Published online May 14, 2022. doi: 10.3748/wjg.v28.i18.1922
Peer-review started: November 13, 2021
First decision: January 9, 2022
Revised: January 22, 2022
Accepted: March 26, 2022
Article in press: March 26, 2022
Published online: May 14, 2022
Processing time: 179 Days and 13 Hours
Inflammatory bowel disease (IBD) is a chronic illness characterized by relapsing inflammation of the intestines. The disorder is stratified according to the severity and is marked by its two main phenotypical representations: Ulcerative colitis and Crohn’s disease. Pathogenesis of the disease is ambiguous and is expected to have interactivity between genetic disposition, environmental factors such as bacterial agents, and dysregulated immune response. Treatment for IBD aims to reduce symptom extent and severity and halt disease progression. The mainstay drugs have been 5-aminosalicylates (5-ASAs), corticosteroids, and immunosuppressive agents. Parenteral, oral and rectal routes are the conventional methods of drug delivery, and among all, oral administration is most widely adopted. However, problems of systematic drug reactions and low specificity in delivering drugs to the inflamed sites have emerged with these regular routes of delivery. Novel drug delivery systems have been introduced to overcome several therapeutic obstacles and for localized drug delivery to target tissues. Enteric-coated microneedle pills, various nano-drug delivery techniques, prodrug systems, lipid-based vesicular systems, hybrid drug delivery systems, and biologic drug delivery systems constitute some of these novel methods. Microneedles are painless, they dislodge their content at the affected site, and their release can be prolonged. Recombinant bacteria such as genetically engineered Lactococcus Lactis and eukaryotic cells, including GM immune cells and red blood cells as nanoparticle carriers, can be plausible delivery methods when evaluating biologic systems. Nano-particle drug delivery systems consisting of various techniques are also employed as nanoparticles can penetrate through inflamed regions and adhere to the thick mucus of the diseased site. Prodrug systems such as 5-ASAs formulations or their derivatives are effective in reducing colonic damage. Liposomes can be modified with both hydrophilic and lipophilic particles and act as lipid-based vesicular systems, while hybrid drug delivery systems containing an internal nanoparticle section for loading drugs are potential routes too. Leukosomes are also considered as possible carrier systems, and results from mouse models have revealed that they control anti- and pro-inflammatory molecules.
Core Tip: Current literature on novel drug delivery systems is extensive. However, limited content is available that focuses on the novel drug delivery strategies, focally the enteric-coated microneedles pills and biological drug delivery systems. In our review, we discuss multiple novel approaches comprehensively, after outlining the disease course in inflammatory bowel diseases (IBD) and how the conventional routes of drug administrations are deficient in addressing some aspects of disease treatment. We specify that novel carrier systems can be considered for targeting inflamed sites in IBD and discuss the implications of choosing such routes.
- Citation: Yasmin F, Najeeb H, Shaikh S, Hasanain M, Naeem U, Moeed A, Koritala T, Hasan S, Surani S. Novel drug delivery systems for inflammatory bowel disease. World J Gastroenterol 2022; 28(18): 1922-1933
- URL: https://www.wjgnet.com/1007-9327/full/v28/i18/1922.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i18.1922
Inflammatory bowel disease (IBD) is chronic, relapsing intestinal inflammation[1]. The disorder is most common in Western industrialized countries[2] and is mainly characterized by the two idiopathic phenotypes of IBD; Ulcerative colitis (UC) and Crohn’s disease (CD)[3]. The illness can be classified as mild, moderate, or severe based on clinical, para clinical parameters and symptoms such as abdominal spasms, rectal bleeding[4]. Intestinal and extraintestinal complications also manifest in the disease[2], and IBD is known to significantly affect the quality of life of the patients[4].
The main aim of IBD treatment is to ameliorate patients’ quality of life[5] by diminishing the extent and symptoms of intestinal inflammation[3], revitalizing nutrition lost, assisting psychosocially, and altering disease course in those undergoing severe forms[5]. Therapy is decided after determining the type of disease and site involved[3]. Routine treatment options for mild attacks include 5-aminosalicylates (5-ASA) such as mesalazine or olsalazine, while for moderate to severe IBD, corticosteroids are prescribed. Probiotics, antibiotics, chemically modified drugs, enzymatic, prodrug therapy, enteric-coated drugs can be used in the early stages, too[5]. For severe disease stages, immunosuppressive agents like azathioprine and anti-TNF-α-antibodies are specified[2]. Surgery is performed in the refractory or fulminate disease stages[2] and is reported to be required by 30%-40% UC and all CD patients at least once in a lifetime[3].
The IBD medication administration's modes are the conventional parenteral, peroral, and rectal routes[2], while the peroral route is most preferred, indicated for targeting the colon[3]. However, there is variable intestinal absorption via this route, and treating intermittent inflamed areas is challenging by oral drug delivery systems[2]. Systematic drug reactions by absorption of luminal released active agents through the healthy and inflamed mucosal wall into systematic circulation, inability to deliver to an affected gastrointestinal section only[2] and the reliance of the conventional delivery systems on unstable parameters such as intestinal motility, time, pH, and microbial and enzymatic degradation for optimal drug delivery[5] have rendered these standard methods unsatisfactory.
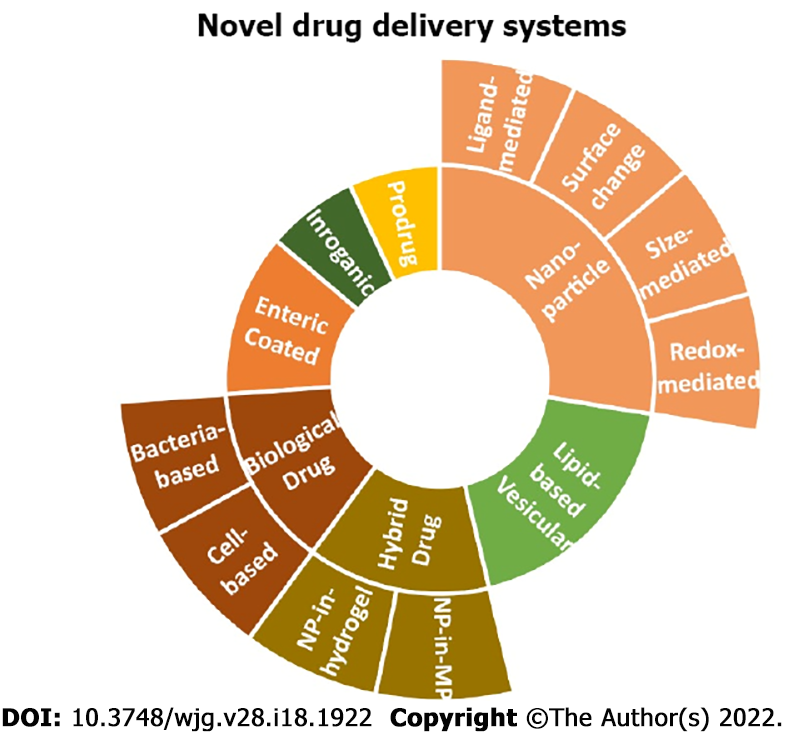
To balance therapeutic efficacy and risk of adverse side effects[2], novel drug delivery systems have been suggested that provide a more localized delivery and circumvent unnecessary drug exposure[6]. Inspired by the German physician Paul Ehrlich who first proposed drug targeting of active agents to pathogenic or cancerous cells while preventing side effects, the new synthetic/biologic drug delivery systems known[2] incorporate liposomes, solid lipid nanoparticles, nanostructured lipid carriers, nano emulsions, hydrogel, and eukaryotic systems via resealed erythrocytes, macrophages, prokaryotic cells such as bacterial capsules[5]. These innovative approaches minimize drug loss, increase drug bioavailability[5], increase the local concentration of drugs at inflamed tissue and increase patient’s compliance lowering the frequency of drug intake[2]. This review presents an overview of enteric-coated microneedle pills, nano-drug delivery systems, including size-mediated targeting, surface-charged targeting, redox-mediated targeting, and ligand-mediated targeting techniques. Furthermore, prodrug delivery systems, lipid-based vesicular delivery systems, hybrid drug delivery systems, and biologic drug delivery systems are also discussed. All that is being seen as a more disease-oriented[2] approach for restoring gut homeostasis in IBD.
In IBD, the intestinal barrier is disrupted, characterized by a reduced number of secretory cells, which lead to a decrease in antimicrobial secretion, and a loss of epithelium. A decrease in goblet cells at the inflamed site causes a reduction in mucus. Bacteria from the lumina side and immune-related cells infiltrate the mucosa and constitute the pathophysiological state in IBD[2]. The two main phenotypes of IBD are UC and CD, respectively. Inflammation in UC is superficial, limited to the colon. The disease starts in the rectum and extends continuously through the colon. The extent of colonic involvement defines disease distribution; proctitis to left-sided colitis or extensive colitis (pancolitis)[7]. In CD, the inflammation extends to the serosa. The disease is transmural and is found anywhere along the gastrointestinal length, mostly the ileum and colon. Endoscopy reveals mucosa's “sandpaper” appearance in UC, while in CD, the mucosa is cobblestone[8]. The etiology of IBD is unknown, and the interaction of genetic disposition with environmental factors in the microbiome leads to improper immune activation resulting in clinical presentation observed in IBD[9].
Rare genetic variants have been identified in early onset and severe IBD patients indicating a relation with pathways leading to intestinal inflammation. Genome-wide association studies have also identified 201 Loci implicated in IBD[9]. NOD2 is the first gene associated with IBD[9], and is related to CD susceptibility[3]. Environmental agents can trigger IBD in genetically susceptible individuals; medications can alter the intestinal microbiome, which is linked with an increased risk of developing IBD. An increase in bacteria with inflammatory potentialities and a decrease in anti-inflammatory bacteria are reported[9]. Mucolytic bacteria are also found to be increased in the intestine[9]. A dysregulation between anti and pro-inflammatory signals, causing migration of leukocytes to the intestinal mucosa exacerbated by an enhanced T-cell immune response, is noted in UC and CD[9]. Th-2 cell response is dominant in UC and antibodies recruitment. IL-5 is also elevated in UC. However, antigens eliciting the response are not known[3]. Moreover, a Th-1 inflammatory response is signified in CD, resulting in granulomatous inflammation with increased levels of proinflammatory cytokines such as IFN-γ, IL-2, and TNF-α. Suppressor cytokines produced by T regulatory cells are reduced[3].
Microneedle pills were first considered as a fix to deliver drugs through the transdermal system in 1998[10]. Increasing the permeability of the human skin, a painless drug delivery system was formulated. However, the lack of stratum corneum in the GI tract established a strong case for microneedles in oral delivery. Coated microneedles provide extended controlled release reducing the required dosage frequency of many medications[11]. An ingestible microneedle pill for adalimumab, a tumor necrosis factor blocker, developed by Rani Therapeutics LLC[12] demonstrated a higher serum adalimumab concentration for the first eight hours to its administration as compared to the subcutaneous route.
Microneedles are either solid or hollow with the biologics contained in the reservoir, released upon breach of the mucosal lining of the GI tract. Microneedles are prepared using biodegradable polymers and coated by a pH-resistant coating to withstand the acidic range of pH of the gut[13]. Following ingestion, microneedles travel to the required location and spill their contents upon peristalsis and or penetrate the tissue and break off in hollow and solid microneedles, respectively[13].
The retention time of the microneedles was seven days at the minimum in a study[13], with no tissue damage reported. The safety profile and the extended retention warrants its potential use as a drug delivery system in IBD. However, patient-to-patient variation in intraluminal pressure presents a significant challenge along with the impact of food in the GI tract on the efficacy of the drug delivery system[14].
Size-dependent targeting is the most common mechanism for NPs drug delivery systems targeting the inflamed colon. During inflammation, enhanced permeability and retention (EPR) is observed, which increases penetration of nano-particles in the inflamed regions[15-18]. Furthermore, increased secretion of mucus leads to a thick mucus layer which increases the probability of adhesion of nanoparticles[19,20]. The size of mucous fiber networks varies from 10-200 nm; hence modifying the particle size to nano-formulations assists in movement inside the fiber network[21-24]. It is reported that diarrhea affects particles > 200 μm, which are subsequently washed away[25] whereas, neutrophils, macrophages, M cells, and enterocytes rapidly internalize particles < 500 nm via the process of transcytosis[26]. Lamprecht et al[27] compared fluorescent polystyrene particles of sizes 10 mm, 1 mm, and 100 nm following oral gavage among healthy and trinitrobenzene sulfonic acid (TNBS)-induced colitic rats. The 100 nm formulation displayed the greatest bio adhesion to the inflamed colon compared to healthy rats. Another study done on oxazolone-induced colitis in mice reported that budesonide-filled poly lactic-co-glycolic acid (PLGA) NPs of size 200 nm accumulated at the inflamed colon[28]. Similarly, Nagasaki’s group[29,30] developed a nanoparticle sized 40 nm (RNP O), which localized in the inflamed colonic mucosa, and reduced inflammation by consuming the excessive ROS. The increased epithelial enhanced permeability and retention (epEPR) during inflammation leads to increased uptake of NPs and prolongs the retention time[31]. This consequently leads to the accumulation of drugs and increased efficacy. Another study investigated the penetration of fluorescently labeled PLGA particles, sized 250 nm and 3 nm, into rectal mucosa in healthy and IBD human subjects. They reported that significantly more 3 nm particles were found accumulated in the mucosal surface, while most of the 250 nm particles had penetrated inside and translocated to the serosal surface[32]. The probability of systemic absorption increases with smaller particles and could lead to adverse side effects. This difference in the behavior of NPs accumulation in humans and rodent colitis models requires further studies to reach a definite conclusion.
The surface charge of the nanoparticles can be altered to control interaction with the colonic mucosa to increase the efficacy of the drug delivery system. The colonic mucosa comprises negatively charged carbohydrates molecules, sulfates, sialic acid, and colonic mucins, which enable the positively charged NPs to adhere to the mucosal surface due to electrostatic interaction. In a study, clodronate loaded nanoparticles based on cationic polymethacrylate (Eudragit RL) and free clodronate were administered in mice induced with 2,4,6-TNBS and oxazolone colitis. A significant reduction in myeloperoxidase activity was observed in colitis-induced mice models, whereas free clodronate failed to produce ameliorating effect[33]. In an ex vivo study, Lautenschlager demonstrated that cationic chitosan modified PLGA nanoparticles adhered to intestinal mucosa[34]. However, the cation became immobilized after attaching to the mucosa, which led to a premature release of the drug. This was due to the strong electrostatic attraction experienced by the negatively charged mucosa[34]. In comparison, negatively charged NPs precisely attach to specific positively charged proteins and show greater penetration into the intestinal mucosa. During the inflammatory process of colonic mucosa, the epithelium is damaged, and positively charged proteins such as transferrin and eosinophil cationic proteins accumulate[35,36]. This enables nanoparticles with the negative surface charge to adhere to the positively charged mucosal surface due to electrostatic interaction. Jubeh et al[37] studied the varied adhesion of anionic, neutral, and cationic liposomes in healthy and inflamed colon explants from rats inflicted with DNBS colitis. They reported that anionic liposomes were 2-times more likely to adhere to the inflamed mucosa than neutral or cationic liposomes. In healthy colonic mucosa, the cationic liposomes displayed preferential binding, which was 3-fold higher than neutral or anionic liposomes[38]. Similarly, Beloqui et al[39] reported that negatively charged NPs, comprised of budesonide, invaded deeper into mucosa in the colitis group while staying on the mucosal surface in a healthy cohort.
Endogenous ROS are produced because of the physiological metabolism of oxygen. In patients suffering from IBD, increased production of ROS is observed, which is localized to the site of inflammation and correlated with disease progression[40,41]. This increase is due to the inflammatory cells present in the intestinal mucosa[42]. Subsequently, redox-dependent nanoparticle drug delivery systems have emerged[43,44]. Zhang et al developed oxidation-sensitive β-cyclodextrin material (OxβCD), which was loaded with Tpl. This drug delivery system could successfully consume ROS and mitigate symptoms in 3 mouse colitis models[45,46]. Sun et al[47] synthesized redox-sensitive NPs composed of carboxymethyl inulin. The NPs accumulate in the inflamed regions of the intestine. Vong et al[30] developed NPs composed of nitroxide radicals (RNPo), which exhaust the ROS in the inflamed gut regions and thereby reduce inflammation. The group of mice treated with oral RNPo for one week showed reduced disease activity compared with the group administered mesalamine[30]. Li et al[48] synthesized Ac2-26-encapsulated nano therapy using OxβCD. This drug delivery system is ROS-responsive and stimulates the release of Ac2-26 in response to increased ROS concentration in GIT.
In conclusion, since a high concentration of ROS is found in the inflamed regions of GIT[42], redox-dependent targeting via NPs may produce beneficial results. However, volatility in low pH, high enzyme environment, and rapid drug release hinder the use of redox-mediated drug delivery systems.
Under inflammatory processes, ligand-dependent NPs have shown to be a precise method of drug localization, minimal side effects, and increased therapeutic efficacy[49]. Overexpression of inflammatory markers on cell surface provides molecular targets for anchoring NPs[50,51]. PLA NPs, which carry CD98 Fab antibodies, and chitosan/polyethyleneimine NPs modified with CD98 antibody were developed to treat mice with colitis. During inflammation, cells in colonic mucosa overexpress CD98 glycoprotein, which becomes the target of action for the ligand-modified NPs[52,53]. Similarly, mannose receptors are overexpressed on macrophages, which are targeted by mannosylated poly (amido amine)-modified NPs[54]. Moreover, increased expression of CD44 is observed in the macrophages and inflamed epithelial cells during colitis[55-57]. Hyaluronic acid can attach to CD44; hence, HA-modified NPs have been developed and tested successfully in mice with UC[58]. Increased expression of folate receptors has also been observed during inflammation[59-61]. Naserifar et al[62] successfully demonstrated the efficacy of oral administration of folic acid conjugated PLGA NPs loaded with resveratrol in TNBS rats.
Besides overexpression of ligands/receptors on colonic epithelial cells, endothelial adhesion molecules (ECAMs) are also upregulated. Vascular adhesion molecules-1 are ECAMs against which polylactic acid (PLA)-PEG particles modified with monoclonal antibodies have been developed. The drug delivery system showed increased adhesion to the endothelium in dextran sodium sulfate (DSS)-induced colitis[63]. In conclusion, ligand-modified NPs promise to be a potential alternative for targeted drug delivery in cases of IBD. However, concerns regarding IV administration, antibody precipitated immune reaction, ligand instability inside GIT hamper its utilization.
In prodrug systems, a pharmacologically active agent is protected in a temporarily inactive form that gains bioactivity on exposure to enzymes overexpressed in inflammatory tissues. This allows drug delivery to specific sites[64]. Several 5-ASA formulations have been used as prodrugs for selective delivery to the colon, azo conjugates being the most common and effective[65]. Many 5-ASA derivates were also shown to be transportable substrates for Intestinal H+-coupled oligopeptide transporter 1, the expression of which is increased in chronic inflammatory conditions such as IBD[66]. Furthermore, a synthetic prodrug of 4-ASA, an isomer of 5-ASA, was prepared and shown to be suitable for the treatment of colon inflammatory diseases[67]. To reduce endoplasmic reticulum (ER) stress, an aggravating factor for IBD, Kim et al[68] synthesized prodrugs for 4-phenyl butyric acid, which effectively reduced the colonic damage and inflammation in a rat colitis model. Moreover, Shen et al[69] designed a novel pH/reactive oxygen species (ROS) dual-responsive prodrug micelle GC-B-Que. In his vivo experiments, he found that the GC-B-Que micelles tended to accumulate in sites of inflammation within the intestine and showed better therapeutic efficacy than free drugs[69]. Using a prodrug approach for management of IBD allows accumulation of drugs at sites of inflammation, avoiding unwanted side effects on healthy intestinal tissue.
Liposomes are lipid bilayer vesicles that consist of an aqueous core. They can carry both hydrophilic and lipophilic drugs[70]. For use as a drug delivery system for IBD, liposomes can be modified with lactoferrin which can specifically bind to low-density lipoprotein receptor-related protein that is expressed on the inflammatory macrophages. Zhao et al[71] designed a lactoferrin-modified liposome (LF-lipo) for delivering patchouli alcohol to colonic macrophages for anti-inflammatory activity. The results showed decreased disease activity index and body weight loss in DSS salt -induced colitis mice. In another study, an oxymatrine loaded, nitric oxide releasing liposome, tested in DSS-induced ulcerative colitis mice, was shown to significantly alleviate inflammation. Moreover, the results also revealed that liposomes could accumulate in the inflammatory colon efficiently and can be maintained for more than 36 h[72]. Furthermore, in an in vivo experiment, curcumin-loaded liposomes (CUR-LPs) were shown to allow sustained release of CUR in the simulated gastrointestinal tract, attenuate the clinical symptoms of ulcerative colitis and prevent DSS-induced colon tissue damage and colon shortening[73]. Liposomes have also been loaded with heparin for intrarectal delivery in the form of enema, which has demonstrated dose-dependent anti-inflammatory activity in vivo[74]. Krill oil incorporation into liposomes has demonstrated a high capacity to entrap the IBD drug budesonide, and results of an in vitro study indicate that using KO liposomes as an IBD drug carrier may yield increased drug accumulation in the inflamed intestinal region[75]. Keratinocyte growth factor (KGF), previously shown to be an effective drug for ulcerative colitis, faces several obstacles for clinical such as poor stability, short half-life, and easy degradation; therefore, liposomes were considered as a potential delivery system. KGF was encapsulated into liposomes (KGF-Lips), and neutrophil membrane vesicles were then inlaid in KGF-Lips to construct a neutrophile-like liposome (KGF-Neu) which was shown to significantly improve the chemical stability of KGF[76]. Anemia is frequently seen in patients with IBD, which is responsible for a significant loss of quality of life. Treatment with oral liposomal iron was shown to be effective in improving mild iron deficiency anemia and quality of life, as well as in decreasing fatigue in patients with inactive or mildly active IBD[77].
Although difficult to prepare, hybrid drug delivery systems hold great promise as they integrate the advantages of several different carriers into one. An external compartment is used to protect drugs from gastric degradation and denaturation at low pH, and the internal section of NPs is developed for loading drugs[78]. Kotla et al[79] fabricated hyaluronan (HA) functionalized polymeric nano-drug delivery system (Cur-HA NPs) using curcumin as a model fluorescent drug. HA functionalization was shown to increase cellular interaction and uptake following the use of Cur-HA NPs on colon epithelial-like (HT-29) monolayers cell cultures indicating that this system is effective for oral delivery of drugs to treat local colonic disease[79]. Furthermore, encapsulation of curcumin with bovine b-lactoglobulin has been shown to increase its aqueous solubility, and encapsulation with succinylated- b-lactoglobulin prevented its release when subjected to gastric fluids[80]. In another study, Zhang et al[81] developed a phospholipid vesicle co-hybridized with HA and ethanol (HA-ES) for transdermal delivery of eugenol (EUG)/cinnamaldehyde (CAH). Results of the subsequent study showed that HA-ES as carriers effectively improved the percutaneous absorption of EUG and CAH in a rat ulcerative colitis model[81]. To improve the colon targeted oral delivery of Berberine, Zhang et al[82] designed a micro-and nano- encapsulated hybrid delivery system, which resulted in significant alleviation of acute colitis in a DSS induced colitis mice model. To overcome limitations faced by nanoparticle systems, Naeem et al[83] developed a “nanoparticles-in-enteric microparticles” (NPsinMPs) system. A (PLGA) polymer-based NP system was encapsulated in pH-sensitive Eudragit FS30D microparticles. NPisMPs provided complete protection of NPs in both gastric acid and intestinal-like pH. Moreover, NPs, after releasing from NPsinMPs, accumulated in the inflamed colon of mice after oral administration and significantly alleviated murine experimental colitis when compared to the bare nanoparticle[83].
Apart from maintaining intestinal homeostasis and synthesizing bile acids[84], bacteria could also be used as a potential delivery system in IBD. Recently, scientists ventured to use bioengineered bacteria as a delivery system of therapeutics in various diseases[85]. Lactococcus Lactis (L. Lactis) was the front runner in the race of effective biological vectors. L. Lactis is present in abundance in plants in an inactive state, gaining the active status in the gut of ruminants[86].
In 2006, genetically engineered L. Lactis was graded safe as a drug delivery system[87]. Due to the immunomodulatory functions of L. Lactis, its use as a vector to transport cytokines has been reported in numerous studies. One of the first applications of genetically modified (GM) L. Lactis in mice to produce interleukin-10 (IL-10) for IBD treatment was reported in 2000[88]. In 2003, recombinant L. Lactis strain labeled Thy12 successfully produced IL-10 in pigs[89].
A phase I clinical trial aimed to treat CD patients with genetically engineered L. Lactis. The results backed up its safety profile and solidified the effectiveness of the containment strategy[90]. In another study, orally administered L. Lactis was seen to ameliorate inflammation of the colon in mice by delivering anti-murine TNF nanobodies to the inflamed site[91]. Similarly, a study of mice with induced colitis showed mitigation of the disease upon application of L. Lactis carrying the anti-TNF scFv expression vector[92]. In a 2020 study of a DSS induced colitis mouse model, inflammation was seen to be reduced by blocking the IL-1 signaling using genetically modified Lactic acid bacteria (gmLAB)[93].
These studies show the therapeutic nature of recombinant bacteria as a vector in the treatment of IBD. L. Lactis is demonstrated to be safe and potent in inducing anti-inflammatory effects. However, studies should investigate further into the risk of unintended transgene escape, and focus attempts must be made to develop various other anti-inflammatory substances by L. Lactis[89].
Apart from bacteria, eukaryotic cells can be utilized as a delivery system of therapeutics for IBD. GM immune and red blood cells (RBCs) are used as nanoparticle (NP) carriers via injection from these cells. Endothelial cells in blood vessels present as a barricade in the route of NPs to their target destination. In a study of a mouse model of acute inflammation, activated neutrophils took up intravenously administered NPs and crossed the barrier into the inflamed tissues[94]. This study highlighted the ability to “hijack” neutrophils to transport NPs to the diseased site.
Another study by Corbo et al[95] implemented the mechanism of α4β7 integrins, which promote entry of T cells to intestinal sites[96]. The study employed this principle to investigate the effect of specialized leukosomes (SLKs) in alleviating inflammation in mice models of IBD. SLKs were associated with modulating both pro and anti-inflammatory molecules. Immune cells are presumably blocked by SLKs, as indicated by the reduction in CD45+ cells in the colon of mice treated with SLKs. However, further studies must be initiated examining the mechanism of functioning of SLKs in-depth. Furthermore, a risk-benefit analysis must be conducted to assess its safety.
Similarly, RBCs are increasingly being popular based on their high bioavailability in blood. Due to the rapid clearance of NPs in blood, a study successfully reported a novel method of adhering them to circulating RBCs to enhance their vascular circulation[97]. A clinical trial by Castro et al[98] assessed the impact of autologous RBCs loaded with dexamethasone 21-Phosphate (Dex 21-P) on children with steroid-dependent Crohn’s disease. During treatment, the pediatric Crohn’s Disease Activity Index was sufficiently reduced, and 78% of the patients stopped taking steroids. Overall, 44% showed remission, indicating the safe and efficacious nature of RBCs laden with Dex 21-P. Another study corroborated these findings as steroids were withdrawn entirely in all the patients and were in clinical remission after three infusions[99]. Further large-scale controlled studies should examine the safety and potency of this therapy in IBD patients. In future studies focus should be on the delivery of drugs directly to the inflamed sites and on the concentration of dexamethasone to increase the bioavailability.
Various nanoparticle drug-delivery systems exist. Size-dependent is the most popular mechanism. During inflammation, increased permeability, and retention (EPR) and enhanced mucosal secretions lead to increased efficacy of size-dependent targeting. However, systemic absorption of nanoparticles could lead to adverse side effects, which require further investigation. In addition to size, the surface charge of nanoparticles can also be modified to increase effectiveness. Due to electrostatic attraction, positively charged nanoparticles can adhere to the negatively charged colonic mucosa.
Moreover, redox-dependent nanoparticles use the increased amounts of ROS produced by intestinal mucosal cells during inflammation. However, volatility in low pH and quick drug release limit their use. Lastly, ligand-dependent NPs precisely target colonic mucosa with increased efficacy. NPs attach to the inflammatory markers’ ligands/receptors and ECAMs on the cell surface. A ligand-dependent deliveries system is a promising alternative; however, the concern of antibody precipitated immune reaction and instability inside GIT warrants further investigations. The existing targeted novel drug delivery systems are illustrated in Figure 1.
In our review, we divulged several drug delivery systems that have been under investigation in previous studies. These carrier systems were found to be potential options for drug delivery, with enteric-coated microneedles and recombinant bacteria termed safe upon use, while further analysis needs to be undertaken to assess the reliability of eukaryotic cells as delivery systems. Due to increased epithelial permeability and retention, Nanoparticles are well-absorbed in inflamed mucosa and have shown to be efficacious. However, accumulation of NPs might be observed that requires further study. Redox-dependent nanoparticle drug delivery systems have also emerged, such as NPs composed of nitroxide radicals (RNPo), which have successfully reduced inflammation by exhausting ROS, but their utilization might be limited due to factors such as rapid drug release. Ligand-depending NPs are efficient localized treatment, and HA-coated NPs and folic acid conjugated PLGA NPs loaded with resveratrol in TNBS have shown positive results in animal models. 5-ASA formulations, its isomers comprise some of the prodrug systems well in practice, and a novel pH/ROS dual-responsive prodrug micelle GC-B-Que has been found to display high therapeutic efficacy. Liposomes modified with lactoferrin, CUR-LPs, and heparin loaded liposomes are lipid based vesicular systems that have been anti-inflammatory. Importantly, oral liposomal iron is advantageous in alleviating anemia often observed in IBD patients. Hybrid methods of drug delivery have been proven to be effective in alleviating colitis across various studies. Leukosomes as carrier systems have also been found to be effective in mediating inflammatory response in IBD, but more clinical trials involving human subjects are warranted. Conventional drug delivery systems have certain caveats associated with their use, and novel drug delivery systems are being resorted to avoiding the adverse outcomes reported with the former routes; however, more research is needed to substantiate the efficacy of the novel carrier systems. Overall, given the challenge of delivering a drug to the inflamed site in IBD and the unique presentations of the disease in various patients, the new strategies can be instrumental in improving disorder symptoms and disease course.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: American College of Chest Physicians.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Zhang Y, China; Zhou Y, China; S-Editor: Wu YXJ L-Editor: A P-Editor: Wu YXJ
| 1. | Matsuoka K, Kanai T. The gut microbiota and inflammatory bowel disease. Semin Immunopathol. 2015;37:47-55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 407] [Cited by in RCA: 573] [Article Influence: 52.1] [Reference Citation Analysis (1)] |
| 2. | Lautenschläger C, Schmidt C, Fischer D, Stallmach A. Drug delivery strategies in the therapy of inflammatory bowel disease. Adv Drug Deliv Rev. 2014;71:58-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 204] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 3. | Kesisoglou F, Zimmermann EM. Novel drug delivery strategies for the treatment of inflammatory bowel disease. Expert Opin Drug Deliv. 2005;2:451-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 4. | Talaei F, Atyabi F, Azhdarzadeh M, Dinarvand R, Saadatzadeh A. Overcoming therapeutic obstacles in inflammatory bowel diseases: a comprehensive review on novel drug delivery strategies. Eur J Pharm Sci. 2013;49:712-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 5. | Singh D, Srivastava S, Pradhan M, Kanwar JR, Singh MR. Inflammatory Bowel Disease: Pathogenesis, Causative Factors, Issues, Drug Treatment Strategies, and Delivery Approaches. Crit Rev Ther Drug Carrier Syst. 2015;32:181-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 6. | Steidler L. Microbiological and immunological strategies for treatment of inflammatory bowel disease. Microbes Infect. 2001;3:1157-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | OrdaÌs I, Eckmann L, Talamini M, Baumgart DC, Sandborn WJ. Ulcerative colitis. In: The Lancet. Lancet; 2012. page 1606-19. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1151] [Cited by in RCA: 1547] [Article Influence: 119.0] [Reference Citation Analysis (5)] |
| 8. | Yu YR, Rodriguez JR. Clinical presentation of Crohn's, ulcerative colitis, and indeterminate colitis: Symptoms, extraintestinal manifestations, and disease phenotypes. Semin Pediatr Surg. 2017;26:349-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 232] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 9. | Ramos GP, Papadakis KA. Mechanisms of Disease: Inflammatory Bowel Diseases. Mayo Clin Proc. 2019;94:155-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 646] [Article Influence: 107.7] [Reference Citation Analysis (2)] |
| 10. | Henry S, McAllister DV, Allen MG, Prausnitz MR. Microfabricated microneedles: a novel approach to transdermal drug delivery. J Pharm Sci. 1998;87:922-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 790] [Cited by in RCA: 670] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 11. | Gill HS, Prausnitz MR. Coating formulations for microneedles. Pharm Res. 2007;24:1369-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 201] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 12. | Confidential Rani-1. Available from: http://a.wethrive.com/ext/rani/docs/rani_pre-clinical_data_July_2014_1.pdf. |
| 13. | Traverso G, Schoellhammer CM, Schroeder A, Maa R, Lauwers GY, Polat BE, Anderson DG, Blankschtein D, Langer R. Microneedles for drug delivery via the gastrointestinal tract. J Pharm Sci. 2015;104:362-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 125] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 14. | Vllasaliu D, Thanou M, Stolnik S, Fowler R. Recent advances in oral delivery of biologics: nanomedicine and physical modes of delivery. Expert Opin Drug Deliv. 2018;15:759-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 15. | Powell JJ, Faria N, Thomas-McKay E, Pele LC. Origin and fate of dietary nanoparticles and microparticles in the gastrointestinal tract. J Autoimmun. 2010;34:J226-J233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 318] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 16. | Tolstanova G, Deng X, French SW, Lungo W, Paunovic B, Khomenko T, Ahluwalia A, Kaplan T, Dacosta-Iyer M, Tarnawski A, Szabo S, Sandor Z. Early endothelial damage and increased colonic vascular permeability in the development of experimental ulcerative colitis in rats and mice. Lab Invest. 2012;92:9-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Xiao B, Merlin D. Oral colon-specific therapeutic approaches toward treatment of inflammatory bowel disease. Expert Opin Drug Deliv. 2012;9:1393-1407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 18. | Collnot EM, Ali H, Lehr CM. Nano- and microparticulate drug carriers for targeting of the inflamed intestinal mucosa. J Control Release. 2012;161:235-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 180] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 19. | Erratum for the Report "Global distribution of earthworm diversity" by H. R. P. Phillips, C. A. Guerra, M. L. C. Bartz, M. J. I. Briones, G. Brown, T. W. Crowther, O. Ferlian, K. B. Gongalsky, J. van den Hoogen, J. Krebs, A. Orgiazzi, D. Routh, B. Schwarz, E. M. Bach, J. Bennett, U. Brose, T. Decaëns, B. König-Ries, M. Loreau, J. Mathieu, C. Mulder, W. H. van der Putten, K. S. Ramirez, M. C. Rillig, D. Russell, M. Rutgers, M. P. Thakur, F. T. de Vries, D. H. Wall, D. A. Wardle, M. Arai, F. O. Ayuke, G. H. Baker, R. Beauséjour, J. C. Bedano, K. Birkhofer, E. Blanchart, B. Blossey, T. Bolger, R. L. Bradley, M. A. Callaham, Y. Capowiez, M. E. Caulfield, A. Choi, F. V. Crotty, A. Dávalos, D. J. Diaz Cosin, A. Dominguez, A. E. Duhour, N. van Eekeren, C. Emmerling, L. B. Falco, R. Fernández, S. J. Fonte, C. Fragoso, A. L. C. Franco, M. Fugère, A. T. Fusilero, S. Gholami, M. J. Gundale, M. Gutiérrez López, D. K. Hackenberger, L. M. Hernández, T. Hishi, A. R. Holdsworth, M. Holmstrup, K. N. Hopfensperger, E. Huerta Lwanga, V. Huhta, T. T. Hurisso, B. V. Iannone III, M. Iordache, M. Joschko, N. Kaneko, R. Kanianska, A. M. Keith, C. A. Kelly, M. L. Kernecker, J. Klaminder, A. W. Koné, Y. Kooch, S. T. Kukkonen, H. Lalthanzara, D. R. Lammel, I. M. Lebedev, Y. Li, J. B. Jesus Lidon, N. K. Lincoln, S. R. Loss, R. Marichal, R. Matula, J. H. Moos, G. Moreno, A. Morón-Ríos, B. Muys, J. Neirynck, L. Norgrove, M. Novo, V. Nuutinen, V. Nuzzo, M. Rahman P, J. Pansu, S. Paudel, G. Pérès, L. Pérez-Camacho, R. Piñeiro, J.-F. Ponge, M. I. Rashid, S. Rebollo, J. Rodeiro-Iglesias, M. Á. Rodríguez, A. M. Roth, G. X. Rousseau, A. Rozen, E. Sayad, L. van Schaik, B. C. Scharenbroch, M. Schirrmann, O. Schmidt, B. Schröder, J. Seeber, M. P. Shashkov, J. Singh, S. M. Smith, M. Steinwandter, J. A. Talavera, D. Trigo, J. Tsukamoto, A. W. de Valença, S. J. Vanek, I. Virto, A. A. Wackett, M. W. Warren, N. H. Wehr, J. K. Whalen, M. B. Wironen, V. Wolters, I. V. Zenkova, W. Zhang, E. K. Cameron, N. Eisenhauer. Science. 2020;369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Youshia J, Lamprecht A. Size-dependent nanoparticulate drug delivery in inflammatory bowel diseases. Expert Opin Drug Deliv. 2016;13:281-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 62] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 21. | Lai SK, Wang YY, Hanes J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv Drug Deliv Rev. 2009;61:158-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1469] [Cited by in RCA: 1277] [Article Influence: 79.8] [Reference Citation Analysis (0)] |
| 22. | García-Díaz M, Birch D, Wan F, Nielsen HM. The role of mucus as an invisible cloak to transepithelial drug delivery by nanoparticles. Adv Drug Deliv Rev. 2018;124:107-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 23. | Murgia X, Loretz B, Hartwig O, Hittinger M, Lehr CM. The role of mucus on drug transport and its potential to affect therapeutic outcomes. Adv Drug Deliv Rev. 2018;124:82-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 220] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 24. | Lieleg O, Ribbeck K. Biological hydrogels as selective diffusion barriers. Trends Cell Biol. 2011;21:543-551. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 333] [Cited by in RCA: 271] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 25. | Viscido A, Capannolo A, Latella G, Caprilli R, Frieri G. Nanotechnology in the treatment of inflammatory bowel diseases. J Crohns Colitis. 2014;8:903-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 26. | Meissner Y, Pellequer Y, Lamprecht A. Nanoparticles in inflammatory bowel disease: particle targeting vs pH-sensitive delivery. Int J Pharm 2006; 316: 138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 85] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 27. | Lamprecht A, Schäfer U, Lehr CM. Size-dependent bioadhesion of micro- and nanoparticulate carriers to the inflamed colonic mucosa. Pharm Res. 2001;18:788-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 359] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 28. | Ali H, Weigmann B, Neurath MF, Collnot EM, Windbergs M, Lehr CM. Budesonide loaded nanoparticles with pH-sensitive coating for improved mucosal targeting in mouse models of inflammatory bowel diseases. J Control Release. 2014;183:167-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 110] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 29. | Vong LB, Mo J, Abrahamsson B, Nagasaki Y. Specific accumulation of orally administered redox nanotherapeutics in the inflamed colon reducing inflammation with dose-response efficacy. J Control Release. 2015;210:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 30. | Vong LB, Tomita T, Yoshitomi T, Matsui H, Nagasaki Y. An orally administered redox nanoparticle that accumulates in the colonic mucosa and reduces colitis in mice. Gastroenterology. 2012;143:1027-36.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 138] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 31. | Lamprecht A. IBD: selective nanoparticle adhesion can enhance colitis therapy. Nat Rev Gastroenterol Hepatol. 2010;7:311-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 32. | Schmidt C, Lautenschlaeger C, Collnot EM, Schumann M, Bojarski C, Schulzke JD, Lehr CM, Stallmach A. Nano- and microscaled particles for drug targeting to inflamed intestinal mucosa: a first in vivo study in human patients. J Control Release. 2013;165:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 173] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 33. | Niebel W, Walkenbach K, Béduneau A, Pellequer Y, Lamprecht A. Nanoparticle-based clodronate delivery mitigates murine experimental colitis. J Control Release. 2012;160:659-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 34. | Lautenschläger C, Schmidt C, Lehr CM, Fischer D, Stallmach A. PEG-functionalized microparticles selectively target inflamed mucosa in inflammatory bowel disease. Eur J Pharm Biopharm. 2013;85:578-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 35. | Antoni L, Nuding S, Wehkamp J, Stange EF. Intestinal barrier in inflammatory bowel disease. World J Gastroenterol. 2014;20:1165-1179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 248] [Cited by in RCA: 302] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 36. | Larsson JM, Karlsson H, Sjövall H, Hansson GC. A complex, but uniform O-glycosylation of the human MUC2 mucin from colonic biopsies analyzed by nanoLC/MSn. Glycobiology. 2009;19:756-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 209] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 37. | Jubeh TT, Barenholz Y, Rubinstein A. Differential adhesion of normal and inflamed rat colonic mucosa by charged liposomes. Pharm Res. 2004;21:447-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 116] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 38. | Jubeh TT, Nadler-Milbauer M, Barenholz Y, Rubinstein A. Local treatment of experimental colitis in the rat by negatively charged liposomes of catalase, TMN and SOD. J Drug Target. 2006;14:155-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 39. | Beloqui A, Coco R, Alhouayek M, Solinís MÁ, Rodríguez-Gascón A, Muccioli GG, Préat V. Budesonide-loaded nanostructured lipid carriers reduce inflammation in murine DSS-induced colitis. Int J Pharm. 2013;454:775-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 40. | Zhu H, Li YR. Oxidative stress and redox signaling mechanisms of inflammatory bowel disease: updated experimental and clinical evidence. Exp Biol Med (Maywood). 2012;237:474-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 333] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 41. | Lih-Brody L, Powell SR, Collier KP, Reddy GM, Cerchia R, Kahn E, Weissman GS, Katz S, Floyd RA, McKinley MJ, Fisher SE, Mullin GE. Increased oxidative stress and decreased antioxidant defenses in mucosa of inflammatory bowel disease. Dig Dis Sci. 1996;41:2078-2086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 340] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 42. | Mahida YR, Wu KC, Jewell DP. Respiratory burst activity of intestinal macrophages in normal and inflammatory bowel disease. Gut. 1989;30:1362-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 101] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 43. | Bertoni S, Machness A, Tiboni M, Bártolo R, Santos HA. Reactive oxygen species responsive nanoplatforms as smart drug delivery systems for gastrointestinal tract targeting. Biopolymers. 2020;111:e23336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 44. | Wang X, Yan J, Wang L, Pan D, Xu Y, Wang F, Sheng J, Li X, Yang M. Oral delivery of anti-TNF antibody shielded by natural polyphenol-mediated supramolecular assembly for inflammatory bowel disease therapy. Theranostics. 2020;10:10808-10822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 45. | Zhang Q, Tao H, Lin Y, Hu Y, An H, Zhang D, Feng S, Hu H, Wang R, Li X, Zhang J. A superoxide dismutase/catalase mimetic nanomedicine for targeted therapy of inflammatory bowel disease. Biomaterials. 2016;105:206-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 159] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 46. | Zhang S, Langer R, Traverso G. Nanoparticulate Drug Delivery Systems Targeting Inflammation for Treatment of Inflammatory Bowel Disease. Nano Today. 2017;16:82-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 151] [Article Influence: 18.9] [Reference Citation Analysis (1)] |
| 47. | Sun Q, Luan L, Arif M, Li J, Dong QJ, Gao Y, Chi Z, Liu CG. Redox-sensitive nanoparticles based on 4-aminothiophenol-carboxymethyl inulin conjugate for budesonide delivery in inflammatory bowel diseases. Carbohydr Polym. 2018;189:352-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 48. | Li C, Zhao Y, Cheng J, Guo J, Zhang Q, Zhang X, Ren J, Wang F, Huang J, Hu H, Wang R, Zhang J. A Proresolving Peptide Nanotherapy for Site-Specific Treatment of Inflammatory Bowel Disease by Regulating Proinflammatory Microenvironment and Gut Microbiota. Adv Sci (Weinh). 2019;6:1900610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 137] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 49. | Zhang X, Wu W. Ligand-mediated active targeting for enhanced oral absorption. Drug Discov Today. 2014;19:898-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 50. | Cox D, Brennan M, Moran N. Integrins as therapeutic targets: lessons and opportunities. Nat Rev Drug Discov. 2010;9:804-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 363] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 51. | Kim E, Schueller O, Sweetnam PM. Targeting the leukocyte activation cascade: getting to the site of inflammation using microfabricated assays. Lab Chip. 2012;12:2255-2264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 52. | Xiao B, Yang Y, Viennois E, Zhang Y, Ayyadurai S, Baker M, Laroui H, Merlin D. Glycoprotein CD98 as a receptor for colitis-targeted delivery of nanoparticle. J Mater Chem B. 2014;2:1499-1508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 53. | Xiao B, Laroui H, Viennois E, Ayyadurai S, Charania MA, Zhang Y, Zhang Z, Baker MT, Zhang B, Gewirtz AT, Merlin D. Nanoparticles with surface antibody against CD98 and carrying CD98 small interfering RNA reduce colitis in mice. Gastroenterology. 2014;146:1289-300.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 153] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 54. | Xiao B, Laroui H, Ayyadurai S, Viennois E, Charania MA, Zhang Y, Merlin D. Mannosylated bioreducible nanoparticle-mediated macrophage-specific TNF-α RNA interference for IBD therapy. Biomaterials. 2013;34:7471-7482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 159] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 55. | Fromont Hankard G, Cezard JP, Aigrain Y, Navarro J, Peuchmaur M. CD44 variant expression in inflammatory colonic mucosa is not disease specific but associated with increased crypt cell proliferation. Histopathology. 1998;32:317-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 56. | Farkas S, Hornung M, Sattler C, Anthuber M, Gunthert U, Herfarth H, Schlitt HJ, Geissler EK, Wittig BM. Short-term treatment with anti-CD44v7 antibody, but not CD44v4, restores the gut mucosa in established chronic dextran sulphate sodium (DSS)-induced colitis in mice. Clin Exp Immunol. 2005;142:260-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 57. | Dreaden EC, Morton SW, Shopsowitz KE, Choi JH, Deng ZJ, Cho NJ, Hammond PT. Bimodal tumor-targeting from microenvironment responsive hyaluronan layer-by-layer (LbL) nanoparticles. ACS Nano. 2014;8:8374-8382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 144] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 58. | Xiao B, Xu Z, Viennois E, Zhang Y, Zhang Z, Zhang M, Han MK, Kang Y, Merlin D. Orally Targeted Delivery of Tripeptide KPV via Hyaluronic Acid-Functionalized Nanoparticles Efficiently Alleviates Ulcerative Colitis. Mol Ther. 2017;25:1628-1640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 144] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 59. | Chapkin RS, Kamen BA, Callaway ES, Davidson LA, George NI, Wang N, Lupton JR, Finnell RH. Use of a novel genetic mouse model to investigate the role of folate in colitis-associated colon cancer. J Nutr Biochem. 2009;20:649-655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 60. | Spencer SP, Belkaid Y. Dietary and commensal derived nutrients: shaping mucosal and systemic immunity. Curr Opin Immunol. 2012;24:379-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 61. | Zhang M, Xu C, Liu D, Han MK, Wang L, Merlin D. Oral Delivery of Nanoparticles Loaded With Ginger Active Compound, 6-Shogaol, Attenuates Ulcerative Colitis and Promotes Wound Healing in a Murine Model of Ulcerative Colitis. J Crohns Colitis. 2018;12:217-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 150] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 62. | Naserifar M, Hosseinzadeh H, Abnous K, Mohammadi M, Taghdisi SM, Ramezani M, Alibolandi M. Oral delivery of folate-targeted resveratrol-loaded nanoparticles for inflammatory bowel disease therapy in rats. Life Sci. 2020;262:118555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 63. | Sakhalkar HS, Hanes J, Fu J, Benavides U, Malgor R, Borruso CL, Kohn LD, Kurjiaka DT, Goetz DJ. Enhanced adhesion of ligand-conjugated biodegradable particles to colitic venules. FASEB J. 2005;19:792-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 64. | Manda JN, Markovic M, Zimmermann EM, Ben-Shabat S, Dahan A, Aponick A. Phospholipid Cyclosporine Prodrugs Targeted at Inflammatory Bowel Disease (IBD) Treatment: Design, Synthesis, and in Vitro Validation. ChemMedChem. 2020;15:1639-1644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 65. | Kotla NG, Rana S, Sivaraman G, Sunnapu O, Vemula PK, Pandit A, Rochev Y. Bioresponsive drug delivery systems in intestinal inflammation: State-of-the-art and future perspectives. Adv Drug Deliv Rev. 2019;146:248-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 150] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 66. | Yuri T, Kono Y, Okada T, Terada T, Miyauchi S, Fujita T. Transport Characteristics of 5-Aminosalicylic Acid Derivatives Conjugated with Amino Acids via Human H+-Coupled Oligopeptide Transporter PEPT1. Biol Pharm Bull. 2020;43:697-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 67. | Sadeghi F, Eidizade A, Saremnejad F, Hadizadeh F, Khodaverdi E, Akhgari A. Synthesis of a novel PEGylated colon-specific azo-based 4- aminosalicylic acid prodrug. Iran J Basic Med Sci. 2020;23:781-787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 68. | Kim S, Lee S, Lee H, Ju S, Park S, Kwon D, Yoo JW, Yoon IS, Min DS, Jung YS, Jung Y. A Colon-Targeted Prodrug, 4-Phenylbutyric Acid-Glutamic Acid Conjugate, Ameliorates 2,4-Dinitrobenzenesulfonic Acid-Induced Colitis in Rats. Pharmaceutics. 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 69. | Shen C, Zhao L, Du X, Tian J, Yuan Y, Jia M, He Y, Zeng R, Qiao R, Li C. Smart Responsive Quercetin-Conjugated Glycol Chitosan Prodrug Micelles for Treatment of Inflammatory Bowel Diseases. Mol Pharm. 2021;18:1419-1430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 49] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 70. | Zhang JX, Wang K, Mao ZF, Fan X, Jiang DL, Chen M, Cui L, Sun K, Dang SC. Application of liposomes in drug development--focus on gastroenterological targets. Int J Nanomedicine. 2013;8:1325-1334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 71. | Zhao Y, Yang Y, Zhang J, Wang R, Cheng B, Kalambhe D, Wang Y, Gu Z, Chen D, Wang B, Huang Y. Lactoferrin-mediated macrophage targeting delivery and patchouli alcohol-based therapeutic strategy for inflammatory bowel diseases. Acta Pharm Sin B. 2020;10:1966-1976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 72. | Tang Q, Zhang W, Zhang C, Guan Y, Ding J, Yuan C, Tan C, Gao X, Tan S. Oxymatrine loaded nitric oxide-releasing liposomes for the treatment of ulcerative colitis. Int J Pharm. 2020;586:119617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 73. | Wang C, Han Z, Wu Y, Lu X, Tang X, Xiao J, Li N. Enhancing stability and anti-inflammatory properties of curcumin in ulcerative colitis therapy using liposomes mediated colon-specific drug delivery system. Food Chem Toxicol. 2021;151:112123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 74. | Ahmad A, Vaghasiya K, Kumar A, Alam P, Raza SS, Verma RK, Khan R. Enema based therapy using liposomal formulation of low molecular weight heparin for treatment of active ulcerative colitis: New adjunct therapeutic opportunity. Mater Sci Eng C Mater Biol Appl. 2021;121:111851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 75. | Kim JH, Hong SS, Lee M, Lee EH, Rhee I, Chang SY, Lim SJ. Krill Oil-Incorporated Liposomes As An Effective Nanovehicle To Ameliorate The Inflammatory Responses Of DSS-Induced Colitis. Int J Nanomedicine. 2019;14:8305-8320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 76. | Zhao YZ, ZhuGe DL, Tong MQ, Lin MT, Zheng YW, Jiang X, Yang WG, Yao Q, Xiang Q, Li XK, Xu HL. Ulcerative colitis-specific delivery of keratinocyte growth factor by neutrophils-simulated liposomes facilitates the morphologic and functional recovery of the damaged colon through alleviating the inflammation. J Control Release. 2019;299:90-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 77. | de Alvarenga Antunes CV, de Alvarenga Nascimento CR, Campanha da Rocha Ribeiro T, de Alvarenga Antunes P, de Andrade Chebli L, Martins Gonçalves Fava L, Malaguti C, Maria Fonseca Chebli J. Treatment of iron deficiency anemia with liposomal iron in inflammatory bowel disease: efficacy and impact on quality of life. Int J Clin Pharm. 2020;42:895-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 78. | Li X, Lu C, Yang Y, Yu C, Rao Y. Site-specific targeted drug delivery systems for the treatment of inflammatory bowel disease. Biomed Pharmacother. 2020;129:110486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 79. | Kotla NG, Burke O, Pandit A, Rochev Y. An Orally Administrated Hyaluronan Functionalized Polymeric Hybrid Nanoparticle System for Colon-Specific Drug Delivery. Nanomaterials (Basel). 2019;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 80. | Pujara N, Giri R, Wong KY, Qu Z, Rewatkar P, Moniruzzaman M, Begun J, Ross BP, McGuckin M, Popat A. pH - Responsive colloidal carriers assembled from β-lactoglobulin and Epsilon poly-L-lysine for oral drug delivery. J Colloid Interface Sci. 2021;589:45-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 81. | Zhang Y, Zhang H, Zhang K, Li Z, Guo T, Wu T, Hou X, Feng N. Co-hybridized composite nanovesicles for enhanced transdermal eugenol and cinnamaldehyde delivery and their potential efficacy in ulcerative colitis. Nanomedicine. 2020;28:102212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 82. | Zhang L, Li M, Zhang G, Gao C, Wang S, Zhang T, Ma C, Wang L, Zhu Q. Micro- and Nanoencapsulated Hybrid Delivery System (MNEHDS): A Novel Approach for Colon-Targeted Oral Delivery of Berberine. Mol Pharm. 2021;18:1573-1581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 83. | Naeem M, Lee J, Oshi MA, Cao J, Hlaing SP, Im E, Jung Y, Yoo JW. Colitis-targeted hybrid nanoparticles-in-microparticles system for the treatment of ulcerative colitis. Acta Biomater. 2020;116:368-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 84. | Weingarden AR, Vaughn BP. Intestinal microbiota, fecal microbiota transplantation, and inflammatory bowel disease. Gut Microbes. 2017;8:238-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 336] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 85. | Wegmann U, Carvalho AL, Stocks M, Carding SR. Use of genetically modified bacteria for drug delivery in humans: Revisiting the safety aspect. Sci Rep. 2017;7:2294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 86. | Bolotin A, Wincker P, Mauger S, Jaillon O, Malarme K, Weissenbach J, Ehrlich SD, Sorokin A. The complete genome sequence of the lactic acid bacterium Lactococcus lactis ssp. lactis IL1403. Genome Res. 2001;11:731-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 768] [Cited by in RCA: 754] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 87. | Steidler L, Rottiers P. Therapeutic drug delivery by genetically modified Lactococcus lactis. Ann N Y Acad Sci. 2006;1072:176-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 88. | Steidler L, Hans W, Schotte L, Neirynck S, Obermeier F, Falk W, Fiers W, Remaut E. Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. Science. 2000;289:1352-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1049] [Cited by in RCA: 1013] [Article Influence: 40.5] [Reference Citation Analysis (0)] |
| 89. | Steidler L, Neirynck S, Huyghebaert N, Snoeck V, Vermeire A, Goddeeris B, Cox E, Remon JP, Remaut E. Biological containment of genetically modified Lactococcus lactis for intestinal delivery of human interleukin 10. Nat Biotechnol. 2003;21:785-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 376] [Cited by in RCA: 364] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 90. | Braat H, Rottiers P, Hommes DW, Huyghebaert N, Remaut E, Remon JP, van Deventer SJ, Neirynck S, Peppelenbosch MP, Steidler L. A phase I trial with transgenic bacteria expressing interleukin-10 in Crohn's disease. Clin Gastroenterol Hepatol. 2006;4:754-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 525] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 91. | Vandenbroucke K, de Haard H, Beirnaert E, Dreier T, Lauwereys M, Huyck L, Van Huysse J, Demetter P, Steidler L, Remaut E, Cuvelier C, Rottiers P. Orally administered L. lactis secreting an anti-TNF Nanobody demonstrate efficacy in chronic colitis. Mucosal Immunol. 2010;3:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 202] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 92. | Chiabai MJ, Almeida JF, de Azevedo MGD, Fernandes SS, Pereira VB, de Castro RJA, Jerônimo MS, Sousa IG, de Souza Vianna LM, Miyoshi A, Bocca AL, Maranhão AQ, Brigido MM. Mucosal delivery of Lactococcus lactis carrying an anti-TNF scFv expression vector ameliorates experimental colitis in mice. BMC Biotechnol. 2019;19:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 93. | Namai F, Shigemori S, Ogita T, Sato T, Shimosato T. Microbial therapeutics for acute colitis based on genetically modified Lactococcus lactis hypersecreting IL-1Ra in mice. Exp Mol Med. 2020;52:1627-1636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 94. | Chu D, Gao J, Wang Z. Neutrophil-Mediated Delivery of Therapeutic Nanoparticles across Blood Vessel Barrier for Treatment of Inflammation and Infection. ACS Nano. 2015;9:11800-11811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 207] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 95. | Corbo C, Cromer WE, Molinaro R, Toledano Furman NE, Hartman KA, De Rosa E, Boada C, Wang X, Zawieja DC, Agostini M, Salvatore F, Abraham BP, Tasciotti E. Engineered biomimetic nanovesicles show intrinsic anti-inflammatory properties for the treatment of inflammatory bowel diseases. Nanoscale. 2017;9:14581-14591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 96. | DeNucci CC, Pagán AJ, Mitchell JS, Shimizu Y. Control of alpha4beta7 integrin expression and CD4 T cell homing by the beta1 integrin subunit. J Immunol. 2010;184:2458-2467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 65] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 97. | Chambers E, Mitragotri S. Prolonged circulation of large polymeric nanoparticles by non-covalent adsorption on erythrocytes. J Control Release. 2004;100:111-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 167] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 98. | Castro M, Rossi L, Papadatou B, Bracci F, Knafelz D, Ambrosini MI, Calce A, Serafini S, Isacchi G, D'Orio F, Mambrini G, Magnani M. Long-term treatment with autologous red blood cells loaded with dexamethasone 21-phosphate in pediatric patients affected by steroid-dependent Crohn disease. J Pediatr Gastroenterol Nutr. 2007;44:423-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 99. | Annese V, Latiano A, Rossi L, Lombardi G, Dallapiccola B, Serafini S, Damonte G, Andriulli A, Magnani M. Erythrocytes-mediated delivery of dexamethasone in steroid-dependent IBD patients-a pilot uncontrolled study. Am J Gastroenterol. 2005;100:1370-1375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 3.0] [Reference Citation Analysis (0)] |