Published online May 28, 2020. doi: 10.3748/wjg.v26.i20.2533
Peer-review started: December 23, 2019
First decision: April 1, 2020
Revised: April 7, 2020
Accepted: April 29, 2020
Article in press: April 29, 2020
Published online: May 28, 2020
Processing time: 157 Days and 10.5 Hours
Stress-induced gastric mucosal lesion (SGML) is one of the most common visceral complications after trauma. Exploring the nervous mechanisms of SGML has become a research hotspot. Restraint water-immersion stress (RWIS) can induce GML and has been widely used to elucidate the nervous mechanisms of SGML. It is believed that RWIS-induced GML is mainly caused by the enhanced activity of vagal parasympathetic nerves. Many central nuclei, such as the dorsal motor nucleus of the vagus, nucleus of the solitary tract, supraoptic nucleus and paraventricular nucleus of the hypothalamus, mediodorsal nucleus of the thalamus, central nucleus of the amygdala and medial prefrontal cortex, are involved in the formation of SGML in varying degrees. Neurotransmitters/neuromodulators, such as nitric oxide, hydrogen sulfide, vasoactive intestinal peptide, calcitonin gene-related peptide, substance P, enkephalin, 5-hydroxytryptamine, acetylcholine, catecholamine, glutamate, γ-aminobutyric acid, oxytocin and arginine vasopressin, can participate in the regulation of stress. However, inconsistent and even contradictory results have been obtained regarding the actual roles of each nucleus in the nervous mechanism of RWIS-induced GML, such as the involvement of different nuclei with the time of RWIS, the different levels of involvement of the sub-regions of the same nucleus, and the diverse signalling molecules, remain to be further elucidated.
Core tip: The nervous mechanisms of gastric mucosal lesion in rats subjected to restraint water-immersion stress were investigated. Abnormal regulation of the enteric nervous system, mainly due to the enhanced activity of the parasympathetic nervous system, can induce gastrointestinal dysfunction. The central nucleus such as dorsal motor nucleus of the vagus, nucleus ambiguous, nucleus of the solitary tract, paraventricular nucleus, supraoptic nucleus, mediodorsal nucleus of the thalamus, central nucleus of the amygdala and medial prefrontal cortex are all involved in the formation of stress-induced gastric mucosal lesion. Nitric oxide, 5-hydroxytryptamine, hydrogen sulfide, calcitonin gene-related peptide, vasoactive intestinal peptide, acetylcholine, catecholamine, glutamate, γ-aminobutyric acid, oxytocin and arginine vasopressin may be involved in the physiological process.
- Citation: Zhao DQ, Xue H, Sun HJ. Nervous mechanisms of restraint water-immersion stress-induced gastric mucosal lesion. World J Gastroenterol 2020; 26(20): 2533-2549
- URL: https://www.wjgnet.com/1007-9327/full/v26/i20/2533.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i20.2533
Stress is defined as a disordered state of homeostasis caused by internal or external noxious stimuli, and the gastrointestinal tract is the main organ that responds to stress[1,2]. Stress-induced gastric mucosal lesion (SGML) is an acute GML mainly characterized by inflammatory erosion and gastrointestinal bleeding, which has a high mortality rate and is one of the most common visceral complications following trauma[3-5]. Thus, exploring the mechanisms that underlie the occurrence and development of SGML as well as the identification of more effective, safer and more affordable drugs have become one of the research hotspots in modern biology.
There are nerve plexuses in the gastrointestinal tract, with great independence in food digestion, nutrient uptake and waste removal, while the central nervous system (CNS) provides external nerves that regulate and control these functions[6]. Neural control of the gut is hierarchic with four basic levels of integrative organization. The first level is the enteric nervous system (ENS) that behaves like a local minibrain; the second level is in the prevertebral sympathetic ganglia; the third and fourth levels are within the CNS. Sympathetic and parasympathetic signals to the digestive tract exist at level 3, and represent the final common pathways for information outflow from the CNS to the gut. The fourth level includes higher brain centers that provide input for integrative functions at level 3[7,8]. The nervous mechanisms of SGML have been explored by many scholars from all over the world using methods such as basic physiology, electrophysiology, immunocytochemistry and pharmacology on the basis of neurotransmitters (neuromodulators), receptors, agonists and blockers[9-18]. In this study, the nervous mechanism of restraint water-immersion stress (RWIS)-induced gastric dysfunction was investigated, in order to provide ideas to further reveal the nervous mechanism underlying the occurrence and development of SGML.
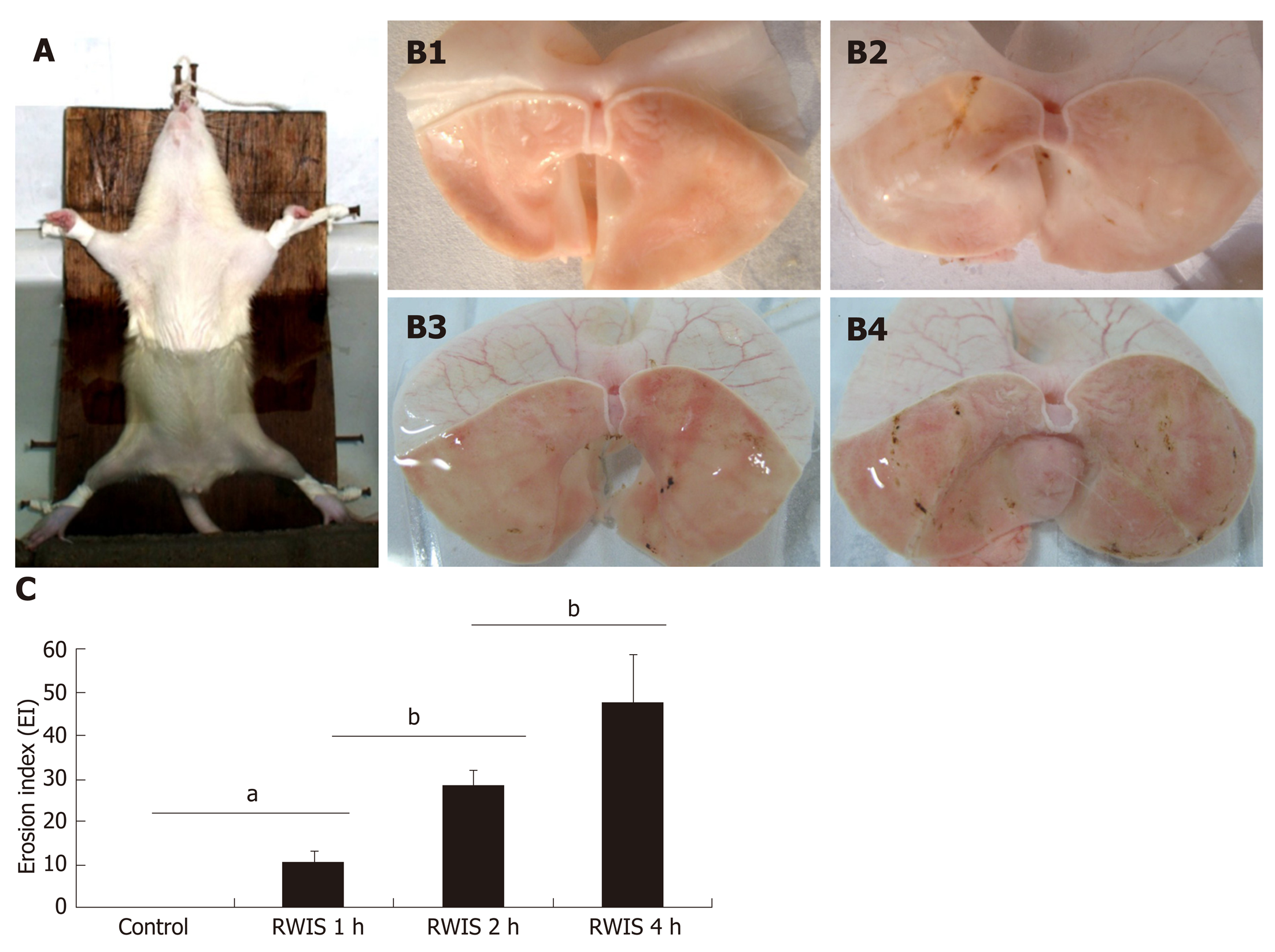
RWIS was first discovered by Takagi, and has been widely used to elucidate the mechanism of SGML and screen for potential therapeutic drugs[19-21]. RWIS is a powerful multiple stress model that integrates psychological factors (e.g., fear, anger, anxiety and despair) and physiological factors (e.g., hunger, struggle and cold water). During RWIS, the anesthetized rats were fixed on a wooden board in the supine position, and then placed in cold water at 21 ± 1 °C below the sternum xiphoid in an "erect position" after resuscitation (Figure 1A). RWIS can irritate the rats, and leads to a decline in body temperature, gastric dysfunction and dotted or strip-like ulcers on the gastric mucosa within a few hours after induction (Figure 1B). In addition, RWIS can increase the gastric ulcer index values[22] in a time-dependent manner (Figure 1C).
ENS consists of both submucosal plexuses and intermuscular plexuses. The submucosal plexuses are located in the submucosal muscles, which mainly regulate gastrointestinal mucosa contraction and glandular secretion in the lamina propria. To regulate the movement of gastrointestinal smooth muscles, glandular secretion, gastrointestinal blood flow, intestinal epithelial substance transport, gastrointestinal immune response and inflammatory process, the intermuscular plexuses distributed throughout the entire digestive tract between longitudinal muscles and circular muscles can form a network with sensory neurons, intermediate neurons and motor neurons via synapses. Moreover, the ENS neurons are interconnected to form an independent nervous system similar to the brain and spinal cord, which is also known as the "gut brain" due to a high degree of independence as well as the largest and most complex autonomic nervous system in the peripheral nerves. At the same time, ENS has a large number of afferent nerves derived from the CNS, such as the vagus nerve and spinal nerve, both of which can interact and serve as the gut-brain axis[23]. Abnormal regulation of ENS can induce gastrointestinal dysfunction, by elevating the amplitudes and index values of gastric motility (gastric hyperkinesia), decreasing gastric mucosal blood flow (GMBF) and gastric mucus secretion, increasing the production of gastric acid and proliferation of gastric mucosal cells[14,24-27]. Huang et al[15-17] examined the molecular signaling pathways related to the maintenance of gastrointestinal tract homeostasis and gastric mucosal barrier integrity, as well as the energy-related proteins that retain the levels of intracellular adenosine triphosphate (ATP). However, the exact functions of these molecules in SGML need to be further evaluated using molecular biology techniques.
Similar to the CNS, there are many types of neurons in the ENS, which differ from each other not only in morphology and structure, but also in neurotransmitter diversity. Apart from acetylcholine (ACh) and norepinephrine (NE), numerous endogenous substances, such as substance P (SP), vasoactive intestinal peptide (VIP), calcitonin gene-related peptide (CGRP), ATP, 5-hydroxytryptamine (5-HT) and nitric oxide (NO), have recently been found to play important roles as neurotransmitters in the gastrointestinal neural network. These neurotransmitters are also termed the non-adrenergic non-cholinergic (NANC) nerves. Gastrointestinal excitatory motor neurons release excitatory transmitters, such as ACh and SP, thus promoting gastrointestinal smooth muscle contraction and glandular secretion. On the contrary, inhibitory motor neurons release inhibitory transmitters, such as VIP and NO, thus suppressing gastrointestinal smooth muscle contraction and glandular secretion[28]. All these gastrointestinal excitatory and inhibitory motor neurons can interact with each other under a complex and delicate balance. If this balance is broken, gastrointestinal dysfunction may be induced.
NO: NO is an active and unstable inorganic gaseous molecule secreted by NANC nerves in the ENS, which serves as a major inhibitory neurotransmitter in the gastrointestinal tract. Nitric oxide synthase (NOS) is a key rate-limiting enzyme in NO production, and NOS assessment can indirectly determine the changes in NO, which can be classified into eNOS, nNOS and iNOS. eNOS is mainly distributed in vascular endothelial cells, and the synthesized NO is mainly used to regulate GMBF and promote gastric mucosal repair. nNOS is mainly distributed in cells within the intermuscular and submucosal plexuses, endocrine cells and vascular endothelial cells, and the synthesized NO interacts with gastrointestinal hormones (VIP and ATP) to jointly regulate gastrointestinal motility[29]. iNOS is highly expressed upon the stimulation of endotoxins and cytokines, which produces a large amount of NO within a short period of time, and ultimately results in cell damage. Previous studies have demonstrated that NO can inhibit gastric acid secretion and neutrophil adhesion, improve gastric mucosal blood circulation and eliminate oxygen free radicals, thereby protecting the gastric mucosa from injury[30,31]. It was reported by Nishiad et al[31] that the expression level of iNOS increased significantly in the gastric mucosa of RWIS rats, while that of eNOS reduced significantly, indicating that the changes in iNOS and eNOS activities in the gastric mucosa are closely related to the incidence of GML. In SGML, L-NAME (NOS inhibitor) can decrease the production of NO, thus exacerbating acute GML and inhibiting the healing process of chronic gastric ulcers, while L-Arg (NO precursor) can obviously prevent the injury[32-34]. Wei et al[35] found that NO is involved in RWIS, and can promote the SGML healing process.
The mechanisms of NO in protecting gastric mucosa are as follows: (1) NO can reduce vascular permeability, inhibit platelet adhesion and aggregation in gastric mucosal vascular endothelium, and prevent thrombosis. (2) Under physiological conditions, gastric mucosal vascular endothelium synthesizes NO, which in turn regulates vascular smooth muscle tension and maintains GMBF. (3) In acute GML, NO increases GMBF by dilating the mucosal blood vessels, thus promoting gastric mucosal repair[33]. In addition, the secretion of gastric acid can also be inhibited by NO. Upon the reaction of stimulus against gastric mucosa, enterochromaffin cells and mastocytes can release histamine to stimulate parietal cells for gastric acid production, thus aggravating the mucosal lesion. In addition, endogenous NO can inhibit the stimulation of histamine through parietal cells, thus reducing gastric acid secretion and protecting gastric mucosa. It has been found that, through in vivo and in vitro experiments, the NO donor FK409 and sodium nitroprusside can markedly suppress the gastrin-induced increase in histamine release and gastric acid secretion in rats, and L-NAME further increases gastric acid secretion[30]. Gastric mucous cells promote NO synthesis by expressing high-level NOS, and enhance the mucous barrier through the NO effects of promoting mucin synthesis and secretion. Based on the findings of previous experiments, RWIS-induced GMLs can weaken the synthesis and secretion of gastric mucus by reducing nNOS activity, while the NO donor L-Arg can increase nNOS activity and mucus secretion[36].
5-HT: 5-HT is an important neurotransmitter that widely exists in the brain and digestive tract. It has been estimated that 90% of 5-HT is synthesized and secreted by enterochromaffin cells in the gastrointestinal tract[37]. There are four types of 5-HT receptors, of which 5-HT3R and 5-HT4R are the two receptor subtypes most closely related to gastrointestinal function. 5-HT3R is mainly found within the neurons of myenteric plexuses in the stomach and colon[38], while 5-HT4R is mainly distributed within the neurons of myenteric plexuses in the ascending colon, duodenum and gastric smooth muscles, as well as the intestinal submucosa[39]. Both 5-HT3R and 5-HT4R are involved in gastrointestinal motility, gastric acid and mucus secretion, and regulation of local mucosal blood flow. Due to the absence of internal and external nerve fibers in the mucous epithelium, it is a transepithelial phenomenon that ENS senses and reacts to chemical stimulus in the enteric cavity, which may be partly mediated by enterochromaffin cells. Upon response to stimulus, these cells may release 5-HT, and subsequently activate the submucosal primary afferent nerve fibers through 5-HT3R distributed on the vagal afferent nerve fibers, and transmit intestinal information to the center, thereby regulating local excitement and inhibition[40]. Serotonin reuptake transporter (SERT) is a type of translocator responsible for re-uptake of 5-HT from the synaptic cleft, which can rapidly eliminate 5-HT from the synapse and reduce 5-HT concentration in the intestinal tract and tissue space. Thus, pharmacological blockage of SERT function can obviously decrease gastrointestinal motility[41]. Chen et al[42] reported that, in SERT-knockout mice, watery diarrhea is likely to be caused by the enhanced action of the 5-HT signaling pathway, and slow transit constipation may be caused by the insensitivity of 5-HT receptors due to excessive 5-HT production. In addition, Morita et al[43] showed that 5-HT3R and 5-HT4R antagonists can inhibit the antral motility during the interdigestive period (phase III) and colonic motility in dogs. The above data indicate that the 5-HT signaling system is involved in the sensory and motor functions of the gastrointestinal tract. Apart from its motor and sensory functions in the gastrointestinal tract[44], 5-HT inhibits gastric acidity by increasing the synthesis of mucus[45]. It has been reported that 5-HT simulates prostaglandin (PG) synthesis by enhancing the activity of the cyclooxygenase pathway, which in turn stimulates mucosal blood flow and contributes to the secretion of mucus along with bicarbonate. Therefore, the 5-HT signaling system plays a vital role in the regulation of gastrointestinal motility, secretion and visceral sensation. An abnormal 5-HT signaling pathway may trigger gastrointestinal endocrine disorders, leading to a variety of gastrointestinal diseases. In addition, indomethacin-induced intestinal lesions in mice can be prevented by pretreatment with p-chlorophenylalanine (a 5-HT synthesis inhibitor). The administration of the 5-HT4 receptor agonist (mosapride) or 5-HT3 receptor antagonists (e.g., ondansetron and ramosetron) can dose-dependently reduce the severity of indomethacin-induced intestinal lesions, whereas a high dose of GR113808 (a 5-HT4 receptor antagonist) significantly aggravated these lesions. These findings suggest that endogenous 5-HT exerts a dual role in the pathogenesis of indomethacin-induced intestinal lesions: Pro-ulcerogenic action via 5-HT3 receptors and anti-ulcerogenic action via 5-HT4 receptors[46]. Additionally, the cold-restraint stress significantly increased mean ulcer index values in gastric tissue, while a decrease in enterochromaffin cell count was observed with a concomitant decrease in 5-HT content and adherent mucosal thickness. Pretreatment with Aegle marmelos reduced mean ulcer index values, and increased enterochromaffin cell count, 5-HT content and adherent mucosal thickness in ulcerated gastric tissue, suggesting that the high enterochromaffin cell count and 5-HT levels exert protective effects against cold-restraint stress-induced gastric mucosal injury[47].
Hydrogen sulfide: Hydrogen sulfide (H2S) is a new type of endogenous gaseous signaling molecule, and many tissues in the body can catalyze L-cysteine to H2S via cystathionine-synthase (CBS) and cystathionine-γ-lyase (CSE). In recent years, it was found that H2S is involved in various physiological and pathological processes in the body, which in turn regulates the motility, resists the inflammation, affects the visceral sensitivity, and promotes glandular secretion. Specifically, its regulation of digestive tract motility is mainly manifested as the suppression of intestinal motility. Exogenous administration of sodium hydrosulfide (NaHS; a H2S donor) can enhance the secretion of colon mucosal and submucosal chloride in guinea pigs in a concentration-dependent manner, and this secretion can be inhibited by H2S CSE and CBS blockers[48]. Krueger et al[49] found that H2S promotes intestinal secretion via activation of transient receptor potential vanilloid-1 (TRPV1) receptor and the release of SP, thus activating cholinergic neurons. In addition, Dhaese et al[50] found that NaHS can relax the gastric fundus smooth muscles of mice in a concentration-dependent manner, which can be weakened by myosin light chain phosphatase (MLCP) inhibitors, instead of ATP-sensitive potassium channel (KATP) blockers. This indicates that H2S-induced dilation of gastric fundus smooth muscles is realized, at least partly, by activating MLCP, but is not related to KATP.
H2S is able to protect the gastrointestinal tract and promote the repair of GML. Nonsteroidal anti-inflammatory drugs (NSAIDs) can markedly down-regulate the expression of CSE in gastric mucosa and reduce the synthesis of endogenous H2S. However, NaHS can be used to decrease the synthesis of tumor necrosis factor-alpha (TNF-α), intercellular adhesion molecule-1 and lymphocyte function associated antigen-1, reduce the adhesion of white blood cells to mesenteric vessels, increase GMBF and suppress GML caused by NSAIDs[51]. Both exogenous administration and endogenous release of H2S are gastroprotective against cold restraint stress-induced gastric injury. It has been reported that NaHS attenuates the ulcer index by reducing gastric acid output, mucosal carbonyl content, pepsin activity and ROS generation. Moreover, H2S also reduces TNF-α level and myeloperoidase activity[52]. Magierowski et al[53] demonstrated that treatment with NaHS plays an important physiological role in gastric mucosal protection against stress-induced lesions. The protective effect of NaHS is often accompanied by an enhancement in gastric microcirculation, possibly mediated by a significant local increase in the gastric mucosal production of H2S. Furthermore, the mechanism of H2S-induced gastroprotection may involve activation of the endogenous prostaglandin/cyclooxygenase (PG/COX) system, increase biosynthesis of prostaglandin E2 (PGE2), afferent sensory fibers release of CGRP via VR-1 receptors and an anti-inflammatory effect resulting in the inhibition of pro-inflammatory cytokines (e.g., TNF-α).
CGRP: CGRP is the main transmitter of capsaicin-sensitive sensory nerves, with a wide variety of functions, which is widely distributed throughout the cardiovascular, gastrointestinal and respiratory systems and commonly recognized as a protector of gastric mucosa. CGRP is the most effective vasodilator discovered to date, and it can improve blood flow through two mechanisms. (1) CGRP binds to receptors on endothelial cells and up-regulates NOS to produce NO, thereby relaxing the vascular smooth muscles. (2) CGRP directly binds to receptors on vascular smooth muscle cells and increases GMBF by activating KATP channels without involving the endothelium[54]. In addition, CGRP also exerts anti-apoptotic, anti-platelet aggregation, anti-oxidation, anti-proliferation and anti-aging properties. Previous findings have shown that CGRP can significantly inhibit gastric acid secretion[55]. Following craniocerebral injury and severe burn, the concentrations of hydrogen ions in gastric juice may be elevated. It has also been found that excess gastric acid production is related to neuroendocrine disorders, and a decrease in CGRP secretion is caused by nerve center and hypothalamic injury[56]. The gastric acid secretion in mice can be facilitated by an intracerebroventricular injection of thyrotropin-releasing hormone, but inhibited by CGRP. A large amount of experimental data has proved that noxious stimuli, such as GML, can trigger the sensory neurons to release CGRP, activate intermediate neurons and motor neurons. Besides, it also directly acts on smooth muscles and inhibits gastrointestinal motility, whose mechanisms are as follows: (1) The longitudinal muscles and circular muscles of the intestine are directly dilated. And (2) The NANC inhibitory neurons are stimulated, and other inhibitory neurotransmitters are released, such as NO and VIP, thereby inhibiting gas-trointestinal motility[57].
CGRP can help to improve local gastric microcirculation and protect against stress- or drug-induced GML by increasing gastrointestinal blood flow and regulating gastrointestinal motility. A previous study demonstrated that CGRP down-regulation is related to the pathogenesis of gastric ulcers[58]. After stimulation, capsaicin-sensitive sensory nerve fibers may release CGRP, and then CGRP increases the levels of prostacyclin and PGE2 in gastric mucosa, thereby inhibiting the activation of neutrophils and degranulation of mastocytes, reducing the secretion of inflammatory mediators (e.g., histamine), and alleviating gastrointestinal inflammation[59]. In a mouse model of ischemia-reperfusion injury, intraperitoneal treatment with CGRP significantly reduced gastric mucosal edema, hemorrhage, apoptosis, mucosal separation and inflammatory cell infiltration[60]. RWIS-induced gastric ulcers were inversely correlated with the decrease in CGRP-like immunoreactivity observed in the whole thickness of the stomach corpus. Systemic administration of CGRP exhibited a significant decrease in the lesion index of RWIS-induced ulcers, and such protection was inhibited by the functional ablation of afferent neurons induced by capsaicin pretreatment. These findings suggest that endogenous CGRP plays a defensive role in RWIS-induced ulcers[61].
VIP: As one of the most important peptide neurotransmitters, VIP is widely distributed in the circulatory, immune, reproductive and digestive systems, as well as the central and peripheral nervous systems. VIP possesses dual functions in the body, and acts as both a gastrointestinal hormone and a neuropeptide, and has been considered a type of brain-gut peptide. VIP is mainly produced by the central and peripheral nervous systems, released by the parasympathetic postganglionic fibers and coexists with ACh, which plays a regulatory role in local mucosal immu-noregulation. In the digestive system, VIP is predominantly distributed in the endocrine cells of gastrointestinal mucosa as well as the submucosal plexuses and smooth muscle layer. Moreover, VIP regulates gastrointestinal absorption, inhibits gastric acid secretion, protects the gastrointestinal mucosa from acid-induced damage, and promotes the secretion of water, electrolytes, pancreatic juice and intestinal juice in the intestine. Additionally, VIP is able to induce smooth muscle relaxation and exert a potent vasodilator effect[62]. The regulatory effect of VIP on gastrointestinal smooth muscle motility is closely related to the main inhibitory neurotransmitter NO, in which VIP can promote NO synthesis, relax circular muscles, inhibit gastric motility and reduce gastric tightness.
In addition, VIP exerts an impact on the integrity of the gastrointestinal mucous membrane barrier. For instance, VIP can induce the secretion of water and bicarbonate in the pancreas, thus promoting the formation and repair of the gastrointestinal mucous membrane barrier. Moreover, VIP can stimulate the production of intestinal juice, and inhibit the secretion of gastric acid and gastrin, thereby protecting the gastric mucosa, suppressing the occurrence of gastric and duodenal ulcers, and promoting the repair of ulcers[63]. In ethanol-induced GML, the content of VIP decreases obviously in the gastric mucosa, by reducing the release of NO and increasing the production of endothelin[64]. It has been reported that cold-restraint stress induces gastric lesions and mast cell degranulation, and exacerbates lipid peroxidation in gastric tissue. VIP can prevent stress-induced ulcers and mast cell degranulation, and protect gastric tissue from lipid peroxidation. When VIP was administered after stress-induced ulcers, it was demonstrated to be therapeutically beneficial, suggesting that VIP is valuable for the prevention of gastric mucosal damage induced by cold-restraint stress[65]. Recent studies have shown that VIP significantly suppressed cold-restraint stress gastric lesions and markedly decreased the content of histamine in the tissue. Histamine plays an important role in the development of gastric ulcers, and mast cell-derived histamine might be essential during this process. The mechanisms of the action of VIP on gastric tissue histamine levels can be explained by its inhibitory effect on the release of gastrin hormone, mast cells and enterochromaffin-like cells[66].
Some studies have found that RWIS leads to the elevation of blood corticosterone and adrenocorticotropic hormone levels in rats, and their levels in plasma also gradually rise over a prolonged period of stress[67]. This seems to indicate that the activity of the hypothalamic-pituitary-adrenal (HPA) axis is enhanced during RWIS. However, severing the subphrenic vagus nerves or consuming atropine can significantly alleviate and even cure RWIS-induced GML, but removing the pituitary glands and adrenal glands or administering phenoxybenzamine (adrenergic α-receptor blocker) has little impact on RWIS-induced GML, gastric hyperkinesia and RWIS-induced gastric acid secretion. This suggests that the HPA axis does not play a major role in RWIS-induced GML, and the peripheral nervous mechanism of RWIS-induced GML is mainly through the enhanced parasympathetic activity[14-16]. Therefore, the nervous mechanism of RWIS-induced gastrointestinal dysfunction in rats is mainly the "enhanced activity of parasympathetic nervous system", rather than the traditional ideas of the "enhanced activity of sympathetic-adrenal medulla system" and "HPA axis".
Medullary gastrointestinal center: In recent years, the mechanisms of the primary central-brainstem loop in regulating gastric function have been clarified using electrophysiological and immunohistochemical techniques. Parasympathetic control is dominant in the nervous regulation of gastric function. The parasympathetic nerves that control the stomach and intestines are from the dorsal motor nucleus of vagus (DMV) and nucleus ambiguous (NA) of the medulla oblongata (both of which are the initial nuclei of parasympathetic preganglionic fibers and essential visceral motor nuclei). The nucleus of the solitary tract (NTS), located at the dorsolateral side of the DMV, is an important primary visceral sensory nucleus; while area postrema (AP), located at the dorsomedial side of the NTS, has a chemosensory function. Eventually, a dorsal vagal complex (DVC) is formed. The DVC and NA are the lower centers of visceral parasympathetic nerves that integrate both sensory afference and visceral efference, which are also the primary centers for the regulation of gastric function[68,69].
The DVC and vagal efferent play an outstanding role in the regulation of gastric mucosal resistance to injury. However, the role of the vagal nerve is likely to be dual, as it can mediate both mucosal damaging and protective effects. It has been demonstrated that thyrotropin-releasing hormone (TRH) acting centrally in the DVC can stimulate gastric acid and pepsin secretion, induce gastric emptying and trigger ulceration via activation of parasympathetic outflow to the stomach[70]. Similarly, activation of the dorsal motor nucleus of vagus by RX-77368 (a TRH analogue) through an intracisternal injection at high dose could promote the formation of GML. Furthermore, electrical stimulation of the vagus has been found to induce GML and mast cell degranulation[71]. In addition, TRH, or its analogue RX-77368, injected at the dorsal motor nucleus of vagus, has been reported to protect the gastric mucosa against ethanol injury through stimulation of vagal cholinergic pathways, as both vagotomy and atropine can reverse the gastroprotective effects[72,73]. Biochemical and pharmacological studies have demonstrated that the mechanisms of vagal-mediated gastroprotective effects may be due to the activation of vagal cholinergic pathways, secretion of gastric PG and production of NO[74]. Our previous study revealed that RWIS induced significant gastric mucosal damage and activated astrocytes by increasing the levels of glial fibrillary acidic protein and neurons, as indicated by Fos expression in the DMV and NTS. Intracerebroventricular administration of the astroglial toxin L-α-aminoadipate could alleviate RWIS-induced gastric mucosal damage. Immunohistochemistry results showed that L-α-aminoadipate decreased the activation of both astrocytes and neurons induced by RWIS. These findings provide strong evidence that astrocytic and neuronal activation in the DMV and NTS may be closely related to RWIS-induced gastric mucosal damage[75].
With the expression of immediate early genes (e.g., c-Fos) as an index, Zhang and colleagues[18] evaluated the activities of neurons in the medullary gastrointestinal center at different time periods of stress using an immunohistochemistry-based technique. They found that c-Fos was highly expressed in DVC and NA at different time periods of RWIS (30, 60, 120 and 180 min), and its expression intensity was highest in the DMV, followed by the NA and NTS. The above findings preliminarily reveal the temporal-spatial rule of nuclear activity in the primary center that regulates gastrointestinal function in rats during RWIS, and further confirm that RWIS-induced gastric dysfunction is mainly caused by the enhanced parasympathetic activity. In other words, there is a neural circuit ("medullary gastrointestinal center-gastrointestinal wall plexus loop") between the medulla oblongata and the gastrointestinal tract. Under RWIS, the information of gastrointestinal motility is transmitted as follows: Information → vagal afferent nerves → NTS → DMV/NA, while those of medullary efferents are disseminated as follows: DMV/NA → vagal efferent nerves → gastrointestinal wall plexuses, thereby causing gastric hyperkinesia, increasing gastric acid secretion and reducing gastric mucus secretion, and ultimately leads to GML and fecal impaction.
However, in the case of electrical stimulation of NTS in normal rats, gastric motility may be inhibited, and a possible reason for this is that excitement of the NTS activates inhibitory neurons in the DMV, thus suppressing gastric motility through a non-cholinergic neural pathway[68,76]. In addition, gastric motility was significantly inhibited when electrical or chemical stimulation induced neuronal excitation in the NA and DMV, indicating that excitation of the NA and DMV also exerts an inhibitory effect on gastric motility[77]. However, the above findings are contradictory to the immunohistochemical results showing that the expression levels of c-Fos in the NA, DMV and NTS were obviously enhanced in RWIS-induced rats. This is probably due to the fact that the activity of the higher center (e.g., anterior hypothalamus) eliminates the inhibition of medullary visceral centers on the stomach during RWIS, thereby causing gastric hyperkinesia and increasing gastric acid secretion.
Anterior hypothalamus: The hypothalamus region is generally divided into the anterior hypothalamus and posterior hypothalamus. With regard to the regulation of visceral function, the anterior hypothalamus may act as the parasympathetic center, while the posterior hypothalamus serves as the sympathetic center. At different time points of RWIS (30, 60, 120, and 180 min), Fos is expressed in different degrees in the supraoptic nucleus (SON), paraventricular nucleus (PVN) and suprachiasmatic nucleus (SCN) in the anterior hypothalamus, most noticeably in the PVN and SON[78]. Chemical or electrical stimulation of the PVN can markedly aggravate gastric ulcers in RWIS rats, whereas electrical damage of the PVN can alleviate ulcers[79]. Bilateral vagotomy can suppress GML triggered by electrical stimulation of the PVN[80,81], indicating that PVN stimulation-induced GML is mediated by parasympathetic pathways. Lu et al[82] found that chemical (L-Glu) or electrical stimulation of the SON could promote gastric acid secretion. These results further confirm the above-mentioned statements that the nervous mechanism of RWIS-induced gastrointestinal dysfunction is mainly through the "enhanced activity of parasympathetic nervous system". Therefore, the excessive activities of the SON and PVN in the anterior hypothalamus may be one of the central mechanisms underlying stress-induced gastrointestinal dysfunction.
Central nucleus of the amygdala: The amygdaloid complex is an important subcortical nucleus in the limbic system, which is involved in the regulation of mood, emotion and visceral activities related to emotional stimulation, and has thereby attracted considerable attention[83,84]. The central nucleus of the amygdala (CEA) is an important nucleus of the amygdaloid complex, which is closely associated with the autonomic responses to stress and maintenance of gastric mucosal integrity[85-88]. At different time points (30, 60, 120 and 180 min) of stress, the expression level of Fos was significantly enhanced in the CEA, and was second only to that in the anterior hypothalamus[89]. According to previous anatomical studies, CEA has complex fiber connections with the NTS and DMV in the medullary gastrointestinal center[90,91]. It has also been found in physiological studies that electrical stimulation of different regions in the CEA can promote the secretion of gastric acid[92], enhance or decrease gastric motility[93,94], induce gastric ulcers[95], and alter the activity of neurons in NTS and DMV in the medullary gastrointestinal center[90,91]. Moreover, electrical stimulation of CEA-induced vagus nerve-mediated gastric ulcers, demonstrates that the CEA may play a role similar to the hypothalamus. One possible reason is that its neuronal activity diminishes the inhibitory effect of the medullary gastrointestinal center on the gastrointestinal tract, but further research is needed to verify this hypothesis.
Medial prefrontal cortex: The medial prefrontal cortex (mPFC), the highest-level association cortex in mammals, plays a key role in many advanced brain functions. RWIS includes psychological stress and physiological stress. According to the immunohistochemical findings, the expression levels of Fos were relatively high in the prelimbic cortex (PL) and infralimbic cortex (IL) of the mPFC in RWIS rats, in addition to the DMV, NTS, NA, SON and PVN. In both IL + PL-sham-operated and IL + PL bilateral-lesioned groups, exposure to RWIS for 4 h could affect the curves of gastric motility by changing from high-frequency, short-term and low-amplitude fast waves to low-frequency, long-term and high-amplitude slow waves, which represent an obvious sign of gastric hypermotility. Synaptic plasticity can maintain the stability of neural circuits, and the brain can adapt to changes in both internal and external environments through neural plasticity changes in synaptic structure and function when coping with stress. Compared to the control group, the concentration of postsynaptic density protein 95 (PSD95) decreased in the mPFC of the RWIS 4-h group[96]. From the dendritic spine staining results, it was found that the number of dendritic spines per unit length (10 μm) in the 1-, 2- and 4-h RWIS groups was significantly lower than that in the control group. Moreover, according to electron microscopic observations of PFC synapses, the synapses displayed an unclear outline, and there were more perforated synapses and more vacuolated mitochondria observed in the 4-h RWIS group, indicating that oxidative stress causes certain damage in mitochondria. In addition, the number of synapses per unit area, number of presynaptic vesicles and thickness of the PSD in the 4-h RWIS group were obviously decreased compared to those in the control group. From the results of proteomic analysis, it was observed that there were 129 differentially expressed proteins (88 up-regulated and 41 down-regulated) between the control group and the 4-h RWIS group. According to the GO analysis, 22% (29) of the differentially expressed proteins were directly related to nervous system functions, including synaptic plasticity (6), axon morphology and growth (12), neurodevelopment and apoptosis (10), and neural signal transmission (1), while some were associated with synaptic vesicle circulation and toxicological metabolism. These findings indicate that the mPFC is involved in the regulation of RWIS-induced gastric dysfunction, but its exact role requires further study.
Mediodorsal nucleus of the thalamus: Using Fos, SYN and SYN-I as indices, it has been demonstrated that the PFC is involved in the responses to RWIS. However, gastric motility was not significantly affected following an injection of sodium L-glutamate, electrical stimulation of PL or IL of the PFC, suggesting that other nuclei are required to transmit RWIS signals into the PFC. After electrophoresis of the PFC, no labeled nerve cell bodies were observed in the PVN or SON, but were found in the mediodorsal nucleus of the thalamus (MD), indicating that the MD may emit fibers to the PFC.
MD is the largest subnucleus in the medial thalamic nucleus group, as well as the only thalamic nucleus with a fiber connection to the PFC. We also found that the expression level of Fos was significantly upregulated in the MD of RWIS rats compared to that in the control group, and the expression levels of SYN and SYN-I in the 1-h RWIS group were significantly higher than those in the control group, indicating that MD is involved in RWIS. As has been previously reported in the literature, the MD exhibits complex efferent and afferent fiber connections with the PFC, hypothalamus and brain stem, thus forming multiple neural circuits[97]. In addition, the MD is a higher-order thalamic relay nucleus that forms the cortex-thalamus-cortex circuit, which serves as an important integration site of visceral and physical activities[98]. A recent study reported that the density of dendritic spines on the dendritic shaft of neurons in the MD was markedly decreased in the 1-h RWIS group compared to that in the control group[99]. Electrophysiological findings showed that the firing rate of MD neurons was significantly reduced, the mean interspike interval was significantly prolonged and the burst rate gradually declined in awake rats during RWIS, suggesting that RWIS exerts an inhibitory effect on the neurons in the MD. Quantitative proteomic analysis revealed that a total of 65 dysregulated proteins were identified, which are mainly involved in the signaling pathways associated with neurological diseases. Moreover, 31 upregulated proteins were primarily related to cell division, while 34 downregulated proteins were related to neuronal morphogenesis and neurotransmitter regulation. Furthermore, glycogen synthase kinase-3 beta might be related to the central mechanism through which RWIS gives rise to stress-induced gastric ulcers.
ACh: ACh is the first discovered, classical neurotransmitter that is distributed throughout the motor and sensory systems, brainstem reticular structure, limbic system and cerebral cortex. ACh has been shown to affect the sensory, motor, learning, memory and other functions in the CNS, especially excitation function. Continuous intravenous injection of ACh can cause severe GML in rats[100]. Moreover, injecting ACh into the NTS can significantly enhance the protective effect of electroacupuncture against GML, while injecting atropine can weaken this effect, suggesting that the effect of ACh is mediated by the cholinergic M receptor[101]. In addition, injecting low-dose ACh into the lateral ventricle evidently strengthens the aggravating effect of electrical stimulation of the PVN on SGML, which is also mediated by the M receptor[79]. We studied and proved that the excessive activity of cholinergic neurons in the middle segment of the DMV and NA is one of the primary central mechanisms responsible for RWIS-induced gastric dysfunction.
Catecholamine: Catecholamine (CA) is a collective term for NE, epinephrine (E) and dopamine (DA), and is one of the classic neurotransmitters abundantly distributed in the central and peripheral nervous systems. Numerous morphological and electrophysiological studies have confirmed that NE and DA are important endogenous inhibitory neurotransmitters that protect the integrity of gastric mucosa during stress[102,103]. A micro-injection of NE into the lateral ventricle can reduce the aggravating effect of electrical stimulation of the PVN on SGML in a dose-dependent manner, which is mediated by β-adrenergic receptors[79]. DA antagonists applied in the center may worsen SGML, while DA agonists can relieve SGML[104]. Moreover, after 6-OHDA is injected into the ventral tegmental area to deplete endogenous DA, or haloperidol (a DA receptor antagonist) is injected into the NA, the protective effect of neurotensin against RWIS-induced GML is obviously inhibited[105]. Zhao et al[106] found that catecholaminergic neurons in the nucleus of the medullary visceral center participate in the regulation of RWIS-induced GML, whereas catecholaminergic neurons in the nucleus of the anterior hypothalamus are rarely or not involved. Therefore, the neurons responsible for RWIS are not located in the anterior hypothalamus, but instead the neuronal activity in the nucleus may be regulated by medullary catecholaminergic neurons.
Glutamate: Glutamate (Glu) is the most abundant and widely distributed neurotransmitter in the brain and CNS, and conveys peripheral nociceptive information to the center. It has been closely associated with rapid excitatory synaptic transmission, neuronal development and death, synaptic plasticity and the incidence of some neurological diseases[107-110]. Yao et al[111] found that the contents of excitatory amino acids (Asp, Glu and Gln) in the brain tissues of rats were significantly higher in the 5-h RWIS group than those in the control group. Micro-injection of L-Glu into the middle segment of rat DMV could enhance vagus nerve-mediated gastric motility, while injection of L-Glu into the final segment of the DMV and micro-injection of L-Glu into rat NTS could inhibit such gastric motility[10,112]. The amount of H+ in the secreted gastric juice of rats injected with Glu into the AP evidently rises compared to that in rats injected with normal saline, and this effect is mediated by the vagus nerve. Krowicki et al[113] injected L-Glu into the NA of rats, and the results showed that the peak of intragastric pressure and contraction of pyloric smooth muscles were greatly increased. Furthermore, we found that micro-injection of L-Glu into rat AP, right NA, DMV and NTS might suppress gastric motility in a dose-dependent manner.
γ-aminobutyric acid: γ-aminobutyric acid (GABA) is one of the major inhibitory neurotransmitters in the CNS, and is widely distributed in various regions of the brain, and has been associated with higher brain functions (e.g., attention, working memory, mood and motorial inhibition). Shi et al[114] found that gastric ulcers were significantly relieved after injecting GABA and pentobarbital sodium (a GABAA receptor agonist) into the lateral ventricle, and they were aggravated after injecting bicuculline (a GABAA receptor blocker). This indicates that GABAA receptor mediates the inhibitory effect of GABA on RWIS-induced GML.
Oxytocin: Oxytocin (OT) is a neuropeptide composed of 9 amino acids, which is mainly synthesized in the magnocellular part of the hypothalamic SON and PVN[115]. When an OT receptor (OTR) blocker was injected into the animal DMV, gastric secretion was suppressed and motility was enhanced. When OT was injected into the DMV, the opposite results were obtained[116]. The injection of OT into the lateral ventricle significantly relieved GML induced by cold restraint stress or subcutaneous injection of mercaptoethylamine[117]. RWIS could induce the expression levels of Fos in 34% and 28% of OT neurons residing in the PVN and SON of rats, indicating that RWIS activates OT neurons in both the PVN and SON[118]. It has been reported that OTR neurons are almost uniformly distributed in the start-final segments of the nucleus of the medullary gastrointestinal center. Moreover, the number of Fos+OTR-immunoreactive neurons (Fos+OTR-IR) in the 1-h RWIS group was obviously increased compared to that in the control group, and the proportions of Fos+OTR-IR neurons in Fos-IR neurons were approximately 58% and 45% in the DMV and NTS, respectively, in the 1-h RWIS group, indicating that the activities of neurons in the DMV and NTS are regulated by OTR-mediated OT[118].
Arginine vasopressin: Arginine vasopressin (AVP) is also a peptide hormone mainly secreted by large-cell neuroendocrine cells of the PVN and SON in the hypothalamus. In addition to the PVN and SON, AVP neurons are also distributed in the SCN, peripheral hypothalamic area, lateral hypothalamic area, posterior hypothalamic area and the CEA. RWIS can induce the expression levels of Fos in AVP neurons to 40% and 53% in the PVN and SON, respectively, in rats, indicating that RWIS activates the AVP neurons in both the PVN and SON[118]. Similar to OTR, AVP neurons (V1bR) are almost uniformly distributed in the start-final segments of the nucleus of the medullary gastrointestinal center. Moreover, the number of Fos + OTR-positive neurons in the 1-h RWIS group was obviously increased compared to that in the control group, and the proportions of Fos + V1bR immunoreactive positive neurons in Fos-positive neurons in the DMV and NTS were approximately 72% and 52%, respectively, in the 1-h RWIS group, indicating that the activities of neurons in the DMV and NTS are regulated by V1bR-mediated AVP[118]. Micro-injection of AVP into the DMV in rats can significantly inhibit gastric mobility and promote gastric acid secretion. Following micro-injection of AVP into the DMV, gastric motility was significantly inhibited. Furthermore, the inhibitory effect of AVP on gastric motility could be completely blocked by both SR49059 (a specific AVP receptor antagonist) and hexamethonium (a specific neuronal nicotinic cholinergic receptor antagonist). These data indicate that AVP inhibits gastric motility by acting on the specific AVP receptor in the DMV, with the potential involvement of parasympathetic preganglionic cholinergic fibers[119].
In addition, the types of neurotransmitters/neuromodulators in the medullary gastrointestinal center in response to RWIS have been studied and it was found that SP, enkephalin, H2S and other neurotransmitters/neuromodulators are involved in the regulation of RWIS-induced gastric dysfunction[120,121].
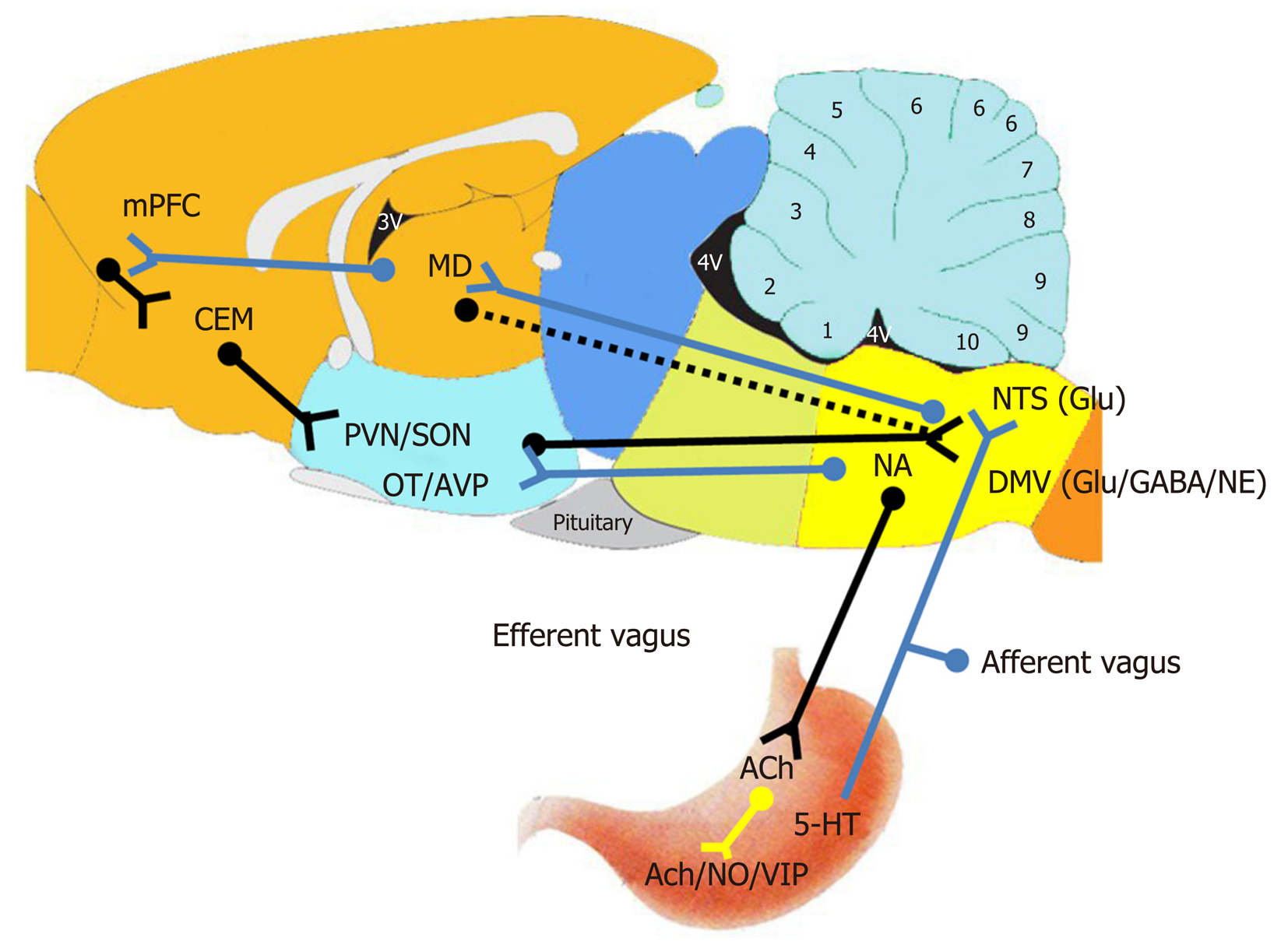
Some scholars have summarized the regulatory pathways of gastrointestinal function under normal physiological conditions[6,122,123], and the major nuclei involved[124]. According to these studies, several hypotheses have been put forward, and a preliminary consensus has been reached by domestic and foreign scholars on the peripheral and central nervous mechanisms of RWIS-induced gastrointestinal dysfunction. It was shown that the sensory information from the gastrointestinal tract and other internal organs are transmitted to the NTS through visceral sensory fibers in the vagus nerve, integrated by NTS secondary neurons in the brain stem, and then conveyed to relevant nuclei, such as the DMV, via neurotransmitters (e.g., Glu). DMV regulates the gastric motility through parasympathetic nerves, and parasympathetic neurons control the gastric function via two different pathways. (1) The M receptor is activated to enhance gastric motility and secretion via the cholinergic excitatory pathway. And (2) Gastric function is inhibited mainly by releasing NO or vasopressin (VP) via activating the NANC pathway (Figure 2). With regard to the advanced central nervous mechanism of RWIS-induced gastrointestinal dysfunction, the potential advanced central nervous regulatory pathway of gastrointestinal function in rats under RWIS is hypothesized based on the previous works of our research group and relevant scholars (Figure 2). The MD receives the advanced neural activity information from the PFC (possibly IL) via the cortical-thalamic pathway, integrates such information with that from subcortical structures (e.g., the hypothalamus and medulla oblongata), and then feeds the integrated information back to the PFC via the thalamic-cortical pathway. At the same time, there is a two-way fiber connection between the IL and CEA, and the final integrated information from the PFC is directly or indirectly fed back to the medullary gastrointestinal center through the CEA, thereby inducing gastric dysfunction.
To sum up, the nervous mechanism of RWIS-induced gastrointestinal dysfunction in rats is mainly the "enhanced activity of parasympathetic nervous system", rather than the traditional ideas of the "enhanced activity of sympathetic-adrenal medulla system" and "HPA axis", and multiple nuclei in the brain, characterized by multi-stage and autonomous regulation.
The nuclei in the brain interact with each other either cooperatively or anta-gonistically to form a complex information network. As a result, the same nucleus may exert inhibitory and excitatory effects on gastric function. For instance, the DMV not only inhibits gastric motility, but also promotes gastric acid secretion. Hence, the role of a certain nucleus has not been specifically defined by scholars in the discussion of multiple formation mechanisms of SGML. Therefore, further research is needed to resolve this uncertainty.
In addition, the levels of nuclei involvement varied with the time of stress. At different time points (30, 60, 120 and 180 min) of RWIS in rats, the expression levels of c-Fos were markedly different in the DMV, NTS and NA, and the highest expression level was observed in the DMV, followed by NTS and NA. Moreover, the different sub-regions of the same nucleus also exhibit different levels of involvement[18]. Notably, the expression levels of c-Fos differed significantly in the hypothalamic SON, PVN, SCN, CEA and mPFC, and its expression intensities from high to low were as follows: SON, SCN and PVN at 30 min; SON, PVN, SCN and mPFC at 60 min; SON, SCN, CEA and PVN at 120 min; SON, PVN and CEA at 180 min[98]. Thus, the temporal-spatial relation of nuclear activity in RWIS-induced GML remains to be elucidated further.
SGML is the result of the combined action of the central and peripheral nervous mechanisms, which involves a wide variety of signaling molecules as well as a complex information network that leads to GML under RWIS. The signaling molecules, such as SP, have dual effects on gastric function. Therefore, these signaling molecules may not only promote or inhibit the gastric function, but also directly act on a single site in the stomach, which require more in-depth studies in the near future.
We would like to thank Prof. Hong-Bin Ai for suggestions on improving the quality of the manuscript. We would also like to thank all graduate students in our Lab who have made continuous efforts to reveal the nervous mechanism of RWIS-induced GML.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Rompianesi G S-Editor: Gong ZM L-Editor: Webster JR E-Editor: Zhang YL
| 1. | Stanghellini V, Chan FK, Hasler WL, Malagelada JR, Suzuki H, Tack J, Talley NJ. Gastroduodenal Disorders. Gastroenterology. 2016;150:1380-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 818] [Cited by in RCA: 966] [Article Influence: 107.3] [Reference Citation Analysis (0)] |
| 2. | Ranasinghe N, Devanarayana NM, Rajindrajith S, Perera MS, Nishanthinie S, Warnakulasuriya T, de Zoysa PT. Functional gastrointestinal diseases and psychological maladjustment, personality traits and quality of life. BMC Gastroenterol. 2018;18:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 3. | Zuckerman GR, Shuman R. Therapeutic goals and treatment options for prevention of stress ulcer syndrome. Am J Med. 1987;83:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 4. | Spirt MJ. Stress-related mucosal disease: risk factors and prophylactic therapy. Clin Ther. 2004;26:197-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 88] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 5. | Sanchez-Mendoza ME, Cruz-Antonio L, Baez DA, Olivares-Corichi IM, RojasMartinez R, Martinez-Cabrera D, Arrieta J. Gastroprotective activity of methyleugenol from peperomia hispidula on ethanol-induced gastric lesions in rats. Int J Pharmacol. 2015;11:697-704. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Browning KN, Travagli RA. Central nervous system control of gastrointestinal motility and secretion and modulation of gastrointestinal functions. Compr Physiol. 2014;4:1339-1368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 365] [Article Influence: 36.5] [Reference Citation Analysis (0)] |
| 7. | Boeckxstaens G, Camilleri M, Sifrim D, Houghton LA, Elsenbruch S, Lindberg G, Azpiroz F, Parkman HP. Fundamentals of Neurogastroenterology: Physiology/Motility - Sensation. Gastroenterology. 2016;150:1292-1304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 84] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 8. | Vanner S, Greenwood-Van Meerveld B, Mawe G, Shea-Donohue T, Verdu EF, Wood J, Grundy D. Fundamentals of Neurogastroenterology: Basic Science. Gastroenterology. 2016;150:1280-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 82] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 9. | Pirnik Z, Kiss A. Fos expression variances in mouse hypothalamus upon physical and osmotic stimuli: co-staining with vasopressin, oxytocin, and tyrosine hydroxylase. Brain Res Bull. 2005;65:423-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Cruz MT, Murphy EC, Sahibzada N, Verbalis JG, Gillis RA. A reevaluation of the effects of stimulation of the dorsal motor nucleus of the vagus on gastric motility in the rat. Am J Physiol Regul Integr Comp Physiol. 2007;292:R291-R307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 11. | Rodzian MN, Aziz Ibrahim IA, Nur Azlina MF, Nafeeza MI. Pure tocotrienol concentrate protected rat gastric mucosa from acute stress-induced injury by a non-antioxidant mechanism. Pol J Pathol. 2013;64:52-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Izgüt-Uysal VN, Gemici B, Birsen I, Acar N, Üstünel I. The protective effect of apelin against water-immersion and restraint stress-induced gastric damage. J Physiol Sci. 2014;64:279-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 13. | Zheng YF, Xie JH, Xu YF, Liang YZ, Mo ZZ, Jiang WW, Chen XY, Liu YH, Yu XD, Huang P, Su ZR. Gastroprotective effect and mechanism of patchouli alcohol against ethanol, indomethacin and stress-induced ulcer in rats. Chem Biol Interact. 2014;222:27-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 14. | Li YM, Lu GM, Zou XP, Li ZS, Peng GY, Fang DC. Dynamic functional and ultrastructural changes of gastric parietal cells induced by water immersion-restraint stress in rats. World J Gastroenterol. 2006;12:3368-3372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Huang P, Zhou ZR, Zheng MQ, Shi FX. Effect of the IGF-1/PTEN/Akt/FoxO signaling pathway in the duodenal mucosa of rats subjected to water immersion and restraint stress. Genet Mol Res. 2012;11:4775-4788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 16. | Huang P, Zhou Z, Wang H, Wei Q, Zhang L, Zhou X, Hutz RJ, Shi F. Effect of the IGF-1/PTEN/Akt/FoxO signaling pathway on the development and healing of water immersion and restraint stress-induced gastric ulcers in rats. Int J Mol Med. 2012;30:650-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Zhou ZR, Huang P, Song GH, Zhang Z, An K, Lu HW, Ju XL, Ding W. Comparative proteomic analysis of rats subjected to water immersion and restraint stress as an insight into gastric ulcers. Mol Med Rep. 2017;16:5425-5433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Zhang YY, Cao GH, Zhu WX, Cui XY, Ai HB. Comparative study of c-Fos expression in rat dorsal vagal complex and nucleus ambiguus induced by different durations of restraint water-immersion stress. Chin J Physiol. 2009;52:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Saxena B, Singh S. Comparison of three acute stress models for simulating the pathophysiology of stress-related mucosal disease. Drug Discov Ther. 2017;11:98-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 20. | Ye Y, Wang XR, Zheng Y, Yang JW, Yang NN, Shi GX, Liu CZ. Choosing an Animal Model for the Study of Functional Dyspepsia. Can J Gastroenterol Hepatol. 2018;2018:1531958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Landeira-Fernandez J. Analysis of the cold-water restraint procedure in gastric ulceration and body temperature. Physiol Behav. 2004;82:827-833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Guth PH, Aures D, Paulsen G. Topical aspirin plus HCl gastric lesions in the rat. Cytoprotective effect of prostaglandin, cimetidine, and probanthine. Gastroenterology. 1979;76:88-93. [PubMed] |
| 23. | Sgambato D, Capuano A, Sullo MG, Miranda A, Federico A, Romano M. Gut-Brain Axis in Gastric Mucosal Damage and Protection. Curr Neuropharmacol. 2016;14:959-966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Kitagawa H, Fujiwara M, Osumi Y. Effect of water immersion stress on gastric secretion and mucosal blood flow in rats. Gastroenterology. 1979;77:298-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 144] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Garrick T, Leung FW, Buack S, Hirabayashi K, Guth PH. Gastric motility is stimulated but overall blood flow is unaffected during cold restraint in the rat. Gastroenterology. 1986;91:141-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 26. | Garrick T, Buack S, Bass P. Gastric motility is a major factor in cold restraint-induced lesion formation in rats. Am J Physiol. 1986;250:G191-G199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 25] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 27. | Nishida K, Ohta Y, Kobayashi T, Ishiguro I. Involvement of the xanthine-xanthine oxidase system and neutrophils in the development of acute gastric mucosal lesions in rats with water immersion restraint stress. Digestion. 1997;58:340-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 73] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 28. | Filaretova LP, Filaretov AA, Makara GB. Corticosterone increase inhibits stress-induced gastric erosions in rats. Am J Physiol. 1998;274:G1024-G1030. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 32] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 29. | Fedail JS, Zheng K, Wei Q, Kong L, Shi F. Roles of thyroid hormones in follicular development in the ovary of neonatal and immature rats. Endocrine. 2014;46:594-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 30. | Wang L, Zhou Y, Peng J, Zhang Z, Jiang DJ, Li YJ. Role of endogenous nitric oxide synthase inhibitor in gastric mucosal injury. Can J Physiol Pharmacol. 2008;86:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 31. | Nishida K, Ohta Y, Ishiguro I. Role of gastric mucosal constitutive and inducible nitric oxide synthases in the development of stress-induced gastric mucosal lesions in rats. Biochem Biophys Res Commun. 1997;236:275-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 32. | Kwiecien S, Ptak-Belowska A, Krzysiek-Maczka G, Targosz A, Jasnos K, Magierowski M, Szczyrk U, Brzozowski B, Konturek SJ, Konturek PC, Brzozowski T. Asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, interacts with gastric oxidative metabolism and enhances stress-induced gastric lesions. J Physiol Pharmacol. 2012;63:515-524. [PubMed] |
| 33. | Szlachcic A, Krzysiek-Maczka G, Pajdo R, Targosz A, Magierowski M, Jasnos K, Drozdowicz D, Kwiecien S, Brzozowski T. The impact of asymmetric dimethylarginine (ADAMA), the endogenous nitric oxide (NO) synthase inhibitor, to the pathogenesis of gastric mucosal damage. Curr Pharm Des. 2013;19:90-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Szlachcic A, Sliwowski Z, Krzysiek-Maczka G, Majka J, Surmiak M, Pajdo R, Drozdowicz D, Konturek SJ, Brzozowski T. New satiety hormone nesfatin-1 protects gastric mucosa against stress-induced injury: mechanistic roles of prostaglandins, nitric oxide, sensory nerves and vanilloid receptors. Peptides. 2013;49:9-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 35. | Wei Q, Korejo NA, Jiang J, Xu M, Zheng K, Mao D, Shi F. Mitigation of stress from gastric mucosal injuries by mulberry extract may occur via nitric oxide synthase signaling in mice. Tissue Cell. 2018;54:59-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 36. | Ohta Y, Nishida K. L-arginine protects against stress-induced gastric mucosal lesions by preserving gastric mucus. Clin Exp Pharmacol Physiol. 2002;29:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Sikander A, Rana SV, Prasad KK. Role of serotonin in gastrointestinal motility and irritable bowel syndrome. Clin Chim Acta. 2009;403:47-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 197] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 38. | Kato S. Role of serotonin 5-HT₃ receptors in intestinal inflammation. Biol Pharm Bull. 2013;36:1406-1409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 39. | Sveshnikov DS, Torshin VI, Smirnov VM, Kuchuk AV, Myasnikov IL. [The significance of different 5-HT-receptors in regulation of gastrointestinal motility]. Patol Fiziol Eksp Ter. 2014;45-51. [PubMed] |
| 40. | Wood JD. Enteric nervous system, serotonin, and the irritable bowel syndrome. Curr Opin Gastroenterol. 2001;17:91-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 41. | Coates MD, Johnson AC, Greenwood-Van Meerveld B, Mawe GM. Effects of serotonin transporter inhibition on gastrointestinal motility and colonic sensitivity in the mouse. Neurogastroenterol Motil. 2006;18:464-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | Chen JJ, Li Z, Pan H, Murphy DL, Tamir H, Koepsell H, Gershon MD. Maintenance of serotonin in the intestinal mucosa and ganglia of mice that lack the high-affinity serotonin transporter: Abnormal intestinal motility and the expression of cation transporters. J Neurosci. 2001;21:6348-6361. [PubMed] |
| 43. | Morita H, Mochiki E, Takahashi N, Kawamura K, Watanabe A, Sutou T, Ogawa A, Yanai M, Ogata K, Fujii T, Ohno T, Tsutsumi S, Asao T, Kuwano H. Effects of 5-HT2B, 5-HT3 and 5-HT4 receptor antagonists on gastrointestinal motor activity in dogs. World J Gastroenterol. 2013;19:6604-6612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 44. | Tack J, Sarnelli G. Serotonergic modulation of visceral sensation: upper gastrointestinal tract. Gut. 2002;51 Suppl 1:i77-i80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 59] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 45. | Guha D, Ghosh A. Effect of cerebellar modulation on gastroduodenal PGE2 and 5-HT content of rat. Indian J Exp Biol. 1995;33:361-364. [PubMed] |
| 46. | Kato S, Matsuda N, Matsumoto K, Wada M, Onimaru N, Yasuda M, Amagase K, Horie S, Takeuchi K. Dual role of serotonin in the pathogenesis of indomethacin-induced small intestinal ulceration: pro-ulcerogenic action via 5-HT3 receptors and anti-ulcerogenic action via 5-HT4 receptors. Pharmacol Res. 2012;66:226-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 47. | Singh P, Dutta SR, Guha D. Gastric mucosal protection by aegle marmelos against gastric mucosal damage: role of enterochromaffin cell and serotonin. Saudi J Gastroenterol. 2015;21:35-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 48. | Schicho R, Krueger D, Zeller F, Von Weyhern CW, Frieling T, Kimura H, Ishii I, De Giorgio R, Campi B, Schemann M. Hydrogen sulfide is a novel prosecretory neuromodulator in the Guinea-pig and human colon. Gastroenterology. 2006;131:1542-1552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 173] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 49. | Krueger D, Foerster M, Mueller K, Zeller F, Slotta-Huspenina J, Donovan J, Grundy D, Schemann M. Signaling mechanisms involved in the intestinal pro-secretory actions of hydrogen sulfide. Neurogastroenterol Motil. 2010;22:1224-1231, e319-e320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 50. | Dhaese I, Lefebvre RA. Myosin light chain phosphatase activation is involved in the hydrogen sulfide-induced relaxation in mouse gastric fundus. Eur J Pharmacol. 2009;606:180-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 51. | Fiorucci S, Antonelli E, Distrutti E, Rizzo G, Mencarelli A, Orlandi S, Zanardo R, Renga B, Di Sante M, Morelli A, Cirino G, Wallace JL. Inhibition of hydrogen sulfide generation contributes to gastric injury caused by anti-inflammatory nonsteroidal drugs. Gastroenterology. 2005;129:1210-1224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 314] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 52. | Aboubakr EM, Taye A, El-Moselhy MA, Hassan MK. Protective effect of hydrogen sulfide against cold restraint stress-induced gastric mucosal injury in rats. Arch Pharm Res. 2013;36:1507-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 53. | Magierowski M, Jasnos K, Kwiecien S, Drozdowicz D, Surmiak M, Strzalka M, Ptak-Belowska A, Wallace JL, Brzozowski T. Endogenous prostaglandins and afferent sensory nerves in gastroprotective effect of hydrogen sulfide against stress-induced gastric lesions. PLoS One. 2015;10:e0118972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 54. | Kondo T, Oshima T, Koseki J, Hattori T, Kase Y, Tomita T, Fukui H, Watari J, Miwa H. Effect of rikkunshito on the expression of substance P and CGRP in dorsal root ganglion neurons and voluntary movement in rats with experimental reflux esophagitis. Neurogastroenterol Motil. 2014;26:913-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 55. | Kwiecien S, Magierowska K, Magierowski M, Surmiak M, Hubalewska-Mazgaj M, Pajdo R, Sliwowski Z, Chmura A, Wojcik D, Brzozowski T. Role of sensory afferent nerves, lipid peroxidation and antioxidative enzymes in the carbon monoxide-induced gastroprotection against stress ulcerogenesis. J Physiol Pharmacol. 2016;67:717-729. [PubMed] |
| 56. | Saeki T, Ohno T, Boku K, Saigenji K, Katori M, Majima M. Mechanism of prevention by capsaicin of ethanol-induced gastric mucosal injury--a study in the rat using intravital microscopy. Aliment Pharmacol Ther. 2000;14 Suppl 1:135-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 57. | Holzer P, Guth PH. Neuropeptide control of rat gastric mucosal blood flow. Increase by calcitonin gene-related peptide and vasoactive intestinal polypeptide, but not substance P and neurokinin A. Circ Res. 1991;68:100-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 78] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 58. | Peskar BM, Ehrlich K, Peskar BA. Role of ATP-sensitive potassium channels in prostaglandin-mediated gastroprotection in the rat. J Pharmacol Exp Ther. 2002;301:969-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 80] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 59. | Namai F, Yamamoto Y, Sato T, Ogita T, Shimosato T. Recombinant mouse calcitonin gene-related peptide secreted by Lactococcus lactis inhibits lipopolysaccharide-induced inflammatory response in macrophages. Anim Sci J. 2018;89:1707-1711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 60. | Zhang S, Liu H, Xu Q, Huang F, Xu R, Liu Q, Lv Y. Role of capsaicin sensitive sensory nerves in ischemia reperfusion-induced acute kidney injury in rats. Biochem Biophys Res Commun. 2018;506:176-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 61. | Evangelista S, Renzi D. A protective role for calcitonin gene-related peptide in water-immersion stress-induced gastric ulcers in rats. Pharmacol Res. 1997;35:347-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 62. | Li DM, Li LX. The role of neuropeptides in nasal mucosa hypersensitivity (in Chinese). Guowai Yixue Er Bi Yan Hou Kexue Fence. 2002;26:79-83. [DOI] [Full Text] |
| 63. | Delgado M, Munoz-Elias EJ, Gomariz RP, Ganea D. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide enhance IL-10 production by murine macrophages: in vitro and in vivo studies. J Immunol. 1999;162:1707-1716. [PubMed] |
| 64. | Yuan HX, Yang M. Effect of the No.2 prescription for the treatment of the stomachon on gastrointestinal hormones in CAG model rats (in Chinese). Shijie Huaren Xiaohua Zazhi. 2000;8:1424-1425. [DOI] [Full Text] |
| 65. | Tunçel N, Erkasap N, Sahintürk V, Ak DD, Tunçel M. The protective effect of vasoactive intestinal peptide (VIP) on stress-induced gastric ulceration in rats. Ann N Y Acad Sci. 1998;865:309-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 66. | Tunçel N, Tunçel M, Aboul-Enein HY. Effects of the vasoactive intestinal peptide on stress-induced mucosal ulcers and modulation of methylation of histamine in gastric tissue of the rats. Farmaco. 2003;58:449-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 67. | Arai I, Muramatsu M, Aihara H. Body temperature dependency of gastric regional blood flow, acid secretion and ulcer formation in restraint and water-immersion stressed rats. Jpn J Pharmacol. 1986;40:501-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 68. | Wang YH, Ai HB, Zhang YY, Cui XY. Effects and mediated pathway of electrical stimulation of nucleus ambiguus on gastric motility and mucus secretion in rats. Scand J Clin Lab Invest. 2007;67:489-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 69. | Zhang G, Yu L, Chen ZY, Zhu JS, Hua R, Qin X, Cao JL, Zhang YM. Activation of corticotropin-releasing factor neurons and microglia in paraventricular nucleus precipitates visceral hypersensitivity induced by colorectal distension in rats. Brain Behav Immun. 2016;55:93-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 70. | Taché Y, Stephens RL, Ishikawa T. Central nervous system action of TRH to influence gastrointestinal function and ulceration. Ann N Y Acad Sci. 1989;553:269-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 71. | Király A, Süto G, Tam B, Hermann V, Mózsik G. Vagus-mediated activation of mucosal mast cells in the stomach: effect of ketotifen on gastric mucosal lesion formation and acid secretion induced by a high dose of intracisternal TRH analogue. J Physiol Paris. 2000;94:131-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 72. | Tache Y. Brainstem neuropeptides and vagal protection of the gastric mucosal against injury: role of prostaglandins, nitric oxide and calcitonin-gene related peptide in capsaicin afferents. Curr Med Chem. 2012;19:35-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 73. | Gyires K. Neuropeptides and gastric mucosal homeostasis. Curr Top Med Chem. 2004;4:63-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 74. | Holzer P, Lippe IT. Stimulation of afferent nerve endings by intragastric capsaicin protects against ethanol-induced damage of gastric mucosa. Neuroscience. 1988;27:981-987. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 162] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 75. | Sun H, Liu Z, Ma X. Interactions between astrocytes and neurons in the brainstem involved in restraint water immersion stress-induced gastric mucosal damage. Neuroreport. 2016;27:151-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 76. | Davis SF, Derbenev AV, Williams KW, Glatzer NR, Smith BN. Excitatory and inhibitory local circuit input to the rat dorsal motor nucleus of the vagus originating from the nucleus tractus solitarius. Brain Res. 2004;1017:208-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 97] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 77. | Sun HZ, Zhao SZ, Cui XY, Ai HB. Effects and mechanisms of L-glutamate microinjected into nucleus ambiguus on gastric motility in rats. Chin Med J (Engl). 2010;123:1052-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 78. | Zhang YY, Zhu WX, Cao GH, Cui XY, Ai HB. c-Fos expression in the supraoptic nucleus is the most intense during different durations of restraint water-immersion stress in the rat. J Physiol Sci. 2009;59:367-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 79. | Zhang JF, Zheng F. The role of paraventricular nucleus of hypothalamus in stress-ulcer formation in rats. Brain Res. 1997;761:203-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 80. | Ferguson AV, Marcus P, Spencer J, Wallace JL. Paraventricular nucleus stimulation causes gastroduodenal mucosal necrosis in the rat. Am J Physiol. 1988;255:R861-R865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 81. | Hierlihy LE, Wallace JL, Ferguson AV. Autonomic pathways in development of neural stimulation-induced gastric mucosal damage. Am J Physiol. 1994;266:G179-G185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 82. | Lu CL, Li ZP, Zhu JP, Zhao DQ, Ai HB. Studies on functional connections between the supraoptic nucleus and the stomach in rats. J Physiol Sci. 2011;61:191-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 83. | Ulrich-Lai YM, Herman JP. Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci. 2009;10:397-409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2503] [Cited by in RCA: 2205] [Article Influence: 137.8] [Reference Citation Analysis (0)] |
| 84. | Kim MJ, Loucks RA, Palmer AL, Brown AC, Solomon KM, Marchante AN, Whalen PJ. The structural and functional connectivity of the amygdala: from normal emotion to pathological anxiety. Behav Brain Res. 2011;223:403-410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 688] [Cited by in RCA: 623] [Article Influence: 44.5] [Reference Citation Analysis (0)] |
| 85. | Pacák K, Palkovits M. Stressor specificity of central neuroendocrine responses: implications for stress-related disorders. Endocr Rev. 2001;22:502-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 457] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 86. | Roozendaal B, McEwen BS, Chattarji S. Stress, memory and the amygdala. Nat Rev Neurosci. 2009;10:423-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1110] [Cited by in RCA: 1237] [Article Influence: 77.3] [Reference Citation Analysis (0)] |
| 87. | Myers B, McKlveen JM, Herman JP. Neural Regulation of the Stress Response: The Many Faces of Feedback. Cell Mol Neurobiol. 2012;32:683-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 127] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 88. | Myers B, Greenwood-Van Meerveld B. Role of anxiety in the pathophysiology of irritable bowel syndrome: importance of the amygdala. Front Neurosci. 2009;3:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 89. | He F, Ai H, Wang M, Wang X, Geng X. Altered Neuronal Activity in the Central Nucleus of the Amygdala Induced by Restraint Water-Immersion Stress in Rats. Neurosci Bull. 2018;34:1067-1076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 90. | Liubashina O, Jolkkonen E, Pitkänen A. Projections from the central nucleus of the amygdala to the gastric related area of the dorsal vagal complex: a Phaseolus vulgaris-leucoagglutinin study in rat. Neurosci Lett. 2000;291:85-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 91. | Zhang X, Cui J, Tan Z, Jiang C, Fogel R. The central nucleus of the amygdala modulates gut-related neurons in the dorsal vagal complex in rats. J Physiol. 2003;553:1005-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 92. | Innes DL, Tansy MF. Gastric mucosal ulceration associated with electrochemical stimulation of the limbic brain. Brain Res Bull. 1980;5 Suppl 1:33-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 93. | Liubashina O, Bagaev V, Khotiantsev S. Amygdalofugal modulation of the vago-vagal gastric motor reflex in rat. Neurosci Lett. 2002;325:183-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 94. | He F, Ai HB. Effects of electrical stimulation at different locations in the central nucleus of amygdala on gastric motility and spike activity. Physiol Res. 2016;65:693-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 95. | Henke PG, Ray A, Sullivan RM. The amygdala. Emotions and gut functions. Dig Dis Sci. 1991;36:1633-1643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 96. | Zhao DQ, Gong SN, Ma YJ, Zhu JP. Medial prefrontal cortex exacerbates gastric dysfunction of rats upon restraint water‑immersion stress. Mol Med Rep. 2019;20:2303-2315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 97. | Vertes RP. Analysis of projections from the medial prefrontal cortex to the thalamus in the rat, with emphasis on nucleus reuniens. J Comp Neurol. 2002;442:163-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 196] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 98. | Mitchell AS. The mediodorsal thalamus as a higher order thalamic relay nucleus important for learning and decision-making. Neurosci Biobehav Rev. 2015;54:76-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 198] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 99. | Gong SN, Zhu JP, Ma YJ, Zhao DQ. Proteomics of the mediodorsal thalamic nucleus of rats with stress-induced gastric ulcer. World J Gastroenterol. 2019;25:2911-2923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 100. | Gatón J, Fernández de la Gándara F, Velasco A. The role of the neurotransmitters acetylcholine and noradrenaline in the pathogenesis of stress ulcers. Comp Biochem Physiol C. 1993;106:125-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 101. | Yu Z, Wang P, Tan XQ, Xu XL, Li CW, Huang BL. The effects of microinjection of acetylcholine into nucleus tractus salitarious on electroacupuncture against stress mucosa ijury in rats (in Chinese). Xianning Xueyuan Xuebao (Yixueban). 2007;21:34-36. [DOI] [Full Text] |
| 102. | Glavin GB, Szabo S. Dopamine in gastrointestinal disease. Dig Dis Sci. 1990;35:1153-1161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 75] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 103. | Glavin GB. Central dopamine involvement in experimental gastrointestinal injury. Prog Neuropsychopharmacol Biol Psychiatry. 1992;16:217-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 104. | Glavin GB. Dopamine: a stress modulator in the brain and gut. Gen Pharmacol. 1992;23:1023-1026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 105. | Xing LP, Balaban C, Seaton J, Washington J, Kauffman G. Mesolimbic dopamine mediates gastric mucosal protection by central neurotensin. Am J Physiol. 1991;260:G34-G38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 106. | Zhao DQ, Lu CL, Ai HB. The role of catecholaminergic neurons in the hypothalamus and medullary visceral zone in response to restraint water-immersion stress in rats. J Physiol Sci. 2011;61:37-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 107. | Nakanishi S. Molecular diversity of glutamate receptors and implications for brain function. Science. 1992;258:597-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1900] [Cited by in RCA: 1928] [Article Influence: 58.4] [Reference Citation Analysis (0)] |
| 108. | Moriyoshi K, Masu M, Ishii T, Shigemoto R, Mizuno N, Nakanishi S. Molecular cloning and characterization of the rat NMDA receptor. Nature. 1991;354:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1337] [Cited by in RCA: 1373] [Article Influence: 40.4] [Reference Citation Analysis (0)] |
| 109. | Nakanishi S, Nakajima Y, Masu M, Ueda Y, Nakahara K, Watanabe D, Yamaguchi S, Kawabata S, Okada M. Glutamate receptors: brain function and signal transduction. Brain Res Brain Res Rev. 1998;26:230-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 248] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 110. | Nakanishi S, Masu M. Molecular diversity and functions of glutamate receptors. Annu Rev Biophys Biomol Struct. 1994;23:319-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 356] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 111. | Yao SX, Ai HB. Effects of water-immersion restraint stress on the contents of free amino acid in brain tissue of rats (in Chinese). Shengwu Yixue Gongcheng Yanjiu. 2006;25:96-99. [DOI] [Full Text] |
| 112. | Zhou SY, Lu YX, Yao H, Owyang C. Spatial organization of neurons in the dorsal motor nucleus of the vagus synapsing with intragastric cholinergic and nitric oxide/VIP neurons in the rat. Am J Physiol Gastrointest Liver Physiol. 2008;294:G1201-G1209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 113. | Krowicki ZK, Arimura A, Nathan NA, Hornby PJ. Hindbrain effects of PACAP on gastric motor function in the rat. Am J Physiol. 1997;272:G1221-G1229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 114. | Shi Y, Ai HB, Cui XY. Effect of intracerebroventricular injection of γ-aminobutyric acid on restrain water-immersion stress-induced gastric ulcer in rats (in Chinese). Shengwu Yixue Gongcheng Yanjiu. 2007;26:55-58. [DOI] [Full Text] |
| 115. | Sofroniew MV, Glasmann W. Golgi-like immunoperoxidase staining of hypothalamic magnocellular neurons that contain vasopressin, oxytocin or neurophysin in the rat. Neuroscience. 1981;6:619-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 148] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 116. | Rogers RC, Hermann GE. Oxytocin, oxytocin antagonist, TRH, and hypothalamic paraventricular nucleus stimulation effects on gastric motility. Peptides. 1987;8:505-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 134] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 117. | Grassi M, Drago F. Effects of oxytocin on emotional stress and stress-induced gastric lesions. J Physiol Paris. 1993;87:261-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 118. | Zhao DQ, Ai HB. Oxytocin and vasopressin involved in restraint water-immersion stress mediated by oxytocin receptor and vasopressin 1b receptor in rat brain. PLoS One. 2011;6:e23362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 119. | Zhu J, Chang L, Xie J, Ai H. Arginine Vasopressin Injected into the Dorsal Motor Nucleus of the Vagus Inhibits Gastric Motility in Rats. Gastroenterol Res Pract. 2016;2016:4618672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 120. |
Zhao DQ, Bi YJ, Gong SN, Li PF. Neurons in the medulla oblongata related to gastric mucosal lesion of rats subjected to restraint water-immersion stress.
|
| 121. | Sun HZ, Zheng S, Lu K, Hou FT, Bi JX, Liu XL, Wang SS. Hydrogen sulfide attenuates gastric mucosal injury induced by restraint water-immersion stress via activation of KATP channel and NF-κB dependent pathway. World J Gastroenterol. 2017;23:87-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 122. | Travagli RA, Hermann GE, Browning KN, Rogers RC. Brainstem circuits regulating gastric function. Annu Rev Physiol. 2006;68:279-305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 416] [Cited by in RCA: 388] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 123. | Pagani FD, Norman WP, Kasbekar DK, Gillis RA. Localization of sites within dorsal motor nucleus of vagus that affect gastric motility. Am J Physiol. 1985;249:G73-G84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 124. | Laughton WB, Powley TL. Localization of efferent function in the dorsal motor nucleus of the vagus. Am J Physiol. 1987;252:R13-R25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 26] [Article Influence: 0.7] [Reference Citation Analysis (0)] |