Published online Jun 28, 2019. doi: 10.3748/wjg.v25.i24.2977
Peer-review started: April 1, 2019
First decision: April 16, 2019
Revised: April 24, 2019
Accepted: May 18, 2019
Article in press: May 18, 2019
Published online: June 28, 2019
Processing time: 90 Days and 23.3 Hours
Hepatocellular carcinoma (HCC) arises on the background of chronic liver disease. Despite the development of effective anti-viral therapeutics HCC is continuing to rise, in part driven by the epidemic of non-alcoholic fatty liver disease. Many patients present with advanced disease out with the criteria for transplant, resection or even locoregional therapy. Currently available therapeutics for HCC are effective in a small minority of individuals. However, there has been a major global interest in immunotherapies for cancer and although HCC has lagged behind other cancers, great opportunities now exist for treating HCC with newer and more sophisticated agents. Whilst checkpoint inhibitors are at the forefront of this revolution, other therapeutics such as inhibitory cytokine blockade, oncolytic viruses, adoptive cellular therapies and vaccines are emerging. Broadly these may be categorized as either boosting existing immune response or stimulating de novo immune response. Although some of these agents have shown promising results as monotherapy in early phase trials it may well be that their future role will be as combination therapy, either in combination with one another or in combination with treatment modalities such as locoregional therapy. Together these agents are likely to generate new and exciting opportunities for treating HCC, which are summarized in this review.
Core tip: A significant proportion of patients with hepatocellular carcinoma (HCC) present with advanced disease, for which there are limited systemic therapeutic options. Complicating this, HCC often develops on a background of cirrhosis, which can preclude the use of certain cytotoxic agents. Immunotherapy has previously not been an available therapeutic option in HCC. However, checkpoint inhibition therapy was recently licensed as a second line option for advanced disease. Multiple other promising agents are in development which boost existing immune response or stimulate a de novo immune response. These agents are discussed herein.
- Citation: Johnston MP, Khakoo SI. Immunotherapy for hepatocellular carcinoma: Current and future. World J Gastroenterol 2019; 25(24): 2977-2989
- URL: https://www.wjgnet.com/1007-9327/full/v25/i24/2977.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i24.2977
Hepatocellular carcinoma (HCC) is the predominant form of primary liver cancer, constituting 75%-85% of cases. It presents a significant health burden as the sixth most commonly diagnosed cancer worldwide in 2018. In addition, reflecting its poor outcome, it was the fourth leading cause of cancer death[1]. The incidence of HCC varies country by country depending on the relative prevalence of key risk factors. These include chronic infection with either hepatitis B virus (HBV) or hepatitis C virus (HCV), as well as aflatoxin exposure[2]. These are more common in lower human development index countries. Although vaccination against HBV is recommended to reduce HCC development and has been used successfully in countries such as Taiwan, problems such as logistics of delivery and vaccine availability are significant factors which limit this approach[3].
HCV and HBV related cirrhosis are associated with the highest incidences of HCC. However, other aetiologies of cirrhosis, including non-alcoholic fatty liver disease (NAFLD), alcohol related liver disease and hereditary haemochromatosis are also strongly associated with an increased incidence of HCC[4]. Furthermore, any optimism about the revelatory impact new HCV drugs will have on HCC burden is forestalled by the global rise in NAFLD, type 2 diabetes mellitus and the “metabolic syndrome” as endemic risk factors for HCC development[5,6]. In particular the incidence of HCC is rising, particularly in countries with a high socio-demographic index, and consistent with this HCC may arise on the background of a non-cirrhotic liver in NALFD[7,8].
Surgery is the most successful treatment for HCC, either liver transplantation or liver resection depending on liver function, the presence of portal hypertension and tumour burden. Selection for surgery remains based upon Barcelona clinic liver cancer (BCLC) criteria for the most part, although “extended criteria” may be used by experienced centres[9]. Unfortunately given that liver cancer usually occurs on the background of cirrhosis, the residual liver post-resection still presents an environment predisposing to the development of subsequent tumours. Thus recurrence is a significant problem[10]. Transplantation obviates this concern to an extent by removing the background liver but patients may be outwith criteria for transplant at presentation or subsequently become ineligible while waiting for a suitable donor organ. Post-liver transplantation HCC recurrence appears to occur in 10%-20% of patients[11].
Locoregional therapy is the main alternative therapy depending on the stage of the underlying liver disease. This largely comprises two major types: (1) Percutaneous ablation such as microwave ablation or radiofrequency ablation (RFA); (2) Intra-arterial chemoembolotherapy, namely transcatheter chemoembolization[9]. Ablation may even be a first line option over surgery in selected early stage tumours with comparable mortality rates, albeit higher recurrence rates[12]. Nonetheless, locoregional therapy is for the most part not curative treatment with recurrence being common.
Unfortunately the majority (> 70%) of patients present with advanced disease outwith the criteria for transplant, surgery or locoregional therapeutic options[13]. For these patients there remains a paucity of approved therapeutic options. Sorafenib is an oral multi-tyrosine kinase inhibitor (TKI), targeting a number of signaling pathways such as vascular endothelial growth factor (VEGF), and increasing median survival by 3 mo[14]. On this basis it is recommended as the standard first line systemic therapy for patients with Child-Pugh A cirrhosis and BCLC-C[9]. Another oral multi-TKI lenvatinib is now recommended as alternative first line therapy based on non-inferiority to sorafenib[9,15]. Based upon survival benefits versus placebo in patients previously treated with sorafenib both oral multi-TKIs regorafenib and cabozantinib have been added as second line systemic therapeutic options[16,17]. Importantly, liver cirrhosis precludes the potential use of many cytotoxic drugs and so, combined with the resistance of HCC to a number of reagents, the development of prospective chemotherapy regimens has been relatively difficult[18].
In recent years cancer immunotherapy has seen a rapid expansion in terms of the number of agents which confer a prognostic benefit by awakening the immune system to mount a response against developing cancers, with particular success in metastatic melanoma[19]. Given the paucity of therapeutic options it is therefore logical that that these immunotherapeutic targets should be explored in HCC, particularly given the correlation between immunological findings and outcomes in HCC[20]. Recently, nivolumab was added as the first Food and Drug Administration (FDA) approved immunotherapy for HCC[15]. This expansion in the therapeutic armoury is a welcome one. In this article we review the basis for immunotherapy in HCC, the agents studied to date as well as potential future developments.
In addition to its many metabolic functions the liver has an important immuno-regulatory role. Its dual supply of arterial and portal systemic blood makes it a unique recipient for gut pathogen exposure. This anatomy is combined with a honeycomb-like vasculature of sinusoids densely laden with specialized immunocytes including macrophages (Kupffer cells), liver sinusoidal endothelial cells (LSECs), natural killer (NK) cells and innate T cells[21].
The LSECs account for roughly 50% of the non-parenchymal cells within the liver[22]. In conjunction with Kupffer cells and dendritic cells (DCs) one of their roles is to act as antigen presenting cells as part of the hepatic reticulo-endothelial system[23]. Residing in the space of Dissé between the parenchymal cells and LSECs are hepatic stellate cells which contribute an immune sentinel role in this nuanced interplay[24]. Further to the LSECs is an abundance of resident liver lymphocytes, including NK and innate T cells, which serve a number of roles including innate immune response against viruses, intracellular bacteria, tumours and parasites[25,26]. There is thus a rich effector population which needs to be responsive to pathogens, but also immuno-regulatory when exposed to the non-pathogenic antigens that flood the liver via the portal vein. These include innocuous nutrient antigens, bacterial degradation products, damaged cells and of course pathogenic or toxic components. It is for this reason that the immune response within the liver requires such precise homeostatic control. The inherent immune tolerogenicity which the liver has developed to adapt to this unique environment of antigen exposure has been well described[23,27]. This manifest immunotolerant capacity is evident in the liver’s relatively low rates of allograft rejection compared to other organ transplants[28,29].
There are two aspects to immunobiology in the context of cirrhosis. One is that in cirrhosis there is an active immune-mediated inflammatory process and that as decompensation develops it becomes progressively systemic[30]. However, the precise nature of the immune activity in cirrhosis depends on the underlying liver disease. Combined with a dysregulated immune response that predisposes to infection, this has been elsewhere described as “cirrhosis-associated immune dysfunction”[31]. It is well established the predisposition to bacterial infection and this is most evident in acute-on-chronic liver failure[32,33]. Additionally, the structural damage of cirrhosis compromises reticulo-endothelial function leading to impaired immune surveillance[21]. However, immune dysregulation is also manifest in non-cirrhotic patients, with irregularities such as elevated levels of endogenous cytokines, and a pro-inflammatory environment especially in autoimmune liver disease and viral hepatitis[34].
In the majority of cases HCC is associated with chronic liver disease and in particular cirrhosis. The underlying inflammatory process described above drives hepatocellular DNA damage, endoplasmic reticulum stress and subsequent necrosis of the hepatocyte which leads to regenerative nodular formation, dysplastic nodules and ultimately carcinoma[35]. HCV and HBV also drive an immune-mediated inflammatory response which promotes neoplastic change, the latter also mediating its carcinogenic properties via direct oncogenic transformation following incorporation into host cell DNA[36]. Furthermore, once HCC has developed the tumour can be associated with a rich immune cell infiltrate. Detailed analysis of HCCs indicated that approximately 25% have high inflammatory scores, with high or moderate levels of lymphocyte infiltration[37,38]. As one might expect, tumour infiltrating lymphocytes (TILs) form a large component in solid tumours, in an attempt by the host to mediate an antitumour reaction[39]. Unfortunately this cellular response can be dysfunctional with a higher proportion of CD4+ (helper or T regulatory cells) to CD8+ cells. This promotes immune tolerance and has been shown to confer a worse prognosis[40]. Additionally the innate immune system may be attenuated as evidenced by the hypofunctionality of NK cells in HCC[41,42].
However, although TILs can be identified, within the tumour microenvironment in cirrhosis, they often prove insufficient to control tumour growth[43]. Expansion of myeloid derived suppressor cells (MDSCs) as well as Tregs appears to further enable the evasion of tumour cells from immune detection[44]. This creates an immuno-suppressive immune environment through the secretion of transforming growth factor (TGF-β). In addition there are multiple mechanisms of immune evasion including secretion of other immunoregulatory cytokines such as interleukin-10 (IL-10), downregulation of ligands that activate immune cells including MHC class I and NKG2D ligands and expression of ligands that directly inhibit lymphocytes, including both T cells and NK cells[45-48]. Thus HCC is a challenging environment for the immune system. Nevertheless, immunotherapy is one of the most promising avenues for future therapies.
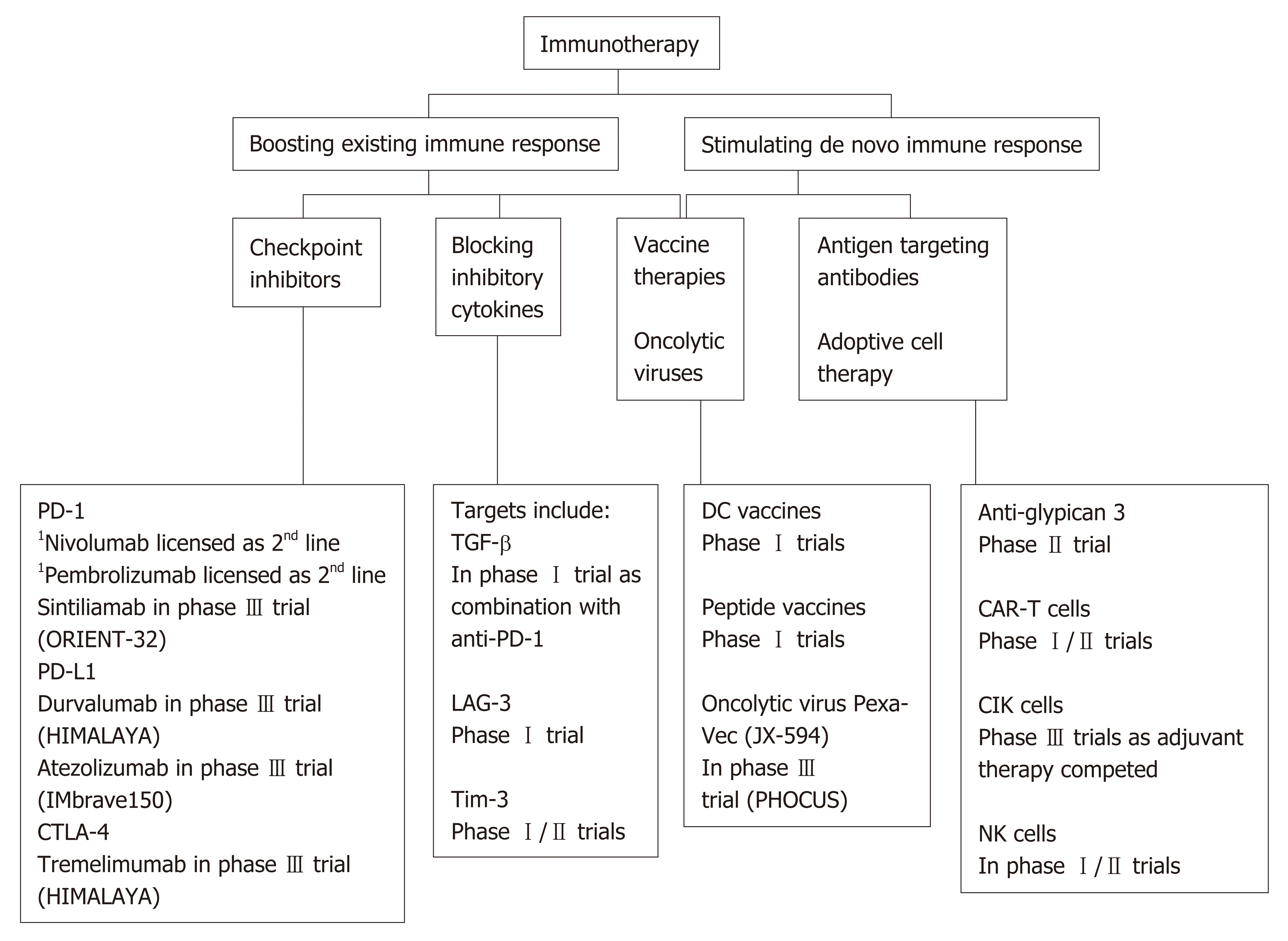
Current approaches of immunotherapy were shown in Figure 1. Current immuno-therapeutic strategies are based on two fundamental principles: (1) The ability to unmask current immune responses; or (2) The need to stimulate new or different immune responses. Unleashing current immune response relies on there being a pre-existing immune reactivity to cancer which is being held in check by micro-environmental factors, such as inhibitory receptors on T cells especially programmed cell death protein 1 (PD-1) and cytotoxic T-lymphocyte associated antigen 4 (CTLA-4), or alternatively immunosuppressive cytokines such as TGF-β. Checkpoint inhibitors fall within this category, and importantly for these therapies to work the precise molecules that the cells are targeting do not need to be known. Conversely, antibodies that directly target molecules expressed on HCC, such as alpha-fetoprotein (AFP) or glypican-3 (GPC-3) are within the second category. These strategies can be enhanced by coupling these antibodies to effector cells, such as T cells or even NK cells. Vaccine therapeutics and the use of oncolytic viruses, discussed below, may straddle these two mechanisms by unmasking pre-existing and inducing de novo T cell responses to antigens expressed by HCC. Additionally, tumour ablation liberates antigens into the periphery and can augment CTL responses, that have been correlated with survival[49-51].
A rapidly growing list of blocking antibodies to immune checkpoints has been approved by the FDA in recent years for cancer treatment. In general, these are thought to be most effective for tumours with a high mutagenic load, such as melanoma[52]. Although these have been in trials for some time, it was not until recently that the first of these checkpoint inhibitors was approved for use in HCC, when the PD-1 inhibitor nivolumab (Opdivo®) gained FDA approval (Table 1).
| Trial identifier | Targets | Drugs | Other treatment | Patient group | Status | n | Estimated completion date |
| NCT03794440 | PD-1 | Sintilimab | vs Sorafenib | Advanced HCC | Recruiting | 566 | Dec 2022 |
| VEGF | Bevacizumab biosimilar | ||||||
| NCT03298451 | CTLA-4 | Tremelimumab | vs Sorafenib | HCC BCLC stage B not eligibile for locoregional therapy | Recruiting | 1310 | Jun 2021 |
| PD-L1 | Durvalumab | ||||||
| NCT02702401 | PD-1 | Pembrolizumab | vs Placebo | Advanced HCC | Results available | 408 | Dec 2019 |
| NCT02576509 | PD-1 | Nivolumab | vs Sorafenib | Advanced HCC | Active, not recruiting | 726 | July 2020 |
| NCT03755739 | PD-1 | Pembrolizumab | Peripheral vs hepatic infusion following TACE | Advanced HCC | Recruiting | 200 | Nov 2021 |
| NCT03062358 | PD-1 | Pembrolizumab | vs Placebo | Advanced HCC | Recruiting | 450 | Jan 2022 |
| NCT03713593 | PD-1 | Pembrolizumab | vs Lenvatinib monotherapy | Advanced HCC | Recruiting | 750 | July 2022 |
| VEGR | Lenvatinib | ||||||
| NCT03847428 | PD-L1 | Durvalumab | Combination with resection/MWA vs resection/MWA alone | HCC eligible for curative resection/MWA | Not yet recruiting | 888 | June 2023 |
| VEGF | Bevacizumab | ||||||
| NCT03764293 | PD-1 | Camrelizumab | vs Sorafenib | Advanced HCC | Not yet recruiting | 510 | Jan 2022 |
| TKI | Apatinib | ||||||
| NCT03434379 | PD-L1 | Atezolizumab | vs Sorafenib | Advanced HCC | Recruiting | 480 | June 2022 |
| VEGF | Bevacizumab |
Programmed cell death protein 1: PD-1 is a cell surface protein expressed on an extensive number of immune cell types, predominantly CD8+ T cells but also CD4+ T cells, B cells, NKs, Tregs, MDSCs and DCs[53-55]. It is upregulated following activation of T cells and when it binds to PD-L1 (or PD-L2) on target cells this inhibits effector T cell responses. Therefore, blocking its action is an attractive target of immunotherapy. Nivolumab’s grading as an approved second line therapy for HCC is supported by evidence from the CheckMate040 trial. This was a phase I/II, open label, non-comparative, dose escalation and expansion trial in advanced HCC of mixed underlying chronic liver diseases (n = 262)[56]. 46 (96%) of 48 patients discontinued treatment in the dose escalation phase, 42 (88%) due to disease progression. However, the objective response rate was 20% in the dose-expansion phase. Incorporation of nivolumab into the AASLD guideline on HCC as second line systemic therapy was made in advance of the first phase III trial results on the basis of CheckMate040[15]. CheckMate459 (NCT02576509), is a phase III, randomized, open label trial of nivolumab versus sorafenib which has closed to recruitment and results are awaited at the present time.
There are a number of registered phase III trials looking at PD-1 checkpoint blockade. The ORIENT-32 study (NCT03794440) is a randomized, open-label, multicentre trial in China randomizing patients to a combination of sintilimab (PD-1 inhibitor) and bevacizumab (anti-VEGF antibody) versus a control arm of sorafenib. The RATIONALE-301 study (NCT03412773) is a phase III trial randomizing patients to the PD-1 inhibitor tislelizumab monotherapy versus sorafenib. Pembrolizumab (Keytruda®), another IgG4 isotype antibody targeting the PD-1 receptor of lymphocytes, has been similarly studied as monotherapy. In the 104 patients enrolled in the open-label, phase II trial KEYNOTE-240 (NCT02702401) there were mixed results. An objective response in 17% of patients (complete in 1%, partial in 16%) was offset by serious treatment-related adverse events in 15%, including 1 death associated with ulcerative oesophagitis attributed to treatment[57]. However, based on the promising response rates pembrolizumab was granted accelerated approval for HCC. Unfortunately Merck and Co. has recently announced that the subsequent phase III trial of pembrolizumab versus placebo did not meet its co-primary endpoints of overall survival (OS) and progression-free survival in patients with advanced HCC[58]. These results, although disappointing, would appear to be consistent with the opinion that checkpoint blockade may well be most efficacious as combination therapy[54,59]. Combination of PD-1/PD-L1 blockade may be with VEGF inhibition (NCT03794440, NCT03713593, NCT03764293, NCT03434379), as well as with locoregional treatment or resection (NCT03847428, NCT03755739), or indeed with another checkpoint inhibitor. However, care needs to be taken as combination therapy with checkpoint inhibitors can lead to higher rates of side-effects including an immune-mediated hepatitis[60].
Cytotoxic T-lymphocyte associated antigen 4: CTLA-4 is another membrane bound molecule which keeps the immune response in check. It has a multifaceted role, actively competing for binding to the co-stimulatory molecule CD28, and leading to increased secretion of the immunoregulatory cytokine IL-10, as well as serving as a key mediator by which regulatory T cells (Tregs) dampen immune response[61,62]. Inhibition of CTLA-4 is associated with improved clinical outcomes in other malignancies such as metastatic melanoma. In 2011, ipilimumab (YERVOY®) was the first checkpoint inhibitor approved by the FDA. Reports of therapeutic CTLA-4 blockade in HCC have also shown promise. In 2013 there was a reported phase I trial of 20 patients with advanced HCC and a background of HCV who received the CTLA-4 inhibitor tremelimumab[63]. Partial response was 17.6% and disease control was 76.4%. Time to progression was 6.48 mo (95% confidence interval 3.95-9.14). Although intense elevations in transaminases were common, particularly after first dose, no course of steroids were required for hepatotoxicity. The randomized phase III HIMALAYA trial (NCT03298451) is recruiting patients for randomization to a combination of CTLA-4 inhibitor tremelimumab and PD-L1 inhibitor durvalumab versus sorafenib. It leads on from the early clinical data of 40 patients enrolled in the phase I trial of durvalumab and tremelimumab in advanced HCC[64]. Patient selection for combination checkpoint blockade will no doubt be key with 20% of patients having at least one grade 3 adverse event.
Furthermore, the combination of checkpoint blockade with locoregional therapy is attractive, with the potential for CTLA-4 inhibition to uncouple the systemic immunogenic response which occurs with tumour necrosis. A phase II trial which enrolled 32 patients (predominantly with HCV) treated with tremelimumab and followed by subtotal RFA or chemoembolization, demonstrated that of the 19 patients with lesions evaluable 5 of them (26%) showed partial response[65].
T-cell immunoglobulin and mucin-domain containing-3: TIM-3 is another trans-membrane protein which is known to be expressed on CD4+ T Helper 1 cells and CD8+ cytotoxic cells[66]. Initially identified due to a putative pathogenic association with autoimmune disease, interest in this as a therapeutic has grown due to its role in the ability of tumour cells to evade immunosurveillance. A propensity of CD8+ cells to co-express both PD-1 and TIM-3 seems to contribute to the dysfunctional phenotype of CD8+ T cells[67,68]. We await with interest a phase II trial of dual blockade of anti-TIM-3 and PD-1 in HCC (NCT03680508) which has not yet begun recruitment.
Transforming growth factor-β: TGF-β is a membrane bound molecule expressed on and associated with a Treg subset which suppresses CD4+ T cell response in tumour tissue, promoting progression, in both murine models and HCC patients[69]. Co-expression of PD-1 on these CD4+CD69+ Tregs makes for another potential combination therapy. Results are awaited of a phase I trial assigning patients in parallel to both the anti-TGF-β monoclonal antibody NIS793 and PD-1 inhibitor spartalizumab (NCT02947165) due for completion in April 2021.
Lymphocyte activation gene 3: Closely related to CD4, lymphocyte activation gene 3 (LAG-3) is a membrane protein that binds the same ligand, MHC-II[70]. Not only do these proteins suppress T cell activity and cytokine release, but they are also of considerable interest due to their upregulation in T cell exhaustion in the context of chronic viral infection or cancer[71,72]. The synergistic effect of LAG-3 with PD-1 to induce tumour regression raises another further potential combination therapy[73]. Although engineered LAG-3 binding therapy for solid tumours remains in early phase trials, given its significant upregulation in tumour infiltrating CD8+ T cells of HCC patients, its potential in liver cancer is eagerly awaited[74].
In contrast to the active augmentation of immune response seen with checkpoint inhibition therapy, adoptive cell transfer aims to improve HCC outcomes by passively administering autologous lymphocytes following ex vivo cultivation[75]. This is a long-standing therapeutic strategy starting over 30 years ago, with infusion of TILs leading to improved responses in metastatic melanoma[76]. The broad cell subsets that have been studied in HCC to date include NK cells, cytokine-induced killer (CIK) cells or TILs, and finally chimeric antigen receptor T cells (CAR-T cells).
The first of these, NK cells, form as much as 50% of innate immune cell rich infiltrate within the liver[26]. With their ability to kill cells without prior activation or priming they are best known for forming part of the host defence against infection and tumour development[26]. In a murine model expanded NK cells exert a significant cytotoxic effect against HCC cells, reducing tumour growth and improving OS. Furthermore, they enchanced the effect of sorafenib in the same study[77]. Although clinical data on use is limited, there has been a successfully conducted phase I trial in patients with liver cirrhosis with HCC undergoing liver transplantation. NK cells derived from donor liver perfusate, stimulated with IL-2 and administered showed upregulation of peripheral NK cell cytotoxicity and no adverse events[78]. We await ongoing trials of high affinity NK cells versus sorafenib (NCT03563170) and combination therapy of NK cell transfer with irreversible electroporation (IRE) vs IRE alone (NCT03008343).
Next are the CIK cells which represent another novel immunotherapeutic option. By incubating peripheral blood monocytes with cytokines including IL-1, IL-2, IFN-γ and a monoclonal antibody against the T cell marker CD3, these cells show a significant inhibitory effect on tumorigenesis[79]. These MHC-unrestricted cytotoxic lymphocytes are made up of a heterogeneous group of efficient cytotoxic effector cells comprising predominantly CD3+CD56+ T cells, and some CD3-CD56+ NK cells. Trials into reinfusion of CIK cells have predominantly been studied as an adjunctive therapy following surgical resection, with a theoretical base in murine models showing an effect of these cells on micrometastases[80]. Early trials randomizing post-curative resection patients to adjuvant CIK cell therapy or no adjuvant showed promising results with a significantly reduced risk of recurrence, but without an improvement in OS[81,82]. The largest study to date, involving 230 patients, was a multicenter, randomized, open label phase 3 trial studying CIK cell therapy as adjuvant to RFA, ethanol injection or curative resection. This showed an improvement of 14 mo in recurrence free survival[83]. A systematic review and meta-analysis of CIK cell therapy in HCC in Asia reached similar conclusions that in selected patients, progression free survival and recurrence free survival are improved[84].
Antigen specific T cells have also been studied. These include native TILs and also CAR-T cells. A phase I trial studied administration of autologous TILs in 15 patients with HCC post-resection. This showed successful expansion in 88% and there were no serious adverse events (SAEs) reported[85]. The incorporation of a chimeric antigen receptor into T cells to modulate their antigen selectivity and signaling offers another exciting prospect for immunotherapy in HCC. Although discovered 30 years ago, CAR-T cell therapy for HCC remained relatively in its infancy until more recently[86]. The FDA approved the first two CAR-T cell therapies Kymriah® and Yescarta® for lymphoma in 2018 and 2017. A plethora of trials into solid tumours have followed in parallel with these breakthroughs in lymphoma.
HCC has a number of tumour associated antigens (TAAs). Selection of an appropriate antigen for CAR-T cells is integral to their success as a prospective immuno-therapeutic option. Given its high expression and association with poor prognosis in HCC, GPC-3, a member of the glypican family, has been a natural target antigen to study[87,88]. There remains one published phase I trial of 13 patients, 8 of whom had lymphodepletion with fludarabine and cyclophosphamide. These were patients with advanced HCC, portal vein invasion or extrahepatic metastases. This has only been published in abstract form to date, but no dose limiting toxicity was identified and there was one SAE of grade 3 fever found[89]. We await the published results of a further phase I clinical trial (NCT02723942) that was completed in 2017. There are currently five Phase I/II trials recruiting, four of which examining GPC-3 and one EpCAM (NCT03198546, NCT03130712, NCT02715362, NCT03013712, NCT02723942). AFP is another potential target TAA that is being explored[90,91]. Unfortunately the propensity of AFP to be found on healthy hepatocytes has stymied its potential as a target antigen of CAR-T cell or other targeted immunotherapies.
Tumour vaccines are agents which increase specific immune responses to tumour antigens. Registered clinical trials for such tumour vaccines in HCC are currently relatively few compared to those studying adoptive cellular therapies and checkpoint inhibitors, in part because of previously disappointing trial results, and also relative lack of efficacy of other tumour vaccines. This may be related to the previous difficulty in identifying the correct tumour antigens, which has now become possible through recent technological breakthroughs allowing massive parallel DNA sequencing. Thus, priming an immune response whether in isolation, or more likely in combination with an immune modulator remains an attractive therapeutic strategy for HCC. A number of agents have been examined to date with regards to this.
Dendritic cells: DCs are professional antigen presenting cells, responsible for a multitude of tasks, including absorption, processing and presentation of TAAs. Allogeneic DCs form one broad subset of vaccines by providing both the antigen and the secondary co-stimulation required to prime an effective T cell response. Isolating DCs from peripheral blood, expanding them ex vivo and stimulating with cytokines such as granulocyte-macrophage colony-stimulating factor (GM-CSF) produces primed DCs for reinfusion. The injection of these cells to induce recruitment of effector cells and provoke a cascade of tumour lysis and further TAA release, is another attractive, targeted mechanism[92]. A number of techniques may be employed to optimize this TAA priming and enhance the efficacy of the vaccine. DCs can be transduced with DNA or RNA encoding known TAAs, or they may be incubated with tumour lysate or fusion of DCs and tumour cells[93].
A recently published phase I trial studied intra-tumoral injection of ilixadencel (pro-inflammatory allogeneic DCs stimulated by GM-CSF and IL-4) either as monotherapy or in combination with sorafenib in 17 patients. The primary objective was to evaluate tolerability. Only one grade 3 adverse event was recorded. 73% of the 15 evaluable patients demonstrated increased tumour specific CD8+ T cells in peripheral blood, suggesting a successful immune provoked response at least[94].
Peptide vaccines: Peptide vaccines constitute an alternative option in terms of generating an effective immune reaction. However, although there has been success in terms of immunological surrogates such as generating GPC-3 reactive cytotoxic T lymphocytes in one phase I trial, this has not translated into clinical successes[95]. Despite a plethora of TAAs identified in HCC only trials utilizing AFP, GPC-3 and MRP3 have shown any success inducing a T cell response rate over 70%, with other TAAs such as SSX-2, NY-ESO-1, hTERT and MAGE-A all inducing much lower rates[96].
Oncolytic viruses: A more recent development in the arena of tumour vaccines is the use of oncolytic viruses. These therapeutically useful viruses are targeted to preferentially replicate in cancer cells. To date they have been predominantly introduced by intra-tumoral injection. The modified poxvirus JX-594 remains the lead oncolytic virus of interest in clinical trials with regards to HCC. As an im-munotherapeutic agent it piqued considerable interest when it conferred a dose-related survival benefit (median of 14.1 mo compared to 6.7 mo) in a phase II dose-finding trial of 30 patients[97]. The global, randomized, open-label, phase III study of Pexa-Vec (JX-594; an oncolytic vaccinia virus which selectively targets cancer cells) is currently recruiting patients with advanced HCC to two arms of vaccination with sorafenib vs. sorafenib alone[98]. We eagerly await the results of this particularly as a combination therapy.
Immunotherapy for HCC is still in its infancy compared to other tumours. Encouraging results with PD-1 inhibitors are emerging, and prospects for combination therapies arising. This makes immunological sense, as the immune system is a multi-faceted and integrated effector system. Optimising this response is challenging, especially because of the immune environment on which HCC arises, and the challenges of treating an individual with cirrhosis, which substantially decreases the therapeutic index of these agents. Nevertheless, the massive interest in immunotherapy, gives hope that better combinations of drugs will be found to treat this challenging disease.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: United Kingdom
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Jin C, Liu YY, Luo HS, Niu ZS, Yang L S-Editor: Yan JP L-Editor: A E-Editor: Zhang YL
| 1. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55839] [Article Influence: 7977.0] [Reference Citation Analysis (132)] |
| 2. | Global Burden of Disease Liver Cancer Collaboration. Akinyemiju T, Abera S, Ahmed M, Alam N, Alemayohu MA, Allen C, Al-Raddadi R, Alvis-Guzman N, Amoako Y, Artaman A, Ayele TA, Barac A, Bensenor I, Berhane A, Bhutta Z, Castillo-Rivas J, Chitheer A, Choi JY, Cowie B, Dandona L, Dandona R, Dey S, Dicker D, Phuc H, Ekwueme DU, Zaki MS, Fischer F, Fürst T, Hancock J, Hay SI, Hotez P, Jee SH, Kasaeian A, Khader Y, Khang YH, Kumar A, Kutz M, Larson H, Lopez A, Lunevicius R, Malekzadeh R, McAlinden C, Meier T, Mendoza W, Mokdad A, Moradi-Lakeh M, Nagel G, Nguyen Q, Nguyen G, Ogbo F, Patton G, Pereira DM, Pourmalek F, Qorbani M, Radfar A, Roshandel G, Salomon JA, Sanabria J, Sartorius B, Satpathy M, Sawhney M, Sepanlou S, Shackelford K, Shore H, Sun J, Mengistu DT, Topór-Mądry R, Tran B, Ukwaja KN, Vlassov V, Vollset SE, Vos T, Wakayo T, Weiderpass E, Werdecker A, Yonemoto N, Younis M, Yu C, Zaidi Z, Zhu L, Murray CJL, Naghavi M, Fitzmaurice C. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results From the Global Burden of Disease Study 2015. JAMA Oncol. 2017;3:1683-1691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1459] [Cited by in RCA: 1500] [Article Influence: 187.5] [Reference Citation Analysis (0)] |
| 3. | Chang MH, You SL, Chen CJ, Liu CJ, Lee CM, Lin SM, Chu HC, Wu TC, Yang SS, Kuo HS, Chen DS; Taiwan Hepatoma Study Group. Decreased incidence of hepatocellular carcinoma in hepatitis B vaccinees: A 20-year follow-up study. J Natl Cancer Inst. 2009;101:1348-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 427] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 4. | Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: Incidence and risk factors. Gastroenterology. 2004;127:S35-S50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1691] [Cited by in RCA: 1793] [Article Influence: 85.4] [Reference Citation Analysis (2)] |
| 5. | Zoller H, Tilg H. Nonalcoholic fatty liver disease and hepatocellular carcinoma. Metabolism. 2016;65:1151-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 133] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 6. | Margini C, Dufour JF. The story of HCC in NAFLD: From epidemiology, across pathogenesis, to prevention and treatment. Liver Int. 2016;36:317-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 183] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 7. | Liu Z, Jiang Y, Yuan H, Fang Q, Cai N, Suo C, Jin L, Zhang T, Chen X. The trends in incidence of primary liver cancer caused by specific etiologies: Results from the Global Burden of Disease Study 2016 and implications for liver cancer prevention. J Hepatol. 2019;70:674-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 518] [Article Influence: 86.3] [Reference Citation Analysis (0)] |
| 8. | Dyson J, Jaques B, Chattopadyhay D, Lochan R, Graham J, Das D, Aslam T, Patanwala I, Gaggar S, Cole M, Sumpter K, Stewart S, Rose J, Hudson M, Manas D, Reeves HL. Hepatocellular cancer: The impact of obesity, type 2 diabetes and a multidisciplinary team. J Hepatol. 2014;60:110-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 433] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 9. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5593] [Cited by in RCA: 6063] [Article Influence: 866.1] [Reference Citation Analysis (3)] |
| 10. | Portolani N, Coniglio A, Ghidoni S, Giovanelli M, Benetti A, Tiberio GA, Giulini SM. Early and late recurrence after liver resection for hepatocellular carcinoma: Prognostic and therapeutic implications. Ann Surg. 2006;243:229-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 706] [Cited by in RCA: 728] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 11. | Vivarelli M, Cucchetti A, La Barba G, Ravaioli M, Del Gaudio M, Lauro A, Grazi GL, Pinna AD. Liver transplantation for hepatocellular carcinoma under calcineurin inhibitors: Reassessment of risk factors for tumor recurrence. Ann Surg. 2008;248:857-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 204] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 12. | Majumdar A, Roccarina D, Thorburn D, Davidson BR, Tsochatzis E, Gurusamy KS. Management of people with early- or very early-stage hepatocellular carcinoma: An attempted network meta-analysis. Cochrane Database Syst Rev. 2017;3:CD011650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 13. | Thomas MB, Jaffe D, Choti MM, Belghiti J, Curley S, Fong Y, Gores G, Kerlan R, Merle P, O'Neil B, Poon R, Schwartz L, Tepper J, Yao F, Haller D, Mooney M, Venook A. Hepatocellular carcinoma: Consensus recommendations of the National Cancer Institute Clinical Trials Planning Meeting. J Clin Oncol. 2010;28:3994-4005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 323] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 14. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10271] [Article Influence: 604.2] [Reference Citation Analysis (2)] |
| 15. | Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, Roberts LR, Heimbach JK. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68:723-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2121] [Cited by in RCA: 3242] [Article Influence: 463.1] [Reference Citation Analysis (1)] |
| 16. | Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki JP, Ollivier-Hourmand I, Kudo M, Cheng AL, Llovet JM, Finn RS, LeBerre MA, Baumhauer A, Meinhardt G, Han G; RESORCE Investigators. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389:56-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2160] [Cited by in RCA: 2715] [Article Influence: 339.4] [Reference Citation Analysis (0)] |
| 17. | Abou-Alfa GK, Meyer T, Cheng A-L, B A, El-Khoueiry, Rimassa L, Ryoo BY, Cicin I, Merle P, Park JW, Blanc JF, Bolondi L, Klümpen HJ, Chan SL, Dadduzio V, Hessel C, Borgman-Hagey AE, Schwab G, Kelley RK. Cabozantinib (C) versus placebo (P) in patients (pts) with advanced hepatocellular carcinoma (HCC) who have received prior sorafenib: Results from the randomnized phase III CELESTIAL trial. J Clin Oncol. 2018;36:94. [RCA] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 18. | Asghar U, Meyer T. Are there opportunities for chemotherapy in the treatment of hepatocellular cancer? J Hepatol. 2012;56:686-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 155] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 19. | Pasquali S, Hadjinicolaou AV, Chiarion Sileni V, Rossi CR, Mocellin S. Systemic treatments for metastatic cutaneous melanoma. Cochrane Database Syst Rev. 2018;2:CD011123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 104] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 20. | El-Khoueiry A. The Promise of Immunotherapy in the Treatment of Hepatocellular Carcinoma. Am Soc Clin Oncol Educ Book. 2017;37:311-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Jenne CN, Kubes P. Immune surveillance by the liver. Nat Immunol. 2013;14:996-1006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 619] [Cited by in RCA: 780] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 22. | Racanelli V, Rehermann B. The liver as an immunological organ. Hepatology. 2006;43:S54-S62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 870] [Cited by in RCA: 968] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 23. | Thomson AW, Knolle PA. Antigen-presenting cell function in the tolerogenic liver environment. Nat Rev Immunol. 2010;10:753-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 528] [Cited by in RCA: 599] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 24. | Winau F, Quack C, Darmoise A, Kaufmann SH. Starring stellate cells in liver immunology. Curr Opin Immunol. 2008;20:68-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 25. | Kumar N, Khakoo SI. Hepatocellular carcinoma: Prospects for natural killer cell immunotherapy. HLA. 2018;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Gao B, Jeong WI, Tian Z. Liver: An organ with predominant innate immunity. Hepatology. 2008;47:729-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 612] [Cited by in RCA: 721] [Article Influence: 42.4] [Reference Citation Analysis (1)] |
| 27. | Bowen DG, Zen M, Holz L, Davis T, McCaughan GW, Bertolino P. The site of primary T cell activation is a determinant of the balance between intrahepatic tolerance and immunity. J Clin Invest. 2004;114:701-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 95] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 28. | Takatsuki M, Uemoto S, Inomata Y, Egawa H, Kiuchi T, Fujita S, Hayashi M, Kanematsu T, Tanaka K. Weaning of immunosuppression in living donor liver transplant recipients. Transplantation. 2001;72:449-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 214] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 29. | Starzl TE, Demetris AJ, Trucco M, Murase N, Ricordi C, Ildstad S, Ramos H, Todo S, Tzakis A, Fung JJ. Cell migration and chimerism after whole-organ transplantation: The basis of graft acceptance. Hepatology. 1993;17:1127-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 561] [Cited by in RCA: 504] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 30. | Arroyo V, García-Martinez R, Salvatella X. Human serum albumin, systemic inflammation, and cirrhosis. J Hepatol. 2014;61:396-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 417] [Article Influence: 37.9] [Reference Citation Analysis (0)] |
| 31. | Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: Distinctive features and clinical relevance. J Hepatol. 2014;61:1385-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 691] [Cited by in RCA: 851] [Article Influence: 77.4] [Reference Citation Analysis (1)] |
| 32. | Fernández J, Gustot T. Management of bacterial infections in cirrhosis. J Hepatol. 2012;56 Suppl 1:S1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 250] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 33. | Fernández J, Acevedo J, Wiest R, Gustot T, Amoros A, Deulofeu C, Reverter E, Martínez J, Saliba F, Jalan R, Welzel T, Pavesi M, Hernández-Tejero M, Ginès P, Arroyo V; European Foundation for the Study of Chronic Liver Failure. Bacterial and fungal infections in acute-on-chronic liver failure: Prevalence, characteristics and impact on prognosis. Gut. 2018;67:1870-1880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 414] [Cited by in RCA: 398] [Article Influence: 56.9] [Reference Citation Analysis (2)] |
| 34. | Oliviero B, Varchetta S, Paudice E, Michelone G, Zaramella M, Mavilio D, De Filippi F, Bruno S, Mondelli MU. Natural killer cell functional dichotomy in chronic hepatitis B and chronic hepatitis C virus infections. Gastroenterology. 2009;137:1151-1160, 1160.e1-1160.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 338] [Article Influence: 21.1] [Reference Citation Analysis (1)] |
| 35. | Severi T, van Malenstein H, Verslype C, van Pelt JF. Tumor initiation and progression in hepatocellular carcinoma: Risk factors, classification, and therapeutic targets. Acta Pharmacol Sin. 2010;31:1409-1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 130] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 36. | Hernandez-Gea V, Toffanin S, Friedman SL, Llovet JM. Role of the microenvironment in the pathogenesis and treatment of hepatocellular carcinoma. Gastroenterology. 2013;144:512-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 593] [Article Influence: 49.4] [Reference Citation Analysis (0)] |
| 37. | Sia D, Jiao Y, Martinez-Quetglas I, Kuchuk O, Villacorta-Martin C, Castro de Moura M, Putra J, Camprecios G, Bassaganyas L, Akers N, Losic B, Waxman S, Thung SN, Mazzaferro V, Esteller M, Friedman SL, Schwartz M, Villanueva A, Llovet JM. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology. 2017;153:812-826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 690] [Cited by in RCA: 663] [Article Influence: 82.9] [Reference Citation Analysis (0)] |
| 38. | Cancer Genome Atlas Research Network. Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma. Cell. 2017;169:1327-1341.e23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1578] [Cited by in RCA: 1732] [Article Influence: 216.5] [Reference Citation Analysis (1)] |
| 39. | Qin LX. Inflammatory immune responses in tumor microenvironment and metastasis of hepatocellular carcinoma. Cancer Microenviron. 2012;5:203-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 40. | Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C, Yao J, Jin L, Wang H, Yang Y, Fu YX, Wang FS. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328-2339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 598] [Cited by in RCA: 693] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 41. | Chuang WL, Liu HW, Chang WY. Natural killer cell activity in patients with hepatocellular carcinoma relative to early development and tumor invasion. Cancer. 1990;65:926-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 42. | Wu Y, Kuang DM, Pan WD, Wan YL, Lao XM, Wang D, Li XF, Zheng L. Monocyte/macrophage-elicited natural killer cell dysfunction in hepatocellular carcinoma is mediated by CD48/2B4 interactions. Hepatology. 2013;57:1107-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 215] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 43. | Behboudi S, Boswell S, Williams R. Cell-mediated immune responses to alpha-fetoprotein and other antigens in hepatocellular carcinoma. Liver Int. 2010;30:521-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 44. | Hoechst B, Ormandy LA, Ballmaier M, Lehner F, Krüger C, Manns MP, Greten TF, Korangy F. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4(+)CD25(+)Foxp3(+) T cells. Gastroenterology. 2008;135:234-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 601] [Cited by in RCA: 637] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 45. | Li H, Li CW, Li X, Ding Q, Guo L, Liu S, Liu C, Lai CC, Hsu JM, Dong Q, Xia W, Hsu JL, Yamaguchi H, Du Y, Lai YJ, Sun X, Koller PB, Ye Q, Hung MC. MET Inhibitors Promote Liver Tumor Evasion of the Immune Response by Stabilizing PDL1. Gastroenterology. 2019;156:1849-1861.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 148] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 46. | Li Z, Li B, Peng D, Xing H, Wang G, Li P, Wang J, Ye G, Chen J. Expression and clinical significance of PD-1 in hepatocellular carcinoma tissues detected by a novel mouse anti-human PD-1 monoclonal antibody. Int J Oncol. 2018;52:2079-2092. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 47. | Makarova-Rusher OV, Medina-Echeverz J, Duffy AG, Greten TF. The yin and yang of evasion and immune activation in HCC. J Hepatol. 2015;62:1420-1429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 251] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 48. | Kamimura H, Yamagiwa S, Tsuchiya A, Takamura M, Matsuda Y, Ohkoshi S, Inoue M, Wakai T, Shirai Y, Nomoto M, Aoyagi Y. Reduced NKG2D ligand expression in hepatocellular carcinoma correlates with early recurrence. J Hepatol. 2012;56:381-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 49. | Nobuoka D, Motomura Y, Shirakawa H, Yoshikawa T, Kuronuma T, Takahashi M, Nakachi K, Ishii H, Furuse J, Gotohda N, Takahashi S, Nakagohri T, Konishi M, Kinoshita T, Komori H, Baba H, Fujiwara T, Nakatsura T. Radiofrequency ablation for hepatocellular carcinoma induces glypican-3 peptide-specific cytotoxic T lymphocytes. Int J Oncol. 2012;40:63-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 50. | Zerbini A, Pilli M, Penna A, Pelosi G, Schianchi C, Molinari A, Schivazappa S, Zibera C, Fagnoni FF, Ferrari C, Missale G. Radiofrequency thermal ablation of hepatocellular carcinoma liver nodules can activate and enhance tumor-specific T-cell responses. Cancer Res. 2006;66:1139-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 195] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 51. | Flecken T, Schmidt N, Hild S, Gostick E, Drognitz O, Zeiser R, Schemmer P, Bruns H, Eiermann T, Price DA, Blum HE, Neumann-Haefelin C, Thimme R. Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology. 2014;59:1415-1426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 298] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 52. | Eroglu Z, Zaretsky JM, Kim DW, Algazi A, Johnson DB, Liniker E, Ben Kong, Munhoz R, Rapisuwon S, Gherardini PF, Chmielowski B, Wang X, Shintaku IP, Wei C, Sosman JA, Joseph RW, Postow MA, Carlino MS, Hwu WJ, Scolyer RA, Messina J, Cochran AJ, Long GV, Ribas A, Hu-Lieskovan S. High response rate to PD-1 blockade in desmoplastic melanomas. Nature. 2018;553:347-350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 204] [Cited by in RCA: 263] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 53. | Francisco LM, Salinas VH, Brown KE, Vanguri VK, Freeman GJ, Kuchroo VK, Sharpe AH. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med. 2009;206:3015-3029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1346] [Cited by in RCA: 1623] [Article Influence: 101.4] [Reference Citation Analysis (0)] |
| 54. | Hato T, Goyal L, Greten TF, Duda DG, Zhu AX. Immune checkpoint blockade in hepatocellular carcinoma: Current progress and future directions. Hepatology. 2014;60:1776-1782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 55. | Nguyen LT, Ohashi PS. Clinical blockade of PD1 and LAG3--potential mechanisms of action. Nat Rev Immunol. 2015;15:45-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 496] [Article Influence: 49.6] [Reference Citation Analysis (0)] |
| 56. | Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Lang L, Neely J, Tang H, Dastani HB, Melero I, El-Khoueiry AB, Dela Cruz C. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3278] [Cited by in RCA: 3313] [Article Influence: 414.1] [Reference Citation Analysis (1)] |
| 57. | Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, Knox J, Daniele B, Webber AL, Ebbinghaus SW, Ma J, Siegel AB, Cheng AL, Kudo M; KEYNOTE-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19:940-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1184] [Cited by in RCA: 1900] [Article Influence: 271.4] [Reference Citation Analysis (0)] |
| 58. | Merck. Merck Provides Update on KEYNOTE-240, a Phase 3 Study of KEYTRUDA® (pembrolizumab) in Previously Treated Patients with Advanced Hepatocellular Carcinoma. 2019;Merck Business Wire [Internet]. Available from: https://www.mrknewsroom.com/news-release/oncology/merck-provides-update-keynote-240-phase-3-study-keytruda-pembrolizumab-previou. |
| 59. | Greten TF, Duffy AG, Korangy F. Hepatocellular carcinoma from an immunologic perspective. Clin Cancer Res. 2013;19:6678-6685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 60. | Sznol M, Ferrucci PF, Hogg D, Atkins MB, Wolter P, Guidoboni M, Lebbé C, Kirkwood JM, Schachter J, Daniels GA, Hassel J, Cebon J, Gerritsen W, Atkinson V, Thomas L, McCaffrey J, Power D, Walker D, Bhore R, Jiang J, Hodi FS, Wolchok JD. Pooled Analysis Safety Profile of Nivolumab and Ipilimumab Combination Therapy in Patients With Advanced Melanoma. J Clin Oncol. 2017;35:3815-3822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 237] [Article Influence: 29.6] [Reference Citation Analysis (1)] |
| 61. | Han Y, Chen Z, Yang Y, Jiang Z, Gu Y, Liu Y, Lin C, Pan Z, Yu Y, Jiang M, Zhou W, Cao X. Human CD14+ CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2,3-dioxygenase production in hepatocellular carcinoma. Hepatology. 2014;59:567-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 185] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 62. | Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, Nomura T, Sakaguchi S. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322:271-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2013] [Cited by in RCA: 2254] [Article Influence: 132.6] [Reference Citation Analysis (0)] |
| 63. | Sangro B, Gomez-Martin C, de la Mata M, Iñarrairaegui M, Garralda E, Barrera P, Riezu-Boj JI, Larrea E, Alfaro C, Sarobe P, Lasarte JJ, Pérez-Gracia JL, Melero I, Prieto J. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol. 2013;59:81-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 634] [Cited by in RCA: 751] [Article Influence: 62.6] [Reference Citation Analysis (0)] |
| 64. | Kelley RK, Abou-Alfa GK, Bendell JC, Kim T-Y, Borad MJ, Yong W-P, Morse M, Kang Y-K, Rebelatto M, Makowsky M, Xiao F, Morris SR, Sangro B. Phase I/II study of durvalumab and tremelimumab in patients with unresectable hepatocellular carcinoma (HCC): Phase I safety and efficacy analyses. J Clin Oncol. 2017;35:4073. [RCA] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 117] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 65. | Duffy AG, Ulahannan SV, Makorova-Rusher O, Rahma O, Wedemeyer H, Pratt D, Davis JL, Hughes MS, Heller T, ElGindi M, Uppala A, Korangy F, Kleiner DE, Figg WD, Venzon D, Steinberg SM, Venkatesan AM, Krishnasamy V, Abi-Jaoudeh N, Levy E, Wood BJ, Greten TF. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66:545-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 454] [Cited by in RCA: 640] [Article Influence: 80.0] [Reference Citation Analysis (0)] |
| 66. | Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel RA, Freeman GJ, Kuchroo VK. Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature. 2002;415:536-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1124] [Cited by in RCA: 1292] [Article Influence: 56.2] [Reference Citation Analysis (0)] |
| 67. | Anderson AC. Tim-3: An emerging target in the cancer immunotherapy landscape. Cancer Immunol Res. 2014;2:393-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 239] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 68. | Fourcade J, Sun Z, Benallaoua M, Guillaume P, Luescher IF, Sander C, Kirkwood JM, Kuchroo V, Zarour HM. Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med. 2010;207:2175-2186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 890] [Cited by in RCA: 1052] [Article Influence: 70.1] [Reference Citation Analysis (0)] |
| 69. | Han Y, Yang Y, Chen Z, Jiang Z, Gu Y, Liu Y, Xu S, Lin C, Pan Z, Zhou W, Cao X. Human hepatocellular carcinoma-infiltrating CD4⁺CD69⁺Foxp3⁻ regulatory T cell suppresses T cell response via membrane-bound TGF-β1. J Mol Med (Berl). 2014;92:539-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 70. | Triebel F, Jitsukawa S, Baixeras E, Roman-Roman S, Genevee C, Viegas-Pequignot E, Hercend T. LAG-3, a novel lymphocyte activation gene closely related to CD4. J Exp Med. 1990;171:1393-1405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 506] [Cited by in RCA: 683] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 71. | Blackburn SD, Shin H, Haining WN, Zou T, Workman CJ, Polley A, Betts MR, Freeman GJ, Vignali DA, Wherry EJ. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat Immunol. 2009;10:29-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1703] [Cited by in RCA: 1642] [Article Influence: 102.6] [Reference Citation Analysis (0)] |
| 72. | Long L, Zhang X, Chen F, Pan Q, Phiphatwatchara P, Zeng Y, Chen H. The promising immune checkpoint LAG-3: From tumor microenvironment to cancer immunotherapy. Genes Cancer. 2018;9:176-189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 278] [Article Influence: 39.7] [Reference Citation Analysis (0)] |
| 73. | Matsuzaki J, Gnjatic S, Mhawech-Fauceglia P, Beck A, Miller A, Tsuji T, Eppolito C, Qian F, Lele S, Shrikant P, Old LJ, Odunsi K. Tumor-infiltrating NY-ESO-1-specific CD8+ T cells are negatively regulated by LAG-3 and PD-1 in human ovarian cancer. Proc Natl Acad Sci USA. 2010;107:7875-7880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 572] [Cited by in RCA: 717] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 74. | Li FJ, Zhang Y, Jin GX, Yao L, Wu DQ. Expression of LAG-3 is coincident with the impaired effector function of HBV-specific CD8(+) T cell in HCC patients. Immunol Lett. 2013;150:116-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 108] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 75. | Baruch EN, Berg AL, Besser MJ, Schachter J, Markel G. Adoptive T cell therapy: An overview of obstacles and opportunities. Cancer. 2017;123:2154-2162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 76. | Rosenberg SA, Packard BS, Aebersold PM, Solomon D, Topalian SL, Toy ST, Simon P, Lotze MT, Yang JC, Seipp CA. Use of tumor-infiltrating lymphocytes and interleukin-2 in the immunotherapy of patients with metastatic melanoma. A preliminary report. N Engl J Med. 1988;319:1676-1680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1557] [Cited by in RCA: 1603] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 77. | Kamiya T, Chang YH, Campana D. Expanded and Activated Natural Killer Cells for Immunotherapy of Hepatocellular Carcinoma. Cancer Immunol Res. 2016;4:574-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 78. | Ohira M, Nishida S, Matsuura T, Fan J, Tekin A, Selvaggi G, Levi D, Tryphonopoulos P, Ruiz P, Morita Y, Fukazawa K, Ricordi C, Ohdan H, Tzakis AG. Phase I immunotherapy using liver natural killer cells for preventing recurrence of hepatocellular carcinoma in liver transplantation. Liver Transplant. 2012;18:S1-S306. |
| 79. | Wang FS, Liu MX, Zhang B, Shi M, Lei ZY, Sun WB, Du QY, Chen JM. Antitumor activities of human autologous cytokine-induced killer (CIK) cells against hepatocellular carcinoma cells in vitro and in vivo. World J Gastroenterol. 2002;8:464-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 77] [Cited by in RCA: 82] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 80. | Lafreniere R, Rosenberg SA. Successful immunotherapy of murine experimental hepatic metastases with lymphokine-activated killer cells and recombinant interleukin 2. Cancer Res. 1985;45:3735-3741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 81. | Takayama T, Sekine T, Makuuchi M, Yamasaki S, Kosuge T, Yamamoto J, Shimada K, Sakamoto M, Hirohashi S, Ohashi Y, Kakizoe T. Adoptive immunotherapy to lower postsurgical recurrence rates of hepatocellular carcinoma: A randomised trial. Lancet. 2000;356:802-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 653] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 82. | Hui D, Qiang L, Jian W, Ti Z, Da-Lu K. A randomized, controlled trial of postoperative adjuvant cytokine-induced killer cells immunotherapy after radical resection of hepatocellular carcinoma. Dig Liver Dis. 2009;41:36-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 83. | Lee JH, Lee JH, Lim YS, Yeon JE, Song TJ, Yu SJ, Gwak GY, Kim KM, Kim YJ, Lee JW, Yoon JH. Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma. Gastroenterology. 2015;148:1383-91.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 388] [Article Influence: 38.8] [Reference Citation Analysis (0)] |
| 84. | Cai XR, Li X, Lin JX, Wang TT, Dong M, Chen ZH, Jia CC, Hong YF, Lin Q, Wu XY. Autologous transplantation of cytokine-induced killer cells as an adjuvant therapy for hepatocellular carcinoma in Asia: An update meta-analysis and systematic review. Oncotarget. 2017;8:31318-31328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 85. | Jiang S, Xia J. 352PD A phase I clinical trial utilizing autologous tumor-infiltrating lymphocytes in patients with primary hepatocellular carcinoma. Ann Oncol. 2016;26:105. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 86. | Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci USA. 1989;86:10024-10028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 902] [Cited by in RCA: 1113] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 87. | Shirakawa H, Suzuki H, Shimomura M, Kojima M, Gotohda N, Takahashi S, Nakagohri T, Konishi M, Kobayashi N, Kinoshita T, Nakatsura T. Glypican-3 expression is correlated with poor prognosis in hepatocellular carcinoma. Cancer Sci. 2009;100:1403-1407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 211] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 88. | Zhu ZW, Friess H, Wang L, Abou-Shady M, Zimmermann A, Lander AD, Korc M, Kleeff J, Büchler MW. Enhanced glypican-3 expression differentiates the majority of hepatocellular carcinomas from benign hepatic disorders. Gut. 2001;48:558-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 221] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 89. | Zhai B, Shi D, Gao H, Qi X, Jiang H, Zhang Y, Chi J, Ruan H, Wang H, Ru Q, Li Z. A phase I study of anti-GPC3 chimeric antigen receptor modified T cells (GPC3 CAR-T) in Chinese patients with refractory or relapsed GPC3+ hepatocellular carcinoma (r/r GPC3+ HCC). J Clin Oncol. 2017;35:3049. [RCA] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 90. | Sun L, Guo H, Jiang R, Lu L, Liu T, He X. Engineered cytotoxic T lymphocytes with AFP-specific TCR gene for adoptive immunotherapy in hepatocellular carcinoma. Tumour Biol. 2016;37:799-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 91. | Gerry A, Sanderson J, Maroto M, Ferronha T, Ranganathan S, Norry E, Pandite L, Amado R, Jakobsen B. Targeting alpha fetoprotein with TCR engineered T cells in HCC. J Clin Oncol. 2016;34:3051. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 92. | Gustafsson K, Ingelsten M, Bergqvist L, Nyström J, Andersson B, Karlsson-Parra A. Recruitment and activation of natural killer cells in vitro by a human dendritic cell vaccine. Cancer Res. 2008;68:5965-5971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 93. | Shang N, Figini M, Shangguan J, Wang B, Sun C, Pan L, Ma Q, Zhang Z. Dendritic cells based immunotherapy. Am J Cancer Res. 2017;7:2091-2102. [PubMed] |
| 94. | Rizell M, Sternby Eilard M, Andersson M, Andersson B, Karlsson-Parra A, Suenaert P. Phase 1 Trial With the Cell-Based Immune Primer Ilixadencel, Alone, and Combined With Sorafenib, in Advanced Hepatocellular Carcinoma. Front Oncol. 2019;9:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 95. | Sawada Y, Yoshikawa T, Nobuoka D, Shirakawa H, Kuronuma T, Motomura Y, Mizuno S, Ishii H, Nakachi K, Konishi M, Nakagohri T, Takahashi S, Gotohda N, Takayama T, Yamao K, Uesaka K, Furuse J, Kinoshita T, Nakatsura T. Phase I trial of a glypican-3-derived peptide vaccine for advanced hepatocellular carcinoma: Immunologic evidence and potential for improving overall survival. Clin Cancer Res. 2012;18:3686-3696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 233] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 96. | Sun Z, Zhu Y, Xia J, Sawakami T, Kokudo N, Zhang N. Status of and prospects for cancer vaccines against hepatocellular carcinoma in clinical trials. Biosci Trends. 2016;10:85-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 97. | Heo J, Reid T, Ruo L, Breitbach CJ, Rose S, Bloomston M, Cho M, Lim HY, Chung HC, Kim CW, Burke J, Lencioni R, Hickman T, Moon A, Lee YS, Kim MK, Daneshmand M, Dubois K, Longpre L, Ngo M, Rooney C, Bell JC, Rhee BG, Patt R, Hwang TH, Kirn DH. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat Med. 2013;19:329-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 490] [Cited by in RCA: 614] [Article Influence: 51.2] [Reference Citation Analysis (0)] |
| 98. | Abou-Alfa GK, Galle PR, Chao Y, Brown KT, Heo J, Borad MJ, Luca A, Pelusio A, Agathon D, Lusky M, Breitbach C, Burke J, Qin S. PHOCUS: A phase 3 randomized, open-label study comparing the oncolytic immunotherapy Pexa-Vec followed by sorafenib (SOR) vs SOR in patients with advanced hepatocellular carcinoma (HCC) without prior systemic therapy. J Clin Oncol. 2016;34:TPS4146-TPS4146. [RCA] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |