 2023;7:24-29. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 85. | Martin P, DiMartini A, Feng S, Brown R Jr, Fallon M. Evaluation for liver transplantation in adults: 2013 practice guideline by the American Association for the Study of Liver Diseases and the American Society of Transplantation. Hepatology. 2014;59:1144-1165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 702] [Article Influence: 63.8] [Reference Citation Analysis (1)] |
| 86. | Müllhaupt B, Dimitroulis D, Gerlach JT, Clavien PA. Hot topics in liver transplantation: organ allocation--extended criteria donor--living donor liver transplantation. J Hepatol. 2008;48 Suppl 1:S58-S67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 87. | Akdur A, Ayvazoğlu Soy HE, Karakaya E, Yıldırım S, Moray G, Haberal M. Living and Deceased Liver Donation Criteria of Baskent University. Exp Clin Transplant. 2022;20:80-87. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 88. | Sandhu L, Sandroussi C, Guba M, Selzner M, Ghanekar A, Cattral MS, McGilvray ID, Levy G, Greig PD, Renner EL, Grant DR. Living donor liver transplantation vs deceased donor liver transplantation for hepatocellular carcinoma: Comparable survival and recurrence. Liver Transpl. 2012;18:315-322. [RCA] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 89. | Bjøro K, Schrumpf E. Liver transplantation for primary sclerosing cholangitis. J Hepatol. 2004;40:570-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 90. | Update National Liver Review Board Guidance Documents and Policy Clarification-OPTN. Available from: https://optn.transplant.hrsa.gov/policies-bylaws/public-comment/update-national-liver-review-board-guidance-documents-and-policy-clarification/. |
| 91. | Khungar V, Goldberg DS. Liver Transplantation for Cholestatic Liver Diseases in Adults. Clin Liver Dis. 2016;20:191-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 92. | Liver Transplant Evaluation-Penn Medicine. Available from: https://www.pennmedicine.org/for-patients-and-visitors/find-a-program-or-service/transplant-institute/Liver-transplant/treatments-and-procedures/Liver-evaluation. |
| 93. | Grover S, Sarkar S. Liver transplant-psychiatric and psychosocial aspects. J Clin Exp Hepatol. 2012;2:382-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 94. | Nghiem DM, Gomez J, Gloston GF, Torres DS, Marek RJ. Psychological Assessment Instruments for Use in Liver and Kidney Transplant Evaluations: Scarcity of Evidence and Recommendations. J Pers Assess. 2020;102:183-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 95. | The Lancet Gastroenterology Hepatology. Liver transplantation in the USA: ethical issues. Lancet Gastroenterol Hepatol. 2020;5:1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 96. | Sells RA. Ethical issues in transplantation. Baillieres Clin Gastroenterol. 1994;8:465-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 97. | Berg CL. Being Well Informed Is Critical to Informed Consent for Living Liver Donors. Transplantation. 2022;106:1103-1104. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 98. | Goldberg D, Bittermann T, Makar G. Lack of standardization in exception points for patients with primary sclerosing cholangitis and bacterial cholangitis. Am J Transplant. 2012;12:1603-1609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 99. | Mathur AK, Schaubel DE, Gong Q, Guidinger MK, Merion RM. Racial and ethnic disparities in access to liver transplantation. Liver Transpl. 2010;16:1033-1040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 100. | Andersen IM, Fosby B, Boberg KM, Clausen OP, Jebsen P, Melum E, Line PD, Foss A, Schrumpf E, Karlsen TH. Indications and Outcomes in Liver Transplantation in Patients With Primary Sclerosing Cholangitis in Norway. Transplant Direct. 2015;1:e39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 101. | Berenguer M, Di Maira T, Baumann U, Mirza DF, Heneghan MA, Klempnauer JL, Bennet W, Ericzon BG, Line PD, Lodge PA, Zieniewicz K, Watson CJE, Metselaar HJ, Adam R, Karam V, Aguilera V; all the other contributing centers (www. eltr.org) and the European Liver and Intestine Transplant Association (ELITA). Characteristics, Trends, and Outcomes of Liver Transplantation for Primary Sclerosing Cholangitis in Female Versus Male Patients: An Analysis From the European Liver Transplant Registry. Transplantation. 2021;105:2255-2262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 102. | Ziogas IA, Alexopoulos SP, Matsuoka LK, Geevarghese SK, Gorden LD, Karp SJ, Perkins JD, Montenovo MI. Living vs deceased donor liver transplantation in cholestatic liver disease: An analysis of the OPTN database. Clin Transplant. 2020;34:e14031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 103. | Jadaun SS, Mehtani R, Hasnain A, Bhatia S, Moond V, Kumar M, Kuhad V, Singh S, Agarwal S, Gupta S, Saigal S. Good outcomes of living donor liver transplant in primary sclerosing cholangitis: an experience from North India. Hepatol Int. 2023;17:499-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 104. | Emek E, Serin A, Sahin T, Yazici P, Yuzer Y, Tokat Y, Bozkurt B. Experience in Liver Transplantation Due to Primary Sclerosing Cholangitis: A Single Center Experience. Transplant Proc. 2019;51:2439-2441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 105. | Martin EF, Levy C. Timing, Management, and Outcomes of Liver Transplantation in Primary Sclerosing Cholangitis. Semin Liver Dis. 2017;37:305-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 106. | Sharma S, Gurakar A, Jabbour N. Biliary strictures following liver transplantation: past, present and preventive strategies. Liver Transpl. 2008;14:759-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 275] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 107. | Verdonk RC, Buis CI, van der Jagt EJ, Gouw AS, Limburg AJ, Slooff MJ, Kleibeuker JH, Porte RJ, Haagsma EB. Nonanastomotic biliary strictures after liver transplantation, part 2: Management, outcome, and risk factors for disease progression. Liver Transpl. 2007;13:725-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 108. | Fosby B, Karlsen TH, Melum E. Recurrence and rejection in liver transplantation for primary sclerosing cholangitis. World J Gastroenterol. 2012;18:1-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 103] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 109. | Henson JB, Patel YA, King LY, Zheng J, Chow SC, Muir AJ. Outcomes of liver retransplantation in patients with primary sclerosing cholangitis. Liver Transpl. 2017;23:769-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 110. | Aberg F, Höckerstedt K, Roine RP, Sintonen H, Isoniemi H. Influence of liver-disease etiology on long-term quality of life and employment after liver transplantation. Clin Transplant. 2012;26:729-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 111. | Ruppert K, Kuo S, DiMartini A, Balan V. In a 12-year study, sustainability of quality of life benefits after liver transplantation varies with pretransplantation diagnosis. Gastroenterology. 2010;139:1619-1629, 1629.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 112. | Kizilisik AT, Grewal HP, Shokouh-Amiri MH, Vera SR, Hathaway DK, Gaber AO. Impact of long-term immunosuppressive therapy on psychosocial and physical well being in liver transplant recipients. Prog Transplant. 2003;13:278-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 113. | Gavriilidis P, Edwin B, Pelanis E, Hidalgo E, de'Angelis N, Memeo R, Aldrighetti L, Sutcliffe RP. Navigated liver surgery: State of the art and future perspectives. Hepatobiliary Pancreat Dis Int. 2022;21:226-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 114. | Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32:773-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3943] [Cited by in RCA: 3751] [Article Influence: 375.1] [Reference Citation Analysis (0)] |
| 115. | Thomas J, Patel S, Troop L, Guru R, Faist N, Bellott BJ, Esterlen BA. 3D Printed Model of Extrahepatic Biliary Ducts for Biliary Stent Testing. Materials (Basel). 2020;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 116. | Vesterhus M, Karlsen TH. Emerging therapies in primary sclerosing cholangitis: pathophysiological basis and clinical opportunities. J Gastroenterol. 2020;55:588-614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 117. | He C, Yang Y, Zheng K, Chen Y, Liu S, Li Y, Han Q, Zhao RC, Wang L, Zhang F. Mesenchymal stem cell-based treatment in autoimmune liver diseases: underlying roles, advantages and challenges. Ther Adv Chronic Dis. 2021;12:2040622321993442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 118. | Jalan-Sakrikar N, De Assuncao TM, Navarro-Corcuera A, Hamdan FH, Loarca L, Kirkeby LA, Resch ZT, O'Hara SP, Juran BD, Lazaridis KN, Rosen CB, Heimbach JK, Taner T, Shah VH, LaRusso NF, Huebert RC. Induced Pluripotent Stem Cells From Subjects With Primary Sclerosing Cholangitis Develop a Senescence Phenotype Following Biliary Differentiation. Hepatol Commun. 2022;6:345-360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 119. | Somers EB, O'Shannessy DJ. Folate receptor alpha, mesothelin and megakaryocyte potentiating factor as potential serum markers of chronic kidney disease. Biomark Insights. 2014;9:29-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |