Published online Jun 28, 2020. doi: 10.3748/wjg.v26.i24.3318
Peer-review started: November 19, 2019
First decision: April 1, 2020
Revised: May 11, 2020
Accepted: June 4, 2020
Article in press: June 4, 2020
Published online: June 28, 2020
Processing time: 220 Days and 21.4 Hours
Lymph node dissection is always a hot issue in radical resection of hilar cholangiocarcinoma (HCCA). There are still controversies regarding whether some lymph nodes should be dissected, of which the para-aortic lymph nodes are the most controversial. This review synthesized findings in the literature using the PubMed database of articles in the English language published between 1990 and 2019 on the effectiveness of extended lymphadenectomy including para-aortic lymph nodes dissection in radical resection of HCCA. Hepatobiliary surgeons have basically achieved a consensus that enough lymph nodes should be obtained to accurately stage HCCA. Only a very small number of studies have focused on the effectiveness of extended lymphadenectomy including para-aortic nodes dissection on HCCA. They reported that extended lymphadenectomy can bring some survival benefits for patients with potential para-aortic lymph node metastasis and more lymph nodes can be obtained to make the patient's tumor staging more accurate without increasing the related complications. Extended lymphadenectomy should not be adopted for HCCA patients with intraoperatively confirmed distant lymph node metastases. For these patients, radical resection combined with postoperative adjuvant chemotherapy seems to be a better choice. A prospective, multicenter, randomized, controlled clinical study of regional lymphotomy and extended lymphadenectomy in HCCA should be conducted to guide clinical practice. A standardized extended lymphadenectomy may help to more accurately stage HCCA. Future studies are required to further assess whether extended lymphadenectomy can improve long-term survival in negative celiac, superior mesenteric, and para-aortic lymph node diseases.
Core tip: For patients with resectable hilar cholangiocarcinoma (HCCA), extended lymphadenectomy including the No.16 group may obtain more lymph nodes to more accurately stage the tumor and to reduce the influence of total lymph node count on the lymph node ratio when compared with single enlarged No.16 lymph node biopsy. In addition, it also may help to prevent the occurrence of lymph node micrometastases, which will avoid the difficult to determine cause of postoperative enlargement of the No.16 group lymph nodes. Therefore, extended lymphadenectomy including the No.16 group is potentially more consistent with the principle of lymph node dissection in radical resection of HCCA.
- Citation: Li J, Zhou MH, Ma WJ, Li FY, Deng YL. Extended lymphadenectomy in hilar cholangiocarcinoma: What it will bring? World J Gastroenterol 2020; 26(24): 3318-3325
- URL: https://www.wjgnet.com/1007-9327/full/v26/i24/3318.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i24.3318
Lymph node metastasis is common in hilar cholangiocarcinoma (HCCA) due to the thin bile duct wall, and it is one of the most important factors affecting the prognosis of HCCA[1,2]. According to reports, the incidence of lymph node metastasis is as high as 31%-58% in resectable HCCA[2]. Therefore, the dissection of lymph nodes is always a hot issue in the radical resection of HCCA. A study has shown that lymph node metastasis of HCCA is positively correlated with the degree of infiltration (T) and Bismuth subtype[3]. The incidence of lymph node metastasis was found to be 0% in T1, 36.7% in T2, 23.8% in T3, and 57.7% in T4, respectively. For Bismuth subtype, it was 21.1% for Bismuth I, 27.3% for Bismuth II, 41.5% for Bismuth III, and 55.6% for Bismuth IV, respectively[3]. Additionally, the anatomy surrounding the hilar bile duct is complex; therefore, the extent of lymph nodes dissection in radical resection of HCCA is still controversial.
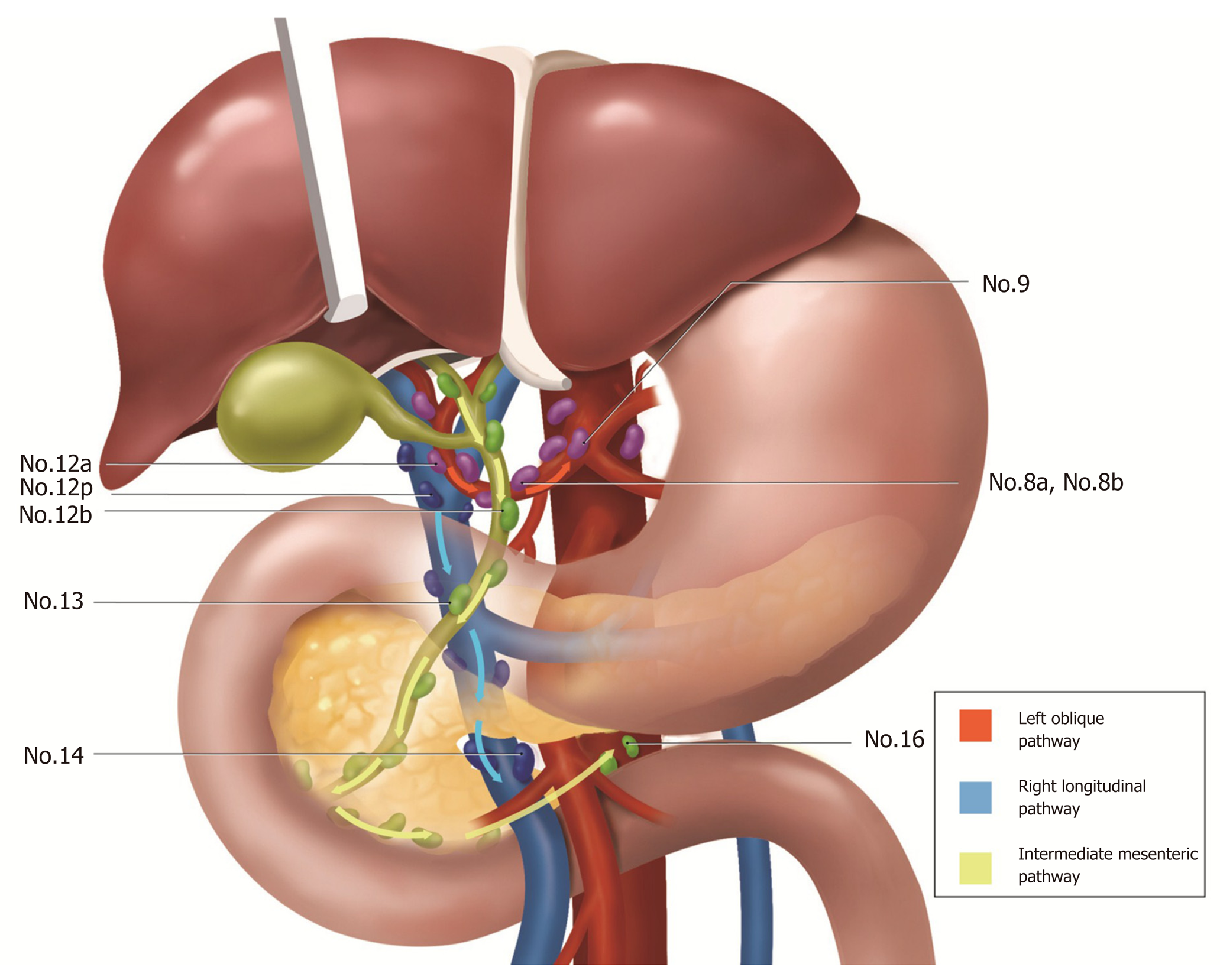
It is important to understand the lymphatic drainage pathway of the hilar bile duct for guiding lymph node dissection in radical resection of HCCA. The lymphatic metastasis pathway of HCCA is closely related to the lymphatic drainage of the hilar bile duct. From the early 1990s to 2013, Japanese researchers such as Ito, Shirai, Kayahara, and Sato conducted in-depth studies of the lymphatic drainage pathway of the hilar bile duct[4-7]. They discovered that the lymphatic drainage of the hilar bile duct followed three paths: (1) That from the hepatic artery (No. 12a) along the common hepatic artery (No. 8) to the celiac lymph nodes (No. 9); (2) That which first descends along the bile duct (No. 12b) and then runs on the posterior surface of the pancreas head (No. 13), and then to the para-aortic lymph node (No. 16); and (3) The lesser known pathway which descends along the portal vein (No. 12p) to reach the superior mesenteric vein and then enters the superior mesenteric nodes (No. 14) (Figure 1). Of these, the first two paths are the main drainage paths and play important roles in the lymphatic drainage of the hilar bile duct.
The staging systems for HCCA are different in different guidelines. According to the 7th edition of the TNM staging system released by the American Joint Committee on Cancer (AJCC) and the Union for International Cancer Control (UICC) in 2010, N1 was defined as a regional lymph node metastasis including the cystic duct, common bile duct, hepatic artery, and/or portal vein lymph nodes. N2 was defined as lymph node metastasis including the para-aorta, para-caval vein, celiac trunk, and/or superior mesenteric vein. Additionally, N1 and N2 are staging indications of stage IIIB and IVB, respectively[8]. Compared to the AJCC staging system, the 3rd edition of the TNM staging system issued by the Japan Society of Hepatobiliary and Pancreatic Surgery (JSHBPS) in 2015, N1 was defined as regional lymph node metastasis including the cystic duct, common bile duct, hepatic artery, portal vein, and the posterior of the pancreatic head. Metastasis beyond the above lymph nodes was considered as distant metastasis (M1)[9]. The JSHBPS recommended that the No. 8, 12, and 13 group lymph nodes should be dissected[10].
In HCCA, lymph nodes metastasis around the bile duct are most commonly encountered (27.1%-42.7%), followed by the portal vein (30.9%-35.7%), common hepatic artery (27.3%-31.3%), para-aorta (17.3%), posterior pancreatic head (14.5%-50%), and celiac trunk (6.4%-14.3%)[11,12]. The guideline for hepatobiliary cancer from the National Comprehensive Cancer Network (NCCN) suggests that the standard lymph node dissection of HCCA includes lymph nodes along the hepatoduodenal ligament, and the posterior of the pancreatic head, i.e., No. 12 and No. 13 lymph nodes. Lymph node metastasis beyond the above was considered a contraindication of radical surgery[13]. Unlike the NCCN guidelines, the JSHBPS provided a more detailed description for the dissection extent of lymph nodes in radical resection of HCCA. Mantel et al[14] reported that lymph node metastasis or micrometastasis often occurs in lymph nodes around the portal vein, common hepatic artery, and the posterior of the pancreatic head. In N0 patients, the 5-year survival rate of patients with lymph node micrometastasis was significantly lower than those without micrometastasis (27% vs 54%, P = 0.01), and not significantly different to N1 patients (27% vs 15%, P = 0.54)[14]. Consequently, the JSHBPS recommended that the lymph nodes in the first and second stations, i.e., the No. 8, 12, and 13 groups[10], should be dissected for HCCA, which seems to be more reasonable. However, the JSHBPS also believed that No. 16 lymph nodes metastasis is a contraindication for radical resection[10].
Nagakawa et al[15] found that in patients with resectable HCCA, 4.6%-20% of patients had superior mesenteric lymph node metastasis (No. 14), and 12.6% of patients had para-aortic lymph node metastasis (No. 16)[11,15]. Additionally, Kitagawa et al[16] defined the extended lymphadenectomy as removal of lymph nodes including para-aortic, superior mesenteric vein, and celiac trunk lymph nodes. Their study showed that the 3-year and 5-year survival rates were 31.8% and 14.6% for 52 patients without lymph node metastasis, 31.8% and 14.7% for 39 patients with regional lymph node metastases, and 12.3% and 12.3% for 19 patients with para-aortic lymph nodes metastases, respectively[16]. Of the 19 patients with para-aortic lymph node metastases, 7 patients had no obvious signs of lymph node involvement during surgery and was confirmed by postoperative pathology examination. The survival of 7 patients was significantly better than that of 12 patients who proved to have positive lymph node metastases intraoperatively, and was equivalent to that of patients with regional lymph node metastases[16]. That is, for patients with potential para-aortic lymph node metastasis, extended lymphadenectomy can still bring some survival benefits, and make the patient's tumor staging more accurate without increasing the incidence of related complications[12,17-19]. The 5-year survival rates of HCCA including regional lymphadenectomy were reported to be 7%-20%[20-23]. In contrast, 5-year survival rates of HCCA with extended lymphadenectomy were reported to be 26%-49%[3,24,25]. Therefore, Aoba et al[3] suggested that extended lymphadenectomy is recommended in resectable HCCA. However, some scholars believe that patients with No. 16 lymph nodes metastasis found intra-operatively should be regarded as a contraindication for radical surgery[14] as the literature related to gallbladder or pancreatic carcinoma reported that extended lymphadenectomy cannot provide survival benefits for patients[26-29]. At the same time, some scholars found that there was no significant difference in the survival between patients with N1 and N2 lymph node metastases[2,3,30]. These findings challenged the accuracy of the 7th AJCC staging system which stages HCCA by the lymph node metastasis site. Recently, Ma et al[31] reported that extended lymphadenectomy significantly increases lymph node retrieval, thereby preventing understaging and improving survival prediction. Extended lymphadenectomy may improve overall survival in patients with M0 disease who underwent R0 resection, but does not improve overall survival for M1 patients. Thus, they concluded that extended lymphadenectomy should not be adopted for HCCA patients with intraoperatively confirmed distant lymph node metastases, which is consistent with previous reports[14]. Three meta-analyses[32-34] have suggested the potential benefit of chemotherapy as adjuvant therapy in patients with resected node-positive disease. For these patients, radical resection combined with postoperative adjuvant chemotherapy seems to be a better choice.
As extended lymphadenectomy indicated improvement in overall survival over patients who underwent regional lymphadenectomy in M0 and R0 resection disease before propensity score matching, but not after propensity score matching in Ma et al[31]’s report, they suggested that future studies are required to further assess whether extended lymphadenectomy should be performed in negative celiac, superior mesenteric, and para-aortic lymph nodes HCCA patients.
On the one hand, there is no consensus on the dissection extent of lymph nodes, even in different guidelines[10,13], for the surgical treatment of HCCA. The accuracy of taking the involved site of lymph node metastases as the basis of tumor staging is controversial and the evidence is not effective to guide clinical practice. On the other hand, total lymph node count (TLNC) and lymph node ratio (LNR) play a positive role in prognostic stratification for gastrointestinal tumors. Hepatobiliary surgeons try to apply TLNC and LNR in HCCA to guide the dissection extent of lymph node and prognostic stratification[3,20,24,35-38].
In 2007, Schwarz et al[36] conducted a retrospective cohort study of 1518 patients with extrahepatic cholangiocarcinoma in the SEER database (surveillance, epidemiology, and end result database) and found that the survival of patients with TLNC > 10 was significantly better than that of patients with TLNC < 10. In 2010, Ito et al[37] conducted a cohort study of 320 patients with HCCA and showed that patients with TLNC > 7 had a significantly better survival than patients with TLNC < 7 in R0N0 patients. In 2015, a systematic review of 20 retrospective studies showed that a TLNC of 7-9 can maximally identify the total number of positive lymph nodes and minimize understaging of the tumor[38]. In 2016, a retrospective cohort study of 437 patients reported by Bagante et al[11] showed that as the TLNC increased, not only did the 5-year survival rate of N0 patients significantly improve, but also the detection rate of N1 patients significantly increased. Bagante et al[11] suggested that the minimum number of lymph nodes dissected is 4.
Although the suggested TLNC in the above studies was different, they all reached the conclusion that accurate LN staging can be performed for HCCA only when an adequate TLNC is obtained. Some lymph node metastases may be misdiagnosed when the TLNC is insufficient, resulting in inaccurate staging (underestimation) of the tumor, making the patient's actual survival lower than expected, manifesting as a poor prognosis in patients with fewer lymph node dissected. At the same time, Bagante et al[11] found that in patients with lymph node metastases, the 5-year survival of patients with ≤ 3 lymph node metastases was significantly better than that of patients with > 3 lymph node metastases (18.6% vs 11.1%, P = 0.02). As a result of the above observation, the 8th edition of the AJCC TNM staging system released in 2017 recommends that the dissected number of lymph nodes should be at least 5, and changes the criteria of lymph node staging, i.e., N1: 1-3 lymph node metastases, N2: > 3 lymph node metastases[13].
Some other scholars believe that the LNR should be used as a staging criterion for HCCA. In 2010 and 2013, Giuliante et al[1] and Guglielmi et al[35] analyzed the LNR of 62 patients and 75 patients with HCCA who underwent radical resection, respectively. The results showed that patients with an LNR of 0.25 or lower had significantly better survival. Therefore, Guglielmi suggested that the LNR had a predictive value for prognosis in HCCA. In 2011, Oshiro et al[22] investigated the LNR of 60 patients with resected extrahepatic cholangiocarcinoma (hilar, n = 31; distal, n = 29) and found that patients with an LNR of 0.2 or lower had significantly better survival. Therefore, Oshiro et al[22] also considered LNR to be an effective prognostic factor after radical resection of extrahepatic cholangiocarcinoma. In 2013, Aoba et al[3]’s study of 320 patients also showed that those with an LNR ≤ 0.2 had a significantly better survival than patients with an LNR > 0.2. Additionally, Aoba et al[3] also found that patients with a TLNC ≥ 16 had a significantly lower LNR than patients with a TLNC of 3-5 or 6-10. In multivariate analysis, LNR is not an independent predictor of prognosis. It was confirmed that the LNR had a better stratification effect than that of the site of lymph node metastasis involvement on prognosis in a number of studies[1,2,11,20,24,30,39], but the median TLNC in the above studies was mostly 3.
Indeed, the LNR can standardize lymph node metastasis in all patients, and minimize the inconsistency and variability in lymph node assessment. However, the LNR is largely influenced by TLNC (i.e., the larger the TLNC, the smaller the LNR). Unlike gastric cancer or colorectal cancer, the TLNC in HCCA is usually less than 10. In this case, the LNR is likely to result in errors. Therefore, the clinical value of the LNR in HCCA needs to be verified with a sufficient TLNC. It must be stated that extended lymphadenectomy can significantly increase the number of lymph node dissections. Studies have confirmed that at least 15 lymph nodes can be obtained by extended lymphadenectomy in the radical resection of HCCA[38]. At the same time, Aoba et al[3] found that the survival of patients undergoing extended lymphadenectomy was significantly better than that of unresected patients. In patients with multiple lymph node metastases, patients with distant lymph node metastases (No. 14/16) had comparable survival to those with regional lymph node metastases[3]. These findings suggest that in addition to obtaining sufficient numbers of lymph nodes to accurately stage tumors, extended lymphadenectomy can also provide certain survival benefits for patients with distant lymph node metastases.
Following the exploration of lymph node dissection in radical resection and the redefinition of lymph node staging for HCCA, hepatobiliary surgeons have basically reached a consensus that enough lymph nodes should be obtained to accurately stage HCCA. However, there are still controversies regarding whether some lymph nodes should be dissected, of which the No. 16 group is the most controversial.
For scholars who believe that No.16 lymph node dissection does not bring survival benefits to patients with HCCA, we reviewed the literature and found that their views were either directly derived from previous reports of gallbladder or pancreatic carcinoma[36], or No. 16 lymph node metastases were treated as a surgical contraindication[14,20] without an in-depth exploration. Only a very small number of studies have focused on extended lymphadenectomy including the No. 16 group. They found that extended lymphadenectomy can provide a certain survival benefit for patients with HCCA, but the 5-year survival rate was only 12.3%[12,25]. However, Ma et al[31] reported that extended lymphadenectomy indicated improvement in overall survival over patients who underwent regional lymphadenectomy in M0 and R0 resection disease before propensity score matching, but not after propensity score matching. Simultaneously, studies have reported that the lymph node metastasis rate for the posterior of the pancreatic head (No. 13) could be as high as 50%, and the lymph nodes in the No. 16 group are the next drainage station for the No. 13 lymph nodes. Additionally, it is common to find enlarged No. 16 lymph nodes in clinical practice and the No. 16 lymph node metastasis rate is relatively low (17.3%)[12]. It is also very common to find enlarged No. 16 lymph nodes in the follow-up of patients with HCCA due to post-operative inflammation or metastasis. This has resulted in difficulty in judging the cause (due to inflammation or metastasis) and to determine the next treatment step for post-operative HCCA patients. Therefore, future studies are required to further assess whether extended lymphadenectomy should be performed in negative celiac, superior mesenteric, and para-aortic lymph node diseases. Fortunately, a prospective, multicenter, randomized, controlled clinical study of regional lymphotomy and extended lymphadenectomy in HCCA (registration number: ChiCTR1800015688) is being conducted in China. It will evaluate the clinical safety of extended lymphadenectomy in HCCA resection and the effect of different lymph node dissections on the survival of patients.
Although the pathologists and medical oncologists typically prefer more lymph nodes for accurate staging of any cancer, the surgeon should balance other factors, e.g., the anatomic location, difficulty in removing more lymph nodes, intra-operative and long-term complications associated with extensive lymph node dissection, etc. Fortunately, Ma et al[31] reported that incidence of lymphorrhagia, duration of postoperative stay, 30- or 90-d mortality, and other complications were comparable between the traditional regional lymphadenectomy and extended lymphadenectomy groups. Thus, extended lymphadenectomy could be performed in selected patients with resectable HCCA.
In summary, we believe that for patients with resectable HCCA, along with the improvement of surgical techniques, extended lymphadenectomy including the No. 16 group may obtain more lymph nodes to more accurately stage the tumor and to reduce the influence of the TLNC on the LNR when compared with single enlarged No. 16 lymph node biopsy. In addition, it may also help to prevent the occurrence of lymph node micrometastases, which will avoid the difficult to determine cause of postoperative enlargement of the No. 16 group of lymph nodes. Extended lymphadenectomy was reported to more accurately stage HCCA and bring survival benefits, and the role of extended lymphadenectomy in HCCA should be further confirmed.
The authors acknowledge the guidance of the Professor of Biliary Surgery in West China Hospital.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Kim HS, Tajiri K, Yang Z S-Editor: Ma RY L-Editor: Webster JR E-Editor: Zhang YL
| 1. | Giuliante F, Ardito F, Guglielmi A, Aldrighetti L, Ferrero A, Calise F, Giulini SM, Jovine E, Breccia C, De Rose AM, Pinna AD, Nuzzo G. Association of Lymph Node Status With Survival in Patients After Liver Resection for Hilar Cholangiocarcinoma in an Italian Multicenter Analysis. JAMA Surg. 2016;151:916-922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 2. | Conci S, Ruzzenente A, Sandri M, Bertuzzo F, Campagnaro T, Bagante F, Capelli P, D'Onofrio M, Piccino M, Dorna AE, Pedrazzani C, Iacono C, Guglielmi A. What is the most accurate lymph node staging method for perihilar cholangiocarcinoma? Comparison of UICC/AJCC pN stage, number of metastatic lymph nodes, lymph node ratio, and log odds of metastatic lymph nodes. Eur J Surg Oncol. 2017;43:743-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 3. | Aoba T, Ebata T, Yokoyama Y, Igami T, Sugawara G, Takahashi Y, Nimura Y, Nagino M. Assessment of nodal status for perihilar cholangiocarcinoma: location, number, or ratio of involved nodes. Ann Surg. 2013;257:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 106] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 4. | Ito M, Mishima Y, Sato T. An anatomical study of the lymphatic drainage of the gallbladder. Surg Radiol Anat. 1991;13:89-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Shirai Y, Yoshida K, Tsukada K, Ohtani T, Muto T. Identification of the regional lymphatic system of the gallbladder by vital staining. Br J Surg. 1992;79:659-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 74] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 6. | Sato T, Ito M, Sakamoto H. Pictorial dissection review of the lymphatic pathways from the gallbladder to the abdominal para-aortic lymph nodes and their relationships to the surrounding structures. Surg Radiol Anat. 2013;35:615-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Kayahara M, Nagakawa T, Ohta T, Kitagawa H, Tajima H, Miwa K. Role of nodal involvement and the periductal soft-tissue margin in middle and distal bile duct cancer. Ann Surg. 1999;229:76-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 102] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471-1474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5537] [Cited by in RCA: 6460] [Article Influence: 430.7] [Reference Citation Analysis (0)] |
| 9. | Miyazaki M, Ohtsuka M, Miyakawa S, Nagino M, Yamamoto M, Kokudo N, Sano K, Endo I, Unno M, Chijiiwa K, Horiguchi A, Kinoshita H, Oka M, Kubota K, Sugiyama M, Uemoto S, Shimada M, Suzuki Y, Inui K, Tazuma S, Furuse J, Yanagisawa A, Nakanuma Y, Kijima H, Takada T. Classification of biliary tract cancers established by the Japanese Society of Hepato-Biliary-Pancreatic Surgery: 3(rd) English edition. J Hepatobiliary Pancreat Sci. 2015;22:181-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 151] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 10. | Miyazaki M, Yoshitomi H, Miyakawa S, Uesaka K, Unno M, Endo I, Ota T, Ohtsuka M, Kinoshita H, Shimada K, Shimizu H, Tabata M, Chijiiwa K, Nagino M, Hirano S, Wakai T, Wada K, Isayama H, Okusaka T, Tsuyuguchi T, Fujita N, Furuse J, Yamao K, Murakami K, Yamazaki H, Kijima H, Nakanuma Y, Yoshida M, Takayashiki T, Takada T. Clinical practice guidelines for the management of biliary tract cancers 2015: the 2nd English edition. J Hepatobiliary Pancreat Sci. 2015;22:249-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 165] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 11. | Bagante F, Tran T, Spolverato G, Ruzzenente A, Buttner S, Ethun CG, Groot Koerkamp B, Conci S, Idrees K, Isom CA, Fields RC, Krasnick B, Weber SM, Salem A, Martin RC, Scoggins C, Shen P, Mogal HD, Schmidt C, Beal E, Hatzaras I, Vitiello G, IJzermans JN, Maithel SK, Poultsides G, Guglielmi A, Pawlik TM. Perihilar Cholangiocarcinoma: Number of Nodes Examined and Optimal Lymph Node Prognostic Scheme. J Am Coll Surg. 2016;222:750-759.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 59] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 12. | Kitagawa Y, Nagino M, Kamiya J, Uesaka K, Sano T, Yamamoto H, Hayakawa N, Nimura Y. Lymph node metastasis from hilar cholangiocarcinoma: audit of 110 patients who underwent regional and paraaortic node dissection. Ann Surg. 2001;233:385-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 222] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 13. | Chun YS, Pawlik TM, Vauthey JN. 8th Edition of the AJCC Cancer Staging Manual: Pancreas and Hepatobiliary Cancers. Ann Surg Oncol. 2018;25:845-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 546] [Article Influence: 68.3] [Reference Citation Analysis (0)] |
| 14. | Mantel HT, Wiggers JK, Verheij J, Doff JJ, Sieders E, van Gulik TM, Gouw AS, Porte RJ. Lymph Node Micrometastases are Associated with Worse Survival in Patients with Otherwise Node-Negative Hilar Cholangiocarcinoma. Ann Surg Oncol. 2015;22 Suppl 3:S1107-S1115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 15. | Nagakawa T, Kayahara M, Ikeda S, Futakawa S, Kakita A, Kawarada H, Matsuno M, Takada T, Takasaki K, Tanimura H, Tashiro S, Yamaoka Y. Biliary tract cancer treatment: results from the Biliary Tract Cancer Statistics Registry in Japan. J Hepatobiliary Pancreat Surg. 2002;9:569-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 16. | Tojima Y, Nagino M, Ebata T, Uesaka K, Kamiya J, Nimura Y. Immunohistochemically demonstrated lymph node micrometastasis and prognosis in patients with otherwise node-negative hilar cholangiocarcinoma. Ann Surg. 2003;237:201-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | Ogura Y, Kawarada Y. Surgical strategies for carcinoma of the hepatic duct confluence. Br J Surg. 1998;85:20-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Miyazaki M, Ito H, Nakagawa K, Ambiru S, Shimizu H, Okaya T, Shinmura K, Nakajima N. Parenchyma-preserving hepatectomy in the surgical treatment of hilar cholangiocarcinoma. J Am Coll Surg. 1999;189:575-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 89] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 19. | Kosuge T, Yamamoto J, Shimada K, Yamasaki S, Makuuchi M. Improved surgical results for hilar cholangiocarcinoma with procedures including major hepatic resection. Ann Surg. 1999;230:663-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 242] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 20. | Guglielmi A, Ruzzenente A, Campagnaro T, Valdegamberi A, Bagante F, Bertuzzo F, Conci S, Iacono C. Patterns and prognostic significance of lymph node dissection for surgical treatment of perihilar and intrahepatic cholangiocarcinoma. J Gastrointest Surg. 2013;17:1917-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 21. | Ocuin LM, Bağci P, Fisher SB, Patel SH, Kooby DA, Sarmiento JM, Cardona K, Russell MC, Staley CA, Volkan Adsay N, Maithel SK. Discordance between conventional and detailed lymph node analysis in resected biliary carcinoma at or above the cystic duct: are we understaging patients? Ann Surg Oncol. 2013;20:4298-4304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 22. | Oshiro Y, Sasaki R, Kobayashi A, Murata S, Fukunaga K, Kondo T, Oda T, Ohkohchi N. Prognostic relevance of the lymph node ratio in surgical patients with extrahepatic cholangiocarcinoma. Eur J Surg Oncol. 2011;37:60-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 23. | de Jong MC, Marques H, Clary BM, Bauer TW, Marsh JW, Ribero D, Majno P, Hatzaras I, Walters DM, Barbas AS, Mega R, Schulick RD, Choti MA, Geller DA, Barroso E, Mentha G, Capussotti L, Pawlik TM. The impact of portal vein resection on outcomes for hilar cholangiocarcinoma: a multi-institutional analysis of 305 cases. Cancer. 2012;118:4737-4747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 150] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 24. | Hakeem AR, Marangoni G, Chapman SJ, Young RS, Nair A, Hidalgo EL, Toogood GJ, Wyatt JI, Lodge PA, Prasad KR. Does the extent of lymphadenectomy, number of lymph nodes, positive lymph node ratio and neutrophil-lymphocyte ratio impact surgical outcome of perihilar cholangiocarcinoma? Eur J Gastroenterol Hepatol. 2014;26:1047-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Murakami Y, Uemura K, Sudo T, Hashimoto Y, Nakashima A, Kondo N, Sakabe R, Kobayashi H, Sueda T. Is para-aortic lymph node metastasis a contraindication for radical resection in biliary carcinoma? World J Surg. 2011;35:1085-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Kondo S, Nimura Y, Hayakawa N, Kamiya J, Nagino M, Uesaka K. Regional and para-aortic lymphadenectomy in radical surgery for advanced gallbladder carcinoma. Br J Surg. 2000;87:418-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 151] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 27. | Onoyama H, Ajiki T, Takada M, Urakawa T, Saitoh Y. Does radical resection improve the survival in patients with carcinoma of the gallbladder who are 75 years old and older? World J Surg. 2002;26:1315-1318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 28. | Yoshida T, Matsumoto T, Sasaki A, Morii Y, Aramaki M, Kitano S. Prognostic factors after pancreatoduodenectomy with extended lymphadenectomy for distal bile duct cancer. Arch Surg. 2002;137:69-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 115] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 29. | Araida T, Yoshikawa T, Azuma T, Ota T, Takasaki K, Hanyu F. Indications for pancreatoduodenectomy in patients undergoing lymphadenectomy for advanced gallbladder carcinoma. J Hepatobiliary Pancreat Surg. 2004;11:45-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 30. | Mao K, Liu J, Sun J, Zhang J, Chen J, Pawlik TM, Jacobs LK, Xiao Z, Wang J. Patterns and prognostic value of lymph node dissection for resected perihilar cholangiocarcinoma. J Gastroenterol Hepatol. 2016;31:417-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 31. | Ma WJ, Wu ZR, Hu HJ, Wang JK, Yin CH, Shi YJ, Li FY, Cheng NS. Extended Lymphadenectomy Versus Regional Lymphadenectomy in Resectable Hilar Cholangiocarcinoma. J Gastrointest Surg. 2019;Online ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 32. | Horgan AM, Amir E, Walter T, Knox JJ. Adjuvant therapy in the treatment of biliary tract cancer: a systematic review and meta-analysis. J Clin Oncol. 2012;30:1934-1940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 560] [Cited by in RCA: 513] [Article Influence: 39.5] [Reference Citation Analysis (0)] |
| 33. | Ghidini M, Tomasello G, Botticelli A, Barni S, Zabbialini G, Seghezzi S, Passalacqua R, Braconi C, Petrelli F. Adjuvant chemotherapy for resected biliary tract cancers: a systematic review and meta-analysis. HPB (Oxford). 2017;19:741-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 34. | Rangarajan K, Simmons G, Manas D, Malik H, Hamady ZZ. Systemic adjuvant chemotherapy for cholangiocarcinoma surgery: A systematic review and meta-analysis. Eur J Surg Oncol. 2020;46:684-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 35. | Guglielmi A, Ruzzenente A, Campagnaro T, Pachera S, Conci S, Valdegamberi A, Sandri M, Iacono C. Prognostic significance of lymph node ratio after resection of peri-hilar cholangiocarcinoma. HPB (Oxford). 2011;13:240-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 36. | Schwarz RE, Smith DD. Lymph node dissection impact on staging and survival of extrahepatic cholangiocarcinomas, based on U.S. population data. J Gastrointest Surg. 2007;11:158-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Ito K, Ito H, Allen PJ, Gonen M, Klimstra D, D'Angelica MI, Fong Y, DeMatteo RP, Brennan MF, Blumgart LH, Jarnagin WR. Adequate lymph node assessment for extrahepatic bile duct adenocarcinoma. Ann Surg. 2010;251:675-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 114] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 38. | Kambakamba P, Linecker M, Slankamenac K, DeOliveira ML. Lymph node dissection in resectable perihilar cholangiocarcinoma: a systematic review. Am J Surg. 2015;210:694-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 39. | Sakata J, Wakai T, Matsuda Y, Ohashi T, Hirose Y, Ichikawa H, Kobayashi T, Minagawa M, Kosugi S, Koyama Y, Akazawa K, Ajioka Y. Comparison of Number Versus Ratio of Positive Lymph Nodes in the Assessment of Lymph Node Status in Extrahepatic Cholangiocarcinoma. Ann Surg Oncol. 2016;23:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |