Published online Feb 27, 2021. doi: 10.4254/wjh.v13.i2.166
Peer-review started: June 27, 2020
First decision: November 4, 2020
Revised: December 2, 2020
Accepted: December 13, 2020
Article in press: December 13, 2020
Published online: February 27, 2021
Processing time: 242 Days and 20.1 Hours
The prognosis of patients with advanced or unresectable extrahepatic cholangiocarcinoma is poor. More than 50% of patients with jaundice are inoperable at the time of first diagnosis. Endoscopic treatment in patients with obstructive jaundice ensures bile duct drainage in preoperative or palliative settings. Relief of symptoms (pain, pruritus, jaundice) and improvement in quality of life are the aims of palliative therapy. Stent implantation by endoscopic retrograde cholangiopancreatography is generally preferred for long-term palliation. There is a vast variety of plastic and metal stents, covered or uncovered. The stent choice depends on the expected length of survival, quality of life, costs and physician expertise. This review will provide the framework for the endoscopic minimally invasive therapy in extrahepatic cholangiocarcinoma. Moreover, additional therapies, such as brachytherapy, photodynamic therapy, radiofrequency ablation, chemotherapy, molecular-targeted therapy and/or immunotherapy by the endoscopic approach, are the nonsurgical methods associated with survival improvement rate and/or local symptom palliation.
Core Tip: Cholangiocarcinoma is an aggressive tumor with a poor prognosis mainly due to its late diagnosis. The development of new minimally invasive techniques provides these patients a chance to relieve symptoms and attain a better quality of life. We herein discuss the palliation of obstructive jaundice by radiofrequency ablation, photodynamic therapy and brachytherapy in advanced extrahepatic cholangiocarcinoma.
- Citation: Tantau AI, Mandrutiu A, Pop A, Zaharie RD, Crisan D, Preda CM, Tantau M, Mercea V. Extrahepatic cholangiocarcinoma: Current status of endoscopic approach and additional therapies. World J Hepatol 2021; 13(2): 166-186
- URL: https://www.wjgnet.com/1948-5182/full/v13/i2/166.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i2.166
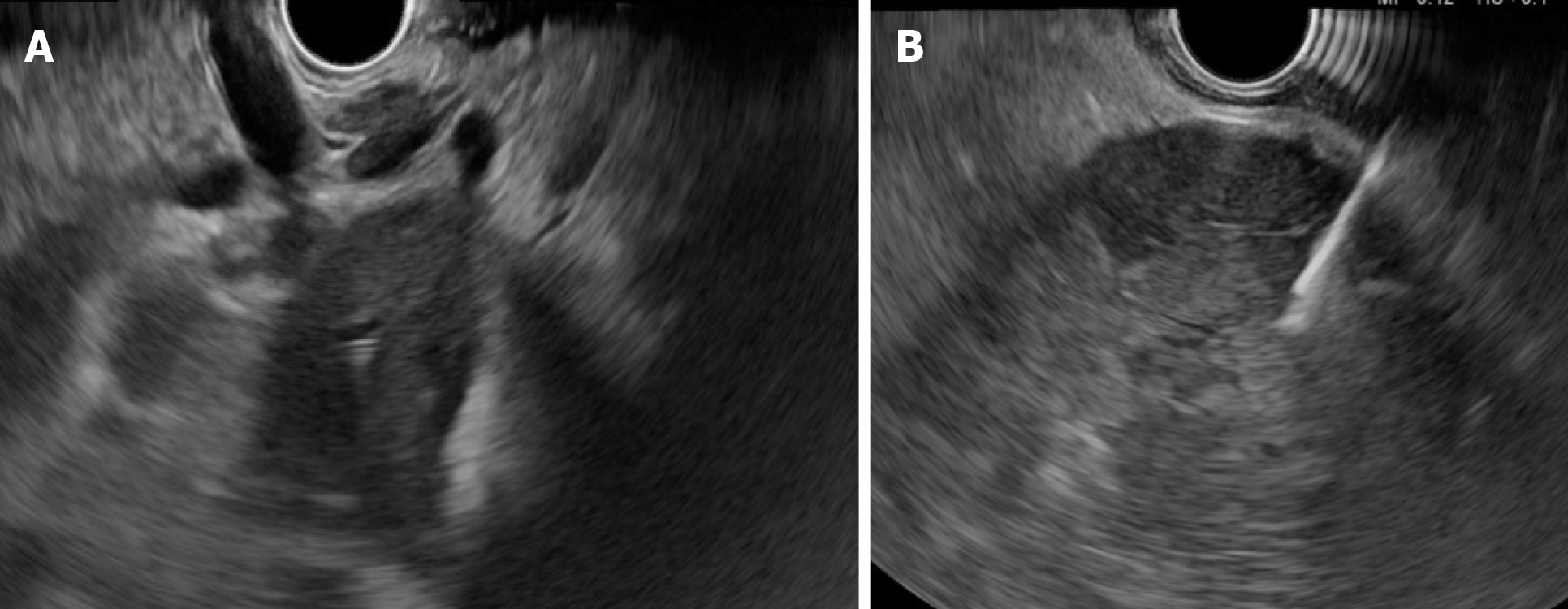
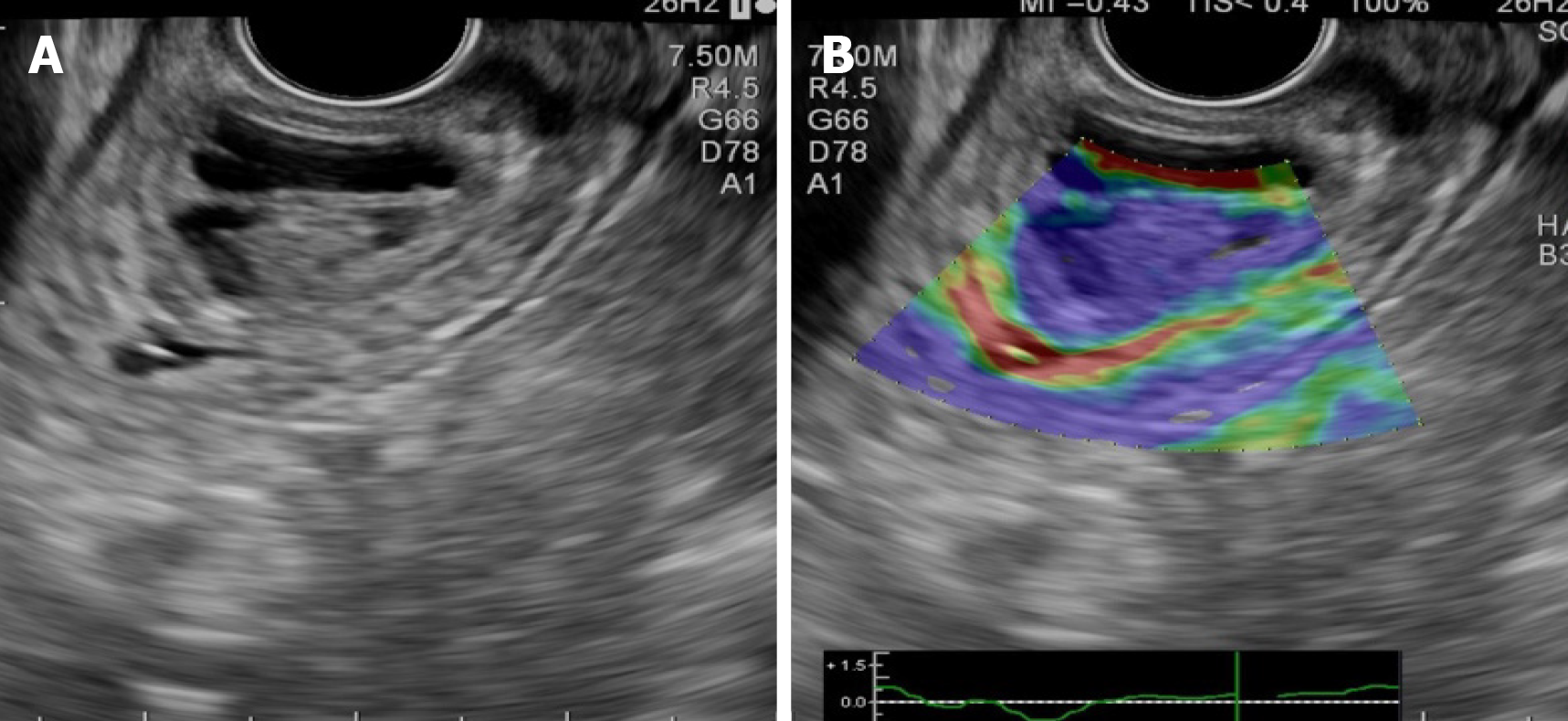
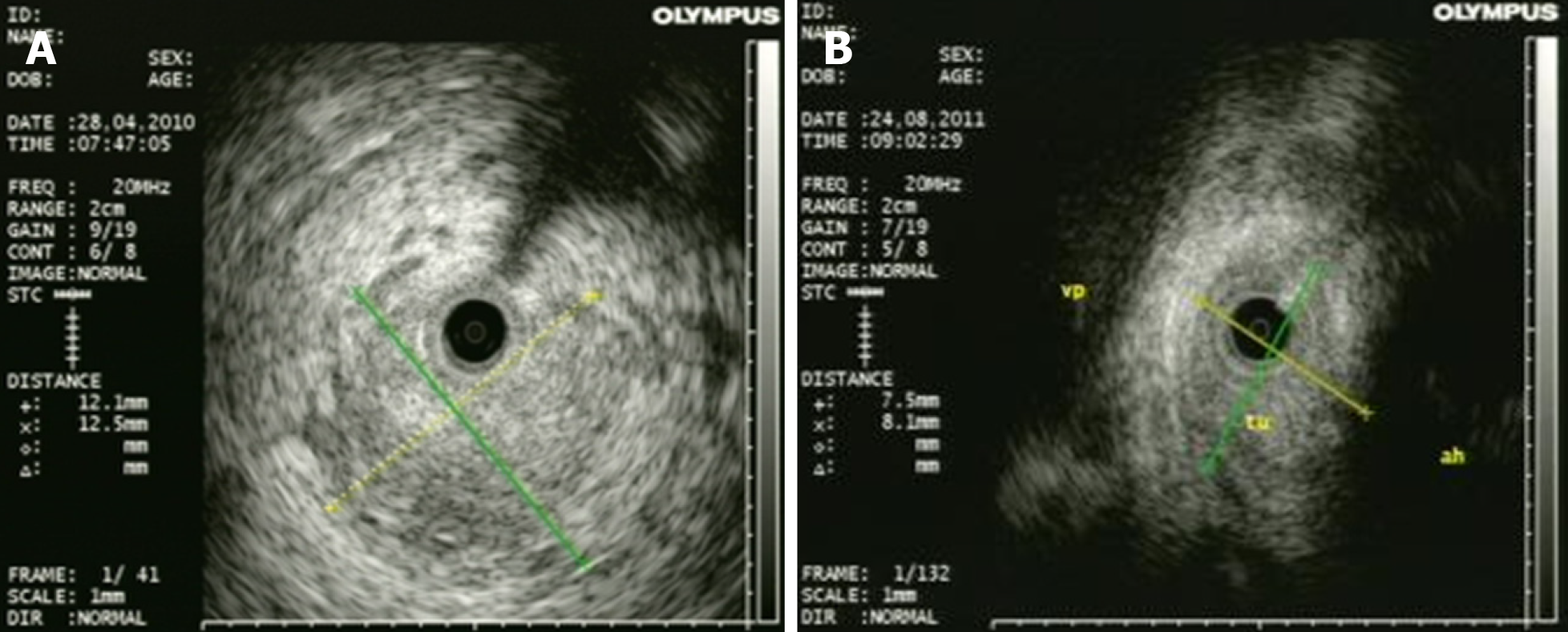
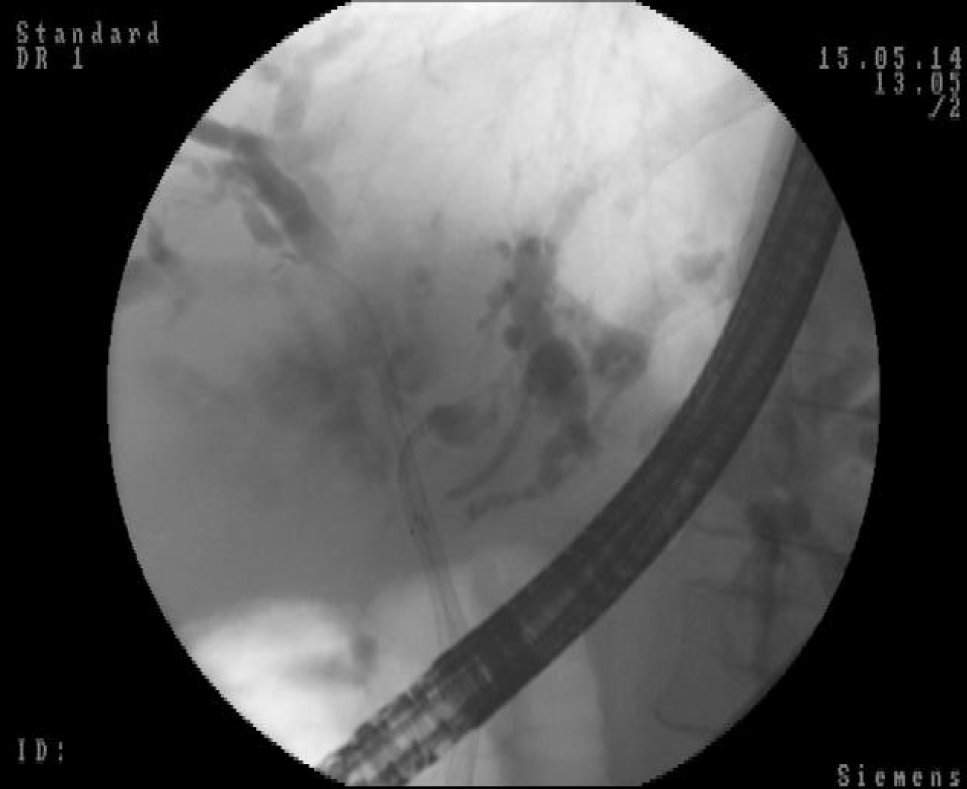
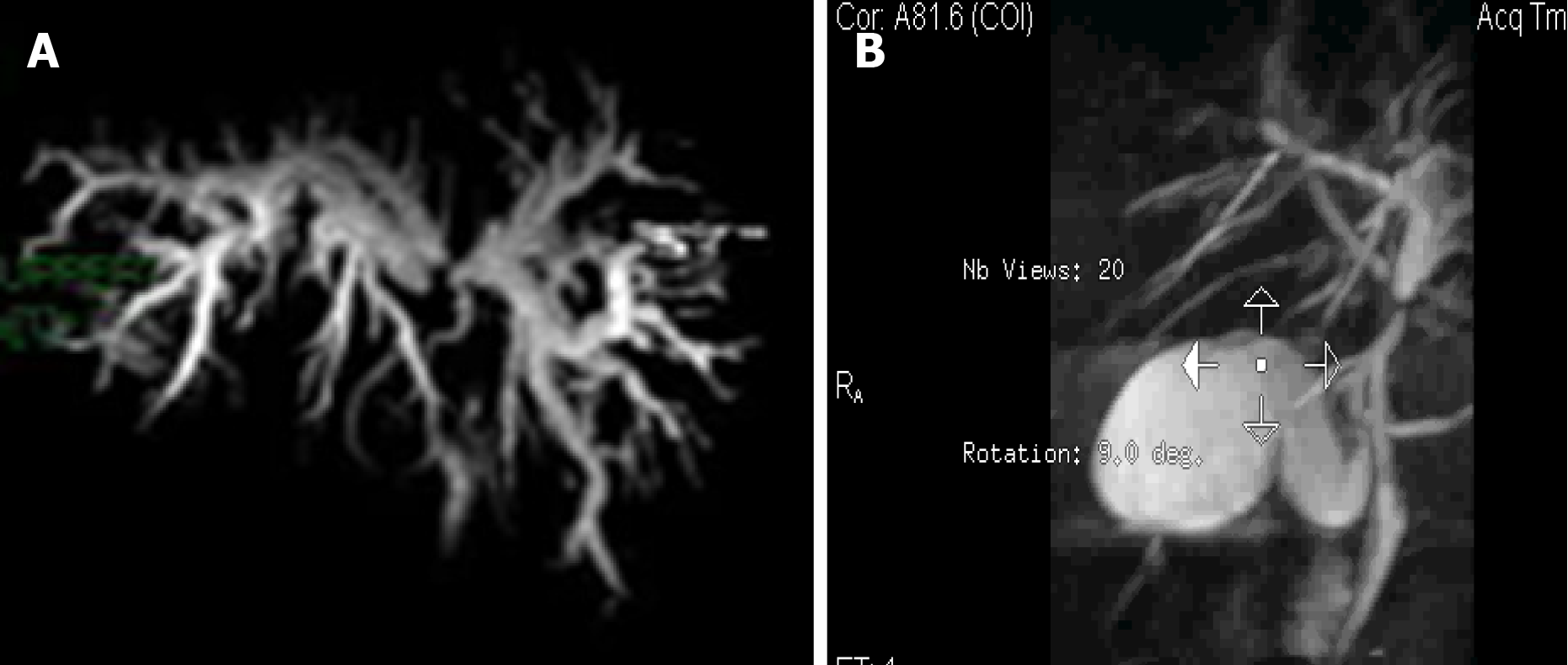
Cholangiocarcinomas (CCAs) have a very high mortality rate worldwide[1,2]. Diagnosis is challenging and delayed in many cases due to the common asymptomatic clinical behavior of early-stage disease, the lack of a standardized screening protocol for early-stage disease and the limitations inherent to using CA19-9 as a cancer marker[3].The ability to achieve a definite cytopathological or histopathological diagnosis in patients with suspected CCA ranges widely in the literature from 26% to 80%[4-8]. Magnetic resonance imaging plus magnetic resonance cholangiopancreatography is the preferred imaging modality as it can assess resectability and tumor extent with a high accuracy[9-15]. Endoscopic ultrasound (EUS) and fine needle aspiration guided by EUS is a useful technique in the diagnosis and staging of CCA (Figure 1 and 2) and should always be taken into consideration for CCA clinical management. For patients with obstructive jaundice, in particular, intraductal ultrasonography has been suggested for the assessment of bile duct strictures and local tumor staging[16] (Figure 3).
CCAs are divided into three types: Intrahepatic CCA, distal CCA (dCCA) and perihilar CCAs (pCCA) or Klatskin tumors. The majority of CCAs are pCCAs (60%-75% of cases). dCCA is present in 15%to 25% of cases, and intrahepatic CCA accounts for 5%to 15% of cases[17-20].
Surgery is the only curative treatment for extrahepatic CCA with the goal of R0 resection. Unfortunately, only a minority of patients (approximately 35%) have early stage disease and are candidates for this curative treatment option[21].Furthermore, only a few patients with pCCA are candidates for liver transplantation following neoadjuvant chemotherapy[22].
More than 50% of patients with jaundice are reportedly inoperable at the time of first diagnosis. Locally advanced, unresectable CCA cases include patients with macroscopic residual disease following resection, locally advanced, categorically unresectable disease at presentation or locally recurrent disease after potentially curative treatment. Prognosis of these patients is poor with a median survival time of < 6 mo[23]. Relief of symptoms (pain, pruritus, jaundice) and improvement in quality of life are the aims of palliative therapy.
Each subtype of CCA has different clinical management[24]. Therefore, an individualized approach is mandatory for pCCA or dCCA. In patients with extrahepatic CCA who are not candidates for surgery or liver transplantation, consideration should be given to enrollment in a clinical trial, particularly those evaluating targeted therapy[25].
Additional treatment measures in locally advanced extrahepatic CCA may include the following: Stenting, radiofrequency ablation (RFA), photodynamic therapy (PDT), radiation therapy, chemotherapy, molecular-targeted therapy and/or immunetherapy[25].
Preoperative or palliative biliary drainage using stents are two main approaches for extrahepatic CCA[26]. Stents can be placed via endoscopic retrograde cholangiopancreatography (ERCP) or percutaneous transhepatic cholangiography to relieve biliary obstruction. Stenting may relieve the jaundice and pruritus and improve the quality of life[26]. In ERCP, a unilateral or bilateral plastic or metallic stent can be used[23,24,26].
RFA and PDT are effective in restoring biliary drainage and improving quality of life in patients with nonresectable disseminated extrahepatic CCA[23,27]. Local radiotherapy combined with metallic stent placement is a new and efficient method in advanced extrahepatic CCA[28]. Several clinical trials are evaluating the effect of specific molecular agents targeting various signaling pathways in advanced extrahepatic CCA[25]. Our proposal is to highlight the utility and the efficiency of different endoscopic techniques and additional measures in extrahepatic CCA.
Endoscopic treatment of CCA with obstructive jaundice ensures bile duct drainage in preoperative or palliative settings[23,26]. Endoscopic procedures are the preferred palliative treatment options for patients with advanced or unresectable CCA. In patients with advanced pCCA, endoscopic biliary drainage via ERCP is more difficult than those with dCCA[23,25,26]. If the transpapillary approach failed, then other procedures can be considered: Percutaneous transhepatic biliary drainage (PTBD), endoscopic ultrasound-guided biliary drainage (EUS-BD) or hepatico-gastrostomy or locoregional therapies including transluminal PDT and RFA[27].
There is some controversy in the literature as to how preoperative biliary drainage should be accomplished prior to laparotomy for patients with obstructive jaundice[29-30]. In a European multicenter study, Gouma et al[31] showed that the postoperative outcomes in patients with pCCA who underwent surgery and preoperative biliary drainage were not improved. However, the rate of mortality was lower in patients who received en bloc right hepatectomy. In dCCA, preoperative bile duct drainage is not always necessary unless neoadjuvant chemotherapy is planned and might be associated with an increased risk of cholangitis and postoperative infectious complications[32].
Acute cholangitis, sepsis, bilirubin > 10-15 mg/dL, scheduled neoadjuvant therapy and the need for extensive hepatic resection are indications for preoperative biliary drainage. The goal is to reduce peri- and postoperative complications[23,24,26,29]. Cholestasis, liver dysfunction and biliary cirrhosis can develop rapidly with unrelieved obstruction and may influence postoperative morbidity and mortality after surgery[23-26,33]. The definitive operation is deferring until bilirubin levels are less than 2 to 3 mg/dL[33].
Some centers prefer preoperative biliary decompression in order to decrease the total bilirubin level to under 3 mg/dL, whereas others recommend resection in patients without biliary drainage. In our center the decision to perform preoperative biliary drainage is made in the setting of a multidisciplinary team, and it is not generally recommended unless severe liver dysfunction is suspected.
It should be taken into account that the stent may induce different artifacts in subsequent images. Therefore, previous high-quality imaging is required (computed tomography, magnetic resonance imaging, magnetic resonance cholangiopancreatography, endoscopic ultrasound and intraductal ultrasonography) to assess the tumor resectability[23-26,33]. The biliary stent may be a hindrance for the surgeon to find the proximal tumor extent. Resection of pCCA always requires a concomitant major liver resection. Liver segments that will remain after surgery should be drained sufficiently with a plastic stent to improve postoperative liver function and regeneration[29].
There are different data regarding the benefits of preoperative biliary drainage in jaundice patients with pCCA without absolute indications for biliary drainage[29]. The most recent studies concluded that routine biliary drainage does not impart any advantage because it does not improve the morbidity or mortality of patients with resected pCCA[31,34,35]. A recent meta-analysis and a systematic review showed that preoperative biliary drainage have not changed the incidence of postoperative complications, hospitalization time, R0 or survival rate. However, in jaundice patients, preoperative biliary drainage decreased postoperative mortality[36].
In dCCA, a European multicenter study did not find any differences regarding mortality rate in patients with preoperative biliary drainage[37]. Moreover, in a recent retrospective study, preoperative endoscopic biliary drainage was associated with a decrease in the survival rate[38].
In PTBD, some studies reported that catheter tract recurrence rates were up to 6%[32], and the median time of recurrence was months. Furthermore, the technical success rate regarding the decrease of biliary level is higher with the endoscopic approach than with PTBD[39,40]. In a recent randomized prospective study, the risk of cholangitis in patients who underwent surgery was higher in the PTBD group compared with the endoscopic biliary drainage group (59% vs 37%) (P = 0.1)[41].
PTBD is no longer recommended for preoperative biliary drainage in patients with extrahepatic CCA, and an endoscopic approach is currently preferred[24,26]. The risk of endoscopic plastic stent occlusion is 60%. Therefore, there are several groups of experts who recommend preoperative nasobiliary drainage. Kawashima et al[42] compared preoperative nasobiliary drainage with endobiliary stenting drainage in 164 patients with pCCA. They found a longer stent patency and a lower risk of cholangitis in the nasobiliary group than the endobiliary stenting group.
The relief of symptoms (pain, pruritus, jaundice) and improvement in quality of life are the goals of palliative therapy. Radiotherapy, PDT, RFA, local ablation and embolization are nonsurgical local therapies that can prolong the time to local failure (in patients with macroscopically positive margins) or to palliate local symptoms, pain or jaundice (in patients with unresectable or recurrent disease).
In patients with pCCA and dCCA who are not suitable for surgery or liver transplantation, the guidelines recommend endoscopic bile duct drainage as the first approach[23,24,26,34]. In patients with a good performance status an additional treatment, such as chemotherapy, radiotherapy, molecularly targeted therapy and/or immunotherapy, is recommended[25].
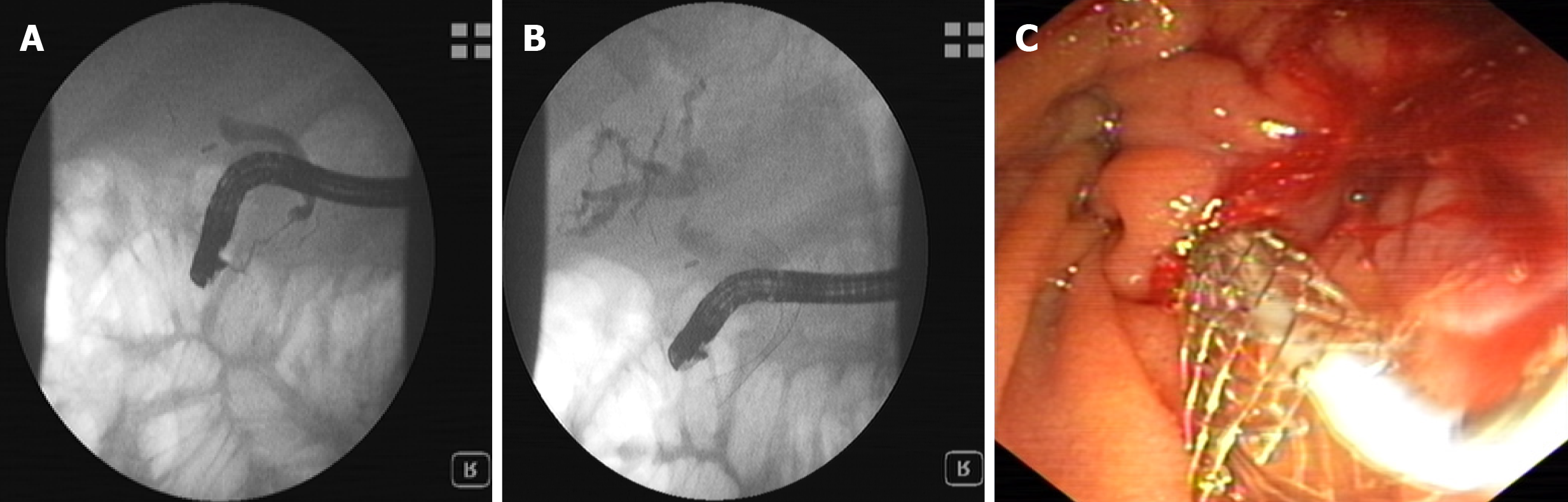
Stent implantation by ERCP should be the standard procedure[24-26] (Figure 4 and 5). Placement of a stent is generally preferred for long-term palliation. This approach has similar successful palliation and survival rates and less morbidity compared with the surgical approach[43]. The endoscopic drainage with one or more stents is technically possible in 70% to 100% of cases. The extent of decompression that is necessary to restore sufficient bile flow while avoiding the risk of bacterial cholangitis, the optimal approach to placement of the stents and the use of plastic or metal uncovered/covered stents are the major issues of biliary endoscopic stenting[44].
The goal of palliative drainage is to drain more than one half of the biliary tree, although it has been shown that the jaundice may be clinically improved if only a quarter of the liver is drained[45]. A target stenting using previous superior imaging methods is preferred[44]. In cases of cholangitis, drainage of all suspected infected intrahepatic segmental branches should be performed[24].
In complex and difficult cases a multimodality biliary drainage (transpapillary drainage in combination with PTBD) should be considered[44]. Rendezvous technique, anterograde PTBD and transluminal stenting through the stomach, duodenum or jejunum walls are the procedures using EUS-BD in these cases. This approach can be performed even when a passage of a wire through a biliary stricture is not possible[46]. In a meta-analysis conducted by Leng et al[47], the technical success rate of PTBD varied from 60% to 90% and the morbidity rate from 18%to 67%. In some difficult cases, an external drainage has been required. Therefore, the quality of life of these patients is decreased. EUS-BD technical success varied from 70% to 100%, and the rate of complications was up to 77%[48,49]. A few comparative studies are available[50-54] (Table 1). The technical success rates are similar in most studies with a higher incidence of complications for PTBD than EUS-BD[50-54].
| Ref. | Procedure | Patients, n | Technical success % | Morbidity % |
| Artifon et al[50], 2012 | PTBD, EUS-BD | 12, 13 | 100, 100 | 25.00, 15.30 |
| Bapaye et al[51], 2013 | PTBD, EUS-BD | 26, 25 | 46.00, 92.00 | 46.00, 20.00 |
| Khashab et al[52], 2015 | PTBD, EUS-BD | 51, 22 | 100, 86.40 | 39.20, 18.20 |
| Dhir et al[53], 2015 | ERCP SEMS, EUS-BD | 104, 104 | 94.23, 93.26 | 8.65, 8.65 |
In most cases, unilateral stent placement should be adequate for biliary drainage via ERCP because only 25% to 30% of the liver needs to be drained to relieve jaundice[54-56]. However, unilateral drainage alone may not relieve jaundice completely and may increase the risk of cholangitis due to contrast medium injection into undrained bile ducts[45]. Unilateral stenting is technically easier and less expensive than bilateral stenting with reintervention for stent dysfunction also being considerably easier[45]. In our practice, we prefer to place a unilateral self-expandable metallic stent (SEMS) in order to provide good efficacy of biliary drainage with minimum risk of cholangitis. In clinical practice, many endoscopists prefer to place bilateral stents (plastic or metal) in an attempt to maximize biliary drainage and to prevent cholangitis.
Previous studies have demonstrated that bilateral stenting is associated with longer stent patency compared to unilateral stenting[57,58]. In a recent multicenter prospective randomized study conducted by Lee et al[59], the same survival rate in patients with bilateral SEMS biliary drainage but with a longer stent patency vs unilateral SEMS biliary stenting were shown. No significant difference between unilateral and bilateral SEMS regarding the technical success or complications was shown[59]. These results highlighted the superiority of bilateral stenting. However, several study results have similarly supported the superiority of unilateral stenting[54-56,60].
In a recent meta-analysis involving 782 patients, bilateral biliary drainage had a lower re-intervention rate compared to unilateral drainage in patients with pCCA with no significant difference in technical success and early or late complication rates[61].
Endoscopic biliary drainage can be performed using plastic or SEMS. There are a variety of plastic and metal stents, covered or uncovered. While some studies showed benefits of metallic stents regarding the successful drainage and early complication rate, stent patency and survival rate[55-59,62], a systematic review concluded that neither stent type offered a survival advantage[63]. The decision to use one vs another should be guided by the expected length of survival, quality of life, costs and physician expertise. Usually, SEMS should be considered for patients with a life expectancy of longer than 3 mo[44]. The results of different meta-analyses that compared SEMS with plastic stents for endoscopic drainage of distal malignant biliary obstruction are illustrated in Table 2[62-66].
| Ref. | Studies included | Patients, n | Procedures | Results |
| Almadi et al[62], 2017 | 20 | 1713 | Endoscopic or percutaneous palliative biliary drainage with plastic stent vs SEMS | Stent patency 4.45 mo (95%CI: 0.31-8.59) in favor of SEMS; Overall survival 0.67 (95%CI: 0.66-1.99), no difference |
| Moole et al[33], 2017 | 11 | 947 | Endoscopic palliative biliary drainage with plastic stent vs SEMS | Stent occlusion OR 0.48 (95%CI: 0.34-0.67) in favor of SEMS; Overall survival/time to death: (1) SEMS, 157.3 d (95%CI: 148.9-165.6), (2) Plastic, 120.6 d (95%CI: 114.3-126.9), P = 0.0024 |
| Zorrón Pu et al[64], 2015 | 13 | 1133 | Endoscopic palliative biliary drainage with plastic stent vs SEMS | Stent dysfunction, RD -0.26 (95%CI: -0.32 to -0.20) in favor of SEMS; Survival longer in the SEMS group (187 d vs 162 d, P < 0.0001) |
| Sawas et al[65], 2015 | 19 | 1989 | Endoscopic or percutaneous palliative biliary drainage with plastic stent vs SEMS | Stent occlusion, HR 0.42 (95%CI: 0.27-0.64) in favor of SEMS; 30-d survival, HR 0.82 (95%CI: 0.45-1.48), no difference |
| Hong et al[66], 2013 | 10 | 785 | Endoscopic palliative biliary drainage with plastic stent vs SEMS | Stent patency, HR 0.37 (95%CI: 0.28-0.48) in favor of SEMS; Survival, HR 0.81 (95%CI: 0.68-0.96) in favor of SEMS |
Plastic (polyethylene) stents are inexpensive, effective and easily removable or exchangeable[38-44]. The major disadvantage is a higher rate of occlusion by sludge and/or bacterial biofilm with cholangitis development and necessity of multiple ERCPs[60,62-66]. Instead, metal stents have a longer patency (approximately 8-12 mo vs 2-5 mo for plastic stents)[61-66], higher costs and may not be removable. The high occlusion rate of plastic stents (average 42%) can be reduced by changing the stents every 3-6 mo[60,62-66]. Another way is to wait for a complication before changing the stent because many patients will die before the stents will obstruct. The preferred approach for patients who are expected to live beyond a few months is to replace the plastic stent with a metal one as soon as is feasible[44].
In dCCA, uncovered SEMS are used in patients with an intact gallbladder[26]. For patients who have undergone prior cholecystectomy, the choice of a covered vs uncovered SEMS is individualized given the location and geometry of the stenosis. Patients with extrinsic compression may be adequately treated with an uncovered SEMS, while those with intrinsic and/or papillary tumors may benefit from a covered SEMS in an attempt to minimize tumor ingrowth[26,67,68]. The patency rates are not higher for covered stents despite showing significantly less tumor ingrowth. Tumor overgrowth and stent obstruction by debris and biliary sludge are associated with a low patency rate for uncovered SEMS[68]. Covered SEMS should be used for pCCA. Deployment may inadvertently result in the occlusion of a major hepatic duct[24,26,44,68].
The stent in stent technique (Y stenting) and the side-by-side technique (Figure 6) are two endoscopic techniques for biliary drainage in CCA. By using the Y stent technique, Hwang et al[69] demonstrated an 86.7% technical success rate and a 100% functional success rate regardless of the stent type. For side-by-side stenting technique in pCCA, Lee et al[70] reported a 91% technical success rate and a 100% functional success rate with no statistically significant difference between stent patency and median survival of the 8-mm and 10-mm groups.
The reported rate of stent dysfunction following pCCA biliary drainage was 45%-57% due to tumor ingrowth, tumor overgrowth or stent migration[55-58]. Given the fact that SEMS may be successfully revised in the majority of cases and that the second SEMS have a higher patency compared with plastic stents, it seems that SEMS are the best choice in cases of SEMS dysfunction[55-59].
Guidelines recommend prophylactic antibiotics in patients with plastic or metal stents for long-term palliation of obstructive jaundice after the first episode of cholangitis[24,26,44]. In 5%-10% of cases, endoscopic biliary drainage by ERCP will fail or will be incomplete[54-69]. In this case, multimodality drainage should be considered[24,26,44].
Several studies have shown a higher rate of successful palliation of jaundice and lower rates of cholangitis in the percutaneous approach rather than the endoscopic approach of biliary drainage in patients with malignant hilar obstruction (pCCA/gallbladder cancer)[71-73]. Bile leaks and bleeding are more frequent and morbidity and mortality are higher than the endoscopic approach[73]. Percutaneous stents are usually left to open drainage externally from the body and are less comfortable for the patient. Another technique is the combination of ERCP with percutaneous drainage.
EUS-BD: EUS-BD has been proposed as an effective alternative for PTBD after failed ERCP[74-80]. The use of EUS-BD is feasible for a left system drainage procedure in patients with advanced CCA who failed transpapillary drainage[74-80]. For extrahepatic CCA, the procedure of choice is EUS-guided hepatico-gastrostomy, which allows left system access only. It is less invasive given that it affords a more accurate control as well as more access sites to the bile duct than the classical alternatives of PTBD or surgery[77]. After the identification of the biliary duct, the technique consists of puncturing and dilatation by EUS with stent placement across the bile duct into the digestive lumen. Literature data showed a 94.0% per-protocol success rate and a 90.2% intention-to-treat basis success rate[75-81].
Peritoneal bile leakage and cholangitis are the most frequent complications[75-81]. Early migration or the clogging of the plastic stents may lead to cholangitis[76]. Bile peritonitis and biloma are more frequent in transmural SEMS placement[77,80]. However, most complications are mild and can be conservatively treated[81]. By combining an uncovered metal stent with a covered metal stent inside, the risk of leakage is minimized. The uncovered stent is initially deployed to provide anchorage and prevent migration. The covered stent is inserted coaxially and dropped in the first stent. A fully covered SEMS[77] or a double pig-tail stent through the expanded SEMS may be used to prevent stent migration[78].
The advantages of EUS-guided hepatico-gastrostomy over rendezvous or anterograde stent insertion are particularly relevant in patients with prior duodenal or biliary SEMS who experience recurrent biliary obstruction[79,81]. Dhir et al[82] compared ERCP-guided biliary drainage with EUS-guided approach in patients with malignant distal obstruction who required SEMS placement. They found that the short-term outcome of EUS-BD is comparable to that of ERCP. Postprocedural pancreatitis rates were higher in the ERCP group[82,83]. Clinical efficacy of a novel technique of EUS-BD for right intrahepatic bile duct obstruction was evaluated[84,85]. Most of the studies have only shown the role of EUS-BD in distal biliary obstruction, and the utility of EUS-BD for pCCA is limited. Recent studies have reported the efficacy of EUS-BD in a setting of failed ERCP for biliary drainage in proximal malignant obstruction[86,87].
Kongkam et al[88] proposed a new concept of a combination of ERCP and EUS-BD for biliary drainage in pCCA as a primary biliary drainage method whereby ERCP with a single SEMS is placed into either the right or the left intrahepatic bile duct. In cases of failure of all interventional options, surgical bypass should be considered as the last rescue procedure. It is typically only performed during an unsuccessful attempt at resection, or it may be necessary in jaundice patients in whom stenting is not possible due to tumor location[1,6,7,18].
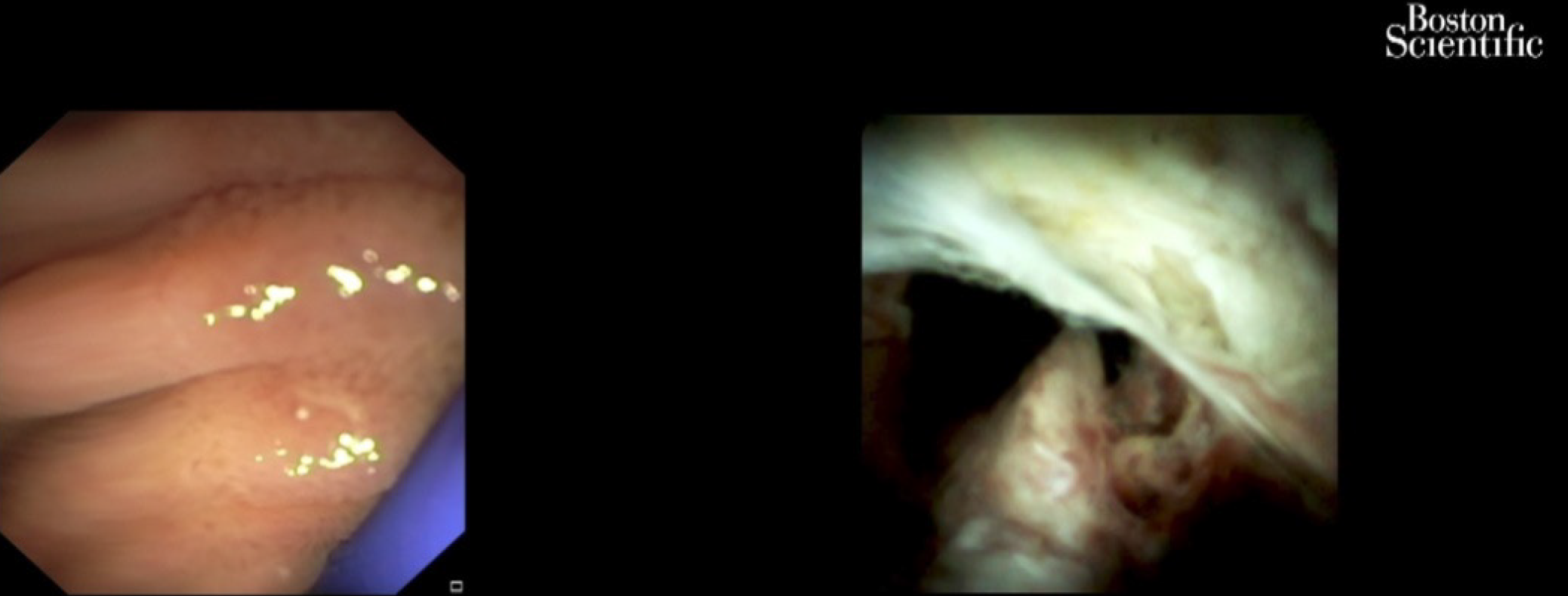
Peroral cholangioscopy (POC) allowing direct visualization of the biliary tract with targeted biopsy of suspicious lesions is a useful diagnostic procedure in the evaluation of biliary strictures (Figure 7). A recent study[89] showed that POC use for the assessment of intraductal spread in potentially resectable pCCA can accurately detect and can change surgical management. In the future, preoperative staging of CCAs should combine radiological with endoscopic (i.e. POC evaluation) in order to optimize surgical results.
Another study[90] compared the performance characteristics of single-operator cholangioscopy-guided biopsies and transpapillary biopsies with standard sampling techniques for the detection of CCA. It showed that single-operator cholangioscopy-guided and transpapillary biopsies improved sensitivity for the detection of CCAs in combination with other ERCP-based techniques compared to brush cytology alone. However, it seemed that these modalities did not significantly improve the sensitivity for the detection of malignancy in primary sclerosing cholangitis.
A very recent publication[91] evaluated a newly developed POC classification system by comparing classified lesions with histological and genetic findings. Thirty biopsies were analyzed from 11 patients with biliary tract cancer who underwent POC. An original classification of POC findings was made based on the biliary surface’s form (F factor, 4 grades) and vessel structure (V factor, 3 grades). Histological malignancy rate increased with increasing F- and V-factor scores. The system was validated by comparing it to the histological diagnosis and genetic mutation analysis in simultaneously biopsied specimens. F-V classification is the first reported system to quantify and classify biliary tract cancer based on POC findings.
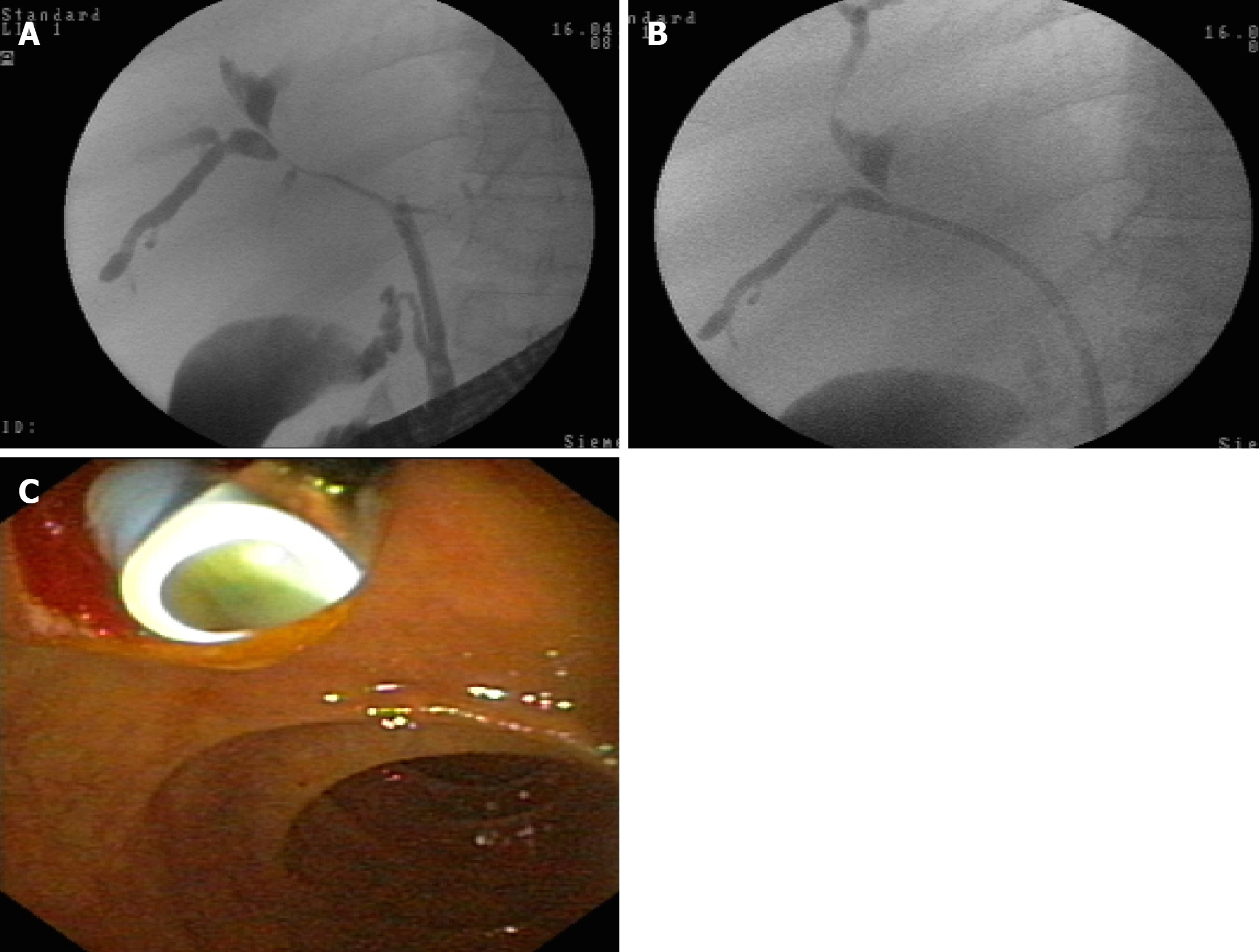
Percutaneous image-guided RFA is a potential “new tool” for the endobiliary treatment of pCCA[92]. After selective intrahepatic duct cannulation, the 0.035-inch guidewire is placed across the stricture point. The lesion is identified during cholangiography. After the previous sphincterotomy, the RFA is performed using a specific catheter. It is mandatory for all of tumor area to be caught during the procedure. The coagulated tissue will be removed using a balloon probe, and a stent will be inserted[93]. There are only a few studies regarding the successful therapy with intraductal RFA for pCCA[94,95]. A recent study[95] including 65 patients with unresectable extrahepatic CCA showed that the mean survival time was significantly greater among those who underwent RFA plus stenting compared with stenting alone (13 mo vs 8 mo). At 12 mo, the survival rate was 63% in the RFA group compared with 12% in the stenting-only group. Stent patency was also longer in the RFA group (7 mo vs 3 mo). The adverse event rate did not differ significantly between groups (6% and 9%).
These results are overlapping with those of a meta-analysis, which was comprised of 505 patients and evaluated the effectiveness of biliary stent placement with RFA on stent patency and patient survival[27]. The pooled weighted mean difference in stent patency was 50.6 d, favoring patients receiving RFA and an improved survival in patients treated with RFA. RFA was associated with a higher risk of postprocedural abdominal pain. There was no significant difference between the RFA and stent placement-only groups with regard to the risk of cholangitis, acute cholecystitis, pancreatitis and hemobilia[27].
A prospective open-label multicenter study included 12 patients with histologically proven endobiliary adenoma remnant (ductal extent < 20 mm) after endoscopic papillectomy for ampullary tumor. RFA was performed during ERCP with biliary ± pancreatic stent placed at the end of the procedure. All underwent one successful intraductal RFA session with biliary stent placement and recovered uneventfully. Five (25%) received a pancreatic stent. The rates of residual neoplasia were 15% and 30% at 6 and 12 mo, respectively. Only two patients (10%) were referred for surgery. Eight patients (40%) experienced at least one adverse event between intraductal RFA and 12 mo of follow-up. No major adverse events occurred. Intraductal RFA of residual endobiliary dysplasia after endoscopic papillectomy can be offered as an alternative to surgery with a 70% chance of dysplasia eradication at 12 mo after a single session and a good safety profile[96].
PDT is the use of photosensitizing agents that accumulate into the tumor. The agents are activated by laser light. Free oxygen radicals are released and destroy the neoplastic cells[69]. Apoptotic death of cells is another mechanism produced by PDT with an immunomodulatory effect. Hematoporphyrin derivatives, ∂-aminolevulinic acid and meso-tetra (hydroxyphenyl) chlorin are the photosensitizing agents used for CCA treatment[24,97]. Strong phototoxic skin reactions that can persist for weeks are a disadvantage of the use of photosensitive substances such as photofrin (porfimer sodium). The advantage of the ∂-aminolevulinic acid, which is a second generation photosensitizer, is the lack of prolonged photosensitization and laser light exposure.
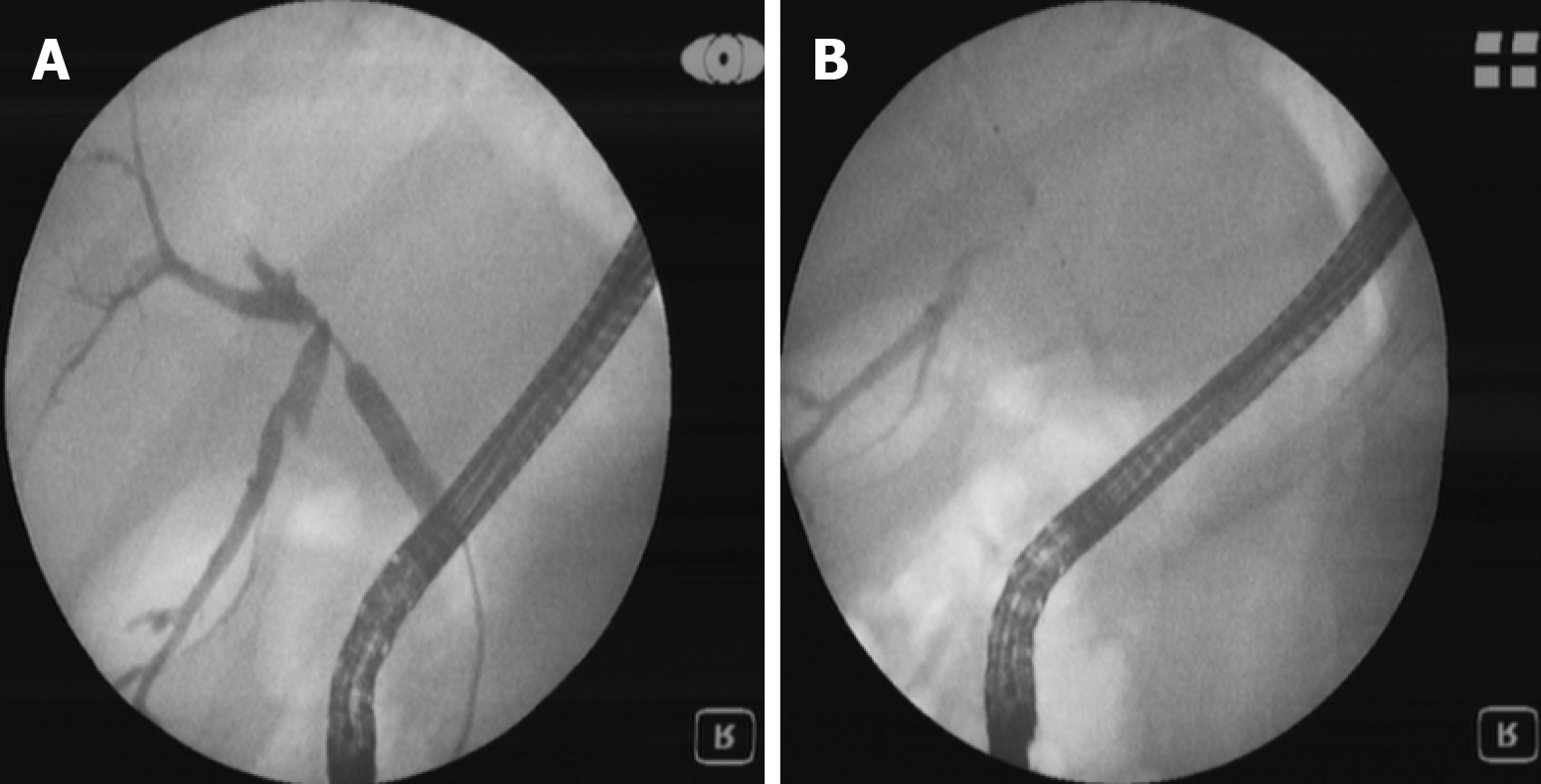
The endoscopic PDT technique involves intravenous 48-h administration of the photosensitizing agent prior to the laser light illumination. The specific substance is retained in tumor cells and into the skin longer than 48-72 h like in the normal tissues. With a guidewire and a catheter, the light laser fiber is placed across the tumoral stricture (Figure 8). The power density used is 300-400 mW/cm with a power energy of 180-200 J/cm. The irradiation time is 400-600 s[97]. Due to the fact that light laser fiber is stiff, the breakage may occur in up to one third of the procedures, making the procedure a bit more cumbersome and affecting treatment cost[24,44]. The PDT is only performed in some specialized centers.
In addition to facilitating biliary decompression after stenting in patients with locally advanced disease, survival might be improved in patients who undergo PDT[98-107] (Figure 9). The data showed a survival benefit for this approach with favorable early results including longer survival and quality of life[98,105,106] (Table 3). The survival benefit was related to the prolonged relief of obstruction rather than to a reduction of the tumor. Although the factors that are associated with prolonged survival are not completely known, at least some data suggest that the absence of a visible mass on radiographic studies correlates with longer survival after PDT[44,107].
| Ref. | No. patients | Median survival, d/mo | Adjuvant therapy |
| Ortner et al[106] | PDT 20, Control 19 | 493 d, 98 d | PDT -, Control - |
| Zoepf et al[103] | PDT 16, Control 16 | 630 d, 210 d | PDT -, Control - |
| Dumoulin et al[100] | PDT 24, Control 23 | 9.9 mo, 5.6 mo | PDT -, Control - |
| Kahaleh et al[102] | PDT 19, Control 21 | 8.0 mo, 5.0 mo | PDT, CTX 11; RTX 9; CTX 11, RTX 10 |
| Witzigman et al[99] | PDT 68, Control 56 | 12.0 mo, 6.4 mo | PDT, CTX 6; RTX 2; CTX 5, RTX 1 |
Cholangitis and a liver abscess are the main complications of photodynamic therapy[98-107]. Data suggest that combining photodynamic therapy with systemic combination chemotherapy improved outcomes over PDT alone for patients with nonresectable tumors without increasing toxicity rates, although randomized trials have not been conducted[108-112]. At the moment, PDT is being studied preoperatively as a means of improving the likelihood of achieving a margin-negative resection[113].
In a recent meta-analysis conduct by Lu et al[114], overall survival was significantly better in patients who received photodynamic therapy than those who did not. Among the eight trials (642 subjects), five assessed the changes of serum bilirubin levels and/or Karnofsky performance status as other indications for improvement. The incidence of phototoxic reaction was 11.11%. The incidence for other events in photodynamic therapy and the stent-only group was 13.64% and 12.79%, respectively.
A new model of a photosensitizer-embedded self-expanding metal stent (PDT-stent) that provides a photodynamic effect without a systemic injection has been developed. The treatment could be repeated due to the incorporation of the polymeric photosensitizer into the mesh of the stent. The stent maintained its photodynamic power for at least 8 wk. This type of stent after light exposure creates cytotoxic free radical, such as singlet oxygen, in the surrounding tissue and induces destruction of tumoral cells on animal models[115]. Unfortunately, PDT is not widely available and is expensive and uncomfortable for the patient.
The purpose of brachytherapy (BT) is to deliver a high local dose of radiation to the tumoral tissue while sparing healthy tissue around it. It can be adapted for right and left hepatic duct and for common bile duct lesions. It plays a limited but specific role in the curative intent treatment in selected cases of early disease as well as in postoperative small residual tumoral tissue. The indications for BT are as radical or palliative treatment. For radical treatment, it is recommended in small inoperable tumors or in combination with external beam radiation therapy and/or chemotherapy in advanced disease for unresectable tumors. BT may be used as adjuvant treatment after nonradical excision, possibly combined with external beam radiation therapy. The most common indication for BT occurs as palliative in unresectable Klatskin tumors. The purpose is to prevent locoregional disease progression and to facilitate the bile outflow. The major aim is to improve the quality of life and to increase survival. The treatment decision should be personalized[116].
ERCP-directed tumor therapy using iridium-192 ribbons via nasobiliary catheters in patients with pCCA as part of a neo-adjuvant treatment protocol that include external beam radiation therapy, radiation-sensitizing chemotherapy and low-dose-rate BT (< 3000 cGy) followed by liver transplant was first described in 2006[117]. High-dose-rate (HDR)-BT using 930-1600 cGy fractionated in 1-4 doses over 1-2 d was introduced in 2009[117]. The benefits of this technique are lack of irradiation of medical staff, lower time span (5-10 min), a better distribution of doses in the tumor and protection of the stomach and duodenum[117]. Using ERCP, an 8.5 Fr or 10 Fr nasobiliary tube is placed into the biliary system with the proximal end of tube at least 2 cm beyond the proximal end of the tumor. In cases of bilateral duct involvement, a second 10 Fr tube is placed. After HDR-BT is completed, the tubes and brachycatheter are removed. Nasobiliary BT catheter displacement, cholangitis, abdominal pain, duodenopathy and gastropathy are possible complications[118,119].
Some studies demonstrated longer survival in patients with CCA due to the BT. Extrahepatic localization of CCA, the absence of metastases, increasing calendar year of treatment and liver transplantation with postoperative radiation therapy were factors significantly associated with improved survival[118,119]. However, another study did not find any benefit regarding the survival in patients treated with PTBD-guided iridium-192, intraluminal BT compared with patients with only PTBD[120]. These results are in accordance with another study that found a correlation only with local tumor control[121].
In a recent study[122], 122 patients with CCA were successfully treated with HDR-BT using the nasobiliary technique. The BT was not completed in three patients because either the catheter migrated between the ERCP and the treatment (two patients) or the HDR after loader was physically unable to extend the source wire into the treatment site (one patient). These three patients benefited from an external beam boost instead of HDR-BT. Intraluminal HDR-BT with a nasobiliary catheter is a minimally invasive method for administering neoadjuvant radiotherapy.
The assessment of patients with CCA before starting chemotherapy includes the Eastern Cooperative Oncology Group patient scale used for the evaluation of the patient performance status, disease distribution and accessibility of tumor profiling[123]. The current data support the use of first-line cisplatin and gemcitabine combination regimen chemotherapy. The multicenter phase III ABC-02 study illustrated the superiority of the combination regimen regarding median overall survival (11.7 mo) over the gemcitabine monotherapy (8.1 mo)[124,125].
New combinations and more intensive triple chemotherapy are being explored. The combinations include: Cisplatin-gemcitabine combined with nab (nanoparticle albumin-bound)-paclitaxel[126]; S1 (tegafur, gimeracil and oteracil)[127]; and FOLFIRINOX ( 5-FU, oxaliplatin and irinotecan; AMEBICA study , NCT02591030). Acelarin is a nucleotide-analogue independent of hENT2 (also known as SLC29A2) cellular transport and is not metabolized by cytidine deaminase, resulting in greater intracellular concentrations. Cisplatin with acelarin was compared with the classic combination regimen of cisplatin and gemcitabine in a phase III study[128].
A recent phase III clinical trial ABC-06[129] randomly assigned 162 patients with advanced biliary cancer (72% with CCA) who obtained symptom control from first-line cisplatin-gemcitabine (81 patients) or second-line chemotherapy with FOLFOX (folinic acid, 5-FU and oxaliplatin) (81 patients). The results showed a benefit from second-line chemotherapy regarding survival at 6 mo (35.5% vs 50.6%) and 12 mo (11.4% vs 25.9%), but no significant differences regarding overall survival (5.3 mo vs 6.2 mo) were observed.
A very difficult to handle and a major issue in the management of patients with CCA is the poor response to pharmacological treatment. A cause could be the poor understanding of the mechanisms of chemoresistance. To identify the so-called “resistome” that includes a set of proteins involved in the lack of response to chemotherapies is required to increase efficacy. Genes involved in mechanisms of chemoresistance are usually expressed by normal cholangiocytes because one of their roles is the protection against potentially harmful compounds present in bile. Their expression during carcinogenesis contributes to intrinsic chemoresistance, and upregulation in response to treatment leads to acquired chemoresistance[130-132].
Recent molecular studies have increased the understanding of the pathogenetic mechanism of CCAs, but to date the clinical data on immune-directed therapies in CCA are limited.
Inhibitors of isocitrate dehydrogenase (IDH) 1, IDH2 and pan-IDH1-IDH2 are currently being tested in patients with intrahepatic CCA. Ivosidenib (IDH1 inhibitor) was tested in 73 patients with IDH1-mutant advanced CCA in a phase I study with no major adverse events reported[133]. A recent preliminary phase III trial showed a benefit for ivosidenib over placebo in terms of progression free-survival. One hundred eighty-five patients with IDH1 mutant CCA were randomly assigned to ivosidenib or placebo. This study highlighted the importance of molecular profiling in CCA[134] .
There are some phase II studies with encouraging preliminary data for fibroblast growth factor receptor inhibitors in patients with CCA. Some fibroblast growth factor receptors inhibitors are currently being evaluated as first-line treatment, for example the FIGHT-302 study (NCT03656536) and the PROOF study (NCT03773302)[135-137].
CCAs are heterogeneous and highly aggressive tumors with a poor prognosis despite the progress of the research in this field. Surgical resection is still the only potential curative treatment method. The recent findings on understanding the mechanism of chemoresistance and molecular targeted therapy could bring a new horizon in the approach of these tumors. Currently, endoscopic treatment in patients with CCA and jaundice remain the first choice of biliary duct decompression, either preoperatively or with a palliative purpose. The combination of endoscopic procedures with nonsurgical local methods or additional therapies may increase the quality of life and the rate of survival in patients with locally advanced, unresectable or recurrent disease.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Romania
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Fabris L, Garbuzenko DV, Khaliq S, Tajiri K S-Editor: Gao CC L-Editor: Filipodia P-Editor: Wang LL
| 1. | Saha SK, Zhu AX, Fuchs CS, Brooks GA. Forty-Year Trends in Cholangiocarcinoma Incidence in the U.S.: Intrahepatic Disease on the Rise. Oncologist. 2016;21:594-599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 364] [Cited by in RCA: 563] [Article Influence: 62.6] [Reference Citation Analysis (0)] |
| 2. | Khan SA, Taylor-Robinson SD, Toledano MB, Beck A, Elliott P, Thomas HC. Changing international trends in mortality rates for liver, biliary and pancreatic tumours. J Hepatol. 2002;37:806-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 421] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 3. | Patel AH, Harnois DM, Klee GG, LaRusso NF, Gores GJ. The utility of CA 19-9 in the diagnoses of cholangiocarcinoma in patients without primary sclerosing cholangitis. Am J Gastroenterol. 2000;95:204-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 285] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 4. | Trikudanathan G, Navaneethan U, Njei B, Vargo JJ, Parsi MA. Diagnostic yield of bile duct brushings for cholangiocarcinoma in primary sclerosing cholangitis: a systematic review and meta-analysis. Gastrointest Endosc. 2014;79:783-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 136] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 5. | Barr Fritcher EG, Voss JS, Brankley SM, Campion MB, Jenkins SM, Keeney ME, Henry MR, Kerr SM, Chaiteerakij R, Pestova EV, Clayton AC, Zhang J, Roberts LR, Gores GJ, Halling KC, Kipp BR. An Optimized Set of Fluorescence In Situ Hybridization Probes for Detection of Pancreatobiliary Tract Cancer in Cytology Brush Samples. Gastroenterology 2015; 149: 1813-1824. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 99] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 6. | Gonda TA, Viterbo D, Gausman V, Kipp C, Sethi A, Poneros JM, Gress F, Park T, Khan A, Jackson SA, Blauvelt M, Toney N, Finkelstein SD. Mutation Profile and Fluorescence In Situ Hybridization Analyses Increase Detection of Malignancies in Biliary Strictures. Clin Gastroenterol Hepatol 2017; 15: 913-919. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Zhimin G, Noor H, Jian-Bo Z, Lin W, Jha RK. Advances in diagnosis and treatment of hilar cholangiocarcinoma -- a review. Med Sci Monit. 2013;19:648-656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Tamada K, Ushio J, Sugano K. Endoscopic diagnosis of extrahepatic bile duct carcinoma: Advances and current limitations. World J Clin Oncol. 2011;2:203-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 58] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 9. | Ni Q, Wang H, Zhang Y, Qian L, Chi J, Liang X, Chen T, Wang J. MDCT assessment of resectability in hilar cholangiocarcinoma. Abdom Radiol (NY). 2017;42:851-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Blechacz B, Komuta M, Roskams T, Gores GJ. Clinical diagnosis and staging of cholangiocarcinoma. Nat Rev Gastroenterol Hepatol. 2011;8:512-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 427] [Cited by in RCA: 533] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 11. | Sadeghi A, Mohamadnejad M, Islami F, Keshtkar A, Biglari M, Malekzadeh R, Eloubeidi MA. Diagnostic yield of EUS-guided FNA for malignant biliary stricture: a systematic review and meta-analysis. Gastrointest Endosc 2016; 83: 290-8. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 97] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 12. | Raine T, Thomas JP, Brais R, Godfrey E, Carroll NR, Metz AJ. Test performance and predictors of accuracy of endoscopic ultrasound-guided fine-needle aspiration for diagnosing biliary strictures or masses. Endosc Int Open. 2020;8:E1537-E1544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Mohamadnejad M, DeWitt JM, Sherman S, LeBlanc JK, Pitt HA, House MG, Jones KJ, Fogel EL, McHenry L, Watkins JL, Cote GA, Lehman GA, Al-Haddad MA. Role of EUS for preoperative evaluation of cholangiocarcinoma: a large single-center experience. Gastrointest Endosc. 2011;73:71-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 155] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 14. | Ito K, Ito H, Allen PJ, Gonen M, Klimstra D, D'Angelica MI, Fong Y, DeMatteo RP, Brennan MF, Blumgart LH, Jarnagin WR. Adequate lymph node assessment for extrahepatic bile duct adenocarcinoma. Ann Surg. 2010;251:675-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 114] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 15. | Malikowski T, Levy MJ, Gleeson FC, Storm AC, Vargas EJ, Topazian MD, Abu Dayyeh BK, Iyer PG, Rajan E, Gores GJ, Roberts LR, Chandrasekhara V. Endoscopic Ultrasound/Fine Needle Aspiration Is Effective for Lymph Node Staging in Patients With Cholangiocarcinoma. Hepatology. 2020;72:940-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 16. | Tantau M, Pop T, Badea R, Spirchez Z, Moşteanu O, Tantau A. Intraductal ultrasonography for the assessment of preoperative biliary and pancreatic strictures. J Gastrointestin Liver Dis. 2008;17:217-222. [PubMed] |
| 17. | Valery PC, Laversanne M, Clark PJ, Petrick JL, McGlynn KA, Bray F. Projections of primary liver cancer to 2030 in 30 countries worldwide. Hepatology. 2018;67:600-611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 235] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 18. | DeOliveira ML, Cunningham SC, Cameron JL, Kamangar F, Winter JM, Lillemoe KD, Choti MA, Yeo CJ, Schulick RD. Cholangiocarcinoma: thirty-one-year experience with 564 patients at a single institution. Ann Surg. 2007;245:755-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 882] [Cited by in RCA: 1017] [Article Influence: 56.5] [Reference Citation Analysis (1)] |
| 19. | Lim JH. Cholangiocarcinoma: morphologic classification according to growth pattern and imaging findings. AJR Am J Roentgenol. 2003;181:819-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 215] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 20. | Mihalache F, Tantau M, Diaconu B, Acalovschi M. Survival and quality of life of cholangiocarcinoma patients: a prospective study over a 4 year period. J Gastrointestin Liver Dis. 2010;19:285-290. [PubMed] |
| 21. | Jarnagin WR, Fong Y, DeMatteo RP, Gonen M, Burke EC, Bodniewicz BS J, Youssef BA M, Klimstra D, Blumgart LH. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507-17; discussion 517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 973] [Cited by in RCA: 964] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 22. | Darwish Murad S, Kim WR, Harnois DM, Douglas DD, Burton J, Kulik LM, Botha JF, Mezrich JD, Chapman WC, Schwartz JJ, Hong JC, Emond JC, Jeon H, Rosen CB, Gores GJ, Heimbach JK. Efficacy of neoadjuvant chemoradiation, followed by liver transplantation, for perihilar cholangiocarcinoma at 12 US centers. Gastroenterology 2012; 143: 88-98. quiz e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 395] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 23. | Prachayakul V, Chaisayan S, Aswakul P, Deesomsak M. Clinical characteristics and treatment outcomes of patients with unresectable cholangiocarcinoma in Thailand: are there differences dependent on stent type? Asian Pac J Cancer Prev. 2013;14:529-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Prachayakul V. Current Status of Endoscopic Treatment of Advanced Hilar Cholangiocarcinoma. J Gastroint Dig Syst. 2014;4:168. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 25. | Rizvi S, Khan SA, Hallemeier CL, Kelley RK, Gores GJ. Cholangiocarcinoma - evolving concepts and therapeutic strategies. Nat Rev Clin Oncol. 2018;15:95-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1052] [Cited by in RCA: 1141] [Article Influence: 163.0] [Reference Citation Analysis (0)] |
| 26. | Brandi G, Venturi M, Pantaleo MA, Ercolani G; GICO. Cholangiocarcinoma: Current opinion on clinical practice diagnostic and therapeutic algorithms: A review of the literature and a long-standing experience of a referral center. Dig Liver Dis. 2016;48:231-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 27. | Sofi AA, Khan MA, Das A, Sachdev M, Khuder S, Nawras A, Lee W. Radiofrequency ablation combined with biliary stent placement versus stent placement alone for malignant biliary strictures: a systematic review and meta-analysis. Gastrointest Endosc 2018; 87: 944-951. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 28. | Mansour JC, Aloia TA, Crane CH, Heimbach JK, Nagino M, Vauthey JN. Hilar cholangiocarcinoma: expert consensus statement. HPB (Oxford). 2015;17:691-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 280] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 29. | Liu F, Li Y, Wei Y, Li B. Preoperative biliary drainage before resection for hilar cholangiocarcinoma: whether or not? Dig Dis Sci. 2011;56:663-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 30. | Takahashi Y, Nagino M, Nishio H, Ebata T, Igami T, Nimura Y. Percutaneous transhepatic biliary drainage catheter tract recurrence in cholangiocarcinoma. Br J Surg. 2010;97:1860-1866. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 132] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 31. | Gouma DJ. Multicentre European study of preoperative biliary drainage for hilar cholangiocarcinoma (Br J Surg 2013; 100: 274-283). Br J Surg. 2013;100:283-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 32. | Sakata J, Shirai Y, Wakai T, Nomura T, Sakata E, Hatakeyama K. Catheter tract implantation metastases associated with percutaneous biliary drainage for extrahepatic cholangiocarcinoma. World J Gastroenterol. 2005;11:7024-7027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 82] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 33. | Moole H, Jaeger A, Cashman M, Volmar FH, Dhillon S, Bechtold ML, Puli SR. Are self-expandable metal stents superior to plastic stents in palliating malignant distal biliary strictures? Med J Armed Forces India. 2017;73:42-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Zhao XQ, Dong JH, Jiang K, Huang XQ, Zhang WZ. Comparison of percutaneous transhepatic biliary drainage and endoscopic biliary drainage in the management of malignant biliary tract obstruction: a meta-analysis. Dig Endosc. 2015;27:137-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 79] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 35. | Inamdar S, Slattery E, Bhalla R, Sejpal DV, Trindade AJ. Comparison of Adverse Events for Endoscopic vs Percutaneous Biliary Drainage in the Treatment of Malignant Biliary Tract Obstruction in an Inpatient National Cohort. JAMA Oncol. 2016;2:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 36. | Yan K, Tian J, Xu J, Fu Y, Zhang H, Song J. The value of preoperative biliary drainage in hilar cholangiocarcinoma: a systematic review and meta-analysis of 10 years’ literatures. Int J ClinExp Med. 2018;119:3462-3472. |
| 37. | Farges O, Regimbeau JM, Fuks D, Le Treut YP, Cherqui D, Bachellier P, Mabrut JY, Adham M, Pruvot FR, Gigot JF. Multicentre European study of preoperative biliary drainage for hilar cholangiocarcinoma. Br J Surg. 2013;100:274-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 173] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 38. | Miura F, Sano K, Wada K, Shibuya M, Ikeda Y, Takahashi K, Kainuma M, Kawamura S, Hayano K, Takada T. Prognostic impact of type of preoperative biliary drainage in patients with distal cholangiocarcinoma. Am J Surg 2017; 214: 256-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 39. | George C, Byass OR, Cast JE. Interventional radiology in the management of malignant biliary obstruction. World J Gastrointest Oncol. 2010;2:146-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 40. | Paik WH, Park YS, Hwang JH, Lee SH, Yoon CJ, Kang SG, Lee JK, Ryu JK, Kim YT, Yoon YB. Palliative treatment with self-expandable metallic stents in patients with advanced type III or IV hilar cholangiocarcinoma: a percutaneous versus endoscopic approach. Gastrointest Endosc. 2009;69:55-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 188] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 41. | Coelen RJS, Roos E, Wiggers JK, Besselink MG, Buis CI, Busch ORC, Dejong CHC, van Delden OM, van Eijck CHJ, Fockens P, Gouma DJ, Koerkamp BG, de Haan MW, van Hooft JE, IJzermans JNM, Kater GM, Koornstra JJ, van Lienden KP, Moelker A, Damink SWMO, Poley JW, Porte RJ, de Ridder RJ, Verheij J, van Woerden V, Rauws EAJ, Dijkgraaf MGW, van Gulik TM. Endoscopic versus percutaneous biliary drainage in patients with resectable perihilar cholangiocarcinoma: a multicentre, randomised controlled trial. Lancet Gastroenterol Hepatol. 2018;3:681-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 128] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 42. | Kawashima H, Itoh A, Ohno E, Itoh Y, Ebata T, Nagino M, Goto H, Hirooka Y. Preoperative endoscopic nasobiliary drainage in 164 consecutive patients with suspected perihilar cholangiocarcinoma: a retrospective study of efficacy and risk factors related to complications. Ann Surg. 2013;257:121-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 116] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 43. | Smith AC, Dowsett JF, Russell RC, Hatfield AR, Cotton PB. Randomised trial of endoscopic stenting versus surgical bypass in malignant low bileduct obstruction. Lancet. 1994;344:1655-1660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 603] [Cited by in RCA: 554] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 44. | Anderson CA, Stuart KE, Palta M, Goldberg RM, Tanabe KK, Savarese D. Treatment options for locally advanced un resectable but nonmetastatic cholangiocarcinoma, 2018; 633. Available from: https://www.uptodate.com/contents/treatment-options-for-locally-advanced-unresectable-but-nonmetastatic-cholangiocarcinoma. |
| 45. | Kerdsirichairat T, Arain MA, Attam R, Glessing B, Bakman Y, Amateau SK, Freeman ML. Endoscopic Drainage of >50% of Liver in Malignant Hilar Biliary Obstruction Using Metallic or Fenestrated Plastic Stents. Clin Transl Gastroenterol. 2017;8:e115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 46. | Testoni PA, Mariani A, Aabakken L, Arvanitakis M, Bories E, Costamagna G, Devière J, Dinis-Ribeiro M, Dumonceau JM, Giovannini M, Gyokeres T, Hafner M, Halttunen J, Hassan C, Lopes L, Papanikolaou IS, Tham TC, Tringali A, van Hooft J, Williams EJ. Papillary cannulation and sphincterotomy techniques at ERCP: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2016;48:657-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 384] [Article Influence: 42.7] [Reference Citation Analysis (1)] |
| 47. | Leng JJ, Zhang N, Dong JH. Percutaneous transhepatic and endoscopic biliary drainage for malignant biliary tract obstruction: a meta-analysis. World J Surg Oncol. 2014;12:272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 48. | Fabbri C, Luigiano C, Lisotti A, Cennamo V, Virgilio C, Caletti G, Fusaroli P. Endoscopic ultrasound-guided treatments: are we getting evidence based--a systematic review. World J Gastroenterol. 2014;20:8424-8448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 105] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 49. | Gupta K, Perez-Miranda M, Kahaleh M, Artifon EL, Itoi T, Freeman ML, de-Serna C, Sauer B, Giovannini M; InEBD STUDY GROUP. Endoscopic ultrasound-assisted bile duct access and drainage: multicenter, long-term analysis of approach, outcomes, and complications of a technique in evolution. J Clin Gastroenterol. 2014;48:80-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 110] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 50. | Artifon EL, Aparicio D, Paione JB, Lo SK, Bordini A, Rabello C, Otoch JP, Gupta K. Biliary drainage in patients with unresectable, malignant obstruction where ERCP fails: endoscopic ultrasonography-guided choledochoduodenostomy versus percutaneous drainage. J Clin Gastroenterol. 2012;46:768-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 177] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 51. | Bapaye A, Dubale N, Aher A. Comparison of endosonography-guided vs. percutaneous biliary stenting when papilla is inaccessible for ERCP. United European Gastroenterol J. 2013;1:285-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 52. | Khashab MA, Valeshabad AK, Afghani E, Singh VK, Kumbhari V, Messallam A, Saxena P, El Zein M, Lennon AM, Canto MI, Kalloo AN. A comparative evaluation of EUS-guided biliary drainage and percutaneous drainage in patients with distal malignant biliary obstruction and failed ERCP. Dig Dis Sci. 2015;60:557-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 158] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 53. | Dhir V, Itoi T, Khashab MA, Park DH, Yuen Bun Teoh A, Attam R, Messallam A, Varadarajulu S, Maydeo A. Multicenter comparative evaluation of endoscopic placement of expandable metal stents for malignant distal common bile duct obstruction by ERCP or EUS-guided approach. Gastrointest Endosc. 2015;81:913-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 129] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 54. | Yasuda I, Mukai T, Moriwaki H. Unilateral versus bilateral endoscopic biliary stenting for malignant hilar biliary strictures. Dig Endosc. 2013;25 Suppl 2:81-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 55. | Iwano H, Ryozawa S, Ishigaki N, Taba K, Senyo M, Yoshida K, Sakaida I. Unilateral versus bilateral drainage using self-expandable metallic stent for unresectable hilar biliary obstruction. Dig Endosc. 2011;23:43-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 56. | Mukai T, Yasuda I, Nakashima M, Doi S, Iwashita T, Iwata K, Kato T, Tomita E, Moriwaki H. Metallic stents are more efficacious than plastic stents in unresectable malignant hilar biliary strictures: a randomized controlled trial. J Hepatobiliary Pancreat Sci. 2013;20:214-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 167] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 57. | Naitoh I, Ohara H, Nakazawa T, Ando T, Hayashi K, Okumura F, Okayama Y, Sano H, Kitajima Y, Hirai M, Ban T, Miyabe K, Ueno K, Yamashita H, Joh T. Unilateral versus bilateral endoscopic metal stenting for malignant hilar biliary obstruction. J Gastroenterol Hepatol. 2009;24:552-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 155] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 58. | Liberato MJ, Canena JM. Endoscopic stenting for hilar cholangiocarcinoma: efficacy of unilateral and bilateral placement of plastic and metal stents in a retrospective review of 480 patients. BMC Gastroenterol. 2012;12:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 112] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 59. | Lee TH, Kim TH, Moon JH, Lee SH, Choi HJ, Hwangbo Y, Hyun JJ, Choi JH, Jeong S, Kim JH, Park DH, Han JH, Park SH. Bilateral versus unilateral placement of metal stents for inoperable high-grade malignant hilar biliary strictures: a multicenter, prospective, randomized study (with video). Gastrointest Endosc. 2017;86:817-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 60. | De Palma GD, Galloro G, Siciliano S, Iovino P, Catanzano C. Unilateral versus bilateral endoscopic hepatic duct drainage in patients with malignant hilar biliary obstruction: results of a prospective, randomized, and controlled study. Gastrointest Endosc. 2001;53:547-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 336] [Cited by in RCA: 290] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 61. | Ashat M, Arora S, Klair JS, Childs CA, Murali AR, Johlin FC. Bilateral vs unilateral placement of metal stents for inoperable high-grade hilar biliary strictures: A systemic review and meta-analysis. World J Gastroenterol. 2019;25:5210-5219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 62. | Almadi MA, Barkun A, Martel M. Plastic vs. Self-Expandable Metal Stents for Palliation in Malignant Biliary Obstruction: A Series of Meta-Analyses. Am J Gastroenterol. 2017;112:260-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 190] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 63. | Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7953] [Cited by in RCA: 8102] [Article Influence: 506.4] [Reference Citation Analysis (2)] |
| 64. | Zorrón Pu L, de Moura EG, Bernardo WM, Baracat FI, Mendonça EQ, Kondo A, Luz GO, Furuya Júnior CK, Artifon EL. Endoscopic stenting for inoperable malignant biliary obstruction: A systematic review and meta-analysis. World J Gastroenterol. 2015;21:13374-13385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 64] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 65. | Sawas T, Al Halabi S, Parsi MA, Vargo JJ. Self-expandable metal stents versus plastic stents for malignant biliary obstruction: a meta-analysis. Gastrointest Endosc 2015; 82: 256-267. e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 172] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 66. | Hong WD, Chen XW, Wu WZ, Zhu QH, Chen XR. Metal versus plastic stents for malignant biliary obstruction: an update meta-analysis. Clin Res Hepatol Gastroenterol. 2013;37:496-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 67. | Ridtitid W, Rerknimitr R, Janchai A, Kongkam P, Treeprasertsuk S, Kullavanijaya P. Outcome of second interventions for occluded metallic stents in patients with malignant biliary obstruction. Surg Endosc. 2010;24:2216-2220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 68. | Hong W, Sun X, Zhu Q. Endoscopic stenting for malignant hilar biliary obstruction: should it be metal or plastic and unilateral or bilateral? Eur J Gastroenterol Hepatol. 2013;25:1105-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 69. | Hwang JC, Kim JH, Lim SG, Kim SS, Yoo BM, Cho SW. Y-shaped endoscopic bilateral metal stent placement for malignant hilar biliary obstruction: prospective long-term study. Scand J Gastroenterol. 2011;46:326-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 70. | Lee TH, Park DH, Lee SS, Choi HJ, Lee JK, Kim TH, Kim JH, Jeong S, Park SH, Moon JH. Technical feasibility and revision efficacy of the sequential deployment of endoscopic bilateral side-by-side metal stents for malignant hilar biliary strictures: a multicenter prospective study. Dig Dis Sci. 2013;58:547-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 71. | Saluja SS, Gulati M, Garg PK, Pal H, Pal S, Sahni P, Chattopadhyay TK. Endoscopic or percutaneous biliary drainage for gallbladder cancer: a randomized trial and quality of life assessment. Clin Gastroenterol Hepatol 2008; 6: 944-950. e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 72. | Piñol V, Castells A, Bordas JM, Real MI, Llach J, Montañà X, Feu F, Navarro S. Percutaneous self-expanding metal stents versus endoscopic polyethylene endoprostheses for treating malignant biliary obstruction: randomized clinical trial. Radiology. 2002;225:27-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 93] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 73. | Arvanitakis M, Van Laethem JL, Pouzere S, Le Moine O, Deviere J. Predictive factors for survival in patients with inoperable Klatskin tumors. Hepatogastroenterology. 2006;53:21-27. [PubMed] |
| 74. | Artifon EL, Ferreira FC, Sakai P. Endoscopic ultrasound-guided biliary drainage. Korean J Radiol. 2012;13 Suppl 1:S74-S82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 75. | Horaguchi J, Fujita N, Noda Y, Kobayashi G, Ito K, Obana T, Takasawa O, Koshita S, Kanno Y. Endosonography-guided biliary drainage in cases with difficult transpapillary endoscopic biliary drainage. Dig Endosc. 2009;21:239-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 76. | Bories E, Pesenti C, Caillol F, Lopes C, Giovannini M. Transgastric endoscopic ultrasonography-guided biliary drainage: results of a pilot study. Endoscopy. 2007;39:287-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 164] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 77. | Park DH, Koo JE, Oh J, Lee YH, Moon SH, Lee SS, Seo DW, Lee SK, Kim MH. EUS-guided biliary drainage with one-step placement of a fully covered metal stent for malignant biliary obstruction: a prospective feasibility study. Am J Gastroenterol. 2009;104:2168-2174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 133] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 78. | Perez-Miranda M, de la Serna C, Diez-Redondo P, Vila JJ. Endosonography-guided cholangiopancreatography as a salvage drainage procedure for obstructed biliary and pancreatic ducts. World J Gastrointest Endosc. 2010;2:212-222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 37] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 79. | Park DH, Song TJ, Eum J, Moon SH, Lee SS, Seo DW, Lee SK, Kim MH. EUS-guided hepaticogastrostomy with a fully covered metal stent as the biliary diversion technique for an occluded biliary metal stent after a failed ERCP (with videos). Gastrointest Endosc. 2010;71:413-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 84] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 80. | Kawakubo K, Isayama H, Kato H, Itoi T, Kawakami H, Hanada K, Ishiwatari H, Yasuda I, Kawamoto H, Itokawa F, Kuwatani M, Iiboshi T, Hayashi T, Doi S, Nakai Y. Multicenter retrospective study of endoscopic ultrasound-guided biliary drainage for malignant biliary obstruction in Japan. J Hepatobiliary Pancreat Sci. 2014;21:328-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 172] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 81. | Park DH, Jang JW, Lee SS, Seo DW, Lee SK, Kim MH. EUS-guided biliary drainage with transluminal stenting after failed ERCP: predictors of adverse events and long-term results. Gastrointest Endosc. 2011;74:1276-1284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 244] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 82. | Kaffes AJ, Sriram PV, Rao GV, Santosh D, Reddy DN. Early institution of pre-cutting for difficult biliary cannulation: a prospective study comparing conventional vs. a modified technique. Gastrointest Endosc. 2005;62: 669-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 83. | Park DH. Endoscopic ultrasound-guided biliary drainage of hilar biliary obstruction. J Hepatobiliary Pancreat Sci. 2015;22:664-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 84. | Ogura T, Sano T, Onda S, Imoto A, Masuda D, Yamamoto K, Kitano M, Takeuchi T, Inoue T, Higuchi K. Endoscopic ultrasound-guided biliary drainage for right hepatic bile duct obstruction: novel technical tips. Endoscopy. 2015;47:72-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 85. | Park SJ, Choi JH, Park DH, Choi JH, Lee SS, Seo DW, Lee SK, Kim MH. Expanding indication: EUS-guided hepaticoduodenostomy for isolated right intrahepatic duct obstruction (with video). Gastrointest Endosc. 2013;78:374-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 86. | Nakai Y, Kogure H, Isayama H, Koike K. Endoscopic Ultrasound-Guided Biliary Drainage for Unresectable Hilar Malignant Biliary Obstruction. Clin Endosc. 2019;52:220-225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 87. | Minaga K, Takenaka M, Kitano M, Chiba Y, Imai H, Yamao K, Kamata K, Miyata T, Omoto S, Sakurai T, Watanabe T, Nishida N, Kudo M. Rescue EUS-guided intrahepatic biliary drainage for malignant hilar biliary stricture after failed transpapillary re-intervention. Surg Endosc. 2017;31:4764-4772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 88. | Kongkam P, Tasneem AA, Rerknimitr R. Combination of endoscopic retrograde cholangiopancreatography and endoscopic ultrasonography-guided biliary drainage in malignant hilar biliary obstruction. Dig Endosc. 2019;31 Suppl 1:50-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 89. | Pereira P, Santos S, Morais R, Gaspar R, Rodrigues-Pinto E, Vilas-Boas F, Macedo G. Role of Peroral Cholangioscopy for Diagnosis and Staging of Biliary Tumors. Dig Dis. 2020;38:431-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 90. | Kaura K, Sawas T, Bazerbachi F, Storm AC, Martin JA, Gores GJ, Abu Dayyeh BK, Topazian MD, Levy MJ, Petersen BT, Chandrasekhara V. Cholangioscopy Biopsies Improve Detection of Cholangiocarcinoma When Combined with Cytology and FISH, but Not in Patients with PSC. Dig Dis Sci. 2020;65:1471-1478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 91. | Fukasawa Y, Takano S, Fukasawa M, Maekawa S, Kadokura M, Shindo H, Takahashi E, Hirose S, Kawakami S, Hayakawa H, Yamaguchi T, Nakayama Y, Inoue T, Sato T, Enomoto N. Form-Vessel Classification of Cholangioscopy Findings to Diagnose Biliary Tract Carcinoma's Superficial Spread. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 92. | Steel AW, Postgate AJ, Khorsandi S, Nicholls J, Jiao L, Vlavianos P, Habib N, Westaby D. Endoscopically applied radiofrequency ablation appears to be safe in the treatment of malignant biliary obstruction. Gastrointest Endosc. 2011;73:149-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 225] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 93. | Rafi S, Piduru SM, El-Rayes B, Kauh JS, Kooby DA, Sarmiento JM, Kim HS. Yttrium-90 radioembolization for unresectable standard-chemorefractory intrahepatic cholangiocarcinoma: survival, efficacy, and safety study. Cardiovasc Intervent Radiol. 2013;36:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 94. | Mouli S, Memon K, Baker T, Benson AB 3rd, Mulcahy MF, Gupta R, Ryu RK, Salem R, Lewandowski RJ. Yttrium-90 radioembolization for intrahepatic cholangiocarcinoma: safety, response, and survival analysis. J Vasc Interv Radiol. 2013;24:1227-1234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 164] [Article Influence: 13.7] [Reference Citation Analysis (1)] |
| 95. | Hoffmann RT, Paprottka PM, Schön A, Bamberg F, Haug A, Dürr EM, Rauch B, Trumm CT, Jakobs TF, Helmberger TK, Reiser MF, Kolligs FT. Transarterial hepatic yttrium-90 radioembolization in patients with unresectable intrahepatic cholangiocarcinoma: factors associated with prolonged survival. Cardiovasc Intervent Radiol. 2012;35:105-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 165] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 96. | Camus M, Napoléon B, Vienne A, Le Rhun M, Leblanc S, Barret M, Chaussade S, Robin F, Kaddour N, Prat F. Efficacy and safety of endobiliary radiofrequency ablation for the eradication of residual neoplasia after endoscopic papillectomy: a multicenter prospective study. Gastrointest Endosc. 2018;88:511-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 97. | Talreja JP, Kahaleh M. Photodynamic therapy for cholangiocarcinoma. Gut Liver. 2010;4 Suppl 1:S62-S66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 98. | Lee TY, Cheon YK, Shim CS. Current status of photodynamic therapy for bile duct cancer. Clin Endosc. 2013;46:38-44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 99. | Witzigmann H, Berr F, Ringel U, Caca K, Uhlmann D, Schoppmeyer K, Tannapfel A, Wittekind C, Mossner J, Hauss J, Wiedmann M. Surgical and palliative management and outcome in 184 patients with hilar cholangiocarcinoma: palliative photodynamic therapy plus stenting is comparable to r1/r2 resection. Ann Surg. 2006;244:230-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 198] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 100. | Dumoulin FL, Gerhardt T, Fuchs S, Scheurlen C, Neubrand M, Layer G, Sauerbruch T. Phase II study of photodynamic therapy and metal stent as palliative treatment for nonresectable hilar cholangiocarcinoma. Gastrointest Endosc. 2003;57:860-867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 101] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 101. | Wiedmann M, Caca K, Berr F, Schiefke I, Tannapfel A, Wittekind C, Mössner J, Hauss J, Witzigmann H. Neoadjuvant photodynamic therapy as a new approach to treating hilar cholangiocarcinoma: a phase II pilot study. Cancer. 2003;97:2783-2790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 117] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 102. | Kahaleh M, Mishra R, Shami VM, Northup PG, Berg CL, Bashlor P, Jones P, Ellen K, Weiss GR, Brenin CM, Kurth BE, Rich TA, Adams RB, Yeaton P. Unresectable cholangiocarcinoma: comparison of survival in biliary stenting alone versus stenting with photodynamic therapy. Clin Gastroenterol Hepatol. 2008;6:290-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 127] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 103. | Zoepf T, Jakobs R, Arnold JC, Apel D, Riemann JF. Palliation of nonresectable bile duct cancer: improved survival after photodynamic therapy. Am J Gastroenterol. 2005;100:2426-2430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 233] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 104. | Gao F, Bai Y, Ma SR, Liu F, Li ZS. Systematic review: photodynamic therapy for unresectable cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2010;17:125-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 105. | Tomizawa Y, Tian J. Photodynamic therapy for unresectable cholangiocarcinoma. Dig Dis Sci. 2012;57:274-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 106. | Ortner ME, Caca K, Berr F, Liebetruth J, Mansmann U, Huster D, Voderholzer W, Schachschal G, Mössner J, Lochs H. Successful photodynamic therapy for nonresectable cholangiocarcinoma: a randomized prospective study. Gastroenterology. 2003;125:1355-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 383] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 107. | Prasad GA, Wang KK, Baron TH, Buttar NS, Wongkeesong LM, Roberts LR, LeRoy AJ, Lutzke LS, Borkenhagen LS. Factors associated with increased survival after photodynamic therapy for cholangiocarcinoma. Clin Gastroenterol Hepatol. 2007;5:743-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 108. | Wentrup R, Winkelmann N, Mitroshkin A, Prager M, Voderholzer W, Schachschal G, Jürgensen C, Büning C. Photodynamic Therapy Plus Chemotherapy Compared with Photodynamic Therapy Alone in Hilar Nonresectable Cholangiocarcinoma. Gut Liver. 2016;10:470-475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 109. | Park DH, Lee SS, Park SE, Lee JL, Choi JH, Choi HJ, Jang JW, Kim HJ, Eum JB, Seo DW, Lee SK, Kim MH, Lee JB. Randomised phase II trial of photodynamic therapy plus oral fluoropyrimidine, S-1, versus photodynamic therapy alone for unresectable hilar cholangiocarcinoma. Eur J Cancer. 2014;50:1259-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 110. | Knüppel M, Kubicka S, Vogel A, Malek NP, Schneider M, Papendorf F, Greten T, Wedemeyer J, Schneider A. Combination of conservative and interventional therapy strategies for intra- and extrahepatic cholangiocellular carcinoma: a retrospective survival analysis. Gastroenterol Res Pract. 2012;2012:190708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 111. | Fuks D, Bartoli E, Delcenserie R, Yzet T, Celice P, Sabbagh C, Chatelain D, Joly JP, Cheron N, Dupas JL, Regimbeau JM. Biliary drainage, photodynamic therapy and chemotherapy for unresectable cholangiocarcinoma with jaundice. J Gastroenterol Hepatol. 2009;24:1745-1752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 112. | Hong MJ, Cheon YK, Lee EJ, Lee TY, Shim CS. Long-term outcome of photodynamic therapy with systemic chemotherapy compared to photodynamic therapy alone in patients with advanced hilar cholangiocarcinoma. Gut Liver. 2014;8:318-323. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 113. | Wagner A, Wiedmann M, Tannapfel A, Mayr C, Kiesslich T, Wolkersdörfer GW, Berr F, Hauss J, Witzigmann H. Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy--Long-Term Outcome of a Phase II Pilot Study. Int J Mol Sci. 2015;16:26619-26628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 114. | Lu Y, Liu L, Wu JC, Bie LK, Gong B. Efficacy and safety of photodynamic therapy for unresectable cholangiocarcinoma: A meta-analysis. Clin Res Hepatol Gastroenterol. 2015;39:718-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 115. | Bae BC, Yang SG, Jeong S, Lee DH, Na K, Kim JM, Costamagna G, Kozarek RA, Isayama H, Deviere J, Seo DW, Nageshwar Reddy D. Polymeric photosensitizer-embedded self-expanding metal stent for repeatable endoscopic photodynamic therapy of cholangiocarcinoma. Biomaterials. 2014;35:8487-8495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 116. | Skowronek J, Zwierzchowski G. Brachytherapy in the treatment of bile duct cancer - a tough challenge. J Contemp Brachytherapy. 2017;9:187-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 117. | Mukewar S, Gupta A, Baron TH, Gores G, Furutani K, Haddock MG, Hallemeier CL. Endoscopically inserted nasobiliary catheters for high dose-rate brachytherapy as part of neoadjuvant therapy for perihilar cholangiocarcinoma. Endoscopy. 2015;47:878-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 118. | Fletcher MS, Brinkley D, Dawson JL, Nunnerley H, Williams R. Treatment of hilar carcinoma by bile drainage combined with internal radiotherapy using 192iridium wire. Br J Surg. 1983;70:733-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 60] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 119. | Karani J, Fletcher M, Brinkley D, Dawson JL, Williams R, Nunnerley H. Internal biliary drainage and local radiotherapy with iridium-192 wire in treatment of hilar cholangiocarcinoma. Clin Radiol. 1985;36: 603-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 58] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 120. | Chen Y, Wang XL, Yan ZP, Cheng JM, Wang JH, Gong GQ, Qian S, Luo JJ, Liu QX. HDR-192Ir intraluminal brachytherapy in treatment of malignant obstructive jaundice. World J Gastroenterol. 2004;10:3506-3510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 41] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 121. | Yoshioka Y, Ogawa K, Oikawa H, Onishi H, Kanesaka N, Tamamoto T, Kosugi T, Hatano K, Kobayashi M, Ito Y, Takayama M, Takemoto M, Karasawa K, Nagakura H, Imai M, Kosaka Y, Yamazaki H, Isohashi F, Nemoto K, Nishimura Y; Japanese Radiation Oncology Study Group (JROSG). Impact of intraluminal brachytherapy on survival outcome for radiation therapy for unresectable biliary tract cancer: a propensity-score matched-pair analysis. Int J Radiat Oncol Biol Phys. 2014;89:822-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 122. | Deufel CL, Furutani KM, Dahl RA, Grams MP, McLemore LB, Hallemeier CL, Neben-Wittich M, Martenson JA, Haddock MG. Technique for the administration of high-dose-rate brachytherapy to the bile duct using a nasobiliary catheter. Brachytherapy. 2018;17:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 123. | Banales JM, Marin JJG, Lamarca A, Rodrigues PM, Khan SA, Roberts LR, Cardinale V, Carpino G, Andersen JB, Braconi C, Calvisi DF, Perugorria MJ, Fabris L, Boulter L, Macias RIR, Gaudio E, Alvaro D, Gradilone SA, Strazzabosco M, Marzioni M, Coulouarn C, Fouassier L, Raggi C, Invernizzi P, Mertens JC, Moncsek A, Rizvi S, Heimbach J, Koerkamp BG, Bruix J, Forner A, Bridgewater J, Valle JW, Gores GJ. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020;17:557-588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1555] [Cited by in RCA: 1549] [Article Influence: 309.8] [Reference Citation Analysis (0)] |
| 124. | Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, Madhusudan S, Iveson T, Hughes S, Pereira SP, Roughton M, Bridgewater J; ABC-02 Trial Investigators. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med. 2010;362:1273-1281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2617] [Cited by in RCA: 3167] [Article Influence: 211.1] [Reference Citation Analysis (1)] |
| 125. | Okusaka T, Nakachi K, Fukutomi A, Mizuno N, Ohkawa S, Funakoshi A, Nagino M, Kondo S, Nagaoka S, Funai J, Koshiji M, Nambu Y, Furuse J, Miyazaki M, Nimura Y. Gemcitabine alone or in combination with cisplatin in patients with biliary tract cancer: a comparative multicentre study in Japan. Br J Cancer. 2010;103:469-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 570] [Cited by in RCA: 568] [Article Influence: 37.9] [Reference Citation Analysis (0)] |
| 126. | Shroff RT, Javle MM, Xiao L, Kaseb AO, Varadhachary GR, Wolff RA, Raghav KPS, Iwasaki M, Masci P, Ramanathan RK, Ahn DH, Bekaii-Saab TS, Borad MJ. Gemcitabine, Cisplatin, and nab-Paclitaxel for the Treatment of Advanced Biliary Tract Cancers: A Phase 2 Clinical Trial. JAMA Oncol. 2019;5:824-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 345] [Article Influence: 69.0] [Reference Citation Analysis (0)] |
| 127. | Sakai D, Kanai M, KobayashiS, Eguchi H, Baba H, Seo S, Taketomi A, Takayama T, Yamaue H, IshiokaC, Sho M, Takeyama Y, FujimotoJ, Toyoda M, Shimizu J, GotoT, Yoshimura K, Hatano E, NaganoH, Ioka T. Randomized phase III study of gemcitabine, cisplatin plus S-1 (GCS) versus gemcitabine, cisplatin (GC) for advanced biliary tract cancer (KHBO1401-MITSUBA). Ann Oncol. 2018;29:viii205-viii270. [RCA] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 62] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 128. | McNamara MG, Bridgewater J, Palmer D, Wasan H, Ryder WD, Gnanaranjan C, Ghazaly E, Evans TRJ, Valle JW. A new ProTide, NUC-1031, combined with cisplatin for the first- line treatment of advanced biliary tract cancer (ABC-08). Ann Oncol. 2018;29:viii259. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 129. | Lamarca A, Palmer DH, Wasan HS, Ross PJ, Ma YT, Arora A, Falk S, Gillmore R, Wadsley J, Patel K, Anthoney A, Maraveyas A, Waters JS, Hobbs C, Barber S, Ruder D, Ramage J, Davies LM, Bridgewater JA, Valle JW. A randomised phase III, multi- centre, open- label study of Active Symptom Control (ASC) alone or ASC with oxaliplatin/5- FU chemotherapy (ASC+mFOLFOX) for patients (pts) with locally advanced/metastatic biliary tract cancers (ABC) previously- treated with cisplatin/gemcitabine (CisGem) chemotherapy. J Clin Oncol. 2019;37:4003. [RCA] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 143] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 130. | Marin JJG, Lozano E, Briz O, Al-Abdulla R, Serrano MA, Macias RIR. Molecular Bases of Chemoresistance in Cholangiocarcinoma. Curr Drug Targets. 2017;18:889-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 131. | Marin JJG, Lozano E, Herraez E, Asensio M, Di Giacomo S, Romero MR, Briz O, Serrano MA, Efferth T, Macias RIR. Chemoresistance and chemosensitization in cholangiocarcinoma. Biochim Biophys Acta Mol Basis Dis. 2018;1864:1444-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 92] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 132. | Fouassier L, Marzioni M, Afonso MB, Dooley S, Gaston K, Giannelli G, Rodrigues CMP, Lozano E, Mancarella S, Segatto O, Vaquero J, Marin JJG, Coulouarn C. Signalling networks in cholangiocarcinoma: Molecular pathogenesis, targeted therapies and drug resistance. Liver Int. 2019;39 Suppl 1:43-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 133. | Lowery MA, Abou-Alfa G, Burris HA, Janku F, Shroff RT, Clearly JM, Azad NS, Goyal L, Maher EA, Gore L, Hollebecque A, Beeram M, Trent JC, Jiang L, Ishii Y, Auer J, Gliser C, Agresta SV, Pandya SS, Zhu AX. Phase I study of AG-120, an IDH1 mutant enzyme inhibitor: Results from the cholangiocarcinoma dose escalation and expansion cohorts. J Clin Oncol. 2017;35:4015. [RCA] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 134. | Lowery MA, Abou-Alfa GK, Valle JW, Kelley RK, Goyal L, Shroff RT, Javle MM, Borad MJ, Cleary JM, El-Khoueiry AB, Bendell JC, Macarulla T, Vogel A, Korth C, Jiang L, Gliser C, Wu B, Agresta SV, Pandya SS, Zhu AX. ClarIDHy: A phase 3, multicenter, randomized, double-blind study of AG-120 vs placebo in patients with an advanced cholangiocarcinoma with an IDH1 mutation. J Clin Oncol. 2019;35:TPS4142. [RCA] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 135. | Javle MM, Shroff RT, Zhu A, Sadeghi S, Choo SP, Borad MJ, Lowery MA, El-Khoueiry A, Macarulla T, Phillip PA, Oh D-Y, Van Cutsem E, Yeh K-H, Isaacs R, McGarry C, Sen S, Bekaii-Saab TS. A phase 2 study of BGJ398 in patients (pts) with advanced or metastatic FGFRaltered cholangiocarcinoma (CCA) who failed or are intolerant to platinum- based chemotherapy. J Clin Oncol. 2016;34:335. [RCA] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 136. | Mazzafero V, Shaib W, Harris W, Personeni N, El-Raves B, Teicher A, Hall T, Wang Y Schwartz B, Kazakin J, Droz Dit Busset M, Cotsoglou C, Papadopoulos K. ARQ 087, an oral pan- fibroblast growth factor receptor (FGFR) inhibitor, in patients with advanced and/or metastatic intrahepatic cholangiocarcinoma (iCCA). Ann Oncol. 2016;27:ii109. [RCA] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 137. | Meric-Bernstam F, Arkenau H, Tran B, Bahleda R, Kelley R, Hierro C, Ahn D, Zhu A, Javle M, Winkler R, He H, Huang J, Goyal L. Efficacy of TAS-120, an irreversible fibroblast growth factor receptor (FGFR) inhibitor, in cholangiocarcinoma patients with FGFR pathway alterations who were previously treated with chemotherapy and other FGFR inhibitors. Ann Oncol. 2018;29:v100. [RCA] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |