Published online Oct 28, 2022. doi: 10.3748/wjg.v28.i40.5845
Peer-review started: February 6, 2022
First decision: April 10, 2022
Revised: May 6, 2022
Accepted: June 13, 2022
Article in press: June 13, 2022
Published online: October 28, 2022
Processing time: 263 Days and 19.8 Hours
Inflammatory bowel disease (IBD) constitutes a substantial risk factor for col
To investigate Cx43 and TET-2 expression levels and presence of 5-hydroxymethylcytosine (5-hmC) marks under inflammatory conditions both in vitro and in vivo.
TET-2 expression was evaluated in parental HT-29 cells and in HT-29 cells expressing low or high levels of Cx43, a putative tumor-suppressor gene whose expression varies in IBD and colorectal cancer, and which has been implicated in the inflammatory process and in tumor onset. The dextran sulfate sodium-induced colitis model was reproduced in BALB/c mice to evaluate the expression of TET-2 and Cx43 under inflammatory conditions in vivo. In addition, archived colon tissue sections from normal, IBD (ulcerative colitis), and sporadic colon adenocarcinoma patients were obtained and evaluated for the expression of TET-2 and Cx43. Expression levels were reported at the transcriptional level by quantitative real-time polymerase chain reaction, and at the translational level by Western blotting and immunofluorescence.
Under inflammatory conditions, Cx43 and TET-2 expression levels increased compared to non-inflammatory conditions. TET-2 upregulation was more pronounced in Cx43-deficient cells. Moreover, colon tissue sections from normal, ulcerative colitis, and sporadic colon adenocarcinoma patients corroborated that Cx43 expression increased in IBD and decreased in adenocarcinoma, compared to tissues from non-IBD subjects. However, TET-2 expression and 5-hmC mark levels decreased in samples from patients with ulcerative colitis or cancer. Cx43 and TET-2 expression levels were also investigated in an experimental colitis mouse model. Interestingly, mice exposed to carbenoxolone (CBX), a GJ inhibitor, had upregulated TET-2 levels. Collectively, these results show that TET-2 levels and activity increased under inflammatory conditions, in cells downregulating gap junctional protein Cx43, and in colon tissues from mice exposed to CBX.
These results suggest that TET-2 expression levels, as well as Cx43 expression levels, are modulated in models of intestinal inflammation. We hypothesize that TET-2 may demethylate genes involved in inflammation and tumorigenesis, such as Cx43, potentially contributing to intestinal inflammation and associated carcinogenesis.
Core Tip: Chronic inflammation of the colon is a risk factor for colorectal cancer. Intestinal inflammation has been associated with reduced gap junction-mediated intercellular communication and downregulation connexins [mainly connexin 43 (Cx43)]. The involvement of the demethylating enzyme ten-eleven translocation-2 (TET-2) (methylcytosine dioxygenase) in the inflammatory process has motivated the investigation of Cx43 and TET-2 expression in colitis and colorectal carcinoma. In vitro and in vivo data report on the upregulation of Cx43 in inflammatory states and a downregulation of TET-2. In human samples of colon adenocarcinoma, both TET-2 and Cx43 were downregulated, potentially implicating them in the malignant transformation of inflamed intestinal tissues.
- Citation: El-Harakeh M, Saliba J, Sharaf Aldeen K, Haidar M, El Hajjar L, Awad MK, Hashash JG, Shirinian M, El-Sabban M. Expression of the methylcytosine dioxygenase ten-eleven translocation-2 and connexin 43 in inflammatory bowel disease and colorectal cancer. World J Gastroenterol 2022; 28(40): 5845-5864
- URL: https://www.wjgnet.com/1007-9327/full/v28/i40/5845.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i40.5845
The gastrointestinal tract is constantly exposed to environmental insults that may potentially lead to pathologies. Inflammatory bowel disease (IBD) is a relapsing and remitting inflammatory disorder affecting distinct parts of the gastrointestinal tract. It comprises two primarily encountered disorders: Crohn’s disease and ulcerative colitis, which can be distinguished by their localization in the different parts of the gut and the endoscopic appearance of the inflamed segment[1-3]. IBD increases the risk of developing colon adenocarcinoma[4], and colitis-associated colon adenocarcinoma remains a fun
Gap junctions (GJs), components of junctional complexes, have a potential role in regulating epithelial integrity and function. GJs are formed by the docking of two hemichannels (connexin hexamers) contributed from two adjacent cells, allowing the direct exchange of ions and small signaling molecules (< 2 kDa)[6]. Connexins play a pivotal role in cellular proliferation, differentiation, and function and are recognized as putative tumor-suppressor genes[7-9]. The library of connexin-related diseases has been emerging for many years, and includes various types of syndromes and disorders, such as inflammatory diseases and cancer. Several studies have revealed that the inflammatory response relies in part on connexins and GJ-mediated intercellular communication (GJIC)[10]. More specifically, alteration in the expression of connexin 43 (Cx43) in the gastrointestinal tract is associated with IBD, gastrointestinal infections, and impaired motility[11,12]. We have previously shown an alteration of Cx43 expression and localization, as well as a direct heterocellular communication between intestinal epithelial cells (IECs) and macrophages, which may contribute to IBD pathogenesis[12].
In complex diseases, such as IBD and colon adenocarcinoma, in addition to the heritable component, environmental and epigenetic (DNA methylation and demethylation, histone marks, higher order chromatin structure, etc.) factors are likely to influence onset[13]. DNA methylation state is controlled by the interplay between DNA methyltransferases and demethylating enzymes, such as ten-eleven translocation (TET) proteins. Methylation occurs at normally demethylated CpG-rich regions named “CpG islands”, of which 70% overlap with human gene promoters, and results in gene silencing[14]. DNA methylation profiles from IBD patients are considerably altered compared to healthy counterparts[15]. Moreover, in IBD-associated colon adenocarcinoma, DNA methylation status was distinct from sporadic colon adenocarcinoma, with different gene expression profiles[16]. DNA methylation is counteracted by demethylation mechanisms, catalyzed by TET enzymes (TET-1, TET-2, TET-3), thus activating gene transcription[17]. Following a series of oxidation reactions by TET enzymes, previously methylated 5-methylcytosine is converted to 5-hydroxymethylcytosine (5-hmC) and further oxidized products that will be recognized and excised by thymine-DNA glycosylase/base enzyme repair pathway to result in an unmethylated cytosine residue[18]. TET genes are mutated in a variety of diseases and cancers. In inflammatory diseases, TET-2 is crucial for the repression of the pro-inflammatory cytokine, interleukin (IL)-6[19], while in several cancers, TET-2 expression is downregulated[20]. In particular, tumors of the digestive tract express different levels of TET enzymes, which are explored as potential factors informing prognosis[21]. Downregulation of TET-1, TET-2, and TET-3 have been reported in gastric cancers with concomitant loss of 5-hmC marks[22] and in colorectal carcinoma[23], with TET-2 transcriptional levels potentially serving as indicators for treatment outcome and disease recurrence[23]. In addition, a recent study reported that the human gene coding for TET-2 contains three promoter elements which are differently regulated in different tissues and developmental stages[24] and may play a role in cancer development.
While many studies have reported the Cx43 promoter to be hypermethylated during the transition from inflammation to cancer[25-27], the expression statuses of Cx43 and TET-2 in IBD and colorectal cancer have not been simultaneously described. This study investigated the effect of inflammation and Cx43 expression levels on the intestinal cell membrane integrity and described TET-2 expression and 5-hmC marks under inflammatory states in vitro, in vivo, and in human samples of ulcerative colitis and sporadic colon carcinoma.
The HT-29 and Caco-2 cell lines derived from colorectal adenocarcinoma tissues were used as models for human IECs. When grown in monolayer, these cells become more differentiated and IEC-like[12,28,29] and are widely used as a model of intestinal transport and pathology, including inflammation. Human embryonic kidney cells (packaging HEK 293T cells) were used for production of viral sup-ernatant for transduction purposes.
In addition, the human monocytic cell line (THP-1) was used as an in vitro model for activated macrophages[12,30,31] for the production of conditioned inflammatory medium. After exposure to phorbol myristate acetate (PMA) and lipopolysaccharide (LPS), THP-1 cells become activated; they adhere to the cell culture vessel and show increased transcriptional levels of connexins, Toll-Like Receptor (TLR)-2, TLR-4, NF-κB p65, COX-2, inflammatory cytokines tumor necrosis factor-α (TNF-α), and IL-1β[12]. In this study, suspension THP-1 cells were activated with 50 ng/mL PMA (Sigma-Aldrich, St Louis, MO, United States) for 24 h, followed with 1 μg/mL of LPS (Sigma-Aldrich) for 4 additional hours. When activated THP-1 cells adhered to the cell culture plate, they were washed with PMA- and LPS-free media and left to grow for 72 h. Conditioned media was then collected, filtered, and applied in vitro onto colon cell lines to create an inflammatory milieu.
Cells were maintained in complete RPMI-1640 (Sigma-Aldrich) for HT-29 and THP-1 cells or Dulbecco's Modified Eagle's medium (DMEM AQ; Sigma-Aldrich) for HEK 293T cells supplemented with 10% FBS (Sigma-Aldrich), 100 U/mL penicillin G, and 100 μg/mL streptomycin (Sigma-Aldrich). Cells were grown at 37 °C in a humidified incubator with 5% CO2 atmosphere.
The Cx43-pDendra2N construct (Evrogen, Moscow, Russia) was previously cloned into pCSCW lentiviral vectors[12]. The plasmid was then used to transform DH5α competent Escherichia coli bacteria, which were then left to proliferate. Plasmid was then isolated and purified using the EndoFree Maxi plasmid purification kit (Qiagen, Hilden, Germany) to be used for transfection along with other plasmids (gag/pol/env) into HEK 293T cells for production of viral supernatant, which carries the Cx43-pDendra2 chimeric proteins. The viral supernatant was used to transduce HT-29 cells. Following the transduction, HT-29 cells were cultured, expanded, and highly positive cells (referred to as HT-29 Cx43D cells thereafter) were sorted using the BD FACSAria™ III sorter (BD Biosciences, Franklin Lakes, NJ, United States). Viral supernatants were obtained from HEK 293T cells and used to transduce HT-29 cells. These cells, named HT-29 Cx43D, were then isolated using a BD Fluorescence-Activated Cell Sorting Aria SORP cell sorter in the single-cell mode. Green fluorescence in HT-29 Cx43D cells attests to the successful upregulation of exogenous Cx43 in these cells.
HT-29 cells knocking down Cx43, referred to as HT-29 Cx43- cells, were generated using the CRISPR/Cas9 gene-editing system. After cloning the Cx43 target sequence (20 bp) into the guide RNA scaffold of the pX330-CRISPR plasmid, the bacterial transformation was performed as above and 10-15 colonies were picked to check for the correct insertion of the guide RNA by sequencing. Transfection of HT-29 cells with positive clones was performed, and cells were selected with puromycin (1 μg/mL) until isolated colonies were obtained. Several positive clones were validated for Cx43 knockdown at the RNA and protein levels. One specific clone resulted in more than 90% down-regulation of Cx43 mRNA, generating the experimental HT-29 Cx43- cells. This downregulation was confirmed by western blot and immunofluorescence assays.
Parental HT-29, HT-29 Cx43D, and HT-29 Cx43- cells were seeded in 24-well plates at a density of 25000 cells/cm2. At 24 h, 48 h, and 72 h, cells were washed with PBS and trypsinized. Viable cells were counted using the trypan blue exclusion dye.
Trans-epithelial electrical resistance: This method evaluates barrier integrity of epithelial cells grown in monolayer; by describing the impedance of barrier-forming cell cultures. Briefly, electrodes are placed on both sides of the cellular barrier and an electric current is applied. The resulting current established in the circuit is measured and trans-epithelial electrical resistance (TEER) is calculated. The higher the TEER, the better the membrane integrity[32]. In this study, HT-29, HT-29 Cx43D, and HT-29 Cx43- cells were cultured on Transwell® inserts with 0.4 μm-pore size filters (Corning, Corning, NY, United States). TEER was measured on confluent cells in the presence or absence of 2% dextran sulfate sodium (DSS) using an EVOM voltmeter with an ENDOHM-12 (World Precision Instruments, Sarasota, FL, United States). Electrical resistance was expressed as Ω × cm2. DSS was applied onto the cells to reproduce in vitro the membrane breach it is known to induce in vivo and characterize the loss of membrane integrity. TEER was calculated by subtracting the resistance of blank filters from that of filters covered with a monolayer of parental HT-29, HT-29 Cx43D, or HT-29 Cx43- cells.
Epithelial cells form tight junctions that prevent paracellular transit. Evans blue is a dye that strongly binds to serum albumin in vivo and in vitro, becoming a protein tracer[33]. When Evans Blue is added on the apical aspect of cells in culture, it is retained in this compartment due to the established epithelial barrier. Any perturbation of barrier integrity will result in seeping of Evans Blue to the basolateral aspect of the cells. HT-29, HT-29 Cx43D, and HT-29 Cx43- cells were grown on transparent PET membrane cell culture inserts with 0.4 μm-pore size (Corning) until confluent (monolayer). Evans Blue solution (Sigma-Aldrich) was prepared in a 1% bovine serum albumin (cell culture grade; GIBCO®, Paisley, Scotland) at a concentration of 170 μg/mL and then filtered through 0.22 μm-filters (Corning®, Wiesbaden, Germany). At confluence, cells were exposed to 2% DSS for 24 h. Cells were then washed twice with PBS and 400 μL of Evans Blue solution were added on top of the cells (seeded in inserts). Plate wells were rinsed and 1 mL of PBS was added into each well. Cells were then incubated at 37 °C. Every 30 min, 200 μL of solution from the well were collected and replaced with fresh 200 μL PBS, for a total of 2 h. The optic density of collected solution was recorded at 630 nm and concentrations of Evans Blue were calculated.
This study was approved by the Institutional Animal Care and Utilization Committee of the American University of Beirut (IACUCC# 18-03-476). The acute DSS-induced colitis mouse model was performed as previously reported[34]. The DSS colitis model in mice is a well-established model for IBD. Using this mouse model, studies have shown disruption of the epithelial barrier function with the infiltration of immune cells, as well as an uncommon production of cytokines[35,36]. In addition, DSS has been previously used in vitro to induce a cell membrane breach in a monolayer of colon cells, mimicking the intestinal mucosal barrier. DSS exposure resulted in impairment of protein trafficking and alterations in membrane composition in the intestinal Caco-2 cell line[37,38], resembling the intestinal mucosa integrity breach that occurs in IBD.
In this study, carbenoxolone (CBX) was used as a non-specific GJ inhibitor, injected intraperitoneally at the dose of 30 mg/kg every other day. Briefly, adult BALB/c male mice were distributed into the following experimental groups, each comprised of five mice: (1) Control group that received normal drinking water with no CBX injections; (2) CBX group that received normal drinking water with CBX injections starting on day 11; (3) DSS group where mice were exposed to 2.5% DSS in their drinking water for 10 d, followed by normal drinking water as of day 11; and (4) DSS + CBX group that received DSS-containing drinking water for 10 d followed by CBX injections. All four groups were given normal drinking water from day 11 to day 21 (end of experimental duration) and body weights were measured daily. All mice were given standard chow ad libitum and were housed in a temperature-controlled environment on a 12-h automated light/dark cycle. On day 21, mice were anesthetized with isoflurane and then euthanized by cervical dislocation. Colon lengths were measured using a ruler and colon tissues were collected for histological and molecular examinations to assess tissue integrity and expression of inflammatory markers, Cx43, and TET-2.
Cells in culture: Cells were washed with PBS and total RNA was extracted using the RNeasy® Plus mini kit (Qiagen) as per manufacturers’ instructions.
Cryopreserved tissues: Colon tissues from experimental mice were collected and snap-frozen in liquid nitrogen. RNA was then isolated using TRI reagent (Sigma), as per manufacturers’ protocol. Since DSS treatment inhibits mRNA amplification from tissues by quantitative polymerase chain reaction (qPCR) by inhibiting the activities of both polymerase and reverse transcriptase, RNA purification for all samples in all conditions was done by the lithium chloride method according to Viennois et al[38].
One μg of total RNA was reverse-transcribed to cDNA using the iScriptTM cDNA synthesis kit (Bio-Rad, Hercules, CA, United States). qPCR was conducted using a homemade SYBR green mix in a CFX96 system (Bio-Rad). Products were amplified using primers that recognize Cx43, TET-2, IL-1β, TNF-α, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Table 1). PCR settings were as follows: A pre-cycle at 95 °C for 5 min followed by 40 cycles consisting of 95 °C for 10 s, 52-62 °C for 30 s, and 72 °C for 30 s with a final extra-elongation at 72 °C for 5 min. The fluorescence threshold cycle value (Ct) was obtained for each gene and normalized to the corresponding GAPDH Ct value. All experiments were carried out in technical duplicates and independently performed at least three times.
| Gene | Primer sequences, 5’ to 3’ | Annealing temperature, °C |
| hCx43 | F: CTTCACTACTTTTAAGCAAAAGAG | 52 |
| R: TCCCTCCAGCAGTTGAG | ||
| hTET-1 | F: TTCGTCACTGCCAACCTTAG | 60 |
| R: ATGCCTCTTTCACTGGGTG | ||
| hTET-2 | F: CACTGCATGTTTGGACTTCTG | 60 |
| R: TGCTCATCCTCAGGTTTTCC | ||
| hTET-3 | F: GCCCACAAGGACCAGCATAA | 60 |
| R: CGCAGCGATTGTCTTCCTTG | ||
| hGAPDH | F: TGGTGCTCAGTGTAGCCCAG | 52-62 |
| R: GGACCTGACCTGCCGTCTAG | ||
| mCx43 | F: ACAGCGGTTGAGTCAGCTTG | 60 |
| R: GAGAGATGGGGAAGGACTTGT | ||
| mTET-2 | F: CAAGGTGTGCTTGGCAATTTT | 60 |
| R: CATCACTGTGCGTCAATCAAGA | ||
| mIl-1β | F: CTCCACCTCAATGGACAGAA | 57 |
| R: GCCGTCTTTCATTACACAGG | ||
| mTNF-α | F: AATGGCCTCCCTCTCATCAGT | 57 |
| R: CCACTTGGTGGTTTGCTACGA | ||
| mGAPDH | F: CATGGCCTTCCGTGTTCCTA | 58 |
| R: CCTGCTTCACCACCTTCTTGAT |
Cells in culture: Cells were washed with ice-cold PBS, and scraped on ice in lysis buffer (0.5 M Tris-HCl buffer, pH 6.8; 2% SDS, and 20% glycerol) containing protease and phosphatase inhibitors (Roche, Basel, Switzerland).
Cryopreserved tissues: Snap-frozen mouse colon tissues were homogenized on ice in ice-cold RIPA buffer containing 10% of a 0.5 M Tris-HCl buffer, pH 7.5, 3% of 5 M NaCl, 1% NP-40, 10% sodium deoxycholate, and 1% SDS as well as phosphatase and protease inhibitors.
The lysate was then sonicated. Proteins were quantified, loaded onto 10% SDS-polyacrylamide gels, and subjected to electrophoresis. Migrated proteins were transferred to polyvinylidene fluoride membranes (Bio-Rad) and membranes were blocked with 5% fat-free milk in PBS. Membranes were then incubated with either human or mouse primary antibodies for 3 h at room temperature or overnight at 4 °C. Primary antibodies were hybridized with horseradish peroxidase-conjugated secondary antibodies (anti-rabbit: SC-2357 and anti-mouse SC-2005; Santa Cruz Biotechnology, Dallas, TX, United States) and blots were visualized using an enhanced chemiluminescence detection kit. GAPDH (MAB5476; Abnova, Taipei, Taiwan) and β-actin (A2228; Sigma-Aldrich) were used as loading controls. The quantification of bands was performed using the ImageJ software (United States National Institutes of Health, Bethesda, MD, United States). Immunoreactivity of proteins under study was reported as a ratio of protein-of-interest expression to the housekeeping gene used as loading control.
Archived formaldehyde-fixed paraffin-embedded (FFPE) colon blocks (ulcerative colitis, sporadic colon adenocarcinoma and normal) were obtained from the American University of Beirut Medical Center. All patients’ identifiers were kept confidential from the study team.
Sections obtained from animals and human samples were stained with hematoxylin and eosin (H&E) for observation under the light microscope (CX41; Olympus, Tokyo, Japan).
Cells in culture: Cells were grown on coverslips and then fixed either with 4% paraformaldehyde and permeabilized with 0.05% Triton X-100 for 20 min for Cx43 probing (SAB4501175; Sigma-Aldrich) or with ice-cold methanol at -20 °C for TET-2 (ab94580; Abcam, Cambridge, United Kingdom) and 5-hmC (ab214728; Abcam) probing before blocking of non-specific binding.
FFPE tissues: Human and murine 5-μm-thick tissue sections were immune-stained using antibodies against Cx43, TET-2, 5-hmC, and CD68 (ab201340, Abcam, United States). Briefly, paraffin blocks were sectioned using a microtome and sections were mounted onto glass slides. Specimens were deparaffinized in xylene and rehydrated using a gradient series of alcohol to water. Antigen retrieval was performed by incubating sections in sodium citrate buffer (pH 6.0) in a steamer for 30 min. Sections were allowed to cool, then washed twice with deionized water before blocking of non-specific binding.
Non-specific binding was blocked with 5% normal goat serum (Chemicon, Burlington, MA, United States) in PBS for 1 h in a humidified chamber and incubated with the primary antibody against Cx43, TET-2, and 5-hmC overnight at 4 °C. Cells were then washed and incubated with IgG-conjugated secondary antibody: Either Texas Red (T862; Life Technologies, Carlsbad, CA, United States) or Alexa Fluor 488 (A11070; Life Technologies) at 1 μg/mL for 1 h at room temperature. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole at 1 μg/mL for 10 min. Coverslips were then mounted onto the glass slides using Prolong Anti-fade kit (Life Technologies).
Slides were examined under a fluorescence microscope and images were acquired using a 63×/1.46 Oil Plan-Apochromatic objective (on the laser scanning confocal microscope LSM 710, operated by the Zeiss LSM 710 software; Carl Zeiss, Oberkochen, Germany).
Numerical values were expressed as mean ± SEM or mean ± SD. The P value was determined and considered significant for P < 0.05. Differences between experimental groups were assessed using Student t-test, one-way analysis of variance (ANOVA) or by two-way ANOVA followed by Tukey's multiple comparisons test.
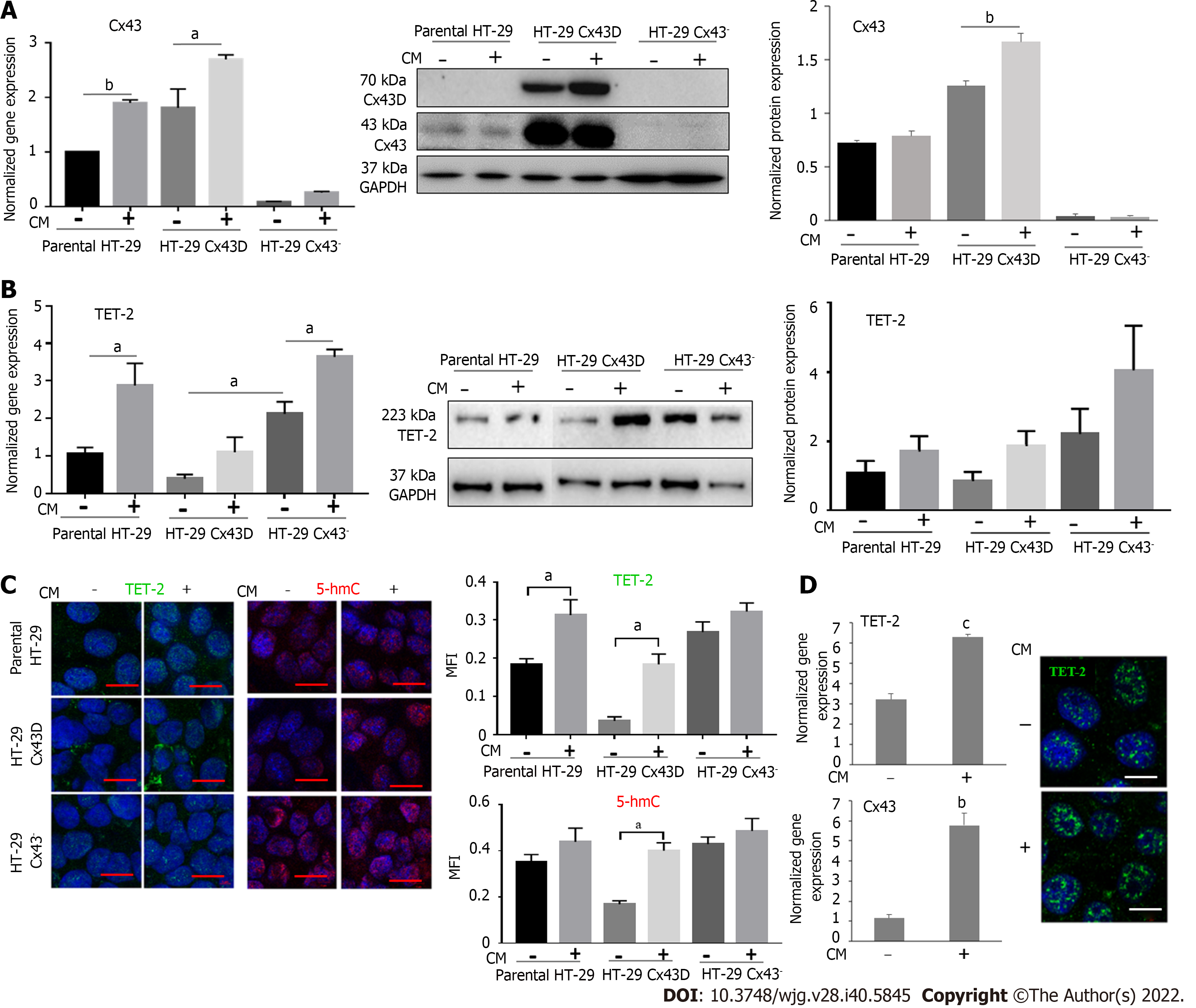
In an attempt to investigate TET-2 modulation in the context of different Cx43 expression levels in HT-29 cells, Cx43 was upregulated (HT-29 Cx43D cells) or downregulated (HT-29 Cx43- cells). Increases and decreases in Cx43 levels were reflected at the translational level: Western blot analysis (Figure 1A) shows upregulation of exogenous Cx43 protein levels in HT-29 Cx43D and loss of endogenous Cx43 protein expression in HT-29 Cx43- cells compared to parental HT-29 cells (P < 0.001). Figure 1B also shows increased expression of Cx43 in HT-29 Cx43D cells and their localization at the cell periphery, where they form GJ plaques, and a decrease in Cx43 levels in HT-29 Cx43- cells.
Cells manipulated for Cx43 gene showed differential proliferation rates 48 h and 72 h post-seeding. As depicted in Figure 1C, Cx43 upregulation (HT-29 Cx43D cells) significantly decreased the number of viable cells compared to parental HT-29 cells (P < 0.05 at 48 h and P < 0.001 at 72 h). However, cells devoid of Cx43 (HT-29 Cx43- cells) demonstrated a significantly greater proliferation rate compared to parental HT-29 cells at 48 h and 72 h post-seeding (P < 0.001). The increased proliferation rate of HT-29 Cx43- cells was also accompanied by increased migratory potential, as suggested by the wound healing and the invasion assays (Supplementary Figure 1). Compared to parental cells, HT-29 Cx43- cells seemed more efficient at closing the artificially created gap (P < 0.05).
These observations are in accordance with the tumor suppressor role of Cx43, greatly inhibiting the proliferation of HT-29 cells.
Transcriptional levels of TET-1, TET-2, and TET-3 were evaluated in HT29 cells, manipulated for Cx43 expression. TET-2 specifically demonstrated the highest transcriptional levels between all three TET genes (Figure 1D). TET-2 protein expression was verified by immunofluorescence in parental HT-29 cells (Figure 1D). While TET-1 and TET-3 levels were not significantly different in HT-29 cells expressing lower or higher levels of Cx43, TET-2 levels significantly increased in HT-29 Cx43- cells compared to parental HT-29 (P < 0.05) and to HT-29 Cx43D (P < 0.001) cells.
For the remainder of this study, TET-2 expression levels and 5-hmC mark were evaluated given the greater expression of TET-2 in HT-29 cells and the prevalence of loss-of-function mutations of TET-2 in cancer[21,39-41].
In order to explore a potential pattern of expression for TET-2 and Cx43 in inflammation, parental HT-29, HT-29 Cx43D, and HT-29 Cx43- cells were screened for TET-2 mRNA and protein expressions, as well as 5-hmC marks by immunofluorescence using an antibody against 5-hmC, a product of the reaction catalyzed by TET-2. In Cx43-expressing cells, the addition of inflammatory media (supernatant from activated THP-1 cells) resulted in upregulation of Cx43 (Figure 2A). Similarly, under inflammatory conditions, all three cellular subsets upregulated their TET-2 expression (P < 0.05) at the transcriptional and translational levels (Figure 2B). Interestingly, HT-29 cells down-regulating Cx43 showed higher expression of TET-2 compared to parental cells or cells overexpressing Cx43 (Figure 2B). The 5-hmC marks also increased with increased TET-2 levels in HT-29 Cx43- cells. Levels of TET-2 and 5-hmC both increased in cells exposed to inflammation, as shown by quantified immunofluorescence micrographs (Figure 2C). Changes in TET-2 expression were less pronounced in HT-29 Cx43D cells than in HT-29 Cx43- cells. This trend was also evident with the 5-hmC marks (Figure 2C) where increased expression of TET-2 correlated with increased amounts of 5-hmC.
In order to strengthen these findings, experiments were performed on Caco-2 cells. Levels of Cx43 and TET-2 were also upregulated in Caco-2 cells exposed to inflammatory medium (Figure 2D), with a five-fold increase in Cx43 levels (P < 0.001) and two-fold increase in TET-2 levels (P < 0.01).
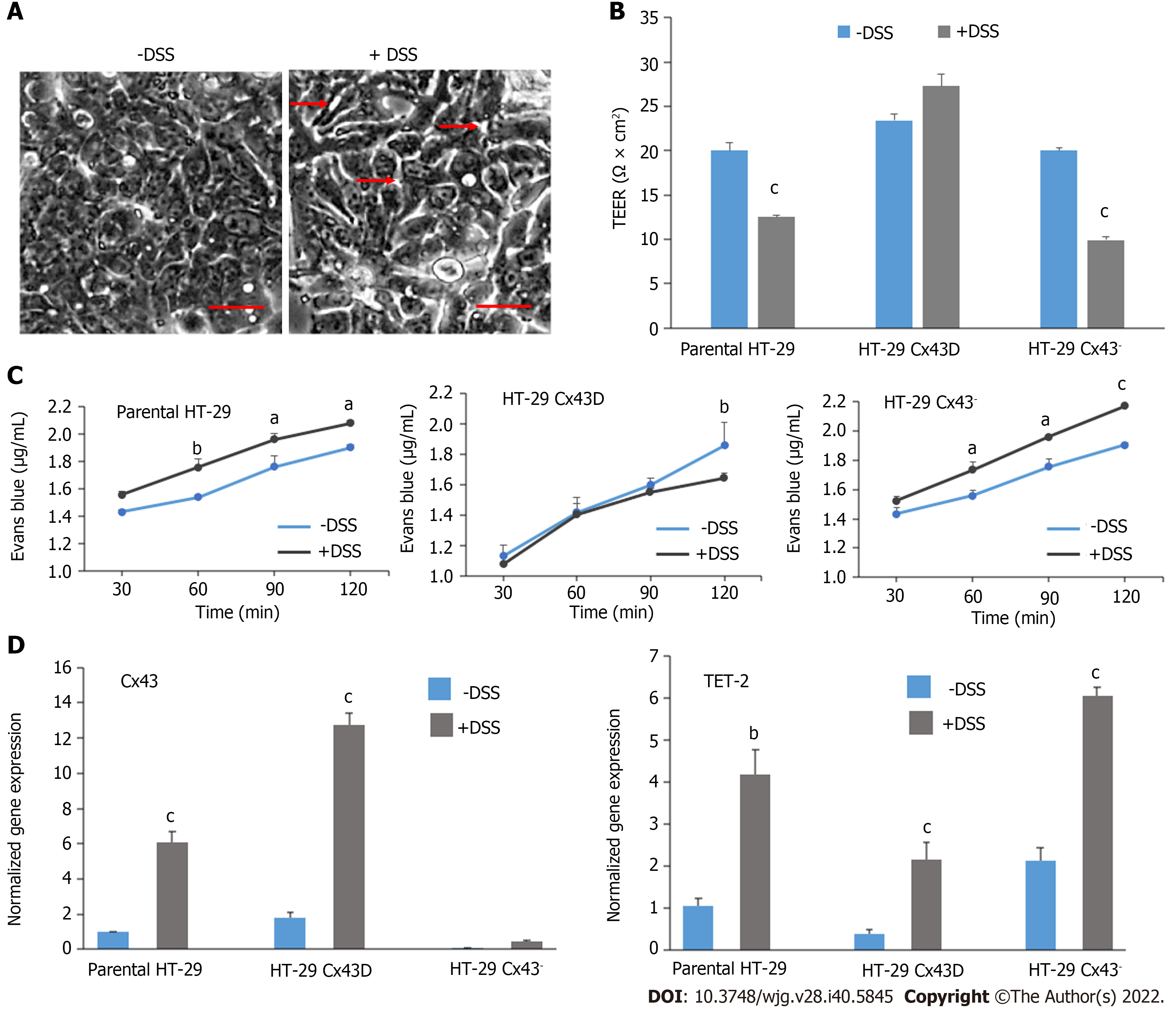
Parental HT-29, HT-29 Cx43D, and HT-29 Cx43- cells were grown as monolayers and exposed to 2% DSS for 24 h to evaluate if differential Cx43 levels would have any bearing on DSS-induced membrane integrity breach. DSS was previously shown to impair IEC membrane integrity in vitro[42] and it was used in our study to induce membrane injury similar to that observed upon chronic inflammation. Figure 3A shows that DSS resulted in decreased cell contacts and increased spaces between cells (red arrows), indicating loss of membrane integrity. The TEER assay was performed to assess how Cx43 modulation affects membrane integrity in the presence of DSS. Figure 3B shows that, in the absence of DSS, differentially expressed Cx43 had no major impact on epithelial barrier integrity, with an average TEER of 20 Ω × cm2 in parental and in HT-29 Cx43- cells and 23 Ω × cm2 in HT-29 Cx43D cells. However, the addition of DSS resulted in a significant decrease in TEER values by approximately 37% in parental cells and 50% in HT-29 Cx43- cells (P < 0001). Therefore, overexpression of Cx43 in HT-29 Cx43D cells seemed to be protective against DSS-induced loss of membrane integrity (Figure 3B).
The Evans Blue permeability assay was then performed to evaluate the monolayer integrity under DSS exposure and in cells with different Cx43 expression status. The level of cell permeability or “leakage” was correlated to the concentration of Evans Blue measured at the bottom of the well. As shown in Figure 3C, after 60 min of incubation with Evans Blue, the concentration of the dye significantly and rapidly increased in DSS-treated parental HT-29 (P < 0.05) and HT-29 Cx43- cells (P < 0.0001). However, within the two-hour timeframe, cells overexpressing Cx43 (HT-29 Cx43D cells) had not leaked substantial levels of Evans Blue dye.
DSS was subsequently used to reproduce the murine colitis model to evaluate modulation of Cx43 and TET-2 levels in the inflamed colons of mice. Figure 3D shows that upon exposure of HT-29 cells to DSS in vitro, levels of Cx43 and TET-2 vary. Specifically, in Cx43-expressing cells, DSS exposure results in upregulation of Cx43 (P < 0.001). Levels of TET-2 also significantly increase in all HT-29 cellular subsets (P < 0.01).
In summary, in vitro results suggest that inflammation leads to upregulation of Cx43 and of TET-2 in HT-29 cells. TET-2 upregulation was more pronounced in HT-29 cells devoid of Cx43. Moreover, Cx43 knockdown rendered HT-29 cells more sensitive to DSS-induced membrane integrity breach, in favor of a role of Cx43 protein in the maintenance of epithelial barrier integrity. Modulation of Cx43 and TET-2 in vitro displayed similar trends whether cells were exposed to conditioned inflammatory media (obtained from activated THP-1 cells) or to DSS (subsequently used in the colitis mouse model).
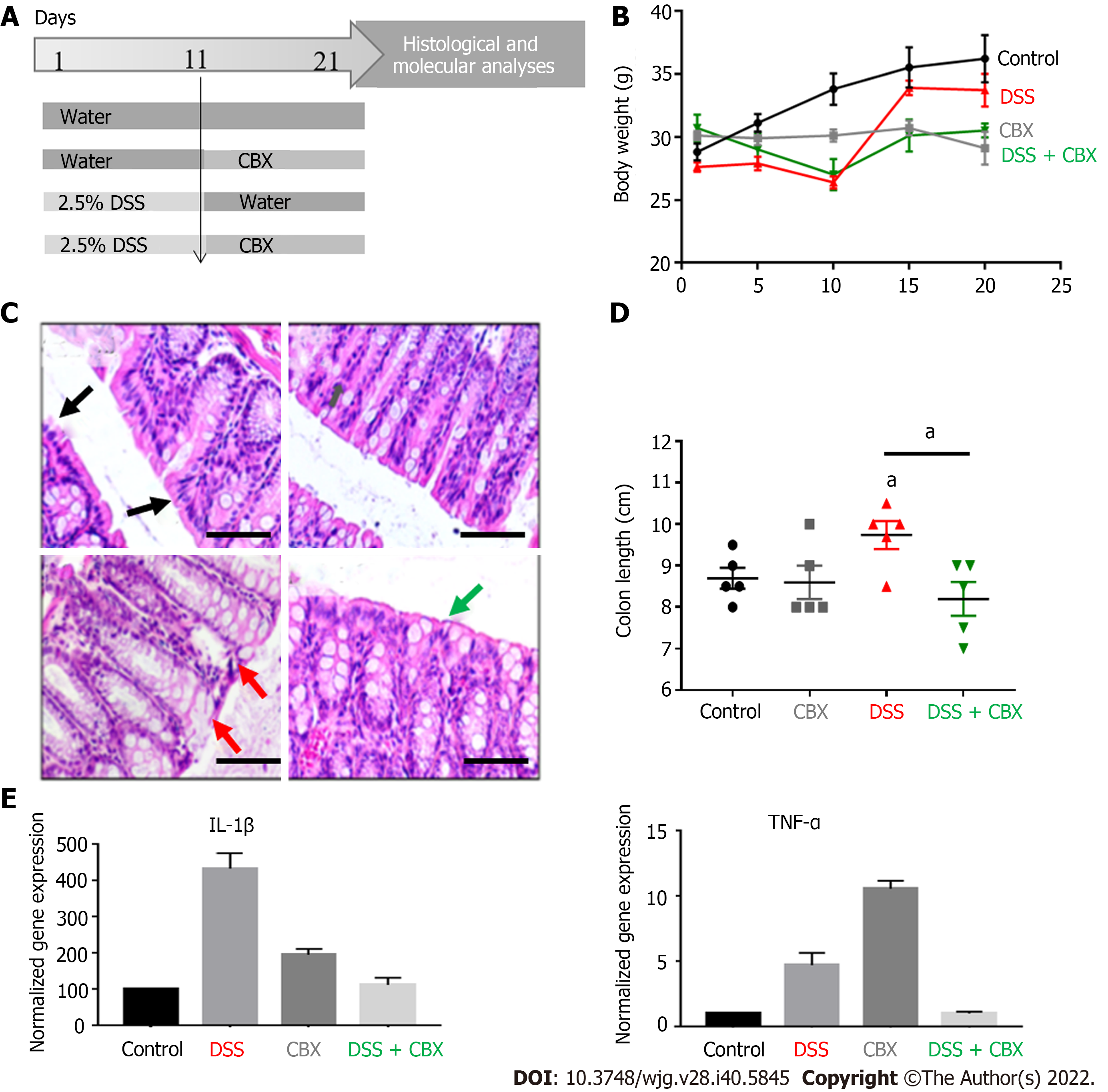
An established and widely used DSS-induced colitis mouse model was reproduced[34] to evaluate Cx43 and TET-2 expression. A subset of mice was also administered CBX, a GJ inhibitor, to examine TET-2 expression modulation in varying functional states of GJs.
The experimental design was as described in Figure 4A, where each group comprised five mice. Body weight was recorded daily, throughout the 21-d experimental duration. Figure 4B reflects weight changes; while mice in the control group (Group 1) consistently gained weight over the 21-d experiment, mice exposed to CBX alone (Group 2) maintained their baseline weight throughout the experiment. On the other hand, DSS-exposed mice lost a moderate amount of weight until day 10, after which they were switched to normal drinking water (Group 3) and their average weight picked up to almost control group levels. Mice in the DSS group who were injected with CBX starting day 11 recovered some of the weight lost until day 10, but did not reach the normal weight recorded in the control group.
At the end of the experimental duration, mice were euthanized and their colons examined and collected for measurement purposes and a biopsy was used for histological and molecular analyses. Histological examination of H&E stained sections of colons showed a disruption of epithelial barrier as well as an increase in infiltrating cells in crypts of colons from DSS-treated mice compared to control. CBX-treated animals seem to have retained (albeit not fully) some normalcy (Figure 4C). DSS-treated mice showed significantly longer colons compared to the control group (P < 0.05). CBX significantly reduced the length of DSS-treated colons to approximately normal levels (P < 0.05) (Figure 4D).
Levels of inflammatory cytokines, IL-1β and TNF-α, previously shown to be modulated in IBD models[12,38,43], were evaluated by qPCR. In accordance with weight changes and colon length indicative of intestinal damage, DSS-treated mice of Group 3 had increased expression of both IL-1β and TNF-α, compared to control Group 1 (P < 0.05) (Figure 4E).
These data propose that CBX alleviates DSS-induced inflammation in mice, as demonstrated by normalized colon length and histology, as well as decreased levels of inflammatory mediator transcripts in colon tissues.
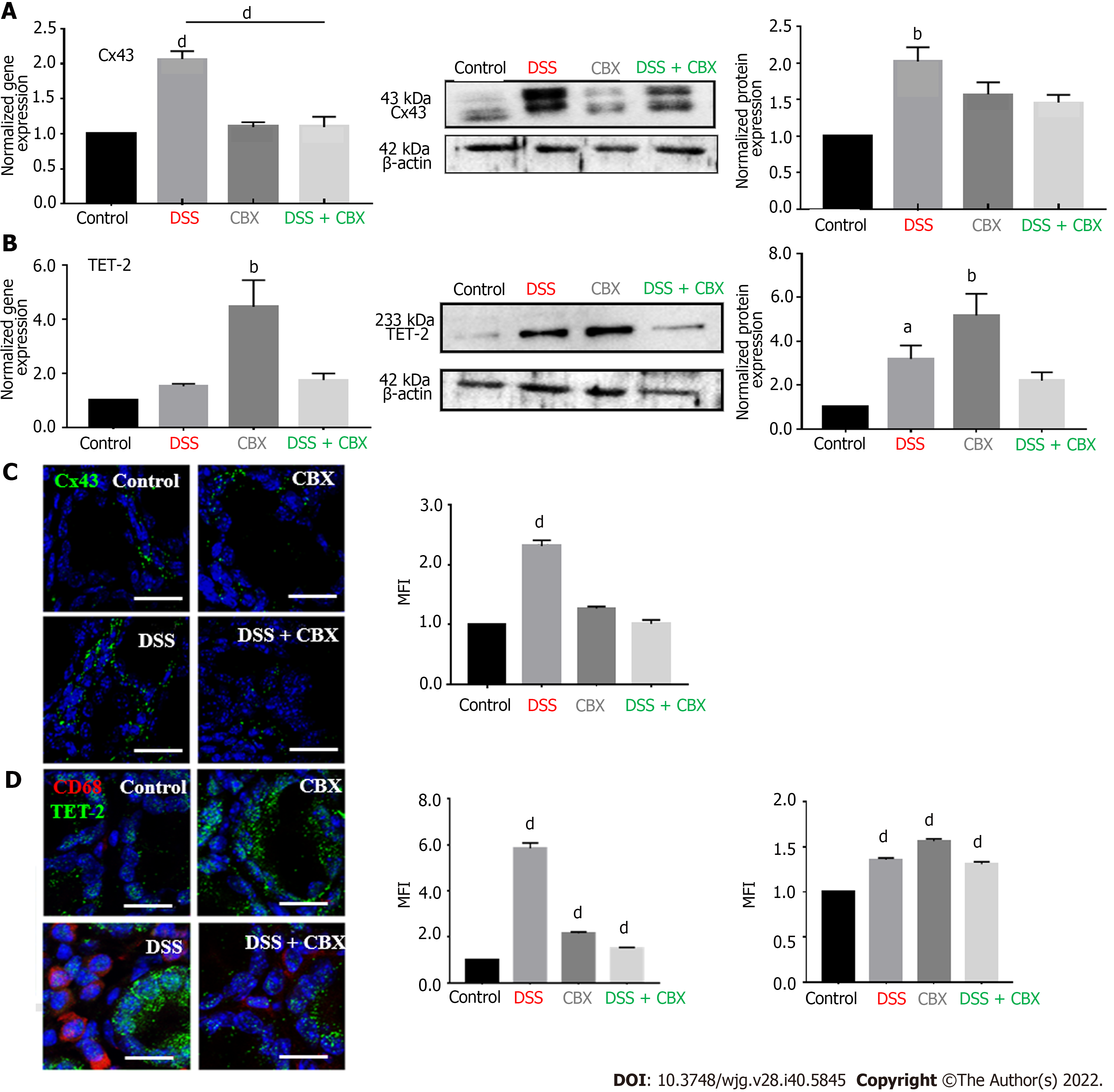
Colon tissues obtained from mice on day 21 (end of experimental duration) were processed for total RNA and protein extraction. Cx43 expression was significantly higher in tissues from mice exposed to DSS both at the transcriptional (Figure 5A, P < 0.0001) and translational levels (Figure 5A, middle and right panels). However, CBX injections restored Cx43 mRNA and protein levels back to control levels. Not only did GJ inhibition attenuate the inflammatory state in the colon, it also led to downregulated Cx43 expression.
Levels of TET-2 were also evaluated in the colon tissues obtained from mice in all four experimental groups. Gene expression analysis pointed to an increase of TET-2 mRNA levels in the CBX-treated group (Group 2, Figure 5B, P < 0.001). In the tissues of mice from Group 4 (DSS + CBX), TET-2 levels were similar to those in the DSS group (slightly, but not significantly, higher than control levels). The same pattern was observed at the protein level (Figure 5B, middle and right panels). Changes in TET-2 levels were accompanied by changes in Cx43 levels, which increased in DSS-treated (inflamed) colons and decreased upon addition of CBX (Figure 5C). Inflammation was further underscored by immunostaining using CD68 antibody. As shown in Figure 5D, levels of infiltrated CD68+ cells (macrophages) increased significantly by six-fold in colons of DSS-treated mice as opposed to those of control or CBX-treated mice alone (P < 0.0001). In accordance with CBX effects on inflammatory mediators and histological aspects, CBX injections following DSS exposure resulted in a decreased amount of macrophages. Sections from colons of mice in the DSS group also showed considerable levels of TET-2 protein in epithelial cells with neighboring macrophages, both indicative of an active inflammatory process. TET-2 levels increased further in colons of CBX-treated mice. In addition, and consistent with transcriptional and translational data, CBX-treated mice had upregulated TET-2 protein levels in their colons (Figure 5D). Immunofluorescent micrographs reflect TET-2 level increase in mucosal cells, though not to the same extent as qPCR data; due to the contribution of TET-2 mRNA from CD68+ macrophages in the vicinity of IECs.
These data indicate that inhibiting GJs in inflamed tissues mitigates DSS-induced inflammation. In addition, the increase in Cx43 expression upon exposure to DSS is accompanied by an increase in TET-2 protein expression levels, and disabling GJs by CBX is paralleled by a significant increase in TET-2 levels. Data therefore suggest that, under inflammatory conditions (DSS), GJ inhibition by CBX restores Cx43 levels down to control. In accordance with in vitro data where HT-29 Cx43- cells had upregulated TET-2 levels, chemical inhibition of Cx43 in vivo also was accompanied by increased TET-2 levels.
Collectively, in vitro and in vivo data indicate that under inflammatory conditions, levels of both TET-2 and Cx43 increase.
In an attempt to correlate in vitro and in vivo findings with clinical data, FFPE colon biopsies from normal non-dysplastic non-inflamed colons (n = 7), ulcerative colitis (n = 5), and colon adenocarcinoma (n = 7) patients were obtained anonymously. Sections were stained with H&E for light microscopy, as well as with antibodies against CD68 (macrophage marker), Cx43, and TET-2 for immunofluorescence analyses.
Histological staining exposed a clearly disorganized intestinal mucosal layer in ulcerative colitis and sporadic colon adenocarcinoma tissues as compared to normal tissues (Figure 6A). The disrupted architecture was due to extensive immune activity in the tissues underlying the mucosa. Figure 6B shows CD68+ macrophages infiltrating tissues from ulcerative colitis patients (P < 0.0001), underscoring the inflammatory profile. Tissues obtained from patients with sporadic adenocarcinoma of the colon also displayed macrophage foci, but to a much lesser extent than ulcerative colitis tissues (P < 0.001). Although comparable to control in terms of fluorescence intensity, CD68+ cells in the adenocarcinoma specimen were not uniformly distributed in the tissue.
Moreover, Cx43 and TET-2 expression and localization were evaluated by immunofluorescence (Figure 6C and D). Consistent with data obtained in vitro (Figures 2 and 3) and in vivo (Figure 5), Cx43 protein levels were significantly increased in ulcerative colitis (i.e. inflamed) tissues (P < 0.005) and sharply decreased in adenocarcinoma. Unlike in vitro and in vivo data that reflect parallel patterns of Cx43 and TET-2 expression in inflammatory conditions, Cx43 level increase in ulcerative colitis was accompanied by a decrease in TET-2 expression (P < 0.001). This observation, however, resembles the in vitro scenario illustrated in Figure 2. In fact, while TET-2 expression had increased upon exposure of cells to an inflammatory milieu, Cx43 upregulation in HT-29 Cx43D cells was associated with lower overall TET-2 expression compared to parental cells, and Cx43 knockdown resulted in elevated TET-2 levels.
In various pathologies, including inflammation and cancer, the alteration of the expression, regulation, and association of junctional complexes with other proteins have been reported. Epigenetic methylation and demethylation processes are also major players in a cell’s malignant transformation. Specifically, GJ-forming Cx43 and demethylating enzyme TET-2 have both been documented to be impaired in inflammation and cancer[10-12,19,20].
Studies have shown that the inflammatory response relies in part on intercellular communication mediated by channel-forming connexins. The role of GJ-mediated signaling in the initiation of inflammation is mainly through the transfer of ATP between neighboring cells. ATP molecules are then released in the extracellular environment where they act as signaling molecules that activate purinergic receptors, amplifying the inflammatory response[10]. Wong et al[11] partly elucidated the role of Cx43 in intestinal epithelial barrier injury. TLR activation in the disrupted intestinal epithelium translates intracellularly into Cx43 transcription, translation, post-translational modification, and assembly into connexons. GJIC regulates intestinal epithelial function during both acute and chronic inflammation[11]. Furthermore, Al-Ghadban et al[12] demonstrated a role of GJs in the pathogenesis of IBD by a direct communication between IECs and macrophages enhanced by basement membrane degradation[12]. Long-term IBD associates with amplified risk of colon carcinogenesis, and the deregulation of connexin expression is regarded as one of the hallmarks of different cancers. Shilova et al[44] reviewed in vitro and in vivo studies that point at a correlation between the loss of connexin expression and cancer onset. In fact, downregulation of Cx26, Cx32, and Cx43 accompanies the development of human bladder cancer, hepatocarcinoma, and breast cancer, respectively[44]. A recent study on breast cancer proposed that Cx43 upregulation in a triple negative breast cancer cell line led to a reversal of its mesenchymal phenotype in vitro and to a decreased metastatic potential in vivo[7]. While connexin downregulation is observed in many cancers, elevated Cx43 levels have also been associated with increased malignancy. In a hepatocellular carcinoma cell line, Cx43 was shown to enhance malignancy by inhibiting Cx32-mediated GJIC[45], and, in prostate cancer, Cx43 promoted invasion and metastasis[46]. Variability of data on connexin involvement in cancer onset and progression warrants more research to identify the underlying factors that dictate a tumor-suppressing or tumor-enhancing activity of connexins. Epigenetic regulation of various genes could explain the differential expression of tumor-suppressor and tumor-enhancer genes in inflammation and cancer onset and progression.
The role of demethylating enzyme TET-2 in IBD and colorectal adenocarcinoma has not been fully described. In fact, TET proteins have mostly been studied in hematological malignancies. TET-1 was characterized as a partner gene to the Mixed-Lineage Leukemia gene in acute myeloid leukemia (AML), and the TET-2 gene was described in a study on the myelodysplastic syndrome[47]. Recent investigations report that frequent point mutations in the TET-2 gene can lead to the truncation of the resulting protein and loss of enzymatic activity, reflected by reduced global levels of the 5-hmC marks in AML patients[41,48]. However, although mutations in the TET-1 and TET-3 genes are rare in hematological malignancies compared to TET-2, both still exhibit a tumor-suppressor role: TET-1 in B-cell lymphomas[41] and TET-3, together with TET-2, in murine aggressive myeloid cancer[49]. Mutations and reduced expression of TET proteins were also observed in solid tumors, and decreased translational levels of TETs seem like an important hallmark of different cancers, including cancers of the digestive system[50-52].
In addition to carcinogenesis, a study by Zhang et al[19] shows an anti-inflammatory role for TET-2, which, under inflammatory conditions, repressed IL-6 transcription in dendritic cells and macrophages[19]. Moreover, TET-2-deficient bone marrow-derived dendritic cells and macrophages had high transcriptional levels of IL-6 upon LPS challenge. Another study also reported an increase of TET-2 transcriptional levels in colon tissues from patients with IBD[53].The present study examined the expression of Cx43 and TET-2 in an in vitro model of colon epithelium, where HT-29 cells were used in their parental state or with upregulated or knocked-out Cx43 in the presence or absence of inflammation (induced by DSS or by the addition of an inflammatory medium). A DSS-induced colitis murine model was used to describe how inflammation modulates the expression of Cx43 and TET-2. Data were then compared to Cx43 and TET-2 expression levels in tissues obtained from patients with ulcerative colitis or sporadic colorectal cancer.
Results from this study indicate that knockout of Cx43 in the HT-29 intestinal cell line leads to disruption of membrane integrity exacerbated by the DSS chemical insult. In Cx43-deficient cells and in mice that were subjected to chemical inhibition of GJs by CBX, loss of GJs was paralleled with increased TET-2 expression, further intensified in the presence of inflammation. In fact, inflammation triggered an upregulation of Cx43 and TET-2 expression in all cell subsets, which is in accordance with the literature[19,47]. An exploratory analysis of human colon samples obtained from ulcerative colitis patients revealed an increase in Cx43 and a decrease in TET-2 protein levels accompanied by the disruption of colon architecture. Though these findings are not aligned with the overall trend implicating TET-2 in intestinal inflammatory conditions, little is known about characteristics of patients whose colon biopsies were examined, including potential pharmacological agents that could lead to TET-2 degradation via one of the four pathways reviewed by Cong et al[54] in October 2020: A caspase-dependent pathway, calpain1-promoted degradation of TET-2, proteasome-dependent degradation, and p53-facilitated autophagy of TET-2 proteins[54]. Moreover, TET-2 seems to be implicated in both initiation of the inflammatory process by activating innate pro-inflammatory signaling pathways and its resolution by inducing the repression of pro-inflammatory mediators[55]. Therefore, the exact status of TET-2 in the colon of IBD patients remains to be elucidated, in light of multiple factors governing post-transcriptional and post-translational TET-2 regulation. In specimens from sporadic colon adenocarcinoma, both TET-2 and Cx43 proteins were considerably downregulated, underscoring the tumor-suppressor role of Cx43.
In the context of IBD (specifically ulcerative colitis) and colon cancer, we propose that Cx43 upregulation in inflamed colons could attenuate or slow down the onset of malignancy. Therefore, loss of Cx43 in tissues from colon cancer could be attributed to a malignant switch, turning off tumor-suppressor genes and activating tumor-promoting genes. The presence of CD68+ cells (macrophages) was concomitant with loss of TET-2 expression in the colon adenocarcinoma samples. This could be explained by an immune-active phenotype in tumors, with tumor-associated macrophages having little to no TET-2 expression[55]. A 2018 study reported increased levels of TET-2 in T lymphocytes from colorectal tumor tissues, leading to demethylation and activation of FOXP3 and regulation of regulatory T cell (Treg) function[56], further implicating TET-2 in immune-related processes. In particular, TET-2 has been reported to regulate the innate immune response[57,58]. While it serves to activate the inflammatory response, TET-2 has also been implicated in inflammation resolution, where in response to IL-1/RMyD88 signaling, TET-2 downregulates the expression of inflammatory cytokines, such as IL-6 in innate myeloid cells and IL-1β in macrophages by recruiting HDACs for histone deacetylation[19,57]. This property might explain low levels of TET-2 in human ulcerative colitis samples where unresolved inflammation can be attributed to persistent inflammatory cytokines.
TET-2 status and role in digestive cancers remain unclear and variable, depending on the cancer site and grade, but also on possible mutations that could lead to cancer progression and resistance to chemotherapy, as recently reviewed[21].
Data presented in this manuscript show that exposure of intestinal cells to inflammation is associated with Cx43 and TET-2 upregulation in vitro and in vivo. We propose that, as part of its potential anti-inflammatory role and through its demethylating activity, TET-2 might be responsible for promoting the expression of anti-inflammatory genes and for indirectly repressing the expression of pro-inflammatory genes under inflammatory conditions. Similarly, we hypothesize that the demethylating TET-2 enzyme might promote the expression of tumor-suppressor genes (among others, Cx43) in the inflamed colon. Extrapolating from observations made on human colon samples, a malignant switch could happen in chronically inflamed colons. This malignant switch could be associated with downregulation of TET-2 (and potentially other TET enzymes[20,23]) in ulcerative colitis, possibly resulting in the hypermethylation of genes that are relevant in the context of carcinogenesis, such as Cx43. Although we present evidence on the modulation of TET-2 expression and we reiterate the role of Cx43 in intestinal inflammation, further investigation is under way to more solidly explore the mechanism of action behind a potential interplay between Cx43 and TET-2 that might, at least partly and indirectly, bridge chronic inflammation (such as in IBD) and IBD-induced carcinogenesis in the colon.
Patients with inflammatory bowel disease (IBD) have a higher propensity to acquire colorectal cancer. The link between inflammation and cancer has long been established; however, the molecular players in the switch to cancer are still poorly defined.
In previous work, we have demonstrated a potential role for connexin 43 (Cx43) in inflamed intestinal cells. Recognizing the tumor-suppressor role of Cx43 in several cancers, we set out to explore whether the loss of Cx43 is associated with the switch to carcinogenesis. One potential mechanism for downregulating Cx43 expression is through methylation. We hypothesized that ten-eleven translocation-2 (TET-2), a demethylating enzyme, previously described to have a role in inflammation, may be involved in this process.
Using cell culture, a colitis animal model, and archived human tissues, we assessed the expression of both Cx43 and TET-2 in intestinal inflammation. Specific objectives include: (1) Assessment of the expression levels of Cx43 and TET-2 under inflammatory conditions; (2) Assessment of the expression and activity of TET-2 in HT-29 cell lines with up- or down-regulated for Cx43 expression; (3) Reproduction of a colitis mouse model and assessment of Cx43 and TET-2 levels in colons of mice; and (4) Explore Cx43 and TET-2 expression levels in archived biopsies obtained from patients with ulcerative colitis and colon adenocarcinoma.
This study employed several modalities to verify the hypothesis, which include intestinal epithelial cell (IEC) line modified for Cx43 expression grown under inflammatory conditions. A dextran sulfate sodium-induced colitis mouse model was reproduced and tissues from different experimental conditions were analyzed. Gene expression profile, protein expression levels, morphology and cellular localization were described. In addition, archived formalin-fixed paraffin-embedded tissues were sectioned and evaluated for Cx43 and TET-2 expression.
In vitro, TET-2 expression was elevated under inflammatory conditions and even more so in HT-29 cells devoid of Cx43. The barrier function of IECs was breached when Cx43 levels were down-regulated. These results were corroborated in the murine colitis model. In archived biopsies from ulcerative colitis patients, Cx43 expression was upregulated compared to non-inflamed counterparts. In sporadic colon adenocarcinoma sections, both TET-2 and Cx43 expression levels were decreased.
Under inflammatory conditions, levels of Cx43 and of the demethylating enzyme TET-2 are upregulated. Through demethylation, TET-2 would turn on the expression of several factors involved in inflammation (presumably Cx43 included). When TET-2 levels were diminished in sporadic colon adenocarcinoma, we also observed that Cx43 was downregulated, which may indicate a role of TET-2 in shutting down Cx43 and its tumor-suppressing potential.
In vitro, manipulation of TET-2 levels in intestinal cells may yield further insight into the mechanism of action. Methylation studies will also be undertaken. The animal model will be expanded to allow for the development of colon carcinoma, and timed evaluation of molecular players will be performed. More stringent criteria will be implemented for prospective tissue collection from non-inflamed subjects, patients with IBD, and with IBD-associated colorectal cancer.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Lebanon
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): E
P-Reviewer: Feng Y, China; Ruan X, China; Wan XH, China A-Editor: Nakaji K, Japan S-Editor: Fan JR L-Editor: Filipodia P-Editor: Cai YX
| 1. | Khor B, Gardet A, Xavier RJ. Genetics and pathogenesis of inflammatory bowel disease. Nature. 2011;474:307-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2019] [Cited by in RCA: 1883] [Article Influence: 134.5] [Reference Citation Analysis (2)] |
| 2. | Gajendran M, Loganathan P, Catinella AP, Hashash JG. A comprehensive review and update on Crohn's disease. Dis Mon. 2018;64:20-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 328] [Article Influence: 41.0] [Reference Citation Analysis (1)] |
| 3. | Gajendran M, Loganathan P, Jimenez G, Catinella AP, Ng N, Umapathy C, Ziade N, Hashash JG. A comprehensive review and update on ulcerative colitis. Dis Mon. 2019;65:100851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 343] [Article Influence: 57.2] [Reference Citation Analysis (0)] |
| 4. | Fleming M, Ravula S, Tatishchev SF, Wang HL. Colorectal carcinoma: Pathologic aspects. J Gastrointest Oncol. 2012;3:153-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 274] [Reference Citation Analysis (0)] |
| 5. | Du L, Kim JJ, Shen J, Chen B, Dai N. KRAS and TP53 mutations in inflammatory bowel disease-associated colorectal cancer: a meta-analysis. Oncotarget. 2017;8:22175-22186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 6. | Beyer EC, Berthoud VM. Gap junction gene and protein families: Connexins, innexins, and pannexins. Biochim Biophys Acta Biomembr. 2018;1860:5-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 149] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 7. | Kazan JM, El-Saghir J, Saliba J, Shaito A, Jalaleddine N, El-Hajjar L, Al-Ghadban S, Yehia L, Zibara K, El-Sabban M. Cx43 Expression Correlates with Breast Cancer Metastasis in MDA-MB-231 Cells In Vitro, In a Mouse Xenograft Model and in Human Breast Cancer Tissues. Cancers (Basel). 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 8. | Kou Y, Ji L, Wang H, Wang W, Zheng H, Zou J, Liu L, Qi X, Liu Z, Du B, Lu L. Connexin 43 upregulation by dioscin inhibits melanoma progression via suppressing malignancy and inducing M1 polarization. Int J Cancer. 2017;141:1690-1703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 9. | Zhou JZ, Riquelme MA, Gu S, Kar R, Gao X, Sun L, Jiang JX. Osteocytic connexin hemichannels suppress breast cancer growth and bone metastasis. Oncogene. 2016;35:5597-5607. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 76] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 10. | Willebrords J, Crespo Yanguas S, Maes M, Decrock E, Wang N, Leybaert L, Kwak BR, Green CR, Cogliati B, Vinken M. Connexins and their channels in inflammation. Crit Rev Biochem Mol Biol. 2016;51:413-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 94] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 11. | Wong J, Chopra J, Chiang LLW, Liu T, Ho J, Wu WKK, Tse G, Wong SH. The Role of Connexins in Gastrointestinal Diseases. J Mol Biol. 2019;431:643-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Al-Ghadban S, Kaissi S, Homaidan FR, Naim HY, El-Sabban ME. Cross-talk between intestinal epithelial cells and immune cells in inflammatory bowel disease. Sci Rep. 2016;6:29783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 13. | Amatullah H, Jeffrey KL. Epigenome-metabolome-microbiome axis in health and IBD. Curr Opin Microbiol. 2020;56:97-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 14. | Grosser C, Wagner N, Grothaus K, Horsthemke B. Altering TET dioxygenase levels within physiological range affects DNA methylation dynamics of HEK293 cells. Epigenetics. 2015;10:819-833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Ryan FJ, Ahern AM, Fitzgerald RS, Laserna-Mendieta EJ, Power EM, Clooney AG, O'Donoghue KW, McMurdie PJ, Iwai S, Crits-Christoph A, Sheehan D, Moran C, Flemer B, Zomer AL, Fanning A, O'Callaghan J, Walton J, Temko A, Stack W, Jackson L, Joyce SA, Melgar S, DeSantis TZ, Bell JT, Shanahan F, Claesson MJ. Colonic microbiota is associated with inflammation and host epigenomic alterations in inflammatory bowel disease. Nat Commun. 2020;11:1512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 185] [Article Influence: 37.0] [Reference Citation Analysis (1)] |
| 16. | Pekow J, Hernandez K, Meckel K, Deng Z, Haider HI, Khalil A, Zhang C, Talisila N, Siva S, Jasmine F, Li YC, Rubin DT, Hyman N, Bissonnette M, Weber C, Kibriya MG. IBD-associated Colon Cancers Differ in DNA Methylation and Gene Expression Profiles Compared With Sporadic Colon Cancers. J Crohns Colitis. 2019;13:884-893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 17. | Liu XS, Wu H, Ji X, Stelzer Y, Wu X, Czauderna S, Shu J, Dadon D, Young RA, Jaenisch R. Editing DNA Methylation in the Mammalian Genome. Cell. 2016;167:233-247.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 850] [Article Influence: 94.4] [Reference Citation Analysis (0)] |
| 18. | Wawrzyniak M, Scharl M. Genetics and epigenetics of inflammatory bowel disease. Swiss Med Wkly. 2018;148:w14671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 19. | Zhang Q, Zhao K, Shen Q, Han Y, Gu Y, Li X, Zhao D, Liu Y, Wang C, Zhang X, Su X, Liu J, Ge W, Levine RL, Li N, Cao X. Tet2 is required to resolve inflammation by recruiting Hdac2 to specifically repress IL-6. Nature. 2015;525:389-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 633] [Article Influence: 63.3] [Reference Citation Analysis (0)] |
| 20. | Rasmussen KD, Helin K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 2016;30:733-750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 583] [Cited by in RCA: 787] [Article Influence: 87.4] [Reference Citation Analysis (0)] |
| 21. | Wang F, Zhang J, Qi J. Ten-eleven translocation-2 affects the fate of cells and has therapeutic potential in digestive tumors. Chronic Dis Transl Med. 2019;5:267-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 22. | Du C, Kurabe N, Matsushima Y, Suzuki M, Kahyo T, Ohnishi I, Tanioka F, Tajima S, Goto M, Yamada H, Tao H, Shinmura K, Konno H, Sugimura H. Robust quantitative assessments of cytosine modifications and changes in the expressions of related enzymes in gastric cancer. Gastric Cancer. 2015;18:516-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 23. | Rawłuszko-Wieczorek AA, Siera A, Horbacka K, Horst N, Krokowicz P, Jagodziński PP. Clinical significance of DNA methylation mRNA levels of TET family members in colorectal cancer. J Cancer Res Clin Oncol. 2015;141:1379-1392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 24. | Lou H, Li H, Ho KJ, Cai LL, Huang AS, Shank TR, Verneris MR, Nickerson ML, Dean M, Anderson SK. The Human TET2 Gene Contains Three Distinct Promoter Regions With Differing Tissue and Developmental Specificities. Front Cell Dev Biol. 2019;7:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Aasen T, Leithe E, Graham SV, Kameritsch P, Mayán MD, Mesnil M, Pogoda K, Tabernero A. Connexins in cancer: bridging the gap to the clinic. Oncogene. 2019;38:4429-4451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 144] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 26. | Wang Y, Huang LH, Xu CX, Xiao J, Zhou L, Cao D, Liu XM, Qi Y. Connexin 32 and 43 promoter methylation in Helicobacter pylori-associated gastric tumorigenesis. World J Gastroenterol. 2014;20:11770-11779. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 30] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 27. | Yi ZC, Wang H, Zhang GY, Xia B. Downregulation of connexin 43 in nasopharyngeal carcinoma cells is related to promoter methylation. Oral Oncol. 2007;43:898-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Ferraretto A, Bottani M, De Luca P, Cornaghi L, Arnaboldi F, Maggioni M, Fiorilli A, Donetti E. Morphofunctional properties of a differentiated Caco2/HT-29 co-culture as an in vitro model of human intestinal epithelium. Biosci Rep. 2018;38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 29. | Lea T, Verhoeckx K, Cotter P, López-Expósito I, Kleiveland C, Lea T, Mackie A, Requena T, Swiatecka D, Wichers H. Caco-2 Cell Line. In: The Impact of Food Bioactives on Health: in vitro and ex vivo models [Internet]. Cham (CH): Springer; 2015. Chapter 10. [PubMed] |
| 30. | Hyun J, Romero L, Riveron R, Flores C, Kanagavelu S, Chung KD, Alonso A, Sotolongo J, Ruiz J, Manukyan A, Chun S, Singh G, Salas P, Targan SR, Fukata M. Human intestinal epithelial cells express interleukin-10 through Toll-like receptor 4-mediated epithelial-macrophage crosstalk. J Innate Immun. 2015;7:87-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 31. | Sáez PJ, Shoji KF, Aguirre A, Sáez JC. Regulation of hemichannels and gap junction channels by cytokines in antigen-presenting cells. Mediators Inflamm. 2014;2014:742734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 32. | Poenar DP, Yang G, Wan WK, Feng S. Low-Cost Method and Biochip for Measuring the Trans-Epithelial Electrical Resistance (TEER) of Esophageal Epithelium. Materials (Basel). 2020;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 33. | Yao L, Xue X, Yu P, Ni Y, Chen F. Evans Blue Dye: A Revisit of Its Applications in Biomedicine. Contrast Media Mol Imaging. 2018;2018:7628037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 101] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 34. | Mizoguchi A. Animal models of inflammatory bowel disease. Prog Mol Biol Transl Sci. 2012;105:263-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 186] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 35. | Dieleman LA, Ridwan BU, Tennyson GS, Beagley KW, Bucy RP, Elson CO. Dextran sulfate sodium-induced colitis occurs in severe combined immunodeficient mice. Gastroenterology. 1994;107:1643-1652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 508] [Cited by in RCA: 543] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 36. | Kitajima S, Takuma S, Morimoto M. Tissue distribution of dextran sulfate sodium (DSS) in the acute phase of murine DSS-induced colitis. J Vet Med Sci. 1999;61:67-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 67] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Toutounji M, Wanes D, El-Harakeh M, El-Sabban M, Rizk S, Naim HY. Dextran Sodium Sulfate-Induced Impairment of Protein Trafficking and Alterations in Membrane Composition in Intestinal Caco-2 Cell Line. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 38. | Viennois E, Chen F, Laroui H, Baker MT, Merlin D. Dextran sodium sulfate inhibits the activities of both polymerase and reverse transcriptase: lithium chloride purification, a rapid and efficient technique to purify RNA. BMC Res Notes. 2013;6:360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 135] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 39. | Feng Y, Li X, Cassady K, Zou Z, Zhang X. TET2 Function in Hematopoietic Malignancies, Immune Regulation, and DNA Repair. Front Oncol. 2019;9:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 87] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 40. | Ko M, Huang Y, Jankowska AM, Pape UJ, Tahiliani M, Bandukwala HS, An J, Lamperti ED, Koh KP, Ganetzky R, Liu XS, Aravind L, Agarwal S, Maciejewski JP, Rao A. Impaired hydroxylation of 5-methylcytosine in myeloid cancers with mutant TET2. Nature. 2010;468:839-843. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1103] [Cited by in RCA: 1057] [Article Influence: 70.5] [Reference Citation Analysis (0)] |
| 41. | Weissmann S, Alpermann T, Grossmann V, Kowarsch A, Nadarajah N, Eder C, Dicker F, Fasan A, Haferlach C, Haferlach T, Kern W, Schnittger S, Kohlmann A. Landscape of TET2 mutations in acute myeloid leukemia. Leukemia. 2012;26:934-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 192] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 42. | Homaidan FR, Chakroun, Dbaibo GS, El-Assaad W, El-Sabban ME. IL-1 activates two phospholipid signaling pathways in intestinal epithelial cells. Inflamm Res. 2001;50:375-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 43. | Homaidan FR, El-Sabban ME, Chakroun I, El-Sibai M, Dbaibo GS. IL-1 stimulates ceramide accumulation without inducing apoptosis in intestinal epithelial cells. Mediators Inflamm. 2002;11:39-45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 44. | Shilova ON, Shilov ES, Lieber A, Deyev SM. Disassembling a cancer puzzle: Cell junctions and plasma membrane as targets for anticancer therapy. J Control Release. 2018;286:125-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 45. | Zhang D, Kaneda M, Nakahama K, Arii S, Morita I. Connexin 43 expression promotes malignancy of HuH7 hepatocellular carcinoma cells via the inhibition of cell-cell communication. Cancer Lett. 2007;252:208-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 46. | Zhang A, Hitomi M, Bar-Shain N, Dalimov Z, Ellis L, Velpula KK, Fraizer GC, Gourdie RG, Lathia JD. Connexin 43 expression is associated with increased malignancy in prostate cancer cell lines and functions to promote migration. Oncotarget. 2015;6:11640-11651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 47. | Langemeijer SM, Kuiper RP, Berends M, Knops R, Aslanyan MG, Massop M, Stevens-Linders E, van Hoogen P, van Kessel AG, Raymakers RA, Kamping EJ, Verhoef GE, Verburgh E, Hagemeijer A, Vandenberghe P, de Witte T, van der Reijden BA, Jansen JH. Acquired mutations in TET2 are common in myelodysplastic syndromes. Nat Genet. 2009;41:838-842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 612] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 48. | Zhao Z, Chen L, Dawlaty MM, Pan F, Weeks O, Zhou Y, Cao Z, Shi H, Wang J, Lin L, Chen S, Yuan W, Qin Z, Ni H, Nimer SD, Yang FC, Jaenisch R, Jin P, Xu M. Combined Loss of Tet1 and Tet2 Promotes B Cell, but Not Myeloid Malignancies, in Mice. Cell Rep. 2015;13:1692-1704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 85] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 49. | Zibara K, Awada Z, Dib L, El-Saghir J, Al-Ghadban S, Ibrik A, El-Zein N, El-Sabban M. Anti-angiogenesis therapy and gap junction inhibition reduce MDA-MB-231 breast cancer cell invasion and metastasis in vitro and in vivo. Sci Rep. 2015;5:12598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 50. | Yang H, Liu Y, Bai F, Zhang JY, Ma SH, Liu J, Xu ZD, Zhu HG, Ling ZQ, Ye D, Guan KL, Xiong Y. Tumor development is associated with decrease of TET gene expression and 5-methylcytosine hydroxylation. Oncogene. 2013;32:663-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 472] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 51. | Lian CG, Xu Y, Ceol C, Wu F, Larson A, Dresser K, Xu W, Tan L, Hu Y, Zhan Q, Lee CW, Hu D, Lian BQ, Kleffel S, Yang Y, Neiswender J, Khorasani AJ, Fang R, Lezcano C, Duncan LM, Scolyer RA, Thompson JF, Kakavand H, Houvras Y, Zon LI, Mihm MC Jr, Kaiser UB, Schatton T, Woda BA, Murphy GF, Shi YG. Loss of 5-hydroxymethylcytosine is an epigenetic hallmark of melanoma. Cell. 2012;150:1135-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 581] [Cited by in RCA: 625] [Article Influence: 48.1] [Reference Citation Analysis (0)] |
| 52. | Liu C, Liu L, Chen X, Shen J, Shan J, Xu Y, Yang Z, Wu L, Xia F, Bie P, Cui Y, Bian XW, Qian C. Decrease of 5-hydroxymethylcytosine is associated with progression of hepatocellular carcinoma through downregulation of TET1. PLoS One. 2013;8:e62828. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 130] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 53. | Cong B, Zhang Q, Cao X. The function and regulation of TET2 in innate immunity and inflammation. Protein Cell. 2021;12:165-173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 54. | Dziaman T, Gackowski D, Guz J, Linowiecka K, Bodnar M, Starczak M, Zarakowska E, Modrzejewska M, Szpila A, Szpotan J, Gawronski M, Labejszo A, Liebert A, Banaszkiewicz Z, Klopocka M, Foksinski M, Marszalek A, Olinski R. Characteristic profiles of DNA epigenetic modifications in colon cancer and its predisposing conditions-benign adenomas and inflammatory bowel disease. Clin Epigenetics. 2018;10:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 55. | Pan W, Zhu S, Qu K, Meeth K, Cheng J, He K, Ma H, Liao Y, Wen X, Roden C, Tobiasova Z, Wei Z, Zhao J, Liu J, Zheng J, Guo B, Khan SA, Bosenberg M, Flavell RA, Lu J. The DNA Methylcytosine Dioxygenase Tet2 Sustains Immunosuppressive Function of Tumor-Infiltrating Myeloid Cells to Promote Melanoma Progression. Immunity. 2017;47:284-297.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 131] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 56. | Ma H, Gao W, Sun X, Wang W. STAT5 and TET2 Cooperate to Regulate FOXP3-TSDR Demethylation in CD4+ T Cells of Patients with Colorectal Cancer. J Immunol Res. 2018;2018:6985031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 57. | Cull AH, Snetsinger B, Buckstein R, Wells RA, Rauh MJ. Tet2 restrains inflammatory gene expression in macrophages. Exp Hematol. 2017;55:56-70.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 215] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 58. | Montagner S, Leoni C, Emming S, Della Chiara G, Balestrieri C, Barozzi I, Piccolo V, Togher S, Ko M, Rao A, Natoli G, Monticelli S. TET2 Regulates Mast Cell Differentiation and Proliferation through Catalytic and Non-catalytic Activities. Cell Rep. 2016;15:1566-1579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 69] [Article Influence: 7.7] [Reference Citation Analysis (0)] |