Published online Jun 7, 2019. doi: 10.3748/wjg.v25.i21.2623
Peer-review started: March 7, 2019
First decision: March 27, 2019
Revised: May 6, 2019
Accepted: May 8, 2019
Article in press: May 8, 2019
Published online: June 7, 2019
Processing time: 92 Days and 0 Hours
Laparoscopic resection of the pancreatic body and tail is the predominant methodology to remove lesions in these locations; its safety and surgical planning are relatively mature, but it remains a complex and high-precision surgical operation, requiring abundant experience and skills in laparoscopic surgery, with a 10% rate of complications.
To verify the feasibility and safety, as well as to examine the complications of endoscopic pancreatectomy and healing mechanisms of pancreatic wounds after endoscopic resection.
Transgastric endoscopic resections of varying sizes of pancreases were performed in 15 healthy Bama miniature pigs. The technical success rate, the incidence of serious complications, and the survival of the animals were studied. The healing of the wounds was evaluated by sacrificing the animals at various time points. Finally, the expression of transforming growth factor-β1 and Smad3/Smad7 in the surgical site was examined by immunohistochemistry to explore the role of these factors in wound healing of the pancreas.
Partial and total resections were successfully performed in two groups of animals, respectively. The technical success rate and the survival rate of the pigs were both 100%. We obtained 12 pancreatic tissue samples by endoscopic resection. The pancreatic wounds were closed with metal clips in one group and the wounds healed well by forming scars. There was a small amount of pancreatic leakage in the other group, but it can be fully encapsulated. The level of transforming growth factor-β1 (TGF-β1) in the wounds increased during the inflammatory and fibrous hyperplasia phases, and decreased in the scar phase. The expression of Smad3 paralleled that of TGF-β1, while the expression of Smad7 had an inverse relationship with the expression of TGF-β1.
Purely transgastric endoscopic resection of the pancreas is a safe, effective, and feasible procedure, but the incidence of pancreatic leakage in total pancreatic tail resection is high. The expression of TGF-β1 and Smad3/Samd7 is related to the progression of pancreatic wound healing.
Core tip: Purely transgastric endoscopic resection of the pancreas is a safe, effective, and feasible procedure. Endoscopic partial resection of the pancreatic tail is safe, but the incidence of pancreatic leakage in total pancreatic tail resection is high. Wound closure with metal clips is effective in reducing pancreatic leakage and in helping to form scar tissue at the wound site. The expression of transforming growth factor-β1 and Smad3/Samd7 varies in different phases of pancreatic wound healing, illustrating the relation of these factors to the progression of wound healing.
- Citation: Wang S, Zhang K, Hu JL, Wu WC, Liu X, Ge N, Guo JT, Wang GX, Sun SY. Endoscopic resection of the pancreatic tail and subsequent wound healing mechanisms in a porcine model. World J Gastroenterol 2019; 25(21): 2623-2635
- URL: https://www.wjgnet.com/1007-9327/full/v25/i21/2623.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i21.2623
In recent years, natural orifice transluminal endoscopic surgery (NOTES) has attracted increasing attention as a new technique for minimally invasive treatment. Compared with traditional surgery, it has the advantages of less trauma, faster recovery, and no external postoperative scar. At present, endoscopic doctors globally are performing a large number of experimental animal studies and a small amount of clinical research about NOTES. Most of the results demonstrate the promising application prospects of this technology. However, some experts still question the safety and feasibility of NOTES technology. Therefore, the application of NOTES technology must be further investigated. The initial outcome of interest is the feasibility and safety of the procedure. We aimed to evaluate its complications in animal experiments to provide a theoretical basis for further clinical application of NOTES and to accumulate more operative experience in this technique.
At present, the resection of the pancreatic body and tail generally requires invasive surgical treatment. Laparoscopic resection of the pancreatic body and tail is the predominant methodology to remove lesions in these locations; its safety and surgical planning are relatively mature, but it remains a complex and high-precision surgical operation, requiring abundant experience and skills in laparoscopic surgery, with a 10% rate of complications. Therefore, invasive surgery is predominantly applied to cases with refractory symptoms or an unclear diagnosis[1]. Can we use an endoscopic treatment to remove lesions in the pancreatic body and tail directly and expedite recovery after treatment? In this study, we explored this question through animal experiments.
In the study, 15 healthy Bama miniature pigs weighing 15 kg to 20 kg were provided and raised by the Laboratory Animal Department of Shengjing Hospital of China Medical University. The experiment was conducted at the Endoscopy Center and the Experimental Animal Center of Shengjing Hospital of China Medical University. The experimental animals were divided into a control group with three pigs and two experimental groups with six pigs each. In the control group, the pancreases were only examined via endoscopy in the abdominal cavity. In experimental group one, the pancreatic tail was partially resected by the endoscopic operation, while in experimental group two, the pancreatic tail was completely removed by the endoscopic operation.
One pig in the control group, two pigs in experimental group one, and two pigs in experimental group two were sacrificed at 3 d, 7 d, and 14 d after the exploration or endoscopic resection.
Preoperative preparation and anesthesia: The pigs were fasted for 2 d prior to surgery and given a full-liquid diet consisting of approximately 10% of sugar in 3000 mL of water. The pigs were given an intramuscular injection of 0.3 mL/kg Shuangmianxin II (846 doses) preoperatively, with a venous needle retained. An intravenous injection of propofol (2 mg/kg) was administered to induce anesthesia and a single repeated dosing regimen was used to maintain anesthesia. A bedside monitor was used to track vital signs.
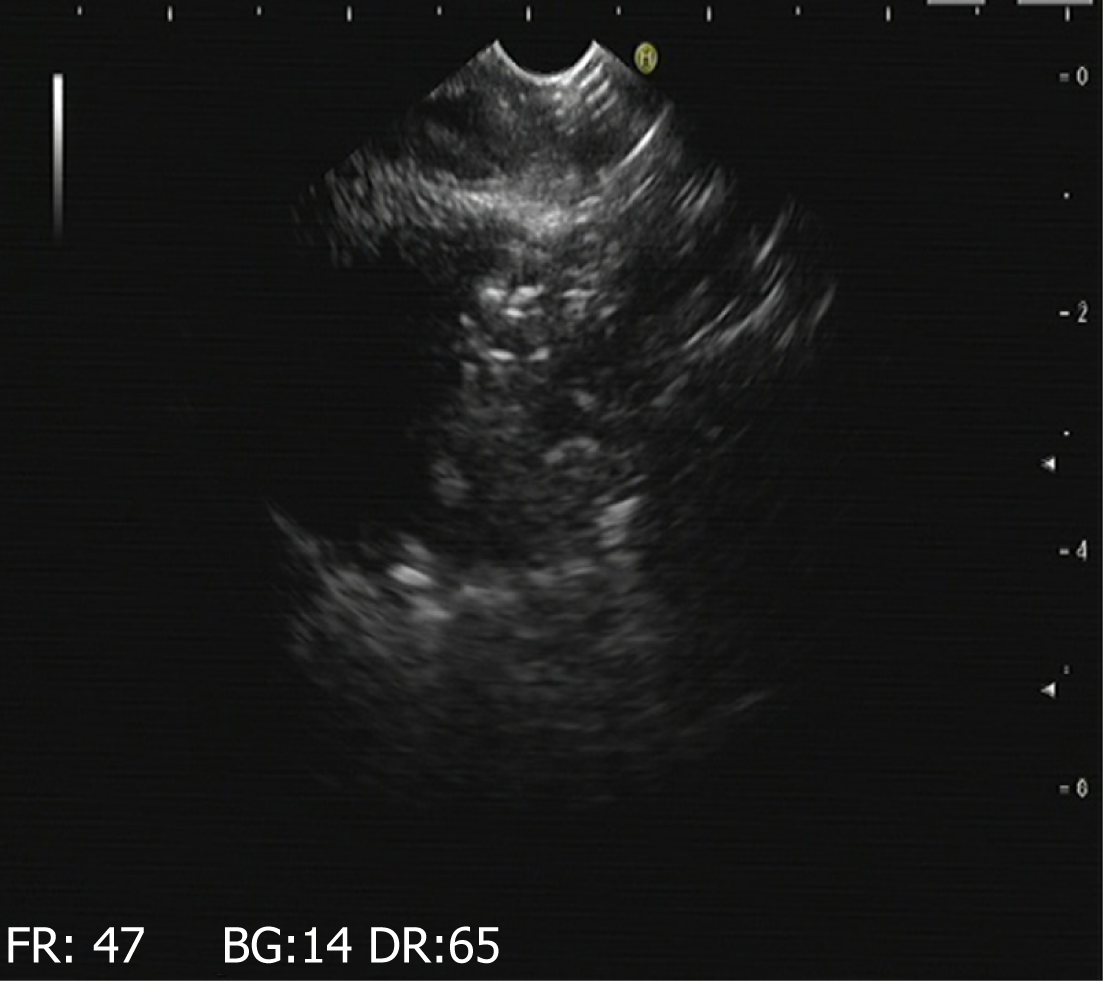
Endoscopic ultrasound to determine the site of intragastric incision: Because the pig's stomach cavity was large and the pancreas was relatively small, we used endoscopic ultrasound to locate the pancreas. First, we used a 19G puncture needle to reach the tail of the pancreas through the stomach wall. We injected approximately 1 mL of methylene blue liquid to facilitate the endoscope to determine the location of the pancreas in the abdominal cavity (Figure 1). During withdrawal of the puncture needle, we continued to inject methylene blue slowly to indwell the melanin-stained channel, especially when the puncture needle passed through the stomach wall. At that time, we injected 3 mL of methylene blue under the mucous membrane of the stomach wall to determine the appropriate incision position in the stomach.
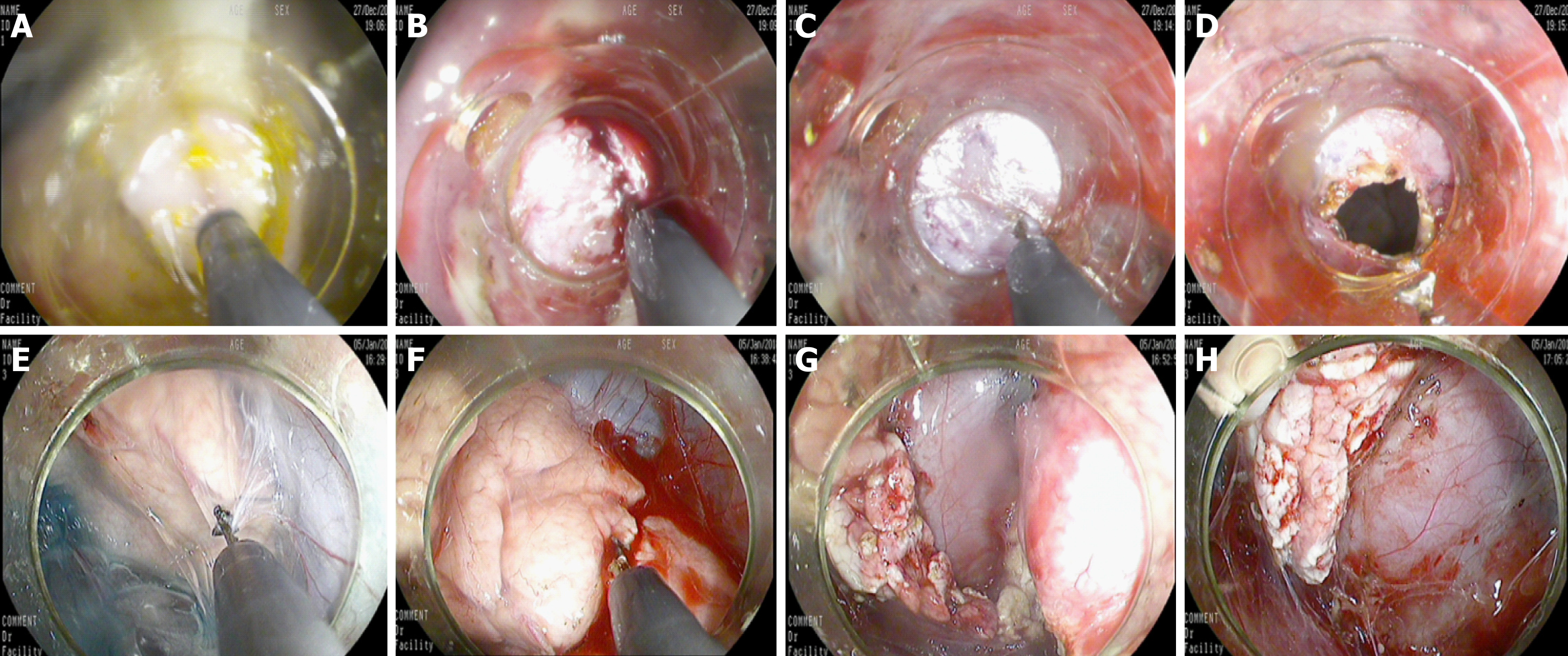
Endoscopic incision of the posterior wall of the stomach: The gastroscope with a transparent cap fixed on the front end was used to localize the marked point. The mucosal, submucosal, and muscular layers were sequentially cut with a triangular knife until the retroperitoneum was reached (Figure 2). A longitudinal or transverse incision could be selected. The incision size was approximately 1 cm to 1.5 cm, which was adequate for the endoscope to pass, could be closed easily, and avoided the stomach contents flowing into the abdominal cavity during the treatment.
Finding and isolating pancreatic tissue: The pancreas could be localized using methylene blue, and the pancreas could be rapidly identified after entering the peritoneum. Exploring in both directions distinguished the head and tail of the pancreas; generally, the tail was located to the right, close to the splenic tissue. The pancreatic membrane of the pancreatic body and tail was separated with a triangular knife to reveal approximately 2 cm of pancreatic parenchyma, avoiding damage to the pancreatic parenchyma.
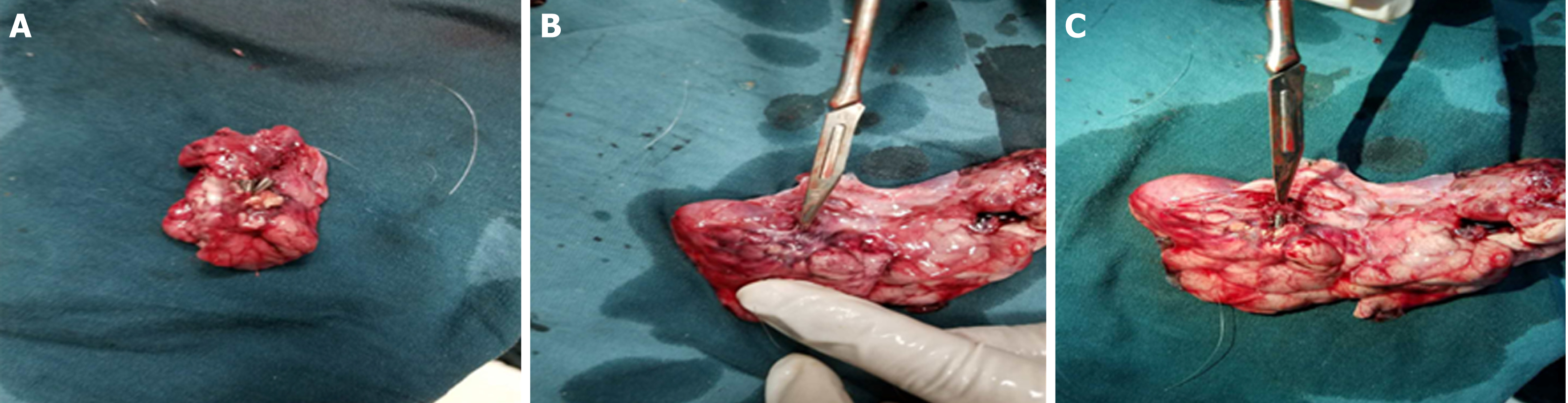
Resection of the pancreatic tail and closure of the wound: In experimental group one, approximately 2 cm of parenchyma of the pancreatic tail (approximately 1/3 of the width of the pancreas) was resected, avoiding the parenchyma around the main pancreatic duct. Hemostatic forceps were used to stop bleeding. In experimental group two, the entire parenchyma of the pancreatic tail was removed, and the parenchyma of the pancreas was transected with a triangular knife (Figure 2). Electrocoagulation was used for hemostasis. Finally, the pancreatic tissue was removed by a snare. The wound was clipped with one or more metal clips to ensure that the end was completely closed (Figure 3). Then, we withdraw the gastroscope into the stomach and observed whether there was bleeding at the gastric incision. After full hemostasis, the wound was sutured along the incised axis with metal clips. An inflation experiment was conducted to determine whether it was completely closed (Figure 3).
Postoperative management: After resuscitating from anesthesia, the experimental animal was sent to the animal department. The pig was fasted for 24 h, with the exception of water. Postoperative survival, signs of infection, eating conditions, weight changes, and complications were observed.
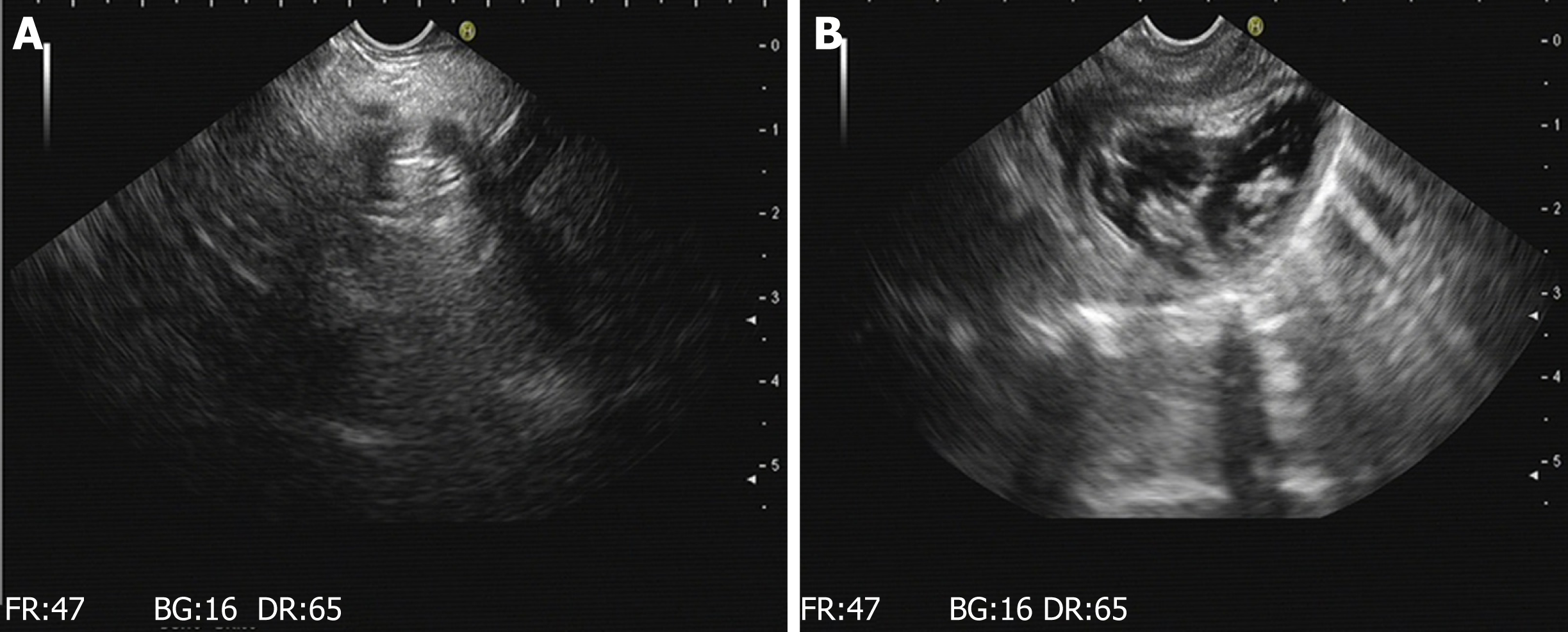
Endoscopic reexamination: According to the experimental design, endoscopy was performed to observe wound healing of the stomach and to determine whether there was any infection, perforation, or displacement of metal clips. Endoscopic ultrasound was performed to observe the remaining pancreatic tissue and to evaluate whether there was any effusion around the pancreatic wound, as well as to locate the metal clips in the wound.
Animal sacrifice and specimen collection: The animals were sacrificed by air embolism and then dissected. We first observed whether there was free gas, fluid, or other findings in the abdominal cavity. Then, the serosal side of the stomach wall of the incision, wound healing, and the residual metal clips of the gastric mucosal incisions, and whether there was exudate, leakage, effusion, or residual metal clips at the pancreatic wound were observed. After this, we removed the remaining pancreatic body and pancreatic head tissue, and retained the pancreatic wound tissue. The specimens were fixed with formalin for subsequent experiments.
Immunohistochemistry: The qualitative expression of transforming growth factor (TGF)-β1, Smad3, and Smad7 in the pancreas was predominantly localized in the cytoplasm. Semi-quantitative grading could be divided into three levels according to the degree of positive staining of the cells: Weak (+), medium (++), and strong (+++), corresponding to the light-stained brown, distinct brown, and deep-stained brown cytoplasm, respectively. The percentage of positive area was divided into negative (-, positive cells below 5%), weakly positive (+, positive cells among 5% to 30%), moderately positive (++, the total number of positive cells at 30% to 50%), and strongly positive (+++, more than 50% of positive cells). Each area of the section was randomly observed for five high magnification fields to evaluate the positive area.
SPSS 20.0 was used for statistical analyses. The data are expressed as the median, and the Kruskal-Wallis H test was used to compare the significant differences between the groups. P-values < 0.05 represent a statistically significant difference.
Fifteen animals were successfully operated under endoscopy. All animals in the two experimental groups were able to successfully undergo partial or total removal of the pancreatic tail through endoscopy. Postoperatively, the animals recovered well. Endoscopic reexamination, sacrificing, and specimen collection of all experimental animals were performed postoperatively based on the study design. In this experiment, a total of 12 pancreatic tissues were removed by endoscopy, in which the resection size of experimental group one was approximately 2.2 ± 0.8 cm and the resection size of experimental group two was approximately 4.5 ± 1.2 cm. All animals had five to six metal clips placed on the closed gastric wound. After the pancreas was removed in experimental group one, three to five metal clips were used to seal the pancreatic wound. After the pancreas was removed in experimental group two, five to seven metal clips were used to close the pancreatic wound.
The results of gastroscopy and endoscopic ultrasound at 3 d, 7 d (Figure 4), and 17 d after operation are shown in Table 1.
| 3 d after operation | 7 d after operation | 14 d after operation | |
| Results of endoscopic observation of the stomach | No shift of metal clips; local mucosa surrounding the wounds converged and the wounds healed well | Detachment of 1-3 metal clips; local mucosa surrounding the wounds converged and red scars on the wound surfaces could be seen | Detachment of all clips; ulcers formed locally and a small amount of white moss was found on the surface |
| Results of endoscopic ultrasound observation of the pancreas | |||
| Experimental group one | A small amount of hypoechoic effusion around the pancreatic wound; the metal clips at the wound site | The presentation was the same as that at 3 d after operation | The presentation was the same as that at 3 d after operation |
| Experimental group two | A small amount of hypoechoic effusion around the pancreatic wound; the metal clips at the wound site | There were visible effusions in the tail of the pancreas and its boundaries were clear; the metal clips at the wound site | There were visible effusion and accumulation; the metal clips at the wound site |
| Histological changes of pancreatic wound | |||
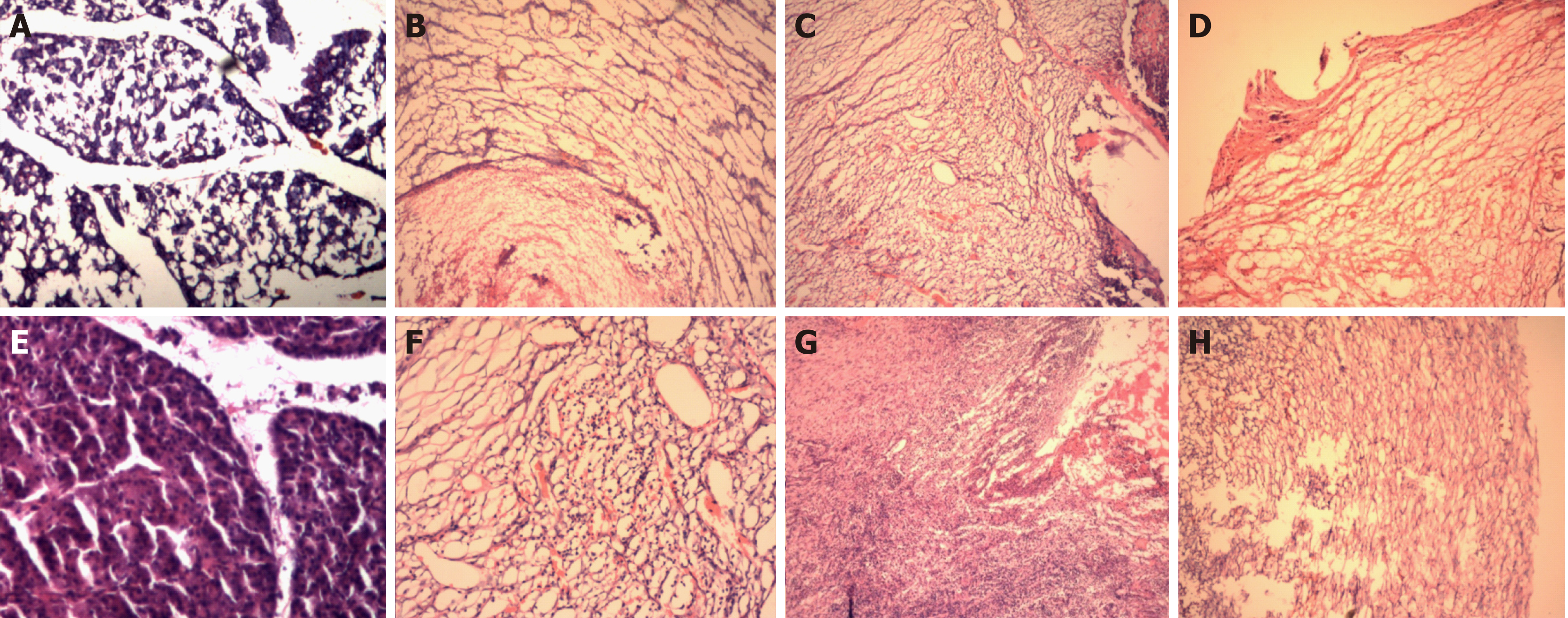
| Experimental group one | More inflammatory cells infiltrating in the interstitium, a small amount of fibroblasts, and a small amount of neovascularization can be seen | Numerous inflammatory cells infiltrating the interstitium, more fibroblasts, and more neovascularization can be seen | Fewer inflammatory cells infiltrating in the interstitium, numerous fibroblasts, and numerous areas of neovascularization |
| Experimental group two | Numerous inflammatory cells infiltrating the interstitium, fewer fibroblasts, and fewer neovascularization | Numerous inflammatory cells infiltrating in the interstitium, more fibroblasts, more neovascularization, and a small amount of necrotic tissue on the margin can be seen | Fewer inflammatory cells infiltrating the interstitium, more fibroblasts, and more neovascularization can be seen |
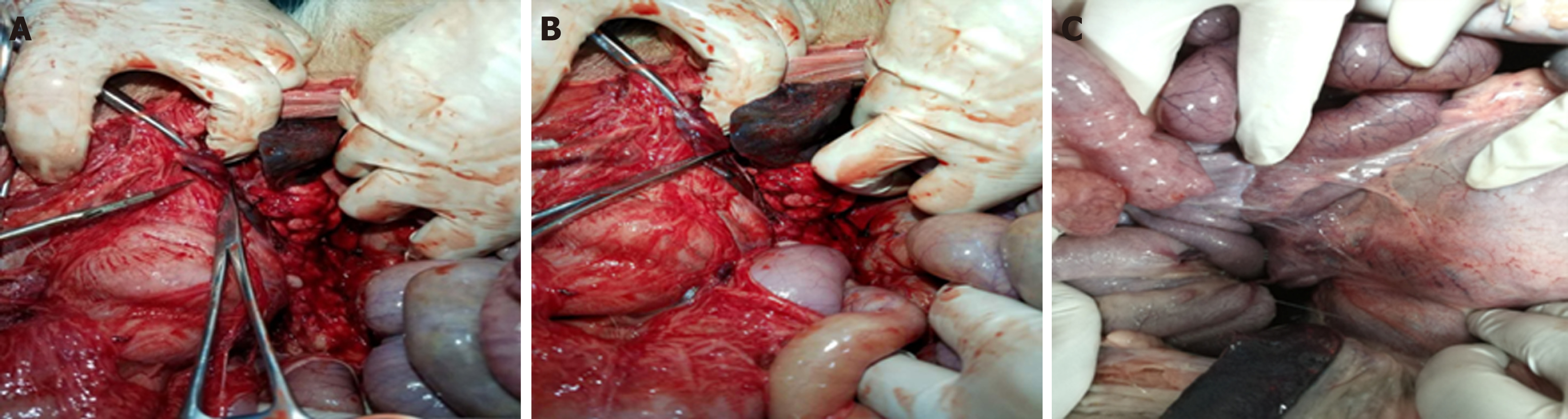
The experimental animals were sacrificed after endoscopic observation according to the study design. All animals had no free gas or free fluid after laparotomy. The serosal side of the posterior gastric wall was observed and there was local hyperemia. Adhesion bands were found between the gastric wall and the retroperitoneum. The band was thick in animals 3 d after surgery and the band was the thinnest in animals 14 d postoperatively (Figure 5). The anterior wall of the stomach was incised to look for gastric mucosal incisions, to observe wound healing and to evaluate for residual metal clips. Gastroscopes allow to observe at a magnification of 40 times. These observation results are thus more accurate than normal observation with the naked eye.
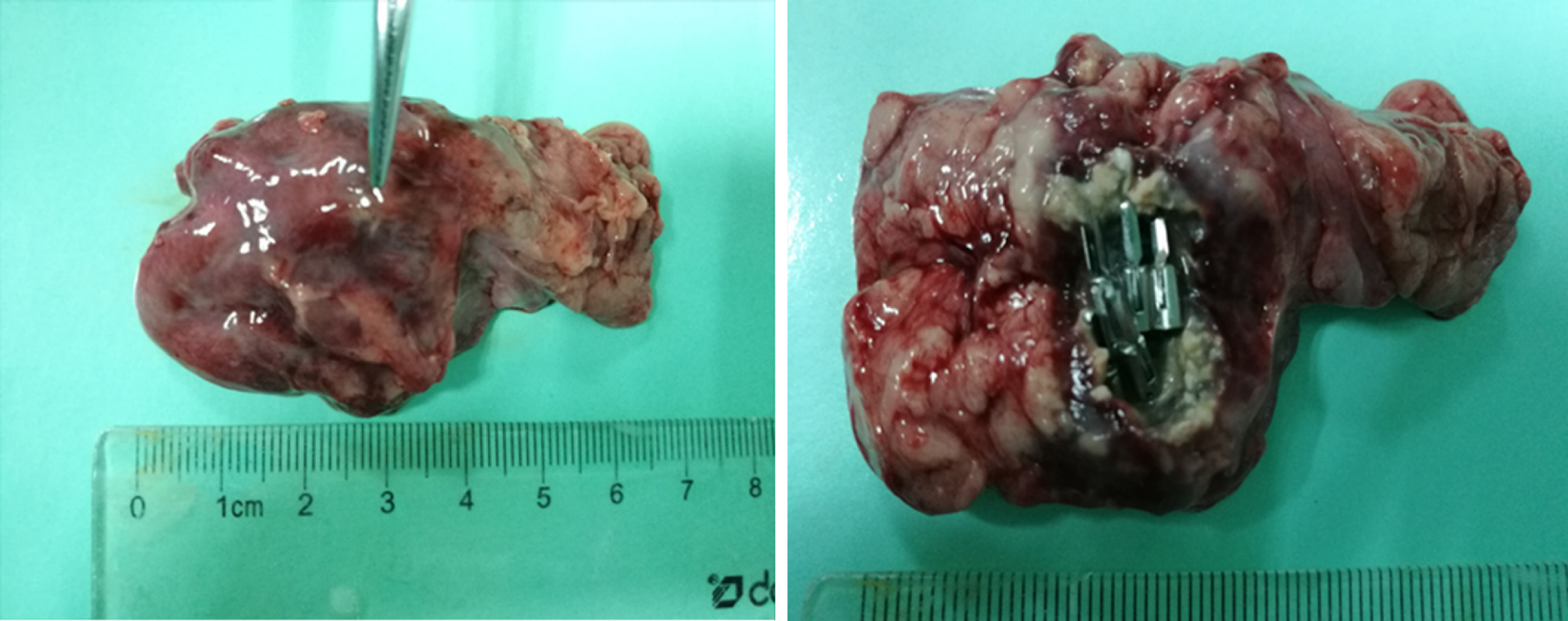
For the control group and experimental group one, no free liquid was seen in the wounds. There were no pancreatic leaks or fluid collections and all metal clips were present. The experimental animals sacrificed 14 d postoperatively had white scars surrounded by metal clips tightly attached to the pancreas seen with the naked eye. No definite scar was visible to the naked eye in animals sacrificed at 3 d and 7 d postoperatively, and the metal clips were visible outside the pancreas (Figure 6). In experimental group two, the local wounds of the animals sacrificed 3 d after surgery were the same as those of experimental group one. In the experimental animals observed at the 7th and 14th days postoperatively, the local swelling of the wound was apparent. The metal clips were covered with scar tissue and needed to be incised to expose the surface. There was a small amount of partially encapsulated liquid, which was a colorless and cool liquid totaling less than 5 mL (Figure 7). An amylase analysis was performed and the value exceeded 2000 U/L.
All experimental animals survived after surgery without severe infection or bleeding. In the control group and experimental group one, there were no complications such as pancreatic leakage or abdominal cavity and wound infections.
In experimental group two, the animals sacrificed 3 d after surgery had no complications. The animals sacrificed 7 d and 14 d postoperatively showed a liquid area with hyperechoic metal clips on endoscopic ultrasonography. However, there was no obvious fluid leakage after the laparotomy. Localized scar tissue wrapped in metal clips and a colorless liquid with congestive changes could be seen, suggesting a small amount of pancreatic fistula.
HE staining: HE staining of the pancreatic sections of the control group revealed normal pancreatic tissue. Microscopically, the pancreatic lobules were intact, the connective tissue between the lobules was diminished, the interstitium was loose, and no inflammatory cell infiltration was seen.
In experimental groups one and two, HE staining of the sections obtained at baseline from the pancreatic head revealed normal pancreatic tissue. The changes revealed by HE staining at 3 d, 7 d and 14 d after operation in experimental groups one and two are shown in Figure 8 and Table 1.
Expression of TGF-β1, Smad3, and Smad7 in pancreatic tissue of the control group and pancreatic wounds: As observed by immunohistochemical staining, the expression of these factors in the pancreatic tissue of the wounds varied over time. The details are shown in Figure 9.
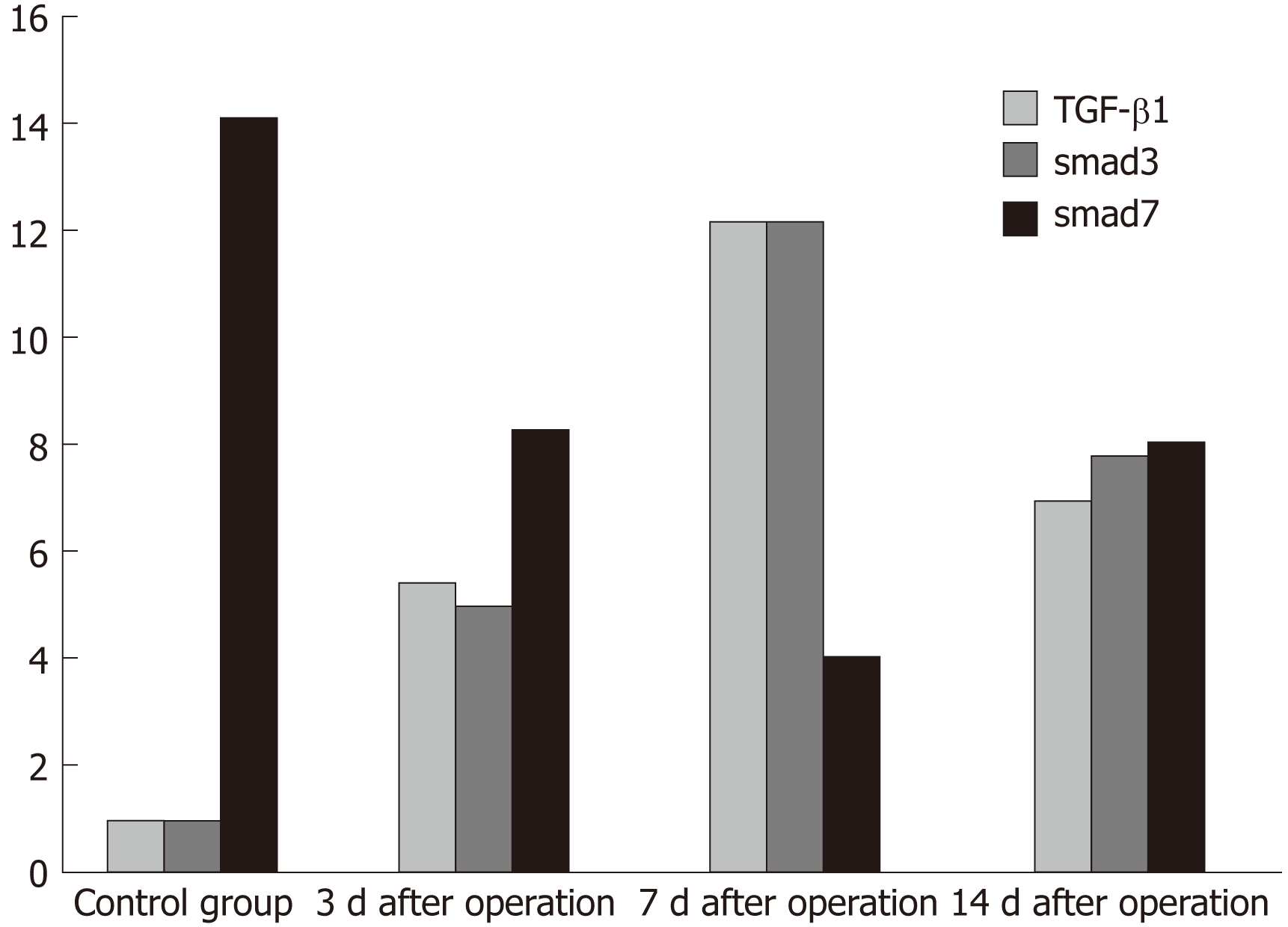
We processed the results of immunohistochemistry and scored the degree of cell positive staining: Negative for 1 point, weakly positive for 2 points, moderately positive for 3 points, and strongly positive for 4 points. We also scored the positive area percentages: Negative for 1 point, weakly positive for 2 points, moderately positive for 3 points, and strongly positive for 4 points. The total score was calculated as the score of positive staining multiplied by the score of percentage of positive area. The scores for specific expression of TGF-β1, Smad3, and Smad7 at different time points are shown in Figure 10.
At present, the common problems of NOTES predominantly involve the following aspects: First, it is essential to select an approach with an excellent visual field suitable for the procedure and to allow for appropriate wound closure[2-6]. This experiment confirmed that the metal clip is safe and effective in the closure of the gastric wound. Second, to avoid intra-abdominal infection, we should minimize the operative time and reduce any factors that can introduce bacteria into the abdominal cavity[7]. For the NOTES of the pancreas, we chose the upper gastrointestinal approach, as the stomach is a relatively clean area of the digestive tract that can be controlled. We controlled the animal's digestive tract by using a 2 d fluid/food method to ensure cleanliness of the digestive tract preoperatively. We used a small incision ensuring that only the endoscope can pass in order to avoid the stomach contents flowing into the abdominal cavity. Third, the method of identifying intra-abdominal anatomy is difficult in NOTES due to insufficient experience[8]. In our study, methylene blue-guided endoscopic ultrasound guidance with fine needle aspiration (EUS-FNA) was used, which can help us to quickly and accurately identify the target organ. The function of EUS-FNA has been fully used[9-13]. Finally, some NOTES procedures are relatively complicated. Existing endoscopes and auxiliary instruments may not be able to complete the procedure in some medical centers[14]. In this study, we used existing devices to minimize procedural difficulty and to achieve effective resection.
The predominant complication following pancreatic resection is pancreatic leakage. The incidence of pancreatic fistula after surgery averages approximately 5% to 10%. Pancreatic leakage may cause many other serious complications and may eventually lead to multiple organ failure and death[15]. Pancreatic leakage predominantly consists of the leakage of pancreatic fluid from the ruptured pancreatic duct or from sutures to the abdominal cavity. A small amount of pancreatic leakage enclosed by tissue can form a pseudocyst and secondary infection can result in the formation of abscesses.
We observed that when a small amount of pancreatic tissue was removed and there was no damage to the main pancreatic duct, we could close the wound with metal clips without leakage. For the pancreatic tail excision, there was injury to the main pancreatic duct. There was no evidence of pancreatic leakage initially, but this complication occurred one week after surgery and continued for two weeks postoperatively. In view of this finding, we believe that the metal clip can close the pancreatic parenchyma without damaging the main pancreatic duct and can play a role in closing the damaged branch of the pancreatic duct. If there is damage to the main pancreatic duct, a small amount of pancreatic leakage may occur. This can be explained as follows: First, it may be due to necrosis of the edge of the pancreatic wound after electrocoagulation, leading to the metal clip no longer remaining firmly affixed. Second, the necrotic pancreatic tissue will release digestive enzymes and relaxation of the metal clip will expose more pancreatic tissue and cause more exudation. However, due to the protection of the metal clip in the initial stage, pancreatic leakage could be encapsulated within a week, not leading to pancreatic ascites, infection, etc. This illustrates the efficacy and relative safety of endoscopic resection of the pancreas. More studies are needed to verify the efficacy of this treatment modality and to further examine complications. When a fluid collection does develop, we cannot do abdominal drainage like surgery. Further studies should evaluate whether the fluid can be absorbed and whether it can be controlled with a pancreatic stent preoperatively. To evaluate the possibility of promoting the healing of wounds, we conducted a follow-up basic experimental study.
We can observe obvious scar repair with the naked eye on the experimental specimens. HE staining also confirmed this result. We next focused on the scar formation process. TGF-β1 is the most commonly used index in the wound repair process, which involves various processes including cell proliferation, differentiation, and apoptosis. This factor plays an important regulatory role in the process of inflammation and fibrosis[16-18]. It has been reported that TGF-β1 also plays an important role in the repair of pancreatic tissue and can participate in many aspects such as wound healing, cell growth, cell cycle regulation, angiogenesis, and immune regulation[19-21]. Studies have shown that the expression of TGF-β1 in wound scar cells is significantly increased compared with normal fibroblasts. In addition, multiple factors including smad3 are also increased[12], suggesting that TGF-β1/Smad signaling plays an important role in the repair of scar tissue. However, the role of TGF-β1 is complex and its role is dose-dependent, cell cycle-dependent, and cell type-dependent, while the synergy between different cytokines impacts the biological effects of TGF-β1. We selected TGF-β1 at different time points for observation. We compared it in the wound repair process and we also examined its role in signal transduction pathways.
How does TGF-β1 impact signal transduction and how does it regulate tissue repair? Previous studies have suggested that the key mediator of transduction of this signal from outside the cell into the nucleus is the Smad protein family, which acts as a signal downstream from protein molecules of TGF-β1 and plays varied roles in the repair process[22-24]. We detected TGF-β1 and Smad3/Smad7 at different time points by immunohistochemistry and found that while TGF-β1 was not normally expressed in normal pancreatic tissues, its expression began in the pancreatic wound, which demonstrates that it participates in wound repair. As time following surgery increases, the expression of TGF-β1 increases, reducing the inflammatory response of the tissue while also promoting the formation of scar tissue. At this later time, we can visualize the inflammatory cells and fibroblasts in the tissue sections stained with HE.
In the final stage of the experiment, the body no longer needs to repair the wound due to the formation of scar tissue, so TGF-β1 begins to decline. As a downstream reaction to proteins of TGF-β1, Smad3 and Smad7 also have various changes. We observed that the change of Smad3 paralleled that of TGF-β1. It was shown that during the process of tissue repair, the increase of TGF-β1 promoted the expression of Smad3. Although the changing trend was the same, the time of peak value was slightly different and Smad3 expression lagged slightly behind TGF-β1. The Smad7 highly expressed in normal pancreatic tissues declined with the progression of the study inversely to the changes of TGF-β1, particularly when the pancreas tissues were repaired. After the formation of scar tissue, Smad7 expression began to rise to regulate the expression of TGF-β1 so that excessive tissue repair is avoided. Therefore, the expression of TGF-β1 in pancreatic wounds paralleled that of Smad3 and is inversely correlated with the expression of Smad7, suggesting that TGF-β1 may contribute to fibrosis of pancreatic wounds through the TGF-β1/Smad signaling pathway. Smad3 may play a role in the signal transduction of TGF-β1, while Smad7 may antagonize receptor-activated Smad protein-mediated signaling, forming a negative feedback loop. The interaction of multiple factors achieves wound healing without excessive fibrosis. The mechanisms need be further studied.
The transgastric endoscopic route is a safe, effective, and feasible method for pancreatic tail resection in pigs. Partial resection of the pancreatic tail is very safe, while total pancreatic tail resection has a high incidence of pancreatic leakage. Metal clips are effective for closure of the pancreatic parenchymal wound and can create optimal conditions for scar formation of pancreatic wounds while reducing pancreatic leakage. The changes of TGF-β1 and Smad3/Smad7 expression in different stages of pancreatic wound healing indicate that TGF-β1 participates in the occurrence and development of fibrosis of pancreatic wound healing through the TGF-β1/Smad signaling pathway.
The resection of the pancreatic body and tail generally requires invasive surgical treatment. Laparoscopic resection of the pancreatic body and tail is the predominant methodology to remove lesions in these locations; its safety and surgical planning are relatively mature, but it remains a complex and high-precision surgical operation, requiring abundant experience and skills in laparoscopic surgery, with a 10% rate of complications. Natural orifice transluminal endoscopic surgery has attracted increasing attention as a new technique for minimally invasive treatment. Compared with traditional surgery, it has the advantages of less trauma, faster recovery, and no external postoperative scar. Can we use an endoscopic treatment to remove lesions in the pancreatic body and tail directly and expedite recovery after treatment? In this study, we explored this question through animal experiments.
Based on this study, we may provide a micro-invasive therapy for the lesions in the pancreatic tail.
To verify the feasibility and safety, as well as to examine the complications of endoscopic pancreatectomy and healing mechanisms of pancreatic wounds after endoscopic resection.
Transgastric endoscopic resections of varying sizes of pancreases were performed in 15 healthy Bama miniature pigs. The technical success rate, the incidence of serious complications, and the survival of the animals were studied. The healing of the wounds was evaluated by sacrificing the animals at various time points. Finally, the expression of transforming growth factor-β1 and Smad3/Smad7 in the surgical site was examined by immunohistochemistry to explore the role of these factors in wound healing of the pancreas.
Partial and total resections were successfully performed in two groups of animals, respectively. The technical success rate and the survival rate of the pigs were both 100%. We obtained 12 pancreatic tissue samples by endoscopic resection. The pancreatic wounds were closed with metal clips in one group and the wounds healed well by forming scars. There was a small amount of pancreatic leakage in the other group, but it can be fully encapsulated. The level of transforming growth factor-β1 (TGF-β1) in the wounds increased during the inflammatory and fibrous hyperplasia phases, and decreased in the scar phase. The expression of Smad3 paralleled that of TGF-β1, while the expression of Smad7 had an inverse relationship with the expression of TGF-β1.
Purely transgastric endoscopic resection of the pancreas is a safe, effective, and feasible procedure, but the incidence of pancreatic leakage in total pancreatic tail resection was high. The expression of TGF-β1 and Smad3/Samd7 is related to the progression of pancreatic wound healing.
Further studies should be conducted to confirm the safety and effectiveness of endoscopic resection for lesions in the pancreatic tail and wound healing mechanism.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Lee MW, Otto G, Sharma M S-Editor: Ma RY L-Editor: Wang TQ E-Editor: Ma YJ
| 1. | Merkow J, Paniccia A, Edil BH. Laparoscopic pancreaticoduodenectomy: a descriptive and comparative review. Chin J Cancer Res. 2015;27:368-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 2. | Sumiyama K, Gostout CJ, Rajan E, Bakken TA, Knipschield MA, Chung S, Cotton PB, Hawes RH, Kalloo AN, Kantsevoy SV, Pasricha PJ. Transgastric cholecystectomy: transgastric accessibility to the gallbladder improved with the SEMF method and a novel multibending therapeutic endoscope. Gastrointest Endosc. 2007;65:1028-1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Wilhelm D, Meining A, von Delius S, Fiolka A, Can S, Hann von Weyhern C, Schneider A, Feussner H. An innovative, safe and sterile sigmoid access (ISSA) for NOTES. Endoscopy. 2007;39:401-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 4. | Ryou M, Fong DG, Pai RD, Sauer J, Thompson CC. Evaluation of a novel access and closure device for NOTES applications: a transcolonic survival study in the porcine model (with video). Gastrointest Endosc. 2008;67:964-969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Bachman SL, Sporn E, Furrer JL, Astudillo JA, Calaluce R, McIntosh MA, Miedema BW, Thaler K. Colonic sterilization for natural orifice translumenal endoscopic surgery (NOTES) procedures: a comparison of two decontamination protocols. Surg Endosc. 2009;23:1854-1859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Perretta S, Dallemagne B, Barry B, Marescaux J. The ANUBISCOPE® flexible platform ready for prime time: description of the first clinical case. Surg Endosc. 2013;27:2630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Wang Z, Phee SJ, Lomanto D, Goel R, Rebala P, Sun ZL, Trasti S, Reddy N, Wong JY, Ho KY. Endoscopic submucosal dissection of gastric lesions by using a master and slave transluminal endoscopic robot: an animal survival study. Endoscopy. 2012;44:690-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 8. | ASGE Technology Committee. Kantsevoy SV, Adler DG, Chand B, Conway JD, Diehl DL, Kwon RS, Mamula P, Rodriguez SA, Shah RJ, Song LM, Tierney WM. Natural orifice translumenal endoscopic surgery. Gastrointest Endosc. 2008;68:617-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Okasha HH, Naguib M, El Nady M, Ezzat R, Al-Gemeie E, Al-Nabawy W, Aref W, Abdel-Moaty A, Essam K, Hamdy A. Role of endoscopic ultrasound and endoscopic-ultrasound-guided fine-needle aspiration in endoscopic biopsy negative gastrointestinal lesions. Endosc Ultrasound. 2017;6:156-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 10. | Hussain I, Ang TL. Cystic pancreatic lymphangioma diagnosed with endoscopic ultrasound-guided fine needle aspiration. Endosc Ultrasound. 2017;6:136-139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 11. | Oh D, Seo DW, Hong SM, Song TJ, Park DH, Lee SS, Lee SK, Kim MH. Endoscopic ultrasound-guided fine-needle aspiration can target right liver mass. Endosc Ultrasound. 2017;6:109-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 12. | De Moura DTH, Moura EGH, Bernardo WM, De Moura ETH, Baraca FI, Kondo A, Matuguma SE, Almeida Artifon EL. Endoscopic retrograde cholangiopancreatography versus endoscopic ultrasound for tissue diagnosis of malignant biliary stricture: Systematic review and meta-analysis. Endosc Ultrasound. 2018;7:10-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 90] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 13. | Lariño-Noia J, Iglesias-Garcia J, de la Iglesia-Garcia D, Dominguez-Muñoz JE. EUS-FNA in cystic pancreatic lesions: Where are we now and where are we headed in the future? Endosc Ultrasound. 2018;7:102-109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 14. | Patel N, Seneci C, Yang GZ, Darzi A, Teare J. Flexible platforms for natural orifice transluminal and endoluminal surgery. Endosc Int Open. 2014;2:E117-E123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Pratt WB, Maithel SK, Vanounou T, Huang ZS, Callery MP, Vollmer CM. Clinical and economic validation of the International Study Group of Pancreatic Fistula (ISGPF) classification scheme. Ann Surg. 2007;245:443-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 293] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 16. | Chen S, Feng T, Vujić Spasić M, Altamura S, Breitkopf-Heinlein K, Altenöder J, Weiss TS, Dooley S, Muckenthaler MU. Transforming Growth Factor β1 (TGF-β1) Activates Hepcidin mRNA Expression in Hepatocytes. J Biol Chem. 2016;291:13160-13174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 17. | Li Z, Zhang L, He W, Zhu C, Yang J, Sheng M. Astragalus membranaceus inhibits peritoneal fibrosis via monocyte chemoattractant protein (MCP)-1 and the transforming growth factor-β1 (TGF-β1) pathway in rats submitted to peritoneal dialysis. Int J Mol Sci. 2014;15:12959-12971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Liu X, Hubchak SC, Browne JA, Schnaper HW. Epidermal growth factor inhibits transforming growth factor-β-induced fibrogenic differentiation marker expression through ERK activation. Cell Signal. 2014;26:2276-2283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Wang W, Vootukuri S, Meyer A, Ahamed J, Coller BS. Association between shear stress and platelet-derived transforming growth factor-β1 release and activation in animal models of aortic valve stenosis. Arterioscler Thromb Vasc Biol. 2014;34:1924-1932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 20. | Massuto DA, Kneese EC, Johnson GA, Burghardt RC, Hooper RN, Ing NH, Jaeger LA. Transforming growth factor beta (TGFB) signaling is activated during porcine implantation: proposed role for latency-associated peptide interactions with integrins at the conceptus-maternal interface. Reproduction. 2010;139:465-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 21. | Wu W, Jiang XY, Zhang QL, Mo Y, Sun LZ, Chen SM. Expression and significance of TGF-beta1/Smad signaling pathway in children with IgA nephropathy. World J Pediatr. 2009;5:211-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Zhao J, Wang L, Cao AL, Jiang MQ, Chen X, Wang Y, Wang YM, Wang H, Zhang XM, Peng W. HuangQi Decoction Ameliorates Renal Fibrosis via TGF-β/Smad Signaling Pathway In Vivo and In Vitro. Cell Physiol Biochem. 2016;38:1761-1774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Yang F, Chung AC, Huang XR, Lan HY. Angiotensin II induces connective tissue growth factor and collagen I expression via transforming growth factor-beta-dependent and -independent Smad pathways: the role of Smad3. Hypertension. 2009;54:877-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 219] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 24. | Tang Y, Wu X, Lei W, Pang L, Wan C, Shi Z, Zhao L, Nagy TR, Peng X, Hu J, Feng X, Van Hul W, Wan M, Cao X. TGF-beta1-induced migration of bone mesenchymal stem cells couples bone resorption with formation. Nat Med. 2009;15:757-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 976] [Cited by in RCA: 891] [Article Influence: 55.7] [Reference Citation Analysis (0)] |