Published online Jun 7, 2020. doi: 10.3748/wjg.v26.i21.2821
Peer-review started: December 26, 2019
First decision: January 13, 2020
Revised: March 27, 2020
Accepted: May 26, 2020
Article in press: May 26, 2020
Published online: June 7, 2020
Untreated neonatal cholestasis can progress to liver cirrhosis and end stage liver disease in infancy due to prolonged hepatocyte and biliary tree injury and may require liver transplantation. Therefore, non-invasive evaluation of hepatic fibrosis is important in infants with cholestasis.
To investigate the usefulness of periportal thickening (PT) measured on liver magnetic resonance imaging (MRI) for the assessment of hepatic fibrosis in infants with cholestasis including biliary atresia (BA).
This retrospective study included infants less than 6 mo who underwent liver MRI and biopsy for the evaluation of infantile cholestasis. PT and spleen size were measured on MRI. Serologic assessment was based on aspartate transaminase to platelet ratio index (APRI). The grade of histopathologic fibrosis was assessed by the METAVIR grading system. Correlation and diagnostic performance of PT, normalized spleen size ratio (SR, using the upper normal size limit), and APRI for diagnosing hepatic fibrosis were obtained by receiver-operating characteristic (ROC) curve analysis.
A total of 155 patients were included, 110 of which were diagnosed with BA. Mean age at the time of MRI was 57.6 ± 34.4 d. There were positive correlations between fibrosis grade and PT and SR, even after adjusting age (all, P < 0.001). For the diagnosis of significant fibrosis (METAVIR grade F2-F4), the area under the ROC curve was 0.899 (95%CI: 0.840–0.941) for PT (cutoff, 4.2 mm), which was higher than 0.741 (95%CI: 0.664–0.808) for SR and 0.712 (95%CI: 0.634–0.782) for APRI (both, P < 0.001). For the diagnosis of cirrhosis (F4), the area under the ROC curve was the highest with SR as 0.790 (95%CI: 0.718–0.852).
Liver MRI findings of PT and SR are useful to assess clinically significant hepatic fibrosis (F2 and higher) in infants with cholestasis including BA.
Core tip: Non-invasive evaluation of hepatic fibrosis is important in infants with cholestasis including biliary atresia. Periportal thickening (PT) and normalized spleen size ratio (SR) measured on liver magnetic resonance imaging (MRI) showed positive correlations with hepatic fibrosis grade, even after adjusting age. For the diagnosis of significant fibrosis (F2-F4), PT using the cutoff of 4.2 mm showed higher diagnostic performance than SR or aspartate transaminase to platelet ratio index. For the diagnosis of cirrhosis (F4), SR was the best. Therefore, liver MRI findings of PT and SR can be useful to assess clinically significant hepatic fibrosis (F2 and higher) in infants with cholestasis.
- Citation: Lee MH, Shin HJ, Yoon H, Han SJ, Koh H, Lee MJ. Periportal thickening on magnetic resonance imaging for hepatic fibrosis in infantile cholestasis. World J Gastroenterol 2020; 26(21): 2821-2830
- URL: https://www.wjgnet.com/1007-9327/full/v26/i21/2821.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i21.2821
Neonatal cholestasis, an intrahepatic or extrahepatic disorder, is defined as persistent conjugated hyperbilirubinemia more than 2 wk and may be caused by various diseases[1]. Untreated neonatal cholestasis can progress to liver cirrhosis or end stage liver disease in infants due to prolonged hepatocyte and biliary tree injury and may require liver transplantation. Ultimately, the degree of hepatic fibrosis in infantile cholestasis is a major determinant of the patient's outcome[2]. For biliary atresia (BA), a previous study reported that severe hepatic fibrosis at the time of Kasai operation, irrespective of age, carried a poor outcome and the degree of hepatic fibrosis, as well as the time of operation, is an important factor for survival after Kasai operation[3,4]. Therefore, knowing the degree of hepatic fibrosis before the operation can help predict the treatment methods and outcomes of patients and prepare for subsequent liver transplants.
Hepatic fibrosis in neonates and infants can be measured by serology, ultrasound elastography, and liver biopsy[5]. Ultrasound elastography is non-invasive, but has the disadvantage that the accuracy of the test may be impaired if the practitioner lacks experience or if ascites exists in the perihepatic space. Liver biopsy is necessary for confirmation of biliary disease including BA as an accurate diagnostic tool and assessment of hepatic fibrosis, but it is an invasive method and has the disadvantage that it cannot be repeatedly performed due to complications of the test itself[6,7].
Magnetic resonance imaging (MRI) has been reported to be useful for differential diagnosis of cholestatic jaundice in infants with high resolution and quantitative evaluation[8]. Several previous studies investigating diagnosis of BA by MRI evaluated visibility of the extrahepatic bile duct on MR cholangiopancreatography (MRCP)[9-11], but it has been reported that the accuracy of BA diagnosis was improved when the triangular cord thickness or periportal thickening (PT), which is usually measured on ultrasonography, was applied to MRI[12]. We thought if these MRI findings were associated with hepatic fibrosis, degree of hepatic fibrosis could be predicted noninvasively and reproducibly by making less susceptible to practitioner’s skill than ultrasonography. However, there was no previous study showing that MRI findings including PT reflect the degree of hepatic fibrosis in patients with infantile cholestasis. Therefore, we investigated whether the PT and spleen size measured on MRI is associated with pathologically assessed hepatic fibrosis in patients with infantile cholestasis.
This retrospective study was approved by our Institutional Review Board, with a waiver for informed consent for reviewing medical records and images of the patients. However, written informed consent for sedation and MRI was received before each examination as routine clinical practice.
We included all consecutive patients between July 2009 and October 2017 who (1) underwent both liver MRI and liver biopsy to evaluate the cause of infantile cholestasis which was defined as prolonged hyperbilirubinemia longer than 2 wk; (2) were younger than 6 mo at the time of MRI as identified by chart review; and (3) had an interval between liver MRI and liver biopsy within 1 mo. We do liver MRI and liver biopsy in infants with persistent jaundice after conservative treatment. We also do liver biopsy during BA and choledochal cyst operation in neonates and infants as a routine protocol. Patient’s sex and age at the time of MRI, laboratory results at the time of MRI and etiology of cholestasis confirmed by pathology and/or operative cholangiography (BA vs non-BA) were evaluated through medical chart review.
All MR images were obtained using a 1.5-T unit (Intera Achieva; Philips Healthcare) with a cardiac coil and patients were sedated by general anesthesia under the supervision of anesthesiologists. MR sequences used at our hospital included respiratory gated T2-weighted fast spine echo (FSE) axial, T1-weighted spin echo axial, T2-weighted single shot FSE sagittal, and respiratory gated FSE 3 dimensional MRCP coronal images. Repetition time/echo time was 2000/100 ms for FSE T2, 600/11 for T1, 700/80 for single shot FSE T2, and 1500/650 for MRCP. Matrix was 256 × 256. Section thickness was 2 mm with 1 mm gap for MRCP and 3 mm without gap for the others. Field of view was 18-24 cm. The total scan time for our protocol was approximately 15 -20 min for most of the cases.
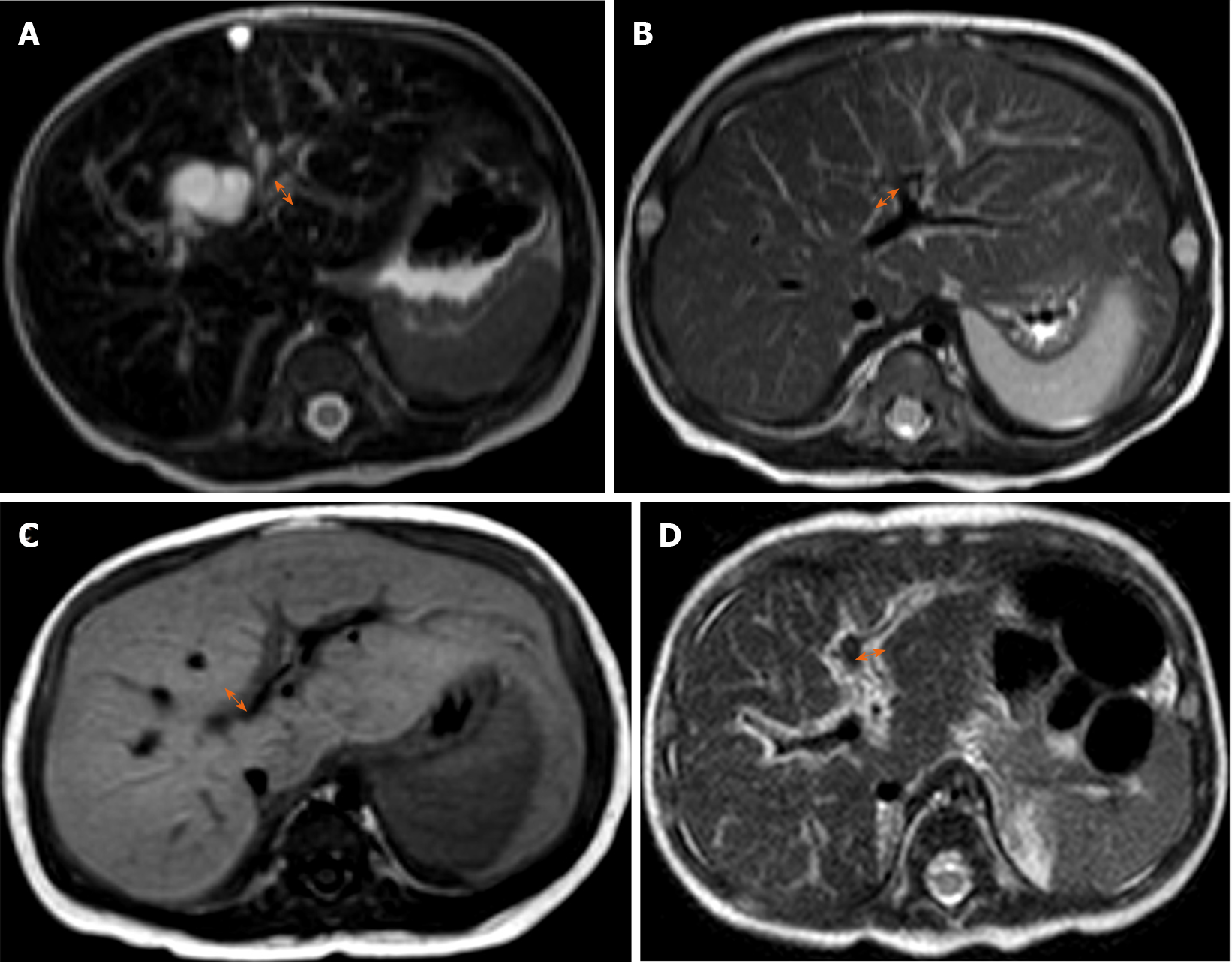
Two radiologists (with 15 and 4 years of clinical experience in pediatric radiology, respectively) who were unaware of the final diagnosis analyzed the MR images in consensus on the picture archiving and communication system (Centricity; GE Healthcare). The PT measured on MRI was defined as the maximal thickness of periportal signal change on MRI which was measured (1) either along the right main or left main portal vein; and (2) on either side of the portal vein where its thickness was the greatest. The maximum thickness of the periportal signal change was separately measured on T1-weighted axial, T2-weighted axial and T2-weighted sagittal images, and then the largest value among the three measurements was selected (Figure 1)[12]. The size of the spleen was defined as the maximum length of spleen either measured on T2-weighted axial or sagittal images, and the larger value was used for analysis. Then we analyzed the normalized spleen size ratio (SR)[13] by calculating the maximum spleen length divided by the upper normal limit of size according to the patient’s age and sex[14].
Basically, ultrasonography-guided liver biopsy was performed using an 18-gauge core biopsy needle (TSK Stericut Standard type with Co-axial; TSK) with a free-hand technique under sedation supervised by anesthesiologists in patient with infantile cholestasis. However, if BA or choledochal cyst was strongly suspected, liver biopsy was performed during operation.
All specimens were evaluated for histopathologic diagnosis of cholestasis and grading hepatic fibrosis by hepatobiliary pathologists. The fibrosis grades of pathologic specimens of all cases were evaluated by METAVIR fibrosis grade, ranging from grade F0 to F4 (F0, no fibrosis; F1, portal fibrosis without septa; F2, portal fibrosis with few septa; F3, numerous septa without cirrhosis; and F4, cirrhosis). Clinically significant fibrosis was defined as grade F2 to F4 and advanced fibrosis was defined as grade F3 to F4[15,16].
Laboratory results included aspartate aminotransferase (AST), alanine transaminase and platelet count. We calculated the AST-to-platelet ratio index (APRI), known as a noninvasive fibrosis score by serology, which is calculated as [(AST/upper limit of normal AST) × 100]/platelet count (109/L).
Statistical analyses were performed using SPSS version 23 (IBM Corp.). The independent two-sample t test for continuous variables and χ2 test for categorical variables were used. Analysis of variance was used for the comparison of parameters with different hepatic fibrosis grades. Correlation between laboratory or imaging parameters and hepatic fibrosis grades was analyzed using Kendall’s tau correlation coefficient. The diagnostic performances of parameters for clinically significant or advanced fibrosis and cirrhosis were evaluated by a receiver operating characteristic (ROC) curve analysis. The optimal cut-off values from the ROC curves were determined by the Youden index. The area under the ROC curve (AUC) of the parameters was compared with a Z test. In all tests, a P value less than 0.05 was considered statistically significant.
During the study period, 155 patients with infantile cholestasis were included. Baseline characteristics of our study are summarized in Table 1. The mean age at the time of MRI was 57.6 ± 34.4 d and 94 patients (60.6%) were female. Among the patients, 110 infants (71%) were diagnosed with BA and the remaining 45 (29%) were diagnosed with diseases other than BA by pathologic confirmation. The final diagnoses of the non-BA patients were choledochal cyst (n = 29), hepatitis (n = 8), paucity of intrahepatic bile ducts (n = 3), and other metabolic diseases (n = 5).
| Overall (n = 155) | BA (n = 110) | Non-BA (n = 45) | P value | |
| Demographics | ||||
| Age (d) | 57.6 ± 34.4 | 59.7 ± 30.3 | 52.4 ± 42.8 | 0.303 |
| Gender (M:F) | 61:94 | 41:69 | 20:25 | 0.470 |
| Laboratory results | ||||
| APRI | 1.32 ± 1.61 | 1.47 ± 1.66 | 0.95 ± 1.42 | 0.069 |
| Imaging findings | ||||
| PT (mm) | 4.9 ± 1.8 | 5.7 ± 1.4 | 2.9 ± 0.9 | < 0.001 |
| SR | 1.02 ± 0.20 | 1.07 ± 0.19 | 0.91 ± 0.17 | < 0.001 |
| METAVIR grades (Patients’ number) | ||||
| F0/F1/F2/F3/F4 | 32/10/66/37/10 | 1/4/59/37/9 | 31/6/7/0/1 | < 0.001 |
The mean age at the time of MRI was not different between the BA and non-BA groups (P = 0.303). The PT and SR were significantly higher in the BA group than in the non-BA group (P < 0.001), but ARPI was not different (Table 1). The number of patients with clinically significant fibrosis (F2–F4) was 113 (72.9%), with 105 from the BA group and 8 from the non-BA group. There were 47 patients (30.3%) with advanced fibrosis (F3–F4), with 46 from the BA group and 1 from the non-BA group. The distribution of each hepatic fibrosis grade is detailed in Table 1. There were 9 BA and 1 non-BA patients with liver cirrhosis (F4).
Table 2 summarizes the results of parameters in each fibrosis grading group. Age, APRI, PT, and SR were different in the fibrosis group comparison. Correlation analysis also demonstrated positive correlation between these parameters and fibrosis grades (all, P < 0.001). However, after adjusting age, partial correlation results demonstrated that only PT (τ = 0.518, P < 0.001) and SR (τ = 0.340, P < 0.001) showed positive correlations with the hepatic fibrosis grades. Representative images of each hepatic fibrosis grade are shown in Figure 2.
| Age (d) | APRI | PT (mm) | SR | |
| Hepatic fibrosis group comparison | ||||
| F0 (n = 32) | 40.8 ± 34.8 | 1.12 ± 1.63 | 3.1 ± 1.1 | 0.89 ± 0.15 |
| F1 (n = 10) | 60.6 ± 44.0 | 0.46 ± 0.38 | 3.3 ± 1.3 | 0.94 ± 0.20 |
| F2 (n = 66) | 52.3 ± 31.0 | 1.14 ± 1.23 | 5.2 ± 1.4 | 1.01 ± 0.17 |
| F3 (n = 37) | 71.9 ± 28.6 | 2.00 ± 2.27 | 5.9 ± 1.6 | 1.12 ± 0.20 |
| F4 (n = 10) | 89.7 ± 26.6 | 1.52 ± 0.55 | 5.7 ± 1.6 | 1.21 ± 0.17 |
| P value | < 0.001 | 0.026 | < 0.001 | < 0.001 |
| Correlation analysis | ||||
| Overall correlation | ||||
| τ | 0.313 | 0.326 | 0.448 | 0.361 |
| P value | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
| Partial correlation with adjusting age | ||||
| τ | 0.071 | 0.518 | 0.340 | |
| P value | 0.405 | < 0.001 | < 0.001 | |
The diagnostic performances in diagnosing clinically significant fibrosis (F0–F1 vs F2–F4), advanced fibrosis (F0–F2 vs F3–F4), and cirrhosis (F0-F3 vs F4) for each parameter are shown in Table 3. The AUCs of PT were 0.899 (95%CI: 0.840–0.941) using a cutoff value of 4.2 mm for clinically significant fibrosis and 0.734 (95%CI: 0.657–0.801) with a cutoff of 5.3 mm for advanced fibrosis (P < 0.001). The AUCs of SR and APRI were 0.741 with a cutoff value of 0.85 and 0.712 with a cutoff of 0.44 for the diagnosis of clinically significant fibrosis, respectively. For diagnosing advanced fibrosis, the AUC of SR was 0.742 with a cutoff value of 1.1 and that of APRI was 0.724 with a cutoff value of 0.78. For diagnosing cirrhosis, the AUC was the highest with SR as 0.790 (95%CI: 0.718-0.852) with a cutoff value of 1.04. However, the AUC of PT was not significant (P = 0.058).
| Variable | Cutoff value | Sensitivity (%) | Specificity (%) | AUC | 95%CI | P value |
| Clinically significant fibrosis (F2-F4) | ||||||
| APRI | 0.44 | 84.1 | 59.5 | 0.712 | 0.634-0.782 | 0.001 |
| PT | 4.2 mm | 81.4 | 85.7 | 0.899 | 0.840-0.941 | < 0.001 |
| SR | 0.85 | 85.0 | 54.8 | 0.741 | 0.664-0.808 | < 0.001 |
| Advanced fibrosis (F3-F4) | ||||||
| APRI | 0.78 | 78.7 | 63.0 | 0.724 | 0.647-0.793 | < 0.001 |
| PT | 5.3 mm | 66.0 | 71.3 | 0.734 | 0.657-0.801 | < 0.001 |
| SR | 1.1 | 61.7 | 78.7 | 0.742 | 0.666-0.809 | < 0.001 |
| Cirrhosis (F4) | ||||||
| APRI | 1.24 | 90.0 | 70.3 | 0.715 | 0.637-0.785 | 0.002 |
| PT | 5.3 mm | 70.0 | 62.1 | 0.656 | 0.675-0.730 | 0.058 |
| SR | 1.04 | 90.0 | 61.4 | 0.790 | 0.718-0.852 | < 0.001 |
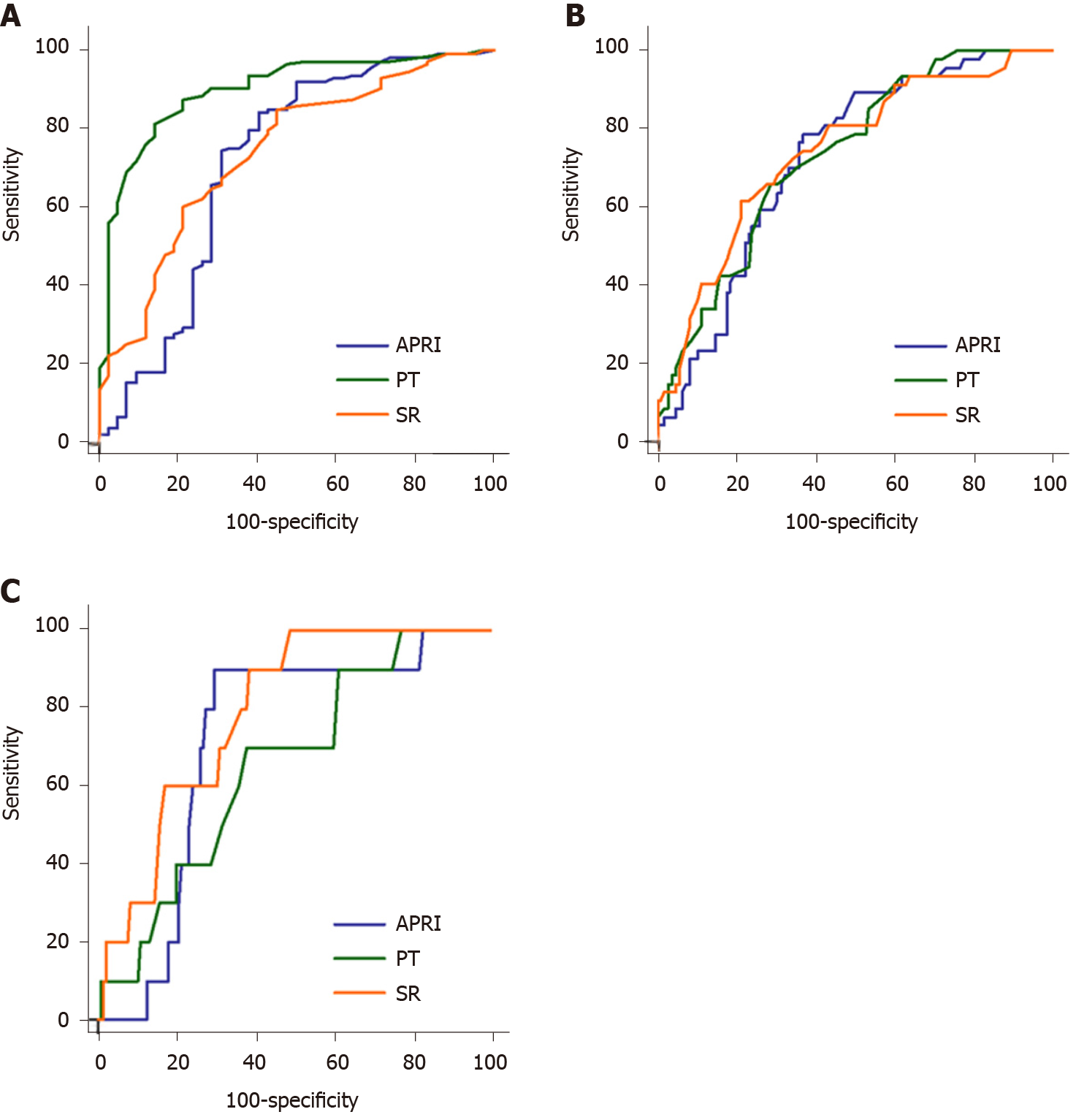
For diagnosing clinically significant fibrosis, there was a significant difference in the AUC between PT and SR (difference between area, 0.158; P < 0.001) and PT and APRI (difference between area, 0.186; P < 0.001), but no significant difference between SR and APRI (difference between area, 0.029; P = 0.613). The AUC of PT (0.899) was significantly higher than those of SR (0.741) and APRI (0.712) (Figure 3A).
In contrast, there were no significant differences in the AUCs between PT, SR and APRI for diagnosing advanced fibrosis. The differences between areas were 0.008 (P = 0.873) between PT and SR, 0.009 (P = 0.859) between PT and APRI, and 0.018 (P = 0.724) between SR and APRI (Figure 3B).
In cirrhosis, the AUC with SR was higher than that with PT (difference between area, 0.135; P = 0.041), but not that with APRI (difference between area, 0.075; P = 0.427). There was no significant difference in the AUCs between PT and APRI (difference of 0.060, P = 0.626) (Figure 3C).
Serologic marker of APRI and splenomegaly are well known markers for the evaluation of hepatic fibrosis in adults, but there is limited study about these parameters in infants. Moreover, this is the first study suggested the usefulness of PT for investigating hepatic fibrosis in infants with cholestasis. We demonstrated that PT measured on MRI showed positive correlation with pathologically assessed hepatic fibrosis grades and had good performance to diagnose both clinically significant fibrosis and advanced fibrosis with cutoff values of 4.2 mm and 5.3 mm, respectively. In addition, hepatic fibrosis was also significantly correlated with SR and APRI, but the diagnostic performance of PT measured on MRI for clinically significant hepatic fibrosis was superior to that of SR and APRI. Moreover, after adjusting age, only PT and SR showed positive correlations with the hepatic fibrosis grades. Therefore, our study shows that MRI findings of PT and SR are useful in predicting the degree of hepatic fibrosis in infants with cholestasis who cannot easily undergo liver biopsy.
To determine grade of hepatic fibrosis in patients with neonatal cholestasis, liver biopsy is the gold standard. However, it is invasive and can result in severe, life-threatening complications in infants and neonates[17-19]. Moreover, this procedure is associated with significant sampling error[20]. Therefore, several methods for non-invasive assessment of hepatic fibrosis in infants and neonates have been proposed. APRI by laboratory test could be a reliable method to assess advanced fibrosis (F3–F4) and liver cirrhosis (F4) at the time of Kasai operation and postoperative follow-up care[21]. APRI has shown significant correlation with hepatic fibrosis grade in children with chronic liver disease[5]. These studies are consistent with our study, in which APRI of our patients showed significant correlation with hepatic fibrosis grade and good performance to diagnose clinically significant fibrosis and advanced fibrosis. However, APRI was not correlated with hepatic fibrosis grades after adjusting age.
Another tool is transient elastography, which could suggest optimal time of Kasai operation in patients with BA[22]. There have been several reports on the utility of this technique to evaluate hepatic fibrosis and cirrhosis in patients with BA[23-25]. However, there are limitations in the aforementioned studies in that they could not evaluate the diagnostic performance of clinically significant fibrosis (F2–F4) and operator dependency of this method[7,23,25,26].
Triangular cord sign or PT was a concept originally designed for ultrasound, which is represented by triangular cone-shaped fibrous tissue in the bifurcation of the portal vein at the porta hepatis. However, due to operator dependence on ultrasound, the concept of PT measured on MRI has been proposed and defined as a thickening of periportal signal change on T1 or T2-weighted images[27,28]. A recent study also proposed that an MRI-based decision tree for diagnosis of BA using PT on MRI with a cutoff value of 5.1 mm showed high sensitivity (97.3%) and specificity (94.8%)[12]. PT measured on ultrasonography was associated with hepatic fibrosis in BA, but there was no significant association between PT and hepatic fibrosis grade[23]. And there was a few previous study on whether PT measured on MRI is associated with hepatic fibrosis in patients with neonatal cholestasis with relatively small population[28]. Because this was a retrospective study and there were limited data about PT on ultrasonography in non-biliary atresia patients, we could not evaluate the diagnostic performance of PT on ultrasonography compared with that on MRI. However, as ultrasonography is a more available and easily accessible technique, further study is needed for this topic.
Degree of hepatic fibrosis at time of Kasai operation is known to be one of the prognostic factors after Kasai operation in patients with BA[3,29]. Many previous studies have focused on age at time of surgery, but a few studies have suggested that severe fibrosis at the time of Kasai operation, irrespective of age, carries a poor outcome[3,30]. Severe fibrosis was associated with unsuccessful outcome of Kasai operation without biliary drainage and all patients with cirrhosis had no biliary drainage after Kasai operation in a previous study[4]. Therefore, in patients with advanced fibrosis and liver cirrhosis at the time of diagnosis, early liver transplantation may improve prognosis over Kasai portoenterostomy[23,31]. A few retrospective studies suggested that anti-inflammatory and immunomodulatory drugs such as steroids might alter progression of hepatic fibrosis and improve biliary drainage and survival rate of native liver of patients[32]. Considering that hepatic fibrosis progresses even after surgery, degree of hepatic fibrosis both before and after surgery could be associated with postoperative complications and prognosis in BA. A prospective study is needed on whether the imaging findings of PT are also relevant to the patient's prognosis.
There were several limitations in this study. First, this study was performed retrospectively, so inherent possibility of bias exists. Second, reproducibility of PT measurements could be problematic, even though we used consensus reading. Third, we could not evaluate the pathologic finding of PT. The meaning of PT as a direct or indirect finding of hepatic or periportal fibrosis should be evaluated. Finally, the number of patients with F1 and F4 was relatively small, leading to unreliable data from these groups.
In conclusion, liver MRI findings of PT and SR can be useful to assess hepatic fibrosis in infants with cholestasis including BA. The finding of PT with a cutoff value of 4.2 mm has better diagnostic performance to predict clinically significant fibrosis than either SR or APRI. And SR had the highest diagnostic performance value for differentiating cirrhosis. The degree of hepatic fibrosis is an important prognostic factor after Kasai operation in patients with BA. Therefore, PT and SR measured on MRI may help predict prognosis of BA and suggest more effective non-invasive treatment options.
Untreated neonatal cholestasis can progress to liver cirrhosis or end stage liver disease in infants due to prolonged hepatocyte and biliary tree injury and may require liver transplantation. Therefore, non-invasive evaluation of hepatic fibrosis is important in infants with cholestasis including biliary atresia.
Some serologic tests and splenomegaly are known markers for the evaluation of hepatic fibrosis in adults, but there is limited study about these parameters in infants. Periportal thickening (PT) was only considered as the finding of biliary atresia and has not been evaluated for assessing hepatic fibrosis in infants with cholestasis.
We investigated whether the PT and spleen size measured on magnetic resonance imaging (MRI) is associated with pathologically assessed hepatic fibrosis in patients with infantile cholestasis including biliary atresia.
This retrospective study included infants less than 6 mo with liver MRI and biopsy for the evaluation of infantile cholestasis. Not only PT and spleen size measured on MRI, but also serologic assessment of aspartate transaminase to platelet ratio index (APRI) were evaluated and compared with histopathologic METAVIR grading of hepatic fibrosis. We calculated normalized spleen size ratio (SR) using the upper normal size limit.
A total of 155 patients were evaluated including 110 with biliary atresia. Mean age at the time of MRI was 57.6 ± 34.4 d. There were positive correlations between fibrosis grade and PT and SR, even after adjusting age. The finding of PT with a cutoff value of 4.2 mm has better diagnostic performance to predict clinically significant fibrosis than either SR or APRI. And SR had the highest diagnostic performance value for differentiating cirrhosis.
Liver MRI findings of PT and SR are useful to assess clinically significant hepatic fibrosis (F2 and higher) in infants with cholestasis including biliary atresia.
The degree of hepatic fibrosis is an important prognostic factor after Kasai operation in patients with biliary atresia. Therefore, PT and SR measured on MRI may help predict prognosis of biliary atresia and suggest more effective non-invasive treatment options.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: South Korea
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Karaksy H, Nobile S, Yuan YS S-Editor: Wang JL L-Editor: A E-Editor: Ma YJ
| 1. | Girard M, Lacaille F. Diagnosis of neonatal cholestasis. Annales Nestlé. 2008;66:109-120. [Cited in This Article: ] |
| 2. | Sokol RJ. Liver cell injury and fibrosis. J Pediatr Gastroenterol Nutr. 2002;35 Suppl 1:S7-10. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 3. | Weerasooriya VS, White FV, Shepherd RW. Hepatic fibrosis and survival in biliary atresia. J Pediatr. 2004;144:123-125. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 80] [Cited by in F6Publishing: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 4. | Salzedas-Netto AA, Chinen E, de Oliveira DF, Pasquetti AF, Azevedo RA, da Silva Patricio FF, Cury EK, Gonzalez AM, Vicentine FP, Martins JL. Grade IV fibrosis interferes in biliary drainage after Kasai procedure. Transplant Proc. 2014;46:1781-1783. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | de Lédinghen V, Le Bail B, Rebouissoux L, Fournier C, Foucher J, Miette V, Castéra L, Sandrin L, Merrouche W, Lavrand F, Lamireau T. Liver stiffness measurement in children using FibroScan: feasibility study and comparison with Fibrotest, aspartate transaminase to platelets ratio index, and liver biopsy. J Pediatr Gastroenterol Nutr. 2007;45:443-450. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 185] [Cited by in F6Publishing: 169] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 6. | Castéra L, Vergniol J, Foucher J, Le Bail B, Chanteloup E, Haaser M, Darriet M, Couzigou P, De Lédinghen V. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology. 2005;128:343-350. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1796] [Cited by in F6Publishing: 1760] [Article Influence: 92.6] [Reference Citation Analysis (0)] |
| 7. | Kettaneh A, Marcellin P, Douvin C, Poupon R, Ziol M, Beaugrand M, de Lédinghen V. Features associated with success rate and performance of FibroScan measurements for the diagnosis of cirrhosis in HCV patients: a prospective study of 935 patients. J Hepatol. 2007;46:628-634. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 187] [Cited by in F6Publishing: 208] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 8. | Takaya J, Nakano S, Imai Y, Fujii Y, Kaneko K. Usefulness of magnetic resonance cholangiopancreatography in biliary structures in infants: a four-case report. Eur J Pediatr. 2007;166:211-214. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Han SJ, Kim MJ, Han A, Chung KS, Yoon CS, Kim D, Hwang EH. Magnetic resonance cholangiography for the diagnosis of biliary atresia. J Pediatr Surg. 2002;37:599-604. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 53] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 10. | Liu B, Cai J, Xu Y, Peng X, Zheng H, Huang K, Yang J. Three-dimensional magnetic resonance cholangiopancreatography for the diagnosis of biliary atresia in infants and neonates. PLoS One. 2014;9:e88268. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 38] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 11. | Sung S, Jeon TY, Yoo SY, Hwang SM, Choi YH, Kim WS, Choe YH, Kim JH. Incremental Value of MR Cholangiopancreatography in Diagnosis of Biliary Atresia. PLoS One. 2016;11:e0158132. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 12. | Kim YH, Kim MJ, Shin HJ, Yoon H, Han SJ, Koh H, Roh YH, Lee MJ. MRI-based decision tree model for diagnosis of biliary atresia. Eur Radiol. 2018;28:3422-3431. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Yoon H, Shin HJ, Kim MJ, Han SJ, Koh H, Kim S, Lee MJ. Predicting gastroesophageal varices through spleen magnetic resonance elastography in pediatric liver fibrosis. World J Gastroenterol. 2019;25:367-377. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 18] [Cited by in F6Publishing: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 14. | Megremis SD, Vlachonikolis IG, Tsilimigaki AM. Spleen length in childhood with US: normal values based on age, sex, and somatometric parameters. Radiology. 2004;231:129-134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 124] [Cited by in F6Publishing: 124] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 15. | Imbert-Bismut F, Ratziu V, Pieroni L, Charlotte F, Benhamou Y, Poynard T; MULTIVIRC Group. Biochemical markers of liver fibrosis in patients with hepatitis C virus infection: a prospective study. Lancet. 2001;357:1069-1075. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1066] [Cited by in F6Publishing: 1113] [Article Influence: 48.4] [Reference Citation Analysis (0)] |
| 16. | Alkhouri N, Sedki E, Alisi A, Lopez R, Pinzani M, Feldstein AE, Nobili V. Combined paediatric NAFLD fibrosis index and transient elastography to predict clinically significant fibrosis in children with fatty liver disease. Liver Int. 2013;33:79-85. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 17. | Castéra L, Nègre I, Samii K, Buffet C. Pain experienced during percutaneous liver biopsy. Hepatology. 1999;30:1529-1530. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 202] [Cited by in F6Publishing: 200] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 18. | Dienstag JL. The role of liver biopsy in chronic hepatitis C. Hepatology. 2002;36:S152-S160. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 19. | Poynard T, Ratziu V, Bedossa P. Appropriateness of liver biopsy. Can J Gastroenterol. 2000;14:543-548. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 130] [Cited by in F6Publishing: 140] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 20. | Bedossa P, Dargère D, Paradis V. Sampling variability of liver fibrosis in chronic hepatitis C. Hepatology. 2003;38:1449-1457. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1193] [Cited by in F6Publishing: 1334] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 21. | Kim SY, Seok JY, Han SJ, Koh H. Assessment of liver fibrosis and cirrhosis by aspartate aminotransferase-to-platelet ratio index in children with biliary atresia. J Pediatr Gastroenterol Nutr. 2010;51:198-202. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 62] [Cited by in F6Publishing: 59] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 22. | Lee HY, Park YA, Han SJ, Koh H. Tentative Proposal of Optimal Timing of Kasai Operation for Biliary Atresia Based on Fibroscan Results. Korean J Pediatr Gastroenterol Nutr. 2011;14:74-80. [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 23. | Shin NY, Kim MJ, Lee MJ, Han SJ, Koh H, Namgung R, Park YN. Transient elastography and sonography for prediction of liver fibrosis in infants with biliary atresia. J Ultrasound Med. 2014;33:853-864. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 24. | Chang HK, Park YJ, Koh H, Kim SM, Chung KS, Oh JT, Han SJ. Hepatic fibrosis scan for liver stiffness score measurement: a useful preendoscopic screening test for the detection of varices in postoperative patients with biliary atresia. J Pediatr Gastroenterol Nutr. 2009;49:323-328. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 46] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Fraquelli M, Rigamonti C, Casazza G, Conte D, Donato MF, Ronchi G, Colombo M. Reproducibility of transient elastography in the evaluation of liver fibrosis in patients with chronic liver disease. Gut. 2007;56:968-973. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 629] [Cited by in F6Publishing: 618] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 26. | de Lédinghen V, Vergniol J. Transient elastography (FibroScan). Gastroenterol Clin Biol. 2008;32:58-67. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 177] [Cited by in F6Publishing: 186] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 27. | Kim MJ, Park YN, Han SJ, Yoon CS, Yoo HS, Hwang EH, Chung KS. Biliary atresia in neonates and infants: triangular area of high signal intensity in the porta hepatis at T2-weighted MR cholangiography with US and histopathologic correlation. Radiology. 2000;215:395-401. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 64] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 28. | Avni FE, Segers V, De Maertelaer V, Cadranel S, Dassonville M, Delaet MH, Nicaise N, Metens T. The evaluation by magnetic resonance imaging of hepatic periportal fibrosis in infants with neonatal cholestasis: preliminary report. J Pediatr Surg. 2002;37:1128-1133. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 29. | Shteyer E, Ramm GA, Xu C, White FV, Shepherd RW. Outcome after portoenterostomy in biliary atresia: pivotal role of degree of liver fibrosis and intensity of stellate cell activation. J Pediatr Gastroenterol Nutr. 2006;42:93-99. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Davenport M, Kerkar N, Mieli-Vergani G, Mowat AP, Howard ER. Biliary atresia: the King's College Hospital experience (1974-1995). J Pediatr Surg. 1997;32:479-485. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 166] [Cited by in F6Publishing: 160] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 31. | Pakarinen MP, Rintala RJ. Surgery of biliary atresia. Scand J Surg. 2011;100:49-53. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 32. | Dillon PW, Owings E, Cilley R, Field D, Curnow A, Georgeson K. Immunosuppression as Adjuvant Therapy for Biliary Atresia. J Pediatr Surg. 2001;36:80-85. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 64] [Article Influence: 2.8] [Reference Citation Analysis (0)] |