Peer-review started: April 30, 2022
First decision: May 29, 2022
Revised: July 2, 2022
Accepted: December 21, 2022
Article in press: December 21, 2022
Published online: January 27, 2023
Processing time: 260 Days and 8.7 Hours
Liver cancer is a severe concern for public health officials since the clinical cases are increasing each year, with an estimated 5-year survival rate of 30%–35% after diagnosis. Hepatocellular carcinoma (HCC) constitutes a significant subtype of liver cancer (approximate75%) and is considered primary liver cancer. Treatment for liver cancer mainly depends on the stage of its progression, where surgery including, hepatectomy and liver transplantation, and ablation and radiotherapy are the prime choice. For advanced liver cancer, various drugs and immunotherapy are used as first-line treatment, whereas second-line treatment includes chemotherapeutic drugs from natural and synthetic origins. Sorafenib and lenvatinib are first-line therapies, while regorafenib and ramucirumab are second-line therapy. Various metabolic and signaling pathways such as Notch, JAK/ STAT, Hippo, TGF-β, and Wnt have played a critical role during HCC pro
Core Tip: Liver cancer is a serious public health concern and its therapy is stage-dependent. Approximately 75% of all liver cancers are hepatocellular carcinoma, which is regarded as primary liver cancer. First and second-line therapies are used to manage the disease but they have their own limitations in terms of toxicity and other severe side effects. Natural products are the prime choice for the future treatment of liver cancer. With advancement in the knowledge about the molecular mechanism of the disease, newer strategies having fewer side effects and greater effectiveness are needed.
- Citation: Singh AK, Singh SV, Kumar R, Kumar S, Senapati S, Pandey AK. Current therapeutic modalities and chemopreventive role of natural products in liver cancer: Progress and promise . World J Hepatol 2023; 15(1): 1-18
- URL: https://www.wjgnet.com/1948-5182/full/v15/i1/1.htm
- DOI: https://dx.doi.org/10.4254/wjh.v15.i1.1
The liver is the human body’s largest solid organ, and has a pivotal role in removing various blood toxins and maintaining bioenergetics and cellular metabolism[1,2]. The liver is structured into four lobes that are made up of multiple lobules, each having a flowing duct toward the common hepatic duct, responsible for bile excretion[3]. Changes in lifestyle patterns and excessive use of medicines, alcohol and intake of various unhygienic supplements, impose further stress and finally damage the liver[4,5]. Excess alcohol and viral infections causing hepatitis are critical factors for liver cancer[6,7]. Each year approximately 0.8 million new clinical cases of liver cancer are diagnosed. From this disease, approximately 830180 people died worldwide in 2020 alone, and this figure seems to be increasing daily, according to World Health Organization surveillance reports[8]. Among various liver cancer types, hepatocellular carcinoma (HCC) is the most common type and accounts for approximately 85% of primary liver cancer cases and often occurs in people with chronic liver diseases. It is the most common and second leading cause of cancer-related deaths in Asian and sub-Saharan African countries. It is the sixth most common in western countries due to escalating hepatitis C burden along with nonalcoholic steatohepatitis and obesity[9,10]. In patients with a preclinical history of chronic liver diseases and cirrhosis, the development of HCC is a complex process, including inflammatory damage leading to hepatocyte necrosis, regeneration and fibrotic deposition[11,12].
In recent years, multiple efforts have been made to manage HCC using various chemotherapeutic approaches, of which, targeted tyrosine kinase inhibitors, immunotherapy and anticancer combination therapies are the main ones[13]. However, chemoresistance and initiation and progression of tumors mainly reprogram cellular metabolism, particularly during HCC development[14]. These metabolic alterations are key factors promoting tumor growth, proliferation and requirements of cancer cells, such as increased energy production, macromolecular biosynthesis and maintenance of redox balance. The liver is the main site for contact with a variety of orally ingested therapeutic drugs, alcohol and other xenobiotics after intestinal absorption and this organ is susceptible to various chemicals[15,16]. These chemicals cause serious complications such as acute and chronic hepatitis, granulomatous hepatitis, cholestasis with or without hepatitis, tumors and vascular disorders[17].
Among various factors responsible for HCC, viral hepatitis and resulting cirrhosis cover a significant proportion of clinical cases. Various viral infections cause upregulation of hexosamine and membrane lipid biosynthesis, by modulating glutamine-fructose-6-phosphate transaminase (GFAT)1 and choline kinase A expression[18,19]. These findings have been further validated with some results where GFAT1 is upregulated in HCC patients and its overexpression enhances tumorigenic phenotypes, as observed during in vitro studies[20]. Hepatitis B virus (HBV) also alters lipid metabolism, where viral proteins are known for inducing lipid accumulation via the upregulation of sterol regulatory element-binding protein (SREBP)1, peroxisome proliferator-activated receptor (PPAR)γ, as well as lipogenic and adipogenic enzymes, which are also reported during HCC progression[21,22]. HCC cells infected with hepatitis C virus (HCV) are also known to exhibit altered glycolysis and gluconeogenesis along with the activation of lipid-metabolism transcription factor PPARγ in human hepatocytes, similar to HCV infection[23]. Some early findings of HCC include the CD36 gene role in free fatty acid uptake and its increased expression during chronic alcohol consumption, thus modulating lipid metabolism, upregulation of SREBP1c and PPARγ, and downregulation of sirtuin 1, collectively leading to impaired fatty acid oxidation[24,25]. Nonalcoholic fatty liver disease also manifests alterations in mitochondrial and other metabolic pathways reminiscent of HCC metabolism. As mentioned earlier, modifications in the processes are primarily analogous to many contexts observed in HCC. However, there is still a need for a better understanding of various underlying mechanisms governing metabolic changes during HCC.
Surgical resection is the primary choice for treating HCC, where recurrence and metastasis mostly occur, thus limiting proper treatment for HCC. Due to the minimal number of drugs available for the treatment of HCC, chemotherapy has remained insufficient for successful management of HCC[26,27]. Although the first-line and second-line therapies can increase life span for several months, these have serious side effects and resistance problems[28]. Since natural products are promising and cost-effective against various illnesses, it seems reasonable to focus on HCC management using natural products where anticancer drugs are limited[2,4,29]. This review focuses on HCC and its associated pathways, descriptive illustration of various natural products, along with their anticancer properties. This review provides information to investigate further regarding liver cancer, signaling pathways, therapeutic targets, current treatment strategies and the chemopreventive role of various natural products.
The liver is highly exposed to foreign materials, and their continuous processing is required for the body’s normal functioning. Alcohol consumption imposes stress on hepatic cells. This condition worsens when combined with a genetic defect in hepatic cells. These factors, either alone or in combination, alter the molecular signaling events responsible for controlled cellular proliferation and differentiation, ultimately leading to hepatic cancer[23]. Targeting these signaling pathways by therapeutic molecules is an important strategy. Inhibition of hepatic cancer-associated signaling pathways ameliorates cancer hallmarks such as increased cellular proliferation, reduced apoptosis, migration, and angiogenesis[24,25]. This section discusses recent advances in molecular signaling pathways associated with the different stages (initiation and development) of liver cancer and their therapeutic target potential. Critical signaling pathways related to HCC include transforming growth factor (TGF)-β, Wnt/B-catenin, Hedgehog, Notch, epidermal growth factor (EGF), hepatocyte growth factor (HGF), vascular endothelial growth factor (VEGF), Janus kinase (JAK)/STAT3, and Hippo signaling pathways[30].
The human liver possesses regeneration potential, and highly controlled molecular mechanisms regulate its repair and regeneration. The Notch signaling pathway is involved in the repair and regeneration of the liver, but its malfunction (loss or gain of function) is associated with hepatic diseases, including cancer[31]. Notch1 upregulation has been found in most hepatic cancer patients. Molecular profiling studies have revealed Notch target genes such as Hes1 and Hey1 in hepatic cancer patients with increased cellular proliferation, reduced apoptosis, increased metastasis and angiogenesis in hepatic cancer cells[32,33]. Notch signaling crosstalk with other molecular pathways (such as hypoxia signaling) is associated with hepatic cancer[34]. Cytokine signaling pathways such as JAK/STAT (Janus kinase/signal transducer and activator of transcription) have been involved in viral escape in virus-induced HCC[35]. Viral invasion and liver injury stimulate hepatocytes and Kupffer cells to secrete sonic hedgehog (SHH) ligands. The ligand triggers Smoothened (Smo) receptor by interacting with the Patched protein, which initiates the Hippo signaling pathway in hepatic cancer cells. The activation of the Hippo signaling pathway results in increased transcription of effector genes (cyclin D, c-Myc, MMP, and CD133, etc.), affecting cell proliferation, invasion, and stemness properties of hepatic cancer cells[36-38].
The TGF-β signaling pathway promotes epithelial to mesenchymal transition, angiogenesis, macrophage maturation, cancer stem cell population, and cellular proliferation in HCC. Crosstalk of TGF-β with other pathways (EGF, Wnt, SHH, etc.) is associated with liver cancer[39,40]. Increased Wnt ligand expression and/or mutations in the molecular components of the Wnt signaling pathway results in hyperactivation of the pathway in hepatic cancer cells. The binding of Wnt ligand to its receptor, followed by production of free β-catenin and its translocation to the nucleus, activates transcription of target genes (CD44, EpCAM, cyclin D1, c-Myc, etc.)[41]. Transcription of target genes ultimately increases cellular proliferation, stemness, angiogenesis and migration potential in hepatic cancer cells. Wnt signaling response to a hypoxic condition in the tumor microenvironment increases stemness potential in hepatic tumor cells. Like other solid tumors, liver cancer cells secrete various growth factors such as platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), HGF, and VEGF. These factors in turn induce angiogenesis to ensure the appropriate supply of nutrients and oxygen. Liver cancer is the result of chronic liver cirrhosis, which ultimately takes the shape of advanced HCC. Available clinical data show that protein mutation increases as the disease progresses from the initiation stage to highly advanced cancer[42]. Mutation in TERT gene (catalytic subunit of telomerase reverse transcriptase) is associated with increased cellular proliferation in liver cancer cells. Clinical data revealed that TERT promoter mutation increased up to 10 times in HCC cells compared with low-grade dysplastic nodules[43]. It indicates that mutation plays an important role in the initiation and progression of the pathological stage of HCC. Besides, other mutations are only involved at the later stage of the disease progression and produce more genetic diversified subtypes[44].
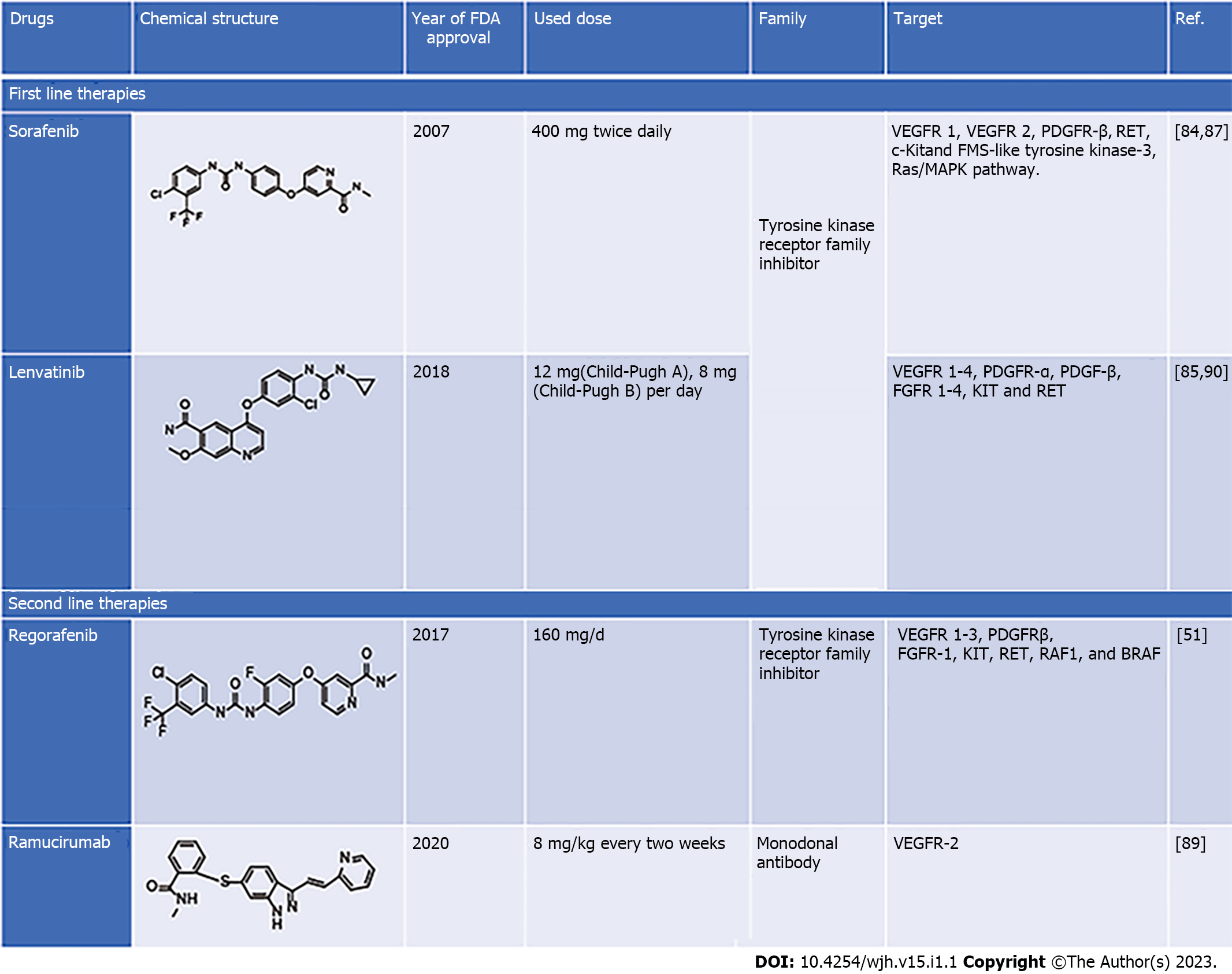
Sorafenib is a first-line chemotherapeutic agent approved for advanced HCC. It is a multikinase inhibitor targeting Raf, EGFR, VEGF receptor (VEGFR), PDGF receptor (PDGFR), FMS-like tyrosine kinase-3 (FLT3) and c-kit[45,46]. Clinical studies have revealed that sorafenib inhibits hepatic tumor growth and angiogenesis in advanced stage, but its prolonged exposure induces resistance[47-50]. Recently it has been reported that the second-line drugs such as lenvatinib, regorafenib and ipilimumab have a better therapeutic outcome, and increase overall disease-free survival in liver cancer patients[51]. Increased tumor growth and distance metastasis in sorafenib resistance patients and lower overall survival rates in sorafenib-treated liver cancer patients necessitate exploring new and potential therapeutic targets in liver cancer. Exploration of newer therapeutic agents and combinatorial drug regimens may also be explored to target the disease and increase the therapeutic outcome in patients. The current treatment strategy for liver cancer (first- and second-line therapies) is discussed in more detail in the subsequent section of this review.
Luo et al[52] identified emerging targets in liver cancer by utilizing comprehensive and integrated multiomics analysis. The study identified potential signaling pathways (Tp53/RB1, Wnt/β-catenin, PI3/Akt/mTOR, JAK/STAT, MAPK and TGF-β) and molecular events (telomere maintenance, cellular differentiation, chromatin remodeling and oxidative stress) in liver cancer. Mutation-mediated protein activation (CCND1, CTNNB1, TERT, PIK3CA, KRAS, KEAP1, NFE2L2, JAK3, FGF4, FGF19, and FGF3) and inactivation (TP53, Rb1, CDKN24, CHN2B, ATM, AXIN1, APC, ZNRF3, HNF1A, APOB, ALB, ARID1A/B, ARID2, SMARC2, BAP1, BRD7, KMT2C, PTEN, TSC1, TSC2, RPS6KA3, and ACVR2A) are associated with the pathophysiology of liver cancer and have emerged as therapeutic targets for hepatic cancer[52]. β2-spectrin (SPTBN1), a cytoskeleton protein is essential for the development of various organs, including the liver. It performs both structural (establishment and maintenance of cellular structure) and functional (apoptosis, cell adhesion, and cell cycle regulation) role[53]. Recently it has been reported that SPTBN1 induces lipogenesis-mediated liver cancer in high-fat diet fed experimental mice. The study proposed SPTBN1 as a potential therapeutic target for liver cancer[54]. Craig et al[55] studied the expression profile of cancer testis antigens (CTA) proteins in HCC. CTA was overexpressed in HCC patients and associated with poor overall survival and prognosis. Further experimental evidence of the study showed that melanoma-associated antigens family A (MAGE-A), a member of the CTA family, is responsible for increasing cellular proliferation, and decreased apoptosis and aggressiveness in HCC experimental models. The study revealed that MAGEA3 is involved in the developing hepatic carcinoma and could serve as a potential novel target for the disease[55]. Glypican (GPC)-3, a heparin sulfate proteoglycan, was significantly overexpressed in > 80% of HCC patients and was positively associated with poor diagnosis in the patients[56,57]. Clinical studies showed that targeting GPC-3 by developed antibodies significantly increased disease progression-free survival in patients with overexpressed GPC-3 in comparison with patients with low GPC-3 levels. Combination of chemotherapy and the immunotoxin (antibody + exotoxin) mediated GPC-3 targeting showed better therapeutic outcomes in liver cancer patients[58-60]. These facts indicate the therapeutic potential of GPC-3 proteins in liver cancer. Interaction between HGF and its receptor c-Met is important in liver regeneration. Overexpression and/or mutation in c-kit have been positively associated with liver cancer[61]. Direct or indirect (via different signaling pathways) interaction among HGF and c-kit increases the cellular growth, angiogenesis and metastasis in liver cancer cells[62]. Preclinical and clinical studies reported that interrupting the association between HGF and c-kit resulted in a potential therapeutic response in liver cancer[63-65]. Thus HGF and/or c-kit are potential therapeutic targets in liver cancer. Various studies showed that cancer cells rewire their metabolic pathways to fulfil their increased need for nutritional requirement. Liver cancer cells also reprogram their lipid metabolic pathway to combat their increased nutritional requirements, which ultimately help in cellular proliferation, growth and survival. Preclinical studies have shown that biosynthesis of lipids and desaturation process play an important role in liver cancer initiation, progression and survival. Pope et al[66]beautifully reviewed aberrant biochemical/molecular players of lipid metabolism as potential therapeutic targets in liver cancer[66,67]. Overexpression of lipid metabolism enzymes such as fatty acid synthetase, ATP citrate lyase, stearoyl-CoA desaturase (SCD)-1, and acetyl CoA carboxylase have been associated with various cancers including liver cancer. Targeting these enzymes with small molecules showed a potential tumor-suppressive nature in experimental models of liver cancer. There is a need to study some enzyme inhibitors in the clinical trial, such as SCD-1 inhibitors[68-70].
miRNAs are short-length noncoding RNAs involved in regulating gene expression and thus controlling the normal physiology and disease pathophysiology by normal and abrupt expression, respectively[71]. Modulating miRNAs by therapeutic molecules, and/or using their respective inhibitors or mimics is an important strategy to target cancer at the gene level[72]. The study showed that aberrant expression of miRNAs (miR34, miR36, miR21, miR203, miR17, miR83, miR93, miR221, etc.) in liver cancer cells is associated with the increased cellular proliferation, metastasis, angiogenesis, drug resistance, cell survival and reduced apoptosis[72]. A miRNA-based mouse model of HCC has been developed to study inflammation, tumor initiation, metabolic alteration, and hepatocyte differentiation[73]. The therapeutic potential of miRNAs in liver cancer has been shown by utilizing the miRNA inhibition/replacement approach. One studyidentified miR-550a, miR-574, miR-424, let-7i, miR-549, miR-518 and miR-512 as being significantly associated with overall survival, using bioinformatics tools that indicated their therapeutic potential. The study proposed that these miRNAs should be studied in detail for their therapeutic potential in liver cancer experimental models[74]. Dai et al[75] compared the publically available liver cancer miRNA expression data with the human HCC (hepatitis B positive and negative) data (generated by the study group). The study identified miR-0308-3p as a novel miRNA associated with HBV-positive HCC. miRNA suppresses liver cancer cell proliferation and arrests cells in the G1/S phase by targeting CDK6 and cyclin1 genes[75]. These results show that the miR-0308-3p is a novel therapeutic target in liver cancer. Shao et al[76]developed personalized miRNA cocktail therapy by combining nanotechnology and gene therapy to treat liver cancer. The research group encapsulated mimics (of miR-199a/b-3p) and inhibitor (of miR-10b) into a polymer-based nanoplatform (PCACP). The in vitro and in vivo experiments showed the better anticancer potential of the PCACP/miR-cocktail system in comparison with mimic or inhibitor treatment alone in liver cancer experimental models[76]. This study showed a novel potential strategy to treat liver cancer by combining nanotechnology and gene therapy. Wang et al[77] studied the relation between LINC01018 (a long noncoding RNA), miR-182-5p and FOXO1 protein in HCC. There was poor expression of the long noncoding RNA and FOXO1, and higher expression of miR-182-5p in the HCC patient samples. Forced expression of LINC01018 in in vitro and in vivo experimental models showed decreased cellular proliferation and induced apoptosis with increased miR-182-5p levels. The study showed liver cancer therapeutic potential of LINC01018 by miR-182-5p sponge-mediated downregulation of FOXO1 expression[77].
The dysregulated cell cycle, apoptosis, and many other key signaling pathways are linked to HCC pathogenesis. Chemotherapeutic approaches similar to different types of cancer are also reported with a limited number of drugs for the cure of HCC and various side effects. Sorafenib, an oral multitargeted tyrosine kinase inhibitor has been used as first-line treatment for advanced HCC, showing increased survival of approximately 12 mo compared with controls[78]. Various antiangiogenic agents such as bevacizumab (human monoclonal antibody directed against VEGF) and erlotinib (EGF receptor tyrosine kinase inhibitor) have also been studied and shown effective results in early studies[79]. Until 2016, sorafenib was the only FDA-approved first-line treatment for HCC, whereas lenvatinib has also been identified and is in use for advanced HCC[80]. Sorafenib acts as an inhibitor of intracellular tyrosine and serine/threonine protein kinases such as VEGF, VEGFR, PDGFR, c-Raf and b-Raf MAP kinases, which in turn induces autophagy. Due to drug resistance and side effects such as liver fibrosis, clinical usage of sorafenib is limited[63]. Long-term exposure to sorafenib also induces cancer cells with less E-cadherin content making them more invasive. Some second-line treatments are also available for HCC, including regorafenib, ramucirumab and cabozantinib, which are rarely used and are less efficient[28,51,79]. It is reported that chemotherapeutic drugs used for HCC treatments are limited in number and seem to be less effective, considering their efficacy, bioavailability and side effects. Considering the side effects of ongoing therapies, scientific pieces of evidence are also suggestive for the use of natural products for the management of HCC, since they can inhibit viral infection, inflammation, oxidative stress, metabolic disorders, angiogenesis and metastatic activity, which are known as prime contributors in HCC[2,80,81]. Hence, there is strong demand for searching novel plant-based drugs for managing HCC with fewer side effects and less chemotoxicity. Therefore, several drugs are used to treat HCC to target the inhibition of some of these processes (Figure 1). The current therapeutic interventions for patients with HCC are divided into first- and second-line therapies. The pharmacological features of these drugs are discussed in the following section of this review.
Sorafenib: Sorafenib (BAY 43-9006, Nexavar) is the first-ever systemic drug as well as a standard therapeutic agent approved by the US FDA for treatment of liver cancer patients who cannot undergo surgical resection or liver transplantation[79]. Sorafenib was the only first-line treatment in the last 10 years until the FDA approved lenvatinib as a frontline therapy in 2018. It is a tyrosine kinase inhibitor that targets VEGFR1 and VEGFR2 and PDGFR-β. It activates AMP-activated protein kinase (AMPK) that can block the formation of tumor blood vessels and inhibit proliferation of liver cancer cells[82]. For individuals with HCC, sorafenib has a clear advantage in terms of survival. Sorafenib improved overall survival considerably compared with placebo in two phase III clinical randomized controlled trials (10.7 mo vs 7.9 mo and 6.5 mo vs 4.2 mo). However, the side effects associated with these clinical trials were diarrhea, tiredness, and hand–foot skin response[80,82].
Several factors hinder more people from obtaining benefits after sorafenib treatment. Because of the genetic variability of HCC and other factors, around 40% of people with HCC can benefit from sorafenib. Sorafenib was more beneficial for some patient categories in several trials. The two clinical studies mentioned above featured only a small number of patients; all of whom had good liver function. These individuals were termed Sorafenib Hepatocellular Carcinoma Assessment Randomized Protocol (SHARP)-eligible patients, and only SHARP-eligible individuals benefited from sorafenib treatment[79,82]. Furthermore, the effectiveness of sorafenib is greater in HCV-infected individuals than in others who have always been resistant to sorafenib. Primary resistance is another term for the unclear mechanism of this phenomenon[82,83]. However, some research has uncovered probable explanations. Gene polymorphism may be a crucial factor influencing sorafenib function. Polymorphisms in the ATP binding cassette (ABC) subfamily B member 1 (ABCB1), ATP binding cassette subfamily G member 2 (ABCG2), solute carrier family 15 member 2 (SLC15A2) and endothelial nitric oxide synthase (eNOS) have been linked to the action of sorafenib[83]. This was confirmed by Silvia and co-workers who reported that β-caryophyllene oxide inhibits ABC proteins and causes HCC cells to become chem
Lenvatinib: Lenvatinib (E7080, Lenvima) is an antitumor drug that belongs to the quinoline carboxiamides. The IUPAC name of lenvatinib is 4-[3-chloro-4-(cyclopropylcarbamoylamino) phenoxy]-7-methoxyquinoline-6-carboxamide. Lenvatinib acts as multikinase inhibitor via targeting VEGFR 1-4, PDGFR-α, PDGFR-β, FGFR 1-4, tyrosine kinase receptor (KIT) and rearranged during transfection receptor (RET) that leads to angiogenesis inhibition, and reduced vascular permeability of the tumor microenvironment[28]. Lenvatinib is an effective drug that increases overall survival in patients with advance HCC and whose tumor cannot be removed by surgery. In a phase I clinical trial, lenvatinib (12 and 8 mg) was effective in patients with advanced HCC and Child–Pugh class A or B. The adverse effects observed during 12 mg daily lenvatinib oral treatment were hypertension, decreased body weight, loss of appetite fatigue, and diarrhea[28,70]. A Phase II clinical trial was conducted to evaluate the effectiveness of lenvatinib in advanced unresectable HCC. The trial was conducted on 46 patients who received 12 mg lenvatinib orally once daily for 28 d, and lenvatinib demonstrated high efficacy with a good toxicity profile. However, the efficacy of lenvatinib was influenced by body weight[85,86].
Regorafenib: Regorafenib (BAY-73-4506) with the brand name Stivagra is an oral multikinase receptor antagonist developed by Bayer and approved by the US FDA in June 2017 to treat unresectable advanced liver cancers. Despite its structural similarity with sorafenib, regorafenib showed more effectiveness in inhibiting the activities of various protein kinases associated with neovascularization (VEGFR 1–3 and tyrosine kinase with immunoglobulin-like loops and epidermal growth factor homology domain-2 (TIE2), oncogenesis (KIT, RET, Raf1 and BRAF) and tumor microenvironment (PDGFR-β, PDGFR-α and FGFR) with better drug tolerance profile[87,88]. HCC patients treated with regorafenib (160 mg/d for 28 d) showed better overall survival, i.e., 10.6 mo compared with 7.8 mo in the placebo group in a randomized, double-blind, placebo-controlled phase III trial. However, the main side effect was hypertension, unlike body weight loss, hepatorenal dysfunction, and fatigue in sorafenib-treated individuals[87,88].
Ramucirumab: Ramucirumab, sold under brand name Cyramza and others, is a recombinant mono
Pirfenidone: Pirifenidone (Esbriet®) is an orally administered antifibrotic, antioxidant, and anti-inflammatory drug that has been studied in clinical and preclinical trials to treat hepatic and idiopathic pulmonary fibrosis[86]. Pirifenidone was effective in causing cell cycle arrest at G0/G1, eventually inhibiting cell proliferation in an in vitro model. Similarly, it induces apoptosis in HepG2 cells via Wnt/β-catenin signaling pathway. Pirifenidone has also been demonstrated to be a potent antifibrotic agent at a dose of 300 mg/kg in a carbon tetrachloride-induced HCC mouse model. However, the cellular mechanisms behind the responses elicited by pirifenidone remain unknown[70,86]. Figure 1 sum
Multiple lines of scientific evidence have suggested the significant contribution of gut microbes to critical aspects of human health. Even though the gut microbiota offers substantial benefits to the host, particularly in terms of immunity and metabolic activities, there is still growing evidence of the role of gut microbes in several pathological conditions. They promote disease progression not just locally, as in chronic inflammatory bowel syndrome, but also in other parts of the body, such as liver, brain and heart[91]. Similarly, there is mounting evidence that the gut microbiota plays a significant role in carcinogenesis via its local and long-distance effects. The liver is intimately connected to the gut through the portal vein. The liver is directly exposed to microbial metabolites and microbe-associated molecular patterns (MAMPs) that can induce inflammatory reactions through pattern-recognition receptors, and receive nutrient-rich blood from the gut. The multilayer epithelial barrier is responsible for minimal hepatic exposure to MAMPs. Although, as in chronic liver diseases, altered gut barrier and microbiota composition increases the incidence of inflammation and progression of liver disorder and thus raises the risk of HCC[92].
According to accumulating scientific evidence, intestinal dysbiosis appears to have a significant role in developing chronic liver disease and HCC. Metagenomic studies have demonstrated significant changes in the gut microbiota composition in a variety of chronic liver diseases as well as in people with cirrhosis[93]. Patients with advanced liver disease and cirrhosis have an increase in potentially harmful bacteria and a decrease in microorganisms with beneficial qualities in their gut microbiomes[94,95].
Toll-like receptor (TLR)4 is found in various liver resident cells such as Kupffer cells, hepatic stellate cells (HSCs), endothelial cells, and hepatocytes. A study conducted by Dapito and colleagues in bone marrow chimeric mice concluded that the presence of TLR4 on these liver-resident cells promotes fibrogenesis and hepatocarcinogenesis[96]. Lipopolysaccharide (LPS), a Gram-negative bacterial cell wall component, is produced through the leaky gut and mainly targets Kupffer cells and HSCs, which appears to increase the incidence of hepatocarcinogenesis. Activation of TLR4 in HSCs causes nuclear factor (NF)-κB-mediated increased expression of epiregulin, a hepatic mitogen belonging to the EGF family, and reported to have strong mitogenic potential in hepatic cells[96,97]. The finding was confirmed when hepatocarcinogenesis decreased in epiregulin-deficient rats treated with N-nitrosodiethylamine (DEN)-CCl4. Another important method through which the LPS–TLR4 axis promotes HCC development is through prevention of NF-κB-mediated hepatocyte apoptosis[96,97].
For centuries, bioactive natural products from plants have been extensively used to treat many human diseases. Recent molecular evidence explains their modes of action, metabolic regulation, and identification of their biological targets. This evidence adds value to their potential use in the chem
In the last two decades, growing evidence has suggested an affirmative role of resveratrol (polyphenolic natural product) in the chemoprevention of liver cancer. Its application is limited due to its poor bioavailability. Previously, resveratrol was shown to negatively regulate the cellular proliferation of rat hepatoma and human hepatoblastoma cell line HepG2 at 1–150 μmol/L concentration[98]. Decreased proliferation and invasion of HepG2 cells and AH109A rat ascites hepatoma cells were also reported. In subsequent studies, resveratrol induced apoptosis in in vitro studies using HepG2and H4IIE rat hepatoma cells[98]. Notas et al[99] showed that even 2 h treatment with resveratrol (10-6–1 μmol/L) in
A curcumin analog, CUR3d, inhibited the proliferation of liver cancer cells at 100 μmol/L, which was due to downregulation of PI3K/Akt and inhibition of the NF-κB pathway, which are responsible for cancer cell growth[102]. In another study, supplementation of curcumin (1 g/kg) significantly inhibited the growth and liver metastasis of colorectal cancer cells[103]. Microemulsion formulation improve 1225 times the water solubility of myricetin and enhanced its antiproliferative activity against human liver cancer cells (HepG2)[104].
Extract of immature plum induced extrinsic apoptosis in HepG2 cells as demonstrated by caspase-1,
| Natural products | Extract/phytochemicals | Experiment model (in vitro/in vivo/clinical trials | Tested concentration | Medicinal effects | Ref. |
| Broccoli | Sulforaphane | In vitro (murine hepatoma Hepa 1c1c7 and human HepG2 cells) | 1-20 μmol/L | Sulforaphane showed positive effect on Phase II detoxification enzyme. Sulphoraphane treatment resulted in increased expression of CYP1A1 and quinone reductase | [116] |
| Grape | Procynidins rich grape crude extract | In vitro (HepG2 human liver cancer cells) | 0-120 μg/mL | Grape extract in concentrations greater than 20 μg/mL (20.4 μmol/L) was cytotoxic to HepG2 human liver cancer cells, with maximal toxicity of 67.2% and ED50 of 49.6 μg/mL (50.5 μmol/L) | [117] |
| Flavan-3-ol rich extract | In vitro HepG2 and breast cancers (MCF-7) cells | 0-60 μg/mL | Grape extract showed dose dependent cytotoxicity via Induction of apoptosis, DNA damage and suppression of oncoprotein Her-2 expression. Treatment also resulted in increased NO production in cancer cell | [118] | |
| Mung bean sprouts | Extract | In vitro [Human cervical (HeLa) and hepatocarcinoma cells (HepG2)] | 9.37 to 300 mg/mL | Mung bean sprouts was found to be a potent anticancer agent. The cytotoxic effect of Mung bean sprouts extract on HeLa, expressed as IC50, was 13.3 mg/ml 163.97 mg/ml while on HepG2 cells was 14.04 mg/ml. It also increases apoptosis, anti-tumor cytokines (TNF-and IFN-β), IFN-γproduction and subsequently up regulated the cell-mediated immunity | [119] |
| Cinnamon | Isoobtusilac-tone A | In vitro (Hep G2 cells) | 100 μmol/L | Induces apoptosis in cancer cell | [120] |
| Ginger | 6-shogaol, 6-gingerol | In vitro (Human hepatoma HepG2 and Hep3B cells) | 10 μmol/L and 50 μmol/L | The migratory and invasive activity of HepG2 and Hep3B cells were decreased in doses dependent manner post 6-shogaol, 6-gingerol treatment. It suppresses the metastatic activity via down regulation of matrix metalloproteinase (MMP)-9 and urokinase type plasminogen and upregulation of tissue inhibitor metalloproteinase protein | [108,121] |
| Asparagus | asparanin A | In vitro (HepG2 cells) | 0-30 μmol/L | Treatment with asparanin A resulted in cell cycle arrest at G2/M phase and apoptosis in HepG2 cells. Following treatment of HepG2 cells with asparanin A, cell cycle-related proteins including cyclin A, Cdk1 and Cdk4 were down-regulated, while p21WAF1/Cip1 and p-Cdk1 (Thr14/Tyr15) were up-regulated | [122] |
| Tomato | Tomatine | In vitro (HepG2 cells) | 10, 50 and 100 μg/mL | Induces antigen-specific cellular immunity and direct destruction of cancer cell membranes | [123] |
| Lycopene | In vivo (N-nitrosodiethylamine induced hepatocarcinogenesis in female Balb/c mice) | 5 mg/kg bw | Lycopene treatment causes modulation of apoptosis related genes (enhanced expression of caspase 3 and 9 and p53 and decreased expression of Bcl-2). Lycopene exhibits pro-oxidant activity in tumor that supports observed enhanced apoptosis. This increased apoptosis is a chemopreventive action of lycopene in liver cancer | [124] | |
| Plum | Extract | Benzopyrene-induced hepatocarcinogenesis in rats | 2.5 or 5 g/kg bw | Plum extracts may counteract toxic effects of carcinogens and benzopyrene, and therefore have chemopreventive efficacy | [125] |
| Pomegranate | Emulsion | Diethylnitrosamine (DENA)-induced hepatocarcinogenesis inrat | 1 or 10 g/kg bw | Treatment for 18 wk in rats resulted in reduced incidence, number, multiplicity, size and volume of hepatic nodules, precursors of HCC. It showed chemoprevention through potent antioxidant activity as the expression of Nrf-2 is increased in pomegranate treated rats | [126] |
| Citrus fruit | Auraptine | Diethylnitrosamine (DEN)-induced hepatocarcinogenesis in rat | 5 or 10 g/kg bw | It suppresses tumor progression in DEN- challenged rats by negative selection for cancer cells with β-catenin mutation | [127] |
| Pepper | Glycoprotein | In vivo (DEN induced hepatocarcinogenesis in mice) | 20 mg/kg bw | Pepper glycoprotein of 24 kDa treatment in rats causes increased activity of natural killer cell and ultimately prevention of DENA induced liver carcinogenesis via immunomodulation and promotion of apoptosis | [128] |
Intraperitoneal resveratrol administration (1 mg/kg) for 7 d in male Wistar rats implanted with AH-130 hepatoma cells arrested tumor growth. Liu et al[109] showed the immunomodulatory role of resveratrol (500, 1000, 1500 mg/kg for 10 d) in BALB/c mice implanted with H22 hepatoma cells. Rajasekaran et al[110] studied the chemopreventive role of resveratrol in a model of DEN-induced HCC in male Wistar rats. It induced apoptosis by PARP cleavage, caspase-3 activation, p53 upregulation and cytochrome c release when given early at a dose of 200 mg/kg. Gao et al[111] tested the chemopreventive property of resveratrol in MHCC97-H-inoculated athymic nude mice. The study identified its antitumor activity by downregulating the HGF/c-Met signaling pathway. Resveratrol-gold nanoparticles have shown improved anticancer effects compared with resveratrol alone in HEPG2 cells and xenografted BALC/c nude mice[112].
Lycium polysaccharide portion (LPP) is the most crucial part of Lycium barbarum that has abundant biological activities such as antioxidant, neuroprotective, immunoprotective, antitumor, and glucose metabolism regulatory activities. LPP inhibited the propagation of hepatocytes and led to apoptosis of liver hepatocytes, thus indicating its anticancer role. A clinical trial showed that consumption of LPP juice leads to elevation in interleukin (IL-2), IgG, serum antioxidants levels, lymphocyte count and reduced levels of lipid peroxides[113]. Berberine mediates anticancer activity by inhibiting antiapoptotic protein Bcl-2, and activating the caspase cascade and proapoptotic pathway of Egr1-NAG-1 (nonsteroidal anti-inflammatory drug-activated gene). Berberine facilitates phosphorylation of AMPK, thus increasing the concentration of p-AMPK/total AMPK. The AMPK-mediated mitocho
Natural products have become a focus of attention in anticancer drug discovery due to unsolved problems related to current chemotherapy, such as drug resistance and toxicity. It should be noted that from 1940 to 2014, approximately 50% of the small molecules approved for cancer treatment were either natural compounds or their derivatives[129,130]. Natural products for anticancer therapy have had some therapeutic limitations, which affect therapeutic outcome, lower bioavailability, and selected and targeted delivery. This section highlights these issues, recent advances in the field, and future potential. Advancements in computational biology/pharmacology/chemistry and high-throughput in vitro screening of natural anticancer drugs have highly accelerated the drug discovery process, resulting in a lead molecule. Most of the time, it is frustrating to obtain unsatisfactory activity of the lead natural molecule in in vivo experiments and/or clinical studies, which results in lesser activity and nonselectivity for a given therapeutic target. It has been proposed that delivering natural products to a targeted site using an appropriate delivery system may improve the efficacy by increasing their bioavailability. The process may also decrease the off-target effects and toxicity related issues in a given therapy[131]. Different means of drug delivery or appropriate vehicles have been discussed elsewhere[132]. The use of these tools/vehicles is dependent on their biocompatibility, degradability and functional limitations. However, the concept is promising but has its limitations (rapid elimination from the body, toxicity and inflammation), which still need to be addressed[132,133].
To consider the efficacy of natural products in living systems, it is essential to understand their pharmacokinetics. Absorption, distribution, metabolism and excretion may primarily affect the therapeutic outcome of the natural products. Absorption of a particular drug is influenced by the mode of administration, i.e., whether it is oral, intravenous or inhalation. In each case, the drug shows different kinetic behavior in relation to its therapeutic outcome. Factors such as permeability of barriers, pH of cellular/body compartments, binding affinity with the off-targets and their fat solubility affect the distribution of the natural products in the body. Drug metabolism in the liver or gut introduces alterations in the structure of natural products, as well as irreversible secretion of the drugs through the hepatobiliary system or kidneys, which affects the plasma level of the drug and its efficacy. Few reports are available on the pharmacokinetics of natural products (such as glycyrrhetinic acid, curcumin, ethiodized oil) in liver cancer experimental models or patients[103,104,134]. Most of the lead anti-liver cancer natural products have not yet been studied for the above pharmacokinetic parameters in experimental models. Information on the pharmacokinetic parameters of the particular natural products may shed light on the efforts that should be taken to improve their therapeutic efficacy in in vivo experimental models and liver cancer patients.
New approaches have been introduced to improve the natural product delivery and specifically target liver cancer cells. Previously it has been reported that tissue-targeted drug delivery significantly enhances the therapeutic efficacy of anti-liver cancer drugs, confining their bioavailability within the tumor. The concept of tissue targeted drug delivery also minimizes the side effects such as toxicity by reducing systemic bioavailability to other organs of the body. Anti-liver cancer drugs combined with a delivery system providing galactose residues have been utilized to target liver cells (with asialoglycoprotein receptors) specifically[135,136]. Liposomes have been used as carriers for anticancer drugs due to various advantages such as improved drug stability in the body without altering the structural integrity of the drug[136,137]. Li et al[138] studied the effect of natural product encapsulated galactosylated liposomes (NPEGLs) to assess their anticancer activity and liver cancer cell selectivity. The study found that anti-liver cancer activity of the NPEGLs was significantly increased compared with normal natural product–liposomes and free natural product treatment in liver cancer cells. Enrichment of NPEGLs with galactosylated stearate significantly increased the uptake of the delivery system by the liver cancer cells compared with gastric and non-small cell lung cancer cells[139].
Toxicity due to the off-target effect of the anticancer therapeutic drug is also an important problem in managing liver cancer at the clinical level. It is challenging to increase bioavailability and decrease off-target effects of anti-liver cancer natural products without compromising therapeutic efficacy. This situation is more difficult when increased effectiveness of the product is required. Nanotechnology-based approaches are promising to provide the solution to this problem. It is possible to deliver the natural products using nanotechnology-based strategies, which not only increase the product’s biological activity but also enhance its bioavailability. Targeted delivery using these strategies also lowers toxicity by reducing the systemic circulation of the product. Gera et al[140] synthesized a phytocomposite nanoparticle and studied its anticancer efficacy in liver cancer cells. The natural-compound-based nanoparticles produced significantly higher antiproliferative activity in liver cancer cells in comparison with free natural product (non-nanoparticle form). The study suggested that the increased activity of the nano-formulation of the natural product in comparison with its non-nano form might be attributed to its well dispersed, small-sized particles, and thereby increased cellular uptake. The study also suggested that the attraction of the formulation towards the acidic environment of liver cancer cells enhances the output of targeted therapy with less or no effect on normal cells. Thus, this type of strategy, in combination with other approaches (such as receptor targeting), could be utilized to selectively target liver cancer cells to avoid the off-target effects and increase the drug’s bioavailability[141,142].
In recent years, liver cancer has emerged as a significant public health concern worldwide. Various factors such as viral infection, alcohol abuse, drug-induced liver injury, or a high fat diet are the leading causes of mortality due to liver diseases. Different signaling pathways, including TGF-β, Wnt/B-catenin, Hedgehog, Notch, EGF, VEGF, JAK and Hippo, are responsible for the progression of liver cancer. First- and second-line treatments produced better therapeutic outcomes than chemotherapy and increased overall disease-free survival in liver cancer patients. Intestinal dysbiosis appears to have a significant role in developing chronic liver diseases. The available modes of treatment include numerous side effects that could be minimized with the use of natural products such as resveratrol, curcumin, diallyl sulfide and many more. However, natural-product-based anticancer therapy also has some limitations, mainly concerning the therapeutic outcome, lower bioavailability, and newer targeted delivery approaches. Targeted drug delivery using NPEGLs and nano-formulations increased the biological activity and bioavailability of the drugs.
Shashank Kumar and Sabyasachi Senapati acknowledge DST-FIST facilities of the Department of Biochemistry and Department of Human Genetics and Molecular Medicine, Central University of Punjab, Bathinda, Punjab. All the authors also acknowledge DST-FIST and UGC-SAP facilities of the Department of Biochemistry, University of Allahabad, Prayagraj, India.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Ling Q, China; Zhao G, China S-Editor: Wu YXJ L-Editor: Kerr C P-Editor: Wu YXJ
| 1. | Kanwal F, Khaderi S, Singal AG, Marrero JA, Loo N, Asrani SK, Amos CI, Thrift AP, Gu X, Luster M, Al-Sarraj A, Ning J, El-Serag HB. Risk factors for HCC in contemporary cohorts of patients with cirrhosis. Hepatology. 2022;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 78] [Article Influence: 39.0] [Reference Citation Analysis (0)] |
| 2. | Ganguly R, Gupta A, Pandey AK. Role of baicalin as a potential therapeutic agent in hepatobiliary and gastrointestinal disorders: A review. World J Gastroenterol. 2022;28:3047-3062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 9] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (2)] |
| 3. | Jeng KS, Chang CF, Sheen IS, Jeng CJ, Wang CH. Upper Gastrointestinal Cancer and Liver Cirrhosis. Cancers (Basel). 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Singh AK, Kumar R, Pandey AK. Hepatocellular Carcinoma: Causes, Mechanism of Progression and Biomarkers. Curr Chem Genom Transl Med. 2018;12:9-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 109] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 5. | Kim JY, Song EH, Lee HJ, Oh YK, Choi KH, Yu DY, Park SI, Seong JK, Kim WH. HBx-induced hepatic steatosis and apoptosis are regulated by TNFR1- and NF-kappaB-dependent pathways. J Mol Biol. 2010;397:917-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Kim KH, Shin HJ, Kim K, Choi HM, Rhee SH, Moon HB, Kim HH, Yang US, Yu DY, Cheong J. Hepatitis B virus X protein induces hepatic steatosis via transcriptional activation of SREBP1 and PPARgamma. Gastroenterology. 2007;132:1955-1967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 225] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 7. | Choudhary NS, Kumar N, Duseja A. Peroxisome Proliferator-Activated Receptors and Their Agonists in Nonalcoholic Fatty Liver Disease. J Clin Exp Hepatol. 2019;9:731-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 8. | Flores JE, Thompson AJ, Ryan M, Howell J. The Global Impact of Hepatitis B Vaccination on Hepatocellular Carcinoma. Vaccines (Basel). 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 44] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 9. | Li XZ, Zhu JQ, Zhang SY, He QQ, Zhang GX, Jia HD, Wang XM. Letter: is it appropriate to use a fatty liver index >60 as an alternative criterion for non-alcoholic fatty liver disease? Aliment Pharmacol Ther. 2022;56:376-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 10. | Klein S, Dufour JF. Nonalcoholic fatty liver disease and hepatocellular carcinoma. Hepat Oncol. 2017;4:83-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Rodina AS, Shubina ME, Kurbatova IV, Topchieva LV, Dudanova OP. [Hepatocellular damage and inflammation in various forms of alcoholic liver disease]. Ter Arkh. 2021;93:15-19. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 12. | Yasmin A, Regan DP, Schook LB, Gaba RC, Schachtschneider KM. Transcriptional regulation of alcohol induced liver fibrosis in a translational porcine hepatocellular carcinoma model. Biochimie. 2021;182:73-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 13. | Liu BJ, Gao S, Zhu X, Guo JH, Kou FX, Liu SX, Zhang X, Wang XD, Cao G, Chen H, Liu P, Xu HF, Gao QZ, Yang RJ. Real-world study of hepatic artery infusion chemotherapy combined with anti-PD-1 immunotherapy and tyrosine kinase inhibitors for advanced hepatocellular carcinoma. Immunotherapy. 2021;13:1395-1405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 14. | Bruix J, Cheng AL, Meinhardt G, Nakajima K, De Sanctis Y, Llovet J. Prognostic factors and predictors of sorafenib benefit in patients with hepatocellular carcinoma: Analysis of two phase III studies. J Hepatol. 2017;67:999-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 453] [Article Influence: 56.6] [Reference Citation Analysis (0)] |
| 15. | Veracruz N, Gish RG, Cheung R, Chitnis AS, Wong RJ. Global incidence and mortality of hepatitis B and hepatitis C acute infections, cirrhosis and hepatocellular carcinoma from 2010 to 2019. J Viral Hepat. 2022;29:352-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 16. | Gore M, Desai NS. Characterization of phytochemicals and evaluation of anti-cancer potential of Blumea eriantha DC. Physiol Mol Biol Plants. 2014;20:475-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Massart J, Begriche K, Corlu A, Fromenty B. Xenobiotic-Induced Aggravation of Metabolic-Associated Fatty Liver Disease. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 18. | Farcas M, Gavrea AA, Gulei D, Ionescu C, Irimie A, Catana CS, Berindan-Neagoe I. SIRT1 in the Development and Treatment of Hepatocellular Carcinoma. Front Nutr. 2019;6:148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 19. | Tommasi S, Pinto R, Pilato B, Paradiso A. Molecular pathways and related target therapies in liver carcinoma. Curr Pharm Des. 2007;13:3279-3287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 20. | Yang C, Peng P, Li L, Shao M, Zhao J, Wang L, Duan F, Song S, Wu H, Zhang J, Zhao R, Jia D, Zhang M, Wu W, Li C, Rong Y, Zhang L, Ruan Y, Gu J. High expression of GFAT1 predicts poor prognosis in patients with pancreatic cancer. Sci Rep. 2016;6:39044. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 21. | Sundaram V, Jalan R, Shah P, Singal AK, Patel AA, Wu T, Noureddin M, Mahmud N, Wong RJ. Acute on Chronic Liver Failure From Nonalcoholic Fatty Liver Disease: A Growing and Aging Cohort With Rising Mortality. Hepatology. 2021;73:1932-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 22. | Yuan RH, Hsu CL, Jhuang YL, Liu YR, Hsieh TH, Jeng YM. Tumor-matrix interaction induces phenotypic switching in liver cancer cells. Hepatol Int. 2022;16:562-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 23. | Nath A, Li I, Roberts LR, Chan C. Elevated free fatty acid uptake via CD36 promotes epithelial-mesenchymal transition in hepatocellular carcinoma. Sci Rep. 2015;5:14752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 270] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 24. | Thompson KJ, Humphries JR, Niemeyer DJ, Sindram D, McKillop IH. The effect of alcohol on Sirt1 expression and function in animal and human models of hepatocellular carcinoma (HCC). Adv Exp Med Biol. 2015;815:361-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Baik M, Yu JH, Hennighausen L. Growth hormone-STAT5 regulation of growth, hepatocellular carcinoma, and liver metabolism. Ann N Y Acad Sci. 2011;1229:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 26. | Azbazdar Y, Karabicici M, Erdal E, Ozhan G. Regulation of Wnt Signaling Pathways at the Plasma Membrane and Their Misregulation in Cancer. Front Cell Dev Biol. 2021;9:631623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 27. | Wörns MA, Weinmann A, Pfingst K, Schulte-Sasse C, Messow CM, Schulze-Bergkamen H, Teufel A, Schuchmann M, Kanzler S, Düber C, Otto G, Galle PR. Safety and efficacy of sorafenib in patients with advanced hepatocellular carcinoma in consideration of concomitant stage of liver cirrhosis. J Clin Gastroenterol. 2009;43:489-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 115] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 28. | Rehman O, Jaferi U, Padda I, Khehra N, Atwal H, Mossabeh D, Bhangu R. Overview of lenvatinib as a targeted therapy for advanced hepatocellular carcinoma. Clin Exp Hepatol. 2021;7:249-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 29. | Xiang M, Su H, Hong Z, Yang T, Shu G. Chemical composition of total flavonoids from Polygonum amplexicaule and their pro-apoptotic effect on hepatocellular carcinoma cells: Potential roles of suppressing STAT3 signaling. Food Chem Toxicol. 2015;80:62-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 30. | Farzaneh Z, Vosough M, Agarwal T, Farzaneh M. Critical signaling pathways governing hepatocellular carcinoma behavior; small molecule-based approaches. Cancer Cell Int. 2021;21:208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 31. | Giovannini C, Gramantieri L, Chieco P, Minguzzi M, Lago F, Pianetti S, Ramazzotti E, Marcu KB, Bolondi L. Selective ablation of Notch3 in HCC enhances doxorubicin's death promoting effect by a p53 dependent mechanism. J Hepatol. 2009;50:969-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 32. | Gramantieri L, Giovannini C, Lanzi A, Chieco P, Ravaioli M, Venturi A, Grazi GL, Bolondi L. Aberrant Notch3 and Notch4 expression in human hepatocellular carcinoma. Liver Int. 2007;27:997-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 33. | Yang SL, Ren QG, Zhang T, Pan X, Wen L, Hu JL, Yu C, He QJ. Hepatitis B virus X protein and hypoxiainducible factor-1α stimulate Notch gene expression in liver cancer cells. Oncol Rep. 2017;37:348-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | Xie J, Wang M, Cheng A, Jia R, Zhu D, Liu M, Chen S, Zhao X, Yang Q, Wu Y, Zhang S, Luo Q, Wang Y, Xu Z, Chen Z, Zhu L, Liu Y, Yu Y, Zhang L, Chen X. The role of SOCS proteins in the development of virus- induced hepatocellular carcinoma. Virol J. 2021;18:74. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 35. | Kaltenecker D, Themanns M, Mueller KM, Spirk K, Suske T, Merkel O, Kenner L, Luís A, Kozlov A, Haybaeck J, Müller M, Han X, Moriggl R. Hepatic growth hormone - JAK2 - STAT5 signalling: Metabolic function, non-alcoholic fatty liver disease and hepatocellular carcinoma progression. Cytokine. 2019;124:154569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 36. | Jeng KS, Jeng CJ, Jeng WJ, Sheen IS, Li SY, Leu CM, Tsay YG, Chang CF. Sonic Hedgehog signaling pathway as a potential target to inhibit the progression of hepatocellular carcinoma. Oncol Lett. 2019;18:4377-4384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 37. | Li Y, Jiang M, Li M, Chen Y, Wei C, Peng L, Liu X, Liu Z, Tong G, Zhou D, He J. Compound Phyllanthus urinaria L Inhibits HBV-Related HCC through HBx-SHH Pathway Axis Inactivation. Evid Based Complement Alternat Med. 2019;2019:1635837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 38. | Singh AK, Bishayee A, Pandey AK. Targeting Histone Deacetylases with Natural and Synthetic Agents: An Emerging Anticancer Strategy. Nutrients. 2018;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 156] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 39. | Steinway SN, Zañudo JG, Ding W, Rountree CB, Feith DJ, Loughran TP Jr, Albert R. Network modeling of TGFβ signaling in hepatocellular carcinoma epithelial-to-mesenchymal transition reveals joint sonic hedgehog and Wnt pathway activation. Cancer Res. 2014;74:5963-5977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 184] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 40. | Gupta A and Pandey AK. Aceclofenac-induced hepatotoxicity: An ameliorative effect of Terminaliabellirica fruit and ellagic acid. World J Hepatol. 2020 27;12(11):949-964 . [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 41. | Liu LJ, Xie SX, Chen YT, Xue JL, Zhang CJ, Zhu F. Aberrant regulation of Wnt signaling in hepatocellular carcinoma. World J Gastroenterol. 2016;22:7486-7499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 74] [Cited by in RCA: 71] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 42. | Marquardt JU, Seo D, Andersen JB, Gillen MC, Kim MS, Conner EA, Galle PR, Factor VM, Park YN, Thorgeirsson SS. Sequential transcriptome analysis of human liver cancer indicates late stage acquisition of malignant traits. J Hepatol. 2014;60:346-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 43. | Nault JC, Calderaro J, Di Tommaso L, Balabaud C, Zafrani ES, Bioulac-Sage P, Roncalli M, Zucman-Rossi J. Telomerase reverse transcriptase promoter mutation is an early somatic genetic alteration in the transformation of premalignant nodules in hepatocellular carcinoma on cirrhosis. Hepatology. 2014;60:1983-1992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 258] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 44. | Pilati C, Letouzé E, Nault JC, Imbeaud S, Boulai A, Calderaro J, Poussin K, Franconi A, Couchy G, Morcrette G, Mallet M, Taouji S, Balabaud C, Terris B, Canal F, Paradis V, Scoazec JY, de Muret A, Guettier C, Bioulac-Sage P, Chevet E, Calvo F, Zucman-Rossi J. Genomic profiling of hepatocellular adenomas reveals recurrent FRK-activating mutations and the mechanisms of malignant transformation. Cancer Cell. 2014;25:428-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 199] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 45. | Liu L, Cao Y, Chen C, Zhang X, McNabola A, Wilkie D, Wilhelm S, Lynch M, Carter C. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer Res. 2006;66:11851-11858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1042] [Cited by in RCA: 1206] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 46. | Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G, Jassem J, Blanc JF, Vogel A, Komov D, Evans TRJ, Lopez C, Dutcus C, Guo M, Saito K, Kraljevic S, Tamai T, Ren M, Cheng AL. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3128] [Cited by in RCA: 3831] [Article Influence: 547.3] [Reference Citation Analysis (1)] |
| 47. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10271] [Article Influence: 604.2] [Reference Citation Analysis (2)] |
| 48. | Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, Liu J, Wang J, Tak WY, Pan H, Burock K, Zou J, Voliotis D, Guan Z. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3854] [Cited by in RCA: 4653] [Article Influence: 273.7] [Reference Citation Analysis (0)] |
| 49. | Chow AK, Ng L, Lam CS, Wong SK, Wan TM, Cheng NS, Yau TC, Poon RT, Pang RW. The Enhanced metastatic potential of hepatocellular carcinoma (HCC) cells with sorafenib resistance. PLoS One. 2013;8:e78675. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 100] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 50. | Chen KF, Chen HL, Tai WT, Feng WC, Hsu CH, Chen PJ, Cheng AL. Activation of phosphatidylinositol 3-kinase/Akt signaling pathway mediates acquired resistance to sorafenib in hepatocellular carcinoma cells. J Pharmacol Exp Ther. 2011;337:155-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 256] [Article Influence: 18.3] [Reference Citation Analysis (1)] |
| 51. | Casadei Gardini A, Frassineti GL, Foschi FG, Ercolani G, Ulivi P. Sorafenib and Regorafenib in HBV- or HCV-positive hepatocellular carcinoma patients: Analysis of RESORCE and SHARP trials. Dig Liver Dis. 2017;49:943-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 52. | Luo XY, Wu KM, He XX. Advances in drug development for hepatocellular carcinoma: clinical trials and potential therapeutic targets. J Exp Clin Cancer Res. 2021;40:172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 141] [Article Influence: 35.3] [Reference Citation Analysis (0)] |
| 53. | Yang P, Yang Y, Sun P, Tian Y, Gao F, Wang C, Zong T, Li M, Zhang Y, Yu T, Jiang Z. βII spectrin (SPTBN1): biological function and clinical potential in cancer and other diseases. Int J Biol Sci. 2021;17:32-49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 54. | Rao S, Yang X, Ohshiro K, Zaidi S, Wang Z, Shetty K, Xiang X, Hassan MI, Mohammad T, Latham PS, Nguyen BN, Wong L, Yu H, Al-Abed Y, Mishra B, Vacca M, Guenigault G, Allison MED, Vidal-Puig A, Benhammou JN, Alvarez M, Pajukanta P, Pisegna JR, Mishra L. β2-spectrin (SPTBN1) as a therapeutic target for diet-induced liver disease and preventing cancer development. Sci Transl Med. 2021;13:eabk2267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 55. | Craig AJ, Garcia-Lezana T, Ruiz de Galarreta M, Villacorta-Martin C, Kozlova EG, Martins-Filho SN, von Felden J, Ahsen ME, Bresnahan E, Hernandez-Meza G, Labgaa I, D'Avola D, Schwartz M, Llovet JM, Sia D, Thung S, Losic B, Lujambio A, Villanueva A. Transcriptomic characterization of cancer-testis antigens identifies MAGEA3 as a driver of tumor progression in hepatocellular carcinoma. PLoS Genet. 2021;17:e1009589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 56. | Yu M, Luo H, Fan M, Wu X, Shi B, Di S, Liu Y, Pan Z, Jiang H, Li Z. Development of GPC3-Specific Chimeric Antigen Receptor-Engineered Natural Killer Cells for the Treatment of Hepatocellular Carcinoma. Mol Ther. 2018;26:366-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 140] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 57. | Ma YS, Liu JB, Wu TM, Fu D. New Therapeutic Options for Advanced Hepatocellular Carcinoma. Cancer Control. 2020;27:1073274820945975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 58. | Sun Q, Hu Y, Gu Y, Huang J, He J, Luo L, Yang Y, Yin S, Dou C, Wang T, Fu X, He L, Qi S, Zhu X, Yang S, Wei X, Cheng W. Deciphering the regulatory and catalytic mechanisms of an unusual SAM-dependent enzyme. Signal Transduct Target Ther. 2019;4:17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 59. | Gao W, Tang Z, Zhang YF, Feng M, Qian M, Dimitrov DS, Ho M. Immunotoxin targeting glypican-3 regresses liver cancer via dual inhibition of Wnt signalling and protein synthesis. Nat Commun. 2015;6:6536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 107] [Cited by in RCA: 116] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 60. | Hassan R, Alewine C, Pastan I. New Life for Immunotoxin Cancer Therapy. Clin Cancer Res. 2016;22:1055-1058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 46] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 61. | Zhang Y, Xia M, Jin K, Wang S, Wei H, Fan C, Wu Y, Li X, Li G, Zeng Z, Xiong W. Function of the c-Met receptor tyrosine kinase in carcinogenesis and associated therapeutic opportunities. Mol Cancer. 2018;17:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 301] [Cited by in RCA: 391] [Article Influence: 55.9] [Reference Citation Analysis (0)] |
| 62. | Lin Y, Fang ZP, Liu HJ, Wang LJ, Cheng Z, Tang N, Li T, Liu T, Han HX, Cao G, Liang L, Ding YQ, Zhou WJ. HGF/R-spondin1 rescues liver dysfunction through the induction of Lgr5+ liver stem cells. Nat Commun. 2017;8:1175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 63. | Wang D, Saga Y, Sato N, Nakamura T, Takikawa O, Mizukami H, Matsubara S, Fujiwara H. The hepatocyte growth factor antagonist NK4 inhibits indoleamine-2,3-dioxygenase expression via the c-Met-phosphatidylinositol 3-kinase-AKT signaling pathway. Int J Oncol. 2016;48:2303-2309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 64. | Ganguly R, Kumar R, Pandey AK. Baicalin provides protection against fluoxetine-induced hepatotoxicity by modulation of oxidative stress and inflammation. World J Hepatol. 2022;14:729-743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 65. | Nelson GM, Ahlborn GJ, Allen JW, Ren H, Corton JC, Waalkes MP, Kitchin KT, Diwan BA, Knapp G, Delker DA. Transcriptional changes associated with reduced spontaneous liver tumor incidence in mice chronically exposed to high dose arsenic. Toxicology. 2009;266:6-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 66. | Pope ED 3rd, Kimbrough EO, Vemireddy LP, Surapaneni PK, Copland JA 3rd, Mody K. Aberrant lipid metabolism as a therapeutic target in liver cancer. Expert Opin Ther Targets. 2019;23:473-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 134] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 67. | Fritz V, Benfodda Z, Rodier G, Henriquet C, Iborra F, Avancès C, Allory Y, de la Taille A, Culine S, Blancou H, Cristol JP, Michel F, Sardet C, Fajas L. Abrogation of de novo lipogenesis by stearoyl-CoA desaturase 1 inhibition interferes with oncogenic signaling and blocks prostate cancer progression in mice. Mol Cancer Ther. 2010;9:1740-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 218] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 68. | Ference BA, Ray KK, Catapano AL, Ference TB, Burgess S, Neff DR, Oliver-Williams C, Wood AM, Butterworth AS, Di Angelantonio E, Danesh J, Kastelein JJP, Nicholls SJ. Mendelian Randomization Study of ACLY and Cardiovascular Disease. N Engl J Med. 2019;380:1033-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 243] [Article Influence: 40.5] [Reference Citation Analysis (0)] |
| 69. | Falvella FS, Pascale RM, Gariboldi M, Manenti G, De Miglio MR, Simile MM, Dragani TA, Feo F. Stearoyl-CoA desaturase 1 (Scd1) gene overexpression is associated with genetic predisposition to hepatocarcinogenesis in mice and rats. Carcinogenesis. 2002;23:1933-1936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 70. | Rimassa L, Assenat E, Peck-Radosavljevic M, Pracht M, Zagonel V, Mathurin P, Rota Caremoli E, Porta C, Daniele B, Bolondi L, Mazzaferro V, Harris W, Damjanov N, Pastorelli D, Reig M, Knox J, Negri F, Trojan J, López López C, Personeni N, Decaens T, Dupuy M, Sieghart W, Abbadessa G, Schwartz B, Lamar M, Goldberg T, Shuster D, Santoro A, Bruix J. Tivantinib for second-line treatment of MET-high, advanced hepatocellular carcinoma (METIV-HCC): a final analysis of a phase 3, randomised, placebo-controlled study. Lancet Oncol. 2018;19:682-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 306] [Article Influence: 43.7] [Reference Citation Analysis (0)] |
| 71. | Kumar S, Pandey AK. Oxidative stress-related microRNAs as diagnostic markers: A newer insight in diagnostics. In: Oxidative Stress: Diagnostic Methods and Applications in Medical Science. Eds. Maurya PK, Chandra P, Springer Nature, Singapore. 2017; 113-125. [DOI] [Full Text] |
| 72. | Kumar S, Prajapati KS, Singh AK, Kushwaha PP, Shuaib M, Gupta S. Long non-coding RNA regulating androgen receptor signaling in breast and prostate cancer. Cancer Lett. 2021;504:15-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 73. | Callegari E, Elamin BK, Giannone F, Milazzo M, Altavilla G, Fornari F, Giacomelli L, D'Abundo L, Ferracin M, Bassi C, Zagatti B, Corrà F, Miotto E, Lupini L, Bolondi L, Gramantieri L, Croce CM, Sabbioni S, Negrini M. Liver tumorigenicity promoted by microRNA-221 in a mouse transgenic model. Hepatology. 2012;56:1025-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 134] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 74. | Yerukala Sathipati S, Ho SY. Novel miRNA signature for predicting the stage of hepatocellular carcinoma. Sci Rep. 2020;10:14452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 75. | Dai X, Huang R, Hu S, Zhou Y, Sun X, Gui P, Yu Z, Zhou P. A novel miR-0308-3p revealed by miRNA-seq of HBV-positive hepatocellular carcinoma suppresses cell proliferation and promotes G1/S arrest by targeting double CDK6/Cyclin D1 genes. Cell Biosci. 2020;10:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 76. | Shao S, Hu Q, Wu W, Wang M, Huang J, Zhao X, Tang G, Liang T. Tumor-triggered personalized microRNA cocktail therapy for hepatocellular carcinoma. Biomater Sci. 2020;8:6579-6591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 77. | Wang S, Xu M, Sun Z, Yu X, Deng Y, Chang H. LINC01018 confers a novel tumor suppressor role in hepatocellular carcinoma through sponging microRNA-182-5p. Am J Physiol Gastrointest Liver Physiol. 2019;317:G116-G126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 78. | Santopaolo F, Lenci I, Milana M, Manzia TM, Baiocchi L. Liver transplantation for hepatocellular carcinoma: Where do we stand? World J Gastroenterol. 2019;25:2591-2602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 83] [Cited by in RCA: 78] [Article Influence: 13.0] [Reference Citation Analysis (3)] |
| 79. | Saito H, Fukuhara T, Furuya N, Watanabe K, Sugawara S, Iwasawa S, Tsunezuka Y, Yamaguchi O, Okada M, Yoshimori K, Nakachi I, Gemma A, Azuma K, Kurimoto F, Tsubata Y, Fujita Y, Nagashima H, Asai G, Watanabe S, Miyazaki M, Hagiwara K, Nukiwa T, Morita S, Kobayashi K, Maemondo M. Erlotinib plus bevacizumab versus erlotinib alone in patients with EGFR-positive advanced non-squamous non-small-cell lung cancer (NEJ026): interim analysis of an open-label, randomised, multicentre, phase 3 trial. Lancet Oncol. 2019;20:625-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 462] [Article Influence: 77.0] [Reference Citation Analysis (0)] |
| 80. | Wang L, Ai J, Shen Y, Zhang H, Peng X, Huang M, Zhang A, Ding J, Geng M. SOMCL-863, a novel, selective and orally bioavailable small-molecule c-Met inhibitor, exhibits antitumor activity both in vitro and in vivo. Cancer Lett. 2014;351:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 81. | Sharma AK, Kumar S, Chashoo G, Saxena AK, Pandey AK. Cell cycle inhibitory activity of Piper longum against A549 cell line and its protective effect against metal-induced toxicity in rats. Indian J Biochem Biophys. 2014;51:358-364. [PubMed] |
| 82. | Fan G, Wei X, Xu X. Is the era of sorafenib over? Ther Adv Med Oncol. 2020;12:1758835920927602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 83. | Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, Xu J, Bai Y, Chi Y, Wang L, Yeh KH, Bi F, Cheng Y, Le AT, Lin JK, Liu T, Ma D, Kappeler C, Kalmus J, Kim TW; CONCUR Investigators. Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2015;16:619-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 455] [Cited by in RCA: 569] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 84. | Di Giacomo S, Briz O, Monte MJ, Sanchez-Vicente L, Abete L, Lozano E, Mazzanti G, Di Sotto A, Marin JJG. Chemosensitization of hepatocellular carcinoma cells to sorafenib by β-caryophyllene oxide-induced inhibition of ABC export pumps. Arch Toxicol. 2019;93:623-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 85. | Casadei-Gardini A, Scartozzi M, Tada T, Yoo C, Shimose S, Masi G, Lonardi S, Frassineti LG, Nicola S, Piscaglia F, Kumada T, Kim HD, Koga H, Vivaldi C, Soldà C, Hiraoka A, Bang Y, Atsukawa M, Torimura T, Tsuj K, Itobayashi E, Toyoda H, Fukunishi S, Rimassa L, Rimini M, Cascinu S, Cucchetti A. Lenvatinib versus sorafenib in first-line treatment of unresectable hepatocellular carcinoma: An inverse probability of treatment weighting analysis. Liver Int. 2021;41:1389-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 86. | Marino D, Zichi C, Audisio M, Sperti E, Di Maio M. Second-line treatment options in hepatocellular carcinoma. Drugs Context. 2019;8:212577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 87. | Bruix J, Qin S, Merle P, Granito A, Huang Y-H, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki J-P, Ollivier-Hourmand I, Kudo M, Cheng A-L, Llovet JM, Finn RS, LeBerre M-A, Baumhauer A, Meinhardt G, Han G. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a 33andomized, double-blind, placebo-controlled, phase 3 trial. The Lancet 2017; 389: 56–66. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2160] [Cited by in RCA: 2717] [Article Influence: 339.6] [Reference Citation Analysis (0)] |
| 88. | Duffy AG, Greten TF. Liver cancer: Regorafenib as second-line therapy in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2017;14:141-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 89. | Zhu AX, Kang YK, Yen CJ, Finn RS, Galle PR, Llovet JM, Assenat E, Brandi G, Pracht M, Lim HY, Rau KM, Motomura K, Ohno I, Merle P, Daniele B, Shin DB, Gerken G, Borg C, Hiriart JB, Okusaka T, Morimoto M, Hsu Y, Abada PB, Kudo M; REACH-2 study investigators. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20:282-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1027] [Cited by in RCA: 1251] [Article Influence: 208.5] [Reference Citation Analysis (0)] |
| 90. | Ikeda K, Kudo M, Kawazoe S, Osaki Y, Ikeda M, Okusaka T, Tamai T, Suzuki T, Hisai T, Hayato S, Okita K, Kumada H. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J Gastroenterol. 2017;52:512-519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 265] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 91. | Gupta B, Rai R, Oertel M, Raeman R. Intestinal Barrier Dysfunction in Fatty Liver Disease: Roles of Microbiota, Mucosal Immune System, and Bile Acids. Semin Liver Dis. 2022;42:122-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 92. | Yu LX, Schwabe RF. The gut microbiome and liver cancer: mechanisms and clinical translation. Nat Rev Gastroenterol Hepatol. 2017;14:527-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 414] [Article Influence: 51.8] [Reference Citation Analysis (0)] |
| 93. | Zhang C, Yang M, Ericsson AC. The Potential Gut Microbiota-Mediated Treatment Options for Liver Cancer. Front Oncol. 2020;10:524205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 94. | Singh AK, Cabral C, Kumar R, Ganguly R, Kumar Rana H, Gupta A, Rosaria Lauro M, Carbone C, Reis F, Pandey AK. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients. 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 252] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 95. | Wei X, Jiang S, Chen Y, Zhao X, Li H, Lin W, Li B, Wang X, Yuan J, Sun Y. Cirrhosis related functionality characteristic of the fecal microbiota as revealed by a metaproteomic approach. BMC Gastroenterol. 2016;16:121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 96. | Dapito DH, Mencin A, Gwak GY, Pradere JP, Jang MK, Mederacke I, Caviglia JM, Khiabanian H, Adeyemi A, Bataller R, Lefkowitch JH, Bower M, Friedman R, Sartor RB, Rabadan R, Schwabe RF. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell. 2012;21:504-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 854] [Cited by in RCA: 1029] [Article Influence: 79.2] [Reference Citation Analysis (0)] |
| 97. | Lv LX, Fang DQ, Shi D, Chen DY, Yan R, Zhu YX, Chen YF, Shao L, Guo FF, Wu WR, Li A, Shi HY, Jiang XW, Jiang HY, Xiao YH, Zheng SS, Li LJ. Alterations and correlations of the gut microbiome, metabolism and immunity in patients with primary biliary cirrhosis. Environ Microbiol. 2016;18:2272-2286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 183] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 98. | Delmas D, Jannin B, Cherkaoui Malki M, Latruffe N. Inhibitory effect of resveratrol on the proliferation of human and rat hepatic derived cell lines. Oncol Rep. 2000;7:847-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 99. | Notas G, Nifli AP, Kampa M, Vercauteren J, Kouroumalis E, Castanas E. Resveratrol exerts its antiproliferative effect on HepG2 hepatocellular carcinoma cells, by inducing cell cycle arrest, and NOS activation. Biochim Biophys Acta. 2006;1760:1657-1666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 84] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 100. | Roncoroni L, Elli L, Dolfini E, Erba E, Dogliotti E, Terrani C, Doneda L, Grimoldi MG, Bardella MT. Resveratrol inhibits cell growth in a human cholangiocarcinoma cell line. Liver Int. 2008;28:1426-1436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 101. | Zhou X, Zhao Y, Wang J, Wang X, Chen C, Yin D, Zhao F, Yin J, Guo M, Zhang L, Du L, Zhang B, Yin X. Resveratrol represses estrogen-induced mammary carcinogenesis through NRF2-UGT1A8-estrogen metabolic axis activation. Biochem Pharmacol. 2018;155:252-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 102. | Bhullar KS, Jha A, Rupasinghe HP. Novel carbocyclic curcumin analog CUR3d modulates genes involved in multiple apoptosis pathways in human hepatocellular carcinoma cells. Chem Biol Interact. 2015;242:107-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 103. | Herrero de la Parte B, Rodeño-Casado M, Iturrizaga Correcher S, Mar Medina C, García-Alonso I. Curcumin Reduces Colorectal Cancer Cell Proliferation and Migration and Slows In Vivo Growth of Liver Metastases in Rats. Biomedicines. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 104. | Guo RX, Fu X, Chen J, Zhou L, Chen G. Preparation and Characterization of Microemulsions of Myricetin for Improving Its Antiproliferative and Antioxidative Activities and Oral Bioavailability. J Agric Food Chem. 2016;64:6286-6294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 105. | Yu MH, Im HG, Kim HI, Lee IS. Induction of apoptosis by immature plum in human hepatocellular carcinoma. J Med Food. 2009;12:518-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 106. | Kushwaha PP, Kumar R, Neog PR, Behara MR, Kumar A, Prajapti KS, Singh AK, Shuaib M, Sharma AK, Pandey AK, Kumar S. Characterization of phytochemicals and validation of antioxidant and anticancer activity in some Indian polyherbal ayurvedic products. Vegetos. 2021;34:286-299. [RCA] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 107. | Ng KT, Guo DY, Cheng Q, Geng W, Ling CC, Li CX, Liu XB, Ma YY, Lo CM, Poon RT, Fan ST, Man K. A garlic derivative, S-allylcysteine (SAC), suppresses proliferation and metastasis of hepatocellular carcinoma. PLoS One. 2012;7:e31655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 108. | Prasad S, Tyagi AK. Ginger and its constituents: role in prevention and treatment of gastrointestinal cancer. Gastroenterol Res Pract. 2015;2015:142979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 180] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 109. | Zhang D, Zhang J, Zeng J, Li Z, Zuo H, Huang C, Zhao X. Nano-Gold Loaded with Resveratrol Enhance the Anti-Hepatoma Effect of Resveratrol In Vitro and In Vivo. J Biomed Nanotechnol. 2019;15:288-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 110. | Liu HS, Pan CE, Yang W, Liu XM. Antitumor and immunomodulatory activity of resveratrol on experimentally implanted tumor of H22 in Balb/c mice. World J Gastroenterol. 2003;9:1474-1476. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 51] [Cited by in RCA: 47] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 111. | Rajasekaran D, Elavarasan J, Sivalingam M, Ganapathy E, Kumar A, Kalpana K, Sakthisekaran D. Resveratrol interferes with N-nitrosodiethylamine-induced hepatocellular carcinoma at early and advanced stages in male Wistar rats. Mol Med Rep. 2011;4:1211-1217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 112. | Gao F, Deng G, Liu W, Zhou K, Li M. Resveratrol suppresses human hepatocellular carcinoma via targeting HGF-c-Met signaling pathway. Oncol Rep. 2017;37:1203-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 113. | Venditti I, Iucci G, Fratoddi I, Cipolletti M, Montalesi E, Marino M, Secchi V, Battocchio C. Direct Conjugation of Resveratrol on Hydrophilic Gold Nanoparticles: Structural and Cytotoxic Studies for Biomedical Applications. Nanomaterials (Basel). 2020;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 114. | Amagase H, Sun B, Borek C. Lycium barbarum (goji) juice improves in vivo antioxidant biomarkers in serum of healthy adults. Nutr Res. 2009;29:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 188] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 115. | Lin YC, Kuo JY, Hsu CC, Tsai WC, Li WC, Yu MC, Wen HW. Optimizing manufacture of liposomal berberine with evaluation of its antihepatoma effects in a murine xenograft model. Int J Pharm. 2013;441:381-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 116. | Ji X, Shen Y, Sun H, Gao X. A novel anti-alpha-fetoprotein single-chain variable fragment displays anti-tumor effects in HepG2 cells as a single agent or in combination with paclitaxel. Tumour Biol. 2016;37:10085-10096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 117. | Anwar-Mohamed A, El-Kadi AO. Sulforaphane induces CYP1A1 mRNA, protein, and catalytic activity levels via an AhR-dependent pathway in murine hepatoma Hepa 1c1c7 and human HepG2 cells. Cancer Lett. 2009;275:93-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 118. | Jo JY, de Mejia EG, Lila MA. Cytotoxicity of bioactive polymeric fractions from grape cell culture on human hepatocellular carcinoma, murine leukemia and non-cancerous PK15 kidney cells. Food Chem Toxicol. 2006;44:1758-1767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 119. | Scola G, Fernandes Correia Laurino CC, Menin E, Salvador M. Suppression of oncoprotein Her-2 and DNA damage after treatment with Flavan-3- ol vitis labrusca extract. Anticancer Agents Med Chem. 2013;13:1088-1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 120. | Hafidh RR, Abdulamir AS, Bakar FA, Jalilian FA, Abas F, Sekawi Z. Novel molecular, cytotoxical, and immunological study on promising and selective anticancer activity of mung bean sprouts. BMC Complement Altern Med. 2012;12:208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 121. | Chen CY, Liu TZ, Chen CH, Wu CC, Cheng JT, Yiin SJ, Shih MK, Wu MJ, Chern CL. Isoobtusilactone A-induced apoptosis in human hepatoma Hep G2 cells is mediated via increased NADPH oxidase-derived reactive oxygen species (ROS) production and the mitochondria-associated apoptotic mechanisms. Food Chem Toxicol. 2007;45:1268-1276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 122. | Weng CJ, Wu CF, Huang HW, Ho CT, Yen GC. Anti-invasion effects of 6-shogaol and 6-gingerol, two active components in ginger, on human hepatocarcinoma cells. Mol Nutr Food Res. 2010;54:1618-1627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 95] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 123. | Liu W, Huang XF, Qi Q, Dai QS, Yang L, Nie FF, Lu N, Gong DD, Kong LY, Guo QL. Asparanin A induces G(2)/M cell cycle arrest and apoptosis in human hepatocellular carcinoma HepG2 cells. Biochem Biophys Res Commun. 2009;381:700-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 124. | Friedman M, Levin CE, Lee SU, Kim HJ, Lee IS, Byun JO, Kozukue N. Tomatine-containing green tomato extracts inhibit growth of human breast, colon, liver, and stomach cancer cells. J Agric Food Chem. 2009;57:5727-5733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 79] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 125. | Gupta P, Bansal MP, Koul A. Evaluating the effect of lycopene from Lycopersicum esculentum on apoptosis during NDEA induced hepatocarcinogenesis. Biochem Biophys Res Commun. 2013;434:479-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 126. | Kim HJ, Yu MH, Lee IS. Inhibitory effects of methanol extract of plum (Prunus salicina L., cv. 'Soldam') fruits against benzo(alpha)pyrene-induced toxicity in mice. Food Chem Toxicol. 2008;46:3407-3413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 127. | Bishayee A, Bhatia D, Thoppil RJ, Darvesh AS, Nevo E, Lansky EP. Pomegranate-mediated chemoprevention of experimental hepatocarcinogenesis involves Nrf2-regulated antioxidant mechanisms. Carcinogenesis. 2011;32:888-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 81] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 128. | Hara A, Sakata K, Yamada Y, Kuno T, Kitaori N, Oyama T, Hirose Y, Murakami A, Tanaka T, Mori H. Suppression of beta-catenin mutation by dietary exposure of auraptene, a citrus antioxidant, in N,N-diethylnitrosamine-induced hepatocellular carcinomas in rats. Oncol Rep. 2005;14:345-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 129. | Lee J, Lee SJ, Lim KT. ZPDC glycoprotein (24 kDa) induces apoptosis and enhances activity of NK cells in N-nitrosodiethylamine-injected Balb/c. Cell Immunol. 2014;289:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 130. | Gupta A, Singh AK, Loka M, Pandey AK, Bishayee A. Ferulic acid-mediated modulation of apoptotic signaling pathways in cancer. Adv Protein Chem Struct Biol. 2021;125:215-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 131. | Stagos D, Amoutzias GD, Matakos A, Spyrou A, Tsatsakis AM, Kouretas D. Chemoprevention of liver cancer by plant polyphenols. Food Chem Toxicol. 2012;50:2155-2170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 132. | Mandlik DS, Mandlik SK. An Overview of Hepatocellular Carcinoma with Emphasis on Dietary Products and Herbal Remedies. Nutr Cancer. 2022;74:1549-1567. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 133. | Obeid MA, Gebril AM, Tate RJ, Mullen AB, Ferro VA. Comparison of the physical characteristics of monodisperse non-ionic surfactant vesicles (NISV) prepared using different manufacturing methods. Int J Pharm. 2017;521:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 134. | Zamboni WC, Torchilin V, Patri AK, Hrkach J, Stern S, Lee R, Nel A, Panaro NJ, Grodzinski P. Best practices in cancer nanotechnology: perspective from NCI nanotechnology alliance. Clin Cancer Res. 2012;18:3229-3241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 181] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 135. | Kumar S, Pandey AK. Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal. 2013;2013:162750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2843] [Cited by in RCA: 1982] [Article Influence: 165.2] [Reference Citation Analysis (1)] |
| 136. | Idée JM, Guiu B. Use of Lipiodol as a drug-delivery system for transcatheter arterial chemoembolization of hepatocellular carcinoma: a review. Crit Rev Oncol Hematol. 2013;88:530-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 154] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 137. | Luo LH, Zheng PJ, Nie H, Chen YC, Tong D, Chen J, Cheng Y. Pharmacokinetics and tissue distribution of docetaxel liposome mediated by a novel galactosylated cholesterol derivatives synthesized by lipase-catalyzed esterification in non-aqueous phase. Drug Deliv. 2016;23:1282-1290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 138. | Jiang PL, Lin HJ, Wang HW, Tsai WY, Lin SF, Chien MY, Liang PH, Huang YY, Liu DZ. Galactosylated liposome as a dendritic cell-targeted mucosal vaccine for inducing protective anti-tumor immunity. Acta Biomater. 2015;11:356-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 139. | Li WJ, Lian YW, Guan QS, Li N, Liang WJ, Liu WX, Huang YB, Cheng Y, Luo H. Liver-targeted delivery of liposome-encapsulated curcumol using galactosylated-stearate. Exp Ther Med. 2018;16:925-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 140. | Gera M, Kim N, Ghosh M, Sharma N, Huynh DL, Chandimali N, Koh H, Zhang JJ, Kang TY, Park YH, Kwon T, Jeong DK. Synthesis and evaluation of the antiproliferative efficacy of BRM270 phytocomposite nanoparticles against human hepatoma cancer cell lines. Mater Sci Eng C Mater Biol Appl. 2019;97:166-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 141. | Kumar R, Gupta A, Singh AK, Bishayee A, Pandey AK. The Antioxidant and Antihyperglycemic Activities of Bottlebrush Plant (Callistemon lanceolatus) Stem Extracts. Medicines (Basel). 2020;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 142. | Gupta A, Kumar R, Ganguly R, Singh AK, Rana HK, Pandey AK. Antioxidant, anti-inflammatory and hepatoprotective activities of Terminalia bellirica and its bioactive component ellagic acid against diclofenac induced oxidative stress and hepatotoxicity. Toxicol Rep. 2021;8:44-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |