Published online May 7, 2022. doi: 10.3748/wjg.v28.i17.1830
Peer-review started: November 3, 2021
First decision: December 26, 2021
Revised: January 3, 2022
Accepted: March 25, 2022
Article in press: March 25, 2022
Published online: May 7, 2022
Processing time: 177 Days and 1.2 Hours
There remains a dearth of Asian epidemiological literature for paediatric inflammatory bowel disease (PIBD).
To describe the presenting features of PIBD from 7 Asia-Pacific pediatric gastroenterology centers via a central standardised electronic data platform.
Clinical, endoscopic and radiologic data at diagnosis from the registry were extracted between 1st January 1995 to 31st December 2019. Disease phenotypic characteristics were classified as per the Paris classification system.
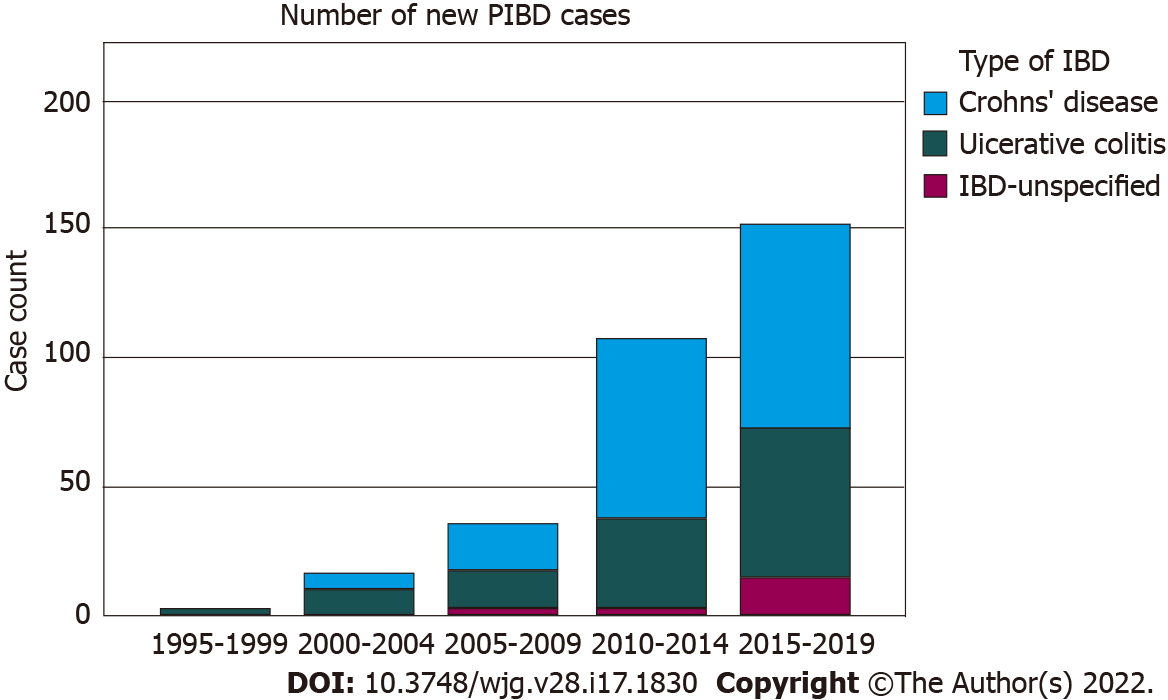
There was a distinct rise in new PIBD cases: Nearly half (48.6%) of the cohort was diagnosed in the most recent 5 years (2015-2019). The ratio of Crohn’s disease (CD):Ulcerative colitis (UC):IBD-Unclassified was 55.9%:38.3%:5.8%. The mean age was 9.07 years with a high proportion of very early onset IBD (VEO-IBD) (29.3%) and EO-IBD (52.7%). An over-representation of the Indian/South Asian ethnic group was observed which accounted for 37.0% of the overall Singapore/Malaysia subcohort (6.8%-9.0% Indians in census). Indian/South Asian CD patients were also most likely to present with symptomatic perianal disease (P = 0.003). CD patients presented with significantly more constitutional symptoms (fever, anorexia, malaise/fatigue and muscle-wasting) than UC and higher inflammatory indices (higher C-reactive protein and lower albumin levels).
We observed a high incidence of VEO-IBD and an over-representation of the Indian ethnicity. South Asian CD patients were more likely to have symptomatic perianal disease.
Core Tip: We describe the presenting features of paediatric inflammatory bowel disease (IBD) in 7 paediatric gastroenterology centers across six Asia-Pacific regions via a centrally-hosted electronic data capture platform. Clinical, endoscopic and radiologic data of 311 paediatric patients diagnosed with IBD between 1995 and 2019 were extracted. The ratio of Crohn’s (CD):Ulcerative colitis:IBD-Unclassified was 55.9%:38.3%:5.8%. The mean age was 9.07 years with a high proportion of very early onset IBD (VEO-IBD) and EO-IBD. An over-representation of the Indian/South Asian ethnic group was observed in the multiethnic subpopulations of Singapore and Malaysia. Patients of Indian/South Asian ethnicity were also most likely to present with symptomatic perianal CD.
- Citation: Huang JG, Wong YKY, Chew KS, Tanpowpong P, Calixto Mercado KS, Reodica A, Rajindrajith S, Chang KC, Ni YH, Treepongkaruna S, Lee WS, Aw MM. Epidemiological characteristics of Asian children with inflammatory bowel disease at diagnosis: Insights from an Asian-Pacific multi-centre registry network. World J Gastroenterol 2022; 28(17): 1830-1844
- URL: https://www.wjgnet.com/1007-9327/full/v28/i17/1830.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i17.1830
The epidemiology of inflammatory bowel disease (IBD) is a dynamic and rapidly evolving landscape. Recent publications over the past decade document the rise in IBD incidence in traditionally low-prevalence regions (Asia, Africa, South America) while the incidence is somewhat stabilising in high-prevalence western nations[1]. In particular, there is an increased impetus to focus on paediatric IBD (PIBD) epidemiology as the rising incidence of childhood-onset disease appears to be driving this overall trend in certain regions[2]. Very-early onset IBD (VEO-IBD), generally defined as disease onset prior to the age of 6 years, was once considered rare but now represents the fastest growing age bracket of IBD incidence in certain countries[3]. Yet in spite of these global trends, there remains a dearth of epidemiological literature for PIBD in Asia, in particular the regions of Central, South and Southeast Asia. This begets the question whether these countries are truly ‘low prevalence’ in the absence of national IBD registries such as those established in Japan[4] and South Korea[5]. Apart from studies published out of these East Asian cohorts, current literature is mostly limited to single centre publications[6-8] from countries in the Asia-Pacific region. Epidemiological data from these publications would only represent ‘the tip of the iceberg’ of the true burden of IBD and may not necessarily encapsulate recent regional epidemiological trends in disease incidence and behaviour.
There are numerous challenges in establishing a robust disease registry within Asia, especially with chronic diseases of emerging importance such as PIBD. For one, there is significant heterogeneity in disease awareness, diagnostic evaluation processes and therapeutic strategies across the vast Asian continent. This inevitably results in variations in case definitions with consequent epidemiological data inconsistencies. The aim of the present study was to describe the presenting features of PIBD managed in 7 paediatric gastroenterology centers across six Asia-Pacific countries and regions via a centrally-hosted electronic data capture platform using international standardised definitions.
The concept and goal of establishing a standardised disease data capture platform across Asian paediatric gastroenterology centres were first discussed amongst paediatric gastroenterologists practising across 7 tertiary centres in 6 Asian countries (Malaysia, Philippines, Singapore, Sri Lanka, Taiwan R.O.C., Thailand) in 2017. A consensus was established amongst the network committee members with regards to the standardisation of disease data collected both retrospectively and prospectively. Anonymised clinical data was to be collected on patient demographics, diagnostic evaluation and disease characteristics based on clinical examination, laboratory, endoscopy and radiology investigations. The diagnosis and classification of IBD into one of three disease subtypes [Crohn’s disease (CD), Ulcerative colitis (UC) and IBD-unclassified (IBD-U)] was determined by each participating centre based on the revised Porto criteria[9] as agreed within the network. The Paris Classification[10] (paediatric modification of the Montreal classification for IBD) system was then utilised to describe disease behaviour and phenotype. Electronic case report forms were thereafter loaded on a secure REDCAP data-capture platform centrally hosted by the Singapore Clinical Research Institute.
Ethics approval for the storage of anonymised clinical data on a multi-centre central data registry hosted by the Singapore Clinical Research Institute was granted by the National Healthcare Group (NHG) Domain Specific Review Board (Approval letter for study code NUH/2019-00060 dated 23rd January 2020), followed by a separate ethics approval for data extraction and analysis (Approval letter for study code 2019/00751 dated 20th October 2021 to 19th October 2022).
For the purposes of analysis of clinical features at diagnosis, data from the registry was extracted for patients diagnosed with IBD between 1st January 1995 to 31st December 2019. The patient’s inclusion criteria were: (1) Gastroenterologist-confirmed diagnosis of IBD at the participating site; (2) Age below 18 years at the point of diagnosis; and (3) Resident (citizens, permanent residents or long-term visitors) of the country of the participating site. Medical tourists (non-resident patients visiting the country solely for the purpose for medical assessment/treatment) were excluded. We stratified the cohort (1995-2019) into five equal time-intervals, each of 5-year duration, for the intent of analysing change in IBD incidence and evolution of diagnostic practices with time.
VEO-IBD was defined by an age of onset less than 6 years and EO-IBD, by an age of onset less than 10 years. Symptomatic perianal disease was defined as perianal pain or discharge due to a fistula or abscess/collection. A ‘perianal manifestation’ of CD, symptomatic or otherwise, was defined broadly as large inflamed perianal tags, poorly healing perianal fissures ascribed to CD or perianal fistula/abscess or collection evident clinically or via imaging. South Asians were defined as people of Indian, Bangladeshi, Pakistani, Sri Lankan (Sinhalese/Tamil) or Maldivian origin. Anthropometric Z-scores for children of age 0-5 years were measured based on the World Health Organization (WHO) Child Growth Standards, while those for children aged 5-19 years were measured on the WHO Reference 2007. Linear growth failure was defined as a height Z-score of -2 and lower.
Disease severity was defined based on the patients’ respective Paediatric Crohn’s Disease Activity Index (PCDAI) (Inactive: ≤ 10, mild: 11-30, moderate-severe: > 30)[11] and Paediatric Ulcerative Colitis Activity Index (Inactive: < 10, mild: 10-34, moderate: 35-64, severe ≥ 65)[12] scores.
The date of IBD diagnosis was taken as the date of diagnostic endoscopy. For the purposes of this publication discussing solely the presenting clinical features at diagnosis, we only included clinical data occurring within 3-mo prior or after the date of diagnosis and before any therapeutic intervention.
Statistical analysis was done through SPSS Version 27 whereby we compared presenting symptoms, biochemical indices and growth indices between the key phenotypic subgroups (CD and UC); the overall effect of ethnicity on IBD phenotype and behaviour. Continuous variables (laboratory investigation values, anthropometric indices) were compared through the student’s t-test while most of the other outcomes (clinical findings, disease phenotype and behaviour) were classified as categorical variables/dichotomous outcomes and these were compared through the chi-square test. A P value of less than 0.05 was deemed as a significant difference in outcomes, otherwise stated as ‘NS’ (non-significant). Whereby multivariate analysis was required in determining the effect of South Asian ethnicity on the incidence of perianal disease in CD adjusting for inter-ethnic variability in disease phenotype, logistic regression was used to calculate an adjusted odds ratio.
Three hundred and eleven children were recruited from seven participating tertiary centres across six countries within the Asia-Pacific region. There was a distinct rise in the number of new PIBD cases recorded across the network, with nearly half (48.6%, 151/311) of the entire cohort being diagnosed in the most recent 5 years (2015-2019) of recruitment (Figure 1). Table 1 illustrates the overall distribution of CD vs UC vs IBD-U, in which the CD:UC ratio was approximately 1.5:1 while the proportion of IBD-U was 5.8%. While there were more males in the overall cohort (58.2% male), we did not observe any significant gender predilection across the three disease subtypes: Male CD 58.0% vs male UC 57.0% vs male IBD-U 66.7% (P = 0.75).
| IBD subtypes | P value (CD vs UC) | ||||
| ALL IBD, n = 311 | CD, n = 174 (55.9%) | UC, n = 119 (38.3%) | IBD-U, n = 18 (5.8%) | ||
| Country (n, row %) | |||||
| Malaysia | 95 | 39 (41.1) | 47 (49.5) | 9 (9.5) | |
| Philippines | 29 | 24 (82.8) | 1 (3.4) | 4 (13.8) | |
| Singapore | 108 | 59 (54.6) | 49 (45.4) | 0 (0.0) | - |
| Sri Lanka | 19 | 10 (52.6) | 7 (36.8) | 2 (10.5) | |
| Taiwan R.O.C. | 19 | 19 (100.0) | 0 (0.0) | - | |
| Thailand | 41 | 23 (56.1) | 15 (36.6) | 3 (7.3) | |
| Mean age at diagnosis, yr (SD) | 9.07 (4.56) | 10.18 (4.34) | 7.73 (4.39) | 7.34 (5.1) | < 0.001 |
| Age groups (n, column %) | |||||
| Very early onset < 6 yr | 91 (29.3) | 30 (17.2) | 51 (42.9) | 10 (55.6) | < 0.001 |
| Early onset < 10 yr | 164 (52.7) | 75 (43.1) | 78 (65.5) | 11 (61.1) | 0.001 |
| Sex (n, column %) | |||||
| Male | 181 (58.2) | 101 (58.0) | 68 (57.1) | 12 (66.7) | NS |
| Female | 130 (41.8) | 73 (42.0) | 51 (42.9) | 6 (33.3) | |
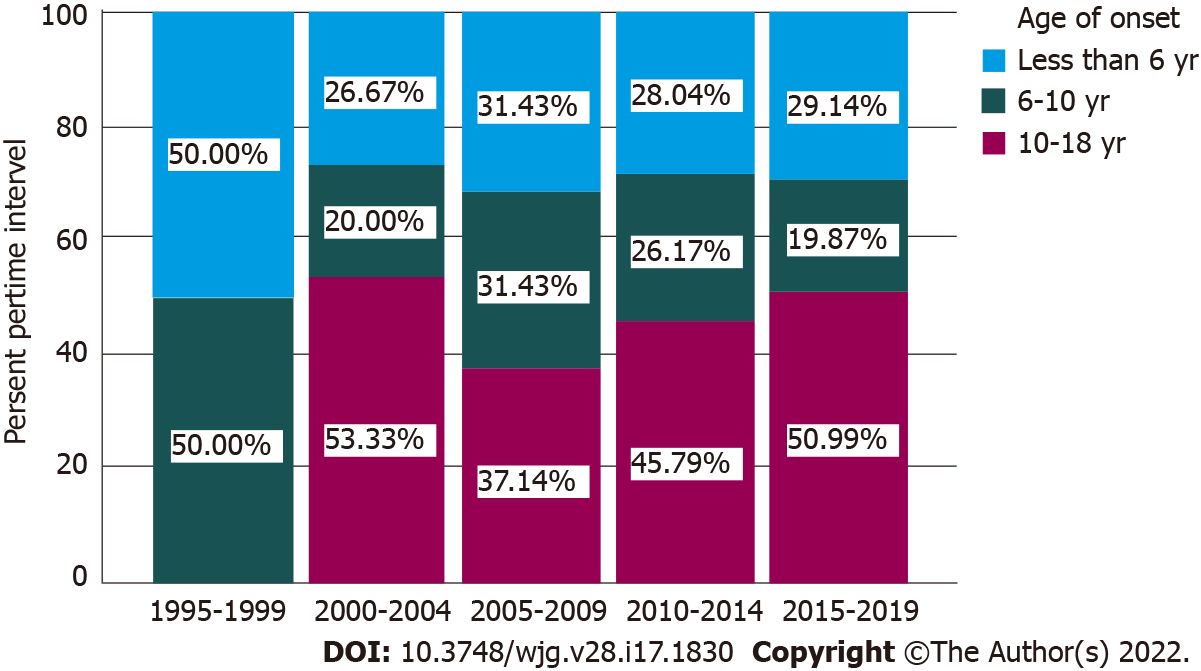
The mean age of disease presentation was 9.07 years for the entire IBD cohort which is within the age definition of EO disease. UC patients presented at a significantly younger age than CD patients (7.73 years vs 10.18 years, P < 0.001), with the mean age of IBD-U patients (7.34 years) similar to that of UC. When stratifying age of onset into VEO disease and EO disease, 29.3% of the entire cohort was classified as VEO-IBD and more than half (52.7%) was early-onset. There was a significantly higher proportion of VEO-IBD and EO-IBD presenting as a UC phenotype as compared to CD (Table 1). Across a period of 20 years, the proportions of VEO-IBD and EO-IBD remained relatively stable, approximately at 30% and 50% of the cohort per time interval respectively (Figure 2).
For a meaningful interpretation of ethnic predilection amongst Asian IBD patients, the multi-racial sub cohorts from Singapore and Malaysia were chosen as the other participating sites were relatively more homogeneous in ethnic group distribution. The three main ethnic groups in these two countries are Chinese, Indian/South Asian and Malay as listed in Table 2[13,14]. A distinct over-representation of the Indian/South Asian ethnic group was observed, with Indians/South Asians accounting for 37.0% of the overall Singapore/Malaysia IBD cohort relative to a minority representation of 6.8%-9.0% in both the countries’ respective population census. There was a significantly higher proportion of UC patients relative to CD amongst the Malay ethnic group (CD:UC ratio 0.7, P = 0.037).
| Total, n = 203 | Singapore population census 2020[1] | Malaysian population census 2020[2] | CD, n = 92 | UC, n = 91 | IBD-U, n = 9 | CD:UC ratio | P value | |
| Ethnic group (n, column %) | ||||||||
| Chinese | 70/203 (36.5) | 74.3 | 22.6 | 38 (41.3) | 31 (34.1) | 1 (11.1) | 1.2 | NS |
| Indian/South Asian | 71/203 (37.0) | 9.0 | 6.8 | 35 (38.0) | 33 (36.3) | 3 (33.3) | 1.0 | NS |
| Malay | 51/203 (26.6) | 13.5 | 69.6 | 19 (20.7) | 27 (29.7) | 5 (55.6) | 0.7 | 0.037 |
Abdominal pain (P = 0.004) and clinically evident abdominal tenderness (P = 0.003), fever (P < 0.001), loss of appetite (P = 0.001) and malaise/fatigue (P = 0.016) were significantly more common amongst CD patients relative to UC. While there was a non-significant higher proportion of CD patients presenting with weight loss (46.6% CD vs 37.0% UC), clinically evident muscle wasting was significantly more common in CD (14.4% CD vs 5.9% UC, P = 0.022).
Although CD patients had numerically lower mean weight Z-scores [(-1.4) vs (-1.0)] and height Z-scores [(-0.8) vs (-0.6)] than UC, only the mean body-mass index Z-score of CD patients was significantly lower [(-1.5) vs (-0.9), P = 0.013]. There were approximately similar rates of linear growth failure across the three disease subtypes: 18.4% CD vs 16.1% UC vs 17.6% IBD-U (Table 3). There was a non-significant higher proportion of patients with joint pains (7.5% CD vs 5% UC) and clinically evident arthritis (6.9% CD vs 2.5% UC) in CD patients. Other extraintestinal manifestations (EIMs) such as oral ulcers (20.7% CD vs 0.8% UC, P < 0.001) and erythema nodosum (4.6% CD vs 0.0% UC, P = 0.018) were more common in CD than UC. Hepatic involvement (hepatomegaly and elevated transaminases) was however more commonly observed in UC than CD, consistent with a significantly higher association of autoimmune liver disease/primary sclerosing cholangitis with UC than CD (17.6% UC vs 1.1% CD, P < 0.001).
| IBD subtypes | P value (CD vs UC) | |||
| CD, n = 174 (55.9%) | UC, n = 119 (38.3%) | IBD-U, n = 18 (5.8%) | ||
| Symptoms at diagnosis (n, column %) | ||||
| Abdominal pain | 107 (61.5) | 53 (44.5) | 7 (38.9) | 0.004 |
| Chronic diarrhoea | 104 (59.8) | 69 (58.0) | 10 (55.6) | 0.760 |
| Bloody diarrhoea | 60 (34.5) | 89 (74.8) | 11 (61.1) | < 0.001 |
| Weight loss | 81 (46.6) | 44 (37.0) | 9 (50.0) | 0.104 |
| Fever | 66 (37.9) | 21 (17.6) | 2 (11.1) | < 0.001 |
| Loss of appetite | 57 (32.8) | 19 (16.0) | 6 (33.3) | 0.001 |
| Malaise/fatigue | 28 (16.1) | 8 (6.7) | 1 (5.6) | 0.016 |
| Oral ulcers | 36 (20.7) | 1 (0.8) | 0 (0.0) | < 0.001 |
| Perianal symptoms (pain/discharge) | 23 (13.2) | 3 (2.5) | 1 (5.6) | 0.002 |
| Joint pains | 13 (7.5) | 6 (5.0) | 0 (0.0) | 0.407 |
| Physical signs at diagnosis (n, column %) | ||||
| Pallor | 68 (39.1) | 32 (26.9) | 5 (27.8) | 0.031 |
| Hepatomegaly | 8 (4.6) | 12 (10.1) | 2 (11.1) | 0.067 |
| Abdominal tenderness | 47 (27.0) | 15 (12.6) | 2 (11.1) | 0.003 |
| Asymptomatic perianal tags | 25 (14.4) | 3 (2.5) | 1 (5.6) | 0.001 |
| Perianal fissure | 20 (11.5) | 2 (1.7) | 1 (5.6) | 0.002 |
| Muscle wasting | 25 (14.4) | 7 (5.9) | 1 (5.6) | 0.022 |
| Arthritis | 12 (6.9) | 3 (2.5) | 0 (0.0) | 0.095 |
| Erythema nodosum | 8 (4.6) | 0 (0.0) | 0 (0.0) | 0.018 |
| Anthropometric parameters (SD) | ||||
| Mean weight Z-score | -1.4 (1.7) | -1.0 (1.7) | -0.7 (2.2) | 0.110 |
| Mean height Z-score | -0.8 (1.5) | -0.6 (1.6) | -0.6 (2.4) | 0.583 |
| Growth failure (Height Z-score less than -2) (%) | 18.4 | 16.1 | 17.6 | 0.907 |
| Mean BMI Z-score | -1.5 (1.8) | -0.9 (1.7) | -0.6 (1.8) | 0.013 |
| Laboratory investigations at diagnosis (SD) | ||||
| Mean haemoglobin, g/dL | 10.6 (1.9) | 11.0 (2.5) | 10.9 (1.5) | 0.221 |
| Mean ESR, mm/hr | 50.8 (30.5) | 42.6 (34.4) | 34.9 (32.2) | 0.063 |
| Mean C-reactive protein, mg/L | 54.6 (55.0) | 20.1 (24.3) | 7.1 (9.1) | < 0.001 |
| Mean albumin, g/dL | 33.0 (8.7) | 35.4 (8.4) | 37.2 (7.1) | 0.040 |
| Mean ALT, U/L | 18.5 (20.5) | 72.7 (162.0) | 20.4 (13.4) | < 0.001 |
| Mean AST, U/L | 27.0 (32.9) | 84.2 (170.7) | 30.5 (10.5) | 0.001 |
| Mean GGT, U/L | 38.3 (79.8) | 126.7 (160.8) | 30.8 (40.0) | < 0.001 |
| Mean faecal calprotectin, μg/g | 783 (1009.0) | 583.4 (353.1) | 593.5 (300.5) | 0.447 |
CD patients also presented with significantly higher C-reactive protein (CRP) levels (mean 54.6 CD vs 20.1 UC, P < 0.001) and lower albumin levels relative to UC. However, there was no significant difference in mean haemoglobin and proportion of severe anaemia (defined as ≤ 7.0g/dL: 4.0% CD vs 5.0% UC, P = 0.601) at presentation between CD and UC.
16.1% of the cohort had a faecal calprotectin (50/211) assayed at baseline. There was no significant difference between the mean values in CD and UC (Table 3). Amongst children with CD, faecal calprotectin values were lower in patients with isolated ileal disease (L1) compared to ileocolonic (L3) disease (468.7 in L1 vs 749.2 μg/g in L3, P = 0.05). No discernible difference in mean faecal calprotectin values was noted amongst children with UC; left sided disease (E1/E2: 547.0 μg/g) vs more extensive disease (E3/E4: 555.6 μg/g).
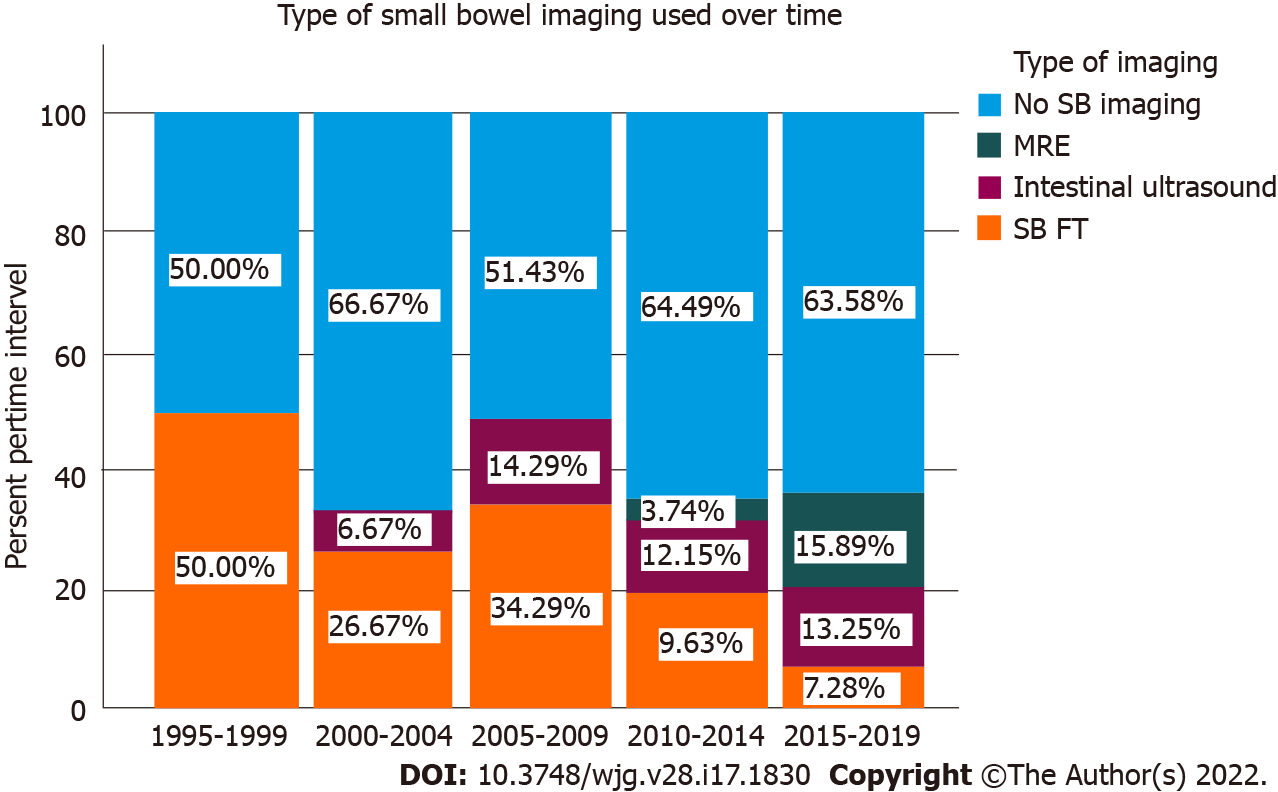
There was a progressive decline in the use of small bowel follow through studies across the cohort. Since 2010, there has been increasing use of either MR enterography or intestinal ultrasound although approximately 60% of the cohort have not had any form of small bowel imaging (Figure 3). Complete endoscopy as per Porto guidelines (upper and lower endoscopy) was performed in 79.1% of the cohort.
The ileocolonic location was the most common disease location in the entire cohort of CD patients, with pure inflammatory disease being most common (90.7%) followed by luminal penetrating disease (5.3%) and stricturing disease (3.3%). Isolated L4 disease (absence of terminal ileal or colonic disease) was relatively uncommon at 4.0% of the CD cohort. Over half of the CD patients had at least moderate disease severity at presentation (54.3%), with no differences in mean PCDAI scores noted with the South Asian subgroup. Upper gastrointestinal involvement was seen in 49.0% of the entire CD cohort. Symptomatic perianal disease was seen in 13.2% of the CD cohort and up to 30.5% of the CD cohort had some form of perianal manifestation as earlier defined.
South Asian CD patients were strongly associated with perianal symptoms at presentation (P = 0.003), the physical finding of asymptomatic perianal tags (15.5% South Asian vs 7.5%, P = 0.042), as well as any form of perianal manifestation (symptomatic and asymptomatic, detected on either clinical examination or perianal imaging; P = 0.003). Adjusting for inter-ethnic differences in CD incidence, the South Asian ethnicity remained strongly associated with symptomatic perianal disease at presentation (B co-efficient 1.264, P = 0.001). Sri Lanka was the only participating South Asian site in this study, and only 1 out of 19 Sri Lankan IBD patients had presented with symptomatic perianal disease. All the South Asian CD patients in this subgroup analysis (Table 4) were either first- or second-generation immigrants within Singapore or Malaysia.
| Disease characteristics | N | SA | P value (SA vs non-SA) |
| CD | n = 174 | n = 35 (20.1) | |
| Disease severity based on PCDAI score (n, column %)1 | |||
| Inactive (≤ 10) | 17 (12.3) | 4 (14.3) | NS |
| Mild (11-30) | 46 (33.3) | 9 (32.1) | |
| Moderate-severe (> 30) | 75 (54.3) | 15 (53.6) | |
| Disease location (n, column %) | |||
| L1 (distal 1/3 ileum +/- limited caecal disease) | 28 (18.7) | 4 (12.5) | 0.505 (SA) |
| L2 (colonic) | 54 (36.0) | 10 (31.3) | |
| L3 (ileocolonic) | 62 (41.3) | 16 (50.0) | |
| Isolated L4 disease | 6 (4.0) | 2 (6.3) | |
| L4a | 59 (40.7) | 13 (39.4) | 0.973 (SA) |
| L4b | 12 (8.3) | 3 (9.1) | |
| No L4 disease | 74 (51.0) | 17 (51.3) | |
| Disease behaviour (n, %) | |||
| B1 (inflammatory) | 136 (90.7) | 31 (93.9) | 0.858 (SA) |
| B2 (stricturing) | 5 (3.3) | 1 (3.0) | |
| B3 (penetrating) | 8 (5.3) | 1 (3.0) | |
| B2/B3 | 1 (0.7) | 0 (0.0) | |
| Perianal involvement (n, %) | |||
| Perianal symptoms at presentation | 23 (13.2) | 10 (28.6) | 0.003 (SA) |
| Any form of perianal manifestation (clinical exam or imaging) | 53 (30.5) | 18 (51.4) | 0.003 (SA) |
| UC | n = 119 | n = 33 (27.7) | |
| Disease severity based on PUCAI score (n, column %)2 | |||
| Inactive (< 10) | 7 (8.3) | 0 (0.0) | 0.353 (SA) |
| Mild (10-34) | 36 (42.9) | 11 (47.8) | |
| Moderate (35-64) | 28 (33.3) | 9 (39.1) | |
| Severe (≥ 65) | 13 (15.5) | 3 (13.0) | |
| Disease location (n, column %) | |||
| E1 (proctitis) | 8 (7.5) | 2 (6.9) | 0.353 (SA) |
| E2 (left sided) | 16 (15.1) | 5 (17.2) | |
| E3 (extensive) | 5 (4.7) | 5 (17.2) | |
| E4 (pancolitis) | 77 (72.6) | 17 (58.6) | |
| Disease severity (n, column %) | |||
| S0 (never severe) | 79 (78.2) | 20 (74.1) | 0.542 (SA) |
| S1 (ever severe) | 22 (21.8) | 7 (25.9) | |
Overall, pan colonic involvement was the most common location in UC (72.6%) and almost half (48.8%) of UC patients had at least moderate disease severity at presentation. South Asian UC patients had slightly less cases of isolated proctitis than the non-South Asian UC sub cohort (6.9% proctitis South Asian vs 7.8% non-South Asian).
This first publication from the Asian PIBD registry gives valuable insights into the distinct epidemiological patterns of a rapidly emerging chronic disease in the Asia-Pacific region. Existing literature from published adult cohorts documents the distinct rapid rise in overall IBD incidence across the Asia-Pacific region[15] ascribed to increased urbanisation and industrialisation of developing areas with a consequent change in lifestyle and environmental factors. Our pooled data across major paediatric gastroenterology centres in the Asia-Pacific region verifies the rise in PIBD incidence, particularly in the last decade of data collection (2010-2019). This is also consistent with rising trends observed in other Asian paediatric cohorts in Saudi Arabia[16], Bahrain[17], Japan[18], South Korea[19] and China[20]. There still remains a knowledge gap on the epidemiological trends of PIBD in many parts of Asia, particularly Central and South Asian regions, although recently published studies out of Kazakhstan[21], India[22] and Nepal[23] reaffirm IBD as an emerging health issue.
Several other factors have been proposed as reasons for the observed rise in IBD incidence within the Asia-Pacific region, namely increased disease awareness amongst healthcare professionals, improved access to healthcare resources and better diagnostic modalities in rapidly industrialising areas. While such factors may account for part of the initial observed rise in incidence in the earlier decades, they do not sufficiently explain the sustained year-on-year rise in PIBD incidence seen in developed regions of our network[6,24] as well as other highly industrialised regions in Japan and South Korea.
The key strength of a multi-centre registry in this ethnically and culturally diverse region would be to allow epidemiological comparisons amongst multi-racial Asian populations (e.g., Singapore/Malaysia) and ethnically homogeneous populations (e.g., Philippines/Sri Lanka/Thailand). In Singapore and Malaysia where the Chinese and Malay populations form the majority ethnic group respectively and the Indians are an ethnic minority, we affirm earlier findings from adult Southeast Asian cohorts[25,26] that there is an ethnic predisposition to IBD incidence with an over-representation of the Indian/South Asian subgroup (37% IBD vs 6%-9% general population). This is similar to observations in the United Kingdom and Canada where IBD is seen with greater frequency amongst both South Asians born in the United Kingdom[27] and South Asian migrants to the United Kingdom[28], as well as the paediatric South Asian community in British Columbia, Canada[29]. Similarly, in a study of 30812 IBD subjects diagnosed in the United States, Malhotra et al[30] found that residents of Indian ancestry carried the highest risk for all types of IBD, compared to residents of Jewish, East Asian or Hispanic ancestries.
With combined Singaporean and Malaysian data, we were able to discern unique characteristics of each ethnic group. The CD:UC ratio varied distinctly across the three ethnic groups (Chinese 1.2; Indian/South Asian 1.0 and Malay 0.7) and this is likely attributed to inter-ethnic differences in environmental factors, such as diet, diversity in gut microbiome[31] including Helicobacter pylori (H. pylori)[32] as well as socio-economic status[33]. This inter-ethnic variation in IBD incidence has been observed between African Americans and white Americans previously[34]; white Americans were reported to have a CD:UC ratio > 1.0 with Hispanic Americans having an inverse ratio[35], paralleling what is observed between Chinese and Malays in our cohort. A similar variegation of CD:UC ratio was seen in a Latin American systematic review by Kotze et al[36], in which only certain states in Brazil reported a CD:UC ratio > 1 whereas most of the other Latin American countries had a CD:UC ratio < 1. The authors concluded similarly that regions within Latin America that experienced higher degrees of economic development and ‘Westernisation’ had a higher proportion of CD cases.
We also observed a strongly significant association between the South Asian ethnicity and symptomatic perianal disease (pain or perianal discharge) at presentation, only in the South Asian populations of Singapore and Malaysia. Interestingly, despite less South Asians having the CD phenotype than Chinese, the South Asian ethnicity was significantly associated with asymptomatic perianal tags. The latter physical finding may be a prelude to developing symptomatic perianal Crohn’s later in the clinical course, as seen in this study by Singer et al[37] showing a higher rate of perianal fistulisation in patients initially presenting with non-penetrating perianal lesions. The observation of increased rates of symptomatic perianal disease was not appreciated in the native South Asian population of Sri Lanka within the limits of small patient numbers recruited in this sub cohort. A previous questionnaire survey conducted in India estimated the incidence of perianal disease at presentation in Indian paediatric CD patients at 18.0%, which is not strikingly higher than paediatric cohorts in Israel (13.3%)[38] and Canada (16.0%)[39]. More recent Indian studies, which included perianal disease developed both at diagnosis and subsequent follow-up, however, have found even lower rates than other Asian cohorts. Banerjee et al[40] reported 7.4% perianal involvement amongst CD patients and a multicentre paediatric study in India reported 10.9% of CD patients with perianal disease(fistula or abscess). These findings suggest South Asian CD patients in their native countries do not experience disproportionately high rates of perianal disease.
On the contrary, countries with a high South Asian immigrant population report findings congruent with our cohort’s. A previous paediatric study in San Francisco likewise demonstrated similar findings in our cohort, where South Asians tended to have more perianal fistulising disease at presentation than their Caucasian counterparts[41]. A recent adult cohort study published by Jangi et al[42] also showed South Asian CD patients in the United States were more likely to experience symptomatic perianal disease than white patients. Current findings from our study and the above-mentioned cohorts may suggest these phenotypic trends may be unique only to South Asians who have migrated from their native countries to relatively more developed and urbanised regions. A possible explanation is that South Asians may carry an intrinsic genetic predisposition for IBD, which results in a distinct disease phenotype in the setting of specific environmental exposures after migration[43]. A review by Foster and Jacobson[44] discusses these hypothetical gene-environmental interactions in South Asian immigrants, postulating the role of dietary changes from a traditionally high carbohydrate/low saturated fat diet to one which is highly processed. Other possible factors discussed include an increased exposure to urban pollutants and a reduced exposure to potentially protective factors such as vitamin D, H. pylori and helminths.
Overall, we do report a moderately high rate of perianal involvement in our CD patients at presentation (13.2% with symptomatic perianal disease and up to 30.5% with some form of perianal manifestation of CD either clinically detected or via imaging. An earlier Singaporean paediatric cohort reported a 21.6% incidence of perianal disease amongst CD patients at diagnosis[6] with mainland Chinese and South Korean paediatric CD cohorts reporting even higher rates of perianal involvement 42.4% Shanghai[45] , 47.1% [(perianal fistulising disease) only South Korean[46]]. Some variability in the reported rates of perianal involvement across CD cohorts could be accounted by differences in disease definitions as well as the extent of perianal imaging performed, which may detect indolent perianal fistulising disease in otherwise asymptomatic patients. A large IBD cohort of 5223 white Americans, 35 United States-born Asians and 81 Asian immigrants also reported Asians having a significantly higher rate of perianal disease (at presentation and follow-up) than whites (33% vs 18%)[47]. Further prospective follow-up of our cohort is required to ascertain if an even greater proportion of CD patients will evolve to develop perianal disease beyond the point of diagnosis.
Interestingly, we also found a very high proportion of VEO-IBD (29.3%) in our cohort, which appears to be stably high across a 20-year period of analysis. In comparison, data from the EPIMAD registry in France (n = 1412) reported a VEO-IBD rate of 3.0% based on a similar age definition for VEO-IBD[48]. Other Asian cohorts have similarly reported comparatively high rates of VEO-IBD, albeit using varying ages for definition. A single centre study in Beijing, Mainland China (n = 184) reported 41.8% of hospitalised children with IBD were VEO[49]; a cohort in Saudi Arabia reported 15.9% of patients were younger than 4 years at time of onset[16] and a recent Indian multicentre paediatric study had reported 19.1% of patients being VEO[22]. It is not certain if this epidemiologic trend is unique to regions where IBD is of emerging importance, as a Canadian study across 5 provinces reported VEO-IBD as the subgroup with the most rapid increase in incidence relative to the other age groups[2].
Aside from the South Asian predisposition and younger age of onset, the clinical presentation and extra-intestinal manifestations of CD and UC were similar to other Asian and Western paediatric and adult cohorts[50]. Abdominal pain and constitutional symptoms (fever, loss of appetite, oral ulcers, malaise) were more common presenting features in CD than UC; bloody diarrhoea and associated liver disease/primary sclerosing cholangitis were more common in UC at presentation, the latter being a well-established association. EIMs such as arthritis and erythema nodosum were more common in CD than UC, as also seen in a Mainland Chinese cohort[20]. We also reaffirm the finding of significant heterogeneity of CRP response between CD and UC, as discussed extensively in published literature[51,52]. Within the limits of a small sample size, we noted that faecal calprotectin values vary with disease location in CD but not in UC. Previous correlation studies have also similarly observed lower values of faecal calprotectin in CD patients with pure ileal disease[53].
While CD patients had numerically lower weight Z-scores and height Z-scores than UC patients, these comparisons were not statistically significant. This is due to a relatively high rate of linear growth failure (16.1%) reported in UC patients in our cohort, which is higher than the typical prevalence of 3%-10% previously described[54]. It is uncertain if this truly reflects a unique UC phenotype or whether some of these UC patients will be re-classified as CD with evolution of disease and/or further diagnostic modalities performed. As approximately 60% of our cohort did not undergo any form of small bowel imaging and 20% did not undergo an upper gastrointestinal endoscopy, we could expect changes in these initial diagnostic labels had more complete evaluation been performed.
There are a few limitations to our study; firstly, as most of the data was collected retrospectively, we were not able to compute patient disease activity indices in all patients because of incomplete clinical, biochemical, imaging or endoscopy records. Secondly, a lack of access and/or consistency in performing small bowel imaging and upper gastrointestinal endoscopy could have resulted in inaccurate disease classification. However, as almost half of our cohort was diagnosed in the recent 5 years, we are confident that the disease phenotype has been more accurately assessed and documented in these later years.
Another limitation was the predominance of Southeast Asian participating centres in our network, with the lack of East and West Asian representation. However, we were able to mitigate this as there already exists a substantial amount of published literature from established Japanese, South Korean and mainland Chinese cohorts as previously cited. Hence, we were able to compare certain epidemiological features such as age of onset and the proportion of perianal disease as discussed above.
Lastly, the number of patients captured within our network may only represent ‘the tip of an ice-berg’. We acknowledge that access to healthcare services may be very limited in rural regions of the participating countries, and there may be an inevitable selection bias in recruiting patients living in the proximity of major Asian city centres (Singapore, Kuala Lumpur, Bangkok, Manila, Colombo, Chinese Taipei) where our participating sites are located. Hence, there could be a number of children with IBD who do not present to centres equipped with adequate disease knowledge and diagnostic capability, and thus remain undiagnosed. At the same time, there could be a number of adolescent IBD patients who are managed by adult gastroenterologists. The general lack of national chronic disease registries for PIBD in the respective countries of this network further adds to the challenges in obtaining complete epidemiologic data. These factors discussed suggest the true burden of PIBD in the Asia-Pacific region is very likely under-estimated.
Our study presents epidemiological data from the largest multi-centre Asian-Pacific paediatric cohort to date, and reaffirms the rising trend of PIBD across our registry, particularly in the past decade (2010-2019). We also report a substantially higher incidence of VEO-IBD than European cohorts and this is similarly observed in other Asian cohorts in mainland China and the Middle East. The unique multi-ethnic demographic composition of our cohort allows for distinct phenotypic differences to be seen between ethnicities, chiefly the over-representation of the Indian/South Asian ethnicity and its strong association with symptomatic perianal CD. Prospective follow-up data from this registry would also ascertain if any of these observed epidemiologic trends within this publication have implications on medium to longer term disease outcomes.
This inaugural publication from our registry likely under-estimates the true burden of IBD in Asia, and the way forward to address these identified knowledge gaps, would be to encourage more tertiary centres from diverse parts of Asia to partake in the Asian PIBD registry. This will improve the representativeness of our cohort and enhance the ability to make both intra- and inter-regional comparisons in disease behaviour and phenotypic trends.
There remains a dearth of epidemiological literature on paediatric inflammatory bowel disease (IBD) in most parts of Asia. While there have been several published cohort studies out of East Asia, little is known on the actual disease burden beyond isolated case series and single-centre studies in the rest of the Asia-Pacific region. This would represent the first cohort study from a multi-centre Asian-Pacific PIBD registry first initiated in 2017.
The main issue was a lack of a standardised data platform in Asian-Pacific paediatric gastroenterology centres. A standardised data platform is vital to allow an accurate and validated way of reporting the disease behaviour and phenotype of PIBD across different healthcare systems in the heterogeneous Asia-Pacific region. This facilitates meaningful epidemiological comparisons with other established PIBD cohorts from North America and Western Europe.
This cohort study describes the epidemiological characteristics of all PIBD patients at the point of initial presentation, whom are managed at one of seven paediatric gastroenterology centres in the Asian-Pacific region (Singapore, Sri Lanka, Malaysia, Thailand, Philippines, Taiwan R.O.C). The objectives were to establish if there were unique disease characteristics of PIBD in the Asia-Pacific region in contrast to East Asian and Caucasian cohorts, as well as inter-ethnic disease phenotypic differences in the multi-ethnic populations of Asia-Pacific.
Standardised disease data collection forms using existing validated disease activity indices and disease classification systems for PIBD were stored electronically on a centrally-hosted secure REDCAP platform. Participating Asian-Pacific sites were invited to enrol patients with an established diagnosis of IBD from the point of initial presentation.
Epidemiological data from our registry demonstrates a rapid rise in PIBD incidence, particularly in the last 5 years. The unique disease characteristics of our Asian-Pacific cohort include a large proportion of very-early onset (VEO)-IBD (29.3%) similar to the high proportions of VEO disease reported from mainland China, Saudi Arabia and India. There is also a relative over-representation of the Indian/ South Asian ethnicity in the multi-ethnic countries of Singapore and Malaysia. Patients of Indian ethnicity with Crohn’s disease (CD) were also most likely to present with symptomatic perianal disease.
The rise in PIBD incidence across the Asian-Pacific region is consistent with the observed rise in incidence in many other global cohorts. The high proportion of very early onset IBD in the Asia-Pacific region may represent a shift in epidemiological trends in PIBD, as other established IBD cohorts similarly report the fastest growth in IBD incidence amongst very young children. The higher incidence of symptomatic perianal CD amongst Indian patients in Singapore/Malaysia suggests inter-ethnic genetic differences may cause variability in disease behaviour.
Future research should be targeted at establishing national disease data registries to fully encapsulate the true burden of IBD in the Asia-Pacific region, and prospective follow-up data from this multi-centre registry is essential to determine if any of the observed epidemiological trends at diagnosis have implications on longer term outcomes.
We thank Dr Dimple Rajgor for helping with editing, formatting, and submission of the manuscript for publication.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Singapore
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Naftali T, Israel; Poddighe D, Kazakhstan; Sugimoto M, Japan S-Editor: Wang JJ L-Editor: Filipodia P-Editor: Wang JJ
| 1. | Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, Panaccione R, Ghosh S, Wu JCY, Chan FKL, Sung JJY, Kaplan GG. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390:2769-2778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2677] [Cited by in RCA: 4102] [Article Influence: 512.8] [Reference Citation Analysis (110)] |
| 2. | Benchimol EI, Bernstein CN, Bitton A, Carroll MW, Singh H, Otley AR, Vutcovici M, El-Matary W, Nguyen GC, Griffiths AM, Mack DR, Jacobson K, Mojaverian N, Tanyingoh D, Cui Y, Nugent ZJ, Coulombe J, Targownik LE, Jones JL, Leddin D, Murthy SK, Kaplan GG. Trends in Epidemiology of Pediatric Inflammatory Bowel Disease in Canada: Distributed Network Analysis of Multiple Population-Based Provincial Health Administrative Databases. Am J Gastroenterol. 2017;112:1120-1134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 240] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 3. | Benchimol EI, Mack DR, Nguyen GC, Snapper SB, Li W, Mojaverian N, Quach P, Muise AM. Incidence, outcomes, and health services burden of very early onset inflammatory bowel disease. Gastroenterology. 2014;147:803-813.e7; quiz e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 193] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 4. | Morita N, Toki S, Hirohashi T, Minoda T, Ogawa K, Kono S, Tamakoshi A, Ohno Y, Sawada T, Muto T. Incidence and prevalence of inflammatory bowel disease in Japan: nationwide epidemiological survey during the year 1991. J Gastroenterol. 1995;30 Suppl 8:1-4. [PubMed] |
| 5. | Yang SK, Yun S, Kim JH, Park JY, Kim HY, Kim YH, Chang DK, Kim JS, Song IS, Park JB, Park ER, Kim KJ, Moon G, Yang SH. Epidemiology of inflammatory bowel disease in the Songpa-Kangdong district, Seoul, Korea, 1986-2005: a KASID study. Inflamm Bowel Dis. 2008;14:542-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 379] [Cited by in RCA: 381] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 6. | Ong C, Aw MM, Liwanag MJ, Quak SH, Phua KB. Rapid rise in the incidence and clinical characteristics of pediatric inflammatory bowel disease in a South-East Asian cohort in Singapore, 1994-2015. J Dig Dis. 2018;19:395-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Lee WS, Ng RT, Chan KW, Lau YL. Variable outcome in infantile-onset inflammatory bowel disease in an Asian cohort. World J Gastroenterol. 2016;22:10653-10662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 8. | Treepongkaruna S, Pienvichit P, Sornmayura P, Pornkul R, Wisedopas N, Phuapradit P. Inflammatory bowel disease in Thai children: presentations and outcomes of treatment. Asian Pac J Allergy Immunol. 2006;24:73-79. [PubMed] |
| 9. | Levine A, Koletzko S, Turner D, Escher JC, Cucchiara S, de Ridder L, Kolho KL, Veres G, Russell RK, Paerregaard A, Buderus S, Greer ML, Dias JA, Veereman-Wauters G, Lionetti P, Sladek M, Martin de Carpi J, Staiano A, Ruemmele FM, Wilson DC; European Society of Pediatric Gastroenterology, Hepatology, and Nutrition. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J Pediatr Gastroenterol Nutr. 2014;58:795-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 822] [Cited by in RCA: 993] [Article Influence: 90.3] [Reference Citation Analysis (0)] |
| 10. | Levine A, Griffiths A, Markowitz J, Wilson DC, Turner D, Russell RK, Fell J, Ruemmele FM, Walters T, Sherlock M, Dubinsky M, Hyams JS. Pediatric modification of the Montreal classification for inflammatory bowel disease: the Paris classification. Inflamm Bowel Dis. 2011;17:1314-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1216] [Cited by in RCA: 1144] [Article Influence: 81.7] [Reference Citation Analysis (0)] |
| 11. | Hyams J, Markowitz J, Otley A, Rosh J, Mack D, Bousvaros A, Kugathasan S, Pfefferkorn M, Tolia V, Evans J, Treem W, Wyllie R, Rothbaum R, del Rosario J, Katz A, Mezoff A, Oliva-Hemker M, Lerer T, Griffiths A; Pediatric Inflammatory Bowel Disease Collaborative Research Group. Evaluation of the pediatric crohn disease activity index: a prospective multicenter experience. J Pediatr Gastroenterol Nutr. 2005;41:416-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 257] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 12. | Turner D, Hyams J, Markowitz J, Lerer T, Mack DR, Evans J, Pfefferkorn M, Rosh J, Kay M, Crandall W, Keljo D, Otley AR, Kugathasan S, Carvalho R, Oliva-Hemker M, Langton C, Mamula P, Bousvaros A, LeLeiko N, Griffiths AM; Pediatric IBD Collaborative Research Group. Appraisal of the pediatric ulcerative colitis activity index (PUCAI). Inflamm Bowel Dis. 2009;15:1218-1223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 227] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 13. | Department of Statistics Malaysia Official Portal. Current Population Estimates, Malaysia 2020. [cited 3 October 2021]. Available from: https://www.dosm.gov.my/v1/index.php?r=column/cthemeByCat&cat=155&bul_id=OVByWjg5YkQ3MWFZRTN5bDJiaEVhZz09&menu_id=L0pheU43NWJwRWVSZklWdzQ4TlhUUT09. |
| 14. | Singapore Department of Statistics. Singapore Census of Population 2020. [cited 3 October 2021]. Available from: https://www.singstat.gov.sg/publications/reference/cop2020/cop2020-sr1. |
| 15. | Mak WY, Zhao M, Ng SC, Burisch J. The epidemiology of inflammatory bowel disease: East meets west. J Gastroenterol Hepatol. 2020;35:380-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 411] [Article Influence: 82.2] [Reference Citation Analysis (0)] |
| 16. | El Mouzan MI, Saadah O, Al-Saleem K, Al Edreesi M, Hasosah M, Alanazi A, Al Mofarreh M, Asery A, Al Qourain A, Nouli K, Al Hussaini A, Telmesani A, AlReheili K, Alghamdi S, Alrobiaa N, Alzaben A, Mehmadi A, Al Hebbi H, Al Sarkhy A, Al Mehaidib A, Al Saleem B, Assiri A, Wali S. Incidence of pediatric inflammatory bowel disease in Saudi Arabia: a multicenter national study. Inflamm Bowel Dis. 2014;20:1085-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 17. | Zayyani NR, Malaty HM, Graham DY. Increasing Incidence of Crohn's Disease with Familial Clustering in the Kingdom of Bahrain: A 25-Year Population-based Study. Inflamm Bowel Dis. 2017;23:304-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Ishige T, Tomomasa T, Hatori R, Tatsuki M, Igarashi Y, Sekine K, Arakawa H. Temporal Trend of Pediatric Inflammatory Bowel Disease: Analysis of National Registry Data 2004 to 2013 in Japan. J Pediatr Gastroenterol Nutr. 2017;65:e80-e82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 19. | Jung YS, Han M, Kim WH, Park S, Cheon JH. Incidence and Clinical Outcomes of Inflammatory Bowel Disease in South Korea, 2011-2014: A Nationwide Population-Based Study. Dig Dis Sci. 2017;62:2102-2112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 20. | Wang XQ, Zhang Y, Xu CD, Jiang LR, Huang Y, Du HM, Wang XJ. Inflammatory bowel disease in Chinese children: a multicenter analysis over a decade from Shanghai. Inflamm Bowel Dis. 2013;19:423-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 21. | Poddighe D, Telman A, Tuleutayev E, Ibrayeva A. Pediatric Ulcerative Colitis in Kazakhstan: First Case Series from Central Asia and Current Clinical Management. Gastroenterol Insights. 2020;11:27-35. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Srivastava A, Sathiyasekharan M, Jagadisan B, Bolia R, Peethambaran M, Mammayil G, Acharya B, Malik R, Sankaranarayanan S, Biradar V, Malhotra S, Philip M, Poddar U, Yachha SK. Paediatric inflammatory bowel disease in India: a prospective multicentre study. Eur J Gastroenterol Hepatol. 2020;32:1305-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Paudel MS, Khanal A, Shrestha B, Purbey B, Paudel BN, Shrestha G, Thapa J, Dewan KR, Gurung R, Joshi N. Epidemiology of Inflammatory Bowel Diseases in Nepal. Cureus. 2021;13:e16692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 24. | Shen YM, Wu JF, Chen HL, Hsu HY, Chang MH, Hsieh TK, Ni YH. Characteristics and incidences of pediatric Crohn's disease in the decades before and after 2000. Pediatr Neonatol. 2011;52:317-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Lee YM, Fock K, See SJ, Ng TM, Khor C, Teo EK. Racial differences in the prevalence of ulcerative colitis and Crohn's disease in Singapore. J Gastroenterol Hepatol. 2000;15:622-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 75] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 26. | Hilmi I, Jaya F, Chua A, Heng WC, Singh H, Goh KL. A first study on the incidence and prevalence of IBD in Malaysia--results from the Kinta Valley IBD Epidemiology Study. J Crohns Colitis. 2015;9:404-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 27. | Montgomery SM, Morris DL, Pounder RE, Wakefield AJ. Asian ethnic origin and the risk of inflammatory bowel disease. Eur J Gastroenterol Hepatol. 1999;11:543-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 28. | Fellows IW, Freeman JG, Holmes GK. Crohn's disease in the city of Derby, 1951-85. Gut. 1990;31:1262-1265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 77] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 29. | Pinsk V, Lemberg DA, Grewal K, Barker CC, Schreiber RA, Jacobson K. Inflammatory bowel disease in the South Asian pediatric population of British Columbia. Am J Gastroenterol. 2007;102:1077-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 122] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 30. | Malhotra R, Turner K, Sonnenberg A, Genta RM. High prevalence of inflammatory bowel disease in United States residents of Indian ancestry. Clin Gastroenterol Hepatol. 2015;13:683-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 31. | Brooks AW, Priya S, Blekhman R, Bordenstein SR. Gut microbiota diversity across ethnicities in the United States. PLoS Biol. 2018;16:e2006842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 207] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 32. | Castaño-Rodríguez N, Kaakoush NO, Lee WS, Mitchell HM. Dual role of Helicobacter and Campylobacter species in IBD: a systematic review and meta-analysis. Gut. 2017;66:235-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 167] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 33. | Khalid MA, Yang L. Income inequality and ethnic cleavages in Malaysia: Evidence from distributional national accounts (1984–2014). J Asian Econ. 2021;72:101252. [DOI] [Full Text] |
| 34. | Loftus EV Jr. Update on the Incidence and Prevalence of Inflammatory Bowel Disease in the United States. Gastroenterol Hepatol (N Y). 2016;12:704-707. [PubMed] |
| 35. | Wang YR, Loftus EV Jr, Cangemi JR, Picco MF. Racial/Ethnic and regional differences in the prevalence of inflammatory bowel disease in the United States. Digestion. 2013;88:20-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 36. | Kotze PG, Underwood FE, Damião AOMC, Ferraz JGP, Saad-Hossne R, Toro M, Iade B, Bosques-Padilla F, Teixeira FV, Juliao-Banos F, Simian D, Ghosh S, Panaccione R, Ng SC, Kaplan GG. Progression of Inflammatory Bowel Diseases Throughout Latin America and the Caribbean: A Systematic Review. Clin Gastroenterol Hepatol. 2020;18:304-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 125] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 37. | Singer AAM, Bloom DA, Adler J. Factors Associated With Development of Perianal Fistulas in Pediatric Patients With Crohn's Disease. Clin Gastroenterol Hepatol. 2021;19:1071-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 38. | Assa A, Rinawi F, Shamir R. The Long-Term Predictive Properties of the Paris Classification in Paediatric Inflammatory Bowel Disease Patients. J Crohns Colitis. 2018;12:39-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 39. | Dhaliwal J, Walters TD, Mack DR, Huynh HQ, Jacobson K, Otley AR, Debruyn J, El-Matary W, Deslandres C, Sherlock ME, Critch JN, Bax K, Seidman E, Jantchou P, Ricciuto A, Rashid M, Muise AM, Wine E, Carroll M, Lawrence S, Van Limbergen J, Benchimol EI, Church P, Griffiths AM. Phenotypic Variation in Paediatric Inflammatory Bowel Disease by Age: A Multicentre Prospective Inception Cohort Study of the Canadian Children IBD Network. J Crohns Colitis. 2020;14:445-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 40. | Banerjee R, Pal P, Nabi Z, Shava U, Ganesh G, Reddy DN. Very early onset inflammatory bowel disease in a South Asian country where inflammatory bowel disease is emerging: a distinct clinical phenotype from later onset disease. Intest Res. 2021;19:398-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 41. | Li BH, Guan X, Vittinghoff E, Gupta N. Comparison of the presentation and course of pediatric inflammatory bowel disease in South Asians with Whites: a single center study in the United States. J Pediatr. 2013;163:1211-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 42. | Jangi S, Ruan A, Korzenik J, de Silva P. South Asian Patients With Inflammatory Bowel Disease in the United States Demonstrate More Fistulizing and Perianal Crohn Phenotype. Inflamm Bowel Dis. 2020;26:1933-1942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 43. | Ray G. Inflammatory bowel disease in India - Past, present and future. World J Gastroenterol. 2016;22:8123-8136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 42] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 44. | Foster A, Jacobson K. Changing incidence of inflammatory bowel disease: environmental influences and lessons learnt from the South asian population. Front Pediatr. 2013;1:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 45. | Wang XQ, Xiao Y, Xu X, Yu Y, Shan CY, Guo Y, Gong L, Zhou T, Gao SS, Yuan YZ, Wang XJ, Xu CD. Study of disease phenotype and its association with prognosis of paediatric inflammatory bowel disease in China. BMC Pediatr. 2018;18:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 46. | Kim HJ, Oh SH, Kim DY, Lee HS, Park SH, Yang SK, Kim KM. Clinical Characteristics and Long-Term Outcomes of Paediatric Crohn's Disease: A Single-Centre Experience. J Crohns Colitis. 2017;11:157-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 47. | Kochar B, Barnes EL, Herfarth HH, Martin CF, Ananthakrishnan AN, McGovern D, Long M, Sandler RS. Asians have more perianal Crohn disease and ocular manifestations compared with white Americans. Inflamm Intest Dis. 2018;2:147-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 48. | Gower-Rousseau C, Vasseur F, Fumery M, Savoye G, Salleron J, Dauchet L, Turck D, Cortot A, Peyrin-Biroulet L, Colombel JF. Epidemiology of inflammatory bowel diseases: new insights from a French population-based registry (EPIMAD). Dig Liver Dis. 2013;45:89-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 145] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 49. | Guan DX, Yu FH, Wang GL, Zhou J, Wang DY, Nie XL, Xu XW. [Single center retrospective study of 184 children with inflammatory bowel disease seen from 2000-2014]. Zhonghua Er Ke Za Zhi. 2017;55:493-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 50. | Sawczenko A, Sandhu BK. Presenting features of inflammatory bowel disease in Great Britain and Ireland. Arch Dis Child. 2003;88:995-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 297] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 51. | Saverymuttu SH, Hodgson HJ, Chadwick VS, Pepys MB. Differing acute phase responses in Crohn's disease and ulcerative colitis. Gut. 1986;27:809-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 102] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Vermeire S, Van Assche G, Rutgeerts P. Laboratory markers in IBD: useful, magic, or unnecessary toys? Gut. 2006;55:426-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 654] [Cited by in RCA: 634] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 53. | D'Haens G, Ferrante M, Vermeire S, Baert F, Noman M, Moortgat L, Geens P, Iwens D, Aerden I, Van Assche G, Van Olmen G, Rutgeerts P. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:2218-2224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 553] [Cited by in RCA: 622] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 54. | Hildebrand H, Karlberg J, Kristiansson B. Longitudinal growth in children and adolescents with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 1994;18:165-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 118] [Article Influence: 3.8] [Reference Citation Analysis (0)] |