Published online Apr 28, 2020. doi: 10.3748/wjg.v26.i16.1847
Peer-review started: February 2, 2020
First decision: February 29, 2020
Revised: March 6, 2020
Accepted: April 17, 2020
Article in press: April 17, 2020
Published online: April 28, 2020
Processing time: 85 Days and 22 Hours
Malignant gastric outlet obstruction (MGOO) is a clinical condition characterized by the mechanical obstruction of the pylorus or the duodenum due to tumor compression/infiltration, with consequent reduction or impossibility of an adequate oral intake. MGOO is mainly secondary to advanced pancreatic or gastric cancers, and significantly impacts on patients’ survival and quality of life. Patients suffering from this condition often present with intractable vomiting and severe malnutrition, which further compromise therapeutic chances. Currently, palliative strategies are based primarily on surgical gastrojejunostomy and endoscopic enteral stenting with self-expanding metal stents. Several studies have shown that surgical approach has the advantage of a more durable relief of symptoms and the need of fewer re-interventions, at the cost of higher procedure-related risks and longer hospital stay. On the other hand, enteral stenting provides rapid clinical improvement, but have the limit of higher stent dysfunction rate due to tumor ingrowth and a subsequent need of frequent re-interventions. Recently, a third way has come from interventional endoscopic ultrasound, through the development of endoscopic ultrasound-guided gastroenterostomy technique with lumen-apposing metal stent. This new technique may ideally encompass the minimal invasiveness of an endoscopic procedure and the long-lasting effect of the surgical gastrojejunostomy, and brought encouraging results so far, even if prospective comparative trial are still lacking. In this Review, we described technical aspects and clinical outcomes of the above-cited therapeutic approaches, and discussed the open questions about the current management of MGOO.
Core tip: In the last decades, surgical gastrojejunostomy and enteral stenting have represented the main palliative strategies for patient with malignant gastric outlet obstruction. Although they showed good clinical efficacy, these approaches suffer from limits secondary to the high procedure-related risks and longer hospital stay (surgery) or the need subsequent re-interventions due to stent dysfunction (endoscopic stenting). The recently proposed endoscopic ultrasonography-guided gastroenterostomy may include both advantages of a minimally invasive endoscopic procedure and the long-lasting benefits of the gastrojejunostomy. However, such procedure is not standardized and prospective comparative studies are needed to define the best strategy for these patients.
- Citation: Troncone E, Fugazza A, Cappello A, Blanco GDV, Monteleone G, Repici A, Teoh AYB, Anderloni A. Malignant gastric outlet obstruction: Which is the best therapeutic option? World J Gastroenterol 2020; 26(16): 1847-1860
- URL: https://www.wjgnet.com/1007-9327/full/v26/i16/1847.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i16.1847
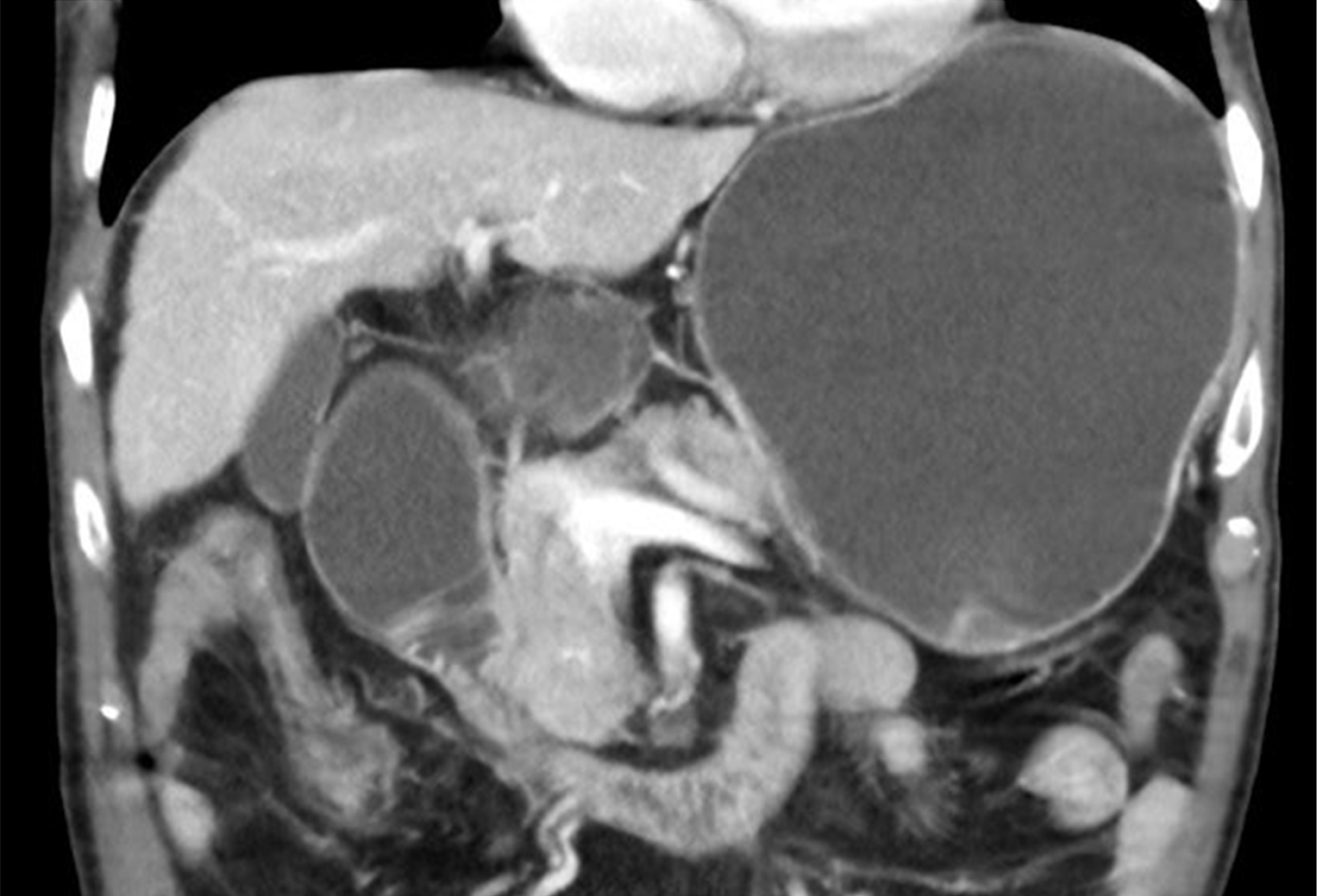
Malignancies of the pancreas, biliary tract and gastro-duodenum are often diagnosed in advanced stages, in many cases are not amenable of curative surgical treatment and thus may require prolonged radio-chemotherapy regimens or palliative care. In this setting, malignant gastric outlet obstruction (MGOO) is defined as the mechanical obstruction of the pylorus or the duodenum secondary to compression/infiltration from advanced loco-regional malignancies that make difficult or even impossible the oral feeding (Figure 1). Patients with MGOO typically present with nausea and vomiting, which could associate with abdominal pain, weight loss, malnutrition and dehydration secondary to poor oral intake[1]. The most frequent cause of MGOO in the Western countries is pancreatic adenocarcinoma, which lead to obstructive symptoms in about 15%–20% of patients during the disease course, while gastric adenocarcinoma is the most common cause in Asiatic population[2-7]. Less common causes are duodenal or ampullary neoplasms, biliary cancers, lymphomas or adenopathies from other malignancies[1]. Effective treatment of MGOO is of paramount importance either for patients who have to face radio-chemotherapy regimens, as for those at late stage of disease who only require supportive care aimed at improving quality of life. For many decades MGOO has been managed with open surgical gastrojejunostomy, during which also biliary bypass was performed in case of concomitant biliary obstruction[8-10]. However, most patients with MGOO often present with advanced disease and are not optimal candidates for open surgery. Due to the high surgical risk and the short life expectation (i.e. 3-4 mo) that characterize the majority of these patients, less invasive approaches have been developed and proposed over time, aimed at providing fast and effective relieve of symptoms and return to an adequate oral feeding with the highest safety, the shortest hospitalization time and the lowest costs. Such an ambitious goal has been pursued with the use of enteral stents, with the development of less invasive surgical techniques (i.e. laparoscopic gastrojejunostomy) and, more recently, with the progress of interventional endoscopic ultrasonography (EUS), and the development of EUS-guided gastroenterostomy techniques (EUS-GE) (Figure 2). A consistent body of literature exists about the outcome and safety of such interventions in the setting of MGOO. Nevertheless, few randomized clinical trials have been conducted to compare these different approaches, and so there is still uncertainty about the best strategy to choose.
In this review, we aimed to summarize the available evidence on the most common palliative strategies for MGOO, focusing on the strength and weakness of each approaches and discussing possible unanswered questions that should be addressed in future studies.
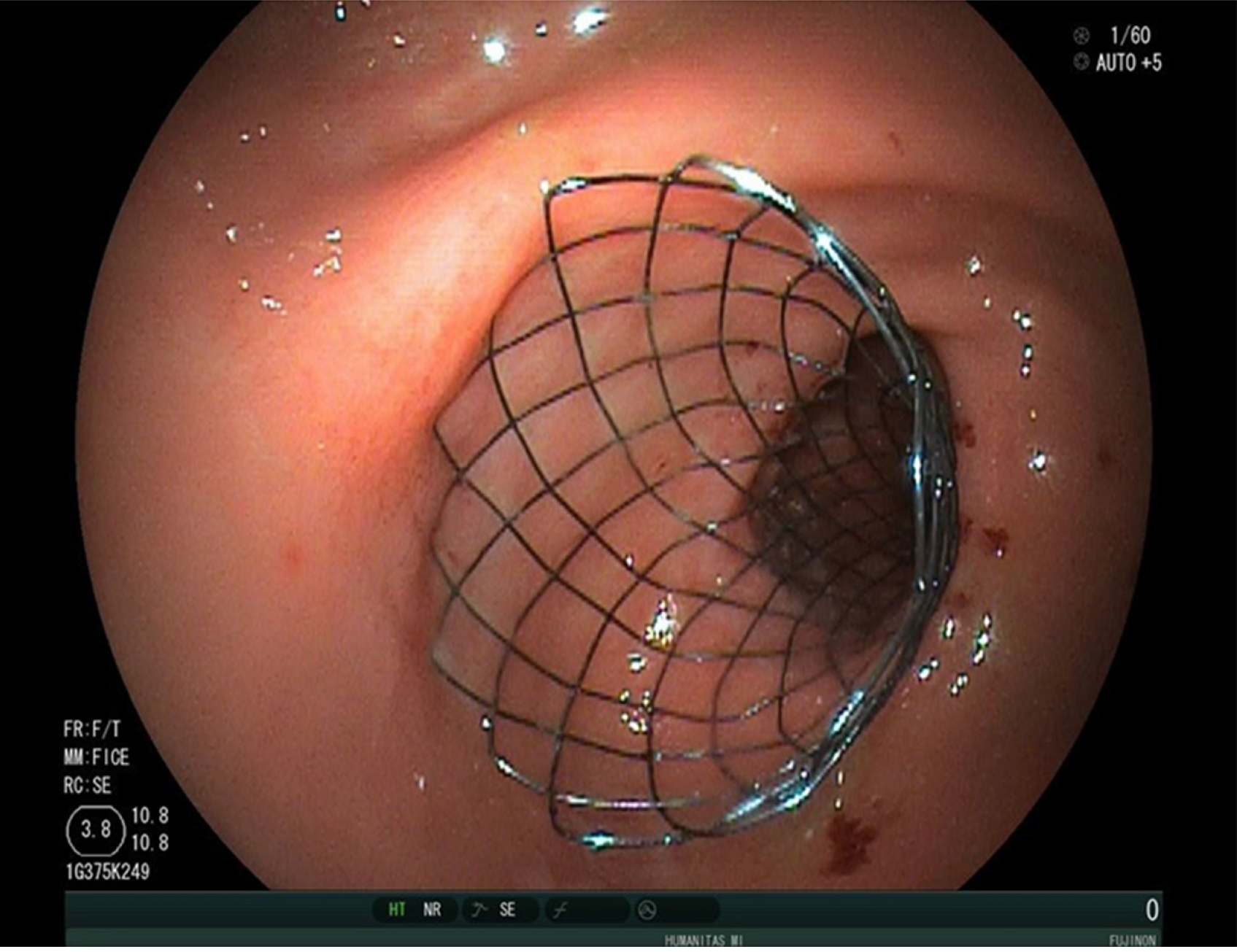
From the late 90’s endoscopic enteral stenting has been proposed as a minimal invasive treatment for MGOO, using the experience gained from the use of expanding and self-expanding metal stents (SEMS) in the setting of malignant esophageal strictures[11-13]. Generally, a wire is passed through the gastro-duodenal stricture under endoscopic and fluoroscopic assistance, and subsequently the metal stent is passed over the wire and released across the stenosis. According to specific endoscopist’s preference, a soft angled wire can be used first to pass the stricture, and then exchanged with a stiffer one using a catheter. Moreover, passing a catheter over the wire allow to inject contrast to define the anatomy (i.e. length, angulation) of the stenosis, in order to optimize the size of the stent. Over the time, different techniques to deploy enteral stents across gastro-duodenal strictures have been described. After positioning a wire across the stricture, interventional radiologists could deploy the stent with an over-the-wire technique exclusively under fluoroscopic assistance[14,15]. Alternatively, the stent can be deployed under endoscopic and fluoroscopic view; using the over-the-wire technique the endoscope is positioned parallel to the wire, while, with the through-the scope technique, the stent is inserted over the wire into the working channel of the endoscope[16-19]. Currently, the endoscopic deployment using through-the scope stents is the most used technique, and requires therapeutic endoscopes with a large working channel (i.e., ≥ 3.7 mm). Most cases are managed with therapeutic gastroscopes, but cases of dilated stomachs or strictures in the distal duodenum could be better managed with a colonoscope or a duodenoscope[20,21]. Moreover, tight and angulated stenosis could be negotiated in an easier way using a sphincterotome, with the additional advantage of the elevator of the duodenoscope. The choice of the stent largely depend on the stricture anatomy and the endoscopist’s preference. Over time, several different types of enteral metal stent have been designed, which differed in type of metal alloy, length, diameter and radial expansive force after deployment[1,18,22-25] (Figure 3). Currently, available enteral SEMS are made of nitinol, an alloy of nickel and titanium, which confers high flexibility useful for sharply angulated strictures, even if with a weaker expansive radial force compared to other metal stents[26]. Several studies on enteral SEMS for MGOO have shown a high rate of technical success (defined as the successful deployment of the stent across the stricture), which is usually above 90%, and a good rate of clinical success, which ranges from 63% to 97%[27]. Clinical success is generally defined as the relief of obstructive symptoms and the improvement of food oral intake. Adler and colleagues developed a clinical score aimed at providing an objective measure of the oral intake before and after the treatment for MGOO[19]. The Gastric Outlet Obstruction Scoring System (GOOSS) assigns a score of 0 in case of no oral intake, 1 for only liquids, 2 for soft solids and 3 for low-residues or full diet, and currently is the most used score to quantify the clinical improvement after treatment for MGOO[19]. However, high heterogeneity exists among published studies on the definition of this outcome, and a systematic review from Larssen et al[28] highlighted that only 40% of studies used a graded scoring system to evaluate the effect of stenting on MGOO. A systematic review of 32 studies (606 patients) reported a technical success rate of 97% and a clinical success rate of 89% (87% in the intention-to-treat analysis), with a mean GOOSS that rose from 0.4 to 2.4 after treatment, and a resolution of symptoms after a mean period of 4 d[29]. Similarly, a systematic review of 1046 patients treated with duodenal SEMS published in 2007 reported a technical success and clinical success rate of 96% and 89% respectively, with a significant improvement of GOOSS[30]. A recent systematic review from van Halsema and colleagues included 19 prospective studies from 2009 to 2016 and analyzed outcomes of more than 1200 patients with MGOO treated with SEMS. The overall pooled technical success rate was 97.3% and the clinical success rate was 85.7%, thus confirming the high efficacy of this technique[27]. Several studies investigated potential predictive factors of clinical failure or stent dysfunction, in order to optimize the outcome of patients with MGOO undergoing stent placement. The presence of carcinomatosis and a poor performance status (Karnofsky performance status < 50 or Eastern Cooperative Oncology Group status ≥ 3) have been identified as predictors of clinical failure and/or stent dysfunction in several studies[31-34], while chemotherapy after stent placement has been reported as protective[35,36]. In particular, a retrospective study of 228 patients found that carcinomatosis is a predictive factor of clinical failure only if associated with ascites, while carcinomatosis without ascites did not decrease clinical success rate compared to patients without peritoneal disease[37]. The site of the gastro-duodenal stricture (distal vs proximal) and the number of strictures (i.e., ≥ 3) are other factors associated with worst outcome in retrospective studies[31,38].
Adverse event (AE) rate related to SEMS placement ranges from 0% to 30% depending on the definition adopted in the specific study, and includes minor AEs (non-life threatening) such as nausea, vomiting, mild abdominal pain, or major AE, such as bleeding, perforation, stent migration/displacement, cholangitis[19,29,30] . Delayed AEs are usually related to stent dysfunction, secondary to migration or occlusion by food impaction and/or tumor ingrowth/overgrowth. In the report of 1281 patients that received duodenal SEMS, stent obstruction was reported in 12.6%, stent migration in 4.3%, bleeding in 4.1% (major bleeding in 0.8%) and perforation in 1.2%[27]. A recent retrospective study of 220 patients reported a SEMS-related AE rate of 2%, with 3 fatal cases of perforation and an overall rate of re-intervention of 13% after 4 mo[39]. As stated above, stent may occlude due to food impaction or secondary to tumor progression and ingrowth, that is the tumor growth through the mesh of the stent. Stent occlusion leads to reappearance of gastric obstruction symptoms and often needs endoscopic re-intervention that, although feasible and effective, may affect negatively patients’ quality of life and increase costs for the health system, representing one of the main weakness of this approach[40,41]. In order to reduce the risk of stent occlusion, several studies have investigated the possible role of covered SEMS in the setting of MGOO. A systematic review and meta-analysis published in 2014 including 9 studies (849 patients) confirmed that covered SEMS have a lower obstruction rate (RR: 0.42, 95%CI: 0.24-0.73, P = 0.002), but at the same time, as expected, have a higher migration risk (RR: 3.48, 95%CI: 2.16-5.62, P < 0.00001)[42]. Interestingly, the Authors reported no significant difference in technical success rate, clinical success rate, post-stenting dysphagia score, stent patency, overall complications and re-intervention rate between covered and uncovered SEMS group. A more recent systematic review including 1624 patients reported similar results, with comparable technical and clinical success rate between covered and uncovered gastroduodenal SEMS[43]. The Authors highlighted a trend toward a lower dysfunction rate in covered SEMS group (RR: 0.63; 95%CI: 0.45-0.88) when performing a sub-analysis of randomized trials. However, it should be noted that the higher risk of migration was confirmed for covered SEMS, together with a higher overall AE rate in this group (RR: 1.75; 95%CI: 1.09-2.83)[43]. Several technical modifications or precautions (e.g., stent clipping or suturing, anti-migratory design) have been proposed to overcome the migration risk saving the possible advantages of the lower occlusion rate[44-46]. Despite these intriguing alternatives, an increased risk of cholangitis and pancreatitis secondary to the compression/occlusion of the ampulla exists with covered SEMS[22,47-49], and, therefore, uncovered SEMS are still considered the first option in this setting.
Advanced gastro-duodenal or pancreato-biliary malignancies frequently cause biliary obstruction, which is estimated to affect 70%-90% of pancreatic cancer patients during the course of the disease and may appear before, concomitantly or after the onset of MGOO[1]. The presence of MGOO could make the papilla not achievable for standard endoscopic drainage with endoscopic retrograde cholangio-pancreatography (ERCP), in particular for type 1 (proximal to the papilla) or type 2 (at the level of the papilla) duodenal stricture, accordingly to the classification proposed by Mutignani et al[50]. Such cases may be very difficult for therapeutic endoscopists and, therefore, performing biliary drainage with biliary SEMS in patients concomitantly treated for MGOO with risk of impending or future biliary obstruction appears a reasonable strategy, when feasible[50,51]. Although technically challenging, ERCP through an indwelling duodenal stent is feasible and effective, as reported in a recent multicenter retrospective studies on 71 patients, with an overall technical success rate of 85%, which was reduced to 76% in case of duodenal obstruction at the level of the ampulla[52]. The recent progress of interventional EUS, and the possibility to perform EUS-guided biliary drainage (EUS-BD) from the stomach (i.e., EUS-guided hepato-gastrostomy) or from the bulb (EUS-guided choledocho-duodenostomy) has radically changed the approach to the patients with concomitant MGOO and biliary obstruction[53]. EUS-BD in patients with MGOO is safe and effective, even when performed in the same session or with an indwelling duodenal stent, and probably could be considered the first-line strategy to achieve biliary drainage in this setting of patients[54-56].
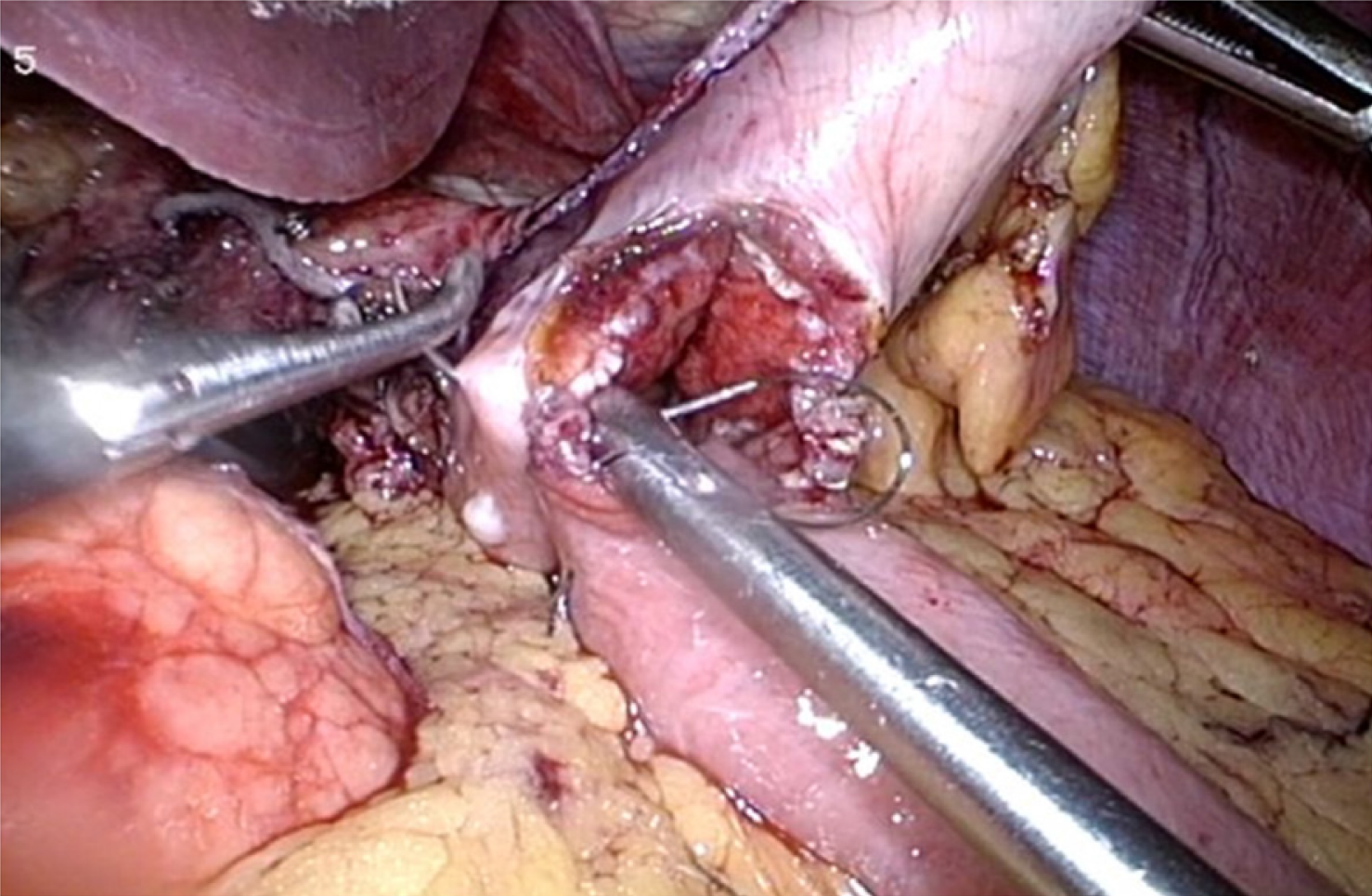
Since the introduction of enteral SEMS for palliation of MGOO, a consistent body of literature has been produced to compare this approach to surgical gastrojejunostomy (GJ), aimed at defining which method, and for which patients, had to be preferred in this clinical setting (Figure 4). Currently, several retrospective cohort studies, but few randomized controlled studies, are available on this topic, with some conflicting results. A multicenter, prospective, randomized study conducted in The Netherlands between 2006 and 2008 randomized 39 patients with MGOO to duodenal stent placement (21 patients) or surgical GJ (18 patients) (SUSTENT Study)[57]. The Authors found a faster relief of symptoms and improvement of GOOSS in the stent group compared to the surgery group (GOOSS score ≥ 2 after a median period of 5 vs 8 d, respectively; P < 0.01), but a more lasting relief in the surgery group, which showed a median period of 72 vs 50 d (P < 0.05) with GOOSS score ≥ 2 after the procedure. Major AEs, recurrent obstructive symptoms and re-interventions were more common after stent placement compared with GJ, and this result was mainly dependent on the risk of stent obstruction in the duodenal stent group[57]. The authors concluded that GJ was the treatment of choice in patients with a life expectancy of 2 mo or longer because of the better long-term results, while stent placement was preferable for patients expected to live less than 2 mo due to the better short-term outcomes. Other randomized trials reported favorable results for the duodenal stenting, mainly in terms of shorter hospitalization time compared to surgical GJ[58,59] (Table 1). However, it should be noted that the cited studies suffer from several limitations, especially for the limited number of patients enrolled and the lack of an adequate statistical power calculation. A recent retrospective study from Jang and colleagues analyzed the outcome of 183 patients who underwent SEMS placement and 127 patients who received surgical GJ over period of 7 years[60]. While the clinical success did not differ significantly between the two groups (79.4% vs 80.1%; P = 0.83), the mean patency duration was significantly longer in the GJ group compared to the stenting group (169.2 vs 96.5 d respectively). Moreover, the GJ group showed longer survival (193.4 vs 119.9 d), and the authors concluded that GJ should be considered the primary treatment option for patients with good performance status and reasonable survival expectancy[60]. It should be underlined that the work from Jang et al[60] suffers from possible selection bias, which is the main limitation of many retrospective studies in this field. Indeed, patients who underwent surgery were “healthier” compared to patients selected for endoscopic palliation (Eastern Cooperative Oncology Group score of 1 vs 2, respectively; P < 0.001), and this could account for the longer survival in this group. Despite the unquestionable value of the study, the presented results are certainly not conclusive, and high quality evidence is still lacking. Recently, results from 27 studies including 2.354 patients (1.306 treated with SEMS and 1.048 with surgical GJ) have been analyzed in a systematic review with meta-analysis, which concluded that patients with acceptable performance status should be primarily considered for a palliative GJ rather than duodenal stenting[61]. In particular, the study confirmed that mean time to oral intake and length of hospital stay were shorter in the SEMS group compared to surgery group, while the frequency of re-interventions was almost three times higher in the SEMS group (OR: 2.95, CI: 1.70-5.14, P < 0.001), thus remarking that stent dysfunction secondary to migration/dislocation or occlusion/obstruction is the main limitation of the duodenal stenting in MGOO[61]. Surgical techniques other than classic open surgical gastrojejunostomy or laparoscopic gastrojejunostomy have been proposed to overcome the risk of post-operative delayed gastric emptying, such as stomach-partitioning GJ with or without Braun entero-enterostomy, and small retrospective studies have compared such technique with enteral stenting[62,63]. Although intriguing, the study design and the limited number of patients included do not yet allow drawing conclusion about the superiority of this approach over classic surgery or stenting, and larger studies are needed. Comparison of costs between GJ and stenting has produced conflicting results, as the shorter hospitalization documented with enteral stents could be counter-balanced by higher costs for re-interventions[4,57,61,64,65]. Taken together, these data highlight that a correct selection of patients is probably the crucial step to achieve a satisfactory clinical outcome for both strategies.
| Ref. | Randomized patients | Treated patients | Type of stent | Surgical technique | Main outcome measures | Technical success | Clinical success | Adverse events | Hospital stay (d, median) | Reintervention | Follow-up |
| Fiori et al[58], 2004 | Stent group: 9 | 9 | Covered SEMS (Ultraflex, Boston) | Open GJ | Gastric emptying (after 15 d) | 9/9 (100%) | 9/9 (100%) | 2/9 (22.2%) | 3.1 | 7/9 (77.7%) | 3 mo |
| Surgery group: 9 | 9 | 9/9 (100%) | 8/9 (88.9%) | 2/9 (22.2%) | 10 | 1/9 (11.1%) | |||||
| Mehta et al[59], 2006 | Stent group: 13 | 12 | Wallstent (Boston)1 | Laparoscopic GJ | Safety quality of life | 10/12 (83.3%) | Significant improvement in Physical Health score after 1 mo in duodenal stent group | 0/10 (0%) | 5.2 | NA | 12 mo |
| Surgery group: 14 | 13 | 13/13 (100%) | 10/13 (76.9%) | 11.4 | |||||||
| Jeurnink et al[57], 2010 | Stent group: 21 | 20 | Wallflex (Boston) | Laparoscopic or open GJ | GOOSS improvement | 20/21 (95.2%) | 17/21 (80.9%) | 8/21 (38.1%) | 7 | 2/21 (9.5%) | Median survival: 72 d (GJ) vs 50 d (SEMS) |
| 17/18 (94.4%) | 14/18 (77.7%) | 5/21 (23.8%) | 15 | 7/182 (38.8%) | |||||||
| Surgery group: 18 | 17 |
The above-cited progress of interventional EUS have enriched the field of therapeutic endoscopy with the possibility to perform trans-luminal procedures, such as trans-gastric or trans-duodenal drainage of peripancreatic fluid collections, gallbladder drainage in patients unfit for surgery or biliary drainage after failed ERCP[66-69]. In the last years, it has been developed and proposed an innovative technique that allows the creation of a stable gastro-jejunal anastomosis through a EUS-guided procedure, named EUS-guided gastroenterostomy (EUS-GE)[70,71]. With this regard, the field of interventional has enormously benefited from the development of dedicated metal stents for trans-luminal interventions. Indeed, lumen-apposing metal stents (LAMS) are fully covered “dumb-bell”-shaped short stent made up of braided nitinol, specifically designed for interventional EUS procedures, with wide anti-migratory flanges which provide a lumen-to-lumen apposition effect[68]. The stent is pre-loaded in a 10.8 French catheter with a through-the-scope delivery system compatible with therapeutic echoendoscope with a working channel ≥ 3.7 mm. In the EUS-GE procedure, the small bowel is punctured from the stomach at the level of the distal duodenum or in the proximal jejunum (i.e. gastroduodenostomy or gastro-jejunostomy, respectively) under EUS and fluoroscopic guidance, with subsequent placement of a LAMS, thus creating a tight and sealed anastomosis, owing to the lumen-to-lumen apposition effect of the stent. The first EUS-GE with LAMS (AXIOSTM stent; Boston Scientific, Marlborough, MA, United States) was described in 2012 in a porcine model by Binmoeller and Shah[72]. In the subsequent years, three different techniques have been described to perform EUS-GE with LAMS: (1) Direct EUS-GE; (2) Assisted EUS-GE, performed using accessory devices (e.g., dilating balloon, single balloon overtube, nasobiliary drain, ultra-slim endoscope) for small bowel loop distension before punction and stent placement; and (3) EUS-guided double balloon-occluded gastrojejunostomy bypass (EPASS)[70,73-75].
Direct EUS-GE requires as first step the puncture of the small bowel loop from the stomach with a 19 Gauge fine needle aspiration (FNA) needle under EUS view. Once confirmed the positioning of the FNA needle into the small bowel through contrast injection under fluoroscopic view, a guide-wire is advanced through the needle into the small bowel, and dilation of the tract with a balloon or a cystotome is performed, to allow the insertion of the stent delivery system (10.8 French) into the small bowel and to open the LAMS. The main technical issues of this technique concern the correct puncturing of the small bowel loop, which is often collapsed and mobile, and the multi-step technique, with several subsequent device exchanges that increase the risk of pushing away the bowel loop with the guide-wire with possible subsequent leakage, perforation or stent mal-deployment. To address these limitations, injection of normal saline (usually about 500 mL) through the duodenal stricture and administering anti-peristaltic drug (e.g. glucagone) could help to reduce peristalsis and to have a good view of the target bowel loop. Moreover, LAMS delivery system has further evolved with the addition of an electrocautery tip [electrocautery-enhanced (EC)-LAMS-HOT-AXIOSTM, Boston Scientific Corp., Marlborough, Massachusetts, United States] which allows a single-stage access to the small bowel distal to the obstruction, without the need of multiple exchanges[67,76-78].
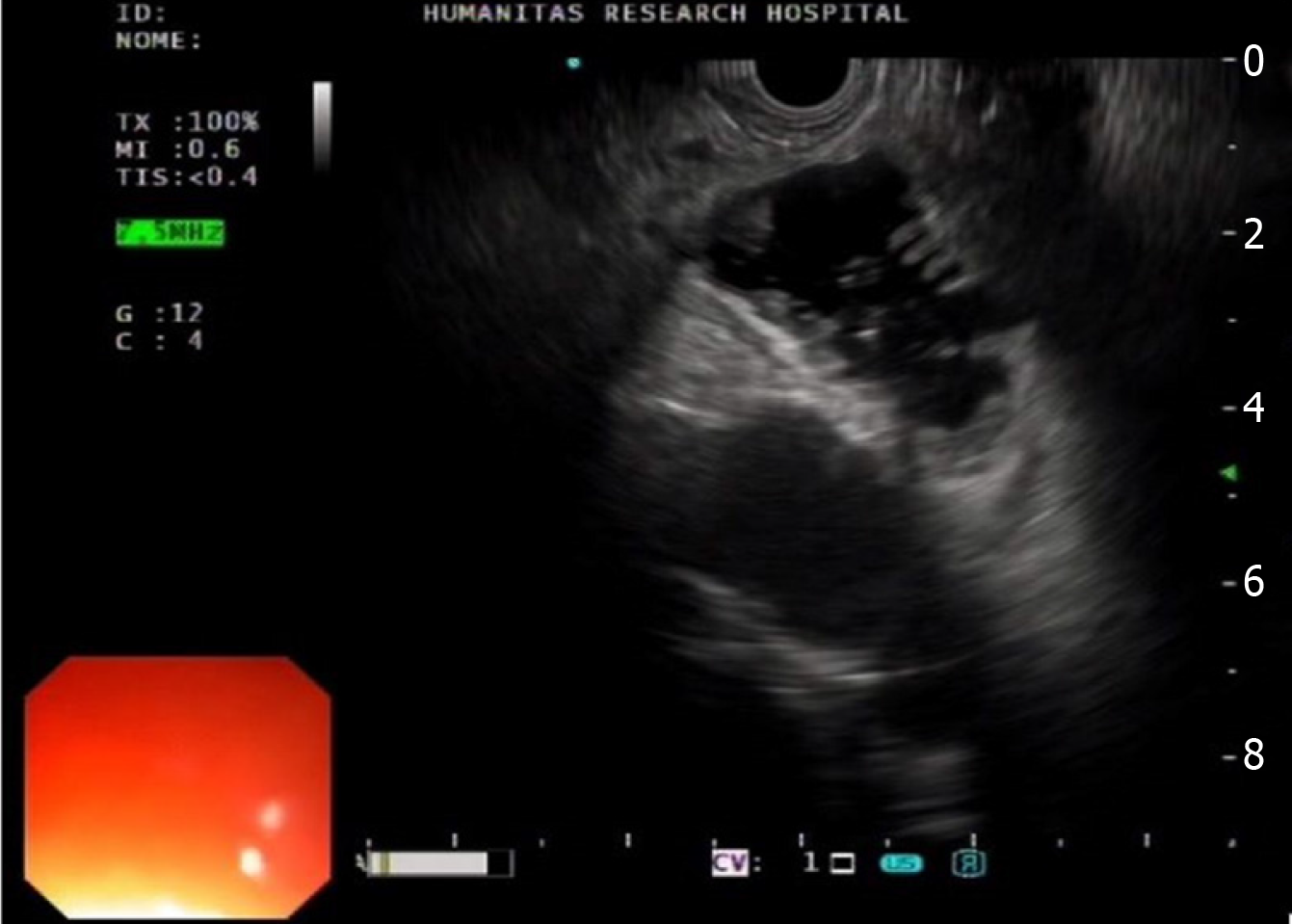
As resumed above, the assisted EUS-GE technique requires the distension of the jejunal loops distal to the strictures with infusion of normal saline directly injected through an ultra-slim endoscope which is advanced (when possible) beyond the stenosis or passing a nasobiliary catheter over a wire. Alternatively, a balloon is passed over a wire into the jejunum and then inflated, providing a guide for the EUS view to identify the target bowel loop (Figure 5). The main limitation of this technique is related to the difficulties in advancing per-orally the mentioned devices through an often tight and angulated duodenal stricture.
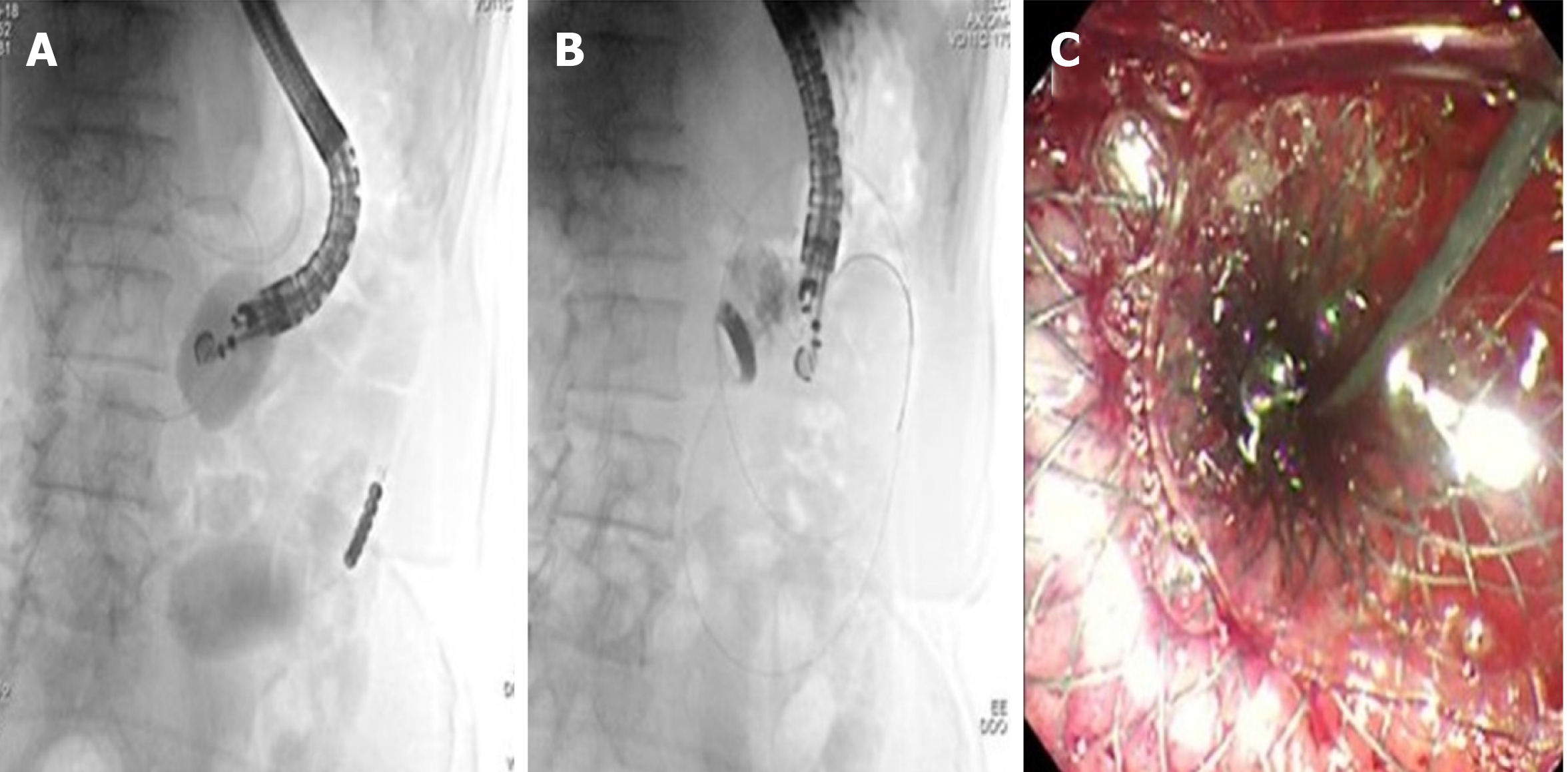
The EPASS technique requires a special double-balloon enteric tube (Tokyo Medical University type; Create Medic Co., Ltd, Yokohama, Japan) specifically designed for this procedure[79,80]. The double-balloon tube is inserted perorally over a previously placed guidewire and advanced through the stenosis. Then, both balloons are filled with saline to hold the small intestine open and fixed, and saline with contrast material is introduced into the space between the two balloons to distend the small bowel lumen (Figure 6). At this point, the echoendoscope is introduced into the stomach and the distended duodenum or jejunum is identified at the EUS image. The subsequent LAMS placement can be performed with the multi-step procedure described above, or with a single-step procedure using the EC-LAMS.
Regardless of the technique adopted, data from several studies reported high technical and clinical success rate for EUS-GE in MGOO using 10 mm o 15 mm diameter LAMS, ranging from 87%-96% and 81%-92% respectively[81]. In 2015, Khashab et al[70] and colleagues reported the first series of EUS-GE in both malignant (3 patients) and benign (7 patients) gastric outlet obstruction using the direct or the balloon-assisted technique[70]. The authors reported a technical success of 90% and clinical success of 100%, with resumption of soft or normal diet in all patients with technical success. Moreover, no AEs were reported and patients did not experienced symptom recurrence during a mean follow-up period of 150 d[70]. Itoi et al[82] reported similar outcomes in a prospective study of 20 EUS-GE performed with the EPASS technique, with a technical success of 90% and a significant improvement of GOOSS. Despite a 10% (2 patients) with stent mal-positioning, no further AEs were reported and no patients reported stent migration or occlusion needing re-interventions[82]. Other series reported a rate of AEs ranging from 0 to 21%, including pneumoperitoneum, gastric leak, bleeding, peritonitis or abdominal pain[74,80,83-85]. A multicenter study comparing the direct and the balloon-assisted technique reported no significant differences in technical and clinical success and AE rate[86]. EUS-GE has been compared to enteral stenting in two retrospective studies[80,85]. Technical success, length of hospitalization and safety were similar, while in the study from Ge et al[85] a higher rate of initial clinical success was found in the EUS-GE group (95.8% vs 76.3%, P = 0.042)[85]. Strikingly, stent failure requiring re-intervention was significantly lower in EUS-GE group compared to enteral stent group in both studies. A multicenter retrospective study compared EUS-GE (30 patients) and surgical GJ (63 patients)[83]. Despite surgical GJ showed a higher technical success (100% vs 87%, P = 0.009), clinical success and symptoms recurrence were similar, even if a trend toward more frequent recurrent obstruction in the EUS-GE group (3% vs 14%, P = 0.08) was reported. A non-significant higher rate of AE rate was found in surgical GJ group (16% vs 25 %, P = 0.3), however it should be noted that surgery group underwent open GJ, and therefore these results may be not generalizable to laparoscopic GJ[83] (Table 2).
| Ref. | Study characteristics | EUS-GE technique | Comparison group | Number of patients | Technical success | Clinical success | Hospital stay (d, median) | Symptom recurrence or re-intervention | Adverse events |
| Chen et al[80], 2017 | Multicenter; Retrospective | EPASS; Balloon-assisted; Direct | Duodenal SEMS | EUS-GE: 30; | 26/30 (86.7%); | 25/30 (83.3%); | 11.3 ± 6.6; | 1/30 (4.3%); | 5/30 (16.7%); |
| SEMS: 52 | 49/52 (94.2%) | 35/52 (67.3%) | 9.5 ± 8.3 | 10/52 (28.6%) | 6/52 (11.5%) | ||||
| Khashab et al[83], 2017 | Multicenter; Retrospective | EPASS; Balloon-assisted; Direct | Open GJ | EUS-GE: 30; | 26/30 (87%); | 26/30 (87%); | 11.6 ± 6.6; | 1/30 (3%); | 5/30 (16.7%); |
| Open GJ: 63 | 63/63 (100%) | 57/63 (90%) | 12 ± 8.2 | 9/63 (14%) | 16/63 (25%) | ||||
| Perez-Miranda et al[84], 2017 | Multicenter; Retrospective | Assisted; Direct | Laparoscopic GJ (LGJ) | EUS-GE: 251 | 23/25 (88%); | 21/25 (90%); | 9.4; | NA; | 3/25 (12%); |
| LGJ: 29 | 29/29 (100%) | 28/29 (90%) | 8.9 | NA | 12/29 (41%) | ||||
| Ge et al[85], 2019 | Single-center; Retrospective | Assisted; Direct | Duodenal SEMS | EUS-GE: 22; SEMS: 78 | 22/22 (100%); | 21/22 (95.5%); | 7.4 ± 9.1; | 2/22 (8.3%); | 5/22 (20.8%); |
| 78/78 (100%) | 60/78 (76.3%) | 7.9 ± 8.2 | 31/78 (32%) | 39/78 (40.2%) |
Taken together, these data propose EUS-GE as a valuable minimal invasive option for patient with MGOO (Figure 7). The main limitations concern the technical difficulty of the procedure, which is not yet standardized and requires high skilled therapeutic endoscopists. Moreover, high quality prospective studies comparing the three different palliative strategies are still lacking.
Palliation of MGOO may be challenging, and multi-disciplinary team is often needed to evaluate the best therapeutic strategy taking into account the patient’s performance status, life expectancy, the need of chemo-radiotherapy, surgical risks and, importantly, the patient’s preference. In this scenario, the therapeutic endoscopist may offer effective minimal invasive approaches. Enteral stenting provides rapid relief of obstructive symptoms and a short hospital stays through a relatively safe endoscopic procedure compared to surgical GJ. On the other hand, SEMS suffer from high rate of stent failure and need of re-intervention on long-term period, mainly secondary to stent ingrowth, and, for this reason, are the first-line strategy in ill patients with short life expectancy (< 3 mo). The recently proposed EUS-GE has the ambition to provide a minimal invasive endoscopic procedure, with the consequent safe and rapid efficacy, and, at the same time, with the long-lasting advantages of GJ, as the metal stent is placed away from the neoplastic stricture and therefore is virtually free from ingrowth risk. Despite these exciting novelties, EUS-GE is still a difficult and not standardized technique, and is currently limited to centers with high experience in therapeutic EUS. In the next years, conducting well-designed prospective studies will be the intriguing challenge to identify the best therapeutic option to treat patients with MGOO.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Arigami T, Guo JT S-Editor: Zhang L L-Editor: A E-Editor: Ma YJ
| 1. | Brimhall B, Adler DG. Enteral stents for malignant gastric outlet obstruction. Gastrointest Endosc Clin N Am. 2011;21:389-403, vii-viii. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 2. | Tringali A, Didden P, Repici A, Spaander M, Bourke MJ, Williams SJ, Spicak J, Drastich P, Mutignani M, Perri V, Roy A, Johnston K, Costamagna G. Endoscopic treatment of malignant gastric and duodenal strictures: a prospective, multicenter study. Gastrointest Endosc. 2014;79:66-75. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 99] [Article Influence: 9.9] [Reference Citation Analysis (18)] |
| 3. | Costamagna G, Tringali A, Spicak J, Mutignani M, Shaw J, Roy A, Johnsson E, De Moura EG, Cheng S, Ponchon T, Bittinger M, Messmann H, Neuhaus H, Schumacher B, Laugier R, Saarnio J, Ariqueta FI. Treatment of malignant gastroduodenal obstruction with a nitinol self-expanding metal stent: an international prospective multicentre registry. Dig Liver Dis. 2012;44:37-43. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 4. | Khashab M, Alawad AS, Shin EJ, Kim K, Bourdel N, Singh VK, Lennon AM, Hutfless S, Sharaiha RZ, Amateau S, Okolo PI, Makary MA, Wolfgang C, Canto MI, Kalloo AN. Enteral stenting versus gastrojejunostomy for palliation of malignant gastric outlet obstruction. Surg Endosc. 2013;27:2068-2075. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 99] [Cited by in F6Publishing: 111] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 5. | Hori Y, Naitoh I, Ban T, Narita K, Nakazawa T, Hayashi K, Miyabe K, Shimizu S, Kondo H, Nishi Y, Yoshida M, Umemura S, Kato A, Yamada T, Ando T, Joh T. Stent under-expansion on the procedure day, a predictive factor for poor oral intake after metallic stenting for gastric outlet obstruction. J Gastroenterol Hepatol. 2015;30:1246-1251. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 6. | Lee KM, Choi SJ, Shin SJ, Hwang JC, Lim SG, Jung JY, Yoo BM, Cho SW, Kim JH. Palliative treatment of malignant gastroduodenal obstruction with metallic stent: prospective comparison of covered and uncovered stents. Scand J Gastroenterol. 2009;44:846-852. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Park CH, Park JC, Kim EH, Chung H, An JY, Kim HI, Shin SK, Lee SK, Cheong JH, Hyung WJ, Lee YC, Noh SH, Kim CB. Impact of carcinomatosis and ascites status on long-term outcomes of palliative treatment for patients with gastric outlet obstruction caused by unresectable gastric cancer: stent placement versus palliative gastrojejunostomy. Gastrointest Endosc. 2015;81:321-332. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 8. | Ly J, O'Grady G, Mittal A, Plank L, Windsor JA. A systematic review of methods to palliate malignant gastric outlet obstruction. Surg Endosc. 2010;24:290-297. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 113] [Cited by in F6Publishing: 112] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 9. | Wilson RG, Varma JS. Laparoscopic gastroenterostomy for malignant duodenal obstruction. Br J Surg. 1992;79:1348. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 31] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Watanapa P, Williamson RC. Surgical palliation for pancreatic cancer: developments during the past two decades. Br J Surg. 1992;79:8-20. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 240] [Cited by in F6Publishing: 259] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 11. | Maetani I, Inoue H, Sato M, Ohashi S, Igarashi Y, Sakai Y. Peroral insertion techniques of self-expanding metal stents for malignant gastric outlet and duodenal stenoses. Gastrointest Endosc. 1996;44:468-471. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 36] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Venu RP, Pastika BJ, Kini M, Chua D, Christian R, Schlais J, Brown RD. Self-expandable metal stents for malignant gastric outlet obstruction: a modified technique. Endoscopy. 1998;30:553-558. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 13. | Kozarek RA. Complications and lessons learned from 10 years of expandable gastrointestinal prostheses. Dig Dis. 1999;17:14-22. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 14. | Jung GS, Song HY, Seo TS, Park SJ, Koo JY, Huh JD, Cho YD. Malignant gastric outlet obstructions: treatment by means of coaxial placement of uncovered and covered expandable nitinol stents. J Vasc Interv Radiol. 2002;13:275-283. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 77] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 15. | Jung GS, Song HY, Kang SG, Huh JD, Park SJ, Koo JY, Cho YD. Malignant gastroduodenal obstructions: treatment by means of a covered expandable metallic stent-initial experience. Radiology. 2000;216:758-763. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 132] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 16. | Nevitt AW, Vida F, Kozarek RA, Traverso LW, Raltz SL. Expandable metallic prostheses for malignant obstructions of gastric outlet and proximal small bowel. Gastrointest Endosc. 1998;47:271-276. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 80] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 17. | Yates MR, Morgan DE, Baron TH. Palliation of malignant gastric and small intestinal strictures with self-expandable metal stents. Endoscopy. 1998;30:266-272. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 82] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 18. | Baron TH. Expandable metal stents for the treatment of cancerous obstruction of the gastrointestinal tract. N Engl J Med. 2001;344:1681-1687. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 304] [Cited by in F6Publishing: 323] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 19. | Adler DG, Baron TH. Endoscopic palliation of malignant gastric outlet obstruction using self-expanding metal stents: experience in 36 patients. Am J Gastroenterol. 2002;97:72-78. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 399] [Cited by in F6Publishing: 366] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 20. | Jeurnink SM, Repici A, Luigiano C, Pagano N, Kuipers EJ, Siersema PD. Use of a colonoscope for distal duodenal stent placement in patients with malignant obstruction. Surg Endosc. 2009;23:562-567. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Park JM, Min BH, Lee SH, Chung KH, Lee JM, Song BJ, Lee JK, Ryu JK, Kim YT. Feasibility of self-expandable metal stent placement with side-viewing endoscope for malignant distal duodenal obstruction. Dig Dis Sci. 2015;60:524-530. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | van Hooft JE, Uitdehaag MJ, Bruno MJ, Timmer R, Siersema PD, Dijkgraaf MG, Fockens P. Efficacy and safety of the new WallFlex enteral stent in palliative treatment of malignant gastric outlet obstruction (DUOFLEX study): a prospective multicenter study. Gastrointest Endosc. 2009;69:1059-1066. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 148] [Cited by in F6Publishing: 142] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 23. | van Hooft J, Mutignani M, Repici A, Messmann H, Neuhaus H, Fockens P. First data on the palliative treatment of patients with malignant gastric outlet obstruction using the WallFlex enteral stent: a retrospective multicenter study. Endoscopy. 2007;39:434-439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 42] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | van den Berg MW, Haijtink S, Fockens P, Vleggaar FP, Dijkgraaf MG, Siersema PD, van Hooft JE. First data on the Evolution duodenal stent for palliation of malignant gastric outlet obstruction (DUOLUTION study): a prospective multicenter study. Endoscopy. 2013;45:174-181. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | van Hooft JE, van Montfoort ML, Jeurnink SM, Bruno MJ, Dijkgraaf MG, Siersema PD, Fockens P. Safety and efficacy of a new non-foreshortening nitinol stent in malignant gastric outlet obstruction (DUONITI study): a prospective, multicenter study. Endoscopy. 2011;43:671-675. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 26. | ASGE Technology Committee, Varadarajulu S, Banerjee S, Barth B, Desilets D, Kaul V, Kethu S, Pedrosa M, Pfau P, Tokar J, Wang A, Song LM, Rodriguez S. Enteral stents. Gastrointest Endosc. 2011;74:455-464. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 78] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 27. | van Halsema EE, Rauws EA, Fockens P, van Hooft JE. Self-expandable metal stents for malignant gastric outlet obstruction: A pooled analysis of prospective literature. World J Gastroenterol. 2015;21:12468-12481. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 65] [Cited by in F6Publishing: 62] [Article Influence: 6.9] [Reference Citation Analysis (37)] |
| 28. | Larssen L, Medhus AW, Hauge T. Treatment of malignant gastric outlet obstruction with stents: an evaluation of the reported variables for clinical outcome. BMC Gastroenterol. 2009;9:45. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | Dormann A, Meisner S, Verin N, Wenk Lang A. Self-expanding metal stents for gastroduodenal malignancies: systematic review of their clinical effectiveness. Endoscopy. 2004;36:543-550. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 336] [Cited by in F6Publishing: 355] [Article Influence: 17.8] [Reference Citation Analysis (1)] |
| 30. | Jeurnink SM, van Eijck CH, Steyerberg EW, Kuipers EJ, Siersema PD. Stent versus gastrojejunostomy for the palliation of gastric outlet obstruction: a systematic review. BMC Gastroenterol. 2007;7:18. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 250] [Cited by in F6Publishing: 222] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 31. | Yamao K, Kitano M, Kayahara T, Ishida E, Yamamoto H, Minaga K, Yamashita Y, Nakajima J, Asada M, Okabe Y, Osaki Y, Chiba Y, Imai H, Kudo M. Factors predicting through-the-scope gastroduodenal stenting outcomes in patients with gastric outlet obstruction: a large multicenter retrospective study in West Japan. Gastrointest Endosc. 2016;84:757-763.e6. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 32. | Pais-Cunha I, Castro R, Libânio D, Pita I, Bastos RP, Silva R, Dinis-Ribeiro M, Pimentel-Nunes P. Endoscopic stenting for palliation of intra-abdominal gastrointestinal malignant obstruction: predictive factors for clinical success. Eur J Gastroenterol Hepatol. 2018;30:1033-1040. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 33. | Rademacher C, Bechtler M, Schneider S, Hartmann B, Striegel J, Jakobs R. Self-expanding metal stents for the palliation of malignant gastric outlet obstruction in patients with peritoneal carcinomatosis. World J Gastroenterol. 2016;22:9554-9561. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 13] [Cited by in F6Publishing: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 34. | Sasaki T, Isayama H, Nakai Y, Togawa O, Kogure H, Kawakubo K, Mizuno S, Yashima Y, Ito Y, Yamamoto N, Sasahira N, Hirano K, Tsujino T, Toda N, Tada M, Omata M, Koike K. Predictive factors of solid food intake in patients with malignant gastric outlet obstruction receiving self-expandable metallic stents for palliation. Dig Endosc. 2012;24:226-230. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 35. | Kim JH, Song HY, Shin JH, Choi E, Kim TW, Jung HY, Lee GH, Lee SK, Kim MH, Ryu MH, Kang YK, Kim BS, Yook JH. Metallic stent placement in the palliative treatment of malignant gastroduodenal obstructions: prospective evaluation of results and factors influencing outcome in 213 patients. Gastrointest Endosc. 2007;66:256-264. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 150] [Cited by in F6Publishing: 144] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 36. | Kim CG, Park SR, Choi IJ, Lee JY, Cho SJ, Park YI, Nam BH, Kim YW. Effect of chemotherapy on the outcome of self-expandable metallic stents in gastric cancer patients with malignant outlet obstruction. Endoscopy. 2012;44:807-812. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 37. | Jeon HH, Park CH, Park JC, Shim CN, Kim S, Lee HJ, Lee H, Shin SK, Lee SK, Lee YC. Carcinomatosis matters: clinical outcomes and prognostic factors for clinical success of stent placement in malignant gastric outlet obstruction. Surg Endosc. 2014;28:988-995. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 38. | Grunwald D, Cohen J, Bartley A, Sheridan J, Chuttani R, Sawhney MS, Pleskow DK, Berzin TM, Mizrahi M. The location of obstruction predicts stent occlusion in malignant gastric outlet obstruction. Therap Adv Gastroenterol. 2016;9:815-822. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 39. | Ratone JP, Caillol F, Zemmour C, Bories E, Pesenti C, Lestelle V, Godat S, Hoibian S, Proux A, Capodano G, Giovannini M. Outcomes of duodenal stenting: Experience in a French tertiary center with 220 cases. Dig Liver Dis. 2020;52:51-56. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 40. | Park JC, Park JJ, Cheoi K, Chung H, Lee H, Shin SK, Lee SK, Lee YC. Clinical outcomes of secondary stent-in-stent self-expanding metal stent placement for primary stent malfunction in malignant gastric outlet obstruction. Dig Liver Dis. 2012;44:999-1005. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 41. | Sasaki T, Isayama H, Nakai Y, Takahara N, Hamada T, Mizuno S, Mohri D, Yagioka H, Kogure H, Arizumi T, Togawa O, Matsubara S, Ito Y, Yamamoto N, Sasahira N, Hirano K, Toda N, Tada M, Koike K. Clinical outcomes of secondary gastroduodenal self-expandable metallic stent placement by stent-in-stent technique for malignant gastric outlet obstruction. Dig Endosc. 2015;27:37-43. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 31] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 42. | Pan YM, Pan J, Guo LK, Qiu M, Zhang JJ. Covered versus uncovered self-expandable metallic stents for palliation of malignant gastric outlet obstruction: a systematic review and meta-analysis. BMC Gastroenterol. 2014;14:170. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 43. | Hamada T, Hakuta R, Takahara N, Sasaki T, Nakai Y, Isayama H, Koike K. Covered versus uncovered metal stents for malignant gastric outlet obstruction: Systematic review and meta-analysis. Dig Endosc. 2017;29:259-271. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 44. | Hori Y, Hayashi K, Naitoh I, Kato H, Nomura T, Miyabe K, Yoshida M, Jinno N, Natsume M, Kato A, Asano G, Takiguchi S, Nakajima K. Feasibility and safety of duodenal covered self-expandable metallic stent fixation: an experimental study. Surg Endosc. 2019;33:4026-4031. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 45. | Lee H, Min BH, Lee JH, Shin CM, Kim Y, Chung H, Lee SH. Covered metallic stents with an anti-migration design vs. uncovered stents for the palliation of malignant gastric outlet obstruction: a multicenter, randomized trial. Am J Gastroenterol. 2015;110:1440-1449. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 46. | Kim ID, Kang DH, Choi CW, Kim HW, Jung WJ, Lee DH, Chung CW, Yoo JJ, Ryu JH. Prevention of covered enteral stent migration in patients with malignant gastric outlet obstruction: a pilot study of anchoring with endoscopic clips. Scand J Gastroenterol. 2010;45:100-105. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 47. | Maetani I, Ukita T, Tada T, Shigoka H, Omuta S, Endo T. Metallic stents for gastric outlet obstruction: reintervention rate is lower with uncovered versus covered stents, despite similar outcomes. Gastrointest Endosc. 2009;69:806-812. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 61] [Cited by in F6Publishing: 67] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 48. | Maetani I, Ukita T, Nambu T, Shigoka H, Omuta S, Endo T, Takahashi K. Comparison of ultraflex and niti-s stents for palliation of unresectable malignant gastroduodenal obstruction. Dig Endosc. 2010;22:83-89. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 49. | Shi-Yi L, Ai-Wu M, Yi-Ping J, Zhen-Lei W, Hao-Sheng J, Yong-Dong L, Xiang Y. Placement of duodenal stents across the duodenal papilla may predispose to acute pancreatitis: a retrospective analysis. Diagn Interv Radiol. 2012;18:360-364. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 50. | Mutignani M, Tringali A, Shah SG, Perri V, Familiari P, Iacopini F, Spada C, Costamagna G. Combined endoscopic stent insertion in malignant biliary and duodenal obstruction. Endoscopy. 2007;39:440-447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 139] [Cited by in F6Publishing: 131] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 51. | Kaw M, Singh S, Gagneja H. Clinical outcome of simultaneous self-expandable metal stents for palliation of malignant biliary and duodenal obstruction. Surg Endosc. 2003;17:457-461. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 82] [Cited by in F6Publishing: 92] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 52. | Staub J, Siddiqui A, Taylor LJ, Loren D, Kowalski T, Adler DG. ERCP performed through previously placed duodenal stents: a multicenter retrospective study of outcomes and adverse events. Gastrointest Endosc. 2018;87:1499-1504. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 26] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 53. | Anderloni A, Troncone E, Fugazza A, Cappello A, Blanco GDV, Monteleone G, Repici A. Lumen-apposing metal stents for malignant biliary obstruction: Is this the ultimate horizon of our experience? World J Gastroenterol. 2019;25:3857-3869. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 16] [Cited by in F6Publishing: 17] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 54. | Anderloni A, Buda A, Carrara S, Di Leo M, Fugazza A, Maselli R, Repici A. Single-session double-stent placement in concomitant malignant biliary and duodenal obstruction with a cautery-tipped lumen apposing metal stent. Endoscopy. 2016;48:E321-E322. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 55. | Anderloni A, Fugazza A, Auriemma F, Maia L, Maselli R, Troncone E, DʼAmico F, Repici A. Cautery-Tipped Lumen Apposing Metal Stent Placement Through the Mesh of an Indwelling Duodenal Self-Expanding Metal Stent. Am J Gastroenterol. 2018;113:644. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 56. | Yamao K, Kitano M, Takenaka M, Minaga K, Sakurai T, Watanabe T, Kayahara T, Yoshikawa T, Yamashita Y, Asada M, Okabe Y, Hanada K, Chiba Y, Kudo M. Outcomes of endoscopic biliary drainage in pancreatic cancer patients with an indwelling gastroduodenal stent: a multicenter cohort study in West Japan. Gastrointest Endosc. 2018;88:66-75.e2. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 57. | Jeurnink SM, Steyerberg EW, van Hooft JE, van Eijck CH, Schwartz MP, Vleggaar FP, Kuipers EJ, Siersema PD; Dutch SUSTENT Study Group. Surgical gastrojejunostomy or endoscopic stent placement for the palliation of malignant gastric outlet obstruction (SUSTENT study): a multicenter randomized trial. Gastrointest Endosc. 2010;71:490-499. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 358] [Cited by in F6Publishing: 338] [Article Influence: 24.1] [Reference Citation Analysis (2)] |
| 58. | Fiori E, Lamazza A, De Cesare A, Bononi M, Volpino P, Schillaci A, Cavallaro A, Cangemi V. Palliative management of malignant rectosigmoidal obstruction. Colostomy vs. endoscopic stenting. A randomized prospective trial. Anticancer Res. 2004;24:265-268. [PubMed] [Cited in This Article: ] |
| 59. | Mehta S, Hindmarsh A, Cheong E, Cockburn J, Saada J, Tighe R, Lewis MP, Rhodes M. Prospective randomized trial of laparoscopic gastrojejunostomy versus duodenal stenting for malignant gastric outflow obstruction. Surg Endosc. 2006;20:239-242. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 163] [Cited by in F6Publishing: 177] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 60. | Jang S, Stevens T, Lopez R, Bhatt A, Vargo JJ. Superiority of Gastrojejunostomy Over Endoscopic Stenting for Palliation of Malignant Gastric Outlet Obstruction. Clin Gastroenterol Hepatol. 2019;17:1295-1302.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 61. | Mintziras I, Miligkos M, Wächter S, Manoharan J, Bartsch DK. Palliative surgical bypass is superior to palliative endoscopic stenting in patients with malignant gastric outlet obstruction: systematic review and meta-analysis. Surg Endosc. 2019;33:3153-3164. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 62. | Lorusso D, Giliberti A, Bianco M, Lantone G, Leandro G. Stomach-partitioning gastrojejunostomy is better than conventional gastrojejunostomy in palliative care of gastric outlet obstruction for gastric or pancreatic cancer: a meta-analysis. J Gastrointest Oncol. 2019;10:283-291. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 63. | Arigami T, Uenosono Y, Ishigami S, Yanagita S, Okubo K, Uchikado Y, Kita Y, Mori S, Kurahara H, Maemura K, Natsugoe S. Clinical Impact of Stomach-partitioning Gastrojejunostomy with Braun Enteroenterostomy for Patients with Gastric Outlet Obstruction Caused by Unresectable Gastric Cancer. Anticancer Res. 2016;36:5431-5436. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 64. | Roy A, Kim M, Christein J, Varadarajulu S. Stenting versus gastrojejunostomy for management of malignant gastric outlet obstruction: comparison of clinical outcomes and costs. Surg Endosc. 2012;26:3114-3119. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 65. | Jeurnink SM, Polinder S, Steyerberg EW, Kuipers EJ, Siersema PD. Cost comparison of gastrojejunostomy versus duodenal stent placement for malignant gastric outlet obstruction. J Gastroenterol. 2010;45:537-543. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 65] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 66. | Dollhopf M, Larghi A, Will U, Rimbaş M, Anderloni A, Sanchez-Yague A, Teoh AYB, Kunda R. EUS-guided gallbladder drainage in patients with acute cholecystitis and high surgical risk using an electrocautery-enhanced lumen-apposing metal stent device. Gastrointest Endosc. 2017;86:636-643. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 101] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 67. | Anderloni A, Fugazza A, Troncone E, Auriemma F, Carrara S, Semeraro R, Maselli R, Di Leo M, D'Amico F, Sethi A, Repici A. Single-stage EUS-guided choledochoduodenostomy using a lumen-apposing metal stent for malignant distal biliary obstruction. Gastrointest Endosc. 2019;89:69-76. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 95] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 68. | Mussetto A, Fugazza A, Fuccio L, Triossi O, Repici A, Anderloni A. Current uses and outcomes of lumen-apposing metal stents. Ann Gastroenterol. 2018;31:535-540. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 69. | Siddiqui UD, Levy MJ. EUS-Guided Transluminal Interventions. Gastroenterology. 2018;154:1911-1924. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 70. | Khashab MA, Kumbhari V, Grimm IS, Ngamruengphong S, Aguila G, El Zein M, Kalloo AN, Baron TH. EUS-guided gastroenterostomy: the first U.S. clinical experience (with video). Gastrointest Endosc. 2015;82:932-938. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 130] [Cited by in F6Publishing: 128] [Article Influence: 14.2] [Reference Citation Analysis (3)] |
| 71. | Khashab MA, Baron TH, Binmoeller KF, Itoi T. EUS-guided gastroenterostomy: a new promising technique in evolution. Gastrointest Endosc. 2015;81:1234-1236. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 37] [Article Influence: 4.1] [Reference Citation Analysis (35)] |
| 72. | Binmoeller KF, Shah JN. Endoscopic ultrasound-guided gastroenterostomy using novel tools designed for transluminal therapy: a porcine study. Endoscopy. 2012;44:499-503. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 100] [Cited by in F6Publishing: 116] [Article Influence: 9.7] [Reference Citation Analysis (16)] |
| 73. | Itoi T, Baron TH, Khashab MA, Tsuchiya T, Irani S, Dhir V, Bun Teoh AY. Technical review of endoscopic ultrasonography-guided gastroenterostomy in 2017. Dig Endosc. 2017;29:495-502. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 78] [Article Influence: 11.1] [Reference Citation Analysis (35)] |
| 74. | Tyberg A, Perez-Miranda M, Sanchez-Ocaña R, Peñas I, de la Serna C, Shah J, Binmoeller K, Gaidhane M, Grimm I, Baron T, Kahaleh M. Endoscopic ultrasound-guided gastrojejunostomy with a lumen-apposing metal stent: a multicenter, international experience. Endosc Int Open. 2016;4:E276-E281. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 174] [Cited by in F6Publishing: 155] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 75. | Itoi T, Tsuchiya T, Tonozuka R, Ijima M, Kusano C. Novel EUS-guided double-balloon-occluded gastrojejunostomy bypass. Gastrointest Endosc. 2016;83:461-462. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 76. | Walter D, Will U, Sanchez-Yague A, Brenke D, Hampe J, Wollny H, López-Jamar JM, Jechart G, Vilmann P, Gornals JB, Ullrich S, Fähndrich M, de Tejada AH, Junquera F, Gonzalez-Huix F, Siersema PD, Vleggaar FP. A novel lumen-apposing metal stent for endoscopic ultrasound-guided drainage of pancreatic fluid collections: a prospective cohort study. Endoscopy. 2015;47:63-67. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 75] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 77. | Walter D, Teoh AY, Itoi T, Pérez-Miranda M, Larghi A, Sanchez-Yague A, Siersema PD, Vleggaar FP. EUS-guided gall bladder drainage with a lumen-apposing metal stent: a prospective long-term evaluation. Gut. 2016;65:6-8. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 129] [Cited by in F6Publishing: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 78. | Anderloni A, Leo MD, Carrara S, Fugazza A, Maselli R, Buda A, Amato A, Auriemma F, Repici A. Endoscopic ultrasound-guided transmural drainage by cautery-tipped lumen-apposing metal stent: exploring the possible indications. Ann Gastroenterol. 2018;31:735-741. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 79. | Itoi T, Itokawa F, Uraoka T, Gotoda T, Horii J, Goto O, Moriyasu F, Moon JH, Kitagawa Y, Yahagi N. Novel EUS-guided gastrojejunostomy technique using a new double-balloon enteric tube and lumen-apposing metal stent (with videos). Gastrointest Endosc. 2013;78:934-939. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 80. | Chen YI, Itoi T, Baron TH, Nieto J, Haito-Chavez Y, Grimm IS, Ismail A, Ngamruengphong S, Bukhari M, Hajiyeva G, Alawad AS, Kumbhari V, Khashab MA. EUS-guided gastroenterostomy is comparable to enteral stenting with fewer re-interventions in malignant gastric outlet obstruction. Surg Endosc. 2017;31:2946-2952. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 117] [Cited by in F6Publishing: 138] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 81. | Carbajo AY, Kahaleh M, Tyberg A. Clinical Review of EUS-guided Gastroenterostomy (EUS-GE). J Clin Gastroenterol. 2020;54:1-7. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 82. | Itoi T, Ishii K, Ikeuchi N, Sofuni A, Gotoda T, Moriyasu F, Dhir V, Teoh AY, Binmoeller KF. Prospective evaluation of endoscopic ultrasonography-guided double-balloon-occluded gastrojejunostomy bypass (EPASS) for malignant gastric outlet obstruction. Gut. 2016;65:193-195. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 137] [Cited by in F6Publishing: 148] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 83. | Khashab MA, Bukhari M, Baron TH, Nieto J, El Zein M, Chen YI, Chavez YH, Ngamruengphong S, Alawad AS, Kumbhari V, Itoi T. International multicenter comparative trial of endoscopic ultrasonography-guided gastroenterostomy versus surgical gastrojejunostomy for the treatment of malignant gastric outlet obstruction. Endosc Int Open. 2017;5:E275-E281. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 148] [Article Influence: 21.1] [Reference Citation Analysis (1)] |
| 84. | Perez-Miranda M, Tyberg A, Poletto D, Toscano E, Gaidhane M, Desai AP, Kumta NA, Fayad L, Nieto J, Barthet M, Shah R, Brauer BC, Sharaiha RZ, Kahaleh M. EUS-guided Gastrojejunostomy Versus Laparoscopic Gastrojejunostomy: An International Collaborative Study. J Clin Gastroenterol. 2017;51:896-899. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 146] [Article Influence: 20.9] [Reference Citation Analysis (1)] |
| 85. | Ge PS, Young JY, Dong W, Thompson CC. EUS-guided gastroenterostomy versus enteral stent placement for palliation of malignant gastric outlet obstruction. Surg Endosc. 2019;33:3404-3411. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 89] [Cited by in F6Publishing: 133] [Article Influence: 26.6] [Reference Citation Analysis (1)] |
| 86. | Chen YI, Kunda R, Storm AC, Aridi HD, Thompson CC, Nieto J, James T, Irani S, Bukhari M, Gutierrez OB, Agarwal A, Fayad L, Moran R, Alammar N, Sanaei O, Canto MI, Singh VK, Baron TH, Khashab MA. EUS-guided gastroenterostomy: a multicenter study comparing the direct and balloon-assisted techniques. Gastrointest Endosc. 2018;87:1215-1221. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 94] [Article Influence: 15.7] [Reference Citation Analysis (0)] |