Published online Oct 7, 2016. doi: 10.3748/wjg.v22.i37.8439
Peer-review started: June 16, 2016
First decision: July 29, 2016
Revised: August 16, 2016
Accepted: August 19, 2016
Article in press: August 19, 2016
Published online: October 7, 2016
Processing time: 110 Days and 6 Hours
Schwannomas are mesenchymal tumors originating from Schwann cells in peripheral nerve sheaths. Although the tumor can be located in any part of the human body, the most common locations are the head, neck, trunk and extremities. Pancreatic schwannomas are rare. To our knowledge, only 64 cases of pancreatic schwannoma have been reported in the English literature over the past 40 years. In this paper, we present a pancreatic schwannoma in a 59-year-old female. Ultrasound, computed tomography and magnetic resonance imaging revealed the tumor located in the pancreatic body; however, accurate diagnosis was hard to obtain preoperatively and a pancreatic cystadenoma was preliminarily considered. During laparotomy, the mass was found in the body of the pancreas. An enlarged gallbladder with multiple stones was also observed. We performed central pancreatectomy, end-to-side pancreaticojejunostomy and cholecystectomy. Notably, central pancreatectomy has been reported in only one case prior to this report. The gross specimen showed a mass with a thin capsule, 1.6 cm × 1.1 cm × 1.1 cm in size. Microscopic examination showed that the tumor was mainly composed of spindle-shaped cells with palisading arrangement and no atypia, which is consistent with a benign tumor. Both hypercellular and hypocellular areas were visible. Immunohistochemical staining revealed strongly positive results for protein S-100. Finally, the tumor was diagnosed as a schwannoma of the pancreatic body. Postoperatively, the patient recovered well and left the hospital 6 d later. During the 53-mo follow-up period, the patient remained well and free of complications.
Core tip: Over the past 40 years, only 64 cases of pancreatic schwannomas have been reported in the English literature. It is a considerable challenge to obtain a precise preoperative diagnosis, despite the application of multiple imaging modalities. We present a patient with a pancreatic schwannoma and enlarged gallbladder with multiple stones. After central pancreatectomy, end-to-side pancreaticojejunostomy and cholecystectomy, the patient recovered quickly and had a good prognosis. In this study, we focused on the diagnosis and treatment of a pancreatic schwannoma and conducted a literature review to deepen the understanding of the subject.
- Citation: Xu SY, Sun K, Owusu-Ansah KG, Xie HY, Zhou L, Zheng SS, Wang WL. Central pancreatectomy for pancreatic schwannoma: A case report and literature review. World J Gastroenterol 2016; 22(37): 8439-8446
- URL: https://www.wjgnet.com/1007-9327/full/v22/i37/8439.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i37.8439
Schwannomas are tumors originating from the Schwann cells of peripheral nerve sheaths[1]. Most schwannomas show either monosomy 22 or loss of 22q material; however, the pathogenesis of the tumor remains unclear[2]. Schwannomas are generally encapsulated tumors, and more than 90% of them are benign[2]. Schwannomas occur in patients with no obvious gender difference and at all ages. However, patients between 20 and 50 years of age were most frequently reported. Surgery is the most common treatment for schwannomas, and patients usually have a good postoperative prognosis[3]. While almost every part of the human body can be involved, the most common locations are the head, neck, trunk and extremities[3]. Pancreatic schwannomas are extremely rare. To our knowledge, only 64 cases of pancreatic schwannomas have been reported in the English literature over the past 40 years[4-61]. Although most patients with pancreatic schwannomas were symptomatic, a considerable number of patients were asymptomatic with tumors that were found incidentally. This paper presents a case of pancreatic schwannoma in a 59-year-old female and a review of the literature.
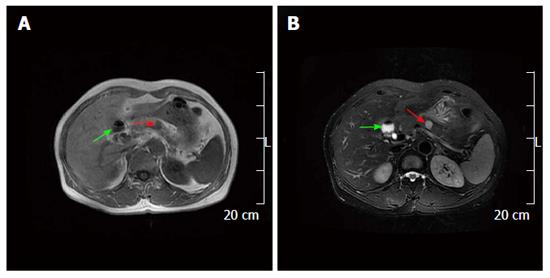
On January 7, 2012, a 59-year-old female was referred to our hospital because of a pancreatic mass found during a routine health examination. The abdomen was soft and nondistended without evidence of a palpable mass. Her family history was not significant. Abnormal laboratory results included: Unconjugated bilirubin, 2 μmol/L (normal, 3-14) and serum kalium, 3.42 mmol/L (normal, 3.5-5.2). Ultrasound revealed a 1.4 cm × 1.3 cm, well-defined cystic lesion in the pancreatic body (Figure 1A), as well as a 6.8 cm sized strong echo in the gallbladder (Figure 1B). An unenhanced computed tomography (CT) scan showed a 1.6 cm × 1.1 cm well-defined hypodense mass in the pancreatic body (Figure 2A). On the contrast-enhanced CT, the mass was not enhanced (Figure 2B). On magnetic resonance imaging (MRI), the mass in the pancreatic body and gallbladder appeared hypointense on T1 weighted images (Figure 3A). The mass in the pancreatic body appeared inhomogeneously hyperintense and the enlarged gallbladder appeared hyperintense on T2 weighted images (Figure 3B). We also performed endoscopic ultrasound-guided fine needle aspiration (EUS-FNA). However, the tumor sample was difficult to acquire, and the procedure was unsuccessful. According to these results, a pancreatic cystadenoma and an enlarged gallbladder with multiple stones were preliminarily considered.
After sufficient preparation and obtaining consent from the patient and her family members, a laparotomy was performed. A 1.5 × 1.0 cm mass surrounded by a thin fibrous capsule was found in the pancreatic body. An enlarged gallbladder with multiple stones was also found. We performed central pancreatectomy, end-to-side pancreaticojejunostomy, cholecystectomy and inserted a pancreatic stent. Intraoperative frozen pathology revealed a schwannoma in the pancreatic body.
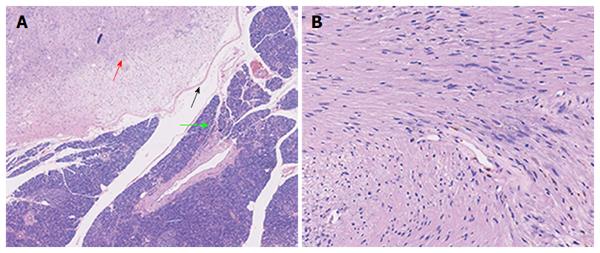
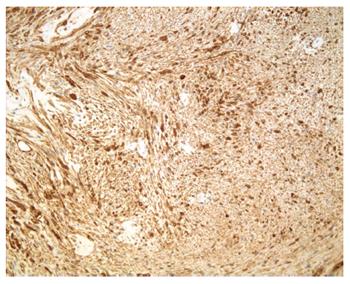
Macroscopically, the mass in the pancreatic body measured 1.6 cm × 1.1 cm × 1.1 cm. Microscopically, the tumor, surrounded by a thin capsule, was mainly composed of spindle-shaped cells with palisading arrangement and no atypia, which was consistent with a benign schwannoma. Both hypercellular and hypocellular areas were visible (Figure 4). Immunohistochemical staining was strongly positive for protein S-100 (Figure 5) and negative for SMA, CD34 and CD117. The final diagnosis was a schwannoma of the pancreatic body. Postoperatively, the patient recovered well and left the hospital 6 d later with no evidence of postoperative pancreatic fistula. During the follow-up period of 53 mo, the patient remained well without any complications.
Schwannomas are neoplasms that originate from the Schwann cells of nerve sheaths[62]. More than 90% of schwannomas are benign and manifest in approximately 5% of cases as benign soft-tissue neoplasms[63]. Schwannomas can occur in patients of any age with no obvious gender difference. However, they are most commonly found in patients aged between 20 years and 50 years. Although nearly any part of the human body can be involved, the head, neck and extremities are the most common sites[64]. In the abdominal cavity, the retroperitoneum (6% of primary retroperitoneal tumors)[65] and stomach[66] are the most common sites involved. Schwannomas of the pancreas are rare. Table 1 summarizes the important available clinicopathological characteristics of the 64 cases reported in the English literature over the past 40 years[4-61] and the case presented in our study. Continuous variables were summarized as the mean ± SD and range. The Student’s t test was used for comparisons of continuous variables. Statistical analyses were conducted using SPSS version 20.0 for Windows (SPSS Inc., Chicago, IL, United States). All tests for significance were two-sided, and P values < 0.05 were considered statistically significant.
| n (%) or mean ± SD (range) | |
| Age (yr) (n = 64) | |
| Mean | 55.22 ± 15.26 (20-87) |
| Sex (male/female), (male %) (n = 64) | 29/35 (45.31) |
| Symptoms1 (n = 64) | |
| Asymptomatic | 24 (37.50) |
| Symptomatic | |
| Abdominal pain | 28 (43.75) |
| Weight loss | 8 (12.50) |
| Back pain | 4 (6.26) |
| Nausea/vomiting | 3 (4.69) |
| Anorexia | 2 (3.13) |
| Abdominal mass | 1 (1.56) |
| Anemia | 2 (3.13) |
| Melena | 2 (3.13) |
| Jaundice | 2 (3.13) |
| Abdominal discomfort | 1 (1.56) |
| Location (n = 65) | |
| Head | 26 (40.00) |
| Head/body | 3 (4.62) |
| Body | 15 (23.08) |
| Body/tail | 7 (10.78) |
| Tail | 7 (10.78) |
| Uncinate process | 7 (10.78) |
| Mean size (cm) | 5.83 ± 4.59 (1-20) |
| Benign | 56 (5.27 ± 3.95 ) (1-20) |
| Malignant | 4 (13.75 ± 6.24) (7-20) |
| Operation (n = 65) | |
| PD2 | 20 (30.77) |
| PPPD | 2 (3.08) |
| DP3 | 16 (24.62) |
| Enucleation | 9 (13.85) |
| Central pancreatectomy | 2 (3.08) |
| Unresectable | 2 (3.08) |
| Refused | 1 (1.54) |
| Not specified | 13 (20.00) |
| Histology (n = 65) | |
| Malignant | 5 (7.69) |
| Benign | 59 (90.77) |
| Not specified | 1 (1.54) |
| Nature of tumor (n = 65) | |
| Solid | 17 (26.15) |
| Cystic | 28 (43.08) |
| Solid and cystic | 14 (21.54) |
| Not specified | 6 (9.23) |
| Mean follow-up months (n = 29) | 20.59 ± 17.76 (3-66) |
| Died of disease | 0 |
Precise preoperative diagnosis of a pancreatic schwannoma is challenging because the clinical symptoms and radiological characteristics of schwannomas are nonspecific. Definitive diagnosis can be achieved only based on the combined results of histopathological and immunohistochemical examinations of surgical specimens. Microscopically, pancreatic schwannomas are encapsulated tumors that consist of hypercellular (Antoni type A area) and hypocellular areas (Antoni type B area) with varying amounts of these histological components[22]. The hypercellular area consists of closely packed spindle cells with occasional nuclear palisading as well as Verocay bodies. The hypocellular area is composed of loosely arranged tumor cells and abundant myxoid stroma[6]. Occasionally these may become cystic, hemorrhagic and calcified[18]. More than 90% of pancreatic schwannomas are benign. However, malignant pancreatic schwannomas have been reported in 5 cases (7.69%)[22,57,59-61]. Immunohistochemically, schwannomas show strongly positive staining for S-100 and negative staining for desmin, smooth muscle myosin, SMA, CD34 and CD117[16,67].
Accurate diagnosis of a pancreatic schwannoma prior to operation is nearly impossible. US, CT and MRI can be performed to establish a probable diagnosis. A pancreatic schwannoma usually appears as a well-defined hypodense lesion on US and shows no echoic enhancement with color Doppler. On unenhanced CT scan, schwannomas are usually well-defined hypodense lesions with encapsulation and/or cystic degeneration. Schwannomas with high Antoni A areas show high density and have a heterogeneous appearance due to high cellularity and increased lipid content. Antoni B areas of schwannomas appear cystic and show low density due to loose stroma and low cellularity[10]. On contrast-enhanced CT, the Antoni A areas are usually enhanced, whereas the Antoni B areas are unenhanced[7]. On MRI, a typical schwannoma appears hypointense in T1-weighted images and appears inhomogeneously hyperintense in T2-weighted images[16]. EUS-FNA may greatly contribute to precise preoperative diagnosis. In a case reported by Li et al[28], a pancreatic schwannoma was accurately diagnosed preoperatively by EUS-FNA. In the present study, we also attempted EUS-FNA. However, a sample of the tumor was difficult to acquire because of the small tumor size and the procedure was unsuccessful.
Surgery is the optimal treatment for pancreatic schwannoma. As tumors could be located in different parts of the pancreas, surgical methods vary accordingly. In the present case, laparotomy permitted discovery of the mass in the body of the pancreas. An enlarged gallbladder with multiple stones was also found. We performed central pancreatectomy, end-to-side pancreaticojejunostomy, cholecystectomy and inserted a pancreatic stent. To date, central pancreatectomy had been reported in only one case[5] prior to this study. Compared with traditional distal pancreatectomy and splenectomy for tumors in the body or tail of the pancreas, central pancreatectomy can not only completely resect the tumor, but also preserve the distal pancreas and spleen, which is beneficial to patients. Following complete tumor excision, patients with pancreatic schwannomas generally have a good prognosis.
In conclusion, a schwannoma of the pancreas is rare. To our knowledge, only 64 cases of pancreatic schwannoma have been reported in the English literature over the past 40 years. Precise preoperative diagnosis is challenging despite the application of multiple imaging modalities. Surgery is the most effective treatment for pancreatic schwannoma. As tumors could be located in different parts of the pancreas, surgical methods vary accordingly. Following complete tumor removal, patients with pancreatic schwannomas generally have a good prognosis.
A 59-year-old female was referred to our hospital because of a pancreatic mass found during a routine health examination.
The abdomen was soft and nondistended without evidence of a palpable mass.
Differential diagnoses included intraductal papillary mucinous neoplasm, mucinous cystic neoplasm, solid pseudopapillary tumor, pancreatic endocrine tumor or pancreatic ductal adenocarcinoma.
Abnormal laboratory results included: Unconjugated bilirubin, 2 μmol/L (normal, 3-14) and serum kalium 3.42 mmol/L (normal, 3.5-5.2).
Ultrasound revealed a 1.4 cm × 1.3 cm, well-defined cystic lesion in the pancreatic body, as well as a 6.8 cm sized strong echo in the gallbladder. An unenhanced computed tomography (CT) scan showed a 1.6 cm × 1.1 cm well-defined hypodense mass in the pancreatic body. On the contrast-enhanced CT, the mass was not enhanced. On magnetic resonance imaging, the mass in the pancreatic body and gallbladder appeared hypointense on T1 weighted images. The mass in the pancreatic body appeared inhomogeneously hyperintense and the enlarged gallbladder appeared hyperintense on T2 weighted images. We also performed Endoscopic ultrasound-guided fine needle aspiration. However, the tumor sample was difficult to acquire, and the procedure was unsuccessful. According to these results, a pancreatic cystadenoma and an enlarged gallbladder with multiple stones were preliminarily considered.
Microscopic examination revealed a tumor composed mainly of spindle-shaped cells with palisading arrangement and no atypia, which is consistent with a benign tumor. Both hypercellular and hypocellular areas were visible. Immunohistochemical staining was strongly positive for protein S-100. Finally, the tumor was diagnosed as a schwannoma of the pancreatic body.
The patient underwent central pancreatectomy, end-to-side pancreaticojejunostomy and cholecystectomy.
Schwannoma of the pancreas is rare. Over the past 40 years, only 64 cases of pancreatic schwannomas have been reported in the English literature.
Precise preoperative diagnosis is challenging despite the application of multiple imaging modalities. Surgery is the most effective treatment for pancreatic schwannoma. As tumors could be located in different parts of the pancreas, surgical approach varies accordingly. Following complete tumor removal, patients with pancreatic schwannomas generally have a good prognosis.
This manuscript is an interesting surgical case report, good literature review, and well written. This study highlights the diagnosis and treatment of a rare pancreatic schwannoma and presents a literature review to deepen the understanding of the subject. The information included is worthwhile to the reader.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Garcia-Olmo D, Kowalewski PK, Mise Y, Popescu I S- Editor: Gong ZM L- Editor: Ma JY E- Editor: Zhang FF
| 1. | Pilavaki M, Chourmouzi D, Kiziridou A, Skordalaki A, Zarampoukas T, Drevelengas A. Imaging of peripheral nerve sheath tumors with pathologic correlation: pictorial review. Eur J Radiol. 2004;52:229-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 206] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 2. | Le Guellec S. [Nerve sheath tumours]. Ann Pathol. 2015;35:54-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 3. | Bhattacharyya AK, Perrin R, Guha A. Peripheral nerve tumors: management strategies and molecular insights. J Neurooncol. 2004;69:335-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 73] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | Nishikawa T, Shimura K, Tsuyuguchi T, Kiyono S, Yokosuka O. Contrast-enhanced harmonic EUS of pancreatic schwannoma. Gastrointest Endosc. 2016;83:463-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Mourra N, Calvo J, Arrive L. Incidental Finding of Cystic Pancreatic Schwannoma Mimicking a Neuroendocrine Tumor. Appl Immunohistochem Mol Morphol. 2016;24:149-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 6. | Duma N, Ramirez DC, Young G, Nikias G, Karpeh M, Bamboat ZM. Enlarging Pancreatic Schwannoma: A Case Report and Review of the Literature. Clin Pract. 2015;5:793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 7. | Ohbatake Y, Makino I, Kitagawa H, Nakanuma S, Hayashi H, Nakagawara H, Miyashita T, Tajima H, Takamura H, Ninomiya I. A case of pancreatic schwannoma - The features in imaging studies compared with its pathological findings: Report of a case. Clin J Gastroenterol. 2014;7:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 8. | Kim JY, Song JS, Park H, Byun JH, Song KB, Kim KP, Kim SC, Hong SM. Primary mesenchymal tumors of the pancreas: single-center experience over 16 years. Pancreas. 2014;43:959-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 9. | J D, R S, K C, Devi NR. Pancreatic schwannoma - a rare case report. J Clin Diagn Res. 2014;8:FD15-FD16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Ciledag N, Arda K, Aksoy M. Pancreatic schwannoma: A case report and review of the literature. Oncol Lett. 2014;8:2741-2743. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Val-Bernal JF, Mayorga M, Sedano-Tous MJ. Schwannomatosis presenting as pancreatic and submandibular gland schwannoma. Pathol Res Pract. 2013;209:817-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Poosawang W, Kiatkungwankai P. Pancreatic schwannoma: A case report and review of literature. J Med Assoc Thai. 2013;96:112-116. [PubMed] |
| 13. | Barresi L, Tarantino I, Granata A, Traina M. Endoscopic ultrasound-guided fine-needle aspiration diagnosis of pancreatic schwannoma. Dig Liver Dis. 2013;45:523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Abu-Zaid A, Azzam A, Abou Al-Shaar H, Alshammari AM, Amin T, Mohammed S. Pancreatic tail schwannoma in a 44-year-old male: a case report and literature review. Case Rep Oncol Med. 2013;2013:416713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | Di Benedetto F, Ballarin R, Spaggiari M, Pecchi A, Gerunda GE. Role of intraoperative ultrasonography for pancreatic schwannoma. J Surg Oncol. 2012;105:859-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Moriya T, Kimura W, Hirai I, Takeshita A, Tezuka K, Watanabe T, Mizutani M, Fuse A. Pancreatic schwannoma: Case report and an updated 30-year review of the literature yielding 47 cases. World J Gastroenterol. 2012;18:1538-1544. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 50] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 17. | Paik KY, Choi SH, Heo JS, Choi DW. Solid tumors of the pancreas can put on a mask through cystic change. World J Surg Oncol. 2011;9:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Kim G, Choi YS, Kim HJ, Do JH, Park ES. Pancreatic benign schwannoma: combined with hemorrhage in an internal cyst. J Dig Dis. 2011;12:138-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Liegl B, Bodo K, Martin D, Tsybrovskyy O, Lackner K, Beham A. Microcystic/reticular schwannoma of the pancreas: a potential diagnostic pitfall. Pathol Int. 2011;61:88-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 20. | Kinhal VA, Ravishankar TH, Melapure AI, Jayaprakasha G, Range Gowda BC. Pancreatic schwannoma: Report of a case and review of literature. Indian J Surg. 2010;72:296-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Dorsey F, Taggart MW, Fisher WE. Image of the month. Pancreatic schwannoma. Arch Surg. 2010;145:913-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 22. | Stojanovic MP, Radojkovic M, Jeremic LM, Zlatic AV, Stanojevic GZ, Jovanovic MA, Kostov MS, Katic VP. Malignant schwannoma of the pancreas involving transversal colon treated with en-bloc resection. World J Gastroenterol. 2010;16:119-122. [PubMed] |
| 23. | Suzuki S, Kaji S, Koike N, Harada N, Hayashi T, Suzuki M, Hanyu F, Ban S. Pancreatic schwannoma: a case report and literature review with special reference to imaging features. JOP. 2010;11:31-35. [PubMed] |
| 24. | Aggarwal G, Satsangi B, Shukla S, Lahoti BK, Mathur RK, Maheshwari A. Rare asymptomatic presentations of schwannomas in early adolescence: three cases with review of literature. Int J Surg. 2010;8:203-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Oshima M, Yachida S, Suzuki Y. Pancreatic schwannoma in a 32-year-old woman mimicking a solid-pseudopapillary neoplasm. Clin Gastroenterol Hepatol. 2010;8:e1-e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 26. | Mummadi RR, Nealon WH, Artifon EL, Fleming JB, Bhutani MS. Pancreatic Schwannoma presenting as a cystic lesion. Gastrointest Endosc. 2009;69:341; discussio 341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Gupta A, Subhas G, Mittal VK, Jacobs MJ. Pancreatic schwannoma: literature review. J Surg Educ. 2009;66:168-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | Li S, Ai SZ, Owens C, Kulesza P. Intrapancreatic schwannoma diagnosed by endoscopic ultrasound-guided fine-needle aspiration cytology. Diagn Cytopathol. 2009;37:132-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 29. | Tafe LJ, Suriawinata AA. Cystic pancreatic schwannoma in a 46-year-old man. Ann Diagn Pathol. 2008;12:296-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Hirabayashi K, Yasuda M, Umemura S, Itoh H, Itoh J, Yazawa N, Imaizumi T, Osamura RY. Cytological features of the cystic fluid of pancreatic schwannoma with cystic degeneration. A case report. JOP. 2008;9:203-208. [PubMed] |
| 31. | Okuma T, Hirota M, Nitta H, Saito S, Yagi T, Ida S, Okamura S, Chikamoto A, Iyama K, Takamori H. Pancreatic schwannoma: report of a case. Surg Today. 2008;38:266-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 32. | Tofigh AM, Hashemi M, Honar BN, Solhjoo F. Rare presentation of pancreatic schwannoma: a case report. J Med Case Rep. 2008;2:268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 33. | Fasanella KE, Lee KK, Kaushik N. Clinical challenges and images in GI. Benign schwannoma of the pancreatic head. Gastroenterology. 2007;132:489, 830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 34. | Di Benedetto F, Spaggiari M, De Ruvo N, Masetti M, Montalti R, Quntini C, Ballarin R, Di Sandro S, Costantini M, Gerunda GE. Pancreatic schwannoma of the body involving the splenic vein: case report and review of the literature. Eur J Surg Oncol. 2007;33:926-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 35. | Yu RS, Sun JZ. Pancreatic schwannoma: CT findings. Abdom Imaging. 2006;31:103-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 36. | Wu YL, Yan HC, Chen LR, Chen J, Gao SL, Li JT. Pancreatic benign schwannoma treated by simple enucleation: case report and review of literature. Pancreas. 2005;31:286-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 37. | Novellas S, Chevallier P, Saint Paul MC, Gugenheim J, Bruneton JN. MRI features of a pancreatic schwannoma. Clin Imaging. 2005;29:434-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 38. | Soumaoro LT, Teramoto K, Kawamura T, Nakamura N, Sanada T, Sugihara K, Arii S. Benign schwannoma of the pancreas. J Gastrointest Surg. 2005;9:288-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 39. | Bui TD, Nguyen T, Huerta S, Gu M, Hsiang D. Pancreatic schwannoma. A case report and review of the literature. JOP. 2004;5:520-526. [PubMed] |
| 40. | Akiyoshi T, Ueda Y, Yanai K, Yamaguchi H, Kawamoto M, Toyoda K, Hayashi T, Ohuchida J. Melanotic schwannoma of the pancreas: report of a case. Surg Today. 2004;34:550-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 41. | von Dobschuetz E, Walch A, Werner M, Hopt UT, Adam U. Giant ancient schwannoma of pancreatic head treated by extended pancreatoduodenectomy. Pancreatology. 2004;4:505-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 42. | Paranjape C, Johnson SR, Khwaja K, Goldman H, Kruskal JB, Hanto DW. Clinical characteristics, treatment, and outcome of pancreatic Schwannomas. J Gastrointest Surg. 2004;8:706-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 43. | Tan G, Vitellas K, Morrison C, Frankel WL. Cystic schwannoma of the pancreas. Ann Diagn Pathol. 2003;7:285-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 44. | Almo KM, Traverso LW. Pancreatic schwannoma: an uncommon but important entity. J Gastrointest Surg. 2001;5:359-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 45. | Lee JS, Kim HS, Jung JJ, Han SW, Kim YB. Ancient schwannoma of the pancreas mimicking a cystic tumor. Virchows Arch. 2001;439:697-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 46. | Morita S, Okuda J, Sumiyoshi K, Taketani M, Moriguchi A, Katsu K, Tanigawa N. Pancreatic Schwannoma: report of a case. Surg Today. 1999;29:1093-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 47. | Brown SZ, Owen DA, O’Connell JX, Scudamore CH. Schwannoma of the pancreas: a report of two cases and a review of the literature. Mod Pathol. 1998;11:1178-1182. [PubMed] |
| 48. | Hsiao WC, Lin PW, Chang KC. Benign retroperitoneal schwannoma mimicking a pancreatic cystic tumor: case report and literature review. Hepatogastroenterology. 1998;45:2418-2420. [PubMed] |
| 49. | Feldman L, Philpotts LE, Reinhold C, Duguid WP, Rosenberg L. Pancreatic schwannoma: report of two cases and review of the literature. Pancreas. 1997;15:99-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 50. | Ferrozzi F, Bova D, Garlaschi G. Pancreatic schwannoma: report of three cases. Clin Radiol. 1995;50:492-495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 51. | Sugiyama M, Kimura W, Kuroda A, Muto T. Schwannoma arising from peripancreatic nerve plexus. AJR Am J Roentgenol. 1995;165:232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 52. | Steven K, Burcharth F, Holm N, Pedersen IK. Single stage pancreaticoduodenectomy (Whipple’s procedure), radical cystectomy and bladder substitution with the urethral Kock reservoir. Case report. Scand J Urol Nephrol. 1994;28:199-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 53. | Melato M, Bucconi S, Marus W, Spivach A, Perulli A, Mucelli RP. The schwannoma: an uncommon type of cystic lesion of the pancreas. Ital J Gastroenterol. 1993;25:385-387. [PubMed] |
| 54. | David S, Barkin JS. Pancreatic schwannoma. Pancreas. 1993;8:274-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 55. | Urban BA, Fishman EK, Hruban RH, Cameron JL. CT findings in cystic schwannoma of the pancreas. J Comput Assist Tomogr. 1992;16:492-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 56. | Burd DA, Tyagi G, Bader DA. Benign schwannoma of the pancreas. AJR Am J Roentgenol. 1992;159:675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 57. | Coombs RJ. Case of the season. Malignant neurogenic tumor of duodenum and pancreas. Semin Roentgenol. 1990;25:127-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 58. | Liessi G, Barbazza R, Sartori F, Sabbadin P, Scapinello A. CT and MR imaging of melanocytic schwannomas; report of three cases. Eur J Radiol. 1990;11:138-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 59. | Walsh MM, Brandspigel K. Gastrointestinal bleeding due to pancreatic schwannoma complicating von Recklinghausen’s disease. Gastroenterology. 1989;97:1550-1551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 60. | Eggermont A, Vuzevski V, Huisman M, De Jong K, Jeekel J. Solitary malignant schwannoma of the pancreas: report of a case and ultrastructural examination. J Surg Oncol. 1987;36:21-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 61. | Móller Pedersen V, Hede A, Graem N. A solitary malignant schwannoma mimicking a pancreatic pseudocyst. A case report. Acta Chir Scand. 1982;148:697-698. [PubMed] |
| 62. | Das Gupta TK, Brasfield RD, Strong EW, Hajdu SI. Benign solitary Schwannomas (neurilemomas). Cancer. 1969;24:355-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 63. | Das Gupta TK, Brasfield RD. Tumors of peripheral nerve origin: benign and malignant solitary schwannomas. CA Cancer J Clin. 1970;20:228-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 64. | Abell MR, Hart WR, Olson JR. Tumors of the peripheral nervous system. Hum Pathol. 1970;1:503-551. [PubMed] |
| 65. | Fu H, Lu B. Giant retroperitoneal schwannoma: a case report. Int J Clin Exp Med. 2015;8:11598-11601. [PubMed] |
| 66. | Tao K, Chang W, Zhao E, Deng R, Gao J, Cai K, Wang G, Zhang P. Clinicopathologic Features of Gastric Schwannoma: 8-Year Experience at a Single Institution in China. Medicine (Baltimore). 2015;94:e1970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 67. | Weiss SW, Langloss JM, Enzinger FM. Value of S-100 protein in the diagnosis of soft tissue tumors with particular reference to benign and malignant Schwann cell tumors. Lab Invest. 1983;49:299-308. [PubMed] |