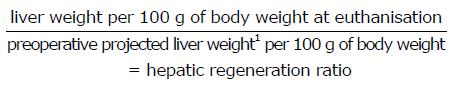Peer-review started: October 13, 2017
First decision: November 7, 2017
Revised: November 20, 2017
Accepted: December 6, 2017
Article in press: December 6, 2017
Published online: January 27, 2018
Processing time: 105 Days and 12.2 Hours
To evaluate the liver regeneration capacity (LRC) after partial hepatectomy (PH) in experimental non-alcoholic steatohepatitis (NASH).
Fifty-four female rats were fed a high-fat, high-cholesterol diet (HFCD, 65% fat, 1% cholesterol) or standard diet (STD) for 16 wk. A 70% PH was performed and the animals were euthanised before PH or 2 or 5 d post-PH. LRC was evaluated using: The total number of Ki-67 positive hepatocytes in the caudate lobe, N(Ki-67, lobe) evaluated in a stereology-based design, the regenerated protein ratio (RPR), prothrombin-proconvertin ratio (PP), and mRNA expression of genes related to regeneration.
The HFCD NASH model showed significant steatosis with ballooning and inflammation, while no fibrosis was present. Mortality was similar in HFCD and STD animals following PH. HFCD groups were compared to respective STD groups and HFCD animals had a significantly elevated alanine transaminase at baseline (P < 0.001), as well as a significantly elevated bilirubin at day 2 after PH (P < 0.05). HFCD animals had a higher N(Ki-67, lobe) at baseline, (P < 0.0001), day 2 after PH (P = 0.06) and day 5 after PH (P < 0.025). We found no significant difference in RPR or PP neither 2 or 5 d post-PH. Expression of liver regeneration genes (e.g., hepatic growth factor) was higher at both day 2 and 5 post-PH in HFCD groups (P < 0.05).
NASH rats had a preserved LRC after hepatectomy when compared to STD rats. The methods and models of NASH are essential in understanding and evaluating LRC.
Core tip: Liver regeneration capacity has been studied in different animal models of non-alcoholic steatohepatitis. This study is the first to use a high fat high cholesterol model which mimic the pathogenesis of human non-alcoholic steatohepatitis better than previous animal models. Liver regeneration capacity was evaluated using: (1) The total number of Ki-67 positive hepatocytes in the caudate lobe, evaluated in a stereology based design; (2) the regenerated protein content to describe the regenerated liver mass; and (3) the plasma concentration of coagulation factors as a marker of liver function. We found a preserved liver regeneration capacity in rats with non-alcoholic steatohepatitis, adding important knowledge to the subject.
- Citation: Haldrup D, Heebøll S, Thomsen KL, Andersen KJ, Meier M, Mortensen FV, Nyengaard JR, Hamilton-Dutoit S, Grønbæk H. Preserved liver regeneration capacity after partial hepatectomy in rats with non-alcoholic steatohepatitis. World J Hepatol 2018; 10(1): 8-21
- URL: https://www.wjgnet.com/1948-5182/full/v10/i1/8.htm
- DOI: https://dx.doi.org/10.4254/wjh.v10.i1.8
The incidence of obesity and non-alcoholic fatty liver disease (NAFLD) is increasing worldwide, affecting approximately one-third of the general population[1]. Patients with NAFLD and especially non-alcoholic steatohepatitis (NASH) have a higher risk of developing primary hepatocellular carcinoma (HCC)[2]. Further, they have an increased risk of other cancers, for example colorectal carcinoma[3], which often metastasize to the liver[4]. Surgical resection of the liver tumor remains the gold standard treatment for both HCC and liver metastases from colorectal cancer[5]. Epidemiological studies have shown that liver resection is associated with increased morbidity and mortality in patients with NAFLD following liver resections[6]. It has been proposed that NASH livers are more vulnerable to surgical interventions because of decreased liver regeneration capacity (LRC)[7].
Previously, LRC has been studied in various rodent models of NAFLD/NASH generally based on the use of the methionine-choline deficiency diet (MCD)[7-12], choline deficiency diet (CDD)[13-16], simple high-fat diets (HFD)[17,18] and the genetic leptin-deficiency model[19-25]. These are widely accepted models of NAFLD/NASH, yet the animals lack many of the clinical and/or histopathological features related to human NAFLD/NASH. These previous studies of LRC have reported conflicting results, even when the same dietary models were used. In the MCD[7-12], CDD[13-16], HFD[17,18] and a high-fat model combined with fructose[26], decreased[9-14,18,26] as well as normal liver regeneration[7,8,15-17] have been demonstrated. Our group has previously studied a high-fat, high-cholesterol diet (HFCD) rat model[27,28] with features that closely resemble human NASH[29]. To our knowledge, HFCD models have never been used to study the LRC experimentally.
We studied partially hepatectomized, HFCD-fed rats, hypothesizing that rats with HFCD-induced NASH would have decreased LRC, as well as lower expression of genes related to regeneration. LRC was evaluated using: (1) The total number of Ki-67 positive hepatocytes N(Ki-67, liver) evaluated in a stereology-based design; (2) the regenerated protein ratio (RPR); and (3) plasma concentration of coagulation factors II, VII, X, prothrombin-proconvertin ratio (PP) before, and 2 or 5 d after hepatectomy.
In total, 54 female Wistar rats (body weight 201-237 g; Taconic M and B, Ejby, Denmark) were housed at 21 °C ± 2 °C with a 12-h artificial light cycle. Three animals were housed in each cage with free access to tap water. All animals were allowed to acclimatize on a standard diet (STD) for a week followed by randomization and allocation. Then, half of the rats were fed STD and the other half HFCD ad libitum for 16 wk (Figure 1). Diets were obtained from Research Diets (NJ, United States). The STD (D14071501) consisted of the following energy sources: carbohydrates 67 g (70 kcal/100 kcal), fat 4 g (10 kcal/100 kcal), and protein 19 g (20 kcal/100 kcal) per 100 g diet. The HFCD (D14071502) consisted of carbohydrates 19 g (15 kcal/100 kcal), protein 27 g (20 kcal/100 kcal), 1 g cholesterol and fat 39 g (65 kcal/100 kcal) per 100 g diet, including 1% cholesterol and 0.25% cholate.
The study was performed in accordance with local and national guidelines for animal welfare and approved by the Animal Experiments Inspectorate (2014-15-2934-00997).
After 16 wk, both STD and HFCD rats were randomly divided into the following groups of nine rats (Figure 1): (1) No hepatectomy before sacrifice (NH); (2) partial hepatectomy (PH), sacrificed two days post-surgery (PH-2); (3) partial hepatectomy, sacrificed five days post-surgery (PH-5).
The non-hepatectomized NH rats served as a “baseline” reference. The PH-2 and PH-5 animals underwent a partial hepatectomy as previously described[30]. Briefly, the abdominal cavity was opened with a longitudinal incision in the linea alba. The left lateral and median lobes were mobilized and ligated followed by a resection, resulting in a 70% reduction of the liver tissue. The abdominal wall was closed with a continuous 4.0 absorbent suture and the skin was closed with staples. The resected liver tissue was weighed after removal. To ensure minimal post-operative pain, Carprofen 5 mg/kg (Rimadyl; Pfizer Animal Health, Exton, United States) was administered prior to the surgical procedure and two days after hepatectomy. Following hepatectomy, rats were fed their initial diet (STD or HFCD) until euthanisation.
At euthanisation the animals were anesthetized with a subcutaneous injection of fentanyl/fluanisone 0.5 mL/kg (Hypnorm; Jansen Pharma, Denmark) and midazolam 2.5 mg/kg (Dormicum; La Roche, Switzerland) and body weights were registered. Blood samples were collected through the retrobulbar venous plexus. Blood for analysis of prothrombin-proconvertin ratio (PP) were collected through the vena cava caudalis. The liver was removed en bloc and weighed. Liver tissue was collected from the right liver lobe and immediately snap-frozen in liquid nitrogen and stored at 80 °C until use. The caudate liver lobe was immersion fixed in phosphate-buffered 4% formaldehyde for a total of 48 h before paraffin embedding. Prior to embedding, and 24 h into the fixation, the lobe was cut with a special designed razor tool to create 2.15 mm thick slabs of liver tissue. The tissue slabs were then put back in the grid in correct order, all facing the same way for stereological examination. The caudate lobe was used for the histological evaluation and the stereological Ki-67 evaluation. Euthanasia was then achieved by cervical dislocation. Euthanisation was carried out between 8 am and 13 pm.
All samples were stained with hematoxylin and eosin (HE) and Masson-trichrome (MT), using standard protocols. The degree of steatosis and the presence of NASH were evaluated by an expert liver pathologist using both the Kleiner and Bedossa criteria[31,32] examining 5 medium-power fields (20 x objective). Steatosis was classified as either: large droplet macrovesicular steatosis (LDMS), small droplet macrovesicular steatosis (SDMS), mixed small and large macrovesicular steatosis (MXMS) or microvesicular steatosis (MVS).
Ki-67 positive hepatocytes: We quantified the total number of Ki-67 positive hepatocytes using a stereological-based design. The paraffin embedded caudate lobes were cut in 3 μm thick slides and immunohistochemically stained with the anti-Ki-67 antibody (clone MIB-5, isotype IgG1; Dako, Denmark) using a standard (in-house) protocol. All Ki-67 stained slides were scanned as virtual images using an Olympus VS 120 slide scanner with a 20x oil lens (numerical aperture 0.85).
The image files were transferred to the newCAST software version 5.2.1 (Visiopharm, Denmark) for quantification, performed as previous described[30]. Briefly, the examiner was blinded to all slides. The software was then set up to perform systematic uniform and random sampling (SURS) of fields of view, an unbiased sampling method. An average of 60 fields was used per slide. A 2D unbiased counting frame for counting cell profiles per area covering 50% of the field of view was used when few positive hepatocyte profiles were visible; when many profiles were visible, a counting frame of 10% was used. Positive Ki-67-stained hepatocyte profiles were defined as a large (approximately 8 μm in diameter) oval cells with an obviously stained border and visible nucleus (Supplementary Figures 1-3).
To calculate the number of Ki-67 positive hepatocyte cell profiles per area the following formula was used:
QA(Ki-67/lobe) = [ΣQ(Ki-67)]/[A(frame)•P(lobe)]
QA(Ki-67) is the number of Ki-67 positive hepatocyte cell profiles per mm2 lobe, A(frame) is the area of the counting frame, and P(lobe) is the number of test points-a maximum of two per counting frame. Counted if the lower left or upper right corner is hitting lobe tissue.
Total number of Ki-67 positive hepatocytes in the caudate lobe: To account for the larger cell size of the HFCD livers the total number of Ki-67 positive hepatocytes in the caudate lobe, N(Ki-67, lobe), was estimated.
First, the number of Ki-67 positive hepatocytes per volume liver lobe was calculated; SURS was set up, and approximately 30 positive hepatocyte cell profiles were sampled and the diameter measured at 20 x magnification. The diameter was defined as the length of diameter perpendicular to the longest axis of the cell (Supplementary Figure 4). This was used in the following formula[33]:
NV(Ki-67/lobe) = [QA(Ki-67/lobe)]/[D(cell) + t(section)]
Nv is the number of Ki-67 positive hepatocytes per mm3 lobe, QA(Ki-67/lobe) is the number of Ki-67 positive cell profiles per mm2 lobe, D(cell) is average diameter of the counted cells, t(section) is the thickness of the tissue sections (3 μm).
This is a model-based approach for number estimation biased by tissue shrinkage, projection effects, and deviations from model assumptions.
N(Ki-67, lobe) was then estimated; the volume of the caudate lobe was estimated based on the weight and the density of the rat liver. The density of the liver was set to 1.05 g/cm3[34] and used in the following formula:
N(Ki-67,lobe) = NV(Ki-67/lobe)•V(lobe)
N(Ki-67, lobe) is the total number of Ki-67 positive hepatocytes in the caudate lobe, NV(Ki-67/lobe) is the number of Ki-67 positive hepatocyte profiles per volume lobe, V(lobe) is total volume of the caudate lobe.
Hepatic regeneration ratio: The hepatic regeneration ratio (HRR) was calculated for each animal, and defined as:
Math 1
1Preoperative projected liver weight: Weight of resected liver at hepatectomy/0.7
Net regeneration: We calculated the net regeneration (NET), defined as:
Math 2
Total protein analysis: The Pierce™ BCA total protein assay kit (Thermo Fisher Scientific, IL, United States) was used to measure the amount of total protein in the liver tissue. Prior to the total protein measurement, the tissue was homogenized in a lysis buffer as previously described[35], only, mortar and pestle was used in this study. The total concentration of protein was multiplied by the weight of the whole liver at euthanisation to determine the absolute amount of protein in the whole liver.
Regenerated protein ratio: The regenerated protein ratio (RPR) was calculated as follows:
Math 3
We used a guanidinium thiocyanate-phenol-chloroform extraction protocol for RNA isolation as previously described[36]. The final RNA concentration was determined using a NanoDrop™ 2000 Spectrometer (Thermo Fisher Scientific). RNA concentrations were normalized to 1000 ng/μL and cDNA synthesized with High-Capacity RNA-to-cDNA™ Kit (Thermo Fisher Scientific) on a polymerase chain reaction (PCR) Express Thermal Cycler (Thermo Hybaid, DE, United States), according to manufacturer’s protocol.
Reverse transcriptase quantitative PCR (qPCR) was run on a 96-well StepOnePlusTM Real-Time PCR System Thermal Cycling Block (Thermo Fischer) using TaqMan Gene Expression Assays (Thermo Fisher Scientific). All assays contained the FAM dye. Samples were duplicated and we measured the mean cycle threshold (Ct) for each gene and standardized it to reference gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Data was analyzed using the ΔΔ-Ct method as described by Livak et al[37]. The STD NH-group was set as a reference group with a fold change of 1 and the relative expressional levels were compared with this group for each gene. Selected genes are displayed in Supplementary Table 1.
Routine analysis and PP measurements were performed at the Department of Clinical Biochemistry, Aarhus University Hospital, on the day of euthanisation. Plasma samples were analyzed for alanine transaminase (ALT), bilirubin and albumin on a Cobas 6000 (Roche Diagnostics, Basel, Switzerland), using a routine protocol. PP was analyzed on a CS 2100i instrument (Sysmex, Siemens Siemens Healthcare). The remaining plasma and serum samples were stored at 80 °C until analysis. Serum samples were evaluated for the rat acute phase protein alfa-2-macroglobulin (α2M), by ELISA (Immunology Consultants Laboratory, OR, United States).
Statistical analyses were performed using STATA 11.0, graphs were drawn in Excel 14.4.1. Normality of data was checked by qq-plots. For continuous variables, comparisons were made using the ANOVA test for significance. Post-hoc comparisons were performed by Student’s t-test. Categorical data were analyzed using Fisher’s exact test. The qPCR data exhibited skewed distributions with variance heterogeneity. Therefore, these data were analysed using the nonparametric Kruskal-Wallis one-way analysis of variance on ranks test; when significant, post-hoc tests were performed using the Mann-Whitney rank sum test. Variables are expressed as means (± SD). Significance level was set at P < 0.05.
One HFCD animal died during surgery and one STD animal died of internal bleeding due to insufficent ligature of the right median lobe at day one after hepatectomy. Thus, none of the animals died from liver insufficiency or failure. At baseline, HFCD liver weight increased 2-fold compared with STD liver. HFCD animals had a significantly higher liver weight at all times (P < 0.001, Table 1).
| Standard diet | High fat high cholesterol diet | |||||
| NH (n = 9) | PH-2 (n = 9) | PH-5 (n = 8) | NH (n = 9) | PH-2 (n = 9) | PH-5 (n = 8) | |
| Weight at euthanisation, g | 294 ± 22 | 289 ± 18 | 305 ± 12 | 289 ± 21 | 271 ± 181 | 290 ± 15 |
| Liver weight at euthanisation, g | 8.1 ± 0.6 | 6.1 ± 0.5 | 7.5 ± 1.3 | 15.4 ± 2.01 | 9.0 ± 0.81 | 12.3 ± 1.71 |
| Recected liver at PH, g | - | 5.2 ± 0.3 | 5.0 ± 0.5 | - | 11.2 ± 2.01 | 11.4 ± 0.71 |
| Hepatic regeneration ratio, % | 1.0 | 0.82 ± 0.08 | 1.04 ± 0.17 | 1.0 | 0.61 ± 0.11 | 0.77 ± 0.121 |
| Net regeneration (g) | - | 3.8 ± 0.5 | 4.2 ± 1.2 | - | 5.3 ± 0.9 | 7.4 ± 1.61 |
| Total protein, g | 0.83 ± 0.11 | 0.51 ± 0.08 | 0.69 ± 0.18 | 1.10 ± 0.13 | 0.60 ± 0.12 | 0.90 ± 0.14 |
| Ki-67 Index, positive profiles, mm2 | 1.6 ± 0.6 | 216 ± 83.2 | 27.2 ± 22.3 | 9.0 ± 3.21 | 227.5 ± 80 | 31.7 ± 11.1 |
| Spleen weigth at euthanisation, g | 0.64 ± 0.12 | 0.83 ± 0.12 | 1.10 ± 0.16 | 1.25 ± 0.361 | 1.04 ± 0.31 | 1.34 ± 0.44 |
| Alanine transaminase, u/L | 24.1 ± 5.2 | 64.9 ± 9.0 | 28.8 ± 13.4 | 79.9 ± 27.41 | 130.1 ± 25.41 | 45.8 ± 20.5 |
| ΔAlanine transaminase, u/L | - | 41 ± 9 | 5 ± 13 | - | 50 ± 25 | -34 ± 211 |
| Albumin (g/L) | 19.0 ± 1.9 | 14.7 ± 1.0 | 12.4 ± 1.5 | 15.1 ± 1.91 | 12.2 ± 0.8 | 11.5 ± 2.3 |
| α2-macroglobulin, μg/mL | 18.1 ± 0.7 | 102 ± 12.6 | 98.2 ± 13 | 22.6 ± 3.41 | 157.8 ± 401 | 94.5 ± 64.5 |
| Prothrombin-proconvertin ratio | 0.32 ± 0.03 | 0.33 ± 0.04 | 0.32 ± 0.03 | 0.34 ± 0.03 | 0.38 ± 0.06 | 0.33 ± 0.03 |
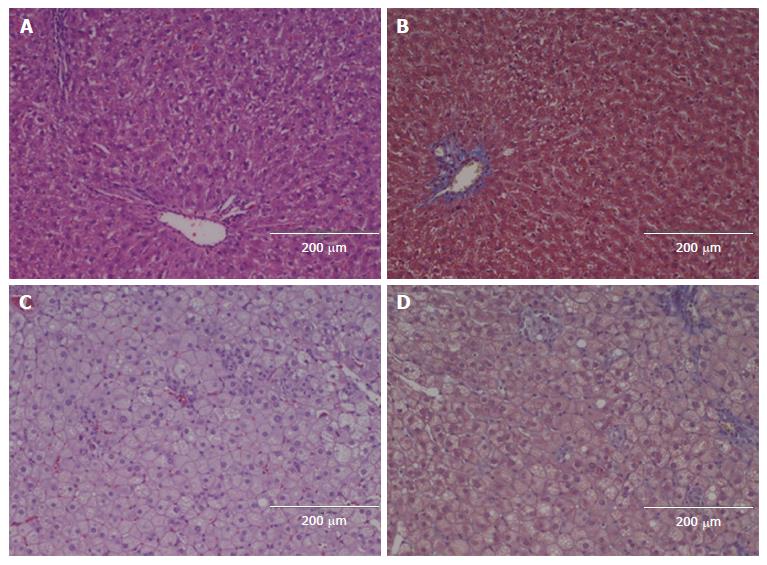
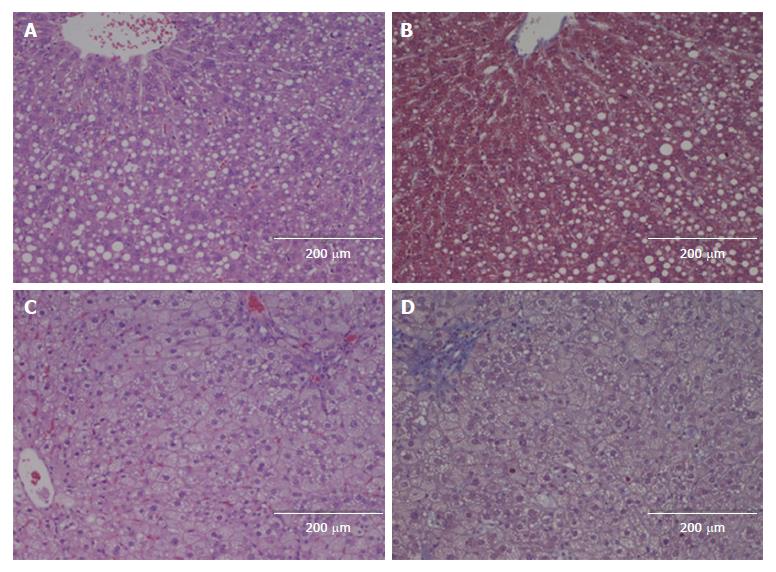
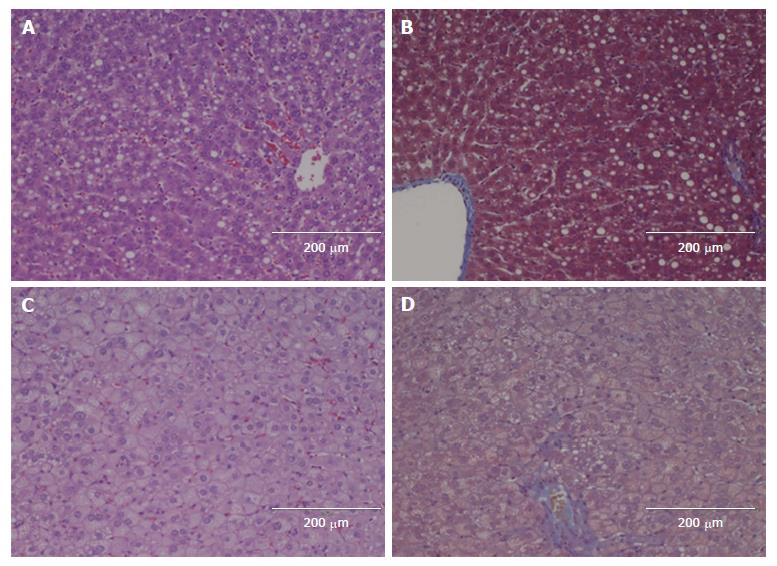
All the HFCD livers showed marked steatosis (grade 3) (Figures 2-4). Prior to hepatectomy, steatosis was of SDMS type, however, changing to SDMS/MXMS during regeneration. None of the HFCD animals had evidence of fibrosis. According to the Kleiner criteria, nine had borderline NASH and 17 had NASH; with Bedossa criteria, 11 had NAFLD and 15 NASH (Table 2). No morphological abnormalities were observed in the liver tissues of the control animals.
| High fat high cholesterol diet | |||
| NH (n = 9) | PH-2 (n = 9) | PH-5 (n = 8) | |
| Kleiner criteria score | |||
| Steatosis | 3 | 3 | 3 |
| Ballooning | 0.6 | 0.7 | 0.5 |
| Inflammation | 2.7 | 1.6 | 0.8 |
| Fibrosis | 0 | 0 | 0 |
| NAS score | 6.2 | 5.2 | 4.3 |
| Histological grading | |||
| Borderline NASH/NASH, Kleiner criteria | 1/8 | 4/5 | 4/4 |
| NAFLD/NASH, Bedossa criteria | 3/6 | 4/5 | 4/4 |
At baseline, a significant difference in Ki-67 liver proliferation index was observed between STD and HFCD animals (P < 0.001, Table 1); however, there was no difference between the HFCD and STD groups at either day 2 or day 5. Peak values were seen in the PH-2 groups.
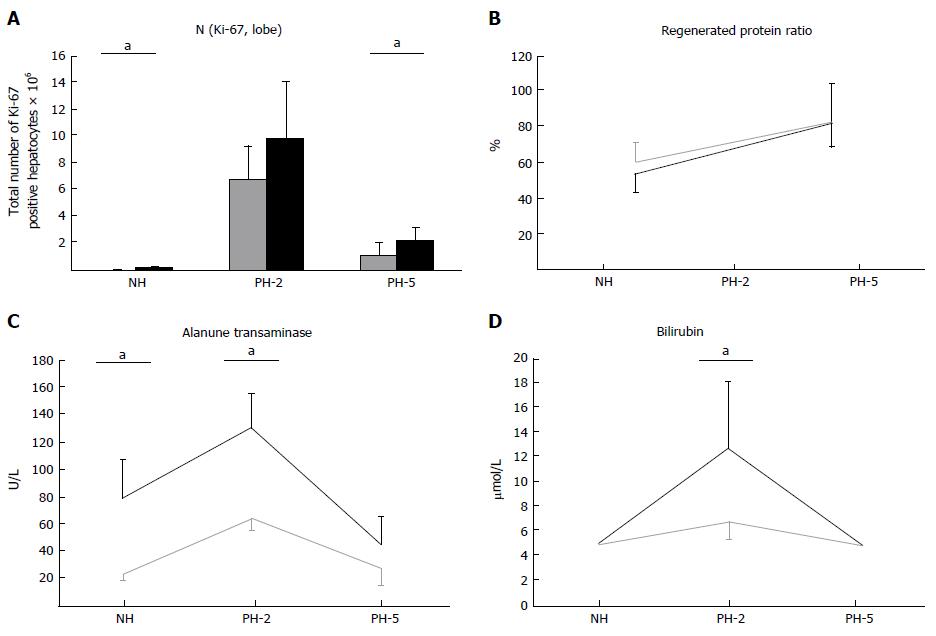
The total number of Ki-67 positive hepatocytes in the caudate lobe [N(Ki-67, lobe)] was significantly higher in HFCD animals at baseline (P < 0.0001) and at day 5 (P < 0.026), while a trend was observed at day 2 (P = 0.06) (Figure 5A).
The hepatectomized HFCD rats had a lower hepatic regeneration capacity as determined by HRR than the STD rats (P < 0.01, all). However, Net regeneration (NET) showed no difference at day 2 and was significantly higher in HFCD animals at day 5 (P < 0.018).
No differences were observed in the regeneration of hepatic protein, as determined by RPR (Figure 5B). The HFCD groups had a higher amount of total protein in the liver at baseline and at day 5 after PH (both P < 0.03, Table 1).
At baseline, ALT was significantly higher in the HFCD than in the STD animals (P < 0.001, Figure 5C) whereas bilirubin was unchanged (Figure 5D). Peak ALT and bilirubin levels were seen two days after hepatectomy with HFCD animals having significantly higher levels than the STD group (P < 0.05, Figure 5C and D). However, when calculating ΔALT between baseline and day two, no difference was found between HFCD and STD animals (Table 1). Surprisingly, ALT levels were lower in the PH-5 HFCD group than in the non-hepatectomized HFCD group (P < 0.001, Figure 5C). Bilirubin was normalized at day 5 in both the HFCD and STD groups (Figure 5D).
We did not find any significant differences in PP levels between the groups (Table 1).
Plasma albumin was significantly lower in the HFCD animals (P < 0.05) at baseline and at day 2 after hepatectomy (P < 0.001) compared with STD animals, whereas no difference was observed at day 5 after PH. In the HFCD groups, albumin levels dropped after hepatectomy but were stable during regeneration (Table 1).
At baseline, α2M was significantly higher in the HFCD than the STD animals (P < 0.01, Table 1). α2M increased two days after hepatectomy with HFCD animals having significantly higher values than the STD group (P < 0.001, Table 1). The PH-5 HFCD group had a α2M value similar to PH-5 STD controls (Table 1).
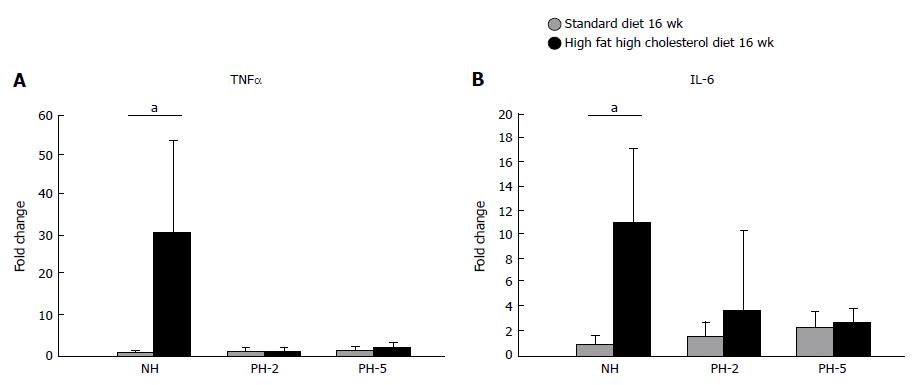
At baseline, the HFCD animals had increased tumor necrosis factor α (TNFα) and interleukin-6 (IL6) (Figure 6) gene expression when compared with STD animals, although interestingly, this difference was normalized during regeneration.
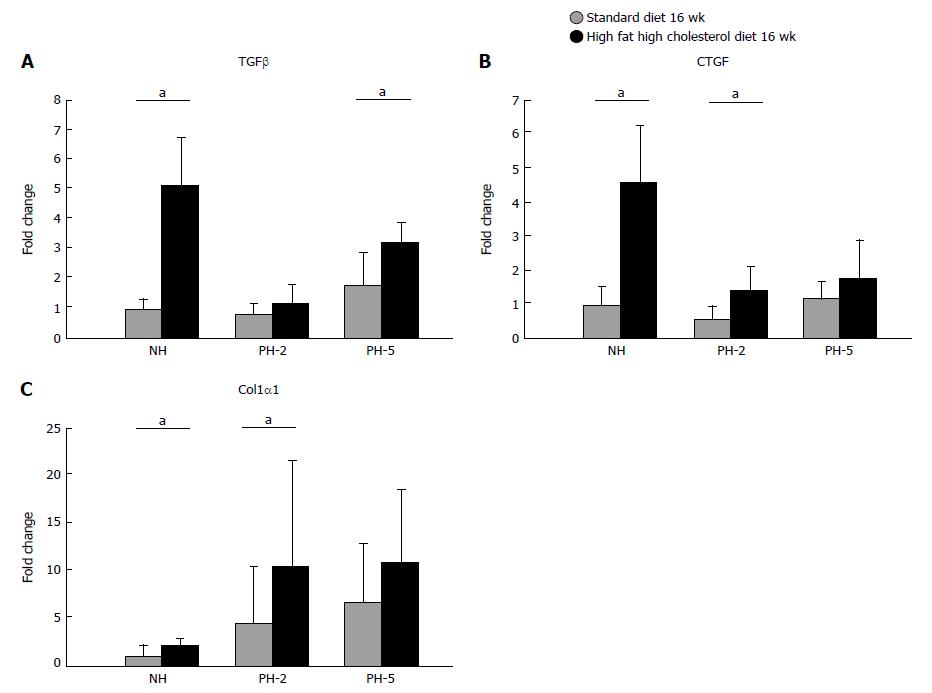
At baseline transforming growth factor β (TGFβ), connective tissue growth factor (CTGF) and Collagen 1α1 (COL1α1) mRNA expression were significantly higher in the HFCD group. At day 2, CTGF and COL1α1 mRNA expression were significantly higher in the HFCD group, while no significant difference was found when looking at TGFβ. At day five, only TGFβ was significantly higher when comparing HFCD and STD PH-5 groups (Figure 7) (P < 0.05).
At baseline, HFCD animals displayed a significantly higher mRNA expression of hepatocyte growth factor (HGF) and transforming growth factor α (TGFα), but no significant differences were seen in Proto-oncogene, tyrosine kinase (MET) and epidermal growth factor (EGF) mRNA expression. At day 2 and 5 HFCD groups had a higher expression of HGF, TGFα and EGF mRNA, while no significant difference was found when looking at MET compared with respective control groups (Figure 8) (P < 0.05).
We investigated LRC after a 70% PH in rats with HFCD-induced NASH. Surprisingly, we found a similar LRC when comparing HFCD and STD rats.
This is the first study to evaluate LRC in an HFCD-induced NASH model. Previous studies were conducted in other less suitable models. The MCD model potently induces NASH, but the rodents suffer severe weight loss and cachexia, in contrast to human NASH. In the CDD and HFD models only simple steatosis is induced[17,18,26,38-41]. Similarly, leptin-deficient rodents are gene-modified and thus do not reflect the etiological features of human NAFLD/NASH[29,42]. Our model successfully established NAFLD with significant steatosis and increased liver weight. NASH changes were present in the majority of HFCD animals with inflammation and ballooning. Prior to hepatectomy, the HFCD animals had elevated ALT levels with increased expression of genes related to inflammation and fibrogenesis, although none of the HFCD animals showed histological fibrosis. The type of steatosis changed from SDMS to SDMS/MXMS during regeneration, which might be explained by the fact that transient large fat droplet accumulation is a natural part of liver regeneration[43], as also seen in our STD animals.
As markers of liver injury, we observed elevated ALT and bilirubin levels two days post-hepatectomy in the HFCD groups compared with STD animals as previously found in MCD studies[9,11,12]. However, baseline ALT levels were already increased in the HFCD animals, and the relative ALT (ΔALT) increase following hepatectomy was similar in the HFCD and STD groups. A previous study using a steatosis-only HFD model found elevated ALT levels at day 1 after hepatectomy only in HFD animals compared to controls, but normal ALT levels both prior to hepatectomy and at day 3[26].
The Ki-67 liver proliferation index was similar between STD and HFCD animals during regeneration, however, this index does not take the larger cell size of the HFCD animals into account; which was the reason for estimating the total number of Ki-67 positive hepatocytes N(Ki-67, lobe).
N(Ki-67, lobe) was significantly higher in the HFCD animals at baseline and at day 5, with a trend on day 2. We used the same factor (1.05 g/cm3) for both control and HFCD groups even though we expected the density to be lower in the HFCD livers due to fat accumulation. In adipose tissue, the density is approximately 0.9 g/cm3[44]. This would probably decrease the total number of positive profiles by 5%-10% in the HFCD animals. Nonetheless, N(Ki-67, lobe) underlines the fact that the HFCD animals at the very least have a similar, if not higher, proliferative response during regeneration. Whether this is due to increased apoptosis, a delayed regenerative response or a higher regeneration remains elusive.
Other studies have reported both similar and opposing results using high-powered fields (HPF) when estimating proliferative indexes such as Ki-67. In rats fed a MCD diet for 5 wk, Veteläinen et al[12] found a decreased Ki-67 index at days 1, 2 and 3 after partial hepatectomy, while rats with simple steatosis after only one week of MCD diet had a normal Ki-67 index. Marsman et al[9] found a decreased Ki-67 index at day 1, but a normal or higher Ki-67 index between days 2 and 5 in MCD rats fed for 5 wk. Using a proliferating cell nuclear antigen (PNCA) labeling index (a marker of DNA synthesis), Tanoue et al[26] found normal PNCA index in HFD rats, whereas rats on a high fructose diet had a decreased PNCA index, although they had better histological scores than HFD rats. They speculated that the etiology of steatosis had more impact on the proliferative index than the degree of steatosis.
Clearly, this diversity in published results indicates that the Ki-67 index is greatly influenced by factors in the experimental design, such as the study duration, composition of the diet and not just the histopathological findings. Also, the methods of evaluation are important. By using a stereology-based design instead of semi-quantitative counting methods or counting HPF, we eliminated the selection bias of fields of view on obtained quantitative data. We sampled the whole caudate liver lobe, whereas previous studies have used single or multiple slabs from different liver lobes[9,12,26]. We believe that our stereological-based design is superior to HPF-based and semi-quantitative scoring systems since it eliminates several potential sources of error even though it is not perfectly unbiased.
Use of HRR and equivalent ratios have been widely reported in the literature of LRC in experimental NAFLD/NASH[7-10,12,14,17,18,20-22,24,39,45,46]. However, it is clearly problematic to use this measure, as the HRR is based on weight alone. Being fed a HFCD diet after PH, the HFCD liver will continue to accumulate fat. Using the HRR to compare steatotic livers with healthy livers, one must assume that the steatotic liver regenerates the same ratio of liver- and fat tissue as it has prior to hepatectomy.
In our study, we found a decreased LRC in HFCD rats looking exclusively at HRR. Looking at NET we found similar values at day 2 but at significantly higher value in HFCD animals at day 5. Both HHR and NET are measures that might be biased by fat accumulation. When considering other variables, such as the RPR, no difference was found between the two groups and this observation supports that the HRR value could be incorrect due to fat accumulation. To our knowledge, this study is the first to address this important issue - while previous studies have tended to use the outcome of the HRR uncritically[7-10,12,14,17,18,20-22,24,39,45,46].
Prothrombin-proconvertin ratio measures coagulation factors II, VII and X. Coagulation factors are exclusively synthesized in the liver and PP levels were therefore used as an indicator of hepatic protein synthesis. PP was not significantly affected in HFCD animals or at any time point following hepatectomy, which indicates that this specific metabolic liver function was already restored day 2 post-hepatectomy similar to findings during regeneration in healthy rats[47,48]. In contrast, albumin was decreased at baseline and at day 2 after hepatectomy in HFCD compared with STD animals; however, this difference disappeared at day 5.
For model evaluation and hepatectomy effects on liver inflammation and fibrosis, we measured the expression of genes related to inflammation (TNFα and IL-6) and fibrogenesis (TGFβ, CTGF, COL1α1). TNFα and IL-6 levels were elevated prior to hepatectomy in the HFCD animals but no difference was observed between HFCD and STD groups after PH. Thus, it seems that the inflammatory process is on hold during regeneration.
Despite the absence of histological fibrosis, we demonstrated increased hepatic levels of pro-fibrogenic cytokines (COL1α1, CTGF and TGFβ) in HFCD animals indicating active fibrogensis although not yet visible in the histology.
We studied the pathways of MET and the epidermal growth factor receptor (EGFR), which are generally accepted as the main mitogenic pathways of liver regeneration[49]. MET binds it’s ligand HGF, and EGFR binds several ligands, among others TGFα and EGF. Surprisingly, we did not find a decrease in the expression of the HGF/MET pathway in the HFCD animals. MET expression was unchanged and HGF expression higher at all time points in the HFCD animals compared to STD animals, which shows that this pathway is indeed not downregulated. HGF is proposed to have an anti-fibrotic effect[50], and we speculate that the elevation of HGF was a response to the on-going fibrogenesis in the HFCD animals. Also, we investigated the ligands for the EGFR pathway TFGα and EGF. In the HFCD groups, mRNA expression of both ligands was either higher or similar to STD groups in keeping with previous studies[51], indicating that this pathway is also not down-regulated.
The study has certain limitations. Liver regeneration is a process that commences immediately after liver injury and it would have been preferable to investigate liver regeneration as early as a few hours after PH as well as at day 1 after PH. Thus, early differences in HFCD animals may have been overlooked. Further, the evaluation of regeneration is compromised by the fat accumulation, which may disturb the results; however, no former study has addressed this important issue. The different measures of regeneration such as HHR, NET, RPR and N(Ki-67, lobe) are not flawless and have all limitations, but they leave the overall impression that the regenerative response in HFCD animals is at the very least, comparable to the STD animals.
In conclusion, we believe that the model and degree of NASH as well as methods of LRC evaluation are essential in understanding and evaluating LRC.
The HFCD induced significant steatosis and NASH changes along with increased expression of pro-inflammatory and pro-fibrogenic genes. However, we found that the HFCD rats had a preserved liver regeneration as assessed by total number of Ki-67 positive hepatocytes, RPR and PP, which was supported by the gene expression of growth factors during regeneration.
Epidemiological studies showed that liver resections are associated with increased morbidity and mortality in patients with non-alcoholic fatty liver disease (NAFLD)/ non-alcoholic steatohepatitis (NASH). It has been suggested that NASH livers are more vulnerable to surgical interventions because of decreased liver regeneration capacity (LRC). LRC has been studied in different animal models of NAFLD/NASH. However, these models may have significant limitations. Some models induce NASH but with severe weight loss, while other models induce simple steatosis only, further, genetic modified models may not reflect the etiological features of human NASH. In the present study we used a high fat high cholesterol diet (HFCD) rat model, which mimic human NASH better than previous models.
This is the first study of LRC in rats with NASH induced by a HFCD. Previous experimental NAFLD/NASH studies showed contradictory findings with decreased LRC or unchanged LRC, even when the same animal models were used. Clearly, the model and methods of evaluation may significantly influence the results and conclusions. For future treatment strategies of liver resections, it is important to understand whether the LRC of NAFLD/NASH livers is compromised.
The aim of the present study was to evaluate LRC in rats with NASH induced by a HFCD. Authors the methods of evaluation and the chosen model of NAFLD/NASH significantly influences the results and further research on the subject should be aware of this.
Rats were fed a high-fat, high-cholesterol diet (65% fat, 1% cholesterol) or standard diet (STD) for 16 wk. After the feeding phase 1/3 of the animals were euthanised immediately and served as a baseline reference. The remaining 2/3 of the animals underwent 70% partial hepatectomy (PH) and the hepatectomized animals were euthanised either 2 or 5 d post-PH. The degree of steatosis and the presence of NASH were evaluated by an expert liver pathologist using both the Kleiner and Bedossa criteria. LRC was evaluated using: the total number of Ki-67 positive hepatocytes in the caudate lobe, N(Ki-67, lobe) evaluated in a stereology-based design, the regenerated protein ratio (RPR), prothrombin-proconvertin ratio (PP), and mRNA expression of genes related to regeneration. The study is the first to use a stereology based design to evaluate cell proliferation. The authors believe this design superior to former methods of evaluation.
The study is also the first to address that future research should be cautious using the regenerated liver weight only to evaluate LRC. The NASH liver weight is biased by fat accumulation and when using the liver weight only one cannot account for whether the NASH liver regenerates fat- or liver tissue. Thus, we estimated the total protein concentration in the livers and used this to describe the regenerated liver mass. Biochemical tests were used as markers of liver injury. The data was analyzed using STATA. Normality of data was checked by qq-plots. For continuous variables, comparisons were made using the ANOVA test for significance. Post-hoc comparisons were performed by Student’s t-test. Categorical data were analyzed using Fisher’s exact test. The qPCR data exhibited skewed distributions with variance heterogeneity. Therefore, these data were analysed using the non-parametric Kruskal-Wallis one-way analysis of variance on ranks test; when significant, post-hoc tests were performed using the Mann-Whitney rank sum test.
The HFCD NASH model showed significant steatosis with ballooning and inflammation, while no fibrosis was present. Mortality was similar in HFCD and STD animals following PH. Further, HFCD animals had significantly elevated markers of liver injury after PH. HFCD animals had a higher N(Ki-67, lobe) at baseline, day 2 after PH and day 5 after PH. However, we found no significant difference in RPR or PP neither 2 or 5 d post-PH. Expression of liver regeneration genes was higher at both day 2 and 5 post-PH in HFCD groups.Authors evaluated LRC at day 2 and 5 after PH; however, it would have been interesting also to evaluate the very early stages of liver regeneration including time points as early as a few hours after PH and at day 1 after PH. Further, it would be of interest to investigate this rat model after more prolonged HFCD diet treatment when fibrosis may be more pronounced and if this decreases LRC. In addition, finding and identifying relevant new and better methods of LRC evaluation may ease the interpretation of the results.
The novel finding is that in a HFCD NASH model without fibrosis authors observed preserved LRC. The etiology and methods of evaluation is of great importance when evaluating LRC in animal models. Further, the fat accumulation in the NAFLD/NASH liver is a bias when estimating LRC and it needs to be addressed in future studies. In animal models the etiology of NAFLD/NASH and methods of evaluation is of significant importance in understanding LRC. Seemingly, NASH without fibrosis induced by a HFCD does not decrease LRC. HFCD induced NASH without fibrosis does not compromise LRC in rats following hepatectomy. HFCD induced NASH without fibrosis does not compromise LRC in rats following hepatectomy. When evaluating LRC the fat accumulation of the liver must be addressed, thus we have used both a stereological design to evaluate cell proliferation and measured the total protein concentration in the liver as a marker of regenerated liver mass. Prior to the study hypothesized LRC to be decreased, but in contrast we found a preserved LRC. It is too early to draw conclusions for clinical practice, but this study adds insight to the subject. Speculating, the reasons for increased morbidity and mortality in patients with NAFLD/ NASH following liver resections should be sought elsewhere than in decreased LRC.
Identifying and/or optimising relevant animal models of NAFLD/NASH as well as methods of evaluation for LRC. Using a pure stereological design for evaluation of cell proliferation, as this is perfectly unbiased. Using different markers LRC and being aware of the potential bias fat accumulation brings when evaluating LRC based on liver weight alone.
The authors wish to thank Claus Uhrenholt Christensen, Rikke Andersen, Birgitte Nielsen, Lene Vestergaard and Mette Mejlby Hansen who provided expert laboratory assistance.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Denmark
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Streba LAM, Xu CF S- Editor: Cui LJ L- Editor: A E- Editor: Li D
| 1. | European Association for the Study of the Liver (EASL). European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388-1402. [PubMed] |
| 2. | Oda K, Uto H, Mawatari S, Ido A. Clinical features of hepatocellular carcinoma associated with nonalcoholic fatty liver disease: a review of human studies. Clin J Gastroenterol. 2015;8:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 3. | Sanna C, Rosso C, Marietti M, Bugianesi E. Non-Alcoholic Fatty Liver Disease and Extra-Hepatic Cancers. Int J Mol Sci. 2016;17:pii: E717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 149] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 4. | Landreau P, Drouillard A, Launoy G, Ortega-Deballon P, Jooste V, Lepage C, Faivre J, Facy O, Bouvier AM. Incidence and survival in late liver metastases of colorectal cancer. J Gastroenterol Hepatol. 2015;30:82-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 5. | Smith JJ, D’Angelica MI. Surgical management of hepatic metastases of colorectal cancer. Hematol Oncol Clin North Am. 2015;29:61-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 6. | de Meijer VE, Kalish BT, Puder M, Ijzermans JN. Systematic review and meta-analysis of steatosis as a risk factor in major hepatic resection. Br J Surg. 2010;97:1331-1339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 194] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 7. | Zhang BH, Weltman M, Farrell GC. Does steatohepatitis impair liver regeneration? A study in a dietary model of non-alcoholic steatohepatitis in rats. J Gastroenterol Hepatol. 1999;14:133-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 8. | Picard C, Lambotte L, Starkel P, Sempoux C, Saliez A, Van den Berge V, Horsmans Y. Steatosis is not sufficient to cause an impaired regenerative response after partial hepatectomy in rats. J Hepatol. 2002;36:645-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 66] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 9. | Marsman HA, de Graaf W, Heger M, van Golen RF, Ten Kate FJ, Bennink R, van Gulik TM. Hepatic regeneration and functional recovery following partial liver resection in an experimental model of hepatic steatosis treated with omega-3 fatty acids. Br J Surg. 2013;100:674-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 10. | Hsiao IT, Lin KJ, Chang SI, Yen TC, Chen TC, Yeh TS. Impaired liver regeneration of steatotic rats after portal vein ligation: a particular emphasis on (99m)Tc-DISIDA scintigraphy and adiponectin signaling. J Hepatol. 2010;52:540-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 11. | Boeykens N, Ponsaerts P, Van der Linden A, Berneman Z, Ysebaert D, De Greef K. Injury-dependent retention of intraportally administered mesenchymal stromal cells following partial hepatectomy of steatotic liver does not lead to improved liver recovery. PLoS One. 2013;8:e69092. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Veteläinen R, van Vliet AK, van Gulik TM. Severe steatosis increases hepatocellular injury and impairs liver regeneration in a rat model of partial hepatectomy. Ann Surg. 2007;245:44-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 123] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 13. | Uzun MA, Koksal N, Kadioglu H, Gunerhan Y, Aktas S, Dursun N, Sehirli AO. Effects of N-acetylcysteine on regeneration following partial hepatectomy in rats with nonalcoholic fatty liver disease. Surg Today. 2009;39:592-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Tsuchiya T, Miyazawa M, Abe T, Saito T, Kanno H, Ishii S, Suzuki M, Kenjo A, Yamada F, Gunji T. Hepatic regeneration and ischemia/reperfusion injury in fatty-liver rats. Transplant Proc. 2000;32:2324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 15. | Rao MS, Papreddy K, Abecassis M, Hashimoto T. Regeneration of liver with marked fatty change following partial hepatectomy in rats. Dig Dis Sci. 2001;46:1821-1826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 16. | Ninomiya M, Shimada M, Terashi T, Ijichi H, Yonemura Y, Harada N, Soejima Y, Suehiro T, Maehara Y. Sustained spatial disturbance of bile canalicular networks during regeneration of the steatotic rat liver. Transplantation. 2004;77:373-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Tsai CY, Lin YS, Yeh TS, Cheong CF, Chang CH, Chen TC, Chen MF. Disrupted hepatic adiponectin signaling impairs liver regeneration of steatotic rats. Chang Gung Med J. 2011;34:248-259. [PubMed] |
| 18. | Lai HS, Lin WH, Chen PR, Wu HC, Lee PH, Chen WJ. Effects of a high-fiber diet on hepatocyte apoptosis and liver regeneration after partial hepatectomy in rats with fatty liver. JPEN J Parenter Enteral Nutr. 2005;29:401-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Yang SQ, Lin HZ, Mandal AK, Huang J, Diehl AM. Disrupted signaling and inhibited regeneration in obese mice with fatty livers: implications for nonalcoholic fatty liver disease pathophysiology. Hepatology. 2001;34:694-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 20. | Abshagen K, Mertens F, Eipel C, Vollmar B. Limited therapeutic efficacy of thrombopoietin on the regeneration of steatotic livers. Int J Clin Exp Pathol. 2013;6:1759-1769. [PubMed] |
| 21. | Yamauchi H, Uetsuka K, Okada T, Nakayama H, Doi K. Impaired liver regeneration after partial hepatectomy in db/db mice. Exp Toxicol Pathol. 2003;54:281-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Uetsuka K, Shirai M, Yamauchi H, Nakayama H, Doi K. Impaired proliferation of non-parenchymal cells participates in an impairment of liver regeneration in db/db mice. Exp Mol Pathol. 2005;79:51-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 23. | Redaelli CA, Semela D, Carrick FE, Ledermann M, Candinas D, Sauter B, Dufour JF. Effect of vascular endothelial growth factor on functional recovery after hepatectomy in lean and obese mice. J Hepatol. 2004;40:305-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 24. | Selzner M, Clavien PA. Failure of regeneration of the steatotic rat liver: disruption at two different levels in the regeneration pathway. Hepatology. 2000;31:35-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 191] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 25. | Leclercq IA, Vansteenberghe M, Lebrun VB, VanHul NK, Abarca-Quinones J, Sempoux CL, Picard C, Stärkel P, Horsmans YL. Defective hepatic regeneration after partial hepatectomy in leptin-deficient mice is not rescued by exogenous leptin. Lab Invest. 2006;86:1161-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 26. | Tanoue S, Uto H, Kumamoto R, Arima S, Hashimoto S, Nasu Y, Takami Y, Moriuchi A, Sakiyama T, Oketani M. Liver regeneration after partial hepatectomy in rat is more impaired in a steatotic liver induced by dietary fructose compared to dietary fat. Biochem Biophys Res Commun. 2011;407:163-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 27. | Heebøll S, El-Houri RB, Hellberg YE, Haldrup D, Pedersen SB, Jessen N, Christensen LP, Grønbaek H. Effect of resveratrol on experimental non-alcoholic fatty liver disease depends on severity of pathology and timing of treatment. J Gastroenterol Hepatol. 2016;31:668-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 28. | Thomsen KL, Grønbæk H, Glavind E, Hebbard L, Jessen N, Clouston A, George J, Vilstrup H. Experimental nonalcoholic steatohepatitis compromises ureagenesis, an essential hepatic metabolic function. Am J Physiol Gastrointest Liver Physiol. 2014;307:G295-G301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 29. | Xu ZJ, Fan JG, Ding XD, Qiao L, Wang GL. Characterization of high-fat, diet-induced, non-alcoholic steatohepatitis with fibrosis in rats. Dig Dis Sci. 2010;55:931-940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 147] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 30. | Andersen KJ, Knudsen AR, Kannerup AS, Sasanuma H, Nyengaard JR, Hamilton-Dutoit S, Erlandsen EJ, Jørgensen B, Mortensen FV. The natural history of liver regeneration in rats: description of an animal model for liver regeneration studies. Int J Surg. 2013;11:903-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6807] [Cited by in RCA: 8248] [Article Influence: 412.4] [Reference Citation Analysis (5)] |
| 32. | Bedossa P, Poitou C, Veyrie N, Bouillot JL, Basdevant A, Paradis V, Tordjman J, Clement K. Histopathological algorithm and scoring system for evaluation of liver lesions in morbidly obese patients. Hepatology. 2012;56:1751-1759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 649] [Article Influence: 49.9] [Reference Citation Analysis (0)] |
| 33. | Cruz-Orive LM. Distribution-free estimation of sphere size distributions from slabs showing overprojection and truncation, with a review of previous methods. J Microsc. 1983;131:265-290. [RCA] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 112] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 34. | Sohlenius-Sternbeck AK. Determination of the hepatocellularity number for human, dog, rabbit, rat and mouse livers from protein concentration measurements. Toxicol In Vitro. 2006;20:1582-1586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 181] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 35. | Brix-Christensen V, Gjedsted J, Andersen SK, Vestergaard C, Nielsen J, Rix T, Nyboe R, Andersen NT, Larsson A, Schmitz O. Inflammatory response during hyperglycemia and hyperinsulinemia in a porcine endotoxemic model: the contribution of essential organs. Acta Anaesthesiol Scand. 2005;49:991-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 36. | Chomczynski P, Sacchi N. The single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction: twenty-something years on. Nat Protoc. 2006;1:581-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1288] [Cited by in RCA: 1371] [Article Influence: 72.2] [Reference Citation Analysis (0)] |
| 37. | Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149116] [Cited by in RCA: 133995] [Article Influence: 5583.1] [Reference Citation Analysis (1)] |
| 38. | Sydor S, Gu Y, Schlattjan M, Bechmann LP, Rauen U, Best J, Paul A, Baba HA, Sowa JP, Gerken G. Steatosis does not impair liver regeneration after partial hepatectomy. Lab Invest. 2013;93:20-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 39. | DeAngelis RA, Markiewski MM, Taub R, Lambris JD. A high-fat diet impairs liver regeneration in C57BL/6 mice through overexpression of the NF-kappaB inhibitor, IkappaBalpha. Hepatology. 2005;42:1148-1157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 125] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 40. | Hamano M, Ezaki H, Kiso S, Furuta K, Egawa M, Kizu T, Chatani N, Kamada Y, Yoshida Y, Takehara T. Lipid overloading during liver regeneration causes delayed hepatocyte DNA replication by increasing ER stress in mice with simple hepatic steatosis. J Gastroenterol. 2014;49:305-316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 41. | Veteläinen R, van Vliet A, van Gulik TM. Essential pathogenic and metabolic differences in steatosis induced by choline or methione-choline deficient diets in a rat model. J Gastroenterol Hepatol. 2007;22:1526-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 81] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 42. | Kucera O, Cervinkova Z. Experimental models of non-alcoholic fatty liver disease in rats. World J Gastroenterol. 2014;20:8364-8376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 148] [Cited by in RCA: 143] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 43. | Stein TA, Burns GP, Tropp BE, Wise L. Hepatic fat accumulation during liver regeneration. J Surg Res. 1985;39:338-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 44. | Farvid MS, Ng TW, Chan DC, Barrett PH, Watts GF. Association of adiponectin and resistin with adipose tissue compartments, insulin resistance and dyslipidaemia. Diabetes Obes Metab. 2005;7:406-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 98] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 45. | Shirai M, Yamauchi H, Nakayama H, Doi K, Uetsuka K. Expression of epidermal growth factor receptor protein in the liver of db/db mice after partial hepatectomy. Exp Toxicol Pathol. 2007;59:157-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 46. | Murata H, Yagi T, Iwagaki H, Ogino T, Sadamori H, Matsukawa H, Umeda Y, Haga S, Takaka N, Ozaki M. Mechanism of impaired regeneration of fatty liver in mouse partial hepatectomy model. J Gastroenterol Hepatol. 2007;22:2173-2180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 47. | Yildirim SI, Poulsen HE. Quantitative liver functions after 70% hepatectomy. Eur J Clin Invest. 1981;11:469-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 48. | Antović J, Djordjević V, Kocić G, Koraćević D, Bjelaković G, Bakić M. Blood coagulation factors changes during liver regeneration in rats. Arch Int Physiol Biochim Biophys. 1993;101:357-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 49. | Michalopoulos GK. Liver regeneration after partial hepatectomy: critical analysis of mechanistic dilemmas. Am J Pathol. 2010;176:2-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 578] [Cited by in RCA: 571] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 50. | Kim MD, Kim SS, Cha HY, Jang SH, Chang DY, Kim W, Suh-Kim H, Lee JH. Therapeutic effect of hepatocyte growth factor-secreting mesenchymal stem cells in a rat model of liver fibrosis. Exp Mol Med. 2014;46:e110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 51. | Webber EM, FitzGerald MJ, Brown PI, Bartlett MH, Fausto N. Transforming growth factor-alpha expression during liver regeneration after partial hepatectomy and toxic injury, and potential interactions between transforming growth factor-alpha and hepatocyte growth factor. Hepatology. 1993;18:1422-1431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 97] [Article Influence: 3.0] [Reference Citation Analysis (0)] |