Published online Jul 7, 2020. doi: 10.3748/wjg.v26.i25.3686
Peer-review started: March 9, 2020
First decision: March 31, 2020
Revised: June 4, 2020
Accepted: June 17, 2020
Article in press: June 17, 2020
Published online: July 7, 2020
Processing time: 120 Days and 0.3 Hours
Gastroenteropancreatic neuroendocrine tumours (GEP-NETs) are slow-growing cancers that arise from diffuse endocrine cells in the gastrointestinal tract (GI-NETs) or the pancreas (P-NETs). They are relatively uncommon, accounting for 2% of all gastrointestinal malignancies. The usual treatment options in advanced GEP-NET patients with metastatic disease include chemotherapy, biological therapies, and peptide receptor radionuclide therapy. Understanding the impact of treatment on GEP-NET patients is paramount given the nature of the disease. Health-related quality of life (HRQoL) is increasingly important as a concept reflecting the patients’ perspective in conjunction with the disease presentation, severity and treatment.
To conduct a systematic literature review to identify literature reporting HRQoL data in patients with GEP-NETs between January 1985 and November 2019.
The PRISMA guiding principles were applied. MEDLINE, Embase and the Cochrane library were searched. Data extracted from the publications included type of study, patient population data (mid-gut/hind-gut/GI-NET/P-NET), sample size, intervention/comparators, HRQoL instruments, average and data spread of overall and sub-scores, and follow-up time for data collection.
Forty-three publications met the inclusion criteria. The heterogeneous nature of the different study populations was evident; the percentage of female participants ranged between 30%-60%, whilst average age ranged from 53.8 to 67.0 years. Eight studies investigated GI-NET patients only, six studies focused exclusively on P-NET patients and the remaining studies involved both patient populations or did not report the location of the primary tumour. The most commonly used instrument was the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-C30 (n = 28) with consistent results across studies; the GI-NET-specific module Quality of Life Questionnaire-GINET21 was used in six of these studies. A number of randomised trials demonstrated no HRQoL changes between active treatment and placebo arms. The Phase III NETTER-1 study provides the best data available for advanced GEP-NET patients; it shows that peptide receptor radionuclide therapy can significantly improve GEP-NET patients’ HRQoL.
HRQoL instruments offer a means to monitor patients’ general disease condition, disease progression and their physical and mental well-being. Instruments including the commonly used European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-C30 and GINET21 lack, however, validation and a defined minimal clinical important difference specifically for GI-NET and P-NET patients.
Core tip: Gastroenteropancreatic neuroendocrine tumours (GEP-NETs) are slow-growing cancers that arise from diffuse endocrine cells in the gastrointestinal tract and pancreas. An increased emphasis has been placed on health-related quality of life assessment in clinical studies, using reliable and validated patient-reported outcome instruments. Long-term therapeutic options provide symptomatic relief for patients with GEP-NETs, and can slow down or stabilise disease progression, but are not curative. Thus, understanding the impact of the long-term treatment options is particularly important. The aim of this study was to perform a systematic review to assess health-related quality of life focusing on patients with inoperable metastatic GEP-NETs undergoing different treatments in order to uncover areas for future research.
- Citation: Watson C, Tallentire CW, Ramage JK, Srirajaskanthan R, Leeuwenkamp OR, Fountain D. Quality of life in patients with gastroenteropancreatic tumours: A systematic literature review. World J Gastroenterol 2020; 26(25): 3686-3711
- URL: https://www.wjgnet.com/1007-9327/full/v26/i25/3686.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i25.3686
Gastroenteropancreatic tumours (GEP-NETs) account for 2% of all gastrointestinal malignancies, with a United Kingdom age-standardised incidence rate of 8.6/100000/year[1]. GEP-NETs tend to be slow-growing cancers arising from diffuse endocrine cells in the gastrointestinal tract (GI-NETs) or pancreas (P-NETs). GEP-NETs are categorised as “functioning” or “non-functioning”. Functioning tumours cause symptoms due to the hypersecretion of peptides and hormones[2,3], whilst non-functioning tumours have no hormone-related clinical features. Specific symptoms vary by primary tumour location[4,5]. Around 3 in 10 patients with GI-NET develop symptoms of diarrhoea, abdominal pain and flushing due to increased serotonin production[6], known as carcinoid syndrome (CS). P-NETs can produce a variety of hormones, the most common of which are insulin and gastrin. Other abnormal hormone production associated with GEP-NETs include glucagon, vasoactive intestinal peptide, adrenocorticotropic hormone, somatostatin, and parathyroid hormone-related protein[7].
Surgery is the only curative treatment for GEP-NETs and can be performed when tumours are localised and resectable. The majority of GEP-NET patients, however, are diagnosed with metastatic disease requiring systemic treatment[8,9], including chemotherapy, biological therapies, and peptide receptor radionuclide therapy (PRRT)[10]. These long-term therapeutic options provide symptomatic relief, and can slow down or stabilise disease progression, but are not curative. The 5-year survival rate for patients with metastatic GI-NETs is approximately 75% and the 5-year survival rate for patient with metastatic P-NETs is > 60%, whilst the prevalence of GEP-NETs has been on the rise in the recent decades[2,11]. For instance, the median overall time of survival from the date of diagnosis of distant metastatic disease was reported to be 103 mo (8.5 years) in patients with metastatic midgut GI-NETs[12]. It is therefore vital to assess the patient’s health-related quality of life (HRQoL) in general and related to the disease whilst taking treatments which can have a marked impact on well-being.
In oncology, an increased emphasis has been placed on HRQoL assessment as a secondary endpoint in clinical studies, using reliable and thoroughly validated patient-reported outcome (PRO) instruments for which the minimal important difference for improvement/worsening has been defined[13-17]. High-quality information on HRQoL serves a variety of purposes such as the development of targeted interventions, informed decision making about treatment options and allocation of healthcare resources[18]. Indeed, the United States Food and Drug Administration and European Medicines Agency have published guidance on the use of PRO measures in oncology studies emphasising the value of patient perspective when evaluating medicinal products[19,20].
To date, systematic assessments of HRQoL in patients with GEP-NETs have been performed to a limited extent. In the past, where the literature was systematically evaluated, the review focused on the methodological quality of the identified studies[18]. To our knowledge, there has been no systematic literature review published to provide a comprehensive overview of the different treatment options and associated impact on the patients’ HRQoL. The aim of our study was to perform a systematic review to assess HRQoL focusing on patients with inoperable metastatic GEP-NETs undergoing different treatments in order to uncover areas for future research.
This systematic review adheres to the established international guidelines for conducting systematic reviews, such as those provided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses report (PRISMA Statement[21]), and the Cochrane Handbook for Systematic Reviews of Interventions[22]. Methods of analysis and inclusion criteria were defined in advance and outlined in a designated protocol.
An extensive search of MEDLINE, Embase and the Cochrane library was performed by an experienced information specialist to identify relevant studies. We examined articles published in English between January 1985 and November 2019. The protocol outlined the electronic search strategy in full; searches were supplemented by manual searches of relevant review journals.
After de-duplication, article titles and abstracts were independently reviewed by two analysts, with discrepancies resolved via discussion with a third reviewer. Full-text articles were then obtained for records that met the inclusion criteria to re-evaluate eligibility.
Eligibility for this systematic review was defined according to the PICOS[21]. Participants were adult patients (≥ 18 years) with inoperable GEP-NETs. Outcome measures included HRQoL, PROs, utility estimates, validity, and minimally important difference. The study design incorporated studies reporting utility estimates and HRQoL data, including economic evaluations, observational studies, and relevant randomised control trials. Cross-sectional studies were included, as they can provide useful HRQoL data at a specific point in time, although not providing longitudinal changes due to treatment over time. Studies were also required to report at least one relevant outcome for ≥ 15 patients. There were no limits defined in terms of intervention or comparator PICOS elements.
Articles were excluded if patients were diagnosed with neuroendocrine tumours that did not originate in the gastrointestinal tract or the pancreas, and when the HRQoL data for patients with GEP-NETs were not separated by sub-analysis (e.g., the population included patients with lung and/or thyroid neuroendocrine tumours). Studies indexed as case reports, case series, editorials and letters, conference abstracts, systematic reviews and non-human studies were also excluded. Finally, studies where the treatment applied did not target the malignancies specifically, such as psychotherapeutic interventions, were excluded.
A dedicated data extraction sheet was devised to capture all relevant data, including publication details, study design, country, intervention(s), comparator(s), eligibility, population description (including any specific sub-groups), sample size, age (average and range), sex ratio, tumour location, description of HRQoL measure (including valuation technique, method of elicitation, timepoints, follow-up duration, response rates), utility tool assessment (including disease state, timepoint, average utility) and HRQoL assessment (including scale domain where relevant, timepoint, average score, data spread). Disagreements were resolved by discussion between the two reviewers, with third reviewer involvement as required.
The quality and relevance of HRQoL studies were assessed based on the key criteria described by Papaioannou et al[23] and Brazier et al[24], where applicable. Papers reporting insufficient information to assess study methods and populate the data extraction sheet were considered lower quality and were excluded on this basis.
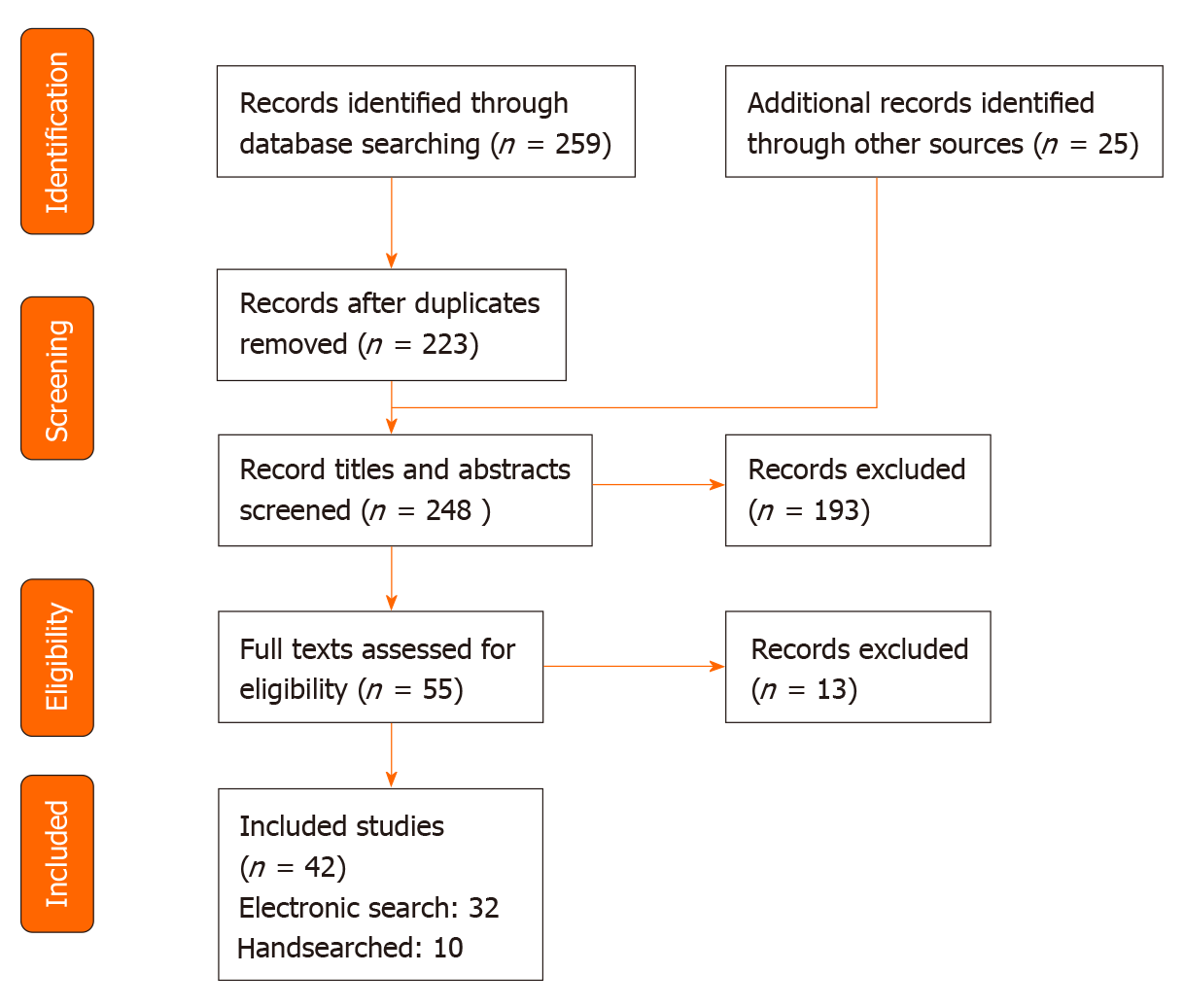
A total of 42 studies published between 1985 and 2019 met the inclusion criteria of this study, of which 32 were found electronically and 10 were found by handsearching the reference lists of relevant articles (Figure 1). Of these studies, 6 can be classed as cross-sectional studies. There was a combined total of 3552 participants in the 42 studies, excluding healthy participants used for comparisons and the duplicate patients taken from the same data, with study sample sizes ranging from 17 to 253 patients. The heterogeneous nature of the different study populations was evident; the percentage of female participants ranged between 30%-60%, whilst average age ranged from 53.8 to 67.0 years (Table 1). In terms of disease location, 8 studies focused on GI-NET patients only and 6 studies focused on P-NET patients; the remaining studies were either a combination of both populations or did not report the specific disease location.
| Ref. | Intervention | Comparator(s) | Study design, “Open” Label / “Blinded” / “Double blinded” | Total sample size | Patient sub-group | Pt sub-group sample size | Sex (% female) | Average age (yr) | Mean/median | Total GI-NET, % | P-NET, % | Functioning /Non-functioning, “function” | Additional information about disease1: “Stage (TNM)” “progressive” “stable”, (“advanced” “metastatic/metastases” “spread”) (“grow” “grade” “differentiated”) |
| Zandee et al[58], 2019 | 177Lu-DOTATATE | NA | Retrospective study | 34 | NA | NA | 50 | 59 | Mean | NA | 100 | Functioning (100%) | Grade 1 or 2 tumours with metastases (stage IV). Metastatic disease sites: Liver (97%), bone (21%), lung (6%) |
| Ramage et al[54], 2019 | Everolimus | NA | Phase IV, open | 48 | All Patients | 46 | 30 | 64.3 | Median | NR | 100 | NR | Advanced and/or metastatic, well-differentiated, grade 1 and 2 |
| Continuing treatment at month 6 | 30 | NR | NR | NR | NR | NR | |||||||
| Ballal | 225Ac-DOTATATE | NA | Prospective study, open | 21 | NA | NA | 57 | 54 | Median | 38 | 57 | NR | Metastatic GEP-NETs with stable disease after completing 177Lu-PRRT (57%) or progressive disease on 177Lu-PRRT (43%). Grades 1 (n = 9), 2 (n = 11), and 3 (n = 1). Metastatic disease sites: Liver (86%), lymph node (71%), bone (33%) |
| Marinova et al[61], 2019 | 177Lu-DOTATATE | NA | Retrospective study | 70 | NA | NA | 44.3 | 64.2 | Mean | 100 | NR | Functioning (83%), non-functioning (17%) | Metastatic. Grade 1 (50%), grade 2 (35.7%), unknown (14.3%). Morphological or clinical progression prior to 1st cycle PRRT (78.6%) |
| Rinke et al[16], 2019 | Long-acting octreotide | Placebo (sodium chloride) | Clinical trial (RCT), Phase IIIb trial, double blind, placebo controlled | 85 | Long-acting octreotide | 42 | 49.4 | 62.4 | Mean | NR | NR | Functioning (39%), non-functioning (61%) | Metastatic, liver metastases (86%) |
| Placebo | 43 | 60.3 | Mean | NR | NR | ||||||||
| NA | NA | NR | NA | NR | NR | ||||||||
| Martini et al[59], 2018 | DOTATATE or 90Y-DOTATOC | General population norms | Retrospective study | 61 | Small intestine NET patients | 37 | 40.5 | 62.8 | Mean | 100 | 0 | NR | No brain metastases; otherwise not defined |
| P-NET patients | 24 | 37.5 | 61.0 | Mean | 0 | 100 | |||||||
| Lewis et al[3], 2018 | Not defined | Patients with CS vs patients without CS | Prospective study | 50 | Without CS (CS) | 25 | 48.0 | 62.0 | Median | 60.0 | 40.0 | NR | Advanced, well-differentiated, with liver metastases |
| With CS (CS) | 25 | 36.0 | 67.0 | Median | 96.0 | 4.0 | NR | ||||||
| Strosberg et al[17], 2018 | 177Lu-DOTATATE | High-dose octreotide | Clinical trial (RCT), phase III, open | 231 | 177Lu-DOTATATE | 117 | NR | NR | NR | 100 | NR | NR | Advanced, progressive, low- or intermediate-grade |
| octreotide LAR | 114 | NR | NR | NR | 100 | NR | |||||||
| Karppinen et al[46], 2018 | NA | Control population | Cross-sectional | 134 | Impaired excretion | 87 | 55 | 66.8 | Mean | 100 | NR | Flushing (31%), diarrhoea (63%) | Locally advanced or metastatic disease (91%), grade 1 (54.1%), grade 2 (45.9%) |
| Cella et al[44], 2018 | Telotristat ethyl | Placebo | Clinical trial (RCT), phase III trial, double blind | 135 | NA | 135 | 48.1 | 63.6 | Mean | NR | NR | NR | Well-differentiated, metastatic NETs and CS inadequately controlled by somatostatin analogues (for ≥ 3 mo) |
| Durable responders (DR) | 48 | 52.1 | 63.4 | Mean | NR | NR | |||||||
| Non-durable responders (DR) | 87 | 46 | 63.6 | Mean | NR | NR | |||||||
| Meng et al[28], 2017 | Somatostatin analogue lanreotide | Placebo | Clinical trial (RCT), phase III trial, double blind | 204 | Lanreotide | 101 | 48 | 62.1 | Mean | NR | NR | Non-functioning | Advanced, well-differentiated or moderately differentiated, somatostatin receptor-positive NETs of grade 1 or 2 |
| Placebo | 103 | NR | NR | ||||||||||
| Overall | 204 | NR | NR | ||||||||||
| Vinik et al[35], 2016 | Sunitinib plus best supportive care | Placebo plus best supportive care | Clinical trial (RCT), phase III trial, double blind | 171 | NA | NA | NR | NR | NR | NA | 100 | NR | Advanced and/or metastatic, pathologically confirmed, well-differentiated P-NETs with disease progression as assessed by RECIST |
| Haugland et al[9], 2016 | None | None | Cross-sectional | 196 | None | NA | 50.5 | 65.0 | Mean | 100.0 | 0.0 | NR | NR |
| Pavel et al[56], 2016 | Everolimus | NA | Clinical trial, Phase IIIb, open, expanded access study | 246 | P-NET | 126 | 46 | 61 | Median | NR | 100 | Functioning (n = 38), non-functioning (n = 88) | Metastatic, well differentiated (n = 81), moderately differentiated (n = 26), poorly differentiated (n = 1), unknown (n = 16), missing (n = 2) |
| Non P-NET | 120 | 50.8 | 66 | Median | NR | 0 | Functioning (n = 66), non-functioning (n = 55) | Metastatic, well differentiated (n = 65), moderately differentiated (n = 34), poorly differentiated (n = 2), unknown (n = 19) | |||||
| Delpassand et al[66], 2014 | 177Lu-DOTATATE | NA | Clinical trial, phase II, open | 37 | Patients with available data participated in quality of life questionnaire | 27 | 56.8 | 63.4 | Mean | 43.2 | 37.8 | NR | Grade 1 and grade 2, disseminated, progressive, somatostatin receptor-positive. Multiple metastases in the liver: grade 2 (n = 4), grade 3 (n = 26), grade 4 (n = 7). Metastatic disease sites: Liver (n = 34), lymph nodes (n = 16), bone (n = 11), pancreas (n = 8), lung (n = 3) |
| Ducreux et al[41], 2014 | Bevacizumab combined with 5-FU/ streptozocin (Mitry et al[40] 2014) | Capecitabine plus bevacizumab | Clinical trial, phase II, open | 34 | NA | NA | 35 | 55 | Median | NR | 100 | Functioning (n = 7): CS (n = 3), Zollinger–Ellison syndrome (n = 1), other (n = 3) | Progressive, metastatic disease. Metastatic disease sites: Liver (n = 33), lymph node (n = 14), peritoneum (n = 2), lung (n = 3), bone (n = 2), other (n = 2) |
| Mitry et al[40], 2014 | Bevacizumab combined with 5-FU/ streptozocin (reported in a companion paper focusing on P-NET patients (Ducreux et al[41], 2014) | Capecitabine plus bevacizumab | Clinical trial, phase II, open | 49 | NA | NA | 47 | 60 | Median | 100 | NA | NR | Progressive, metastatic well-differentiated. Metastatic disease sites: Liver (n = 46), lymph node (n = 24), peritoneum (n = 23), lung (n = 7), bone (n = 7), other (n = 5) |
| Meyer et al[42], 2014 | Capecitabine and streptozocin plus cisplatin (stratified) | Capecitabine and streptozocin (stratified) | Clinical trial (RCT), Phase II | 86 | Capecitabine and streptozocin | 44 | 39 | 57 | Median | 21 | 46.0 | Functioning (n = 31; 36%) | Advanced and/or metastatic. Metastatic disease sites: Local/regional (n = 2), distant (n = 42), liver (included in distant; n = 41) |
| Capecitabine and streptozocin plus cisplatin | 42 | 45 | 59 | Median | 19 | 50.0 | |||||||
| Yadegarfar et al[33], 2013 | Somatostatin analogues or interferon therapy (88 patients) | Peptide-receptor radiotherapy (102 patients), chemotherapy (23 patients), surgery (20 patients) or ablative/ other therapies (20 patients) | Phase IV, open | 253 | P-NET | 70 | NR | NR | NR | NR | NR | Functioning: Secreting 5-hydroxy-indoloacetic acid (n = 111), gastrin (n = 4), glucagon (n = 3), insulin (n = 5), vasoactive intestinal peptide (n = 1). Non-functioning (secreting only chromogranin-A; n = 124) | Any gut-primary with metastases, lung-primary with liver/abdominal metastases and pancreas with/without metastases |
| Casciano et al[36], 2012 | Everolimus (Afinitor) | Sunitinib (Sutent) | Economic analysis of phase III studies | NA | NA | NA | NA | NA | NA | NA | NA | NR | Advanced, progressive |
| Kvols et al[29], 2012 | Pasireotide | Octreotide | Prospective study, phase II, open | 44 | Pasireotide responders | 12 (Baseline), 12 (Month 1), 10 (Month 2), 11 (Month 3) | 44 | 61 | Mean | NR | NR | NR | Advanced, metastatic (symptoms refractory or resistant to octreotide LAR therapy). Metastatic disease sites: Liver (87%), lymph node (n = 16), peritoneum (n = 8), lung (n = 5), bone (n = 3), abdomen (n = 3), pleura (n = 3), retroperitoneal (n = 2), pancreas (n = 2), kidney (n = 1) |
| Pasireotide non responders | 13 (Baseline), 13 (Month 1), 13 (Month 2), 13 (Month 3) | NR | NR | ||||||||||
| Patients who discontinued treatment before 3 mo | 19 (Baseline), 15 (Month 1), 8 (Month 2) | NR | NR | ||||||||||
| Raymond et al[34], 2011 | Sunitinib plus Best Supportive Care | Placebo plus Best Supportive Care | Clinical trial (RCT), phase III, double blind | 171 | sunitinib | 86 | 51 | 56 | Median | NR | 100 | Sunitinib Arm: Functioning: Gastrinoma (n = 9), glucagonoma (n = 3), insulinoma (n = 2), somatostatinoma (n = 1), other, multisecretory, or unknown (n = 10). Non-functioning (n = 42) | Advanced and/or metastatic, progressive, well-differentiated |
| Placebo | 85 | 53 | 57 | Median | NR | 100 | Placebo Arm: Functioning: Gastrinoma (n = 10), glucagonoma (n = 2), insulinoma (n = 2), vasoactive intestinal peptide–secreting tumour (n = 2), other, multisecretory, or unknown (n = 5). Non-functioning (n = 44) | ||||||
| Pezzilli et al[49], 2009 | NA | NA | Prospective study | 51 | NA | NA | 58.8 | 61 | Mean | NR | 100 | NR | Advanced disease (lymph node involvement/liver metastases) (n = 22) |
| Rinke et al[27], 2009 | Octreotide LAR | Placebo | Clinical trial (RCT), phase IIIb, double blind | 85 | Octreotide LAR | 42 | 52.4 | 63.5 | Median | 100 | NR | Octreotide LAR arm: Functioning: CS (n = 17) 40.5% | Well-differentiated, metastatic. Metastatic disease sites: Liver (n = 73), regional lymph node involvement (n = 6) |
| Placebo | 43 | 46.5 | 61 | Median | 100 | NR | Placebo arm: Functioning: CS (n = 16) 37.2% | ||||||
| Korse et al[30], 2009 | Long-acting sandostatin LAR | Short-acting Sandostatin | Clinical trial, phase II | 39 | NA | NA | 51.3 | 61 | Mean | 51.3 | NR | Functioning: CS (n = 39) | Metastatic: Liver metastases (n = 35) |
| van der Horst-Schrivers et al[67] , 2009 | NA | NA | Prospective study | 43 | NA | NA | 37.2 | 60.6 | Mean | NR | NR | NR | Metastatic midgut carcinoid tumours: Liver metastases (n = 37) |
| Haugland et al[47], 2009 | NA | NA | Cross-sectional | 5341 | NET patients | 189 | 50 | 65 | Mean | 100 | NR | NR | GI-NET patients (excluded radical surgery that may have been curative), otherwise undefined |
| General population | 5152 | 48.8 | NR | NR | NA | NA | |||||||
| Larsson and Janson[50], 2008 | Erythropoietin | None | Pilot study | 18 | None | NA | 50.0 | 63.0 | Mean | 100 | NR | NR | NR |
| Kulke et al[55], 2008 | Sunitinib | NA | Clinical trial, phase II, open | 107 | NA | NA | 40.2 | Carcinoid tumour = 58, P-NET= 56 | Median | 37.3 | 61.7 | P-NET: Functioning: Gastrinoma n = 5 (7.6%), insulinoma n = 3 (4.5%), VIPoma n = 2 (3.0%), glucagonoma n = 4 (6.1%), other n = 5 (7.6%). Non-functioning n = 46 (69.7%) | Advanced carcinoid or P-NET |
| Fröjd et al[37] , 2007 | Various included | None | Longitudinal, prospective, comparative study | 59 | T1-T4 successful | 36 | 47.0 | 60.0 | Mean | NR | NR | NR | Metastatic (70% metastatic at start; 78% metastatic at end) |
| Davies et al[68], 2006 | NA | NA | Cross-sectional, open | 50 | Patients | 35 | 42.9 | 60 | Mean | NR | NR | NR | Metastatic |
| Healthcare workers | 15 | NR | NR | NR | NA | NA | |||||||
| Frilling et al[48], 2006 | 90Y-DOTATOC then 177Lu-DOTATOC | NA | Prospective study | 20 | Excluding pt (no 3) with Paraganglioma-neck | 19 | 30 | 53.8 | Median | NR | NR | NR | Advanced, progressive, and metastatic |
| Arnold et al[31], 2005 | Octreotide plus interferon alpha | Octreotide monotherapy | Clinical trial (RCT), open | 114 | Octreotide monotherapy | 51 | 47.1 | 58 | Median | NR | NR | Octreotide: Functioning (n = 23), non-functioning (n = 28). Octreotide plus Interferon-alpha: functioning (n = 24), non-functioning (n = 30) | Metastatic or locally advanced disease without curative therapeutic option |
| Octreotide plus interferon alpha | 54 | 44.4 | 57 | Median | NR | NR | |||||||
| Nonrandomised | 9 | 33.3 | 60 | Median | NR | NR | |||||||
| Teunissen et al[62], 2004 | 177Lu-DOTATOC | None | Clinical trial | 50 | None | NA | 56.0 | 58.3 | Mean | NR | 26.0 | NR | Metastatic |
| Pasieka et al[43], 2004 | I-MIBG | NA | Clinical trial, open | 19 | I-MIBG | 10 | 40 | 59 | Mean | 90 | 10 | NR | Progressive |
| Octreotide | 9 | 55.6 | 55.6 | Mean | 66.6 | 33.3 | |||||||
| Kwekkeboom et al[60], 2003 | Somatostatin analogue 177Lu-DOTATOC | NA | Clinical trial, open | 35 | None | NA | 60 | 54 | Mean | NR | NR | NR | Metastatic |
| Larsson et al[25], 2001 | Interferon, somatostatin analogue, interferon, and a somatostatin | None | Prospective study | 24 | None | NA | 42.0 | 62.0 | Median | 100 | NR | NR | NR |
| O'Toole et al[32], 2000 | Octreotide followed by lanreotide | Lanreotide followed by octreotide | Clinical trial (RTC), double blind | 33 | Patients received octreotide followed by Lanreotide | 16 | 50 | 63 | NR | 62.5 | 0 | ||
| Patients received lanreotide followed by octreotide | 17 | 53 | 64 | NR | 76 | 5.9 | NR | NR | |||||
| Wymenga et al[57], 1999 | Lanreotide prolonged release | NA | Clinical trial, phase II, open | 55 | NA | NA | 49.1 | 59.7 | Mean | NR | 5.5 | NR | Advanced (Stage IV), metastatic. Metastatic disease sites: Lymph nodes (n = 39), distant organs (n = 44) |
| Larsson et al[26], 1999 | Ongoing treatment: interferon and octreotide, other | NA | Cross-sectional | 119 | Patients with carcinoid tumours | 64 | 43.7 | 64 | Median | 53.8 | NA | NR | NR |
| Patients with EPT | 55 | 37.5 | 54 | Median | NR | 46.20 | |||||||
| Overall | 119 | 43.7 | 61 | Median | 53.8 | 46.20 | |||||||
| Larsson et al[38] , 1999 | Interferon and/or a somatostatin analogue | NA | Clinical trial | 99 | NA | NA | 39.4 | 59 | Mean | NR | NR | NR | Stage = not terminal, no other status provided. |
| HADS Anxiety (cases) | 19 | NR | NR | NR | NR | NR | |||||||
| HADS Depression (cases) | 13 | NR | NR | NR | NR | NR | |||||||
| Larsson et al[69], 1998 | Interferon and/or a somatostatin analogue | NA | Cross-sectional | 17 | NA | NA | 47 | 58 | Mean | NR | NR | NR | NR |
Of the total studies, 14 included somatostatin analogues as the intervention, including octreotide (n = 7), lanreotide (n = 3), pasireotide (n = 1), and undefined (n = 3). Such treatments slow down the production of hormones, such as serotonin, and thus reduce the symptoms of CS. In two studies, somatostatin analogues were combined with immunotherapy drugs such as interferon[25,26]. Three studies compared somatostatin analogues with placebos, allocated within the context of a randomised controlled trial[16,27,28]. Five studies used somatostatin analogues as comparators for an alternative somatostatin therapy, 177Lu-DOTATATE or octreotide combined with interferon alpha[17,29-32].
In PRRT, a radioactive substance can be combined with a relevant peptide (or its analogue), such as somatostatin, so that it preferentially binds with a high affinity to the tumour. Nine studies considered PRRT as the intervention: 225Ac-DOTATATE (n = 1), 177Lu-DOTATATE (n = 5), 90Y-DOTATOC (n = 2) and/or 177Lu-DOTATOC (n = 3) (Table 1). 177Lu-DOTATATE was compared with octreotide administered at a high dose in one prospective study[17]. One retrospective study considered PRRT as the comparator[33], with somatostatin analogues or interferon therapy as the intervention.
Everolimus, sunitinib and erythropoietin were studied as interventions in three, three and one studies, respectively. Two studies compared sunitinib with a placebo within the context of randomised control trials[34,35]. Sunitinib was investigated as a comparator of everolimus in an additional study[36]. In some studies, interferon was used within the populations[25,31,33,37,38], but this was not applied to a specific subgroup.
Six studies used chemotherapy as the intervention: These combined bevacizumab with 5-FU/streptozocin (n = 2), capecitabine and streptozocin plus cisplatin (n = 1), and bevacizumab plus capecitabine (n = 1). Two studies followed patients with various ongoing treatments[37,39]. Three studies used chemotherapy treatments as the comparators to chemotherapeutic interventions; two studies combined capecitabine with the anti-angiogenic drug bevacizumab and used the same population data[40,41], whilst one study combined capecitabine and streptozocin as the comparator[42].
Two additional studies identified within the scope of this review focused on meta-iodobenzylguanidine (MIBG) therapy and telotristat ethyl[43,44], respectively. MIBG is a radiometabolic therapy with limited antitumor effect on GEP-NETs. Telotristat ethyl is used to inhibit L-tryptophan hydroxylases TPH-1 and TPH-2 and reduce the production of serotonin, thereby alleviating CS symptoms[44]; this was assessed in the context of a randomised controlled trial, randomised 1:1:1 to receive 250 mg TID, 500 mg TID, and placebo.
In total, 30 studies employed a version of the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ), in which linear transformation is used to standardise the raw score, so that scores range from 0 to 100. The majority of these studies utilised the EORTC QLQ-C30 HRQoL measure (n = 28). One study developed a disease-specific quality of life score questionnaire for patients with GI-NETs to supplement EORTC QLQ-C30. Six studies employed the GI-NET specific module QLQ-GI.NET21, of which one study apparently did not use the HRQoL measure in conjunction with EORTC QLQ-C30[8], thus will not show valid data. Others administered the EQ-5D or EQ-VAS to assess patient utilities and/or the Hospital Anxiety and Depression Scale (HADS) (Although HADS is not strictly a HRQoL measure, it provides insight into the anxiety and depressive symptoms of GEP-NET patients’ underlying HRQoL) in addition to the EORTC measures (n = 5)[45]. Larsson et al[25] reported mean baseline HADS anxiety and depression scores of 4.6 (SD: 4.0) and 3.2 (SD: 2.7), respectively. Four studies applied the Short Form-36 (SF-36) questionnaire[9,46-48], three of which were cross-sectional studies. Other measures used include the SF-12[49], the Functional Assessment of Cancer Therapy-Anaemia[50] and the Functional Assessment of Chronic Illness Therapy-Diarrhoea (FACIT-D). Pezzilli et al[49] reported no significant difference between P-NET patients and a sample of age- and sex-matched subjects in terms of physical domain (44.7 ± 11.0 vs 46.1 ± 9.9, P = 0.610), but a significantly lower mental score in the same patients compared to the normative population (42.4 ± 13.0 vs 48.2 ± 9.8, P = 0.036).
The average HRQoL global health score at baseline and at different timepoints during each trial, obtained by asking patients to rate their overall health, have been reported for the EORTC QLQ-C30 (Table 2). A higher score represents a higher (“better”) level of functioning, including the global health score. This particular tool became the primary focus of this manuscript, as EORTC QLQ-C30 was the single most commonly cited HRQoL dimension (n = 24), with global health score providing a single HRQoL dimension for comparison between studies. The average value for the baseline global health score had a range of 56–70. A higher score for symptoms represents a “worse” level of symptoms. Lowest average score ranges were observed for baseline EORTC QLQ-C30 scores in the following symptoms: nausea and vomiting (4.1–20), insomnia (11.0–38.6), appetite loss (9.2–27.8), constipation (0–24.4) and financial difficulties (6–23.6). Diarrhoea demonstrated a wide range of mean values (16.7–78.3), which can be attributed to the heterogeneity of disease symptoms in GI-NET and P-NET patients, indicating that diarrhoea in GEP-NET patients is multifactorial and very variable. Considering disease location, the average baseline global health scores were comparable, ranging from 56–68 for GI-NETs (4 studies) and 56–67 for P-NETs (5 studies).
| Ref. | Intervention/comparator | Time point | Global health score | Statistical analysis and P values, as presented in the literature |
| Arnold et al[31], 2005 | Octreotide plus interferon alpha | Baseline | 57.9 | 0.04 (between treatment arms at 3 mo) |
| 3 mo | 51.2 | |||
| Octreotide monotherapy | Baseline | 63 | ||
| 3 mo | 74.4 | |||
| Cella et al[44], 2018 | Telotristat ethyl | - | Overall: 55.4; Durable responders: 40; Non-durable responders: 66 | NR (NS) |
| Fröjd et al[37], 2007 | Various (i.e. interferon, octreotide, chemotherapy and other) | Timepoint 1 | 58 | NR |
| Timepoint 2 | 61 | NR | ||
| Timepoint 3 | 58 | NR | ||
| Timepoint 4 | 58 | NR | ||
| van der Horst-Schrivers et al[67], 2009 | NA | - | 61.8 | NR |
| Korse et al[30], 2009 | Long-acting sandostatin LAR | Baseline | 70 | 0.275 (repeated measurement analysis using mixed linear models) |
| 3 mo | 71 | |||
| 6 mo | 67 | |||
| 9 mo | 68 | |||
| 12 mo | 64 | |||
| Larsson et al[50], 2008 | Erythropoietin | Baseline | 56 | NR (NS) |
| 4 mo | 55 | |||
| 8 mo | 66 | |||
| 2 yr | 70 | |||
| Larsson et al[25], 2001 | Interferon, somatostatin analogue, interferon, and a somatostatin. | Baseline | 68 | NR (NS) |
| 3 mo | 59 | |||
| 6 mo | 58 | |||
| 9 mo | 50 | |||
| 12 mo | 63 | |||
| Larsson et al[26], 1999 | Interferon and/or a somatostatin analogue for at least 4 weeks - Five patients were treated with interferon, three with a somatostatin analogue, and nine with a combination | - | 64.71 | NR |
| Larsson et al[38], 1999 | Interferon and/or a somatostatin analogue | - | Overall: 66.7; HADS Anxiety (cases): 46; HADS Anxiety (non-cases): 71.6; HADS Depression (cases): 43; HADS Depression (non-cases): 70.2 | < 0.001 between HADS anxiety “cases” (n = 19) and “non-cases” (n = 80) and 0.001 between HADS depression “cases” (n = 13) and “non-cases” (n = 86) |
| Larsson et al[69], 1998 | Interferon and/or a somatostatin analogue and/or a combination | - | 64.71 | NR |
| Lewis et al[3], 2018 | NA | - | Without CS: 63.0; With CS: 61.7 | 0.04 (between subpopulations) |
| Marinova et al[61], 2019 | 177Lu-DOTATATE | Baseline | 62.6 | |
| 3 mo | 66.7 | < 0.05 (compared to baseline using mixed longitudinal model) | ||
| 6 mo | 69.6 | < 0.01 (compared to baseline using mixed longitudinal model) | ||
| 9 mo | 69.4 | < 0.01 (compared to baseline using mixed longitudinal model) | ||
| Meyer et al[42], 2014 | Capecitabine and streptozocin plus cisplatin. | Baseline | 69.3 | |
| ≤ 9 wk - after 3 × 3 weekly cycles | 52.2 | 0.052 (compared to baseline) | ||
| 6 mo | 56 | 0.68 (compared to baseline) | ||
| Capecitabine and streptozocin | Baseline | 67 | ||
| ≤ 9 wk - after 3 × 3 weekly cycles | 62.2 | 0.5 (compared to baseline) | ||
| 6 mo | 68.9 | 0.75 (compared to baseline) | ||
| Mitry et al[40], 2014 | Bevacizumab plus capecitabine | Baseline | 67 | |
| 6 mo | 63 | NR (NS) | ||
| 12 mo | 71 | NR (NS) | ||
| Pavel et al[56], 2016 | Everolimus | End of treatment (compared to baseline) | P-NET: -3.9; non P-NET: -13 | NR |
| Ramage et al[54], 2019 | Everolimus | 1 mo | FAS Population (paired scores baseline and 1 m): 58 | NR (NS) |
| Continuing tx at 6m (paired scores baseline and 1 m): 56.9 | NR (NS) | |||
| 2 mo | FAS Population (paired scores baseline and 2 m): 57.6 | NR (NS) | ||
| Continuing tx at 6m (paired scores baseline and 2 m): 56.9 | NR (NS) | |||
| 3 mo | FAS Population (paired scores baseline and 3 m): 56.7 | NR (NS) | ||
| Continuing tx at 6m (paired scores baseline and 3 m): 56.3 | NR (NS) | |||
| 4 mo | FAS Population (paired scores baseline and 4 m): 56 | NR (NS) | ||
| Continuing tx at 6 m (paired scores baseline and 4 m): 56.8 | NR (NS) | |||
| 5 mo | FAS Population (paired scores baseline and 5 m): 56.6 | NR (NS) | ||
| Continuing tx at 6 m (paired scores baseline and 5 m): 58.6 | NR (NS) | |||
| 6 mo | FAS Population (paired scores baseline and 6 m): 56.9 | NR (NS) | ||
| Continuing tx at 6 m (paired scores baseline and 6 m): 56.9 | NR (NS) | |||
| Raymond et al[34], 2011 | Sunitinib | Baseline | 67 | |
| "post-baseline" | 62.4 | NR | ||
| Placebo | Baseline | 64 | ||
| "post-baseline" | 61.3 | NR | ||
| Rinke et al[16], 2019 | Long-acting octreotide | Baseline | 64 | |
| Placebo | Baseline | 65.7 | ||
| 12 wk | +4.13 (difference in global health score compared to placebo) | NR | ||
| 24 wk | +5.05 (difference in global health score compared to placebo) | NR | ||
| 36 wk | +1.85 (difference in global health score compared to placebo) | NR | ||
| Strosberg et al[17], 2018 | 177Lu-DOTATATE | Baseline | 67 | NR |
| High-dose octreotide | Baseline | 64 | NR | |
| Teunissen et al[62], 2004 | 177Lu-DOTATOC | Baseline | 69 | |
| 6 wk | 78.2 | < 0.01 (Analysis of variance (two-sided) compared to baseline) | ||
| Vinik et al[35], 2016 | Sunitinib | Baseline | 67 | NR (NS difference between arms) |
| Up to cycle 10 (about 9 mo) | 60.4 | |||
| Placebo | Baseline | 64 | NR (NS difference between arms) | |
| Up to cycle 10 (about 9 mo) | 61.3 | |||
| Wymenga et al[57], 1999 | Lanreotide prolonged release | Baseline | 60.4 | |
| 1 mo | 69.7 | 0.001 (compared to baseline using t tests or Wilcoxon signed rank) | ||
| End of treatment | 65.5 | NR (NS) | ||
| Yadegarfar et al[33], 2013 | Somatostatin analogues or interferon therapy | Baseline | 61 | |
| 3 mo | 67 | NR | ||
| 6 mo | 67 | NR | ||
| Zandee et al[58], 2019 | 177Lu-DOTATATE | Baseline | 61.7 | |
| 3 mo (after final PRRT cycle) | 79.5 | 0.002 (compared to baseline using paired t test) |
Whilst the most popular HRQoL measure was EORTC QLQ-C30, studies that employed the QLQ-GI.NET21 module provided domain scores specific to the disease that is the focus of this report, including: endocrine systems, gastrointestinal systems, disease-related worries, bone/muscle pain etc. Table 3 presents the scores given for these domains. The average HRQoL score ranges for body image (13.3–29.1), weight gain (9-27.8) information/communication (1.1–16.0) and treatment-related symptoms (6.5–26.24) showed that these were only minor problems experienced by patients with GI-NETS. Other domains displayed a wide range of scores, e.g. sexual function (28.2–68.4), social function (30.0–72.9) and disease-related worries (37.6–84.2), making these some of the most important problems experienced by many patients. The QLQ-GI.NET21 module has been validated as a responsive tool for assessing HRQoL of patients with GEP-NETs[33], although it is accepted that numbers were too small for a proper validation in patients with P-NETs alone. This is of relevance, as P-NET patients often present with a more aggressive disease and concomitant treatments may be used.
| Ref. | Intervention/ comparator | Time point | Endocrine Symptoms | Gastrointestinal Symptoms | Social function | Disease related worries | Muscle/bone pain symptom | Weight gain | Information/Communication function | Body Image | Sexual function | Treatment related symptoms scale |
| Ramage et al[54], 2019 | Everolimus | Baseline (paired scores with 3 mo) | 14.8 | 24.4 | 44.1 | 51.1 | 35.2 | NR | 1.9 | 25.9 | 37 | NA |
| 3 mo | 12.3 | 20.2 | 42.6 | 44.8 | 35.2 | NR | 4.6 | 24.1 | 42.6 | 22 | ||
| Baseline (paired scores with 6 mo) | 14.4 | 25.8 | 44.8 | 50.9 | 33.3 | NR | 1.1 | 20 | 35.6 | NA | ||
| 6 mo | 12.6 | 21.8 | 34.4 | 43.7 | 30 | NR | 1.1 | 21.1 | 37.8 | 14.1 | ||
| Lewis et al[3], 2018 | NA | Single timepoint (patients without CS) | 16.7 | 18.9 | 60.0 | 56.9 | 41.3 | 18.7 | 10.7 | 15.3 | 60.8 | 17.5 |
| Single timepoint (patients with CS) | 28.4 | 24.0 | 68.4 | 38.7 | 46.7 | 13.3 | 16.0 | 13.3 | 68.4 | 10.1 | ||
| Yadegarfar et al[33], 2013 | Various | Baseline (P-NETs) | 22 | 26 | 39 | 56 | 25 | 11 | 10 | 25 | 32 | 18 |
| 3 mo (P-NETs) | 16 | 18 | 33 | 44 | 31 | 9 | 4 | 21 | 31 | 22 | ||
| 6 mo (P-NETs) | 18 | 22 | 30 | 50 | 32 | 13 | 10 | 19 | 31 | 23 | ||
| Ballal et al[8], 2019 | 225Ac-DOTATATE TAT | Baseline | 21.4 | 40.2 | 64.3 | 61.2 | 38.8 | 25 | NR | 28.8 | 40 | 6.53 |
| End of assessment | 3.57 | 22.8 | 72.9 | 84.2 | 26.8 | 27.8 | NR | 15.6 | 45 | 26.24 | ||
| Strosberg et al[17], 2018 | 177Lu-DOTATATE | Baseline | NR | 22.8 | 33.4 | 43.7 | 29 | NR | 5.4 | 20 | 30.6 | 11.6 |
| High-dose octreotide | Baseline | NR | 23.8 | 37.1 | 43.8 | 34.6 | NR | 12.3 | 20.3 | 28.2 | 11.9 | |
| Cella et al[44], 2018 | Telotristat ethyl | Baseline (Durable responders) | 37.8 | 33.8 | 46.7 | 41.1 | 35 | NR | 5 | 29.1 | 47.2 | 18.1 |
| Baseline (Nondurable responders) | 29.5 | 28.5 | 38.5 | 37.6 | 30.3 | NR | 7 | 26.9 | 32.6 | 12.8 | ||
| Difference in change from baseline between subpopulations | -1.9 | -9.6 | -2.8 | 1.9 | -4.5 | NR | 3.9 | 1 | -1.6 | -3.4 |
In addition, a 10-point change in an EORTC domain score is frequently considered a minimal clinically important difference and, using Kaplan-Meier methodology, time to deterioration (TTD) can be measured using a 10-point decline in analogy with time to event curves[51,52]. Two studies identified in the literature search applied the use of minimal important difference in the change in HRQoL domains as a means of assessing the impact of interventions using TTD, and time until definitive deterioration (TUDD), i.e. ≥ 10 point worsening of a HRQoL domain score from baseline without further improvement[16,17].
The SF-36 measures eight dimensions of HRQoL, where item scores are linearly transformed into scales ranging from 0 to 100, with higher scores indicating better HRQoL. In cross-sectional studies, HRQoL has been shown to be significantly reduced in patients with GEP-NETs compared to the general population in every SF-36 dimension[46,47]: 77.9 vs 70.0 for physical functioning, 65.0 vs 40.2 for role physical, 69.1 vs 64.3 for bodily pain, 74.3 vs 54.8 for general health, 60.9 vs 49.5 for vitality, 83.9 vs 75.5 for social functioning, 77.4 vs 64.4 for role emotional and 80.6 vs 74.9 for mental health. Mean scores for each scale were consistently worse in a sub-population of patients with impaired bowel/bladder function, which was strongly correlated with high scores for diarrhoea[46], a symptom of CS which reduces patient HRQoL. Elsewhere, Haugland et al[9] reported only the physical component (39.6) and the mental component (45.9) scores; these were similar to baseline scores previously reported[53]. Frilling et al[48] reported improved HRQoL (assessed using SF-36) in 50% of patients in their prospective study of 90Y-DOTATOC therapy followed by 177Lu-DOTATOC, but sample size was low (n = 20).
Various studies used other HRQoL tools to evaluate HRQoL in GEP-NET patients; direct comparisons between these studies were limited by the small number of studies using each tool (n ≤ 2) and the heterogenous population and treatment interventions. Pezzilli et al[49] was the only identified publication to use SF-12 in a population of P-NET patients, reporting a good physical HRQoL that was not significantly different to the normative population (44.7 ± 11.0 vs 46.1 ± 9.9, P = 0.610), but a significantly impaired mental component (42.4 ± 13.0 vs 48.2 ± 9.8, P = 0.036). Pezzilli et al[49] was also the only study to employ the Beck Depression Inventory and State Anxiety Inventory Y-1; 8 patients (18.2%) identified with moderate depression, 9 patients (20.5%) with mild moderate depression, and 27 patients (61.4%) with no depression. Anxiety results were similar in the patients and in the normative population. Ramage et al[54] used the EQ-5D-5L (in addition to EORTC QLQ-C30 and GINET21), with no significant difference for the composite health index or self-rated (global) health score noted between baseline and 6-mo treatment with everolimus. The EQ-5D VAS was employed in two studies: Kulke et al[55] examined patients receiving repeated 6-weekly cycles of oral sunitinib, noting no significant difference in VAS scores over the study period, whilst Pavel et al[56] reported a reduced mean VAS from baseline to end of treatment with oral everolimus (10 mg daily, in 28-d cycles) in non-P-NET patients (63.9 ± 19.0 vs 55.3 ± 23.0, respectively) compared to P-NET patients (68.8 ± 19.9 vs 66.5 ± 20.6, respectively). The FACIT-D measure is a symptom-specific measure used by Kvols et al[29] in a Phase II, open-label, multicentre prospective study of self-administered pasireotide. Clinical responders were noted to have improved mean scores at the end of treatment compared to baseline. Kulke et al[55] reported that the mean FACIT-fatigue score remained stable for each treatment cycle of sunitinib with a “modest” score increase during the dosing period. Elsewhere, Larsson et al[50] used the Functional Assessment of Cancer Therapy-Anaemia measure to assess the HRQoL of GEP-NET patients with anaemia being treated with erythropoietin; no significant changes were observed over the study period. Finally, O’Toole et al[32] showed no significant difference in HRQoL scores between lanreotide and octreotide using the Index de Santé Perceptuel de Nottingham measure. These studies provide a basis for future randomised control trials to test whether interventions improve HRQoL for patients with GEP-NETs.
All studies included in this review reported the HRQoL of patients with inoperable tumours; the majority of which described the disease as metastatic and/or advanced (Table 1), and thus the disease could be categorised as stage IV. Out of these studies, only two provided explicit information regarding disease stage for the entire study population[57,58]. Notably, no studies investigated how disease stage and tumour functionality affect HRQoL in GEP-NET patients, with HRQoL data most often presented according to treatment comparator.
Few studies reported a significant difference (P < 0.05) in the patients’ global health score between baseline and after a period of treatment (Table 2).
HRQoL was maintained with few deteriorations in patients receiving long-acting octreotide compared to the deterioration observed in patients who received a placebo[16,27]. Octreotide improved HRQoL scores for the clinically important GEP-NET symptoms such as fatigue, insomnia, diarrhoea and pain; whereas significantly earlier TUDD was observed in the placebo arm for fatigue, pain and insomnia in patients with GI-NETs than those treated with long-acting octreotide (P < 0.05)[16]. An improvement in the EORTC global health score was shown after 3 mo on octreotide but not when octreotide was combined with interferon alpha[31]. Flushing was shown to be reduced significantly following 12 mo of treatment with long-acting octreotide[30]. Further studies are deemed necessary to assess the HRQoL of GEP-NET patients treated with pasireotide, who are refractory or resistant to octreotide; symptom-specific scores and total FACIT-D assessments showed some variability and no clear trends but sample size was small (n = 30)[29].
Another somatostatin analogue shown to bring about significant improvement in the global health score of GEP-NET patients was prolonged-release lanreotide[57], possibly driven by improvements in symptom scales or single-term measures such as fatigue, pain, nausea/vomiting, sleeping problems, appetite, constipation, diarrhoea and financial problems; although dyspnoea became more severe during the 6-mo treatment period. O’Toole et al[32] found no significant HRQoL differences between lanreotide and octreotide, although most patients prefer lanreotide due to its simplified mode of administration. However, examining evidence from the CLARINET study, and mapping EORTC QLQ-C30 to EQ-5D-based utility values, no significant differences in HRQoL were noted between the lanreotide and placebo arms[28].
PRRT treatment for GEP-NETs has been shown to result in stable HRQoL scores during and after a 3-mo treatment period, compared to baseline levels using the EORTC QLQ-C30 measure[59,60]. In more recent studies, treatment with 177Lu-DOTATATE was shown to be a safe and effective therapy in the treatment of functioning P-NETs[58] leading to a significant increase in the global health score observed at follow-up compared to baseline (79.5 vs 61.7, P = 0.002). Furthermore, significant improvements in the physical functioning, role functioning, emotional functioning, and social functioning were reported. Of all symptom scales, only “fatigue” was found to have significantly decreased (27.3 vs 17.2, P = 0.02). These results are consistent with a recent retrospective analysis study conducted by Marinova[61]; HRQoL was significantly improved in GI-NET patients receiving 177Lu-DOTATATE compared to baseline, revealing an increased global health score (3 mo after 1st, 2nd, and 3rd treatment cycle P = 0.049, P = 0.004, and P = 0.041, respectively).
In the Phase III NETTER-1 study, 231 patients were recruited with 117 randomised to the 177Lu-DOTATATE arm (200 mCi every 8 wk for four treatments) and 114 to the high-dose octreotide long-acting repeatable arm (60 mg every 4 wk)[17]. HRQoL analysis, conducted on an intention-to-treat basis, demonstrated that 177Lu-DOTATATE provides a significant and clinically robust HRQoL benefit for patients with progressive GI-NETs (compared to high-dose octreotide). TTD, defined as the time from randomisation to first HRQoL deterioration of ≥ 10 points from baseline score, was significantly longer in the 177Lu-DOTATATE arm (P < 0.001); the hazard ratio was 0.406 for global health status, 0.518 for physical functioning, 0.580 for role functioning, 0.621 for fatigue, 0.566 for pain, 0.473 for diarrhoea, 0.425 for body image and 0.572 for disease-related worries. Differences in median TTD were clinically significant in several domains: 28.8 mo vs 6.1 mo for global health status, and 25.2 mo vs 11.5 mo for physical functioning. Significant improvements in TUDD were also observed for the global health score (P < 0.001), as well as for physical functioning (P = 0.002), role functioning (P < 0.001), emotional functioning (P = 0.020), social functioning (P = 0.001), pain (P = 0.002), insomnia (P = 0.026), appetite loss (P = 0.009), constipation (P = 0.042), diarrhoea (P = 0.001), GI scale (P = 0.005), treatment scale (P = 0.002), social function scale (P = 0.011), disease-related worries scale (P = 0.001), and body image (P = 0.002).
Other PRRT interventions included the use of 177Lu-DOTATOC[62], 90Y-DOTATOC[59] and 225Ac-DOTATATE[8]. 177Lu-DOTATOC treatment improved functional scales and most single item scores in GEP-NET patients, whilst symptom scale scores for fatigue and pain decreased significantly from 31.9 to 22.8 (P < 0.01) and 23.3 to 15.0 (P < 0.05), respectively[62]. Martini et al[59] examined HRQoL following 4–6 cycles of 177Lu-DOTATATE or 90Y-DOTATOC. The most significant domain change for GI-NETs was noted for diarrhoea (-16.3 points, P = 0.008). Compared to 90Y-DOTATOC, 177Lu-DOTATATE demonstrated a reduced score for fatigue (-27.7 points, P = 0.020) and better physical functioning (+22.4 points, P = 0.050), cognitive functioning (+23.1 points, P = 0.003) and global health score (+17.3 points, P = 0.029) in P-NET patients. 225Ac-DOTATATE therapy has the potential to overcome the resistance of GEP-NET to 177Lu-DOTATATE beta therapy and promote remission with minimal or transient toxicity, whilst reducing endocrine (P = 0.001) and gastrointestinal symptoms (P < 0.001), although treatment-related symptoms were worsened by the end of the assessment (P = 0.001)[8].
A significant difference in the EORTC QLQ-C30 physical functioning scale was noted by Ramage et al[54] following 6-mo treatment with everolimus, but no other significant differences were noted during this treatment period; in line with what was also shown by analysis conducted in conjunction with the RADIANT trial[56]. Likewise, sunitinib has no significant impact on global health score of EORTC QLQ-C30, but it has been shown to improve the HRQoL symptom scores of insomnia and diarrhoea compared to placebo in P-NETs[34,35]. In another study[55], there was no significant difference in EQ-VAS scores over 6 treatment cycles of sunitinib. There was no significant impact on the HRQoL of patients with GI-NETs when treated with erythropoietin for the anaemia associated with interferon therapy[50].
Chemotherapy in the BETTER trial was chosen based on NET location; GI-NETs were treated with capecitabine and bevacizumab, whilst P-NETs patients received 5-fluorouracil/streptozocin with bevacizumab. Differences in the EORTC QLQ-C30 global health score between baseline, 3-mo, 6-mo and 12-mo follow-up assessments were not significant[40,41]. The NET01 trial suggested that the addition of cisplatin to the combined capecitabine and streptozocin regimen worsened HRQoL[42]. After three cycles, the mean global health score was 67.0 vs 62.2 (P = 0.5) in the capecitabine and streptozocin arm and 69.3 vs 52.2 (P = 0.052) in the capecitabine and streptozocin plus cisplatin arm. After 6 cycles, mean global health score remained virtually unchanged in the capecitabine and streptozocin arm from 66.7 vs 68.9 (P = 0.75) and 59.8 vs 56.0 (P = 0.68) in the capecitabine and streptozocin plus cisplatin arm. All three studies were based on small HRQoL sample sizes (less than 100).
Of the remaining treatments identified within the scope of this study, the HRQoL was improved or remained the same in all GEP-NET patients treated with MIBG therapy. Three studies used more than one treatment in the populations studied without breaking down HRQoL results into treatment categories[26,37,38]. Using HADS, Larsson et al[25] noted a lower anxiety rating at 12 mo (P < 0.05) following treatment with interferon, somatostatin analogue or a combination of both, whilst the depression score was significantly higher at 9 mo (P < 0.01) when compared to baseline. The study by Fröjd et al[37] suggested a slight increase in HADS anxiety over time (timepoints unspecified) in a longitudinal study of octreotide and/or interferon or chemotherapy or no treatment, but sample size was small and significance was not reported. Finally, telotristat ethyl therapy was not shown to be associated with any measurable short-term HRQoL benefits when assessed using the EORTC QLQ-30 measure, beyond diarrhoea[44].
Given the indolent nature and slow progression of disease in most GEP-NET patients, assessment of HRQoL is vital for monitoring impact of treatment. The lack of conclusive data on outcomes in GEP-NETs can be attributed, in part, to the small sample size of clinical trials involving GEP-NETs that makes it difficult to capture clinically meaningful changes or a significant benefit in terms of HRQoL[6]. Moreover, the work by Martini et al[18] showed that a transfer of HRQoL results into clinical practice is hindered by the scarcity of robust studies and the quality of HRQoL assessment[18]. To our knowledge, our study is the first to report a systematic review investigating the impact of treatments on the HRQoL of GEP-NET patients and the development status of instruments used in terms of validation and minimal important difference.
Martini et al[18] performed a systematic review examining methodological robustness of HRQoL studies of patients with GEP-NETs. They concluded that heterogeneity and limited methodological quality of considered studies did not permit them to draw conclusions regarding the impact of HRQoL in clinical practice. In agreement, our review confirmed heterogeneity of studies and patient characteristics, including variety in intervention or comparator treatment, disease severity and location of primary tumour, age, gender and other symptoms or comorbidities. Sample size for studies considered eligible for inclusion ranged from 17 to 253 patients, with a median of 57, highlighting the usual small number of study participants. A similar median value of 51 (range 9–663 patients) was reported by Martini et al[18]. Further, where comparator groups were used, HRQoL scores were sometimes reported for the entire study population and not for the respective treatment groups in the study[29,33,40,41,44]. Due to the wide range of comparators identified and the significant heterogeneity across the studies included in this review, which would violate the assumptions of meta-analysis, HRQoL related to various GEP-NET treatments was assessed in a qualitative manner rather than quantitative (e.g. meta-analysis).
In line with Martini et al[18], we identified the well-validated EORTC QLQ-C30 as the most commonly used HRQoL instrument in GEP-NET patients. The EORTC instrument has been used in 30 out of 43 published studies; however, validation and definition of minimal important difference have not been reported to date specifically for GEP-NET patients. The EORTC QLQ-GINET21 module (n = 6) and the SF-12/36 tools (n = 5) have occasionally been used, with other instruments used in two or less publications.
Where change in the EORTC QLQ-C30 global health score could be calculated from baseline scores (Table 2), six studies demonstrated a trend towards an improved score, three showed a decline, with the remaining studies presenting mixed or inconsistent responses depending on intervention and/or follow-up time point. However, results should be interpreted with caution for the reasons given above. The mixed HRQoL trends are comparable to those reported by Jimenez-Fonseca et al[63], who highlighted the existing evidence for improved treatment efficacy, safety or toxicity for a range of therapies, including somatostatin analogues, sunitinib and everolimus, but with mixed HRQoL response. Jimenez-Fonseca et al[63] suggested the generic EORTC QLQ-C30 has not been developed specifically for GEP-NET patients and, therefore, may lack the sensitivity to detect subtle HRQoL changes in association with disease items that are relevant to GEP-NET patients.
Given the potentially long treatment duration in GEP-NET patients, one challenge of HRQoL analysis is taking the potential occurrence of a response shift into consideration. This can occur as self-assessment of HRQoL is inherently subjective; patients can adapt to their disease and the treatment toxicities resulting in changing standards of HRQoL over time[51]. Thus, the choice of the reference score to qualify a change such as deterioration is a major concern. With this in mind, it is difficult to determine the change in HRQoL over time in most studies identified in this systematic review. One approach that deals with response shift effect as a concept is evaluation of TTD, making use of minimal important difference as a criterion for signalling deterioration. In the Phase III trial NETTER-1, it was shown that adjusting for significant covariates (HRQoL baseline values, OctreoScan maximum tumour uptake score, and/or age), the impact of treatment remained statistically significant for global health score, physical functioning, diarrhoea, and body image domains[17]. Osoba et al[52] rated patients’ perceived change in EORTC QLQ-C30 in breast cancer or small-cell lung cancer patients, concluding a “moderate” change equated to a 10–20 change in mean scores. Use of a minimal important difference of at least 10 points as a criterion for deterioration yielded TTD curves with a time course similar to that of the curves for centrally reviewed radiological progression[17]. It should be noted, however, that the “minimal important difference” was not defined specifically for patients with midgut NETs participating in the NETTER-1 study. Moreover, the TTD and TUDD criterion of a drop in ≥ 10 points in a domain score was used to show more than half of the EORTC QLQ-30 domains improved for patients treated with 177Lu-DOTATATE compared to a control arm; the same TUDD criterion was also applied by Rinke et al[16], who showed long-acting octreotide significantly extended TUDD for fatigue, pain and insomnia HRQoL domains compared to a placebo.
Treatment of GEP-NETs with 177Lu-DOTATATE consistently improved global health score compared to baseline[58,61,62] and increased TTD. The Phase III NETTER-1 study provides the best information available regarding HRQoL in patients with advanced progressive GEP-NETs not only in terms of benefit but also in terms of TTD[17]. However, the minimal important difference (and clinically meaningful thresholds) for improvement/worsening has not been defined yet for the EORTC QLQ-C30 specifically in GEP-NET patients, although it has for a number of other common cancers[64,65]. It is conceivable that the abbreviated version of the EORTC QLQ-C30 (possibly combined with GINET21 and computer adaptive testing) can be developed especially for use in clinical practice. Thus, there is scope for further research in this area and exploration of HRQoL as a potential tool for monitoring disease progression. Longitudinal studies with a long-term follow-up and involving clinical practice patients may provide further insight into the potential role of HRQoL in patient monitoring and management.
In conclusion, study and patient heterogeneity, small sample sizes and limited methodological quality in terms of assessing HRQoL has impaired definitive statements being made regarding the impact of the various treatment options for GEP-NETs in terms of HRQoL[66-69]. A number of randomised trials demonstrated only small or no HRQoL changes between active treatment and placebo arms. The Phase III NETTER-1 is the only study to have shown more significant and robust differences between active and placebo groups. It demonstrated that PRRT can delay the time to worsening of HRQoL in GEP-NET patients. However, validation of QLQ-C30 and GINET21 specifically for GI-NET and P-NET patients, and definition of minimal important difference and clinical thresholds, are still lacking for these patients. As such, further research is necessary in order to offer a benchmark for interpreting the clinical importance of differences in HRQoL domain scores between groups or changes in these scores over time in conjunction with different treatment modalities.
Gastroenteropancreatic neuroendocrine tumours (GEP-NETs) are slow-growing cancers that arise from diffuse endocrine cells in the gastrointestinal tract (GI-NETs) and pancreas (P-NETs). The majority of patients who are diagnosed with GEP-NETs have metastatic disease. Despite this, systemic therapies provide a 5-year survival rate of about 75% for patients with GI-NETs and > 60% for patients with P-NETs.
Health related quality of life (HRQoL) is an increasingly important concept, reflecting the patients’ perspective in conjunction with the disease presentation, severity, and treatment. Although not curative, long-term therapeutic options are available for patients with GEP-NETs to provide symptomatic relief, and these can even slow down and stabilise disease for multiple years. Since patients can live many years by managing their disease in this way, understanding the impact of treatment on GEP-NETs patients’ HRQoL is paramount especially given the indolent course the disease may take in patients.
The aim of this study was to perform a systematic review to assess the HRQoL of patients with inoperable metastatic GEP-NETs undergoing various treatments in order to uncover areas for future research. This systematic literature review is a response to the increased importanceof assessing HRQoL with validated patient-reported outcome instruments for which the minimal important difference has been defined.
The systematic review performed here adhered to the established international guidelines for conducting systematic reviews. A search strategy was developed and refined to recover relevant publications reporting HRQoL data for adult patients with GEP-NETs. Data were extracted from every record returned by the literature search that met the inclusion criteria of the study.
A total of 42 studies met the inclusion criteria of the study. The EORTC QLQ-C30 and gastrointestinal GI-NET module (QLQ-GI.NET21) were the most used HRQoL instruments, exhibiting a range of outcomes compared to baseline for the specific interventions.
The worst HRQoL observed at baseline were related to the following symptoms: Nausea and vomiting (4.1–20.0), insomnia (11.0–38.6), appetite loss (9.2–27.8), constipation (0–24.4) and financial difficulties (6.0–23.6). Diarrhoea demonstrated a wide range of mean values (16.7–78.3), which can be attributed to the heterogeneity of involved GEP-NET patients.
The average value for the baseline global health score had a range of 56–70. Where change in the EORTC QLQ-C30 global health score could be calculated from baseline scores, five studies demonstrated a significant trend towards an improved score. Treatment of GEP-NETs with 177Lu-DOTATATE consistently improved global health score compared to baseline and increased time-to-deterioration, i.e. time until HRQoL worsens by at least 10 points for each domain (e.g. global health, physical functioning). The Phase III NETTER-1 study resulted in time-to-deterioration curves consistent with the curves for progression free survival for 177Lu-DOTATATE. However, the EORTC QLQ-C30 and QLQ-GINET21 have not been validated, and the minimal important differences have not yet been defined, specifically for GI-NET and P-NET patients.
Study and patient heterogeneity, small sample sizes and limited methodological quality in terms of assessing HRQoL has impaired definitive statements being made regarding the impact of the various treatment options for GEP-NETs in terms of HRQoL. A notable finding was that PRRT can delay the time to worsening of HRQoL in GEP-NET patients. However, validation of EORTC QLQ-C30 and GINET21 specifically for GI-NET and P-NET patients and the definitions of minimal important difference and clinical thresholds are still lacking for these patients. As such, further research is necessary in order to offer a benchmark for interpreting the clinical importance of differences in HRQoL domain scores between treatment groups or changes in these scores over time in conjunction with different treatment modalities.
Further research is necessary in order to offer a benchmark for interpreting the clinical importance of differences in HRQoL domain scores over time and between treatment groups.
We would like to acknowledge Dr Persefoni Ioannou for providing editorial support and guiding the research from the outset of this project.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Karaivazoglou K, Moschovi MA, Wang ZJ S-Editor: Zhang L L-Editor: A E-Editor: Ma YJ
| 1. | Genus TSE, Bouvier C, Wong KF, Srirajaskanthan R, Rous BA, Talbot DC, Valle JW, Khan M, Pearce N, Elshafie M, Reed NS, Morgan E, Deas A, White C, Huws D, Ramage J. Impact of neuroendocrine morphology on cancer outcomes and stage at diagnosis: a UK nationwide cohort study 2013-2015. Br J Cancer. 2019;121:966-972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 2. | Öberg K, Knigge U, Kwekkeboom D, Perren A; ESMO Guidelines Working Group. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23 Suppl 7:vii124-vii130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 337] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 3. | Lewis AR, Wang X, Magdalani L, D'Arienzo P, Bashir C, Mansoor W, Hubner R, Valle JW, McNamara MG. Health-related quality of life, anxiety, depression and impulsivity in patients with advanced gastroenteropancreatic neuroendocrine tumours. World J Gastroenterol. 2018;24:671-679. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 4. | Wilander E, Lundqvist M, Oberg K. Gastrointestinal carcinoid tumours. Histogenetic, histochemical, immunohistochemical, clinical and therapeutic aspects. Prog Histochem Cytochem. 1989;19:1-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Ahlman H, Wängberg B, Jansson S, Stenqvist O, Geterud K, Tylén U, Caidahl K, Scherstén T, Tisell LE. Management of disseminated midgut carcinoid tumours. Digestion. 1991;49:78-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 6. | Gelhorn HL, Kulke MH, O'Dorisio T, Yang QM, Jackson J, Jackson S, Boehm KA, Law L, Kostelec J, Auguste P, Lapuerta P. Patient-reported Symptom Experiences in Patients With Carcinoid Syndrome After Participation in a Study of Telotristat Etiprate: A Qualitative Interview Approach. Clin Ther. 2016;38:759-768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 7. | Jacobsen MB, Hanssen LE. Clinical effects of octreotide compared to placebo in patients with gastrointestinal neuroendocrine tumours. Report on a double-blind, randomized trial. J Intern Med. 1995;237:269-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Ballal S, Yadav MP, Bal C, Sahoo RK, Tripathi M. Broadening horizons with 225Ac-DOTATATE targeted alpha therapy for gastroenteropancreatic neuroendocrine tumour patients stable or refractory to 177Lu-DOTATATE PRRT: first clinical experience on the efficacy and safety. Eur J Nucl Med Mol Imaging. 2020;47:934-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 9. | Haugland T, Wahl AK, Hofoss D, DeVon HA. Association between general self-efficacy, social support, cancer-related stress and physical health-related quality of life: a path model study in patients with neuroendocrine tumors. Health Qual Life Outcomes. 2016;14:11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 10. | Uri I, Grozinsky-Glasberg S. Current treatment strategies for patients with advanced gastroenteropancreatic neuroendocrine tumors (GEP-NETs). Clin Diabetes Endocrinol. 2018;4:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 11. | Kane A, Thorpe MP, Morse MA, Howard BA, Oldan JD, Zhu J, Wong TZ, Petry NA, Reiman R, Borges-Neto S. Predictors of Survival in 211 Patients with Stage IV Pulmonary and Gastroenteropancreatic MIBG-Positive Neuroendocrine Tumors Treated with 131I-MIBG. J Nucl Med. 2018;59:1708-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Strosberg J, Gardner N, Kvols L. Survival and prognostic factor analysis of 146 metastatic neuroendocrine tumors of the mid-gut. Neuroendocrinology. 2009;89:471-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 123] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 13. | Brundage M, Blazeby J, Revicki D, Bass B, de Vet H, Duffy H, Efficace F, King M, Lam CL, Moher D, Scott J, Sloan J, Snyder C, Yount S, Calvert M. Patient-reported outcomes in randomized clinical trials: development of ISOQOL reporting standards. Qual Life Res. 2013;22:1161-1175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 163] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 14. | Efficace F, Rees J, Fayers P, Pusic A, Taphoorn M, Greimel E, Reijneveld J, Whale K, Blazeby J; European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Group. Overcoming barriers to the implementation of patient-reported outcomes in cancer clinical trials: the PROMOTION Registry. Health Qual Life Outcomes. 2014;12:86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 15. | Reeve BB, Mitchell SA, Dueck AC, Basch E, Cella D, Reilly CM, Minasian LM, Denicoff AM, O'Mara AM, Fisch MJ, Chauhan C, Aaronson NK, Coens C, Bruner DW. Recommended patient-reported core set of symptoms to measure in adult cancer treatment trials. J Natl Cancer Inst. 2014;106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 220] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 16. | Rinke A, Neary MP, Eriksson J, Hunger M, Doan T, Karli D, Arnold R. Health-Related Quality of Life for Long-Acting Octreotide versus Placebo in Patients with Metastatic Midgut Neuroendocrine Tumors in the Phase 3 PROMID Trial. Neuroendocrinology. 2019;109:141-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 17. | Strosberg J, Wolin E, Chasen B, Kulke M, Bushnell D, Caplin M, Baum RP, Kunz P, Hobday T, Hendifar A, Oberg K, Sierra ML, Thevenet T, Margalet I, Ruszniewski P, Krenning E; NETTER-1 Study Group. Health-Related Quality of Life in Patients With Progressive Midgut Neuroendocrine Tumors Treated With 177Lu-Dotatate in the Phase III NETTER-1 Trial. J Clin Oncol. 2018;36:2578-2584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 277] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 18. | Martini C, Gamper EM, Wintner L, Nilica B, Sperner-Unterweger B, Holzner B, Virgolini I. Systematic review reveals lack of quality in reporting health-related quality of life in patients with gastroenteropancreatic neuroendocrine tumours. Health Qual Life Outcomes. 2016;14:127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 19. | United States Food and Drug Administration. Value and use of patient-reported outcomes (PROs) in assessing effects of medical devices. CDRH strategic priorities 2016-2017. |
| 20. | European Medicines Agency. Integrating patients' views in clinical studies of anticancer medicines 2016. Available from: https://www.ema.europa.eu/en/news/integrating-patients-views-clinical-studies-anticancer-medicines. |
| 21. | Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13930] [Cited by in RCA: 13320] [Article Influence: 832.5] [Reference Citation Analysis (0)] |
| 22. | Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane handbook for systematic reviews of interventions. 2nd Edition. Chichester (UK): John Wiley and Sons, 2019. Available from: https://training.cochrane.org/handbook. |
| 23. | Papaioannou D, Brazier J, Paisley S. NICE DSU Technical Support Document 9: The Identification, Review and Synthesis of Health State Utility Values from the Literature [Internet]. London: National Institute for Health and Care Excellence (NICE) 2010. Available from: https://www.ncbi.nlm.nih.gov/books/NBK425822/?term=collection-id_nicedsutsd%5BAttribute%5D. |
| 24. | Brazier J, Ara R, Azzabi I, Busschbach J, Chevrou-Séverac H, Crawford B, Cruz L, Karnon J, Lloyd A, Paisley S, Pickard AS. Identification, Review, and Use of Health State Utilities in Cost-Effectiveness Models: An ISPOR Good Practices for Outcomes Research Task Force Report. Value Health. 2019;22:267-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 25. | Larsson G, Sjödén PO, Oberg K, Eriksson B, von Essen L. Health-related quality of life, anxiety and depression in patients with midgut carcinoid tumours. Acta Oncol. 2001;40:825-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 26. | Larsson G, Sjödén PO, Oberg K, von Essen L. Importance-satisfaction discrepancies are associated with health-related quality of life in five-year survivors of endocrine gastrointestinal tumours. Ann Oncol. 1999;10:1321-1327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 27. | Rinke A, Müller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, Mayer C, Aminossadati B, Pape UF, Bläker M, Harder J, Arnold C, Gress T, Arnold R; PROMID Study Group. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27:4656-4663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1609] [Cited by in RCA: 1739] [Article Influence: 108.7] [Reference Citation Analysis (0)] |
| 28. | Meng Y, McCarthy G, Berthon A, Dinet J. Patient-reported health state utilities in metastatic gastroenteropancreatic neuroendocrine tumours - an analysis based on the CLARINET study. Health Qual Life Outcomes. 2017;15:131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 29. | Kvols LK, Oberg KE, O'Dorisio TM, Mohideen P, de Herder WW, Arnold R, Hu K, Zhang Y, Hughes G, Anthony L, Wiedenmann B. Pasireotide (SOM230) shows efficacy and tolerability in the treatment of patients with advanced neuroendocrine tumors refractory or resistant to octreotide LAR: results from a phase II study. Endocr Relat Cancer. 2012;19:657-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 148] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 30. | Korse CM, Bonfrer JM, Aaronson NK, Hart AA, Taal BG. Chromogranin A as an alternative to 5-hydroxyindoleacetic acid in the evaluation of symptoms during treatment of patients with neuroendocrine Tumors. Neuroendocrinology. 2009;89:296-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 31. | Arnold R, Rinke A, Klose KJ, Müller HH, Wied M, Zamzow K, Schmidt C, Schade-Brittinger C, Barth P, Moll R, Koller M, Unterhalt M, Hiddemann W, Schmidt-Lauber M, Pavel M, Arnold CN. Octreotide versus octreotide plus interferon-alpha in endocrine gastroenteropancreatic tumors: a randomized trial. Clin Gastroenterol Hepatol. 2005;3:761-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 188] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 32. | O'Toole D, Ducreux M, Bommelaer G, Wemeau JL, Bouché O, Catus F, Blumberg J, Ruszniewski P. Treatment of carcinoid syndrome: a prospective crossover evaluation of lanreotide versus octreotide in terms of efficacy, patient acceptability, and tolerance. Cancer. 2000;88:770-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 33. | Yadegarfar G, Friend L, Jones L, Plum LM, Ardill J, Taal B, Larsson G, Jeziorski K, Kwekkeboom D, Ramage JK; EORTC Quality of Life Group. Validation of the EORTC QLQ-GINET21 questionnaire for assessing quality of life of patients with gastrointestinal neuroendocrine tumours. Br J Cancer. 2013;108:301-310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 88] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 34. | Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, Valle J, Metrakos P, Smith D, Vinik A, Chen JS, Hörsch D, Hammel P, Wiedenmann B, Van Cutsem E, Patyna S, Lu DR, Blanckmeister C, Chao R, Ruszniewski P. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:501-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2032] [Cited by in RCA: 1827] [Article Influence: 130.5] [Reference Citation Analysis (0)] |
| 35. | Vinik A, Bottomley A, Korytowsky B, Bang YJ, Raoul JL, Valle JW, Metrakos P, Hörsch D, Mundayat R, Reisman A, Wang Z, Chao RC, Raymond E. Patient-Reported Outcomes and Quality of Life with Sunitinib Versus Placebo for Pancreatic Neuroendocrine Tumors: Results From an International Phase III Trial. Target Oncol. 2016;11:815-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 36. | Casciano R, Chulikavit M, Perrin A, Liu Z, Wang X, Garrison LP. Cost-effectiveness of everolimus vs sunitinib in treating patients with advanced, progressive pancreatic neuroendocrine tumors in the United States. J Med Econ. 2012;15 Suppl 1:55-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 37. | Fröjd C, Larsson G, Lampic C, von Essen L. Health related quality of life and psychosocial function among patients with carcinoid tumours. A longitudinal, prospective, and comparative study. Health Qual Life Outcomes. 2007;5:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 38. | Larsson G, von Essen L, Sjödén PO. Health-related quality of life in patients with endocrine tumours of the gastrointestinal tract. Acta Oncol. 1999;38:481-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 39. | Ramage J, Yadegarfar G, Friend L, Jones L, Plum L, Ardill J, Taal B, Larsson G, Jesiorski K. Clinical and psychometric validation of the disease-specific questionnaire module (EORTC qlqgiNET21) in assessing quality of life of patients with neuroendocrine tumors. 9th Annual ENETS Conference (2012). Available from: https://www.enets.org/clinical-and-psychometric-validation-of-the-disease-specific-questionnaire-module-eortc-qlq-ginet21-in-assessing-quality-of-life-of-patients-with-neuroendocrine-tumors.html. |
| 40. | Mitry E, Walter T, Baudin E, Kurtz JE, Ruszniewski P, Dominguez-Tinajero S, Bengrine-Lefevre L, Cadiot G, Dromain C, Farace F, Rougier P, Ducreux M. Bevacizumab plus capecitabine in patients with progressive advanced well-differentiated neuroendocrine tumors of the gastro-intestinal (GI-NETs) tract (BETTER trial)--a phase II non-randomised trial. Eur J Cancer. 2014;50:3107-3115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 41. | Ducreux M, Dahan L, Smith D, O'Toole D, Lepère C, Dromain C, Vilgrain V, Baudin E, Lombard-Bohas C, Scoazec JY, Seitz JF, Bitoun L, Koné S, Mitry E. Bevacizumab combined with 5-FU/streptozocin in patients with progressive metastatic well-differentiated pancreatic endocrine tumours (BETTER trial)--a phase II non-randomised trial. Eur J Cancer. 2014;50:3098-3106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 42. | Meyer T, Qian W, Caplin ME, Armstrong G, Lao-Sirieix SH, Hardy R, Valle JW, Talbot DC, Cunningham D, Reed N, Shaw A, Navalkissoor S, Luong TV, Corrie PG. Capecitabine and streptozocin ± cisplatin in advanced gastroenteropancreatic neuroendocrine tumours. Eur J Cancer. 2014;50:902-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 43. | Pasieka JL, McEwan AJ, Rorstad O. The palliative role of 131I-MIBG and 111In-octreotide therapy in patients with metastatic progressive neuroendocrine neoplasms. Surgery. 2004;136:1218-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 44. | Cella D, Beaumont JL, Hudgens S, Marteau F, Feuilly M, Houchard A, Lapuerta P, Ramage J, Pavel M, Hörsch D, Kulke MH. Relationship Between Symptoms and Health-related Quality-of-life Benefits in Patients With Carcinoid Syndrome: Post Hoc Analyses From TELESTAR. Clin Ther. 2018;40:2006-2020.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 45. | Zigmond AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67:361-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28548] [Cited by in RCA: 31757] [Article Influence: 756.1] [Reference Citation Analysis (0)] |
| 46. | Karppinen N, Lindén R, Sintonen H, Tarkkanen M, Roine R, Heiskanen I, Matikainen N, Schalin-Jäntti C. Health-Related Quality of Life in Patients with Small Intestine Neuroendocrine Tumors. Neuroendocrinology. 2018;107:366-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 47. | Haugland T, Vatn MH, Veenstra M, Wahl AK, Natvig GK. Health related quality of life in patients with neuroendocrine tumors compared with the general Norwegian population. Qual Life Res. 2009;18:719-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 48. | Frilling A, Weber F, Saner F, Bockisch A, Hofmann M, Mueller-Brand J, Broelsch CE. Treatment with (90)Y- and (177)Lu-DOTATOC in patients with metastatic neuroendocrine tumors. Surgery. 2006;140:968-76; discussion 976-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 49. | Pezzilli R, Campana D, Morselli-Labate AM, Fabbri MC, Brocchi E, Tomassetti P. Patient-reported outcomes in subjects with neuroendocrine tumors of the pancreas. World J Gastroenterol. 2009;15:5067-5073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 27] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 50. | Larsson G, Janson ET. Anemia in patients with midgut carcinoid, treated with alpha interferon: effects by erythropoietin treatment on the perceived quality of life. Eur J Cancer Care (Engl). 2008;17:200-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 51. | Anota A, Hamidou Z, Paget-Bailly S, Chibaudel B, Bascoul-Mollevi C, Auquier P, Westeel V, Fiteni F, Borg C, Bonnetain F. Time to health-related quality of life score deterioration as a modality of longitudinal analysis for health-related quality of life studies in oncology: do we need RECIST for quality of life to achieve standardization? Qual Life Res. 2015;24:5-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 52. | Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16:139-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2281] [Cited by in RCA: 2363] [Article Influence: 87.5] [Reference Citation Analysis (0)] |
| 53. | Haugland T, Veenstra M, Vatn MH, Wahl AK. Improvement in Stress, General Self-Efficacy, and Health Related Quality of Life following Patient Education for Patients with Neuroendocrine Tumors: A Pilot Study. Nurs Res Pract. 2013;2013:695820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 54. | Ramage JK, Punia P, Faluyi O, Frilling A, Meyer T, Saharan R, Valle JW. Observational Study to Assess Quality of Life in Patients with Pancreatic Neuroendocrine Tumors Receiving Treatment with Everolimus: The OBLIQUE Study (UK Phase IV Trial). Neuroendocrinology. 2019;108:317-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 55. | Kulke MH, Lenz HJ, Meropol NJ, Posey J, Ryan DP, Picus J, Bergsland E, Stuart K, Tye L, Huang X, Li JZ, Baum CM, Fuchs CS. Activity of sunitinib in patients with advanced neuroendocrine tumors. J Clin Oncol. 2008;26:3403-3410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 467] [Cited by in RCA: 443] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 56. | Pavel M, Unger N, Borbath I, Ricci S, Hwang TL, Brechenmacher T, Park J, Herbst F, Beaumont JL, Bechter O. Safety and QOL in Patients with Advanced NET in a Phase 3b Expanded Access Study of Everolimus. Target Oncol. 2016;11:667-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 57. | Wymenga AN, Eriksson B, Salmela PI, Jacobsen MB, Van Cutsem EJ, Fiasse RH, Välimäki MJ, Renstrup J, de Vries EG, Oberg KE. Efficacy and safety of prolonged-release lanreotide in patients with gastrointestinal neuroendocrine tumors and hormone-related symptoms. J Clin Oncol. 1999;17:1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 134] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 58. | Zandee WT, Brabander T, Blazevic A, Kam BLR, Teunissen JJM, Feelders RA, Hofland J, De Herder WW. Symptomatic and radiological response to 177Lu-DOTATATE for the treatment of functioning pancreatic neuroendocrine tumors. J Clin Endocrinol Metab. 2019;104:1336-1344 [. [RCA] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 59. | Martini C, Buxbaum S, Rodrigues M, Nilica B, Scarpa L, Holzner B, Virgolini I, Gamper EM. Quality of Life in Patients with Metastatic Gastroenteropancreatic Neuroendocrine Tumors Receiving Peptide Receptor Radionuclide Therapy: Information from a Monitoring Program in Clinical Routine. J Nucl Med. 2018;59:1566-1573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 60. | Kwekkeboom DJ, Bakker WH, Kam BL, Teunissen JJ, Kooij PP, de Herder WW, Feelders RA, van Eijck CH, de Jong M, Srinivasan A, Erion JL, Krenning EP. Treatment of patients with gastro-entero-pancreatic (GEP) tumours with the novel radiolabelled somatostatin analogue [177Lu-DOTA(0),Tyr3]octreotate. Eur J Nucl Med Mol Imaging. 2003;30:417-422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 279] [Cited by in RCA: 275] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 61. | Marinova M, Mücke M, Fischer F, Essler M, Cuhls H, Radbruch L, Ghaei S, Conrad R, Ahmadzadehfar H. Quality of life in patients with midgut NET following peptide receptor radionuclide therapy. Eur J Nucl Med Mol Imaging. 2019;46:2252-2259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 62. | Teunissen JJ, Kwekkeboom DJ, Krenning EP. Quality of life in patients with gastroenteropancreatic tumors treated with [177Lu-DOTA0,Tyr3]octreotate. J Clin Oncol. 2004;22:2724-2729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 140] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 63. | Jiménez-Fonseca P, Carmona-Bayonas A, Martín-Pérez E, Crespo G, Serrano R, Llanos M, Villabona C, García-Carbonero R, Aller J, Capdevila J, Grande E; Spanish Neuroendocrine Tumor Group (GETNE). Health-related quality of life in well-differentiated metastatic gastroenteropancreatic neuroendocrine tumors. Cancer Metastasis Rev. 2015;34:381-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 64. | Giesinger JM, Loth FLC, Aaronson NK, Arraras JI, Caocci G, Efficace F, Groenvold M, van Leeuwen M, Petersen MA, Ramage J, Tomaszewski KA, Young T, Holzner B; EORTC Quality of Life Group. Thresholds for clinical importance were established to improve interpretation of the EORTC QLQ-C30 in clinical practice and research. J Clin Epidemiol. 2020;118:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 227] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 65. | Cocks K, King MT, Velikova G, de Castro G, Martyn St-James M, Fayers PM, Brown JM. Evidence-based guidelines for interpreting change scores for the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. Eur J Cancer. 2012;48:1713-1721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 383] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 66. | Delpassand ES, Samarghandi A, Zamanian S, Wolin EM, Hamiditabar M, Espenan GD, Erion JL, O'Dorisio TM, Kvols LK, Simon J, Wolfangel R, Camp A, Krenning EP, Mojtahedi A. Peptide receptor radionuclide therapy with 177Lu-DOTATATE for patients with somatostatin receptor-expressing neuroendocrine tumors: the first US phase 2 experience. Pancreas. 2014;43:518-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 105] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 67. | van der Horst-Schrivers AN, van Ieperen E, Wymenga AN, Boezen HM, Weijmar-Schultz WC, Kema IP, Meijer WG, de Herder WW, Willemse PH, Links TP, de Vries EG. Sexual function in patients with metastatic midgut carcinoid tumours. Neuroendocrinology. 2009;89:231-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 68. | Davies AH, Larsson G, Ardill J, Friend E, Jones L, Falconi M, Bettini R, Koller M, Sezer O, Fleissner C, Taal B, Blazeby JM, Ramage JK; EORTC Quality of Life Group. Development of a disease-specific Quality of Life questionnaire module for patients with gastrointestinal neuroendocrine tumours. Eur J Cancer. 2006;42:477-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 69. | Larsson G, von Essen L, Sjödén PO. Quality of life in patients with endocrine tumors of the gastrointestinal tract: patient and staff perceptions. Cancer Nurs. 1998;21:411-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |