Published online Nov 7, 2020. doi: 10.3748/wjg.v26.i41.6510
Peer-review started: July 10, 2020
First decision: August 8, 2020
Revised: August 21, 2020
Accepted: September 29, 2020
Article in press: September 29, 2020
Published online: November 7, 2020
Processing time: 119 Days and 5.3 Hours
I read with interest an article “Updated meta-analysis of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis” by Fan and colleagues in World J Gastroenterol 2015; 21(24): 7577-7583. Although I appreciate their work, I have found problems with the data extracted and analyzed by the authors, and will give my comment in this letter. It would be valuable if the authors could provide an accurate estimation of their extracted data.
Core Tip: Data extraction errors appeared to exist in a published meta-analysis entitled “Updated meta-analysis of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis” by Fan and colleagues in World J Gastroenterol 2015; 21(24): 7577-7583.
- Citation: Chu JT. Comment on “Updated meta-analysis of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis”. World J Gastroenterol 2020; 26(41): 6510-6513
- URL: https://www.wjgnet.com/1007-9327/full/v26/i41/6510.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i41.6510
I read with interest the article “Updated meta-analysis of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis” by Fan et al[1]. In this study, the authors performed an updated meta-analysis to evaluate the prophylactic effect of pancreatic duct (PD) stents in post-endoscopic pancreatitis (PEP). This is a significant study. However, after reading the article carefully, I found some worthwhile issues which I would like to discuss with the authors.
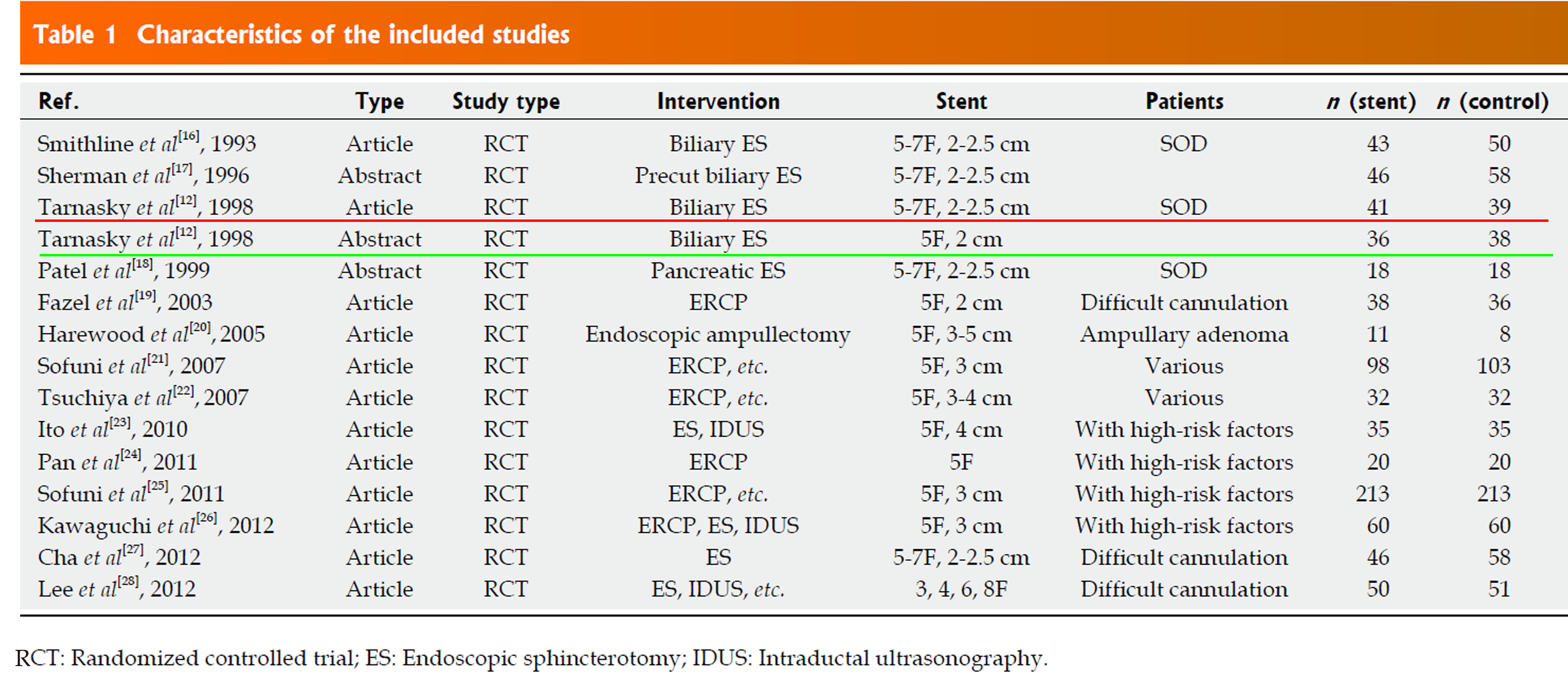
First, in the meta-analysis, the authors finally included 15 randomized controlled trials. Two of the 15 studies were labeled with the same reference, but the extracted data were different (Figure 1). This makes me doubt the accuracy of the data. I retrieved published clinical trials on prophylactic PD stents to prevent PEP from MEDLINE (between 1980 and May 2013), EMBASE (between 1980 and May 2013), and the Cochrane clinical trial databases. As a result, I have found that the data underlined in green do not exist. Therefore, I concluded that there were errors in the extracted data.
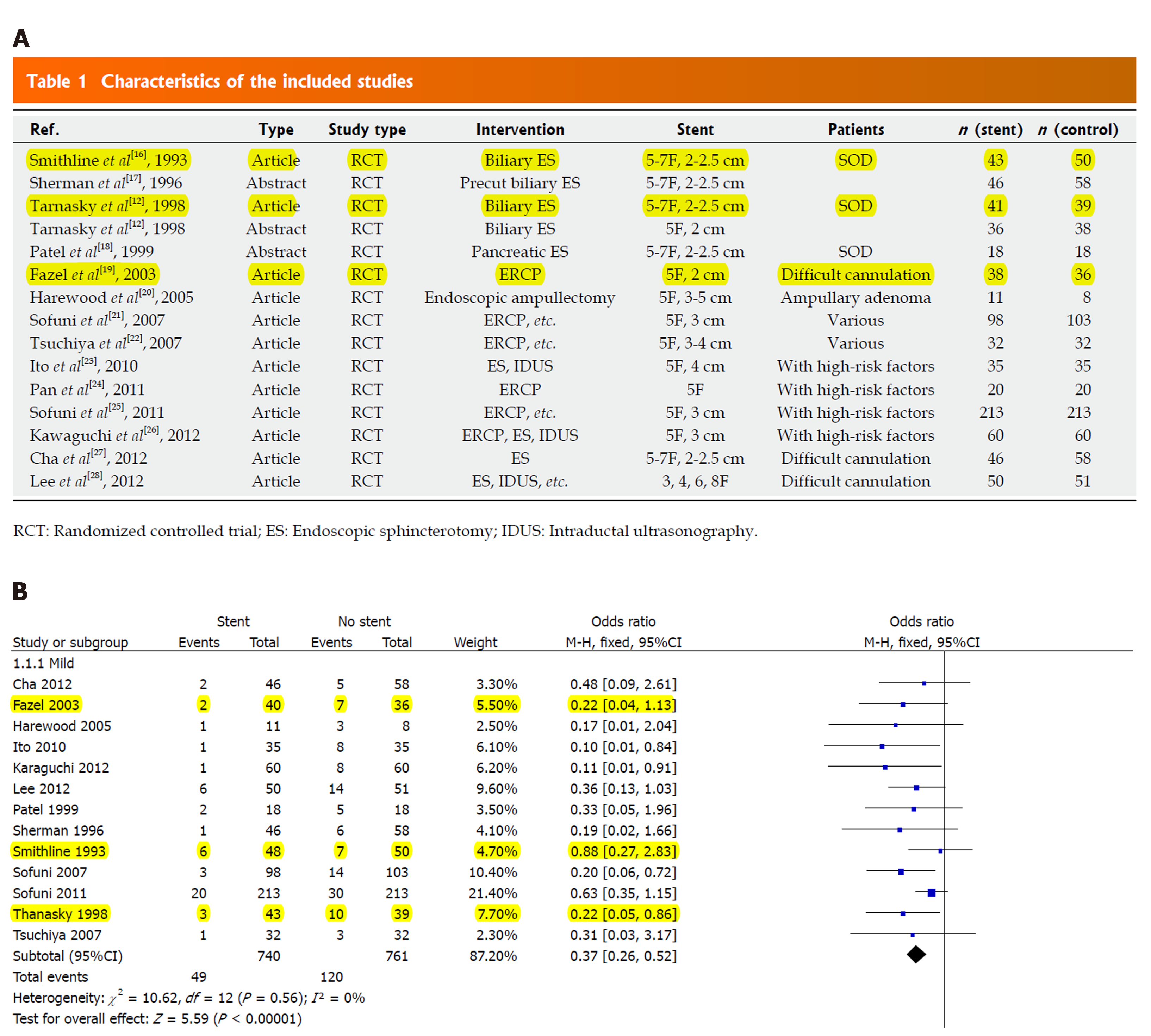
Second, in their study, the initial extracted data were inconsistent with the later data in the meta-analysis (Figure 2). The reason was not mentioned by the authors. Smithline et al[2] reported in 1993 a total of 98 alternately randomized patients: 50 to the no-stent group and 48 to the stent group. Stent placement was unsuccessful in 5 patients. If the number of patients who underwent treatment was used as the result in the meta-analysis, the total number of patients in the stent group should be 48. However, if the number of patients who were successfully treated was used as the result, the total number of patients in the stent group should be 43. In the meta-analysis, the authors did not describe the analysis method used.
In the study by Thanasky et al[3] in 1998, 80 patients were randomized to the stent (n = 41) or no stent groups (n = 39). One patient had mild pancreatitis in the stent group, another 2 patients in the stent group developed mild pancreatitis after stent extraction. So, the total number of patients in the stent group should be 41, and the number of event patients in the stent group should be 3.
In the study by Fazel et al[4] in 2003, two patients randomized to the PD stent group were excluded. If the intent-to-treat analysis was included, the total number of stents would be 40, and the number of event patients would be 2; the total number of people in the control group should be 36, and the number of event patients should be 7. But the authors did not state which analysis method was used.
In summary, we admire the efforts by the authors to clarify the role of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis. Nevertheless, it would be valuable if the authors could provide an accurate estimation of the extracted data to address my questions.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Abdollahi M, Saito S-Editor: Gong ZM L-Editor: MedE-Ma JY P-Editor: Ma YJ
| 1. | Fan JH, Qian JB, Wang YM, Shi RH, Zhao CJ. Updated meta-analysis of pancreatic stent placement in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis. World J Gastroenterol. 2015;21:7577-7583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 45] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 2. | Smithline A, Silverman W, Rogers D, Nisi R, Wiersema M, Jamidar P, Hawes R, Lehman G. Effect of prophylactic main pancreatic duct stenting on the incidence of biliary endoscopic sphincterotomy-induced pancreatitis in high-risk patients. Gastrointest Endosc. 1993;39:652-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 140] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 3. | Tarnasky PR, Palesch YY, Cunningham JT, Mauldin PD, Cotton PB, Hawes RH. Pancreatic stenting prevents pancreatitis after biliary sphincterotomy in patients with sphincter of Oddi dysfunction. Gastroenterology. 1998;115:1518-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 267] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 4. | Fazel A, Quadri A, Catalano MF, Meyerson SM, Geenen JE. Does a pancreatic duct stent prevent post-ERCP pancreatitis? A prospective randomized study. Gastrointest Endosc. 2003;57:291-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 207] [Article Influence: 9.4] [Reference Citation Analysis (0)] |