Published online Sep 28, 2019. doi: 10.3748/wjg.v25.i36.5559
Peer-review started: May 15, 2019
First decision: June 16, 2019
Revised: June 27, 2019
Accepted: August 7, 2019
Article in press: August 7, 2019
Published online: September 28, 2019
Processing time: 137 Days and 0.7 Hours
Small-for-size grafts (SFSGs) in living donor liver transplantation (LDLT) could optimize donor postoperative outcomes and also expand the potential donor pool. Evidence on whether SFSGs would affect medium-term and long-term recipient graft survival is lacking.
To evaluate the impact of small-for-size liver grafts on medium-term and long-term graft survival in adult to adult LDLT.
A systematic review and meta-analysis were performed by searching eligible studies published before January 24, 2019 on PubMed, EMBASE, and Web of Science databases. The primary outcomes were 3-year and 5-year graft survival. Incidence of small-for-size syndrome and short term mortality were also extracted.
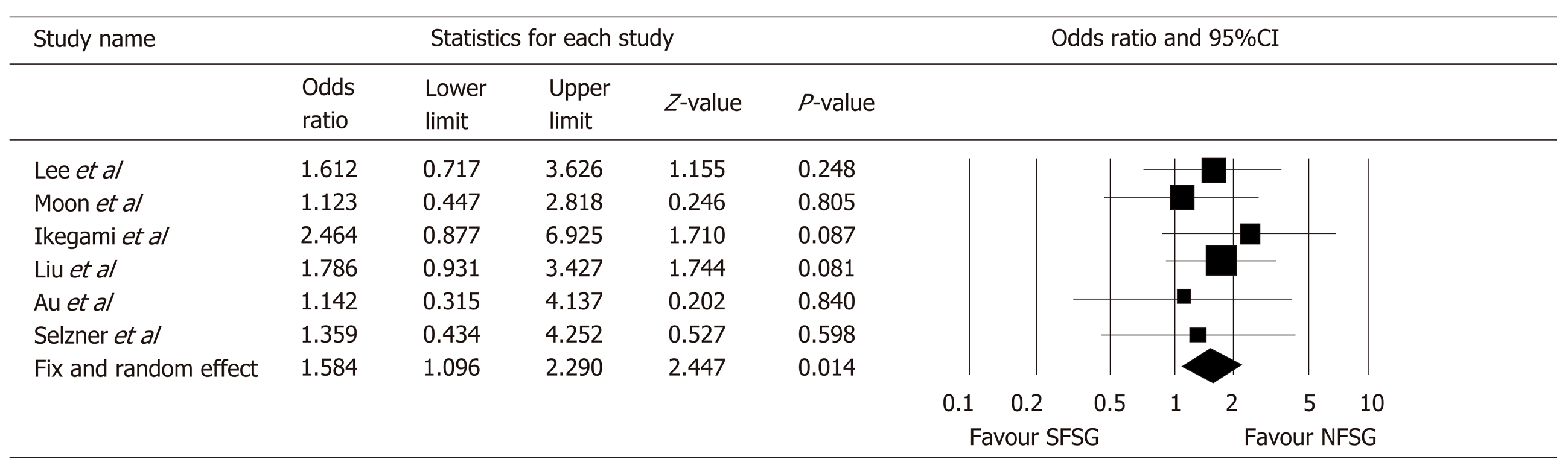
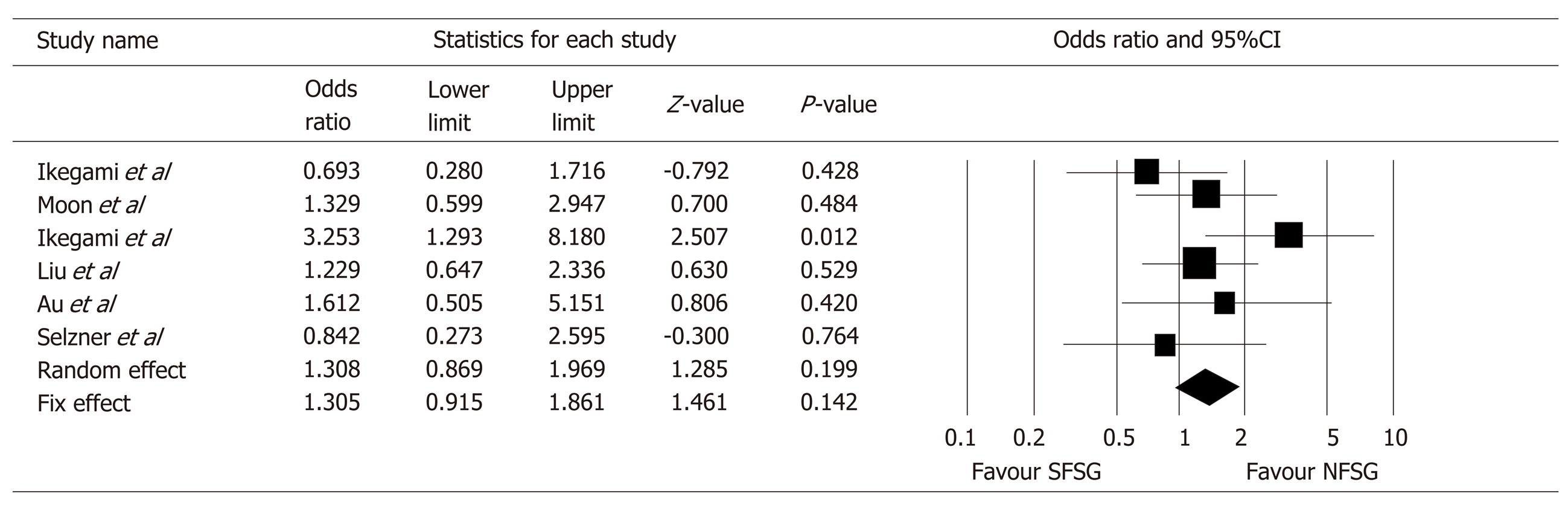
This meta-analysis is reported according to the guidelines of the PRISMA 2009 Statement. Seven retrospective observational studies with a total of 1821 LDLT recipients were included in the meta-analysis. SFSG is associated with significantly poorer medium-term graft survival. The pooled odds ratio for 3-year graft survival was 1.58 [95% confidence interval 1.10-2.29, P = 0.014]. On the other hand, pooled results of the studies showed that SFSG had no significant discriminatory effect on 5-year graft survival with an odds ratio of 1.31 (95% confidence interval 0.87-1.97, P = 0.199). Furthermore, incidence of small-for-size syndrome detected in recipients of SFSG ranged from 0-11.4% in the included studies.
SFSG is associated with inferior medium-term but not long-term graft survival. Comparable long-term graft survival based on liver graft size shows that smaller grafts could be accepted for LDLT with appropriate flow modulatory measures. Close follow-up for graft function is warranted within 3 years after liver transplantation.
Core tip: Small-for-size grafts in living donor liver transplantation is associated with inferior medium-term but not long-term graft survival. Comparable long-term graft survival based on liver graft size shows that smaller grafts could be accepted for living donor liver transplantation with appropriate flow modulatory measures. Close follow-up for graft function is warranted within 3 years after transplantation.
- Citation: Ma KW, Wong KHC, Chan ACY, Cheung TT, Dai WC, Fung JYY, She WH, Lo CM, Chok KSH. Impact of small-for-size liver grafts on medium-term and long-term graft survival in living donor liver transplantation: A meta-analysis. World J Gastroenterol 2019; 25(36): 5559-5568
- URL: https://www.wjgnet.com/1007-9327/full/v25/i36/5559.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i36.5559
Living donor liver transplantation (LDLT) has been a well-recognized alternative to whole graft transplantation from deceased donor in face of organ shortage in the past two decades. Since the first successful LDLT from adult to child reported by Strong et al[1] in 1989 in Australia, the operation has been rapidly taken up by various centers. Multiple technical advancements including concomitant caudate lobe resection, transplantation of right posterior sector grafts, and dual left grafts have been reported since then to optimize patient outcomes[2-4].
In contrast to deceased donor liver transplantation, LDLT requires balancing the outcomes of both recipients and donors. The minimum graft size required to meet the metabolic demands of recipients thus has been a topic of debate for LDLT. When a small-for-size graft (SFSG) is unable to meet the demands, small-for-size syndrome (SFSS) occurs. SFSS is characterized by postoperative coagulopathy, prolonged cholestasis, and liver dysfunction[5-7], which would result in poor postoperative outcomes. In fact, there was evidence showing that SFSG resulted in significantly worse graft survival[8]. Therefore, the graft size has been limited to graft-to-recipient weight ratio (GRWR) of ≥ 0.8[8], or graft volume to standard liver volume (GV/SLV) of 30%-40%[9,10]. The early trend in the development of LDLT was to obtain larger grafts from living donors; techniques to obtain the right lobe for LDLT thus have been developed[11].
On the other hand, it has been reported that the complication rate of right lobe donors was higher than that of left lobe donors[12,13]. Studies have also shown that SFSG was not the only factor leading to SFSS[14,15]. These findings combined with improved surgical techniques resulted in a paradigm shift of trying to obtain smaller grafts in LDLT. Techniques have been developed to modulate portal vein flow to prevent the damage to SFSG; this includes hemi-portocaval shunt[16], splenic artery ligation[17], splenectomy, and splenic artery embolization[18,19]. It has therefore been suggested that adoption of a smaller graft combined with the application of such techniques to modulate hepatic inflow would be a possible method to expand the potential donor pool and optimize outcomes of both donor and recipients of LDLT.
However, there is no clear evidence on whether SFSG (GRWR < 0.8 or GV/SLV < 35%) is safe for liver transplantation recipients and whether the recipient and graft survival would be compromised. We hypothesize that a SFSG would possibly be detrimental to long-term graft survival. Thus, a well-conducted meta-analysis is required to gather existing evidence and analyze the impact of SFSG on the survival outcomes of adult recipients in LDLT.
Systematic review and meta-analysis were performed according to the PRISMA guidelines. Literature search was conducted among publications dated to January 24, 2019 in three databases: PubMed, EMBASE, and Web of Science. Publication year and language were not limited in the search. The following keywords were used for the database search “living donor liver transplantation” and ”small-for-size”. Title and abstracts of the articles identified in the databases were screened independently by two authors (MKW and WKH). References of the eligible studies were manually checked to avoid missing relevant papers. Full-text articles with potential relevance to the study were obtained for further screening. Approval from Institutional Review Board (IRB) for performing this meta-analysis was not required in our center as it did not involve patients from our locality. The research protocol of the current study has not been registered and this study received no funding from any party.
A study was regarded as suitable for inclusion if survival outcome (3-year or 5-year overall graft survival) was compared between the LDLT groups using SFSG and normal-for-size grafts (NFSG). The following types of articles were excluded: Conference abstracts, case reports/series, reviews, meta-analyses, com-mentary/editorial letters, animal/non-human studies, articles with no full-text, and non-English studies with no English translation available. Furthermore, the study was excluded if there was the presence of any one of the following conditions: (1) Outcome data contamination by case-mix analysis of living donors and deceased donor (split liver); (2) GRWR < 0.8% or GV/SLV < 35% was not used as criteria for grouping patients; or (3) Hazard ratio and 95% confidence interval (CI) were not available nor deducible. We carefully checked the source and period of the potentially relevant articles to avoid duplicates. When overlapping cohorts were examined by the same institutions, the most recent and relevant articles were included.
Basic information such as author name, center and country of origin, year of the study, and population characteristics were extracted. Total number of patients in small-for-size and normal size group were extracted from the included studies. 3-year and 5-year graft survival of each group were also extracted. In case of insufficient data or incomplete information, we confirmed the data to the corresponding authors by e-mails. In this study, GRWR of less than 0.8% of body weight or GV/SLV of less than 35% were considered SFSG.
Statistical analyses were performed using software Comprehensive Meta-analysis version 3.0. Number of event (i.e., number of patients with 3-year and 5-year graft failure) was calculated by 3-year or 5-year graft survival multiplied by the number of the patients in each group (small-size graft and normal size). After deriving odds ratio (OR) and 95%CI from each study, the overall effect of SFSG was calculated using both fixed and random-effect model. Heterogeneity of the studies were assessed by I2 value in which the number of less than 0.2 was considered not significant. Publication bias of the studies were assessed by Funnel plot and Eggar’s test, and P values of less than 0.05 were considered significant.
The quality of each included study was assessed by Newcastle-Ottawa Score. This score grades a study according to three criteria: patient selection, comparability, and outcome[20]. The score ranges from lowest of 3 to highest of 9. Scores over 6 are regarded as satisfactory quality. Assessment was performed independently by two authors (MKW and WKH). Any disagreement was revolved by decision of the corresponding author (CKS).
Using the described Medical Subject Headings term, there were 1245 articles matched with the keywords. After exclusion of 1174 papers, 71 potential studies remained for further assessment. After full manuscript appraisal of each paper, another 64 studies were excluded. In the end, data from seven centers with 1821 LDLT recipients were considered eligible for meta-analysis (Figure 1). All of the included studies were retrospective. The majority of the studies were from Asian-pacific regions and one from North America. Study and population characteristics were tabulated in Table 1. Four studies used graft weight to body weight ratio to define SFSG while the remaining three used percentage of the estimated SLV. The flow diagram of study selection is shown in Figure 1.
| Study | Country | Definition of SFSG | Number of patient in SFSG group | Number of patient in NFSG group | Incidence of SFSS (%) | Short-term mortality in SFSG group | Medium-term mortality in SFSG group (OR, 95%CI) | Long-term mortality in SFSG group (OR, 95%CI) |
| Ikegami et al[24], (2008) | Japan | < 35% GV/SLV | 33 | 87 | 0 | 12.5% (1 yr) | 2.46 (0.88-6.93) | 3.25 (1.29-8.18) |
| Moon et al[25], (2010) | Korea | < 0.8% GRWR | 35 | 392 | 5.7 | - | 1.12 (0.45-2.82) | 1.33 (0.60-2.95) |
| Au et al[22], (2015) | Hong Kong | < 35% GV/SLV | 21 | 212 | - | - | 1.14 (0.32-4.14) | 1.61 (0.51-5.15) |
| Ikegami et al[23], (2016) | Japan | < 35% GV/SLV | 88 | 119 | 11.4 | - | - | 0.69 (0.28-1.72) |
| Lee et al[21], (2014) | Korea | < 0.8% GRWR | 50 | 267 | 8 | 2% (1 yr) | 1.61 (0.72-3.63) | - |
| Liu et al[20], (2015) | China | < 0.8% GRWR | 65 | 181 | 11 | 7.7% (30 d) | 1.79 (0.93-3.43) | 1.23 (0.65-2.34) |
| Selzner et al[26], (2009) | Canada | < 0.8% GRWR | 22 | 249 | 9 | 4.5% (30 d) | 1.36 (0.43-4.25) | 0.82 (0.27-2.60) |
Incidence of SFSS in the SFSG recipients was reported in six studies, which ranged from 0-11.4%. Dahm’s criteria[5] of SFSS was adopted by three[22,25,26] out of the six articles that reported the incidence of SFSS. Short-term outcome in terms of 30-d mortality and 1-year mortality using SFSG were reported in two studies, respectively (Table 1).
Concerning the medium-term graft survival, six out of the seven papers provided comparative data between SFSS and NFSG recipients for analysis. All of these studies reported an inferior 3-year graft survival using SFSG with the OR ranging from 1.12 to 2.46. The resulting OR after meta-analysis was 1.58 (95%CI 1.10-2.29, P = 0.014, I2 = 0%) (both fixed and random effect model) (Figure 2).
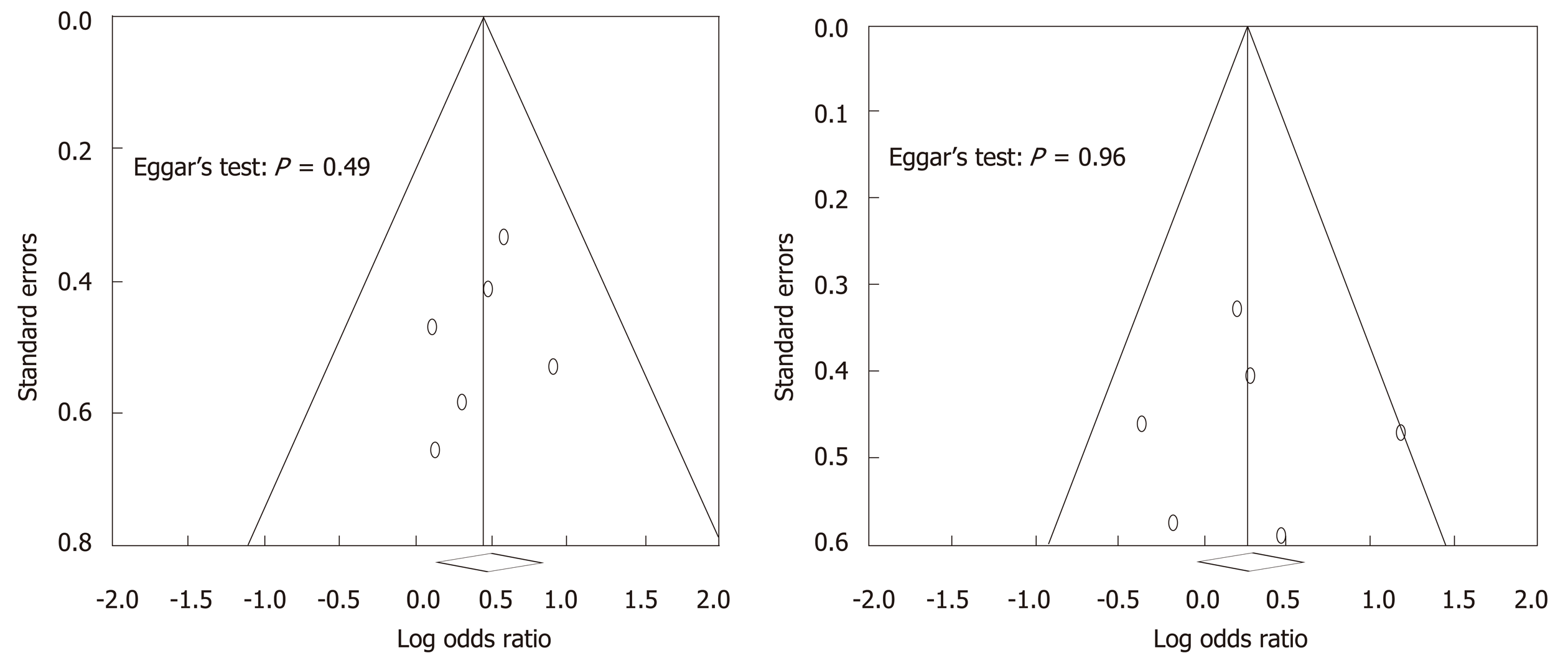
Concerning the long-term outcomes of SFSS recipients, six papers provided data comparing SFSS and NFSG recipient. The studies from Kyushu University[24] and Toronto General Hospital[27] showed a tendency of better 5-year graft survival (OR 0.69 and OR 0.84, respectively). Because meta-analysis using fix-effect model demonstrated significant heterogeneity (I2 = 21.8%), analysis was performed using random-effect model instead, and the resulting OR of 5-year graft survival in using SFSG was 1.31 (95%CI 0.87-1.97, P = 0.199, I2 = 2.1%) (Figure 3). There was no significant publication bias demonstrated by Funnel plots of 3-year and 5-year graft survival meta-analysis (Figure 4).
All studies included in this meta-analysis had a Newcastle-Ottawa Score over 6. The mean Newcastle-Ottawa Score was 7.4 (range 6-8) (Table 2). Characteristics of the patient population and treatment protocol were clearly described. However, most studies did not document the duration of follow-up and dropout rate during the follow-up period. Nonetheless, overall quality of the studies was satisfactory.
This study analyzed extracted data from seven LDLT centers, containing over 1800 recipients and it demonstrated that SFSG was associated with an inferior 3-year graft survival, but no significant discriminatory effect on 5-year graft survival was demonstrated. In regions of low deceased donor rate, living donor liver graft represents a significant organ source[28]. As inferred by its name, SFSS is the manifestation of the physiological mismatch secondary to a SFSG. Given the high morbidity and mortality associated with SFSS reported in early series[29], insufficient graft size became a major reason leading to donor rejection especially in female donors. This situation frequently results in wait list mortality of the desperate patients.
As our understanding of the relationship between SFSG and SFSS increases, a number of techniques have been proposed to alleviate the physiological mismatch associated with the use of SFSG[16]. In the author’s center, measurement of portal venous flow and portal venous pressure assessment are performed once the actual donor graft size to GV/SLV ratio is less than 40%. If the portal flow is over 400 mL/min/100 g and the portal venous/hepatic venous pressure gradient is over 15 mmHg, the splenic artery is clipped to reduce portal venous flow[17]. Apart from intra-operative modulatory procedures, we adopted a strict postoperative fluid management protocol, such as keeping the patient 5-degree to the right side and head up, maintaining low central venous pressure (i.e., at 5 mmHg) by diuresis and albumin infusion to ensure optimal graft perfusion pressure. With these measures we and many other centers are able to recruit SFSGs as small as 0.6% of body weight or less than 25% estimated SLV.
It was generally believed that SFSG was associated with a higher chance of SFSS and hence morbidity and mortality. However, the risk of clinically detectable SFSS associated with SFSG in the included studies was only 0-11.4%. This low incidence suggested that SFSS in SFSG recipient could be safely avoided if appropriate perioperative precautions had been taken. In addition, the short-term mortality associated with SFSG was comparable to the figure quoted by most other series using NFSG.
Unlike the case of renal transplantation, in which SFS kidney graft is associated with higher incidence of long-term graft loss[30], long-term outcomes of SFS liver graft were infrequently reported in the literature. Although many publications reported long-term treatment outcomes, the majority of them were not suitable for meta-analysis. For instance, some reported long-term outcomes using overall patient survival instead of graft survival[31,32], while another used a different definition of SFSG (i.e., 0.85)[33], and different end-point (i.e., 10-year) for survival analysis[34]. All these limited the number of papers to be included in the current meta-analysis.
In this study, all included studies reported an insignificant trend of poorer 3-year (medium-term) graft survival in the SFSG group. This consistent inferior trend reported by all of the included studies resulted in a significantly high odd ratio (i.e., OR 1.58, P = 0.014) of graft failure in the 3-year meta-analysis. This SFSG associated inferiority in graft survival was not demonstrated in 5-year (long-term) analysis. The authors postulate that the SFSG might have physiological implications on the degree of graft regeneration and fibrosis, vascular patency, and bile duct integrity. If graft function could be maintained during the modulation period, long-term graft survival could be expected. Therefore, we suggest a short follow-up interval for surveillance of graft function (i.e., blood taking no longer than 3 mo) to detect transaminitis and ductopathy, low threshold of biopsy to rule out acute cellular rejection and biliary obstruction, and careful titration of immunosuppressant to avoid rejection and opportunistic infection particularly in the first 3 years of transplantation.
There were some limitations in this current meta-analysis. Firstly, the number of eligible articles was small and the reasons for that had been explained in previous paragraphs. Secondly, all of the studies recruited were retrospective in nature, which limited the power of the analysis; however, randomized controlled trials comparing SFSG and NFSG LDLT outcomes is impossible in clinical settings for obvious ethical reasons. Last but not least, the inherent heterogeneities between studies in patient characteristics, transplantation indication, threshold and method of intra-operative portal pressure modulation, and definition of graft failure would lead to bias of the results. Nonetheless, this is the first meta-analysis consisting of a reasonable number of patients gathered from seven well-conducted retrospective analyses that suggests the effect of SFSG on medium-term and long-term graft survival. SFSG is associated with inferior medium-term but not long-term graft survival. Close follow-up for graft function is warranted within 3 years after transplantation.
Left side donor hepatectomy has become more popular given the paramount importance of donor safety. There is limited understanding concerning the medium-term and long-term effect of small-for-size grafts (SFSGs), which is commonly encountered in living donor liver transplantation (LDLT). This study aims to provide more evidence concerning this issue.
The objective is to see if SFSG imposes a negative effect on graft survival. This will help future decision making on performing left donor hepatectomy.
The research objective is to answer whether SFSG is associated with an inferior graft survival through meta-analysis.
Literature comparing the survival outcomes between SFSGs and normal-for-size grafts were limited. In addition, most studies were of small sample size. Meta-analysis allows pooling of the results from these studies. This is the first meta-analysis performed on this important topic.
After extensive literature review following the preset search protocol, there were seven studies comprising of over 1800 LDLT recipients eligible to be included for meta-analysis. It was demonstrated that SFSG is associated with inferior medium-term (3-year) graft survival, but no significant effect on long-term (5-year) graft survival. This result would support the use of SFSG and potentially help promoting left shifting. However, it is beyond the capacity of this study to define what is the smallest graft size that is safe for LDLT.
This is the first study trying to illustrate the effect of SFSG on graft survival using meta-analysis. With the results in this study, SFSG is associated with inferior medium-term but not long-term graft survival. Therefore, for patients who receive a SFSG, graft function should be followed more vigilantly by means of biochemical and radiological investigations. However, SFSG should not be considered an “inferior graft” when compared to normal-for-size grafts judging from the comparable long-term outcome.
The result from this study seems to suggest that left lobe donor hepatectomy should be considered even if it is a SFSG to the transplant recipient for the benefit of better donor safety margin. Due to the heterogeneity of the patients in this study, whether this statement is valid in all liver transplantation indications (i.e., hepatocellular carcinoma) remains to be answered. In addition, the smallest graft size that is safe for LDLT is yet to be defined.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Enosawa S, Mrzljak A, Nacif LS S-Editor: Yan JP L-Editor: Filipodia E-Editor: Zhang YL
| 1. | Strong RW, Lynch SV, Ong TH, Matsunami H, Koido Y, Balderson GA. Successful liver transplantation from a living donor to her son. N Engl J Med. 1990;322:1505-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 604] [Cited by in RCA: 545] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 2. | Miyagawa S, Hashikura Y, Miwa S, Ikegami T, Urata K, Terada M, Kubota T, Nakata T, Kawasaki S. Concomitant caudate lobe resection as an option for donor hepatectomy in adult living related liver transplantation. Transplantation. 1998;66:661-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 83] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 3. | Sugawara Y, Makuuchi M, Takayama T, Mizuta K, Kawarasaki H, Imamura H, Hashizume K. Liver transplantation using a right lateral sector graft from a living donor to her granddaughter. Hepatogastroenterology. 2001;48:261-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 4. | Lee S, Hwang S, Park K, Lee Y, Choi D, Ahn C, Nah Y, Koh K, Han S, Park S, Min P. An adult-to-adult living donor liver transplant using dual left lobe grafts. Surgery. 2001;129:647-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 98] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 5. | Dahm F, Georgiev P, Clavien PA. Small-for-size syndrome after partial liver transplantation: Definition, mechanisms of disease and clinical implications. Am J Transplant. 2005;5:2605-2610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 473] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 6. | Tucker ON, Heaton N. The 'small for size' liver syndrome. Curr Opin Crit Care. 2005;11:150-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 134] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 7. | Selvaggi G, Tzakis A. Surgical considerations in liver transplantation: Small for size syndrome. Panminerva Med. 2009;51:227-233. [PubMed] |
| 8. | Kiuchi T, Kasahara M, Uryuhara K, Inomata Y, Uemoto S, Asonuma K, Egawa H, Fujita S, Hayashi M, Tanaka K. Impact of graft size mismatching on graft prognosis in liver transplantation from living donors. Transplantation. 1999;67:321-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 741] [Cited by in RCA: 722] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 9. | Lo CM, Fan ST, Liu CL, Chan JK, Lam BK, Lau GK, Wei WI, Wong J. Minimum graft size for successful living donor liver transplantation. Transplantation. 1999;68:1112-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 209] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 10. | Kawasaki S, Makuuchi M, Matsunami H, Hashikura Y, Ikegami T, Nakazawa Y, Chisuwa H, Terada M, Miyagawa S. Living related liver transplantation in adults. Ann Surg. 1998;227:269-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 317] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 11. | Lo CM, Fan ST, Liu CL, Wei WI, Lo RJ, Lai CL, Chan JK, Ng IO, Fung A, Wong J. Adult-to-adult living donor liver transplantation using extended right lobe grafts. Ann Surg. 1997;226:261-9; discussion 269-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 419] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 12. | Lo CM. Complications and long-term outcome of living liver donors: A survey of 1,508 cases in five Asian centers. Transplantation. 2003;75:S12-S15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 207] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 13. | Umeshita K, Fujiwara K, Kiyosawa K, Makuuchi M, Satomi S, Sugimachi K, Tanaka K, Monden M; Japanese Liver Transplantation Society. Operative morbidity of living liver donors in Japan. Lancet. 2003;362:687-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 209] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 14. | Yagi S, Uemoto S. Small-for-size syndrome in living donor liver transplantation. Hepatobiliary Pancreat Dis Int. 2012;11:570-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 15. | Lei JY, Wang WT, Yan LN. Risk factors of SFSS in adult-to-adult living donor liver transplantation using the right liver: A single-center analysis of 217 cases. Hepatogastroenterology. 2012;59:1491-1497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 16. | Troisi R, Ricciardi S, Smeets P, Petrovic M, Van Maele G, Colle I, Van Vlierberghe H, de Hemptinne B. Effects of hemi-portocaval shunts for inflow modulation on the outcome of small-for-size grafts in living donor liver transplantation. Am J Transplant. 2005;5:1397-1404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 207] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 17. | Lo CM, Liu CL, Fan ST. Portal hyperperfusion injury as the cause of primary nonfunction in a small-for-size liver graft-successful treatment with splenic artery ligation. Liver Transpl. 2003;9:626-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 119] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 18. | Troisi RI, Berardi G, Tomassini F, Sainz-Barriga M. Graft inflow modulation in adult-to-adult living donor liver transplantation: A systematic review. Transplant Rev (Orlando). 2017;31:127-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 19. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6:e1000097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52948] [Cited by in RCA: 47198] [Article Influence: 2949.9] [Reference Citation Analysis (0)] |
| 20. | Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8858] [Cited by in RCA: 12666] [Article Influence: 844.4] [Reference Citation Analysis (0)] |
| 21. | Liu C, Song JL, Lu WS, Yang JY, Jiang L, Yan LN, Zhang JY, Lu Q, Wen TF, Xu MQ, Wang WT. Hepatic Arterial Buffer Response Maintains the Homeostasis of Graft Hemodynamics in Patient Receiving Living Donor Liver Transplantation. Dig Dis Sci. 2016;61:464-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Lee SD, Kim SH, Kim YK, Lee SA, Park SJ. Graft-to-recipient weight ratio lower to 0.7% is safe without portal pressure modulation in right-lobe living donor liver transplantation with favorable conditions. Hepatobiliary Pancreat Dis Int. 2014;13:18-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 23. | Au KP, Chan SC, Chok KS, Chan AC, Wong TC, Sharr WW, Lo CM. Durability of small-for-size living donor allografts. Liver Transpl. 2015;21:1374-1382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 24. | Ikegami T, Yoshizumi T, Sakata K, Uchiyama H, Harimoto N, Harada N, Itoh S, Nagatsu A, Soejima Y, Maehara Y. Left lobe living donor liver transplantation in adults: What is the safety limit? Liver Transpl. 2016;22:1666-1675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 25. | Ikegami T, Masuda Y, Ohno Y, Mita A, Kobayashi A, Urata K, Nakazawa Y, Miwa S, Hashikura Y, Miyagawa S. Prognosis of adult patients transplanted with liver grafts < 35% of their standard liver volume. Liver Transpl. 2009;15:1622-1630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 26. | Moon JI, Kwon CH, Joh JW, Jung GO, Choi GS, Park JB, Kim JM, Shin M, Kim SJ, Lee SK. Safety of small-for-size grafts in adult-to-adult living donor liver transplantation using the right lobe. Liver Transpl. 2010;16:864-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 27. | Selzner M, Kashfi A, Cattral MS, Selzner N, Greig PD, Lilly L, McGilvray ID, Therapondos G, Adcock LE, Ghanekar A, Levy GA, Renner EL, Grant DR. A graft to body weight ratio less than 0.8 does not exclude adult-to-adult right-lobe living donor liver transplantation. Liver Transpl. 2009;15:1776-1782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 93] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 28. | Chen CL, Kabiling CS, Concejero AM. Why does living donor liver transplantation flourish in Asia? Nat Rev Gastroenterol Hepatol. 2013;10:746-751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 29. | Sugawara Y, Makuuchi M, Takayama T, Imamura H, Dowaki S, Mizuta K, Kawarasaki H, Hashizume K. Small-for-size grafts in living-related liver transplantation. J Am Coll Surg. 2001;192:510-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 164] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 30. | Kasiske BL, Snyder JJ, Gilbertson D. Inadequate donor size in cadaver kidney transplantation. J Am Soc Nephrol. 2002;13:2152-2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 105] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 31. | Uemura T, Wada S, Kaido T, Mori A, Ogura Y, Yagi S, Fujimoto Y, Ogawa K, Hata K, Yoshizawa A, Okajima H, Uemoto S. How far can we lower graft-to-recipient weight ratio for living donor liver transplantation under modulation of portal venous pressure? Surgery. 2016;159:1623-1630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 32. | Lee EC, Kim SH, Shim JR, Park SJ. Small-for-size grafts increase recurrence of hepatocellular carcinoma in liver transplantation beyond milan criteria. Liver Transpl. 2018;24:35-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Ben-Haim M, Emre S, Fishbein TM, Sheiner PA, Bodian CA, Kim-Schluger L, Schwartz ME, Miller CM. Critical graft size in adult-to-adult living donor liver transplantation: Impact of the recipient's disease. Liver Transpl. 2001;7:948-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 226] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 34. | Shoreem H, Gad EH, Soliman H, Hegazy O, Saleh S, Zakaria H, Ayoub E, Kamel Y, Abouelella K, Ibrahim T, Marawan I. Small for size syndrome difficult dilemma: Lessons from 10 years single centre experience in living donor liver transplantation. World J Hepatol. 2017;9:930-944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |