Published online Dec 14, 2019. doi: 10.3748/wjg.v25.i46.6767
Peer-review started: October 20, 2019
First decision: November 10, 2019
Revised: November 21, 2019
Accepted: December 7, 2019
Article in press: December 7, 2019
Published online: December 14, 2019
Processing time: 55 Days and 2.1 Hours
Recent advances in endoscopic technology, especially magnifying endoscopy with narrow band imaging (ME-NBI) enable us to detect superficial esophageal squamous cell carcinoma (ESCC), but determining the appropriate method of resection, endoscopic resection (ER) vs surgical resection, is often challenging. Recently, several studies have reported that 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) is a useful indicator for decision-making regarding treatment for superficial ESCC. Although, there are not enough reports on association between FDG-PET uptake and clinicopathological characteristics of superficial ESCC. And, there are not enough reports on evaluating the usefulness of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC. This study evaluated clinical relevance of FDG-PET and ME-NBI in decision-making regarding the treatment strategy for ESCC.
To investigate the association between FDG uptake and the clinicopathological characteristics of superficial ESCC and its usefulness of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC.
A database of all patients with superficial ESCC who had undergone both ME-NBI and FDG-PET for pre-treatment staging at Aichi Cancer Center Hospital between January 2008 and November 2018 was retrospectively analyzed. FDG uptake was defined positive or negative whether the primary lesion was visualized or could be distinguished from the background, or not. The invasion depth of ESCC was classified according to the Japan Esophageal Society. Primary endpoint is to evaluate the association between FDG uptake and clinicopathological characteristics of superficial ESCC. Secondary endpoint is to investigate the efficacy of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC.
A total of 82 lesions in 82 patients were included. FDG-PET showed positive uptake in 29 (35.4%) lesions. Univariate analysis showed that uptake of FDG-PET had significant correlations with circumferential extension (P = 0.014), pathological depth of tumor invasion (P < 0.001), infiltrative growth pattern (P < 0.001), histological grade (P = 0.002), vascular invasion (P = 0.001), and lymphatic invasion (P < 0.001). On multivariate analysis, only depth of tumor invasion was independently correlated with FDG-PET/computed tomography visibility (P = 0.018). The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of Type B2 in ME-NBI for the invasion depth of T1a muscularis mucosae and T1b upper submucosal layer were 68.4%/79.4%/50.0%/89.3%/76.8%, respectively, and those of Type B3 for the depth of T1b middle and deeper submucosal layers (SM2 and SM3) were 46.7%/100%/100%/89.3%/90.2%, respectively. On the other hand, those of FDG-PET for SM2 and SM3 were 93.3%/77.6%/48.2%/98.1%/80.5%, respectively, whereas, if the combination of positive FDG uptake and type B2 and B3 was defined as an indicator for radical esophagectomy or definitive chemoradiotherapy, the sensitivity, specificity, PPV, NPV, and accuracy were 78.3%/91.5%/78.3%/91.5%/87.8%, respectively.
FDG uptake was correlated with the invasion depth of superficial ESCC. Combined use of FDG-PET and ME-NBI, especially with the microvascular findings of Type B2 and B3, is useful to determine whether ER is indicated for the lesion.
Core tip: The role of 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) for determining treatment strategies in patients with superficial esophageal squamous cell carcinoma (ESCC) remains unclear. This study aimed to demonstrate whether the combination of FDG uptake with findings of magnifying endoscopy with narrow band imaging (ME-NBI) was useful for determining the treatment strategy. If the combination of FDG uptake with Type B2 and B3 on ME-NBI was taken as an indicator for radical esophagectomy or definitive chemoradiotherapy, the sensitivity, specificity, and accuracy were 78.3%, 91.5%, and 87.8%, respectively. Combined FDG-PET and ME-NBI was useful for determining the treatment strategies in patients with ESCC.
- Citation: Toriyama K, Tajika M, Tanaka T, Ishihara M, Hirayama Y, Onishi S, Mizuno N, Kuwahara T, Okuno N, Matsumoto S, Sasaki E, Abe T, Yatabe Y, Hara K, Matsuo K, Tamaki T, Niwa Y. Clinical relevance of fluorodeoxyglucose positron emission tomography/computed tomography and magnifying endoscopy with narrow band imaging in decision-making regarding the treatment strategy for esophageal squamous cell carcinoma. World J Gastroenterol 2019; 25(46): 6767-6780
- URL: https://www.wjgnet.com/1007-9327/full/v25/i46/6767.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i46.6767
Recent advances in endoscopic technology such as image-enhanced endoscopy and magnifying examination enable us to detect superficial esophageal squamous cell carcinoma (ESCC)[1-4]. Furthermore, development of endoscopic treatment had led to an alternative therapy for superficial ESCC. Endoscopic submucosal dissection (ESD), in particular, allows en bloc resection of large lesions and reduces the risk of local recurrence compared with endoscopic mucosal resection (EMR)[5-7]. Furthermore, due to the development of methods to prevent esophageal stricture after ESD for large lesions, the risk of stricture has been dramatically decreased[8,9]. So, now in Japan, for superficial ESCC, tumor invasion depth within the lamina propria mucosae (LPM) is an absolute indication for ESD, regardless of its size and location, and tumor invasion depth into the muscularis mucosae (MM) or upper submucosal layers (SM1) is a relative indication for ESD[10,11]. However, if histological findings after treatment demonstrate that tumor involved SM1 or more, additional treatment, such as radical esophagectomy or definitive chemoradiotherapy (CRT), is recommended for these patients[10,11]. Furthermore, with MM involvement, if the tumor is accompanied by lymphovascular invasion, additional treatment is strongly recommended[10,11]. However, it is currently difficult to evaluate lymphovascular invasion using endoscopic techniques. If additional treatment is recommended, patients unavoidably have to wait for several weeks until treatment starts. If complications such as perforation or esophageal stricture occur, patients must spend much more time feeling uneasy.
18F-Fluorodeoxyglucose positron emission tomography (FDG-PET) is a useful imaging modality that reflects tumor volume and activity, and it is also useful for initial staging, detection of recurrence, and evaluation of response to therapy in advanced ESCC[12-15]. Recently, several studies reported the clinical utility of FDG-PET in superficial ESCC[16-19] and mentioned that FDG-PET uptake could be one of the indicators used for making treatment decisions. However, the utility of FDG-PET in decision-making for endoscopic treatment for superficial ESCC is controversial.
In this study, the aim was to investigate the association between FDG accumulation and clinicopathological characteristics of superficial ESCC, as well as its utility for determining the treatment strategy for superficial ESCC.
A total of 82 consecutive patients diagnosed with thoracic ESCC for the first time and who underwent radical esophagectomy or endoscopic resection (ER), including ESD and EMR using a cap-fitted, with pathologically diagnosed superficial (mucosal or submucosal) cancer in Aichi Cancer Center Hospital, Nagoya, Japan, from January 2008 to November 2018 were retrospectively reviewed. To evaluate clinical staging, each patient was examined by computed tomography (CT) and FDG-PET/CT from the neck to the abdomen and esophagogastroduodenoscopy with narrow band imaging (NBI) magnification before treatment. Before radical esophagectomy or ER, none of these patients underwent CRT, chemotherapy, radiotherapy, or surgery for lesions of the head and neck, mediastinum, thorax, and lung. When patients had synchronous multiple esophageal cancers, the deepest or largest lesions were assessed in this study. All patients were confirmed to not have diabetes mellitus or abnormal glucose tolerance. Staging of cancers clinically and pathologically was in accordance with the TNM classification (8th edition) of the Union for International Cancer Control[20]. The characteristics of these 82 patients are summarized in Table 1. The Institutional Review Board at Aichi Cancer Center Hospital approved this study (ACC 2018-1-400).
| Variable | Total (n = 82) |
| Clinical variables | |
| Sex | |
| Male | 63 |
| Female | 19 |
| Age (yr), mean ± SD | 64.7 ± 9.06 |
| Tumor location | |
| Upper thoracic | 6 |
| Middle thoracic | 46 |
| Lower thoracic | 30 |
| Circumferential extension | |
| < 3/4 | 71 |
| ≥ 3/4 | 11 |
| Macroscopic type | |
| 0-I | 5 |
| 0-IIa | 2 |
| 0-IIc | 75 |
| Primary treatment | |
| EMR-C | 7 |
| ESD | 57 |
| Surgery | 18 |
| Depth of tumor invasion by, EP-LPM | 49 |
| NBI with magnification | |
| MM-SM1 | 26 |
| SM2-SM3 | 7 |
| Lymph node metastasis | |
| Positive | 3 |
| Negative | 79 |
| Pathological variables | |
| Depth of tumor invasion | |
| pTis | 4 |
| pT1a | 61 |
| pT1b | 17 |
| Diameter of primary tumor (mm), mean ± SD | 30.5 ± 18.5 |
| Lymph node metastasis | |
| Positive | 8 |
| Negative | 10 |
| Infiltrative growth pattern | |
| INFa | 50 |
| INFb | 32 |
| Histological grade | |
| Well | 30 |
| Moderate | 47 |
| Poor | 5 |
| Vascular invasion | |
| Positive | 15 |
| Negative | 67 |
| Lymphatic invasion | |
| Positive | 19 |
| Negative | 63 |
An FDG-PET/CT device (TruePoint Biograph 40 with TrueV; Siemens Medical Solutions United States, Knoxville, IL, United States) was used to scan all patients. Patients fasted for 6 h before intravenous injection of 185-370 MBq 18F-FDG, depending on body weight, and images were acquired 90 min after intravenous infusion of the tracer. The parameters of PET scans were as follows: 3D acquisition mode 168 × 168 matrix, trans-axial field of view (FOV) 605 mm, axial FOV 216 mm, and 2-3 min/bed position depending on the body mass index. CT scans for attenuation correction were obtained using the following parameters: 120 KVp; 100 mA, CARE DOSE4D pitch factor 1.2, rotation time 0.5 s, trans-axial scan field 70 cm, 512 × 512 matrix, and slice thickness 2 mm. Reconstruction of PET images was performed using a Gaussian filter of 4.0 mm full width at half maximum value. All images were reconstructed by 3D ordered subset expectation maximization with point spread function modelling, 21 subsets, and 3 iterations.
One certified nuclear medicine physician evaluated PET-CT in consideration of chest CT scan results before treatment. In this study, the FDG uptake was evaluated to be positive or negative by visual evaluation. It was defined positive when the primary lesion was visualized or could be distinguished from the background. On the other hand, the opposite was defined negative. The 3-dimensional image was made in the standardized uptake value (SUV) mode for semiquantitative evaluation on a workstation (syngo MultiModality Workplace, Siemens Healthcare, Erlangen, Germany; software program TrueD, Siemens Healthcare; soft workstation, Siemens, Medical Solutions). FDG uptake was automatically calculated as SUV by the following formula: SUV = tissue concentration (Bq/g)/ [injection dose (Bq)/body weight (g)]. The maximum SUV (SUVmax) of the primary esophageal tumor was automatically obtained from a region of interest, designated as a site of abnormal accumulation on the 3-dimensional images.
For all lesions, before treatment, the depth of tumor invasion was assessed by endoscopy with NBI magnification (ME-NBI) using the classification established by the Japan Esophageal Society[21]. This classification defines microvessels in cancerous lesions as Type B, and Type B is subclassified into Types B1, B2, and B3, which are predictive of invasion depth of T1a- Epithelium (EP) or T1a- LPM, T1a-MM or T1b- SM1, and T1b-SM2 or deeper, respectively. Therefore, in this study, pre-treatment diagnosis of the invasion depth of tumor was decided according to the classification with NBI magnification. For Type B1 and Type B2, ER is indicated, while ER is contra-indicated for B3. This time, macroscopic type was assessed according to the Japanese Classification of Esophageal Cancer, 11th Edition[22]. The circumferential extent of the tumor was classified into two groups, under three-quarters (< 3/4) or three-quarters or more (≥ 3/4), evaluated by endoscopic vital staining using iodine solution. All of the procedures in this study were performed by experienced endoscopists having performed more than 1000 gastrointestinal endoscopies.
Resected specimens were fixed in formalin, sectioned into 2 to 3-mm-thick slices, and then embedded in paraffin. Each slice was microscopically assessed for tumor length, depth of invasion, nodal metastasis, status of vascular invasion (lymphatic spaces and blood vessels), histological grade, and infiltrative growth pattern (INF) pathologically. The pathological stages were diagnosed by the TNM classification (8th edition) of the Union for International Cancer Control[20].
In this study, primary endpoint is to evaluate the association between FDG uptake and clinicopathological characteristics of superficial ESCC. Secondary endpoint is to investigate the efficacy of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC.
For statistical comparisons of clinicopathological factors associated with FDG-PET uptake of primary lesions, the χ2 test or Fisher’s exact test was used for categorical data, and the Mann-Whitney U test was used for continuous data. Continuous variables are expressed as mean ± SD. Logistic regression analysis of clinicopathological factors was performed to assess their independent effects on FDG-PET uptake of primary lesions. The sensitivity, specificity, accuracy, positive predictive value (PPV), and negative predictive value (NPV) of FDG-PET and endoscopy with NBI magnification were calculated with standard formulas. Overall survival was calculated from treatment to death or to the last known date of follow-up. Actual survival was calculated by the Kaplan-Meier method and evaluated statistically by the log-rank test. Significance was defined as probability values < 0.05. Statistical analyses were performed with SPSS software (SPSS for Mac, version 25.0; SPSS, Chicago, IL, Untied States).
Of 82 superficial ESCCs, FDG uptake was positive in 29 (35.4%) and negative in 53 (64.6%) lesions. The associations between primary lesion visibility and clinical and pathological findings are summarized in Table 2. Primary lesion visibility was significantly higher in lesions whose extent was greater than three-quarters (P = 0.014) or showed pT1b tumor invasion (P < 0.001), INFb infiltrative growth pattern (P < 0.001), moderate and poorly differentiated histological grade (P = 0.002), positive vascular invasion (P = 0.001), or positive lymphatic invasion (P < 0.001) on univariate analysis. On multivariate analysis, only depth of tumor invasion into the submucosa (pT1b) was independently correlated with FDG-PET/CT visibility [odds ratio (OR) = 30.21; 95%CI: 1.81-504.05; P = 0.018] (Table 3). SUVmax values were distributed from 2.36 to 12.14, with a mean ± SD value of 4.00 ± 1.73, and those of positive FDG uptake lesion was significantly higher compared to those of negative lesion (5.15 ± 2.44 vs 3.37 ± 1.16; P < 0.001). The circumferential extent, depth of tumor invasion, infiltrative growth pattern, vascular invasion, and lymphatic invasion were significantly associated with the SUVmax values. Lesions that extended more than three-quarters of the circumference had higher SUVmax values than those extending less than three-quarters (5.91 ± 3.21 vs 3.70 ± 1.16; P = 0.003). Tumor invasion of pT1b showed higher SUVmax values than pTis and pT1a (5.62 ± 3.01 vs 3.58 ± 0.81; P < 0.01). The INFb infiltrative growth pattern had higher SUVmax values than INFa (4.72 ± 2.52 vs 3.50 ± 0.63; P = 0.03). Positive vascular invasion showed higher SUVmax values than negative vascular invasion (5.70 ± 3.24 vs 3.62 ± 0.81, P = 0.01). Positive lymphatic invasion showed higher SUVmax values than negative lymphatic invasion (5.17 ± 3.01 vs 3.64 ± 0.86; P = 0.04).
| Variable | Positive (n = 29) | Negative (n = 53) | P value |
| Clinical variables | |||
| Sex | 0.88 | ||
| Male | 22 | 41 | |
| Female | 7 | 12 | |
| Age (yr), mean ± SD | 64.4 ± 11.3 | 64.8 ± 7.66 | 0.86 |
| Tumor location | |||
| Upper thoracic | 2 | 4 | |
| Middle thoracic | 16 | 30 | 0.98 |
| Lower thoracic | 11 | 19 | |
| Circumferential extension | |||
| < 3/4 | 21 | 50 | 0.014 |
| ≥ 3/4 | 8 | 3 | |
| Macroscopic type | |||
| 0-I | 3 | 2 | |
| 0-IIa | 25 | 50 | 0.46 |
| 0-IIc | 1 | 1 | |
| Pathological variables | |||
| Depth of tumor invasion | |||
| Tis + T1a | 14 | 51 | < 0.001 |
| T1b | 15 | 2 | |
| Length diameter of primary tumor (mm), mean ± SD | 36.4 ± 21.87 | 27.2 ± 15.70 | 0.052 |
| Infiltrative growth pattern | |||
| INFa | 9 | 41 | < 0.001 |
| INFb | 20 | 12 | |
| Histological grade | |||
| Well | 4 | 26 | 0.002 |
| Moderate+poor | 25 | 27 | |
| Vascular invasion | |||
| Positive | 11 | 4 | 0.001 |
| Negative | 18 | 49 | |
| Lymphatic invasion | |||
| Positive | 14 | 5 | < 0.001 |
| Negative | 15 | 48 |
| Variable | OR | 95%CI | P value |
| Circumferential extension | |||
| < 3/4 | 3.26 | 0.52-20.49 | 0.21 |
| ≥ 3/4 | |||
| Histological grade | |||
| Well | 3.09 | 0.71-13.48 | 0.13 |
| Moderate + poor | |||
| Depth of tumor invasion | |||
| pTis + pT1a | 30.21 | 1.81-504.05 | 0.018 |
| pT1b | |||
| Infiltrative growth pattern | |||
| INFa | 1.52 | 0.38-6.18 | 0.56 |
| INFb | |||
| Vascular invasion | |||
| Positive | 3.42 | 0.24-49.86 | 0.37 |
| Negative | |||
| Lymphatic invasion | |||
| Positive | 0.26 | 0.012-5.41 | 0.38 |
| Negative |
The sensitivity, specificity, PPV, NPV, and accuracy of Type B1 for the depth of T1a EP-LPM were 87.5%, 79.4%, 85.7%, 81.8%, and 84.1%, respectively, whereas those of Type B2 for the depth of T1a MM and T1b SM1 were 68.4%, 79.4%, 50.0%, 89.3%, and 76.8%, respectively. Those of Type B3 for the depth of T1b middle and deeper submucosal layers (SM2 and SM3) were 46.7%, 100%, 100%, 89.3%, and 90.2%, respectively (Table 4).
| Type B1 for EP-LPM | Type B2 for MM-SM1 | Type B3 for SM2-3 | |
| Sensitivity | 87.5% (42/48) | 68.4% (13/19) | 46.7% (7/15) |
| Specificity | 79.4% (27/34) | 79.4% (50/63) | 100% (67/67) |
| PPV | 85.7% (42/49) | 50.0% (13/26) | 100% (7/7) |
| NPV | 81.8% (27/33) | 89.3% (50/56) | 89.3% (67/75) |
| Accuracy | 84.1% (69/82) | 76.8% (63/82) | 90.2% (74/82) |
If positive FDG uptake was taken as an indicator for T1b SM2-3, the sensitivity, specificity, PPV, NPV, and accuracy of FDG-PET were 93.3%,77.6%, 48.2%, 98.1%, and 80.5%, respectively, whereas if positive uptake of FDG was taken an indicator for radical esophagectomy or definitive CRT, i.e., contraindication for ER, the values of FDG-PET were 78.3%, 81.4%, 62.1%, 90.6%, and 80.5%, respectively (Table 5). Furthermore, if positive FDG uptake was combined with type B2 and B3 and defined as an indicator for radical esophagectomy or definitive CRT, the sensitivity, specificity, PPV, NPV, and accuracy were 78.3%, 91.5%, 78.3%, 91.5%, and 87.8%, respectively (Table 6).
| Invasion depth of SM2-3 | Lesions indicated for surgery or CRT | P value | |
| Sensitivity | 93.3% (14/15) | 78.3% (18/23) | 0.37 |
| Specificity | 77.6% (52/67) | 81.4% (48/59) | 0.60 |
| PPV | 48.2% (14/29) | 62.1% (18/29) | 0.29 |
| NPV | 98.1% (52/53) | 90.6% (48/53) | 0.12 |
| Accuracy | 80.5% (66/82) | 80.5% (66/82) | 1.00 |
| FDG-PET | FDG-PET + ME-NBI | P value | |
| Sensitivity | 78.3% (18/23) | 78.3% (18/23) | 1.00 |
| Specificity | 81.4% (48/59) | 91.5% (54/59) | 0.12 |
| PPV | 62.1% (18/29) | 78.3% (18/23) | 0.24 |
| NPV | 90.6% (48/53) | 91.5% (54/59) | 0.86 |
| Accuracy | 80.5% (66/82) | 87.8% (72/82) | 0.20 |
The mean follow-up period after treatment was 41.8 mo. Four patients died during the follow-up period. Three patients died of ESCC recurrence, and one patient died of other benign disease. Of the three recurrent patients, one died of metachronous ESCC unrelated to the primary lesion. The second patient who underwent radical esophagectomy was diagnosed pre-treatment with invasion depth of SM3, and 12 mo after surgery, lymph node metastasis appeared. The third patient who underwent EMR was diagnosed with MM-SM1, and the histological findings showed LPM without lymphovascular invasion. However, 21 mo later, lymph node metastasis appeared, and radical esophagectomy with neoadjuvant chemotherapy was performed immediately. The patient died 24 mo after surgery. Of the three recurrent patients, only the lesion with invasion depth of SM3 showed FDG uptake.
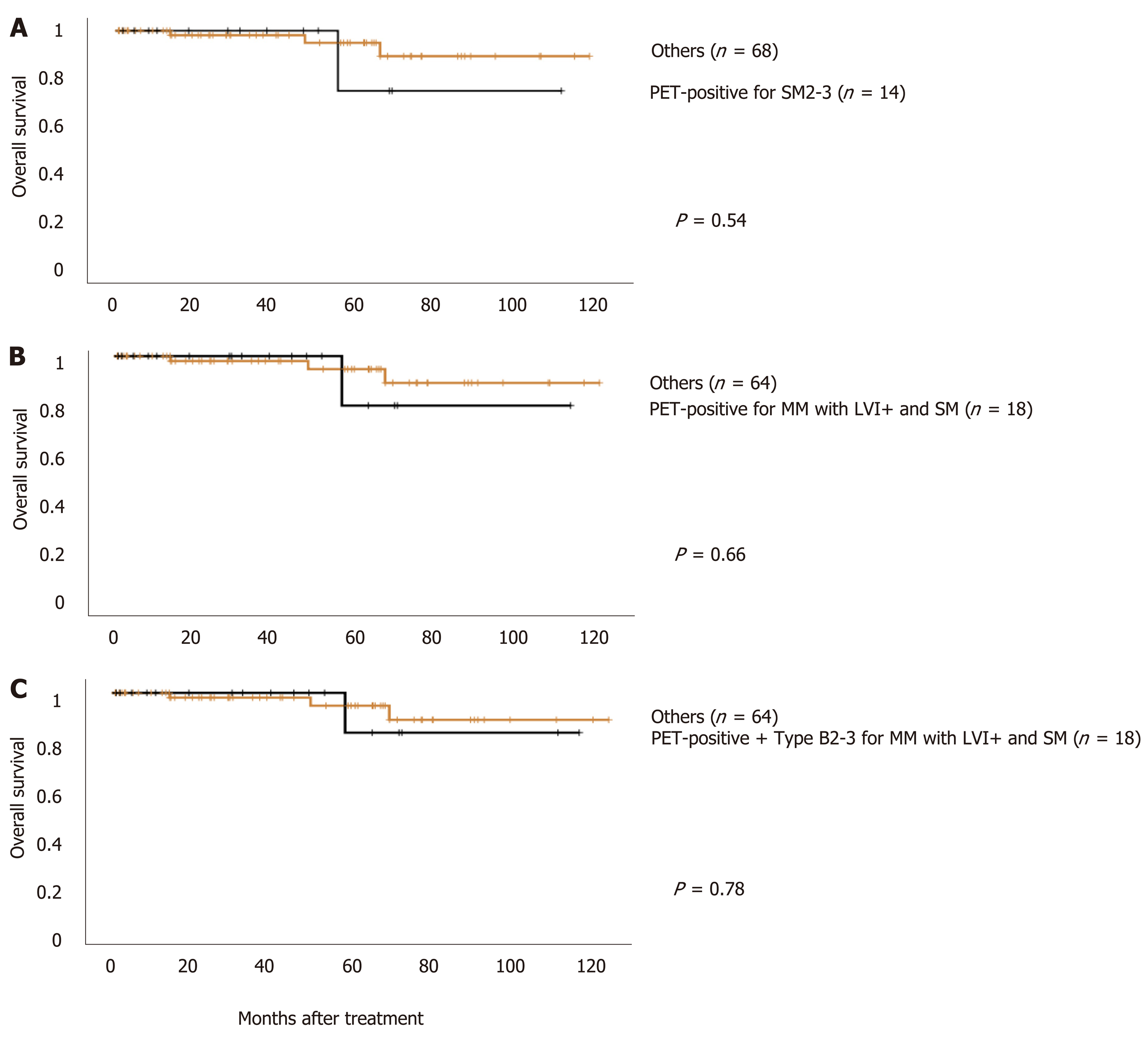
If positive FDG uptake was taken an indicator of T1b SM2-3, patients with positive FDG uptake had a worse prognosis than patients with negative uptake [5-year overall survival (5-year OS) rate, 75.0% vs 94.9%, Figure 1A]. Similarly, if positive FDG uptake was taken as an indicator for radical esophagectomy or definitive CRT, patients with positive FDG uptake showed a worse prognosis than patients with negative uptake (5-year OS rate, 80.0% vs 94.7%, Figure 1B). Furthermore, if the combined finding of type B2 and B3 and positive FDG uptake was defined as an indicator for radical esophagectomy or definitive CRT, patients with the combined finding showed a worse prognosis than patients without (5- year OS rate, 83.3% vs 94.6 %, Figure 1C).
This study investigated the association between FDG uptake and the invasion depth of superficial ESCC in patients who underwent radical esophagectomy or ER. The results showed that FDG uptake was correlated with circumferential extension, depth of tumor invasion, INF, histological grade, vascular invasion, and lymphatic invasion. Of them, only depth of tumor invasion showed a significant correlation with FDG uptake on multivariate analysis. Recently, several studies reported the clinical importance of FDG-PET in superficial ESCC. Miyata et al[16] reported that FDG uptake correlated with depth of tumor invasion and macroscopic type in 41 surficial ESCCs, and they identified FDG uptake as the only independent risk factor for deep submucosal invasion (OR = 7.407; P = 0.0279). Kita et al[19] reported that FDG uptake correlated with histology, depth of tumor invasion, lymph node metastasis, lymphatic invasion, and vascular invasion in 35 FDG-positive ESCCs of 80 superficial ESCCs. They also demonstrated that, in FDG uptake-positive patients, depth of tumor invasion and histology were prognostic factors. Nakajo et al[23] reported that FDG uptake correlated with tumor length, depth of tumor invasion, macroscopic type, lymph node metastasis, and vascular invasion in 23 of 37 superficial ESCCs. These results were almost concordant with those of the present study, especially depth of tumor invasion, since submucosal deep invasion seems to reflect FDG uptake. From this perspective, Kita et al[19] and Nakajo et al[23] mentioned that FDG uptake can be one of the indicators for choosing between radical esophagectomy and endoscopic treatment.
Nakajima et al[17] took a closer look at the indications for ER of superficial ESCC. They investigated the relationship between tumor depth and FDG uptake in 146 superficial ESCCs before treatment, and they proposed a diagnostic algorithm for superficial ESCC. First, they recommended ME-NBI, and if the lesions are difficult to differentiate between type B1 and type B2, endoscopic ultrasound (EUS) should be considered. If the lesion is difficult to diagnose as T1a-EP/LPM after this two-step examination, FDG-PET should be considered. In cases of negative FDG-PET, ESD should be performed; in cases of positive FDG-PET, radical esophagectomy or definitive CRT should be considered. However, with development of devices and techniques for ESD and methods for preventing esophageal stenosis after ESD, the indications for ESD for superficial ESCC have been increasing[24]. Now, the absolute indication for ESD is, regardless of its size and location, tumor within LPM, and tumor involving MM or SM1 layers is a relative indication for ESD[10,11]. Therefore, we think that the most important judgement for determining the treatment strategy is to distinguish SM2-3 from MM/SM1, not to distinguish EP-LPM from MM/SM1. Superficial ESCC invaded to SM2-3 should not undergo ESD, and that invaded to MM/SM1 should be considered for ESD, because lesions without lymphovascular invasion have little possibility of lymph node metastases.
The present study showed that FDG-PET had high sensitivity of 93.3% to diagnose tumor invasion to SM2-3, but the specificity and PPV were low, at 77.6% and 48.2%, respectively (Table 5). Furthermore, as for the diagnostic performance for tumor invasion to MM with lymphovascular invasion and SM invasion regardless of lymphovascular invasion, that is, superficial ESCC, for which radical esophagectomy or definitive CRT is indicated, the sensitivity decreased to 78.3%, whereas the specificity and PPV improved to 81.4% and 62.1%, respectively. However, these are not fully satisfactory for the clinical situation. Then, the findings of ME-NBI, that is type B2 and B3, were combined with positive FDG uptake, and although the sensitivity did not change, the specificity and PPV improved to 91.5% and 78.3%, respectively (Table 6). In the present study, five of 23 lesions showed positive findings of the previous combined criterion, that is, they were false-positives. Of them, 2 of 5 lesions were superficial spreading type, defined as a superficial type 0-II tumor in which the maximal length of the tumor extends more than 5 cm longitudinally. FDG uptake partially reflects tumor volume, and superficial extensive spreading-type ESCC has a certain tumor volume. Therefore, such tumors can probably be detected by FDG-PET. Superficial extensive spreading-type ESCC is hard to dissect endoscopically and has a high risk of esophageal stricture after ESD, notwithstanding the development of methods to prevent it. Therefore, these lesions could be excluded. The remaining three cases included one case that showed poorly differentiated SCC, another case showed ulceration in the lesion, and the last case showed protruded type. These unique findings may affect the uptake of FDG in superficial ESCC.
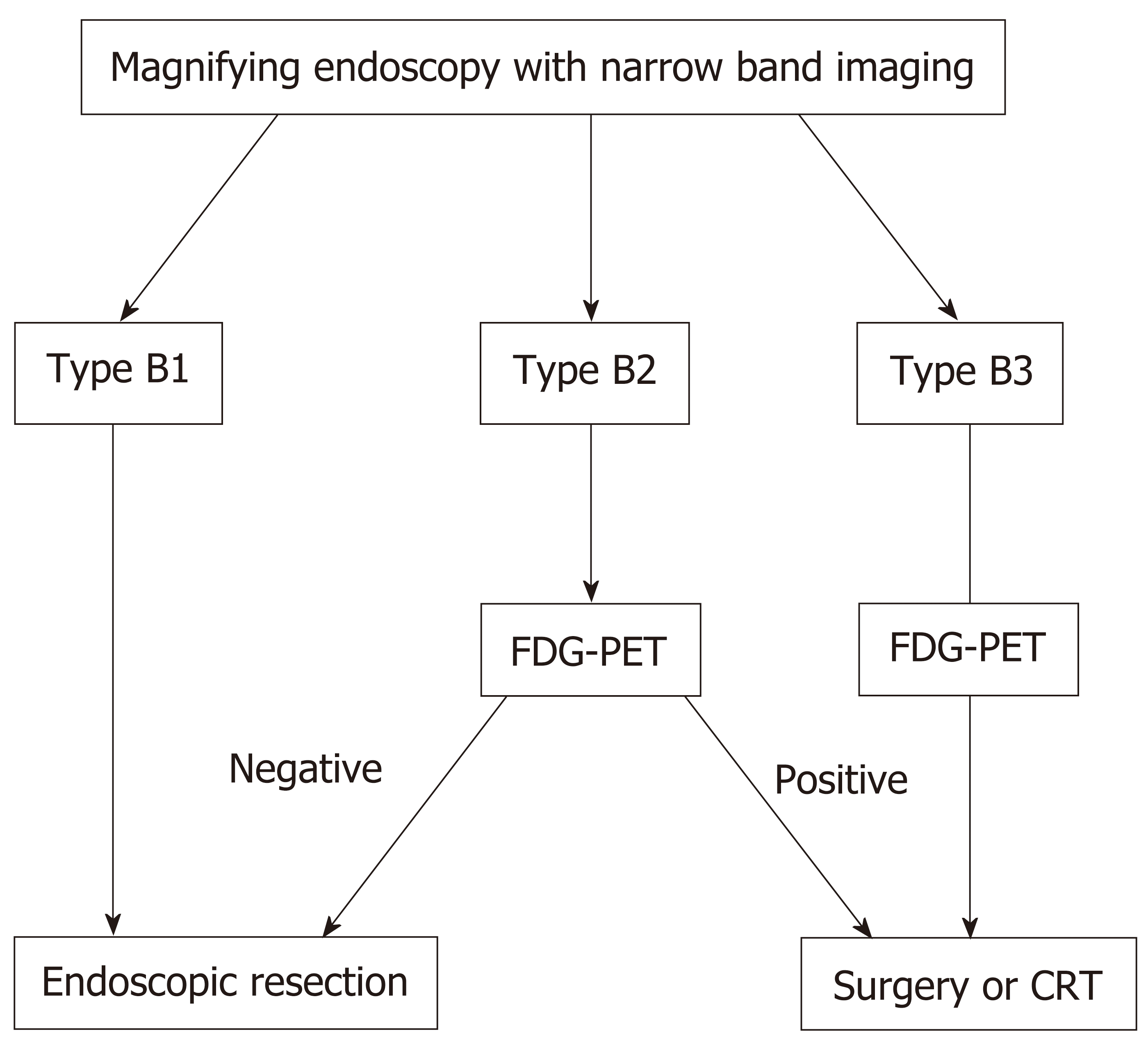
The present study showed that FDG uptake may reflect not only tumor invasion depth, but also lymphovascular invasion in MM. These findings suggest that high FDG uptake indicates proliferation and aggressiveness of the primary tumor. Therefore, high FDG uptake by primary tumors might also be associated with tumor infiltration of deep layers, occult lymph node micro-metastases, and, eventually, predict a poor prognosis. We will propose a diagnostic algorithm for deciding a treatment strategy for superficial ESCC (Figure 2).
In Japan, a Phase II trial to evaluate the efficacy and safety of combined treatment of ER and CRT for clinical stage I (T1b) esophageal cancer (Japan Clinical Oncology Group Study, JCOG0508) has been completed[25]. The final analysis showed that 5-year survival data were comparable to surgery or CRT for clinical stage I ESCC[26]. The results from this trial will be expected to provide a non-surgical treatment option for patients with clinical stage I (T1b) esophageal cancer. The more that the indications for ER of superficial ESCC increase, the greater will be the importance of FDG-PET/CT.
There were several limitations in the present study. First, this was a retrospective study in a single facility. Second, EUS was not performed in this series because EUS is not routinely performed in the staging of esophageal cancer in our institute, only for restricted cases. A recent meta-analysis showed that EUS has a high positive likelihood ratio and can reliably confirm that the cancer is limited to the surface[27], but it requires skill, and the result may depend on the examiner. We consider that FDG-PET is more objective and less invasive than EUS. Third, there was the possibility that endoscopic diagnosis with ME-NBI did not necessarily reflect the deepest area where tumor had invaded.
In conclusion, FDG uptake was correlated with the invasion depth of superficial ESCC. Combined use of FDG-PET and ME-NBI, especially with the microvascular findings of types B2 and B3, is useful to determine the indications for ER.
Recent advances in endoscopic technology, especially magnifying endoscopy with narrow band imaging (ME-NBI) have enabled us to detect superficial esophageal squamous cell carcinoma (ESCC), and 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) have reported to be a useful indicator for decision-making regarding treatment for superficial ESCC. Although, there are not enough reports on association between FDG-PET uptake and clinicopathological characteristics of superficial ESCC.
ME-NBI enabled us to detect superficial ESCC, but determining the appropriate method of resection, endoscopic resection (ER) vs surgical resection, is often challenging. Also, the position of FDG-PET in the decision-making regarding the treatment strategy for superficial ESCC is not clear. We investigated the association between FDG uptake and the clinicopathological characteristics of superficial ESCC and its usefulness of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC.
The aim was to investigate the association between FDG accumulation and the clinicopathological characteristics of superficial ESCC, as well as its utility for determining the treatment strategy for superficial ESCC to avoid unnecessary treatment. Primary endpoint is to evaluate the association between FDG uptake and clinicopathological characteristics of superficial ESCC. Secondary endpoint is to investigate the efficacy of combination of FDG-PET and ME-NBI for determining the treatment strategy for superficial ESCC.
A database of all patients with superficial ESCC who had undergone both ME-NBI and FDG-PET for pre-treatment staging at Aichi Cancer Center Hospital between January 2008 and November 2018 was retrospectively analyzed. FDG uptake was defined positive or negative whether the primary lesion was visualized or could be distinguished from the background, or not. The invasion depth of ESCC was classified according to the classification established by the Japan Esophageal Society.
A total of 82 lesions in 82 patients were included. FDG-PET showed positive uptake in 29 (35.4%) lesions. Univariate analysis showed that uptake of FDG-PET had significant correlations with circumferential extension (P = 0.014), pathological depth of tumor invasion (P < 0.001), infiltrative growth pattern (P < 0.001), histological grade (P = 0.002), vascular invasion (P = 0.001), and lymphatic invasion (P < 0.001). On multivariate analysis, only depth of tumor invasion was independently correlated with FDG-PET/computed tomography visibility (P = 0.018). The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of Type B2 in ME-NBI for the invasion depth of T1a muscularis mucosae (MM) and T1b upper submucosal layer (SM1) were 68.4%/79.4%/50.0%/89.3%/76.8%, respectively, and those of Type B3 for the depth of T1b middle and deeper submucosal layers (SM2 and SM3) were 46.7%/100%/100%/89.3%/90.2%, respectively. On the other hand, those of FDG-PET for SM2 and SM3 were 93.3%/77.6%/48.2%/98.1%/80.5%, respectively, whereas, if the combination of positive FDG uptake and type B2 and B3 was defined as an indicator for radical esophagectomy or definitive chemoradiotherapy (CRT), the sensitivity, specificity, PPV, NPV, and accuracy were 78.3%/91.5%/78.3%/91.5%/87.8%, respectively.
FDG uptake was correlated with the invasion depth of superficial ESCC. Combined use of FDG-PET and ME-NBI, especially with the microvascular findings of Type B2 and B3, is useful to determine whether ER is indicated for the lesion. We will propose a diagnostic algorithm for deciding a treatment strategy for superficial ESCC. First, when the lesion showed that ME-NBI is type B1, the lesion is indication for endoscopic treatment, and it showed type B3, the lesion is indication for surgical resection or CRT. Second, when the lesion showed that ME-NBI is type B2, if FDG-PET is negative, endoscopic treatment is recommended, if FDG-PET is positive, surgical resection or CRT is recommended.
Now, the absolute indication for endoscopic submucosal dissection (ESD) is, regardless of its size and location, tumor within LPM, and tumor involving MM or SM1 layers is a relative indication for ESD. Therefore, the most important judgement for determining the treatment strategy is to distinguish SM2-3 from MM/SM1. So, the results of this study will help the decision-making regarding the treatment strategy for ESCC. However, this study was retrospective setting. So, in the future, prospective study using ME-NBI, FDG-PET and endoscopic ultrasound is needed to confirm the results obtained in this study.
Manuscript source: Invited Manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Demir Y, Surucu E, Tabibian JH S-Editor: Tang JZ L-Editor: A E-Editor: Zhang YL
| 1. | Kubo K, Fujino MA. Ultra-high magnification endoscopy of the normal esophageal mucosa. Gastrointest Endosc. 1997;46:96-97. [PubMed] |
| 2. | Kumagai Y, Inoue H, Nagai K, Kawano T, Iwai T. Magnifying endoscopy, stereoscopic microscopy, and the microvascular architecture of superficial esophageal carcinoma. Endoscopy. 2002;34:369-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 165] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 3. | Kawahara Y, Uedo N, Fujishiro M, Goda K, Hirasawa D, Lee JH, Miyahara R, Morita Y, Singh R, Takeuchi M, Wang S, Yao T. The usefulness of NBI magnification on diagnosis of superficial esophageal squamous cell carcinoma. Dig Endosc. 2011;23 Suppl 1:79-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Inoue H, Kaga M, Ikeda H, Sato C, Sato H, Minami H, Santi EG, Hayee B, Eleftheriadis N. Magnification endoscopy in esophageal squamous cell carcinoma: a review of the intrapapillary capillary loop classification. Ann Gastroenterol. 2015;28:41-48. [PubMed] |
| 5. | Takahashi H, Arimura Y, Masao H, Okahara S, Tanuma T, Kodaira J, Kagaya H, Shimizu Y, Hokari K, Tsukagoshi H, Shinomura Y, Fujita M. Endoscopic submucosal dissection is superior to conventional endoscopic resection as a curative treatment for early squamous cell carcinoma of the esophagus (with video). Gastrointest Endosc. 2010;72:255-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 243] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 6. | Ishihara R, Iishi H, Uedo N, Takeuchi Y, Yamamoto S, Yamada T, Masuda E, Higashino K, Kato M, Narahara H, Tatsuta M. Comparison of EMR and endoscopic submucosal dissection for en bloc resection of early esophageal cancers in Japan. Gastrointest Endosc. 2008;68:1066-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 231] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 7. | Fujishiro M, Yahagi N, Kakushima N, Kodashima S, Muraki Y, Ono S, Yamamichi N, Tateishi A, Shimizu Y, Oka M, Ogura K, Kawabe T, Ichinose M, Omata M. Endoscopic submucosal dissection of esophageal squamous cell neoplasms. Clin Gastroenterol Hepatol. 2006;4:688-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 258] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 8. | Abe S, Iyer PG, Oda I, Kanai N, Saito Y. Approaches for stricture prevention after esophageal endoscopic resection. Gastrointest Endosc. 2017;86:779-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 9. | Oliveira JF, Moura EG, Bernardo WM, Ide E, Cheng S, Sulbaran M, Santos CM, Sakai P. Prevention of esophageal stricture after endoscopic submucosal dissection: a systematic review and meta-analysis. Surg Endosc. 2016;30:2779-2791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 10. | Kitagawa Y, Uno T, Oyama T, Kato K, Kato H, Kawakubo H, Kawamura O, Kusano M, Kuwano H, Takeuchi H, Toh Y, Doki Y, Naomoto Y, Nemoto K, Booka E, Matsubara H, Miyazaki T, Muto M, Yanagisawa A, Yoshida M. Esophageal cancer practice guidelines 2017 edited by the Japan Esophageal Society: part 1. Esophagus. 2019;16:1-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 377] [Cited by in RCA: 390] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 11. | Kitagawa Y, Uno T, Oyama T, Kato K, Kato H, Kawakubo H, Kawamura O, Kusano M, Kuwano H, Takeuchi H, Toh Y, Doki Y, Naomoto Y, Nemoto K, Booka E, Matsubara H, Miyazaki T, Muto M, Yanagisawa A, Yoshida M. Esophageal cancer practice guidelines 2017 edited by the Japan esophageal society: part 2. Esophagus. 2019;16:25-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 233] [Cited by in RCA: 328] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 12. | Kato H, Kuwano H, Nakajima M, Miyazaki T, Yoshikawa M, Ojima H, Tsukada K, Oriuchi N, Inoue T, Endo K. Comparison between positron emission tomography and computed tomography in the use of the assessment of esophageal carcinoma. Cancer. 2002;94:921-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 184] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 13. | Guo H, Zhu H, Xi Y, Zhang B, Li L, Huang Y, Zhang J, Fu Z, Yang G, Yuan S, Yu J. Diagnostic and prognostic value of 18F-FDG PET/CT for patients with suspected recurrence from squamous cell carcinoma of the esophagus. J Nucl Med. 2007;48:1251-1258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 56] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 14. | Brücher BL, Weber W, Bauer M, Fink U, Avril N, Stein HJ, Werner M, Zimmerman F, Siewert JR, Schwaiger M. Neoadjuvant therapy of esophageal squamous cell carcinoma: response evaluation by positron emission tomography. Ann Surg. 2001;233:300-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 260] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 15. | Downey RJ, Akhurst T, Ilson D, Ginsberg R, Bains MS, Gonen M, Koong H, Gollub M, Minsky BD, Zakowski M, Turnbull A, Larson SM, Rusch V. Whole body 18FDG-PET and the response of esophageal cancer to induction therapy: results of a prospective trial. J Clin Oncol. 2003;21:428-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 223] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 16. | Miyata H, Doki Y, Yasuda T, Yamasaki M, Higuchi I, Makari Y, Matsuyama J, Hirao T, Takiguchi S, Fujiwara Y, Monden M. Evaluation of clinical significance of 18F-fluorodeoxyglucose positron emission tomography in superficial squamous cell carcinomas of the thoracic esophagus. Dis Esophagus. 2008;21:144-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 17. | Nakajima M, Muroi H, Yokoyama H, Kikuchi M, Yamaguchi S, Sasaki K, Kato H. 18 F-Fluorodeoxyglucose positron emission tomography can be used to determine the indication for endoscopic resection of superficial esophageal cancer. Cancer Med. 2018;7:3604-3610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Furukawa T, Hamai Y, Hihara J, Emi M, Yamakita I, Ibuki Y, Okada M. Clinical Significance of FDG-PET to Predict Pathologic Tumor Invasion and Lymph Node Metastasis of Superficial Esophageal Squamous Cell Carcinoma. Ann Surg Oncol. 2016;23:4086-4092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Kita Y, Okumura H, Uchikado Y, Sasaki K, Omoto I, Matsumoto M, Setoyama T, Tanoue K, Mori S, Owaki T, Ishigami S, Ueno S, Kajiya Y, Natsugoe S. Clinical significance of 18F-fluorodeoxyglucose positron emission tomography in superficial esophageal squamous cell carcinoma. Ann Surg Oncol. 2013;20:1646-1652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Brierley J, Gospodarowicz M, Wittekind C. International Union Against Cancer: TNM Classification of Malignant Tumours. 8th ed Wiley Blackwell, 2017: 56-63. [DOI] [Full Text] |
| 21. | Oyama T, Inoue H, Arima M, Momma K, Omori T, Ishihara R, Hirasawa D, Takeuchi M, Tomori A, Goda K. Prediction of the invasion depth of superficial squamous cell carcinoma based on microvessel morphology: magnifying endoscopic classification of the Japan Esophageal Society. Esophagus. 2017;14:105-112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 223] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 22. | Japan Esophageal Society. Japanese classification of esophageal cancer. 11th English ed. Tokyo: Kanehara & Co Ltd; 2015; . |
| 23. | Nakajo M, Nakajo M, Tani A, Kajiya Y, Shimaoka S, Matsuda A, Nioh T, Nihara T, Suenaga T, Tanaka S, Shirahama H, Higashi M, Koriyama C. Clinical significance of primary lesion FDG uptake for choice between oesophagectomy and endoscopic submucosal dissection for resectable oesophageal squamous cell carcinomas. Eur Radiol. 2011;21:2396-2407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Ishihara R. Prevention of esophageal stricture after endoscopic resection. Dig Endosc. 2019;31:134-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 25. | Kurokawa Y, Muto M, Minashi K, Boku N, Fukuda H; Gastrointestinal Oncology Study Group of Japan Clinical Oncology Group (JCOG). A phase II trial of combined treatment of endoscopic mucosal resection and chemoradiotherapy for clinical stage I esophageal carcinoma: Japan Clinical Oncology Group Study JCOG0508. Jpn J Clin Oncol. 2009;39:686-689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 26. | Minashi K, Nihei K, Mizusawa J, Takizawa K, Yano T, Ezoe Y, Tsuchida T, Ono H, Iizuka T, Hanaoka N, Oda I, Morita Y, Tajika M, Fujiwara J, Yamamoto Y, Katada C, Hori S, Doyama H, Oyama T, Nebiki H, Amagai K, Kubota Y, Nishimura K, Kobayashi N, Suzuki T, Hirasawa K, Takeuchi T, Fukuda H, Muto M. Efficacy of Endoscopic Resection and Selective Chemoradiotherapy for Stage I Esophageal Squamous Cell Carcinoma. Gastroenterology. 2019;157:382-390.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 139] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 27. | Ishihara R, Matsuura N, Hanaoka N, Yamamoto S, Akasaka T, Takeuchi Y, Higashino K, Uedo N, Iishi H. Endoscopic imaging modalities for diagnosing invasion depth of superficial esophageal squamous cell carcinoma: a systematic review and meta-analysis. BMC Gastroenterol. 2017;17:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |