Published online Mar 7, 2019. doi: 10.3748/wjg.v25.i9.1088
Peer-review started: November 16, 2018
First decision: January 6, 2019
Revised: January 24, 2019
Accepted: January 28, 2019
Article in press: January 28, 2019
Published online: March 7, 2019
Processing time: 111 Days and 2.4 Hours
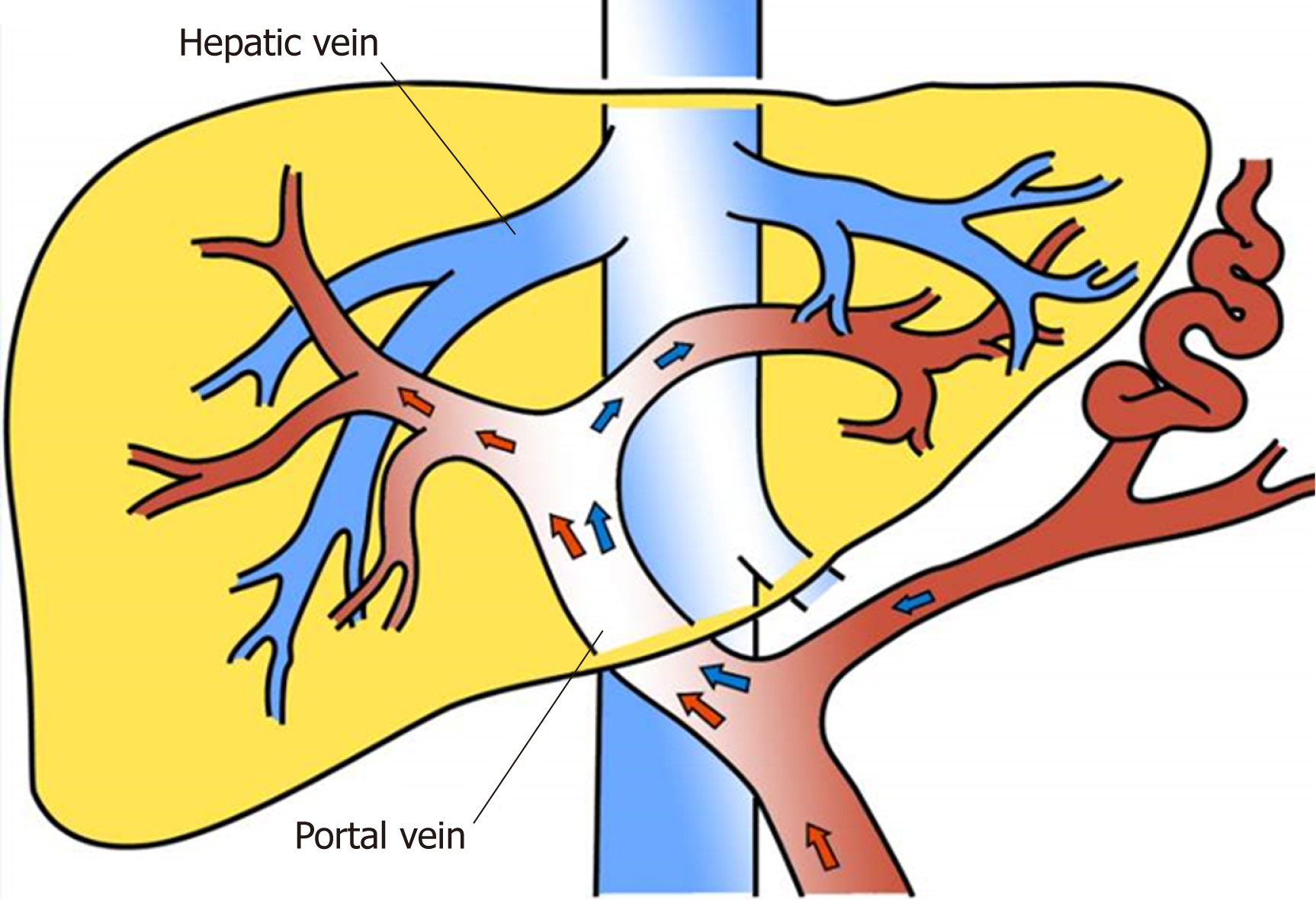
Transjugular intrahepatic portosystemic shunt (TIPS) is currently used for the treatment of complications of portal hypertension. The incidence of hepatic encephalopathy (HE) remains a problem in TIPS placement. It has been reported that the right branch mainly receives superior mesenteric venous blood while the left branch mainly receives blood from the splenic vein. We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt; therefore, targeted puncture of the left branch of the intrahepatic portal vein during TIPS may reduce the risk of HE.
To evaluate the influence of targeted puncture of left branch of portal vein in TIPS on HE.
A retrospective analysis of 1244 patients with portal-hypertension-related complications of refractory ascites or variceal bleeding who underwent TIPS from January 2000 to January 2013 was performed. Patients were divided into group A (targeting left branch of portal vein, n = 937) and group B (targeting right branch of portal vein, n = 307). TIPS-related HE and clinical outcomes were analyzed.
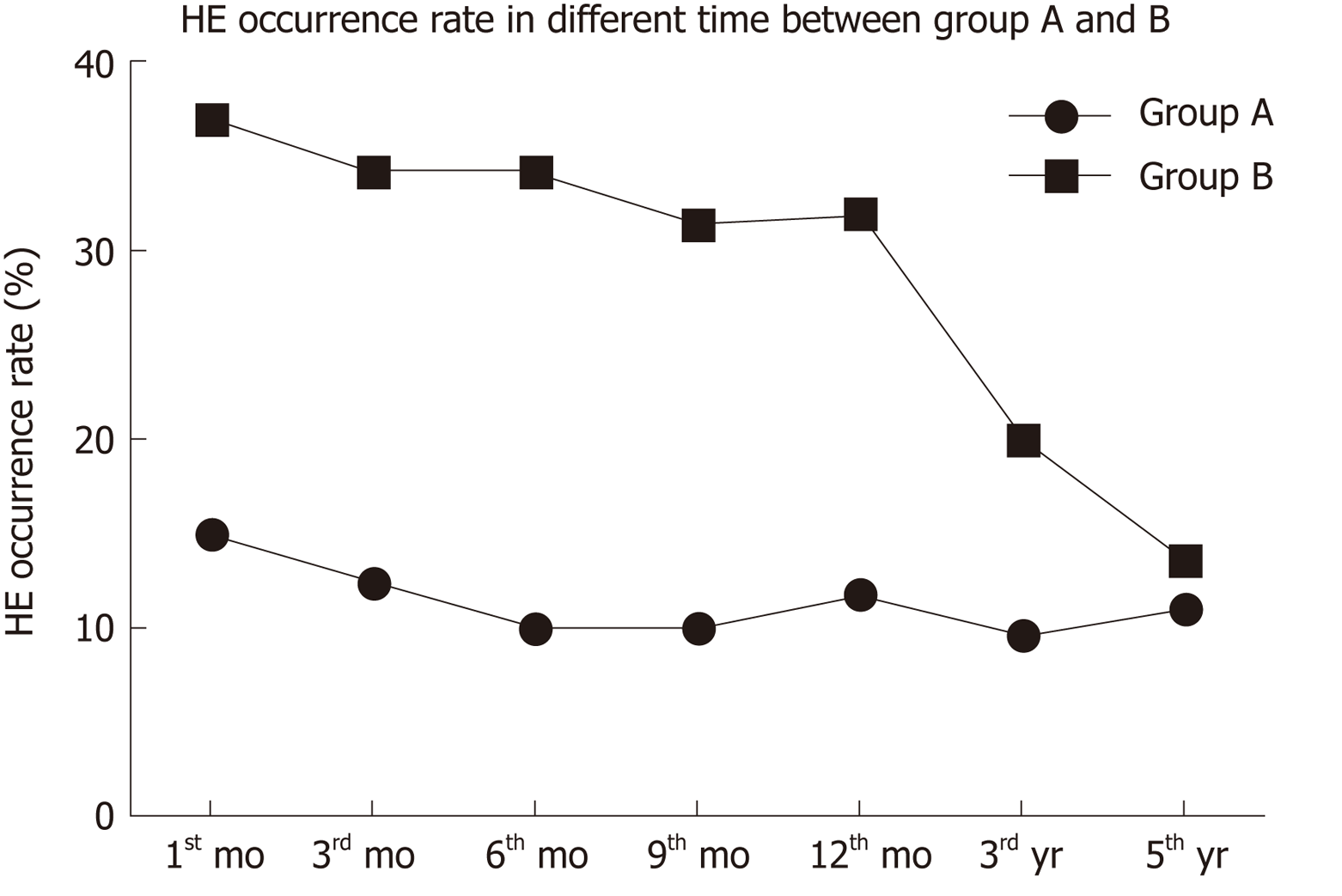
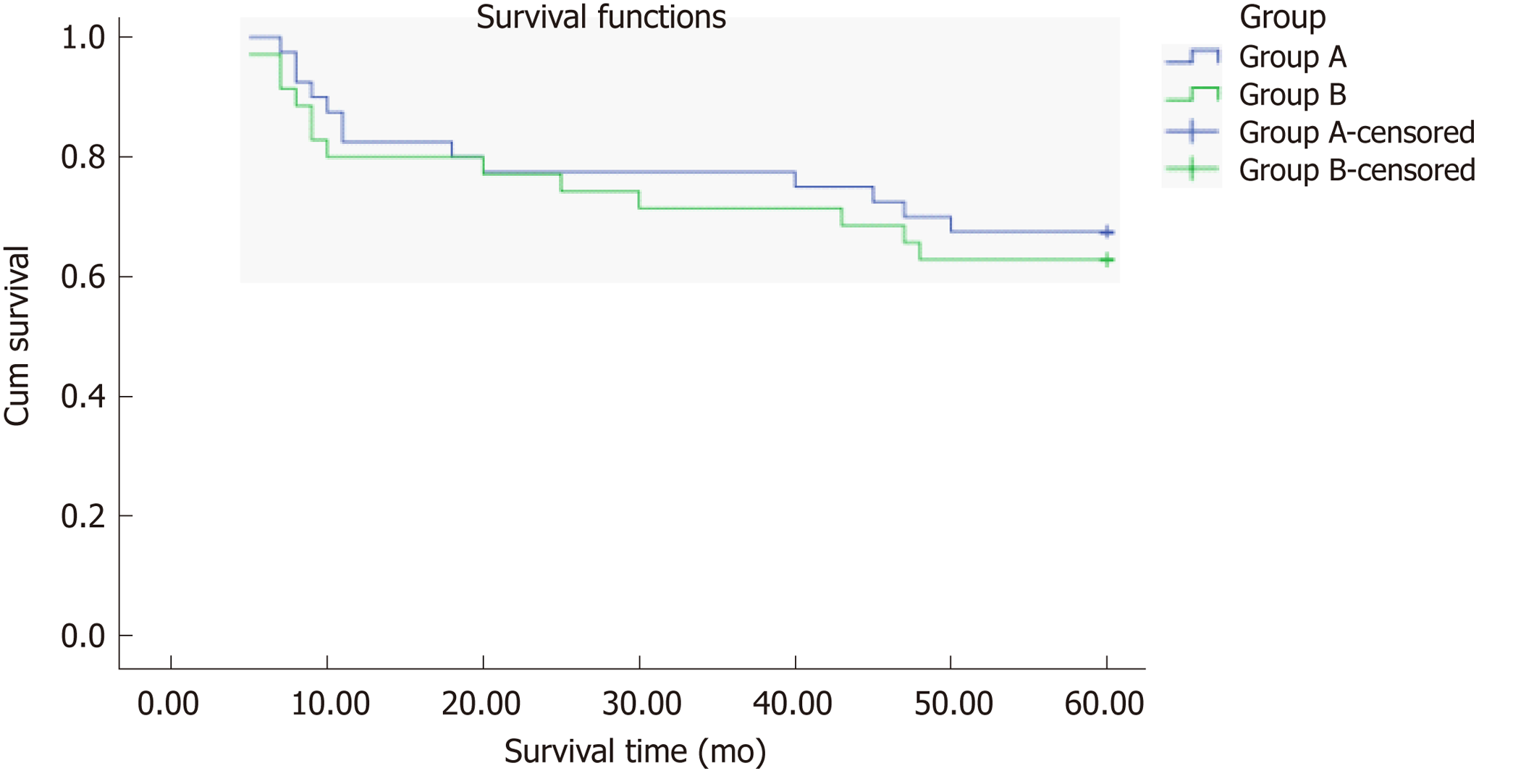
The symptoms of ascites and variceal bleeding disappeared within a short time. By the endpoint of follow-up, recurrent bleeding and ascites did not differ significantly between groups A and B (P = 0.278, P = 0.561, respectively). Incidence of HE differed significantly between groups A and B at 1 mo (14.94% vs 36.80%, χ2 = 4.839, P = 0.028), 3 mo (12.48% vs 34.20%, χ2 = 5.054, P = 0.025), 6 mo (10.03% vs 32.24%, χ2 = 6.560, P = 0.010), 9 mo (9.17% vs 31.27%, χ2 = 5.357, P = 0.021), and 12 mo (8.21% vs 28.01, χ2 = 3.848, P = 0.051). There were no significant differences between groups A and B at 3 years (6.61% vs 7.16%, χ2 = 1.204, P = 0.272) and 5 years (5.01% vs 6.18%, χ2 = 0.072, P = 0.562). The total survival rate did not differ between groups A and B (χ2 = 0.226, P = 0.634, log-rank test).
Targeted puncture of the left branch of the intrahepatic portal vein during TIPS may reduce the risk of HE but has no direct influence on prognosis of portal-hypertension-related complications.
Core tip: We studied a large cohort of patients with cirrhosis who underwent transjugular intrahepatic portosystemic shunt for recurrent variceal bleeding and/or ascites. Targeted puncture of the left branch of the intrahepatic portal vein during transjugular intrahepatic portosystemic shunt did not directly influence the prognosis of portal-hypertension-related complications of refractory ascites or variceal bleeding but may reduce the risk of hepatic encephalopathy.
- Citation: Luo SH, Chu JG, Huang H, Zhao GR, Yao KC. Targeted puncture of left branch of intrahepatic portal vein in transjugular intrahepatic portosystemic shunt to reduce hepatic encephalopathy. World J Gastroenterol 2019; 25(9): 1088-1099
- URL: https://www.wjgnet.com/1007-9327/full/v25/i9/1088.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i9.1088
Transjugular intrahepatic portosystemic shunt (TIPS) is currently used for the treatment of complications of portal hypertension[1]. The establishment of TIPS is widely accepted as an alternative to surgery in the management of complications from portal hypertension such as variceal bleeding, refractory ascites, Budd-Chiari syndrome, hepatorenal syndrome, hepatic hydrothorax, and even hepatopulmonary syndrome[2]. After TIPS was introduced as an alternative treatment for complications related to portal hypertension, it was progressively recognized as an effective therapeutic option in a growing number of clinical situations[3,4].
With the advances in materials, many experimental and clinical studies[5] have been conducted using covered stent grafts, especially stent grafts covered with polytetrafluoroethylene, to improve the long-term patency of TIPS[6]. The incidence of hepatic encephalopathy (HE) remains a problem in TIPS placement and affects the quality of life and long-term outcomes of patients.
It has been reported[7] that the reflux blood from the splenic and superior mesenteric veins is distributed hydrodynamically in the main portal vein, that is, alongside the trunk on both sides of the wall of the portal vein. However, it is not fully mixed and enters the left and right branches of the portal vein. The right branch mainly receives superior mesenteric venous blood while the left branch mainly receives blood from the splenic vein[8].
We hypothesized (Figure 1) that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE. The purpose of this study was to evaluate the influence of targeted puncture of the left or right branches of the portal vein on the incidence of HE in patients who required TIPS placement for portal-hypertension-related complications of ascites or variceal bleeding.
Between January 2000 and January 2013, 1244 patients were referred to us on an intention-to-treat basis and underwent a TIPS procedure. Indications for stent graft shunt were variceal hemorrhage, refractory ascites, or both. The outcomes of recurrent variceal bleeding and/or ascites, mortality, and HE were compared between two groups. The Institutional Review Board approved the study protocol. The patients’ medical records and images were reviewed to gather information regarding the underlying etiology, clinical presentation, age, sex, and severity of cirrhosis (Table 1).
| Characteristics | Group A | Group B | P value |
| Gender, M/F | 398/539 | 166/141 | 0.308 |
| Age in yr, mean ± SD | 44.48 ± 18.23 | 40.80 ± 17.76 | 0.381 |
| Child-Pugh A/B/C | 79/729/137 | 25/160/122 | 0.753 |
| MELD score, mean ± SD | 13.19 ± 7.35 | 12.26 ± 8.47 | 0.591 |
| Viral hepatitis | 585 | 194 | 0.947 |
| Chronic ethanol consumption | 210 | 61 | 0.651 |
| Autoimmune hepatitis | 53 | 23 | 0.463 |
| Metabolic liver disease | 89 | 29 | 0.527 |
| Variceal hemorrhage | 653 | 236 | 0.163 |
| Refractory ascites | 384 | 117 | 0.125 |
| Laboratory tests | |||
| Alanine transaminase in U/L | 69.13 ± 12.21 | 56.24 ± 11.17 | 0.615 |
| Aspartate transaminase in U/L | 56.14 ± 14.19 | 49.53 ± 12.24 | 0.653 |
| Alkaline phosphatase in U/L | 129.63 ± 26.32 | 136.46 ± 27.02 | 0.534 |
| γ-glutamyl transpeptidase in U/L | 236.23 ± 62.46 | 248.13 ± 53.32 | 0.561 |
| Total bilirubin in μmol/L | 24.32 ± 5.63 | 26.46 ± 6.37 | 0.482 |
| Albumin in g/L | 29.14 ± 7.22 | 31.23 ± 6.52 | 0.237 |
| Prothrombin time in s | 16.03 ± 7.31 | 18.46 ± 5.17 | 0.316 |
| Clinical presentations | |||
| Abdominal distention | 627 | 193 | 0.164 |
| Abdominal pain | 48 | 17 | 0.237 |
| Weakness | 473 | 136 | 0.253 |
| Poor appetite | 729 | 275 | 0.428 |
| Jaundice | 15 | 6 | 0.107 |
| Splenomegaly | 482 | 174 | 0.316 |
| Lower limbs edema | 136 | 57 | 0.263 |
| Endoscopic therapy | 1736 | 625 | 0.421 |
| Ascites paracentesis | 1492 | 479 | 0.304 |
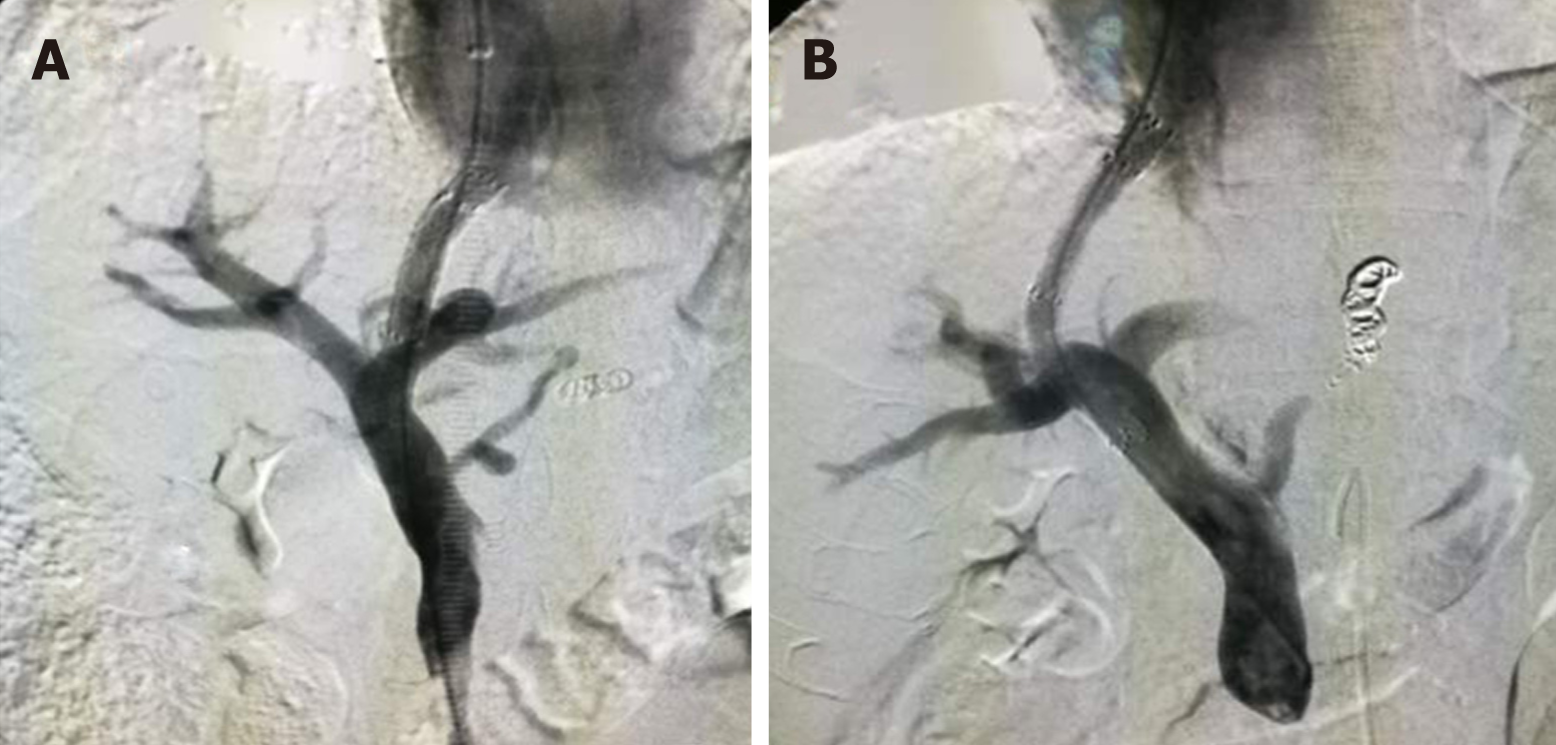
This study was a single-center and retrospective study that compared the influence of targeted puncture of the left and right branches of the portal vein on the incidence of HE in patients who required TIPS placement for portal-hypertension-related complications of ascites or variceal bleeding. The patients were divided into two groups: A (targeting of left branch of portal vein, n = 937) and B (targeting of right branch of portal vein, n = 307) (Figure 2). The outcomes of HE, recurrent variceal bleeding and/or ascites, and mortality were compared and analyzed between the groups. The inclusion criteria were: Recurrent variceal bleeding after a session of variceal sclerotherapy, refractory ascites, or both that required TIPS placement with portal-hypertension-related complications. The exclusion criteria were: Variceal bleeding as an emergency indication, portal vein thrombosis, history of HE, severe right-sided heart failure, severe liver failure (bilirubin > 4 mg/dL), polycystic liver disease, dilated biliary ducts, age > 75 years, Child-Pugh score > 11, Model of End-Stage Liver Disease score > 18, hepatic carcinoma, sepsis, spontaneous bacterial peritonitis, and patients who underwent liver transplantation.
TIPS was performed under standard local anesthesia as described previously[9]. The entire length of the intrahepatic tract was covered by the stent graft (BARD, Fluency, Voisins le Bretonneux, France; or Viatorr, W.L. Gore & Associates, Flagstaff, AZ, United States). Hepatic venous pressure gradient and portal vein pressure were measured during the procedure, and the shunts were dilated to their full nominal diameter to reach a target portosystemic gradient (PSG) of < 12 mmHg. Obvious gastroesophageal collateral vessels observed during the TIPS procedure were embolized with coils (Cook Inc., Bloomington, IL, United States; or Interlock Coil, Boston Scientific Corporation, Natikeshi, MA, United States). Subsequent direct portography was performed to evaluate whether the portal venous system was completely patent. After the TIPS procedure, intravenous heparin (4000 U/d; Chase Sun Pharma Co. Ltd., Tianjin, China) was administered for 3 d and oral warfarin was given at 2.5 mg/d (Orion Pharma Co. Ltd., Orionintie, Finland) to achieve an international normalized ratio ≤ 2.0, if prolonged international normalized ratio, oral warfarin was not given.
After TIPS deployment, baseline duplex sonography was performed on the day. Shunt velocities were compared with this baseline result during follow-up. Patients were placed into a routine follow-up protocol identical for each group. They were seen as outpatients 1 mo after the procedure and then 3, 6 and 9 mo and 1, 3 and 5 years, or whenever needed. Each consultation included a clinical examination, blood chemistry, upper abdominal ultrasonography, and assessment of HE. TIPS angiography was performed in patients with recurrent symptoms or suspected shunt dysfunction. TIPS revision was performed when a hemodynamically significant shunt stenosis (> 50%) was present with recurrent variceal bleeding, recurrent or gradually worsening ascites, and PSG ≥ 15 mmHg unless grade III/IV encephalopathy was present (Practice Guideline of the European Association for the Study of the Liver and the American Association for the Study of Liver Diseases). Patients lost to follow-up were censored at the time of the last known imaging of the shunt (duplex ultrasonography or shunt venography).
Results were expressed as mean ± standard deviation and compared using the independent sample t test or one-way analysis of variance, and categorical variables were expressed as frequencies and compared using χ2 tests. The differences between the groups were compared using one-way analysis of variance followed by least significant difference t tests. Differences were considered significant at P < 0.05. The statistical analyses were performed with SPSS version 20.0 (SPSS, Armonk, NY, United States).
None of the 1244 patients died within 30 d after TIPS, with an early survival rate of 100%. Both TIPS procedures demonstrated similar efficacy in decreasing PSG before and after TIPS placement from 27.08 ± 5.47 to 10.75 ± 3.67 mmHg in group A (P = 0.003) and from 26.42 ± 3.53 to 10.96 ± 2.41 mmHg in group B (P = 0.001) (Table 2).
| Groups | PSG in mmHg | t value | P value | |
| Before | After | |||
| Group A | 27.08 ± 5.47 | 10.75 ± 3.67 | 13.79 | 0.001 |
| Group B | 26.42 ± 3.53 | 10.96 ± 2.41 | 18.64 | 0.003 |
| (T value) 0.622 | 0.284 | |||
| (P value) 0.536 | 0.777 | |||
HE occurrence in group A was lower than in group B at 1, 3, 6, 9, and 12 mo, and showed a downward trend (Figure 3). At 3 and 5 years, there was no significant difference in HE occurrence between the two groups. After drug treatment, the symptoms disappeared in patients with covert and grade II HE. In patients with grade III or IV HE, the symptoms disappeared after shunt reduction, but three patients who underwent shunt reduction still had hepatic myelopathy (Table 3).
| Time | Group | HE occurrence | HE occurrence rate, % | Covert | HE grade | χ2 | P value | |||
| Yes | No | II | III | IV | ||||||
| 1 mo | A | 140 | 797 | 14.94 | 44 | 78 | 10 | 8 | 4.839 | 0.028 |
| B | 113 | 194 | 36.80 | 27 | 56 | 18 | 12 | |||
| 3 mo | A | 117 | 820 | 12.48 | 34 | 59 | 17 | 7 | 5.054 | 0.025 |
| B | 105 | 202 | 34.20 | 26 | 58 | 11 | 10 | |||
| 6 mo | A | 94 | 843 | 10.03 | 53 | 32 | 6 | 3 | 6.560 | 0.010 |
| B | 99 | 208 | 32.24 | 36 | 46 | 9 | 8 | |||
| 9 mo | A | 86 | 851 | 9.17 | 42 | 31 | 7 | 6 | 5.357 | 0.021 |
| B | 96 | 211 | 31.27 | 46 | 37 | 9 | 4 | |||
| 12 mo | A | 77 | 860 | 8.21 | 34 | 35 | 6 | 3 | 3.848 | 0.051 |
| B | 86 | 221 | 28.01 | 28 | 39 | 11 | 8 | |||
| 3 yr | A | 62 | 875 | 6.61 | 25 | 31 | 6 | 0 | 1.204 | 0.272 |
| B | 22 | 285 | 7.16 | 12 | 6 | 4 | 0 | |||
| 5 yr | A | 47 | 890 | 5.01 | 23 | 24 | 0 | 0 | 0.072 | 0.562 |
| B | 19 | 288 | 6.18 | 12 | 5 | 2 | 0 | |||
At 1 mo after TIPS placement, in group A, 140 patients manifested HE; among them, 44 cases were covert, 78 were grade II, 10 were grade III, and eight were grade IV. In group B, 113 patients manifested HE; among them, 27 cases were covert, 56 were grade II, 18 were grade III, and 12 were grade IV. The incidence of HE in group A was lower than in group B (14.94% vs 36.80%, χ2 = 4.839, P = 0.028). The symptoms in the 48 patients with grade III and IV HE disappeared after shunt reduction in both groups.
At 3 mo after TIPS placement, in group A, 117 patients manifested HE; among them, 34 cases were covert, 59 were grade II, 17 were grade III, and seven were grade IV. In group B, 105 patients manifested HE; among them, 26 cases were covert, 58 were grade II, 11 were grade III, and 10 were grade IV. The incidence of HE in group A was lower than that in group B (12.48% vs 34.20%, χ2 = 5.054, P = 0.025). The symptoms in the 42 patients with grade III and IV HE disappeared after shunt reduction in both groups.
At 6 mo after TIPS placement, in group A, 94 patients manifested HE; among them, 53 cases were covert, 32 were grade II, six were grade III, and three were grade IV. In group B, 99 patients manifested HE; among them, 36 cases were covert, 46 were grade II, nine were grade III, and eight were grade IV. The incidence of HE in group A was lower than that in group B (10.03% vs 32.24%, χ2 = 6.560, P = 0.010). The symptoms in the 26 patients with grade III and IV HE disappeared after shunt reduction.
At 9 mo after TIPS placement, in group A, 86 patients manifested HE; among them, 42 cases were covert, 31 were grade II, seven were grade III, and six were grade IV. In group B, 96 patients manifested HE; among them, 46 cases were covert, 37 were grade II, nine were grade III, and four were grade IV. The incidence of HE in group A was lower than that in group B (9.17% vs 31.27%, χ2 = 5.357, P = 0.021). The symptoms in the 26 patients with grade III and IV HE disappeared after shunt reduction.
At 12 mo after TIPS placement, in group A, 77 patients manifested HE; among them, 34 cases were covert, 35 were grade II, six were grade III, and three were grade IV. In group B, 86 patients manifested HE; among them, 28 cases were covert, 39 were grade II, 11 were grade III, and eight were grade IV. The incidence of HE in group A was lower than that in group B (8.21% vs 28.01%, χ2 = 3.848, P = 0.051). The symptoms in the 25 patients with grade III and IV HE disappeared after shunt reduction, although three patients who underwent shunt reduction still had hepatic myelopathy.
At 3 years after TIPS placement, in group A, 62 patients manifested HE; among them, 25 cases were covert, 31 were grade II, six were grade III, and none were grade IV. In group B, 22 patients manifested HE; among them, 12 cases were covert, six were grade II, four were grade III, and none were IV. There was no significant difference in the incidence of HE between group A and group B (6.61% vs 7.16%, χ2 = 1.204, P = 0.272).
At 5 years after TIPS placement, in group A, 47 patients manifested HE; among them, 23 cases were covert, 24 were grade II, and none were grade III or IV. In group B, 19 patients manifested HE; among them, 12 cases were covert, five were grade II, and two were grade III. There was no significant difference in the incidence of HE between group A and group B (5.01% vs 6.18%, χ2 = 0.072, P = 0.562).
The symptom of ascites in 357 cases in group A and 119 cases in group B disappeared within the first week without paracentesis, and there was no significant difference between the groups (P = 0.364). No patient experienced re-bleeding within a week. By the endpoint of follow-up, 112 cases had recurrent bleeding in group A compared with 49 cases in group B, and there was no significant difference between the groups (P = 0.278). There were 185 patients with recurrent ascites in group A and 64 patients in group B, and there was no significant difference between the groups (P = 0.561). After stent revision, the symptoms disappeared (Table 4).
| Symptoms | Group A | Group B | P value |
| Absorption of ascites within the first week | 357 | 11 | 0.364 |
| Recurrence of bleeding | 112 | 49 | 0.278 |
| Recurrence of ascites | 185 | 64 | 0.561 |
During follow-up, at 1 year, 141/937 patients (84.95%) were lost to follow-up in group A and 62/307 patients (79.80%) in group B. At 3 years, 211/937 patients (77.48%) were lost to follow-up in group A and 88/307 patients (71.33%) in group B. The endpoint of this study was at 5 years, 305/937 patients (67.44%) were lost to follow-up in group A and 114/307 patients (62.86%) in group B. The 1-, 3-, and 5-year survival rates did not differ between groups A and B (χ2 = 0.326, P = 0.568; χ2 = 0.364, P = 0.564 and χ2 = 0.178, P = 0.673, respectively), and the total survival rates did not differ between groups A and B (χ2 = 0.226, P = 0.634, log-rank test) (Figure 4). Among them, 221 patients died from hepatic tumor, 151 from multiorgan failure, and 47 from other causes (Table 5).
| Time | Group | Survival | Survival rate, % | χ2 | P value | |
| Yes | No | |||||
| 1 yr | A | 796 | 141 | 84.95 | 0.326 | 0.568 |
| B | 245 | 62 | 79.80 | |||
| 3 yr | A | 726 | 211 | 77.48 | 0.364 | 0.564 |
| B | 219 | 88 | 71.33 | |||
| 5 yr | A | 632 | 305 | 67.44 | 0.178 | 0.673 |
| B | 193 | 114 | 62.86 | |||
The use of TIPS in the treatment of portal-hypertension-related complications has progressively increased and has achieved good results[9,10]. However, the clinical benefit of this intervention has been hampered due to a high rate of HE, up to 20%-40% at 12 mo follow-up[3]. HE has become an important issue to be taken into consideration in TIPS candidates and a problem to be addressed after the procedure, which influence its widespread use in clinical practice.
Numerous studies[11,12] have evaluated the risk factors associated with post-TIPS HE in patients with portal hypertension due to cirrhosis, such as the selection of candidates for TIPS placement, the patient’s age, and liver function, as measured by Child-Pugh or Model of End-Stage Liver Disease scores. However, few studies focus on whether targeted puncture of right or left intrahepatic branch of portal vein in TIPS may reduce the incidence of post-TIPS HE[13].
HE is mainly due to absorption of toxic substances from the intestinal portal vein system, through the shunt without the liver first pass effect into the systemic circulation, caused by dysfunction of the central nervous system; a syndrome with mental and nervous symptoms[14]. One of the toxic substances that causes HE is blood ammonia[15]. There are two aspects of the source of blood ammonia in the body, endogenous ammonia is produced by catabolism in vivo, and exogenous ammonia is produced by catabolism of nitrogenous substances in the intestine[16]. In the latter, 90% is found in blood urea and is diffused into the intestinal cavity through the gastrointestinal mucosal blood vessels and decomposed by bacterial urease[17].
The superior mesenteric vein and splenic vein are composed of two branches of main portal vein; the former mainly collects blood reflux in the small intestine, colon, and pancreatic head; the latter mainly collects blood from the spleen, pancreatic body and tail; and other parts or inferior mesenteric vein collect blood reflux in the left colon[18]. Thus, exogenous ammonia is absorbed into the body mainly through the superior mesenteric vein. It is reported that the reflux blood from the splenic and superior mesenteric veins is distributed hydrodynamically in the main portal vein, that is, alongside the trunk on both sides of the wall of the portal vein. However, it is not fully mixed and enters the left and right branches of the portal vein. The right branch mainly receives superior mesenteric venous blood, while the left branch mainly receives blood from the splenic vein[7,8]. We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE.
Normally, urea is produced by the circulation of ornithine through the liver[19]. However, almost no exogenous ammonia was produced in the blood collected from the splenic vein. The concentration of ammonia in the superior mesenteric vein was higher than that in the splenic vein and the left and right branches of the portal vein, and the latter was higher than in the vena cava[20]. The anatomy of the portal vein and its mechanism of ammonia production are consistent with those of the portal vein. Similar results[21] have been found in humans: blood ammonia concentration, superior mesenteric vein > portal vein > splenic vein > peripheral vein, and the differences are significant. Later, the isotope hypothesis[22,23] was used to confirm the hypothesis. The results showed that the concentration of ammonia in the superior mesenteric vein was higher than that in the splenic vein and vena cava, suggesting that exogenous ammonia removal was not timely and would cause systemic circulation of ammonia to increase rapidly.
In early TIPS technology, it was easier to use the right than left branch of the portal vein for puncture target[24]. Currently, experienced interventional radiologists can choose the left or right branch as a puncture target, with no technical difficulty and a near 100% success rate[25]. Further consideration is how to improve the clinical success rate and reduce the incidence of HE and liver failure. It was hypothesized[26] that the blood components of the left and right branches of the intrahepatic portal vein are different. We believe that the choice of TIPS method plays a decisive role in treatment outcome.
In addition, from the anatomical point of view[27], the right branch of the portal vein supplies more of the right liver, and if it is partially or completely diverted, liver function impairment is aggravated. Moreover, the mesenteric vein blood, which contains a large number of toxins (including ammonia) and the liver factor[28], enters the right branch of the portal vein, and then is diverted, which increases the concentration of circulating blood ammonia. Liver failure and the high concentration of ammonia in the systemic circulation are the main causes of portal body shunt encephalopathy[29,30]. The pathogenesis of HE is complex, especially in TIPS treatment. In addition to being shunted around the liver, blood ammonia is not cleared, but because the liver blood supply is reduced after shunting, liver dysfunction and decreased removal of ammonia lead to the occurrence of HE. The hepatic dysfunction after shunting is related to the shunt flow and the quality of the shunt. As with insulin, high concentrations of glucagon and other liver factors in the blood likely lead to the occurrence of liver failure[31].
We can only correctly select the TIPS procedure after fully understanding the important differences between the delivery and concentration of substances in the left and right branches of the portal vein and the complications of shunting. The results of the present study provide evidence that after TIPS treatment, targeted puncture of the left branch of the intrahepatic portal vein may reduce the risk of HE. In group A, the incidence of HE was lower than that in group B and the occurrence of HE showed a downward trend. Our results confirmed in the TIPS process that targeted puncture of the left portal vein diverted the non-nutritive blood that came from the splenic vein into the TIPS shunt and minimized the incidence of HE.
The previous literature[15] only compared the overall incidence of HE in 3 years, and showed that HE occurred one year after TIPS[32]. Therefore, we compared the occurrence of HE after 1, 3, 6, 9 and 12 mo, and the total incidence of HE after 5 years. We showed that targeted puncture of the left branch of the intrahepatic portal vein reduced the risk of HE, but it had no direct influence on prognosis of portal-hypertension-related complications of refractory ascites or variceal bleeding.
In our study, the survival rate and recurrence rate for ascites and bleeding did not differ between the two groups. These results indicate the prognostic importance of TIPS placement for portal-hypertension-related complications. We believe that, as long as intrahepatic vein angiography shows that the anatomical structure meets the requirements, we should puncture the left portal vein, which significantly decreases the incidence of HE.
This study had several limitations. First, randomized controlled trials are needed to verify our results. Second, targeted puncture of the left intrahepatic portal vein during TIPS procedure is difficult for those who are used to targeted puncture of the right intrahepatic portal vein, which will take some time for such change. Finally, our hypothesis need to be validated by animal experiments and further study.
In conclusion, targeted puncture of the left branch of the intrahepatic portal vein during TIPS does not directly influence the prognosis of portal-hypertension-related complications of refractory ascites or variceal bleeding but may reduce the risk of HE.
Transjugular intrahepatic portosystemic shunt (TIPS) is currently used for the treatment of complications of portal hypertension. The incidence of hepatic encephalopathy (HE) remains a problem in TIPS placement. It has been reported that the right branch mainly receives superior mesenteric venous blood, while the left branch mainly receives blood from the splenic vein. We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt; therefore, targeted puncture of the left branch of the intrahepatic portal vein during TIPS may reduce the risk of HE.
TIPS is currently used for the treatment of complications of portal hypertension. With advances in materials, many experimental and clinical studies have been conducted using covered stent grafts, especially stent grafts covered with polytetrafluoroethylene, to improve the long-term patency of TIPS. However, the incidence of HE remains a problem in TIPS placement and affects the quality of life and long-term outcomes of patients.
It has been reported that the reflux blood from the splenic and superior mesenteric veins is distributed hydrodynamically in the main portal vein, that is, alongside the trunk on both sides of the wall of the portal vein. However, it is not fully mixed and enters the left and right branches of the portal vein. The right branch mainly receives superior mesenteric venous blood, while the left branch mainly receives blood from the splenic vein. We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE. The purpose of this study was to compare the effect of the left and right branches of the portal vein as TIPS shunt on the incidence of HE in patients who required TIPS placement for portal-hypertension-related complications of ascites or variceal bleeding. In the future, randomized controlled trials are needed to verify our results, and our hypothesis needs to be validated by animal experiments and further study.
The main objective was to establish whether the left branch of the intrahepatic portal vein as TIPS shunt reduced the risk of HE. If we realized this objective for future clinical practice in TIPS, we should target puncturing the left branch of the intrahepatic portal vein during TIPS as far as possible, because we hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE.
We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE. To achieve this objective, we conducted a single-center retrospective study that compared the influence of targeted puncture of the left and right branches of the portal vein on the incidence of HE in patients who required TIPS placement for portal-hypertension-related complications of ascites or variceal bleeding. The patients were divided into two groups: A (targeting of left branch of portal vein, n = 937) and B (targeting of right branch of portal vein, n = 307) (Figure 2). The outcomes of HE, recurrent variceal bleeding and/or ascites, and mortality were compared and analyzed between the groups. This study was not reported previously. Results were expressed as mean ± standard deviation and compared using the independent sample t test or one-way analysis of variance, and categorical variables were expressed as frequencies and compared using χ2 tests. The differences between the groups were compared using one-way analysis of variance followed by least significant difference t tests. Differences were considered significant at P < 0.05. The statistical analyses were performed with SPSS version 20.0.
This study showed that targeted puncture of the left branch of the intrahepatic portal vein during TIPS reduced the risk of HE, although it did not directly influence the prognosis of portal-hypertension-related complications of refractory ascites or variceal bleeding. It verified the hypothesis that the reflux blood from the splenic and superior mesenteric veins is distributed hydrodynamically in the main portal vein, that is, alongside the trunk on both sides of the wall of the portal vein. However, it is not fully mixed and enters the left and right branches of the portal vein. The right branch mainly receives superior mesenteric venous blood, while the left branch mainly receives blood from the splenic vein indirectly.
We found that the left branch of the intrahepatic portal vein during TIPS reduced the risk of HE. One of the toxic substances that causes HE is blood ammonia. HE is mainly caused by absorption of toxic substances from the intestinal portal vein system, through the shunt without the liver first pass effect into the systemic circulation, caused by dysfunction of the central nervous system. It has been reported that the reflux blood from the splenic and superior mesenteric veins is distributed hydrodynamically in the main portal vein, that is, alongside the trunk on both sides of the wall of the portal vein. However, it is not fully mixed and enters the left and right branches of the portal vein. The right branch mainly receives superior mesenteric venous blood, while the left branch mainly receives blood from the splenic vein. We hypothesized that targeted puncture of the left portal vein would divert the non-nutritive blood from the splenic vein into the TIPS shunt and reduce the incidence of HE. As far as possible, we should target puncturing the left branch of the intrahepatic portal vein during TIPS procedure for clinical practice in the future.
We can learn from this study that, to reduce the risk of HE, we should target puncturing the left branch of the intrahepatic portal vein during TIPS for clinical practice in the future. We believe that, as long as intrahepatic vein angiography shows that the anatomical structure meets the requirements, we should puncture the left portal vein, which significantly decreases the incidence of HE. In future research, randomized controlled trials are needed to verify our results, and it will take some time to switch to the targeted puncture of the left intrahepatic portal vein during TIPS procedure. Finally, our hypothesis needs to be validated by animal experiments to find direct evidence for hydrodynamics of blood.
The authors thank all the patients who were involved in this study and colleagues of the Department of Radiology of Air Force Medical Center of PLA for their contributions to the data collection.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Abd-Elsalam S, Milovanovic T S- Editor: Ma RY L- Editor: Filipodia E- Editor: Huang Y
| 1. | García-Pagán JC, Caca K, Bureau C, Laleman W, Appenrodt B, Luca A, Abraldes JG, Nevens F, Vinel JP, Mössner J, Bosch J; Early TIPS (Transjugular Intrahepatic Portosystemic Shunt) Cooperative Study Group. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362:2370-2379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 826] [Cited by in RCA: 842] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 2. | Ascha M, Abuqayyas S, Hanouneh I, Alkukhun L, Sands M, Dweik RA, Tonelli AR. Predictors of mortality after transjugular portosystemic shunt. World J Hepatol. 2016;8:520-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 3. | Nardelli S, Gioia S, Pasquale C, Pentassuglio I, Farcomeni A, Merli M, Salvatori FM, Nikolli L, Torrisi S, Greco F, Nicoletti V, Riggio O. Cognitive Impairment Predicts The Occurrence Of Hepatic Encephalopathy After Transjugular Intrahepatic Portosystemic Shunt. Am J Gastroenterol. 2016;111:523-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 78] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 4. | Rössle M. TIPS: 25 years later. J Hepatol. 2013;59:1081-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 287] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 5. | Weber CN, Nadolski GJ, White SB, Clark TW, Mondschein JI, Stavropoulos SW, Shlansky-Goldberg RD, Trerotola SO, Soulen MC. Long-Term Patency and Clinical Analysis of Expanded Polytetrafluoroethylene-Covered Transjugular Intrahepatic Portosystemic Shunt Stent Grafts. J Vasc Interv Radiol. 2015;26:1257-65; quiz 1265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 6. | Jung HS, Kalva SP, Greenfield AJ, Waltman AC, Walker TG, Athanasoulis CA, Wicky ST. TIPS: comparison of shunt patency and clinical outcomes between bare stents and expanded polytetrafluoroethylene stent-grafts. J Vasc Interv Radiol. 2009;20:180-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 7. | Ursic M, Ravnik D, Hribernik M, Pecar J, Butinar J, Fazarinc G. Gross anatomy of the portal vein and hepatic artery ramifications in dogs: corrosion cast study. Anat Histol Embryol. 2007;36:83-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Maruyama H, Okugawa H, Ishibashi H, Takahashi M, Kobayashi S, Yoshizumi H, Yokosuka O. Carbon dioxide-based portography: an alternative to conventional imaging with the use of iodinated contrast medium. J Gastroenterol Hepatol. 2010;25:1111-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 9. | Trebicka J. Emergency TIPS in a Child-Pugh B patient: When does the window of opportunity open and close? J Hepatol. 2017;66:442-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 10. | Miraglia R, Maruzzelli L, Tuzzolino F, Petridis I, D'Amico M, Luca A. Transjugular Intrahepatic Portosystemic Shunts in Patients with Cirrhosis with Refractory Ascites: Comparison of Clinical Outcomes by Using 8- and 10-mm PTFE-covered Stents. Radiology. 2017;284:281-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 11. | Fonio P, Discalzi A, Calandri M, Doriguzzi Breatta A, Bergamasco L, Martini S, Ottobrelli A, Righi D, Gandini G. Incidence of hepatic encephalopathy after transjugular intrahepatic portosystemic shunt (TIPS) according to its severity and temporal grading classification. Radiol Med. 2017;122:713-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 12. | Berlioux P, Robic MA, Poirson H, Métivier S, Otal P, Barret C, Lopez F, Péron JM, Vinel JP, Bureau C. Pre-transjugular intrahepatic portosystemic shunts (TIPS) prediction of post-TIPS overt hepatic encephalopathy: the critical flicker frequency is more accurate than psychometric tests. Hepatology. 2014;59:622-629. [PubMed] |
| 13. | Chen L, Xiao T, Chen W, Long Q, Li R, Fang D, Wang R. Outcomes of transjugular intrahepatic portosystemic shunt through the left branch vs. the right branch of the portal vein in advanced cirrhosis: a randomized trial. Liver Int. 2009;29:1101-1109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Chen HJ, Liu PF, Chen QF, Shi HB. Brain Microstructural Abnormalities in Patients With Cirrhosis Without Overt Hepatic Encephalopathy: A Voxel-Based Diffusion Kurtosis Imaging Study. AJR Am J Roentgenol. 2017;209:1128-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Fiati Kenston SS, Song X, Li Z, Zhao J. Mechanistic insight, diagnosis, and treatment of ammonia-induced hepatic encephalopathy. J Gastroenterol Hepatol. 2019;34:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 16. | Levitt DG, Levitt MD. A model of blood-ammonia homeostasis based on a quantitative analysis of nitrogen metabolism in the multiple organs involved in the production, catabolism, and excretion of ammonia in humans. Clin Exp Gastroenterol. 2018;11:193-215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 17. | Miner-Williams W, Deglaire A, Benamouzig R, Fuller MF, Tomé D, Moughan PJ. Endogenous proteins in terminal ileal digesta of adult subjects fed a casein-based diet. Am J Clin Nutr. 2012;96:508-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Miyamoto R, Inagawa S, Nagai K, Maeda M, Kemmochi A, Yamamoto M. Three-dimensional reconstruction of vascular arrangement including the hepatic artery and left gastric vein during gastric surgery. Springerplus. 2016;5:835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | De Chiara F, Heebøll S, Marrone G, Montoliu C, Hamilton-Dutoit S, Ferrandez A, Andreola F, Rombouts K, Grønbæk H, Felipo V, Gracia-Sancho J, Mookerjee RP, Vilstrup H, Jalan R, Thomsen KL. Urea cycle dysregulation in non-alcoholic fatty liver disease. J Hepatol. 2018;69:905-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 138] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 20. | Olde Damink SW, Jalan R, Redhead DN, Hayes PC, Deutz NE, Soeters PB. Interorgan ammonia and amino acid metabolism in metabolically stable patients with cirrhosis and a TIPSS. Hepatology. 2002;36:1163-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 161] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 21. | Chu J, Sun X, Piao L, Chen Z, Huang H, Lu C, Xu J. [Portosystemic shunt via the left branch of portal vein for the prevention of encephalopathy following transjugular intrahepatic portosystemic shunt]. Zhonghua Gan Zang Bing Za Zhi. 2002;10:437-440. [PubMed] |
| 22. | Garcier JM, Bousquet J, Alexandre M, Filaire M, Viallet JF, Vanneuville G, Boyer L. Visualisation of the portal flows by porto-scanner. Surg Radiol Anat. 2000;22:239-242. [PubMed] |
| 23. | Mogicato G, Vautravers G, Meynaud-Collard P, Deviers A, Sautet J. Blood flows in tributaries of the portal vein: anatomical and angiographic studies in normal beagle dogs. Anat Histol Embryol. 2015;44:460-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Luo SH, Chu JG, Huang H, Yao KC. Effect of initial stent position on patency of transjugular intrahepatic portosystemic shunt. World J Gastroenterol. 2017;23:4779-4787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | RiChard J, Thornburg B. New Techniques and Devices in Transjugular Intrahepatic Portosystemic Shunt Placement. Semin Intervent Radiol. 2018;35:206-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Mari L, Acocella F. Vascular anatomy of canine hepatic venous system: a basis for liver surgery. Anat Histol Embryol. 2015;44:212-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Gill AB, Hilliard NJ, Hilliard ST, Graves MJ, Lomas DJ, Shaw A. A semi-automatic method for the extraction of the portal venous input function in quantitative dynamic contrast-enhanced CT of the liver. Br J Radiol. 2017;90:20160875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 28. | Stefanello S, Mezzomo MP, Zeni D, Ebling RC, Soares AV, Kozloski GV. Oxygen uptake and net flux of metabolites by splanchnic tissues of sheep in response to short-term mesenteric infusion of nitrogenous compounds. J Anim Physiol Anim Nutr (Berl). 2018;102:853-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Cruz NF, Dienel GA, Patrick PA, Cooper AJL. Organ Distribution of 13N Following Intravenous Injection of [13N]Ammonia into Portacaval-Shunted Rats. Neurochem Res. 2017;42:1683-1696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Sawhney R, Holland-Fischer P, Rosselli M, Mookerjee RP, Agarwal B, Jalan R. Role of ammonia, inflammation, and cerebral oxygenation in brain dysfunction of acute-on-chronic liver failure patients. Liver Transpl. 2016;22:732-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 31. | Samson WK, Stein LM, Elrick M, Salvatori A, Kolar G, Corbett JA, Yosten GL. Hypoglycemia unawareness prevention: Targeting glucagon production. Physiol Behav. 2016;162:147-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 32. | Pereira K, Carrion AF, Salsamendi J, Doshi M, Baker R, Kably I. Endovascular Management of Refractory Hepatic Encephalopathy Complication of Transjugular Intrahepatic Portosystemic Shunt (TIPS): Comprehensive Review and Clinical Practice Algorithm. Cardiovasc Intervent Radiol. 2016;39:170-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |