Published online Feb 14, 2019. doi: 10.3748/wjg.v25.i6.644
Peer-review started: November 28, 2018
First decision: January 18, 2019
Revised: January 25, 2019
Accepted: January 28, 2019
Article in press: January 28, 2019
Published online: February 14, 2019
Processing time: 78 Days and 16.5 Hours
Development of non-invasive methods to risk-stratify patients and predict clinical endpoints have been identified as one of the key research priorities in primary sclerosing cholangitis (PSC). In addition to serum and histological biomarkers, there has been much recent interest in developing imaging biomarkers that can predict disease course and clinical outcomes in PSC. Magnetic resonance imaging/magnetic resonance cholangiopancreatography (MRI/MRCP) continue to play a central role in the diagnosis and follow-up of PSC patients. Magnetic resonance (MR) techniques have undergone significant advancement over the last three decades both in MR data acquisition and interpretation. The progression from a qualitative to quantitative approach in MR acquisition techniques and data interpretation, offers the opportunity for the development of objective and reproducible imaging biomarkers that can potentially be incorporated as an additional endpoint in clinical trials. This review article will discuss how the role of MR techniques have evolved over the last three decades from emerging as an alternative diagnostic tool to endoscopic retrograde cholangiopancreatography, to being instrumental in the ongoing search for imaging biomarker of disease stage, progression and prognosis in PSC.
Core tip: Magnetic resonance imaging/magnetic resonance cholangiopancreatography (MRI/MRCP) remains the cornerstone in the diagnosis and follow-up of primary sclerosing cholangitis (PSC) patients. However, heterogeneity in acquisition, image processing and interpretation varies significantly. There is ongoing interest in establishing non-invasive methods to predict clinical endpoints in PSC. A number of recent publications have focused on objectively quantifying output from various magnetic resonance (MR) techniques and have suggested MR parameters as potential prognostic risk-stratification tool in PSC. Our aim is to revisit the historical use of imaging in PSC and consolidate the evolving role of the different MR techniques to date in the quest for establishing a validated imaging biomarker for PSC.
- Citation: Selvaraj EA, Culver EL, Bungay H, Bailey A, Chapman RW, Pavlides M. Evolving role of magnetic resonance techniques in primary sclerosing cholangitis. World J Gastroenterol 2019; 25(6): 644-658
- URL: https://www.wjgnet.com/1007-9327/full/v25/i6/644.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i6.644
Primary sclerosing cholangitis (PSC) is a rare, chronic, immune-mediated liver disease, characterised by intrahepatic and extrahepatic bile duct inflammation, leading to chronic cholestasis, biliary fibrosis and liver cirrhosis with portal hypertension[1]. It has a male preponderance, with a mean age of diagnosis of 40 years, and a strong association with concomitant inflammatory bowel disease (IBD)[2]. The population incidence ranges from 0 to 1.3 per 100000 persons annually, and the prevalence ranges from 0 to 16.2 per 100000 persons[3]. A clinical definition for PSC was based upon three landmark papers in the 1980s from the United States, United Kingdom and Norway[4-6]. Subsequently, four sub-types of PSC were described. ‘Classical’ large-duct PSC (LD-PSC), accounting for 90% of patients, involves either the intrahepatic bile ducts, extra hepatic bile ducts or both. It is usually diagnosed on the basis of cholestatic liver biochemistry and characteristic changes in the bile ducts on cholangiography. Magnetic resonance imaging/magnetic resonance cholangiopancreatography (MRI/MRCP) is the standard imaging modality to confirm a diagnosis of LD-PSC[7]. ‘Small duct’ PSC that has normal cholangiography but affects only the small intrahepatic bile ducts on liver histology, accounts for 5% of patients[8]. PSC with ‘autoimmune hepatitis (AIH) overlap’, confirmed histologically in those with elevated transaminases and/or immunoglobulin G levels and an abnormal cholangiogram, presents in 5% of patients[9]. Lastly, ‘PSC with high immunoglobulin G subclass 4 (IgG4) levels’ in the serum and/or tissue, is reported in 12%-18% of LD-PSC patients, with a distinct clinical phenotype and natural history of disease[10,11].
PSC is insidious, with nearly half of patients being asymptomatic at diagnosis, identified after investigation for abnormal liver biochemistry[12]. PSC is considered a premalignant condition, associated with the development of hepatobiliary and colorectal cancers, the most common being cholangiocarcinoma[13]. In the absence of effective medical therapies to date, liver transplantation is the only proven life-extending intervention. PSC accounts for 10%-15% of all liver transplant activity in Europe and the median transplant-free survival of patients with PSC is 14.5 years[13]. There is interest in developing non-invasive clinical risk-stratification methods and surrogate markers in the disease.
This article will review how the role of magnetic resonance (MR) techniques have evolved over the last three decades from emerging as an alternative diagnostic tool to endoscopic retrograde cholangiopancreatography (ERCP), to being instrumental in the ongoing search for an imaging biomarker of disease stage, progression and prognosis in PSC. A summary of the MR techniques that will be discussed in this review is presented in Table 1.
| MR technique | Description of technique | Role in PSC |
| T2-weighted MRCP | Non-contrast sequences that depict fluid-filled structures such as bile ducts as high-intensity (white) compared to low-intensity (grey/black) of adjacent structures. | Visualisation of biliary anatomy. |
| Three-dimensional MRCP | Respiratory-triggered, single volume thin slab acquisitions producing isotropic images. | Preferred sequences for optimal multi angle visualisation of the biliary anatomy. |
| Two-dimensional MRCP | Specific sequences combining coronal thin-slab and rotating oblique-coronal thick-slab image acquisition. | Single shot T2w MRCP sequences are used when three-dimensional MRCP has artefacts or not feasible. |
| T2-weighted liver axial | Measure of T2 relaxation time in liver parenchyma. Both fat and water appear bright. | Sequence for optimal visualisation of the liver parenchyma. |
| T1-weighted liver axial | Measure of T1 relaxation time in liver parenchyma. Fat appears bright, water appears dark. | Sequence for optimal visualisation of the liver parenchyma. |
| MR elastrography | Generates an elastogram map. Specific regions can be selected to obtain mean liver stiffness (kilopascals; kPa). | Quantification and distribution of liver fibrosis. |
| Diffusion-weighted MRI | Captures changes in the diffusion properties of water protons in tissue represented as the apparent diffusion coefficient. | Can be used to assess liver parenchymal morphological changes (e.g., tumours) and as surrogate for liver fibrosis. |
| Dynamic contrast-enhanced MRI | Measures T1 changes in liver parenchyma following bolus administration of gadolinium in different phases of uptake and elimination. | Delineates flow in vessels, permeability and enhancement of parenchyma. Can be used to quantify liver function using flow and permeability parameters as surrogate for liver fibrosis. |
The earliest description of PSC was found in a publication by Hofmann 1867[14]. The German pathologist reported two post-mortem descriptions of obstruction of the common hepatic duct by thickening of the duct walls with the absence of malignancy and stones. However, it was not until 1924-1925 that the first well-documented case was reported by the French surgeons Delbet[15] and Lafourcade[16]. The first in the English literature was reported by Miller in 1927[17]. In 1958, Schwarts and Dale reported 13 cases who they felt were consistent with a diagnosis of PSC on review of worldwide literature[18].
PSC was historically a condition recognised intra-operatively when the accessible portion of the extrahepatic biliary system was involved. Subsequently, with the development of operative cholangiography, surgeons were better able to visualise the intra- and extrahepatic bile ducts, extent of the disease and plan optimum site for biliary drainage using T-tube cholangiograms. Reported series of PSC cases were small prior to 1980[5]. The advent of percutaneous transhepatic cholangiography (PTC) and ERCP paved the way for non-operative methods of obtaining a cholangiogram. This led to more detailed description of PSC with a rise in reporting in the medical literature worldwide. The first classification of intrahepatic and extrahepatic features of PSC using cholangiograms obtained from T-tube, PTC, or ERCP was reported in 1984 by Chen and Goldberg using a case series of 19 patients[19].
ERCP had been the standard of reference for obtaining a cholangiogram in diagnosing PSC until the emergence of MRCP. Whilst MRI of the liver was being performed for liver disease, it was not until 1991 that the first “MR cholangiography” sequence was performed by Wallner et al[20]. They developed a T2-weighted rapid sequential gradient-echo two-dimensional (2D) acquisition and a three-dimensional (3D) post-processing technique to produce coronal and sagittal images, without the need for ionising radiation or intravenous biliary contrast. Static fluid-filled structures in the abdomen have long T2 relaxation time in comparison to adjacent tissue. MRCP exploits these differences by using heavily T2-weighted sequences that depicts higher signal intensity (white) of slow-moving or static fluid within the bile and pancreatic ducts in comparison to lower signal intensity (grey/black) of adjacent solid structures. Specific image acquisition sequences ensure that flowing blood has minimal or no measurable signal in order not to mistake blood vessels for bile or pancreatic ducts. Acquisition is performed in a fasted state (often for at least 4 h) to reduce signal overlap from fluid in the surrounding stomach and duodenum, reduce peristalsis and promote gallbladder distension. Some centres use a negative oral contrast agent, such as 200-400 mL of pineapple juice, 20-30 min prior to MRCP. The high concentration of manganese in pineapple juice is thought to have a paramagnetic effect, suppressing signal from overlapping fluid in the stomach and duodenum[21]. 2D and 3D MRCP has undergone significant advances since its first description with shorter acquisition time, better image quality and improved reconstruction algorithms. Optimal imaging protocol depends on the specific scanner used and parameters including field strength (e.g., 1.5 or 3T), the manufacturer, institutional preference and experience. Good quality 3D MRCP acquisition also depends on good navigator-based respiratory-triggering of the diaphragm.
The International PSC Study Group has recently published a position statement from multidisciplinary experts offering recommendations on the minimum standard for performing MRI/MRCP in PSC as well as a more complete workup[22].
Bile duct imaging: T2-weighted MRCP is preferred to T1-weighted for improved visualisation of biliary ducts. 3D MRCP is preferred over 2D MRCP as the thinner 1mm sections result in higher spatial resolution with excellent signal-to-noise ratio. Post-processing 3D reconstruction using isotropic data allows creation of multiple projections. However, the trade-off for this is longer acquisition time and motion artefact. If a hepatobiliary contrast agent is used, 3D MRCP sequences should be acquired first or else the bile signal will be suppressed.
Liver parenchyma imaging: Cross sectional T2 and T1-weighted acquisition is recommended. T2-weighted coronal plane acquisitions covering most of the liver from anterior to posterior is important for evaluation of peripheral intrahepatic ducts. Fat-suppressed T1-weighted image is acquired as it adds information on the liver parenchyma. Gadolinium-based intravenous contrast agents form part of a more complete workup of PSC patients to detect and differentiate mass lesions and inflammation. MR contrast agents can be classified as purely extracellular or extracellular with a hepatocyte-specific (hepatobiliary) component. Depending on the contrast agent used, post-contrast images are acquired in different phases including arterial, portal venous, equilibrium (parenchyma), delayed and hepatobiliary phases. Some centres perform diffusion-weighted imaging (DWI) routinely for parenchymal and lesion characterisation.
Cholangiography is required to make a diagnosis of LD-PSC. ERCP is invasive with potential serious complications including pancreatitis, cholangitis, perforation and bleeding[23]. Over the last twenty years, MRCP has replaced ERCP as the first line imaging method for the diagnosis of PSC. A large meta-analysis, including 189 PSC patients, compared the diagnostic accuracy of MRCP against combined clinical, biochemical, and ERCP or PTC endpoint as the reference standard for diagnosis. The study concluded that the sensitivity and specificity of MRCP for the diagnosis of PSC were 0.86 (95%CI: 0.80-0.90) and 0.94 (95%CI: 0.86-0.98) respectively, with an area under the receiver operating curve of 0.91, supporting a high diagnostic accuracy[24]. Advancement in imaging techniques with higher quality images and spatial resolution is likely to have increased the diagnostic accuracy further. Both the European Association for the Study of the Liver (EASL) and American Association for the Study of Liver Diseases (AASLD) recommend MRCP as the first-choice imaging modality in PSC[7,25]. Performing MRCP first has been shown to be a more cost-effective strategy[26,27]. Moreover, incomplete biliary tract distension mimicking the ductal irregularities of PSC can give rise to false-positive diagnosis on ERCP cholangiogram and false-negative diagnosis if a high-grade stricture causes inadequate opacification of the intrahepatic ducts[28,29]. However, ERCP is still performed when diagnostic doubt exists after MRCP scanning.
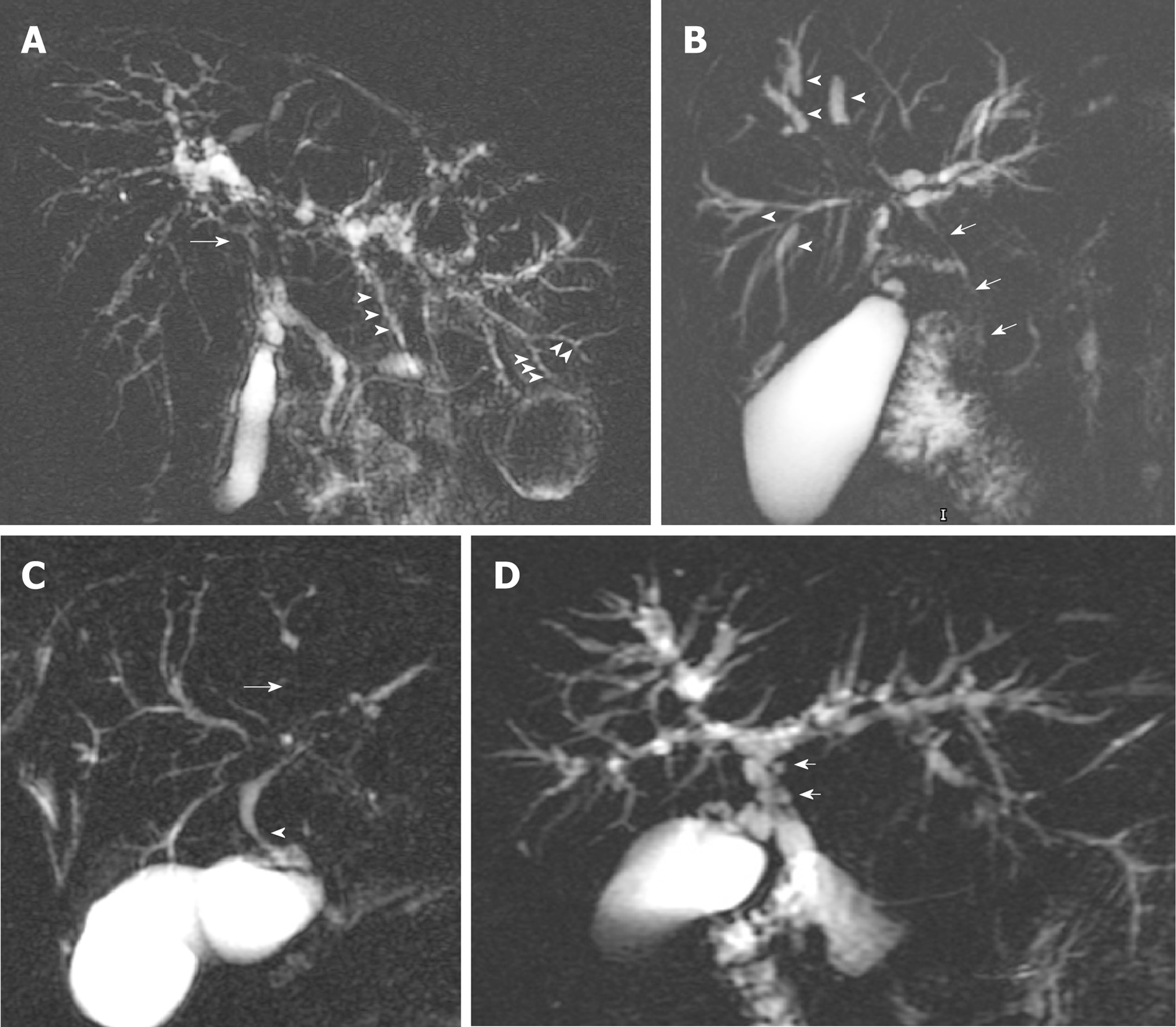
Identification of multifocal fibrotic strictures and areas of dilatation and ductal wall thickening of the intrahepatic or extrahepatic biliary systems, or both, underpins the diagnosis of LD-PSC. The majority of patients have involvement of both the intra- and extrahepatic bile ducts, with less than 25% with intrahepatic duct disease only[25]. Exclusive involvement of the extrahepatic duct is uncommon (less than 5%) and should prompt a search for an alternative cause[1]. Figure 1 illustrates typical MRCP features of LD-PSC. The MRI/MRCP features of PSC that have been reported in the literature are summarised in Table 2[22,30-32].
| Bile duct changes |
| Multiple annular or short segmental strictures (1-2 mm) with slightly dilated ducts among them: “beaded” appearance |
| Obliteration of small peripheral ducts “pruned tree” |
| Periductal inflammation |
| Thickening of walls of large ducts |
| Strictures seen at bile duct bifurcation |
| Angles between peripheral and central bile ducts become obtuse |
| Exclusive involvement of extrahepatic bile duct is infrequent |
| Bile duct dilatations are usually subtle |
| Retraction of papilla |
| Webs, diverticula and pigmented stones |
| Liver parenchymal changes |
| Segmental or lobular atrophy with compensatory hypertrophy attributed to chronic biliary obstruction |
| Patchy areas of peripheral parenchymal enhancement |
| Caudate lobe hypertrophy1 |
| Spherical liver shape2 |
| Peripheral wedge-shaped areas with focal increased signal intensity on T2-weighted images3 |
| T2-weighted hyperintensity around portal vein branches |
| Regional changes |
| Gallbladder enlargement |
| Enlarged regional lymph nodes |
| Signs of portal hypertension including splenomegaly and collateral vessels |
As for any other imaging modality, MRI/MRCP is subject to inter-observer variability. The cholangiographic features of PSC on its own do not necessarily distinguish PSC from secondary sclerosing cholangitis, particularly in the absence of IBD diagnosis. Immunoglobulin G4-related sclerosing cholangitis (IgG4-SC) can often mimic PSC on MRCP. Features that support a diagnosis of IgG4-SC over PSC include dilatation proximal to confluent stricture, symmetrical bile duct wall thickening with smoother outer and inner margins, presence of continuous as opposed to skip disease in the bile ducts, common bile duct thickness greater than 2.5 mm, gallbladder and pancreatic involvement[33,34]. Other mimickers include ischaemic cholangiopathy, acquired immune deficiency syndrome-related cholangiopathy, secondary sclerosing cholangitis after repeated ascending cholangitis and portal biliopathy.
Liver biopsy, assessed using Ludwig staging system, has been shown to be an independent predictor of survival in PSC[35,36]. More recently, in a multicentre PSC cohort, three separate histological scoring systems (Nakanuma, Ishak and Ludwig), have been shown to have independent prognostic value in monitoring disease progression[37]. However, liver biopsy is not recommended in the current EASL and AASLD guidelines for the diagnosis and follow-up of PSC due to its invasive nature and risk of complications[7,25]. Moreover, distribution of disease in PSC is patchy and liver biopsy is prone to sampling variability[38]. A liver biopsy is usually performed when MRCP/ERCP is normal to diagnose small-duct PSC and/or there is suspicion of an autoimmune overlap syndrome or IgG4-SC.
Several promising serum biomarkers have been studied as surrogate markers for liver fibrosis. The ELF test is based on three direct markers of fibrogenesis: hyaluronic acid (HA), tissue inhibitor of metalloproteinases-1 (TIMP-1) and procollagen III amino terminal peptide (PIIINP). It has been reported in two large, retrospective, cohort studies to be a strong predictor of clinical outcomes defined as liver-transplant or death, and independent of other risk factors or prognostic scores that predict outcomes[39,40]. Fibrosis-4 index (FIB-4) and aspartate aminotransferase to platelet ratio index (APRI) have been studied in other chronic liver diseases as a marker of liver fibrosis, but their roles in PSC have not been reported to date.
Liver stiffness has also been shown to be a surrogate marker for liver fibrosis. Studies have shown correlation with stages of liver fibrosis, liver decompensation and survival[41,42]. Liver stiffness can be measured using shear-wave-based technology such as vibration controlled transient elastography (VCTE) or magnetic resonance elastography (MRE). Two retrospective studies have shown good correlation between baseline VCTE measurements and changes in VCTE measurements with stages of fibrosis and clinical outcomes[43,44]. Although widely available with relatively low-cost, false positive elevation of VCTE measurements can be caused by biliary obstruction and active inflammation such as occurs in PSC patients[45,46]. Technical failures and unreliable results have been reported to be as high as 10% in PSC patients[43]. There is limited data reported on the use of other ultrasound-based techniques such as point shear wave elastography and 2D shear wave elastography in PSC.
MRE has been shown to predict liver decompensation in a large single-centre retrospective study involving 266 PSC patients with median follow-up of 2 years[47]. In a small sub analysis of this study, MRE was also shown to correlate with different stages of liver fibrosis. In comparison to VCTE, it has the added advantage of being able to visualise the whole liver and identify patchy areas of fibrosis in PSC. MRE is able to assess more than 1000 times the volume of liver than VCTE[48]. Unlike VCTE, MRE can be performed regardless of patient’s body habitus or presence of ascites. However, MRE is not widely available, is more costly and time-consuming[49]. Whilst MRE was found to have better diagnostic accuracy than VCTE for staging of liver fibrosis in non-alcoholic fatty liver disease[50], there has been no head-to-head comparison performed in a chronic cholestatic disease such as PSC. MRE has also been shown to correlate better with Mayo PSC risk score than VCTE and liver stiffness quantified by MRE is an independent predictor of worse score[51].
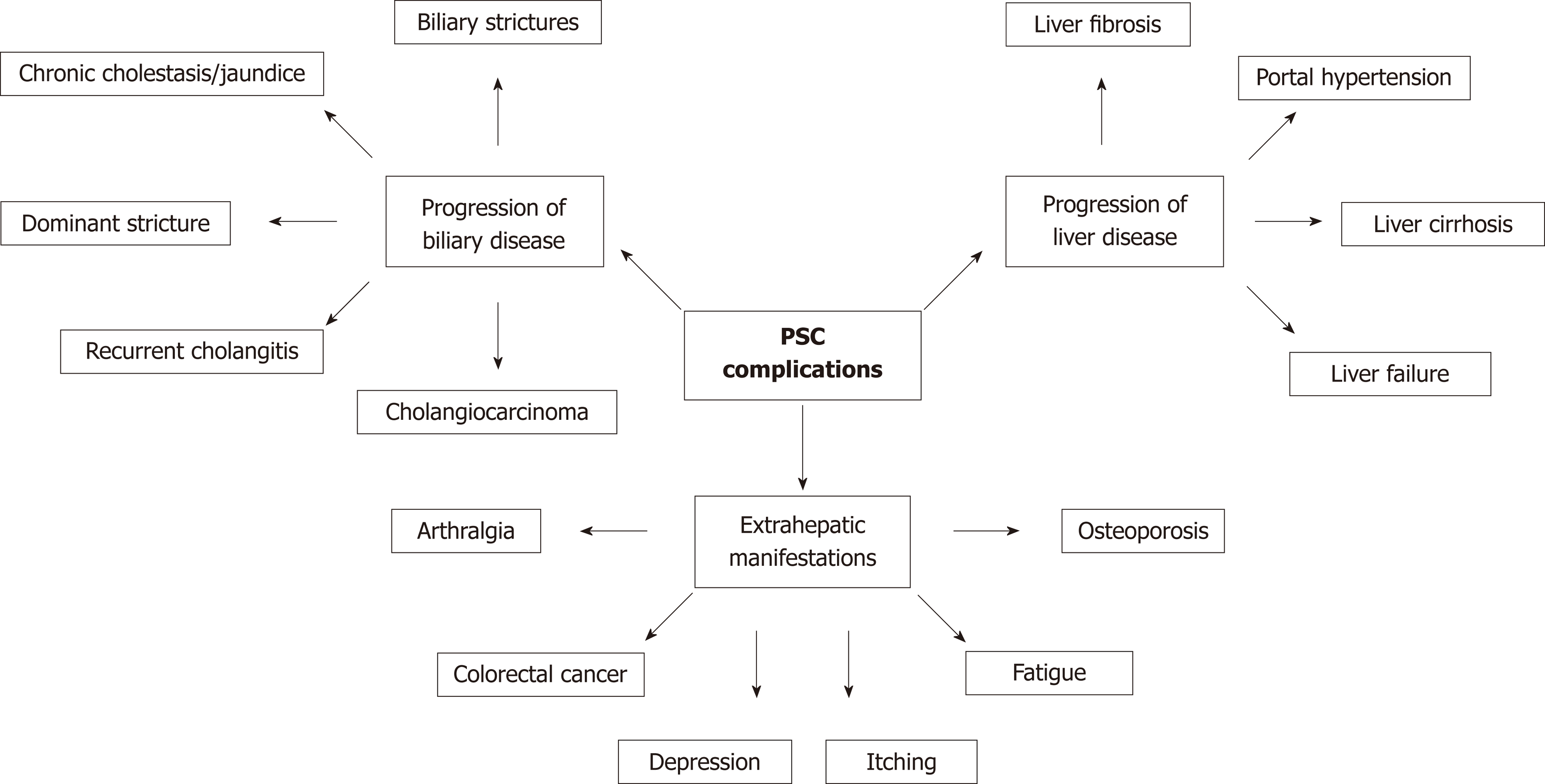
Disease progression in PSC can affect the biliary tree and/or the liver. A third domain of disease complications or progression includes extra-hepatic manifestations and symptoms, which can be independent of the stage of the biliary or liver disease. The complications of disease progression in PSC are summarised in Figure 2. Predicting which patients are at risk of developing these complications is challenging in clinical practice. Development of non-invasive methods to risk-stratify patients with PSC and predict clinical events was identified as a research priority in a 2016 survey by PSC Support, a registered United Kingdom charity[52]. It highlighted that patients experience significant anxiety due to the uncertainty about the future of their disease; in particular, the risk of disease progression, malignancy, and liver transplantation. In addition to the stage of liver fibrosis, a combination of serum biomarkers, clinical risk prediction model and cholangiographic features can be used to risk-stratify patients.
Serum alkaline phosphatase (ALP) has been the most widely studied serum biomarker in PSC. ALP levels can fluctuate throughout the disease course but persistently low ALP has been shown to correlate with better clinical outcomes. A cut-off value of 1.5 × the upper limit of normal (ULN) has been demonstrated in several patient cohorts to have prognostic implications[53-57]. However, patients can have normal serum ALP with advanced liver disease[58]. In one study with 10-year follow-up, 62% of patients did not experience any liver-related endpoints despite having a serum ALP that did not improve to levels less than 1.5 × ULN[57].
Several clinical risk scores have been developed to predict disease progression and clinical outcomes in PSC. The revised Mayo PSC risk score, based on age, bilirubin, albumin, AST and variceal bleeding, is the most commonly used clinical risk model[59]. The model was developed using multi-centre large cohort data (n = 405) and was subsequently validated in a separate cohort (n = 105). The risk score provides survival estimates up to 4-year follow-up but does not include time to liver transplant. Given that it is made up of markers predictive of advanced disease, it is not surprising that it has insufficient power and is not clinically useful in discriminating and predicting the clinical course of early disease.
The Amsterdam-Oxford risk score based on seven variables (PSC subtype, age at diagnosis, albumin, platelet, AST, ALP and bilirubin) predicted long-term transplant-free survival in a large derivation cohort (n = 692) and external validation cohort (n = 264)[60]. The PSC risk estimate tool (PREsTo) was recently developed using machine learning techniques. It consists of 9 variables (bilirubin, albumin, ALP times the ULN, platelet, AST, haemoglobin, sodium, age and number of years since diagnosis)[61]. The model was derived using 509 patients and validated in an international multicentre cohort (278 patients) who did not have markers of advanced disease. It accurately predicted the 5-year risk of liver decompensation. None of the prognostic scores that have been developed to date has entered radiological features as a variable into their modelling methods, probably because of the significant inter-observer variability in radiological interpretation even among experienced experts[62].
Given that cholangiography is required for the diagnosis of the majority cases of PSC, it would seem intuitive to use cholangiographic features as predictors of disease stage and prognosis. Whilst there are limited studies evaluating the use of ERCP cholangiogram findings, there is an increasing trend of utilising MR techniques to study both the liver parenchyma and cholangiography of PSC patients simultaneously to propose imaging biomarkers in PSC. The non-invasive nature of MR techniques makes this an attractive option as a surrogate marker.
ERCP: Craig et al[63] retrospectively reviewed ERCP cholangiograms of a cohort of 174 PSC patients with relatively advanced disease and found that both high-grade intrahepatic duct strictures and diffuse intrahepatic duct strictures were associated with a lower 3-year survival[63]. Similarly, Olsson et al[64] concluded that high-grade intrahepatic strictures predicted shorter survival in a study involving 94 PSC patients. The Amsterdam cholangiographic classification system was developed by Ponsioen et al[65], incorporating the previously reported classifications by Majoie et al[66] and Chen-Goldberg[19]. It is based on scoring intrahepatic and extrahepatic stricture and dilatation severity on ERCP cholangiograms as outlined in Table 3. In a large single-centre study with a long follow-up period, 133 patients’ cholangiograms were scored. Cholangiographic scores were inversely correlated to survival, and together with age at ERCP, a prognostic model was derived[65]. It remains to be externally validated, perhaps reflecting the shift away from invasive biomarkers of disease.
| Type | Intrahepatic | Extrahepatic |
| 0 | No visible abnormalities | No visible abnormalities |
| I | Multiple calibre changes; minimal dilatation | Slight irregularities of duct contour; no stricture |
| II | Multiple strictures; saccular dilatations, decreased arborisation | Segmental strictures |
| III | Only central branches filled despite adequate filling pressure; severe pruning | Strictures of almost entire length of duct |
| IV | - | Extremely irregular margins; diverticulum-like outpouchings |
MRI/MRCP: MRI/MRCP presents a more favourable option than ERCP as a marker of disease activity and prognosis in PSC as it allows co-assessment of liver parenchyma and biliary abnormalities. Petrovic et al[67] retrospectively examined the relationship between MRI/MRCP features and survival as predicted by the Mayo risk score. The severity of biliary stricture was graded using the Amsterdam cholangiographic classification. In this study involving 47 patients with PSC, delayed (3-min post contrast) peribiliary hyperenhancement in the liver parenchyma using extracellular gadolinium contrast, showed weak correlation with Mayo risk score. There was no correlation with peribiliary hyperenhancement at 2-min post contrast, intrahepatic or extrahepatic duct grading of strictures. Extrapolating ERCP-based cholangiographic findings to MR-based cholangiography cast doubts on the reproducibility of stricture grading, particularly with variability in contrast injection technique and volume used during ERCP. Tenca et al[68] reported only moderate agreement between ERCP and MRCP cholangiograms using a modified Amsterdam scoring system. Weak correlations were demonstrated between severity of biliary changes and serum ALP as well as clinical endpoints defined by liver transplantation or death.
Change in the morphological appearance of the biliary tree and liver on interval MRI/MRCP is often used to comment on whether the disease is stable or has progressed. Ruiz et al[69] designed the first MRI-based score to determine radiological disease progression on follow-up MRI/MRCP of 142 well-characterised PSC patients. They designed an interpretation standard model that converted radiological descriptors into categorical variables in this study, which allowed them to systematically analyse the bile ducts and liver parenchyma. An MRI progression risk score model was built using factors that predicted radiological progression between two successive MRIs, as shown in Figure 3.
This study demonstrated radiological progression in 58% of patients (n = 37) over a 4-year follow-up period. Both scores had area under receiver operating characteristic curve of 80% and 83% respectively for predicting radiological progression. However, the study did not take into account inter-observer variability, had no correlation with clinical outcomes and did not have ERCP as the reference standard. The MRI score is awaiting external validation. Kitzing et al[70] subsequently examined serial MRI/MRCP images and reported that liver morphological changes on surveillance imaging, specifically liver atrophy, was associated with adverse clinical outcome and shorter transplant-free survival over a mean intervening period of 5 years.
Several studies have evaluated the changes seen on contrast-enhanced MRI sequences in PSC with mixed evidence. Bader et al[71] studied 52 patients with PSC and reported that there were no correlations between liver parenchymal signal abnormalities or biliary ductal features and Childs-Pugh or Model for End-stage Liver Disease (MELD) score in a retrospective single time point analysis of MR images. Whilst delayed phase peribiliary hyperenhancement showed weak correlation with Mayo risk score as described earlier, Ni Mhuircheatiagh et al[72] reported that the presence and extent of arterial phase peribiliary hyperenhancement on MRI was associated with a higher Mayo risk score in a cohort of 60 PSC patients. They postulated that this is potentially a marker of active biliary inflammation and poorer prognosis. Bookwalter et al[73] retrospectively reviewed MRI that included dynamic contrast enhanced sequences, MRCP and MRE of 55 PSC patients to examine the relationship between liver parenchymal changes, biliary features and liver stiffness at a segmental, lobar and global level. They found weak correlation at segmental level between liver stiffness and liver parenchymal signal changes and ductal strictures. However, they found no significant correlation between the presence and absence of periductal enhancement in any of the three contrast enhanced phases with Mayo risk score or MELD score.
A dominant bile duct stricture in PSC is defined as a stricture less than 1.5 mm diameter in the common bile duct, or less than 1 mm in the left or right main hepatic ducts on cholangiography[7]. However, there are currently no validated criteria for MR definition of a dominant stricture. Deterioration in clinical and biochemical parameters prompts evaluation for a dominant stricture and/or cholangiocarcinoma on MRI/MRCP. The presence of a dominant stricture either at diagnosis or follow-up is associated with an increased risk of developing cholangiocarcinoma and mortality[74,75].
Over one third of cholangiocarcinoma cases were detected within the first year following PSC diagnosis in a retrospective, international, observational cohort study involving 594 PSC patients[13]. This is likely due to length-time bias and detection of cholangiocarcinoma only when it becomes clinically overt. Serum tumour marker carbohydrate antigen (CA)19-9 is widely used in surveillance strategy but it lacks both sensitivity and specificity for the detection of cholangiocarcinoma[76,77]. The cholangiocarcinoma is usually too advanced for curable treatment by the time CA19-9 becomes persistently elevated[78]. Annual MRI/MRCP as a surveillance strategy is often performed, but with limited proven benefit. Nevertheless, the current recommendation for cholangiocarcinoma surveillance is annual CA19-9 and MRI/MRCP[79]. There is an unmet need for earlier detection of cholangiocarcinoma and closer monitoring of newly diagnosed patients.
A population-based study of long-term IBD patients in Norway reported that 8.1% of 322 patients had MRCP lesions indicating PSC, a 3-fold higher prevalence than detected clinically before MRCP screening. Nearly two-thirds of these detected cases had ‘subclinical’ PSC with mild changes on cholangiography and no biochemical abnormalities[80]. A prospective controlled UK study demonstrated 14% of 51 patients with extensive ulcerative colitis and normal liver biochemistry had biliary abnormalities suggestive of PSC on MRCP, and over long-term (10-year) follow-up, one-third developed abnormal liver biochemistry, one-fifth developed progressive bile duct disease and over half developed malignancy, including two biliary and one colorectal carcinoma[81].
DW-MRI manipulates the altered diffusion properties of water protons in fibrotic tissue and allows assessment of liver fibrosis[82]. Addition of the short sequence to routine MRI/MRCP enables whole liver assessment of fibrosis distribution, particularly useful in a patchy disease such as PSC[32,83]. Since TE and MRE have shown better diagnostic accuracy for the staging of liver fibrosis[83,84], DWI has fallen out of favour but is still performed for better characterisation of lesions involving the liver parenchyma. In a recent prospective study involving 47 PSC patients, DWI-MRI performed better than dynamic contrast-enhanced MRI (DCE-MRI) in detecting and staging liver fibrosis using VCTE as the reference standard[85].
Administration of hepatocyte-specific contrast agents such as Gd-BOPTA (gadobenate dimeglumine) and Gd-EOB-DTPA (gadoxetate disodium) allows assessment of liver function by analysing the liver uptake and elimination of contrast. Several studies have attempted to quantify liver parenchymal changes with the administration of a contrast agent.
Ringe et al[86] demonstrated that hepatobiliary excretion of hepatocyte-specific contrast is significantly reduced in patients with PSC when compared to normal controls and correlated with bilirubin levels in PSC. Noren et al[87] quantitatively compared hepatocyte-specific contrast uptake with histopathological stage of fibrosis in a prospective study involving 38 patients with compensated chronic liver diseases of varying aetiology. They demonstrated that quantitative measurement of signal intensities using DCE-MRI was able to distinguish advanced fibrosis (F3-4) from no and moderate fibrosis (F0-2). Nilsson et al[88] developed a non-invasive imaging-based method using DCE-MRI to assess liver function at the segmental and global level, and showed significantly heterogeneity in the liver parenchyma of PSC patients compared to controls. This small study (involving PSC patients with mild disease) reported correlation between MRI-derived liver function indices and disease severity using Mayo risk score. Segmental liver function correlated with level of downstream biliary obstruction.
Hinrichs et al[89] reported that reduction in T1 relaxation time after hepatocyte-specific contrast administration correlated with liver biochemistry tests, MELD and Mayo risk score. They proposed that global liver function could be non-invasively assessed using this specific T1 mapping sequence technique in PSC. Nolz et al[90] performed retrospective quantitative analysis of liver parenchymal enhancement in T1-weighted MRCP images in a small cohort of PSC patients, and calculated the difference in signal intensity (SI) ratio between the hepatobiliary phase and unenhanced parenchyma [termed relative enhancement (RE)]. They demonstrated significant reduction in RE in localised areas of impaired liver parenchyma in comparison to normal areas, thus allowing regional functional assessment.
RE = [(hepatobiliary SI - unenhanced SI)/unenhanced SI × 100]
Keller et al[91] adapted the above technique to MRI scans performed with extracellular gadolinium-based instead of hepatocyte-specific contrast. They retrospectively reviewed scans and liver biopsies of 40 PSC patients to evaluate the utility of several quantitative MRI-derived parameters as markers of liver inflammation and fibrosis (LIF). Relative liver enhancement (RLE) in the delayed phase of T1-weighted MRI was shown to strongly correlate with stage of liver fibrosis. The same group also demonstrated an increased RLE within T2 hyperintense areas in the liver parenchyma on T2-weighted MRI and postulated that this could be early changes of patchy inflammation[92]. Schulze et al[93] calculated RLE in the hepatobiliary MRI phase in a prospective study using hepatocyte-specific contrast agent and evaluated its role as a prognostic marker. Moderate correlation was demonstrated with serum markers (ALP, albumin, bilirubin, INR) and prognostic scorings systems (MELD, Mayo risk score, Amsterdam-Oxford). They proposed a cut-off RLE value that predicted clinical endpoints with low sensitivity (74%) and reasonable specificity (94%), which remains to be externally validated.
LiverMultiscan™ (Perspectum Diagnostics, Oxford, United Kingdom) is a software product that enables post-processing of liver MRI using T1 and T2* maps[94,95]. In a small proof of principle study, the LIF score derived from the iron corrected T1 (cT1) measurements, has been shown to strongly correlate with clinical outcomes in patients with chronic liver disease of mixed aetiologies[96]. This technique looks promising for the evaluation of patients with non-alcoholic fatty liver disease[97].
More recently, Arndtz et al[98,99] reported the distribution of imaging metrics derived from quantitative maps of T1, T2* and cT1 of LiverMultiscan™ in autoimmune hepatitis (AIH), primary biliary cholangitis (PBC) and PSC. Using a machine-learning technique to analyse the skewness and kurtosis of the distribution as well as local regional variance, they demonstrated that addition of imaging metrics to serum ALT or ALP performed slightly better than serum ALT or ALP alone in disease differentiation between parenchymal liver disease (62 AIH patients) and biliary liver disease (124 PSC and PBC patients).
Acquisition and 3D image reconstruction protocols still vary significantly across centres and there is no standard model for interpreting MRCP data. Current clinical utility of MRI/MRCP allows only qualitative assessment of the bile ducts and liver parenchyma, and is therefore susceptible to subjectivity in interpretation. Assessment of the distal common bile duct and subtle changes in the smaller peripheral intrahepatic bile ducts still remains a challenge despite the use of modern 3 Tesla (T) MRI scanners[22]. The position statement from the International PSC Study Group outlines areas of unmet need for imaging techniques in PSC, including (1) early detection of disease; (2) the determination of disease stage, activity and prognosis; (3) the assessment of treatment response; (4) a clinically meaningful definition of dominant bile duct stenosis; and (5) the early detection of cholangiocarcinoma.
The role of MRI/MRCP in establishing the diagnosis of PSC is well documented and has long superseded ERCP as the gold standard for obtaining cholangiography. Disease staging is based on the severity of the liver fibrosis component of PSC, which has prognostic implications. MRE appears to be a promising technique that generates a liver stiffness map of the whole liver for assessment of patchy liver fibrosis. However, there have been no comparative studies between MRE and other surrogate markers of fibrosis in PSC. Whilst there is some exciting work published on MR quantitative methods involving the liver parenchyma in PSC, there has been surprisingly little advancement in the last three decades on quantitative methods involving the bile ducts. Published studies to date have only proposed cholangiography-based scoring systems derived from interpretation of qualitative descriptors by two specialist radiologists assessing the morphological appearance of bile ducts and liver parenchyma. This method is highly variable even among experts and therefore limits its inclusion into reliable prognostic models. MRI/MRCP shows promising potential for prediction of disease course and clinical endpoints in PSC as MR techniques evolve towards ‘quantifying’ the disease. However, further development and validation of objective and reproducible MR-based parameters are needed before it can establish its role as an imaging biomarker in PSC.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: United Kingdom
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Ahmed M, Arrive L, Chow WK, Kitamura K S- Editor: Ma RY L- Editor: A E- Editor: Yin SY
| 1. | Hirschfield GM, Karlsen TH, Lindor KD, Adams DH. Primary sclerosing cholangitis. Lancet. 2013;382:1587-1599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 398] [Cited by in RCA: 426] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 2. | Boonstra K, Weersma RK, van Erpecum KJ, Rauws EA, Spanier BW, Poen AC, van Nieuwkerk KM, Drenth JP, Witteman BJ, Tuynman HA, Naber AH, Kingma PJ, van Buuren HR, van Hoek B, Vleggaar FP, van Geloven N, Beuers U, Ponsioen CY; EpiPSCPBC Study Group. Population-based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology. 2013;58:2045-2055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 505] [Article Influence: 42.1] [Reference Citation Analysis (0)] |
| 3. | Boonstra K, Beuers U, Ponsioen CY. Epidemiology of primary sclerosing cholangitis and primary biliary cirrhosis: a systematic review. J Hepatol. 2012;56:1181-1188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 499] [Cited by in RCA: 441] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 4. | Schrumpf E, Elgjo K, Fausa O, Gjone E, Kolmannskog F, Ritland S. Sclerosing cholangitis in ulcerative colitis. Scand J Gastroenterol. 1980;15:689-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 126] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Chapman RW, Arborgh BA, Rhodes JM, Summerfield JA, Dick R, Scheuer PJ, Sherlock S. Primary sclerosing cholangitis: a review of its clinical features, cholangiography, and hepatic histology. Gut. 1980;21:870-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 522] [Cited by in RCA: 513] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 6. | Wiesner RH, LaRusso NF. Clinicopathologic features of the syndrome of primary sclerosing cholangitis. Gastroenterology. 1980;79:200-206. [PubMed] |
| 7. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of cholestatic liver diseases. J Hepatol. 2009;51:237-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1382] [Cited by in RCA: 1204] [Article Influence: 75.3] [Reference Citation Analysis (1)] |
| 8. | Angulo P, Maor-Kendler Y, Lindor KD. Small-duct primary sclerosing cholangitis: a long-term follow-up study. Hepatology. 2002;35:1494-1500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 132] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 9. | Kaya M, Angulo P, Lindor KD. Overlap of autoimmune hepatitis and primary sclerosing cholangitis: an evaluation of a modified scoring system. J Hepatol. 2000;33:537-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 90] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 10. | Mendes FD, Jorgensen R, Keach J, Katzmann JA, Smyrk T, Donlinger J, Chari S, Lindor KD. Elevated serum IgG4 concentration in patients with primary sclerosing cholangitis. Am J Gastroenterol. 2006;101:2070-2075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 231] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 11. | Björnsson E, Chari S, Silveira M, Gossard A, Takahashi N, Smyrk T, Lindor K. Primary sclerosing cholangitis associated with elevated immunoglobulin G4: clinical characteristics and response to therapy. Am J Ther. 2011;18:198-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 82] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 12. | Lazaridis KN, LaRusso NF. Primary Sclerosing Cholangitis. N Engl J Med. 2016;375:1161-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 361] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 13. | Weismüller TJ, Trivedi PJ, Bergquist A, Imam M, Lenzen H, Ponsioen CY, Holm K, Gotthardt D, Färkkilä MA, Marschall HU, Thorburn D, Weersma RK, Fevery J, Mueller T, Chazouillères O, Schulze K, Lazaridis KN, Almer S, Pereira SP, Levy C, Mason A, Naess S, Bowlus CL, Floreani A, Halilbasic E, Yimam KK, Milkiewicz P, Beuers U, Huynh DK, Pares A, Manser CN, Dalekos GN, Eksteen B, Invernizzi P, Berg CP, Kirchner GI, Sarrazin C, Zimmer V, Fabris L, Braun F, Marzioni M, Juran BD, Said K, Rupp C, Jokelainen K, Benito de Valle M, Saffioti F, Cheung A, Trauner M, Schramm C, Chapman RW, Karlsen TH, Schrumpf E, Strassburg CP, Manns MP, Lindor KD, Hirschfield GM, Hansen BE, Boberg KM; International PSC Study Group. Patient Age, Sex, and Inflammatory Bowel Disease Phenotype Associate With Course of Primary Sclerosing Cholangitis. Gastroenterology. 2017;152:1975-1984.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 364] [Article Influence: 45.5] [Reference Citation Analysis (1)] |
| 14. | Hoffmann C. Verschluß der Gallenwege durch Verdickung der Wandungen. Arch Pathol Anat Physiol Klin Med 1867; 49: 206-215. . |
| 15. | Delbet M. Retrecissement due choledoque: Cholecystoduo-denostomie. Bull Mem Soc Nat Chir. 1924;50:1144. |
| 16. | Lafourcade M. Deux observations d'obliteration cicatricelle de choledoque. Bull Mem Soc Nat Chir. 1925;50. |
| 17. | Miller RT. Benign Stricture of the Bile Ducts. Ann Surg. 1927;86:296-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 18. | Schwartz SI, Dale WA. Primary sclerosing cholangitis; review and report of six cases. AMA Arch Surg. 1958;77:439-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 44] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Chen LY, Goldberg HI. Sclerosing cholangitis: broad spectrum of radiographic features. Gastrointest Radiol. 1984;9:39-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 33] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Wallner BK, Schumacher KA, Weidenmaier W, Friedrich JM. Dilated biliary tract: evaluation with MR cholangiography with a T2-weighted contrast-enhanced fast sequence. Radiology. 1991;181:805-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 209] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 21. | Arrivé L, Coudray C, Azizi L, Lewin M, Hoeffel C, Monnier-Cholley L, Lacombe C, Vautier S, Poupon J, Tubiana JM. [Pineapple juice as a negative oral contrast agent in magnetic resonance cholangiopancreatography]. J Radiol. 2007;88:1689-1694. [PubMed] |
| 22. | Schramm C, Eaton J, Ringe KI, Venkatesh S, Yamamura J, MRI working group of the IPSCSG. Recommendations on the use of magnetic resonance imaging in PSC-A position statement from the International PSC Study Group. Hepatology. 2017;66:1675-1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 23. | Chandrasekhara V, Khashab MA, Muthusamy VR, Acosta RD, Agrawal D, Bruining DH, Eloubeidi MA, Fanelli RD, Faulx AL, Gurudu SR, Kothari S, Lightdale JR, Qumseya BJ, Shaukat A, Wang A, Wani SB, Yang J, DeWitt JM. Adverse events associated with ERCP. Gastrointest Endosc. 2017;85:32-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 536] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 24. | Dave M, Elmunzer BJ, Dwamena BA, Higgins PD. Primary sclerosing cholangitis: meta-analysis of diagnostic performance of MR cholangiopancreatography. Radiology. 2010;256:387-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 156] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 25. | Chapman R, Fevery J, Kalloo A, Nagorney DM, Boberg KM, Shneider B, Gores GJ; American Association for the Study of Liver Diseases. Diagnosis and management of primary sclerosing cholangitis. Hepatology. 2010;51:660-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 835] [Article Influence: 55.7] [Reference Citation Analysis (0)] |
| 26. | Talwalkar JA, Angulo P, Johnson CD, Petersen BT, Lindor KD. Cost-minimization analysis of MRC versus ERCP for the diagnosis of primary sclerosing cholangitis. Hepatology. 2004;40:39-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 64] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 27. | Meagher S, Yusoff I, Kennedy W, Martel M, Adam V, Barkun A. The roles of magnetic resonance and endoscopic retrograde cholangiopancreatography (MRCP and ERCP) in the diagnosis of patients with suspected sclerosing cholangitis: a cost-effectiveness analysis. Endoscopy. 2007;39:222-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | Fulcher AS, Turner MA, Franklin KJ, Shiffman ML, Sterling RK, Luketic VA, Sanyal AJ. Primary sclerosing cholangitis: evaluation with MR cholangiography-a case-control study. Radiology. 2000;215:71-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 108] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 29. | Angulo P, Pearce DH, Johnson CD, Henry JJ, LaRusso NF, Petersen BT, Lindor KD. Magnetic resonance cholangiography in patients with biliary disease: its role in primary sclerosing cholangitis. J Hepatol. 2000;33:520-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 69] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Parlak E, Dişibeyaz S, Odemiş B, Köksal AŞ, Oğuz D, Ciçek B, Saşmaz N, Sahin B. Demonstration of retraction of the main papilla toward the biliary system in patients with primary sclerosing cholangitis with magnetic resonance cholangiopancreatography. Dig Endosc. 2012;24:384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 31. | Kovač JD, Weber MA. Primary Biliary Cirrhosis and Primary Sclerosing Cholangitis: an Update on MR Imaging Findings with Recent Developments. J Gastrointestin Liver Dis. 2016;25:517-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 32. | Kovač JD, Ješić R, Stanisavljević D, Kovač B, Maksimovic R. MR imaging of primary sclerosing cholangitis: additional value of diffusion-weighted imaging and ADC measurement. Acta Radiol. 2013;54:242-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 33. | Tokala A, Khalili K, Menezes R, Hirschfield G, Jhaveri KS. Comparative MRI analysis of morphologic patterns of bile duct disease in IgG4-related systemic disease versus primary sclerosing cholangitis. AJR Am J Roentgenol. 2014;202:536-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 34. | Ohara H, Okazaki K, Tsubouchi H, Inui K, Kawa S, Kamisawa T, Tazuma S, Uchida K, Hirano K, Yoshida H, Nishino T, Ko SB, Mizuno N, Hamano H, Kanno A, Notohara K, Hasebe O, Nakazawa T, Nakanuma Y, Takikawa H; Research Committee of IgG4-related Diseases; Research Committee of Intractable Diseases of Liver and Biliary Tract; Ministry of Health, Labor and Welfare, Japan; Japan Biliary Association. Clinical diagnostic criteria of IgG4-related sclerosing cholangitis 2012. J Hepatobiliary Pancreat Sci. 2012;19:536-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 252] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 35. | Wiesner RH, Grambsch PM, Dickson ER, Ludwig J, MacCarty RL, Hunter EB, Fleming TR, Fisher LD, Beaver SJ, LaRusso NF. Primary sclerosing cholangitis: natural history, prognostic factors and survival analysis. Hepatology. 1989;10:430-436. [PubMed] |
| 36. | de Vries EM, Verheij J, Hubscher SG, Leeflang MM, Boonstra K, Beuers U, Ponsioen CY. Applicability and prognostic value of histologic scoring systems in primary sclerosing cholangitis. J Hepatol. 2015;63:1212-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 37. | de Vries EM, de Krijger M, Färkkilä M, Arola J, Schirmacher P, Gotthardt D, Goeppert B, Trivedi PJ, Hirschfield GM, Ytting H, Vainer B, Buuren HR, Biermann K, Harms MH, Chazouilleres O, Wendum D, Kemgang AD, Chapman RW, Wang LM, Williamson KD, Gouw AS, Paradis V, Sempoux C, Beuers U, Hübscher SG, Verheij J, Ponsioen CY. Validation of the prognostic value of histologic scoring systems in primary sclerosing cholangitis: An international cohort study. Hepatology. 2017;65:907-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 73] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 38. | Olsson R, Hägerstrand I, Broomé U, Danielsson A, Järnerot G, Lööf L, Prytz H, Rydén BO, Wallerstedt S. Sampling variability of percutaneous liver biopsy in primary sclerosing cholangitis. J Clin Pathol. 1995;48:933-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 90] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 39. | Vesterhus M, Hov JR, Holm A, Schrumpf E, Nygård S, Godang K, Andersen IM, Naess S, Thorburn D, Saffioti F, Vatn M, Gilja OH, Lund-Johansen F, Syversveen T, Brabrand K, Parés A, Ponsioen CY, Pinzani M, Färkkilä M, Moum B, Ueland T, Røsjø H, Rosenberg W, Boberg KM, Karlsen TH. Enhanced liver fibrosis score predicts transplant-free survival in primary sclerosing cholangitis. Hepatology. 2015;62:188-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 100] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 40. | de Vries EMG, Färkkilä M, Milkiewicz P, Hov JR, Eksteen B, Thorburn D, Chazouillères O, Pares A, Nygård S, Gilja OH, Wunsch E, Invernizzi P, Carbone M, Bernuzzi F, Boberg KM, Røsjø H, Rosenberg W, Beuers UH, Ponsioen CY, Karlsen TH, Vesterhus M. Enhanced liver fibrosis test predicts transplant-free survival in primary sclerosing cholangitis, a multi-centre study. Liver Int. 2017;37:1554-1561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 41. | Singh S, Fujii LL, Murad MH, Wang Z, Asrani SK, Ehman RL, Kamath PS, Talwalkar JA. Liver stiffness is associated with risk of decompensation, liver cancer, and death in patients with chronic liver diseases: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2013;11:1573-1584.e1-2; quiz e88-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 239] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 42. | Singh S, Venkatesh SK, Wang Z, Miller FH, Motosugi U, Low RN, Hassanein T, Asbach P, Godfrey EM, Yin M, Chen J, Keaveny AP, Bridges M, Bohte A, Murad MH, Lomas DJ, Talwalkar JA, Ehman RL. Diagnostic performance of magnetic resonance elastography in staging liver fibrosis: a systematic review and meta-analysis of individual participant data. Clin Gastroenterol Hepatol. 2015;13:440-451.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 417] [Article Influence: 41.7] [Reference Citation Analysis (0)] |
| 43. | Corpechot C, Gaouar F, El Naggar A, Kemgang A, Wendum D, Poupon R, Carrat F, Chazouillères O. Baseline values and changes in liver stiffness measured by transient elastography are associated with severity of fibrosis and outcomes of patients with primary sclerosing cholangitis. Gastroenterology. 2014;146:970-9; quiz e15-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 207] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 44. | Ehlken H, Wroblewski R, Corpechot C, Arrivé L, Rieger T, Hartl J, Lezius S, Hübener P, Schulze K, Zenouzi R, Sebode M, Peiseler M, Denzer UW, Quaas A, Weiler-Normann C, Lohse AW, Chazouilleres O, Schramm C. Validation of Transient Elastography and Comparison with Spleen Length Measurement for Staging of Fibrosis and Clinical Prognosis in Primary Sclerosing Cholangitis. PLoS One. 2016;11:e0164224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 45. | Ehlken H, Lohse AW, Schramm C. Transient elastography in primary sclerosing cholangitis-the value as a prognostic factor and limitations. Gastroenterology. 2014;147:542-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 46. | Millonig G, Reimann FM, Friedrich S, Fonouni H, Mehrabi A, Büchler MW, Seitz HK, Mueller S. Extrahepatic cholestasis increases liver stiffness (FibroScan) irrespective of fibrosis. Hepatology. 2008;48:1718-1723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 463] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 47. | Eaton JE, Dzyubak B, Venkatesh SK, Smyrk TC, Gores GJ, Ehman RL, LaRusso NF, Gossard AA, Lazaridis KN. Performance of magnetic resonance elastography in primary sclerosing cholangitis. J Gastroenterol Hepatol. 2016;31:1184-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 77] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 48. | Shire NJ, Yin M, Chen J, Railkar RA, Fox-Bosetti S, Johnson SM, Beals CR, Dardzinski BJ, Sanderson SO, Talwalkar JA, Ehman RL. Test-retest repeatability of MR elastography for noninvasive liver fibrosis assessment in hepatitis C. J Magn Reson Imaging. 2011;34:947-955. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 49. | Dulai PS, Sirlin CB, Loomba R. MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: Clinical trials to clinical practice. J Hepatol. 2016;65:1006-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 265] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 50. | Hsu C, Caussy C, Imajo K, Chen J, Singh S, Kaulback K, Le MD, Hooker J, Tu X, Bettencourt R, Yin M, Sirlin CB, Ehman RL, Nakajima A, Loomba R. Magnetic Resonance vs Transient Elastography Analysis of Patients With Nonalcoholic Fatty Liver Disease: A Systematic Review and Pooled Analysis of Individual Participants. Clin Gastroenterol Hepatol. 2018;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 301] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 51. | Jhaveri KS, Hosseini-Nik H, Sadoughi N, Janssen H, Feld JJ, Fischer S, Menezes R, Cheung AC. The development and validation of magnetic resonance elastography for fibrosis staging in primary sclerosing cholangitis. Eur Radiol. 2019;29:1039-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 52. | PSC Support. PSC Support Patient Unmet Needs and Research Implications: pscsupport.org.uk; 2016 Available from: http://www.pscsupport.org.uk/sites/default/files/files/PSC%20Support%20Patient%20Survey%20Results(2).pdf. . |
| 53. | de Vries EM, Wang J, Leeflang MM, Boonstra K, Weersma RK, Beuers UH, Geskus RB, Ponsioen CY. Alkaline phosphatase at diagnosis of primary sclerosing cholangitis and 1 year later: evaluation of prognostic value. Liver Int. 2016;36:1867-1875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 54. | Rupp C, Rössler A, Halibasic E, Sauer P, Weiss KH, Friedrich K, Wannhoff A, Stiehl A, Stremmel W, Trauner M, Gotthardt DN. Reduction in alkaline phosphatase is associated with longer survival in primary sclerosing cholangitis, independent of dominant stenosis. Aliment Pharmacol Ther. 2014;40:1292-1301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 55. | Lindström L, Hultcrantz R, Boberg KM, Friis-Liby I, Bergquist A. Association between reduced levels of alkaline phosphatase and survival times of patients with primary sclerosing cholangitis. Clin Gastroenterol Hepatol. 2013;11:841-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 56. | Hilscher M, Enders FB, Carey EJ, Lindor KD, Tabibian JH. Alkaline phosphatase normalization is a biomarker of improved survival in primary sclerosing cholangitis. Ann Hepatol. 2016;15:246-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 57. | Al Mamari S, Djordjevic J, Halliday JS, Chapman RW. Improvement of serum alkaline phosphatase to <1.5 upper limit of normal predicts better outcome and reduced risk of cholangiocarcinoma in primary sclerosing cholangitis. J Hepatol. 2013;58:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 146] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 58. | Balasubramaniam K, Wiesner RH, LaRusso NF. Primary sclerosing cholangitis with normal serum alkaline phosphatase activity. Gastroenterology. 1988;95:1395-1398. [PubMed] |
| 59. | Kim WR, Therneau TM, Wiesner RH, Poterucha JJ, Benson JT, Malinchoc M, LaRusso NF, Lindor KD, Dickson ER. A revised natural history model for primary sclerosing cholangitis. Mayo Clin Proc. 2000;75:688-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 60. | de Vries EM, Wang J, Williamson KD, Leeflang MM, Boonstra K, Weersma RK, Beuers U, Chapman RW, Geskus RB, Ponsioen CY. A novel prognostic model for transplant-free survival in primary sclerosing cholangitis. Gut. 2018;67:1864-1869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 61. | Eaton JE, Vesterhus M, McCauley BM, Atkinson EJ, Schlicht EM, Juran BD, Gossard AA, LaRusso NF, Gores GJ, Karlsen TH, Lazaridis KN. Primary Sclerosing Cholangitis Risk Estimate Tool (PREsTo) Predicts Outcomes of the Disease: A Derivation and Validation Study Using Machine Learning. Hepatology. 2018;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 62. | Zenouzi R, Liwinski T, Yamamura J, Weiler-Normann C, Sebode M, Keller S, Lohse AW, Schramm C; International PSC Study Group (IPSCSG). Follow-up magnetic resonance imaging/3D-magnetic resonance cholangiopancreatography in patients with primary sclerosing cholangitis: challenging for experts to interpret. Aliment Pharmacol Ther. 2018;48:169-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 63. | Craig DA, MacCarty RL, Wiesner RH, Grambsch PM, LaRusso NF. Primary sclerosing cholangitis: value of cholangiography in determining the prognosis. AJR Am J Roentgenol. 1991;157:959-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 51] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 64. | Olsson RG, Asztély MS. Prognostic value of cholangiography in primary sclerosing cholangitis. Eur J Gastroenterol Hepatol. 1995;7:251-254. [PubMed] |
| 65. | Ponsioen CY, Vrouenraets SM, Prawirodirdjo W, Rajaram R, Rauws EA, Mulder CJ, Reitsma JB, Heisterkamp SH, Tytgat GN. Natural history of primary sclerosing cholangitis and prognostic value of cholangiography in a Dutch population. Gut. 2002;51:562-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 198] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 66. | Majoie CB, Reeders JW, Sanders JB, Huibregtse K, Jansen PL. Primary sclerosing cholangitis: a modified classification of cholangiographic findings. AJR Am J Roentgenol. 1991;157:495-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 54] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 67. | Petrovic BD, Nikolaidis P, Hammond NA, Martin JA, Petrovic PV, Desai PM, Miller FH. Correlation between findings on MRCP and gadolinium-enhanced MR of the liver and a survival model for primary sclerosing cholangitis. Dig Dis Sci. 2007;52:3499-3506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 68. | Tenca A, Mustonen H, Lind K, Lantto E, Kolho KL, Boyd S, Arola J, Jokelainen K, Färkkilä M. The role of magnetic resonance imaging and endoscopic retrograde cholangiography in the evaluation of disease activity and severity in primary sclerosing cholangitis. Liver Int. 2018;38:2329-2339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 69. | Ruiz A, Lemoinne S, Carrat F, Corpechot C, Chazouillères O, Arrivé L. Radiologic course of primary sclerosing cholangitis: assessment by three-dimensional magnetic resonance cholangiography and predictive features of progression. Hepatology. 2014;59:242-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 70. | Kitzing YX, Whitley SA, Upponi SS, Srivastava B, Alexander GJ, Lomas DJ. Association between progressive hepatic morphology changes on serial MR imaging and clinical outcome in primary sclerosing cholangitis. J Med Imaging Radiat Oncol. 2017;61:636-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 71. | Bader TR, Beavers KL, Semelka RC. MR imaging features of primary sclerosing cholangitis: patterns of cirrhosis in relationship to clinical severity of disease. Radiology. 2003;226:675-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 59] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 72. | Ni Mhuircheartaigh JM, Lee KS, Curry MP, Pedrosa I, Mortele KJ. Early Peribiliary Hyperenhancement on MRI in Patients with Primary Sclerosing Cholangitis: Significance and Association with the Mayo Risk Score. Abdom Radiol (NY). 2017;42:152-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 73. | Bookwalter CA, Venkatesh SK, Eaton JE, Smyrk TD, Ehman RL. MR elastography in primary sclerosing cholangitis: correlating liver stiffness with bile duct strictures and parenchymal changes. Abdom Radiol (NY). 2018;43:3260-3270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 74. | Chapman MH, Webster GJ, Bannoo S, Johnson GJ, Wittmann J, Pereira SP. Cholangiocarcinoma and dominant strictures in patients with primary sclerosing cholangitis: a 25-year single-centre experience. Eur J Gastroenterol Hepatol. 2012;24:1051-1058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 152] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 75. | Rudolph G, Gotthardt D, Klöters-Plachky P, Kulaksiz H, Rost D, Stiehl A. Influence of dominant bile duct stenoses and biliary infections on outcome in primary sclerosing cholangitis. J Hepatol. 2009;51:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 108] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 76. | Petersen-Benz C, Stiehl A. Impact of dominant stenoses on the serum level of the tumor marker CA19-9 in patients with primary sclerosing cholangitis. Z Gastroenterol. 2005;43:587-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 77. | Levy MJ, Baron TH, Clayton AC, Enders FB, Gostout CJ, Halling KC, Kipp BR, Petersen BT, Roberts LR, Rumalla A, Sebo TJ, Topazian MD, Wiersema MJ, Gores GJ. Prospective evaluation of advanced molecular markers and imaging techniques in patients with indeterminate bile duct strictures. Am J Gastroenterol. 2008;103:1263-1273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 132] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 78. | Chapman RW, Williamson KD. Are Dominant Strictures in Primary Sclerosing Cholangitis a Risk Factor for Cholangiocarcinoma? Curr Hepatol Rep. 2017;16:124-129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 79. | Razumilava N, Gores GJ, Lindor KD. Cancer surveillance in patients with primary sclerosing cholangitis. Hepatology. 2011;54:1842-1852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 207] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 80. | Lunder AK, Hov JR, Borthne A, Gleditsch J, Johannesen G, Tveit K, Viktil E, Henriksen M, Hovde Ø, Huppertz-Hauss G, Høie O, Høivik ML, Monstad I, Solberg IC, Jahnsen J, Karlsen TH, Moum B, Vatn M, Negård A. Prevalence of Sclerosing Cholangitis Detected by Magnetic Resonance Cholangiography in Patients With Long-term Inflammatory Bowel Disease. Gastroenterology. 2016;151:660-669.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 135] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 81. | Culver EL, Bungay H, Betts M, Manganis C, Buchel O, Shrumpf E, Cummings JF, Keshav S, Travis SP, Chapman RW. Complications of primary sclerosing cholangitis in patients with ulcerative colitis and normal liver function tests: a prospective magnetic resonance cholangiographic study with long-term follow-up. Journal of Crohn's and Colitis. 2017;11 Suppl 1:S215. [DOI] [Full Text] |
| 82. | Taouli B, Koh DM. Diffusion-weighted MR imaging of the liver. Radiology. 2010;254:47-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 623] [Cited by in RCA: 609] [Article Influence: 40.6] [Reference Citation Analysis (2)] |
| 83. | Kovač JD, Daković M, Stanisavljević D, Alempijević T, Ješić R, Seferović P, Maksimović R. Diffusion-weighted MRI versus transient elastography in quantification of liver fibrosis in patients with chronic cholestatic liver diseases. Eur J Radiol. 2012;81:2500-2506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 84. | Wang QB, Zhu H, Liu HL, Zhang B. Performance of magnetic resonance elastography and diffusion-weighted imaging for the staging of hepatic fibrosis: A meta-analysis. Hepatology. 2012;56:239-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 195] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 85. | Keller S, Sedlacik J, Schuler T, Buchert R, Avanesov M, Zenouzi R, Lohse AW, Kooijman H, Fiehler J, Schramm C, Yamamura J. Prospective comparison of diffusion-weighted MRI and dynamic Gd-EOB-DTPA-enhanced MRI for detection and staging of hepatic fibrosis in primary sclerosing cholangitis. Eur Radiol. 2019;29:818-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 86. | Ringe KI, Hinrichs J, Merkle EM, Weismüller TJ, Wacker F, Meyer BC. Gadoxetate disodium in patients with primary sclerosing cholangitis: an analysis of hepatobiliary contrast excretion. J Magn Reson Imaging. 2014;40:106-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 87. | Norén B, Forsgren MF, Dahlqvist Leinhard O, Dahlström N, Kihlberg J, Romu T, Kechagias S, Almer S, Smedby Ö, Lundberg P. Separation of advanced from mild hepatic fibrosis by quantification of the hepatobiliary uptake of Gd-EOB-DTPA. Eur Radiol. 2013;23:174-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 88. | Nilsson H, Blomqvist L, Douglas L, Nordell A, Jacobsson H, Hagen K, Bergquist A, Jonas E. Dynamic gadoxetate-enhanced MRI for the assessment of total and segmental liver function and volume in primary sclerosing cholangitis. J Magn Reson Imaging. 2014;39:879-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Hinrichs H, Hinrichs JB, Gutberlet M, Lenzen H, Raatschen HJ, Wacker F, Ringe KI. Functional gadoxetate disodium-enhanced MRI in patients with primary sclerosing cholangitis (PSC). Eur Radiol. 2016;26:1116-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 90. | Nolz R, Asenbaum U, Schoder M, Wibmer A, Einspieler H, Prusa AM, Peck-Radosavljevic M, Ba-Ssalamah A. Diagnostic workup of primary sclerosing cholangitis: the benefit of adding gadoxetic acid-enhanced T1-weighted magnetic resonance cholangiography to conventional T2-weighted magnetic resonance cholangiography. Clin Radiol. 2014;69:499-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 91. | Keller S, Aigner A, Zenouzi R, Kim AC, Meijer A, Weidemann SA, Krech T, Lohse AW, Adam G, Schramm C, Yamamura J. Association of gadolinium-enhanced magnetic resonance imaging with hepatic fibrosis and inflammation in primary sclerosing cholangitis. PLoS One. 2018;13:e0193929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 92. | Keller S, Venkatesh SK, Avanesov M, Weinrich JM, Zenouzi R, Schramm C, Adam G, Yamamura J. Gadolinium-based relative contrast enhancement in primary sclerosing cholangitis: additional benefit for clinicians? Clin Radiol. 2018;73:677.e1-677.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 93. | Schulze J, Lenzen H, Hinrichs JB, Ringe B, Manns MP, Wacker F, Ringe KI. An Imaging Biomarker for Assessing Hepatic Function in Patients With Primary Sclerosing Cholangitis. Clin Gastroenterol Hepatol. 2019;17:192-199.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 94. | Banerjee R, Pavlides M, Tunnicliffe EM, Piechnik SK, Sarania N, Philips R, Collier JD, Booth JC, Schneider JE, Wang LM, Delaney DW, Fleming KA, Robson MD, Barnes E, Neubauer S. Multiparametric magnetic resonance for the non-invasive diagnosis of liver disease. J Hepatol. 2014;60:69-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 327] [Cited by in RCA: 359] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 95. | Tunnicliffe EM, Banerjee R, Pavlides M, Neubauer S, Robson MD. A model for hepatic fibrosis: the competing effects of cell loss and iron on shortened modified Look-Locker inversion recovery T1 (shMOLLI-T1) in the liver. J Magn Reson Imaging. 2017;45:450-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 96. | Pavlides M, Banerjee R, Sellwood J, Kelly CJ, Robson MD, Booth JC, Collier J, Neubauer S, Barnes E. Multiparametric magnetic resonance imaging predicts clinical outcomes in patients with chronic liver disease. J Hepatol. 2016;64:308-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 170] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 97. | Pavlides M, Banerjee R, Tunnicliffe EM, Kelly C, Collier J, Wang LM, Fleming KA, Cobbold JF, Robson MD, Neubauer S, Barnes E. Multiparametric magnetic resonance imaging for the assessment of non-alcoholic fatty liver disease severity. Liver Int. 2017;37:1065-1073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 98. | Arndtz K, Hodson J, Eddowes PJ, Kelly MD, Green D, Banerjee R, Neubauer S, Hirschfield GM. Multi-parametric MRI imaging correlates with clinically meaningful surrogates of disease activity in autoimmune hepatitis. Hepatology. 2017;66:188a-a. |
| 99. | Arndtz K, Irving B, Eddowes PJ, Green D, Kelly MD, Jayaratne N, Banerjee R, Brady M, Hirschfield GM. Novel quantitative magnetic resonance imaging features with liver function tests to distinguish parenchymal and biliary disease. 22nd Conference of Medical Image Understanding and Analysis (MIUA); United Kingdom. Southampton: Springer, 2018: 37-43. . |