Published online Jul 14, 2019. doi: 10.3748/wjg.v25.i26.3380
Peer-review started: April 12, 2019
First decision: May 9, 2019
Revised: May 18, 2019
Accepted: May 31, 2019
Article in press: June 1, 2019
Published online: July 14, 2019
Processing time: 93 Days and 7.4 Hours
Bile duct cancer is characterized by fast metastasis and invasion and has been regarded as one of the most aggressive tumors due to the absence of effective diagnosis at an early stage. Therefore, it is in the urgent demand to explore novel diagnostic approaches and therapeutic strategies for bile duct cancer to improve patient survival. Raddeanin A (RA) is extracted from the anemone raddeana regel and has been demonstrated to play antitumor roles in various cancers.
To investigate the effects of RA treatment on bile duct cancer cells.
In this study, four cholangiocarcinoma cell lines (RBE, LIPF155C, LIPF178C, and LICCF) treated with RA were used to test the cell viability. The RA-associated cell functional analysis, 5-fluorouracil (5-Fu) effectiveness as well as cell cycle- and apoptosis-related protein expression were investigated.
RA reduced cell viability in a dose-dependent pattern in four cell lines, and the migration and colony formation abilities were also impaired by RA in RBE and LIPF155C cell lines. RA sensitized cell lines to 5-Fu treatment and enhanced the effects of 5-Fu in cholangiocarcinoma. Also, RA decreased protein expression of Wee1, while the combinational effect of RA and 5-Fu decreased protein expressions of cyclooxygenase-2, B cell lymphoma 2, and Wee1 but increased protein levels of Bax, cyclin D1, and cyclin E.
Taken together, the results suggest that RA acts as an anti-cancer agent and enhancer of 5-Fu in bile duct cancer cells via regulating multiple cell cycle and apoptosis-related proteins. This finding provides novel clues to exploring a novel antitumor drug for bile duct cancer.
Core tip: We report that raddeanin A (RA) reduced cell viability, migration, and colony formation abilities in cholangiocarcinoma cells and enhanced the antitumor effect of 5-Fu in cholangiocarcinoma cells. This study also demonstrated that the anticancer effects of RA were associated with apoptosis-related proteins Bax and B cell lymphoma 2 as well as cell cycle-related proteins Wee1, cyclin D1, cyclin E, and cyclooxygenase-2.
- Citation: Guo SS, Wang Y, Fan QX. Raddeanin A promotes apoptosis and ameliorates 5-fluorouracil resistance in cholangiocarcinoma cells. World J Gastroenterol 2019; 25(26): 3380-3391
- URL: https://www.wjgnet.com/1007-9327/full/v25/i26/3380.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i26.3380
Bile duct cancer (cholangiocarcinoma) is characterized by fast metastasis and invasion and, more importantly, the absence of effective diagnosis at an early stage[1], and it thus has been regarded as one of the most aggressive tumors that occur in bile duct epithelial cells[2]. To date, although surgical resection is one of the few curative treatments for bile duct cancer, the patients are often diagnosed at the advanced stage and have lost the chance of surgical resection at diagnosis[3]. In addition, the 5-year survival rate and effective therapeutic level for bile duct cancer have not greatly improved in recent years[3]. Therefore, it is in the urgent need to explore novel diagnostic approaches and therapeutic strategies for bile duct cancer to improve patient survival.
Raddeanin A (RA), an active triterpenoid saponin, is extracted from the anemone raddeana regel that is a traditional medicinal herb in Chinese medicine[4]. An increasing amount of evidence has demonstrated that RA plays a cytotoxic role in facilitating apoptosis of tumor cells and suppressing their proliferation, migration, and invasion[5], thus exhibiting anticancer effects on various cancer types. In gastric cancer, RA results in tumor cell apoptosis via regulating the molecules involved in the caspase-cascade pathway, such as B cell lymphoma 2 (Bcl-2) family[4]. Also, RA has been demonstrated to cause apoptosis in a concentration-dependent pattern in colorectal cancer cells via mechanisms associated with suppressing NF-κB and activated Wnt/β-catenin signaling in colorectal cancer cells[6]. Furthermore, the PI3K/AKT/mTOR pathway is involved in the RA-induced apoptosis in breast cancer cells[7]. Collectively, RA-associated apoptosis in various types of cancer cells display a cancer type-dependent pattern, suggesting the necessity of proapoptotic effect of RA in cholangiocarcinoma cells.
Malignant tumor formation is a highly complicated process, involving a series of carcinogens, tumor promotion, apoptosis, and metastatic process[8]. Cyclooxygenase-2 (Cox-2), a cyclooxygenase enzyme, catalyzes the first step of prostanoids synthesis[9]. Cox-2 is highly expressed in cancer and stroma cells during tumor progression and displays anti-apoptosis roles in tumor cells[10]. In addition, a group of factors that are essential for the regulation of the cell cycle have been demonstrated to be closely related to malignancies. For example, aberrant overexpression of cyclins D1 and E, two members of the cyclin family[11], contributes to carcinogenesis and malignant progression due to the dysfunction of the cell cycle[12,13]. Wee1 is essential for G2 cell cycle checkpoint in response to DNA damage and repair[14] and overexpressed Wee1 has been linked to a number of cancers, such as carcinoma[15] and glioblastoma[16]. Furthermore, apoptosis has been shown to be closely related to a series of cancers[17], in which the mitochondrial-related proteins bcl-2 and Bax are highly involved in the regulation of mitochondrial membrane properties, leading to activation of subsequent pathways of apoptosis[18,19].
Currently, 5-fluorouracil (5-Fu) is one of the most common chemotherapeutic compounds for cancer treatment[20]. The resistance to chemotherapy is a primary challenge for cancer treatment[21]. Thus, any approach or compound that can sensitize 5-Fu-resistant cells would be beneficial to cancer patients. Given its potential anti-cancer effect, the objective of this study was to investigate the effects of RA treatment on bile duct cancer cells including related chemotherapy resistance as well as the underlying mechanisms. This study also aimed to provide clues to the application of RA for the therapy of bile duct cancer.
Cholangiocarcinoma cell lines (RBE, LIPF155C, LIPF178C, and LICCF) were purchased from the cell back of China Center for Type Culture Collection (CCTCC at Wuhan University). The normal intrahepatic biliary epithelial cell line HIBEpiC was obtained from the ATCC (Manassas, VA, United States).
The ATPlite assay (Perkin Elmer, United States) was applied to evaluate cell viability according to the manufacturer’s instructions (n = 3 independent experiments). Cells were seeded in 96-well plates (Corning-Costar, United States) and cultured for 24 h. RA at doses ranging from 0 to 160 μg/mL was applied to treat the cells for 24 h. Cell viability was evaluated using a microplate reader (BioRad, United States).
Wound-healing migration and Transwell invasion assays were used to test migratory cell ability in RBE and LIPF155C cell lines (n = 3 independent experiments). The assays were performed as previously reported[22].
The clonogenic assay was performed to test colony formation ability in RBE and LIPF155C cell lines (n = 3 independent experiments). RBE and LIPF155C cells (70% confluence) were treated transiently with metformin (0.5 mM). Cells were then detached and seeded in 6-well plates (600 cells/well) (Corning-Costar, United States) in drug-free media. Fresh media (25%) were added every three days. Cell colony was stained with cristal violet and the number of colonies was counted ten days later.
Hoechst staining assay was performed to test cell apoptosis (n = 3 independent experiments). The assay was performed as previously reported[23]. Hoechst (Sigma, United States) stained cells were visualized by fluorescence microscopy (Olympus, Japan).
Cell protein was isolated by using the cell lysis buffer (Beyotime Institute of Bio-technology, China). Western blot analysis was performed as previously reported[22]. The primary antibodies for Cox-2, Bcl-2, Bax, GAPDH, cyclin D1, cyclin E, and Wee1 were obtained from Santa Cruz Biotechnology (United States). Quantification of optical density was evaluated using Uvitec Alliance software (Eppendorf, Germany) (n = 3 independent experiments).
Data are expressed as the mean ± standard error of the mean and analyzed via SPSS 19.0 (SPSS Inc, United States). The two-group comparison was performed by two-tailed t-tests and multiple comparisons were analyzed by ANOVA and the post-hoc Tukey test. In this study, statistical significance was set at P < 0.05.
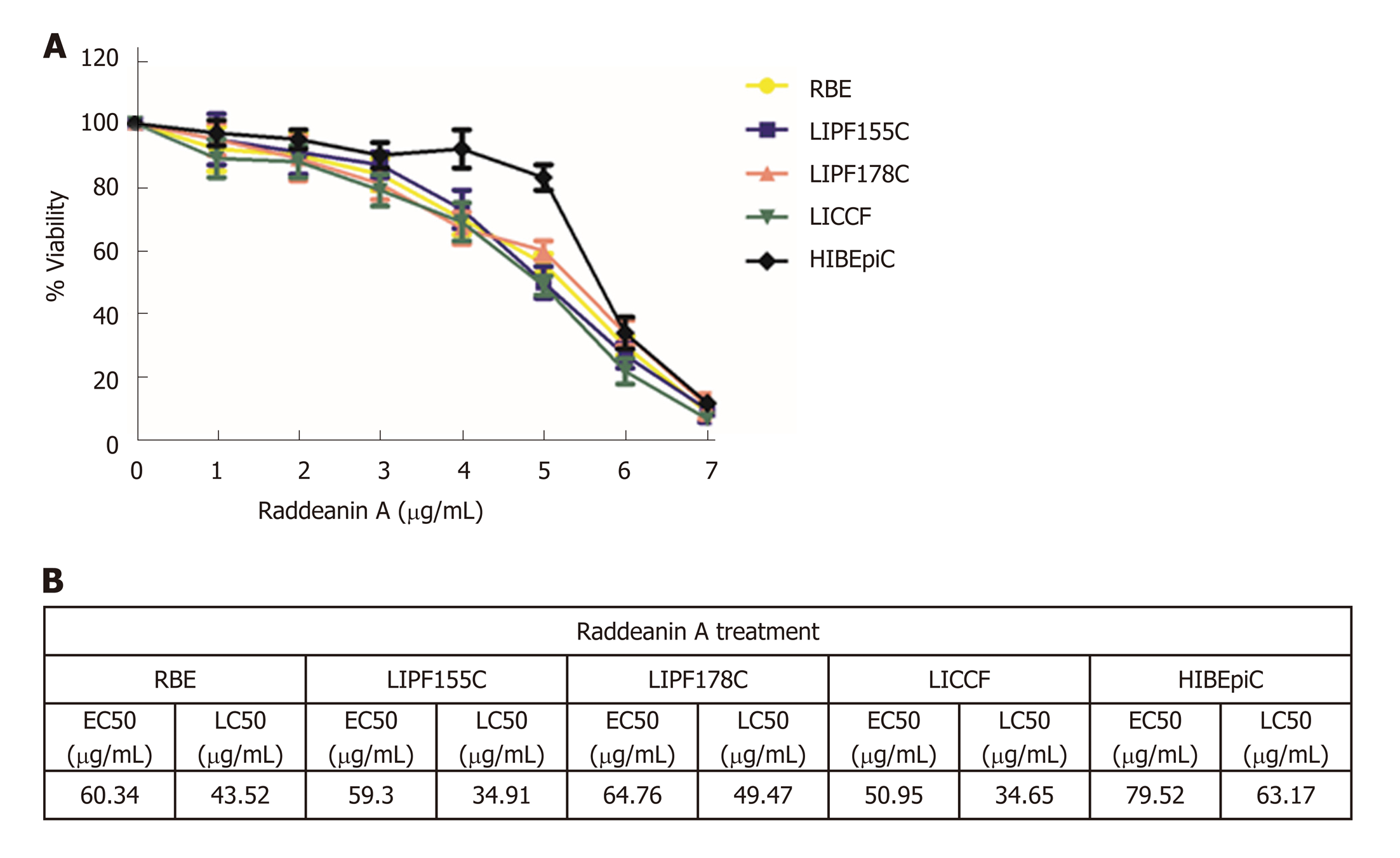
RA at doses ranging from 0-160 μg/mL was applied for 24 h to test the cell viability of four cholangiocarcinoma cell lines (RBE, LIPF155C, LIPF178C, and LICCF) and one normal intrahepatic biliary epithelial cell line (HIBEpiC) via the ATPlite assay. RA was found to reduce cell viability in a dose-dependent pattern (Figure 1A). The half-maximal effective concentration (EC50) and the half-maximal lethal concentration (LC50) for each line were also evaluated (Figure 1B). EC50 and LC50 ranges in the four tumoral cell lines were 50.95-64.76 μg/mL and 34.65-49.47 μg/mL, respectively, while HIBEpiC showed higher values of EC50 (79.52 μg/mL) and LC50 (63.17 μg/mL) compared with those of tumoral cell lines, indicating that the RA does that reduced cell viability of tumoral cell lines were not toxic for the normal cell line.
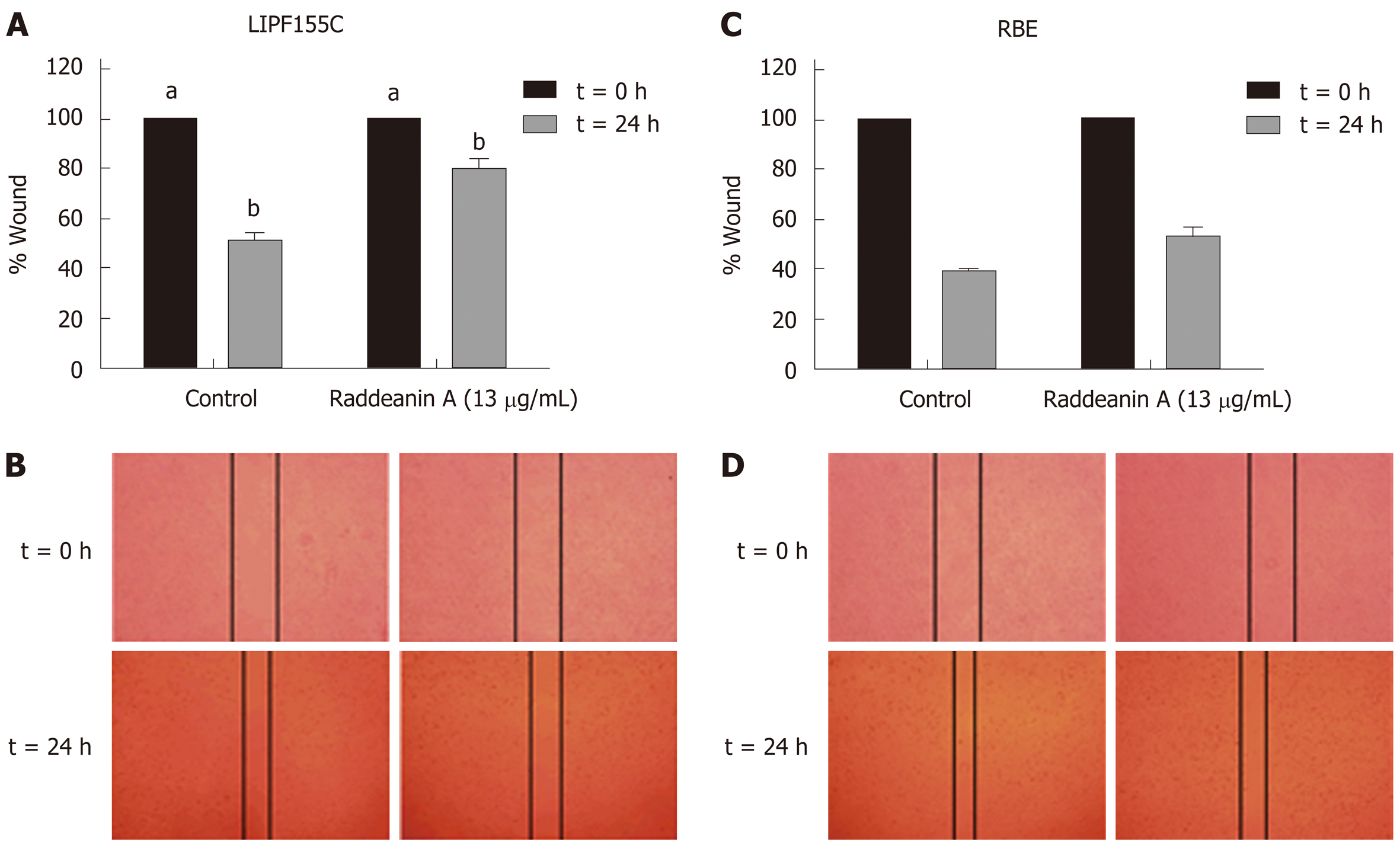
Based on the results from the cell viability assay, RA at a dose of 13 μg/mL that reduced about 25% cell viability was chosen to use in subsequent experiments. The wound healing (Figure 2A-D) and Transwell migration (Figure 3A) assays were conducted to test the migration ability of the two cholangiocarcinoma cell lines RBE and LIPF155C. The results revealed that the migration ability of both lines was impaired by 13 μg/mL RA. In addition, clonogenic assay results indicated that RA reduced colony formation capability of the two cholangiocarcinoma cell lines (Figure 3B).
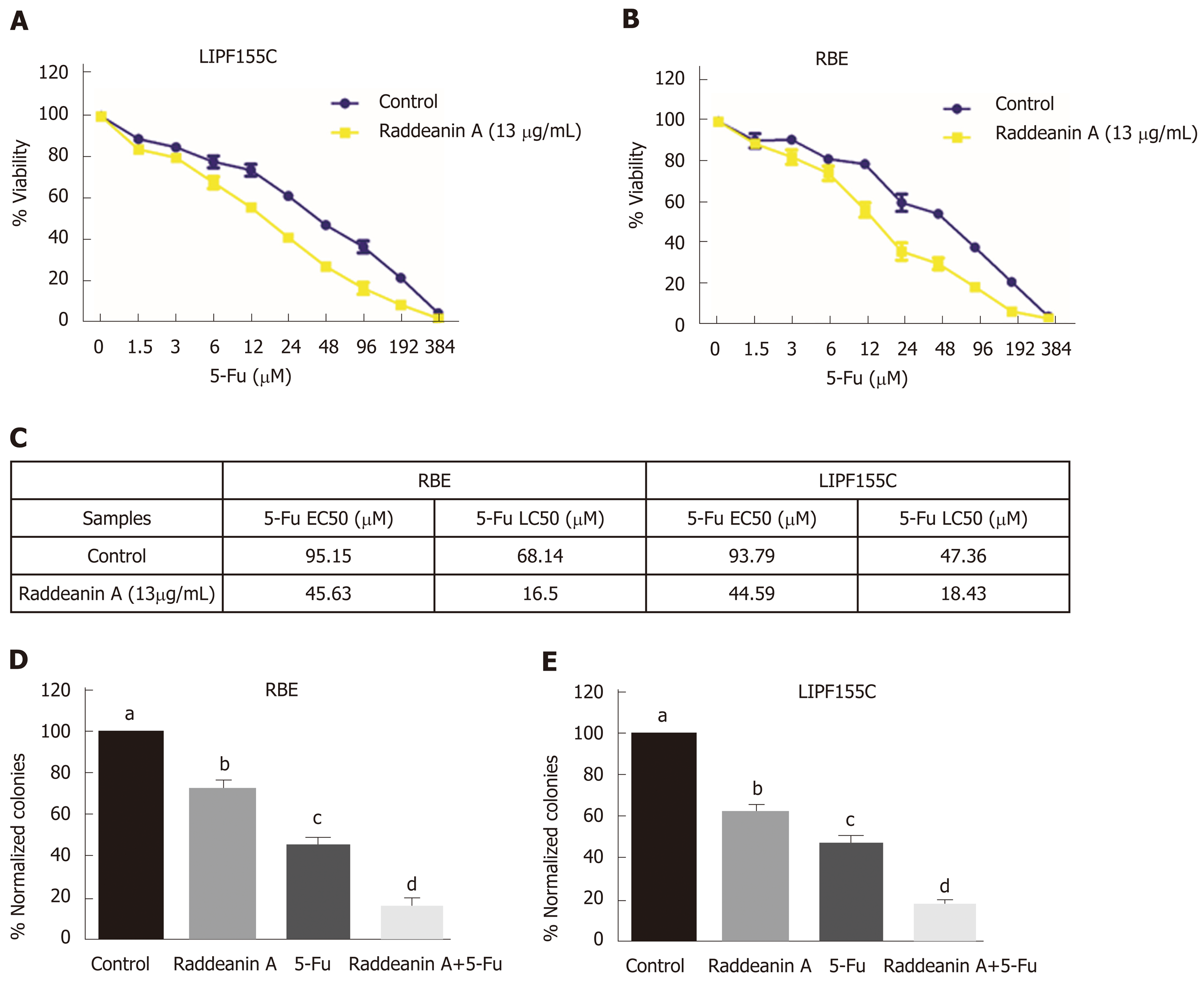
Chemoresistance is currently one of the major issues for therapy of bile duct cancer. Thus, the effects of RA on 5-Fu effectiveness were evaluated by using the combination of RA (13 μg/mL) and different increasing doses of 5-Fu, a common proapoptotic compound, in RBE and LIPF155C lines. RA was found to sensitize both tumoral cell lines (Figure 4A and B), namely, greatly reduced the EC50 (about 2-fold) and LC50 (more than 3-fold) (Figure 4C). Clonogenic assays revealed that 5-Fu displayed more potent roles to reduce cell colony formation compared with RA; however, RA was able to enhance such inhibitory effect of 5-Fu in both cell lines compared with the effect of using 5-FU alone (Figure 4D and E).
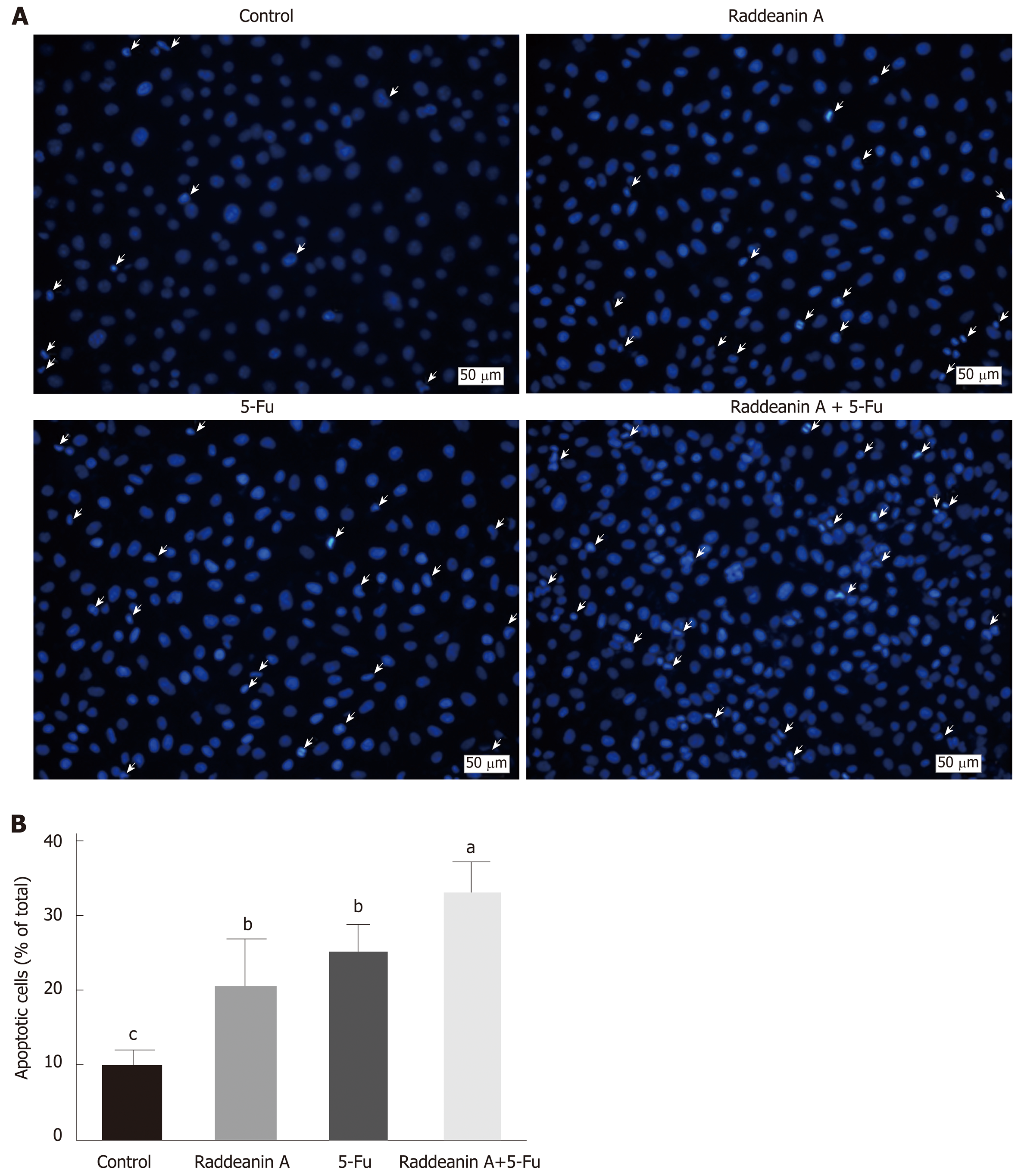
Hoechst staining assay was performed to evaluate the effects of RA on 5-Fu-resistant cell line RBE/5-Fu (Figure 5A and B). RBE/5-Fu (50 μmol/L) cells were treated with 13 μg/mL RA and the results suggested that either RA or 5-Fu promoted cell death. Furthermore, the effects of the combination of both were more potent than those of each one.
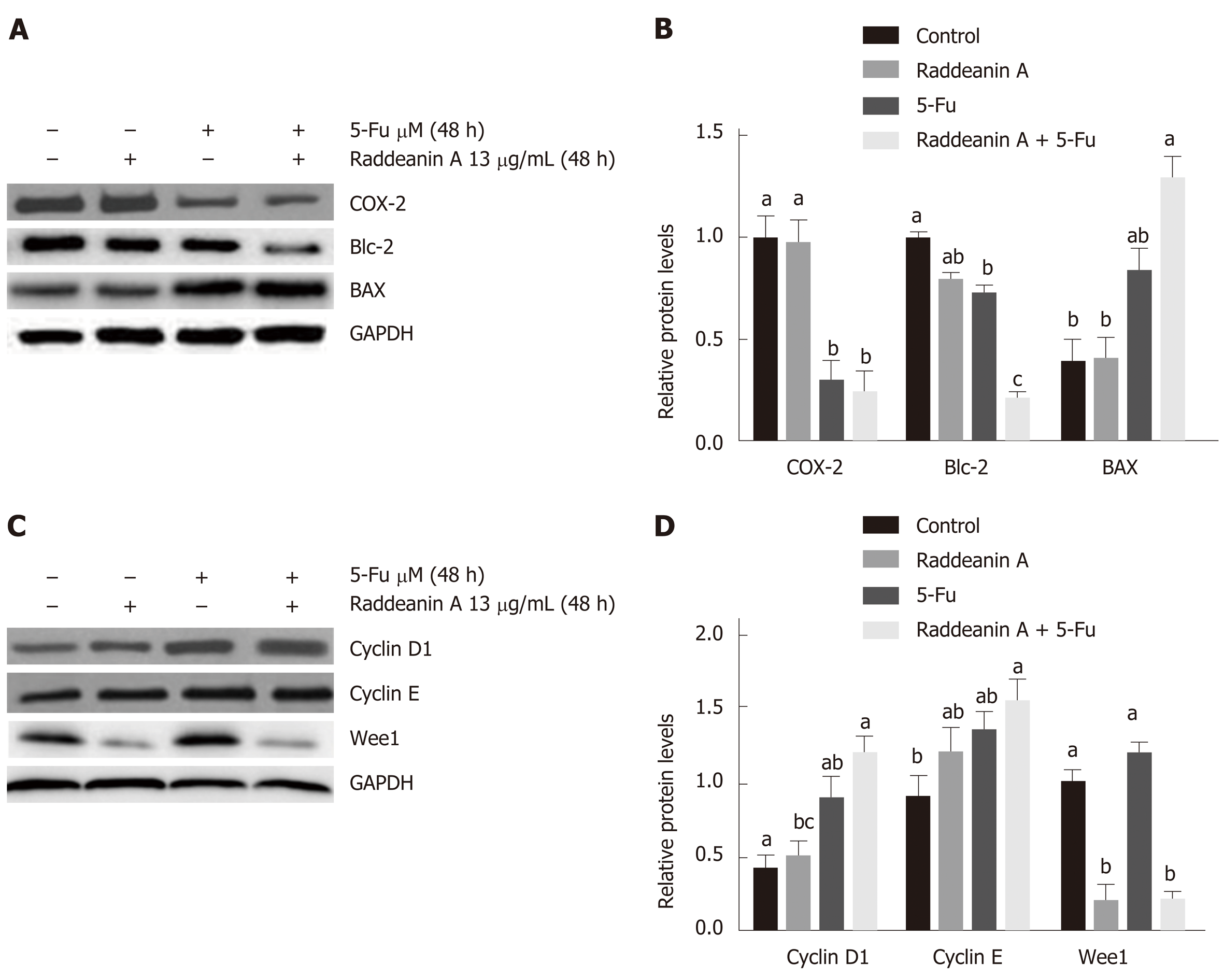
The RBE cell line was treated with 13 μg/mL RA for 48 h and cell cycle- and apoptosis-related protein expression was detected by Western blot (Figure 6A-D). Compared with the control group, RA treatment only decreased Wee1 protein level while 5-Fu treatment alone reduced Cox-2 and Bcl-2 expression and increased Bax and cyclin D1 protein expression. RA treatment combined with 5-Fu decreased protein expression of Cox-2, Bcl-2, and Wee1 whereas increased protein levels of Bax, cyclin D1, and cyclin E. 5-Fu treatment was associated with a lower protein level of Cox-2 and higher Wee1 expression compared with the effect of RA.
As a malignant cancer of the biliary tract, there are still limited precise diagnosis and effective therapy for bile duct cancer[24], although the incidence and mortality rates of bile duct cancer are increasing in recent years[25]. Therefore, there is an urgent demand to seek a novel strategy to improve diagnostic efficiency and therapeutic effect. RA has been demonstrated to play cytotoxic roles in various tumor cell lines via initiating apoptosis and impairing the cell cycle[6]. In our study, the cell viability, migration, and colony formation abilities in four cholangiocarcinoma cell lines were significantly reduced by RA treatment. Also, RA was found to sensitize the 5-Fu treated tumoral cell lines as well as facilitate apoptosis of 5-Fu-resistant cell line. Further Western blot results revealed that Wee1 expression level was dampened by RA (13 μg/mL) treatment and that the combinational effects of RA (13 μg/mL) and 5-Fu (35 μM) were associated with a group of cell cycle- and apoptosis-related factors. The results collectively suggest that the anti-bile duct cancer effects of RA may involve a series of regulatory mechanisms on the cell proliferation and apoptosis.
Wee1, a member of the family of protein kinases, is related to the regulation of G2 checkpoint in response to DNA damage[26,27]. Increasing evidence in recent years demonstrates that the overexpression of Wee1 kinase is associated with various malignancies, such as breast cancer[28], hepatocellular carcinoma[15], and malignant melanoma[16]. In this study, RA treatment was found to be related to decreased Wee1 protein level in RBE cell line, which indicates that the effects of RA potentially relies on the Wee1-dependent mechanism. Downregulation of Wee1 has been reported to contribute to the apoptosis of ovarian tumor[29] and neuroblastoma cells[30]. In particular, the checkpoint in the cell cycle is an important self-check mechanism to terminate the cell cycle process in response to DNA damage[27]. Despite DNA damage, the down-regulated Wee1 expression in tumor cells plays essential roles in main-taining cell proliferation via halting cell cycle arrest, thereby facilitating apoptosis and mitotic arrest during mitosis[27,31]. Collectively, RA-induced decreased cell viability and impaired cell functions in our study may result from the activation of Wee1 signaling that triggers apoptosis of cholangiocarcinoma cells.
5-Fu is a worldwide used chemotherapeutic treatment for many tumors, such as ovarian, breast, pancreatic, and colorectal cancers[32]. It has been well-documented that 5-Fu generates DNA damage through breaking DNA double-strand structure[33] and exerts anticancer effects via suppression of thymidylate synthase[34]. However, increasing reports revealed that the drug resistance has been becoming a significant limitation to the clinical application of 5-Fu in cancer treatment[34], which was also observed in cholangiocarcinoma[35,36]. In this study, the results suggest that RA sensitized cholangiocarcinoma cell lines and also enhanced the anti-cancer effects of 5-Fu. Furthermore, the combinational effects of RA and 5-Fu were found to be associated with alterations of a group of the cell cycle and apoptosis protein expression, such as decreased expression of Cox-2, Bcl-2, and Wee1 and increased levels of Bax, cyclin D1, and cyclin E.
As an important inducible enzyme, Cox-2 has been demonstrated to be involved in angiogenesis and tumorigenesis[37]. Cox-2 expression can be activated by various factors, such as mitogens, oncogenes, as well as carcinogens[38]. Selective Cox-2 inhibitors, for example, rofecoxib and celecoxib, are capable of suppressing established tumor growth and preventing tumorigenesis[37,39]. The down-regulated expression of Cox-2 induced by the combinational treatment in this study suggests that cox-2 may play key roles in the inhibition of cholangiocarcinoma cell growth.
Mitochondrial apoptosis pathway is an important programmed cell death pathway[40] and involves a great number of morphological changes and pathological alterations, including cancers[41,42]. Activation of the mitochondrial apoptotic pathway causes alterations of mitochondrial membrane properties through the Bcl-2 family, including translocation of Bax and suppression of Bcl-2. Thus, Bax/bcl-2 ratio is widely used as a predictive marker for evaluation of cancer therapy[43,44]. In this study, RA treatment in cholangiocarcinoma cell lines is associated with increased Bax/Bcl-2 ratio, namely, increased Bax and decreased bcl-2 level, which is consistent with the findings from previous studies[4,45]. The results indicate that the mitochondrial apoptosis pathway may be involved in the anti-tumor effects of RA in bile duct cancer.
In addition to apoptosis, the cell cycle was also found to be related to the effects of RA on the cholangiocarcinoma cell lines in this study. It is well-studied that cell cycle progression is associated with sophisticated molecular and cellular cascades[46] and participates in the development of various types of cancer[27]. During these processes, the cyclin family and their catalytic subunits cyclin-dependent kinases form a series of molecular complexes in every phase of the cell cycle to regulate this complicated cell cycle progression[27]. The cyclin D-Cdk4/6-cyclin E-Cdk2 signaling pathway plays an essential role in G1-S transition and the response to DNA damage[47,48]. Some evidence reveals that increased cyclins E and D1 protein levels are highly related to the initiation of apoptosis and sensitization to radiation in tumor cells[49,50], suggesting that increased cyclin E and D1 may act as proapoptotic factors in cholangiocarcinoma cells simultaneously treated with RA and 5-Fu.
In conclusion, our results suggest that RA treatment causes increased apoptosis and impaired cell functions in cholangiocarcinoma cell lines via a Wee1-dependent mechanism and that RA is an enhancer of 5-Fu in bile duct cancer through activating multiple cell cycle and apoptosis-related factors, such as Cox-2, Bax, Bcl-2, and cyclins E/D1. These findings together indicate that RA is a potential novel therapeutic treatment for bile duct cancer.
Bile duct cancer is characterized by fast metastasis and invasion and has been thought of as aggressive cancer due to the lack of effective diagnosis at an early stage. The 5-year survival rate of bile duct cancer has not substantially improved in clinical practice. In addition, it is reported that 5-fluorouracil (5-Fu) has been widely applied in treatments for various cancers, achieving a great therapeutic effect. Furthermore, raddeanin A (RA) plays essential proapoptotic roles in various types of tumor cells.
Exploring and developing effective diagnostic ways and therapies for bile duct cancer is greatly urgent for both basic science and clinical management.
The objective of this study was to determine the effects of RA on bile duct cancer cells and the underlying mechanisms.
In this study, RA at different concentrations was administered to four cholangiocarcinoma cell lines (RBE, LIPF155C, LIPF178C, and LICCF). Cell viability, Wound-healing migration, Transwell invasion, and Hoechst staining assays were performed to evaluate cell activities. Western blot analysis was performed to the apoptosis-related pathway.
RA inhibited cell viability in a dose-dependent pattern in the four cell lines. In RBE and LIPF155C cell lines, the migration and colony formation abilities were impaired by RA. Also, RA sensitized cell lines to 5-Fu treatment, promoting the effects of 5-Fu in cholangiocarcinoma cell lines. The role of RA was associated with reduced Wee1 expression. Furthermore, the combinational effect of RA and 5-Fu led to the inhibition of cyclooxygenase-2, B cell lymphoma 2, and Wee1 as well as the elevation of Bax, cyclin D1, and cyclin E.
RA administration could effectively regulate apoptosis and cell functions in cholangiocarcinoma cell lines in a Wee1-dependent pattern and promote the effect of 5-Fu in bile duct cancer. Collectively, RA is a promising treatment for bile duct cancer.
This study was aimed to provide clues to the application of RA for the therapy of bile duct cancer.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Katuchova J, Tanabe S S-Editor: Yan JP L-Editor: Wang TQ E-Editor: Zhang YL
| 1. | Doherty B, Nambudiri VE, Palmer WC. Update on the Diagnosis and Treatment of Cholangiocarcinoma. Curr Gastroenterol Rep. 2017;19:2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 156] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 2. | Ishigami K, Nosho K, Koide H, Kanno S, Mitsuhashi K, Igarashi H, Shitani M, Motoya M, Kimura Y, Hasegawa T, Kaneto H, Takemasa I, Suzuki H, Nakase H. MicroRNA-31 reflects IL-6 expression in cancer tissue and is related with poor prognosis in bile duct cancer. Carcinogenesis. 2018;39:1127-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 3. | Blechacz B. Cholangiocarcinoma: Current Knowledge and New Developments. Gut Liver. 2017;11:13-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 347] [Article Influence: 43.4] [Reference Citation Analysis (0)] |
| 4. | Xue G, Zou X, Zhou JY, Sun W, Wu J, Xu JL, Wang RP. Raddeanin A induces human gastric cancer cells apoptosis and inhibits their invasion in vitro. Biochem Biophys Res Commun. 2013;439:196-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 5. | Wang MK, Ding LS, Wu FE. [Antitumor effects of raddeanin A on S180, H22 and U14 cell xenografts in mice]. Ai Zheng. 2008;27:910-913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 155] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 6. | Wang Y, Bao X, Zhao A, Zhang J, Zhang M, Zhang Q, Ma B. Raddeanin A inhibits growth and induces apoptosis in human colorectal cancer through downregulating the Wnt/β-catenin and NF-κB signaling pathway. Life Sci. 2018;207:532-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 7. | Wang Q, Mo J, Zhao C, Huang K, Feng M, He W, Wang J, Chen S, Xie Z, Ma J, Fan S. Raddeanin A suppresses breast cancer-associated osteolysis through inhibiting osteoclasts and breast cancer cells. Cell Death Dis. 2018;9:376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F, Reichert M, Beatty GL, Rustgi AK, Vonderheide RH, Leach SD, Stanger BZ. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148:349-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1667] [Article Influence: 128.2] [Reference Citation Analysis (0)] |
| 9. | Ghosh N, Chaki R, Mandal V, Mandal SC. COX-2 as a target for cancer chemotherapy. Pharmacol Rep. 2010;62:233-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 249] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 10. | Khan Z, Khan N, Tiwari RP, Sah NK, Prasad GB, Bisen PS. Biology of Cox-2: An application in cancer therapeutics. Curr Drug Targets. 2011;12:1082-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 112] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 11. | Malumbres M, Barbacid M. Cell cycle, CDKs and cancer: A changing paradigm. Nat Rev Cancer. 2009;9:153-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2982] [Cited by in RCA: 2823] [Article Influence: 176.4] [Reference Citation Analysis (0)] |
| 12. | Landis MW, Pawlyk BS, Li T, Sicinski P, Hinds PW. Cyclin D1-dependent kinase activity in murine development and mammary tumorigenesis. Cancer Cell. 2006;9:13-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 242] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 13. | Bani-Hani KE, Almasri NM, Khader YS, Sheyab FM, Karam HN. Combined evaluation of expressions of cyclin E and p53 proteins as prognostic factors for patients with gastric cancer. Clin Cancer Res. 2005;11:1447-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | De Witt Hamer PC, Mir SE, Noske D, Van Noorden CJ, Würdinger T. WEE1 kinase targeting combined with DNA-damaging cancer therapy catalyzes mitotic catastrophe. Clin Cancer Res. 2011;17:4200-4207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 203] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 15. | Masaki T, Shiratori Y, Rengifo W, Igarashi K, Yamagata M, Kurokohchi K, Uchida N, Miyauchi Y, Yoshiji H, Watanabe S, Omata M, Kuriyama S. Cyclins and cyclin-dependent kinases: Comparative study of hepatocellular carcinoma versus cirrhosis. Hepatology. 2003;37:534-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 166] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 16. | Mir SE, De Witt Hamer PC, Krawczyk PM, Balaj L, Claes A, Niers JM, Van Tilborg AA, Zwinderman AH, Geerts D, Kaspers GJ, Peter Vandertop W, Cloos J, Tannous BA, Wesseling P, Aten JA, Noske DP, Van Noorden CJ, Würdinger T. In silico analysis of kinase expression identifies WEE1 as a gatekeeper against mitotic catastrophe in glioblastoma. Cancer Cell. 2010;18:244-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 248] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 17. | Yu J, Zhang L. Apoptosis in human cancer cells. Curr Opin Oncol. 2004;16:19-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 18. | Jin Z, El-Deiry WS. Overview of cell death signaling pathways. Cancer Biol Ther. 2005;4:139-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 730] [Cited by in RCA: 887] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 19. | Ashkenazi A, Fairbrother WJ, Leverson JD, Souers AJ. From basic apoptosis discoveries to advanced selective BCL-2 family inhibitors. Nat Rev Drug Discov. 2017;16:273-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 652] [Article Influence: 81.5] [Reference Citation Analysis (0)] |
| 20. | González-Vallinas M, Molina S, Vicente G, de la Cueva A, Vargas T, Santoyo S, García-Risco MR, Fornari T, Reglero G, Ramírez de Molina A. Antitumor effect of 5-fluorouracil is enhanced by rosemary extract in both drug sensitive and resistant colon cancer cells. Pharmacol Res. 2013;72:61-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 21. | Subbarayan PR, Lee K, Ardalan B. Arsenic trioxide suppresses thymidylate synthase in 5-FU-resistant colorectal cancer cell line HT29 In Vitro re-sensitizing cells to 5-FU. Anticancer Res. 2010;30:1157-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 22. | Pulito C, Mori F, Sacconi A, Goeman F, Ferraiuolo M, Pasanisi P, Campagnoli C, Berrino F, Fanciulli M, Ford RJ, Levrero M, Pediconi N, Ciuffreda L, Milella M, Steinberg GR, Cioce M, Muti P, Strano S, Blandino G. Metformin-induced ablation of microRNA 21-5p releases Sestrin-1 and CAB39L antitumoral activities. Cell Discov. 2017;3:17022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 23. | Zheng XT, Wu ZH, Wei Y, Dai JJ, Yu GF, Yuan F, Ye LC. Induction of autophagy by salidroside through the AMPK-mTOR pathway protects vascular endothelial cells from oxidative stress-induced apoptosis. Mol Cell Biochem. 2017;425:125-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 24. | Blechacz B, Gores GJ. Cholangiocarcinoma: Advances in pathogenesis, diagnosis, and treatment. Hepatology. 2008;48:308-321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 553] [Cited by in RCA: 536] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 25. | Lazaridis KN, Gores GJ. Cholangiocarcinoma. Gastroenterology. 2005;128:1655-1667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 340] [Cited by in RCA: 344] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 26. | Do K, Doroshow JH, Kummar S. Wee1 kinase as a target for cancer therapy. Cell Cycle. 2013;12:3159-3164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 221] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 27. | Otto T, Sicinski P. Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer. 2017;17:93-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1413] [Cited by in RCA: 1377] [Article Influence: 172.1] [Reference Citation Analysis (0)] |
| 28. | Iorns E, Lord CJ, Grigoriadis A, McDonald S, Fenwick K, Mackay A, Mein CA, Natrajan R, Savage K, Tamber N, Reis-Filho JS, Turner NC, Ashworth A. Integrated functional, gene expression and genomic analysis for the identification of cancer targets. PLoS One. 2009;4:e5120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 105] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 29. | Wang F, Zhu Y, Huang Y, McAvoy S, Johnson WB, Cheung TH, Chung TK, Lo KW, Yim SF, Yu MM, Ngan HY, Wong YF, Smith DI. Transcriptional repression of WEE1 by Kruppel-like factor 2 is involved in DNA damage-induced apoptosis. Oncogene. 2005;24:3875-3885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 67] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Creevey L, Ryan J, Harvey H, Bray IM, Meehan M, Khan AR, Stallings RL. MicroRNA-497 increases apoptosis in MYCN amplified neuroblastoma cells by targeting the key cell cycle regulator WEE1. Mol Cancer. 2013;12:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 31. | Hirai H, Arai T, Okada M, Nishibata T, Kobayashi M, Sakai N, Imagaki K, Ohtani J, Sakai T, Yoshizumi T, Mizuarai S, Iwasawa Y, Kotani H. MK-1775, a small molecule Wee1 inhibitor, enhances anti-tumor efficacy of various DNA-damaging agents, including 5-fluorouracil. Cancer Biol Ther. 2010;9:514-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 191] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 32. | Pinedo HM, Peters GF. Fluorouracil: Biochemistry and pharmacology. J Clin Oncol. 1988;6:1653-1664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 593] [Cited by in RCA: 559] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 33. | De Angelis PM, Svendsrud DH, Kravik KL, Stokke T. Cellular response to 5-fluorouracil (5-FU) in 5-FU-resistant colon cancer cell lines during treatment and recovery. Mol Cancer. 2006;5:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 182] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 34. | Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: Mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3:330-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3255] [Cited by in RCA: 3630] [Article Influence: 165.0] [Reference Citation Analysis (0)] |
| 35. | Wu WR, Zhang R, Shi XD, Zhu MS, Xu LB, Zeng H, Liu C. Notch1 is overexpressed in human intrahepatic cholangiocarcinoma and is associated with its proliferation, invasiveness and sensitivity to 5-fluorouracil in vitro. Oncol Rep. 2014;31:2515-2524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 36. | Lim JH, Ryu JK, Choi YJ, Kwon J, Kim JY, Lee YB, Kim JH, Yoon WJ, Kim YT, Yoon YB. A Case of Common Bile Duct Cancer That Completely Responded to Combination Chemotherapy of Gemcitabine and TS-1. Gut Liver. 2013;7:371-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 37. | Leahy KM, Ornberg RL, Wang Y, Zweifel BS, Koki AT, Masferrer JL. Cyclooxygenase-2 inhibition by celecoxib reduces proliferation and induces apoptosis in angiogenic endothelial cells in vivo. Cancer Res. 2002;62:625-631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer J, Koki AT. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer. 2000;89:2637-2645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 39. | Harris RE, Alshafie GA, Abou-Issa H, Seibert K. Chemoprevention of breast cancer in rats by celecoxib, a cyclooxygenase 2 inhibitor. Cancer Res. 2000;60:2101-2103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 40. | Igney FH, Krammer PH. Death and anti-death: Tumour resistance to apoptosis. Nat Rev Cancer. 2002;2:277-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1399] [Cited by in RCA: 1448] [Article Influence: 63.0] [Reference Citation Analysis (0)] |
| 41. | Pujals A, Renouf B, Robert A, Chelouah S, Hollville E, Wiels J. Treatment with a BH3 mimetic overcomes the resistance of latency III EBV (+) cells to p53-mediated apoptosis. Cell Death Dis. 2011;2:e184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 42. | Gobé G, Rubin M, Williams G, Sawczuk I, Buttyan R. Apoptosis and expression of Bcl-2, Bcl-XL, and Bax in renal cell carcinomas. Cancer Invest. 2002;20:324-332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 83] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 43. | Sakinah SA, Handayani ST, Hawariah LP. Zerumbone induced apoptosis in liver cancer cells via modulation of Bax/Bcl-2 ratio. Cancer Cell Int. 2007;7:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 95] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 44. | Mackey TJ, Borkowski A, Amin P, Jacobs SC, Kyprianou N. bcl-2/bax ratio as a predictive marker for therapeutic response to radiotherapy in patients with prostate cancer. Urology. 1998;52:1085-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 141] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 45. | Teng YH, Li JP, Liu SL, Zou X, Fang LH, Zhou JY, Wu J, Xi SY, Chen Y, Zhang YY, Xu S, Wang RP. Autophagy Protects from Raddeanin A-Induced Apoptosis in SGC-7901 Human Gastric Cancer Cells. Evid Based Complement Alternat Med. 2016;2016:9406758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 46. | Shapiro GI. Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol. 2006;24:1770-1783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 783] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 47. | Sears RC, Nevins JR. Signaling networks that link cell proliferation and cell fate. J Biol Chem. 2002;277:11617-11620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 254] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 48. | Bartek J, Lukas J. Pathways governing G1/S transition and their response to DNA damage. FEBS Lett. 2001;490:117-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 332] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 49. | Coco Martin JM, Balkenende A, Verschoor T, Lallemand F, Michalides R. Cyclin D1 overexpression enhances radiation-induced apoptosis and radiosensitivity in a breast tumor cell line. Cancer Res. 1999;59:1134-1140. [PubMed] |
| 50. | Mazumder S, DuPree EL, Almasan A. A dual role of cyclin E in cell proliferation and apoptosis may provide a target for cancer therapy. Curr Cancer Drug Targets. 2004;4:65-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 97] [Article Influence: 4.6] [Reference Citation Analysis (0)] |