Published online Oct 21, 2020. doi: 10.3748/wjg.v26.i39.5944
Peer-review started: June 28, 2020
First decision: July 28, 2020
Revised: August 8, 2020
Accepted: September 25, 2020
Article in press: September 25, 2020
Published online: October 21, 2020
Processing time: 114 Days and 22.8 Hours
Inflammatory bowel disease (IBD) includes Crohn’s disease (CD), ulcerative colitis and unclassified entities. CD commonly involves the terminal ileum and colon but at the time of diagnosis it can be confined to the small bowel (SB) in about 30% of the patients, especially in the young ones. Management of isolated SB-CD can be challenging and objective evaluation of the SB mucosa is essential in differentiating CD from other enteropathies to achieve therapeutic decisions and to plan the follow-up. The introduction of cross-sectional imaging techniques and capsule endoscopy (CE) have significantly expanded the ability to diagnose SB diseases providing a non-invasive test for the visualization of the entire SB mucosa. The main CE limitations are the low specificity, the lack of therapeutic capabilities and the impossibility to take biopsies. Device assisted enteroscopy (DAE) enables histological confirmation when traditional endoscopy, capsule endoscopy and cross-sectional imaging are inconclusive and also allows therapeutic interventions such as balloon stricture dilation, intralesional steroid injection, capsule retrieval and more recently stent insertion. In the current review we will discuss technical aspect, indications and safety profile of DAE in children and adults with IBD.
Core Tip: Evaluation of the small bowel (SB) mucosa is essential in differentiating Crohn’s disease (CD) from other enteropathies to achieve therapeutic decisions and to plan follow-up. The introduction of cross-sectional imaging techniques and capsule endoscopy have significantly improved the diagnostic approach to SB disease providing a non-invasive diagnostic method for the visualization of the entire SB mucosa. However, Device assisted enteroscopy (DAE) has further revolutionized management of SB-CD enabling histological confirmation and allowing therapeutic interventions. In the current review we will discuss technical aspect, indications and safety profile of DAE in children and adults with inflammatory bowel disease.
- Citation: Di Nardo G, Esposito G, Ziparo C, Micheli F, Masoni L, Villa MP, Parisi P, Manca MB, Baccini F, Corleto VD. Enteroscopy in children and adults with inflammatory bowel disease. World J Gastroenterol 2020; 26(39): 5944-5958
- URL: https://www.wjgnet.com/1007-9327/full/v26/i39/5944.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i39.5944
Inflammatory bowel disease (IBD) represents a group of chronic inflammatory disorders that involve the colon, small bowel (SB) and the entire gastrointestinal tract and include Crohn’s disease (CD), ulcerative colitis (UC) and unclassified entities[1,2]. CD is a chronic immuno-mediated inflammation that most commonly involves the terminal ileum and colon, but at the time of diagnosis, it can be confined to the SB, as seen in approximately 30% of CD patients, especially young patients[2-8]. Isolated SB-CD can be challenging to diagnose and manage for several reasons. First, the SB is less easily accessible by endoscopy, making it easy to miss a SB-CD diagnosis with conventional endoscopy contributing to a delay in diagnosis as observed in many patients with CD[5]. Second, SB ulcerations induced by infection (such as tuberculosis) or drugs can sometimes be difficult to differentiate from CD[1,8]. Third, compared with the other phenotypes, SB-CD is associated with an increased risk of relapse and stricture development[1,3,6,7]. Fourth, SB cancer associated with CD is a rare but difficult problem because only a minority of these cases are diagnosed preoperatively and at an early stage[9]. Finally, in the paediatric population, SB-CD has particular clinical relevance for its negative impact on growth and pubertal development[2,7]. Thus, objective evaluation of the SB mucosa is essential in differentiating CD from other enteropathies to make therapeutic decisions and to plan follow-up.
For many years, investigation of the SB has been a challenge because of its anatomy, location, and relative tortuosity. The introduction of cross-sectional imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI), enterography/enteroclysis and SB ultrasound, have enhanced SB assessment with great accuracy in evaluating transmural and extraluminal disease, but subtle mucosal changes can still be missed[8,10,11].
Capsule endoscopy (CE) and device-assisted enteroscopy (DAE) have significantly expanded the ability to diagnose SB diseases. CE provides a non-invasive test for the visualization of the entire SB mucosa, which can aid in the diagnosis of SB-CD and monitoring the therapeutic response. The main CE limitations in IBD patients are a low specificity, a lack of therapeutic capabilities and the inability to perform biopsies[12,13].
DAE enables histological confirmation when other modalities, such as traditional endoscopy, CE and cross-sectional imaging, are inconclusive and allows therapeutic interventions, such as balloon stricture dilation, intralesional steroid injection, capsule retrieval and, more recently, stent insertion[12-14].
In the current review, we will discuss the technical aspects, indications and safety profiles of DAE in children and adults with IBD.
DAE is a generic term for any endoscopic technique that includes assisted progression (i.e., balloons, overtubes or other devices) and includes double-balloon enteroscopy (DBE), single-balloon enteroscopy (SBE), balloon-guided endoscopy (BGE) and spiral enteroscopy (SE). Table 1 shows the main characteristics of the currently available DAE systems.
| Company | DAE system type | Endoscope model | Optical field of view | Optical depth of field | Endoscope distal outer diameter | Endoscope channel inner diameter | Endoscope working length | Overtube outer diameter | Image enhancement |
| Fujifilm Corporation (Tokyo, Japan) | DBE | EN-580T | 140 | 2-100 mm | 9, 4 mm | 3, 2 mm | 200 cm | 13, 2 mm | FICE |
| DBE | EN-580XP | 140 | 2-100 mm | 7, 5 mm | 2, 2 mm | 200 cm | 11, 6 mm | FICE | |
| DBE | EI-580BT | 140 | 2-100 mm | 9, 4 mm | 3, 2 mm | 156 cm | 13, 2 mm | FICE | |
| Pentax Medical (Tokyo, Japan) | DBE on demand using BGE | G-EYE34-i10L/F | 140 | 2-100 mm | 11, 5 mm | 3, 8 mm | 170 cm | NA | i-scan OE |
| DBE on demand using BGE | G-EYE38-i10L/F | 140 | 2-100 mm | 13, 2 mm | 3, 8 mm | 170 cm | NA | i-scan OE | |
| DBE on demand using BGE | G-EYE38-i10F2 | 140 | 2-100 mm | 13, 2 mm | 3, 8 mm | 150 cm | NA | i-scan OE | |
| Olympus (Tokyo, Japan) | SBE | SIF-Q180 | 140 | 3-100 mm | 9, 2 mm | 2, 8 mm | 200 cm | 13, 2 mm | NBI |
| SBE | SIFH290S | 140 | 3-100 mm | 9, 2 mm | 3, 2 mm | 152 cm | 13, 2 mm | NBI | |
| SE | SIF-Y0019 Motorized spiral overtube | 140 | 2-100 mm | 11, 3 mm | 3, 2 mm | 168 cm | 31, 1 mm max1;18, 1 mm distal2 | NBI | |
| NaviAid, Smart Medical Systems, (Ra’anana, Israel) | BGE | No specific scope required | NA | NA | NA | NA | NA | NA | NA |
| Spirus Medical (Stoughton, United States) | SE | No specific scope required | NA | NA | NA | NA | NA | 14, 5 mm | NA |
The DBE system was presented for the first time in Japan in 2001, and the first paediatric report was described in 2003. The DBE system is constituted of a high-resolution enteroscope with a latex balloon attached to the end of the enteroscope and a second balloon attached to the tip of a polyurethane overtube. Both balloons are inflated and deflated using an external pressure-controlled pump system. Currently, three DBE systems are available. The P type is the thinnest; however, the small diameter of its working channel (2.2 mm) limits the possibility of performing advanced therapeutic procedures. The T type has an outer diameter of 9.4 mm and a working channel diameter of 3.2 mm. The main advantage of the short type (working length of 152 cm) is the compatibility with all conventional devices; it can also be used for endoscopic retrograde cholangiopancreatography in patients with altered anatomy and difficult or failed colonoscopy[14-18].
SBE was introduced in 2007, and in contrast to DBE, there is no balloon at the tip of the enteroscope; therefore, handling of the balloon control unit is simplified. The enteroscope is a high-resolution video endoscope, the overtube and balloon are made of silicon, and a control unit with a safety pressure setting that controls the balloon inflation and deflation[14-18].
DBE and SBE use the push-and-pull method, however, the number of balloons makes a slight difference in the insertion technique between the two balloon assisted enteroscopy (BAE) systems.
Two operators are generally needed to perform DBE. For the antegrade approach, the endoscope and overtube are advanced to the duodenum beyond the major papilla, at which point the balloon located on the overtube is inflated to hold the small bowel tightly. The enteroscope is then advanced to the distal side of the SB, and its balloon is inflated to prevent slippage of the scope backward. The balloon on the overtube is then deflated, and the overtube is advanced towards the tip of the enteroscope. The balloon on the overtube is then reinflated. The enteroscope-overtube is then withdrawn to fold the SB along the overtube. This process can be repeated until the maximal insertion point or the target lesion is reached[16].
SBE is usually performed by two operators, but it may be easier than DBE to perform with a single-operator. The enteroscope and overtube are advanced similar to the DBE insertion technique. However, the enteroscope tip must be angled during advancement of the overtube and only after the angulation of the tip, the overtube is advanced towards the tip of the enteroscope, and the overtube balloon is then inflated. The enteroscope-overtube is then withdrawn to fold the SB along the overtube. The overtube balloon is left inflated and the enteroscope is advanced from the overtube tip. This cycle of forward advancement and withdrawal is repeated until the maximal insertion point or target lesion is reached[16].
Retrograde insertion is more difficult than antegrade insertion, even in experienced hands. For both techniques (DBE and SBE), the enteroscope and overtube are advanced to the caecum. After inflation of the overtube balloon, the enteroscope-overtube is withdrawn to decrease the ileo-colic angle. With the overtube balloon inflated, the enteroscope is then passed through the ileocecal valve, and endoscope balloon is inflated within the ileum to hold the position. The overtube is then advanced into the ileum with the balloon deflated. For SBE, although the insertion technique is the same, backward slippage of the tip of the enteroscope to the caecum during insertion of the overtube is frequent because of the lack of a balloon at the enteroscope tip, which would enable holding the position[16].
DBE and SBE have the same procedural technique in children and adults. BAE is suitable and safe for children aged > 3 years and weight > 14 kg. However, due to a smaller abdominal cavity, thinner intestinal walls and a narrower intestinal lumen, BAE requires a greater level of skill in younger children[18].
BGE is performed by using an on-demand through-the-scope (TTS) balloon inserted in the working channel of any endoscope. The balloon is then inflated, allowing anchoring in the SB, and progression is obtained with repeated push-and-pull maneuvres by sliding the endoscope over the catheter. The balloon catheter can be removed to perform therapeutic interventions. The main limitation of this technique is the low stability of the endoscope during the therapeutic procedure due to the lack of an anchoring balloon[14,15]. To overcome this drawback, a colonoscope with an integrated latex-free balloon at the bending section has recently been developed (see Table 1). These colonoscopes, combined with a disposable advancing balloon (AB) applied through the instrument channel, provide the assembly to perform deep SB double-balloon endoscopy. The balloon endoscopes as well as the AB devices are controlled simultaneously by the inflation system. The feasibility and safety of BGE using the NaviAid AB device has recently been evaluated in children with IBD[19].
SE involves the use of an overtube with a raised spiral ridge. The SB is pulled and pleated onto the overtube by continuous rotation of the spiral, and advancement into the SB is allowed by clockwise rotation of the overtube during insertion and anticlockwise rotation during withdrawal. SE overtubes have been replaced by a new motorized spiral enteroscopy (NMSE) system. NMSE system is composed of a reusable endoscope with an integrated motor permitting the rotation of a short spiral overtube placed on the insertion tube portion of the endoscope and a motor control unit with a foot pedal and visual force gauge. The foot pedal activates the drive motor located in the endoscope handle, which controls the rotational direction and speed of a coupler located in the middle of the insertion tube of the endoscope. The bowel is pleated or unpleated on the insertion tube of the endoscope with clockwise or anticlockwise rotation, respectively. After reaching the required depth of insertion, the endoscope will be withdrawn using motorized anticlockwise spiral rotation[14,20]. Preliminary data shows that NMSE offers advantages over traditional methods, in particular concerning the duration of the procedure and the relative ease of use; otherwise, it has similar diagnostic and therapeutic yields as both SBE and DBE[20,21]. Nevertheless, data on SE in IBD patients are not yet available, and the large diameter of the SE overtube makes this technique unsuitable for children.
The approach (oral, anal or both) is determined on clinical judgement, cross-sectional imaging (CT/MRI - enterography) and CE. Usually, the oral approach is the first choice due to its lower technical difficulty[16,17,22]. Both oral and anal approaches are used if inspection of the whole intestine is clinically needed. An ink tattoo or clip is left at the deepest point of insertion achieved during the first approach.
Overnight fasting of 12 h for solid food and 4 h for clear liquids before starting the procedure is enough for the oral approach, the same bowel cleansing suggested for colonoscopy required for retrograde enteroscopy. General anaesthesia is recommended for long procedures or for patients in whom sedation is not appropriate (i.e., paediatric and high-risk patients). Fluoroscopy is not always needed, except when adhesions or massive SB-CD involvement is expected and when enteroscopy is aimed to perform stricture dilation[16,17,22]. Insufflation of CO2 is recommended due to its capability to allow deeper SB intubation of the scope and minimize postprocedural discomfort[22,23].
Five studies evaluated the impact of DAE on adults with suspected IBD (Table 2)[24-28].
| Ref. | DAE system type | Study design | CD patients (n) | Diagnostic yield (%) | Impact on patient management (%) | |||
| Suspected | Known | Suspected | Known | Suspected | Known | |||
| Mensink et al[36], J Gastroenterol 2009 | DBE | Retrospective | 0 | 40 | 60 | 75 | ||
| Kondo et al[33], J Gastroenterol 2010 | DBE | Retrospective | 25 | 50 | 47 | 53 | ||
| Möschler et al[34], Endoscopy 2011 | DBE | Prospective | 193 | 47 | NA | |||
| Schulz et al[24], Dig Endosc 2014 | DBE | Retrospective | 16 | 0 | 69 | NA | ||
| Navaneethan et al[25], Endosc Int Open 2014 | SBE or DBE | Retrospective | 22 | 43 | 27 | 53 | NA | 53 |
| Christian et al[35], World J Gastrointest Endosc 2016 | Retrograde SBE | Retrospective | 29 | 41.4 | 17 | |||
| Rahman et al[26], Gastrointest Endosc 2015 | DBE | Retrospective | 43 | 38 | 79 | 87 | 77 | 82 |
| Tun et al[27], Eur J Gastroenterol Hepatol 2016 | DBE | Retrospective | 100 | 0 | NA | 45 | ||
| Holleran et al[28], Scand J Gastroenterol 2018 | SBE | Retrospective | 13 | 39 | 39 | 77 | 69 | |
In a retrospective German study, 16 adult patients with clinical suspicion of isolated SB-CD underwent DBE after negative gastroscopy and colonoscopy. Abnormal SB findings were detected in 7 patients (44%) but pathognomonic histological findings of CD were found in only one case (6%). However, a diagnosis of CD was confirmed in 11 out of 16 (69%) patients taking into account clinical, endoscopic and radiological features[24].
Navaneethan et al[25] retrospectively reviewed a BAE registry, which included DBE and SBE procedures performed on adult patients, to assess the diagnostic yield and clinical impact of BAE in suspected SB-CD. They identified 22 patients with suspected SB-CD and inconclusive results from conventional upper and lower gastrointestinal endoscopy, radiological cross-sectional imaging studies, and CE. These patients underwent BAE, which provided a histological diagnosis of CD in 6 patients (27.3%) and non-steroidal anti-inflammatory drug-induced enteropathy in 3 patients (13.6%), whereas no lesions were found in 13 patients (59.1%). One newly diagnosed CD patient underwent successful balloon dilation of a jejunal stricture without complications. The authors also evaluated the agreement rate between CT or MRI enterography and BAE findings which was quite low (36.4%).
In a multicenter retrospective study, 43 adult patients with suspected CD based on abnormal cross-sectional imaging or CE were evaluated by DBE. The diagnostic yield reached 79%. SB-CD diagnosis was confirmed in 17 patients, and DBE examination was normal in 12 cases. The remaining 5 patients received alternative diagnoses, such as NSAID ulceration, stricture/solitary ulcer, anastomotic ulcer, or Meckel’s diverticulum. Another main outcome measure of this study was a comparison of DBE and CE findings. Overall, only 46% of lesions were confirmed on DBE. In 33 (77%) patients, BAE modified clinical management by the exclusion of CD in 11 patients, confirming CD in 17, diagnosing stenosis in 2, nonspecific ulcer in 2 and stopping the NSAID treatment in 1[26].
Tun et al[27] retrospectively evaluated the impact of DBE and histology on the diagnosis and management of 100 adult patients with suspected SB-CD for whom, based on clinical and laboratory data and after colonoscopy and radiological imaging studies or CE, a diagnosis of CD was not achieved. Abnormal macroscopic DBE findings were detected in 60 patients (ulcers, n = 47; stricture, n = 11; abnormal mucosa, n = 2), and biopsy samples was obtained. Twenty-three showed no histological abnormalities despite positive macroscopic appearances on DBE, whilst among the remaining 37 patients, according to histological examination, the diagnosis of CD was confirmed in 8 patients (22%), and 15 (41%) had histology suggestive of CD.
Nevertheless, combining clinical, biochemical, endoscopic, and histological findings, 45% of all patients received treatment for CD. After a median follow-up period of 27 mo, the diagnosis of CD was confirmed in 38% of DBE-positive patients[27].
The diagnostic yield and the impact on clinical outcome of the use of SBE for suspected SB-CD were evaluated in an Italian retrospective study, which included 13 adult patients. The diagnostic yield was 39%, detecting four patients with active ileitis and one with ileal stricture. For the remaining eight patients, a new diagnosis of CD was reached in 4 patients (8%) and excluded in the other 4 patients (8%)[28].
Five studies, two on SBE, two on DBE and one on BGE, evaluated the impact of DAE on children with suspected IBD[19,29-32] (Table 3).
| Ref. | DAE system type | Study design | Patients (n) | Previous diagnostic work-up | Impact on patient management (%) | Therapeutic procedure | Complications |
| Di Nardo et al[29], Gastrointest Endosc 2012 | SBE | Prospective | Suspected CD = 16, known CD = 14 | MRI and CE | Suspected CD = 87%, known CD = 64% | Balloon dilation = 5 pts | Not reported |
| de Ridder et al[30], Gastrointest Endosc 2012 | SBE | Prospective | Suspected CD = 14, known CD = 6 | MRI and US | Suspected CD = 57%, known CD = 83% | NA | Not reported |
| Uchida et al[32], Pediatr Int 2012 | DBE | Prospective | Suspected CD = 8, known CD = 4 | Upper GI endoscopy, colonoscopy and SB-contrast study | Suspected CD = 75%, known CD = 75% | Balloon dilation = 1 pt | Not reported |
| Urs et al[31], J Pediatr Gastroenterol Nutr 2014 | DBE | Prospective | Suspected CD = 3, known CD = 5 | CE | Suspected CD = 66%, known CD = 100% | NA | Not reported |
| Broide et al[19], J Pediatr Gastroenterol Nutr 2020 | BGE | Prospective | Suspected IBD = 15, known IBD = 16 | NA | NA | NA | Not reported |
In a study previously published by our group, 16 paediatric patients with suspected CD and unspecific findings after extensive assessment with upper and lower GI endoscopy, MRE and CE were assessed by SBE. SBE provided a histological diagnosis of CD in 12 patients and eosinophilic enteropathy in 2 patients, no lesions were found in the remaining 2 patients. Moreover, SBE allowed dilation of SB strictures identified on MRI in 2 suspected CD patients[29].
de Ridder et al[30] evaluated the diagnostic yield of SBE for paediatric CD. In this study, patients were evaluated directly by two-route SBE, not preceded by conventional endoscopy or CE, and in 8 out of 14 patients with suspected CD, the diagnosis was confirmed after SBE.
Urs et al[31] performed DBE in 3 patients with suspected CD after CE examination indicating mucosal abnormalities. DBE led to a CD diagnosis in 2 out of 3 patients, and CD was excluded in the other child due to the lack of mucosal lesions and a normal histology.
The study from Uchida et al[32], evaluated the efficacy and safety of DBE in 8 children with suspected CD after inconclusive gastroscopy, colonoscopy and an SB-contrast study. DBE confirmed a CD diagnosis in 2 out of 8 patients, led to an alternative diagnosis in four patients and did not find mucosal lesions or histological abnormalities in the remaining 2 patients.
Recently, Broide et al[19] evaluated the feasibility and safety of BAE using a NaviAid AB device in children with suspected IBD. Technical success was achieved in 95.23% and 85.7% of the anterograde and retrograde approaches, respectively. Moreover, the total procedure time was significantly shorter and the learning curve was faster than with BAE, as its operation is intuitive and simple. Unfortunately, the diagnostic yield of this technique was not assessed. In the 15 patients with suspected IBD, 3 patients were diagnosed with UC and 3 patients with CD; the remaining 9 patients showed no intestinal abnormalities.
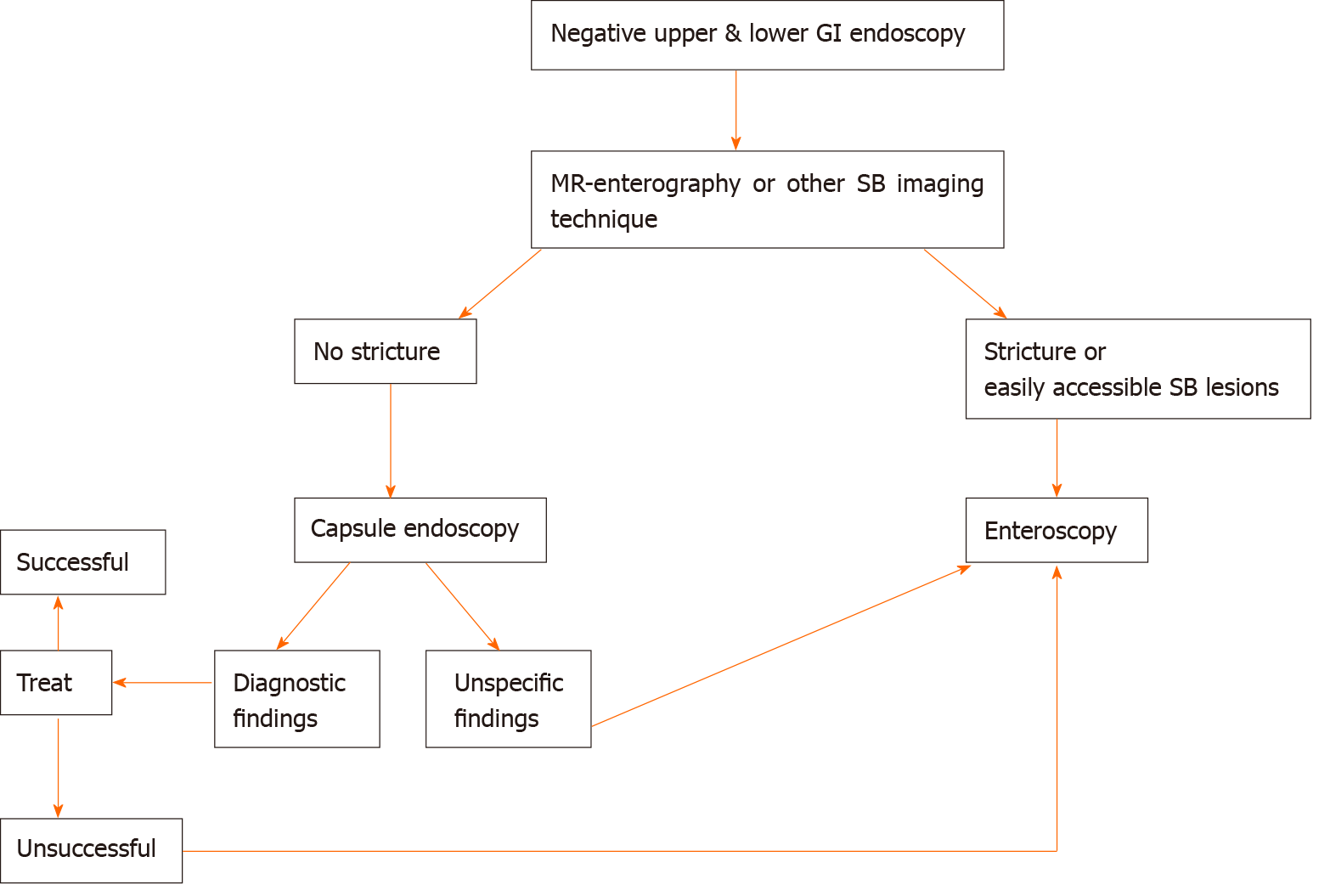
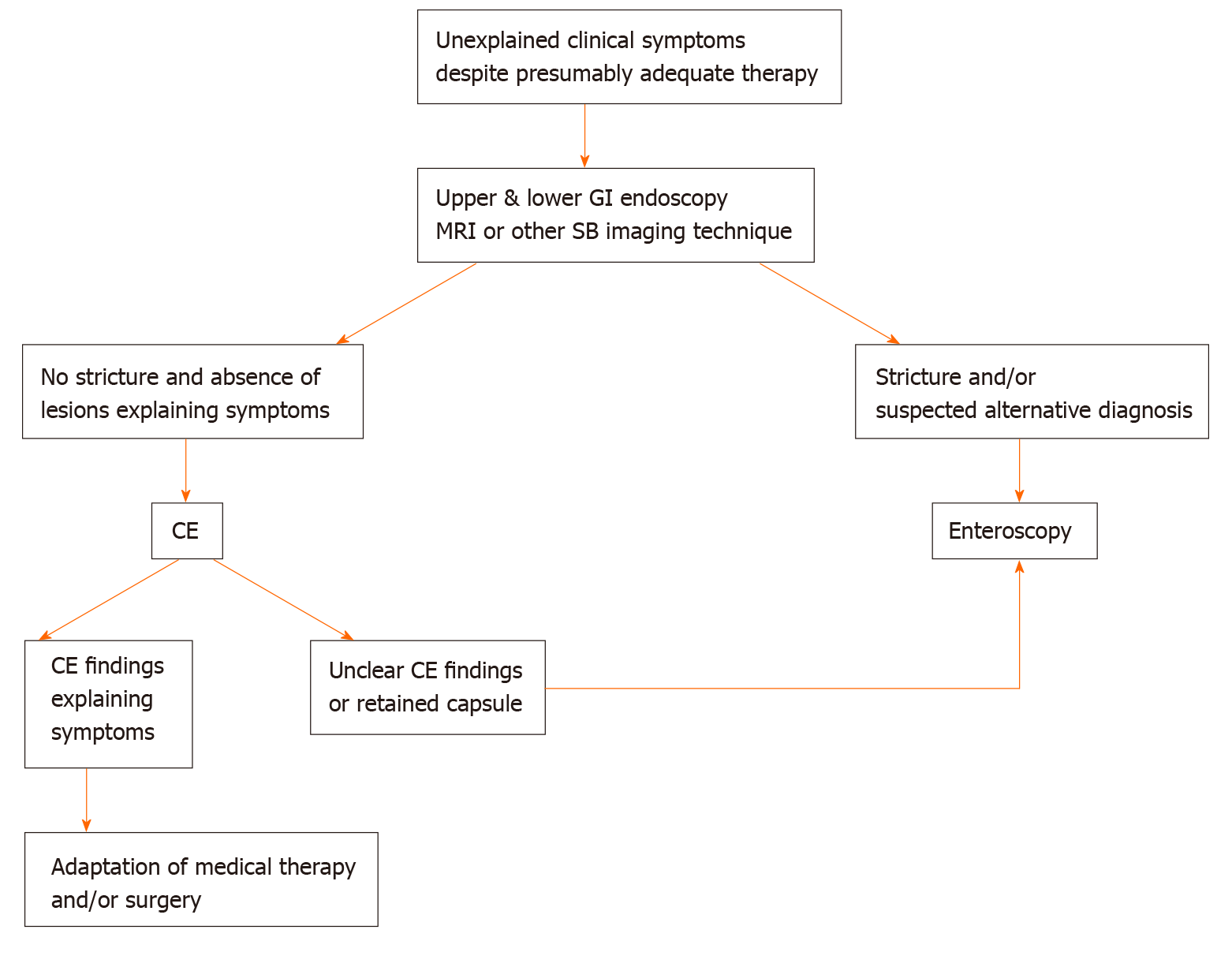
According to the previously described literature and our previously published algorithm (Figure 1), in children with suspected IBD, we suggest the following approach. DAE is recommended when conventional endoscopy, imaging of the SB and CE are inconclusive and tissue sampling and/or therapeutic procedures would alter disease management. DAE should be the preferred primary endoscopic procedure only if a stricture or an easy-to-reach lesion (i.e., proximal SB wall thickness) is suspected at imaging. This is reasonable due to DAE diagnostic and therapeutic possibilities and to the high capsule retention risk[6,18].
Three studies evaluated the impact of DAE on both suspected and already known IBD adult patients (Table 2)[33-35].
Kondo et al[33] analyzed a total of 1444 cases of DBE performed for various indications collected in a multicenter database to investigate the efficacy of DBE for the diagnosis and treatment of CD on adult patient. A total of 50 known and 25 newly diagnosed patients with CD were included. Active inflammatory lesions (ulcer and erosion/redness) were found in 51.2% of the symptomatic patients, but they were also detected in 33.3% of the asymptomatic CD patients. Overall, the treatment was altered for 53.3% of the patients, resulting in introduction of anti-TNF antibody (20.4%) or other medication (17.5%), dose reduction (1.9%), endoscopic balloon dilation (EBD, 7.8%), and surgical treatment (5.8%).
In 2011, Möschler et al[34] published the results derived from a large prospective German database that reported data from 2245 DBE examinations carried out on 1765 adult patients over a 2-year period. Overall, 193 patients (11%) with known or suspected CD underwent endoscopic examination of the SB, showing pathological findings in 91 of them, with a diagnostic yield of 47%.
In a retrospective study, Christian et al[35] evaluated the diagnostic and therapeutic yields of SBE using solely a retrograde approach. Overall, 136 retrograde SBE procedures were considered. Twenty-nine (21.3%) were performed on adult patients with suspected or known CD. Twelve new diagnoses of CD were established, with a diagnostic yield of 41.4%, and the therapeutic yield was 17.2%.
Four studies evaluated the impact of BAE on adults with known IBD (Table 2)[25,26,28,36].
Mensink et al[36] assessed the clinical impact of endoscopic evaluation of the SB by DBE for patients with known CD and clinical suspicion of SB activity. They retrospectively analyzed 52 DBE procedures performed in 40 adult patients. Twenty-four patients (60%) showed macroscopically active inflammation of the SB, and 18 of them (75%) had to switch therapy with persistent clinical improvement in 83% of patients after a mean follow-up of 13 mo. In particular, amongst the 18 patients, 11 introduced anti-TNF therapy, 2 switched from infliximab to adalimumab, 1 introduced steroid therapy, 2 underwent surgical resection, and 2 underwent balloon dilation.
In the study from Navaneethan et al[25], BAE was performed in 43 adult patients with an established diagnosis of CD whose main indications were disease activity and extent assessment in uninvestigated CD, anaemia or obscure gastrointestinal bleeding, confirmation and treatment of SB strictures diagnosed on radiological examination, and evaluation of activity and extent of CD in postoperative patients. BAE had impact on clinical management of 23 patients (53.4%): 18 patients (41.8%) had active inflammation with ulcers or strictures, which led to a switch of medical therapy or surgery, 5 (11.6%) with documented stenosis in the absence of active ulcers underwent EBD to treat obstructive symptoms. Overall, thirteen patients (30%) required surgery: Two due to lack of other therapeutic strategies, five as a result of medical treatment escalation failure, five based on patient’s choice, and one due to bowel perforation after BAE. Finally, the authors found that the agreement rate between CT or MRI enterography and BAE findings were higher (75.6 %) in already diagnosed CD patients.
In the study by Rahman et al[26], the authors analyzed the diagnostic yield and the clinical impact of DBE on the management of 38 adult patients with known CD and clinical suspicion of SB disease activity. In this setting, the diagnostic yield was 87% (33/38 patients), revealing an active disease in 11 patients (29%), 5 with CD stricture (13%), 3 (8%) with functional obstruction due to fixed/angulated bowel, 3 (8%) with anastomotic ulcers, and 2 (5%) with nonspecific ulceration. DBE examination was normal in 9 cases (23.6%). Therefore, DBE resulted in a clinical management change for 82% of patients with known CD. Thirteen patients (34%) needed an increase or a switch of therapy or surgery, and three patients (8%) underwent endoscopic dilation without any related complications. One patient (1.2%) underwent surgery due to perforation consequent to diagnostic DBE and directly related to an ulcer at an anastomosis site.
Holleran et al[28] evaluated the diagnostic yield and the impact on clinical outcome of the use of SBE for 39 patients with established SB-CD. In this setting, the diagnostic yield was significantly higher than for adult patients with suspected CD (77% vs 39%, P < 0.01). The most frequent findings were ileal or anastomotic strictures in 38% and 26%, respectively, and active ileitis in 21% of patients. SBE had an immediate clinical impact on 69% (n = 33) of patients, including stricture dilation in 27%, adjustment of medications in 48%, and referral for surgical resection in 6%. Long-term follow-up (mean duration of 11 mo, range of 3-22 mo), performed in 34 patients (65%) of the 52 patients, showed a significant change in the mean Harvey-Bradshaw index score from 6.6 to 4.2 after the procedure (P < 0.0001).
Regarding the therapeutic role of enteroscopy in adult patients with IBD complications, several studies evaluated the clinical impact of EBD using BAE for SB stricture in CD (Table 4)[25,28,33,37-45]. Among them, Hirai et al[45] conducted, to our knowledge, the largest multicenter study currently available, which prospectively enrolled 112 patients with symptomatic SB strictures related to CD to clarify the efficacy and safety of EBD. Ninety-five patients (85%) were included, and balloon dilation was successful in 89 (94%) of them. The primary endpoint related to short-term outcomes was an improvement of symptoms, which was achieved in 66 patients (70%). The dilation diameter was significantly larger (15.20 ± 1.70 mm vs 13.65 ± 2.59 mm, P = 0.03) in the short-term symptomatic improvement group than in the no improvement group. There were no other significant differences in the groups’ baseline characteristics or stricture features.
| Ref. | DAE system type | Study design | CD patients (n) | Dilation diameter (mm), mean (range) | Technical success (%) | Clinical efficacy (%) | Dilatations (n), mean per patient (range) | Perforation (%) | Observation period (mo), mean (range) | Symptom recurrence (%) |
| Fukumoto et al[37], Gastrointest Endosc 2007 | DBE | Retrospective | 23 | NA | NA | 74 | 35, (1.52; 1-6) | 0 | 12, (1-40) | 26 |
| Pohl et al[38], Eur J Gastroenterol Hepatol 2007 | Push enteroscopy | NA | 10 | 17, (12-20) | 80 | 60 | 15, (1.5; 1-3) | 0 | 10, (4-16) | 40 |
| Ohmiyaet al[39], Gastrointest Endosc 2009 | DBE | Retrospective | 16 | NA, (8-20) | 96 | 69 | NA | 0 | 16, (2-43) | 31 |
| Despottet al[40], Gastrointest Endosc 2009 | DBE | Prospective | 11 | 15.4, (12-20) | 73 | 73 | 18, (2; 1-3) | 9 | 20.5, (2-41) | 25 |
| Hirai et al[41], Dig Endosc 2010 | DBE | Retrospective | 25 | NA, (12-18) | 72 | 72 | 55, (2.2; 1-4) | 0 | 11, (6-29) | 22 |
| Kondo et al[33], J Gastroenterol 2010 | DBE | Retrospective | 8 | NA | 100 | 87.5 | 18, 1.5 (1-2) | 0 | NA | NA |
| Gill et al[42], Ther Adv Gastroenterol 2014 | DBE | Retrospective | 10 | 13.5, (10-16.5) | 80 | 70 | 17, (1.8; 1-4) | 20 | NA | 14 |
| Hirai et al[43], Dig Endosc 2014 | DBE | Retrospective | 65 | NA, (12-18) | 80 | 80 | NA | 1.5 | NA | 48 |
| Navaneethan et al[25], Endosc Int Open 2014 | BAE (SBE/DBE) | Retrospective | 6 | NA | 100 | 100 | 7, (1.16; 1-2) | 16 | NA | NA |
| Sunada et al[44], Inflamm Bowel Dis 2016 | DBE | Retrospective | 85 | 12.4, (8–20) | NA | 87 | 321, (3.8; 1–14) | 5 | 41.9, (0–141) | 78.5 |
| Holleran et al[28], Scand J Gastroenterol 2018 | SBE | Retrospective | 13 | 13, (12-15) | 100 | 80 | 14, (1; 1-2) | 0 | 8, (2-16) | 23 |
| Hirai et al[45], Journal of Crohn's and Colitis 2018 | SBE and DBE | Prospective | 95 | 15, (8-20) | 94 | 70 | 90, (1; 1-2) | 0 | 24, (NA) | NA |
The long-term outcomes of EBD for SB stricture in CD adult patients were extensively evaluated by two retrospective Japanese studies[43,44].
Hirai et al[43] evaluated 65 CD patients with obstructive symptoms caused by endoscopically manageable SB strictures (stricture length ≤ 5 cm, not associated with fistulae, abscesses or deep ulcers, and without severe curvature of the stricture) with clinical success in 80.0% of patients (52/65). During the observation period after the initial EBD (mean 41.8 ± 24.9 mo), seventeen patients (26.2%) underwent surgery. The cumulative surgery-free rate was 79% and 73% at 2 and 3 years, respectively, and it was significantly higher among successful EBD cases (P < 0.0001). Moreover, the cumulative re-dilation-free rate was 64% at 2 years and 47% at 3 years. No significant differences in terms of concomitant treatment or initial dilation method were detected between patients with and without the need for re-dilation.
More recently, Sunada et al[44] analyzed data regarding 85 patients who underwent DBE-assisted EBD for SB-CD strictures and were then followed-up for a mean period of 41.9 mo (range, 0-141). The surgery-free rate after the initial EBD was 87.3% at 1 year, 78.1% at 3 years, and 74.2% at 5 years. Univariate analysis showed that the presence of an internal fistula beside the refractory stricture was significantly associated with the need for surgical intervention (hazard ratio: 5.50; 95%CI: 2.16-14.0; P = 0.01).
Five studies, two on SBE, two on DBE and one on BGE evaluated the impact of BAE in children with established IBD (Table 3)[19,29-32].
In our study, the SBE findings of 14 patients with longstanding CD and symptoms unexplained by conventional endoscopy led to the introduction of or to a change in therapeutic approach in 11 patients. Moreover, SBE allowed successful and safe dilation of SB strictures identified on MRE in 3 patients[29].
In the study of de Ridder et al[30] SBE findings led to a change in therapy in five out of six patients with established CD.
In the paper from Urs et al[31], in the established CD group, 2 patients had adverse reactions to infliximab with poor response to adalimumab and 3 patients underwent DBE for disease evaluation and consideration of escalation of treatment. DBE led to a change of treatment in all 5 patients.
Uchida et al[32] evaluated the efficacy and safety of DBE in 4 children with established CD. After DBE, one patient underwent balloon dilatation and a change in medical therapy, and in two patients, surgical resection was planned. In one patient, DBE was performed for assessment of intestinal lesions due to persistent abdominal pain, and only small erosion near the ileal stoma orifice was found; thus, therapy after DBE was not changed.
Recently, Broide et al[19] evaluated the feasibility and safety of BAE using a NaviAid AB device in 16 children with known IBD (undetermined colitis = 7; CD = 9). In this group, 6 patients with undetermined colitis at baseline were confirmed to have active UC, and 1 patient exhibited mucosal healing. Eight of the 9 patients with known CD were confirmed to have active CD, and 1 patient exhibited mucosal healing.
According to the previously described literature and our previously published algorithm (Figure 2), we suggest the following approach for children with known IBD. DAE is recommended when endoscopic visualization and biopsies of the small intestine are needed to exclude an alternative diagnosis (lymphoma, tuberculosis or carcinoma) or to perform a therapeutic procedure including SB stricture dilation and removal of retained capsule. Endoscopists should keep in mind that in established CD adhesions may limit examination and that active stricturing CD significantly increases the perforation risk[6,18].
The most common complications related to DAE are perforation, bleeding, and pancreatitis, with an overall rate on adult patients of approximately 1%[34,46].
A large retrospective Japanese study identified 29068 patients who underwent diagnostic BAE, reporting 32 cases of perforation (0.11%). Nine hundred forty-two patients underwent a subsequent therapeutic BAE, but no perforations occurred in this group. Univariate logistic regression analysis showed that patients with IBD, irrespective of steroid therapy, had a significantly higher risk of perforation than patients without (8.6-fold and 2.5-fold, respectively)[47]. In most published studies, the perforation rate among CD patients who underwent EBD varied from 0% to 10% of subjects[25,28,33,37-41,44,45], although one small cohort reported a perforation rate of 20%[42] (Table 4). Bleeding after balloon dilation of CD strictures occurs in approximately 2.5% of patients, and it only often requires conservative management[25]. Finally, pancreatitis has been reported to occur in 0.3% of patients, especially after procedures with an anterograde approach[34,48,49]. Adverse event rates for the different types of DAE have been shown to be similar[50].
In paediatric literature, major complications have been reported only for therapeutic procedures. A large retrospective study on 257 DBE procedures in children reported an overall complication rate of 5.4% (10.4% in patients under 10 years)[51]. No major complications related to either diagnostic or therapeutic procedures have been reported in the paediatric IBD setting.
This review analyzed the use of enteroscopy in children and adults with IBD.
Regarding the use of DAE for diagnostic purposes in adult patients, recent ECCO-ESGAR guidelines recommend its use for: (1) Patients with negative endoscopy and suspicion of CD on MRI or CE, if endoscopic and histological diagnostic confirmation is needed; and (2) Patients who need endoscopic intervention in the SB[52].
According to the studies considered in this review, as shown in Table 2, the diagnostic yields of DAE in adult patients with suspected CD and with known CD is 27%-79% and 53%-87%, respectively, and are higher if the indication for DAE is based on previous SB investigations that may identify suspected lesions and guide the choice of insertion route[25,26,28]. Meanwhile, the diagnostic yield of DAE drops drastically when the indication is placed exclusively based on non-specific abdominal symptoms[34]. Similarly the agreement rate between imaging and DAE findings appears to be higher in already diagnosed CD patients than in suspected CD patients (75.6% vs 36.4% respectively)[25]. In published studies a significant impact of DAE on patient management, ranges from 17% to 82%, has been reported with a persistent clinical improvement reaching 83% after a mean follow-up of 13 mo in the study by Mensink et al[36] (Table 2)[25-28,33,35].
Endoscopic ballon dilation overall has a technical success rate from 72% to 100%, and a clinical success rate greater than 60%[25,28,33,37-45], although no standardized definitions of both these short-term outcomes has been clearly stated yet. The majority of studies evaluated clinical success basing on the obstructive symptoms reported by the patients often comparing the clinical improvement before and after dilation[40,45]. The most commonly used definition of technical success was a chance to get the successful inflation of a balloon catheter within the stenotic bowel segment and the subsequently endoscope passage through the dilated segment. Studies considered in this review showed that the mean stricture dilation diameter varied from 12.4 to 17 mm, up to a maximum of 20 mm, and the achievement of a larger dilation diameter seems to be related to a better short-term clinical improvement[45]. The recurrence of obstructive symptoms after EBD has been variously reported (14% to 78.5% of adult subjects) according to the time considered that in most cases was very short (less than fifteen months). However, in two studies the average duration of the observation period after initial EBD was greater than three years[43,44]. They showed the highest recurrence rates of obstructive symptoms (48% and 78.5%, respectively) but most patients underwent successful re-dilation with a high cumulative surgery-free rate (over 78% at three years). Although balloon dilation of CD strictures appears effective in the short term, it needs to take into consideration the high recurrence rate with the possibility of repeated endoscopic procedures and/or surgery. To our best knowledge, no RCT comparing surgery vs balloon dilation has been conducted. As suggested by the ECCO guidelines[53], EBD or surgery are both suitable treatment options for patients with short (< 5 cm) strictures of the terminal ileum in CD, and the choice of treatment depends on local expertise and patient choice (very low level of evidence). Overall, DAE is a relatively safe procedure if it is performed in expert hands. However, needs deep sedation with the presence of anesthesiologist, may require a bi-directional approach, and has a high complication rate, making this technique not very widespread. Moreover, in children with CD EBD it could delay or avoid invasive surgery to dilate the stenosis and potentially positively affect the natural history of chronic disease all the more important the more it is in childhood.
The present review underlines several limitations related to the studies currently available in the literature. First, most of them are retrospective and include a small number of patients. Second, the studies are often extremely heterogeneous in terms of patient selection, including IBD and non-IBD patients. Third, there are no standardized definitions of the principal outcome measures, such as technical success and clinical efficacy. Fourth, observational period after therapeutic endoscopic procedures, such as EBD, are often too short, not allowing a long-term evaluation. Fifth, into the manuscripts some results are not always clearly stated but have been calculated on the basis of results by the authors of the review.
In conclusion, enteroscopy seems a promising technique especially in patients with suspected isolated SB-CD and inconclusive results from conventional studies (including ileocolonoscopy and radiological cross-sectional imaging) in whom histological diagnosis would alter patient management. Moreover, DAE could have many roles in patients with known CD, in terms of adjustment of medical therapy to obtain more lasting and deep clinical improvement, concomitant diagnosis and treatment of stenosing complications and accurate localization of lesions to allow targeted surgical intervention. Because mucosal healing is increasingly becoming a goal of therapy in CD, DBE may have a role in assessment response to therapy in the future in select cases. More standardized and wider studies are needed to confirm these evidences.
We thank Altobello Leo and Di Maio Adele for their tireless work to improve the care of pediatric patients.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Hijaz NM S-Editor: Huang P L-Editor: A P-Editor: Ma YJ
| 1. | Fong SC, Irving PM. Distinct management issues with Crohn's disease of the small intestine. Curr Opin Gastroenterol. 2015;31:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 2. | Sauer CG, Kugathasan S. Pediatric inflammatory bowel disease: highlighting pediatric differences in IBD. Med Clin North Am. 2010;94:35-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 3. | Hall B, Holleran G, McNamara D. Small bowel Crohn's disease: an emerging disease phenotype? Dig Dis. 2015;33:42-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 4. | Peyrin-Biroulet L, Loftus EV Jr, Colombel JF, Sandborn WJ. The natural history of adult Crohn's disease in population-based cohorts. Am J Gastroenterol. 2010;105:289-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 654] [Cited by in RCA: 756] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 5. | Pimentel M, Chang M, Chow EJ, Tabibzadeh S, Kirit-Kiriak V, Targan SR, Lin HC. Identification of a prodromal period in Crohn's disease but not ulcerative colitis. Am J Gastroenterol. 2000;95:3458-3462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 95] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Di Nardo G, de Ridder L, Oliva S, Casciani E, Escher JC, Cucchiara S. Enteroscopy in paediatric Crohn's disease. Dig Liver Dis. 2013;45:351-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | Chouraki V, Savoye G, Dauchet L, Vernier-Massouille G, Dupas JL, Merle V, Laberenne JE, Salomez JL, Lerebours E, Turck D, Cortot A, Gower-Rousseau C, Colombel JF. The changing pattern of Crohn's disease incidence in northern France: a continuing increase in the 10- to 19-year-old age bracket (1988-2007). Aliment Pharmacol Ther. 2011;33:1133-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 131] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 8. | Leighton JA, Pasha SF. Inflammatory Disorders of the Small Bowel. Gastrointest Endosc Clin N Am. 2017;27:63-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Cuffari C, Dubinsky M, Darbari A, Sena L, Baldassano R. Crohn's jejunoileitis: the pediatrician's perspective on diagnosis and management. Inflamm Bowel Dis. 2005;11:696-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Cullen G, Donnellan F, Doherty GA, Smith M, Cheifetz AS. Evaluation of the small bowel in inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2013;7:239-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Ou G, Enns R. Capsule Endoscopy and Small Bowel Enteroscopy: Have They Rendered the Radiologist Obsolete? Gastrointest Endosc Clin N Am. 2019;29:471-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 12. | Pennazio M, Spada C, Eliakim R, Keuchel M, May A, Mulder CJ, Rondonotti E, Adler SN, Albert J, Baltes P, Barbaro F, Cellier C, Charton JP, Delvaux M, Despott EJ, Domagk D, Klein A, McAlindon M, Rosa B, Rowse G, Sanders DS, Saurin JC, Sidhu R, Dumonceau JM, Hassan C, Gralnek IM. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2015;47:352-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 482] [Cited by in RCA: 560] [Article Influence: 56.0] [Reference Citation Analysis (1)] |
| 13. | Di Nardo G, Aloi M, Oliva S, Civitelli F, Casciani E, Cucchiara S. Investigation of small bowel in pediatric Crohn's disease. Inflamm Bowel Dis. 2012;18:1760-1776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 14. | Pennazio M, Venezia L, Cortegoso Valdivia P, Rondonotti E. Device-assisted enteroscopy: An update on techniques, clinical indications and safety. Dig Liver Dis. 2019;51:934-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Schneider M, Höllerich J, Beyna T. Device-assisted enteroscopy: A review of available techniques and upcoming new technologies. World J Gastroenterol. 2019;25:3538-3545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 38] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 16. | Kim J. Training in Endoscopy: Enteroscopy. Clin Endosc. 2017;50:328-333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 17. | ASGE Technology Committee, Chauhan SS, Manfredi MA, Abu Dayyeh BK, Enestvedt BK, Fujii-Lau LL, Komanduri S, Konda V, Maple JT, Murad FM, Pannala R, Thosani NC, Banerjee S. Enteroscopy. Gastrointest Endosc. 2015;82:975-990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 18. | Di Nardo G, Calabrese C, Conti Nibali R, De Matteis A, Casciani E, Martemucci L, Pagliaro G, Pagano N. Enteroscopy in children. United European Gastroenterol J. 2018;6:961-969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 19. | Broide E, Shalem T, Richter V, Matalon S, Shirin H. The Safety and Feasibility of a New Through-the-scope Balloon-assisted Enteroscopy in Children. J Pediatr Gastroenterol Nutr. 2020;71:e6-e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Mans L, Arvanitakis M, Neuhaus H, Devière J. Motorized Spiral Enteroscopy for Occult Bleeding. Dig Dis. 2018;36:325-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 21. | Rahmi G, Samaha E, Vahedi K, Ponchon T, Fumex F, Filoche B, Gay G, Delvaux M, Lorenceau-Savale C, Malamut G, Canard JM, Chatellier G, Cellier C. Multicenter comparison of double-balloon enteroscopy and spiral enteroscopy. J Gastroenterol Hepatol. 2013;28:992-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 22. | Yamamoto H, Ogata H, Matsumoto T, Ohmiya N, Ohtsuka K, Watanabe K, Yano T, Matsui T, Higuchi K, Nakamura T, Fujimoto K. Clinical Practice Guideline for Enteroscopy. Dig Endosc. 2017;29:519-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 118] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 23. | Li X, Zhao YJ, Dai J, Li XB, Xue HB, Zhang Y, Xiong GS, Ohtsuka K, Gao YJ, Liu Q, Song Y, Fang JY, Ge ZZ. Carbon dioxide insufflation improves the intubation depth and total enteroscopy rate in single-balloon enteroscopy: a randomised, controlled, double-blind trial. Gut. 2014;63:1560-1565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 24. | Schulz C, Mönkemüller K, Salheiser M, Bellutti M, Schütte K, Malfertheiner P. Double-balloon enteroscopy in the diagnosis of suspected isolated Crohn's disease of the small bowel. Dig Endosc. 2014;26:236-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Navaneethan U, Vargo JJ, Menon KV, Sanaka MR, Tsai CJ. Impact of balloon-assisted enteroscopy on the diagnosis and management of suspected and established small-bowel Crohn's disease. Endosc Int Open. 2014;2:E201-E206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Rahman A, Ross A, Leighton JA, Schembre D, Gerson L, Lo SK, Waxman I, Dye C, Semrad C. Double-balloon enteroscopy in Crohn's disease: findings and impact on management in a multicenter retrospective study. Gastrointest Endosc. 2015;82:102-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 27. | Tun GS, Rattehalli D, Sanders DS, McAlindon ME, Drew K, Sidhu R. Clinical utility of double-balloon enteroscopy in suspected Crohn's disease: a single-centre experience. Eur J Gastroenterol Hepatol. 2016;28:820-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Holleran G, Valerii G, Tortora A, Scaldaferri F, Conti S, Amato A, Gasbarrini A, Costamagna G, Riccioni ME. The use of single balloon enteroscopy in Crohn's disease and its impact on clinical outcome. Scand J Gastroenterol. 2018;53:925-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 29. | Di Nardo G, Oliva S, Aloi M, Rossi P, Casciani E, Masselli G, Ferrari F, Mallardo S, Stronati L, Cucchiara S. Usefulness of single-balloon enteroscopy in pediatric Crohn's disease. Gastrointest Endosc. 2012;75:80-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 30. | de Ridder L, Mensink PB, Lequin MH, Aktas H, de Krijger RR, van der Woude CJ, Escher JC. Single-balloon enteroscopy, magnetic resonance enterography, and abdominal US useful for evaluation of small-bowel disease in children with (suspected) Crohn's disease. Gastrointest Endosc. 2012;75:87-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 31. | Urs AN, Martinelli M, Rao P, Thomson MA. Diagnostic and therapeutic utility of double-balloon enteroscopy in children. J Pediatr Gastroenterol Nutr. 2014;58:204-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Uchida K, Yoshiyama S, Inoue M, Koike Y, Yasuda H, Fujikawa H, Okita Y, Araki T, Tanaka K, Kusunoki M. Double balloon enteroscopy for pediatric inflammatory bowel disease. Pediatr Int. 2012;54:806-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 33. | Kondo J, Iijima H, Abe T, Komori M, Hiyama S, Ito T, Nakama A, Tominaga K, Kubo M, Suzuki K, Iwanaga Y, Ebara R, Takeda A, Tsuji S, Nishida T, Tsutsui S, Tsujii M, Hayashi N. Roles of double-balloon endoscopy in the diagnosis and treatment of Crohn's disease: a multicenter experience. J Gastroenterol. 2010;45:713-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 34. | Möschler O, May A, Müller MK, Ell C; German DBE Study Group. Complications in and performance of double-balloon enteroscopy (DBE): results from a large prospective DBE database in Germany. Endoscopy. 2011;43:484-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 116] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 35. | Christian KE, Kapoor K, Goldberg EM. Performance characteristics of retrograde single-balloon endoscopy: A single center experience. World J Gastrointest Endosc. 2016;8:501-507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 36. | Mensink PB, Groenen MJ, van Buuren HR, Kuipers EJ, van der Woude CJ. Double-balloon enteroscopy in Crohn's disease patients suspected of small bowel activity: findings and clinical impact. J Gastroenterol. 2009;44:271-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 37. | Fukumoto A, Tanaka S, Yamamoto H, Yao T, Matsui T, Iida M, Goto H, Sakamoto C, Chiba T, Sugano K. Diagnosis and treatment of small-bowel stricture by double balloon endoscopy. Gastrointest Endosc. 2007;66 Suppl 3:S108-S112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 38. | Pohl J, May A, Nachbar L, Ell C. Diagnostic and therapeutic yield of push-and-pull enteroscopy for symptomatic small bowel Crohn's disease strictures. Eur J Gastroenterol Hepatol. 2007;19:529-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 69] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 39. | Ohmiya N, Arakawa D, Nakamura M, Honda W, Shirai O, Taguchi A, Itoh A, Hirooka Y, Niwa Y, Maeda O, Ando T, Goto H. Small-bowel obstruction: diagnostic comparison between double-balloon endoscopy and fluoroscopic enteroclysis, and the outcome of enteroscopic treatment. Gastrointest Endosc. 2009;69:84-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 40. | Despott EJ, Gupta A, Burling D, Tripoli E, Konieczko K, Hart A, Fraser C. Effective dilation of small-bowel strictures by double-balloon enteroscopy in patients with symptomatic Crohn's disease (with video). Gastrointest Endosc. 2009;70:1030-1036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 92] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 41. | Hirai F, Beppu T, Sou S, Seki T, Yao K, Matsui T. Endoscopic balloon dilatation using double-balloon endoscopy is a useful and safe treatment for small intestinal strictures in Crohn's disease. Dig Endosc. 2010;22:200-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 42. | Gill RS, Kaffes AJ. Small bowel stricture characterization and outcomes of dilatation by double-balloon enteroscopy: a single-centre experience. Therap Adv Gastroenterol. 2014;7:108-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 43. | Hirai F, Beppu T, Takatsu N, Yano Y, Ninomiya K, Ono Y, Hisabe T, Matsui T. Long-term outcome of endoscopic balloon dilation for small bowel strictures in patients with Crohn's disease. Dig Endosc. 2014;26:545-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 44. | Sunada K, Shinozaki S, Nagayama M, Yano T, Takezawa T, Ino Y, Sakamoto H, Miura Y, Hayashi Y, Sato H, Lefor AK, Yamamoto H. Long-term Outcomes in Patients with Small Intestinal Strictures Secondary to Crohn's Disease After Double-balloon Endoscopy-assisted Balloon Dilation. Inflamm Bowel Dis. 2016;22:380-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 45. | Hirai F, Andoh A, Ueno F, Watanabe K, Ohmiya N, Nakase H, Kato S, Esaki M, Endo Y, Yamamoto H, Matsui T, Iida M, Hibi T, Watanabe M, Suzuki Y, Matsumoto T. Efficacy of Endoscopic Balloon Dilation for Small Bowel Strictures in Patients With Crohn's Disease: A Nationwide, Multi-centre, Open-label, Prospective Cohort Study. J Crohns Colitis. 2018;12:394-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 46. | Rondonotti E, Sunada K, Yano T, Paggi S, Yamamoto H. Double-balloon endoscopy in clinical practice: where are we now? Dig Endosc. 2012;24:209-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 47. | Odagiri H, Matsui H, Fushimi K, Kaise M, Yasunaga H. Factors associated with perforation related to diagnostic balloon-assisted enteroscopy: analysis of a national inpatient database in Japan. Endoscopy. 2015;47:143-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 48. | Mensink PB, Haringsma J, Kucharzik T, Cellier C, Pérez-Cuadrado E, Mönkemüller K, Gasbarrini A, Kaffes AJ, Nakamura K, Yen HH, Yamamoto H. Complications of double balloon enteroscopy: a multicenter survey. Endoscopy. 2007;39:613-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 241] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 49. | Gerson LB, Tokar J, Chiorean M, Lo S, Decker GA, Cave D, Bouhaidar D, Mishkin D, Dye C, Haluszka O, Leighton JA, Zfass A, Semrad C. Complications associated with double balloon enteroscopy at nine US centers. Clin Gastroenterol Hepatol 2009; 7: 1177-1182, 1182.e1-1182. e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 95] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 50. | Rondonotti E, Spada C, Adler S, May A, Despott EJ, Koulaouzidis A, Panter S, Domagk D, Fernandez-Urien I, Rahmi G, Riccioni ME, van Hooft JE, Hassan C, Pennazio M. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Technical Review. Endoscopy. 2018;50:423-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 290] [Article Influence: 41.4] [Reference Citation Analysis (0)] |
| 51. | Yokoyama K, Yano T, Kumagai H, Mizuta K, Ono S, Imagawa T, Yamamoto H, Yamagata T. Double-balloon Enteroscopy for Pediatric Patients: Evaluation of Safety and Efficacy in 257 Cases. J Pediatr Gastroenterol Nutr. 2016;63:34-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 52. | Maaser C, Sturm A, Vavricka SR, Kucharzik T, Fiorino G, Annese V, Calabrese E, Baumgart DC, Bettenworth D, Borralho Nunes P, Burisch J, Castiglione F, Eliakim R, Ellul P, González-Lama Y, Gordon H, Halligan S, Katsanos K, Kopylov U, Kotze PG, Krustinš E, Laghi A, Limdi JK, Rieder F, Rimola J, Taylor SA, Tolan D, van Rheenen P, Verstockt B, Stoker J; European Crohn’s and Colitis Organisation [ECCO] and the European Society of Gastrointestinal and Abdominal Radiology [ESGAR]. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis. 2019;13:144-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1242] [Cited by in RCA: 1171] [Article Influence: 195.2] [Reference Citation Analysis (0)] |
| 53. | Adamina M, Bonovas S, Raine T, Spinelli A, Warusavitarne J, Armuzzi A, Bachmann O, Bager P, Biancone L, Bokemeyer B, Bossuyt P, Burisch J, Collins P, Doherty G, El-Hussuna A, Ellul P, Fiorino G, Frei-Lanter C, Furfaro F, Gingert C, Gionchetti P, Gisbert JP, Gomollon F, González Lorenzo M, Gordon H, Hlavaty T, Juillerat P, Katsanos K, Kopylov U, Krustins E, Kucharzik T, Lytras T, Maaser C, Magro F, Marshall JK, Myrelid P, Pellino G, Rosa I, Sabino J, Savarino E, Stassen L, Torres J, Uzzan M, Vavricka S, Verstockt B, Zmora O. ECCO Guidelines on Therapeutics in Crohn's Disease: Surgical Treatment. J Crohns Colitis. 2020;14:155-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 367] [Article Influence: 73.4] [Reference Citation Analysis (0)] |