Published online Feb 7, 2019. doi: 10.3748/wjg.v25.i5.632
Peer-review started: October 2, 2018
First decision: November 7, 2018
Revised: December 16, 2018
Accepted: December 19, 2018
Article in press: December 19, 2018
Published online: February 7, 2019
Processing time: 120 Days and 14.6 Hours
To identify factors other than active disease and anemia that contribute to fatigue in pediatric inflammatory bowel disease (IBD).
We performed an electronic search in Medline and EMBASE from their inception to May 2017 using the search term “fatigue” or the related keywords “physical impairment” and “inflammatory bowel disease” with the filter “child” (age 0-18 years). Cross-sectional and case-control studies were included. We restricted our search to studies published in English. We used the PRISMA checklist and flow diagram. Duplicate articles were manually deleted in End Note. To identify further relevant studies, we checked the reference lists of the selected articles.
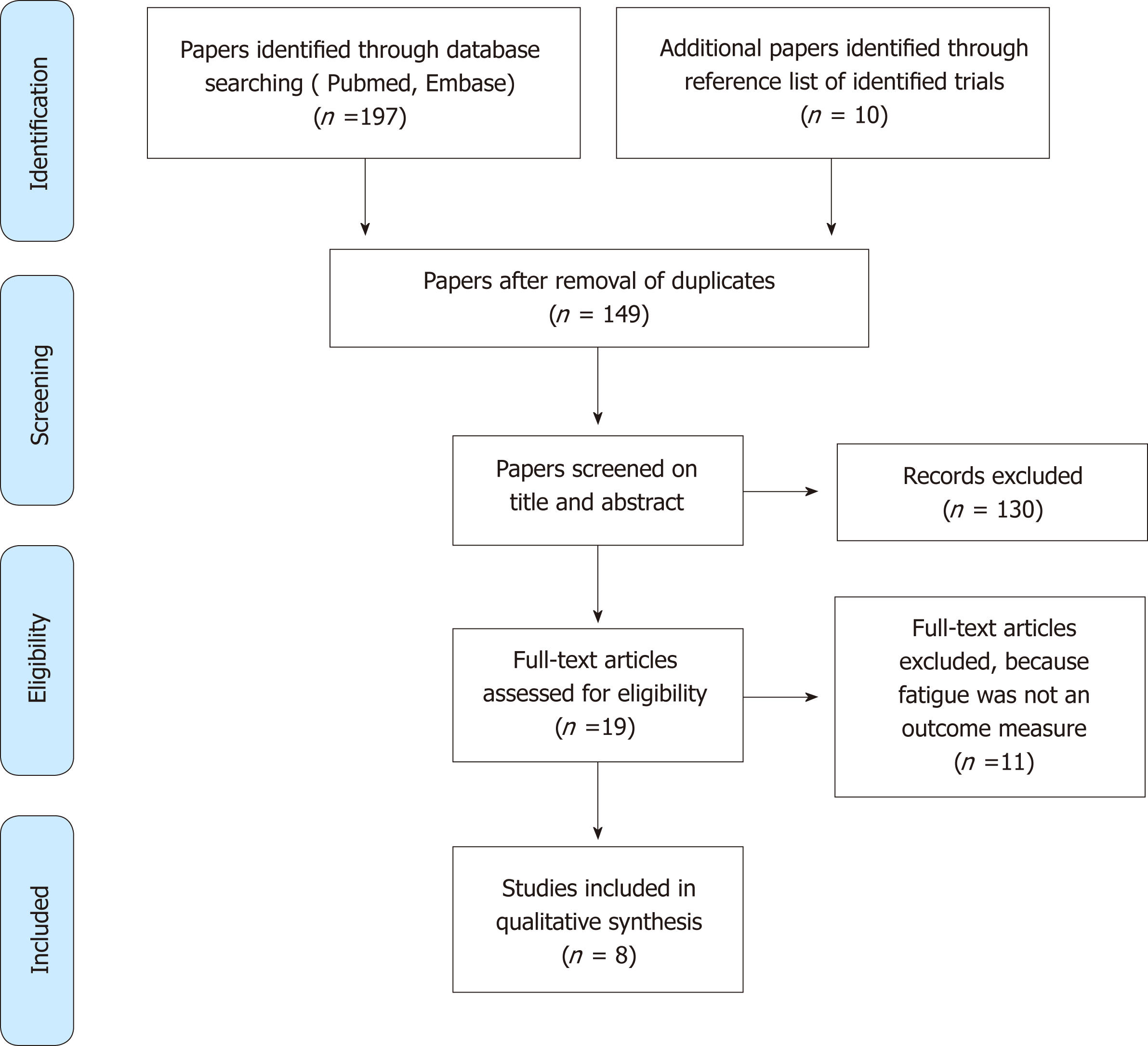
We identified 149 papers, of which 19 were retrieved for full text review. Eleven studies were subsequently excluded because fatigue was not evaluated as an outcome measure. Eight papers focused on the desired topic and were discussed in the final analysis. A lack of uniformity of outcome measures made the pooling of data impossible. In all but one study, questionnaires were used to evaluate fatigue. In the remaining study, an accelerometer was used to measure daily activities, sleeping time and their relationships with fatigue in a more quantifiable manner. Adolescents with IBD are significantly more fatigued than healthy controls. In addition to active disease, increased anxiety or depression and disturbed family relationships were frequently reported predictors of fatigue. Quantitative measurement of physical activity in patients with Crohn’s disease showed a reduction in the number of steps per day, and patients with ulcerative colitis had a shorter duration of physical activity during the day.
Fatigue in pediatric IBD is related to a combination of biological, functional and behavioral factors, which should all be taken into account when managing fatigue.
Core tip: Children and adolescents with inflammatory bowel disease (IBD) often report fatigue as their most severe and distressing symptom. Fatigue is often attributed to active disease and anemia. We systematically reviewed the literature to identify additional factors that contribute to fatigue in pediatric IBD. After a strict selection process, eight studies were suitable for detailed data extraction. Increased anxiety or depression and disturbed family relations were frequently reported predictors of fatigue. This systematic review demonstrates the importance of evaluating biological, functional, and psychobehavioral factors to facilitate the optimal management of fatigue.
- Citation: Van de Vijver E, Van Gils A, Beckers L, Van Driessche Y, Moes ND, van Rheenen PF. Fatigue in children and adolescents with inflammatory bowel disease. World J Gastroenterol 2019; 25(5): 632-643
- URL: https://www.wjgnet.com/1007-9327/full/v25/i5/632.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i5.632
Inflammatory bowel disease (IBD) is a chronic disease of the gastrointestinal tract. The disease is characterized by relapsing periods of inflammation and remission and usually presents with abdominal pain, diarrhea, rectal bleeding and weight loss[1]. The ultimate goal in IBD treatment is to reach clinical remission as quickly as possible. Fatigue and decreased physical fitness may continue to affect a patient’s daily life despite disease remission. Ten percent of patients with IBD are diagnosed before the age of 19 years[2].
Fatigue refers to a subjectively overwhelming sense of tiredness, lack of energy, and feeling of exhaustion that decreases one’s capacity for physical and mental activity[3]. It is a common, independent, and nonspecific symptom identified in numerous chronic health conditions in childhood[4]. In adults with chronic disease, fatigue can be a major source of disablement and is often reported as being among the most severe and distressing symptoms[5]. It affects physical, emotional, cognitive, and social functioning, impacting quality of life. Nevertheless, fatigue has typically been ignored in the assessment of symptom severity or outcome in many diseases in which it is observed[5].
The quantification of fatigue is challenging due to the lack of a consensus framework, vague terminology, and the multidimensional nature of symptoms. Subjective methods, such as self-reported or parent-reported surveys[6,7], are commonly used but can be distorted by response and recall bias. More objective methods, such as polysomnography and performance tests[8-10], are expensive and time-consuming. Furthermore, the prevalence of fatigue varies among healthy pediatric age groups; it is common in infancy, early childhood, and late adolescence and less frequently observed during mid-childhood; it is more common in girls than in boys[9].
We aimed to systematically review the literature to identify factors that contribute to fatigue in children and adolescents with IBD.
We searched for studies published in Medline and EMBASE up to May 2017. The search strategy for Medline was as follows: (“fatigue” [MeSH Terms] OR “fatigue” [All Fields]) AND (“inflammatory bowel diseases”[MeSH Terms] OR (“inflammatory” [All Fields] AND “bowel” [All Fields] AND “disease” [All Fields] OR “inflammatory bowel disease” [All Fields]); (“physical examination” [MeSH Terms] OR (“physical” [All Fields] AND “examination”[All Fields]) OR “physical examination” [All Fields] OR “physical” [All Fields]) AND impairment [All Fields]) AND ("inflammatory bowel diseases"[MeSH Terms] OR ("inflammatory"[All Fields] AND "bowel"[All Fields] AND "diseases"[All Fields]) OR "inflammatory bowel diseases"[All Fields] OR ("inflammatory"[All Fields] AND "bowel"[All Fields] AND "disease"[All Fields]) OR "inflammatory bowel disease"[All Fields]) , with the filter “child” (age 0-18 years). For EMBASE, the search strategy was as follows: (“fatigue”/exp OR fatigue) AND Inflammatory AND (“bowel”/exp OR bowel) AND (“disease”/exp OR disease). We restricted our search to studies published in English. Duplicate articles identified in both Medline and EMBASE were manually deleted in End Note. To identify additional relevant studies, we checked the reference lists of the selected articles.
We selected cross-sectional or case-control studies reporting on fatigue (or its synonyms) in patients under the age of 19 years with IBD. Two reviewers (Van de Vijver E and Van Gils A) independently screened the abstracts of all identified articles to determine their eligibility. Any disagreements regarding the inclusion of articles were solved by discussion until consensus was reached.
Study quality was assessed using the online criteria for case-control and cross-sectional studies[11]. Each item was scored as “yes”, “no”, or “not reported”.
The guidelines of the PRISMA 2009 Statement were adopted.
This study includes paper retrieved by electronic searches up to May 2017. In total, 149 papers were identified, of which 19 were retrieved for full-text review. Eleven were subsequently excluded because fatigue was not evaluated as an outcome measure. Eight focused on the desired topic and were discussed in the final analysis (Figure 1).
The selected studies varied considerably with regard to the fatigue assessment method, which made pooling of data impossible (see Table 1).
| First author (year of publication) | Study objectives | Age (yr) | Patient population | Percentage of patients with active disease | Main findings related to fatigue |
| Marcus et al (2009)[6] | To evaluate the degree of fatigue and health-related quality-of-life in children with IBD | 10-17 | 52 CD; 13 UC; 5 IBD-U; 157 healthy controls | Remission 56%; Mild 22%; Moderate 17%; Severe 5% | Adolescents with IBD have significantly more fatigue than healthy controls; PedsQL total fatigue, general fatigue, and sleep/rest fatigue were all impaired in patients with IBD; Adolescents with IBD are fatigued even when clinical remission is reached |
| Nicholas et al (2007)[13] | To understand the lived experience and elements of quality-of-life in adolescents and adolescents with IBD | 7-19 | 61 CD; 19 UC | Not reported | Young patients with IBD commonly feel “sick and tired” and have “no energy” |
| Pirinen et al (2010)[16] | To evaluate the effect of disease severity on (the frequency of) sleep problems and daytime-tiredness among adolescents with IBD | 10-18 | 53 CD; 83 UC; 24 IBDU; 236 healthy controls | Not reported | Adolescents with IBD do not report more sleeping problems or overtiredness than their healthy peers Adolescents with active disease have significantly more trouble sleeping, more daytime sleepiness and are overtired compared to adolescents with mild IBD symptoms; Adolescents with severe IBD symptoms have worse quality of sleep and more sleep disturbances than those with less severe IBD |
| Werkstetteret al (2012)[8] | To evaluate whether physical activity is reduced in patients with IBD compared to control subjects | 6-20 | 27 CD; 12 UC; 39 healthy controls | Remission 66%; Mild 34% | Patients with IBD show a trend toward less physical activity, especially among girls and those with mild disease activity; There is no relation between inflammatory markers (CRP) and physical activity |
| Rogler et al (2013)[7] | To examine the determinants of health- related quality-of-life in adolescents and adolescents with IBD | 11-15 | 64 CD; 46 UC | PCDAI > 15 36%; PUCAI ≥ 10 28% | Patients with IBD (in particular boys) have moderate impairments in physical well-being; Impairment in physical well-being is associated with active inflammation; And its symptoms |
| Loonen et al (2002)[12] | To evaluate the impact of IBD on health- related quality of life | 8-18 | 41 CD; 40 UC; 2 IBD-U | Mild 60%; Moderate 23%; Severe 15%; Missing 2% | Adolescents with IBD have impairments in motor functioning (running, walking, playing) and complain more of tiredness, especially those with Crohn’s disease. |
| Tojek et al (2002)[14] | To examine family dysfunction, maternal physical symptoms and maternal positive affect as correlates of health status in adolescents with IBD | 11-18 | 36 CD; 26 UC | Not reported | Family dysfunction is related to an increased frequency of fatigue in adolescents; Maternal positive affect is inversely related to fatigue (not significant); Fatigue is independent of maternal negative affect |
| Ondersma et al (1996)[15] | To examine how psychological factors relate to disease severity among adolescents with IBD | 11-17 | 34 CD; 22 UC | Not reported | There is a relationship between negative affect and physical symptoms of fatigue |
Six papers reported fatigue or physical activity related to IBD as their primary outcome. The remaining two studies reported quality-of-life as the primary outcome; one used a quality-of-life questionnaire and evaluated the domain “motor functioning” separately, while the other conducted a semi structured interview with questions about the functional impact of the disease. The methodological quality of the studies is summarized in Table 2.
| First author (year of publication) and study type | Patient selection | Disease activity score | Fatigue score | Study quality |
| Marcus et al (2009)[6] Case-control study | Patients: recruited during scheduled clinical appointments at University Hospital, United States; Healthy controls: adolescent children of hospital employees | CD: PCDAI; CU and IBDU: PGA | PedsQL Multidimensional Fatigue Scale, IMPACT-III, PedsQL 4.0 Generic Core Scales Children’s Depression Inventory: Short Form | Good: no sample size justification |
| Nicholas et al (2007)[13] Cross-sectional study | Patients: recruited from the database of Reference Children’s Hospital, Canada | No distinction made | Semi structured interview designed by author | Poor: Patients purposively selected, questionnaires not validated, participation rate not reported |
| Pirinen et al (2010)[16] Case-control study | Patients: recruited from the database of the Population Register Center, Finland; Healthy controls: matched | VAS disease severity | Youth self-reported questionnaire, Sleep Self Report, child behavior checklist | Medium: Subjective score to assess disease severity, exact sleep duration unknown |
| Werkstetter et al (2012)[8] Case-control study | Patients: recruited from University Hospital, Germany; Healthy controls: matched | CD: PCDAI; UC: PUCAI | SenseWear Pro2 accelerometer, German KINDL, IMPACT III | Good: no sample size justification |
| Rogler et al (2013)[7] Cross-sectional study | Patients: recruited from Swiss IBD cohort study, Switzerland | CD: PCDAI; UC: PUCAI | KIDSCREEN-27 | Medium: numbers in text and table do not match |
| Loonen et al (2002)[12] Cross-sectional study | Patients: recruited from a database of two large tertiary referral centers, Netherlands | 5-item symptom card (completed by patients) | TACQOL, IMPACT-II | Good: validated questionnaires, the results compared with healthy controls |
| Tojek et al (2002)[14] Cross-sectional study | Patients: recruited from routine outpatient visit in 2 urban pediatric gastroenterology hospitals, United States | No distinction made | Questions designed by author | Medium: parental factors can influence adolescent’s health, the converse remains possible, only mothers investigated, questionnaires not validated |
| Ondersma et al (1996)[15] Cross-sectional study | Patients: recruited from 2 pediatric gastroenterology hospitals, United States | No distinction made | 10-item Subjective Illness Questionnaire (parts or RCMAS and CDI) | Medium: no sample size justification, parts of validated questionnaires |
Seven of eight papers used subjective methods, such as questionnaires, to evaluate fatigue[6,7,12-16]. Most research teams used self-reported surveys [IMPACT-III, semistructured interviews, Youth Self Report (YSR), Sleep Self Report (SSR), RCMAS, KINDL, KIDSCREEN and TACQOL][7,8,12,13,15,16], while others used a combination of parent proxy-reported and self-reported surveys (PEDSQL multidimensional fatigue scale and PedsQL 4.0 generic care scale)[6]. Only one paper used a parent proxy-reported questionnaire [Child Behavior Checklist (CBCL)][16]. Table 3 describes the myriad of fatigue-related diagnostic tests that were used in the included studies.
| Abbreviation | Full name | Details |
| CBCL | Child Behavior Checklist | Caregiver report form that categorizes problem behaviors in preschool and school-aged children in the following 8 syndromes: aggressive, anxious-depressed, attention, rule-breaking, somatic complaints, social, thought, withdrawn-depressed. |
| CDI | Children’s Depression Inventory | Adolescent self-reported assessment. For each of 26 items, respondents endorsed one of three sentences indicating varying levels of depression. |
| IMPACT-III | Not applicable | IBD disease-specific health-related quality-of-life questionnaire for pediatric patients. It is composed of 35 items in the following 6 domains: IBD-related symptoms (7 items), systemic symptoms (3), emotional functioning (7), social functioning (12), body image (3) and treatment/intervention-related concerns (3). Each item is scored on a 5-point Likert scale, coded from 0 to 4 points. Higher scores indicate better quality of life. |
| KIDSCREEN 27 | Not applicable | Self-reported survey is a quality of life questionnaire consisting of 27 items measuring physical well-being, psychological well-being, autonomy and parent relations, peers and social support, and school environment. |
| KINDL | Not applicable | Adolescent self-reported survey consists of 24 Likert-scaled items, which are subdivided into the following six dimensions (subscales) of quality of life: physical well-being, emotional well- being; self-worth, well-being in the family, well-being regarding friendships and well-being at school. |
| McMaster Family Assessment Device | Not applicable | Adolescent self-reported 60-item instrument that assesses six domains, namely, problem solving, communication, roles, affective responsiveness, affective involvement, behavior control and general functioning of family functioning as well as general family dysfunction. |
| PedsQL generic scale | Pediatric Quality of Life Inventory | Parent reported and self-reported assessment. A modular approach to measuring health-related quality of life (HRQOL) in healthy children and adolescents and those with acute and chronic health conditions. It contains the following four multidimensional scales: physical functioning, emotional functioning, social functioning, school functioning. |
| PedsQL Multidimensional Fatigue Scale | Pediatric Quality of Life Inventory Multidimensional Fatigue Scale | Age-appropriate versions and parallel forms for children and parents. It measures the perceptions of fatigue by children and their parents and has been validated in a variety of pediatric chronic diseases. |
| RCMAS | Revised Children’s Manifest Anxiety scale | Adolescent self-reported assessment that is a true/false anxiety measure containing 28 items. The measured key areas are physiological anxiety, worry, social anxiety and defensiveness. The scale differentiates between anxiety-disordered and normal Children. |
| SSR | Sleep Self Report | Adolescent self-reported assessment to discern sleep patterns and possible difficulties with sleep. |
| TACQOL | TNO-AZL Children’s Quality of life Questionnaire | Generic health-related quality of life questionnaire enabling comparisons between groups of children with varying chronic diseases. It includes 7 scales, involving general physical function, motor function, daily function, cognitive function, social contact, and positive and negative moods. |
| YSR | Youth Self Report | Adolescent self-reported assessment with the following eight empirically-based syndrome scales: anxious/depressed, withdrawn/depressed and somatic complaints composing the internalizing (i.e., emotional) broad-band scale; rule-breaking behavior and aggressive behavior composing the externalizing (i.e., behavioral) broad-band scale; and these two scales, together with the syndrome scales of social, thought and attention problems, compose the total problems scale. |
Scientists from Chicago and Texas performed a cross-sectional study among 70 children with IBD and 157 healthy controls and their parents[6]. They categorized fatigue as general fatigue (e.g., “feeling tired”), sleep/rest fatigue (e.g., “feeling tired when waking up”) or cognitive fatigue (e.g., “attention problem”)[6] based on the PedsQL Multidimensional Fatigue Scale[11]. General fatigue and sleep/rest fatigue were more frequently observed in pediatric IBD patients than in healthy control subjects, even when their disease was in remission. Differences in cognitive fatigue were not observed[6]. A Canadian team from Toronto conducted in-depth interviews among 80 children and adolescents who were purposively selected for their variation in age and condition and found that children and teenagers with IBD commonly mentioned that “exhaustion” and “malaise” (“having no energy and being tired”) had large impacts on their lives[13]. A Finnish research team evaluated sleep problems and daytime tiredness in 160 adolescents by both a parent proxy-reported survey (CBCL) and a self-reported questionnaire (YSR)[17]. Twenty-five percent of parents reported that their adolescent child had trouble sleeping. This was a significantly greater percentage than was found among the parents of healthy controls. Overall, parents of adolescents with IBD more commonly reported sleeping during the day and night and overtiredness than did parents of healthy controls. The self-reported questionnaire did not confirm the high prevalence of sleep-related problems among IBD patients when compared to healthy subjects (11% vs 16%)[16].
A Swiss research team evaluated 110 adolescents with IBD who were included in the national IBD Cohort Study[7]. They assessed fatigue as physical activity using the KIDSCREEN 27. Physical well-being (e.g., “feeling fit, being physical active, able to run”) was only moderately disturbed in IBD patients compared to healthy controls[7].
A German research group conducted the only study that evaluated fatigue in an objective manner with a wearable device. They assessed physical activity using the SenseWear Pro2 armband (a portable motion sensor) and reported a trend towards a shorter duration of physical activity and significantly prolonged sleep duration in patients with mild IBD compared to controls, but there were no statistically significant differences[8].
Disease activity: All included studies observed a positive correlation between disease activity and fatigue, but the scoring systems used to discriminate active disease from disease remission differed among the papers. The team from Chicago and Texas used the Pediatric Crohn’s Disease Activity Index (PCDAI) and defined disease remission as a score < 10. They reported that children with active Crohn’s disease (CD) had significantly more symptoms of general fatigue (“feeling tired”) and sleep/rest fatigue (“feeling tired when waking up”) than children and teenagers in remission[6]. The Finnish study among 160 children and teenagers used a visual analogue scale (VAS) to measure disease activity. Children with severe IBD (VAS scores above 3) had significantly more trouble falling asleep (41% vs 22%), felt significantly more overtired (80% vs 44%) and had significantly longer sleep duration than adolescents with less active disease (VAS score below 3). In that study, the results of the self-reported questionnaires and the parent reports were very similar when the adolescents had higher VAS scores, but this was less true in the parent-adolescent pairs with mild IBD symptoms[10,16].
Medication: The research team from Chicago and Texas evaluated the association between fatigue and medication and concluded that the use of mesalamine, thiopurine or anti-tumor necrosis factor (TNF) were not predictors of fatigue as measured with the PedsQL Fatigue Scale[6].
Family support: A group from Detroit found a significant association between fatigue and dysfunction in the family[14]. The researchers used the McMaster Family Assessment Device[18]. They also evaluated two additional items created by the authors themselves, which assessed the frequency of IBD-related pain and IBD-related fatigue over the past 3 mo. They found that maternal positive affect, including being attentive, active, and interested, was inversely related to fatigue but the association was not significant. Fathers were not included in the study because they almost never accompanied their children to the clinic, and a considerable proportion of the adolescents did not have fathers living with them[14].
Psychological variables: In another paper, the Detroit team assessed 56 adolescents with IBD (aged between 11 and 17 years) with the Revised Children’s Manifest Anxiety Scale and found that adolescents with a negative affect (i.e., those who reported anxiety and depression) also experienced more pain and fatigue[15]. The group from Chicago and Texas used the Children’s Depression Inventory and found that adolescents with primarily inactive IBD did not report more depressive symptoms than healthy controls (1.4% vs 1.3%)[6].
Disease type: The studies that used questionnaires to assess fatigue did not observe differences between CD and ulcerative colitis (UC) patients[8,12,16]. The German research group that evaluated physical activity with a wearable device found that patients with CD tended towards taking fewer steps per day[8], and UC patients had a shorter duration of physical activity compared with healthy controls[8].
Eight studies were included in this systematic review. These studies were selected for their focus on fatigue in adolescents with IBD.
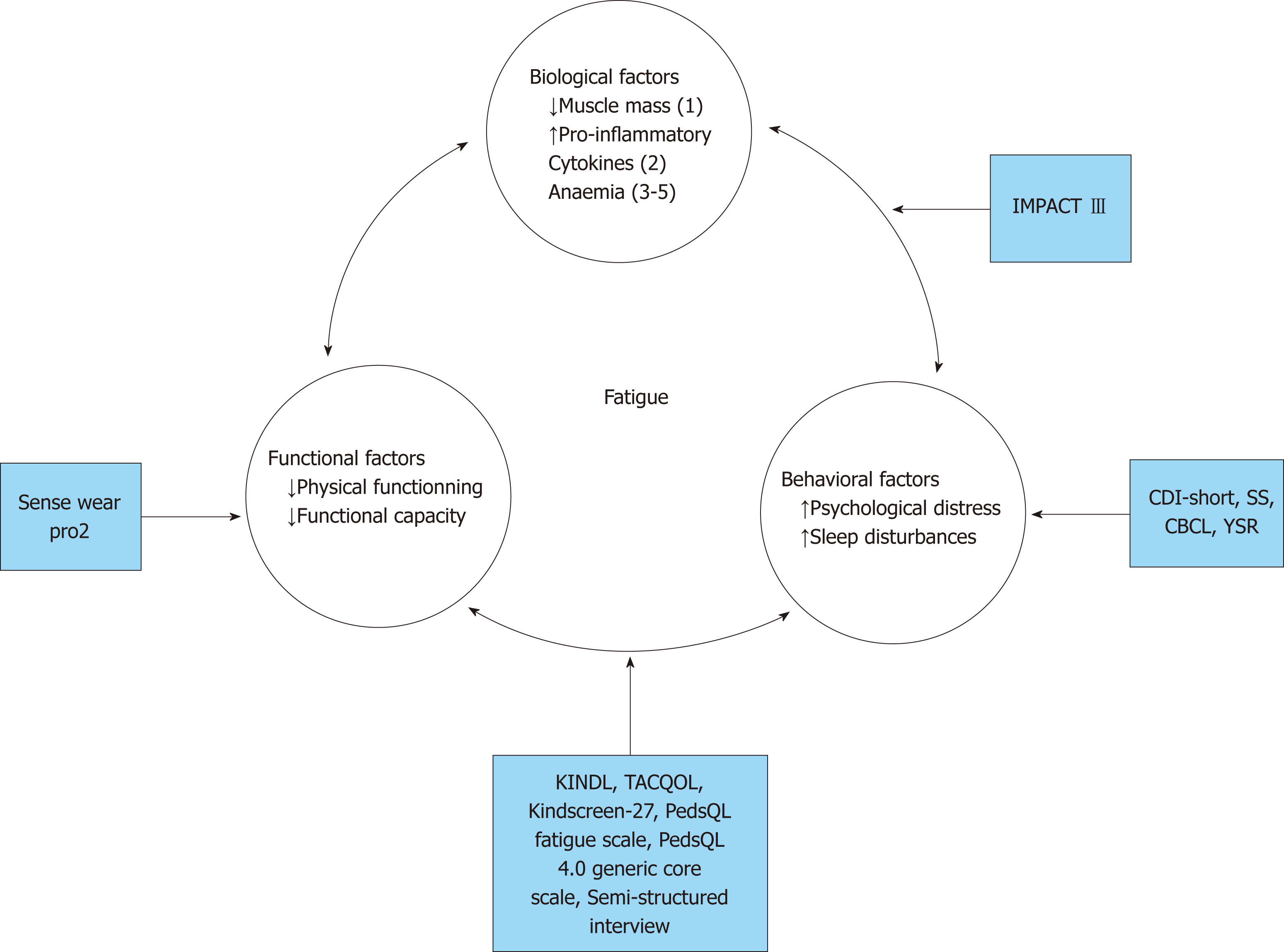
This review demonstrates that fatigue, exhaustion, diminished physical activity and trouble sleeping are more common in children and adolescents with IBD than in their healthy peers. Fatigue is likely to be a multifactorial phenomenon and includes biological factors (such as disease activity), psychobehavioral factors (such as anxiety, depression and family support) and functional factors (such as decreased functional capacity). The model depicted in Figure 2 addresses the various etiological factors and the connection with the fatigue-related diagnostic tests mentioned in this paper. The model highlights the importance of the multifaceted nature of fatigue, and this fatigue model could act as a guide on which to base treatment interventions.
Biological factors: Fatigue is a common finding[6] in children and adolescents with IBD, and several studies have shown a positive relationship between the degree of disease activity and fatigue. Adolescents with active IBD experience more fatigue than their peers in disease remission, who, in turn, experience more fatigue than healthy controls. It is plausible that active disease impairs sleep quality due to nocturnal abdominal pain and diarrhea. Inflammation and immune activation, together with the subsequent activation of glial cells and mitochondrial damage, likely account for the severe levels of intractable fatigue and disability seen in patients with autoimmune diseases[19].
Adolescents in clinical remission are fatigued, but patients in deep remission were not assessed: deep remission could have an impact on less fatigue.
Reduced muscle mass[20] and anemia[21-23], both of which are frequently observed in patients with IBD, even when their disease is in remission, may also have affect fatigue, but so far, these factors have not been investigated in the adolescent IBD population.
Psychobehavioral factors: The papers that sought correlations between psychobehavioral factors and fatigue showed conflicting results. One paper[14] linked anxiety, depression and lack of family support with IBD-related fatigue, while another paper failed to show that depression occurs more often in adolescents with IBD than in their healthy peers[6]. Sleep disorders can affect the feeling of being tired, as shown in 2 of the included papers. Sleep deprivation leads to more anxiety and depression and to an increase in somatic complaints and aggressive behavior[13,16,17]. Sleep itself was not often a research objective; only one study had sleeping problems as an outcome measurement[16], while a German study only reported a trend towards prolonged sleep duration in patients with mild IBD compared to healthy controls[8].
Functional factors: Only one paper focused on functional capacity in relation to fatigue and used a wearable device to plot activity over time; this study did not find a significant difference between patients with IBD and healthy controls. It is rather surprising that only one paper looked at functional capacity in adolescents with IBD because it is a frequently used outcome measurement in other chronic diseases[24-27].
In 2010, a systematic review[3] identified 10 papers about fatigue in patients with IBD and mentioned that the topic deserved more attention, as the prevalence of fatigue approached 50% in patients with IBD in remission and up to 86% in patients with active IBD. Cuzber-Dochan and colleagues published a systematic review in 2013 that included 28 papers on adults, and they concluded that the use of terminology regarding fatigue is inconsistent and that knowledge of the causes, severity and ways of measuring IBD fatigue is incomplete[28]. Three years later, the same research grouped repeated the literature search and identified a number of psychosocial and physical factors that could potentially be modified through targeted health interventions to improve fatigue in IBD. As in this study, they concluded that fatigue is multifactorial and is associated with active disease, poor sleep quality, anxiety and depression, but the complex interplay between these factors has yet to be deciphered[29].
In studies among adolescents, disease activity and sleep quality are also related to fatigue, but the relationship with anxiety and depression is unclear. Approximately one-quarter of adolescents with IBD have somatic or cognitive symptoms of depression[30], and this is comparable with the prevalence observed in their healthy peers.
Depression among adult patients with IBD, on the other hand, is more common compared to among control subjects[12,31-33].
Previous studies described a poor to low degree of parent-adolescent agreement on psychosocial symptoms[17,30]. Moreover, adolescents and parents report different symptoms. Therefore, to gain a comprehensive picture of the complaints in adolescents with IBD, both the adolescents and their parents need to be questioned[34].
The cross-sectional design of most included studies precludes the ability to draw conclusions concerning the causal relations between variables. Prospective observational cohort studies are needed to gain more insight into the direction and mechanism of the identified associations. If prospective cohort studies are conducted in ethnically and socioeconomically diverse groups of children and adolescents, causative factors of fatigue can be identified, and these could potentially lead to more efficacious ways of treating fatigue in adolescents with IBD.
Future research opportunities: The mechanism underlying fatigue in children and adolescents with IBD remains poorly understood. Fatigue is a subjective sensation and presents with a multitude of symptoms, which makes it difficult to describe, measure and quantify. Past studies have mainly focused on one aspect of fatigue. Future studies should explore fatigue manifestations at several levels simultaneously, including illness-related aspects (such as ongoing inflammation, disease activity, medication use and pain), physical functioning (health-related quality of life, sleep quality and disability), and psychobehavioral factors.
Fatigue is a common problem in children and teenagers with IBD, and it is significantly more prevalent among young patients with IBD than in the healthy control population. It is multidimensional and caused by both physical and psychosocial factors. The most predictive factor seems to be disease activity. Health care providers need to pay attention to this problem because it is associated with reduced quality of life, increased sleeping problems and increased anxiety. The multifactorial nature of fatigue necessitates multilevel testing.
Children and adolescents with inflammatory bowel disease (IBD) regularly report fatigue as their most severe and distressing symptom. Fatigue is often attributed to active disease and anemia, but also in quiescent IBD, fatigue can trouble daily life.
The ultimate goal in IBD management is not only to reach disease remission, but also to counteract fatigue and decreased physical fitness
We aimed to systematically review the literature to identify factors that contribute to fatigue in children and adolescents with IBD.
We performed an electronic search in Medline and EMBASE from their inception to May 2017 using the search term “fatigue” or the related keyword “physical impairment” and “inflammatory bowel disease” with the filter “child” (age 0-18 years). Cross-sectional and case-control studies were included. We restricted our search to studies published in English. To identify further relevant studies, we checked the reference lists of the selected articles.
We ultimately identified eight papers that matched the search criteria. A lack of uniformity of outcome measures made the pooling of data impossible. In all but one study, questionnaires were used to evaluate fatigue. In the remaining study, an accelerometer was used to measure daily activities, sleeping time and their relationships with fatigue in a more quantifiable manner. Adolescents with IBD are significantly more fatigued than healthy controls. In addition to active disease, increased anxiety or depression and disturbed family relationships were frequently reported predictors of fatigue. Quantitative measurement of physical activity in patients with Crohn’s disease showed a reduction in the number of steps per day, and patients with ulcerative colitis had a shorter duration of physical activity during the day.
Fatigue is a common problem in children and teenagers with IBD, and it is significantly more prevalent among young patients with IBD than in the healthy control population. It is multidimensional and caused by both physical and psychosocial factors. The most predictive factor seems to be disease activity. Health care providers need to pay attention to this problem because it is associated with reduced quality of life, increased sleeping problems and increased anxiety.
The multifactorial nature of fatigue necessitates multilevel testing. Fatigue in pediatric IBD is related to a combination of biological, functional and behavioral factors, which should all be taken into account when managing fatigue.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Belgium
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Day AS, Sandberg KC, Serban ED S- Editor: Ma RY L- Editor: A E- Editor: Huang Y
| 1. | Rosen MJ, Dhawan A, Saeed SA. Inflammatory Bowel Disease in Children and Adolescents. JAMA Pediatr. 2015;169:1053-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 520] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 2. | Ghione S, Sarter H, Fumery M, Armengol-Debeir L, Savoye G, Ley D, Spyckerelle C, Pariente B, Peyrin-Biroulet L, Turck D, Gower-Rousseau C; Epimad Group. Dramatic Increase in Incidence of Ulcerative Colitis and Crohn's Disease (1988-2011): A Population-Based Study of French Adolescents. Am J Gastroenterol. 2018;113:265-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 120] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 3. | van Langenberg DR, Gibson PR. Systematic review: fatigue in inflammatory bowel disease. Aliment Pharmacol Ther. 2010;32:131-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 166] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 4. | Crichton A, Knight S, Oakley E, Babl FE, Anderson V. Fatigue in child chronic health conditions: a systematic review of assessment instruments. Pediatrics. 2015;135:e1015-e1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Farrell D, McCarthy G, Savage E. Self-reported Symptom Burden in Individuals with Inflammatory Bowel Disease. J Crohns Colitis. 2016;10:315-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 114] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 6. | Marcus SB, Strople JA, Neighbors K, Weissberg-Benchell J, Nelson SP, Limbers C, Varni JW, Alonso EM. Fatigue and health-related quality of life in pediatric inflammatory bowel disease. Clin Gastroenterol Hepatol. 2009;7:554-561. [PubMed] |
| 7. | Rogler D, Fournier N, Pittet V, Bühr P, Heyland K, Friedt M, Koller R, Rueger V, Herzog D, Nydegger A, Schäppi M, Schibli S, Spalinger J, Rogler G, Braegger CP; Swiss IBD Cohort Study Group. Coping is excellent in Swiss Children with inflammatory bowel disease: results from the Swiss IBD cohort study. J Crohns Colitis. 2014;8:409-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Werkstetter KJ, Ullrich J, Schatz SB, Prell C, Koletzko B, Koletzko S. Lean body mass, physical activity and quality of life in paediatric patients with inflammatory bowel disease and in healthy controls. J Crohns Colitis. 2012;6:665-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 88] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 9. | Aadland E, Andersen LB, Skrede T, Ekelund U, Anderssen SA, Resaland GK. Reproducibility of objectively measured physical activity and sedentary time over two seasons in children; Comparing a day-by-day and a week-by-week approach. PLoS One. 2017;12:e0189304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 10. | Alhassan S, Lyden K, Howe C, Kozey Keadle S, Nwaokelemeh O, Freedson PS. Accuracy of accelerometer regression models in predicting energy expenditure and METs in children and youth. Pediatr Exerc Sci. 2012;24:519-536. [PubMed] |
| 11. | Varni JW, Burwinkle TM, Szer IS. The PedsQL Multidimensional Fatigue Scale in pediatric rheumatology: reliability and validity. J Rheumatol. 2004;31:2494-2500. [PubMed] |
| 12. | Loonen HJ, Grootenhuis MA, Last BF, de Haan RJ, Bouquet J, Derkx BH. Measuring quality of life in children with inflammatory bowel disease: the impact-II (NL). Qual Life Res. 2002;11:47-56. [PubMed] |
| 13. | Nicholas DB, Otley A, Smith C, Avolio J, Munk M, Griffiths AM. Challenges and strategies of children and adolescents with inflammatory bowel disease: a qualitative examination. Health Qual Life Outcomes. 2007;5:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 96] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 14. | Tojek TM, Lumley MA, Corlis M, Ondersma S, Tolia V. Maternal correlates of health status in adolescents with inflammatory bowel disease. J Psychosom Res. 2002;52:173-179. [PubMed] |
| 15. | Ondersma SJ, Lumley MA, Corlis ME, Tojek TM, Tolia V. Adolescents with inflammatory bowel disease: the roles of negative affectivity and hostility in subjective versus objective health. J Pediatr Psychol. 1997;22:723-738. [PubMed] |
| 16. | Pirinen T, Kolho KL, Simola P, Ashorn M, Aronen ET. Parent and self-report of sleep-problems and daytime tiredness among adolescents with inflammatory bowel disease and their population-based controls. Sleep. 2010;33:1487-1493. [PubMed] |
| 17. | Pirinen T, Kolho KL, Ashorn M, Aronen ET. Sleep and emotional and behavioral symptoms in adolescents with inflammatory bowel disease. Sleep Disord. 2014;2014:379450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 18. | Georgiades K, Boyle MH, Jenkins JM, Sanford M, Lipman E. A multilevel analysis of whole family functioning using the McMaster Family Assessment Device. J Fam Psychol. 2008;22:344-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Morris G, Berk M, Walder K, Maes M. Central pathways causing fatigue in neuro-inflammatory and autoimmune illnesses. BMC Med. 2015;13:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 123] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 20. | Bechtold S, Alberer M, Arenz T, Putzker S, Filipiak-Pittroff B, Schwarz HP, Koletzko S. Reduced muscle mass and bone size in pediatric patients with inflammatory bowel disease. Inflamm Bowel Dis. 2010;16:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 21. | de Laffolie J, Laass MW, Scholz D, Zimmer KP, Buderus S; CEDATA-GPGE Study Group. Prevalence of Anemia in Pediatric IBD Patients and Impact on Disease Severity: Results of the Pediatric IBD-Registry CEDATA-GPGE®. Gastroenterol Res Pract. 2017;2017:8424628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Goodhand JR, Kamperidis N, Rao A, Laskaratos F, McDermott A, Wahed M, Naik S, Croft NM, Lindsay JO, Sanderson IR, Rampton DS. Prevalence and management of anemia in children, adolescents, and adults with inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:513-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 101] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 23. | Pels LP, Van de Vijver E, Waalkens HJ, Uitentuis J, JGonera-de ong G, van Overbeek LA, Norbruis OF, Rings EH, van Rheenen PF. Slow hematological recovery in children with IBD-associated anemia in cases of "expectant management". J Pediatr Gastroenterol Nutr. 2010;51:708-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 24. | Alameri HF, Sanai FM, Al Dukhayil M, Azzam NA, Al-Swat KA, Hersi AS, Abdo AA. Six Minute Walk Test to assess functional capacity in chronic liver disease patients. World J Gastroenterol. 2007;13:3996-4001. [PubMed] |
| 25. | Cracowski C, Wuyam B, Klein V, Lévy P. Lung function and exercise capacity in thalassaemia major. Eur Respir J. 1998;12:1130-1136. [PubMed] |
| 26. | Donadio MV, Heinzmann-Filho JP, Vendrusculo FM, Frasson PXH, Marostica PJC. Six-Minute Walk Test Results Predict Risk of Hospitalization for Youths with Cystic Fibrosis: A 5-Year Follow-Up Study. J Pediatr. 2017;182:204-209.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 27. | Hassan J, van der Net J, Helders PJ, Prakken BJ, Takken T. Six-minute walk test in children with chronic conditions. Br J Sports Med. 2010;44:270-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 28. | Czuber-Dochan W, Ream E, Norton C. Review article: Description and management of fatigue in inflammatory bowel disease. Aliment Pharmacol Ther. 2013;37:505-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 29. | Hindryckx P, Laukens D, D'Amico F, Danese S. Unmet Needs in IBD: the Case of Fatigue. Clin Rev Allergy Immunol. 2018;55:368-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 30. | Szigethy EM, Youk AO, Benhayon D, Fairclough DL, Newara MC, Kirshner MA, Bujoreanu SI, Mrakotsky C, Bousvaros A, Srinath AI, Keljo DJ, Kupfer DJ, DeMaso DR. Depression subtypes in pediatric inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2014;58:574-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 31. | Reed-Knight B, Lobato D, Hagin S, McQuaid EL, Seifer R, Kopel SJ, Boergers J, Nassau JH, Suorsa K, Bancroft B, Shapiro J, Leleiko NS. Depressive symptoms in youth with inflammatory bowel disease compared with a community sample. Inflamm Bowel Dis. 2014;20:614-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 32. | Ross SC, Strachan J, Russell RK, Wilson SL. Psychosocial functioning and health-related quality of life in paediatric inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2011;53:480-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 33. | Greenley RN, Hommel KA, Nebel J, Raboin T, Li SH, Simpson P, Mackner L. A meta-analytic review of the psychosocial adjustment of youth with inflammatory bowel disease. J Pediatr Psychol. 2010;35:857-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 192] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 34. | Pirinen T, Kolho KL, Simola P, Ashorn M, Aronen ET. Parent-adolescent agreement on psychosocial symptoms and somatic complaints among adolescents with inflammatory bowel disease. Acta Paediatr. 2012;101:433-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |