Published online Oct 14, 2019. doi: 10.3748/wjg.v25.i38.5883
Peer-review started: June 3, 2019
First decision: July 21, 2019
Revised: August 8, 2019
Accepted: September 13, 2019
Article in press: September 13, 2019
Published online: October 14, 2019
Antiviral treatment of patients with chronic hepatitis B (CHB) in the grey zone of treatment comands risk management in order to optimize the health outcome. In this sense, the identification of HBV mutants related with an increased risk of hepatocellular carcinoma (HCC) could be useful to identify subpopulations with potential indication of antiviral treatment.
To analyze the prevalence/persistence of hepatitis B virus (HBV) preS and basal core promoter (BCP)/precore/core variants associated to HCC development in CHB patients in the grey zone.
Work was designed as a longitudinal retrospective study, including 106 plasma samples from 31 patients with CHB in the grey zone of treatment: Hepatitis B e antigen negative, HBV-DNA levels between 12-20000 IU/mL, normal or discordant transaminase levels during follow up and mild/moderate necro-inflammatory activity in liver biopsy or Fibroscan (up to 9.5 kPa). Serum HBV-DNA was tested using the Abbott Real Time HBV Assay and the BCP/precore/core and the hepatitis B surface antigen (HBsAg) coding regions were analyzed in positive samples by PCR/bulk-sequencing to identify the HCC-related HBV mutants.
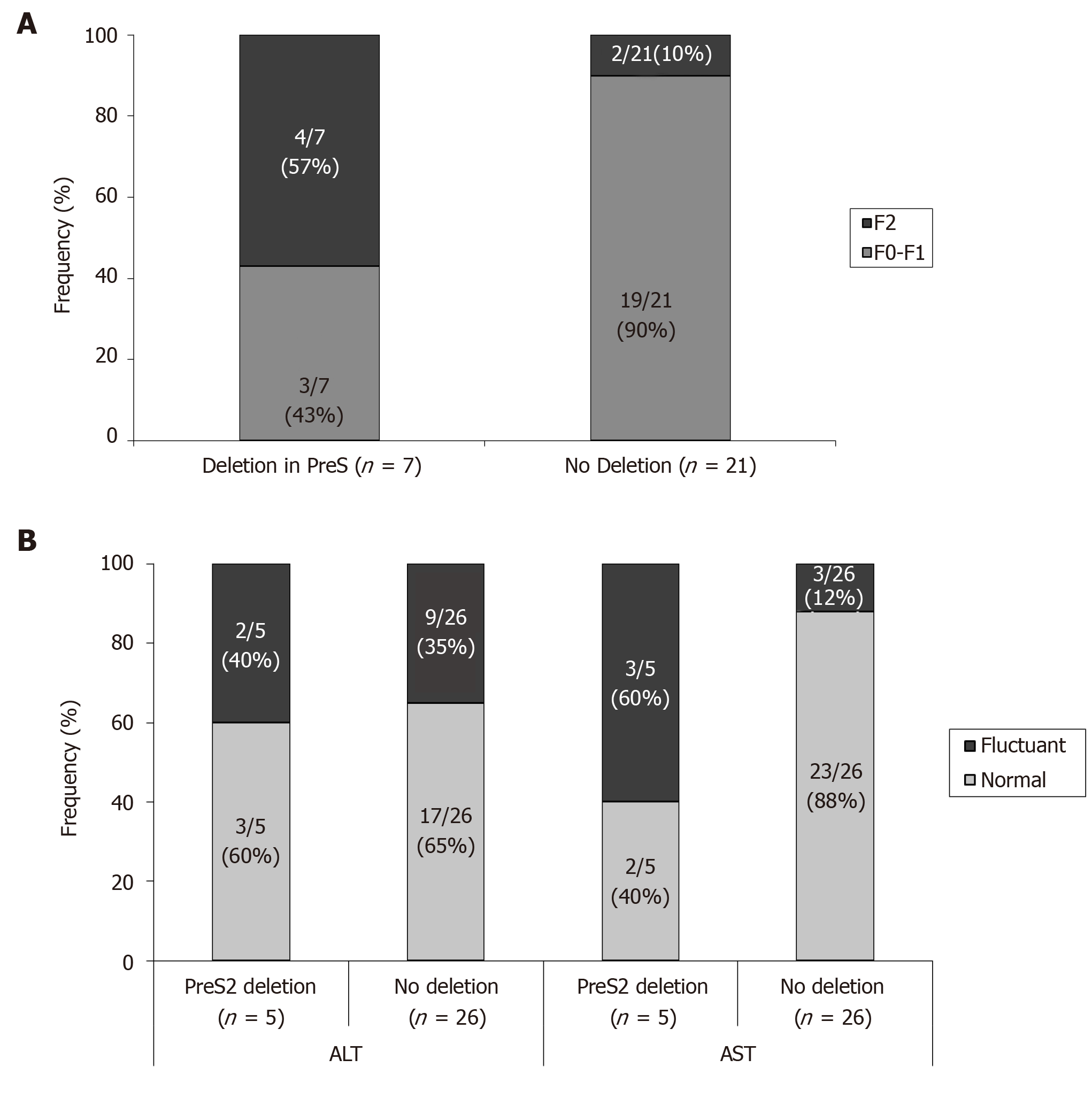
High-risk HCC related mutants were detected in 24 (77%) patients: 19 (61%) in the BCP/precore/core, and 7 (23%) in the HBsAg coding region (2 preS1 and 5 preS2 deletions). The prevalence of preS deletions was genotype-dependent: 3/5 (60%) patients with preS2 deletions and 1/2 with preS1 deletions were infected with the HBV-E genotype. Since HBV-E was the most prevalent in sub-Saharan patients, a correlation between preS deletions and ethnicity was also found: 6/8 (75%) sub-Saharan vs 1/19 (5%) Caucasian patients had preS deletions (P = 0.00016). Remarkably, this correlation was maintained in those patients infected with HBV-A, a minor genotype in sub-Saharan patients: 2/2 patients infected with HBV-A from West Africa vs 0/6 of Caucasian origin had preS deletions. The HCC related variants were the major strains and persisted over time (up to 48 mo). Patients with preS deletions had a significant higher prevalence of F2 fibrosis stage than the negatives (57% vs 10%, P = 0.0078).
HBV genetic analysis of selected populations, like sub-Saharans infected with HBV-E/A genotypes, will allow identification of subpopulations with risk of HCC development due to accumulation of high-risk HBV variants, thus commanding their increased clinical surveillance.
Core tip: The antiviral treatment in patients with chronic hepatitis B in the “grey zone” of treatment is controversial and not clearly indicated. The genetic analysis of hepatitis B virus (HBV) basal core promoter/precore/core and preS regions has shown a high prevalence and persistence of preS deletions in the sub-Saharan population infected with HBV-E/A genotypes. By contrast, Caucasian patients, who have shown a good clinical evolution in previous studies, were negative for these variants. The recognition of these subpopulations warrant to increase the clinical surveillance in order to minimize the risk of liver cancer development due to accumulation of hepatocellular carcinoma-related HBV genetic variants.
- Citation: Gil-García AI, Madejón A, Francisco-Recuero I, López-López A, Villafranca E, Romero M, García A, Olveira A, Mena R, Larrubia JR, García-Samaniego J. Prevalence of hepatocarcinoma-related hepatitis B virus mutants in patients in grey zone of treatment. World J Gastroenterol 2019; 25(38): 5883-5896
- URL: https://www.wjgnet.com/1007-9327/full/v25/i38/5883.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i38.5883
In most cases, patients with chronic hepatitis B (CHB) treated with nucleos(t)ide analogues (NAs) such as tenofovir or entecavir achieve a maintained virological suppression[1-3]. Although the inhibition of viral replication has an evident beneficial effect on the progression of the liver disease, the antiviral treatment does not achieve eradication of the hepatitis B virus (HBV) infection due to the persistence of intracellular covalently closed circular DNA in the nucleus of infected hepatocytes[4]. Thus, therapy is generally recommended for prolonged periods of time and the discontinuation of NAs is controversial because the early interruption of treatment is usually associated with a reactivation of viral replication that may induce severe flares if not controlled by the host immunity, requiring the re-start of the antiviral treatment[5]. According to EASL guidelines treatment may be discontinued in hepatitis B e antigen (HBeAg) negative patients only when hepatitis B surface antigen (HBsAg) loss is achieved or after 3 years of virological suppression[1]. Conversely, AASLD guidelines still recommends lifelong antiviral treatment in patients with HBeAg negative CHB[2].
The indications for CHB treatment are mainly based on the combination of three criteria: serum HBV-DNA levels, alanine aminotransferase (ALT) values and severity of liver disease. Patients should be considered for treatment when they have HBV-DNA levels above 2000 IU/mL, persistent or intermittent serum ALT abnormal levels and severity of the liver disease assessed by liver biopsy (or non-invasive markers validated in HBV-infected patients) showing moderate to severe active necroinflammation and/or at least moderate fibrosis using a standardised scoring system[1,2]. Conversely, there are HBeAg-negative patients with maintained minimal or mild fibrosis, for whom the treatment is not clearly indicated: A) patients with maintained normal ALT levels and low or undetectable serum HBV-DNA: B) patients with marginally elevated ALT and/or HBV-DNA titters ranging 2000-20000 IU/mL; and C) persistently normal ALT and HBV-DNA titters higher than 2000 IU/mL[6,7].
The indication of treatment in these patients, in the so named “grey zone” of treatment, requires to balance the risks and benefits for health outcomes. Although low levels of HBV replication and the absence of significant liver damage are good prognostic factors in the natural history of CHB, it cannot be ruled out that some of these patients may be infected with viral strains with mutations associated with high-risk of hepatocellular carcinoma (HCC) development, which are an additional risk of progression for liver disease[8,9]. There are several studies that associate certain mutations in the HBV genome with an increased risk of HCC development, but the results are controversial because the HBV genotypes or even different HBeAg status have different mutation patterns. One of the mechanisms by which HBV can promote direct carcinogenesis is the ability of wild-type and mutated/truncated viral proteins like hepatitis B x-protein, hepatitis B core-protein (HBc) and preS region (preS) to affect cell functions, activate oncogenic pathways and sensitize liver cells to mutagens[10]. The most common HBV specific mutations are at the preS2 start codon, preS deletions, and point mutations in the basal core promoter (BCP) and in the precore/core coding region[11-13].
For these reasons, the aim of this work is to analyse the prevalence and persistence over time of HBV mutants that predispose to the development of HCC in patients with CHB but without clear indication of treatment, and to analyze their role as a tool in the selection of suitable subpopulations for antiviral treatment.
This work was designed as an observational, longitudinal, retrospective study including 106 plasma samples from 31 CHB patients in the grey zone of treatment: HBeAg negative, HBV-DNA levels between 12-20000 IU/mL, normal or discordant ALT levels during follow up and mild/moderate necro-inflammatory activity in liver biopsy or Fibroscan (up to 9.5 Kpa). Patients with chronic hepatitis C, D or HIV coinfection, or with toxic, alcoholic or autoimmune hepatitis were excluded. The average number of analysed samples per patient was [mean ± standard deviation (SD)] 3.2 ± 1.2 (ranging: 2-6 samples) and the follow-up time period per patient was (mean ± SD) 2.1 ± 2.2 years (ranging: 0.5-8.4 years). No patient was under antiviral treatment for HBV at the time in which samples were collected. The clinical features of the patients, obtained from the full clinical charts, with a follow-up time (mean ± SD) of 6.1 ± 3.9 years (ranging: 0.7-12.8 years) are shown in Table 1. The study was approved by the Ethical Committee of the Hospital Universitario La Paz/Carlos III in Madrid, according to the ethical guidelines of the 1975 Declaration of Helsinki. All the participants received and signed written consent for their participation.
| Demographic data | Value |
| Male/female | 16/15 |
| Caucasian/sub-Saharan/chinese1 | 22/8/1 |
| Age (years: mean ± SD) | 45.1 ± 10.2 |
| Virological data | |
| HBV-DNA positivity (persistent/fluctuant)2 | 28/3 |
| HBV-DNA titters (IU/mL: Mean ± SD)3 | 1.4 × 103 ± 3.5 × 104 |
| Clinical data4 | |
| Persistently normal/marginally altered AST or ALT | 18/13 |
| Persistently normal/marginally altered AST | 25/6 |
| Persistently normal/marginally altered ALT | 20/11 |
| AST (IU/L: mean ± SD) | 25.6 ± 9.5 |
| ALT (IU/L: mean ± SD) | 28.5 ± 14.3 |
| Fibrosis stage (Fibroscan) | |
| F0-F1 | 22 |
| F2 | 6 |
| Not available | 3 |
Serum HBV-DNA was quantified using the Abbott Real Time HBV Assay (Abbott Laboratories, Abbott Park, IL, United States). In positive samples, the viral genome was purified from 200 L of serum using the QIAmp DNA Kit (QIAGEN GmbH, Hilden, Germany), and BCP/precore/core and HBsAg coding regions were amplified using the PCR Master Mix (Promega Corporation, Fitchburg, WI, United States) using genotype-specific primers.
PCR products were purified using the Illustra Exo ProStar (GE Healthcare Life Sciences, Fairfield, Connecticut, United States), sequenced using Bright Dye Terminator Cycle Sequencing (NimaGen, Nijmegen, Netherlands) and analyzed with the DNA sequencer ABI PRISM 3730xl DNA Analyzer (Applied Biosystems, Foster City, CA, United States).
Analysis of the DNA sequences was done using Lasergene SeqMan Pro V7.1.0 (ADNSTAR, United States) software. The phylogenetic relations were established using the MEGA version 4.0 software (Center for Evolutionary Medicine and Informatic, Tempe, AZ, United States).
Assignation of the HCC risk development associated to each HBV mutation was performed according to the previously published data[11,13] (Table 2).
| HCC risk | HBV region | Genetic variant |
| High | ||
| PreS2 | PreS2 deletions | |
| BCP | C1653T, T1753V (A/C/G), A1762T + G1764A | |
| Precore | G1896A, G1899A | |
| Suspect | ||
| PreS1 | PreS1 deletions | |
| PreS2 | T53C | |
| BCP | C1773T | |
| Precore | A1846T | |
| Core | C1914G, C2289A | |
| Minor | ||
| PreS1 | T3098C, T3139A | |
| PreS2 | PreS2 start codon | |
| S | T766A, T791A | |
| BCP | T1674C, G1727, C1741, C1761, C1766T, T1768A | |
| Precore | C1858T, G1862T | |
| Core | C1909, A1934T, C2002T, T2003A, C2100A, A2159G, A2189C, A2246C | |
HBV genotype was analyzed in all cases using the line probe assay INNO-LiPA HBV Genotyping assay (Innogenetics N.V., Ghent, Belgium). Patients with negative results in this technique were typed by the sequence analysis of HBsAg coding region using the Geno2pheno[HBV] open access of the Genafor website (www.genafor.org).
Data were analyzed using the SPSS v13 software (SPSS Inc. North Chicago, IL, United States) on an intention to treat (ITT) basis. All parameters expressed as absolute number or percentages were analyzed using the Spearman´s ran-correlation, the Wilcoxon matched-pairs and the U Mann-Whitney systems. The mean comparisons were performed using the Student-t test.
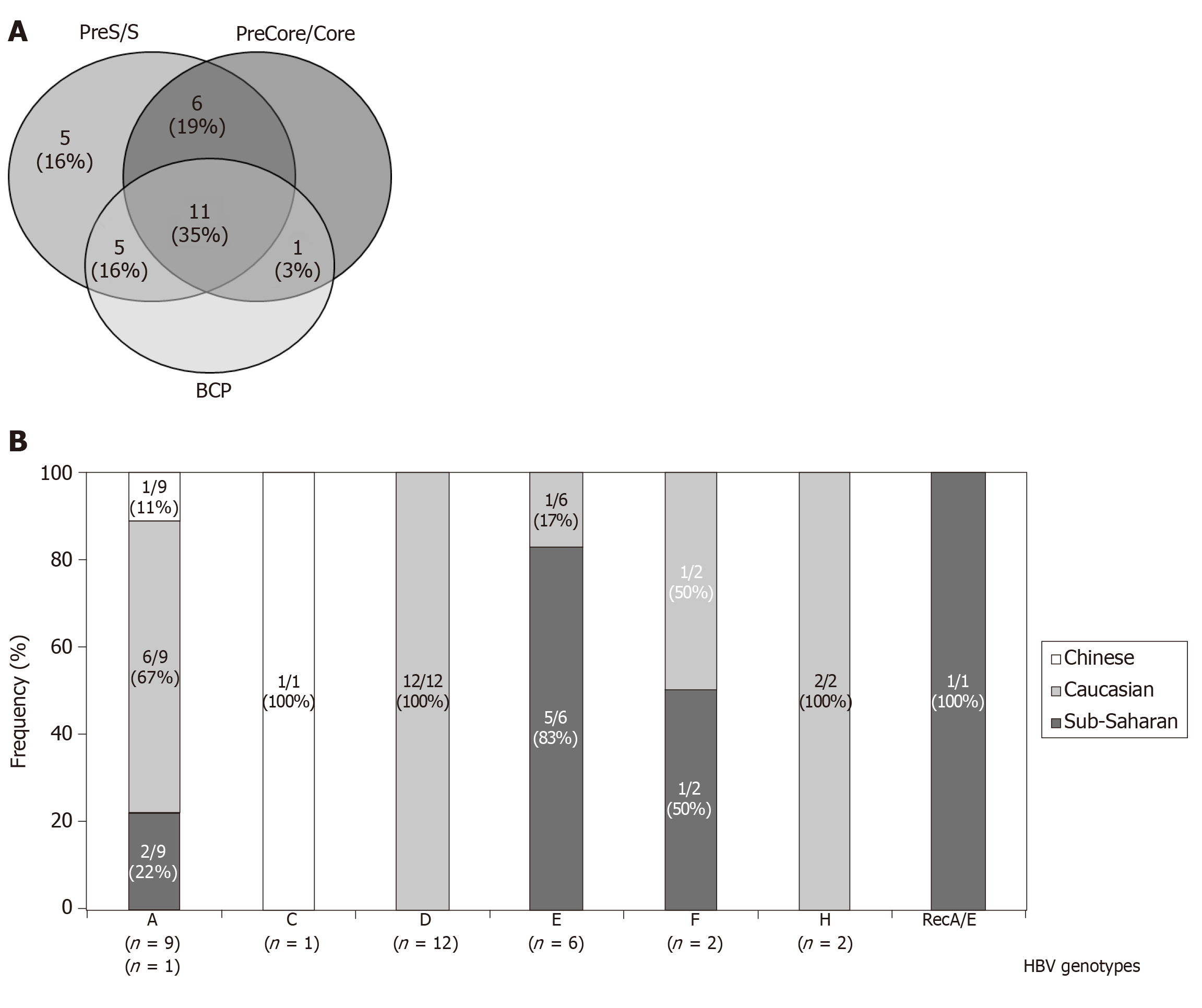
In only 3 (10%) patients no amplification of any of the HBV analyzed regions was achieved. In the remaining 28 patients the efficacy of the HBV-DNA amplification was different depending on the region of the HBV genome analyzed. Thus, we have data from the complete HBsAg and BCP/precore/core regions in at least one sample of the follow-up in 11 (35%) patients, and partial information in 17 (55%) patients (Figure 1A). HBV-DNA titters were significantly higher in the samples positive for the in-house PCR techniques than in the negatives (mean ± SD: 7.6 × 103 ± 1.3 × 104 IU/mL vs 3.1 × 103 ± 4.6 × 103 IU/mL, respectively. P = 0.016).
HBV genotype was determined in 30 (97%) patients [12 (39%) by LIPA and 27 (87%) by HBsAg sequence analysis]: 9/31 (29%) were infected with HBV-A genotype, 1 (3%) with HBV-C, 12 (39%) with HBV-D, 6 (19%) with HBV-E, 2 (6%) with HBV-F, 2 (6%) with HBV-H and 1 (3%) with a recombinant HBV-A/E. Serial changes of the genotyping results during the follow-up were observed in 4 patients: One sub-Saharan patient (who evolve from E to F genotypes), one Chinese (A to C), and 2 Caucasians (from D to A and from A to H, respectively). Differences in the genotype distribution among ethnic groups, especially in the frequency of D and E genotypes, were observed (Figure 1B). In only one case, corresponding to the A/E recombinant, discrepancies between LIPA and HBsAg analysis were remarkably noted.
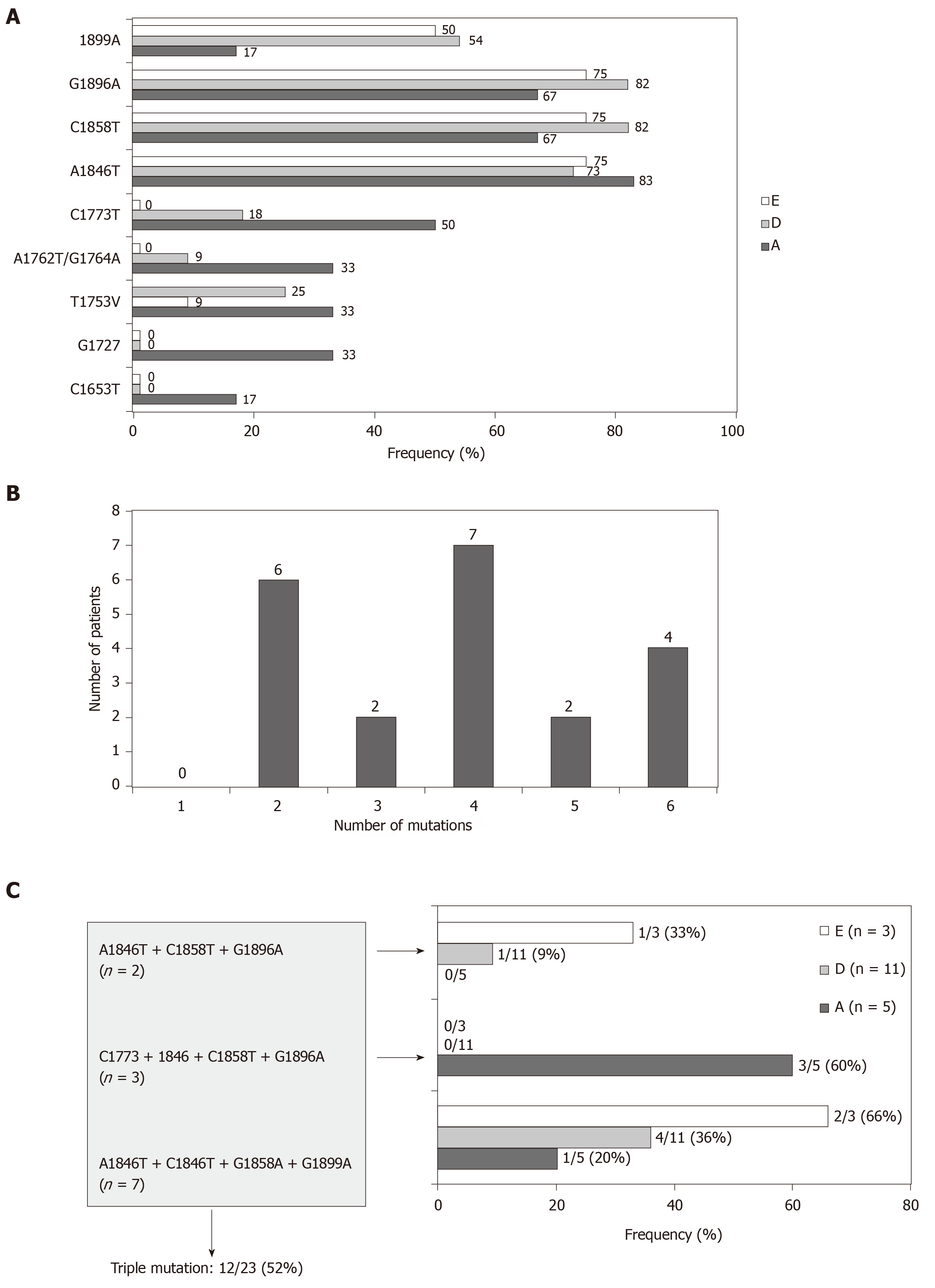
In 21/23 (91%) patients in whom the BCP and/or precore/core regions were amplified, the presence of the most common genetic variants related with the HBeAg negative/anti-HBe status was confirmed. Only partial sequence data (BCP or precore/core sequences) were available for the remaining 2 patients lacking confirmation of these variants. The frequency of mutants related to the HBeAg negative status was higher in the precore/core region than in the BCP in all genotypes, especially in genotypes D and E (Figure 2A). Thus, in 10 patients with mutations in the precore/core region no changes were observed in the BCP region. In addition, coexistence of several mutations in the BCP/precore/core region was observed in the 21 patients (Figure 2B), with special importance for the combination of the A1846T + C1858T + G1896A mutations, which was present in the majority of the patients irrespective of the genotype (Figure 2C).
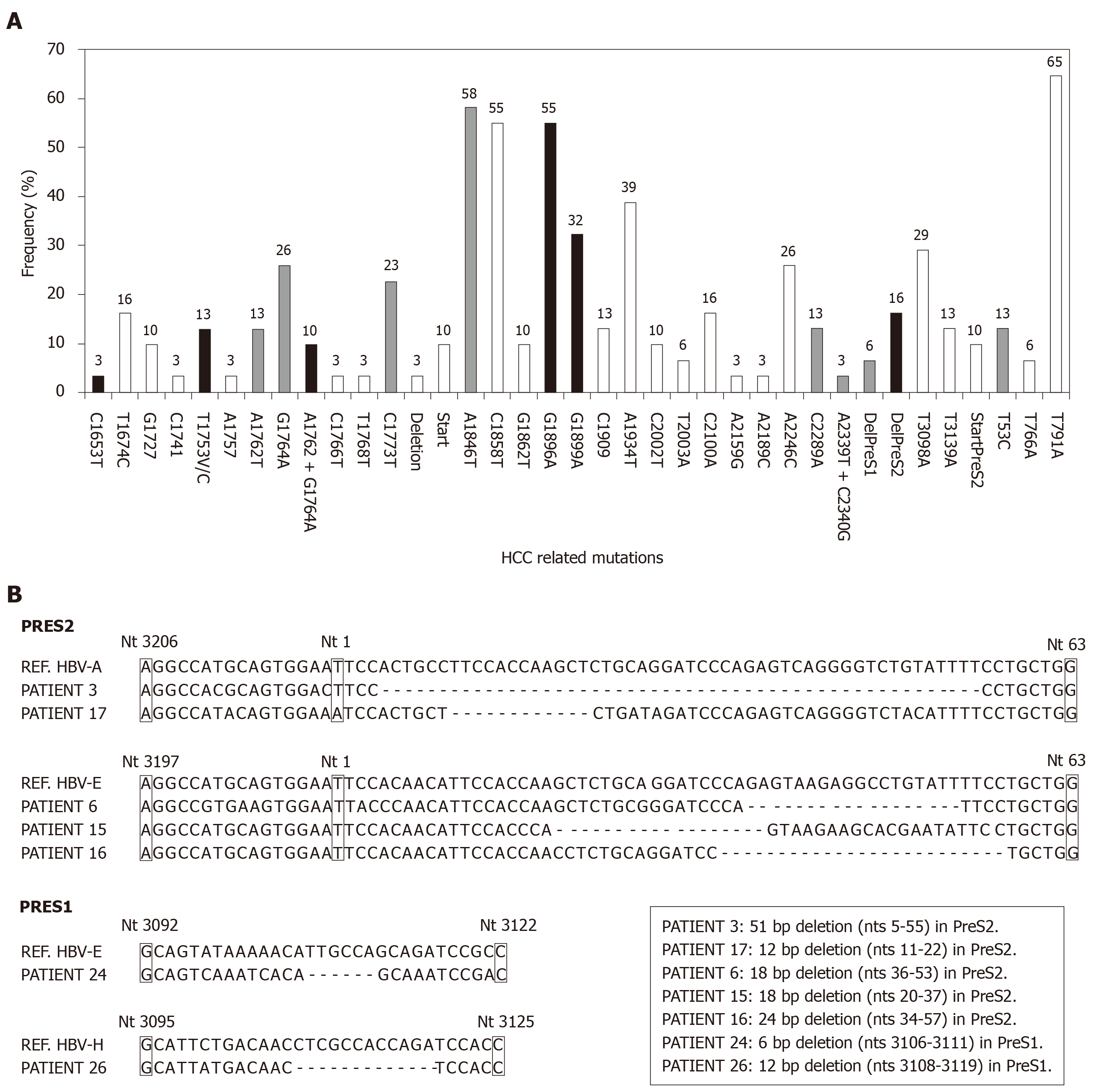
BCP/precore/core region: Taken globally, 19 (61%, on an ITT basis) patients have any high-risk variant (C1653T, T1753V, A1762T/G1764A, G1896A and G1899A) and this percentage increases up to 21 (68%) patients by including the genetic variants with suspect correlation with HCC (C1773T, A1846T, C1914G, C2289A and A2339T+C2340G) in the BCP or precore/core regions. The distribution of the individual mutations in the study population is shown in Figure 3A. Although the presence of single high-risk mutations was the most frequent situation (9 patients), the existence of coinfection with several high-risk mutations was also a common event (Figure 4A).
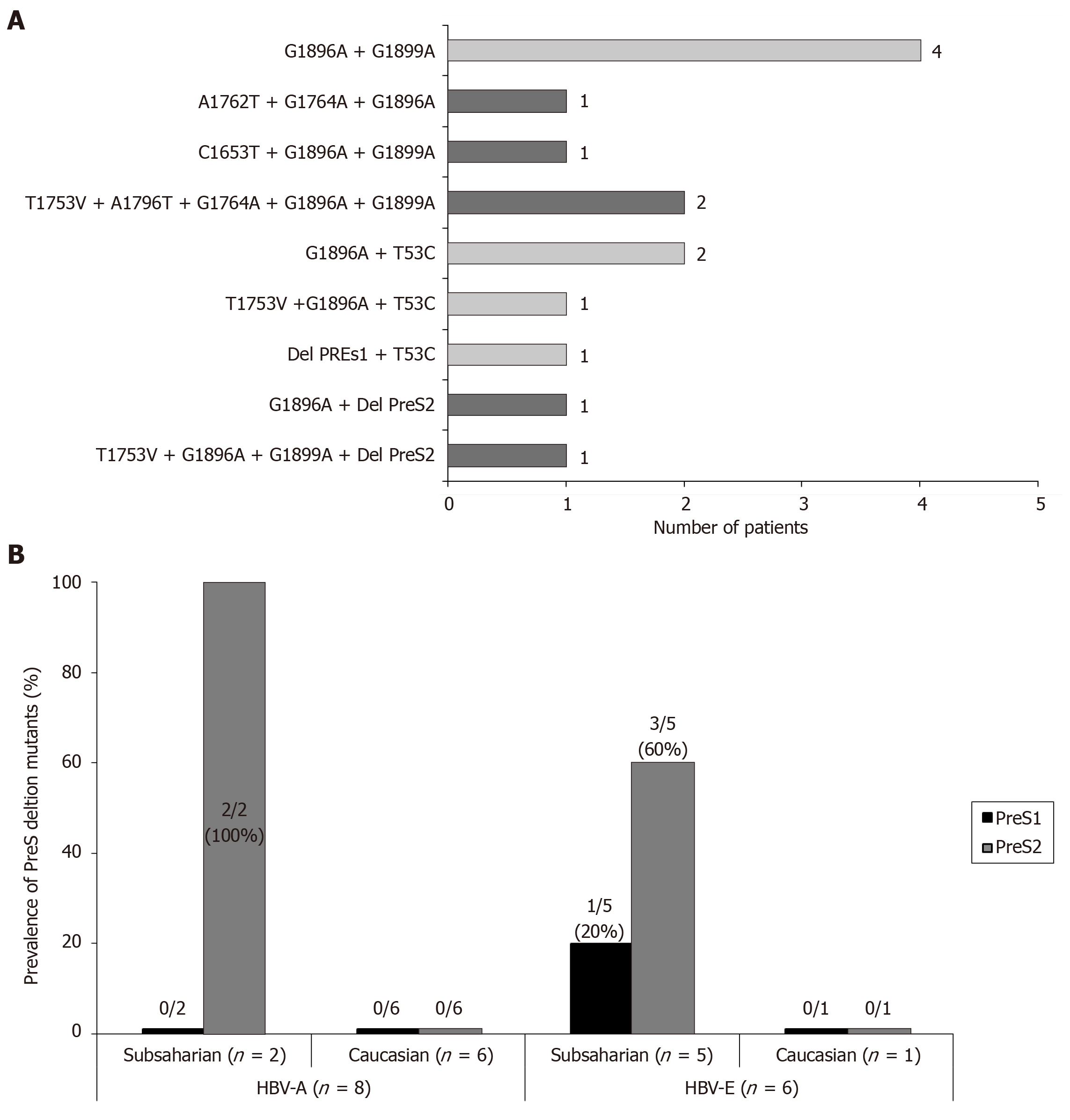
PreS1/preS2/S region: PreS1 deletions were found in 2 (6%), and preS2 deletions in 5 (16%) and the T53C mutation in 4 (13%) patients, respectively. The distribution of the individual mutations in the study population is shown in Figure 3A and the sequences of the deletions of the preS region in Figure 3B. In only 1/10 patient the simultaneous presence of preS1 deletion and the T53C mutation was confirmed. Moreover, the previously described mutation in the preS2 start codon was identified in 3 patients (1 HBV-A, 1 HBV-E and 1 HBV-F). A high correlation between lack of start codon and the preS2 deletion was found, with coexistence of both changes in 2 of the 3 patients with the preS2 start codon mutation. The distribution of HCC related mutations in the HBsAg coding region was genotype-dependent. In this way, 3/5 (60%) patients with preS2 deletions and 1/2 patient with preS1 deletions were infected with the HBV-E genotype. Moreover, it is very remarkable the correlation of these preS deletions with ethnicity: 6/8 (75%) sub-Saharan had preS deletions vs 1/19 (5%) of the Caucasian patients (P = 0.00016). This correlation was observed not only in the HBV-E, which is the predominant genotype in sub-Saharan population, but also in the HBV-A genotype, that is minoritarian within this ethnic group. So, among HBV-A infected patients, only those from West Africa (n = 2) had preS deletions, but none of the patients (n = 6) with Caucasian origin (Figure 4B). In 5/10 (50%) patients with high or medium risk mutations in the HBsAg, data of genetic analysis of the precore/core region were also available. In 2/5 (40%) patients with high-risk deletion in the preS2 coding region, and in 3/4 (75%) patients with the T53C substitution, were simultaneously detected high-risk mutations in the BCP/precore region. Especially significant was the sub-Saharan patient infected with an HBV-E genotype who had a preS2 deletion and was simultaneously positive for 3 high-risk mutations in the BCP/precore/core region (T1753V, G1896A and G1899A) (Figure 4A).
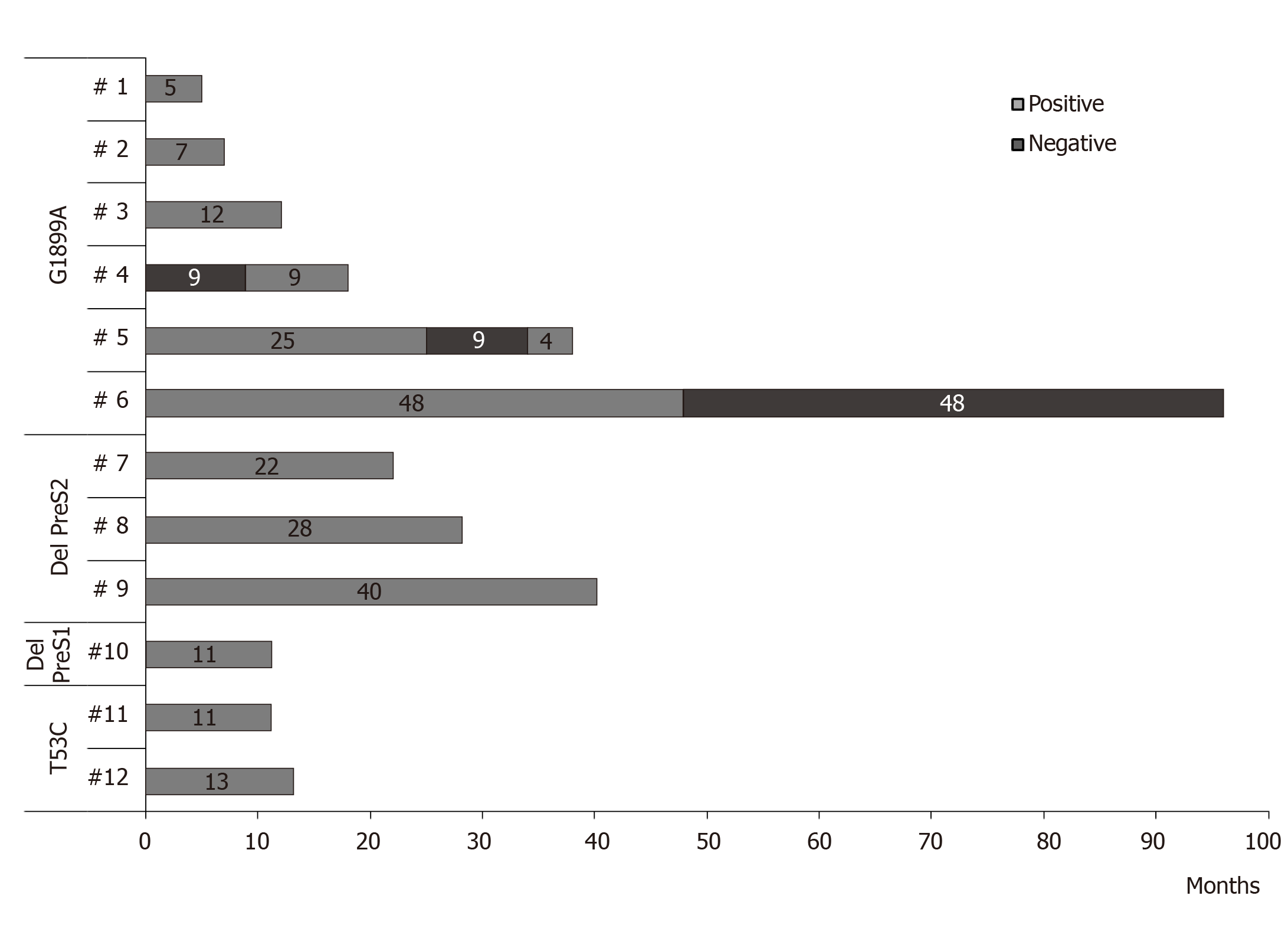
Persistence of HCC related HBV strains: In 12 patients with HCC related mutations, it was possible to analyze the persistence of these changes over time by using bulk sequencing [follow-up time (mean ± SD): 24.9 ± 24.0 mo; ranging: 5-96 mo]. In 11 cases, the mutations persist during the complete follow-up period, or punctually disappear to latter re-appear in the last samples tested. In the remaining case, the G1899A mutation disappears in stable manner. No changes in the pattern of preS2 deletions were observed in none case (Figure 5).
Liver function tests: Although all the patients are in the grey zone of treatment, with low-moderate fibrosis levels (F0-F2, according the selection criteria of patients), it was observed a significant higher percentage of patients with F2 stage of fibrosis in patients with preS deletions than in the negative ones (Figure 6A). In addition, the behaviour of aspartate aminotransferase (AST) and ALT was quite different between patients with or without preS2 deletions. So, the percentage of patients of patients with fluctuant AST or ALT trend to be higher in patients with preS2, although these differences reached significant values only when analyzing AST behaviour (Figure 6B). Only one patient, with sub-Saharan origin, infected with HBV-E genotype, and with a preS2 deletion, reported alpha fetoprotein abnormal values at the end of follow-up, although at the moment of its inclusion in this work no radiologic evidences of any neoplasic event was confirmed.
In this work we performed a longitudinal retrospective study to analyze the prevalence and persistence over time of HBV mutants related with risk of HCC development in HBeAg-negative/anti-HBe-positive patients within the grey zone of treatment, in order to identify subpopulations susceptible to be treated.
Our results confirm the presence of high-risk HCC mutations in BCP/precore or preS regions, in 19 (61%) patients, using a statistical analysis of ITT, with a higher prevalence in the BCP/precore that in the preS region. The most frequently detected high-risk mutations in the BCP/precore region were the G1896A and G1899A. Indeed, finding these mutations in this group of patients is common and frequently related with the Mediterranean variants in patients with HBeAg-negative serological profile[14]. Thus, the G1896A mutation introduces a stop codon at position 28 of the HBc protein that prevents the synthesis of HBeAg[15,16]. The simultaneous presence of A1762T and G1764A mutations that are related to a decrease in HBeAg levels[17-19] and an increase of the HBV-DNA titters, was also confirmed in 10% of the patients[20]. The prevalence of these variants in our cohort of patients was genotype-dependent: BCP variants (C1653T, T1753V, A1762T/G1764A and C1773T) were more frequent in HBV-A genotype, as previously described, except for the C1653T mutation, which was more frequent in HBV-D. Precore/core variant G1899A was more frequent in HBV-E genotype in our cohort, tallying with the published data[21], but also found in a high proportion of patients infected with HBV-A genotype. The prevalence of the G1896A mutation in our patients is consistent with other studies carried out in Spanish population, in which this variant was found with similar frequency in genotypes A and D[22], contrasting with other works conducted in different geographical regions, in which no correlation with the HBV-A genotype was found[23,24]. These discrepancies probably reflect that the HBeAg-negative serological profile may present a different genetic basis in at least some HBV genotypes, with a greater representativeness than in other populations of the G1896A mutation in Mediterranean population, in which this serological pattern appears be more related to the simultaneous presence of A1762T/G1764A double mutation[22].
Although the BCP and precore/core variants has been usually associated to HCC development in meta-analysis studies, a recent work performed in HBeAg-negative Spanish Caucasian patients in the grey zone without antiviral treatment reflects that, in the long term, their clinical evolution is favorable, without progression of liver disease or HCC development in the vast majority of them[25]. Considering that the main objective of antiviral treatment is to prevent the fibrosis progression, these data support the idea that treatment does not provide substantial benefits in this group of patients. In this case, the low progression of HBV disease can be related with the low viral replication levels.
HBV-DNA titters have been classically considered as a prognostic marker of HCC development risk[26]. The maintenance of HBV-DNA levels undetectable or below 2,000 IU/mL can compensate the risk of harbouring HCC-related variants. However, it cannot be ruled that the accumulation of mutations in both BCP/precore/core region and other viral regions like HBsAg, maintained over time, in combination with persistence of viral replication, even at low or moderate levels are risk factors for HCC development[9]. For this reason, we have focused our work on the analysis of the prevalence and persistence over time of the BCP/precore/core and HBsAg regions mutants, present as main strains of HBV detectable by means of bulk sequencing techniques.
The prevalence of preS mutations, which have been correlated with HCC in previous meta-analysis, showed a strong correlation in our cohort with the geographical origin and, in consequence, with the HBV genotypes: 75% of our sub-Saharan patients, the majority infected with HBV-E genotype, had deletions in the preS1 or preS2 regions. In addition, the only 2 sub-Saharan patients infected with HBV-A had also deletions in preS2, while this change was not found in any of the remaining patients infected with HBV-A, all of them with Caucasian origin. HBV-E genotype has not been extensively studied until now. Previous studies performed in Germany confirm our results showing that patients in the grey zone of treatment infected with the HBV-E genotype have a higher prevalence of preS1 and preS2 deletions in comparison with HBV-A and D genotypes[27]. However, the absolute prevalence rate of these mutations in HBeAg negative patients differs between different studies, ranging from 16% in the German series to 75% in our cohort. The phylogenetic analysis of HBV-E strains from different regions of Africa, indicates that the spread of HBV-E genotype in West Africa is a relatively recent event, as reflected by the conspicuously low genetic diversity of this genotype despite the higher prevalence in this geographical region[28,29]. This fact together with the high degree of isolation and scattering of human populations in these regions favours the appearance of viral strains relatively stable over time, limited to specific geographical locations. In this sense, it must be noted that the country origin of the sub-Saharan patients included in our study is located in the South/Central regions of West Africa, corresponding to the area in which the genetic variability of HBV-E is lower, among the countries with high prevalence of this viral genotype[30,31]. In this context, the striking high proportion of preS deletions found in sub-Saharan patients in our series could reflect the existence of isolated viral strains of HBV-E in this region with a higher prevalence than that described for neighbouring countries. It may constitute a subpopulation of patients with increased risk of developing HCC by simultaneously sharing high-risk mutations in different regions of HBV genome.
Especially significant was the persistence of the high-risk mutations over time, as major strains of the viral quasispecies, as demonstrated by its identification with bulk sequencing techniques. As regards amino acid substitutions, these were generally very stable, although changes in the composition were observed. Thus, G1899A mutation remained continuously detectable during a follow-up period of up to 48 mo, with sporadic disappearances of the mutant strain in 3 patients and a definitive loss in only 1 patient. This persistence of point mutations in the BCP/precore/core region can be related to the low level of viral replication, which hinders the appearance and selection of mutations that restore the wild-type genotype in the population. Regarding deletion mutations in the preS region, these were maintained without sporadic eliminations during the follow-up in all the patients who carried them, with proven periods of up to 40 mo. The persistence, without fluctuations, of these deletions seems to influence the presence in the geographical origin of the patients in which these strains are majority. Under these conditions, exclusive infection with deleted strains would make the emergence of wild-type strains impossible due to the absence of a genetic template on which to act. The persistence over time of these mutations favours their accumulation, as major strains, in chronic infected patients. Thus, it was possible to confirm in our patients, not only the coexistence of the mutations A1762T/G1764A and G1896A/G1899A widely documented and associated with a high-risk of carcinogenesis, but also the accumulation of 3-5 risk mutations in other 5 patients, including a case that presented 3 risk mutations in the BCP/precore/core in combination with a deletion in the preS region. In any case, the persistence and accumulation over time of these mutations may have clinical relevance, since it perpetuates the molecular processes by which these viral variants favour the development of HCC.
The selection in this work of patients in the grey zone of treatment, with low-moderate fibrosis levels and normal or marginally elevated transaminase levels, makes it difficult to evaluate unless through a long-term follow-up, the possible harmful effect of the presence of these mutations on the progression of liver disease. However, and even recognizing the limitations of transient elastometry techniques for discriminate patients with low or moderate stages of fibrosis, our data showed a higher prevalence of F2 stage in patients with preS2 deletion than in those in which this mutation was not detected. On the other hand, the behaviour of AST and ALT was quite different between both types of patients, with is a higher percentage of patients with fluctuant AST or ALT levels during follow up in patients harbouring preS deletions.
These results focus an increased risk of HCC development in sub-Saharan patients. Africa represents one of the most HBV endemic regions in the world and HCC mainly due to CHB infection is a major cause of premature death, suggesting that viral strains located in Africa, which mostly belong to HBV-A, are more likely to cause HCC[32]. In this way, occult HBV infection was shown to be present in 75% of Black Africans with HCC, and genotype A was shown to be 4.5 times more likely than other genotypes to cause HCC in Black Africans, whilst tumours occurred at a significantly younger age[33]. Our findings also correlate a higher risk of HCC development with HBV-E, which almost exclusively occurs in African people. Moreover, it has been linked the distribution of HBV genotype E infection with African countries with high incidences of HCC[34].
In conclusion our data indicate that the presence of preS mutations should be assessed in patients with sub-Saharan origin, especially if they are infected with HBV-E and HBV-A genotype, in order to identify subpopulations of patients in which the antiviral treatment can be indicated to minimize the risk of HCC due to accumulation of high-risk HBV genetic variants. In this sense, the development of detection systems for deletion mutations in preS region is technically feasible with conventional techniques of Molecular Biology available for most centers. Its use, especially if it is restricted to preselected subpopulations, can help the treatment decision in patients in grey area of treatment.
The most important limitations of this study are: (1) The number of patients. It must be increased to elucidate the significance of prevalence data of some mutations; (2) The necessity to perform longitudinal studies with longer follow-up periods to clarify the progression of liver disease in this kind of patients. However, our data confirm the accumulation and persistence of HBV mutants related with HCC in different regions of HBV genome especially in the subpopulation of sub-Saharan origin.
The indication of treatment in patients with chronic hepatitis B in the grey zone is not clear, and it is necessary to balance the risks and benefits for health outcomes, including the evaluation of the risk of hepatocellular carcinoma development.
To optimize the management of patients in the grey zone of treatment in order to identify suitable subpopulations for antiviral treatment.
To analyze the prevalence, and persistence over time, of hepatitis B virus (HBV) mutants that predispose to the development of hepatocellular carcinoma in patients in the grey zone of treatment.
We analyzed the presence of basal core promoter/precore/core and preS deletion mutants related with hepatocellular carcinoma development in 106 samples from 31 patient in the grey zone of treatment.
A significant number of the patients analyzed in this work shows hepatocellular carcinoma related mutations. All these hepatocellular carcinoma related mutants are major viral strains and persist over time. Some patients have hepatocellular carcinoma related mutants in basal core promoter/precore/core and preS regions simultaneously. The presence of preS deletions is associated with sub-Saharan subpopulations infected with HBV-A and HBV-E genotypes.
The presence of preS mutations should be assessed in patients with sub-Saharan origin, especially if they are infected with HBV-E and HBV-A genotype, in order to identify subpopulations of patients in whom the antiviral treatment could be indicated to minimize the risk of hepatocellular carcinoma due to accumulation of high-risk HBV genetic variants.
The analysis of preS deletions could be indicated in the management of sub-Saharan patients in grey zone of treatment. Analysis of higher sample size populations during long-time longitudinal follow-up should be further performed in these patient subpopulation.
The authors thank Dr. Pablo Castán for his careful language assistance.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Spain
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Hann HW, Iliescu L S-Editor: Wang J L-Editor: Filipodia E-Editor: Qi LL
| 1. | European Association for the Study of the Liver. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67:370-398. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2771] [Cited by in F6Publishing: 3132] [Article Influence: 447.4] [Reference Citation Analysis (0)] |
| 2. | Terrault NA, Bzowej NH, Chang KM, Hwang JP, Jonas MM, Murad MH; American Association for the Study of Liver Diseases. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261-283. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1532] [Cited by in F6Publishing: 1460] [Article Influence: 182.5] [Reference Citation Analysis (2)] |
| 3. | Ghany MG. Current treatment guidelines of chronic hepatitis B: The role of nucleos(t)ide analogues and peginterferon. Best Pract Res Clin Gastroenterol. 2017;31:299-309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 50] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 4. | Yang HC, Kao JH. Persistence of hepatitis B virus covalently closed circular DNA in hepatocytes: molecular mechanisms and clinical significance. Emerg Microbes Infect. 2014;3:e64. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 92] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 5. | Marciano S, Gadano A. Why not to stop antiviral treatment in patients with chronic hepatitis B. Liver Int. 2018;38 Suppl 1:97-101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Papatheodoridis GV. Hepatitis B virus treatment: Which patients can have treatment deferred? Clin Liver Dis (Hoboken). 2013;2:15-17. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Gish RG, Given BD, Lai CL, Locarnini SA, Lau JY, Lewis DL, Schluep T. Chronic hepatitis B: Virology, natural history, current management and a glimpse at future opportunities. Antiviral Res. 2015;121:47-58. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 176] [Cited by in F6Publishing: 183] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 8. | Lin CL, Kao JH. Risk stratification for hepatitis B virus related hepatocellular carcinoma. J Gastroenterol Hepatol. 2013;28:10-17. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 9. | Papatheodoridis GV, Chan HL, Hansen BE, Janssen HL, Lampertico P. Risk of hepatocellular carcinoma in chronic hepatitis B: assessment and modification with current antiviral therapy. J Hepatol. 2015;62:956-967. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 320] [Cited by in F6Publishing: 357] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 10. | Levrero M, Zucman-Rossi J. Mechanisms of HBV-induced hepatocellular carcinoma. J Hepatol. 2016;64:S84-S101. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 479] [Cited by in F6Publishing: 571] [Article Influence: 71.4] [Reference Citation Analysis (0)] |
| 11. | Liao Y, Hu X, Chen J, Cai B, Tang J, Ying B, Wang H, Wang L. Precore mutation of hepatitis B virus may contribute to hepatocellular carcinoma risk: evidence from an updated meta-analysis. PLoS One. 2012;7:e38394. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Liu S, Zhang H, Gu C, Yin J, He Y, Xie J, Cao G. Associations between hepatitis B virus mutations and the risk of hepatocellular carcinoma: a meta-analysis. J Natl Cancer Inst. 2009;101:1066-1082. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 279] [Cited by in F6Publishing: 314] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 13. | Yeung P, Wong DK, Lai CL, Fung J, Seto WK, Yuen MF. Association of hepatitis B virus pre-S deletions with the development of hepatocellular carcinoma in chronic hepatitis B. J Infect Dis. 2011;203:646-654. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 81] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 14. | Lazarevic I. Clinical implications of hepatitis B virus mutations: recent advances. World J Gastroenterol. 2014;20:7653-7664. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 96] [Cited by in F6Publishing: 91] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 15. | Brunetto MR, Stemler M, Bonino F, Schodel F, Oliveri F, Rizzetto M, Verme G, Will H. A new hepatitis B virus strain in patients with severe anti-HBe positive chronic hepatitis B. J Hepatol. 1990;10:258-261. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 175] [Cited by in F6Publishing: 179] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 16. | Carman WF, Jacyna MR, Hadziyannis S, Karayiannis P, McGarvey MJ, Makris A, Thomas HC. Mutation preventing formation of hepatitis B e antigen in patients with chronic hepatitis B infection. Lancet. 1989;2:588-591. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 852] [Cited by in F6Publishing: 835] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 17. | Buckwold VE, Xu Z, Chen M, Yen TS, Ou JH. Effects of a naturally occurring mutation in the hepatitis B virus basal core promoter on precore gene expression and viral replication. J Virol. 1996;70:5845-5851. [PubMed] [Cited in This Article: ] |
| 18. | Günther S, Piwon N, Will H. Wild-type levels of pregenomic RNA and replication but reduced pre-C RNA and e-antigen synthesis of hepatitis B virus with C(1653) --> T, A(1762) --> T and G(1764) --> A mutations in the core promoter. J Gen Virol. 1998;79:375-380. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 88] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 19. | Laras A, Koskinas J, Hadziyannis SJ. In vivo suppression of precore mRNA synthesis is associated with mutations in the hepatitis B virus core promoter. Virology. 2002;295:86-96. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 47] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | Cao GW. Clinical relevance and public health significance of hepatitis B virus genomic variations. World J Gastroenterol. 2009;15:5761-5769. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 102] [Cited by in F6Publishing: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 21. | Olinger CM, Venard V, Njayou M, Oyefolu AO, Maïga I, Kemp AJ, Omilabu SA, le Faou A, Muller CP. Phylogenetic analysis of the precore/core gene of hepatitis B virus genotypes E and A in West Africa: new subtypes, mixed infections and recombinations. J Gen Virol. 2006;87:1163-1173. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 125] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 22. | Jardi R, Rodriguez F, Buti M, Costa X, Valdes A, Allende H, Schaper M, Galimany R, Esteban R, Guardia J. Mutations in the basic core promoter region of hepatitis B virus. Relationship with precore variants and HBV genotypes in a Spanish population of HBV carriers. J Hepatol. 2004;40:507-514. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 40] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Chauhan R, Kazim SN, Bhattacharjee J, Sakhuja P, Sarin SK. Basal core promoter, precore region mutations of HBV and their association with e antigen, genotype, and severity of liver disease in patients with chronic hepatitis B in India. J Med Virol. 2006;78:1047-1054. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Elkady A, Tanaka Y, Kurbanov F, Oynsuren T, Mizokami M. Virological and clinical implication of core promoter C1752/V1753 and T1764/G1766 mutations in hepatitis B virus genotype D infection in Mongolia. J Gastroenterol Hepatol. 2008;23:474-481. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Bonacci M, Lens S, Mariño Z, Londoño MC, Rodríguez-Tajes S, Mas A, García-López M, Pérez-Del-Pulgar S, Sánchez-Tapias JM, Forns X. Anti-viral therapy can be delayed or avoided in a significant proportion of HBeAg-negative Caucasian patients in the Grey Zone. Aliment Pharmacol Ther. 2018;47:1397-1408. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 26. | Ohata K, Hamasaki K, Toriyama K, Ishikawa H, Nakao K, Eguchi K. High viral load is a risk factor for hepatocellular carcinoma in patients with chronic hepatitis B virus infection. J Gastroenterol Hepatol. 2004;19:670-675. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 66] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 27. | Kuhnhenn L, Jiang B, Kubesch A, Vermehren J, Knop V, Susser S, Dietz J, Carra G, Finkelmeier F, Grammatikos G, Zeuzem S, Sarrazin C, Hildt E, Peiffer KH. Impact of HBV genotype and mutations on HBV DNA and qHBsAg levels in patients with HBeAg-negative chronic HBV infection. Aliment Pharmacol Ther. 2018;47:1523-1535. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 28. | Kramvis A, Kew MC. Epidemiology of hepatitis B virus in Africa, its genotypes and clinical associations of genotypes. Hepatol Res. 2007;37:S9-S19. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 210] [Cited by in F6Publishing: 222] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 29. | Andernach IE, Hübschen JM, Muller CP. Hepatitis B virus: the genotype E puzzle. Rev Med Virol. 2009;19:231-240. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 30. | Hübschen JM, Andernach IE, Muller CP. Hepatitis B virus genotype E variability in Africa. J Clin Virol. 2008;43:376-380. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 31. | Mulders MN, Venard V, Njayou M, Edorh AP, Bola Oyefolu AO, Kehinde MO, Muyembe Tamfum JJ, Nebie YK, Maiga I, Ammerlaan W, Fack F, Omilabu SA, Le Faou A, Muller CP. Low genetic diversity despite hyperendemicity of hepatitis B virus genotype E throughout West Africa. J Infect Dis. 2004;190:400-408. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 129] [Cited by in F6Publishing: 144] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 32. | Lemoine M, Thursz MR. Battlefield against hepatitis B infection and HCC in Africa. J Hepatol. 2017;66:645-654. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 65] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 33. | Kew MC. Hepatocellular carcinoma in African Blacks: Recent progress in etiology and pathogenesis. World J Hepatol. 2010;2:65-73. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 34. | Malagnino V, Salpini R, Maffongelli G, Battisti A, Fabeni L, Piermatteo L, Colagrossi L, Fini V, Ricciardi A, Sarrecchia C, Perno CF, Andreoni M, Svicher V, Sarmati L. High rates of chronic HBV genotype E infection in a group of migrants in Italy from West Africa: Virological characteristics associated with poor immune clearance. PLoS One. 2018;13:e0195045. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |