Published online Dec 26, 2020. doi: 10.4252/wjsc.v12.i12.1623
Peer-review started: May 31, 2020
First decision: August 22, 2020
Revised: September 20, 2020
Accepted: October 12, 2020
Article in press: October 12, 2020
Published online: December 26, 2020
Processing time: 210 Days and 1.6 Hours
Autoimmune hepatitis is a serious autoimmune liver disease that threatens human health worldwide, which emphasizes the urgent need to identify novel treatments. Stem cells from human exfoliated deciduous teeth (SHED), which are easy to obtain in a non-invasive manner, show pronounced proliferative and immunomodulatory capacities.
To investigate the protective effects of SHED on concanavalin A (ConA)-induced hepatitis in mice, and to elucidate the associated regulatory mechanisms.
We used a ConA-induced acute hepatitis mouse model and an in vitro co-culture system to study the protective effects of SHED on ConA-induced autoimmune hepatitis, as well as the associated underlying mechanisms.
SHED infusion could prevent aberrant histopathological liver architecture caused by ConA-induced infiltration of CD3+, CD4+, tumor necrosis-alpha+, and interferon-gamma+ inflammatory cells. Alanine aminotransferase and aspartate aminotransferase were significantly elevated in hepatitis mice. SHED infusion could therefore block ConA-induced alanine aminotransferase and aspartate aminotransferase elevations. Mechanistically, ConA upregulated tumor necrosis-alpha and interferon-gamma expression, which was activated by the nuclear factor-kappa B pathway to induce hepatocyte apoptosis, resulting in acute liver injury. SHED administration protected hepatocytes from ConA-induced apoptosis.
SHED alleviates ConA-induced acute liver injury via inhibition of hepatocyte apoptosis mediated by the nuclear factor-kappa B pathway. Our findings could provide a potential treatment strategy for hepatitis.
Core Tip: Autoimmune hepatitis poses an enhancing global burden, underscoring the need to identify novel treatments. To determine the effects of stem cells from human exfoliated deciduous teeth (SHED) on concanavalin A (ConA)-induced autoimmune hepatitis, we pretreated ConA-induced hepatitis mice with SHED. SHED blocked increased expression of alanine aminotransferase and aspartate aminotransferase, and prevented aberrant histopathological liver architecture due to infiltration of CD3+, CD4+, tumor necrosis-alpha+, and interferon-gamma+ inflammatory cells. ConA upregulated nuclear factor-kappa B-activated tumor necrosis-alpha and interferon-gamma expression, inducing hepatocyte apoptosis and acute liver injury. SHED administration protected hepatocytes from apoptosis, providing a potential treatment for hepatitis and acute hepatic injury.
- Citation: Zhou YK, Zhu LS, Huang HM, Cui SJ, Zhang T, Zhou YH, Yang RL. Stem cells from human exfoliated deciduous teeth ameliorate concanavalin A-induced autoimmune hepatitis by protecting hepatocytes from apoptosis. World J Stem Cells 2020; 12(12): 1623-1639
- URL: https://www.wjgnet.com/1948-0210/full/v12/i12/1623.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i12.1623
Autoimmune hepatitis (AIH) is an immune-mediated liver inflammatory disease that has a variety of clinical manifestations, including occult onset without symptoms, acute onset, and even acute liver failure[1]. The course of AIH can undulate, which may begin as acute hepatitis and ultimately lead to fibrosis, cirrhosis, and liver transplantation, eventually leading to death in a short period of time. The therapeutic aim of AIH is to alleviate the state of liver inflammation and prevent fibrosis. Generally, AIH treatment requires long-term or even lifelong use of im-munosuppressants, which may cause serious side effects. Ten to twenty percent of AIH patients are insensitive to immunosuppressant treatment, cannot be well controlled even after treatment, and will ultimately develop end-stage cirrhosis[2,3]. AIH is becoming a worldwide health problem, which highlights the need to explore alternative treatment strategies.
As mesenchymal stem cells (MSCs) have multilineage differentiation and immunomodulatory properties, stem cell therapies for inflammation-related diseases have recently attracted more attention. It has been reported that MSC infusion could alleviate a variety of diseases, such as autoimmune disease, nerve injury, spinal cord injury, and diabetes[4]. Previous studies have shown that injection of bone marrow (BM) MSCs, adipose MSCs, and tonsil-derived stem cells can alleviate concanavalin A (ConA)-induced AIH in mice[5-7]. However, the limited sources of BMMSCs have greatly narrowed their therapeutic applications. Stem cells were found in the pulp of human exfoliated deciduous teeth (SHED), which showed more pronounced proliferative and immunomodulatory capacity compared to BMMSCs[8,9]. Yamaza et al[10] also reported that spleen transplantation of SHED significantly improved hepatic dysfunction, and that these stem cells directly transformed into hepatocytes in carbon tetrachloride-treated mice without cell fusion, thus suggesting that SHED may provide a feasible treatment for liver regeneration. SHED was also reported to differentiate into hepatocytes in vitro, indicating that SHED may represent the appropriate stem cells to treat hepatitis[11]. In light of the above effects related to SHED, we questioned whether these non-traumatically collected stem cells could block the occurrence of AIH.
ConA, from the jack bean Canavalia ensiformis, binds to sugar residues on the surface of multiple cell types[12,13]. It has been reported that ConA-induced AIH has the same pathogenicity and histopathological characteristics as AIH. ConA can induce hepatocyte damage by activating inflammatory cytokines produced by macrophages and T cells. It has been reported that inflammatory cytokines, such as interleukin (IL)-2, IL-6, tumor necrosis-alpha (TNF-α), and interferon-gamma (IFN-γ), play important roles in the development of acute hepatitis[14-16]. Therefore, previous studies used drugs to downregulate the relevant pro-inflammatory factors through different pathways (TRADD/TRAF2 JAK2/STAT, reactive oxygen species [ROS], PI3K, and JNK), so as to reduce liver damage and treat AIH[17-19]. However, the effects of these drugs are limited by some phenomena, and its exact mechanism requires further study. SHED have demonstrated profound immunomodulatory and anti-inflammatory abilities that inhibit T lymphocyte proliferation both in vitro and in vivo[20]. Whether SHED infusion could block ConA-induced acute inflammation needs to be further investigated.
Here, we explored the effects of SHED on ConA-induced AIH in mice, as well as the potential underlying mechanisms. Our results revealed that SHED infusion alleviated ConA-induced acute liver injury via hepatocyte apoptotic inhibition mediated by the nuclear factor-kappa B (NF-κB) pathway.
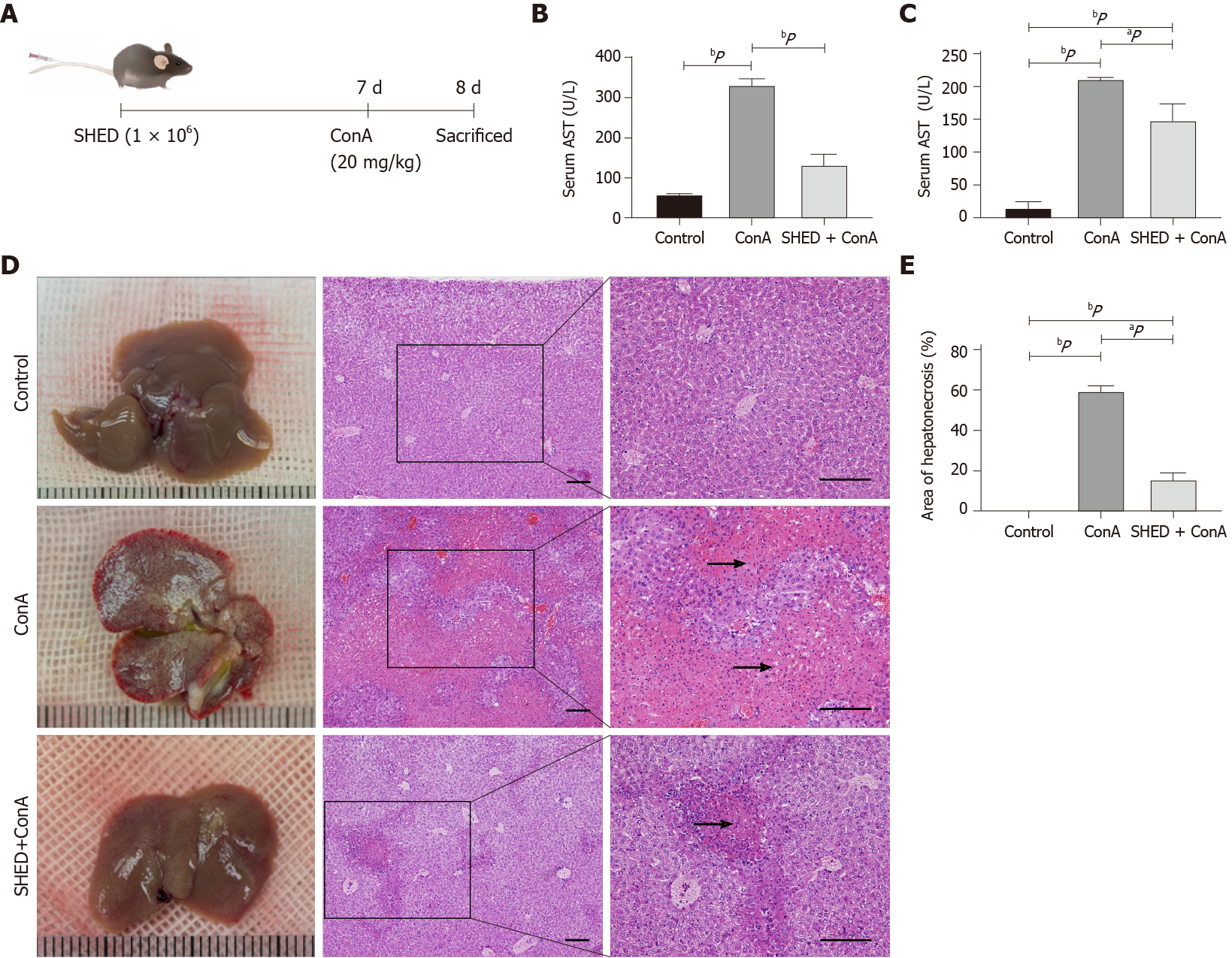
Male Balb/c mice (6-8 wk old; 23 ± 2 g) were purchased from Vital River Laboratory Animal Technology (Beijing, China). Mice were randomly assigned to a control group (n = 10; intravenous injections with saline solution), a ConA group (n = 10; intravenous injections with 20 mg/kg ConA at 20 mg/kg body weight; Sigma-Aldrich, Street Louis, MO, United States), and an SHED + ConA group (n = 10; intravenous injections with SHED cells (1 × 106) 7 d prior to ConA challenge). After injections of ConA for 24 h, the mice were sacrificed for further analysis (Figure 1A). Protocols were approved by the Animal Care and Use Committee of the Health Science Center, Peking University (No. 2015-186).
Mice (n = 3) were intravenously injected with prepared DiR-labeled SHED cells in phosphate-buffered saline (PBS) and scanned at 1 d, 3 d, and 7 d post-injection using an Interactive Video Information System Lumina Series III in vivo Imaging System (Caliper Life Sciences, Perkin Elmer, United States). Ex vivo imaging was carried out immediately afterward by imaging excised major organs (heart, lung, liver, spleen, and kidney).
SHED cells were labeled with 5-(and-6)-carboxy fluorescein diacetate succinimidyl ester (CFSE, Molecular Probes Biotec. Co) and diluted to 1 × 106/mL for transplantation. The liver and spleen were flash-frozen at optimum cutting temperature at 1 d, 3 d, and 7 d post-injection. Nuclear staining was performed with diamidine phenyl indoles (DAPI) mounting medium (Vector Lab, Burlingame, CA, United States). The positions of CFSE-labeled SHED cells could be traced using a confocal laser scanning microscope (Zeiss, Thornwood, NY, United States).
Mouse serum samples were collected for the analysis of inflammatory cytokines, alanine aminotransferase (ALT), and aspartate aminotransferase (AST). ALT and AST levels were evaluated with commercial kits, according to the manufacturer's instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). TNF-α and IFN-γ levels were measured using enzyme-linked immunosorbent assay kits, according to the manufacturer's instructions (eBioscience, Inc., San Diego, CA, United States)[17].
To detect the levels of CD3+/CD4+ T cells and Th1 cells in mouse blood, anti-mouse CD3, CD4, and IFN-γ antibodies were included according to the manufacturer’s instructions. Antibodies against cell surface markers were added for 15 min at room temperature. After lysing red blood cells, leukocytes were fixed and permeabilized by using the Cytofix/Cytoperm Fixation/Permeabilization Kit as described above. The cell pellets were resuspended, fluorescein isothiocyanate-conjugated anti-mouse IFN-γ was added and incubated at 4 °C for 30 min, and flow cytometry (FACS) analysis was performed according to the manufacturer’s instructions.
For immunohistochemistry, sections were dewaxed with xylene, treated with gradient concentrations of ethanol, washed with deionized water, and heated in a microwave in 10 mmol/L sodium citrate (pH 6). Then, 0.05% Tween 20 was added and samples were blocked for 1 h with 5% bovine serum albumin and 0.1% Triton X-100 in PBS. Anti-CD3 (1:100), anti-CD4 (1:100), anti-total caspase3 (1:100), anti-TNF-α (1:100), and anti-IFN-γ (1:100) antibodies were added and incubated overnight at 4 °C. After washing three times, sections were incubated with secondary antibodies at room temperature for 1 h, and then observed under a microscope (Leica, Wetzlar, Germany). Positive areas were measured with Image-Pro Plus software 6.0 (Media Cybernetics, Rockville, MD, United States).
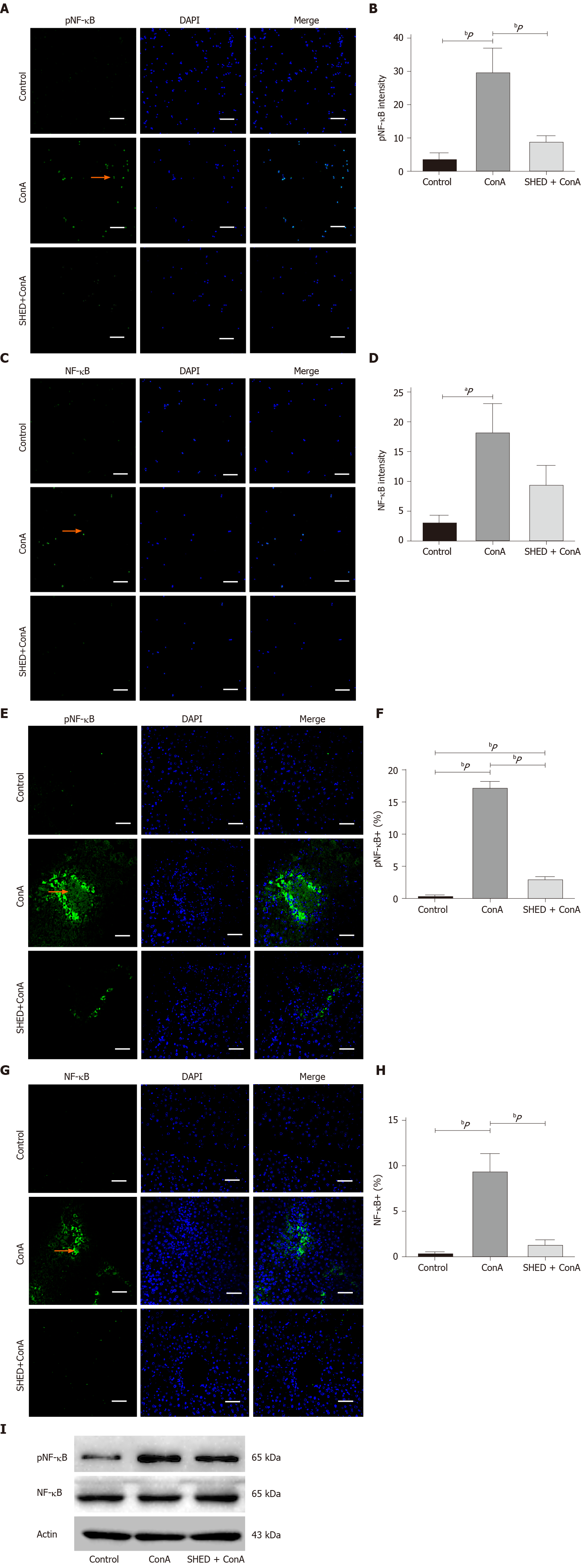
After washing in PBS for 5 min, frozen tissue sections were treated with 0.1% Triton at room temperature for 30 min to promote membrane rupture, and non-specific antigen binding sites were blocked with 5% bovine serum albumin. Rabbit anti-total caspase3 (1:100), anti-phosphorylated (p) NF-κB (1:400), and anti-NF-κB (1:400) antibodies were added and incubated overnight at 4 °C. Nuclear staining was performed with DAPI mounting medium after incubation with secondary antibody for 1 h. All sections were observed with a Zeiss fluorescence microscope (Zeiss, Thornwood, NY, United States).
Murine liver NCTC-1469 cells were purchased from the Institute of Biochemistry and Cell Biology, Chinese Academy of Science (Shanghai, China). SHED were provided by the Oral Stem Cell Bank (Beijing, China) and isolated as previously reported[8]. NCTC-1469 cells were cultured in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 U/mL streptomycin. SHED cells were cultured in α-MEM supplemented with 15% fetal bovine serum, 100 U/mL penicillin, and 100 U/mL streptomycin. NCTC cells (2 × 105 cells/well) were cultivated in 6-well plates and incubated with ConA of different concentrations (10, 20, 50, 80 and 100 μg/mL), which were analyzed for cell proliferative activity by cell counting. We then selected the appropriate treatment concentration (50 μg/mL) to study whether SHED cells inhibit NCTC cell death caused by ConA in vitro. PKH26-labeled NCTC-1469 cells were inoculated into 6-well plates at a density of 2 × 105 cells/well for 12 h. Cells were then cultured with 50 μg/mL ConA and SHED cells (1 × 105 cells/well) for 24 h. Determination of cell seeding density was based on our preliminary experiments by which we found that this ratio is more suitable for the study of cell co-cultures. Cells on the slide were fixed and observed with a fluorescence microscope (Zeiss).
Apoptosis in liver tissues was detected by terminal dUTP nick-end labeling (TUNEL) staining. Paraffin-embedded sections were deparaffinized and digested with 20 μg/mL proteinase K for 30 min at room temperature. After washing with PBS, slices were incubated with the TUNEL reaction overnight at 4 °C, followed by incubation with rabbit anti-mouse fluorescein isothiocyanate, and DAPI for nuclear labeling. TUNEL+ cells were observed and counted in at least three different fields per section.
For the in vitro study, NCTC 1469 cells were inoculated in 6-well plates at a density of 2 × 105 cells/well, followed by the addition of ConA (50 μg/mL) and SHED (1 × 105 cells/well). After 24 h of culture, cells were collected, washed with PBS, and stained using an Annexin V apoptosis detection kit. FACS was used to observe cell fluorescence. Cells on the slide were fixed, and immunofluorescence staining was then used to determine apoptotic levels. After incubation in blocking buffer, cells were incubated with anti–caspase3 (1:400), anti-phosphorylated (p) NF-κB (1:200), and anti-NF-κB (1:200) overnight at 4 °C, followed by incubation with goat anti-rabbit fluorescein isothiocyanate antibody. Nuclear staining was performed with DAPI. Sections were observed with a fluorescence microscope (Zeiss).
Mouse normal liver cells (NCTC 1469) were treated with ConA (50 mg/mL) or ConA + SHED cells (1 × 105 cells/well, transwell). When the cells grew to 80%, proteins were extracted after 24 h of treatment. The proteins were separated by sodium dodecyl sulfate polyacrylamide-gel electrophoresis and transferred to membranes, which were then blocked for 1 h. The membranes were incubated with the following primary antibodies: Anti–pNF-κB (1:1000; Cell Signaling) and anti–NF-κB (1:1000; Cell Signaling). After washing with TBS containing Tween-20, the membranes were incubated with a secondary antibody for 1 h, and chemiluminescence was used to visualize protein bands.
To analyze the effects of SHED infusion on ConA-induced AIH, we infused the SHED cells (1 × 106) 7 d prior to ConA injection (Figure 1A). The results showed that pretreatment of SHED infusion significantly inhibited the transaminase (ALT and AST) release in AIH (Figure 1B and C). Anatomical and histological examinations, which were used to estimate liver injury, showed that the structure of liver lobule and the arrangement of liver cells were clear in the control group, while the structure and arrangement of liver lobules were aberrant, with inflammatory cell infiltration and massive necrotic areas in the ConA group. Moreover, SHED infusion could significantly restore liver structure and reduce inflammatory cell infiltration (Figure 1D and E). DiR-labeled SHED cells were detected in mouse liver and spleen 1 d, 3 d, and 7 d after the transplantation (Supplementary Figure 1A). With the extension of time after SHED injection, the intensity of dye decreased gradually. Moreover, CFSE labeled cells were detected in mouse liver and spleen, which also decreased in a time dependent manner. However, no obvious recruitment of SHED cells was observed in the lung, kidney, and heart (Supplementary Figure 1B and C). These results suggested that SHED cells recruited in the liver of mice may play protective effects on acute liver injury.
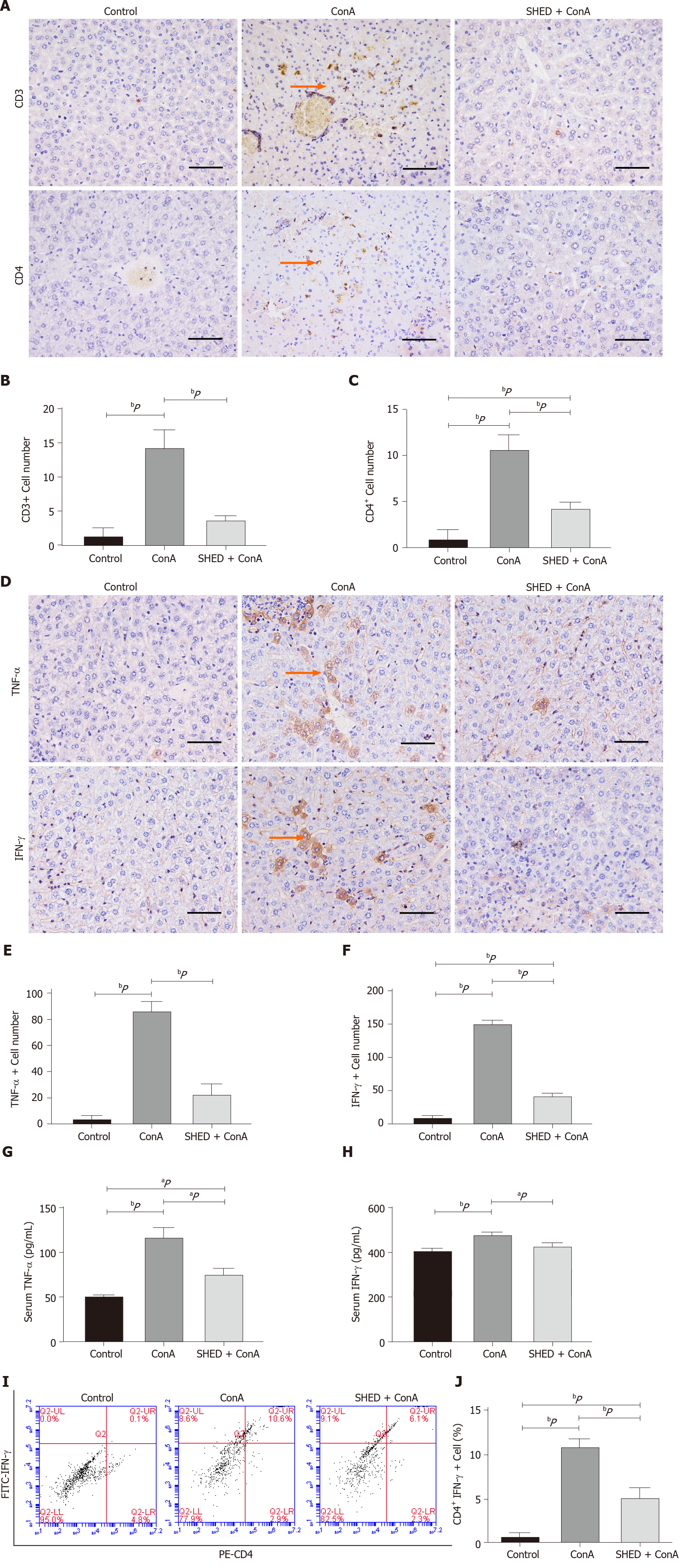
T cell-mediated immune responses play a crucial role in the occurrence and development of AIH. We further explored the underlying mechanism by analyzing T cell infiltration and inflammation. The results showed that the number of CD3+ and CD4+ T cells in the liver tissues of mice in the ConA group was significantly higher than that of the control group, and that SHED pretreatment markedly reduced the number of CD3+ and CD4+ T cells (Figure 2A-C). Similarly, we detected the level of CD3+ and CD4+ T cells in whole blood, and the results were consistent with previous results (Supplementary Figure 2). Hepatitis is associated with changes in inflammatory cytokine levels. TNF-α and IFN-γ were significantly increased in the ConA-treated group, while pretreatment with SHED cells significantly reduced the ConA-induced increase of TNF-α and IFN-γ in liver tissues (Figure 2D-F). In addition, biochemical assays were used to detect the levels of TNF-α and IFN-γ in mouse sera, and the findings were consistent with the results of those in the liver tissues (Figure 2G and H). Besides, we found that the number of CD4+ IFN-γ+ T cells in ConA group increased, and SHED injection could reverse this situation (Figure 2I and J). These results showed that SHED played an immunomodulatory role in AIH mice, which alleviated liver damage by inhibiting the activation of T helper 1 cell-mediated inflammation.
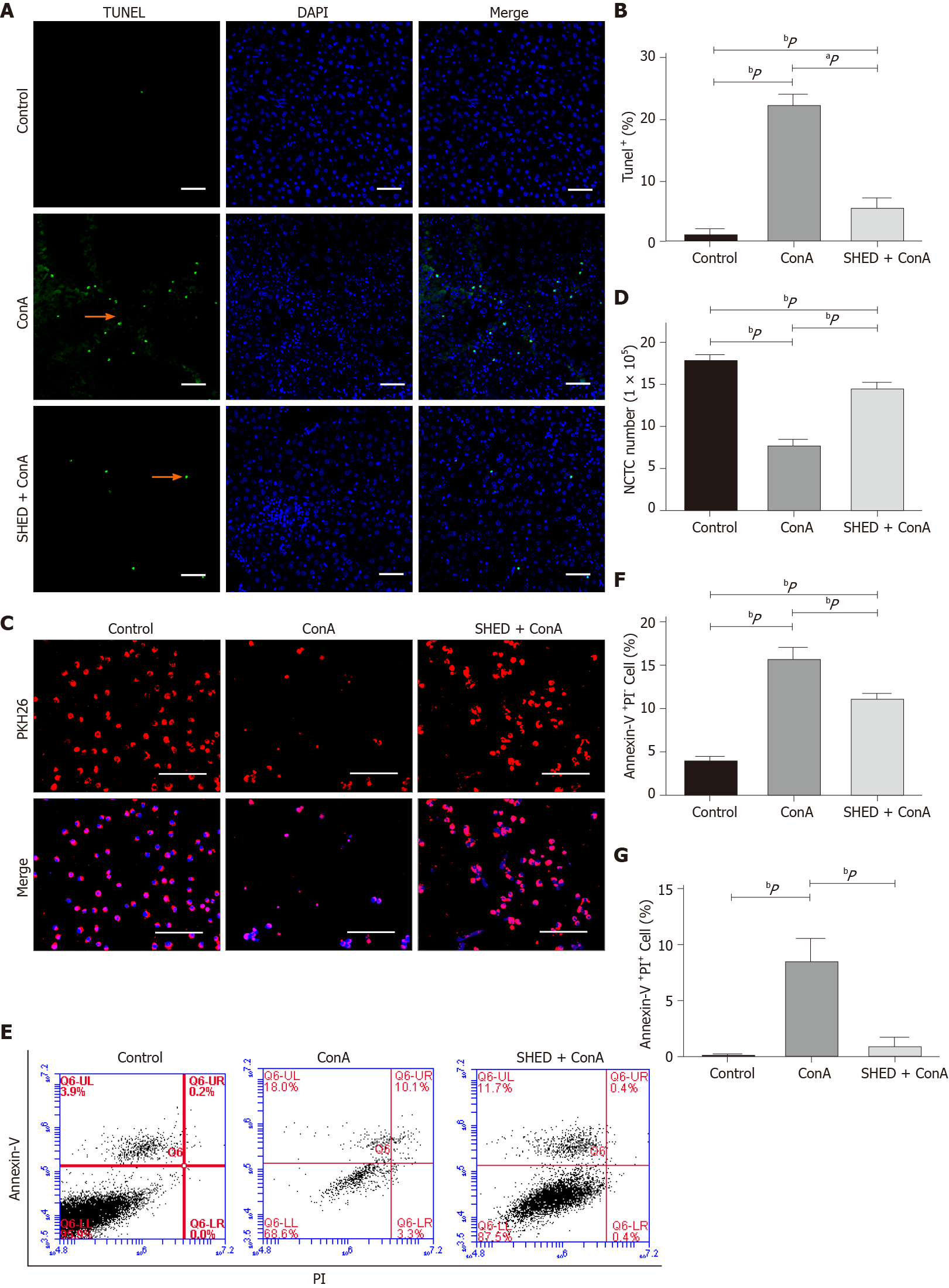
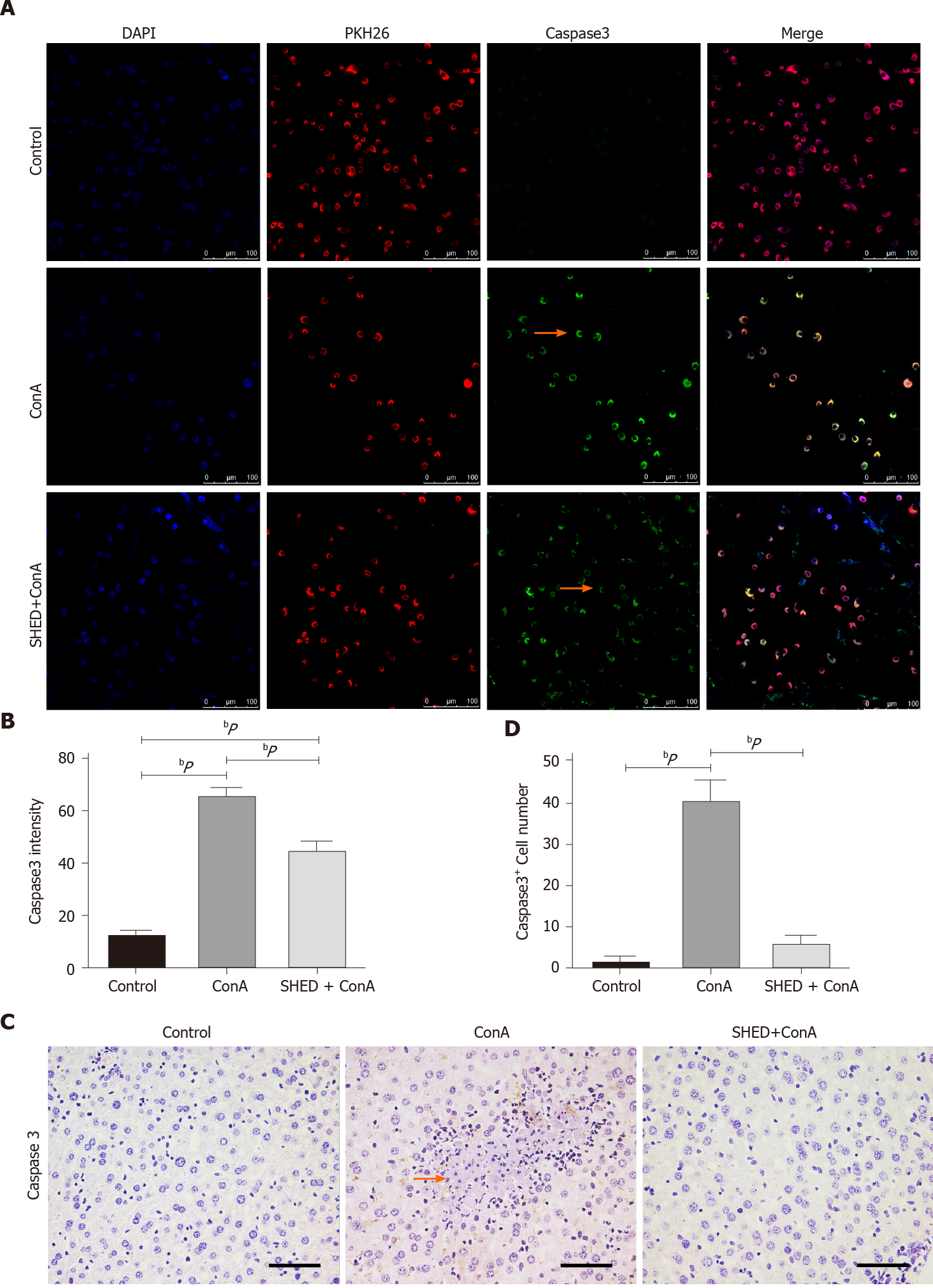
Since ConA could induce hepatocyte damage, we analyzed whether ConA treatment affected liver cell apoptosis. The results revealed that the ConA group showed more apoptotic liver cells, and SHED significantly reduced this apoptotic trend (Figure 3A and B). In order to test whether SHED infusion could protect liver cells from apoptosis, we used mouse liver NCTC 1469 cells co-cultured with SHED in vitro. ConA treatment decreased NCTC cell viability in a dose-dependent manner (Supplementary Figure 3A and B). NCTC cells were stimulated with ConA at a concentration of 50 μg/mL for 24 h, then co-cultured with SHED to analyze its protective effect. The data showed that pretreatment with SHED considerably increased NCTC cell viability, which was inhibited by ConA treatment (Supplementary Figure 3C and D). To verify the effects of SHED, NCTC cells were labeled with PKH26 and co-cultured with SHED, and the results showed that the number of PKH26-labeled NCTC cells significantly decreased after ConA treatment, while SHED co-culture blocked this decrease in live PKH26-labeled NCTC cells (Figure 3C and D). Furthermore, FACS showed that ConA treatment induced remarkable (Annexin-V+PI- and Annexin-V+PI+) cell apoptosis, which could be blocked by SHED pretreatment (Figure 3E-G). Caspase3 is one of the most important end shear enzymes in the process of apoptosis. We found that caspase3 expression was upregulated in ConA-treated NCTC cells, while SHED co-culture decreased this upregulation, as assessed by immunofluorescence staining (Figure 4A and B). Furthermore, we found that the number of caspase3+ cells was also increased in the liver of ConA-induced hepatitis mice. SHED pretreatment could block the increase of caspase3+ cells in the liver (Figure 4C and D). These results indicated that SHED infusion could protect hepatocytes from ConA-induced apoptosis.
Inflammatory cytokines TNF-α and IFN-γ were elevated in ConA-induced hepatitis, as the NF-κB pathway plays a crucial role in this cytokine-mediated inflammation[21]. We analyzed the NF-κB pathway and found that pNF-κB expression was upregulated in ConA-treated NCTC cells, while SHED co-culture decreased this upregulation (Figure 5A and B). NF-κB also showed the same trend (Figure 5C and D). In addition, we found that the numbers of cells positive for pNF-κB (Figure 5E and F) and NF-κB (Figure 5G and H) were significantly increased in the ConA-treated group and significantly decreased in the SHED cell pretreated groups. These results were verified by Western blot analysis (Figure 5I), suggesting that SHED cell treatment may ameliorate liver injury in ConA-induced hepatitis, in part, through the NF-κB pathway.
AIH is an immune-mediated inflammatory disease of the liver that can exist in acute or chronic forms, and has an increasing incidence and mortality rate. It is characterized by the elevation of serum transaminase, immunoglobulin G levels, and positive circulating autoantibody[22]. Here, we reveal that pretreatment with SHED for 7 d has a protective effect against ConA-induced hepatitis by restoring liver structure and reducing serum transaminase levels. Studies have reported that several MSCs could partially alleviate ConA-induced hepatitis in mice when MSCs and ConA were co-transfused[6,7].
SHED are a brilliant resource solution for tissue engineering and immunotherapy because of its wide range of sources, safety, convenience, low immune rejection, high proliferation ability, strong multidirectional differentiation potential, and significant immunomodulatory properties. Their therapeutic potential has been extensively studied in various animal disease models, and previous research has achieved promising insights for clinical treatment[23-25]. SHED therapy has been used to treat refractory liver disease, which involves severe dysfunction and fibrosis, through hepatocyte transdifferentiation and paracrine mechanisms[10,26]. Here, we first report that pretreatment with SHED demonstrates protective effects for acute hepatitis. ConA-induced hepatitis progresses rapidly, and causes death in more than 50% of mice in 24 h without intervention (data not shown). This phenomenon suggests that earlier SHED infusion may benefit acute hepatitis treated with stem cells. According to our results, SHED cells can be specifically recruited to the liver and spleen after tail vein injection, so as to play the role of pretreatment through blood circulation into the lesion site. Spleen and liver accumulation can be related to phagocytic cell uptake by the mononuclear phagocyte system. SHED are derived from the pulp of deciduous teeth, which are easily obtained in a non-invasive manner[27]. These data indicate that pretreatment with SHED may be one alternative strategy to prevent and treat severe acute hepatitis and liver injury.
T cell-secreted TNF-α and IFN-γ play important roles in inflammatory responses[28]. Previous studies have shown that mice pretreated with TNF-α inhibitors or anti-TNF-α antibodies can reduce ConA-induced hepatitis[29]. TNF-α and IFN-γ knockout mice could not induce liver damage following ConA stimulation[28-30]. It has been reported that SHED has superior anti-inflammatory effects compared to BMMSCs[31]. In this study, the number of CD3+ and CD4+ T lymphocytes in the liver decreased following SHED injection, so as to inhibit T helper 1 cell inflammatory factors and downregulate TNF-α and IFN-γ expression in liver tissues and peripheral blood. In vitro, we found that SHED can reverse this decrease in ConA-induced liver cells, and we thus studied its effect on liver cell apoptosis. The results indicated that SHED could reverse ConA-induced liver cell apoptosis.
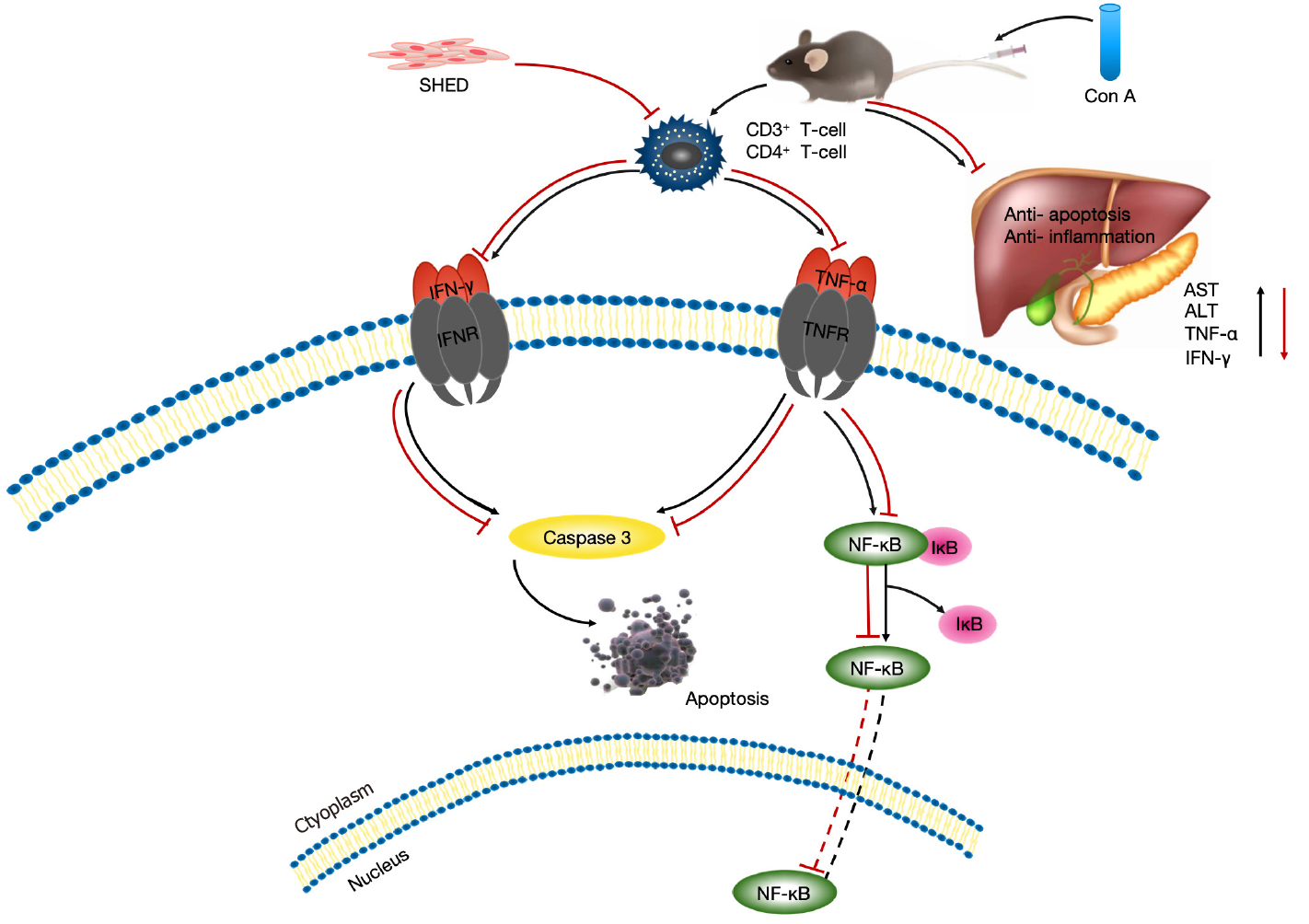
Several pathways, such as NF-κB, Fas/FasL, JNK, MAPK, and ROS, play crucial roles in cell apoptosis[32-34]. Based on the significant protective effects of SHED, we explored the potential underlying mechanisms of SHED against ConA-induced apoptosis. The results showed that pretreatment with SHED could effectively inhibit p65 phosphorylation, suggesting that SHED may inhibit NF-κB activation by reducing TNF-α and IFN-γ expression, thus protecting mice from AIH invasion[34-37]. Since apoptosis may also be regulated by death receptors or the mitochondrial pathway, the caspase family needs to be activated in both cases[38,39]. The results showed that SHED may block caspase3 activation to inhibit external apoptosis (Figure 6). However, details of the mechanism and whether this signaling contributes to the protective effects of SHED on ConA-induced hepatitis require further investigation.
In summary, our results reveal that SHED exhibits protective effects in a ConA-induced AIH mouse model. SHED treatment could inhibit the activation of T helper 1 cells and reduce the secretion of TNF-α and IFN-γ to inhibit NF-κB pathway activation induced by ConA, so as to reduce hepatocyte apoptosis and inflammatory liver damage. Our results suggest that SHED could be used as a potential preventive and therapeutic strategy for acute hepatitis and liver injury.
Autoimmune hepatitis (AIH) is a serious autoimmune liver disease that threatens human health globally, thus emphasizing the need to identify novel treatments. Stem cells from human exfoliated deciduous teeth (SHED), which are easy to non-invasively obtain, showed pronounced proliferative and immunomodulatory capacities.
AIH treatment requires long-term or even lifelong use of immunosuppressants, which may cause serious side effects. Some patients are insensitive to treatment and cannot be well controlled. More appropriate and effective medical technologies to cure diabetes are needed. Mesenchymal stem cell infusion could alleviate a variety of diseases such as AIH. SHED show more pronounced proliferative and im-munomodulatory capacities compared to bone marrow mesenchymal stem cells, and can differentiate into hepatocytes in vitro. Non-traumatically collected stem cells (SHED) may therefore be a promising option for treating AIH.
To investigate the protective effects of SHED on concanavalin A (ConA)-induced hepatitis in mice, and to elucidate the associated regulatory mechanisms.
We used a ConA-induced acute hepatitis mouse model (6–week-old to 8-week-old male Balb/c mice) and an in vitro co-culture system to study the protective effects of SHED on ConA-induced AIH, as well as the underlying mechanisms. Protocols were approved by the Animal Care and Use Committee of the Health Science Center, Peking University (No. 2015-186).
SHED infusion could prevent aberrant histopathological liver architecture caused by ConA-induced infiltration of CD3+, CD4+, tumor necrosis-alpha+, and interferon-gamma+ inflammatory cells. Alanine aminotransferase and aspartate aminotransferase were significantly elevated in hepatitis mice. SHED infusion could block the ConA-induced elevation of alanine aminotransferase and aspartate aminotransferase. Mechanistically, ConA upregulated tumor necrosis-alpha and interferon-gamma expression, which were activated by the nuclear factor-kappa B pathway, to induce hepatocyte apoptosis, resulting in acute liver injury. SHED administration protected hepatocytes from ConA-induced apoptosis.
We demonstrate that SHED exhibits protective effects in a ConA-induced AIH mouse model. SHED treatment could inhibit the activation of Th1 cells, and reduce the secretion of tumor necrosis-alpha and interferon-gamma expression to inhibit ConA-induced nuclear factor-kappa B pathway activation. This reduces both hepatocyte apoptosis and inflammatory liver damage.
These results could provide a potential prevention and therapeutic strategy for hepatitis and acute hepatic injury.
We thank the Oral Stem Cell Bank of China for providing the SHED cells. This article is present on a repository website and can be accessed at https://www.research-square.com/article/rs-17847/v1.
Manuscript source: Unsolicited manuscript
Specialty type: Biochemistry and molecular biology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Durazzo M, Sallustio F S-Editor: Zhang L L-Editor: Wang TQ P-Editor: Wu YXJ
| 1. | Tansel A, Katz LH, El-Serag HB, Thrift AP, Parepally M, Shakhatreh MH, Kanwal F. Incidence and Determinants of Hepatocellular Carcinoma in Autoimmune Hepatitis: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol 2017; 15: 1207-1217. e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 2. | Czaja AJ. Diagnosis and Management of Autoimmune Hepatitis: Current Status and Future Directions. Gut Liver. 2016;10:177-203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 3. | Czaja AJ, Bianchi FB, Carpenter HA, Krawitt EL, Lohse AW, Manns MP, McFarlane IG, Mieli-Vergani G, Toda G, Vergani D, Vierling J, Zeniya M. Treatment challenges and investigational opportunities in autoimmune hepatitis. Hepatology. 2005;41:207-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 4. | Squillaro T, Peluso G, Galderisi U. Clinical Trials with Mesenchymal Stem Cells: An Update. Cell Transplant. 2016;25:829-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1128] [Cited by in RCA: 1032] [Article Influence: 103.2] [Reference Citation Analysis (0)] |
| 5. | Zhu X, He B, Zhou X, Ren J. Effects of transplanted bone-marrow-derived mesenchymal stem cells in animal models of acute hepatitis. Cell Tissue Res. 2013;351:477-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 6. | Higashimoto M, Sakai Y, Takamura M, Usui S, Nasti A, Yoshida K, Seki A, Komura T, Honda M, Wada T, Furuichi K, Ochiya T, Kaneko S. Adipose tissue derived stromal stem cell therapy in murine ConA-derived hepatitis is dependent on myeloid-lineage and CD4+ T-cell suppression. Eur J Immunol. 2013;43:2956-2968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 7. | Ryu KH, Kim SY, Kim YR, Woo SY, Sung SH, Kim HS, Jung SC, Jo I, Park JW. Tonsil-derived mesenchymal stem cells alleviate concanavalin A-induced acute liver injury. Exp Cell Res. 2014;326:143-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 8. | Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci USA. 2003;100:5807-5812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1922] [Cited by in RCA: 1984] [Article Influence: 90.2] [Reference Citation Analysis (0)] |
| 9. | Nakamura S, Yamada Y, Katagiri W, Sugito T, Ito K, Ueda M. Stem cell proliferation pathways comparison between human exfoliated deciduous teeth and dental pulp stem cells by gene expression profile from promising dental pulp. J Endod. 2009;35:1536-1542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 180] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 10. | Yamaza T, Alatas FS, Yuniartha R, Yamaza H, Fujiyoshi JK, Yanagi Y, Yoshimaru K, Hayashida M, Matsuura T, Aijima R, Ihara K, Ohga S, Shi S, Nonaka K, Taguchi T. In vivo hepatogenic capacity and therapeutic potential of stem cells from human exfoliated deciduous teeth in liver fibrosis in mice. Stem Cell Res Ther. 2015;6:171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 11. | Ohkoshi S, Hirono H, Nakahara T, Ishikawa H. Dental pulp cell bank as a possible future source of individual hepatocytes. World J Hepatol. 2018;10:702-707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 12. | Bies C, Lehr CM, Woodley JF. Lectin-mediated drug targeting: history and applications. Adv Drug Deliv Rev. 2004;56:425-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 284] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 13. | Heymann F, Hamesch K, Weiskirchen R, Tacke F. The concanavalin A model of acute hepatitis in mice. Lab Anim. 2015;49:12-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 202] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 14. | Erhardt A, Biburger M, Papadopoulos T, Tiegs G. IL-10, regulatory T cells, and Kupffer cells mediate tolerance in concanavalin A-induced liver injury in mice. Hepatology. 2007;45:475-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 223] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 15. | Wang L, Tu L, Zhang J, Xu K, Qian W. Stellate Cell Activation and Imbalanced Expression of TGF-β1/TGF-β3 in Acute Autoimmune Liver Lesions Induced by ConA in Mice. Biomed Res Int. 2017;2017:2540540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Schümann J, Wolf D, Pahl A, Brune K, Papadopoulos T, van Rooijen N, Tiegs G. Importance of Kupffer cells for T-cell-dependent liver injury in mice. Am J Pathol. 2000;157:1671-1683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 231] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 17. | Li J, Chen K, Li S, Liu T, Wang F, Xia Y, Lu J, Zhou Y, Guo C. Pretreatment with Fucoidan from Fucus vesiculosus Protected against ConA-Induced Acute Liver Injury by Inhibiting Both Intrinsic and Extrinsic Apoptosis. PLoS One. 2016;11:e0152570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Schwabe RF, Brenner DA. Mechanisms of Liver Injury. I. TNF-alpha-induced liver injury: role of IKK, JNK, and ROS pathways. Am J Physiol Gastrointest Liver Physiol. 2006;290:G583-G589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 561] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 19. | Li J, Wang F, Xia Y, Dai W, Chen K, Li S, Liu T, Zheng Y, Wang J, Lu W, Zhou Y, Yin Q, Lu J, Zhou Y, Guo C. Astaxanthin Pretreatment Attenuates Hepatic Ischemia Reperfusion-Induced Apoptosis and Autophagy via the ROS/MAPK Pathway in Mice. Mar Drugs. 2015;13:3368-3387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 111] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 20. | Martinez Saez D, Sasaki RT, Neves AD, da Silva MC. Stem Cells from Human Exfoliated Deciduous Teeth: A Growing Literature. Cells Tissues Organs. 2016;202:269-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 21. | Mohan RR, Mohan RR, Kim WJ, Wilson SE. Modulation of TNF-alpha-induced apoptosis in corneal fibroblasts by transcription factor NF-kappaB. Invest Ophthalmol Vis Sci. 2000;41:1327-1336. [PubMed] |
| 22. | Balitzer D, Shafizadeh N, Peters MG, Ferrell LD, Alshak N, Kakar S. Autoimmune hepatitis: review of histologic features included in the simplified criteria proposed by the international autoimmune hepatitis group and proposal for new histologic criteria. Mod Pathol. 2017;30:773-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 23. | Fujii H, Matsubara K, Sakai K, Ito M, Ohno K, Ueda M, Yamamoto A. Dopaminergic differentiation of stem cells from human deciduous teeth and their therapeutic benefits for Parkinsonian rats. Brain Res. 2015;1613:59-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 24. | Rossato C, Brandão WN, Castro SBR, de Almeida DC, Maranduba CMC, Camara NOS, Peron JPS, Silva FS. Stem cells from human-exfoliated deciduous teeth reduce tissue-infiltrating inflammatory cells improving clinical signs in experimental autoimmune encephalomyelitis. Biologicals. 2017;49:62-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | LEIMDORFER A. Electroencephalographic analysis of action of amidone, morphine and strychnine on the central nervous system. Fed Proc. 1948;7:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 204] [Cited by in RCA: 180] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 26. | Ishkitiev N, Yaegaki K, Imai T, Tanaka T, Fushimi N, Mitev V, Okada M, Tominaga N, Ono S, Ishikawa H. Novel management of acute or secondary biliary liver conditions using hepatically differentiated human dental pulp cells. Tissue Eng Part A. 2015;21:586-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 27. | Xie F, He J, Chen Y, Hu Z, Qin M, Hui T. Multi-lineage differentiation and clinical application of stem cells from exfoliated deciduous teeth. Hum Cell. 2020;33:295-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 28. | Constant SL, Bottomly K. Induction of Th1 and Th2 CD4+ T cell responses: the alternative approaches. Annu Rev Immunol. 1997;15:297-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1152] [Cited by in RCA: 1124] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 29. | Wolf D, Hallmann R, Sass G, Sixt M, Küsters S, Fregien B, Trautwein C, Tiegs G. TNF-alpha-induced expression of adhesion molecules in the liver is under the control of TNFR1--relevance for concanavalin A-induced hepatitis. J Immunol. 2001;166:1300-1307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 94] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 30. | Ksontini R, Colagiovanni DB, Josephs MD, Edwards CK 3rd, Tannahill CL, Solorzano CC, Norman J, Denham W, Clare-Salzler M, MacKay SL, Moldawer LL. Disparate roles for TNF-alpha and Fas ligand in concanavalin A-induced hepatitis. J Immunol. 1998;160:4082-4089. [PubMed] |
| 31. | Otabe K, Muneta T, Kawashima N, Suda H, Tsuji K, Sekiya I. Comparison of Gingiva, Dental Pulp, and Periodontal Ligament Cells From the Standpoint of Mesenchymal Stem Cell Properties. Cell Med. 2012;4:13-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 32. | Li J, Xia Y, Liu T, Wang J, Dai W, Wang F, Zheng Y, Chen K, Li S, Abudumijiti H, Zhou Z, Wang J, Lu W, Zhu R, Yang J, Zhang H, Yin Q, Wang C, Zhou Y, Lu J, Zhou Y, Guo C. Protective effects of astaxanthin on ConA-induced autoimmune hepatitis by the JNK/p-JNK pathway-mediated inhibition of autophagy and apoptosis. PLoS One. 2015;10:e0120440. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 33. | Tao YY, Yan XC, Zhou T, Shen L, Liu ZL, Liu CH. Fuzheng Huayu recipe alleviates hepatic fibrosis via inhibiting TNF-α induced hepatocyte apoptosis. BMC Complement Altern Med. 2014;14:449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Zhang P, Yin Y, Wang T, Li W, Li C, Zeng X, Yang W, Zhang R, Tang Y, Shi L, Li R, Tao K. Maresin 1 mitigates concanavalin A-induced acute liver injury in mice by inhibiting ROS-mediated activation of NF-κB signaling. Free Radic Biol Med. 2020;147:23-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 35. | Chen YY, Li RY, Shi MJ, Zhao YX, Yan Y, Xu XX, Zhang M, Zhao XT, Zhang YB. Demethyleneberberine alleviates inflammatory bowel disease in mice through regulating NF-κB signaling and T-helper cell homeostasis. Inflamm Res. 2017;66:187-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 36. | Mehmood T, Maryam A, Zhang H, Li Y, Khan M, Ma T. Deoxyelephantopin induces apoptosis in HepG2 cells via oxidative stress, NF-κB inhibition and mitochondrial dysfunction. Biofactors. 2017;43:63-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 37. | ELnahas EM, Zeid MS, Kawy HS, Hendawy N, Baher W. Celecoxib attenuates depressive-like behavior associated with immunological liver injury in C57BL/6 mice through TNF-α and NF-κb dependent mechanisms. Life Sci. 2016;163:23-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 38. | Liu T, Xia Y, Li J, Li S, Feng J, Wu L, Zhang R, Xu S, Cheng K, Zhou Y, Zhou S, Dai W, Chen K, Wang F, Lu J, Zhou Y, Guo C. Shikonin Attenuates Concanavalin A-Induced Acute Liver Injury in Mice via Inhibition of the JNK Pathway. Mediators Inflamm. 2016;2016:2748367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 39. | Mao Y, Wang J, Yu F, Cheng J, Li H, Guo C, Fan X. Ghrelin reduces liver impairment in a model of concanavalin A-induced acute hepatitis in mice. Drug Des Devel Ther. 2015;9:5385-5396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |