Published online Sep 7, 2019. doi: 10.3748/wjg.v25.i33.4805
Peer-review started: February 27, 2019
First decision: April 30, 2019
Revised: August 3, 2019
Accepted: August 19, 2019
Article in press: August 19, 2019
Published online: September 7, 2019
Processing time: 192 Days and 21.7 Hours
Obesity is a global health epidemic with considerable economic burden. Surgical solutions have become increasingly popular following technical advances leading to sustained efficacy and reduced risk. Sleeve gastrectomy accounts for almost half of all bariatric surgeries worldwide but concerns regarding its relationship with gastroesophageal reflux disease (GERD) has been a topic of debate. GERD, including erosive esophagitis, is highly prevalent in the obese population. The role of pre-operative endoscopy in bariatric surgery has been controversial. Two schools of thought exist on the matter, one that believes routine upper endoscopy before bariatric surgery is not warranted in the absence of symptoms and another that believes that symptoms are poor predictors of underlying esophageal pathology. This debate is particularly important considering the evidence for the association of laparoscopic sleeve gastrectomy (LSG) with de novo and/or worsening GERD compared to the less popular Roux-en-Y gastric bypass procedure. In this paper, we try to address 3 burning questions regarding the inter-relationship of obesity, GERD, and LSG: (1) What is the prevalence of GERD and erosive esophagitis in obese patients considered for bariatric surgery? (2) Is it necessary to perform an upper endoscopy in obese patients considered for bariatric surgery? And (3) What are the long-term effects of sleeve gastrectomy on GERD and should LSG be done in patients with pre-existing GERD?
Core tip: The convenience and ease of sleeve gastrectomy comes at a risk of de novo or worsening of pre-existing gastroesophageal reflux disease. Candidates for bariatric surgery should have a thorough evaluation of reflux symptoms as well as esophageal anatomy and pathology. This should be followed by an informed and open discussion with the patient about risks and benefits of different bariatric surgical options leading to optimal shared decision making.
- Citation: Bou Daher H, Sharara AI. Gastroesophageal reflux disease, obesity and laparoscopic sleeve gastrectomy: The burning questions. World J Gastroenterol 2019; 25(33): 4805-4813
- URL: https://www.wjgnet.com/1007-9327/full/v25/i33/4805.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i33.4805
Obesity is a modern-day global epidemic with significant health and economic burden. According to the World Health Organization, 650 million adults (13% of all adults) and over 340 million children and adolescents are overweight or obese[1]. In light of the oft-disappointing long-term results of medical and behavioral interventions, an increasingly larger number of obese patients are turning to minimally invasive bariatric surgery. According to the American Society for Metabolic and Bariatric Surgery, approximately 216000 individuals underwent bariatric surgery in 2016 in the United States, a net increase of 36.7% over a five-year period starting in 2011[2]. Laparoscopic sleeve gastrectomy (LSG) is currently the most popular procedure accounting for more than 50% to 60% of bariatric surgeries worldwide[2,3]. Two recent large randomized trials have confirmed that there is no significant difference in excess weight loss between LSG and laparoscopic Roux-en-Y gastric bypass (RYGB) at 5 years of follow-up[4,5]. However, enthusiasm for this relatively simple procedure has been curtailed by concerns of post-operative gastroesophageal reflux disease (GERD), as a result of either persistent or de novo reflux[2]. This remains an issue of significant controversy and active debate in clinical practice. At the Fifth International Consensus Conference on LSG, 52.5% of general surgeons and 23.3% of bariatric experts considered GERD a contraindication to LSG[6]. This article will address 3 burning questions concerning the inter-relationship between obesity, GERD, and LSG.
Obesity is an important risk factor for GERD and is associated with esophageal complications such as erosive esophagitis (EE), Barrett's esophagus, and esophageal adenocarcinoma[7,8]. GERD has been reported in as many as 62.4% to 73% of bariatric surgery candidates[9,10]. The pathophysiological mechanisms predisposing to GERD in obesity include increased intra-abdominal pressure[11], impaired gastric emptying[12], decreased lower esophageal sphincter (LES) pressure, and higher frequency of transient LES relaxation[13,14]. In addition, a higher prevalence of hiatal hernia has been described in obese individuals[15]. Central obesity, rather than body mass index (BMI), is more closely associated with GERD[7,16]. High-resolution manometry suggests that both intragastric pressure and gastroesophageal pressure gradient correlate primarily with waist circumference[10].
Overweight and obesity (especially abdominal visceral obesity) are also risk factors for EE. EE is associated with higher distal acid exposure time (percentage time with pH < 4) and higher percentage of reflux episodes reaching the proximal esophagus[17]. El-Serag et al[7] showed that patients with a BMI > 30 are 2.5 times more likely to have reflux symptoms and EE than those with a normal BMI. A meta-analysis of 6 studies showed that the adjusted risk ratio for EE was 1.76 in patients with BMI > 25 compared to those with BMI < 25[18]. Prospective endoscopic studies in bariatric surgery candidates have documented a high prevalence of EE in obese individuals ranging from 4.2% to 33.9% (Table 1)[9-11,19-25]. Risk factors for EE varied between studies and included increased waist circumference, insulin resistance, and presence of reflux symptoms[11]. It is important to note that the absence of symptoms does not exclude erosive disease. In one study, 12.3% of obese patients with low probability of reflux symptoms (low GERDQ score < 8) had EE[9]. The literature is conclusive on the matter: Obesity is associated with higher prevalence of GERD and erosive esophagitis.
| Publication | Year | Number of subjects | Prevalence of EE (%) | Comments |
| Verset et al[18] | 1997 | 147 | 30.6 | High incidence of peptic lesions that were mainly asymptomatic |
| Ortiz et al[9] | 2006 | 138 | 18.8 | Sensitivity of heartburn as diagnostic criterion of GERD was 29.3%, with a specificity of 85.7% |
| Asymptomatic GER (abnormal esophageal acid exposure and/or EE) more common than symptomatic GER | ||||
| Csendes et al[10] | 2007 | 426 | 26.3 | Out of the 112 EE patients, 77 (68.7%) reported GERD symptoms |
| Merrouche et al[11] | 2007 | 94 | 6.4 | 46% of patients had abnormal 24-pH study |
| Dutta et al[19] | 2009 | 101 | 8.9 | 6.9% EE in age- and sex-matched non-obese control subjects |
| Tai et al[20] | 2010 | 260 | 32.3 | Increased waist circumference, insulin resistance, and presence of reflux symptoms independent risk factors for EE |
| Martin-Perez et al[21] | 2014 | 88 | 4.5 | Esophageal pH monitoring tests positive in 65% of patients |
| Absence of symptoms did not rule out abnormal esophageal function tests | ||||
| Carabotti et al[24] | 2015 | 142 | 4.2 | Majority of endoscopic lesions were asymptomatic |
| Mora et al[23] | 2016 | 196 | 17.3 | Esophageal pH-metry abnormal in 54.2% of patients |
| Symptoms not enough to diagnose underlying GERD or EE | ||||
| Sharara et al[24] | 2019 | 242 | 33.9 | Anthropometric data and GERD questionnaires have limited accuracy for EE |
| 12.3% of patients with low GERDQ (< 8) had EE |
Clinical practice guidelines published in 2013 by the American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic and Bariatric Surgery recommend preoperative endoscopy only when clinically indicated[26]. This is in line with the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) 2008 guidelines and the 2014 interdisciplinary European guidelines endorsed by the International Federation for the Surgery of Obesity and Metabolic Disorders-European Chapter and European Association for the Study of Obesity[27]. On the other hand, the 2015 ASGE guidelines recommend that the decision be individualized[28] while the European Association of Endoscopic Surgeons advises that all patients be evaluated by either endoscopy or upper gastrointestinal series prior to their bariatric surgery[29]. In short, the jury is still out on the matter and a consensus between international and national societies seems unlikely. In a recent series of 1555 patients, asymptomatic patients with significant findings on endoscopy did not require a change in management or surgery[30]. The authors went on to conclude that routine upper endoscopy requires further justifications for asymptomatic patients. On the other hand, several recent studies emphasized the importance of doing an upper endoscopy preoperatively[23-25]. Carabotti et al[24] showed that the incidence of endoscopic lesions was the same between patients who reported symptoms and those who did not; the study also concluded that with the current adopted approach to preoperative endoscopy, 87% of EE cases would have been missed. In our experience, we had similar outcomes when we administered the GERD-Q and the Nocturnal GERD Symptom Severity and Impact Questionnaire (N-GSSIQ) to more than 240 consecutive unselected patients scheduled for bariatric surgery. These validated scores were poorly predictive of endoscopically-proven EE in these patients, even when combined with clinical assessment as part of a composite score[9]. As mentioned earlier, the absence of symptoms does not rule out the presence of GERD[9,23]. A recent survey conducted in the United Kingdom showed that 90% of bariatric units perform preoperative upper endoscopy either routinely or selectively[31]. However, there is also no clear consensus on the indications amongst those who do it selectively. This is particularly important in patients considered for LSG given the evidence linking it to worsening GERD and PPI dependence[32-34]. The reason so much debate surrounds the issue is because significant GERD plays a major role in the choice of the bariatric procedure and the presence of per-operative reflux symptoms appears to be associated with post-operative GERD[35]. In the absence of proper randomized trials and dedicated large long-term follow-up studies, the impact of baseline GERD as well as its post-operative risk should be thoroughly discussed with the patient to help guide the choice of the bariatric procedure. We recommend routine upper endoscopy for all patients scheduled to undergo bariatric surgery to assist with this shared decision process.
Several short-term (less than 2 years) follow-up studies have looked at the effect of sleeve gastrectomy on GERD. Some have shown improvement of GERD symptoms after LSG[36-40] while others reported worsening and de novo GERD[41-45]. Few studies have objectively evaluated the presence of pathologic reflux by 24-h multichannel intraluminal impedance pH monitoring at ≥ 12 mo after LSG reporting conflicting results[46-50]. A systematic review and meta-analysis was inconclusive reporting “high heterogeneity among available studies and paradoxical outcomes of objective esophageal function tests”[32]. Recently, two large randomized controlled trials were published comparing the 5-year follow-up outcome of LSG and RYGB[4,5]. The SM-BOSS trial reported 5-year postoperative GERD remission in 25% in the LSG group compared to 60.4% in RYGB (P = 0.002) with de novo GERD in 31.6% of LSG patients compared to 10.7% in RYGB patients (P = 0.01). The study also reported that 9% of LSG patients had to undergo conversion to RYGB because of GERD (highest reason for conversion in the study population). The SLEEVEPASS trial reported RYGB conversion in 6% due to reflux (the study excluded patients with “severe gastroesophageal reflux with a large hiatal hernia”). These figures are consistent with previous literature that showed a 5%-10% conversion rate from LSG to RYGB due to GERD[35,51]. A systematic review published in 2016 demonstrated that 8 out of 10 studies showed new onset GERD at long-term follow up after LSG with a range of 10% to 23%[52]. A prospective study by Genco et al. of 110 LSG patients followed over a mean of 58 months showed that the incidence of GERD symptoms, EE and PPI intake increased significantly post-operatively. Upward migration of the GEJ Z-line was found in 73.6% of cases on follow-up endoscopy. What was most alarming in this study was the fact that non-dysplastic Barrett's esophagus was newly diagnosed in 17.2% of patients. This finding has been duplicated in another recent small multicenter study from Italy[53].
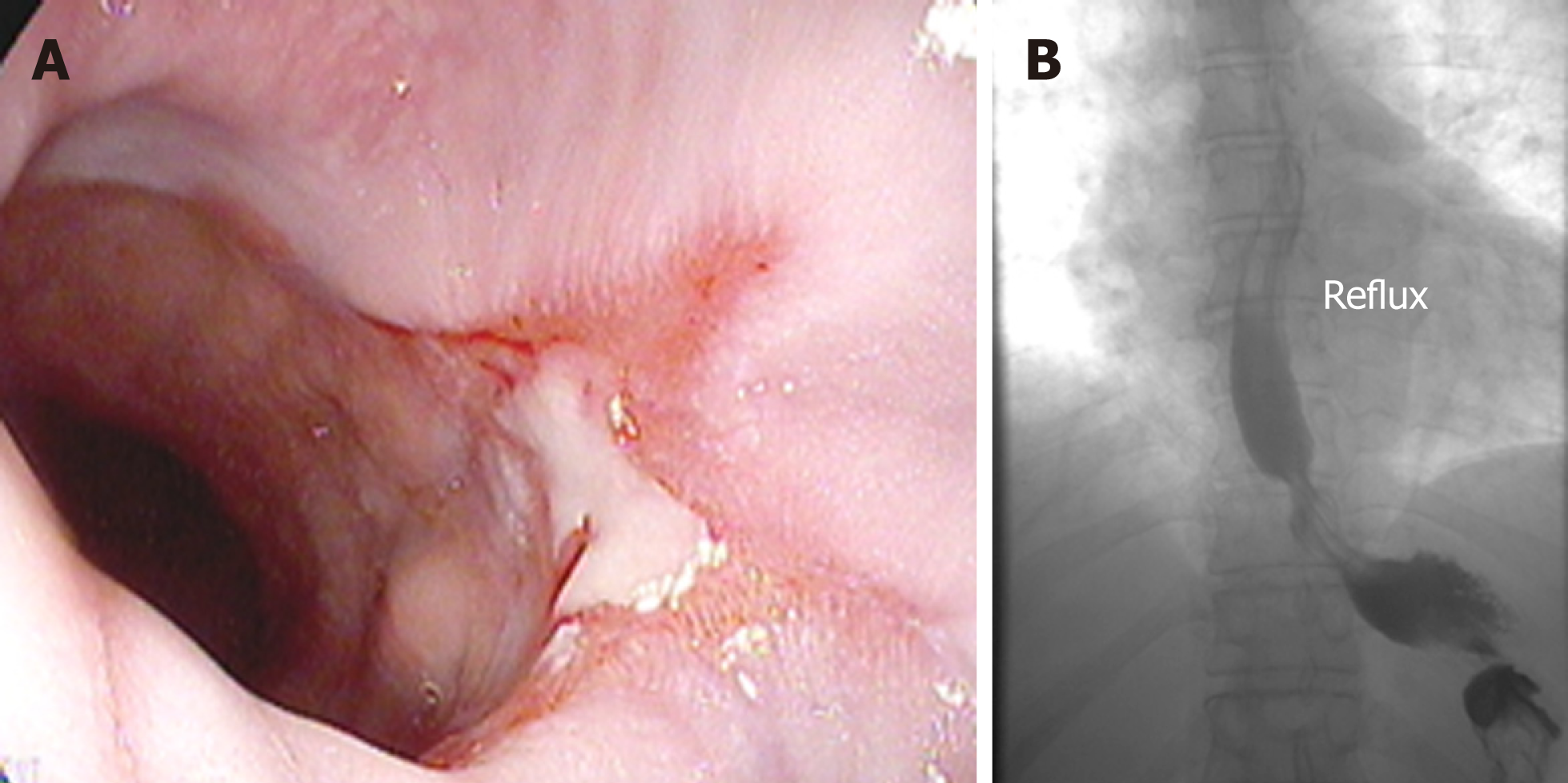
The lines of evidence supporting that LSG is a refluxogenic procedure are multiple and include the observation of increased intragastric pressure and impedance reflux episodes on high-resolution impedance manometry after LSG[54], significant increase in non-acidic reflux with stasis and acidification in esophagus, and the higher rate of de novo reflux in cohort studies and in randomized controlled studies compared to RYGB. In a rat model, LSG was independently associated with histopathologic changes of severe esophagitis compared to high-fat diet fed and to sham-operated rats[55]. The putative pathophysiological mechanisms underlying GERD after LSG are summarized in Table 2[49,56-62]. They include a hypotensive LES, loss of angle of His flap valve, increased gastroesophageal pressure gradient with intra-thoracic migration of the remnant stomach, reduction in the compliance of the gastric remnant provoking an increase in transient LES relaxations, relative gastric stasis in the proximal remnant and increased emptying from the antrum, stasis and acidification in the esophagus, as well as higher intragastric pressure and increased impedance reflux episodes. Figure 1 showcases some of the endoscopic and radiologic findings of GERD post LSG.
| Hypotensive lower esophageal sphincter[48] |
| Loss of angle of His flap valve[55] |
| Increased gastro-esophageal pressure gradient and intra-thoracic migration of the remnant stomach[56] |
| Reduction in the compliance of the gastric remnant provoking an increase in transient lower esophageal sphincter relaxations[57] |
| Lack of gastric compliance and emptying during the first postoperative year[58] |
| Relative gastric stasis in the proximal remnant and increased emptying from the antrum (suggested on time-resolved MRI studies)[59] |
| Excessively large or dilated sleeve retaining increased acid production capacity leading to reflux[60] |
| Overly narrowed or strictured sleeve resulting in reflux and decreased esophageal acid clearance[61] |
Given the evidence for long-term GERD burden post LSG, the 2015 joint statement by the ASMBS, SAGES and ASGE considered EE as a relative contraindication to the surgery[28]. A recent prospective study showed that the presence of pre-operative GERD symptoms and EE at baseline were independently associated with a higher need of postoperative PPI use at 6 mo after LSG[34]. The totality of the evidence suggests that LSG is associated with an increased incidence of GERD. While some obese patients with mild non-erosive reflux disease may benefit from LSG with resolution of GERD symptoms after weight loss, those with severe reflux and erosive disease appear to have a high probability of persistent GERD. The opportunity to save such patients from persistent gastroesophageal reflux, PPI dependence, and possible revisional surgery should be seized and the available evidence openly discussed with the patient.
The popularity of sleeve gastrectomy derives mainly from its relative ease, safety and efficacy. The “Achilles heel” of this procedure appears to be gastroesophageal reflux and its complications. This is an issue of concern particularly for patients with pre-existing GERD or EE. As physicians, we have a duty not to cause harm. We believe that a thorough evaluation of reflux symptoms as well as esophageal anatomy and pathology should be systematically undertaken in all patients considered for bariatric surgery. This should be followed by an informed and open discussion with the patient about risks and benefits of different bariatric surgical options leading to optimal shared decision making.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Lebanon
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Liu F S-Editor: Yan JP L-Editor: A E-Editor: Ma YJ
| 1. | World Health Organization. Obesity and overweight. Fact Sheets. 2018; Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. |
| 2. | English WJ, DeMaria EJ, Brethauer SA, Mattar SG, Rosenthal RJ, Morton JM. American Society for Metabolic and Bariatric Surgery estimation of metabolic and bariatric procedures performed in the United States in 2016. Surg Obes Relat Dis. 2018;14:259-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 316] [Article Influence: 39.5] [Reference Citation Analysis (0)] |
| 3. | Angrisani L, Santonicola A, Iovino P, Vitiello A, Zundel N, Buchwald H, Scopinaro N. Bariatric Surgery and Endoluminal Procedures: IFSO Worldwide Survey 2014. Obes Surg. 2017;27:2279-2289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 580] [Cited by in RCA: 549] [Article Influence: 68.6] [Reference Citation Analysis (0)] |
| 4. | Peterli R, Wölnerhanssen BK, Peters T, Vetter D, Kröll D, Borbély Y, Schultes B, Beglinger C, Drewe J, Schiesser M, Nett P, Bueter M. Effect of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Roux-en-Y Gastric Bypass on Weight Loss in Patients With Morbid Obesity: The SM-BOSS Randomized Clinical Trial. JAMA. 2018;319:255-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 693] [Cited by in RCA: 875] [Article Influence: 125.0] [Reference Citation Analysis (0)] |
| 5. | Salminen P, Helmiö M, Ovaska J, Juuti A, Leivonen M, Peromaa-Haavisto P, Hurme S, Soinio M, Nuutila P, Victorzon M. Effect of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Roux-en-Y Gastric Bypass on Weight Loss at 5 Years Among Patients With Morbid Obesity: The SLEEVEPASS Randomized Clinical Trial. JAMA. 2018;319:241-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 712] [Article Influence: 101.7] [Reference Citation Analysis (0)] |
| 6. | Gagner M, Hutchinson C, Rosenthal R. Fifth International Consensus Conference: Current status of sleeve gastrectomy. Surg Obes Relat Dis. 2016;12:750-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 231] [Article Influence: 25.7] [Reference Citation Analysis (0)] |
| 7. | El-Serag HB, Graham DY, Satia JA, Rabeneck L. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am J Gastroenterol. 2005;100:1243-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 394] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 8. | El-Serag HB. Obesity and disease of the esophagus and colon. Gastroenterol Clin North Am. 2005;34:63-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Sharara AI, Rustom LBO, Bou Daher H, Rimmani HH, Shayto RH, Minhem M, Ichkhanian Y, Aridi H, Al-Abbas A, Shaib Y, Alami R, Safadi B. Prevalence of gastroesophageal reflux and risk factors for erosive esophagitis in obese patients considered for bariatric surgery. Dig Liver Dis. 2019;pii:S1590-8658(19)30550-X. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 10. | Merrouche M, Sabaté JM, Jouet P, Harnois F, Scaringi S, Coffin B, Msika S. Gastro-esophageal reflux and esophageal motility disorders in morbidly obese patients before and after bariatric surgery. Obes Surg. 2007;17:894-900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 136] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 11. | Tai CM, Lee YC, Tu HP, Huang CK, Wu MT, Chang CY, Lee CT, Wu MS, Lin JT, Wang WM. The relationship between visceral adiposity and the risk of erosive esophagitis in severely obese Chinese patients. Obesity (Silver Spring). 2010;18:2165-2169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 12. | Pandolfino JE, Kwiatek MA, Kahrilas PJ. The pathophysiologic basis for epidemiologic trends in gastroesophageal reflux disease. Gastroenterol Clin North Am. 2008;37:827-843, viii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 13. | El-Serag HB, Tran T, Richardson P, Ergun G. Anthropometric correlates of intragastric pressure. Scand J Gastroenterol. 2006;41:887-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 14. | El-Serag HB, Ergun GA, Pandolfino J, Fitzgerald S, Tran T, Kramer JR. Obesity increases oesophageal acid exposure. Gut. 2007;56:749-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 207] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 15. | Wilson LJ, Ma W, Hirschowitz BI. Association of obesity with hiatal hernia and esophagitis. Am J Gastroenterol. 1999;94:2840-2844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 265] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 16. | Ze EY, Kim BJ, Kang H, Kim JG. Abdominal Visceral to Subcutaneous Adipose Tissue Ratio Is Associated with Increased Risk of Erosive Esophagitis. Dig Dis Sci. 2017;62:1265-1271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 17. | Savarino E, Tutuian R, Zentilin P, Dulbecco P, Pohl D, Marabotto E, Parodi A, Sammito G, Gemignani L, Bodini G, Savarino V. Characteristics of reflux episodes and symptom association in patients with erosive esophagitis and nonerosive reflux disease: Study using combined impedance-pH off therapy. Am J Gastroenterol. 2010;105:1053-1061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 178] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 18. | Hampel H, Abraham NS, El-Serag HB. Meta-analysis: Obesity and the risk for gastroesophageal reflux disease and its complications. Ann Intern Med. 2005;143:199-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 828] [Cited by in RCA: 796] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 19. | Ortiz V, Ponce M, Fernández A, Martínez B, Ponce JL, Garrigues V, Ponce J. Value of heartburn for diagnosing gastroesophageal reflux disease in severely obese patients. Obesity (Silver Spring). 2006;14:696-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 20. | Csendes A, Burgos AM, Smok G, Beltran M. Endoscopic and histologic findings of the foregut in 426 patients with morbid obesity. Obes Surg. 2007;17:28-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 73] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 21. | Verset D, Houben JJ, Gay F, Elcheroth J, Bourgeois V, Van Gossum A. The place of upper gastrointestinal tract endoscopy before and after vertical banded gastroplasty for morbid obesity. Dig Dis Sci. 1997;42:2333-2337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 47] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 22. | Dutta SK, Arora M, Kireet A, Bashandy H, Gandsas A. Upper gastrointestinal symptoms and associated disorders in morbidly obese patients: A prospective study. Dig Dis Sci. 2009;54:1243-1246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 23. | Martín-Pérez J, Arteaga-González I, Martín-Malagón A, Díaz-Luis H, Casanova-Trujillo C, Carrillo-Pallarés A A. Frequency of abnormal esophageal acid exposure in patients eligible for bariatric surgery. Surg Obes Relat Dis. 2014;10:1176-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 24. | Carabotti M, Avallone M, Cereatti F, Paganini A, Greco F, Scirocco A, Severi C, Silecchia G. Usefulness of Upper Gastrointestinal Symptoms as a Driver to Prescribe Gastroscopy in Obese Patients Candidate to Bariatric Surgery. A Prospective Study. Obes Surg. 2016;26:1075-1080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 25. | Mora F, Cassinello N, Mora M, Bosca M, Minguez M, Ortega J. Esophageal abnormalities in morbidly obese adult patients. Surg Obes Relat Dis. 2016;12:622-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Mechanick JI, Youdim A, Jones DB, Timothy Garvey W, Hurley DL, Molly McMahon M, Heinberg LJ, Kushner R, Adams TD, Shikora S, Dixon JB, Brethauer S. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient--2013 update: Cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis. 2013;9:159-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 447] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 27. | Fried M, Yumuk V, Oppert JM, Scopinaro N, Torres A, Weiner R, Yashkov Y, Frühbeck G; International Federation for Surgery of Obesity and Metabolic Disorders-European Chapter (IFSO-EC); European Association for the Study of Obesity (EASO); European Association for the Study of Obesity Obesity Management Task Force (EASO OMTF). Interdisciplinary European guidelines on metabolic and bariatric surgery. Obes Surg. 2014;24:42-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 386] [Cited by in RCA: 423] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 28. | Evans JA, Muthusamy VR, Acosta RD, Bruining DH, Chandrasekhara V, Chathadi KV, Eloubeidi MA, Fanelli RD, Faulx AL, Fonkalsrud L, Khashab MA, Lightdale JR, Pasha SF, Saltzman JR, Shaukat A, Wang A, Stefanidis D, Richardson WS, Khothari SN, Cash BD; ASGE Standards of Practice Committee. The role of endoscopy in the bariatric surgery patient. Surg Obes Relat Dis. 2015;11:507-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 29. | Sauerland S, Angrisani L, Belachew M, Chevallier JM, Favretti F, Finer N, Fingerhut A, Garcia Caballero M, Guisado Macias JA, Mittermair R, Morino M, Msika S, Rubino F, Tacchino R, Weiner R, Neugebauer EA; European Association for Endoscopic Surgery. Obesity surgery: Evidence-based guidelines of the European Association for Endoscopic Surgery (EAES). Surg Endosc. 2005;19:200-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 259] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 30. | Salama A, Saafan T, El Ansari W, Karam M, Bashah M. Is Routine Preoperative Esophagogastroduodenoscopy Screening Necessary Prior to Laparoscopic Sleeve Gastrectomy? Review of 1555 Cases and Comparison with Current Literature. Obes Surg. 2018;28:52-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 31. | Zanotti D, Elkalaawy M, Hashemi M, Jenkinson A, Adamo M. Current Status of Preoperative Oesophago-Gastro-Duodenoscopy (OGD) in Bariatric NHS Units-a BOMSS Survey. Obes Surg. 2016;26:2257-2262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 32. | Oor JE, Roks DJ, Ünlü Ç, Hazebroek EJ. Laparoscopic sleeve gastrectomy and gastroesophageal reflux disease: A systematic review and meta-analysis. Am J Surg. 2016;211:250-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 167] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 33. | Felsenreich DM, Kefurt R, Schermann M, Beckerhinn P, Kristo I, Krebs M, Prager G, Langer FB. Reflux, Sleeve Dilation, and Barrett's Esophagus after Laparoscopic Sleeve Gastrectomy: Long-Term Follow-Up. Obes Surg. 2017;27:3092-3101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 220] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 34. | Sharara AI, Rimmani HH, Al Abbas AI, Safadi B, Shayto RH, Aridi H, Shaib Y, R. Erosive esophagitis is prevalent and Predictable by pre-operative gerdq questionnaire in Obese individuals and is associated with the need for Continued ppi use after laparoscopic sleeve Gastrectomy. UEG Week 2017 Oral Presentations. United Eur Gastroent. 2017;5:A1-A160. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 35. | Madhok BM, Carr WR, McCormack C, Boyle M, Jennings N, Schroeder N, Balupuri S, Small PK. Preoperative endoscopy may reduce the need for revisional surgery for gastro-oesophageal reflux disease following laparoscopic sleeve gastrectomy. Clin Obes. 2016;6:268-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 36. | Cottam D, Qureshi FG, Mattar SG, Sharma S, Holover S, Bonanomi G, Ramanathan R, Schauer P. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc. 2006;20:859-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 452] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 37. | Moon Han S, Kim WW, Oh JH. Results of laparoscopic sleeve gastrectomy (LSG) at 1 year in morbidly obese Korean patients. Obes Surg. 2005;15:1469-1475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 255] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 38. | Melissas J, Koukouraki S, Askoxylakis J, Stathaki M, Daskalakis M, Perisinakis K, Karkavitsas N. Sleeve gastrectomy: A restrictive procedure? Obes Surg. 2007;17:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 269] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 39. | Melissas J, Daskalakis M, Koukouraki S, Askoxylakis I, Metaxari M, Dimitriadis E, Stathaki M, Papadakis JA. Sleeve gastrectomy-a "food limiting" operation. Obes Surg. 2008;18:1251-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 198] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 40. | Gibson SC, Le Page PA, Taylor CJ. Laparoscopic sleeve gastrectomy: Review of 500 cases in single surgeon Australian practice. ANZ J Surg. 2015;85:673-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 41. | DuPree CE, Blair K, Steele SR, Martin MJ. Laparoscopic sleeve gastrectomy in patients with preexisting gastroesophageal reflux disease: A national analysis. JAMA Surg. 2014;149:328-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 232] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 42. | Sheppard CE, Sadowski DC, de Gara CJ, Karmali S, Birch DW. Rates of reflux before and after laparoscopic sleeve gastrectomy for severe obesity. Obes Surg. 2015;25:763-768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 43. | Carter PR, LeBlanc KA, Hausmann MG, Kleinpeter KP, deBarros SN, Jones SM. Association between gastroesophageal reflux disease and laparoscopic sleeve gastrectomy. Surg Obes Relat Dis. 2011;7:569-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 109] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 44. | Tai CM, Huang CK, Lee YC, Chang CY, Lee CT, Lin JT. Increase in gastroesophageal reflux disease symptoms and erosive esophagitis 1 year after laparoscopic sleeve gastrectomy among obese adults. Surg Endosc. 2013;27:1260-1266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 133] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 45. | Howard DD, Caban AM, Cendan JC, Ben-David K. Gastroesophageal reflux after sleeve gastrectomy in morbidly obese patients. Surg Obes Relat Dis. 2011;7:709-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 117] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 46. | Rebecchi F, Allaix ME, Giaccone C, Ugliono E, Scozzari G, Morino M. Gastroesophageal reflux disease and laparoscopic sleeve gastrectomy: A physiopathologic evaluation. Ann Surg. 2014;260:909-914; discussion 914-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 143] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 47. | Rebecchi F, Allaix ME, Patti MG, Schlottmann F, Morino M. Gastroesophageal reflux disease and morbid obesity: To sleeve or not to sleeve? World J Gastroenterol. 2017;23:2269-2275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 36] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 48. | Del Genio G, Tolone S, Limongelli P, Brusciano L, D'Alessandro A, Docimo G, Rossetti G, Silecchia G, Iannelli A, del Genio A, del Genio F, Docimo L. Sleeve gastrectomy and development of "de novo" gastroesophageal reflux. Obes Surg. 2014;24:71-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 133] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 49. | Gorodner V, Buxhoeveden R, Clemente G, Solé L, Caro L, Grigaites A. Does laparoscopic sleeve gastrectomy have any influence on gastroesophageal reflux disease? Preliminary results. Surg Endosc. 2015;29:1760-1768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 78] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 50. | Georgia D, Stamatina T, Maria N, Konstantinos A, Konstantinos F, Emmanouil L, Georgios Z, Dimitrios T. 24-h Multichannel Intraluminal Impedance PH-metry 1 Year After Laparocopic Sleeve Gastrectomy: An Objective Assessment of Gastroesophageal Reflux Disease. Obes Surg. 2017;27:749-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 51. | Langer FB, Bohdjalian A, Shakeri-Leidenmühler S, Schoppmann SF, Zacherl J, Prager G. Conversion from sleeve gastrectomy to Roux-en-Y gastric bypass--indications and outcome. Obes Surg. 2010;20:835-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 119] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 52. | Juodeikis Ž, Brimas G. Long-term results after sleeve gastrectomy: A systematic review. Surg Obes Relat Dis. 2017;13:693-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 53. | Sebastianelli L, Benois M, Vanbiervliet G, Bailly L, Robert M, Turrin N, Gizard E, Foletto M, Bisello M, Albanese A, Santonicola A, Iovino P, Piche T, Angrisani L, Turchi L, Schiavo L, Iannelli A. Systematic Endoscopy 5 Years After Sleeve Gastrectomy Results in a High Rate of Barrett's Esophagus: Results of a Multicenter Study. Obes Surg. 2019;29:1462-1469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 185] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 54. | Mion F, Tolone S, Garros A, Savarino E, Pelascini E, Robert M, Poncet G, Valette PJ, Marjoux S, Docimo L, Roman S. High-resolution Impedance Manometry after Sleeve Gastrectomy: Increased Intragastric Pressure and Reflux are Frequent Events. Obes Surg. 2016;26:2449-2456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 55. | Altieri MS, Shroyer KR, Pryor A, Pagnotti GM, Ete Chan M, Talamini M, Telem DA. The association between sleeve gastrectomy and histopathologic changes consistent with esophagitis in a rodent model. Surg Obes Relat Dis. 2015;11:1289-1294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 56. | Klaus A, Weiss H. Is preoperative manometry in restrictive bariatric procedures necessary? Obes Surg. 2008;18:1039-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 63] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 57. | Lazoura O, Zacharoulis D, Triantafyllidis G, Fanariotis M, Sioka E, Papamargaritis D, Tzovaras G. Symptoms of gastroesophageal reflux following laparoscopic sleeve gastrectomy are related to the final shape of the sleeve as depicted by radiology. Obes Surg. 2011;21:295-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 58. | Hayat JO, Wan A. The effects of sleeve gastectomy on gastro-esophageal reflux and gastro-esophageal motility. Expert Rev Gastroenterol Hepatol. 2014;8:445-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 59. | Himpens J, Dapri G, Cadière GB. A prospective randomized study between laparoscopic gastric banding and laparoscopic isolated sleeve gastrectomy: Results after 1 and 3 years. Obes Surg. 2006;16:1450-1456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 494] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 60. | Baumann T, Kuesters S, Grueneberger J, Marjanovic G, Zimmermann L, Schaefer AO, Hopt UT, Langer M, Karcz WK. Time-resolved MRI after ingestion of liquids reveals motility changes after laparoscopic sleeve gastrectomy--preliminary results. Obes Surg. 2011;21:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 61. | Keidar A, Appelbaum L, Schweiger C, Elazary R, Baltasar A. Dilated upper sleeve can be associated with severe postoperative gastroesophageal dysmotility and reflux. Obes Surg. 2010;20:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 62. | Dapri G, Cadière GB, Himpens J. Laparoscopic seromyotomy for long stenosis after sleeve gastrectomy with or without duodenal switch. Obes Surg. 2009;19:495-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |