Published online Aug 14, 2019. doi: 10.3748/wjg.v25.i30.4092
Peer-review started: March 28, 2019
First decision: May 24, 2019
Revised: July 3, 2019
Accepted: July 5, 2019
Article in press: July 5, 2019
Published online: August 14, 2019
Processing time: 139 Days and 20 Hours
Estrogens play important roles in the development and progression of multiple tumor types. Accumulating evidence points to the significance of estrogen action not only in tumors of hormonally regulated tissues such as the breast, endometrium and ovary, but also in the development of colorectal cancer (CRC). The effects of estrogens in physiological and pathophysiological conditions are mediated by the nuclear estrogen receptors α and β, as well as the membrane-bound G protein-coupled estrogen receptor (GPER). The roles of GPER in CRC development and progression, however, remain poorly understood. Studies on the functions of GPER in the colon have shown that this estrogen receptor regulates colonic motility as well as immune responses in CRC-associated diseases, such as Crohn’s disease and ulcerative colitis. GPER is also involved in cell cycle regulation, endoplasmic reticulum stress, proliferation, apoptosis, vascularization, cell migration, and the regulation of fatty acid and estrogen metabolism in CRC cells. Thus, multiple lines of evidence suggest that GPER may play an important role in colorectal carcinogenesis. In this review, we present the current state of knowledge regarding the contribution of GPER to colon function and CRC.
Core tip: G protein-coupled estrogen receptor (GPER) is a membrane-bound estrogen receptor that participates in the rapid non-genomic actions of estrogens involving numerous downstream signaling pathways. GPER is expressed in the gastrointestinal tract and is engaged in physiological and pathophysiological processes in the colon. This review aims to assess the significance of GPER expression and estrogenic signaling in colorectal carcinogenesis.
- Citation: Jacenik D, Beswick EJ, Krajewska WM, Prossnitz ER. G protein-coupled estrogen receptor in colon function, immune regulation and carcinogenesis. World J Gastroenterol 2019; 25(30): 4092-4104
- URL: https://www.wjgnet.com/1007-9327/full/v25/i30/4092.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i30.4092
The roles of estrogen (17β-estradiol) and its receptors in gastrointestinal (GI) diseases are complex. The lower incidence rate of colon cancer in women compared to men suggests a protective role for the female sex hormone estrogen[1,2]. However, because estrogen has at least three receptors, contradictory pro- and anti-tumorigenic mechanisms have been observed depending on the receptor-mediated mechanisms examined. The effects of estrogens are mediated by receptors including the nuclear estrogen receptors (i.e., ERα and ERβ)[3] and the G protein-coupled estrogen receptor (GPER, previously known as GPR30)[4]. GPER is a seven-transmembrane receptor cloned from the ER-positive MCF-7 cell line[5], among other sources[6-11]. As opposed to nuclear estrogen receptors that are primarily responsible for the genomic actions of estrogens, GPER initiates many rapid non-genomic actions of estrogen involving secondary messengers, which can also ultimately lead to secondary gene expression changes[4,12,13]. GPER activity is not only stimulated by 17β-estradiol, but also by numerous xeno- and phyto-estrogens (e.g., bisphenols and genistein), clinically relevant anti-hormonal therapeutic agents (e.g., tamoxifen and fulvestrant) and synthetic GPER-selective ligands (e.g., G-1, G15 and G36) as summarized in Table 1[4]. GPER expression and activity, the latter often defined employing GPER-selective ligands, have been linked to many aspects of normal physiology and patho-physiology[14-16].
| Agonist | Type |
| Estrogen (17β-estradiol) | Natural steroid |
| Bisphenol A | Synthetic xenoestrogen |
| Genistein | Natural phytoestrogen |
| Tamoxifen | Synthetic therapeutic |
| Fulvestrant (ICI 182.780) | Synthetic therapeutic |
| G-1 | Synthetic selective ligand |
| Antagonist | Type |
| G15 | Synthetic selective ligand |
| G36 | Synthetic selective ligand |
Experimental evidence strongly suggests that estrogen, and therefore its receptors, plays important roles in neoplastic transformation of the colon[17,18]. One challenge in understanding these roles is the often-contradictory results regarding estrogen receptors and their roles in GI diseases. Although estrogen is generally thought to be anti-inflammatory, and inflammation typically contributes to carcinogenesis, the roles of individual estrogen receptors are complex, which may result in conflicting observations. Among the various estrogen receptors, ERβ, in particular, has been suggested to act as a tumor suppressor in colorectal cancer (CRC) and serves as a prognostic factor for CRC progression. Stevanato Filho et al[18] observed significantly lower ERβ levels in CRC patients with clinical stage III and IV disease compared to patients with stage I and II disease. The absence of ERβ in CRC is thus a poor prognostic factor associated with higher mortality [hazard ratio = 3.0, 95% confidence interval (CI) = 1.2-7.5]. However, in contrast to its established tumor-protective role, Cho et al[17] used ER-deficient mice to show that, in addition to ERβ, ERα is also crucial for enterocyte growth and differentiation. Both nuclear estrogen receptors appear to be important modulators of colon neoplastic transformation as ERβ or ERα deficiency was associated with tumor progression and abnormal mucosal histology in an APC-dependent tumorigenesis model employing C57BL/6-Min/+ mice[17]. However, in addition to the nuclear estrogen receptors, accumulating evidence suggests that GPER is also involved in many aspects of CRC cell pathophysiology. In this review, we summarize the evidence for GPER expression and function in the colon and in colorectal carcinogenesis.
Colonic motility regulates the frequency and timing of defecation as well as the consistency of stools, with the main symptoms of colon movement disorders being constipation and diarrhea. A large retrospective study demonstrated a positive association between chronic constipation and a higher prevalence, as well as risk, of benign neoplasm and CRC[19]. Moreover, incidence rate ratio analyses have shown that an increased risk of both benign and malignant colorectal neoplasms appears to be directly related to the severity of constipation. Additional evidence linked constipation, defined as fewer than three bowel movements per week, with an overall 2.4-fold increase in CRC development[20]. Interestingly, although women overall have a slightly lower risk of developing CRC than men, women with constipation appear to be more prone to CRC development (odds ratio = 2.7, 95%CI = 1.5-5.0) compared to men with constipation (odds ratio = 1.7, 95%CI = 0.6-4.9). Mechanistically, although colon motor dysfunction may increase exposure of the GI tract to carcinogens, evidence suggests that colonic transit time also influences bacterial metabolism and mucosal turnover, both of which may affect CRC development[21].
Two independent studies have revealed that GPER activity influences colonic motility in vivo[22,23]. Li et al[22] demonstrated that GPER activity affects colonic motility in multiple phases of the estrous cycle in female mice. Colonic transit time, which was significantly longer during the proestrus and estrus phases (vs diestrus phase), was reduced by selective GPER inhibition with the GPER antagonist G15[24,25]. Similarly, in ovariectomized mice, acute estrogen treatment increased colonic transit time, and was reversed by co-administration of G15. Ex vivo, GPER activation with the GPER-selective agonist G-1[26] inhibited circular muscle strip contraction in a nitric oxide (NO)-dependent manner, and stimulated NO production in cultured myenteric nitrergic neurons, which together act to reduce colonic motor function. Further supporting this mechanism, Zielińska et al[23] and Li et al[22], demonstrated, using the colonic bead expulsion test, that GPER stimulation by G-1 or estrogen treatment prolongs colonic transit time in both male and female mice. The inhibitory effect of GPER activation on the number of fecal pellets excreted was further confirmed in vivo employing a mouse model of hypermotility. The potential mechanism by which GPER affects colonic motility appears to involve inhibition of muscle contractility, as determined by electrical field- and bethanechol-stimulated longitudinal smooth muscle contractions[23].
Further indirect evidence highlighting the importance of GPER in the regulation of colonic motor function is derived from studies of irritable bowel syndrome (IBS) patients[27,28]. Alterations in GPER mRNA and protein expression in the colonic mucosa, as well as serum estrogen levels, were reported in samples from IBS patients with constipation (IBS-C) or diarrhea (IBS-D)[27]. An increased number of GPER-positive colonic mast cells in IBS-D patients compared to either healthy control or IBS-C patients has also been observed[28]. GPER-positive cell staining in the colonic mucosa also correlated with increased abdominal pain severity in IBS-D patients and increased expression of GPER in the cytoplasm of mast cells, which are associated with abdominal bloating frequency and dysmotility-like dyspeptic symptom severity and frequency in IBS patients. These results suggest a possible role for GPER in mast cells and immune function at large, which play an important role in intestinal function and disease[29].
Crohn’s disease (CD) and ulcerative colitis (UC), the two most commonly diagnosed types of inflammatory bowel disease (IBD), are characterized by chronic colon inflammation and are associated with a higher risk of CRC development[27]. A meta-analysis performed by Flores et al. reported that patients with histologic inflammation are more prone to colorectal dysplasia and/or cancer development (odds ratio = 2.6, 95%CI = 1.5-4.5) compared to patients without mucosal inflammation[30]. For UC patients, the increased risk of CRC development appears to be related to disease duration[31]. A meta-analysis by von Roon et al[32] further revealed that patients with CD experience a higher relative risk of CRC development, related to both the anatomic localization of disease and patient age. CD patients with disease affecting ileocolic and colon segments experience relative risks of 4.6 (95%CI = 2.1-10.3) and 13.4 (95%CI = 5.7-13.2) for the development of ileocolic and colorectal cancer, respectively. Furthermore, population-based studies have shown that the presence of CD before the age of 25 and 30 correlates with very high relative risks of CRC development (21.5, 95%CI = 11.4-40.4 and 9.5, 95%CI = 3.1-23.2, before the age of 25 and 30, respectively)[32].
Immunomodulatory roles for estrogen and its receptors in the pathogenesis of IBD have been documented[17,33-36], but the specific mechanisms and roles of GPER in modulating immune responses remain unclear. In addition to the nuclear estrogen receptors, GPER is expressed at both the mRNA and protein levels in intestinal samples obtained from both IBD patient subtypes (i.e., CD and UC)[37]. In patients with CD, GPER protein content was increased in non-inflamed colon areas compared to the control group but lower in inflamed colon tissue compared to non-inflamed colon, suggesting the possibility of a complex protective role for GPER in colitis[37]. To date, although there is no direct evidence showing GPER regulates immune responses in CRC or the CRC microenvironment, several studies show that GPER is indeed expressed in multiple immune cells, including monocytes/macrophages, neutrophils, B and T cells[38-44]. Accumulating evidence suggests that GPER modulates cytokine and cytokine receptor expression in immune cells, cancer cells and cancer-associated fibroblasts (CAFs). In breast cancer, estrogen signaling through GPER regulates CAF-mediated progression of breast cancer and is associated with several signaling pathways leading to the modulation of gene expression[45-48]. Pro-tumorigenic effects of GPER activation result from receptor tyrosine kinase [e.g., insulin-like growth factor receptor I and epidermal growth factor receptor (EGFR)] modulation and downstream effector proteins such as AKT and extracellular signal-regulated kinase (ERK)[47]. GPER-mediated ERK activation in breast CAFs leads to changes in the expression of the proto-oncogenes c-Fos, cyclin D1 and connective tissue growth factor (CTGF)[45,46,48], which play an important role in many cancers including CRC[49].
GPER also plays an important role in the regulation of interleukin (IL)-1β and IL-1R expression in CAFs and breast cancer cells, leading to a gene expression profile associated with cancer cell invasiveness[50]. GPER’s immunomodulatory activity occurs not only in CAFs, but also in several immune cell types. In lipopolysaccharide (LPS)-stimulated primary human macrophages, GPER activation leads to inhibition of tumor necrosis factor-α (TNF-α), IL-6, IL-12 and C-C motif ligand (CCL) 5 secretion[38]. A similar effect was observed in the murine macrophage cell line RAW 264.7, where G-1 treatment inhibited TNF-α secretion from LPS-stimulated cells. The anti-inflammatory properties of GPER activation were confirmed in an animal model of multiple sclerosis (experimental autoimmune encephalomyelitis), where G-1 therapy reduced the severity of symptoms and the number of nervous system-infiltrating macrophages[38]. The ability of GPER to inhibit LPS-induced IL-6 expression in murine macrophages was shown to occur via NF-κB signaling[51]. Together, these data suggest a potentially central role for GPER in regulating inflammation during IBD.
GPER activity in macrophages may also be important in tumor development and progression within the GI tract. The importance of tumor-associated macrophages (TAMs), specifically in pancreatic cancer has been recently highlighted[41]. In a murine model of pancreatic cancer, treatment with tamoxifen, which acts as a GPER agonist (see Table 1), reduced the percentage of tissue macrophages as well as the polarization of TAMs to the M2 phenotype. In macrophage-like murine RAW 264.7 cells, GPER regulated focal adhesions, cell-extracellular matrix attachment and invasion[41]. Macrophages are associated with the development and progression of colitis and neoplastic transformation of the colon. Whereas M1 macrophages are elevated in intestinal samples from IBD patients where they promote inflammation, during CRC progression, increased M2/M1 ratios appear to correlate with increased liver me-tastasis[52-54]. Overall, these complex actions of GPER suggest a “protective” role in CRC development and progression.
IL-6 is an important mediator of cancer cell function and tumor development; however, conflicting data exist as to the role of GPER in its regulation. Bisphenol A (BPA), a non-selective ER/GPER agonist (Table 1) induces proliferation, migration and invasion of laryngeal cancer cells, as a result of increased IL-6 mRNA expression, potentially via GPER activation[55]. Furthermore, treatment with the GPER-selective inhibitor G15 and/or siRNA targeting of GPER attenuated cell proliferation, migration and IL-6 expression of laryngeal cancer cells. These processes involved signal transducer and activator of transcription 3 (STAT3) activation by GPER/IL-6 signaling as assessed by siRNA targeting of IL-6 or GPER[55]. As STAT3 is known to regulate both pro- and anti-inflammatory cytokine production, the exact role of GPER in these complex processes needs to be further investigated. It should be noted that BPA action may also depend on both the cell type and the nuclear estrogen receptor status of cells[56]. In contrast to these results, in a more “typical” anti-inflammatory estrogen response, GPER inhibited IL-6 expression via NF-κB inhibition, leading to reduced migration of triple negative MDA-MB-231 breast cancer cells[57]. As a result, in MDA-MB-231 xenograft tumors, GPER activation inhibited both angiogenesis and metastasis. GPER-mediated inhibition of IL-6 production was also demonstrated in TNFα-stimulated breast cancer cells and osteosarcoma cells where GPER inhibited IL-6 expression, suppressing migration and invasion[58,59]. Overall, these results suggest the effects of GPER expression and activity may be tumor type- and context-specific.
Accumulating evidence reveals that GPER is a crucial immunoregulatory factor not only in macrophages, but also in granulocytes, which significantly impact IBD progression. GPER modulates multiple mediators of the immune response in fish acidophilic granulocytes[40], including IL-1β and IL-10, as well as prostaglandin-endoperoxide synthase 2 and prostaglandin D2 synthase expression, through activation of the cAMP/PKA/CREB signaling pathway. These mechanisms are important in IBD[60] and thus should be further examined. In a murine model of castration-resistant prostate cancer, tumor neutrophil influx was observed following G-1 therapy and was associated with extensive tumor necrosis, suggesting a previously unrecognized role for GPER in men[61].
Multiple reports have highlighted that GPER also stimulates anti-inflammatory immune responses through the modulation of T cell function and cytokine expression, representing a potential therapeutic target for certain autoimmune diseases. In one study of autoimmune encephalomyelitis, splenocytes cultured from G-1-treated mice and stimulated with antigen, yielded lower IFN-γ, TNF-α, IL-17, CCL4 and CCL5 protein levels in supernatants compared to splenocytes from vehicle-treated mice[38]. In other studies, GPER activation increased production of IL-10 (an anti-inflammatory cytokine that inhibits several immune cell types) in CD4+ T cells under Th17 polarizing conditions, generating hybrid autoregulatory T cell populations[39]. This effect was abolished by GPER and ERK inhibitors, but not by p38 or Jun N-terminal kinase inhibitors, suggesting that GPER regulates IL-10 production through ERK signaling. GPER’s anti-inflammatory action may be mediated through specific immune cell types as a higher frequency of IL-10-producing CD4+ (but not CD8+) T cells is observed in mouse splenocytes following GPER activation[62]. Moreover, G-1-treated mice showed a significantly higher population of IL-10-producing GPER+ CD4+ T cells, consistent with the ability of GPER to regulate IL-10 production.
In additional T cell studies employing a mouse model of asthma, G-1 reduced the level of Th2 cytokines, such as IL-5 and IL-13, in bronchoalveolar lavage fluid, suggesting negative regulation of acute asthma through IL-10-producing T cells[62]. In IBD, several intestinal T cell populations are dysregulated, with the extent of dysregulation correlating to disease severity. In clinical samples obtained from IBD patients with active disease, higher and lower levels of CD4+ T cells and CD8+ T cells, respectively, are present compared to healthy controls and IBD patients with inactive disease[63]. However, in CRC tumors, only an increased percentage of CD4+ T cells, but not CD8+ T cells was observed[64]. The significance of regulatory T cells in in-flammation and CRC progression, together with the anti-inflammatory properties of GPER, indicates that GPER activation in CD4+ T cells may be a promising target to modulate colon immune responses. However, although GPER activity in T cells generally appears to promote anti-inflammatory responses, which would have a positive impact in inflammatory diseases, such responses may have a potentially negative impact in cancer, where immune monitoring is critical. Taken together, these results suggest that as an important regulator of colon inflammation and immune responses in CRC, GPER action may also be important in the context of long-term immune response deregulation, which is critical in CRC development associated with IBD.
Accumulating evidence indicates important roles for estrogen and its receptors, including GPER, as revealed by selective ligands such as G-1, in the regulation of cancer cell growth, survival and function[65]. GPER appears to regulate cancer cell proliferation and survival not only in estrogen-associated cancers such as breast[66], ovarian[67] or endometrial[68,69], but also in other cancer types not traditionally as-sociated with estrogen (e.g., lung and CRC)[47,70-77]. Several cellular mechanisms are regulated by GPER in cancer cells, including cell cycle, endoplasmic reticulum stress and apoptosis. In both HCT-116 and SW-480 CRC cells, GPER activation by G-1 led to cell cycle arrest and inhibition of proliferation[74]. The higher proportion of HCT-116 cells in the apoptotic sub-G1 phase, as well as lower mitochondrial membrane polarity, following GPER activation, suggests that GPER activation promotes apoptosis in CRC cells. Consistent with this, G-1 treatment induced up-regulation of pro-apoptotic factors such as Bcl-2-associated X protein, cyclin-dependent kinase inhibitor 1 (p21), and cleaved caspase-3, with down-regulation of anti-apoptotic factors, such as B-cell lymphoma 2 (Bcl-2) and procaspase-3.
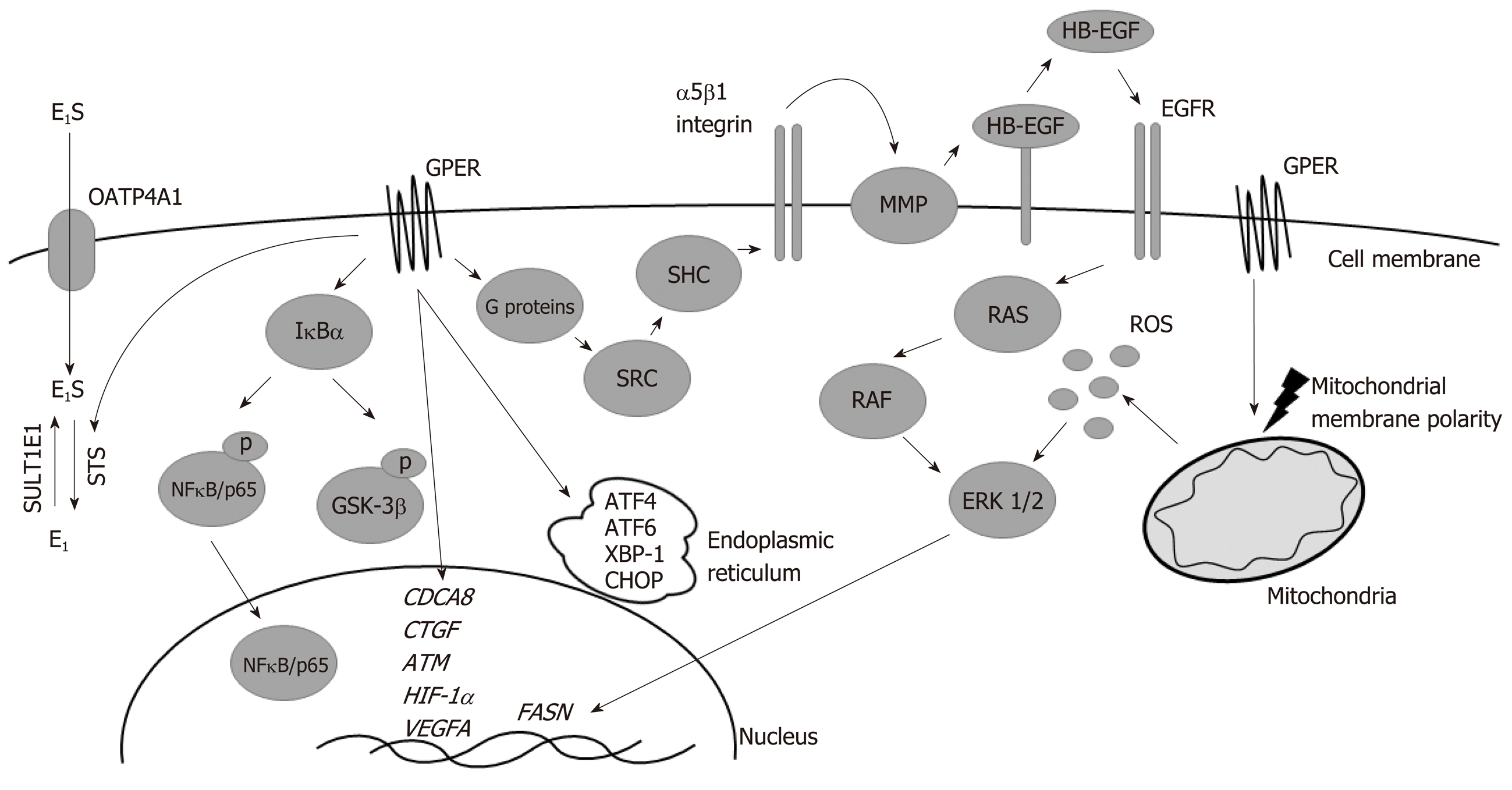
Cancer cell growth arrest and apoptosis are also regulated by endoplasmic reticulum stress signals (reviewed by Sano and Reed[78]). Among several factors modulating endoplasmic reticulum stress, protein expression of ATF4 and 6, XBP-1 and CHOP increased upon G-1 treatment of HCT-116 cells[74] (Figure 1). Elevated reactive oxygen species (ROS) and ERK1/2 phosphorylation, resulting from GPER-mediated signaling, also contributed to growth arrest of HCT-116 cells[74]. These pathways are particularly important in chronic inflammation, where immune cell-released ROS and cytokines activate the NF-κB pathway. GPER mediates inhibition of IκBα, which leads to phosphorylation of NFκB/p65 and nuclear translocation, as well as GSK-3β phosphorylation (Figure 1). Constitutive NF-κB activity in cancer cells drives neoplastic transformation and tumor progression, affecting cancer cell proliferation and survival[79-81] as well as tumor angiogenesis[82], metabolism[83,84], immune response[85] and metastatic potential of cancer cells[82].
GPER may also be a regulator of genetic transmission during cell division in CRC cells. In HT-29 cells, BPA up-regulated mRNA levels of CDCA8 (also known as Borealin), a crucial member of the chromosomal passenger complex that mediates several events during mitosis[86] (Figure 1). Although BPA is classically thought to function through the nuclear estrogen receptors, particularly ERα, it also binds and activates GPER (Table 1)[4]. Since HT-29 cells express only GPER and ERβ, the effects of BPA on CDCA8 expression are likely mediated, at least in part, by GPER[86]. GPER also regulates ataxia telangiectasia mutated (ATM), an important protein in carcinogenesis, through regulation of the cell cycle and DNA repair (reviewed by Branzei and Foiani[87]). In a human breast cancer study, ATM phosphorylation was positively correlated with lymph node metastasis[88]. However, in CRC and colon adenomas, increased ATM promoter methylation was observed compared to control tissue[89]. Estrogen, acting through GPER, represses ATM expression under both normoxic and hypoxic conditions in HT-29 cells[70], with the effect of estrogen on ATM expression being stronger under hypoxic conditions, suggesting an effect of oxygen levels on estrogen’s effects.
Hypoxia plays an important role in tumor progression, affecting tumor vas-cularization, epithelial-mesenchymal transition and metastasis as well as chemo- and radio-resistance[90]. Under hypoxic conditions, the pathways activated by hypoxia-inducible factor (HIF) control numerous cellular proteins, including vascular endothelial growth factor (VEGF), driving tumor growth. GPER mediates estrogen’s suppression or enhancement of HIF-1α and VEGFA expression under normoxic and hypoxic conditions, respectively[70]. Furthermore, GPER mediates opposing functions in HT-29 and DLD1 CRC cells, dependent on oxygen levels, with GPER agonists suppressing proliferation under normoxic conditions, but increasing proliferation under hypoxic conditions.
The production and presence of local estrogens may be important in the development and progression of CRC. The ability of GPER to regulate CRC cell proliferation may thus represent a mechanism through which the known effects of estrogen on CRC are mediated. In vitro and in vivo studies have shown that estrogen promotes CRC in part through the dysregulation of enzymes involved in estrogen metabolism. Specifically, in post-menopausal women and men with CRC, compared to age-matched control groups, higher colonic activity of the enzyme steroid sulfatase (STS), which converts circulating sulfated estrogens to the active forms, has been observed[91]. Furthermore, increased protein and mRNA levels of 17β-hydroxysteroid dehydrogenase (HSD17) B7 and B12 (responsible for the conversion of estrone to estrogen) are present in human CRC tissue samples, whereas mRNA and protein levels of the enzyme catalyzing conversion of estrogen to estrone (i.e., HSD17B2) are decreased. Experimental evidence strongly suggests that supplementation with estrogen or STS overexpression increases CRC cell proliferation in vitro and in vivo. The latter was demonstrated using a murine CRC xenograft model with STS-overexpressing cells[91]. Activation of GPER by estrogen derived from these multiple sources results in the up-regulation of CTGF, EGR1 and ATF3, with CTGF up-regulation being required for enhanced cell proliferation, with estrone sulfate transport and GPER-stimulated STS activity producing a novel estrogen-generating positive feedback loop (Figure 1) that may play an important role in CRC pro-gression[92]. Interestingly, in addition to estrogen and G-1, the breast cancer the-rapeutic agents, tamoxifen and fulvestrant (ICI 182.780), also increased STS activity (in a GPER-dependent manner), suggesting these drugs could have a negative impact on CRC development and progression.
An increasing number of studies describes a modulatory effect of GPER activity on cancer cell migration and invasion in multiple cancer types, including breast (including triple-negative), endometrial, ovarian, lung, thyroid, kidney, and granulosa cell[57,71,77,93-105]. In previous work that demonstrated the dual role of GPER activation on CRC cell proliferation (see above), oxygen-dependent GPER-mediated effects on the migration of HT-29 and DLD-1 cells was also observed[70]. Scratch wound and Boyden chamber assays demonstrated that both G-1 and estrogen inhibited migration under normoxic conditions, whereas GPER stimulation enhanced migration of CRC cells under hypoxic conditions. Fatty acid synthase (FASN), a key lipogenic enzyme that affects neoplastic transformation of the breast, colon and liver, has been described as a metabolic oncogene[106-108] and is regulated by many factors, including estrogens[108,109]. GPER stimulation by G-1 or estrogen increases expression and activity of FASN via EGFR/ERK/c-Fos/AP-1 signaling, resulting in increased growth and migration of CRC cells, which was in turn decreased by a FASN inhibitor[108]. Thus, GPER activity regulates cell motility through multiple complex mechanisms (Figure 1).
There is conflicting evidence that GPER may act as a tumor suppressor as well as a tumor promoter in CRC. A seven-fold down-regulation of GPER mRNA in CRC samples compared to adjacent control tissue has been observed[74]. Immuno-histochemical analysis confirmed a decrease in GPER protein expression in CRC patients that was associated with decreased survival. Lower GPER expression also correlated with tumor progression and lymph node metastasis. Bioinformatic analyses of datasets (accession numbers: GSE2091 and GSE871) confirmed these results and showed statistically significant lower GPER mRNA levels in both colon and rectal adenoma samples compared to control samples. In vitro evidence further de-monstrated that decreased GPER expression in HTC-8 and SW-480 CRC cells correlated with higher promoter methylation of the GPER gene as compared to LS147T CRC cells that exhibit high GPER expression. Beyond promoter methylation, histone H3 acetylation may represent another mechanism regulating GPER expression in CRC cell lines and human tissues as down-regulation of GPER expression in HCT-8 and SW-480 cells was associated with decreased histone H3 acetylation. In contrast to the above results, based on Kaplan-Meier analyses of public data (accession number: GSE39582), high GPER expression was associated with poor relapse-free survival in women with stage 3/4 (but not stage 1/2) CRC, but not in men with disease of any stage[70]. Overall, these results suggest that GPER plays a complex role in colorectal carcinogenesis that is further complicated by sexual dimorphism, potentially as a result of estrogen-dependent signaling in CRC.
Estrogen signaling modulates cancer cell proliferation, invasion and migration, acting not only through the nuclear estrogen receptors α and β, but also through the GPER. GPER is without a doubt an important mediator of colorectal neoplastic trans-formation and progression. Clinical and experimental data are however, not unambiguous, which may result from the use of CRC cell lines with different GPER expression levels or GPER-mediated signaling pathways. On the other hand, recent studies have revealed the dual role of GPER in CRC development, potentially related to the modulation of both anti-tumorigenic and pro-tumorigenic effects, depending in part on oxygen levels in cancer cells. At the molecular level, GPER-mediated signaling in CRC cells regulates endoplasmic reticulum and mitochondrial functions, the metabolism of fatty acids and estrogens and the expression of genes directly involved in cell proliferation and survival (summarized in Table 2). In conclusion, GPER appears to be an important mediator of estrogenic actions in both neoplastic transformation of the colon and tumor progression, effects that need to be considered in the application and development of therapeutic strategies.
| Features of cancer cells | Process | Anti-tumorigenic | Pro-tumorigenic | Ref. |
| Proliferation and tumor growth | Apoptosis | Yes | No | [73] |
| Cell cycle | Yes | No | [73] | |
| DNA repair | No | Yes | [64] | |
| Endoplasmic reticulum stress | Yes | No | [73] | |
| Estrogen metabolism | No | Yes | [90,91] | |
| Mitochondrial membrane polarity | Yes | No | [73] | |
| Oxygen level | Yes under normoxia | Yes under hypoxia | [64] | |
| Migration | Fatty acid metabolism | No | Yes | [107] |
| Oxygen level | Yes under normoxia | Yes under hypoxia | [64] |
We thank Dr. Richard Pepermans for proofreading and editorial corrections.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: United States
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Caruso R, Soriano-Ursúa MA S-Editor: Yan JP L-Editor: A E-Editor: Zhang YL
| 1. | Haziman AA, Ravinderan S, Thangavelu T, Thomas W. A novel role for estrogen-induced signaling in the colorectal cancer gender bias. Ir J Med Sci. 2019;188:389-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 2. | Barzi A, Lenz AM, Labonte MJ, Lenz HJ. Molecular pathways: Estrogen pathway in colorectal cancer. Clin Cancer Res. 2013;19:5842-5848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 173] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 3. | Dahlman-Wright K, Cavailles V, Fuqua SA, Jordan VC, Katzenellenbogen JA, Korach KS, Maggi A, Muramatsu M, Parker MG, Gustafsson JA. International Union of Pharmacology. LXIV. Estrogen receptors. Pharmacol Rev. 2006;58:773-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 384] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 4. | Prossnitz ER, Arterburn JB. International Union of Basic and Clinical Pharmacology. XCVII. G Protein-Coupled Estrogen Receptor and Its Pharmacologic Modulators. Pharmacol Rev. 2015;67:505-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 208] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 5. | Carmeci C, Thompson DA, Ring HZ, Francke U, Weigel RJ. Identification of a gene (GPR30) with homology to the G-protein-coupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics. 1997;45:607-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 397] [Cited by in RCA: 444] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 6. | Feng Y, Gregor P. Cloning of a novel member of the G protein-coupled receptor family related to peptide receptors. Biochem Biophys Res Commun. 1997;231:651-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 85] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 7. | Kvingedal AM, Smeland EB. A novel putative G-protein-coupled receptor expressed in lung, heart and lymphoid tissue. FEBS Lett. 1997;407:59-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 80] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | O'Dowd BF, Nguyen T, Marchese A, Cheng R, Lynch KR, Heng HH, Kolakowski LF, George SR. Discovery of three novel G-protein-coupled receptor genes. Genomics. 1998;47:310-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 244] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 9. | Owman C, Blay P, Nilsson C, Lolait SJ. Cloning of human cDNA encoding a novel heptahelix receptor expressed in Burkitt's lymphoma and widely distributed in brain and peripheral tissues. Biochem Biophys Res Commun. 1996;228:285-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 171] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 10. | Takada Y, Kato C, Kondo S, Korenaga R, Ando J. Cloning of cDNAs encoding G protein-coupled receptor expressed in human endothelial cells exposed to fluid shear stress. Biochem Biophys Res Commun. 1997;240:737-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 168] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 11. | Barton M, Filardo EJ, Lolait SJ, Thomas P, Maggiolini M, Prossnitz ER. Twenty years of the G protein-coupled estrogen receptor GPER: Historical and personal perspectives. J Steroid Biochem Mol Biol. 2018;176:4-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 177] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 12. | Revankar CM, Cimino DF, Sklar LA, Arterburn JB, Prossnitz ER. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science. 2005;307:1625-1630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1634] [Cited by in RCA: 1767] [Article Influence: 88.4] [Reference Citation Analysis (0)] |
| 13. | Prossnitz ER, Hathaway HJ. What have we learned about GPER function in physiology and disease from knockout mice? J Steroid Biochem Mol Biol. 2015;153:114-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 120] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 14. | Meyer MR, Fredette NC, Daniel C, Sharma G, Amann K, Arterburn JB, Barton M, Prossnitz ER. Obligatory role for GPER in cardiovascular aging and disease. Sci Signal. 2016;9:ra105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 59] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 15. | Prossnitz ER, Barton M. Estrogen biology: New insights into GPER function and clinical opportunities. Mol Cell Endocrinol. 2014;389:71-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 310] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 16. | Sharma G, Mauvais-Jarvis F, Prossnitz ER. Roles of G protein-coupled estrogen receptor GPER in metabolic regulation. J Steroid Biochem Mol Biol. 2018;176:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 101] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 17. | Cho NL, Javid SH, Carothers AM, Redston M, Bertagnolli MM. Estrogen receptors alpha and beta are inhibitory modifiers of Apc-dependent tumorigenesis in the proximal colon of Min/+ mice. Cancer Res. 2007;67:2366-2372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Stevanato Filho PR, Aguiar Júnior S, Begnami MD, Ferreira FO, Nakagawa WT, Spencer RMSB, Bezerra TS, Boggiss PE, Lopes A. Estrogen Receptor β as a Prognostic Marker of Tumor Progression in Colorectal Cancer with Familial Adenomatous Polyposis and Sporadic Polyps. Pathol Oncol Res. 2018;24:533-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 19. | Guérin A, Mody R, Fok B, Lasch KL, Zhou Z, Wu EQ, Zhou W, Talley NJ. Risk of developing colorectal cancer and benign colorectal neoplasm in patients with chronic constipation. Aliment Pharmacol Ther. 2014;40:83-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 20. | Roberts MC, Millikan RC, Galanko JA, Martin C, Sandler RS. Constipation, laxative use, and colon cancer in a North Carolina population. Am J Gastroenterol. 2003;98:857-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 21. | Roager HM, Hansen LB, Bahl MI, Frandsen HL, Carvalho V, Gøbel RJ, Dalgaard MD, Plichta DR, Sparholt MH, Vestergaard H, Hansen T, Sicheritz-Pontén T, Nielsen HB, Pedersen O, Lauritzen L, Kristensen M, Gupta R, Licht TR. Colonic transit time is related to bacterial metabolism and mucosal turnover in the gut. Nat Microbiol. 2016;1:16093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 308] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 22. | Li Y, Xu J, Jiang F, Jiang Z, Liu C, Li L, Luo Y, Lu R, Mu Y, Liu Y, Xue B. G protein-coupled estrogen receptor is involved in modulating colonic motor function via nitric oxide release in C57BL/6 female mice. Neurogastroenterol Motil. 2016;28:432-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Zielińska M, Fichna J, Bashashati M, Habibi S, Sibaev A, Timmermans JP, Storr M. G protein-coupled estrogen receptor and estrogen receptor ligands regulate colonic motility and visceral pain. Neurogastroenterol Motil. 2017;29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 24. | Dennis MK, Burai R, Ramesh C, Petrie WK, Alcon SN, Nayak TK, Bologa CG, Leitao A, Brailoiu E, Deliu E, Dun NJ, Sklar LA, Hathaway HJ, Arterburn JB, Oprea TI, Prossnitz ER. In vivo effects of a GPR30 antagonist. Nat Chem Biol. 2009;5:421-427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 438] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 25. | Dennis MK, Field AS, Burai R, Ramesh C, Petrie WK, Bologa CG, Oprea TI, Yamaguchi Y, Hayashi S, Sklar LA, Hathaway HJ, Arterburn JB, Prossnitz ER. Identification of a GPER/GPR30 antagonist with improved estrogen receptor counterselectivity. J Steroid Biochem Mol Biol. 2011;127:358-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 242] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 26. | Bologa CG, Revankar CM, Young SM, Edwards BS, Arterburn JB, Kiselyov AS, Parker MA, Tkachenko SE, Savchuck NP, Sklar LA, Oprea TI, Prossnitz ER. Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat Chem Biol. 2006;2:207-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 605] [Cited by in RCA: 687] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 27. | Jacenik D, Cygankiewicz AI, Krajewska WM. Risk factors in colorectal cancer. In: Fichna J, editor. Introduction to gastrointestinal diseases Vol. 2. Springer International Publishing 2017; 113-128. |
| 28. | Qin B, Dong L, Guo X, Jiang J, He Y, Wang X, Li L, Zhao J. Expression of G protein-coupled estrogen receptor in irritable bowel syndrome and its clinical significance. Int J Clin Exp Pathol. 2014;7:2238-2246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 29. | Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, Stanghellini V, Corinaldesi R, Barbara G. Mucosal immune activation in irritable bowel syndrome: Gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 272] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 30. | Flores BM, O'Connor A, Moss AC. Impact of mucosal inflammation on risk of colorectal neoplasia in patients with ulcerative colitis: A systematic review and meta-analysis. Gastrointest Endosc. 2017;86:1006-1011.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 74] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 31. | Castaño-Milla C, Chaparro M, Gisbert JP. Systematic review with meta-analysis: The declining risk of colorectal cancer in ulcerative colitis. Aliment Pharmacol Ther. 2014;39:645-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 198] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 32. | von Roon AC, Reese G, Teare J, Constantinides V, Darzi AW, Tekkis PP. The risk of cancer in patients with Crohn's disease. Dis Colon Rectum. 2007;50:839-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 211] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 33. | Armstrong CM, Allred KF, Weeks BR, Chapkin RS, Allred CD. Estradiol Has Differential Effects on Acute Colonic Inflammation in the Presence and Absence of Estrogen Receptor β Expression. Dig Dis Sci. 2017;62:1977-1984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 34. | Bábíčková J, Tóthová Ľ, Lengyelová E, Bartoňová A, Hodosy J, Gardlík R, Celec P. Sex Differences in Experimentally Induced Colitis in Mice: A Role for Estrogens. Inflammation. 2015;38:1996-2006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 121] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 35. | Linares PM, Algaba A, Urzainqui A, Guijarro-Rojas M, González-Tajuelo R, Garrido J, Chaparro M, Gisbert JP, Bermejo F, Guerra I, Castellano V, Fernández-Contreras ME. Ratio of Circulating Estrogen Receptors Beta and Alpha (ERβ/ERα) Indicates Endoscopic Activity in Patients with Crohn's Disease. Dig Dis Sci. 2017;62:2744-2754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 36. | Pierdominici M, Maselli A, Varano B, Barbati C, Cesaro P, Spada C, Zullo A, Lorenzetti R, Rosati M, Rainaldi G, Limiti MR, Guidi L, Conti L, Gessani S. Linking estrogen receptor β expression with inflammatory bowel disease activity. Oncotarget. 2015;6:40443-40451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 37. | Włodarczyk M, Sobolewska-Włodarczyk A, Cygankiewicz AI, Jacenik D, Piechota-Polańczyk A, Stec-Michalska K, Krajewska WM, Fichna J, Wiśniewska-Jarosińska M. G Protein-Coupled Receptor 30 (GPR30) Expression Pattern in Inflammatory Bowel Disease Patients Suggests its Key Role in the Inflammatory Process. A Preliminary Study. J Gastrointestin Liver Dis. 2017;26:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 38. | Blasko E, Haskell CA, Leung S, Gualtieri G, Halks-Miller M, Mahmoudi M, Dennis MK, Prossnitz ER, Karpus WJ, Horuk R. Beneficial role of the GPR30 agonist G-1 in an animal model of multiple sclerosis. J Neuroimmunol. 2009;214:67-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 156] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 39. | Brunsing RL, Prossnitz ER. Induction of interleukin-10 in the T helper type 17 effector population by the G protein coupled estrogen receptor (GPER) agonist G-1. Immunology. 2011;134:93-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 40. | Cabas I, Rodenas MC, Abellán E, Meseguer J, Mulero V, García-Ayala A. Estrogen signaling through the G protein-coupled estrogen receptor regulates granulocyte activation in fish. J Immunol. 2013;191:4628-4639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 41. | Cortes E, Sarper M, Robinson B, Lachowski D, Chronopoulos A, Thorpe SD, Lee DA, Del Río Hernández AE. GPER is a mechanoregulator of pancreatic stellate cells and the tumor microenvironment. EMBO Rep. 2019;20:pii: e46556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 63] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 42. | Jacenik D, Cygankiewicz AI, Krajewska WM. The G protein-coupled estrogen receptor as a modulator of neoplastic transformation. Mol Cell Endocrinol. 2016;429:10-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 43. | Rettew JA, McCall SH 4th, Marriott I. GPR30/GPER-1 mediates rapid decreases in TLR4 expression on murine macrophages. Mol Cell Endocrinol. 2010;328:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 44. | Wang C, Dehghani B, Magrisso IJ, Rick EA, Bonhomme E, Cody DB, Elenich LA, Subramanian S, Murphy SJ, Kelly MJ, Rosenbaum JS, Vandenbark AA, Offner H. GPR30 contributes to estrogen-induced thymic atrophy. Mol Endocrinol. 2008;22:636-648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 163] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 45. | Albanito L, Lappano R, Madeo A, Chimento A, Prossnitz ER, Cappello AR, Dolce V, Abonante S, Pezzi V, Maggiolini M. Effects of atrazine on estrogen receptor α- and G protein-coupled receptor 30-mediated signaling and proliferation in cancer cells and cancer-associated fibroblasts. Environ Health Perspect. 2015;123:493-499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 62] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 46. | Madeo A, Maggiolini M. Nuclear alternate estrogen receptor GPR30 mediates 17beta-estradiol-induced gene expression and migration in breast cancer-associated fibroblasts. Cancer Res. 2010;70:6036-6046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 141] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 47. | Pisano A, Santolla MF, De Francesco EM, De Marco P, Rigiracciolo DC, Perri MG, Vivacqua A, Abonante S, Cappello AR, Dolce V, Belfiore A, Maggiolini M, Lappano R. GPER, IGF-IR, and EGFR transduction signaling are involved in stimulatory effects of zinc in breast cancer cells and cancer-associated fibroblasts. Mol Carcinog. 2017;56:580-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 48. | Pupo M, Pisano A, Lappano R, Santolla MF, De Francesco EM, Abonante S, Rosano C, Maggiolini M. Bisphenol A induces gene expression changes and proliferative effects through GPER in breast cancer cells and cancer-associated fibroblasts. Environ Health Perspect. 2012;120:1177-1182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 220] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 49. | Ubink I, Verhaar ER, Kranenburg O, Goldschmeding R. A potential role for CCN2/CTGF in aggressive colorectal cancer. J Cell Commun Signal. 2016;10:223-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 50. | De Marco P, Lappano R, De Francesco EM, Cirillo F, Pupo M, Avino S, Vivacqua A, Abonante S, Picard D, Maggiolini M. GPER signalling in both cancer-associated fibroblasts and breast cancer cells mediates a feedforward IL1β/IL1R1 response. Sci Rep. 2016;6:24354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 51. | Okamoto M, Suzuki T, Mizukami Y, Ikeda T. The membrane-type estrogen receptor G-protein-coupled estrogen receptor suppresses lipopolysaccharide-induced interleukin 6 via inhibition of nuclear factor-kappa B pathway in murine macrophage cells. Anim Sci J. 2017;88:1870-1879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Platt AM, Bain CC, Bordon Y, Sester DP, Mowat AM. An independent subset of TLR expressing CCR2-dependent macrophages promotes colonic inflammation. J Immunol. 2010;184:6843-6854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 172] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 53. | Zigmond E, Varol C, Farache J, Elmaliah E, Satpathy AT, Friedlander G, Mack M, Shpigel N, Boneca IG, Murphy KM, Shakhar G, Halpern Z, Jung S. Ly6C hi monocytes in the inflamed colon give rise to proinflammatory effector cells and migratory antigen-presenting cells. Immunity. 2012;37:1076-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 584] [Article Influence: 44.9] [Reference Citation Analysis (0)] |
| 54. | Cui YL, Li HK, Zhou HY, Zhang T, Li Q. Correlations of tumor-associated macrophage subtypes with liver metastases of colorectal cancer. Asian Pac J Cancer Prev. 2013;14:1003-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 55. | Li S, Wang B, Tang Q, Liu J, Yang X. Bisphenol A triggers proliferation and migration of laryngeal squamous cell carcinoma via GPER mediated upregulation of IL-6. Cell Biochem Funct. 2017;35:209-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 56. | Kurosawa T, Hiroi H, Tsutsumi O, Ishikawa T, Osuga Y, Fujiwara T, Inoue S, Muramatsu M, Momoeda M, Taketani Y. The activity of bisphenol A depends on both the estrogen receptor subtype and the cell type. Endocr J. 2002;49:465-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 104] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 57. | Liang S, Chen Z, Jiang G, Zhou Y, Liu Q, Su Q, Wei W, Du J, Wang H. Activation of GPER suppresses migration and angiogenesis of triple negative breast cancer via inhibition of NF-κB/IL-6 signals. Cancer Lett. 2017;386:12-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 110] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 58. | Okamoto M, Mizukami Y. GPER negatively regulates TNFα-induced IL-6 production in human breast cancer cells via NF-κB pathway. Endocr J. 2016;63:485-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 59. | Zha Z, Su A, Huo S. Activation of GPER suppresses the malignancy of osteosarcoma cells via down regulation of IL-6 and IL-8. Arch Biochem Biophys. 2018;660:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 60. | Shen M, Hu P, Donskov F, Wang G, Liu Q, Du J. Tumor-associated neutrophils as a new prognostic factor in cancer: A systematic review and meta-analysis. PLoS One. 2014;9:e98259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 277] [Cited by in RCA: 259] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 61. | Lam HM, Ouyang B, Chen J, Ying J, Wang J, Wu CL, Jia L, Medvedovic M, Vessella RL, Ho SM. Targeting GPR30 with G-1: A new therapeutic target for castration-resistant prostate cancer. Endocr Relat Cancer. 2014;21:903-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 62. | Itoga M, Konno Y, Moritoki Y, Saito Y, Ito W, Tamaki M, Kobayashi Y, Kayaba H, Kikuchi Y, Chihara J, Takeda M, Ueki S, Hirokawa M. G-protein-coupled estrogen receptor agonist suppresses airway inflammation in a mouse model of asthma through IL-10. PLoS One. 2015;10:e0123210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 63. | Smids C, Horjus Talabur Horje CS, Drylewicz J, Roosenboom B, Groenen MJM, van Koolwijk E, van Lochem EG, Wahab PJ. Intestinal T Cell Profiling in Inflammatory Bowel Disease: Linking T Cell Subsets to Disease Activity and Disease Course. J Crohns Colitis. 2018;12:465-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 99] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 64. | Chirica M, Le Bourhis L, Lehmann-Che J, Chardiny V, Bouhidel F, Foulboeuf L, Gornet JM, Lourenco N, Dulphy N, Toubert A, Allez M. Phenotypic analysis of T cells infiltrating colon cancers: Correlations with oncogenetic status. Oncoimmunology. 2015;4:e1016698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 65. | Zekas E, Prossnitz ER. Estrogen-mediated inactivation of FOXO3a by the G protein-coupled estrogen receptor GPER. BMC Cancer. 2015;15:702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 66. | Filardo EJ. A role for G-protein coupled estrogen receptor (GPER) in estrogen-induced carcinogenesis: Dysregulated glandular homeostasis, survival and metastasis. J Steroid Biochem Mol Biol. 2018;176:38-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 67. | Smith HO, Arias-Pulido H, Kuo DY, Howard T, Qualls CR, Lee SJ, Verschraegen CF, Hathaway HJ, Joste NE, Prossnitz ER. GPR30 predicts poor survival for ovarian cancer. Gynecol Oncol. 2009;114:465-471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 176] [Cited by in RCA: 178] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 68. | Smith HO, Leslie KK, Singh M, Qualls CR, Revankar CM, Joste NE, Prossnitz ER. GPR30: A novel indicator of poor survival for endometrial carcinoma. Am J Obstet Gynecol. 2007;196:386.e1-9; discussion 386.e9-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 170] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 69. | Petrie WK, Dennis MK, Hu C, Dai D, Arterburn JB, Smith HO, Hathaway HJ, Prossnitz ER. G protein-coupled estrogen receptor-selective ligands modulate endometrial tumor growth. Obstet Gynecol Int. 2013;2013:472720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 70. | Bustos V, Nolan ÁM, Nijhuis A, Harvey H, Parker A, Poulsom R, McBryan J, Thomas W, Silver A, Harvey BJ. GPER mediates differential effects of estrogen on colon cancer cell proliferation and migration under normoxic and hypoxic conditions. Oncotarget. 2017;8:84258-84275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 71. | Hao J, Bao X, Jin B, Wang X, Mao Z, Li X, Wei L, Shen D, Wang JL. Ca2+ channel subunit α 1D promotes proliferation and migration of endometrial cancer cells mediated by 17β-estradiol via the G protein-coupled estrogen receptor. FASEB J. 2015;29:2883-2893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 72. | Huff MO, Todd SL, Smith AL, Elpers JT, Smith AP, Murphy RD, Bleser-Shartzer AS, Hoerter JE, Radde BN, Klinge CM. Arsenite and Cadmium Activate MAPK/ERK via Membrane Estrogen Receptors and G-Protein Coupled Estrogen Receptor Signaling in Human Lung Adenocarcinoma Cells. Toxicol Sci. 2016;152:62-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 73. | Liu C, Liao Y, Fan S, Fu X, Xiong J, Zhou S, Zou M, Wang J. G-Protein-Coupled Estrogen Receptor Antagonist G15 Decreases Estrogen-Induced Development of Non-Small Cell Lung Cancer. Oncol Res. 2019;27:283-292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 74. | Liu Q, Chen Z, Jiang G, Zhou Y, Yang X, Huang H, Liu H, Du J, Wang H. Epigenetic down regulation of G protein-coupled estrogen receptor (GPER) functions as a tumor suppressor in colorectal cancer. Mol Cancer. 2017;16:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 75. | Santolla MF, Avino S, Pellegrino M, De Francesco EM, De Marco P, Lappano R, Vivacqua A, Cirillo F, Rigiracciolo DC, Scarpelli A, Abonante S, Maggiolini M. SIRT1 is involved in oncogenic signaling mediated by GPER in breast cancer. Cell Death Dis. 2015;6:e1834. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 79] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 76. | Sathya S, Sudhagar S, Lakshmi BS. Estrogen suppresses breast cancer proliferation through GPER / p38 MAPK axis during hypoxia. Mol Cell Endocrinol. 2015;417:200-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 77. | Yan Y, Jiang X, Zhao Y, Wen H, Liu G. Role of GPER on proliferation, migration and invasion in ligand-independent manner in human ovarian cancer cell line SKOV3. Cell Biochem Funct. 2015;33:552-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 78. | Sano R, Reed JC. ER stress-induced cell death mechanisms. Biochim Biophys Acta. 2013;1833:3460-3470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1214] [Cited by in RCA: 1537] [Article Influence: 128.1] [Reference Citation Analysis (1)] |
| 79. | Bassères DS, Ebbs A, Levantini E, Baldwin AS. Requirement of the NF-kappaB subunit p65/RelA for K-Ras-induced lung tumorigenesis. Cancer Res. 2010;70:3537-3546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 161] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 80. | Meylan E, Dooley AL, Feldser DM, Shen L, Turk E, Ouyang C, Jacks T. Requirement for NF-kappaB signalling in a mouse model of lung adenocarcinoma. Nature. 2009;462:104-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 458] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 81. | Xia Y, Yeddula N, Leblanc M, Ke E, Zhang Y, Oldfield E, Shaw RJ, Verma IM. Reduced cell proliferation by IKK2 depletion in a mouse lung-cancer model. Nat Cell Biol. 2012;14:257-265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 115] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 82. | Huang S, Pettaway CA, Uehara H, Bucana CD, Fidler IJ. Blockade of NF-kappaB activity in human prostate cancer cells is associated with suppression of angiogenesis, invasion, and metastasis. Oncogene. 2001;20:4188-4197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 441] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 83. | Kawauchi K, Araki K, Tobiume K, Tanaka N. p53 regulates glucose metabolism through an IKK-NF-kappaB pathway and inhibits cell transformation. Nat Cell Biol. 2008;10:611-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 520] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 84. | Mauro C, Leow SC, Anso E, Rocha S, Thotakura AK, Tornatore L, Moretti M, De Smaele E, Beg AA, Tergaonkar V, Chandel NS, Franzoso G. NF-κB controls energy homeostasis and metabolic adaptation by upregulating mitochondrial respiration. Nat Cell Biol. 2011;13:1272-1279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 317] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 85. | Iliopoulos D, Hirsch HA, Struhl K. An epigenetic switch involving NF-kappaB, Lin28, Let-7 MicroRNA, and IL6 links inflammation to cell transformation. Cell. 2009;139:693-706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1097] [Cited by in RCA: 1168] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 86. | Ribeiro-Varandas E, Viegas W, Sofia Pereira H, Delgado M. Bisphenol A at concentrations found in human serum induces aneugenic effects in endothelial cells. Mutat Res. 2013;751:27-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 87. | Branzei D, Foiani M. Regulation of DNA repair throughout the cell cycle. Nat Rev Mol Cell Biol. 2008;9:297-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 850] [Cited by in RCA: 911] [Article Influence: 53.6] [Reference Citation Analysis (0)] |
| 88. | Sun M, Guo X, Qian X, Wang H, Yang C, Brinkman KL, Serrano-Gonzalez M, Jope RS, Zhou B, Engler DA, Zhan M, Wong ST, Fu L, Xu B. Activation of the ATM-Snail pathway promotes breast cancer metastasis. J Mol Cell Biol. 2012;4:304-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 89. | Bai AH, Tong JH, To KF, Chan MW, Man EP, Lo KW, Lee JF, Sung JJ, Leung WK. Promoter hypermethylation of tumor-related genes in the progression of colorectal neoplasia. Int J Cancer. 2004;112:846-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 90. | Muz B, de la Puente P, Azab F, Azab AK. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia (Auckl). 2015;3:83-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 842] [Cited by in RCA: 1399] [Article Influence: 139.9] [Reference Citation Analysis (1)] |
| 91. | Gilligan LC, Rahman HP, Hewitt AM, Sitch AJ, Gondal A, Arvaniti A, Taylor AE, Read ML, Morton DG, Foster PA. Estrogen Activation by Steroid Sulfatase Increases Colorectal Cancer Proliferation via GPER. J Clin Endocrinol Metab. 2017;102:4435-4447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 92. | Gilligan LC, Gondal A, Tang V, Hussain MT, Arvaniti A, Hewitt AM, Foster PA. Estrone Sulfate Transport and Steroid Sulfatase Activity in Colorectal Cancer: Implications for Hormone Replacement Therapy. Front Pharmacol. 2017;8:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 93. | Castillo Sanchez R, Gomez R, Perez Salazar E. Bisphenol A Induces Migration through a GPER-, FAK-, Src-, and ERK2-Dependent Pathway in MDA-MB-231 Breast Cancer Cells. Chem Res Toxicol. 2016;29:285-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 94. | Chen ZJ, Wei W, Jiang GM, Liu H, Wei WD, Yang X, Wu YM, Liu H, Wong CK, Du J, Wang HS. Activation of GPER suppresses epithelial mesenchymal transition of triple negative breast cancer cells via NF-κB signals. Mol Oncol. 2016;10:775-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 95. | Deng Q, Jiang G, Wu Y, Li J, Liang W, Chen L, Su Q, Li W, Du J, Wong CKC, Chen Z, Wang H. GPER/Hippo-YAP signal is involved in Bisphenol S induced migration of triple negative breast cancer (TNBC) cells. J Hazard Mater. 2018;355:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 96. | François CM, Wargnier R, Petit F, Goulvent T, Rimokh R, Treilleux I, Ray-Coquard I, Zazzu V, Cohen-Tannoudji J, Guigon CJ. 17β-estradiol inhibits spreading of metastatic cells from granulosa cell tumors through a non-genomic mechanism involving GPER1. Carcinogenesis. 2015;36:564-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 97. | Guan BZ, Yan RL, Huang JW, Li FL, Zhong YX, Chen Y, Liu FN, Hu B, Huang SB, Yin LH. Activation of G protein coupled estrogen receptor (GPER) promotes the migration of renal cell carcinoma via the PI3K/AKT/MMP-9 signals. Cell Adh Migr. 2018;12:109-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 98. | Henic E, Noskova V, Høyer-Hansen G, Hansson S, Casslén B. Estradiol attenuates EGF-induced rapid uPAR mobilization and cell migration via the G-protein-coupled receptor 30 in ovarian cancer cells. Int J Gynecol Cancer. 2009;19:214-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 99. | Jiang QF, Wu TT, Yang JY, Dong CR, Wang N, Liu XH, Liu ZM. 17β-estradiol promotes the invasion and migration of nuclear estrogen receptor-negative breast cancer cells through cross-talk between GPER1 and CXCR1. J Steroid Biochem Mol Biol. 2013;138:314-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 100. | Li Y, Chen Y, Zhu ZX, Liu XH, Yang L, Wan L, Lei TW, Wang XD. 4-Hydroxytamoxifen-stimulated processing of cyclin E is mediated via G protein-coupled receptor 30 (GPR30) and accompanied by enhanced migration in MCF-7 breast cancer cells. Toxicology. 2013;309:61-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 101. | Shang D, Li Z, Zhu Z, Chen H, Zhao L, Wang X, Chen Y. Baicalein suppresses 17-β-estradiol-induced migration, adhesion and invasion of breast cancer cells via the G protein-coupled receptor 30 signaling pathway. Oncol Rep. 2015;33:2077-2085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 102. | Yan Y, Liu H, Wen H, Jiang X, Cao X, Zhang G, Liu G. The novel estrogen receptor GPER regulates the migration and invasion of ovarian cancer cells. Mol Cell Biochem. 2013;378:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 103. | Zhang KS, Chen HQ, Chen YS, Qiu KF, Zheng XB, Li GC, Yang HD, Wen CJ. Bisphenol A stimulates human lung cancer cell migration via upregulation of matrix metalloproteinases by GPER/EGFR/ERK1/2 signal pathway. Biomed Pharmacother. 2014;68:1037-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 104] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 104. | Zhu G, Huang Y, Wu C, Wei D, Shi Y. Activation of G-Protein-Coupled Estrogen Receptor Inhibits the Migration of Human Nonsmall Cell Lung Cancer Cells via IKK-β/NF-κB Signals. DNA Cell Biol. 2016;35:434-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 105. | Zhu P, Liao LY, Zhao TT, Mo XM, Chen GG, Liu ZM. GPER/ERK&AKT/NF-κB pathway is involved in cadmium-induced proliferation, invasion and migration of GPER-positive thyroid cancer cells. Mol Cell Endocrinol. 2017;442:68-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 51] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 106. | Che L, Pilo MG, Cigliano A, Latte G, Simile MM, Ribback S, Dombrowski F, Evert M, Chen X, Calvisi DF. Oncogene dependent requirement of fatty acid synthase in hepatocellular carcinoma. Cell Cycle. 2017;16:499-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 107. | Corominas-Faja B, Vellon L, Cuyàs E, Buxó M, Martin-Castillo B, Serra D, García J, Lupu R, Menendez JA. Clinical and therapeutic relevance of the metabolic oncogene fatty acid synthase in HER2+ breast cancer. Histol Histopathol. 2017;32:687-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 108. | Santolla MF, Lappano R, De Marco P, Pupo M, Vivacqua A, Sisci D, Abonante S, Iacopetta D, Cappello AR, Dolce V, Maggiolini M. G protein-coupled estrogen receptor mediates the up-regulation of fatty acid synthase induced by 17β-estradiol in cancer cells and cancer-associated fibroblasts. J Biol Chem. 2012;287:43234-43245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 109. | Van de Sande T, Roskams T, Lerut E, Joniau S, Van Poppel H, Verhoeven G, Swinnen JV. High-level expression of fatty acid synthase in human prostate cancer tissues is linked to activation and nuclear localization of Akt/PKB. J Pathol. 2005;206:214-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 101] [Article Influence: 5.1] [Reference Citation Analysis (0)] |