Published online Jun 27, 2021. doi: 10.4254/wjh.v13.i6.650
Peer-review started: February 25, 2021
First decision: March 29, 2021
Revised: April 12, 2021
Accepted: May 22, 2021
Article in press: May 22, 2021
Published online: June 27, 2021
Processing time: 117 Days and 20.5 Hours
Rupture of gastric varices (GVs) can be fatal. Balloon-occluded retrograde transvenous obliteration (BRTO), as known as retrograde sclerotherapy, has been widely adopted for treatment of GVs because of its effectiveness, ability to cure, and utility in emergency and prophylactic treatment. Simplifying the route of blood flow from GVs to the gastrorenal shunt is important for the successful BRTO. This review outlines BRTO indications and contraindications, describes basic BRTO procedures and modifications, compares BRTO with other GVs treatments, and discusses various combination therapies. Combined BRTO and partial splenic embolization may prevent exacerbation of esophageal varices and shows promise as a treatment option.
Core Tip: Gastric varices (GVs) are a common complication of liver cirrhosis and their rupture is often fatal. Balloon-occluded retrograde transvenous obliteration (BRTO) has been widely adopted for treatment of GVs because of its effectiveness, ability to cure, and utility in emergency and prophylactic treatment. Various modifications of BRTO and combinations with other therapies are also beneficial. Combined BRTO and partial splenic embolization shows promise as a treatment option.
- Citation: Waguri N, Osaki A, Watanabe Y. Balloon-occluded retrograde transvenous obliteration for treatment of gastric varices. World J Hepatol 2021; 13(6): 650-661
- URL: https://www.wjgnet.com/1948-5182/full/v13/i6/650.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i6.650
Gastric fundal varices (GVs) and esophageal varices (EVs) are two of the main presentations of cirrhosis-induced portal hypertension. Although the bleeding risk of GVs is relatively low, their rupture is associated with high mortality (14%–45%)[1-4], because of their larger shunt diameter and higher flow. Hemodynamically, the two types of varices are completely different. The left and right gastric veins comprise the inflow of EVs, with the azygos vein system serving as the outflow. In contrast, the short and posterior gastric veins comprise the main inflow of GVs, although the left gastric vein may also be involved; the gastrorenal shunt (GRS), which drains blood to the left renal vein via the descending branch of the left inferior phrenic veins (80%–85%), and the gastrocaval shunt (GCS), which runs below the diaphragm and drains into the inferior vena cava (10%–15%) serve as outflow[5]. Eradication of GVs is difficult endoscopically because of the large diameter and high flow velocity of the shunts. Balloon-occluded retrograde transvenous obliteration (BRTO), developed by Kanagawa in 1996, is a sclerotherapy technique that approaches the varices from the outflow side of the GRS[6]. Since then, BRTO has been widely accepted in Japan[7-9], Asia, and the United States[10,11] as an effective treatment for GVs. In Europe, however, BRTO is not well recognized and not a treatment option for GVs[12,13]. In this review, we outline the indications and contraindications for BRTO, describe basic BRTO procedures and modifications, compare BRTO with other GVs treatments, and discuss various combination therapies.
According to Saad et al[14], the two clinical indications for BRTO are bleeding GVs (active, current, prior, and impending) and refractory hepatic encephalopathy involving the portosystemic shunt that forms GVs. Contraindications include: (1) Severe uncontrollable coagulopathy associated with liver failure; (2) Splenic vein thrombosis; (3) Portal vein thrombosis; and (4) Uncontrolled bleeding from EVs. In the case of uncontrolled bleeding from EVs, BRTO is contraindicated as a sole procedure; combined transjugular intrahepatic portosystemic shunt (TIPS) and BRTO or balloon-occluded antegrade transvenous obliteration (BATO) via the TIPS route are recommended instead.
We use BRTO for both emergency and elective treatment of ruptured GVs as well as prophylactic treatment according to the criteria described below[15,16]. Indications for prophylactic treatment of GVs include nodular form and red color spot lesions[17], increasing size over time, and hepatic encephalopathy. However, we do not treat patients with severe hepatic dysfunction (total bilirubin ≥ 4.0 mg/dL, Child-Pugh score ≥ 13), renal dysfunction (eGFR < 30 mL/min/1.73 m2), or other serious diseases with poor prognosis as well as those without a portosystemic shunt amenable to a retrograde approach[15]. We consider the presence of contrast agent flowing freely from the GRS into the portal vein on balloon-occluded retrograde venography (BRTV) a relative contraindication[16].
Although beta blockers are widely used to prevent bleeding in esophagogastric varices, based on a great deal of evidence[13,18], this review omits a description of them as its focus is interventional procedures.
TIPS is widely used in Western countries to treat portal hypertension in patients with esophagogastric varices and refractory ascites[19-24]. TIPS significantly reduces GVs rebleeding compared with pharmacotherapy and endoscopic treatments such as endoscopic variceal band ligation[19-21]. Although TIPS reduces portal venous pressure (PVP), GVs rebleeding and stent dysfunction are common[19-21,25]. Additionally, post-TIPS mortality is relatively high due to serious complications such as intraperitoneal hemorrhage, hemobilia, sepsis, hepatic failure, congestive heart failure, and others[25,26]. However, the use of polytetrafluoroethylene-covered stents has improved the TIPS patency rate[27] and the complication rate has decreased in conjunction with more widespread use. Preemptive TIPS is also recommended to prevent esophagogastric varices rebleeding[13,28].
Endoscopic injection of n-butyl-2-cyanoacrylate (CA) has also been widely used to treat GVs[29]. In patients with acute bleeding, CA injection is reportedly more effective than pharmacotherapy alone[30,31] and is the therapy of choice[32,33]; however, CA injection for elective treatment is not recommended and only used when no other treatment is available[32,33].
BRTO is highly effective to eradicate GVs[6-8,15,34] and can be effective for prophylactic[7-9,34] as well as emergency bleeding treatment[15,35,36]. Several studies have shown that BRTO is superior to endoscopic interventions in terms of bleeding control and prognosis in patients with GVs[35,37,38]. Furthermore, several comparative studies have reported that BRTO has a slight advantage over TIPS in terms of rebleeding, hepatic encephalopathy, hepatic functional reserve, and survival[39-44]. These studies are summarized in Tables 1-4. Table 1 shows the study design and sample size. Table 2 summarizes the sclerosant used for BRTO, types of stents used for TIPS, and the technical success rate of each procedure. Table 3 shows the rebleeding rates of GVs and EVs. Table 4 shows the notable complications after each procedure. Recent meta-analyses[45-47] have concluded that BRTO in patients with GVs bleeding is associated with lower rates of rebleeding and postprocedural hepatic encephalopathy, as well as better survival than TIPS. Although BRTO is effective in eradicating GVs, it is associated with complications such as postprocedural EVs, ectopic varices, and intractable ascites. Further debate over the relative superiority of BRTO or TIPS is not constructive. Rather, clinicians should fully understand the characteristics, risks, and benefits of each and use them suitably according to individual patient therapeutic needs. Clinicians should also consider using them in various therapeutic combinations.
| Ref. | Journal | Country | Study design | Number of cases | |
| BRTO | TIPS | ||||
| Choi et al[39] | KJR 2003 | South Korea | RCT, Single institution | 8 | 13 |
| Ninoi et al[40] | AJR 2004 | Japan | Retrospective, Single institution | 77 (BRTO: 49 / PTS: 28) | 27 |
| Sabri et al[41] | JVIR 2014 | United States | Retrospective, Single institution | 23 | 27 |
| Kim et al[42] | KJR 2017 | United States | Retrospective, Single institution | 25 | 27 |
| Lee et al[43] | JGH 2017 | South Korea | Retrospective, Two institutions | 95 | 47 |
| Gimm et al[44] | Gut and Liver 2018 | South Korea | Retrospective, Single institution | 157 | 19 |
| Ref. | BRTO | Tips | Technical success rate | |
| sclerosant | Stent type | BRTO | TIPS | |
| Choi et al[39] | EO | Bare | 8/8 | 13/13 |
| Ninoi et al[40] | EO | Bare | 49/58 | 27/27 |
| Sabri et al[41] | STS | Covered | 21/23 | 27/27 |
| Kim et al[42] | EO, STS | Covered | 22/25 | 27/27 |
| Lee et al[43] | EO, STS, polidocanol | Covered | 106/123 | 49/60 |
| Gimm et al[44] | EO, STS | Bare, covered | 159/166 | 19/22 |
| Total | 365/403 | 162/176 | ||
| 90.6%1 | 92.0%1 | |||
| Ref. | LF | HE | Ascites | EVs aggravation | ||||
| BRTO | TIPS | BRTO | TIPS | BRTO | TIPS | BRTO | TIPS | |
| Choi et al[39] | 0/8 | 1/13 | 0/8 | 1/13 | 0/8 | 0/13 | 1/8 | 0/13 |
| Ninoi et al[40] | 3/771 | 10/271 | 0/77 | 5/27 | 6/77 | 14/77 | ||
| Sabri et al[41] | 0/23 | 0/27 | 0/23 | 6/27 | ||||
| Kim et al[42] | 0/25 | 0/27 | 0/25 | 6/27 | 1/25 | 1/27 | 1/25 | 0/27 |
| Lee et al[43] | 0/95 | 1/47 | 0/95 | 14/47 | 13/95 | 2/47 | ||
| Gimm et al[44] | 0/157 | 0/19 | 4/157 | 0/19 | 48/157 | 1/19 | 22/157 | 1/19 |
BRTO drug preparation and procedures have been described in detail by Hirota et al[16]. In Japan, BRTO using ethanolamine oleate with iopamidol (EOI) became covered by insurance in 2018 after publication of a prospective multicenter clinical trial[48].
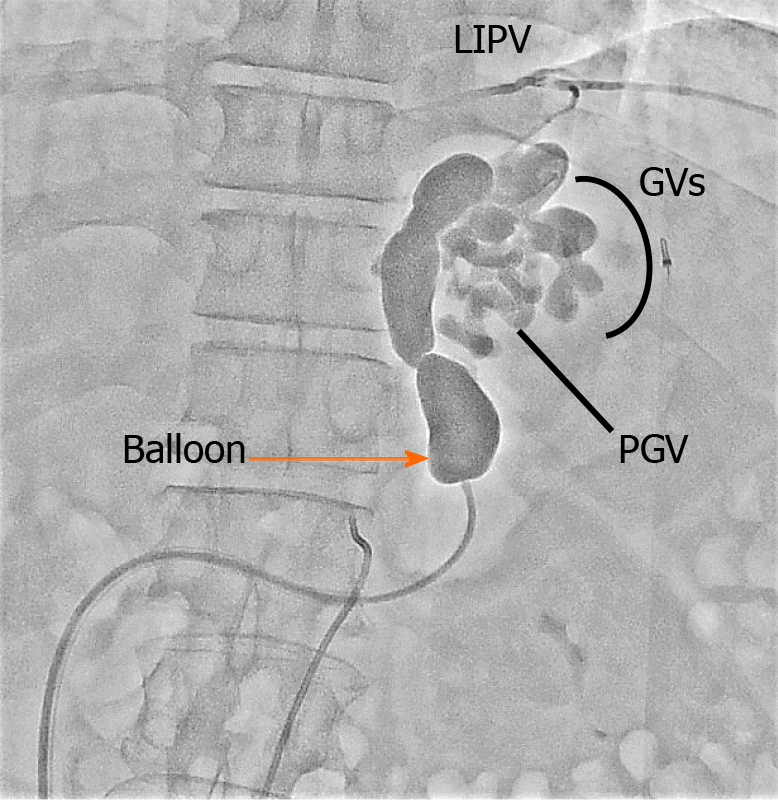
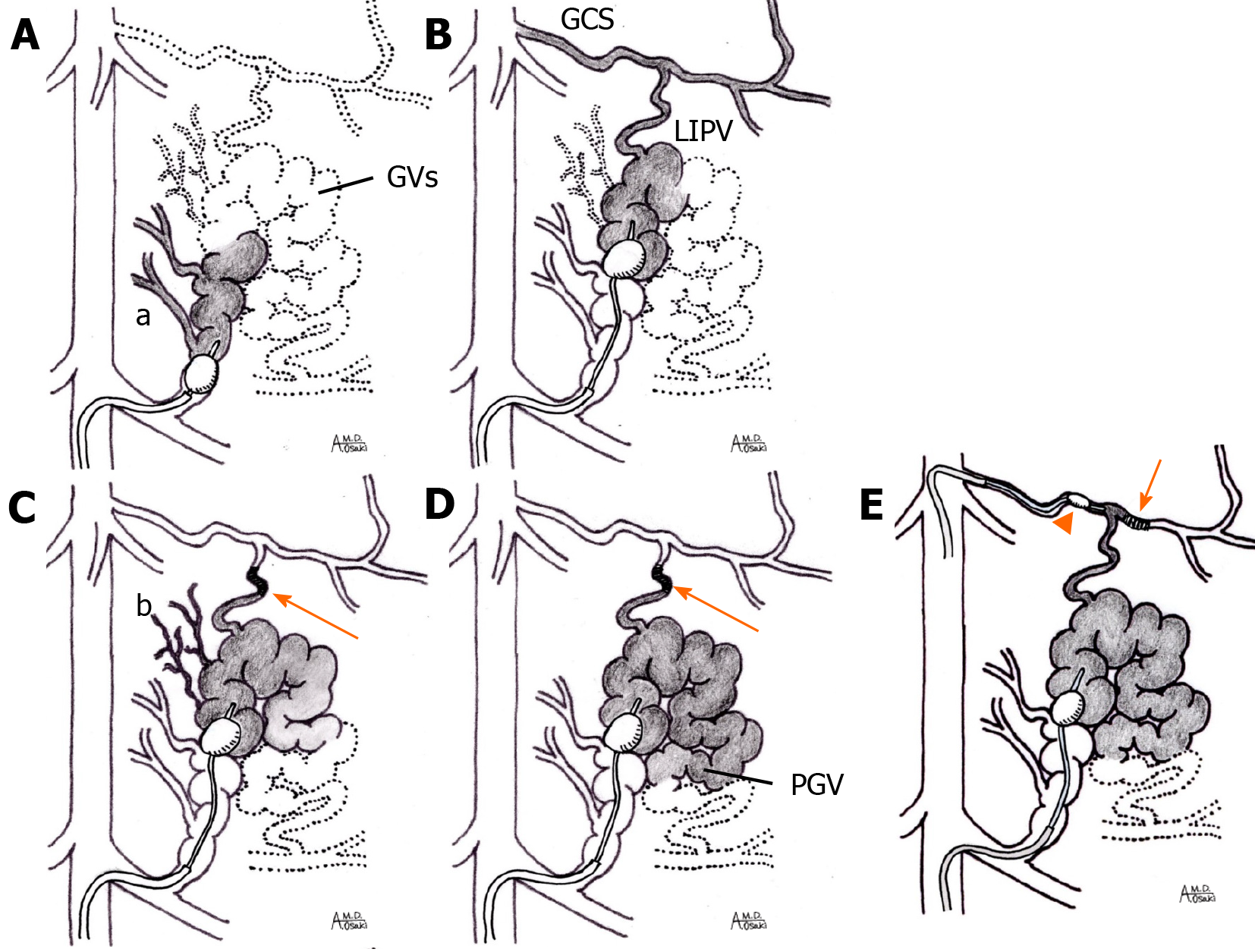
Our conventional BRTO method is described as follows[15,49]: GRS is diagnosed by computed tomography (CT). An 8 Fr long shepherd hook-shaped (Asato; Medikit, Tokyo, Japan) or cobra-shaped (S-one sheath; Terumo Clinical Supply Co., Gifu, Japan) sheath introducer is advanced into the left renal vein via the right femoral or internal jugular vein, respectively. A 6 Fr catheter with a 20 mm diameter balloon or 5.2 Fr catheter with a 9 mm diameter balloon (Selecon MP Catheter; Terumo Clinical Supply Co.) is then advanced into the GRS through the introducer in a retrograde fashion. BRTV (Figure 1) is then performed to identify shunts and their inflow and outflow. Before sclerosing the GRS, the route from the GVs to the GRS needs to be simplified. We use the down-grading method [50], selective coil embolization of the minor accessory draining veins[51], and/or the stepwise injection method [51] to down-grade the target shunt vessels to a relatively simple Hirota grade 1 or 2[52] (Figure 2A-D). If the coexisting GCS has a large diameter and selective coil embolization of the left inferior phrenic vein is impossible, the GCS is occluded with another balloon catheter[53] (Figure 2E). Under temporary balloon occlusion, contrast medium is injected via the balloon catheter to confirm stagnation of variceal flow for ≥ 10 min and evaluate the required volume of sclerosing solution. When stagnation of the contrast medium is confirmed, the same volume (10-40 mL) of 5% EOI is injected and remains stagnant in the vessels with overnight balloon occlusion. Human haptoglobin (4000 units) is administered prior to EOI injection to prevent acute kidney injury secondary to hemolysis caused by EOI[54]. The catheter is removed after overnight occlusion. Thrombosis of the GVs-GRS outflow (therapeutic effect) and thrombus formation elsewhere in the portal system (adverse effect) are confirmed by CT 3 to 7 d after BRTO. Eradication of GVs is confirmed by endoscopy after 2 to 3 mo.
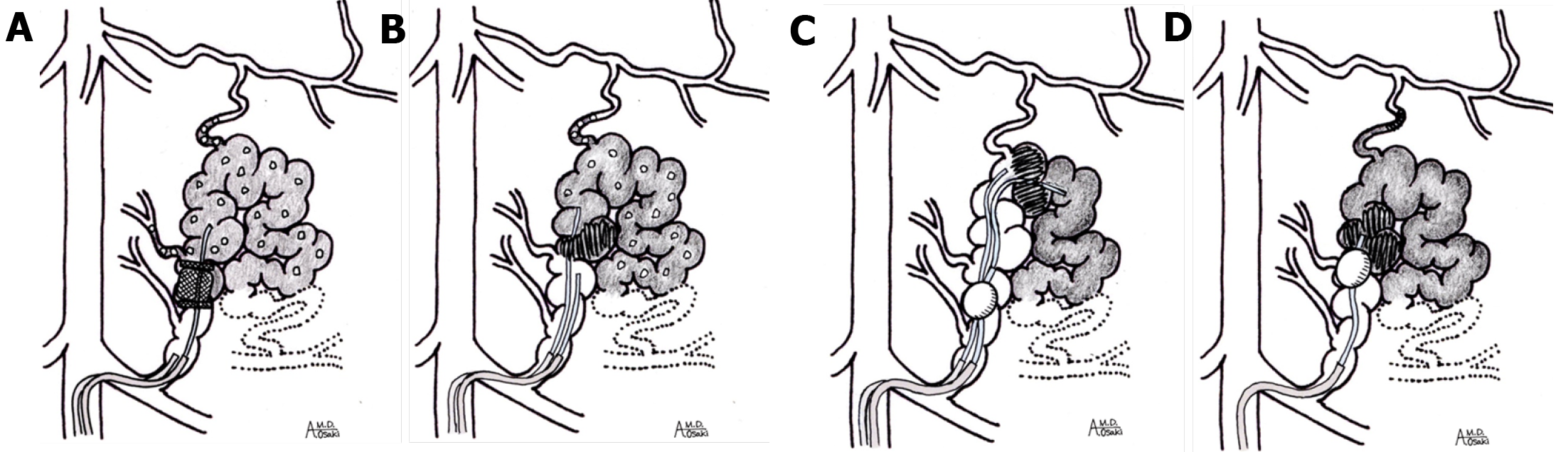
BRTO is commonly performed overnight to prevent the outflow of sclerosant into the systemic circulation[15,16,48]. Alternatively, a vascular plug[55] or microcoils[56] can be placed to occlude the GRS instead, allowing catheter system removal as soon as the treatment is complete (a single day procedure). The original methods of plug-associated retrograde transvenous obliteration (PARTO)[55] and coil-associated retrograde transvenous obliteration (CARTO)[56] (Figure 3A and B) emphasized their advantage of not requiring balloon catheters, sclerosants, or a long period of postprocedural bed rest and monitoring. However, these techniques have the disadvantage of high cost. By embolizing the small drainage vessels with gelatin particles, the selective coil embolization procedure can be omitted, and the procedure becomes easy and effective[55,57]. However, recurrence of GVs is lower when a surfactant such as sodium tetradecyl sulfate is used as a sclerosant in PARTO compared with use of gelatin alone[57]. Recurrence might be due to recanalization through the gelatin sponge which does not provide the permanent endothelial injury and thrombosis caused by sclerosants[58]. Injected gelatin has no direct effect on blood clot formation. Once the injected gelatin particles flow into the systemic circulation, they become emboli to the micro-vessels elsewhere. In contrast, sclerosant has a thrombus-forming effect on small drainage vessels, even in small amounts. However, if a small amount of sclerosant flows into the systemic circulation, it is often diluted with a large amount of blood and the effect of vascular endothelial damage can be ignored. Therefore, we believe that sclerosant should be used in BRTO rather than gelatin sponge alone.
Instead of downgrading by advancing the balloon catheter, a modified CARTO[59] in which embolization is performed using microcoils and sclerosant is injected upstream to the GVs has also been described (Figure 3C). Yamamoto et al[60] described CARTO-II, in which sclerosant is injected from a balloon catheter in the same manner as conventional BRTO, coil-embolization is performed just above the balloon (Figure 3D), and the balloon catheter is finally removed. In CARTO-II, thrombosis has already occurred due to vascular endothelial damage caused by the sclerosant, and coil-embolization is performed to prevent the thrombus from flowing to the systemic circulation after removing the balloon catheter. The same group also reported the utility of a mixture of low-dose gelatin sponge particles and 5% EOI in retrograde transvenous obliteration (GERTO)[61]. GERTO combines the advantages of gelatin particles and sclerosant, blocking small drainage vessels and causing reliable thrombosis via vascular endothelial damage.
Although these various BRTO modifications have appeared, their advantages and disadvantages have not yet been thoroughly evaluated. However, an advantage of both PARTO and CARTO is short indwelling balloon time; their disadvantage is high cost.
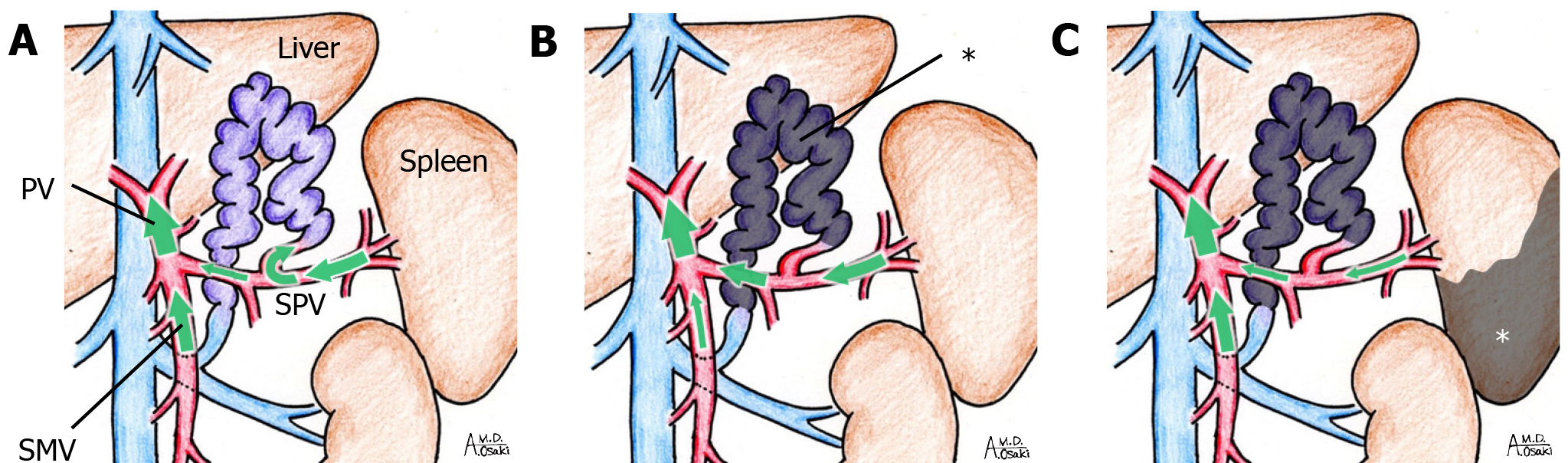
Various additional treatments have been performed in combination with BRTO. If BRTO alone is difficult, additional embolization of gastric vein inflow may be used to completely obliterate the GVs. Percutaneous transhepatic obliteration (PTO) may be used when conditions are unsuitable for BRTO, such as GVs without GRS[40,50,62]. Combined BRTO and PTO can obstruct both the feeding and draining veins of GVs to completely retain the sclerosant with in GVs, which may provide better control of variceal blood flow than either procedure alone[63]. However, the drawback of shunt embolization, including BRTO and PTO, is an increase in PVP. Although BRTO is associated with a lower rate of GVs rebleeding than TIPS[39-44] or endoscopic intervention[37,38], the increased PVP may cause enlargement of EVs[64-66]. Saad et al[67] therefore proposed use of BATO via the TIPS route, combined TIPS and BRTO, or combined BATO and BRTO, depending on the clinical situation. A recent study[68] has proposed a modified method, balloon-assisted antegrade transvenous obliteration (BAATO), in which balloon occlusion of the GRS is performed in retrograde fashion followed by antegrade trans-TIPS catheter injection of CA rather than sclerosant. The distribution of CA in GVs can be controlled by modifying blood flow velocity via balloon size adjustment. Thus, BAATO might be valuable alternative option as well. Although, TIPS certainly offsets the increase in PVP caused by BATO and/or BRTO, it can cause hepatic encephalopathy. Partial splenic embolization (PSE) also has a PVP-reducing effect, although weaker than TIPS, and combination with BRTO can be effective[69]. We previously reported that PSE can diminish the increase in PVP after BRTO[49] and that combined BRTO and PSE is a safe and effective treatment for GVs[15]. PSE is technically easier than TIPS and can be performed rapidly. Furthermore, the incidence of EVs exacerbation is lower and improvement in hepatic functional reserve is greater after combined BRTO and PSE than BRTO alone[15]. Increased portal venous flow after BRTO leads to improvement in the hepatic functional reserve[65,70] and is mainly due to increased splenic venous blood flow (Figure 4A and B) without a substantial increase in hepatopetal mesenteric venous blood flow. We speculate that hepatopetal mesenteric venous blood flow increases after PSE decreases the splenic venous blood flow (Figure 4C), which results in improved hepatic functional reserve. PSE has a PVP-reducing effect and can prevent exacerbation of EVs after BRTO. However, PSE-related complications may occur. According to a systematic review of 30 articles[71], the incidence of post-embolic syndrome, pleural effusion, ascites, thrombosis (mainly portal thrombosis), splenic abscess/bacterial peritonitis, and death after PSE is 73.4%, 9.4%, 8.1%, 2.4%, 1.3%, and 1.0%, respectively. Underlying liver dysfunction and splenic infarction rate (infarcted splenic volume/total splenic volume) greater than 70% may be risk factors for major complications[71,72].
GVs rupture is potentially fatal. Although various GVs treatments have been reported, BRTO is widely used because of its effectiveness, ability to cure, and utility for both emergency and prophylactic treatment. Recent BRTO modifications and combinations with other therapies are also beneficial. Although BRTO combined with TIPS and BRTO combined with PSE seem promising, randomized trials have not been performed and serious complications may occur. Their use should be approached with caution.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Chiu KW, Simón-Talero M, Xiong B S-Editor: Liu M L-Editor: A P-Editor: Wang LL
| 1. | Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992;16:1343-1349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 848] [Article Influence: 25.7] [Reference Citation Analysis (42)] |
| 2. | Trudeau W, Prindiville T. Endoscopic injection sclerosis in bleeding gastric varices. Gastrointest Endosc. 1986;32:264-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 239] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 3. | Abdel-Wahab M, el-Ebidy G, Gad el-Hak N, Abou-Elenin A, Abou-Zid M, el-Ghawalby N, Sultan A, el-Fiky A, Fathy O, Ezzat F. Fundal varices: problem and management. Hepatogastroenterology. 1999;46:849-854. [PubMed] |
| 4. | Greig JD, Garden OJ, Anderson JR, Carter DC. Management of gastric variceal haemorrhage. Br J Surg. 1990;77:297-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Kiyosue H, Mori H, Matsumoto S, Yamada Y, Hori Y, Okino Y. Transcatheter obliteration of gastric varices. Part 1. Anatomic classification. Radiographics. 2003;23:911-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 112] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 6. | Kanagawa H, Mima S, Kouyama H, Gotoh K, Uchida T, Okuda K. Treatment of gastric fundal varices by balloon-occluded retrograde transvenous obliteration. J Gastroenterol Hepatol. 1996;11:51-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 389] [Cited by in RCA: 381] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 7. | Chikamori F, Kuniyoshi N, Shibuya S, Takase Y. Eight years of experience with transjugular retrograde obliteration for gastric varices with gastrorenal shunts. Surgery. 2001;129:414-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 52] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 8. | Ninoi T, Nishida N, Kaminou T, Sakai Y, Kitayama T, Hamuro M, Yamada R, Nakamura K, Arakawa T, Inoue Y. Balloon-occluded retrograde transvenous obliteration of gastric varices with gastrorenal shunt: long-term follow-up in 78 patients. AJR Am J Roentgenol. 2005;184:1340-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 191] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 9. | Takuma Y, Nouso K, Makino Y, Saito S, Shiratori Y. Prophylactic balloon-occluded retrograde transvenous obliteration for gastric varices in compensated cirrhosis. Clin Gastroenterol Hepatol. 2005;3:1245-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Cho SK, Shin SW, Lee IH, Do YS, Choo SW, Park KB, Yoo BC. Balloon-occluded retrograde transvenous obliteration of gastric varices: outcomes and complications in 49 patients. AJR Am J Roentgenol. 2007;189:W365-W372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 95] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 11. | Sabri SS, Swee W, Turba UC, Saad WE, Park AW, Al-Osaimi AM, Caldwell SH, Matsumoto AH, Angle JF. Bleeding gastric varices obliteration with balloon-occluded retrograde transvenous obliteration using sodium tetradecyl sulfate foam. J Vasc Interv Radiol. 2011;22:309-16; quiz 316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 12. | Weil D, Cervoni JP, Fares N, Rudler M, Bureau C, Plessier A, Dao T, Pauwels A, Thabut D, Castellani P, Oberti F, Carbonell N, Elkrief L, Di Martino V, Thevenot T; Club Francophone pour l’Etude de l’Hypertension Portale (CFEHTP). Management of gastric varices: a French national survey. Eur J Gastroenterol Hepatol. 2016;28:576-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2011] [Cited by in RCA: 2294] [Article Influence: 229.4] [Reference Citation Analysis (3)] |
| 14. | Saad WE, Kitanosono T, Koizumi J, Hirota S. The conventional balloon-occluded retrograde transvenous obliteration procedure: indications, contraindications, and technical applications. Tech Vasc Interv Radiol. 2013;16:101-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 15. | Waguri N, Osaki A, Ikarashi S, Ogawa M, Kuraoka N, Ogawa K, Sato M, Aiba T, Yoneyama O, Furukawa K, Sugimura K, Igarashi K. Simultaneous combined balloon-occluded retrograde transvenous obliteration and partial splenic embolization for gastric fundal varices. United European Gastroenterol J. 2016;4:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 16. | Hirota S, Kobayashi K, Kako Y, Takaki H, Yamakado K. Balloon-occluded retrograde transvenous obliteration of varices: focusing on the portal hemodynamics and the recent techniques. Hepatol Int. 2018;12:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | Tajiri T, Yoshida H, Obara K, Onji M, Kage M, Kitano S, Kokudo N, Kokubu S, Sakaida I, Sata M, Tajiri H, Tsukada K, Nonami T, Hashizume M, Hirota S, Murashima N, Moriyasu F, Saigenji K, Makuuchi H, Oho K, Yoshida T, Suzuki H, Hasumi A, Okita K, Futagawa S, Idezuki Y. General rules for recording endoscopic findings of esophagogastric varices (2nd edition). Dig Endosc. 2010;22:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 244] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 18. | Rodrigues SG, Mendoza YP, Bosch J. Beta-blockers in cirrhosis: Evidence-based indications and limitations. JHEP Rep. 2020;2:100063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 100] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 19. | Barange K, Péron JM, Imani K, Otal P, Payen JL, Rousseau H, Pascal JP, Joffre F, Vinel JP. Transjugular intrahepatic portosystemic shunt in the treatment of refractory bleeding from ruptured gastric varices. Hepatology. 1999;30:1139-1143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 119] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 20. | Luca A, D'Amico G, La Galla R, Midiri M, Morabito A, Pagliaro L. TIPS for prevention of recurrent bleeding in patients with cirrhosis: meta-analysis of randomized clinical trials. Radiology. 1999;212:411-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 135] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 21. | Papatheodoridis GV, Goulis J, Leandro G, Patch D, Burroughs AK. Transjugular intrahepatic portosystemic shunt compared with endoscopic treatment for prevention of variceal rebleeding: A meta-analysis. Hepatology. 1999;30:612-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 187] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 22. | García-Pagán JC, Caca K, Bureau C, Laleman W, Appenrodt B, Luca A, Abraldes JG, Nevens F, Vinel JP, Mössner J, Bosch J; Early TIPS (Transjugular Intrahepatic Portosystemic Shunt) Cooperative Study Group. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362:2370-2379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 826] [Cited by in RCA: 842] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 23. | Boyer TD, Haskal ZJ; American Association for the Study of Liver Diseases. The role of transjugular intrahepatic portosystemic shunt in the management of portal hypertension. Hepatology. 2005;41:386-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 298] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 24. | Krajina A, Hulek P, Fejfar T, Valek V. Quality improvement guidelines for Transjugular Intrahepatic Portosystemic Shunt (TIPS). Cardiovasc Intervent Radiol. 2012;35:1295-1300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 25. | Loffroy R, Estivalet L, Cherblanc V, Favelier S, Pottecher P, Hamza S, Minello A, Hillon P, Thouant P, Lefevre PH, Krausé D, Cercueil JP. Transjugular intrahepatic portosystemic shunt for the management of acute variceal hemorrhage. World J Gastroenterol. 2013;19:6131-6143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 25] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 26. | Luca A, Miraglia R, Maruzzelli L, D'Amico M, Tuzzolino F. Early Liver Failure after Transjugular Intrahepatic Portosystemic Shunt in Patients with Cirrhosis with Model for End-Stage Liver Disease Score of 12 or Less: Incidence, Outcome, and Prognostic Factors. Radiology. 2016;280:622-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 27. | Rossi P, Salvatori FM, Fanelli F, Bezzi M, Rossi M, Marcelli G, Pepino D, Riggio O, Passariello R. Polytetrafluoroethylene-covered nitinol stent-graft for transjugular intrahepatic portosystemic shunt creation: 3-year experience. Radiology. 2004;231:820-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 110] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 28. | Trebicka J, Gu W, Ibáñez-Samaniego L, Hernández-Gea V, Pitarch C, Garcia E, Procopet B, Giráldez Á, Amitrano L, Villanueva C, Thabut D, Silva-Junior G, Martinez J, Genescà J, Bureau C, Llop E, Laleman W, Palazon JM, Castellote J, Rodrigues S, Gluud L, Ferreira CN, Barcelo R, Cañete N, Rodríguez M, Ferlitsch A, Mundi JL, Gronbaek H, Hernández-Guerra M, Sassatelli R, Dell'Era A, Senzolo M, Abraldes JG, Romero-Gómez M, Zipprich A, Casas M, Masnou H, Primignani M, Weiss E, Catalina MV, Erasmus HP, Uschner FE, Schulz M, Brol MJ, Praktiknjo M, Chang J, Krag A, Nevens F, Calleja JL, Robic MA, Conejo I, Albillos A, Rudler M, Alvarado E, Guardascione MA, Tantau M, Bosch J, Torres F, Pavesi M, Garcia-Pagán JC, Jansen C, Bañares R; International Variceal Bleeding Observational Study Group and Baveno Cooperation. Rebleeding and mortality risk are increased by ACLF but reduced by pre-emptive TIPS. J Hepatol. 2020;73:1082-1091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 29. | Franco MC, Gomes GF, Nakao FS, de Paulo GA, Ferrari AP Jr, Libera ED Jr. Efficacy and safety of endoscopic prophylactic treatment with undiluted cyanoacrylate for gastric varices. World J Gastrointest Endosc. 2014;6:254-259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 30. | Khawaja A, Sonawalla AA, Somani SF, Abid S. Management of bleeding gastric varices: a single session of histoacryl injection may be sufficient. Eur J Gastroenterol Hepatol. 2014;26:661-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 31. | Mishra SR, Sharma BC, Kumar A, Sarin SK. Primary prophylaxis of gastric variceal bleeding comparing cyanoacrylate injection and beta-blockers: a randomized controlled trial. J Hepatol. 2011;54:1161-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 142] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 32. | Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1108] [Cited by in RCA: 1441] [Article Influence: 180.1] [Reference Citation Analysis (3)] |
| 33. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1777] [Cited by in RCA: 1820] [Article Influence: 260.0] [Reference Citation Analysis (2)] |
| 34. | Koito K, Namieno T, Nagakawa T, Morita K. Balloon-occluded retrograde transvenous obliteration for gastric varices with gastrorenal or gastrocaval collaterals. AJR Am J Roentgenol. 1996;167:1317-1320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 129] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 35. | Akahoshi T, Tomikawa M, Kamori M, Tsutsumi N, Nagao Y, Hashizume M, Maehara Y. Impact of balloon-occluded retrograde transvenous obliteration on management of isolated fundal gastric variceal bleeding. Hepatol Res. 2012;42:385-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 36. | Arai H, Abe T, Shimoda R, Takagi H, Yamada T, Mori M. Emergency balloon-occluded retrograde transvenous obliteration for gastric varices. J Gastroenterol. 2005;40:964-971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 51] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Hong CH, Kim HJ, Park JH, Park DI, Cho YK, Sohn CI, Jeon WK, Kim BI, Hong HP, Shin JH. Treatment of patients with gastric variceal hemorrhage: endoscopic N-butyl-2-cyanoacrylate injection vs balloon-occluded retrograde transvenous obliteration. J Gastroenterol Hepatol. 2009;24:372-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 92] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 38. | Emori K, Toyonaga A, Oho K, Kumamoto M, Haruta T, Inoue H, Morita Y, Mitsuyama K, Tsuruta O, Sata M. Balloon-occluded retrograde transvenous obliteration vs endoscopic injection sclerotherapy for isolated gastric varices: a comparative study. Kurume Med J. 2014;60:105-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 39. | Choi YH, Yoon CJ, Park JH, Chung JW, Kwon JW, Choi GM. Balloon-occluded retrograde transvenous obliteration for gastric variceal bleeding: its feasibility compared with transjugular intrahepatic portosystemic shunt. Korean J Radiol. 2003;4:109-116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 114] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 40. | Ninoi T, Nakamura K, Kaminou T, Nishida N, Sakai Y, Kitayama T, Hamuro M, Yamada R, Arakawa T, Inoue Y. TIPS vs transcatheter sclerotherapy for gastric varices. AJR Am J Roentgenol. 2004;183:369-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 100] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 41. | Sabri SS, Abi-Jaoudeh N, Swee W, Saad WE, Turba UC, Caldwell SH, Angle JF, Matsumoto AH. Short-term rebleeding rates for isolated gastric varices managed by transjugular intrahepatic portosystemic shunt vs balloon-occluded retrograde transvenous obliteration. J Vasc Interv Radiol. 2014;25:355-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 42. | Kim SK, Lee KA, Sauk S, Korenblat K. Comparison of Transjugular Intrahepatic Portosystemic Shunt with Covered Stent and Balloon-Occluded Retrograde Transvenous Obliteration in Managing Isolated Gastric Varices. Korean J Radiol. 2017;18:345-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 43. | Lee SJ, Kim SU, Kim MD, Kim YH, Kim GM, Park SI, Won JY, Lee DY, Lee KH. Comparison of treatment outcomes between balloon-occluded retrograde transvenous obliteration and transjugular intrahepatic portosystemic shunt for gastric variceal bleeding hemostasis. J Gastroenterol Hepatol. 2017;32:1487-1494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 44. | Gimm G, Chang Y, Kim HC, Shin A, Cho EJ, Lee JH, Yu SJ, Yoon JH, Kim YJ. Balloon-Occluded Retrograde Transvenous Obliteration vs Transjugular Intrahepatic Portosystemic Shunt for the Management of Gastric Variceal Bleeding. Gut Liver. 2018;12:704-713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 45. | Wang ZW, Liu JC, Zhao F, Zhang WG, Duan XH, Chen PF, Yang SF, Li HW, Chen FW, Shi HS, Ren JZ. Comparison of the Effects of TIPS vs BRTO on Bleeding Gastric Varices: A Meta-Analysis. Can J Gastroenterol Hepatol. 2020;2020:5143013. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 46. | Paleti S, Nutalapati V, Fathallah J, Jeepalyam S, Rustagi T. Balloon-Occluded Retrograde Transvenous Obliteration (BRTO) Versus Transjugular Intrahepatic Portosystemic Shunt (TIPS) for Treatment of Gastric Varices Because of Portal Hypertension: A Systematic Review and Meta-Analysis. J Clin Gastroenterol. 2020;54:655-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 47. | Yu Q, Liu C, Raissi D. Balloon-occluded Retrograde Transvenous Obliteration Versus Transjugular Intrahepatic Portosystemic Shunt for Gastric Varices: A Meta-Analysis. J Clin Gastroenterol. 2021;55:147-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 48. | Kobayakawa M, Kokubu S, Hirota S, Koizumi J, Nishida N, Yasumoto T, Mochida S, Hidaka H, Tanaka N, Tajima T. Short-Term Safety and Efficacy of Balloon-Occluded Retrograde Transvenous Obliteration Using Ethanolamine Oleate: Results of a Prospective, Multicenter, Single-Arm Trial. J Vasc Interv Radiol 2017; 28: 1108-1115. e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 49. | Waguri N, Hayashi M, Yokoo T, Sato R, Arao Y, Setsu T, Sato M, Kohisa J, Hama I, Ohsugi K, Aiba T, Yoneyama O, Furukawa K, Sugimura K, Igarashi K, Suda T. Simultaneous combined balloon-occluded retrograde transvenous obliteration and partial splenic embolization for portosystemic shunts. J Vasc Interv Radiol. 2012;23:650-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 50. | Fukuda T, Hirota S, Sugimoto K, Matsumoto S, Zamora CA, Sugimura K. "Downgrading" of gastric varices with multiple collateral veins in balloon-occluded retrograde transvenous obliteration. J Vasc Interv Radiol. 2005;16:1379-1383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 51. | Kiyosue H, Mori H, Matsumoto S, Yamada Y, Hori Y, Okino Y. Transcatheter obliteration of gastric varices: Part 2. Strategy and techniques based on hemodynamic features. Radiographics. 2003;23:921-37; discussion 937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 107] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 52. | Hirota S, Matsumoto S, Tomita M, Sako M, Kono M. Retrograde transvenous obliteration of gastric varices. Radiology. 1999;211:349-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 235] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 53. | Nishida N, Ninoi T, Kitayama T, Yamamoto A, Sakai Y, Sato K, Hamuro M, Nakamura K, Inoue Y, Yamada R. Dual balloon-occluded retrograde transvenous obliteration of gastric varix draining into the left adrenal vein and left inferior phrenic vein. Cardiovasc Intervent Radiol. 2004;27:560-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 54. | Hashizume M, Kitano S, Yamaga H, Sugimachi K. Haptoglobin to protect against renal damage from ethanolamine oleate sclerosant. Lancet. 1988;2:340-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 55. | Gwon DI, Ko GY, Yoon HK, Sung KB, Kim JH, Shin JH, Ko HK, Song HY. Gastric varices and hepatic encephalopathy: treatment with vascular plug and gelatin sponge-assisted retrograde transvenous obliteration--a primary report. Radiology. 2013;268:281-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 56. | Lee EW, Saab S, Gomes AS, Busuttil R, McWilliams J, Durazo F, Han SH, Goldstein L, Tafti BA, Moriarty J, Loh CT, Kee ST. Coil-Assisted Retrograde Transvenous Obliteration (CARTO) for the Treatment of Portal Hypertensive Variceal Bleeding: Preliminary Results. Clin Transl Gastroenterol. 2014;5:e61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 57. | Gwon DI, Kim YH, Ko GY, Kim JW, Ko HK, Kim JH, Shin JH, Yoon HK, Sung KB. Vascular Plug-Assisted Retrograde Transvenous Obliteration for the Treatment of Gastric Varices and Hepatic Encephalopathy: A Prospective Multicenter Study. J Vasc Interv Radiol. 2015;26:1589-1595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 68] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 58. | Kim YH, Kim YH, Kim CS, Kang UR, Kim SH, Kim JH. Comparison of Balloon-Occluded Retrograde Transvenous Obliteration (BRTO) Using Ethanolamine Oleate (EO), BRTO Using Sodium Tetradecyl Sulfate (STS) Foam and Vascular Plug-Assisted Retrograde Transvenous Obliteration (PARTO). Cardiovasc Intervent Radiol. 2016;39:840-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 59. | Uotani K, Matsushiro E, Hamanaka A, Yamasaki Y, Kushima T, Sugimoto K. Modified Coil-Assisted Retrograde Transvenous Obliteration (m-CARTO) for Gastric Varices. Cardiovasc Intervent Radiol. 2020;43:1100-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 60. | Yamamoto A, Jogo A, Kageyama K, Sohgawa E, Hamamoto S, Hamuro M, Kamino T, Miki Y. Utility of Coil-Assisted Retrograde Transvenous Obliteration II (CARTO-II) for the Treatment of Gastric Varices. Cardiovasc Intervent Radiol. 2020;43:565-571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 61. | Jogo A, Yamamoto A, Kaminoh T, Nakano M, Kageyama K, Sohgawa E, Hamamoto S, Sakai Y, Hamuro M, Nishida N, Miki Y. Utility of low-dose gelatin sponge particles and 5% ethanolamine oleate iopamidol mixture in retrograde transvenous obliteration (GERTO) for gastric varices. Br J Radiol. 2020;93:20190751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 62. | Ishikawa T, Imai M, Ko M, Sato H, Nozawa Y, Sano T, Iwanaga A, Seki K, Honma T, Yoshida T. Percutaneous transhepatic obliteration and percutaneous transhepatic sclerotherapy for intractable hepatic encephalopathy and gastric varices improves the hepatic function reserve. Biomed Rep. 2017;6:99-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 63. | Arai H, Abe T, Takagi H, Mori M. Efficacy of balloon-occluded retrograde transvenous obliteration, percutaneous transhepatic obliteration and combined techniques for the management of gastric fundal varices. World J Gastroenterol. 2006;12:3866-3873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 40] [Cited by in RCA: 54] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 64. | Elsamman MK, Fujiwara Y, Kameda N, Okazaki H, Tanigawa T, Shiba M, Tominaga K, Watanabe T, Oshitani N, Arafa UA, El-Sayed AA, Nakamura K, Arakawa T. Predictive factors of worsening of esophageal varices after balloon-occluded retrograde transvenous obliteration in patients with gastric varices. Am J Gastroenterol. 2009;104:2214-2221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 65. | Fukuda T, Hirota S, Sugimura K. Long-term results of balloon-occluded retrograde transvenous obliteration for the treatment of gastric varices and hepatic encephalopathy. J Vasc Interv Radiol. 2001;12:327-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 221] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 66. | Jogo A, Nishida N, Yamamoto A, Matsui H, Takeshita T, Sakai Y, Matsuoka T, Nakamura K, Miki Y. Factors associated with aggravation of esophageal varices after B-RTO for gastric varices. Cardiovasc Intervent Radiol. 2014;37:1243-1250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 67. | Saad WE, Kitanosono T, Koizumi J. Balloon-occluded antegrade transvenous obliteration with or without balloon-occluded retrograde transvenous obliteration for the management of gastric varices: concept and technical applications. Tech Vasc Interv Radiol. 2012;15:203-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 68. | Liu J, Yang C, Huang S, Zhou C, Shi Q, Qian K, Song S, Xiong B. The combination of balloon-assisted antegrade transvenous obliteration and transjugular intrahepatic portosystemic shunt for the management of cardiofundal varices hemorrhage. Eur J Gastroenterol Hepatol. 2020;32:656-662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 69. | Chikamori F, Kuniyoshi N, Kawashima T, Takase Y. Gastric varices with gastrorenal shunt: combined therapy using transjugular retrograde obliteration and partial splenic embolization. AJR Am J Roentgenol. 2008;191:555-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 70. | Miyamoto Y, Oho K, Kumamoto M, Toyonaga A, Sata M. Balloon-occluded retrograde transvenous obliteration improves liver function in patients with cirrhosis and portal hypertension. J Gastroenterol Hepatol. 2003;18:934-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 93] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 71. | Talwar A, Gabr A, Riaz A, Desai K, Thornburg B, Mouli S, Lewandowski RJ, Salem R. Adverse Events Related to Partial Splenic Embolization for the Treatment of Hypersplenism: A Systematic Review. J Vasc Interv Radiol 2020; 31: 1118-1131. e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 72. | Sakai T, Shiraki K, Inoue H, Sugimoto K, Ohmori S, Murata K, Takase K, Nakano T. Complications of partial splenic embolization in cirrhotic patients. Dig Dis Sci. 2002;47:388-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 75] [Article Influence: 3.3] [Reference Citation Analysis (0)] |