Published online Jan 21, 2023. doi: 10.3748/wjg.v29.i3.503
Peer-review started: September 13, 2022
First decision: October 19, 2022
Revised: November 30, 2022
Accepted: December 21, 2022
Article in press: December 21, 2022
Published online: January 21, 2023
Processing time: 121 Days and 4.4 Hours
Although different studies have associated coronavirus disease 2019 (COVID-19) with the occurrence of liver injury, the hepatic injury route during the COVID-19 course is not yet fully understood. In order to better understand the mechanisms of the disease, the human gut microbiota has been the subject of extensive discussion in the context of COVID-19 pathophysiology. However, many questions remain, including the risks of liver injury due to COVID-19 specific populations. Further research in this field could allow the discovery of new personalized treatment strategies aimed at improving the microbiota composition, thereby reducing COVID-19 severity and its complications in different populations. In this article, we discussed basic mechanisms of severe acute respiratory syndrome coronavirus 2 infection and recent evidence on the relationship between COVID-19, the gut microbiome and liver injury as well as proposed recommendations for further research.
Core Tip: Although different studies have associated coronavirus disease 2019 (COVID-19) with the occurrence of liver injury, the hepatic injury route during the COVID-19 course is not yet fully understood. Further research is needed to better understand the impacts of changes of the gut microbiota and immunology of COVID-19.
- Citation: Tovani-Palone MR, Pedersini P. Potential role of the microbiome in liver injury during COVID-19: Further research is needed. World J Gastroenterol 2023; 29(3): 503-507
- URL: https://www.wjgnet.com/1007-9327/full/v29/i3/503.htm
- DOI: https://dx.doi.org/10.3748/wjg.v29.i3.503
The gut-liver axis is a well-described bidirectional relationship where a mutual interaction between gut and liver microbiota occurs. It has attracted significant attention in the context of coronavirus disease 2019 (COVID-19). This close anatomical and functional relationship between the gut and its microbiota and liver function results from an interaction between genetic and environmental factors, including diet, medicine use and diseases[1]. Although the human gut microbiota is recognized to have an important role for immunity and protection against pathogens, its diversity decreases in old age, which is the age group with the highest mortality from COVID-19[2]. This suggests a potential protection of balanced gut-liver axis against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, which should be of interest to prevent and reduce the number of fatal cases of COVID-19. On the other hand, any imbalance of this microbiome should affect immunity as well as viral activity against SARS-CoV-2[3]. Moreover, different studies have also reported the occurrence of liver injury to varying degrees in COVID-19 patients, which could be associated with important changes in both the gut-liver axis microbiota and responses at the cellular and molecular level[4,5]. However, research on the risks of liver injury due to COVID-19 in many specific populations is still scarce. Here we discussed basic mechanisms of SARS-CoV-2 infection and recent evidence on the relationship between COVID-19, the gut microbiome and liver injury as well as proposed recommendations for further research.
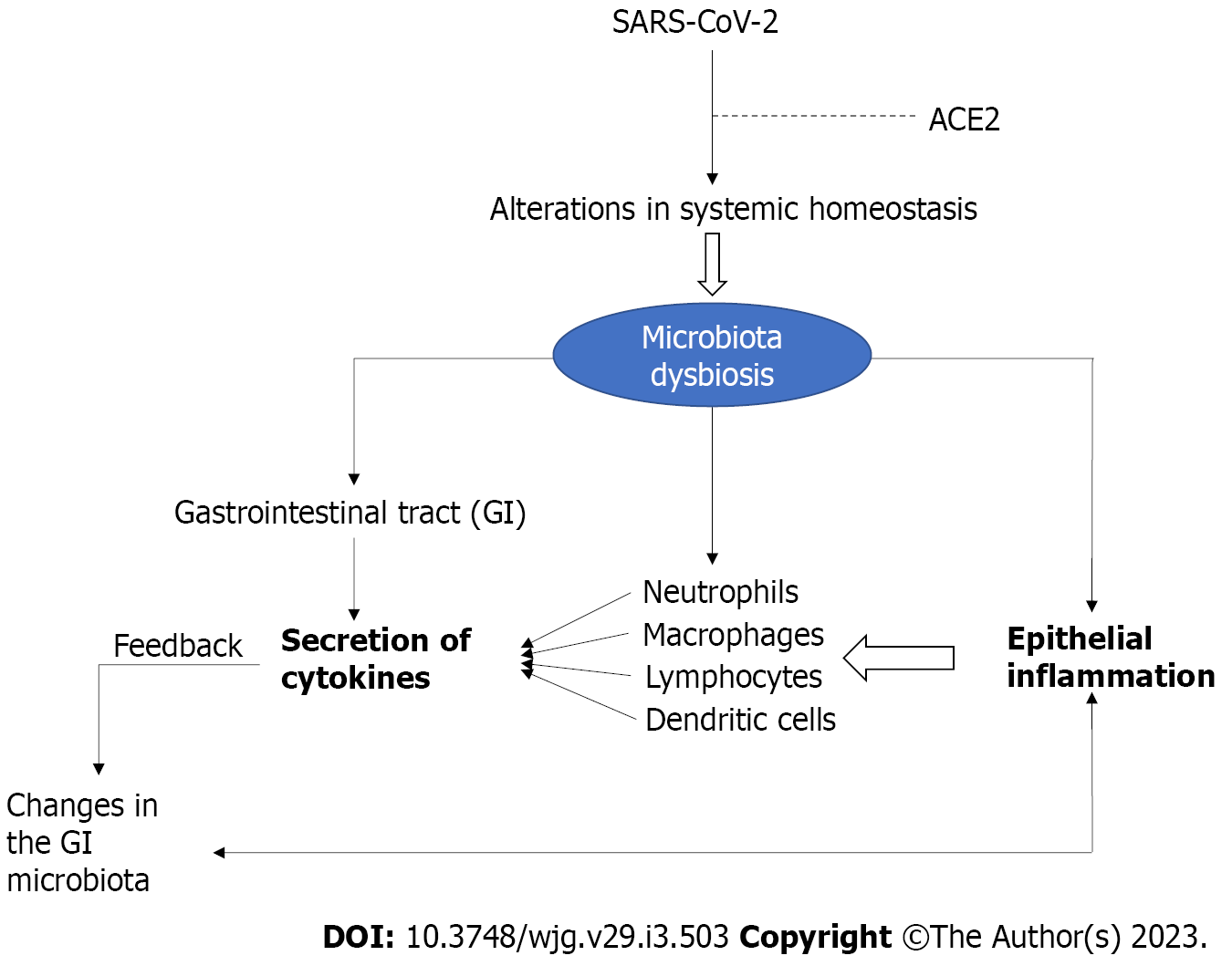
There is a consensus among most scientists that the cellular entry of SARS-CoV-2 primarily occurs via high-affinity interactions between the receptor-binding domain of the SARS-CoV-2 spike protein and the angiotensin converting enzyme 2 (ACE2) receptor, in addition to other molecules[4-7]. This receptor has been identified in different and important organs, including the surface of respiratory tract epithelium, epithelial cells of the upper esophagus, enterocytes of the ileum and colon, in the heart, testicles, cells of smooth muscles, the endothelium of pancreatic, brain and kidney blood vessels[4], and in bile duct epithelial cell and liver[5]. The resulting downregulation of ACE2 activity may lead to an increase in angiotensin 2 through ACE. This is due to the fact that the decrease in ACE2 is associated with a lower conversion of angiotensin to angiotensin 1-7 vasodilator. Thus, there is a gradual tendency towards an increase in plasma concentrations of angiotensin I and angiotensin II, causing an imbalance in the renin-angiotensin system as well as a consequent deregulation of systemic homeostasis[6,8].
According to general statistics, about half of COVID-19 patients are expected to have at least one of these gastrointestinal symptoms: diarrhea, nausea, vomiting, and abdominal pain[4,5]. Research has shown that the ACE2 receptor is the main gateway for SARS-CoV-2 into epithelial cells of the gastrointestinal tract. This receptor is in turn highly expressed on epithelial cells in the small intestine. In addition to the decrease in ACE2 receptor expression due to the invasion of SARS-CoV-2, important changes in the gut microbiota involving different microorganisms (dysbiosis) may also occur (Figure 1), affecting the function of the intestinal barrier and the permeability and homeostatic balance of metabolites in the gut lumen[8,9].
It is also hypothesized that SARS-CoV-2 infection of epithelial cells in the gut, especially in the small intestine, could result in malnutrition as well as potentiate the associated dysbiosis, leading to impaired gut barrier function and systemic inflammation. This in turn may create a positive feedback loop for increased translocation of gut microbes into the systemic circulation and potentiation of inflammation, culminating in systemic inflammation and cytokine storm that may contribute to both worsening gut and systemic damage as well as increasing the severity of COVID-19[8,9]. Therefore, in addition to the classic gastrointestinal disorders and symptoms of COVID-19, accessory digestive organs such as the liver can be affected, as a result of the worsening infection[4].
Although COVID-19 has been associated with liver injury, the hepatic injury route during the COVID-19 course is not yet fully understood. It is believed that such injury is due to specific pathogenic mechanisms of the virus or even the use of hepatotoxic drugs[3,4]. Among the different etiological hypotheses described in the literature in order to advance knowledge about this topic the following stand out: (1) Liver injury resulting from a direct virus cytopathic effect by lysis or by inducing apoptosis; (2) Immune-mediated liver injury from proinflammatory cytokines (interleukin-1, interleukin-6, tumor necrosis factor, chemokines, and inflammatory cells produced against SARS-CoV-2); (3) Liver injury resulting from viral-induced cytotoxic T cells (CD8); (4) Liver injury due to the use of drugs including antivirals, anti-inflammatory drugs, anticoagulants, antibiotics, and drugs used for chronic diseases during SARS-CoV-2 infection; (5) Liver injury caused by hypoxia resulting from pneumonia[4,5]; and (6) Liver injury resulting from the gut vascular barrier and dysbiosis due to the indirect effect of toxic compounds from opportunistic microorganisms[5].
More specifically, researchers in this field believe that the occurrence of prolonged gut microbiome dysbiosis in COVID-19 patients may be associated with two important phenomena: fecal shedding of the virus into the environment and disease severity. Evidence for this pathophysiological mechanism is based on the hypothesis that dysbiosis may lead to epithelial inflammation and an increase in ACE2 expression. Given that ACE2 plays a key role in dietary amino acid homeostasis, patients can be severely affected. In this connection, SARS-CoV-2 binds to ACE2, leading to microflora imbalance. This is because the possible downregulation of ACE2 may reduce the secretion of antimicrobial peptides and in turn lead to increased pathogen survival and gut dysbiosis[5]. It is also worth noting that some drugs used to treat COVID-19, such as corticosteroids, have been shown to interact with the gut microbiome. This is also true for chloroquine, which has been equivocally administered to many patients[3,5] as well as different medicinal herbs[10].
Despite this, in the current context of the ongoing pandemic, although a large amount of research has been published on liver injury due to COVID-19[4,5], there remain many questions to be answered (Table 1), including the risk of this type of injury in specific populations. Important research has demonstrated a greater vulnerability to alterations in the composition of the gut microbiota in different populations. This is true for example for the population of individuals with cleft lip and palate[11] and Hashimoto’s thyroiditis[12]. Therefore, knowing more about interactions between the human microbiota and the host cytokine pathway should be of great relevance. One of the justifications for carrying out further research in this field includes the need to discover new personalized treatment strategies to improve the composition of the gut microbiota in order to more effectively reduce the severity of COVID-19 and its complications[3,5]. This in conjunction with a healthy lifestyle could have positive impacts on both COVID-19 prevention and treatment[13,14].
| No. | Major questions |
| 1 | Mean duration of dysbiosis associated with liver injury due to COVID-19 |
| 2 | Differences in the magnitude of liver injury and changes in the microbiota associated with COVID-19 in patients with varying degrees of disease severity |
| 3 | The impact of different drugs metabolized in the liver on the worsening of liver injury associated with COVID-19 and changes in the gastrointestinal microbiota |
| 4 | Whether changes in the gastrointestinal microbiota and liver injury associated with COVID-19 are also related to long-COVID-19 symptoms |
| 5 | Effective medical protocols and/or treatments to prevent changes in the gastrointestinal microbiota and prevent or treat this type of liver injury |
| 6 | The impact of healthy habits on the prevention of changes in the gastrointestinal microbiota and recovery of liver injury in COVID-19 patients |
In view of the development of new COVID-19 vaccines, another important point to consider is that the microbiome may affect the immune response to vaccines. This is due to the fact that the immunogenicity can be impaired with dysbiosis[5]. Moreover, due to the likely global endemic situation of COVID-19, further microbiological and immunological research may be critical to determine the impact of changes to the balance of the human microbiota and immunology related to COVID-19 in order to achieve better predictions in the fight against possible new SARS-CoV-2 variants.
We thank the Italian Ministry of Health-Ricerca Corrente 2022 and Saveetha Institute of Medical and Technical Sciences for supporting this study.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Microbiology
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Liu DF, China; Peng XC, China; Shiferaw M, Ethiopia S-Editor: Chen YL L-Editor: Filipodia P-Editor: Chen YL
| 1. | Albillos A, de Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol. 2020;72:558-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 542] [Cited by in RCA: 1266] [Article Influence: 253.2] [Reference Citation Analysis (1)] |
| 2. | Jabczyk M, Nowak J, Hudzik B, Zubelewicz-Szkodzińska B. Microbiota and Its Impact on the Immune System in COVID-19-A Narrative Review. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 3. | Donati Zeppa S, Agostini D, Piccoli G, Stocchi V, Sestili P. Gut Microbiota Status in COVID-19: An Unrecognized Player? Front Cell Infect Microbiol. 2020;10:576551. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 4. | Cichoż-Lach H, Michalak A. Liver injury in the era of COVID-19. World J Gastroenterol. 2021;27:377-390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 53] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (2)] |
| 5. | Ozkurt Z, Çınar Tanrıverdi E. COVID-19: Gastrointestinal manifestations, liver injury and recommendations. World J Clin Cases. 2022;10:1140-1163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (7)] |
| 6. | Silhol F, Sarlon G, Deharo JC, Vaïsse B. Downregulation of ACE2 induces overstimulation of the renin-angiotensin system in COVID-19: should we block the renin-angiotensin system? Hypertens Res. 2020;43:854-856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 86] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 7. | Cipriano M, Ruberti E, Tovani-Palone MR. Combined use of lactoferrin and vitamin D as a preventive and therapeutic supplement for SARS-CoV-2 infection: Current evidence. World J Clin Cases. 2022;10:11665-11670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Shen S, Gong M, Wang G, Dua K, Xu J, Xu X, Liu G. COVID-19 and Gut Injury. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 9. | Yu Z, Yang Z, Wang Y, Zhou F, Li S, Li C, Li L, Zhang W, Li X. Recent advance of ACE2 and microbiota dysfunction in COVID-19 pathogenesis. Heliyon. 2021;7:e07548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 10. | Tovani-Palone MR, Ali S. The media and the early dissemination of information on medicines for COVID-19: current scenario in Pakistan. Einstein (Sao Paulo). 2021;19:eCE6354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Vieira NA, Borgo HC, da Silva Dalben G, Bachega MI, Pereira PC. Evaluation of fecal microorganisms of children with cleft palate before and after palatoplasty. Braz J Microbiol. 2013;44:835-838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 12. | Zhao F, Feng J, Li J, Zhao L, Liu Y, Chen H, Jin Y, Zhu B, Wei Y. Alterations of the Gut Microbiota in Hashimoto's Thyroiditis Patients. Thyroid. 2018;28:175-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 137] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 13. | Tovani-Palone MR, Comini N, Giacalone A, Franchi T. Importance of Healthy Habits During the COVID-19 Pandemic: Perspective and Suggestions. Electron J Gen Med. 2022;19:em369. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 14. | Pedersini P, Turroni S, Villafañe JH. Gut microbiota and physical activity: Is there an evidence-based link? Sci Total Environ. 2020;727:138648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |