Published online Nov 27, 2021. doi: 10.4254/wjh.v13.i11.1611
Peer-review started: March 17, 2021
First decision: May 2, 2021
Revised: July 5, 2021
Accepted: September 22, 2021
Article in press: September 22, 2021
Published online: November 27, 2021
Processing time: 252 Days and 6.6 Hours
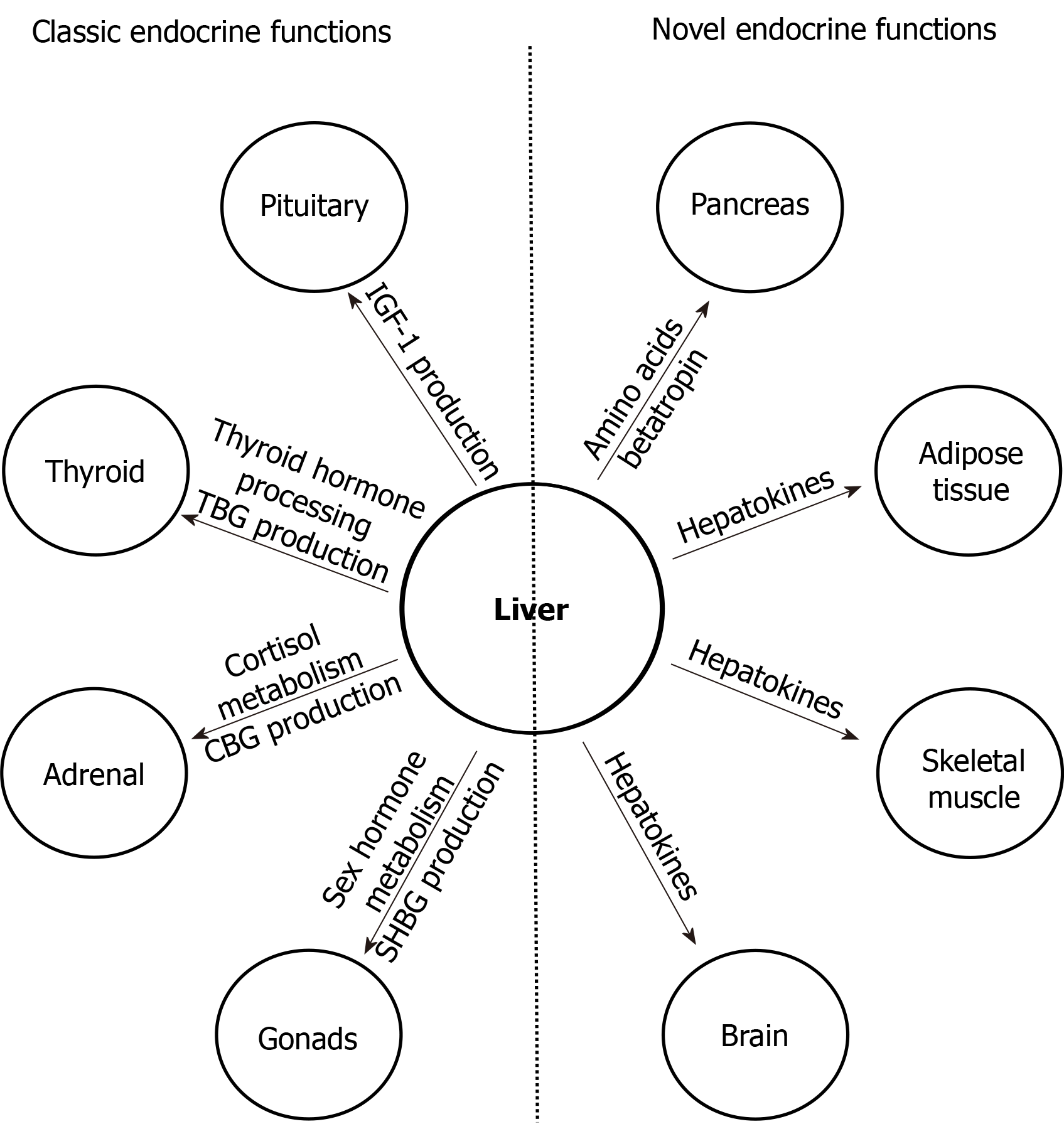
The liver, the largest solid visceral organ of the body, has numerous endocrine functions, such as direct hormone and hepatokine production, hormone metabolism, synthesis of binding proteins, and processing and redistribution of metabolic fuels. In the last 10 years, many new endocrine functions of the liver have been discovered. Advances in the classical endocrine functions include delineation of mechanisms of liver production of endocrine hormones [including 25-hydroxyvitamin D, insulin-like growth factor 1 (IGF-1), and angiotensinogen], hepatic metabolism of hormones (including thyroid hormones, glucagon-like peptide-1, and steroid hormones), and actions of specific binding proteins to glucocorticoids, sex steroids, and thyroid hormones. These studies have furthered insight into cirrhosis-associated endocrinopathies, such as hypogonadism, osteoporosis, IGF-1 deficiency, vitamin D deficiency, alterations in glucose and lipid homeostasis, and controversially relative adrenal insufficiency. Several novel endocrine functions of the liver have also been unraveled, elucidating the liver’s key negative feedback regulatory role in the pancreatic α cell-liver axis, which regulates pancreatic α cell mass, glucagon secretion, and circulating amino acid levels. Betatrophin and other hepatokines, such as fetuin-A and fibroblast growth factor 21, have also been discovered to play important endocrine roles in modulating insulin sensitivity, lipid metabolism, and body weight. It is expected that more endocrine functions of the liver will be revealed in the near future.
Core Tip: The liver has many newly discovered endocrine functions, most of which are in regulating metabolism, underscoring the functioning of the liver as a major metabolic organ. Convincing evidence has shown that the liver regulates endocrine functions in mineral and fuel metabolism, especially in the metabolism of glucose and lipids via hepatokines and amino acids via negative feedback on pancreatic α cells. As research into the endocrine function of the liver is a rapidly evolving field, controversial findings often exist; caution needs to be taken when interpreting novel findings to avoid over-simplification of complex metabolic processes and premature allocation of research resources.
- Citation: Rhyu J, Yu R. Newly discovered endocrine functions of the liver. World J Hepatol 2021; 13(11): 1611-1628
- URL: https://www.wjgnet.com/1948-5182/full/v13/i11/1611.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i11.1611
The liver is a dynamic endocrine organ and mediates critical metabolic pathways via roles in direct hormone and hepatokine production, hormone metabolism, synthesis of binding proteins, detoxification, and processing and redistribution of metabolic fuels[1-4]. It participates in multiple signaling pathways with other endocrine organs, including the pituitary, pancreas, gut, thyroid, adrenal glands, and bone, with hormones in turn modulating the liver’s metabolic and synthetic functions[1,5]. Diseases that affect the liver lead to a variety of endocrine manifestations, including hypogonadism, osteoporosis, effects on glucose metabolism and growth hormone (GH), and controversial effects on cortisol[1,5].
The liver, with its vascularity, is well-positioned to provide and receive endocrine signals, including those from pancreatic and gut hormones[6]. It also receives exposure to antigen-rich blood systemically and from the gastrointestinal system as a lymphoid organ[7] and serves as a principal organ in drug metabolism and clearance[8]. Despite only representing 2.5% of the body weight, the liver receives up to 25% of the total cardiac output at rest[9]. It also receives a unique double afferent blood flow from the hepatic artery and partially deoxygenated portal vein, with around 75% of the blood flow from the latter[9]. The portal vein, in turn, receives blood from the stomach, small and large intestines, pancreas, spleen, and gallbladder[9], with direct physiological implications on the regulation of metabolism by endocrine liver functions[6]. Great progress has been made in the understanding of the endocrine functions of the liver in the last 10 years.
We will first briefly summarize the advances in the understanding of the liver classic endocrine functions (Table 1).
| Hormone | Liver function | Target organ | Action on target organ | Alteration in liver diseases |
| 25-hydroxyvitamin D | Direct production | Gut | Prohormone of calcitriol which stimulates gut calcium absorption | Decreased production resulting in low bone density |
| Insulin-like growth factor 1 | Direct production | Ubiquitous | Promoting growth and differentiation and regulating nutrients metabolism | Decreased production resulting in dysmetabolism |
| Angiotensinogen | Direct production | Cardiovascular system | Precursor of angiotensin II which regulates aldosterone level. Both regulate vascular tone, sodium retention, and cardiac remodeling | Near-normal function |
| Thyroid hormone | Activation through T4 to T3 conversion; inactivation through degradation; TBG production | Ubiquitous | Increasing metabolism and energy expenditure | Low T3 syndrome |
| Glucagon-like peptide 1 (GLP-1) | Metabolism of GLP-1 via dipeptidyl peptidase-4(DPPIV) | Pancreas, gut, and brain | Stimulating insulin production, decreasing gut motility, and suppressing appetite | Increased DPPIV expression resulting in higher risk of diabetes |
| Sex hormones | Hormone metabolism and SHBG production | Ubiquitous | Numerous (details beyond this review) | Hypogonadism |
| Glucocorticoids | Hormone metabolism and CBG production | Ubiquitous | Numerous (details beyond this review) | Relative adrenal insufficiency |
| Mineralocorticoids | Hormone metabolism | Cardiovascular system | Maintaining electrolyte balance and blood pressure | Largely intact |
The liver directly synthesizes multiple hormones, including 25-hydroxyvitamin D, insulin-like growth factor 1 (IGF-1), and angiotensinogen. Given roles in direct hormone production, the liver also has permissive roles of normal hormone function, in particular with effects on bone health, the GH-IGF-1 axis, and renin-angiotensin-aldosterone (RAA) pathway.
Vitamin D: The liver is the primary site of 25-hydroxylation of vitamin D to 25-hydroxyvitamin D (calcidiol), the main storage form of vitamin D[10]. Vitamin D is a secosteroid hormone well known for its role in calcium and bone homeostasis, with pleiotropic effects on cellular proliferation, differentiation, and immunomodulation[11-13]. 25-hydroxyvitamin D (calcidiol) then undergoes 1-alpha-hydroxylation in the kidney to the activated form 1,25-dihydroxyvitamin D (calcitriol)[10], which provides the active hormonal effects of vitamin D. The hydroxylation of vitamin D to produce calcidiol is mainly carried out in the liver by multiple cytochrome P450 mixed-function oxidases (CYPs) located in the mitochondria, endoplasmic reticulum (ER), and microsomes, though studies also show presence of these CYPs in extrahepatic tissues[10,11].
IGF-1: The liver is the primary source of IGF-1, a 70-amino acid polypeptide hormone with endocrine, paracrine, and autocrine effects[14]. IGF-1 affects almost every tissue and organ[15], and its receptors are ubiquitously expressed[16]. Besides mediating the actions of GH, more recently, non-growth-related actions of IGF-1 are found. IGF-1 binds to the insulin receptor and the hybrid IGF-1/insulin receptors, with implications on the metabolic effects of IGF-1[14]. IGF-1, GH, and insulin are hypothesized to constitute a regulated axis to inform cells about nutritional status, helping direct cells grow and differentiate vs induce a state of quiescence, senescence or apoptosis[14]. The IGF-1 receptor also participates in a crosstalk with the thyrotropin receptor by forming heterodimers[17], with implications on cellular growth and pathological implications in Graves’ eye disease.
Angiotensinogen: The liver is the primary source of angiotensinogen, which is involved in the RAA system[18]. The RAA system is vital for maintaining blood pressure homeostasis, via effects on sodium balance, intra- and extra-vascular volume, and systemic vascular tone[19]. Angiotensinogen, an alpha-globulin, is the only known substrate for renin and the main precursor molecule for angiotensin II (AngII), the major biologically active peptide in the RAA pathway[19]. Despite local tissue production of AngII, liver angiotensinogen is the primary source of renal AngII[18]. Hepatocytes tonically secrete angiotensinogen and primarily determine plasma angiotensinogen levels, with small increases in angiotensinogen levels increasing blood pressure and AngII levels[20].
The liver is involved in the metabolism of multiple endocrine hormones, including thyroid hormones, glucagon-like peptide-1, and steroid hormones, with roles in both activation and inactivation of the hormones.
Thyroid hormone: Hepatic metabolism has roles in both activation and inactivation of thyroid hormones. The biologic activity of thyroid hormone is mainly mediated through the active thyroid hormone T3. The thyroid only secretes 20% of the daily T3 requirement, with the remainder 80% converted from T4 by peripheral selenium-containing deiodinase enzymes (DIO), of which three primary deiodinases (type 1, 2, and 3) have been identified[21]. The liver expresses DIO1, along with the kidney and thyroid, which converts T4 to T3, though with less kinetic efficiency compared to DIO2, which is expressed by brown adipose tissue and the pituitary. Subsequently, the thyroid hormone is metabolized by conjugation with sulfate or glucuronic acid, which occurs prominently in the liver[22].
Glucagon-like peptide 1: With the discovery of glucagon-like peptide 1 (GLP-1), increasing research has been studying the gut-pancreas-liver axis, and the liver has been shown to play a key role in the hormone’s metabolism[23]. GLP-1 is an incretin hormone produced by the intestinal L-cells in response to ingestion of nutrients, including carbohydrates, fatty acids, and fiber[24]. It stimulates insulin secretion in a glucose-dependent manner, with associated inhibition of hepatic gluconeogenesis, and promotes insulin gene transcription and growth and proliferation of islet cells[24]. GLP-1 is inactivated by dipeptidyl peptidase-4 (DPPIV), also known as CD26, a ubiquitous membrane-associated peptidase[25]. DPPIV has pleiotropic effects and widespread tissue distribution in all organs, with expression in capillary endothelial cells and high expression in the liver[25].
Steroid hormone metabolism: The liver participates in most steps of steroid hormone regulation, starting from being the primary site of cholesterol biosynthesis[26,27]. At the liver, steroid hormones undergo phase I metabolism by cytochrome P450 enzymes (CYPs), via multiple pathways including hydroxylation or reduction, and phase II metabolism, also via various processes including glucuronidation, sulfation, or methylation[27], ultimately leading to excretion of their conjugates in urine or bile.
Steroid hormone metabolism: Sex hormones: The liver is the main site for metabolic conversion of estrogens, progesterone, and androgens to their metabolites via CYPs, which are abundantly expressed in the liver[28]. In particular, as part of the first phase of metabolism, estrogens undergo hydroxylation by numerous CYPs, including 2-hydroxylation to 2-hydroxyestradiol and 4-hydroxylation to 4-hydroxestradiol, which represent 80% and 20% of biotransformation of estradiol in the liver, respectively. 2-hydroxylation is mainly catalyzed by CYP1A2 and CYP3A4, which are expressed in the liver, and CYP1A1 in extrahepatic tissues[28]. 4-hydroxestradiol, unlike 2-hydroxestradiol, is associated with free radical generation and cellular damage, with associated increased risk of carcinogenesis in the breast and endometrium. Subsequent phase II metabolism of sex hormones, via O-methylation by catechol O-methyltransferase (COMT), glucuronidation, or sulfation, occurs at high levels at the liver, with subsequent elimination in the urine or stool[28-30].
Steroid hormone metabolism: Glucocorticoids and mineralocorticoids: The liver is also the primary site of glucocorticoid and mineralocorticoid metabolism[27]. Cortisol is converted to and from its inactive metabolite cortisone by two isozymes of 11-beta hydroxysteroid dehydrogenase (11-beta-HSD)[31]. 11-beta-HSD type 1 (11-beta-HSD1) is widely distributed, though most abundantly located in the liver and adipose tissue, and is responsible for converting cortisone back to cortisol[31], with in vitro activity being greater in omental than subcutaneous adipose tissue[32]. In healthy individuals, local splanchnic cortisol production, including from the liver, can equal or even exceed that produced by extra-splanchnic tissues, including the adrenal gland[32]. In obese, non-diabetic individuals, the liver has been shown to account for virtually all splanchnic cortisol production[32]. Though primarily secreted from the adrenal glands under the regulation of the RAA axis, animal studies suggest possibility of local hepatic aldosterone production during liver injury, which may contribute to fibro
Lipophilic hormones, including steroid hormones, are not water soluble and need to be carried in the blood stream by binding proteins[2,34]. The liver is the primary source of binding proteins for many hormones. The liver produces specific binding proteins to multiple lipophilic hormones, including glucocorticoids, mineralocorticoids, sex steroids, thyroid hormones (T3 and T4), and vitamin D metabolites[2,34]. Binding globulins for these lipophilic hormones include cortisol binding globulin (CBG, which binds cortisol, aldosterone, and progesterone), sex hormone binding globulin (SHBG, which binds estradiol, testosterone, and other sex hormones), thyroxine binding globulin (TBG, which binds T3 and T4), and vitamin D binding globulin (DBG, which binds vitamin D metabolites)[2,34]. Binding proteins that are produced by the liver also include transthyretin (which binds thyroid hormone and retinol), IGF-1 binding proteins (IGFBP, which binds IGF, including IGF-1), and non-specific binding proteins including albumin and lipoproteins. Binding proteins serve as a circulating reservoir for hormones, potentially regulating tissue distribution and target destination in a manner that can be highly selective and targeted[2,35]. Binding protein expression and production, which occur primarily at the liver, is complex and under the regulation and influence of multiple factors[2]. Most binding protein expression increase in response to estrogens, including physiologically with pregnancy or with oral contraceptives[2,34]. Hepatic failure and protein-losing nephropathies lead to decrease of binding proteins in general[2,34].
The liver mediates the effects of numerous hormonal pathways, whether directly or indirectly; thus, not surprisingly, derangements affecting the liver lead to disruptions of various hormonal pathways. Patients with cirrhosis are characterized by various endocrinopathies, including relative increase in estrogen compared to androgens, hypogonadism, osteoporosis, IGF-1 deficiency, vitamin D deficiency, alterations in glucose and lipid homeostasis, and perhaps more controversially a relative adrenal insufficiency.
Sex hormones: Cirrhosis is characterized by symptoms of estrogen-androgen imbalance, with relatively higher estradiol and lower testosterone concentrations[36]. The etiology of estrogen-testosterone imbalance is at least in part due to conversion of androgens to estrogens in cirrhosis, which in large part occurs peripherally[36]. The pathophysiology of hypogonadism is complex, including potential contribution from hypothalamic-pituitary suppression from a relatively increased estrogen circulation. SHBG is elevated in compensated cirrhotic patients, with subsequent decreases with decompensated cirrhosis, leading to concern for potential underestimation of hypogonadism in cirrhosis[34].
Cortisol: Patients with cirrhosis have relatively lower cortisol levels, also in the setting of lower production of cortisol binding globulin[37]. Some studies suggest the presence of a relative adrenal insufficiency in cirrhosis, also termed critical illness-associated corticosteroid insufficiency[38]. These studies suggest a potential hepatoadrenal syndrome in advanced liver disease, with associated inadequate cortisol production during stress response[38]. The decrease in cortisol binding globulin makes the diagnosis more difficult, though some studies suggest that free cortisol levels are decreased in relative adrenal insufficiency[37]. Hepatoadrenal syndrome and associated low free cortisol are attributed to decreased formation of HDL precursors and formation of proinflammatory cytokines and endotoxins[38].
RAA system: In liver disease, the systemic RAA pathway is upregulated due to systemic and splanchnic arterial vasodilation and associated hypoperfusion of the renal system[39]. Notably, the cirrhotic liver is able to produce angiotensinogen to near-normal plasma levels until the end stages[40].
DPPIV and GLP-1: DPPIV may play a role in linking type 2 diabetes with chronic liver disease. Type 2 diabetes has been associated with a greater than 2-fold increased risk of liver disease[41], and in vitro studies have suggested that elevated glucose can induce DPPIV expression in liver cells[42]. The increased DPPIV activity, which degrades the incretin hormone GLP-1, may contribute towards development of IGT, insulin resistance, lipogenesis, and hepatic injury in liver disease[25,43]. Serum DPPIV levels are notably increased in cirrhosis[25], and increased DPPIV expression in the liver has been observed in hepatitis C, NAFLD, experimental liver regeneration, and cirrhosis[25,43]. Cirrhotic nodules show diffuse and uniform staining of DPPIV, with loss of usual zonal expression of DPPIV[43], and degree of hepatic expression of DPPIV has also been shown to correlate with NAFLD grading[25]. Increased DPPIV expression has also been seen in various malignant tumors, including hepatocellular carcinoma, with DPPIV noted to promote resistance to anticancer agents[25].
Thyroid hormone: Given the liver’s role in thyroid hormone metabolism, including local conversion of T4 to T3 by DIO1[21], patients with cirrhosis may present with abnormalities in thyroid hormone levels[44]. Though a variety of patterns are seen, the most common pattern is a low total T3 (TT3), low free T3 (FT3), elevated reverse T3 (rT3), low total T4 (TT4), variable literature on elevated vs low free T4 (FT4) levels, and possible elevations in TSH[44,45]. The low total hormone levels are attributable to low TBG[44]. The pattern is consistent with low T3 syndrome, which occurs in systemic illnesses, and represents non-thyroidal illness syndrome, previously known as euthyroid sick syndrome[44].
IGF-1: Systemic IGF-1 deficiency in cirrhosis has been associated with an altered metabolic profile, including diabetes, deregulated lipid profile, and cardiovascular disease[14]. Lack of liver-derived IGF-1, in particular, has been associated with resultant insulin insensitivity in the liver, skeletal muscle, and adipose tissue, and corresponding hyperinsulinemia[46]. In NAFLD, the severity of steatosis has been correlated with a decrease in IGF-1 levels, with statistically significant differences in IGF-1 levels between mild-moderate vs severe steatosis[14,47].
Bone health and vitamin D: Chronic liver disease, including cirrhosis regardless of etiology, is associated with osteomalacia, osteopenia, and osteoporosis, and up to 40% of patients with chronic liver disease may develop an osteoporotic fracture[48]. The etiology of hepatic osteodystrophy is not well understood, though potential contributing factors include hypogonadism, and decreased hepatic production of IGF-1 and fibronectin[48]. There is a shift in cytokine production with changes in the receptor activator of nuclear factor kappa-B ligand (RANKL)/osteoprotegerin (OPG) system and an up-regulation of IL-6, which stimulates osteoclasts[48]. Decreased vitamin D synthesis, which is more marked in severely compromised liver function or in cholestatic liver disease, can further contribute to increased osteoporotic risk[49]. History of steroid treatment in chronic liver disease may be a risk factor for osteoporosis as well[48,49]. Different etiologies of liver disease may differ in their pathogenesis of osteoporosis, and in particular, diseases such as hemochromatosis and Wilson’s may also directly impact bone health[48].
Besides the advances in the understanding of classic endocrine functions of the liver, novel liver endocrine functions have been unraveled in the last several years (Table 2), including endocrine regulation of pancreatic α cells, adipose tissue, and insulin sensitivity.
| Liver hormone | Target organ | Action on target organ | Alteration in liver diseases |
| Amino acids | Pancreatic α cells | Stimulate cell proliferation and glucagon secretion | Not studied yet |
| Betatrophin | Pancreatic β cells (?) | Stimulate cell proliferation (?) | Increased in cirrhosis |
| Fetuin | Skeletal muscle; Adipose tissue | Decrease insulin sensitivity; Reduce adiponectin expression | Elevated in nonalcoholic fatty liver disease |
| FGF21 | Adipose tissue; Brain | Increase insulin sensitivity; Reduce food intake | Elevated in nonalcoholic fatty liver disease |
| Activin E | Adipose tissue | Increase fat oxidation | Increased in nonalcoholic fatty liver disease |
| Tsukushi | Adipose tissue | Increase thermogenesis | Increased in nonalcoholic fatty liver disease |
| GPNMB | Adipose tissue | Increase lipogenesis | Increased in nonalcoholic fatty liver disease |
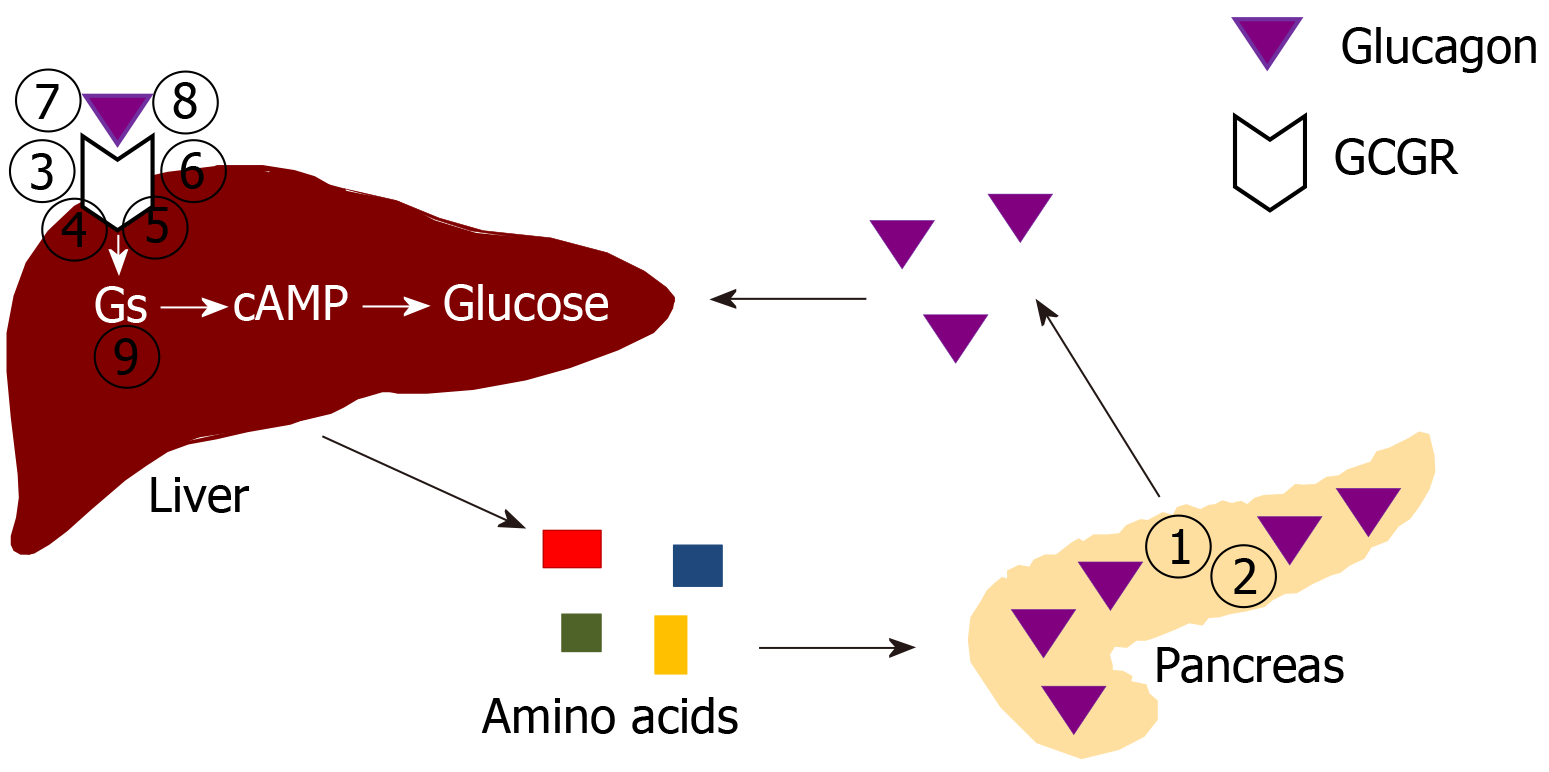
A major novel endocrine function of the liver is its critical role in a pancreatic α cell-liver axis that regulates pancreatic α cell proliferation and circulating glucagon and amino acid levels[50,51]. The pancreatic α cells, unlike the insulin-secreting β cells, have been considered a mysterious cell type until recently[52,53]. The α cells appear first during embryogenesis[54]. The main known function of the α cells is to produce and secrete the hormone glucagon[55]. Glucagon raises circulating glucose levels directly by stimulating gluconeogenesis and glycogenolysis, and indirectly by inhibiting insulin secretion[55,56].
Recently, a new α cell-liver axis has been discovered, endowing the liver with new endocrine functions[50,51]. The first clue of the α cell-liver axis came from glucagon receptor (GCGR) knockout mice[57,58]. The GCGR knockout mice harbor diffusely enlarged pancreas and exhibit extremely high glucagon levels[57-59]. Histologically, the pancreas of GCGR knockout mice contain numerous islets at various sizes, which are composed of mostly α cells as demonstrated by immunochemistry[57-59]. Normally the number of islets is quite small, and the islets are mostly composed of β cells. Mahvash disease, a human autosomal recessive hereditary disease discovered by our group, is caused by biallelic inactivating GCGR mutations, and its universal features are also α cell hyperplasia and hyperglucagonemia[60-62]. GCGR inactivation in zebra fish and non-human primates also result in α cell hyperplasia and hyperglucagonemia[63-66]. Thus, preservation of glucagon function is conserved throughout evolution.
Although a physiological compensation of hyperglucagonemia in animals and humans with inactive GCGR is quite intuitive, the specific mechanism of the compensation was initially not clear[67]. The liver-specific GCGR knockout mice interestingly have similar α cell hyperplasia and hyperglucagonemia, as those in global GCGR knockout mice[57,58,68], suggesting that the liver is the only target organ of glucagon that sends feedback signals to α cells, and that loss of the usual negative feedback mechanism stimulates α cell hyperplasia and glucagon secretion. This theory is also supported by the liver-specific stimulatory G protein α subunit (Gsα) knockout mice, which also exhibit α cell hyperplasia and hyperglucagonemia[69]. As glucagon antagonists were a promising anti-diabetes medication, both academia and pharmacological companies became interested in the α cell-liver axis due to potential applications in diabetes drug development[70,71]. Some of the key original large-scale experiments leading to the discovery of the role of amino acids in regulating α cells were performed by pharmaceutical companies[72-74].
The liver may regulate α cells via neural or humoral mechanisms[67,68]. Islet transplantation experiments demonstrate that the liver uses a humoral mechanism[68]. Wild-type islets transplanted into the kidney of GCGR knockout mice undergo α cell hyperplasia, while GCGR knockout islets transplanted into wild-type kidney undergo reduced α cell proliferation. Thus, it is assumed that the liver sends a humoral factor (hormone) to stimulate pancreatic α cells, a phenomenon that is pronounced in diseases where the usual negative feedback mechanism is affected.
Initially, it was hoped that a single liver hormone would be isolated from differential liver gene expression patterns of wild-type and GCGR knockout mice[67]. Several groups, including ours, performed liver mRNA arrays of GCGR knockout mice and in wild-type mice treated with inhibitory GCGR antibodies, using wild-type mice as control[67,68,72]. Not surprisingly, many genes are overexpressed (potential stimulatory hormones) or underexpressed (potential inhibitory hormones) in the GCGR knockout liver[67,68,72]. Genes involved in gluconeogenesis are downregulated in the GCGR knockout liver[67,68,72]. On the other hand, genes involved in amino acid synthesis (e.g., asparagine synthetase, Asns) are upregulated, and genes involved in amino acid catabolism (e.g., glutaminase 2, Gls2) are downregulated[67,68,72]. Genes regulating lipid metabolism are also differentially expressed[67,68,72]. Most of the genes with significant differential expression were not bona fide hormone candidates because they were not secreted proteins[67,68,72]. InhbA and DefB1 were the only 2 overexpressed secreted proteins by both the GCGR knockout liver and wild-type liver treated with inhibitory GCGR antibodies; however, these two proteins were are also upregulated by glucagon in primary hepatocytes and thus unlikely the pursued liver hormone[67,68,75].
Another possibility was that the liver hormone may not be a direct gene product such as a protein or polypeptide; rather, the hormone may be a small molecule or metabolite[67]. Metabolomes of the GCGR knockout and wild-type mice were compared[72]. Many differences exist but most notable differences were in glucose, amino acid, nucleotide, and bile acid levels[72]. The GCGR knockout mice have lower glucose levels (70% of wild-type value) and higher levels of most amino acids (up to 15-fold for alanine, glutamine, glycine, lysine, and threonine) and 2 bile acids (cholic acid and glycocholic acid, both about 200-fold) [72]. In humans with Mahvash disease, glucose levels are generally normal, but the levels of amino acids, especially alanine and glutamine, are clearly elevated[62,76-78].
Pinpointing the identity of the novel liver hormone requires tremendous amount of work. Parabiosis of GCGR knockout and wild-type mice was considered, but no such models were published[67]. A more practical in vitro islet culture assay was adopted by most groups to screen for the liver hormone that stimulates α cell hyperplasia and hyperglucagonemia[73-75]. With the islet culture assay, it is shown that a < 10 kDa fraction of serum from GCGR knockout mice sufficiently stimulates α cell proliferation[75]. This fraction contains small proteins or peptides, lipids, amino acids, and metabolites[75]. We have discussed earlier that most proteins or peptides are unlikely the liver hormone. Eliminating lipids from the fraction does not change the activity of the fraction in stimulating α cell proliferation[75]. Finally, as amino acids levels are much higher in GCGR knockout serum, cocktails that mimic the amino acids levels in GCGR knockout mice serum have been tested for their ability to stimulate α cell proliferation, and indeed they do[73-75].
Individual amino acids were further tested to see if a particular amino acid is sufficient to stimulate α cell proliferation[73-75,79]. So far, the data on individual amino acids are still somewhat controversial. Most individual amino acid do not stimulate α cell proliferation or glucagon secretion[73-75,79]. Glutamine alone stimulated α cell proliferation in 2 studies, but it did not stimulate glucagon secretion in another, which is intriguing as α cell hyperplasia and hyperglucagonemia coexist in all models of GCGR inhibition[74,75,79]. Alanine alone stimulated α cell proliferation in one study, but not in another, albeit acutely stimulating glucagon release[75,79]. Experimental conditions may explain some of the different results. It is also possible that α cell proliferation and acute glucagon release may be separate processes.
The α cell receptor for amino acids is under active research. In GCCR knockout mice and in wild-type type mice treated with inhibitory GCGR antibodies, the most upregulated α cell gene is the amino acid transporter Slc38a5 (20-80-fold increase)[74,75]. Slc38a5 preferentially transports glutamine and several other amino acids, which is concordant with the stimulatory effect of glutamine on α cell proliferation[74,75]. Slc38a5 knockout mice treated with inhibitory glucagon antibodies and Slc38a5 and GCGR double knockout mice exhibited less prominent α cell hyperplasia ( approximately 50% less) but similar hyperglucagonemia[74]; this data suggested that Slc38a5 is at least partially responsible for amino acid-stimulated α cell hyperplasia and that α cell hyperplasia and hyperglucagonemia may be regulated separately. Slc38a5, however, is not expressed in human α cells[74]. Another amino acid transporter Slc38a4 is enriched in human α cells when mice with human islet implants are treated with inhibitory GCGR antibodies[80]. In humans with Mahvash disease, Slc38a4 is expressed in the α cells[80], supporting a role of the amino acid transporter in mediating amino acid-stimulated α cell hyperplasia in humans as well. The mTOR pathway in α cells is activated by amino acids as well, contributing to α cell hyperplasia[73-75].
As a result of these studies, the α cell-liver axis has largely been clarified (Figure 1). The α cells secrete glucagon, which signals the liver to increase hepatic amino acid breakdown and reduce amino acid synthesis, consequently leading to desirable amino acid levels in the circulation. After glucagon signaling is inhibited, the liver decreases amino acid breakdown and increases amino acid synthesis, thus raising circulating amino acid levels. The amino acid levels, in turn, act on the α cell amino acid transporters to stimulate α cell proliferation. The evolutionarily conserved α cell-liver axis suggests that glucagon’s primary role may be regulating amino acid levels.
Betatrophin (also known as angiopoietin-like protein 8, ANGPTL8) is a 22-kD protein produced and secreted by the liver and adipose tissue[81,82]. Several years ago, betatrophin was touted as the long sought-after liver hormone that stimulates pancreatic β cell proliferation and insulin production in conditions with insulin resistance[83,84]. An insulin resistance mouse model based on insulin receptor antagonist (S961) infusion exhibits remarkable hyperinsulinemia and beta cell hyperproliferation[83]. As S961 does not directly stimulate β cell proliferation, it was hypothesized that a humoral factor mediates the stimulation of β cell proliferation in this mouse model[83]. Screening of liver genes that were differentially expressed as a result of S961 infusion suggested that betatrophin, a secreted protein that is upregulated by S961 infusion, could be the humoral factor[83]. Betatrophin expression correlated well with β cell proliferation rates. The original report found that liver overexpression of betatrophin stimulated β cell proliferation[83].
The potential of betatrophin as the Holy Grail for diabetes treatment attracted much attention, but later experiments strongly argue against this function of betatrophin[85-87]. Betatrophin knockout mice exhibited normal glucose metabolism and similar hyperinsulinemia and β cell hyperproliferation in response to S961 infusion[85,86]. Detailed analysis of pancreas morphometry by several laboratories definitively showed that betatrophin overexpression does not stimulate β cell proliferation[88]. The only exception was that direct delivery of betatrophin to pancreas does stimulate β cell proliferation in rats[89]. In some mouse models of diabetes, betatrophin lowered glucose levels without effects on β cell proliferation[90]. Overall, betatrophin, despite the name, does not appear to stimulate β cell proliferation.
Betatrophin, however, could be a circulating marker of insulin resistance[82]. Early studies of betatrophin levels in various forms of human insulin resistance were quite conflictory, partly due to the differences in measurement methods[82]. Later studies using more standardized methods for measuring betatrophin were summarized by several meta-analyses on the correlation of circulating betatrophin levels and type 2 diabetes, gestational diabetes, polycystic ovary syndrome (PCOS), and obesity — all conditions with insulin resistance[91-95]. Xu et al[91] analyzed 25 such studies and showed a positive and significant correlation between circulating betatrophin levels and insulin resistance. Yue et al[92] analyzed 11 studies on betatrophin in type 2 diabetes and found that betatrophin is significantly elevated in type 2 diabetes. Kong et al[93] analyzed 8 studies on betatrophin in gestational diabetes and concluded that betatrophin is significantly elevated in gestational diabetes. Varikasuvu et al[94] analyzed 11 studies on betatrophin in PCOS and concluded that betatrophin is significantly elevated in PCOS. Similarly, Ye et al[95] analyzed 6 studies on betatrophin in obesity and concluded that betatrophin is significantly elevated in obesity. Thus, overall, circulating betatrophin is likely a marker of insulin resistance in humans. The high betatrophin liver expression in mice treated with S961, in retrospect, could simply be a sign of insulin resistance caused by S961[83]. It is, however, not clear how insulin resistance upregulates betatrophin. In humans, hyperinsulinemia, often associated with insulin resistance, and metformin, an insulin sensitizer, both decrease betatrophin levels, suggesting that insulin resistance per se upregulates betatrophin levels[96]. Betatrophin overexpression could further worsen hepatocyte sensitivity to insulin, the significance of which needs to be further explored[97].
Betatrophin also has a role in lipids regulation[98]. Betatrophin knockout mice exhibit much reduced triglyceride levels due to reduction in liver VLDL secretion[86]; betatrophin also forms a complex with ANGPTL3, which inhibits lipoprotein lipase (LPL) activity[86]. The increased production of VLDL and decreased LPL activity both contribute to hypertriglyceridemia. Betatrophin overexpression doubles triglyceride levels in mice[86]. In humans, circulating betatrophin levels are positively correlated with triglyceride levels in the general population[99]. In people with dyslipidemia, however, betatrophin levels were lower than in controls[100]. Betatrophin may potentially be a target in dyslipidemia treatment[101].
Hepatokines are metabolism-regulating proteins produced and secreted by the liver[102,103]. Several hepatokines have been reported and studied. Five of the most studied hepatokines are discussed in this review: Fetuin-A, fibroblast growth factor 21 (FGF21), activin E, Tsukushi, and glycoprotein nonmetastatic melanoma protein B (GPNMB).
Fetuin-A: Fetuin-A, also known as α2-Heremans-Schmid glycoprotein in humans, is one of the first discovered hepatokines[104]. A 52-kD glycoprotein, fetuin-A has diverse metabolic functions[104]. Under physiological conditions, fetuin-A mostly functions as a carrier protein and regulates osteogenesis and inhibits extra-skeletal calcification[105]. Fetuin-A’s role in regulating insulin sensitivity has also been studied in detail[106,107]. Fetuin-A knockout mice exhibit higher insulin sensitivity and have less tendency to develop obesity[106]. At the molecular level, fetuin-A inhibits insulin receptor phosphorylation in myocytes and adipocytes and adiponectin expression in adipocytes[107]. Fetuin-A levels are elevated in patients with insulin resistance or type 2 diabetes, likely mediated by high free fatty acid levels, and high fetuin-A levels are a risk factor for type 2 diabetes[108,109]. The thiazolidinedione-type diabetes medication pioglitazone directly inhibits hepatic production of fetuin-A, partly contributing to its action in improving insulin sensitivity[110].
FGF21: FGF21 is a hepatokine that was first discovered in 2000, but its metabolic regulation functions were not characterized until recently[111,112]. Although FGF21 is also expressed in adipose tissue and the pancreas, circulating FGF21 is predominantly derived from the liver[113]. Hepatic FGF21 expression is regulated by a number of physiological conditions and factors[114]. Prolonged starvation (> 7 d) and overnutrition both upregulate FGF21 expression[115,116]. Glucagon and the thyroid hormone triiodothyronine (T3) both stimulate FGF21 expression, while insulin may inhibit FGF21 expression in liver[117,118]. High-carbohydrate, high-fat diet, and low protein diets stimulate FGF21 expression as well[119,120]. The microRNAs miR-577 and miR-212 target FGF21 mRNA for degradation, thus suppressing FGF21 expression[121,122]. FGF21 is also upregulated by ER stress[123]. At the molecular level, at least some of the above actions are mediated by the nuclear hormone receptor peroxisome proliferation-activated receptor α (PPARα), which binds to regions of the FGF21 promoter and simulates FGF21 expression[124-126].
The human pre-FGF21 (precursor of mature FGF21) includes a 28-amino-acid signaling peptide and a 181-amino-acid FGF21 proper as the circulating form[127]. FGF21 signals through its transmembrane tyrosine kinase receptors, FGFR1c and FGFR3c, and its transmembrane co-receptor, Klotho-β (KLB)[128]. FGF21 downstream signaling is tissue-specific but generally leads to metabolic benefits such as increased insulin sensitivity and weight loss[129]. In the adipose tissue, FGF21 stimulates the Ras/Raf/MAPK pathway, with phosphorylation of ERK1 and ERK2, and the mTOR pathway, contributing to higher insulin sensitivity[130-132]. Other FGF21 metabolic benefits such as weight loss is mediated by non-adipose tissue such as the brain[133]. FGF21 has been a major interest of metabolic drug development. As the native FGF21 is not stable in the usual formulation, re-engineered FGF21 analogues and PEGylated FGF21 have been developed to be more stable[134]. Activating monoclonal antibodies targeting FGFR1–β-klotho have also been developed[135]. Preclinical and clinical studies have demonstrated clear metabolic benefits of the FGF21 analogs and activating antibodies, such as appetite suppression, weight loss, improved glycemia, and favorable lipid profile[134,135].
Activin E: Activin E belongs to the family of transforming growth factor-β (TGF-β) proteins[136]. Activin E is a secreted homodimer of inhibin-βE, which is mainly expressed in the liver[137]. Each mature inhibin-βE monomer has 113 amino acids[137]. In both mice and humans, inhibin-βE is upregulated by obesity and insulin resistance[138]. In mice, hepatic overexpression of inhibin-βE prevents excess weight gain and improves insulin sensitivity by promoting energy expenditure via increased fat oxidation[139,140]. Inhibin-βE ablation in mice gives conflictory results[138,139]. In one study using the transcriptional activator-like effector nucleases (TALENs) to remove liver specific inhibin-βE expression, inhibin-βE-deficient mice exhibited normal weight but had impaired thermogenesis during cold exposure[139]. In another study, however, use of small interfering RNA (siRNA) to silence Inhibin-βE expression in the liver reduced weight gain in obese mice[138]. Thus, the roles of Activin E in metabolic regulation are still controversial.
Tsukushi: Tsukushi belongs to the family of small leucine-rich proteoglycan (SLRP) extracellular matrix proteins[141]. The secreted human Tsukushi protein has 337 amino acids. Besides its role in regulating embryonic development, Tsukushi is found to be a hepatokine, potentially regulating adipose tissue, weight, and energy expenditure[142]. In both mice and humans, Tsukushi is upregulated by thyroid hormone[142,143]; in mice, Tsukushi is induced by obesity and cold exposure[142]. Tsukushi deficiency in mice protects them from diet-induced obesity by increasing adipose tissue thermogenesis and energy expenditure[142]. Using mice from a different genetic background, another group could not reproduce the metabolic benefits of Tsukushi deficiency[144]. Furthermore, studies have also failed to show deleterious metabolic effects from Tsukushi overexpression[144]. The roles of Tsukushi in metabolic regulation thus also remain controversial.
GPNMB: GPNMB is a transmembrane glycoprotein expressed in the liver and other organs[145]. The cleaved extracellular domain of GPNMB (a glycosylated 480-amino-acid protein) is a hepatokine targeting adipose tissue[146,147]. In 2 obese mouse models, GPNMB expression was upregulated in the liver and secreted GPNMB levels were higher as well. Secreted GPNMB stimulates lipogenesis in vitro and in vivo[147]. A neutralizing antibody targeting GPNMB reduces obesity and improves insulin sensitivity[147]. In both mice and humans, GPNMB levels are positively correlated with obesity and insulin resistance[147]. GPNMB is thus a promising therapeutic target for treatments of obesity and diabetes.
The liver has numerous endocrine functions such as direct hormone and hepatokine production, hormone metabolism, synthesis of binding proteins, and processing and redistribution of metabolic fuels. In the last 10 years, many new endocrine functions of the liver have been discovered (Figure 2). Several novel endocrine functions of the liver have been unraveled. The liver plays a key negative feedback regulatory role in the pancreatic α cell-liver axis which regulates pancreatic α cell mass, glucagon secretion, and circulating amino acid levels. Betatrophin and other hepatokines such as fetuin-A and FGF21 play important endocrine roles in modulating insulin sensitivity, lipid metabolism, and body fat weight. It is expected that more endocrine functions of the liver will be discovered in the near future. As endocrine function of the liver is a rapidly evolving field, controversial findings often exist; caution needs to be taken when interpreting novel findings to avoid over-simplification of complex metabolic processes and premature allocation of research resources.
Provenance and peer review: Invited article; Externally peer reviewed.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Famularo S, Ho CM S-Editor: Gao CC L-Editor: A P-Editor: Li JH
| 1. | Watt MJ, Miotto PM, De Nardo W, Montgomery MK. The Liver as an Endocrine Organ-Linking NAFLD and Insulin Resistance. Endocr Rev. 2019;40:1367-1393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 414] [Article Influence: 69.0] [Reference Citation Analysis (2)] |
| 2. | Bikle DD. The Free Hormone Hypothesis: When, Why, and How to Measure the Free Hormone Levels to Assess Vitamin D, Thyroid, Sex Hormone, and Cortisol Status. JBMR Plus. 2021;5:e10418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 3. | Grant DM. Detoxification pathways in the liver. J Inherit Metab Dis. 1991;14:421-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 125] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 4. | Rui L. Energy metabolism in the liver. Compr Physiol. 2014;4:177-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 1423] [Article Influence: 129.4] [Reference Citation Analysis (0)] |
| 5. | Malespin M, Nassri A. Endocrine Diseases and the Liver: An Update. Clin Liver Dis. 2019;23:233-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Svegliati-Baroni G, Patrício B, Lioci G, Macedo MP, Gastaldelli A. Gut-Pancreas-Liver Axis as a Target for Treatment of NAFLD/NASH. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 7. | Crispe IN. The liver as a lymphoid organ. Annu Rev Immunol. 2009;27:147-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 686] [Cited by in RCA: 743] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 8. | Corsini A, Bortolini M. Drug-induced liver injury: the role of drug metabolism and transport. J Clin Pharmacol. 2013;53:463-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 123] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 9. | Eipel C, Abshagen K, Vollmar B. Regulation of hepatic blood flow: the hepatic arterial buffer response revisited. World J Gastroenterol. 2010;16:6046-6057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 339] [Cited by in RCA: 368] [Article Influence: 24.5] [Reference Citation Analysis (2)] |
| 10. | Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21:319-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 825] [Cited by in RCA: 1154] [Article Influence: 104.9] [Reference Citation Analysis (1)] |
| 11. | Elangovan H, Chahal S, Gunton JE. Vitamin D in liver disease: Current evidence and potential directions. Biochim Biophys Acta Mol Basis Dis. 2017;1863:907-916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 12. | Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol Rev. 2016;96:365-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1206] [Cited by in RCA: 1206] [Article Influence: 134.0] [Reference Citation Analysis (0)] |
| 13. | Konstantakis C, Tselekouni P, Kalafateli M, Triantos C. Vitamin D deficiency in patients with liver cirrhosis. Ann Gastroenterol. 2016;29:297-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 14. | Kineman RD, Del Rio-Moreno M, Sarmento-Cabral A. 40 YEARS of IGF1: Understanding the tissue-specific roles of IGF1/IGF1R in regulating metabolism using the Cre/LoxP system. J Mol Endocrinol. 2018;61:T187-T198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 83] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 15. | Lara-Diaz VJ, Castilla-Cortazar I, Martín-Estal I, García-Magariño M, Aguirre GA, Puche JE, de la Garza RG, Morales LA, Muñoz U. IGF-1 modulates gene expression of proteins involved in inflammation, cytoskeleton, and liver architecture. J Physiol Biochem. 2017;73:245-258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Riedemann J, Macaulay VM. IGF1R signalling and its inhibition. Endocr Relat Cancer. 2006;13 Suppl 1:S33-S43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 211] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 17. | Krieger CC, Place RF, Bevilacqua C, Marcus-Samuels B, Abel BS, Skarulis MC, Kahaly GJ, Neumann S, Gershengorn MC. TSH/IGF-1 Receptor Cross Talk in Graves' Ophthalmopathy Pathogenesis. J Clin Endocrinol Metab. 2016;101:2340-2347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 18. | Matsusaka T, Niimura F, Shimizu A, Pastan I, Saito A, Kobori H, Nishiyama A, Ichikawa I. Liver angiotensinogen is the primary source of renal angiotensin II. J Am Soc Nephrol. 2012;23:1181-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 211] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 19. | Patel S, Rauf A, Khan H, Abu-Izneid T. Renin-angiotensin-aldosterone (RAAS): The ubiquitous system for homeostasis and pathologies. Biomed Pharmacother. 2017;94:317-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 389] [Article Influence: 48.6] [Reference Citation Analysis (0)] |
| 20. | Caulfield M, Lavender P, Newell-Price J, Kamdar S, Farrall M, Clark AJ. Angiotensinogen in human essential hypertension. Hypertension. 1996;28:1123-1125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Luongo C, Dentice M, Salvatore D. Deiodinases and their intricate role in thyroid hormone homeostasis. Nat Rev Endocrinol. 2019;15:479-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 173] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 22. | Findlay KA, Kaptein E, Visser TJ, Burchell B. Characterization of the uridine diphosphate-glucuronosyltransferase-catalyzing thyroid hormone glucuronidation in man. J Clin Endocrinol Metab. 2000;85:2879-2883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Jin T, Weng J. Hepatic functions of GLP-1 and its based drugs: current disputes and perspectives. Am J Physiol Endocrinol Metab. 2016;311:E620-E627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 24. | MacDonald PE, El-Kholy W, Riedel MJ, Salapatek AM, Light PE, Wheeler MB. The multiple actions of GLP-1 on the process of glucose-stimulated insulin secretion. Diabetes. 2002;51 Suppl 3:S434-S442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 410] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 25. | Itou M, Kawaguchi T, Taniguchi E, Sata M. Dipeptidyl peptidase-4: a key player in chronic liver disease. World J Gastroenterol. 2013;19:2298-2306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 129] [Cited by in RCA: 141] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 26. | Trapani L, Segatto M, Pallottini V. Regulation and deregulation of cholesterol homeostasis: The liver as a metabolic "power station". World J Hepatol. 2012;4:184-190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 127] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 27. | Schiffer L, Barnard L, Baranowski ES, Gilligan LC, Taylor AE, Arlt W, Shackleton CHL, Storbeck KH. Human steroid biosynthesis, metabolism and excretion are differentially reflected by serum and urine steroid metabolomes: A comprehensive review. J Steroid Biochem Mol Biol. 2019;194:105439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 253] [Article Influence: 42.2] [Reference Citation Analysis (0)] |
| 28. | Tsuchiya Y, Nakajima M, Yokoi T. Cytochrome P450-mediated metabolism of estrogens and its regulation in human. Cancer Lett. 2005;227:115-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 450] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 29. | Raftogianis R, Creveling C, Weinshilboum R, Weisz J. Estrogen metabolism by conjugation. J Natl Cancer Inst Monogr. 2000;113-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 204] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 30. | Wilson VS, LeBlanc GA. The contribution of hepatic inactivation of testosterone to the lowering of serum testosterone levels by ketoconazole. Toxicol Sci. 2000;54:128-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 31. | Quinkler M, Stewart PM. Hypertension and the cortisol-cortisone shuttle. J Clin Endocrinol Metab. 2003;88:2384-2392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 180] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 32. | Basu R, Basu A, Grudzien M, Jung P, Jacobson P, Johnson M, Singh R, Sarr M, Rizza RA. Liver is the site of splanchnic cortisol production in obese nondiabetic humans. Diabetes. 2009;58:39-45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 33. | Fujisawa G, Muto S, Okada K, Kusano E, Ishibashi S. Mineralocorticoid receptor antagonist spironolactone prevents pig serum-induced hepatic fibrosis in rats. Transl Res. 2006;148:149-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Hammond GL. Plasma steroid-binding proteins: primary gatekeepers of steroid hormone action. J Endocrinol. 2016;230:R13-R25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 209] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 35. | Hammond GL. Potential functions of plasma steroid-binding proteins. Trends Endocrinol Metab. 1995;6:298-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 118] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 36. | Sinclair M, Grossmann M, Gow PJ, Angus PW. Testosterone in men with advanced liver disease: abnormalities and implications. J Gastroenterol Hepatol. 2015;30:244-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 88] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 37. | Fede G, Spadaro L, Tomaselli T, Privitera G, Scicali R, Vasianopoulou P, Thalassinos E, Martin N, Thomas M, Purrello F, Burroughs AK. Comparison of total cortisol, free cortisol, and surrogate markers of free cortisol in diagnosis of adrenal insufficiency in patients with stable cirrhosis. Clin Gastroenterol Hepatol. 2014;12:504-12.e8; quiz e23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 38. | Rakici H. Adrenal Insufficiency in Cirrhosis Patients: Evaluation of 108 Case Series. Euroasian J Hepatogastroenterol. 2017;7:150-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 39. | Di Pascoli M, La Mura V. Renin-angiotensin-aldosterone system in cirrhosis: There's room to try! Dig Liver Dis. 2019;51:297-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 40. | Sansoè G, Aragno M, Wong F. Pathways of hepatic and renal damage through non-classical activation of the renin-angiotensin system in chronic liver disease. Liver Int. 2020;40:18-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 41. | Jarvis H, Craig D, Barker R, Spiers G, Stow D, Anstee QM, Hanratty B. Metabolic risk factors and incident advanced liver disease in non-alcoholic fatty liver disease (NAFLD): A systematic review and meta-analysis of population-based observational studies. PLoS Med. 2020;17:e1003100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 288] [Article Influence: 57.6] [Reference Citation Analysis (0)] |
| 42. | Miyazaki M, Kato M, Tanaka K, Tanaka M, Kohjima M, Nakamura K, Enjoji M, Nakamuta M, Kotoh K, Takayanagi R. Increased hepatic expression of dipeptidyl peptidase-4 in non-alcoholic fatty liver disease and its association with insulin resistance and glucose metabolism. Mol Med Rep. 2012;5:729-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 64] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 43. | Matsumoto Y, Bishop GA, McCaughan GW. Altered zonal expression of the CD26 antigen (dipeptidyl peptidase IV) in human cirrhotic liver. Hepatology. 1992;15:1048-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 44. | Moura Neto A, Zantut-Wittmann DE. Abnormalities of Thyroid Hormone Metabolism during Systemic Illness: The Low T3 Syndrome in Different Clinical Settings. Int J Endocrinol. 2016;2016:2157583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 45. | Green JR. Thyroid function in chronic liver disease. Z Gastroenterol. 1979;17:447-451. [PubMed] |
| 46. | Ohlsson C, Mohan S, Sjögren K, Tivesten A, Isgaard J, Isaksson O, Jansson JO, Svensson J. The role of liver-derived insulin-like growth factor-I. Endocr Rev. 2009;30:494-535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 345] [Cited by in RCA: 311] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 47. | Mallea-Gil MS, Ballarino MC, Spiraquis A, Iriarte M, Kura M, Gimenez S, Oneto A, Guitelman M, Machado R, Miguel CM. IGF-1 Levels in different stages of liver steatosis and its association with metabolic syndrome. Acta Gastroenterol Latinoam. 2012;42:20-26. [PubMed] |
| 48. | Handzlik-Orlik G, Holecki M, Wilczyński K, Duława J. Osteoporosis in liver disease: pathogenesis and management. Ther Adv Endocrinol Metab. 2016;7:128-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 49. | Nair S. Vitamin d deficiency and liver disease. Gastroenterol Hepatol (N Y). 2010;6:491-493. [PubMed] |
| 50. | Wewer Albrechtsen NJ, Pedersen J, Galsgaard KD, Winther-Sørensen M, Suppli MP, Janah L, Gromada J, Vilstrup H, Knop FK, Holst JJ. The Liver-α-Cell Axis and Type 2 Diabetes. Endocr Rev. 2019;40:1353-1366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 121] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 51. | Dean ED. A Primary Role for α-Cells as Amino Acid Sensors. Diabetes. 2020;69:542-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 52. | Gromada J, Franklin I, Wollheim CB. Alpha-cells of the endocrine pancreas: 35 years of research but the enigma remains. Endocr Rev. 2007;28:84-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 451] [Cited by in RCA: 435] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 53. | Habener JF, Stanojevic V. Alpha cells come of age. Trends Endocrinol Metab. 2013;24:153-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 54. | Habener JF, Stanojevic V. α-cell role in β-cell generation and regeneration. Islets. 2012;4:188-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 55. | Quesada I, Tudurí E, Ripoll C, Nadal A. Physiology of the pancreatic alpha-cell and glucagon secretion: role in glucose homeostasis and diabetes. J Endocrinol. 2008;199:5-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 267] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 56. | Habegger KM, Heppner KM, Geary N, Bartness TJ, DiMarchi R, Tschöp MH. The metabolic actions of glucagon revisited. Nat Rev Endocrinol. 2010;6:689-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 269] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 57. | Parker JC, Andrews KM, Allen MR, Stock JL, McNeish JD. Glycemic control in mice with targeted disruption of the glucagon receptor gene. Biochem Biophys Res Commun. 2002;290:839-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 121] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 58. | Gelling RW, Du XQ, Dichmann DS, Romer J, Huang H, Cui L, Obici S, Tang B, Holst JJ, Fledelius C, Johansen PB, Rossetti L, Jelicks LA, Serup P, Nishimura E, Charron MJ. Lower blood glucose, hyperglucagonemia, and pancreatic alpha cell hyperplasia in glucagon receptor knockout mice. Proc Natl Acad Sci U S A. 2003;100:1438-1443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 417] [Cited by in RCA: 462] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 59. | Yu R, Dhall D, Nissen NN, Zhou C, Ren SG. Pancreatic neuroendocrine tumors in glucagon receptor-deficient mice. PLoS One. 2011;6:e23397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 60. | Yu R, Nissen NN, Dhall D, Heaney AP. Nesidioblastosis and hyperplasia of alpha cells, microglucagonoma, and nonfunctioning islet cell tumor of the pancreas: review of the literature. Pancreas. 2008;36:428-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 61. | Zhou C, Dhall D, Nissen NN, Chen CR, Yu R. Homozygous P86S mutation of the human glucagon receptor is associated with hyperglucagonemia, alpha cell hyperplasia, and islet cell tumor. Pancreas. 2009;38:941-946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 113] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 62. | Yu R. Mahvash Disease: 10 Years After Discovery. Pancreas. 2018;47:511-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 63. | Yu R. Pancreatic α-cell hyperplasia: facts and myths. J Clin Endocrinol Metab. 2014;99:748-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 64. | Li M, Dean ED, Zhao L, Nicholson WE, Powers AC, Chen W. Glucagon receptor inactivation leads to α-cell hyperplasia in zebrafish. J Endocrinol. 2015;227:93-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 65. | Bai X, Jia J, Kang Q, Fu Y, Zhou Y, Zhong Y, Zhang C, Li M. Integrated Metabolomics and Lipidomics Analysis Reveal Remodeling of Lipid Metabolism and Amino Acid Metabolism in Glucagon Receptor-Deficient Zebrafish. Front Cell Dev Biol. 2020;8:605979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 66. | Yan H, Gu W, Yang J, Bi V, Shen Y, Lee E, Winters KA, Komorowski R, Zhang C, Patel JJ, Caughey D, Elliott GS, Lau YY, Wang J, Li YS, Boone T, Lindberg RA, Hu S, Véniant MM. Fully human monoclonal antibodies antagonizing the glucagon receptor improve glucose homeostasis in mice and monkeys. J Pharmacol Exp Ther. 2009;329:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 106] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 67. | Yu R, Zheng Y, Lucas MB, Tong YG. Elusive liver factor that causes pancreatic α cell hyperplasia: A review of literature. World J Gastrointest Pathophysiol. 2015;6:131-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 68. | Longuet C, Robledo AM, Dean ED, Dai C, Ali S, McGuinness I, de Chavez V, Vuguin PM, Charron MJ, Powers AC, Drucker DJ. Liver-specific disruption of the murine glucagon receptor produces α-cell hyperplasia: evidence for a circulating α-cell growth factor. Diabetes. 2013;62:1196-1205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 164] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 69. | Chen M, Gavrilova O, Zhao WQ, Nguyen A, Lorenzo J, Shen L, Nackers L, Pack S, Jou W, Weinstein LS. Increased glucose tolerance and reduced adiposity in the absence of fasting hypoglycemia in mice with liver-specific Gs alpha deficiency. J Clin Invest. 2005;115:3217-3227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 121] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 70. | Shen DM, Lin S, Parmee ER. A survey of small molecule glucagon receptor antagonists from recent patents (2006 - 2010). Expert Opin Ther Pat. 2011;21:1211-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 71. | Scheen AJ, Paquot N, Lefèbvre PJ. Investigational glucagon receptor antagonists in Phase I and II clinical trials for diabetes. Expert Opin Investig Drugs. 2017;26:1373-1389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 72. | Yang J, MacDougall ML, McDowell MT, Xi L, Wei R, Zavadoski WJ, Molloy MP, Baker JD, Kuhn M, Cabrera O, Treadway JL. Polyomic profiling reveals significant hepatic metabolic alterations in glucagon-receptor (GCGR) knockout mice: implications on anti-glucagon therapies for diabetes. BMC Genomics. 2011;12:281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 73. | Solloway MJ, Madjidi A, Gu C, Eastham-Anderson J, Clarke HJ, Kljavin N, Zavala-Solorio J, Kates L, Friedman B, Brauer M, Wang J, Fiehn O, Kolumam G, Stern H, Lowe JB, Peterson AS, Allan BB. Glucagon Couples Hepatic Amino Acid Catabolism to mTOR-Dependent Regulation of α-Cell Mass. Cell Rep. 2015;12:495-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 151] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 74. | Kim J, Okamoto H, Huang Z, Anguiano G, Chen S, Liu Q, Cavino K, Xin Y, Na E, Hamid R, Lee J, Zambrowicz B, Unger R, Murphy AJ, Xu Y, Yancopoulos GD, Li WH, Gromada J. Amino Acid Transporter Slc38a5 Controls Glucagon Receptor Inhibition-Induced Pancreatic α Cell Hyperplasia in Mice. Cell Metab. 2017;25:1348-1361.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 149] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 75. | Dean ED, Li M, Prasad N, Wisniewski SN, Von Deylen A, Spaeth J, Maddison L, Botros A, Sedgeman LR, Bozadjieva N, Ilkayeva O, Coldren A, Poffenberger G, Shostak A, Semich MC, Aamodt KI, Phillips N, Yan H, Bernal-Mizrachi E, Corbin JD, Vickers KC, Levy SE, Dai C, Newgard C, Gu W, Stein R, Chen W, Powers AC. Interrupted Glucagon Signaling Reveals Hepatic α Cell Axis and Role for L-Glutamine in α Cell Proliferation. Cell Metab. 2017;25:1362-1373.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 175] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 76. | Larger E, Wewer Albrechtsen NJ, Hansen LH, Gelling RW, Capeau J, Deacon CF, Madsen OD, Yakushiji F, De Meyts P, Holst JJ, Nishimura E. Pancreatic α-cell hyperplasia and hyperglucagonemia due to a glucagon receptor splice mutation. Endocrinol Diabetes Metab Case Rep. 2016;2016. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 77. | Li H, Zhao L, Singh R, Ham JN, Fadoju DO, Bean LJH, Zhang Y, Xu Y, Xu HE, Gambello MJ. The first pediatric case of glucagon receptor defect due to biallelic mutations in GCGR is identified by newborn screening of elevated arginine. Mol Genet Metab Rep. 2018;17:46-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 78. | Gild ML, Tsang V, Samra J, Clifton-Bligh RJ, Tacon L, Gill AJ. Hypercalcemia in Glucagon Cell Hyperplasia and Neoplasia (Mahvash Syndrome): A New Association. J Clin Endocrinol Metab. 2018;103:3119-3123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 79. | Galsgaard KD, Jepsen SL, Kjeldsen SAS, Pedersen J, Wewer Albrechtsen NJ, Holst JJ. Alanine, arginine, cysteine, and proline, but not glutamine, are substrates for, and acute mediators of, the liver-α-cell axis in female mice. Am J Physiol Endocrinol Metab. 2020;318:E920-E929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 80. | Kim J, Dominguez Gutierrez G, Xin Y, Cavino K, Sung B, Sipos B, Kloeppel G, Gromada J, Okamoto H. Increased SLC38A4 Amino Acid Transporter Expression in Human Pancreatic α-Cells After Glucagon Receptor Inhibition. Endocrinology. 2019;160:979-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 81. | Kaestner KH. Betatrophin--promises fading and lessons learned. Cell Metab. 2014;20:932-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 82. | Abu-Farha M, Abubaker J, Tuomilehto J. ANGPTL8 (betatrophin) role in diabetes and metabolic diseases. Diabetes Metab Res Rev. 2017;33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 83. | Yi P, Park JS, Melton DA. Betatrophin: a hormone that controls pancreatic β cell proliferation. Cell. 2013;153:747-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 364] [Cited by in RCA: 358] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 84. | Seymour PA, Serup P. Bulking up on beta cells. N Engl J Med. 2013;369:777-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 85. | Gusarova V, Alexa CA, Na E, Stevis PE, Xin Y, Bonner-Weir S, Cohen JC, Hobbs HH, Murphy AJ, Yancopoulos GD, Gromada J. ANGPTL8/betatrophin does not control pancreatic beta cell expansion. Cell. 2014;159:691-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 170] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 86. | Wang Y, Quagliarini F, Gusarova V, Gromada J, Valenzuela DM, Cohen JC, Hobbs HH. Mice lacking ANGPTL8 (Betatrophin) manifest disrupted triglyceride metabolism without impaired glucose homeostasis. Proc Natl Acad Sci U S A. 2013;110:16109-16114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 268] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 87. | Cox AR, Lam CJ, Bonnyman CW, Chavez J, Rios JS, Kushner JA. Angiopoietin-like protein 8 (ANGPTL8)/betatrophin overexpression does not increase beta cell proliferation in mice. Diabetologia. 2015;58:1523-1531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 88. | Cox AR, Barrandon O, Cai EP, Rios JS, Chavez J, Bonnyman CW, Lam CJ, Yi P, Melton DA, Kushner JA. Resolving Discrepant Findings on ANGPTL8 in β-Cell Proliferation: A Collaborative Approach to Resolving the Betatrophin Controversy. PLoS One. 2016;11:e0159276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 89. | Chen J, Chen S, Huang P, Meng XL, Clayton S, Shen JS, Grayburn PA. In vivo targeted delivery of ANGPTL8 gene for beta cell regeneration in rats. Diabetologia. 2015;58:1036-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 90. | Liu S, Smith EM, King TH, Glenn L, Trevino M, Park SH, Machida Y, Villaflor C, Grzesik W, Morris MA, Imai Y, Nadler JL. Host Factors Alter Effects of Angiopoietin-Like Protein 8 on Glucose Homeostasis in Diabetic Mice. J Diabetes Mellitus. 2016;6:277-290. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 91. | Xu J, Lin Y, Zhou H, Zhao L, Xiang G. The Correlation Between Circulating Betatrophin and Insulin Resistance in General Population: A Meta-Analysis. Horm Metab Res. 2017;49:760-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 92. | Yue S, Wu J, Zhang J, Liu L, Chen L. The Relationship between Betatrophin Levels in Blood and T2DM: A Systematic Review and Meta-Analysis. Dis Markers. 2016;2016:9391837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 93. | Kong FJ, Ma LL, Li G, Chen YX, Zhou JQ. Circulating Betatrophin Levels and Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. PLoS One. 2017;12:e0169941. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 94. | Varikasuvu SR, Panga JR, Satyanarayana MV. Circulating Angiopoietin-like 8 protein (ANGPTL8/Betatrophin) in patients with polycystic ovary syndrome: a systematic review and multi effect size meta-analysis. Gynecol Endocrinol. 2019;35:190-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 95. | Ye J, Qin Y, Wang D, Yang L, Yuan G. The Relationship between Circulating ANGPTL8/Betatrophin Concentrations and Adult Obesity: A Meta-Analysis. Dis Markers. 2019;2019:5096860. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 96. | Wang H, Du L, Wu T, Yang G, Hu W, Wang H, Yang M, Liu D, Gu HF, Zhu Z, Zheng H, Li L. Circulating betatrophin is associated with insulin resistance in humans: cross-sectional and interventional studies in vivo and in vitro. Oncotarget. 2017;8:96604-96614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 97. | Hao Q, Zheng A, Zhang H, Cao H. Down-regulation of betatrophin enhances insulin sensitivity in type 2 diabetes mellitus through activation of the GSK-3β/PGC-1α signaling pathway. J Endocrinol Invest. 2021;44:1857-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 98. | Zhang R. Lipasin, a novel nutritionally-regulated liver-enriched factor that regulates serum triglyceride levels. Biochem Biophys Res Commun. 2012;424:786-792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 282] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 99. | Gao T, Jin K, Chen P, Jin H, Yang L, Xie X, Yang M, Hu C, Yu X. Circulating Betatrophin Correlates with Triglycerides and Postprandial Glucose among Different Glucose Tolerance Statuses--A Case-Control Study. PLoS One. 2015;10:e0133640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 100. | Gómez-Ambrosi J, Pascual-Corrales E, Catalán V, Rodríguez A, Ramírez B, Romero S, Vila N, Ibáñez P, Margall MA, Silva C, Gil MJ, Salvador J, Frühbeck G. Altered Concentrations in Dyslipidemia Evidence a Role for ANGPTL8/Betatrophin in Lipid Metabolism in Humans. J Clin Endocrinol Metab. 2016;101:3803-3811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 101. | Fu Z, Abou-Samra AB, Zhang R. A lipasin/Angptl8 monoclonal antibody lowers mouse serum triglycerides involving increased postprandial activity of the cardiac lipoprotein lipase. Sci Rep. 2015;5:18502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 102. | Jung TW, Yoo HJ, Choi KM. Implication of hepatokines in metabolic disorders and cardiovascular diseases. BBA Clin. 2016;5:108-113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 103. | Jensen-Cody SO, Potthoff MJ. Hepatokines and metabolism: Deciphering communication from the liver. Mol Metab. 2021;44:101138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 143] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 104. | Bourebaba L, Marycz K. Pathophysiological Implication of Fetuin-A Glycoprotein in the Development of Metabolic Disorders: A Concise Review. J Clin Med. 2019;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 105. | Jahnen-Dechent W, Heiss A, Schäfer C, Ketteler M. Fetuin-A regulation of calcified matrix metabolism. Circ Res. 2011;108:1494-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 267] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 106. | Mathews ST, Rakhade S, Zhou X, Parker GC, Coscina DV, Grunberger G. Fetuin-null mice are protected against obesity and insulin resistance associated with aging. Biochem Biophys Res Commun. 2006;350:437-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 93] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 107. | Ren G, Kim T, Papizan JB, Okerberg CK, Kothari VM, Zaid H, Bilan PJ, Araya-Ramirez F, Littlefield LA, Bowers RL, Mahurin AJ, Nickles MM, Ludvigsen R, He X, Grandjean PW, Mathews ST. Phosphorylation status of fetuin-A is critical for inhibition of insulin action and is correlated with obesity and insulin resistance. Am J Physiol Endocrinol Metab. 2019;317:E250-E260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 108. | Ramírez-Vélez R, García-Hermoso A, Hackney AC, Izquierdo M. Effects of exercise training on Fetuin-a in obese, type 2 diabetes and cardiovascular disease in adults and elderly: a systematic review and Meta-analysis. Lipids Health Dis. 2019;18:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 109. | Pan X, Wen SW, Bestman PL, Kaminga AC, Acheampong K, Liu A. Fetuin-A in Metabolic syndrome: A systematic review and meta-analysis. PLoS One. 2020;15:e0229776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 110. | Ochi A, Mori K, Emoto M, Nakatani S, Morioka T, Motoyama K, Fukumoto S, Imanishi Y, Koyama H, Ishimura E, Inaba M. Direct inhibitory effects of pioglitazone on hepatic fetuin-A expression. PLoS One. 2014;9:e88704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 111. | Fisher FM, Maratos-Flier E. Understanding the Physiology of FGF21. Annu Rev Physiol. 2016;78:223-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 652] [Article Influence: 65.2] [Reference Citation Analysis (0)] |
| 112. | Tezze C, Romanello V, Sandri M. FGF21 as Modulator of Metabolism in Health and Disease. Front Physiol. 2019;10:419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 238] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 113. | Markan KR, Naber MC, Ameka MK, Anderegg MD, Mangelsdorf DJ, Kliewer SA, Mohammadi M, Potthoff MJ. Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding. Diabetes. 2014;63:4057-4063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 395] [Cited by in RCA: 480] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 114. | Erickson A, Moreau R. The regulation of FGF21 gene expression by metabolic factors and nutrients. Horm Mol Biol Clin Investig. 2016;30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 30] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 115. | Fazeli PK, Lun M, Kim SM, Bredella MA, Wright S, Zhang Y, Lee H, Catana C, Klibanski A, Patwari P, Steinhauser ML. FGF21 and the late adaptive response to starvation in humans. J Clin Invest. 2015;125:4601-4611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 170] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 116. | Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, Wong RL, Chow WS, Tso AW, Lam KS, Xu A. Serum FGF21 Levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes. 2008;57:1246-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 658] [Cited by in RCA: 719] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 117. | Habegger KM, Stemmer K, Cheng C, Müller TD, Heppner KM, Ottaway N, Holland J, Hembree JL, Smiley D, Gelfanov V, Krishna R, Arafat AM, Konkar A, Belli S, Kapps M, Woods SC, Hofmann SM, D'Alessio D, Pfluger PT, Perez-Tilve D, Seeley RJ, Konishi M, Itoh N, Kharitonenkov A, Spranger J, DiMarchi RD, Tschöp MH. Fibroblast growth factor 21 mediates specific glucagon actions. Diabetes. 2013;62:1453-1463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 192] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 118. | Adams AC, Astapova I, Fisher FM, Badman MK, Kurgansky KE, Flier JS, Hollenberg AN, Maratos-Flier E. Thyroid hormone regulates hepatic expression of fibroblast growth factor 21 in a PPARalpha-dependent manner. J Biol Chem. 2010;285:14078-14082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 113] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 119. | Uebanso T, Taketani Y, Fukaya M, Sato K, Takei Y, Sato T, Sawada N, Amo K, Harada N, Arai H, Yamamoto H, Takeda E. Hypocaloric high-protein diet improves fatty liver and hypertriglyceridemia in sucrose-fed obese rats via two pathways. Am J Physiol Endocrinol Metab. 2009;297:E76-E84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 120. | Martínez-Garza Ú, Torres-Oteros D, Yarritu-Gallego A, Marrero PF, Haro D, Relat J. Fibroblast Growth Factor 21 and the Adaptive Response to Nutritional Challenges. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 121. | Chen XY, Li GM, Dong Q, Peng H. MiR-577 inhibits pancreatic β-cell function and survival by targeting fibroblast growth factor 21 (FGF-21) in pediatric diabetes. Genet Mol Res. 2015;14:15462-15470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 122. | Xiao J, Bei Y, Liu J, Dimitrova-Shumkovska J, Kuang D, Zhou Q, Li J, Yang Y, Xiang Y, Wang F, Yang C, Yang W. miR-212 downregulation contributes to the protective effect of exercise against non-alcoholic fatty liver via targeting FGF-21. J Cell Mol Med. 2016;20:204-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 123. | Schaap FG, Kremer AE, Lamers WH, Jansen PL, Gaemers IC. Fibroblast growth factor 21 is induced by endoplasmic reticulum stress. Biochimie. 2013;95:692-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 133] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 124. | Gälman C, Lundåsen T, Kharitonenkov A, Bina HA, Eriksson M, Hafström I, Dahlin M, Amark P, Angelin B, Rudling M. The circulating metabolic regulator FGF21 is induced by prolonged fasting and PPARalpha activation in man. Cell Metab. 2008;8:169-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 389] [Cited by in RCA: 414] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 125. | Vernia S, Cavanagh-Kyros J, Garcia-Haro L, Sabio G, Barrett T, Jung DY, Kim JK, Xu J, Shulha HP, Garber M, Gao G, Davis RJ. The PPARα-FGF21 hormone axis contributes to metabolic regulation by the hepatic JNK signaling pathway. Cell Metab. 2014;20:512-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 154] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 126. | Videla LA, Fernández V, Vargas R, Cornejo P, Tapia G, Varela N, Valenzuela R, Arenas A, Fernández J, Hernández-Rodas MC, Riquelme B. Upregulation of rat liver PPARα-FGF21 signaling by a docosahexaenoic acid and thyroid hormone combined protocol. Biofactors. 2016;42:638-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 127. | Nishimura T, Nakatake Y, Konishi M, Itoh N. Identification of a novel FGF, FGF-21, preferentially expressed in the liver. Biochim Biophys Acta. 2000;1492:203-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 574] [Cited by in RCA: 667] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 128. | Kilkenny DM, Rocheleau JV. The FGF21 Receptor Signaling Complex: Klothoβ, FGFR1c, and Other Regulatory Interactions. Vitam Horm. 2016;101:17-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 129. | Staiger H, Keuper M, Berti L, Hrabe de Angelis M, Häring HU. Fibroblast Growth Factor 21-Metabolic Role in Mice and Men. Endocr Rev. 2017;38:468-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 205] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 130. | Li H, Wu G, Fang Q, Zhang M, Hui X, Sheng B, Wu L, Bao Y, Li P, Xu A, Jia W. Fibroblast growth factor 21 increases insulin sensitivity through specific expansion of subcutaneous fat. Nat Commun. 2018;9:272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 121] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 131. | Ge X, Chen C, Hui X, Wang Y, Lam KS, Xu A. Fibroblast growth factor 21 induces glucose transporter-1 expression through activation of the serum response factor/Ets-like protein-1 in adipocytes. J Biol Chem. 2011;286:34533-34541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 132] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 132. | Minard AY, Tan SX, Yang P, Fazakerley DJ, Domanova W, Parker BL, Humphrey SJ, Jothi R, Stöckli J, James DE. mTORC1 Is a Major Regulatory Node in the FGF21 Signaling Network in Adipocytes. Cell Rep. 2016;17:29-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 133. | Sarruf DA, Thaler JP, Morton GJ, German J, Fischer JD, Ogimoto K, Schwartz MW. Fibroblast growth factor 21 action in the brain increases energy expenditure and insulin sensitivity in obese rats. Diabetes. 2010;59:1817-1824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 230] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 134. | Geng L, Lam KSL, Xu A. The therapeutic potential of FGF21 in metabolic diseases: from bench to clinic. Nat Rev Endocrinol. 2020;16:654-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 387] [Article Influence: 77.4] [Reference Citation Analysis (0)] |
| 135. | Baruch A, Wong C, Chinn LW, Vaze A, Sonoda J, Gelzleichter T, Chen S, Lewin-Koh N, Morrow L, Dheerendra S, Boismenu R, Gutierrez J, Wakshull E, Wilson ME, Arora PS. Antibody-mediated activation of the FGFR1/Klothoβ complex corrects metabolic dysfunction and alters food preference in obese humans. Proc Natl Acad Sci U S A. 2020;117:28992-29000. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 136. | Fang J, Wang SQ, Smiley E, Bonadio J. Genes coding for mouse activin beta C and beta E are closely linked and exhibit a liver-specific expression pattern in adult tissues. Biochem Biophys Res Commun. 1997;231:655-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 62] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 137. | Namwanje M, Brown CW. Activins and Inhibins: Roles in Development, Physiology, and Disease. Cold Spring Harb Perspect Biol. 2016;8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 190] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 138. | Sugiyama M, Kikuchi A, Misu H, Igawa H, Ashihara M, Kushima Y, Honda K, Suzuki Y, Kawabe Y, Kaneko S, Takamura T. Inhibin βE (INHBE) is a possible insulin resistance-associated hepatokine identified by comprehensive gene expression analysis in human liver biopsy samples. PLoS One. 2018;13:e0194798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 139. | Hashimoto O, Funaba M, Sekiyama K, Doi S, Shindo D, Satoh R, Itoi H, Oiwa H, Morita M, Suzuki C, Sugiyama M, Yamakawa N, Takada H, Matsumura S, Inoue K, Oyadomari S, Sugino H, Kurisaki A. Activin E Controls Energy Homeostasis in Both Brown and White Adipose Tissues as a Hepatokine. Cell Rep. 2018;25:1193-1203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 140. | Sekiyama K, Ushiro Y, Kurisaki A, Funaba M, Hashimoto O. Activin E enhances insulin sensitivity and thermogenesis by activating brown/beige adipocytes. J Vet Med Sci. 2019;81:646-652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 141. | Dellett M, Hu W, Papadaki V, Ohnuma S. Small leucine rich proteoglycan family regulates multiple signalling pathways in neural development and maintenance. Dev Growth Differ. 2012;54:327-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 142. | Wang Q, Sharma VP, Shen H, Xiao Y, Zhu Q, Xiong X, Guo L, Jiang L, Ohta K, Li S, Shi H, Rui L, Lin JD. The hepatokine Tsukushi gates energy expenditure via brown fat sympathetic innervation. Nat Metab. 2019;1:251-260. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 143. | Liu D, Zhang P, Wei X, Deng Y, Liu W, Guo D, Liu J, Xu B, Huang C, Huang J, Lin J, Liu S, Xue Y, Zhang H. Elevated Serum Tsukushi Levels in Patients With Hyperthyroidism. Front Endocrinol (Lausanne). 2020;11:580097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 144. | Mouchiroud M, Camiré É, Aldow M, Caron A, Jubinville É, Turcotte L, Kaci I, Beaulieu MJ, Roy C, Labbé SM, Varin TV, Gélinas Y, Lamothe J, Trottier J, Mitchell PL, Guénard F, Festuccia WT, Joubert P, Rose CF, Karvellas CJ, Barbier O, Morissette MC, Marette A, Laplante M. The Hepatokine TSK does not affect brown fat thermogenic capacity, body weight gain, and glucose homeostasis. Mol Metab. 2019;30:184-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 145. | van der Lienden MJC, Gaspar P, Boot R, Aerts JMFG, van Eijk M. Glycoprotein Non-Metastatic Protein B: An Emerging Biomarker for Lysosomal Dysfunction in Macrophages. Int J Mol Sci. 2018;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 56] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 146. | Kuang H, Lin JD. GPNMB: expanding the code for liver-fat communication. Nat Metab. 2019;1:507-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 147. | Gong XM, Li YF, Luo J, Wang JQ, Wei J, Xiao T, Xie C, Hong J, Ning G, Shi XJ, Li BL, Qi W, Song BL. Gpnmb secreted from liver promotes lipogenesis in white adipose tissue and aggravates obesity and insulin resistance. Nat Metab. 2019;1:570-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |