Published online Oct 26, 2013. doi: 10.4252/wjsc.v5.i4.112
Revised: August 7, 2013
Accepted: August 20, 2013
Published online: October 26, 2013
Processing time: 139 Days and 15.5 Hours
Stress urinary incontinence (SUI), as an isolated symptom, is not a life threatening condition. However, the fear of unexpected urine leakage contributes to a significant decline in quality of life parameters for afflicted patients. Compared to other forms of incontinence, SUI cannot be easily treated with pharmacotherapy since it is inherently an anatomic problem. Treatment options include the use of bio-injectable materials to enhance closing pressures, and the placement of slings to bolster fascial support to the urethra. However, histologic findings of degeneration in the incontinent urethral sphincter invite the use of tissues engineering strategies to regenerate structures that aid in promoting continence. In this review, we will assess the role of stem cells in restoring multiple anatomic and physiological aspects of the sphincter. In particular, mesenchymal stem cells and CD34+ cells have shown great promise to differentiate into muscular and vascular components, respectively. Evidence supporting the use of cytokines and growth factors such as hypoxia-inducible factor 1-alpha, vascular endothelial growth factor, basic fibroblast growth factor, hepatocyte growth factor and insulin-like growth factor further enhance the viability and direction of differentiation. Bridging the benefits of stem cells and growth factors involves the use of synthetic scaffolds like poly (1,8-octanediol-co-citrate) (POC) thin films. POC scaffolds are synthetic, elastomeric polymers that serve as substrates for cell growth, and upon degradation, release growth factors to the microenvironment in a controlled, predictable fashion. The combination of cellular, cytokine and scaffold elements aims to address the pathologic deficits to urinary incontinence, with a goal to improve patient symptoms and overall quality of life.
Core tip: Stress urinary incontinence is a condition which affects millions of women on a world-wide basis. Current surgical strategies to alleviate the symptoms involved with this condition are temporary stop-gap measures. With the advent of tissue engineering strategies in combination with stem cells, the reality of creating a functional replacement for anatomic structures involved in stress urinary incontinence can be a reality.
- Citation: Thaker H, Sharma AK. Regenerative medicine based applications to combat stress urinary incontinence. World J Stem Cells 2013; 5(4): 112-123
- URL: https://www.wjgnet.com/1948-0210/full/v5/i4/112.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v5.i4.112
Symptoms of the lower urinary tract in women, such as urinary incontinence, account for a significant number of outpatient consultations to urogynecologists[1]. Several risk factors have been identified to explain the onset and persistence of this condition, particularly since the prevalence of urinary incontinence among adult women is approximately 16%-30%[2,3]. The occurrence of urinary incontinence is closely correlated with rising age[4], along with obesity through increased pressure on the pelvic floor, diabetes mellitus causing microvascular and neuropathic changes, and prior pelvic surgeries for the fascial, muscular and nerve stress it imparts. Among younger women, pregnancy and subsequent vaginal delivery leads to post-partum incontinence in up to 28% of the population. Bladder outlet compression, pelvic floor strain that elongates the pudendal nerve, and a prolonged second stage of labor all contribute towards urinary leakage[5-7]. Less often, patients with a chronic cough or those with fascial weakness secondary to a defect in collagen metabolism may have incontinence as well[8]. In adult men, direct injury to the neurovascular bundle and fascial planes during radical prostatectomy is a leading cause of incontinence[9].
A Scandinavian study reports that as many as 64% of women with urinary incontinence issues do not consult their primary care physician for a diagnostic work up of their symptoms[10]. A similar study in the United States (US) observed a rate of about 45%[11]. The prevalence of urinary incontinence, therefore, could be much higher than documented. Though not life-threatening, patients endure a dramatic decline to their quality of life. A survey across Europe and the US documented this subjective aspect, showing that confidence, self-perception, levels of physical activity and social engagement were negatively impacted by incontinence[12-14]. Not surprisingly, reports show a higher prevalence of anxiety and depression among these patients as well[15,16]. For the elderly in nursing homes, uncontrollable passage of urine irritates the perineal skin, causing dermatitis and discomfort that is difficult to alleviate[17].
Urinary incontinence is a condition in which there is involuntary leakage of urine that can be attributed to a number of differing factors. Depending on the presentation and history, incontinence is divided into several groups. Stress urinary incontinence (SUI) is an involuntary loss of urine that occurs with increased abdominal exertion, exemplified during coughing, sneezing, laughing or lifting. If the bladder is retaining urine in excessive amounts, small increases to abdominal pressure will produce an unwanted leak. Yet this symptom can still occur within the normal range of bladder fullness, such as in defects to the urethra or the intrinsic sphincter in retaining urine. A laxity of supportive structures to the pelvic floor and bladder means that increased pressures cannot be counteracted. Urge urinary incontinence presents with frequency causing low volume output, urgency and nocturia. The leakage of urine is typically accompanied by or preceded by a sense of urgency. A combination of these two types is described as mixed urinary incontinence, where increased abdominal pressures cause urinary leakage and concomitant urgency. Overactive bladder is characterized by urgency, with or without incontinence. Less common voiding abnormalities include conditions such as nocturnal enuresis and continuous urinary incontinence. An acute onset of urinary incontinence may suggest an underlying neurologic degenerative disease or malignancy, and should therefore be considered in the differential diagnosis[18-20].
Though the development of urinary incontinence is multifactorial in nature[21], all variations of disease in SUI ultimately manifest as either urethral hypermobility or urethral sphincter dysfunction. The effects of aging are particularly well described to demonstrate this. Increasing age leads to urethral musculature degeneration and neurologic injury[22]. The number of striated muscle fibers diminishes with histologic thinning noted at the proximal vesicle neck (supported by the U-shaped detrusor) and at the dorsal wall of the urethra (encircled by striated sphincter muscles). In addition, an age-related weakening of the endopelvic fascia reduces the support to the urethra, causing an inability to maintain the physiologic 90-120 degree vesicourethral angle required to maintain continence[23]. What results is termed urethral hypermobility. The endopelvic fascial layer is a dense, fibrous connective tissue layer surrounding the vagina, with attachments to the arcus tendinous fascia, the pubic bone ventrally, and the ischial spine dorsally[23]. Degenerative changes diminish the ability of the urethra to generate pressure against stress. As an example, a staccato cough creates a 150 cmH2O increase in abdominal pressure, which the urethra must counteract during non-micturation times. A more compliant fascial layer threatens to lose continence during such transient pressures changes.
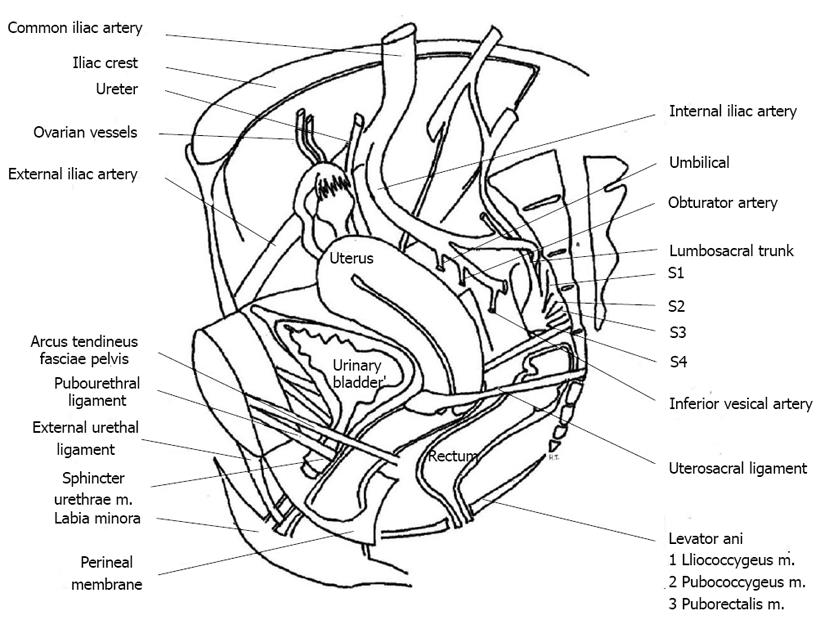
Initiating therapy for SUI requires the identification and understanding of the functional components comprising the urinary sphincter. The nerve supply to the lower urinary tract consists of three important innervations (Figure 1). First, pelvic nerves (S2-S4) provide parasympathetic innervations to the detrusor muscle and urethral smooth muscle sphincter. As an excitatory motor efferent nerve, the pelvic nerves initiate and coordinate micturition by contracting the detrusor and relaxing the sphincter. Second, hypograstric nerves (T2-L3) are sympathetic innervations to the trigone, bladder neck internal sphincter and detrusor muscle. These nerves allow for urinary retention and inhibit detrusor activity. Lastly, the pudendal nerves (S2-S4) of the sacral plexus innervate the external urethral sphincter (EUS) and striated muscles of the pelvic floor. The EUS consists of circular striated “slow twitch” fibers that sustain long periods of contraction, and pelvic basin muscles like the pubococcygeus contains slow and fast twitch fibers for reflex contraction during a cough or sneeze. The pudendal nerve arises from Onuf’s nucleus, traverses Alcock’s canal and enters the ischiorectal fossa to innervate the EUS[6]. This trajectory places the nerve between the sacrospinous and sacrotuberous ligaments, which makes it vulnerable to compression and injury.
Conservative management of SUI involves lifestyle and behavioral changes, weight loss[24], bladder training[25], Kegel exercises to strengthen the pelvic floor and pharmacotherapy to improve urethral muscle tone[26-28]. Unlike other forms of urinary incontinence, there is a dearth of pharmacologic options that are effective for SUI. Alpha-adrenergic receptors at the bladder neck and urethra can be targeted with pseudoepherine and ephedrine to stimulate smooth muscle contraction[27,29]. Though this use may theoretically be suited to improve symptoms, the cardiovascular side effect profile precludes many patients from this therapy[30]. Imipramine, a tricyclic antidepressant with strong anti-cholinergic effects, also promotes urethral sphincter closure and urinary retention. This drug, however, poses a significant risk of orthostatic hypotension, which is often contraindicated in the elderly population. Lately, the use of duloxetine outside of the US has shown some promise, though the mechanism of action hardly addresses the underlying cause[31,32]. Patients with SUI and concomitant urge symptoms have more therapeutic options, including anti-muscarinic agents and oxybutynin[33].
Despite these initial efforts to gain continence, most of these patients are refractory to treatment and are invariably referred for surgical intervention. The goals of surgery are to reduce the number of episodes of incontinence each day or to reduce the volume of leaked urine, with the ultimate aim of achieving complete continence. Surgery can either support the compression of the urethra, or augment coaptation to create a better seal. Pre-operative assessment of surgical candidates begins with a detailed a history with bladder diary recordings, and a physical, including a bimanual exam. A urinalysis to rule out infectious causes is indicated, followed by a cough stress test. In this test, the clinician visualizes the leakage of urine from a full bladder while the patient coughs. The volume of urine leak, collected on chux padding, may signify the degree of stress incontinence. Once the diagnosis of SUI is confirmed, most patients move on to urodynamic testing.
Urodynamic testing is expensive and subject to operator variability[34,35], but it provides two important measurements for the management of SUI. The first is leak point pressure (LPP). LPP measures the intravesical pressure at which urine leakage occurs when the patient is asked to strain or subjected to the Valsalva manuever. The patient must be careful to not induce a detrusor contraction. This is a measurement of intrinsic sphincter deficiency, and a LPP less than 60 cmH2O is suggestive of SUI. The second measurement is the maximum urethral pressure generated, which is part of the urethral pressure profile. Profilometry also documents maximum urethral closure pressure, functional urethral length and the pressure transmission ratio. Maximum urethral pressures less than 20-30 mmHg are suggestive of SUI, and warrants the use of sling placement or periurethral bulking agent injections[36].
The surgical approach to SUI involves either placement of slings to support of the urethra, or injection of biomaterials to increase urethral coaptation. Minimally invasive sling placement is currently the most common intervention for SUI[37]. Through a vaginal incision, a thin mesh of polypropylene thread material is positioned at the midurethral or at the bladder neck[38]. The sling behaves like an immobile floor, onto which the urethra can contract[39]. This strategy assumes the incontinent sphincter is indeed caused by urethral hypermobility, so supporting the plane of contraction would limit the angulation. Midurethral slings may be placed with either a retropubic or transobturator approach. A multicenter, randomized trial shows equivalent outcomes regardless of approach[40], though some studies still report better outcomes with a tension-free vaginal tape retropubic sling[41]. Newer interventions using a single incision sling or an adjustable sling have also shown promise[42].
Midurethral sling procedures have largely replaced the older Marshall-Marchetti-Krantz and Burch techniques of colposuspension[38,43]. Though these procedures have success rates of up to 88%[44], complications of urinary retention, de novo urgency, posterior vaginal wall prolapse and osteitis pubis have been documented[45]. In contrast, a Cochrane review of sling surgeries showed that the minimally invasive approach lessens operative times and decreases post-operative voiding dysfunction[38].
Sling materials have traditionally been sourced from autologous rectus fascia, fascia lata or small intestinal submucosa[46-48]. Cadaveric allografts from the dura, dermis or fascia lata have been shown to be inferior to autologous grafts in the long term based on the frequency of recurrent incontinence[49]. However, harvesting autologous fascial layers requires longer operative times, longer recovery times and more pain for the patient[50], despite evidence suggesting they are equivalent to synthetic slings[51]. For this reason, synthetic materials have become the mainstay in slings.
Sling placement, albeit a minimally invasive procedure, is not without risks and complications. Establishing an appropriate tension across the urethra is challenging, and can result in urinary retention or failure to alleviate symptoms. Both outcomes require release and adjustment of the sling, which is another surgical procedure. Other risks include bladder or urethral laceration, perforation and urinary tract infections (UTI)[52]. Occasionally, these issues are secondary to erosion of the mesh through soft tissue. These patients suffer from vaginal discharge, post-coital spotting, dyspareunia, and frequent UTIs. Despite these potential complications, the majority of patients do achieve freedom from incontinence, and self-reported improvements to sexual activity[53,54], and reductions in coital incontinence[55].
Injecting biomaterials to augment the urethral mucosa has become an alternative to urethral slings. Bulking agents have the greatest benefit for intrinsic sphincter defects with a LPP of less than 60 cmH2O. Regardless of where the injection is placed, studies have shown equivalent success with biomaterials in the periurethral, transurethral, midurethral and proximal neck areas[56,57]. The differences then lie in the biomaterial composition.
Current surgical standards use non-antigenic synthetic materials for slings. Over the past few years, studies have investigated the potential role of stem cells in SUI treatment. In a study by Zou et al[58], acellular silk slings were tested against slings seeded with autologous bone marrow derived mesenchymal stem cells (MSCs) on rats with bilateral sciatic nerve transection. MSCs were isolated from bone marrow aspirates using flow cytometry against CD34, CD44 and CD105 cell surface epitopes. Sciatic nerve transection, among other methods, is a means to produce genuine SUI pathology[59]. At 12 wk post-implantation, MSC/silk slings had double the collagen fiber formation of silk slings alone, evidenced by a higher Young’s modulus (4.468 ± 0.510 MPa) and higher failure force (2.436 ± 0.192 N) as compared to silk slings alone. The mean Young’s modulus of silk slings alone was 3.045 ± 0.388 MPa, with a failure force of 1.521 ± 0.087 N. The collagen formation improved sling integration with the native urethral tissue. However, both MSCs/silk and silk alone constructs performed equally in increasing the LPP (MSCs/silk at 36.3 ± 3.1 cmH2O vs silk alone at 38.0 ± 3.3 cmH2O). Nonetheless, this study demonstrates that the introduction of MSCs into the urethral environment does not cause any significant inflammation, scarring or adverse effects. Other scaffolds may be better suited in lieu of silk. It is important to note that even though a stem cell seeded construct could improve integration of slings into the urethra, the operative risks remain the same as that for current sling placements. Additionally, there is no evidence demonstrating that cellular slings have a decreased risk of mesh erosion over commercially available slings.
Attribution of SUI to intrinsic sphincter degeneration poses a challenging problem from a therapeutic standpoint. Sphincter degeneration involves the loss of multiple functional tissue types. Efforts to recreate the function of urethral sphincters are best demonstrated through artificial fluid-filled cuffs encircling the urethra. Artificial urinary sphincters have three components: a cuff of 4.5 cm in size, a reservoir with 61-70 cmH2O to mimic urethral pressures, and a pump to permit inflation and deflation controlled by the patient. Sphincters are most commonly placed at the bulbar urethra in men who suffer from post-prostatectomy SUI[9]. Though theoretically purposeful, artificial sphincters are associated with a multitude of complications. Acutely, urethral edema produces pain and discomfort for the patient. Chronically, patients experience atrophy and erosion of the sphincter resulting in irritative voiding symptoms, perineal pain and hematuria[60]. There are presently no controlled trials showing an improvement to symptoms using an artificial device over conventional therapy[61]. As such, we consider the employment of stem cells and tissue engineering techniques to reconstruct the urethral sphincter.
Several studies have established a foundation of infusing stem cells directly into the urethral sphincter. Preparations of MSCs, autologous progenitor muscle cells[62], adipose cells[63], processed lipoaspirate[64,65], human amniotic stem cells[66] and fibroblasts[67] have all been used with variable results[62,68,69] to bolster smooth muscle regeneration and to improve LPPs and urethral closure pressures (Table 1). Few studies have assessed the role of stem cells for the subset of male patients with SUI from prostate-related surgery. In one study, transurethral injections of autologous muscle derived fibroblasts and myoblasts produced complete continence in 65% of the 63 participants, quantified by a pre-operative LPP of 46.3 ± 17.1 cmH2O, and a post-operative LPP of 68.2 ± 24.3 cmH2O[70]. Another study using a similar approach reported improvements to merely 12% of 222 male patients, with no improvements in 46%[71]. While both studies showed that stem cell implantation is a safe procedure in eligible patients, the results do not show a clear benefit as seen in trials with women and SUI.
| Study | Stem/progenitor cell source | SUI model | LPP findings in cmH2O at 4 wk status post injection | Absolute difference in LPP in cmH2O and percent improvement |
| Lee et al[117] | Allogenic gastrocnemius MDSC | Rat, sciatic nerve transection | MDSC = 44.1 ± 6.6 Saline = 18.6 ± 5.2 Control (-) = 25.8 ± 2.5 | MDSC = 18.3 ± 9.1, 70.93% |
| Kwon et al[67] | Muscle derived cells and fibroblasts | Rats, sciatic nerve transection | MDSC = 38.2 ± 4.3 Fibroblast = 38.8 ± 1.2 MDSC/fibroblast = 34.5 ± 3.3 Control (-) = 25.8 ± 1.4 Control (+) = 43.3 ± 2.5 | MDSC = 12.4 ± 5.7, 48.06% Fibroblast = 13.0 ± 2.6, 50.38% MDSC/Fibroblast = 8.7 ± 4.7, 33.72% |
| Fu et al[118] | Adipose derived stem cells induced into myoblasts with 5-azacytidine | Rats, vaginal balloon dilation | Myoblast = 32.43 ± 2.05 ADSCs = 30.75 ± 3.17 Control (-) = 36.19 ± 2.25 Control (+) = 45.42 ± 1.71 | Myoblast = -3.76 ± 4.3, -10.38% ADSC = -5.44 ± 5.42, -15.03% |
| Kinebuchi et al[69] | Bone marrow derived mesenchymal stem cells | Rats, urethrolysis and cardiotoxic injection | BMSC = 25.66 ± 4.38 Control (-) = 18.19 ± 1.55 Control (+) = 35.98 ± 5.14 | BMSC = 7.47 ± 5.93, 41.06% |
| Lim et al[119] | Human umbilical cord blood mononuclear cells | Rats, electrocauterization of periurethral soft tissue | HUCS = 91.75 ± 18.99 Control (-) = 65.02 ± 22.09 | HUCS = 26.76 ± 41.08, 41.16% |
| Kim et al[120] | Allogenic mesenchymal stem cells | Rats, pudendal nerve transection | MSC = 43.1 ± 3.2 Control (-) = 22.0 ± 2.2 Control (+) = 29.1 ± 2.1 | MSC: 21.1 ± 5.4, 95.90% |
| Zhao et al[93] | Autologous adipose derived stem cells + NGF | Rats, pudendal nerve transection | ADSC/PLGA = 17.8 ± 3.1 ADSC/NGF = 18.3 ± 2.4 ADSC/PLGA/NGF = 22.5 ± 6.1 Control (-) = 11.6 ± 2.7 | ADSC/PLGA = 6.2 ± 5.8, 53.44% ADSC/NGF = 6.7 ± 5.1, 57.75% ADSC/PLGA/NGF = 10.9 ± 8.8, 93.96% |
| Corcos et al[72] | Bone marrow derived mesenchymal stem cells | Rats, pudendal nerve transection | MSC = 24.28 ± 1.47 Control (-) = 16.21 ± 1.26 | MSC = 8.07 ± 2.73, 49.78% |
| Watanabe et al[121] | Adipose derived mesenchymal stem cells | Rats, pelvic nerve transection | Subtracted LPPs: Collagen = 9.39 ± 2.08 ASC = 8.86 ± 3.13 Control (-) = 10.94 ± 3.55 | |
| Kim et al[122] | Human amniotic fluid stem cells | Rats, pudendal nerve transection | hAFCs = 20.2 ± 3.3 Control (-) = 15.2 ± 3.1 Control (+) = 27.6 ± 3.6 | hAFCs = 5.0 ± 6.4, 32.89% |
| Li et al[123] | Adipose derived stem cells | Rats, vaginal balloon dilation | ADSC = 46.34 ± 2.63 Control (-) = 36.82 ± 1.68 Control (+) = 48.92 ± 2.71 | ADSC = 9.52 ± 4.31, 25.85% |
| Chun et al[66] | Human amniotic fluid stem cells | Rats, pudendal nerve transection | hAFCs = 23.9 ± 1.85 Muscle progenitors = 38.43 ± 0.51 Control (-) = 15.24 ± 1.87 Control (+) = 36.54 ± 1.67 | hAFCs = 8.66 ± 3.72, 56.82% Muscle progenitors = 23.19 ± 2.38, 152.16% |
| Lecoeur et al[124] | Myofiber core of satellite cells | Pigs, endoscopic destruction of striated urethral sphincter | Myofiber = 71.5 ± 17.8 Curarization sphincter = 33.5 ± 14.8 | |
| Mitterberger et al[125] | Autologous myoblasts and fibroblasts | Humans (n = 123) | UCP = 28.8 ± 12.3 Post-operative UCP = 40.5 ± 15.8 after 1 year | 11.7 ± 28.1, 40.62% |
| Mitterberger et al[126] | Autologous myoblasts and fibroblasts | Humans (n = 20) | Pre-operative UCP = 27.0 ± 13.3 Post-operative UCP (1 yr) = 39.4 ± 14.8 Post-operative UCP (2 yr) = 42.2 ± 12.1 | 66.4 ± 28.1, 45.92% (1 yr) 15.2 ± 25.4, 56.29% |
Using MSCs seems to show the greatest promise, as MSCs have displayed the potential to regenerate both muscle and ganglion components in the sphincter. Corcos et al[72] demonstrated in an animal model that injecting BMSCs into denervated urethral sphincters improved LPPs to almost normal, non-SUI levels. This result is argued to be due to the differentiation of MSCs into striated muscle within the urethral microenvironment[72]. Though this evidence is merely histologic, and not in an improvement to symptoms for patients, the concept of creating a functional contractile tissue in the sphincter is worthy of further development.
MSC use in tissue engineering has become a prominent strategy in a multitude of fields, including urogynecology[73-79]. MSCs express cell surface markers CD29, CD44, CD105, CD166, and are negative for hematopoietic markers such as CD14, CD34, CD40 and CD45[80]. MSCs are also negative for leukocyte common antigen CD45, suggesting that these stem cells escape lymphocyte detection, and thus avoid immune rejection[81,82]. Sourced from the bone marrow, MSCs can be easily isolated from other hematopoietic cells through flow cytometry. MSCs have the capacity to divide 24-40 times in vitro, allowing for multiple passages of expansion without losing their multipotent properties or differentiating spontaneously[83]. This advantage, described by Pittenger et al[84], permits the differentiation of MSCs by external forces, such as the microenvironment of target tissue itself. Coupled to this environment, MSCs display an immunomodulatory effect[85] that includes the secretion of cytokines to initiate and support tissue regeneration[86]. For these reasons, MSCs must, by minimal criteria, differentiate into osteoblasts, chondrocytes and adipocytes. Yet, the plasticity inherent in MSCs has pushed researchers to generate neural, cardiac, muscular and other soft tissue lineages. In the realm of urinary tract healing, MSCs are considered a prime candidate since their presence has great therapeutic potential with minimal complications[87,88].
Some centers are already offering stem cell injections into the urethra for patients[89-91]. However, it is unclear whether these cells serve a functional purpose in regenerating damaged sphincters, or whether the cells are merely a bulking agent not unlike injectable biomaterials. In addition, it is recognized that inflammation at the implant site diminishes the ability of injected cells to survive long enough to participate in regeneration[92]. If growth were not sustained over a 7-10 d period, the applications of MSCs would be significantly stunted. Providing a means to enhance cell viability in vivo could be achieved by introducing synthetic scaffolds and growth factors.
The harmony of using scaffolds, stem cells and growth factors together has shown promise in a number of tissue engineering projects. Zhao et al[93] harvested adipose derived stem cells (ADSCs) and seeded them onto poly (lactic-co-glycolic acid) (PLGA) microparticles containing nerve growth factor (NGF). As a synthetic scaffold, PLGA has been shown to be safe in the urinary tract[94,95]. With periurethral injection into mice, this combination improved the LPP to 22.5 ± 6.1 cmH2O over treatments lacking either the PLGA or NGF. This result was explained by NGF prolonging the survival of ADSCs, enhancing the urethral muscle area on histology, and increased the density of neurofilaments supporting the sphincter lamina propria. This is the first iconic study where tissue engineering directly addresses the pathology underlying intrinsic sphincter defects. We propose a similar approach, where scaffolds and MSCs are injected not into the periurethral space, but directly into the urethral sphincter.
The number and variety of polymers synthesized for tissue engineering is rapidly expanding. A popular selection for research is PLGA[96], possessing elastic properties that adapt well to dynamic soft tissue structures. A similar material, poly (1,8-octanediol-co-citrate) (POC), is used by our group for urologic tissue engineering efforts. First established by the Ameer Research Lab[97], POC thin film (POCf) scaffolds are a highly reproducible elastomeric material[98]. The POCf allows for cell growth, cell infiltration, and for unimpaired exchange of oxygen and nutrient delivery. During polymerization of the scaffold, several aspects of construction can be customized to mimic the compliance, elasticity, and tensile strength measured through Young’s modulus. Equimolar amounts of citric acid and 1,8-octanediol are combined, melted and cooled to make a pre-polymer, and parameters such as temperature and time can be adjusted. Higher temperatures with short polymerization times produce dense films, while low temperatures and long polymerization times yield scaffolds that are less cross-linked. With these modifiable ester-bonding schemes, highly adaptable, labile and reproducible scaffolds can be created specifically for urinary tract tissue targets. Tailoring these parameters also reflects the degradation scheme of POCf, which degrades to nontoxic byproducts of CO2 and H2O via non-enzymatic hydrolysis.
Another feature of POC scaffolds, besides providing a highly conducive substrate for cell growth and proliferation, is the ability to deliver growth factors through a controlled release upon scaffold degradation[99]. During the polymerization of POC, small peptides including growth factors and cytokines may be chemically coupled to the scaffold and released upon surface erosion. POCf scaffolds modified with heparan sulfate to hold vascular endothelial growth factor (VEGF), fibroblast growth factor 2 and insulin-like growth factor were studied by Sharma et al[99] in a rat model. Heparan sulfate, a highly sulfated glycosaminoglycan, protects bound growth factors to prevent enzymatic degradation. Delivery of the pro-angiogenic growth factors upon break down of the scaffold led to increased vascular growth in vivo as compared to controls. The difference in results demonstrates that using POCf for a protracted but focused delivery of growth factors improves tissue healing.
The versatility of POCfs used in consonance with MSCs and growth factors offers the basis to potentially correct for sphincter deficiencies. For pregnancy related SUI, one pathway has been identified to be upregulated in response to vaginal distention and subsequent tissue damage[100]. The pathway involves hypoxia inducible factor-1α, a transcription factor stabilized in hypoxic conditions to induce expression of VEGF. This marker of tissue injury, and the resulting drive for angiogenesis, could potentially home stem cells to the site of injury. Studies by Cruz et al[101] have shown that pelvic injury via vaginal distension is a sufficient nidus for MSC homing to the urethra and levator ani. This phenomenon is speculated to be through chemokine ligand-7. Adding these chemokines to the POC delivery system could therefore boost the response of MSCs in tissue regeneration, and recruit circulating progenitors as well[102,103]. Progenitors stationed in the tissue, such as intrinsic satellite cells, may also be recruited to striated muscle reconstruction[104]. Direct and strong evidence exists that VEGF promotes the growth of myoblasts and increases capillary growth to the regenerating tissue. Interestingly, VEGF was capable of advancing the growth of myoblasts sourced even from older mice, where cells have less capacity to proliferate into functional tissue[105]. Growth factors like basic fibroblast growth factor (bFGF), hepatocyte growth factor and insulin-like growth factor have also contributed to muscle regeneration[106]. Characterization studies have also outlined a multitude of paracrine factors secreted by MSCs that are anti-apoptotic, immunomodulatory, anti-fibrotic and pro-angiogenic[107]. As previously confirmed, VEGF from MSCs, along with IL-6, MCP-1 and extracellular matrix components, assist in supporting angiogenesis, laying down extracellular matrix, and preventing apoptosis secondary to hypoxia[108].
The significance of bioactive compounds in regeneration was further strengthened by Choi et al[109]. In this study, plasmid DNA encoding bFGF was injected into rat periurethral submucosa via a PLGA synthetic delivery system. Results indicated that the levels of SM α-actin were elevated due to the bFGF, corresponding to a proliferation of tightly packed smooth muscle. Furthermore, contraction studies, conducted through electrical stimulation, showed a marked elevation in contractile properties for pDNA transfected urethras. Normal, continent urethras generated a contraction force of 36.4 ± 2.5 tension/mg of tissue, which is not dramatically different from the measured 32.3 ± 1.5 tension/mg tissue generated in pDNA/PLGA treated incontinent mice.
Three important concepts come out of this study: (1) that a sustained release of pDNA expressing bFGF through PLGA proved beneficial to regeneration; (2) the use of pDNA ensures that regeneration continues beyond the half-life and denaturation of biogenic compounds; and (3) since neither MSCs nor any other cell lines were not utilized in the injection, the improvement to symptoms can be specifically attributed to bFGF.
Integrating POCfs with MSCs and cytokines addresses the muscular aspect of sphincter regeneration. But a more comprehensive approach pays attention to the vascular and neural components as well. Seeding POC with progenitor cells from the bone marrow alongside MSCs could complete these components. Recent insights into CD34+ hematopoietic stem cells (HSCs), harvested from the same bone marrow origin as MSCs, points to a promising adjunct to MSCS. HSCs express von Willebrand Factor, vascular endothelial-cadherin and Flk-1[110-112]. These markers, in addition to CD133 and CD34[113], allow HSCs to be distinguished from MSCs and other primitive cells[114]. Placement of CD34+ HSCs onto compatible POCfs improved neovascularization and reduced fibrosis when injected into the site of injury[115].
Angiogenesis in the diseased urethral sphincter is beneficial for two reasons. First, it will nourish the proliferation and regeneration of MSCs into muscular components. Second, the blood supply will contribute to the vascular plexus that surrounds the urethral smooth muscle lumen. This plexus, when perfused, helps forms a tight seal of the mucosal surfaces, just as muscle contraction would.
Addressing the need for neural components in tissue engineering has been challenging. One study from our own group demonstrated the ability of MSCs combined with CD34+ cells to form muscular, vascular and even neural tissue in a rat bladder augmentation model[115]. Stem/progenitor cells were seeded onto POC scaffolds prior to implantation, which yielded well-organized fascicles of smooth muscle supported by collagen. CD34+ cells contributed greatly to the levels and distribution of blood vessels in MSCs/CD34+/POC constructs. Novel to this study was the detection of peripheral nerve regeneration from the surrounding healthy tissue. Stained with neuronal specific antibodies β III tubulin and synaptophysin, rat nerve bundles innervated the regenerated tissue significantly more in MSC/CD34+/POC grafts than in controls. The authors suggest that renewal of a blood supply to the area improves delivery of growth factors and cytokines promoting neuronal growth.
In the evolving field of tissue engineering, there has been an overwhelming trend towards therapy against the exact mechanism of disease causing SUI. Individual studies have lent credence to the importance of MSCs, CD34+ HSCs, scaffolds and growth factors in efforts to regenerate the urethral sphincter. A combination of these four components would create a plausible scenario in which to restore function in a structure as complex as the sphincter. Even with the advances in surgical slings, there still remains an inherent need to establish normal physiological function. Paired with POC scaffolds, we exploit the vast potential MSCs to differentiate into muscle, and hematopoietic precursors to proliferate into blood vessels in the presence of cytokines and growth factors. The indices of LPP readings from pre-operative urodynamic studies can be correlated to different levels of POCf elasticities, suited for a specific patient. Immunohistochemical and calcium release assays would support the MSC contractile properties as muscle regenerates, and nicotinic receptors targeted by α-bungarotoxin would illustrate the presence of neuronal fibers[116].
At least one study has observed the restoration of skeletal muscle and ganglionic elements from MSC injection into the rat urethral sphincter. Conducted by Kinebuchi et al[69], this study is a step in the right direction. However, follow up results did not confirm any improvement to LPP when compared to a control of cell free medium injection. The authors attribute the finding to inflammatory changes and to an insufficient bone marrow stem cells volume. In spite of this, the fallbacks can perhaps be accounted for by the absence of scaffolds and growth factors. Applications of this system expand beyond the treatment of SUI in adult women. Foremost, children born with neurogenic bladders secondary to myelomeningocele often have coexistent sphincter dysfunction. Likewise, post-prostatectomy men occasionally complain of incontinence as well. Patients with multiple sclerosis may have S2-S4 damage, leading to neuromuscular degeneration from the loss of incoming sensory nerve impulses and outgoing motor signals. The concept of urinary incontinence is similar to that of vesicoureteral reflux, so tissue engineering strategies provide an additional avenue to explore alongside ureteral reimplantation. To improve the symptoms of lower urinary tract symptoms in these patients, MSCs, HSCs, POC, and growth factors may one day supplement current surgical tactics.
P- Reviewers Gao ZL, Liu SH, Magne D, Yao CL S- Editor Song XX L- Editor A E- Editor Wu HL
| 1. | Kapoor DS, Meher S, Watkins L, Das M. Referral patterns for pelvic floor disorders. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1469-1472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 2. | Stothers L, Friedman B. Risk factors for the development of stress urinary incontinence in women. Curr Urol Rep. 2011;12:363-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 3. | Milsom I. Lower urinary tract symptoms in women. Curr Opin Urol. 2009;19:337-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 4. | Strasser H, Tiefenthaler M, Steinlechner M, Eder I, Bartsch G, Konwalinka G. Age dependent apoptosis and loss of rhabdosphincter cells. J Urol. 2000;164:1781-1785. [PubMed] |
| 5. | Brown SJ, Gartland D, Donath S, MacArthur C. Effects of prolonged second stage, method of birth, timing of caesarean section and other obstetric risk factors on postnatal urinary incontinence: an Australian nulliparous cohort study. BJOG. 2011;118:991-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Sajadi KP, Gill BC, Damaser MS. Neurogenic aspects of stress urinary incontinence. Curr Opin Obstet Gynecol. 2010;22:425-429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 7. | Sangsawang B, Sangsawang N. Stress urinary incontinence in pregnant women: a review of prevalence, pathophysiology, and treatment. Int Urogynecol J. 2013;24:901-912. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 150] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 8. | Aboushwareb T, McKenzie P, Wezel F, Southgate J, Badlani G. Is tissue engineering and biomaterials the future for lower urinary tract dysfunction (LUTD)/pelvic organ prolapse (POP)? Neurourol Urodyn. 2011;30:775-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 9. | Kim JC, Cho KJ. Current trends in the management of post-prostatectomy incontinence. Korean J Urol. 2012;53:511-518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 10. | Visser E, de Bock GH, Kollen BJ, Meijerink M, Berger MY, Dekker JH. Systematic screening for urinary incontinence in older women: who could benefit from it? Scand J Prim Health Care. 2012;30:21-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Harris SS, Link CL, Tennstedt SL, Kusek JW, McKinlay JB. Care seeking and treatment for urinary incontinence in a diverse population. J Urol. 2007;177:680-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 12. | Papanicolaou S, Hunskaar S, Lose G, Sykes D. Assessment of bothersomeness and impact on quality of life of urinary incontinence in women in France, Germany, Spain and the UK. BJU Int. 2005;96:831-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 82] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 13. | Irwin DE, Milsom I, Kopp Z, Abrams P, Cardozo L. Impact of overactive bladder symptoms on employment, social interactions and emotional well-being in six European countries. BJU Int. 2006;97:96-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 239] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 14. | Brown WJ, Miller YD. Too wet to exercise? Leaking urine as a barrier to physical activity in women. J Sci Med Sport. 2001;4:373-378. [PubMed] |
| 15. | Coyne KS, Kvasz M, Ireland AM, Milsom I, Kopp ZS, Chapple CR. Urinary incontinence and its relationship to mental health and health-related quality of life in men and women in Sweden, the United Kingdom, and the United States. Eur Urol. 2012;61:88-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 237] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 16. | Ko Y, Lin SJ, Salmon JW, Bron MS. The impact of urinary incontinence on quality of life of the elderly. Am J Manag Care. 2005;11:S103-S111. [PubMed] |
| 17. | Beeckman D, Verhaeghe S, Defloor T, Schoonhoven L, Vanderwee K. A 3-in-1 perineal care washcloth impregnated with dimethicone 3% versus water and pH neutral soap to prevent and treat incontinence-associated dermatitis: a randomized, controlled clinical trial. J Wound Ostomy Continence Nurs. 2011;38:627-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 63] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 18. | Tapia CI, Khalaf K, Berenson K, Globe D, Chancellor M, Carr LK. Health-related quality of life and economic impact of urinary incontinence due to detrusor overactivity associated with a neurologic condition: a systematic review. Health Qual Life Outcomes. 2013;11:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 19. | Tubaro A, Puccini F, De Nunzio C, Digesu GA, Elneil S, Gobbi C, Khullar V. The treatment of lower urinary tract symptoms in patients with multiple sclerosis: a systematic review. Curr Urol Rep. 2012;13:335-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 20. | Murphy AM, Bethoux F, Stough D, Goldman HB. Prevalence of stress urinary incontinence in women with multiple sclerosis. Int Neurourol J. 2012;16:86-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Petros PE, Ulmsten UI. An integral theory of female urinary incontinence. Experimental and clinical considerations. Acta Obstet Gynecol Scand Suppl. 1990;153:7-31. [PubMed] |
| 22. | Ashton-Miller JA, DeLancey JO. Functional anatomy of the female pelvic floor. Ann N Y Acad Sci. 2007;1101:266-296. [PubMed] |
| 23. | DeLancey JO. Structural support of the urethra as it relates to stress urinary incontinence: the hammock hypothesis. Am J Obstet Gynecol. 1994;170:1713-1720; discussion 1720-1723. [PubMed] |
| 24. | Wing RR, West DS, Grady D, Creasman JM, Richter HE, Myers D, Burgio KL, Franklin F, Gorin AA, Vittinghoff E. Effect of weight loss on urinary incontinence in overweight and obese women: results at 12 and 18 months. J Urol. 2010;184:1005-1010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 25. | Wallace SA, Roe B, Williams K, Palmer M. Bladder training for urinary incontinence in adults. Cochrane Database Syst Rev. 2004;CD001308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 121] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 26. | Shamliyan TA, Kane RL, Wyman J, Wilt TJ. Systematic review: randomized, controlled trials of nonsurgical treatments for urinary incontinence in women. Ann Intern Med. 2008;148:459-473. [PubMed] |
| 27. | Saks EK, Arya LA. Pharmacologic management of urinary incontinence, voiding dysfunction, and overactive bladder. Obstet Gynecol Clin North Am. 2009;36:493-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 28. | Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2010;CD005654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 153] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 29. | Smith AL, Wein AJ. Urinary incontinence: pharmacotherapy options. Ann Med. 2011;43:461-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Mariappan P, Ballantyne Z, N’Dow JM, Alhasso AA. Serotonin and noradrenaline reuptake inhibitors (SNRI) for stress urinary incontinence in adults. Cochrane Database Syst Rev. 2005;CD004742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Li J, Yang L, Pu C, Tang Y, Yun H, Han P. The role of duloxetine in stress urinary incontinence: a systematic review and meta-analysis. Int Urol Nephrol. 2013;45:679-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 32. | Deepak P, Kumar TN, Sen TK. Evaluation of efficacy of duloxetine in stress urinary incontinence in women. Indian J Pharmacol. 2011;43:176-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 33. | Herbison P, Hay-Smith J, Ellis G, Moore K. Effectiveness of anticholinergic drugs compared with placebo in the treatment of overactive bladder: systematic review. BMJ. 2003;326:841-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 208] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 34. | Weber AM, Walters MD. Cost-effectiveness of urodynamic testing before surgery for women with pelvic organ prolapse and stress urinary incontinence. Am J Obstet Gynecol. 2000;183:1338-1346; discussion 1346-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 35. | Weber AM, Taylor RJ, Wei JT, Lemack G, Piedmonte MR, Walters MD. The cost-effectiveness of preoperative testing (basic office assessment vs. urodynamics) for stress urinary incontinence in women. BJU Int. 2002;89:356-363. [PubMed] |
| 36. | Koonings PP, Bergman A, Ballard CA. Low urethral pressure and stress urinary incontinence in women: risk factor for failed retropubic surgical procedure. Urology. 1990;36:245-248. [PubMed] |
| 37. | Jonsson Funk M, Levin PJ, Wu JM. Trends in the surgical management of stress urinary incontinence. Obstet Gynecol. 2012;119:845-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 117] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 38. | Ogah J, Cody JD, Rogerson L. Minimally invasive synthetic suburethral sling operations for stress urinary incontinence in women. Cochrane Database Syst Rev. 2009;CD006375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 39. | Sarlos D, Kuronen M, Schaer GN. How does tension-free vaginal tape correct stress incontinence? investigation by perineal ultrasound. Int Urogynecol J Pelvic Floor Dysfunct. 2003;14:395-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 39] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 40. | Richter HE, Albo ME, Zyczynski HM, Kenton K, Norton PA, Sirls LT, Kraus SR, Chai TC, Lemack GE, Dandreo KJ. Retropubic versus transobturator midurethral slings for stress incontinence. N Engl J Med. 2010;362:2066-2076. [PubMed] |
| 41. | Schierlitz L, Dwyer PL, Rosamilia A, Murray C, Thomas E, De Souza A, Hiscock R. Three-year follow-up of tension-free vaginal tape compared with transobturator tape in women with stress urinary incontinence and intrinsic sphincter deficiency. Obstet Gynecol. 2012;119:321-327. [PubMed] |
| 42. | Araco F, Gravante G, Dati S, Bulzomi’ V, Sesti F, Piccione E. Results 1 year after the Reemex system was applied for the treatment of stress urinary incontinence caused by intrinsic sphincter deficiency. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:783-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | Feki A, Faltin DL, Lei T, Dubuisson JB, Jacob S, Irion O. Sphincter incontinence: is regenerative medicine the best alternative to restore urinary or anal sphincter function? Int J Biochem Cell Biol. 2007;39:678-684. [PubMed] |
| 44. | Lapitan MC, Cody DJ, Grant AM. Open retropubic colposuspension for urinary incontinence in women. Cochrane Database Syst Rev. 2005;CD002912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 45. | Kammerer-Doak DN, Cornella JL, Magrina JF, Stanhope CR, Smilack J. Osteitis pubis after Marshall-Marchetti-Krantz urethropexy: a pubic osteomyelitis. Am J Obstet Gynecol. 1998;179:586-590. [PubMed] |
| 46. | Jankowski R, Pruchnic R, Hiles M, Chancellor MB. Advances toward tissue engineering for the treatment of stress urinary incontinence. Rev Urol. 2004;6:51-57. [PubMed] |
| 47. | VandeVord PJ, Broadrick KM, Krishnamurthy B, Singla AK. A comparative study evaluating the in vivo incorporation of biological sling materials. Urology. 2010;75:1228-1233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | Zoorob D, Karram M. Role of autologous bladder-neck slings: a urogynecology perspective. Urol Clin North Am. 2012;39:311-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 49. | Carbone JM, Kavaler E, Hu JC, Raz S. Pubovaginal sling using cadaveric fascia and bone anchors: disappointing early results. J Urol. 2001;165:1605-1611. [PubMed] |
| 50. | Subak LL, Brubaker L, Chai TC, Creasman JM, Diokno AC, Goode PS, Kraus SR, Kusek JW, Leng WW, Lukacz ES. High costs of urinary incontinence among women electing surgery to treat stress incontinence. Obstet Gynecol. 2008;111:899-907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 51. | Ugurlucan FG, Erkan HA, Onal M, Yalcin O. Randomized trial of graft materials in transobturator tape operation: biological versus synthetic. Int Urogynecol J. 2013;24:1315-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 52. | Brubaker L, Norton PA, Albo ME, Chai TC, Dandreo KJ, Lloyd KL, Lowder JL, Sirls LT, Lemack GE, Arisco AM. Adverse events over two years after retropubic or transobturator midurethral sling surgery: findings from the Trial of Midurethral Slings (TOMUS) study. Am J Obstet Gynecol. 2011;205:498.e1-498.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 135] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 53. | Filocamo MT, Serati M, Frumenzio E, Li arzi V, Cattoni E, Champagne A, Salvatore S, Nicita G, Costantini E. The impact of mid-urethral slings for the treatment of urodynamic stress incontinence on female sexual function: a multicenter prospective study. J Sex Med. 2011;8:2002-2008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 54. | De Souza A, Dwyer PL, Rosamilia A, Hiscock R, Lim YN, Murray C, Thomas E, Conway C, Schierlitz L. Sexual function following retropubic TVT and transobturator Monarc sling in women with intrinsic sphincter deficiency: a multicentre prospective study. Int Urogynecol J. 2012;23:153-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 55. | Jha S, Ammenbal M, Metwally M. Impact of incontinence surgery on sexual function: a systematic review and meta-analysis. J Sex Med. 2012;9:34-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 79] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 56. | Schulz JA, Nager CW, Stanton SL, Baessler K. Bulking agents for stress urinary incontinence: short-term results and complications in a randomized comparison of periurethral and transurethral injections. Int Urogynecol J Pelvic Floor Dysfunct. 2004;15:261-265. [PubMed] |
| 57. | Kuhn A, Stadlmayr W, Lengsfeld D, Mueller MD. Where should bulking agents for female urodynamic stress incontinence be injected? Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:817-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 58. | Zou XH, Zhi YL, Chen X, Jin HM, Wang LL, Jiang YZ, Yin Z, Ouyang HW. Mesenchymal stem cell seeded knitted silk sling for the treatment of stress urinary incontinence. Biomaterials. 2010;31:4872-4879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 59. | Hong SH, Piao S, Kim IG, Lee JY, Cho HJ, Kim SW, Hwang TK, Lee JY. Comparison of three types of stress urinary incontinence rat models: electrocauterization, pudendal denervation, and vaginal distension. Urology. 2013;81:465.e1-465.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 60. | Hussain M, Greenwell TJ, Venn SN, Mundy AR. The current role of the artificial urinary sphincter for the treatment of urinary incontinence. J Urol. 2005;174:418-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 89] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 61. | Lipp A, Shaw C, Glavind K. Mechanical devices for urinary incontinence in women. Cochrane Database Syst Rev. 2011;CD001756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 62. | Sèbe P, Doucet C, Cornu JN, Ciofu C, Costa P, de Medina SG, Pinset C, Haab F. Intrasphincteric injections of autologous muscular cells in women with refractory stress urinary incontinence: a prospective study. Int Urogynecol J. 2011;22:183-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 67] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 63. | Lin G, Wang G, Banie L, Ning H, Shindel AW, Fandel TM, Lue TF, Lin CS. Treatment of stress urinary incontinence with adipose tissue-derived stem cells. Cytotherapy. 2010;12:88-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 139] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 64. | Jack GS, Almeida FG, Zhang R, Alfonso ZC, Zuk PA, Rodríguez LV. Processed lipoaspirate cells for tissue engineering of the lower urinary tract: implications for the treatment of stress urinary incontinence and bladder reconstruction. J Urol. 2005;174:2041-2045. [PubMed] |
| 65. | Obinata D, Matsumoto T, Ikado Y, Sakuma T, Kano K, Fukuda N, Yamaguchi K, Mugishima H, Takahashi S. Transplantation of mature adipocyte-derived dedifferentiated fat (DFAT) cells improves urethral sphincter contractility in a rat model. Int J Urol. 2011;18:827-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 66. | Chun SY, Cho DH, Chae SY, Choi KH, Lim HJ, Yoon GS, Kim BS, Kim BW, Yoo JJ, Kwon TG. Human amniotic fluid stem cell-derived muscle progenitor cell therapy for stress urinary incontinence. J Korean Med Sci. 2012;27:1300-1307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 67. | Kwon D, Kim Y, Pruchnic R, Jankowski R, Usiene I, de Miguel F, Huard J, Chancellor MB. Periurethral cellular injection: comparison of muscle-derived progenitor cells and fibroblasts with regard to efficacy and tissue contractility in an animal model of stress urinary incontinence. Urology. 2006;68:449-454. [PubMed] |
| 68. | Imamura T, Ishizuka O, Kinebuchi Y, Kurizaki Y, Nakayama T, Ishikawa M, Nishizawa O. Implantation of autologous bone-marrow-derived cells reconstructs functional urethral sphincters in rabbits. Tissue Eng Part A. 2011;17:1069-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 69. | Kinebuchi Y, Aizawa N, Imamura T, Ishizuka O, Igawa Y, Nishizawa O. Autologous bone-marrow-derived mesenchymal stem cell transplantation into injured rat urethral sphincter. Int J Urol. 2010;17:359-368. [PubMed] |
| 70. | Mitterberger M, Marksteiner R, Margreiter E, Pinggera GM, Frauscher F, Ulmer H, Fussenegger M, Bartsch G, Strasser H. Myoblast and fibroblast therapy for post-prostatectomy urinary incontinence: 1-year followup of 63 patients. J Urol. 2008;179:226-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 76] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 71. | Gerullis H, Eimer C, Georgas E, Homburger M, El-Baz AG, Wishahi M, Borós M, Ecke TH, Otto T. Muscle-derived cells for treatment of iatrogenic sphincter damage and urinary incontinence in men. ScientificWorldJournal. 2012;2012:898535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 72. | Corcos J, Loutochin O, Campeau L, Eliopoulos N, Bouchentouf M, Blok B, Galipeau J. Bone marrow mesenchymal stromal cell therapy for external urethral sphincter restoration in a rat model of stress urinary incontinence. Neurourol Urodyn. 2011;30:447-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 73. | Kassem M, Kristiansen M, Abdallah BM. Mesenchymal stem cells: cell biology and potential use in therapy. Basic Clin Pharmacol Toxicol. 2004;95:209-214. [PubMed] |
| 74. | Drewa T, Adamowicz J, Sharma A. Tissue engineering for the oncologic urinary bladder. Nat Rev Urol. 2012;9:561-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 75. | Dayanc M, Kibar Y, Ural AU, Onguru O, Yildiz O, Irkilata HC, Avcu F, Soner BC, Ulku C, Seyrek M. The histopathologic, pharmacologic and urodynamic results of mesenchymal stem cell’s injection into the decompensated rabbit’s bladder. Stem Cell Rev. 2012;8:1245-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 76. | Sharma AK, Bury MI, Marks AJ, Fuller NJ, Meisner JW, Tapaskar N, Halliday LC, Matoka DJ, Cheng EY. A nonhuman primate model for urinary bladder regeneration using autologous sources of bone marrow-derived mesenchymal stem cells. Stem Cells. 2011;29:241-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 77. | Woo LL, Tanaka ST, Anumanthan G, Pope JC, Thomas JC, Adams MC, Brock JW, Bhowmick NA. Mesenchymal stem cell recruitment and improved bladder function after bladder outlet obstruction: preliminary data. J Urol. 2011;185:1132-1138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 78. | Drzewiecki BA, Thomas JC, Tanaka ST. Bone marrow-derived mesenchymal stem cells: current and future applications in the urinary bladder. Stem Cells Int. 2011;2010:765167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 79. | Lane FL, Jacobs S. Stem cells in gynecology. Am J Obstet Gynecol. 2012;207:149-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 80. | Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11055] [Cited by in RCA: 12686] [Article Influence: 704.8] [Reference Citation Analysis (2)] |
| 81. | Ryan JM, Barry FP, Murphy JM, Mahon BP. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond). 2005;2:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 598] [Cited by in RCA: 645] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 82. | Tse WT, Pendleton JD, Beyer WM, Egalka MC, Guinan EC. Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation. 2003;75:389-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1141] [Cited by in RCA: 1088] [Article Influence: 49.5] [Reference Citation Analysis (0)] |
| 83. | Sekiya I, Larson BL, Smith JR, Pochampally R, Cui JG, Prockop DJ. Expansion of human adult stem cells from bone marrow stroma: conditions that maximize the yields of early progenitors and evaluate their quality. Stem Cells. 2002;20:530-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 695] [Article Influence: 31.6] [Reference Citation Analysis (0)] |
| 84. | Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143-147. [PubMed] |
| 85. | Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105:1815-1822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3271] [Cited by in RCA: 3279] [Article Influence: 156.1] [Reference Citation Analysis (0)] |
| 86. | Ishikane S, Ohnishi S, Yamahara K, Sada M, Harada K, Mishima K, Iwasaki K, Fujiwara M, Kitamura S, Nagaya N. Allogeneic injection of fetal membrane-derived mesenchymal stem cells induces therapeutic angiogenesis in a rat model of hind limb ischemia. Stem Cells. 2008;26:2625-2633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 83] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 87. | Anumanthan G, Makari JH, Honea L, Thomas JC, Wills ML, Bhowmick NA, Adams MC, Hayward SW, Matusik RJ, Brock JW. Directed differentiation of bone marrow derived mesenchymal stem cells into bladder urothelium. J Urol. 2008;180:1778-1783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 49] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 88. | Tian H, Bharadwaj S, Liu Y, Ma PX, Atala A, Zhang Y. Differentiation of human bone marrow mesenchymal stem cells into bladder cells: potential for urological tissue engineering. Tissue Eng Part A. 2010;16:1769-1779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 110] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 89. | Surcel C, Savu C, Chibelean C, Iordache A, Mirvald C, Sinescu I. Comparative analysis of different surgical procedures for female stress urinary incontinence. Is stem cell implantation the future? Rom J Morphol Embryol. 2012;53:151-154. [PubMed] |
| 90. | Carr LK, Steele D, Steele S, Wagner D, Pruchnic R, Jankowski R, Erickson J, Huard J, Chancellor MB. 1-year follow-up of autologous muscle-derived stem cell injection pilot study to treat stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:881-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 173] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 91. | Peters K, Kaufman M, Dmochowski R, Carr L, Herschorn S, Fischer M, Sirls L, Nagaraju P, Biller D, Ward R. 1340 Autologous muscle derived cell therapy for the treatment of female stress urinary incontinence: A multi-center experience. The Journal of Urology. 2011;185:e535-e536. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 92. | Beauchamp JR, Morgan JE, Pagel CN, Partridge TA. Dynamics of myoblast transplantation reveal a discrete minority of precursors with stem cell-like properties as the myogenic source. J Cell Biol. 1999;144:1113-1122. [PubMed] |
| 93. | Zhao W, Zhang C, Jin C, Zhang Z, Kong D, Xu W, Xiu Y. Periurethral injection of autologous adipose-derived stem cells with controlled-release nerve growth factor for the treatment of stress urinary incontinence in a rat model. Eur Urol. 2011;59:155-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 72] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 94. | Cho ER, Kang SW, Park HJ, Cho YS, Lee YS, Kim JC, Kim BS. Submucosal injection of poly(lactic-co-glycolic acid) microspheres in rabbit bladder as a potential treatment for urinary incontinence and vesicoureteral reflux: preliminary results. J Biomater Sci Polym Ed. 2005;16:1109-1120. [PubMed] |
| 95. | Oh SH, Lee JY, Ghil SH, Lee SS, Yuk SH, Lee JH. PCL microparticle-dispersed PLGA solution as a potential injectable urethral bulking agent. Biomaterials. 2006;27:1936-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 96. | Selim M, Bullock AJ, Blackwood KA, Chapple CR, MacNeil S. Developing biodegradable scaffolds for tissue engineering of the urethra. BJU Int. 2011;107:296-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 97. | Webb AR, Kumar , VA , Ameer GA. Biodegradable poly (diol citrate) nanocomposite elastomers for soft tissue engineering. J Mater Chem. 2007;17:900-906. [RCA] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 98. | Sharma AK, Hota PV, Matoka DJ, Fuller NJ, Jandali D, Thaker H, Ameer GA, Cheng EY. Urinary bladder smooth muscle regeneration utilizing bone marrow derived mesenchymal stem cell seeded elastomeric poly(1,8-octanediol-co-citrate) based thin films. Biomaterials. 2010;31:6207-6217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 99. | Sharma AK, Bury MI, Fuller NJ, Rozkiewicz DI, Hota PV, Kollhoff DM, Webber MJ, Tapaskar N, Meisner JW, Lariviere PJ. Growth factor release from a chemically modified elastomeric poly(1,8-octanediol-co-citrate) thin film promotes angiogenesis in vivo. J Biomed Mater Res A. 2012;100:561-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 100. | Lenis AT, Kuang M, Woo LL, Hijaz A, Penn MS, Butler RS, Rackley R, Damaser MS, Wood HM. Impact of parturition on chemokine homing factor expression in the vaginal distention model of stress urinary incontinence. J Urol. 2013;189:1588-1594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 101. | Cruz M, Dissaranan C, Cotleur A, Kiedrowski M, Penn M, Damaser M. Pelvic organ distribution of mesenchymal stem cells injected intravenously after simulated childbirth injury in female rats. Obstet Gynecol Int. 2012;2012:612946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 102. | Papayannopoulou T, Priestley GV, Bonig H, Nakamoto B. The role of G-protein signaling in hematopoietic stem/progenitor cell mobilization. Blood. 2003;101:4739-4747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 103. | Dissaranan C, Cruz M, Gill B, Salcedo L, Cotleur A, Mendieta R, Balog B, Kiedrowski M, Penn M, Goldman H. 176 intravenous mesenchymal stem cells home to the urethra and facilitate recovery from stress urinary incontinence after childbirth injury via local secretion of paracrine factors. Cell Transplant. 2013;Jul 17; [Epub ahead of print]. [PubMed] |
| 104. | Yiou R, Lefaucheur JP, Atala A. The regeneration process of the striated urethral sphincter involves activation of intrinsic satellite cells. Anat Embryol (Berl). 2003;206:429-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 105. | Delo DM, Eberli D, Williams JK, Andersson KE, Atala A, Soker S. Angiogenic gene modification of skeletal muscle cells to compensate for ageing-induced decline in bioengineered functional muscle tissue. BJU Int. 2008;102:878-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 106. | Takahashi S, Chen Q, Ogushi T, Fujimura T, Kumagai J, Matsumoto S, Hijikata S, Tabata Y, Kitamura T. Periurethral injection of sustained release basic fibroblast growth factor improves sphincteric contractility of the rat urethra denervated by botulinum-a toxin. J Urol. 2006;176:819-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 107. | Meirelles Lda S, Fontes AM, Covas DT, Caplan AI. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009;20:419-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 951] [Cited by in RCA: 1012] [Article Influence: 63.3] [Reference Citation Analysis (0)] |
| 108. | Hung SC, Pochampally RR, Chen SC, Hsu SC, Prockop DJ. Angiogenic effects of human multipotent stromal cell conditioned medium activate the PI3K-Akt pathway in hypoxic endothelial cells to inhibit apoptosis, increase survival, and stimulate angiogenesis. Stem Cells. 2007;25:2363-2370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 350] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 109. | Choi SJ, Oh SH, Kim IG, Chun SY, Lee JY, Lee JH. Functional recovery of urethra by plasmid DNA-loaded injectable agent for the treatment of urinary incontinence. Biomaterials. 2013;34:4766-4776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 110. | Baum CM, Weissman IL, Tsukamoto AS, Buckle AM, Peault B. Isolation of a candidate human hematopoietic stem-cell population. Proc Natl Acad Sci USA. 1992;89:2804-2808. [PubMed] |
| 111. | Yang J, Ii M, Kamei N, Alev C, Kwon SM, Kawamoto A, Akimaru H, Masuda H, Sawa Y, Asahara T. CD34+ cells represent highly functional endothelial progenitor cells in murine bone marrow. PLoS One. 2011;6:e20219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 101] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 112. | Asahara T, Isner JM. Endothelial progenitor cells for vascular regeneration. J Hematother Stem Cell Res. 2002;11:171-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 94] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 113. | Yin AH, Miraglia S, Zanjani ED, Almeida-Porada G, Ogawa M, Leary AG, Olweus J, Kearney J, Buck DW. AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood. 1997;90:5002-5012. [PubMed] |
| 114. | Ghajar CM, Blevins KS, Hughes CC, George SC, Putnam AJ. Mesenchymal stem cells enhance angiogenesis in mechanically viable prevascularized tissues via early matrix metalloproteinase upregulation. Tissue Eng. 2006;12:2875-2888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 179] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 115. | Sharma AK, Bury MI, Fuller NJ, Marks AJ, Kollhoff DM, Rao MV, Hota PV, Matoka DJ, Edassery SL, Thaker H. Cotransplantation with specific populations of spina bifida bone marrow stem/progenitor cells enhances urinary bladder regeneration. Proc Natl Acad Sci USA. 2013;110:4003-4008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 116. | Biérinx AS, Sebille A. The urethral striated sphincter in adult male rat. Anat Embryol (Berl). 2006;211:435-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 117. | Lee JY, Cannon TW, Pruchnic R, Fraser MO, Huard J, Chancellor MB. The effects of periurethral muscle-derived stem cell injection on leak point pressure in a rat model of stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2003;14:31-7; discussion 37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 131] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 118. | Fu Q, Song XF, Liao GL, Deng CL, Cui L. Myoblasts differentiated from adipose-derived stem cells to treat stress urinary incontinence. Urology. 2010;75:718-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 119. | Lim JJ, Jang JB, Kim JY, Moon SH, Lee CN, Lee KJ. Human umbilical cord blood mononuclear cell transplantation in rats with intrinsic sphincter deficiency. J Korean Med Sci. 2010;25:663-670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 120. | Kim SO, Na HS, Kwon D, Joo SY, Kim HS, Ahn Y. Bone-marrow-derived mesenchymal stem cell transplantation enhances closing pressure and leak point pressure in a female urinary incontinence rat model. Urol Int. 2011;86:110-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 121. | Watanabe T, Maruyama S, Yamamoto T, Kamo I, Yasuda K, Saka Y, Ozaki T, Yuzawa Y, Matsuo S, Gotoh M. Increased urethral resistance by periurethral injection of low serum cultured adipose-derived mesenchymal stromal cells in rats. Int J Urol. 2011;18:659-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 122. | Kim BS, Chun SY, Lee JK, Lim HJ, Bae JS, Chung HY, Atala A, Soker S, Yoo JJ, Kwon TG. Human amniotic fluid stem cell injection therapy for urethral sphincter regeneration in an animal model. BMC Med. 2012;10:94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 123. | Li GY, Zhou F, Gong YQ, Cui WS, Yuan YM, Song WD, Xin H, Liu T, Li WR, Gao ZZ. Activation of VEGF and ERK1/2 and improvement of urethral function by adipose-derived stem cells in a rat stress urinary incontinence model. Urology. 2012;80:953.e1-953.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 124. | Lecoeur C, Swieb S, Zini L, Rivière C, Combrisson H, Ghérardi R, Abbou C, Yiou R. Intraurethral transfer of satellite cells by myofiber implants results in the formation of innervated myotubes exerting tonic contractions. J Urol. 2007;178:332-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 125. | Mitterberger M, Marksteiner R, Margreiter E, Pinggera GM, Colleselli D, Frauscher F, Ulmer H, Fussenegger M, Bartsch G, Strasser H. Autologous myoblasts and fibroblasts for female stress incontinence: a 1-year follow-up in 123 patients. BJU Int. 2007;100:1081-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 126. | Mitterberger M, Pinggera GM, Marksteiner R, Margreiter E, Fussenegger M, Frauscher F, Ulmer H, Hering S, Bartsch G, Strasser H. Adult stem cell therapy of female stress urinary incontinence. Eur Urol. 2008;53:169-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 91] [Article Influence: 5.1] [Reference Citation Analysis (0)] |