Published online Oct 26, 2020. doi: 10.4252/wjsc.v12.i10.1080
Peer-review started: May 29, 2020
First decision: June 15, 2020
Revised: June 26, 2020
Accepted: August 24, 2020
Article in press: August 24, 2020
Published online: October 26, 2020
Processing time: 149 Days and 23.4 Hours
Cell therapy, i.e., the use of cells to repair an affected tissue or organ, is at the forefront of regenerative and personalized medicine. Among the multiple cell types that have been used for this purpose [including adult stem cells such as mesenchymal stem cells or pluripotent stem cells], urine-derived stem cells (USCs) have aroused interest in the past years. USCs display classical features of mesenchymal stem cells such as differentiation capacity and immunomodulation. Importantly, they have the main advantage of being isolable from one sample of voided urine with a cheap and unpainful procedure, which is broadly applicable, whereas most adult stem cell types require invasive procedure. Moreover, USCs can be differentiated into renal cell types. This is of high interest for renal cell therapy-based regenerative approaches. This review will firstly describe the isolation and characterization of USCs. We will specifically present USC phenotype, which is not an object of consensus in the literature, as well as detail their differentiation capacity. In the second part of this review, we will present and discuss the main applications of USCs. These include use as a substrate to generate human induced pluripotent stem cells, but we will deeply focus on the use of USCs for cell therapy approaches with a detailed analysis depending on the targeted organ or system. Importantly, we will also focus on the applications that rely on the use of USC-derived products such as microvesicles including exosomes, which is a strategy being increasingly employed. In the last section, we will discuss the remaining barriers and challenges in the field of USC-based regenerative medicine.
Core Tip: Urine-derived stem cells (USCs) display classical features of mesenchymal stem cells such as differentiation capacity and immunomodulation. Importantly, they are easily isolated from voided urine. Moreover, USCs can be differentiated into many cell types including renal cells. This review will describe the isolation and characterization of USCs and detail their differentiation capacity. Then, we will present the main applications of USCs, from reprogramming into human induced pluripotent stem cells to their use for cell therapy approaches (either directly or via their exosomes). Finally, the remaining barriers and challenges in the field of USC-based regenerative medicine will be discussed.
- Citation: Burdeyron P, Giraud S, Hauet T, Steichen C. Urine-derived stem/progenitor cells: A focus on their characterization and potential. World J Stem Cells 2020; 12(10): 1080-1096
- URL: https://www.wjgnet.com/1948-0210/full/v12/i10/1080.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v12.i10.1080
The quest for the optimal cellular product to use for cell therapy strategies has always been challenging for many reasons. First, candidates are numerous and include not only somatic cells but mostly stem cells from adult stem cells to pluripotent ones (either embryonic or induced) in distinct differentiation steps. Moreover, the suitable cell population depends on the targeted organ and its affection. In addition, despite a huge amount of in vivo works (mostly on rodents but some preclinical works have been published[1,2]), the scientific community may not have the necessary hindsight regarding long-term clinical effects (except for procedures already used in clinical routine especially in the hematologic field).
By definition, stem cells are cells able to self-renew and to differentiate into another cell type. Multipotency is defined as the potential to differentiate into more than one cell type. This review focuses on one adult multipotent stem cell type that has aroused interest in the past years especially for their huge advantage of being isolable from one sample of voided urine. Whereas most isolation of adult stem cell types require an invasive procedure [such as mesenchymal stem cells (MSCs) from adipose tissue or bone marrow][3], urine-derived stem cells (USCs) can be isolated and amplified from a noninvasive, unpainful and cheap procedure, applicable to every patient, including children, without specific care issues and facilities.
While USCs display stem cell properties and a variety of markers, their phenotype is not an object of consensus in the literature. They have been used for cell-based strategies in many rodent models of tissue regeneration and display strong beneficial effects[4,5]. They have also been used in indirect approaches, employing their secretome instead of the cells themselves[6,7]. The aim of this review is to list, discuss and propose a summary of USC characterization and applications.
The presence of cells in an adult urine sample was described for the first time in 1976[8]. Since then, many studies reported the presence of epithelial cells in children and/or adult urine samples[9,10]. However, the presence of cells in urine that display stem cell properties was described in 2008 by Zhang et al[11].
USCs are isolated either from voided urine[11] or upper urinary tract collection[12]. Urine is collected into a sterile 50 mL tube with antibiotic and then centrifuged at approximately 400 g for 10 min[13,14]. This first step allows elimination of urinary debris such as dead squamous cells regularly present in urine or potential contamination depending on the collection method. Pellets are usually washed with phosphate buffered saline (PBS) and re-suspended in a specific culture medium (described later) on 6, 12 or 24 well plates, with or without precoating with collagen or 0.1% gelatin[15,16]. From day 1 to day 3, culture supernatant contains a lot of dead and blood cells. The medium is changed every day and then every other day.
First, colonies usually appear around 3 d to 9 d after isolation with zero to several colonies independent of the age or the sex of the donor[15-18]. The overall success rate (being defined by the apparition of at least one colony) is about 50%[16]. Two types of USC colony have been reported: Spindle-shaped USCs and rice-shaped USCs[12,13,15,19-21]. Different USC types can be present in a single urine sample or in one dish. For the first passaging, USCs are detached enzymatically (either clonally or by pooling the colonies together) and amplified until their cryopreservation. It should be noted that after urine sample conservation at 4°C for 24 h, it remains possible to isolate USCs that keep their proliferation capacity and telomerase activity[19].
While different culture media have been described to culture these cells, they usually rely on a mixture of basal medium (either Dulbecco’s modified Eagle’s medium or keratinocyte-serum-Free medium) containing 0.5% to 10% of fetal bovine serum with another medium that is supplemented with growth factors including epidermal growth factor such as the renal epithelial growth medium (Lon-za)[13,15,16,18,22,23].
USC doubling time varies according to passages, ranging from 20 h at P1 to 28 h at P5. These results could be variable according to donor or culture medium[12,19,24]. With passages, cells adopt an elongated “spindle-shaped” fibroblast-like morphology[22].
USCs can be expanded both in normoxia (21% O2) or hypoxia with hypoxia being shown to induce higher proliferation rate but also to increase the USC potential for angiogenesis in a wound healing model[25,26].
Human USCs expressed a variety of markers, and their proposed phenotypes vary according to studies, urine sample and likewise between single colonies appearing in a single urine sample[20,27,28].
A detailed analysis of the USC phenotype is described in Table 1. There are some concordant results in the literature showing that USCs express some MSC markers (CD44, CD73 and VIM) and do not express the pan-hematopoietic marker CD45 or other hematopoietic-derived cell lineage markers (CD11b, CD14 and CD19). Regarding other MSC markers such as CD90, CD105, STRO-1 and other hematopoietic markers such as CD34, the expression level is variable among studies, analysis methods or USC donors. USCs express some markers linked with stemness and pluripotency (such as the embryonic stem cells markers POU5F1, SSEA4 and TRA-1-81) as well as CD117 (c-kit) marker. Regarding their origin, USCs express adhesion markers such as CD29 and/or CD166 but not CD31. They also express some renal specific transporters such as AQP1 and/or AQP2[20]. Importantly, some studies show that at least a subpopulation of USC doubly express CD24/CD133. This combination as well as the transcription factor SIX2 indicates a population of renal progenitor cells, which is crucial for kidney embryonic development[14].
| Marker | Percentage of expression, % | Ref. |
| Embryonic stem cell markers | ||
| c-Myc | [+] | [23] |
| SSEA4 | [+++] | [24,29] |
| [++++] | [12,23,27,28,30,31] | |
| TRA-1-60 | [-] | [23] |
| [+] | [27] | |
| TRA-1-81 | [+] | [23,27,29] |
| Hematopoietic stem cell markers | ||
| CD11b | [-] | [23] |
| CD14 | [-] | [15,23,27,29] |
| CD19 | [-] | [23,29] |
| CD34 | [-] | [12,13,15,18-20,22-24,28,29,32,33] |
| CD45 | [-] | [12,13,18,19,22-24,28,29,31,33] |
| CD117 | [-] | [12,15,19,24,28] |
| [++++] | [27] | |
| Mesenchymal stem cell markers | ||
| CD24 | [+++] | [27] |
| [++++] | [20,27,30-32] | |
| CD44 | [++++] | [12,13,15,19,20,22-24,28-33] |
| CD73 | [++++] | [12,13,15,19,20,22-24,27-33] |
| CD90 | [+] | [18,27] |
| [++] | [12,29] | |
| [+++] | [19,23,24,28] | |
| [++++] | [13,15,20,22,28,30-33] | |
| CD105 | [-] | [27,31] |
| [+] | [22] | |
| [++] | [12,24,30] | |
| [+++] | [18,23] | |
| [++++] | [15,19,20,28,29,32] | |
| CD133 | [-] | [12,15,20,22,33] |
| [++] | [20] | |
| [+++] | [27] | |
| [++++] | [27] | |
| Collagen I | [++++] | [15] |
| SRTO-1 | [-] | [12,23,30] |
| [+] | [31] | |
| VIM | [++++] | [15,20,27] |
| Endothelial/adhesion markers | ||
| CD29 | [++++] | [13,20,22,23,29-33] |
| CD31 | [-] | [12,19,23,29-32,34] |
| CD166 | [++++] | [18,23] |
| Immunogen markers | ||
| CMH-I | [+] | [28] |
| [+++] | [29] | |
| [++++] | [23] | |
| CMH-II | [-] | [18,22-24,28,29,33] |
| Kidney markers | ||
| AQP1 | [++++] | [20] |
| AQP2 | [-] | [20] |
| [++] | [20] | |
| CD13 | [++++] | [18,23,29] |
| CD54 | [+++] | [18] |
| [++++] | [23] | |
| CD224 | [++++] | [23] |
| Pax8 | [-] | [23] |
| NPHS1 | [-] | [20] |
| [++] | [20] | |
| NR3C2 | [++++] | [23] |
| SLC12A1 | [-] | [20] |
| [++] | [20] | |
| UMOD | [-] | [20] |
| [++] | [20] | |
| Pericyte markers | ||
| CD140b | [-] | [19,23] |
| [+] | [32] | |
| CD146 | [++++] | [12,19,23,29-32] |
| NG2 | [-] | [19,23,32] |
| Other markers | ||
| KRT18 | [-] | [20] |
| [++] | [20] | |
USCs originate from the kidney. Indeed, one study showed that after a kidney transplant from a male to a female, isolated USCs were carrying the Y chromosome (PCR and FISH analysis) confirming their renal origin[23]. In addition, USCs isolated from voided urine display the same characteristics and proprieties as USCs isolated specifically from the upper urinary tract. As USCs also meet the criteria of stem cells, these cells are therefore called either USCs, urine progenitor cells or renal progenitor cells depending on the studies and the isolation protocol, which can include a step of cell sorting and may therefore modify the cell phenotype and the characterization proposed.
Regarding stem cell properties, even though telomerase activity varies according to urine sample, USCs display a normal karyotype irrespective of passages[19,22-24,28] and a high telomerase activity, likely excluding contamination of epithelial cells in urine. However, despite high activity of telomerase, USCs did not form teratomas when injected into immunodeficient mice, highlighting the absence of a tumorigenesis phenotype[23,28-35].
As has been repeatedly described for MSCs, USCs also display immunomodulatory properties. Despite their higher MHC-I expression, after a few days in coculture, USCs inhibit lymphocyte proliferation in vitro[15,24]. This potential effect and their low MHC-II expression is of high interest for use of USCs in cell therapy approaches.
One of the advantages of stem cells in cell therapy or regenerative medicine is their multipotential of differentiation. USCs have been reported to be differentiable into a huge variety of cells types, which are summarized in Table 2.
| Cell type | Medium | Supplements | Initial density | Protocol duration | Note | Ref. |
| “MSC-like” differentiated cell types | ||||||
| Adipogenic cells | DMEM | Dexamethasone 1 µmol/L, isobutyl-1 methylxanthine 500 µmol/L, indomethacin 66 µmol/L, hydrocortisone 500 µmol/L | 14000 cells/cm² | 24 d | 2 d in DMEM without supplement before differentiation | [15] |
| DMEM low glucose | Dexamethasone 1 µmol/L, isobutyl-1 methylxanthine 500 µmol/L, indomethacin 100 µmol/L, insulin 10 µg/mL | 20000 cells/cm² | 28 d | / | [30] | |
| Specific adipogenic medium | FBS (optional) | 1000 to 5000 cells/cm² | 14 d | / | [13,22,23,33,39] | |
| Specific adipogenic medium | NA | 80% of confluency | 14 d | / | [24] | |
| Chondrogenic cells | DMEM/Ham's F12 | Fetal calf serum 10%, insulin 6 µg/mL, ascorbic acid 2P 0.2 mmol/L, TGF-b1 10 ng/mL | 1500 cells/cm² | 28 d | / | [15] |
| Specific chondrogenic medium | NA | 50000 cells/cm² | 28 d | / | [22,24] | |
| Specific chondrogenic medium | NA | 300000 cells to 1000000 cells | 28 d | Cell aggregation in conical polypropylene tube before differentiation | [23] | |
| Osteogenic cells | DMEM low glucose | Dexamethasone 10-100 nmol/L, ascorbate2-phosphate 50-1500 µmol/L, glycerophosphate 10 mmol/L, FCS 10%, Vit D3 10 nmol/L (optional) | 1500 to 4000 cells/cm² | 28 d | / | [15,30] |
| DMEM low glucose | FBS + osteogenic supplement | 4000 cells/cm² | 28 d | / | [23] | |
| Specific osteogenic medium | NA | 1000 cells/ cm² | 28 d | / | [39] | |
| Specific osteogenic medium | NA | 80% confluency at P4 | 14 d to 21 d | / | [22,24,33] | |
| Other cell types | ||||||
| Endothelial cells | EBM-2 | VEGF 50 ng/mL + FBS (optional) | 3000 to 5000 cells/cm² | 9 d to 14 d | Coating with fibronectin + 2 d of preculture | [23,31,32] |
| Microbeads | VEGF | 1000 cells/cm² | 14 d | MD | [35] | |
| Neuronal cells | Specific nerve induction medium | NA | 6000 cells/cm² | 2 d | Pre-culture with DMEM + 20% FBS + 10 ng/mL bFGF 24 h | [23] |
| DMEM F12 | hEGF 20 ng/mL, bFGF 40 ng/mL, B27 2%, NEAA 1%, l-glutamine 1%, insulin-transferrin-selenite 1%. | 20%-30% of confluency | 12 d | Coating: Polystyrene | [22] | |
| NeuroCult NS-A differentiation kit | NA | 100000 cells/cm² | 7 d | Preculture | [24] | |
| NM3 basic neuronal induction medium | Y27632 10 μmol/L, A8301 5 μmol/L, CHIR99021 3 μmol/L, TTNPB 1 μmol/L, forskolin5 μmol/L, valproic acid 0.5 mmol/L, sodium butyrate 0.1 mmol/L | MD | MD | Coating with Matrigel and preculture (2 d) | [37] | |
| Skeletal myogenic cells | DMEM | Skeletal supplement + SVF 10%, Horse serum 5%, hydrocortisone 50 µmol/L, dexamethasone 0,1 µmol/L | MD | 28 d | / | [23] |
| Conditioned medium | NA | MD | 28 d | 12 h of culture of medium with skeletal myogenic cells | [23] | |
| Conditioned medium | NA | 80% confluency | 14 d | Culture of medium with skeletal myogenic cells | [24] | |
| Smooth muscle cells | DMEM + EFM (1:1) | FBS, TGF-β1 (2.5 ng/mL), PDG-BB (2.5 to 5 ng/mL) | 1000 to 2000 cells/cm² | 14 d | / | [12,19,23,34,36] |
| Renal differentiation | ||||||
| Podocytes | VRAD: DMEMF12 | FBS 10%, vitamin D3 100 nmol/L, retinoic acid 100 µmol/L | MD | 48 h | MD | [14] |
| Tubular cells | REGM | Hepatocyte growth factor 50 ng/mL | MD | 21 d | MD | [14] |
| Urothelium cells | KFSM + EFM (1:1) | FBS 2%, EGF 30 ng/mL | 3000 cell/cm² | 14 d | MD | [36] |
| KFSM | FBS 2%, EGF 30 ng/mL | 3000 cell/cm² | 14 d | MD | [34] | |
| KFSM + EFM (4:1) | FBS 2%, EGF 30 ng/mL | 3000 cells/cm² | 14 d | / | [36] | |
| DMEM + KFSM (4:1) | FBS 8%, EGF 30 ng/mL | 1,000 cells/cm² | 14 d | / | [19] | |
| Conditioned medium from urothelial cells 8 h + EFM + KFSM (1:1) | EGF 30 ng/mL | 50000 cells/cm² | 7 to 21 d | Dynamic culture | [21] | |
| Conditioned medium of smooth muscle cells 12 h + KFSM (1:1) | NA | 50000 cells/cm² | 7 to 21 d | Dynamic culture | [21] | |
First, it has been shown that USCs can be differentiated towards the osteogenic, chondrogenic and adipogenic lineage (this feature being characteristics of MSCs)[12,34]. Xu et al[36] described USC differentiation into other lineages, such as urothelial cells and smooth myogenic cells, which respectively expressed uroplakin III, AE1/AE3 and desmin, myosin after implantation on 3-D porous small intestinal scaffold. Other reports described the differentiation of USCs into endothelial cells[23,24,31,32,35], neuronal cells[22-24,37] or skeletal myogenic cells[24,35]. Finally, USCs have been differentiated into renal cells, podocytes and tubular epithelial cells[14]; this feature being of great importance in USC potential for renal repair.
Despite the fact that using a variety of protocols, different laboratories have shown the multidifferentiation potential of USCs, it is important to note that: (1) The quality of differentiated cell characterization may vary between laboratories, ranging from expression change using qPCR to in vivo proofs; (2) USC multidifferentiation potential may also depend on the USC isolation technique, culture routine and developed protocols; and (3) A variability of differentiation protocols can exist between different USC donors and/or clones as has been shown for other stem cell types[38].
Induced pluripotent stem cells (iPSCs) are artificial cells obtained through the reprogramming of a somatic cell towards an undifferentiated phenotype; reprogramming is a complex process linked with deep remodeling of the cell’s epigenetic and transcriptomic profile. By definition, iPSCs are able to self-renew indefinitely and to differentiate into all cell types from the three germ layers. Combined or not by the possibility to precisely modify their genome with genome editing approaches such as CRISPR/Cas9, iPSCs are a potential tool for many applications including personalized cell therapy, disease modeling, toxico-logy/pharmacology screening, etc.[39-42].
Huge technical progress have been made since the proof-of-concept article in 2006[43] including safer reprogramming strategies but also substrates more convenient than compared to the skin biopsy mainly used years ago. iPSCs can be derived from keratinocytes, lymphocytes and MSCs among other cell types[44]. Zhou et al[17,45] showed that iPSCs can be generated from cells present in human urine[46]. This was conceptually new and opened the possibility of using a very convenient and accessible substrate, applicable to most patients, including children, without pain or specific care issues. Urine-derived cells have been reprogrammed through several strategies including episomal transfection and mRNA transfection[47] and using feeder-free conditions[16,48,49], potentially lowering the risk of genomic integrity failure[50].
The cells have been differentiated into many cell types and used for various applications, including (as a nonexhaustive list) differentiation into cardiom-yocytes[51,52], motor neurons[53], alveolar epithelial cells[54], hepatocytes[48], retinal organoids[55] and kidney organoids[56].
Organ/tissue repair and reconstruction is one of the great challenges for the whole scientific and medical community. Cell-based tissue engineering may be an alternative for patients suffering from various acute or chronic pathologies linked with tissue lesions. Human cell therapy requires millions of cells ready to be injected, and the choice of the optimal cell population is a significant challenge[57,58]. As described in the first chapter of this review, the proliferation and differentiation capacities combined with the nonteratogenic properties of USCs make these cells or their cellular derivatives a great tool for cellular therapy, especially because these cells do not show immunoreactivity, potentially facilitating their engraftment without causing immunological rejection. Based on their multidifferentiation capacities, USCs can be used for endothelial, myogenic, osteocyte, neurocyte or epithelial regeneration[58-60].
Kidney/genitourinary repair: Kidney and genitourinary repair/bioengineering is an extensive field of research because acute kidney injury (AKI) and chronic kidney disease are major public health issues with high morbidity and mortality. Many studies demonstrated the role and benefits of adult stem cells in kidney repair and regeneration, especially regarding MSCs[61]. Indeed, MSCs have shown to be efficient to reduce renal lesions in many models including rodent models of AKI induced by cisplatin[62], gentamicin[63,64] and ischemia reperfusion[65-67] and also in chronic kidney disease models[68,69].
Importantly, proofs of concepts were also established in preclinical porcine or simian models[70-72]. With this in mind, the use of USCs of urologic origin seems to be a judicious strategy based on their extra capacity of renal differentiation compared to MSCs. Sun et al[73] showed that human USC injection improved renal function and histological damage in cisplatin-induced AKI in rats (injection into the tail vein of 2 × 106 USCs suspended in 0.2 mL of PBS at day 1 after cisplatin). The authors observed an increase of renal tubular epithelial cell proliferation and a parallel decrease of inflammatory and apoptosis markers. Interestingly, the GFP-labeled USCs were detected at day 2 and day 4 after administration in the kidney tissue, but only a small number of these cells was found at day 4[73].
Tian et al[74] demonstrated that administration of USCs to rats with ischemia reperfusion-induced AKI improved renal function. Indeed, 1 x 105 USCs mixed with 50 μL of sucrose solution (10%, w/v), 50 μL hydrogel and 100 μL PBS were injected into the upper, middle and lower cortex of the ischemic kidney. Serum creatinine and blood urea nitrogen levels were significantly decreased in the USC-treated group at day 1, and histological tubular injury score and apoptosis were significantly decreased in the USC-treated group at days 7 and 14[74]. In addition, Zhang et al[30] demonstrated the nephron-protective effect of USCs (injected into renal parenchyma 8 wk after the injury) on renal function via antioxidative stress, anti-inflammatory and antifibrotic activity in an athymic rat model of dual-injury chronic kidney disease (ischemia reperfusion injury combined with gentamicin injection). These studies demonstrated that USC therapy may constitute a new therapeutic intervention for kidney injury and its complications.
Regarding genitourinary repair, USCs have been proven to be efficient in treatment of bladder dysfunction and in cell-based urological tissue engineering[75-77]. Some teams showed that administration of USCs isolated from voided urine was an effective treatment for urogenital diseases and a potential source of cells for urological tissue reconstruction[12,59].
Diabetes and its complications: Diabetes mellitus is a major public health issue, being one of the most lethal noninfectious diseases. USCs have been used as a potential stem cell source for the treatment of urologic complications of diabetes mellitus. Ouyang et al[31] evaluated the benefits of a human USC treatment in a model of erectile dysfunction in type 2 diabetic rats. The authors showed that injection of either USCs or USCs transfected with FGF2 (USCs-FGF2) induced a significantly increased rat intracavernosal pressure and intracavernosal pressure/mean arterial pressure ratio (P < 0.01) 28 d after intracavernous injection (1 mol/L USC/0.2 mL). Although few cells were detected within the implanted sites, the paracrine effect of the USCs or USCs-FGF2 could have induced improvement of erectile function in type 2 diabetic Sprague-Dawley rats by recruiting resident cells and increasing the endothelial phenotype expression (eNOS+) in the smooth muscle.
Moreover, Dong et al[78] evaluated the reparative and protective effects of USCs on various organs and tissues affected by diabetes in a type 2 diabetic rat model (induced by high fat diet and streptozotocin). The authors demonstrated that USCs injected by the tail vein significantly alleviated the histological destruction and functional decline of pancreatic islets, myocardic left ventricle, kidney glomerulus and bladder micturition. USC treatment did not significantly decrease the fasting blood glucose, whereas this treatment reduced the fibrosis and apoptosis of the myocardium, glomerulus and detrusor.
Furthermore, Zhao et al[4] evaluated the benefits of a human USC treatment in streptozotocin (STZ)-induced diabetic mice. In this study, 1.65 × 106/ 0.2 mL PBS of human USCs transplanted into the diabetic mice pancreas prolonged the median animal survival time, improved their blood glucose and the glucose tolerance and increased the serum and pancreas insulin content. The authors suggested that the cells promoted islet vascular regeneration and pancreatic beta-cell survival.
Zhang et al[79] worked on STZ-diabetic Sprague-Dawley rats that received bilateral intracavernous injection of 1 × 106 USCs in 200 μL PBS-1X. The authors showed that the injection upregulated autophagic activity in the cavernosal endothelium, improving cavernosal endothelial and diabetes-induced erectile function. These studies demonstrated that USC therapy may constitute a new therapeutic intervention in diabetes and its complications and could be a judicious alternative to current diabetes therapies.
Combining USCs and scaffolds: Despite the observed positive paracrine effects, there is little or no evidence that the cells can graft into the host tissue after their injection. But increasing the time where the USCs are in contact with the tissue to repair may also increase their potential. Thus, injecting one scaffold previously seeded with the target stem cell type could be one valuable option. Regarding genitourinary repair specifically, the significant challenge using USCs in a reconstructive scenario is that repair may depend on the capacity of the cells to withstand urinary pressure and layered structure while maintaining their properties. Thus, in order to optimize and facilitate USC therapy, several works have shown that the use of a scaffold seeded with USCs improve their engraftment and their differentiation for urologic tissue repair.
Liu et al[5] demonstrated the feasibility of using autologous fluorescent labeled-USCs seeded on small intestinal submucosa scaffold to repair a urethral defect in a rabbit model. The authors demonstrated the presence of labeled-USCs positive for urothelial (AE1/AE3) and smooth muscle (myosin) protein markers 4 wk after engraftment. They observed that the urethral caliber, speed of urothelial regeneration, content of smooth muscle and vessel density significantly increased in the group with autologous USC-seeded small intestinal submucosa without inflammatory cell infiltration and fibrosis compared to the control group.
Bodin et al[34] showed that their 3D culture system based on porous bacterial cellulose induced favorable conditions for USC differentiation. The authors seeded these scaffolds with USC preliminary induced to differentiate into urothelial cells and smooth myogenic cells and implanted them subcutaneously into athymic mice. The authors observed the presence of both urothelial cells and smooth myogenic cells 1 mo after implantation, suggesting that this cell-based tissue engineered conduit may be useful for bladder reconstruction.
In another study, Wu et al[80] evaluated the effects of vascular endothelial growth factor (VEGF) overexpression on USCs. Human USCs overexpressing the mouse VEGF gene were mixed with human umbilical vein endothelial cells (total 5 × 106) in a collagen-I gel. These cell-containing gels were subcutaneously implanted along with six other controls into 18 athymic mice. The authors demonstrated that cell-containing gels enhanced in vivo myogenic differentiation, neovascularization (with expression of endothelial markers) and nerve regeneration within the implanted grafts. In addition, Liu et al[81] suggested that USCs expressing VEGF-165 loaded in collagen hydrogels could be used for the correction of stress urinary incontinence. The authors showed that subcutaneous implantation in nude mice induced major vascularization and improved the recruitment of resident cells, promoting myogenic phenotype differentiation and innervations.
Endothelium repair: The ability of USCs for endothelial, epithelial and myogenic differentiation could be useful for reconstruction of tissues that need neo-vascularization and a repaired vascular network, which is vital for a functional circulatory system. Liu et al[32] demonstrated that USCs could differentiate in vivo into endothelial cells. USCs incubated in collagen-I gel with or without VEGF-enveloped alginate microbeads were subcutaneously injected into nude mice. Four weeks after in vivo injection, cells formed CD31+ intricate tubular networks and presented typical tight junctions, migration and invasion ability as well as the ability to produce endothelial nitric oxide.
In addition, Liu et al[35] used human USCs combined with microbeads of alginate (containing VEGF, insulin growth factor 1, fibroblast growth factor 1, platelet-derived growth factor, hepatocyte growth factor and neural growth factor), which were mixed with the collagen type 1 gel and subcutaneously injected into nude mice. Four weeks after injection, the grafted cell survival was improved with more cells expressing myogenic and endothelial cell transcripts and markers (CD31 and Von Willebrand factor) compared to controls. Neovascularization and innervations (nerve fibers) were enhanced in USCs combined with alginate microbeads containing the cocktail of six growth factors compared to controls. Importantly, the authors suggested that host resident stem cells may have migrated into the graft tissue and then differentiated into endothelial and myogenic lineage cells induced by the growth factors released by the microbeads in particular. They also noted that USCs may possess indirect neurogenic and neuron rescue properties[35]. These results are in agreement with Sun et al[82] who demonstrated that gene modification of USCs could induce a large-scale capacity of differentiation into several lineage cells for tissue repair.
Bone regeneration: Segmental bone/cartilage defects repaired with stem cells is a highly active field of research, and USCs could serve as a potential therapy strategy. Xing et al[83] suggested that USCs loaded on a surface mineralized biphasic calcium phosphate ceramic could represent a new treatment for segmental bone defects. The authors showed that biphasic calcium phosphate ceramic loaded with USCs are effective due to their osteogenic potential for bone regeneration in repairing critical sized segmental bone defects in rabbits.
Moreover, Guan et al[33] used USCs combined with a typical bone tissue engineering scaffold, β-tricalcium phosphate, and implanted the structures into a 6-mm critical size femoral defect rat model. They demonstrated that USCs in the scaffolds could enhance new bone formation, which spanned bone defects in 5 out of 11 rats while β-tricalcium phosphate scaffold alone induced only modest bone formation. Sun et al[39] showed that these beneficial effects could be mediated by efficient proliferative and osteogenic differentiation capacities as well as secretion of agents such as bone morphogenetic protein, which facilitates tissue engineering, in USCs. The authors observed that human USCs induced bone formation (with calcium deposits, osteocalcin and osteopontin expression) in a rat model with cranial defects. The proposed mechanism included the focal adhesion kinase that could mediate bone morphogenetic protein 2-enhanced osteogenic differentiation of human USCs.
In addition, Chen et al[84] combined human USCs with hyaluronic acid (HA) and injected human USCs-HA into a rabbit knee joint with cartilage defect. Twelve weeks after the injection, histologic analyses indicated that human USCs-HA induced more neocartilage formation compared with human USCs alone or HA alone, which only induced modest cartilage regeneration.
Inflammatory diseases: Although the therapies for inflammatory bowel diseases are effective in maintaining remission, they also have many side effects[85]. Stem cells, like USCs, could be beneficial based on their immunoregulatory effect controlling inflammation[86]. Indeed, Zhou et al[87] demonstrated that implantation of USCs in a rodent model reduced inflammatory bowel diseases. Systemic administration of USCs significantly ameliorated the clinical and histopathological severity of colitis and increased the survival rate in both acute and chronic murine colitis models. Indeed, implantation of USCs led to immunomodulatory effect via downregulation of the Th1/Th17 immune responses in a PGE2-dependent manner.
Despite some studies showing that USCs are able to graft or even remain in the organ after their vascular injection (without long-term evidences), most studies showed that their protective effect is primarily mediated by paracrine mechanisms. In this view, there is growing use of a cell’s secretome instead of cells themselves. Indeed, the systemic injection of a conditioned medium from stem cells offers a significant advantage for in vivo survival by preventing the release of biomarkers from tissue damage, thereby reducing cell apoptosis and increasing proliferation[88]. These paracrine effects could be mediated by extracellular vesicles (exosomes) of cellular origin. Exosomes are vesicles of 30 to 120 nm surrounded by a double membrane[13], which could be extracted by various methods including the gold-standard ultra-centrifugation or precipitation[89-91]. They are important in intercellular communication due to richly containing miRNA, proteins, lipids, etc. that they could transfer into recipient cells[6,7,29]. Often characterized by western blot and transmission electron microscopy, exosomes expressed specific markers such as CD9, CD63, CD81 and TSG 101[13,29]. They have proproliferative properties. Indeed, Chen et al[13] demonstrated a higher proliferation of keratinocytes, fibroblasts and endothelial cells in the presence of medium supplemented with USC-exosomes compared to medium only. Exosomes also have angiogenic properties; it has been shown in a cell monolayer scratch-wound assay that USC-exosomes induced cell migration of endothelial cells[13].
Regarding tissue repair, exosomes from USCs have been tested in different clinical contexts. For bone regeneration, Chen et al[92] showed that 100 μg of human USC-extracellular vesicles (EVs) (obtained from USC-conditioned medium) effectively alleviated bone loss and maintained bone strength in osteoporotic mice (ovariectomized mice) by enhancing osteoblastic bone formation and suppressing osteoclastic bone resorption. Interestingly, the authors demonstrated that the antiosteoporotic properties of USC-EVs were not noticeably disturbed by the age, gender or health condition (with or without osteoporosis) of the USC donor, as USC-EVs from a healthy boy, healthy adult man and woman and postmenopausal osteoporotic woman could induce notable antiosteoporotic effects. In neurology, Ling et al[11] showed that exosomes from USCs promoted endogenous neurogenesis and enhanced the repair of neurological functions in post-ischemic stroke rats. Intravenous injection of 1011 total particles of USC-exosomes (in 500 µL) 4 h after transient focal cerebral ischemia reduced infarct volume and promoted functional recovery in rats from 14 d to 28 d after ischemic stroke via promoting neural cell differentiation in the subventricular zone. They demonstrated that USC-exosomes increased both proliferation and neuronal differentiation of oxygen-glucose deprivation/ reoxygenation-stimulated neural stem cells, associated with the repression of histone deacetylase 6 expression[7].
Exosomes from USCs were also linked with promising results in the field of diabetes. Jiang et al[93] showed that intravenous administration of human USCs-exosomes in an STZ-induced rat model reduced urine volume and urinary microalbumin excretion while inhibiting podocyte and tubular epithelial cell apoptosis and promoting angiogenesis of glomerular endothelial cell. In this way, Chen et al[13] demonstrated that human USC-derived exosomes accelerated cutaneous wound healing in STZ-induced diabetic mice by promoting angiogenesis from 6 d to 12 d after the wound. Moreover, Duan et al[29] demonstrated that human USC exosomes overexpressing miR-16-5p and injected into diabetic rats via tail vein conferred protective effects on podocytes in diabetic rats and tended to enhance renal function (with a decrease of circulating glucose, blood urea nitrogen, serum creatinine, creatinine clearance rate, urine protein and urine creatinine). The authors showed that this effect was mediated by VEGF-1 silencing induced by miR-16-5p overexpression.
In the field of kidney injury, Zhang et al[94] demonstrated in a murine model of ischemia reperfusion-induced AKI that extracellular vesicles from MSCs have a therapeutic effect, while Oct-4 overexpression enhanced this therapeutic effect. Additionally, Grange et al[95] showed in an AKI model that urine-derived EVs stimulated tubular cell proliferation, accelerated renal recovery by reducing the expression of injury and inflammatory markers. In this view, combining the use of USCs and exosomes for kidney repair sounds promising.
Numerous experimental studies have shown that administration of USC therapy decreases injury and tissue damage and improves tissue repair and function for many organs. Regarding cell behavior, there is little or no proof that cells can indeed graft into the tissue and differentiate in vivo. This issue could be challenging to check because finding injected labeled cells within one organ may also depend on chance. Using biocompatible and biodegradable scaffold may promote cell engraftment; here again long-term proof of concepts are necessary. However, even if USCs remain in this tissue, their true differentiation capacity still remains to be proven because most results showing USC differentiation rely on in vitro work, at times with poor cell characterization results such as gene expression modification with no differentiated cell functional analysis. USC potential for kidney/genitourinary repair is particularly appealing because these stem cells originate from this region and may therefore have some intrinsic specific properties compared to other stem cell types or to repair organs that are developmentally far from the kidney (for instance not coming from the mesoderm such as brain).
Injection timing, cell administration route and dosage are also critical factors that may be crucial to cell therapy efficacy[96] and are often considered as decided a priori despite a lack of comparative experiments in the literature.
Because urine is a convenient substrate, cell therapy strategies based on USCs could be envisaged in an autologous manner. Regarding USC compatibility, their expression of MHC type I would lead to alloreactivity if USCs from a donor are injected in a different recipient. It is yet not known whether their immunoregulatory profile would counteract this effect and promote cell tolerance; facilitating the creation of haplocompatible USC cell banking that can be fully characterized and used for cell therapy purposes would be needed.
Cell preparation is also a major issue especially because as we highlighted in the first part of this review, the precise characterization of USCs is not concordant. Generating a huge amount of USCs under Good Manufacturing Practices guidelines is another issue but that should easily be resolved based on the large experience the scientific community has with the isolation and expansion under Good Manufacturing Practices conditions of other adult stem cell types. In addition, urine cells can serve as a replacement for current MSCs (derived through invasive methods such as bone marrow harvest and liposuction) to generate a donor biobank using a noninvasive procedure for cell gathering. However, this may require further expandability and qualitative studies[58].
Using stem cell-derived microvesicles containing genetic and protein materials that upon transferring to recipient cells can activate several repair mechanisms has been shown to be efficient to repair tissue and enhance organ function.
For both strategies, these experimental studies have been performed only in small animals so the clinical pertinence is not established yet. USCs or their secreted derivatives must be evaluated in a pertinent preclinical model (pig or monkey) either in the context of tissue reconstruction, diabetes, AKI or kidney transplantation.
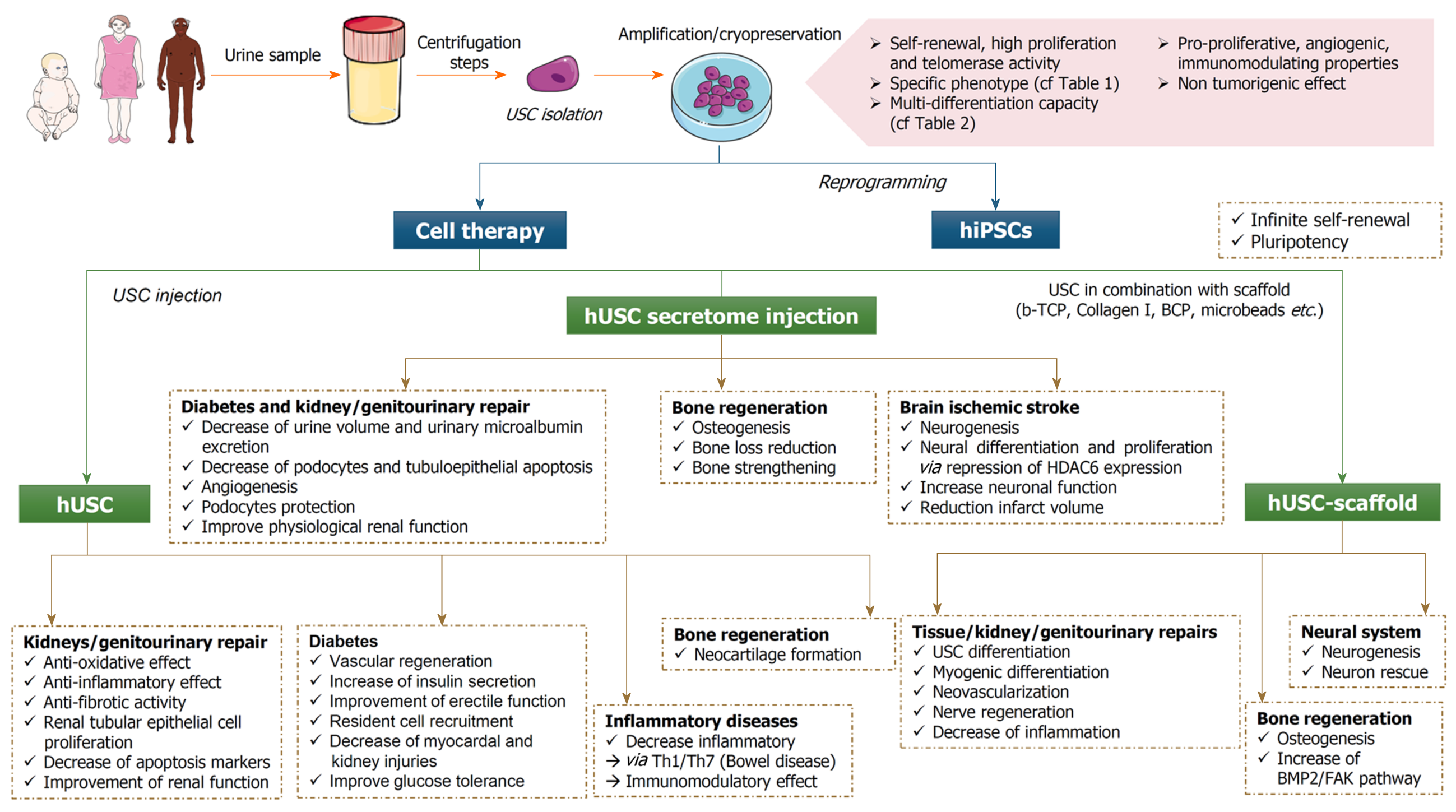
Regarding USC isolation and characterization, careful steps of protocol optimization and quality controls will be mandatory. Either used as a substrate to generate iPSCs or directly for cell therapy approaches, the potential for USCs is high and applicable for different clinical conditions ranging from stroke to diabetes. The USC secretome has also been proven to be efficient to trigger tissue repair and immunomodulation (Figure 1). Of note, USCs can also be used for disease modeling because the desired genotype/patient can be chosen to isolate the cells and based on the numerous cell types they can differentiate into. With the major advantage of being isolable from voided urine, USCs are one new player to consider for cell therapy strategy development.
Manuscript source: Invited manuscript
Specialty type: Engineering, biomedical
Country/Territory of origin: France
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
Corresponding Author’s Membership in Professional Societies: Société Francophone de Transplantation.
P-Reviewer: Cao HC, Emara KM S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Zhang YL
| 1. | Shammaa R, El-Kadiry AE, Abusarah J, Rafei M. Mesenchymal Stem Cells Beyond Regenerative Medicine. Front Cell Dev Biol. 2020;8:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 2. | Wang J, Chen Z, Sun M, Xu H, Gao Y, Liu J, Li M. Characterization and therapeutic applications of mesenchymal stem cells for regenerative medicine. Tissue Cell. 2020;64:101330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 3. | Baer PC, Geiger H. Adipose-derived mesenchymal stromal/stem cells: tissue localization, characterization, and heterogeneity. Stem Cells Int. 2012;2012:812693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 277] [Cited by in RCA: 333] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 4. | Zhao T, Luo D, Sun Y, Niu X, Wang Y, Wang C, Jia W. Human urine-derived stem cells play a novel role in the treatment of STZ-induced diabetic mice. J Mol Histol. 2018;49:419-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 5. | Liu Y, Ma W, Liu B, Wang Y, Chu J, Xiong G, Shen L, Long C, Lin T, He D, Butnaru D, Alexey L, Zhang Y, Zhang D, Wei G. Urethral reconstruction with autologous urine-derived stem cells seeded in three-dimensional porous small intestinal submucosa in a rabbit model. Stem Cell Res Ther. 2017;8:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (1)] |
| 6. | Tao SC, Yuan T, Zhang YL, Yin WJ, Guo SC, Zhang CQ. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics. 2017;7:180-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 380] [Cited by in RCA: 550] [Article Influence: 68.8] [Reference Citation Analysis (0)] |
| 7. | Ling X, Zhang G, Xia Y, Zhu Q, Zhang J, Li Q, Niu X, Hu G, Yang Y, Wang Y, Deng Z. Exosomes from human urine-derived stem cells enhanced neurogenesis via miR-26a/HDAC6 axis after ischaemic stroke. J Cell Mol Med. 2020;24:640-654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 8. | Linder D. Culture of cells from the urine and bladder washings of adults. Somatic Cell Genet. 1976;2:281-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Hintz DS, Sens MA, Jenkins MQ, Sens DA. Tissue culture of epithelial cells from urine. I. Serum-free growth of cells from newborn infants. Pediatr Pathol. 1984;2:153-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 10. | Sutherland GR, Bain AD. Culture of cells from the urine of newborn children. Nature. 1972;239:231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Zhang Y, McNeill E, Tian H, Soker S, Andersson KE, Yoo JJ, Atala A. Urine derived cells are a potential source for urological tissue reconstruction. J Urol. 2008;180:2226-2233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 266] [Article Influence: 15.6] [Reference Citation Analysis (2)] |
| 12. | Bharadwaj S, Liu G, Shi Y, Markert C, Andersson KE, Atala A, Zhang Y. Characterization of urine-derived stem cells obtained from upper urinary tract for use in cell-based urological tissue engineering. Tissue Eng Part A. 2011;17:2123-2132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 134] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 13. | Chen CY, Rao SS, Ren L, Hu XK, Tan YJ, Hu Y, Luo J, Liu YW, Yin H, Huang J, Cao J, Wang ZX, Liu ZZ, Liu HM, Tang SY, Xu R, Xie H. Exosomal DMBT1 from human urine-derived stem cells facilitates diabetic wound repair by promoting angiogenesis. Theranostics. 2018;8:1607-1623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 284] [Article Influence: 40.6] [Reference Citation Analysis (0)] |
| 14. | Lazzeri E, Ronconi E, Angelotti ML, Peired A, Mazzinghi B, Becherucci F, Conti S, Sansavini G, Sisti A, Ravaglia F, Lombardi D, Provenzano A, Manonelles A, Cruzado JM, Giglio S, Roperto RM, Materassi M, Lasagni L, Romagnani P. Human Urine-Derived Renal Progenitors for Personalized Modeling of Genetic Kidney Disorders. J Am Soc Nephrol. 2015;26:1961-1974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 15. | Schosserer M, Reynoso R, Wally V, Jug B, Kantner V, Weilner S, Buric I, Grillari J, Bauer JW, Grillari-Voglauer R. Urine is a novel source of autologous mesenchymal stem cells for patients with epidermolysis bullosa. BMC Res Notes. 2015;8:767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Steichen C, Si-Tayeb K, Wulkan F, Crestani T, Rosas G, Dariolli R, Pereira AC, Krieger JE. Human Induced Pluripotent Stem (hiPS) Cells from Urine Samples: A Non-Integrative and Feeder-Free Reprogramming Strategy. Curr Protoc Hum Genet. 2017;92:21.7.1-21.7.22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 17. | Zhou T, Benda C, Dunzinger S, Huang Y, Ho JC, Yang J, Wang Y, Zhang Y, Zhuang Q, Li Y, Bao X, Tse HF, Grillari J, Grillari-Voglauer R, Pei D, Esteban MA. Generation of human induced pluripotent stem cells from urine samples. Nat Protoc. 2012;7:2080-2089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 433] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 18. | He W, Zhu W, Cao Q, Shen Y, Zhou Q, Yu P, Liu X, Ma J, Li Y, Hong K. Generation of Mesenchymal-Like Stem Cells From Urine in Pediatric Patients. Transplant Proc. 2016;48:2181-2185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 19. | Lang R, Liu G, Shi Y, Bharadwaj S, Leng X, Zhou X, Liu H, Atala A, Zhang Y. Self-renewal and differentiation capacity of urine-derived stem cells after urine preservation for 24 h. PLoS One. 2013;8:e53980. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 96] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 20. | Chen AJ, Pi JK, Hu JG, Huang YZ, Gao HW, Li SF, Li-Ling J, Xie HQ. Identification and characterization of two morphologically distinct stem cell subpopulations from human urine samples. Sci China Life Sci. 2020;63:712-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 21. | Wan Q, Xiong G, Liu G, Shupe TD, Wei G, Zhang D, Liang D, Lu X, Atala A, Zhang Y. Urothelium with barrier function differentiated from human urine-derived stem cells for potential use in urinary tract reconstruction. Stem Cell Res Ther. 2018;9:304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 22. | Guan JJ, Niu X, Gong FX, Hu B, Guo SC, Lou YL, Zhang CQ, Deng ZF, Wang Y. Biological characteristics of human-urine-derived stem cells: potential for cell-based therapy in neurology. Tissue Eng Part A. 2014;20:1794-1806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 23. | Bharadwaj S, Liu G, Shi Y, Wu R, Yang B, He T, Fan Y, Lu X, Zhou X, Liu H, Atala A, Rohozinski J, Zhang Y. Multipotential differentiation of human urine-derived stem cells: potential for therapeutic applications in urology. Stem Cells. 2013;31:1840-1856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 228] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 24. | Kang HS, Choi SH, Kim BS, Choi JY, Park GB, Kwon TG, Chun SY. Advanced Properties of Urine Derived Stem Cells Compared to Adipose Tissue Derived Stem Cells in Terms of Cell Proliferation, Immune Modulation and Multi Differentiation. J Korean Med Sci. 2015;30:1764-1776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 72] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 25. | Zhang XR, Huang YZ, Gao HW, Jiang YL, Hu JG, Pi JK, Chen AJ, Zhang Y, Zhou L, Xie HQ. Hypoxic preconditioning of human urine-derived stem cell-laden small intestinal submucosa enhances wound healing potential. Stem Cell Res Ther. 2020;11:150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 26. | Chun SY, Kim HT, Kwon SY, Kim J, Kim BS, Yoo ES, Kwon TG. The efficacy and safety of Collagen-I and hypoxic conditions in urine-derived stem cell ex vivo culture. Tissue Eng Regen Med. 2016;13:403-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 27. | Rahman MS, Wruck W, Spitzhorn LS, Nguyen L, Bohndorf M, Martins S, Asar F, Ncube A, Erichsen L, Graffmann N, Adjaye J. The FGF, TGFβ and WNT axis Modulate Self-renewal of Human SIX2+ Urine Derived Renal Progenitor Cells. Sci Rep. 2020;10:739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 28. | Chun SY, Kim HT, Lee JS, Kim MJ, Kim BS, Kim BW, Kwon TG. Characterization of urine-derived cells from upper urinary tract in patients with bladder cancer. Urology. 2012;79:1186.e1-1186.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 29. | Duan YR, Chen BP, Chen F, Yang SX, Zhu CY, Ma YL, Li Y, Shi J. Exosomal microRNA-16-5p from human urine-derived stem cells ameliorates diabetic nephropathy through protection of podocyte. J Cell Mol Med. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 30. | Zhang C, George SK, Wu R, Thakker PU, Abolbashari M, Kim TH, Ko IK, Zhang Y, Sun Y, Jackson J, Lee SJ, Yoo JJ, Atala A. Reno-protection of Urine-derived Stem Cells in A Chronic Kidney Disease Rat Model Induced by Renal Ischemia and Nephrotoxicity. Int J Biol Sci. 2020;16:435-446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 31. | Ouyang B, Sun X, Han D, Chen S, Yao B, Gao Y, Bian J, Huang Y, Zhang Y, Wan Z, Yang B, Xiao H, Songyang Z, Liu G, Zhang Y, Deng C. Human urine-derived stem cells alone or genetically-modified with FGF2 Improve type 2 diabetic erectile dysfunction in a rat model. PLoS One. 2014;9:e92825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 95] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 32. | Liu G, Wu R, Yang B, Deng C, Lu X, Walker SJ, Ma PX, Mou S, Atala A, Zhang Y. Human Urine-Derived Stem Cell Differentiation to Endothelial Cells with Barrier Function and Nitric Oxide Production. Stem Cells Transl Med. 2018;7:686-698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 33. | Guan J, Zhang J, Li H, Zhu Z, Guo S, Niu X, Wang Y, Zhang C. Human Urine Derived Stem Cells in Combination with β-TCP Can Be Applied for Bone Regeneration. PLoS One. 2015;10:e0125253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 34. | Bodin A, Bharadwaj S, Wu S, Gatenholm P, Atala A, Zhang Y. Tissue-engineered conduit using urine-derived stem cells seeded bacterial cellulose polymer in urinary reconstruction and diversion. Biomaterials. 2010;31:8889-8901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 164] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 35. | Liu G, Pareta RA, Wu R, Shi Y, Zhou X, Liu H, Deng C, Sun X, Atala A, Opara EC, Zhang Y. Skeletal myogenic differentiation of urine-derived stem cells and angiogenesis using microbeads loaded with growth factors. Biomaterials. 2013;34:1311-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 94] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 36. | Wu S, Liu Y, Bharadwaj S, Atala A, Zhang Y. Human urine-derived stem cells seeded in a modified 3D porous small intestinal submucosa scaffold for urethral tissue engineering. Biomaterials. 2011;32:1317-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 137] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 37. | Xu G, Wu F, Gu X, Zhang J, You K, Chen Y, Getachew A, Zhuang Y, Zhong X, Lin Z, Guo D, Yang F, Pan T, Wei H, Li YX. Direct Conversion of Human Urine Cells to Neurons by Small Molecules. Sci Rep. 2019;9:16707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 38. | Cahan P, Daley GQ. Origins and implications of pluripotent stem cell variability and heterogeneity. Nat Rev Mol Cell Biol. 2013;14:357-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 244] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 39. | Sun X, Zheng W, Qian C, Wu Q, Hao Y, Lu G. Focal adhesion kinase promotes BMP2-induced osteogenic differentiation of human urinary stem cells via AMPK and Wnt signaling pathways. J Cell Physiol. 2020;235:4954-4964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 40. | Bragança J, Lopes JA, Mendes-Silva L, Almeida Santos JM. Induced pluripotent stem cells, a giant leap for mankind therapeutic applications. World J Stem Cells. 2019;11:421-430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (4)] |
| 41. | Ohnuki M, Takahashi K. Present and future challenges of induced pluripotent stem cells. Philos Trans R Soc Lond B Biol Sci. 2015;370:20140367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 120] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 42. | Hockemeyer D, Jaenisch R. Induced Pluripotent Stem Cells Meet Genome Editing. Cell Stem Cell. 2016;18:573-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 364] [Article Influence: 45.5] [Reference Citation Analysis (0)] |
| 43. | Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17989] [Cited by in RCA: 18206] [Article Influence: 958.2] [Reference Citation Analysis (0)] |
| 44. | Raab S, Klingenstein M, Liebau S, Linta L. A Comparative View on Human Somatic Cell Sources for iPSC Generation. Stem Cells Int. 2014;2014:768391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 168] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 45. | Zhou T, Benda C, Duzinger S, Huang Y, Li X, Li Y, Guo X, Cao G, Chen S, Hao L, Chan YC, Ng KM, Ho JC, Wieser M, Wu J, Redl H, Tse HF, Grillari J, Grillari-Voglauer R, Pei D, Esteban MA. Generation of induced pluripotent stem cells from urine. J Am Soc Nephrol. 2011;22:1221-1228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 315] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 46. | Xue Y, Cai X, Wang L, Liao B, Zhang H, Shan Y, Chen Q, Zhou T, Li X, Hou J, Chen S, Luo R, Qin D, Pei D, Pan G. Generating a non-integrating human induced pluripotent stem cell bank from urine-derived cells. PLoS One. 2013;8:e70573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 47. | Gaignerie A, Lefort N, Rousselle M, Forest-Choquet V, Flippe L, Francois-Campion V, Girardeau A, Caillaud A, Chariau C, Francheteau Q, Derevier A, Chaubron F, Knöbel S, Gaborit N, Si-Tayeb K, David L. Urine-derived cells provide a readily accessible cell type for feeder-free mRNA reprogramming. Sci Rep. 2018;8:14363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 48. | Si-Tayeb K, Idriss S, Champon B, Caillaud A, Pichelin M, Arnaud L, Lemarchand P, Le May C, Zibara K, Cariou B. Urine-sample-derived human induced pluripotent stem cells as a model to study PCSK9-mediated autosomal dominant hypercholesterolemia. Dis Model Mech. 2016;9:81-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 49. | Jia B, Chen S, Zhao Z, Liu P, Cai J, Qin D, Du J, Wu C, Chen Q, Cai X, Zhang H, Yu Y, Pei D, Zhong M, Pan G. Modeling of hemophilia A using patient-specific induced pluripotent stem cells derived from urine cells. Life Sci. 2014;108:22-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 50. | Steichen C, Hannoun Z, Luce E, Hauet T, Dubart-Kupperschmitt A. Genomic integrity of human induced pluripotent stem cells: Reprogramming, differentiation and applications. World J Stem Cells. 2019;11:729-747. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 51. | Lin YH, Chen XM, Zhang JW, He XQ, Dai WJ, Chen MS. Preclinical study on induction of pluripotent stem cells from urine of dilated cardiomyopathy patients. Eur Rev Med Pharmacol Sci. 2016;20:1450-1457. [PubMed] |
| 52. | Jouni M, Si-Tayeb K, Es-Salah-Lamoureux Z, Latypova X, Champon B, Caillaud A, Rungoat A, Charpentier F, Loussouarn G, Baró I, Zibara K, Lemarchand P, Gaborit N. Toward Personalized Medicine: Using Cardiomyocytes Differentiated From Urine-Derived Pluripotent Stem Cells to Recapitulate Electrophysiological Characteristics of Type 2 Long QT Syndrome. J Am Heart Assoc. 2015;4:e002159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 53. | Yi H, Xie B, Liu B, Wang X, Xu L, Liu J, Li M, Zhong X, Peng F. Derivation and Identification of Motor Neurons from Human Urine-Derived Induced Pluripotent Stem Cells. Stem Cells Int. 2018;2018:3628578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 54. | Wang C, Hei F, Ju Z, Yu J, Yang S, Chen M. Differentiation of Urine-Derived Human Induced Pluripotent Stem Cells to Alveolar Type II Epithelial Cells. Cell Reprogram. 2016;18:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 55. | Li G, Xie B, He L, Zhou T, Gao G, Liu S, Pan G, Ge J, Peng F, Zhong X. Generation of Retinal Organoids with Mature Rods and Cones from Urine-Derived Human Induced Pluripotent Stem Cells. Stem Cells Int. 2018;2018:4968658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 56. | Mulder J, Sharmin S, Chow T, Rodrigues DC, Hildebrandt MR, D'Cruz R, Rogers I, Ellis J, Rosenblum ND. Generation of infant- and pediatric-derived urinary induced pluripotent stem cells competent to form kidney organoids. Pediatr Res. 2020;87:647-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 57. | Lai MI, Wendy-Yeo WY, Ramasamy R, Nordin N, Rosli R, Veerakumarasivam A, Abdullah S. Advancements in reprogramming strategies for the generation of induced pluripotent stem cells. J Assist Reprod Genet. 2011;28:291-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 58. | Pavathuparambil Abdul Manaph N, Al-Hawwas M, Bobrovskaya L, Coates PT, Zhou XF. Urine-derived cells for human cell therapy. Stem Cell Res Ther. 2018;9:189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 59. | Zhang D, Wei G, Li P, Zhou X, Zhang Y. Urine-derived stem cells: A novel and versatile progenitor source for cell-based therapy and regenerative medicine. Genes Dis. 2014;1:8-17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 105] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 60. | Gao P, Jiang D, Liu W, Li H, Li Z. Urine-derived Stem Cells, A New Source of Seed Cells for Tissue Engineering. Curr Stem Cell Res Ther. 2016;11:547-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 61. | Bochon B, Kozubska M, Surygała G, Witkowska A, Kuźniewicz R, Grzeszczak W, Wystrychowski G. Mesenchymal Stem Cells-Potential Applications in Kidney Diseases. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 62. | Elhusseini FM, Saad MA, Anber N, Elghannam D, Sobh MA, Alsayed A, El-Dusoky S, Sheashaa H, Abdel-Ghaffar H, Sobh M. Long Term Study of Protective Mechanisms of Human Adipose Derived Mesenchymal Stem Cells on Cisplatin Induced Kidney injury in Sprague-Dawley Rats. J Stem Cells Regen Med. 2016;12:36-48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 63. | Liu P, Feng Y, Dong C, Yang D, Li B, Chen X, Zhang Z, Wang Y, Zhou Y, Zhao L. Administration of BMSCs with muscone in rats with gentamicin-induced AKI improves their therapeutic efficacy. PLoS One. 2014;9:e97123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 64. | Liu P, Feng Y, Dong D, Liu X, Chen Y, Wang Y, Zhou Y. Enhanced renoprotective effect of IGF-1 modified human umbilical cord-derived mesenchymal stem cells on gentamicin-induced acute kidney injury. Sci Rep. 2016;6:20287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 65. | Sung PH, Chiang HJ, Wallace CG, Yang CC, Chen YT, Chen KH, Chen CH, Shao PL, Chen YL, Chua S, Chai HT, Chen YL, Huang TH, Yip HK, Lee MS. Exendin-4-assisted adipose derived mesenchymal stem cell therapy protects renal function against co-existing acute kidney ischemia-reperfusion injury and severe sepsis syndrome in rat. Am J Transl Res. 2017;9:3167-3183. [PubMed] |
| 66. | Monteiro Carvalho Mori da Cunha MG, Zia S, Oliveira Arcolino F, Carlon MS, Beckmann DV, Pippi NL, Luhers Graça D, Levtchenko E, Deprest J, Toelen J. Amniotic Fluid Derived Stem Cells with a Renal Progenitor Phenotype Inhibit Interstitial Fibrosis in Renal Ischemia and Reperfusion Injury in Rats. PLoS One. 2015;10:e0136145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 67. | Ko SF, Chen YT, Wallace CG, Chen KH, Sung PH, Cheng BC, Huang TH, Chen YL, Li YC, Chang HW, Lee MS, Yang CC, Yip HK. Inducible pluripotent stem cell-derived mesenchymal stem cell therapy effectively protected kidney from acute ischemia-reperfusion injury. Am J Transl Res. 2018;10:3053-3067. [PubMed] |
| 68. | Geng X, Hong Q, Chi K, Wang S, Cai G, Wu D. Mesenchymal Stem Cells Loaded with Gelatin Microcryogels Attenuate Renal Fibrosis. Biomed Res Int. 2019;2019:6749326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 69. | Sheu JJ, Sung PH, Wallace CG, Yang CC, Chen KH, Shao PL, Chu YC, Huang CR, Chen YL, Ko SF, Lee MS, Yip HK. Intravenous administration of iPS-MSCSPIONs mobilized into CKD parenchyma and effectively preserved residual renal function in CKD rat. J Cell Mol Med. 2020;24:3593-3610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 70. | Moghadasali R, Azarnia M, Hajinasrollah M, Arghani H, Nassiri SM, Molazem M, Vosough A, Mohitmafi S, Najarasl M, Ajdari Z, Yazdi RS, Bagheri M, Ghanaati H, Rafiei B, Gheisari Y, Baharvand H, Aghdami N. Intra-renal arterial injection of autologous bone marrow mesenchymal stromal cells ameliorates cisplatin-induced acute kidney injury in a rhesus Macaque mulatta monkey model. Cytotherapy. 2014;16:734-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 71. | Baulier E, Favreau F, Le Corf A, Jayle C, Schneider F, Goujon JM, Feraud O, Bennaceur-Griscelli A, Hauet T, Turhan AG. Amniotic fluid-derived mesenchymal stem cells prevent fibrosis and preserve renal function in a preclinical porcine model of kidney transplantation. Stem Cells Transl Med. 2014;3:809-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 72. | Moghadasali R, Hajinasrollah M, Argani H, Nassiri SM, Najarasl M, Sodeifi N, Baharvand H, Aghdami N. Autologous transplantation of mesenchymal stromal cells tends to prevent progress of interstitial fibrosis in a rhesus Macaca mulatta monkey model of chronic kidney disease. Cytotherapy. 2015;17:1495-1505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 73. | Sun B, Luo X, Yang C, Liu P, Yang Y, Dong X, Yang Z, Xu J, Zhang Y, Li L. Therapeutic Effects of Human Urine-Derived Stem Cells in a Rat Model of Cisplatin-Induced Acute Kidney Injury In Vivo and In Vitro. Stem Cells Int. 2019;2019:8035076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 74. | Tian SF, Jiang ZZ, Liu YM, Niu X, Hu B, Guo SC, Wang NS, Wang Y. Human urine-derived stem cells contribute to the repair of ischemic acute kidney injury in rats. Mol Med Rep. 2017;16:5541-5548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 75. | Zou Q, Fu Q. Tissue engineering for urinary tract reconstruction and repair: Progress and prospect in China. Asian J Urol. 2018;5:57-68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 76. | Culenova M, Ziaran S, Danisovic L. Cells Involved in Urethral Tissue Engineering: Systematic Review. Cell Transplant. 2019;28:1106-1115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 77. | Qin D, Long T, Deng J, Zhang Y. Urine-derived stem cells for potential use in bladder repair. Stem Cell Res Ther. 2014;5:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 78. | Dong X, Zhang T, Liu Q, Zhu J, Zhao J, Li J, Sun B, Ding G, Hu X, Yang Z, Zhang Y, Li L. Beneficial effects of urine-derived stem cells on fibrosis and apoptosis of myocardial, glomerular and bladder cells. Mol Cell Endocrinol. 2016;427:21-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 79. | Zhang C, Luo D, Li T, Yang Q, Xie Y, Chen H, Lv L, Yao J, Deng C, Liang X, Wu R, Sun X, Zhang Y, Deng C, Liu G. Transplantation of Human Urine-Derived Stem Cells Ameliorates Erectile Function and Cavernosal Endothelial Function by Promoting Autophagy of Corpus Cavernosal Endothelial Cells in Diabetic Erectile Dysfunction Rats. Stem Cells Int. 2019;2019:2168709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 80. | Wu S, Wang Z, Bharadwaj S, Hodges SJ, Atala A, Zhang Y. Implantation of autologous urine derived stem cells expressing vascular endothelial growth factor for potential use in genitourinary reconstruction. J Urol. 2011;186:640-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 81. | Liu G, Wang X, Sun X, Deng C, Atala A, Zhang Y. The effect of urine-derived stem cells expressing VEGF loaded in collagen hydrogels on myogenesis and innervation following after subcutaneous implantation in nude mice. Biomaterials. 2013;34:8617-8629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 82. | Sun B, Dong X, Zhao J, Yang Z, Zhang Y, Li L. Differentiation of human urine-derived stem cells into interstitial cells of Cajal-like cells by exogenous gene modification: A preliminary study. Biochem Biophys Res Commun. 2020;523:10-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 83. | Xing F, Li L, Sun J, Liu G, Duan X, Chen J, Liu M, Long Y, Xiang Z. Surface mineralized biphasic calcium phosphate ceramics loaded with urine-derived stem cells are effective in bone regeneration. J Orthop Surg Res. 2019;14:419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 84. | Chen L, Li L, Xing F, Peng J, Peng K, Wang Y, Xiang Z. Human Urine-Derived Stem Cells: Potential for Cell-Based Therapy of Cartilage Defects. Stem Cells Int. 2018;2018:4686259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 85. | Reenaers C, Belaiche J, Louis E. Impact of medical therapies on inflammatory bowel disease complication rate. World J Gastroenterol. 2012;18:3823-3827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 86. | Chen QQ, Yan L, Wang CZ, Wang WH, Shi H, Su BB, Zeng QH, Du HT, Wan J. Mesenchymal stem cells alleviate TNBS-induced colitis by modulating inflammatory and autoimmune responses. World J Gastroenterol. 2013;19:4702-4717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 84] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 87. | Zhou C, Wu XR, Liu HS, Liu XH, Liu GH, Zheng XB, Hu T, Liang ZX, He XW, Wu XJ, Smith LC, Zhang Y, Lan P. Immunomodulatory Effect of Urine-derived Stem Cells on Inflammatory Bowel Diseases via Downregulating Th1/Th17 Immune Responses in a PGE2-dependent Manner. J Crohns Colitis. 2020;14:654-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 88. | van Poll D, Parekkadan B, Cho CH, Berthiaume F, Nahmias Y, Tilles AW, Yarmush ML. Mesenchymal stem cell-derived molecules directly modulate hepatocellular death and regeneration in vitro and in vivo. Hepatology. 2008;47:1634-1643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 390] [Cited by in RCA: 411] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 89. | Théry C, Amigorena S, Raposo G, Clayton A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. 2006;Chapter 3:Unit 3.22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2443] [Cited by in RCA: 3727] [Article Influence: 196.2] [Reference Citation Analysis (0)] |
| 90. | Merchant ML, Powell DW, Wilkey DW, Cummins TD, Deegens JK, Rood IM, McAfee KJ, Fleischer C, Klein E, Klein JB. Microfiltration isolation of human urinary exosomes for characterization by MS. Proteomics Clin Appl. 2010;4:84-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 165] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 91. | Cheruvanky A, Zhou H, Pisitkun T, Kopp JB, Knepper MA, Yuen PS, Star RA. Rapid isolation of urinary exosomal biomarkers using a nanomembrane ultrafiltration concentrator. Am J Physiol Renal Physiol. 2007;292:F1657-F1661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 383] [Cited by in RCA: 363] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 92. | Chen CY, Rao SS, Tan YJ, Luo MJ, Hu XK, Yin H, Huang J, Hu Y, Luo ZW, Liu ZZ, Wang ZX, Cao J, Liu YW, Li HM, Chen Y, Du W, Liu JH, Zhang Y, Chen TH, Liu HM, Wu B, Yue T, Wang YY, Xia K, Lei PF, Tang SY, Xie H. Extracellular vesicles from human urine-derived stem cells prevent osteoporosis by transferring CTHRC1 and OPG. Bone Res. 2019;7:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 93. | Jiang ZZ, Liu YM, Niu X, Yin JY, Hu B, Guo SC, Fan Y, Wang Y, Wang NS. Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res Ther. 2016;7:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 208] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 94. | Zhang ZY, Hou YP, Zou XY, Xing XY, Ju GQ, Zhong L, Sun J. Oct-4 Enhanced the Therapeutic Effects of Mesenchymal Stem Cell-Derived Extracellular Vesicles in Acute Kidney Injury. Kidney Blood Press Res. 2020;45:95-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 95. | Grange C, Papadimitriou E, Dimuccio V, Pastorino C, Molina J, O'Kelly R, Niedernhofer LJ, Robbins PD, Camussi G, Bussolati B. Urinary Extracellular Vesicles Carrying Klotho Improve the Recovery of Renal Function in an Acute Tubular Injury Model. Mol Ther. 2020;28:490-502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 96. | Cai J, Yu X, Xu R, Fang Y, Qian X, Liu S, Teng J, Ding X. Maximum efficacy of mesenchymal stem cells in rat model of renal ischemia-reperfusion injury: renal artery administration with optimal numbers. PLoS One. 2014;9:e92347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |