Published online Feb 21, 2020. doi: 10.3748/wjg.v26.i7.717
Peer-review started: November 18, 2019
First decision: December 23, 2019
Revised: January 7, 2020
Accepted: January 11, 2020
Article in press: January 11, 2020
Published online: February 21, 2020
Processing time: 94 Days and 18.6 Hours
Chronic constipation is a gastrointestinal functional disease that seriously harms physical and mental health and impacts the quality of life of patients. Its incidence rate is 2%-27%. Slow transit constipation (STC) is a common type of chronic functional constipation, accounting for 10.3%-45.5% of such cases. Scholars have performed many studies on the pathogenesis of STC. These studies have indicated that the occurrence of STC may be related to multiple factors, such as dysfunction of the enteric nervous system, interstitial cells of Cajal (ICC) damage, and changes in neurotransmitters regulating intestinal peristalsis.
To investigate the role of Tenascin-X (TNX) in regulating the TGF-β/Smad signaling pathway in the pathogenesis of STC.
This study included an experimental group and a control group. The experimental group included 28 patients with severe colonic STC, and the control group included 18 patients with normal colon tissues. Immunohistochemistry (IHC) was used to detect c-Kit, a specific marker of the ICC. Western blot, immunofluorescence, and IHC were used to detect the localization and expression of TNX and TGF-β/Smad.
IHC showed that the number of ICC with positive c-Kit expression was significantly reduced in the colon of STC patients (22.17 ± 3.28 vs 28.69 ± 3.53, P < 0.05) and that the distribution was abnormal. Western blot results showed that c-Kit and Smad7 levels were significantly decreased in the colon of STC patients (c-kit: 0.462 ± 0.099 vs 0.783 ± 0.178, P < 0.01; Smad7: 0.626 ± 0.058 vs 0.799 ± 0.03, P < 0.01) and that TNX and Smad2/3 levels were higher in the STC group (TNX: 0.868 ± 0.028 vs 0.482 ± 0.032, P < 0.01). There was no significant difference in TGF-β between the two groups (0.476 ± 0.028 vs 0.511 ± 0.044, P = 0.272). Pearson correlation analysis showed that the TNX protein exhibited a strong correlation with Smad2/3 and Smad7 (P < 0.05, |R| > 0.8) and TGF-β (P < 0.05, |R| = 0.7).
The extracellular matrix protein TNX may activate the TGF-β/Smad signaling pathway by upregulating the Smad 2/3 signaling protein and thereby induce slight or complete epithelial stromal cell transformation, leading to an abnormal distribution and dysfunction of ICC in the diseased colon, which promotes the occurrence and development of STC.
Core tip: The extracellular matrix protein Tenascin-X may activate the TGF-β/Smad signaling pathway by upregulating the Smad2/3 signaling protein and thereby induce slight or complete epithelial stromal cell transformation, leading to an abnormal distribution and dysfunction of interstitial cells of Cajal in the diseased colon, which promotes the occurrence and development of slow transit constipation.
- Citation: Zhang YC, Chen BX, Xie XY, Zhou Y, Qian Q, Jiang CQ. Role of Tenascin-X in regulating TGF-β/Smad signaling pathway in pathogenesis of slow transit constipation. World J Gastroenterol 2020; 26(7): 717-724
- URL: https://www.wjgnet.com/1007-9327/full/v26/i7/717.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i7.717
Chronic constipation is a gastrointestinal functional disease that seriously harms physical and mental health and impacts the quality of life of patients. Its incidence rate is 2%-27%. Slow transit constipation (STC) is a common type of chronic functional constipation, accounting for 10.3%-45.5% of such cases[1]. The clinical features of STC include decreased colonic motility and prolonged colonic transit time. The symptoms of this disease are a decreasing stool frequency, less or no desire to defecate, hard excrement, and abdominal distension. Some patients’ symptoms are so stubborn that they cannot be relieved by increasing the dose of laxatives. Scholars have performed many studies on the pathogenesis of STC. These studies have indicated that the occurrence of STC may be related to multiple factors, such as dysfunction of the enteric nervous system, interstitial cells of Cajal (ICC) damage, and changes in neurotransmitters regulating intestinal peristalsis. Among these factors, abnormal distribution and dysfunction of ICC in the colon are particularly important[2-5], which suggests that abnormal changes in ICC may be related to the mechanism of cell transdifferentiation, rather than apoptosis or death of cells[6].
Tenascin-X (TNX) is an important member of the TN family of extracellular matrix glycoproteins, which are involved in the progression of many diseases. Aktar et al[7] suggested that deficient expression or loss of TNX is highly correlated with Ehlers-Danlos syndrome. Patients with loss of TNX may suffer declining colonic motility as an obvious gastrointestinal symptom. Aktar et al[7] found that TNX-deficient mice show impaired defecation and neural control of distal colonic motility that can be rescued with a 5-HT4 receptor agonist. Fikree et al[8] reported that 37% of joint hypermobility syndrome patients experience GI symptoms including increased nausea, abdominal pain, bloating, constipation, and diarrhea compared with non- joint hypermobility syndrome controls[8]. The above studies suggest that there is a link between the TNX protein and colonic motility. Our research found that the expression of the TNX protein in the colon tissue of STC patients was notably increased in preliminary experiments; therefore, we inferred that this protein may show a certain correlation with the occurrence of STC. Over the past few years, research on TNX has gradually shifted toward whether the protein acts as a promoter of a signaling pathway. It was proven that part of the TNX domain activates the TGF-β signaling pathway, and the TGF-β/Smad signaling pathway was confirmed as an important pattern in cell transition[9-11]. Therefore, we further speculated that the TNX protein may induce the transition of ICC through regulation of the TGF-β/Smad signaling pathway, causing a decrease in the number of functional ICC changes and further leading to the occurrence of STC.
The aims of this study were to investigate the changes in the expression of TNX, TGF-β, Smad2/3, and Smad7 in the colon tissue of STC patients and to explore the influence of the TNX protein and its related signaling pathway on the occurrence of STC. This work will allow the exploration of and speculation about the mechanism of ICC changes at the molecular level.
Patients who were hospitalized in the Colorectal and Anal Surgery Department of Zhongnan Hospital of Wuhan University from January 2015 to October 2018 were selected for this study. The patients were diagnosed with STC according to the Rome III criteria. All patients in the STC group underwent colonic transit tests, defecography, and colonoscopy. The exclusion criteria were as follows: (1) Patients who did not meet the Rome III criteria; (2) Patients with chronic constipation caused by spastic pelvic floor syndrome, rectocele, megacolon, or other organ lesions; (3) Patients with obvious gastric intestinal transit dysfunction; (4) Patients with secondary constipation caused by drugs, endocrine, metabolic, or neurological diseases, renal failure, cirrhosis, severe hypertension, and severe obesity, and those with a body mass index exceeding 30; and (5) Patients with incomplete data that influenced the diagnosis. The control group included patients who experienced regular bowel movements with no history of constipation from whom colon tissue was collected because of abdominal colorectal trauma. All patients were assessed with the Wexner constipation score and the gastrointestinal quality of life index to evaluate their quality of life. The use of the materials was approved by the Ethics Committee of Zhongnan Hospital of Wuhan University and the agreement of the patients.
For immunohistological staining, colon tissue sections were incubated with a 100-fold-diluted TNX antibody (Proteintech, CN, United Stated) or c-Kit antibody (Abcam, United Kingdom) in blocking solution (2% BSA in PBS) at 4 °C overnight, followed by visualization using a poly-HRP anti-rabbit IgG detection kit.
The instructions for the extraction of cellular proteins for pretreatment were followed. The protein concentration was quantified by the BCA assay (Aspen, CN, United States), and equal amounts of denatured proteins (20 μg) were separated via 10%-12% SDS-PAGE and subsequently electrotransferred to PVDF membranes. The proteins of interest were detected with specific antibodies against TNX (Proteintech), TGF-β (Abcam, United Kingdom), Smad2/3 (CST, United States), and Smad7 (Proteintech). Protein bands were visualized after the binding of the secondary antibody with HRP-conjugated anti-rabbit IgG by using ECL reagents.
Data and statistical analyses were performed using SPSS 17.0, and all data are expressed as the mean ± SD. The comparisons of count data were performed by the chi-square test or Fisher's exact test. The data were compared between groups by the t-test, and correlation analysis was performed with the Pearson correlation coefficient. A P value < 0.05 was regarded as significant.
A total of 28 patients with STC and 18 normal controls were collected. Among the 28 STC patients, 7 were male, and 21 were female, with an average age of 56.86 ± 13.57 years. There were 18 subjects in the control group, including 6 males and 12 females; their average age was 50.00 ± 12.02 years. All subjects were examined to determine the Wexner constipation score to evaluate defecation function and gastrointestinal quality of life index to assess the impact of constipation on their quality of life, as shown in Table 1.
| Group | Age (yr) | Sex (Male/Female) | Wexner score | GIQLI | Colonic transit test (-/+) |
| Control | 50.00 ± 12.02 | 6/12 | 2.5 (0-6) | 131.3 (125-138) | - |
| STC | 56.86 ± 13.57 | 7/21 | 14.3 (11-18) | 95.3 (89-102) | + |
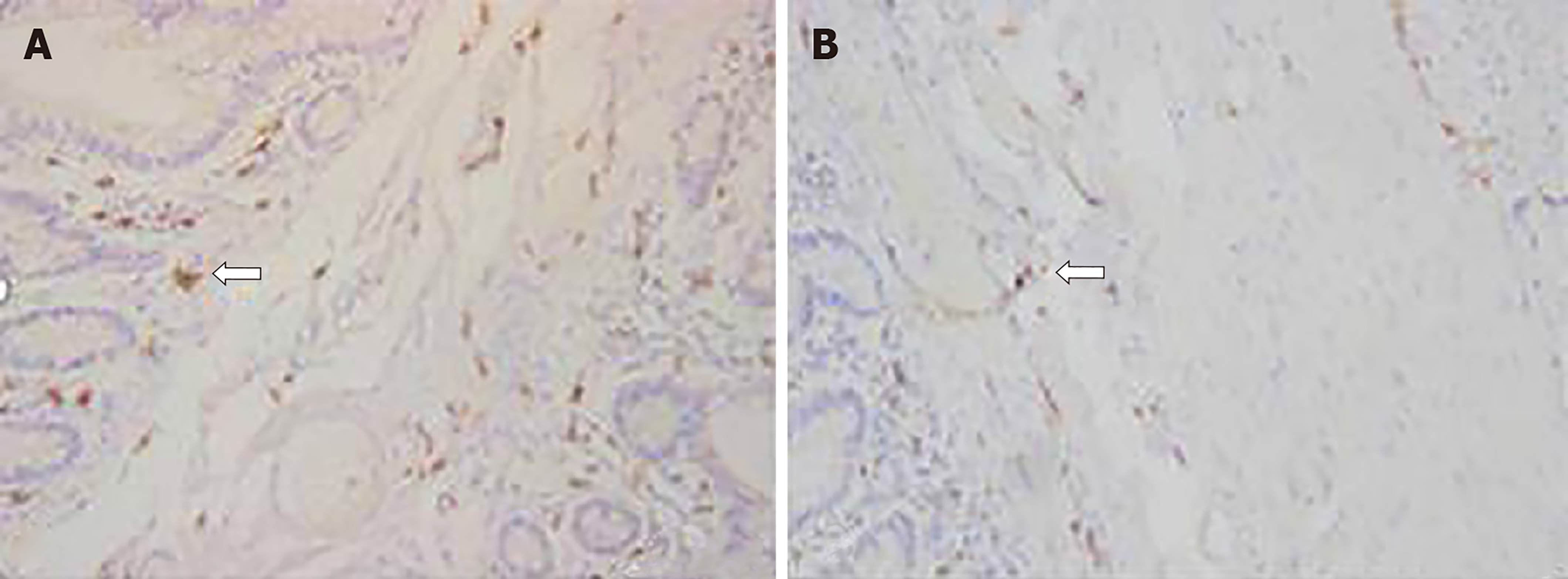
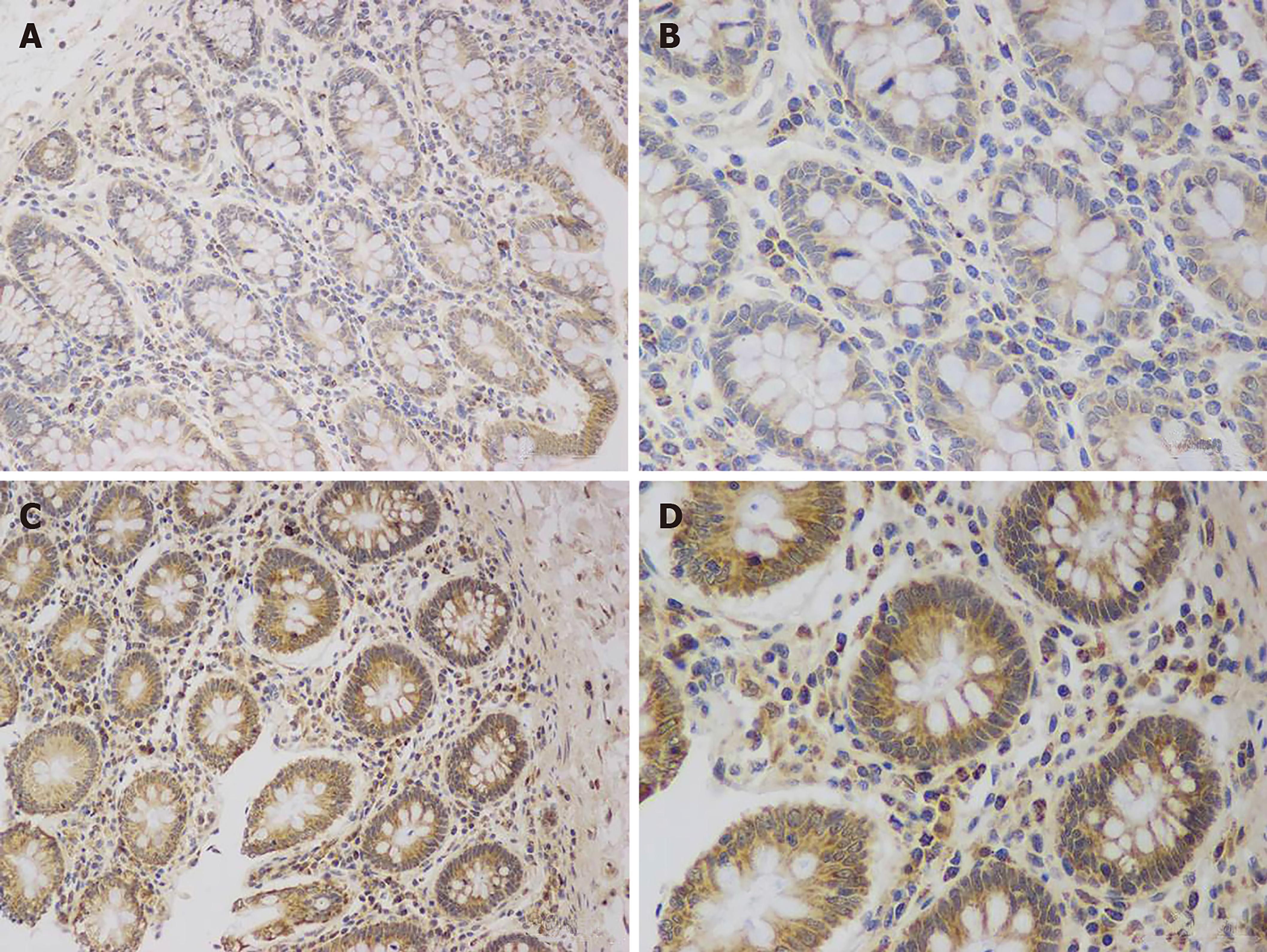
The number of ICC in the colon area of STC patients was significantly decreased compared with that in the control group (0.172 ± 0.013 vs 0.256 ± 0.021, P < 0.05), and their distribution was abnormal (Figure 1). TNX was expressed in both cells and the stroma, but the expression of TNX in the STC group was significantly increased compared with that in the control group (0.397 ± 0.023 vs 0.226 ± 0.017, P < 0.01) (Figure 2).
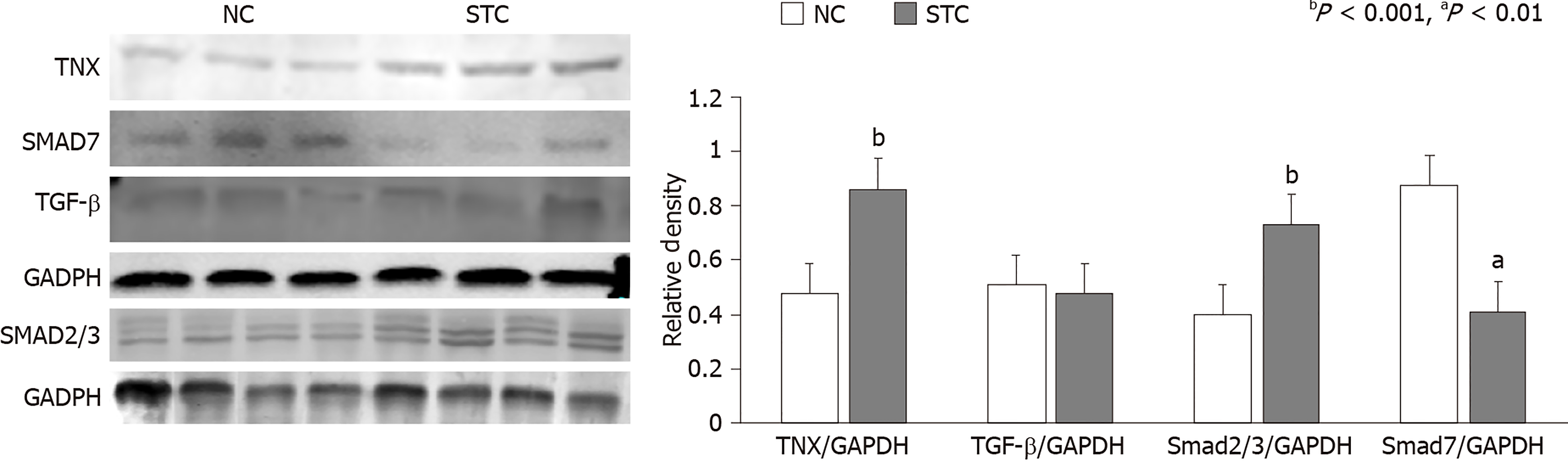
The expression of the TNX and Smad2/3 proteins in the colon tissue of STC patients was significantly higher than that in the control group (TNX: 0.868 ± 0.028 vs 0.482 ± 0.032, P < 0.01; Smad2/3: 0.733 ± 0.045 vs 0.405 ± 0.081, P < 0.01). Consistent with this finding, the expression of the Smad7 protein in the colon tissue of STC patients was decreased (0.626 ± 0.058 vs 0.799 ± 0.035, P < 0.01), and TGF-β expression was not significantly different between the two groups (0.476 ± 0.028 vs 0.511 ± 0.044, P = 0.272) (Figure 3).
Pearson correlation analysis showed that the TNX protein presented a strong correlation with Smad2/3 and Smad7 (P < 0.05, |R| > 0.8) and with TGF-β (P < 0.05, |R| = 0.7).
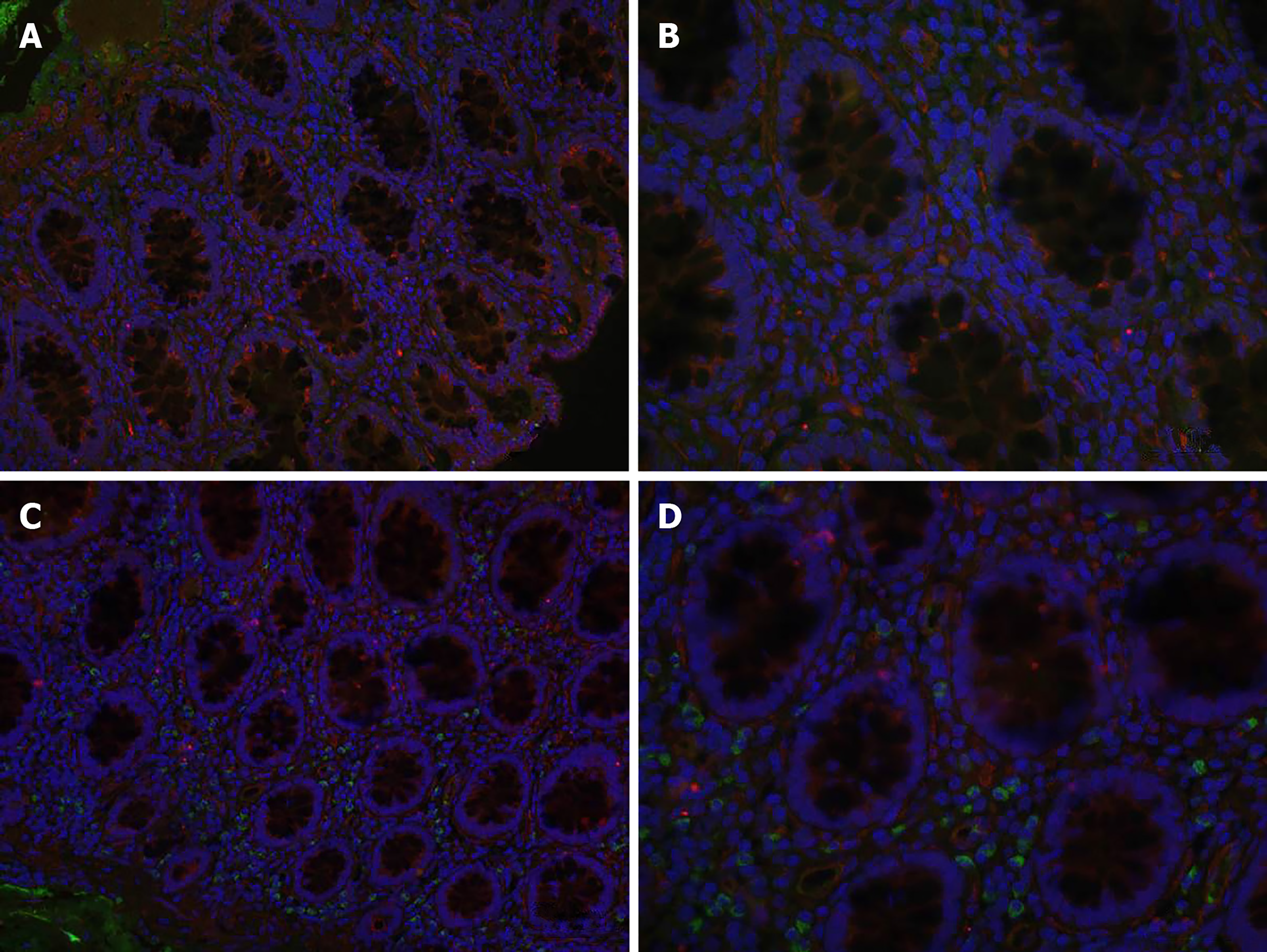
Laser confocal microscopy showed TGF-β expression as red fluorescence and Smad2/3 expression as green fluorescence, and nuclei were indicated by blue fluorescence. Both the STC and normal groups exhibited weak positive expression, which was located in the cytoplasm of stromal cells. The fluorescence intensity and density of Smad2/3 protein expression were significantly higher in the STC group than in the normal group (0.029 ± 0.002 vs 0.017 ± 0.001, P < 0.01). There was no significant difference in the fluorescence intensity or density of TGF-β between the two groups (0.030 ± 0.005 vs 0.033 ± 0.008, P > 0.05) (Figure 4).
With additional research, the etiology of STC is becoming clearer. The theory that 5-HT can stimulate colonic smooth muscle cholinergic nerves has been confirmed. Rubina A demonstrated that 5-HT receptor agonists can improve constipation symptoms in TNX knockout mice and suggested a relationship between the TNX protein and gastrointestinal function for the first time. The important role of the relationship between STC and TNX in gastrointestinal functional disease has not been reported previously, so it is worthy of further study.
TGF-β belongs to a group of the TGF-β superfamily that regulates cell growth, proliferation, and transition through downstream signaling pathways. The precursors of TGF-β are cleaved by protease hydrolysis to form disulfide-bonded active TGF-β dimers and an inactive binding peptide that binds to a TGF-β-binding protein in an inactive form and is stored in the extracellular matrix. Under certain physiological or pathological conditions, active dimers are released from the complex, activate downstream signaling pathways, and regulate cell proliferation and differentiation. The currently recognized downstream pathways include the TGF-β/Smad signaling pathway and a Smad protein-independent MAPK pathway. Alcaraz found that the C-terminal fibrinogen-like domain of TNX could promote epithelial-to-mesenchymal transition by activating latent TGF-β[12].
In this study, it was found that the expression of the TNX protein in the colon of STC patients was significantly higher than that in the normal group by approximately 1.5-2 times. At the same time, it was found that the expression of Samd2/3 in the STC group was increased significantly, whereas the expression of the Smad7 inhibitor decreased, and these differences were correlated with the change in the TNX expression level. However, there was no significant difference in the change in TGF-β. It may be that TNX activates latent TGF-β in the extracellular matrix, rather than inducing the synthesis of TGF-β.
At this stage, it was found that the expression level of the TNX protein was positively correlated with the TGF-β/Smad signaling pathway in the colon tissues of STC patients. The direction of transdifferentiation can be inferred according to the morphological changes in ICC observed by electron microscopy and the patterns of specific expression factors after differentiation, such as α-SMA in the smooth muscle phenotype. Furthermore, ICC transdifferentiation rather than apoptosis or death was verified. Then, based on the cytological animal experiments, the changes in the TGF-β/Smad signaling pathway and the occurrence of STC under the two conditions were explored through gene knockout and overexpression of the TNX protein.
Previous research indicated that Ehlers-Danlos syndrome patients also exhibit colon motility disorders and constipation symptoms, which is contrary to this experimental phenomenon. It is speculated that the changes in ICC are not affected by the TGF-β/Smad signaling pathway but are impacted by inhibition of the synthesis and release of cholinergic nerve acetylcholine, by reducing the maximum number of cholinergic receptors bound in the colon, or by reducing the reactivity of colon smooth muscle to Ach, leading to gastrointestinal electromyographic arrhythmia or abnormal changes in other nerves and their transmitters in the enteric nervous system, which destroys the integrity and coordination of the intestinal nervous system and leads to secondary intestinal transmission dysfunction.
This hypothesis is consistent with the observations that the number of AchE-positive neurons in the colonic myenteric plexus is reduced and that Ach expression is decreased in patients with STC[13]. Therefore, subsequent detection of cholinergic neurons and neurotransmitters can be performed to investigate whether the degree of change is statistically significant. Furthermore, whether the changes in cholinergic nerves are reversed by interfering with the expression of TNX can be examined through in vitro experiments.
At present, the medical treatment of STC is still accepted by the public, but STC is generally poorly treated by dietary structure adjustment, biofeedback, fecal transplantation, drug ladder treatment, or dependence on laxatives. Therefore, surgical treatment has gradually become mainstream. After continuous improvement, the main surgical procedures applied now include total colectomy and ileorectal anastomosis, subtotal colectomy, colonic exclusion, and ileal pouch anastomosis to reduce the number of bowel movements. Although the surgical treatments for STC tend to be varied, they are clearly associated with major surgical trauma and postoperative complications. Therefore, the in-depth exploration of the pathogenesis of STC and the precise localization of therapy still deserve our attention. This study showed that the expression of the TNX protein is increased in the colon tissue of STC patients, which could affect the transdifferentiation of ICC by activating the TGF-β/Smad signaling pathway, upregulating the Smad2/3 excitatory factors, and downregulating the Smad7 inhibitory factor. The transdifferentiation of ICC could be inhibited by interfering with the signaling molecules in this pathway, thereby reducing or reversing the occurrence of STC, providing a new experimental basis for the clinical treatment of STC.
Chronic constipation is a gastrointestinal functional disease that seriously harms physical and mental health and impacts the quality of life of patients. Its incidence rate is 2%-27%. Slow transit constipation (STC) is a common type of chronic functional constipation accounting for 10.3%-45.5% of such cases. At present, the most effective treatment for STC is still surgery, but it is more traumatic. We need to further explore the pathogenesis of the disease, through the intervention of gene or protein expression, to alleviate or prevent the occurrence of the disease.
There is no previous report which has linked constipation to the TGF-β/Smad signaling pathway and Tenascin-X (TNX) protein, and it is also the first time that TNX protein has been introduced into clinical research. The pathogenesis of STC is very complex, surgical treatment is effective, but it is traumatic. Therefore, it is necessary to find a new direction of treatment from the pathogenesis.
The main objective was to explore whether interstitial cells of Cajal (ICC) changes are related to transdifferentiation, and whether TNX protein can cause ICC changes through the TGF-β/Smad signaling pathway. At this stage, it was found that the expression level of the TNX protein was positively correlated with the TGF-β/Smad signaling pathway in the colon tissues of STC patients. Besides, ICC transdifferentiation rather than apoptosis or death was verified. Next, we need to confirm that the direction of transdifferentiation according to the morphological changes in ICC observed by electron microscopy and the patterns of specific expression factors after differentiation, such as α-SMA in the smooth muscle phenotype.
We collected surgical specimens of patients and tested them by different laboratory methods including qualitative and quantitative tests, such as colon immunohistochemistry, immunofluorescence staining, and Western blot. The data were compared between groups by the t-test, and correlation analysis was performed with the Pearson correlation coefficient.
The expression of the TNX protein in the colon of STC patients was significantly higher than that in the normal group by approximately 1.5-2 times. At the same time, it was found that the expression of Samd2/3 in the STC group was increased significantly, whereas the expression of the Smad7 inhibitor decreased, and these differences were correlated with the change in the TNX expression level. The results may provide a new experimental basis for the clinical treatment of STC.
The expression of the TNX protein is increased in the colon tissue of STC patients, which could affect the transdifferentiation of ICC by activating the TGF-β/Smad signaling pathway, upregulating the Smad2/3 excitatory factors, and downregulating the Smad7 inhibitory factor. The transdifferentiation of ICC could be inhibited by interfering with the signaling molecules in this pathway, thereby reducing or reversing the occurrence of STC, providing a new experimental basis for the clinical treatment of STC.
In the future, based on the cytological animal experiments, the changes in the TGF-β/Smad signaling pathway and the occurrence of STC under the two conditions need to be explored through gene knockout and overexpression of the TNX protein. We may be able to regulate the upstream and downstream signaling molecules of this pathway with targeting drugs, so as to alleviate or inhibit the development of STC.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Hashimoto N S-Editor: Zhang L L-Editor: Wang TQ E-Editor: Liu JH
| 1. | Shahid S, Ramzan Z, Maurer AH, Parkman HP, Fisher RS. Chronic idiopathic constipation: more than a simple colonic transit disorder. J Clin Gastroenterol. 2012;46:150-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 2. | Knowles CH, Farrugia G. Gastrointestinal neuromuscular pathology in chronic constipation. Best Pract Res Clin Gastroenterol. 2011;25:43-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Geramizadeh B, Hayati K, Rahsaz M, Hosseini SV. Assessing the interstitial cells of Cajal, cells of enteric nervous system and neurotransmitters in slow transit constipation, using immunohistochemistry for CD117, PGP9.5 and serotonin. Hepatogastroenterology. 2009;56:1670-1674. [PubMed] |
| 4. | Kashyap P, Gomez-Pinilla PJ, Pozo MJ, Cima RR, Dozois EJ, Larson DW, Ordog T, Gibbons SJ, Farrugia G. Immunoreactivity for Ano1 detects depletion of Kit-positive interstitial cells of Cajal in patients with slow transit constipation. Neurogastroenterol Motil. 2011;23:760-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Garrity MM, Gibbons SJ, Smyrk TC, Vanderwinden JM, Gomez-Pinilla PJ, Nehra A, Borg M, Farrugia G. Diagnostic challenges of motility disorders: optimal detection of CD117+ interstitial cells of Cajal. Histopathology. 2009;54:286-294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Sanders KM, Ordög T, Ward SM. Physiology and pathophysiology of the interstitial cells of Cajal: from bench to bedside. IV. Genetic and animal models of GI motility disorders caused by loss of interstitial cells of Cajal. Am J Physiol Gastrointest Liver Physiol. 2002;282:G747-G756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 102] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 7. | Aktar R, Peiris M, Fikree A, Cibert-Goton V, Walmsley M, Tough IR, Watanabe P, Araujo EJA, Mohammed SD, Delalande JM, Bulmer DC, Scott SM, Cox HM, Voermans NC, Aziz Q, Blackshaw LA. The extracellular matrix glycoprotein tenascin-X regulates peripheral sensory and motor neurones. J Physiol. 2018;596:4237-4251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 8. | Fikree A, Grahame R, Aktar R, Farmer AD, Hakim AJ, Morris JK, Knowles CH, Aziz Q. A prospective evaluation of undiagnosed joint hypermobility syndrome in patients with gastrointestinal symptoms. Clin Gastroenterol Hepatol. 2014;12:1680-87.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 9. | Hills CE, Squires PE. The role of TGF-β and epithelial-to mesenchymal transition in diabetic nephropathy. Cytokine Growth Factor Rev. 2011;22:131-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 124] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 10. | Fuxe J, Vincent T, Garcia de Herreros A. Transcriptional crosstalk between TGF-β and stem cell pathways in tumor cell invasion: role of EMT promoting Smad complexes. Cell Cycle. 2010;9:2363-2374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 273] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 11. | Tang WB, Ling GH, Sun L, Liu FY. Smad anchor for receptor activation (SARA) in TGF-beta signaling. Front Biosci (Elite Ed). 2010;2:857-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Alcaraz LB, Exposito JY, Chuvin N, Pommier RM, Cluzel C, Martel S, Sentis S, Bartholin L, Lethias C, Valcourt U. Tenascin-X promotes epithelial-to-mesenchymal transition by activating latent TGF-β. J Cell Biol. 2014;205:409-428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 13. | Porter AJ, Wattchow DA, Brookes SJ, Costa M. Cholinergic and nitrergic interneurones in the myenteric plexus of the human colon. Gut. 2002;51:70-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 93] [Article Influence: 4.0] [Reference Citation Analysis (0)] |