Published online Jan 14, 2016. doi: 10.3748/wjg.v22.i2.776
Peer-review started: June 3, 2015
First decision: July 20, 2015
Revised: August 13, 2015
Accepted: September 28, 2015
Article in press: September 30, 2015
Published online: January 14, 2016
Processing time: 218 Days and 20.4 Hours
Pancreatic cancer is the fourth most common cause of cancer deaths worldwide. Although recent therapeutic developments for patients with pancreatic cancer have provided survival benefits, the outcomes for patients with pancreatic cancer remain unsatisfactory. Molecularly targeted cancer therapy has advanced in the past decade with the use of a number of pathways as candidates of therapeutic targets. This review summarizes the molecular features of this refractory disease while focusing on the recent clinical and experimental findings on pancreatic cancer. It also discusses the data supporting current standard clinical outcomes, and offers conclusions that may improve the management of pancreatic cancer in the future.
Core tip: Pancreatic cancer-related mortality is almost consistently caused by local recurrence and metastasis. The survival of patients after surgical resection remains poor, and the results of adjuvant chemotherapy and radiotherapy are still unsatisfactory. Therefore, new treatments are urgently needed. Recent developments in our knowledge of the underlying biological features of pancreatic cancer may be useful in establishing molecularly targeted therapy as a new strategy, similar to those used to treat other types of malignancies.
- Citation: Matsuoka T, Yashiro M. Molecular targets for the treatment of pancreatic cancer: Clinical and experimental studies. World J Gastroenterol 2016; 22(2): 776-789
- URL: https://www.wjgnet.com/1007-9327/full/v22/i2/776.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i2.776
Pancreatic cancer is the fourth most common cause of cancer deaths, resulting in 330000 deaths per year worldwide[1]. Many patients with pancreatic cancer are diagnosed at advanced incurable stages because of the absence of screening. Although advances in a variety of approaches have improved the management of pancreatic cancer, the 5-year survival rate remains lower than 5%[1]. Surgical resection is currently the only potentially curative treatment. However, even after resection, the 5-year survival rate is less than 20% due to the high frequency of distal metastasis and local recurrence[2].
The treatment of inoperable pancreatic cancer has traditionally involved the use of gemcitabine with low response rates and a marginal survival benefit. The failure of clinical treatment in patients with pancreatic cancer is often due to the heterogeneous nature of the disease. This type of tumor involves not only cancer cells, but stellate cells and stroma, which were known as microenvironment. Stromal proliferation and reduced angiogenesis have been shown to contribute to therapeutic resistance despite the efficacy in experimental studies utilizing cell lines or animal models. A recent European study found that the combination chemotherapy of FOLFORINOX and gemcitabine is more effective than the use of gemcitabine alone. Albumin-bound paclitaxel (nab-paclitaxel), which was approved by the FDA in 2013, can also be used in conjunction with gemcitabine to treat pancreatic cancer[3]. However, in most patients with advanced stages of the disease, these treatments only prolong survival by a few months, while combination therapy can also lead to significantly increased toxicity[4]. The development of effective pancreatic cancer treatments is urgently needed to overcome these obstacles.
To date, knowledge of the molecular basis of tumor initiation has led to the use of various kinds of targeting agents to produce better prognoses for some types of solid tumors. These agents, including those targeting the angiogenesis pathways, the epidermal growth factor receptor (EGFR), the mitogen-activated ERK kinase (MEK), the fibroblast growth factor receptor (FGFR), the phosphatidylinositol-3 kinase/protein kinase-B/mammalian target of rapamycin (PI3K/mTOR), and the cancer stem cell compartment, may lead to significant advancements in pancreatic cancer treatment. In this study, we will review the current clinical and experimental results regarding molecular targets for the treatment of pancreatic cancer, and discuss potential future treatments.
Recent studies have shown that pancreatic cancers include an average of 63 genetic alterations[5]. Therefore, in order to develop effective treatments for pancreatic cancer, the complicated gene alterations and pathological features of this tumor type need to be elucidated. The molecular analysis of pancreatic cancer has often shown the involvement of known cancer genes and traditional cancer signaling pathways. The KRAS gene, which encodes a small GTPase that regulates the downstream signaling of growth factor receptors, is a known mutated oncogene found in most pancreatic cancers at advanced stages[6]. Missense mutations in the KRAS cluster have been found in specific hotspots (most generally codon 12)[7]. Recent studies have demonstrated that KRAS mutations are one of the earliest genetic events seen in human pancreatic intraepithelial neoplasia (PanIN) progression[8,9]. In addition to KRAS mutations, alterations in tumor suppressor genes such as INK4A, BRCA2, and LKB1 occur frequently in pancreatic cancer. The tumor suppressor gene, P16/CDKN2A, which encodes a critical cell cycle regulator, is inactivated in > 90% of pancreatic cancer[10]. Mutation of the p53 gene is closely associated with cellular responses to cytotoxic stress by contributing to both cell cycle arrest and cell apoptosis[11]. Mutations in p53 are also common in pancreatic cancer, as these have been reported in approximately 75% of patients and most frequently characterized by small intragenic mutations coupled with a loss of heterozygosity[5]. The missense mutation of SMAD4, a tumor suppressor gene that encodes the transforming growth factor beta (TGFβ) signaling pathway, is found in approximately 55% of patients with pancreatic cancer[12]. Mutations in SMAD4 are associated with a poorer prognosis and widespread metastases, which suggests potential clinical implications[13]. The mismatch repair gene, MLH1, and the cationic trypsinogen gene, PRSS1, are also often mutated in pancreatic cancer[14]. Based on the typical age of onset related to the aforementioned mutations, these genetic lesions are thought to impact malignant progression rather than cancer initiation.
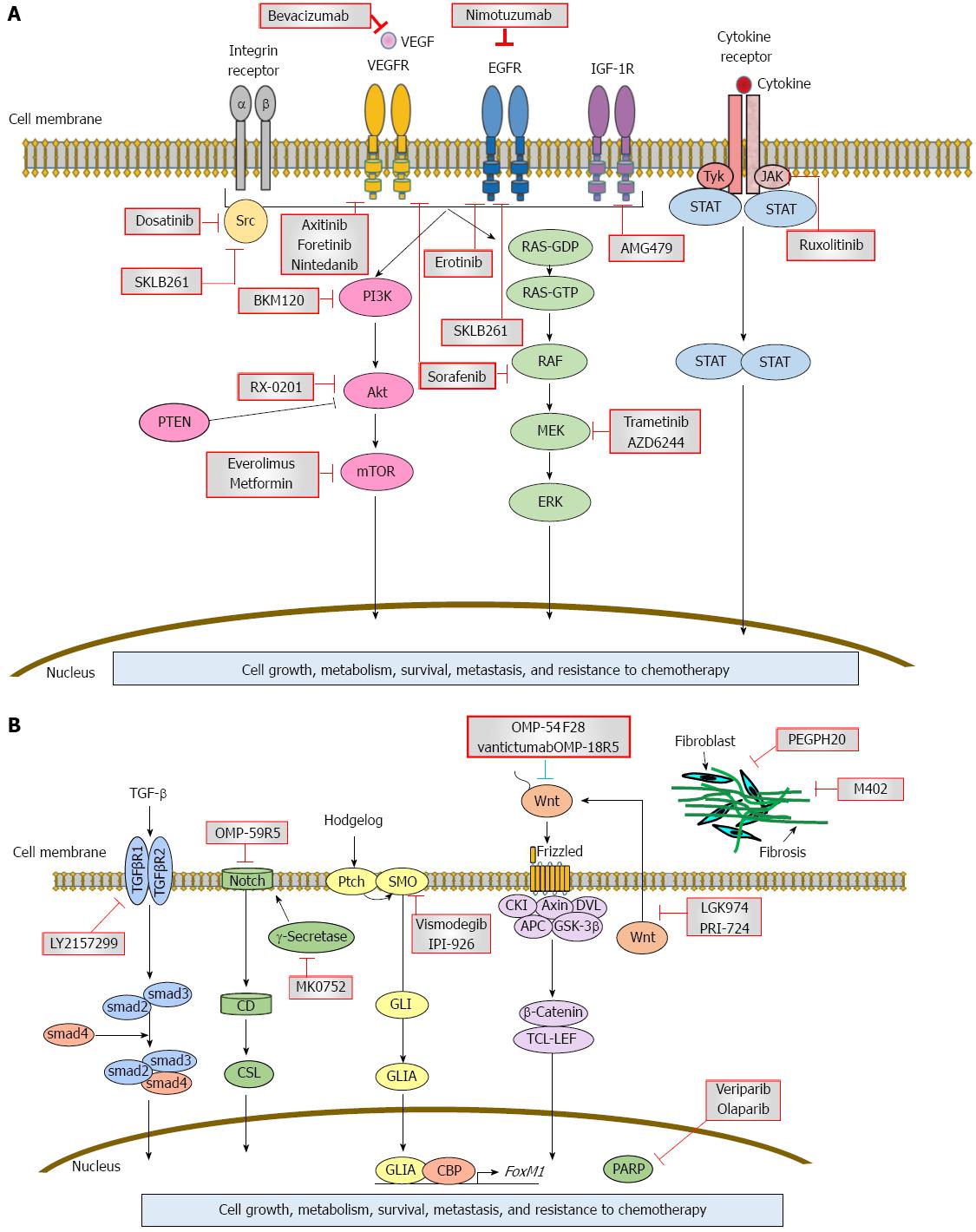
Some pancreatic cancers harbor activating mutations of BRAF rather than KRAS[15]. BRAF encodes RAF, a serine/threonine kinase belonging to a family of MEK (Figure 1A). MEK activates ERK, which forms the MAPK signaling pathway. Thus, active mutations of KRAS and BRAF eventually result in triggering the MAPK signaling, which is critical for the development of pancreatic cancer. Activation of the MAPK pathway is found not only in benign lesions, but also in late-stage pancreatic cancer[16]. Overexpressed MAPK through a constitutively active form of RAF results in PanIN/pancreatic ductal adenocarcinoma formation; conversely, the silencing of MAPK signaling inhibits tumor initiation[17,18]. PI3K signaling is another important pathway that has been studied in great detail in pancreatic cancer along with the MAPK pathway. The PI3K signaling mediates cell growth and survival via several downstream substrates such as Akt, p70-S6K, and mTOR (Figure 1A). Similar to the MAPK pathway, the consistent activation of PI3K has been shown to be closely associated with the carcinogenesis of pancreatic cancer[19]. The PI3K downstream effector, Akt, is amplified in 10%-20% of pancreatic cancers, providing genetic evidence to support the importance of this pathway in this type of cancer[20]. Several growth factor receptors, including vascular endothelial growth factor (VEGF) and insulin-like growth factor 1 receptor (IGF1R), are aberrantly expressed in pancreatic cancer[21]. These pathways mediate the important genes involved in a variety of cellular functions such as growth, apoptosis, differentiation, and metastasis via these two pathways (Figure 1A).
Within the last decade, numerous targeted agents have been examined individually or in combination with cytotoxic agents for the treatment of pancreatic cancer. The growth stimulating signaling described above has been targeted by molecular therapies for many kinds of cancer. Taken together, a number of paracrine signaling pathways, such as Hedgehog, Wnt, Notch, and TGFβ, might also contribute to cancer stem cell signaling and tumorigenesis (Figure 1B)[22]. These characteristics of pancreatic cancer may contribute to the development of molecularly targeted therapies. Figure 1 schematically summarizes the current understanding of inhibitors in pancreatic cancer. Table 1 summarizes clinical trials using molecular targeting agents.
| Target molecule | ClinicalTrials.gov identifier | Sponsor | Agent | Treatmentsetting | Studyphase | Comments |
| EGFR | NCT00561990 | Oncoscience AG | Nimotuzumab | First line | II/III | GEM ± nimotuzumab |
| NCT02395016 | Biotech Pharmaceutical | Nimotuzumab | First line | III | GEM ± nimotuzumab | |
| MEK | NCT01222689 | National Cancer Institute | selumetinib | First line | II | Selumetinib + erlotinib |
| PI3K | NCT01571024 | UNC Lineberger Comprehensive Cancer Center | BKM120 | First line | I | BKM120 + mFOLFOX6 |
| Akt | NCT01028495 | Rexahn Pharmaceuticals | RX-0201 | First line | II | RX0201 + GEM |
| mTOR | NCT00981162 | Roswell Park Cancer Institute | Everolimus | Second line | I/II | Everolimus + sorefenib |
| Angiogenesis | NCT01509911 | Tiltan Pharma Ltd | TL-118 | First line | II | TL-118 + GEM |
| Src | FOLFOX-D, NCT01652976 | University of Florida | Dasatinib | First line | II | 5-Fluorouracil + leucovorin + oxaliplatin + dasatinib |
| Jak | NCT01423604 | Incyte Corporation | Ruxolitinib | Second line | II | Ruxolitinib + capecitabine |
| NCT01822756 | Incyte Corporation | Ruxolitinib | First line | I | Ruxolitinib + gemcitabine or nab-paclitaxel | |
| Notch | NCT01098344 | Cancer Research UK | MK0752 | First line | I | MK0752 + GEM |
| Hedgehog | NCT01130142 | Infinity Pharmaceuticals, Inc. | IPI-926 | First line | I/II | IPI-926 + GEM |
| Wnt | NCT01351103 | Novartis Pharmaceutical | LGK974 | First line | I | LGK974 alone |
| NCT01302405 | Prism Pharma Co., Ltd. | PRI-724 | First line | I | PRI-724 alone | |
| NCT02050178 | OncoMed Pharmaceuticals, Inc | OMP-54 F28 | First line | I | GEM + nab-paclitaxel + OMP-54 F28 | |
| NCT02005315 | OncoMed Pharmaceuticals, Inc | Vantictumab(OMP-18R5) | First line | I | GEM + nab-paclitaxel + vantictumab | |
| Stroma | Halo-109-202, NCT01839487 | Halozyme Therapeutics | PEGPH20(hyaluronidase) | First line | II | GEM + nab-paclitaxel ± PEGPH20 |
| S1313, NCT01959139 | Southwest Oncology Group | PEGPH20 | First line | I/II | FOLFIRINOX ± PEGPH20 | |
| PARP | NCT01585805 | National Cancer Institute | Veriparib | First line | II | GEM + cisplatin ± veriparib |
| NCT01296763 | Sidney Kimmel Comprehensive Cancer Center | Olaparib | First line | I/II | Irinotecan + cisplatin + mitomycin C ± olaparib | |
| Others | NCT01210911 | Academisch Medisch Centrum | Metformin | First line | II | Erlotinib + metformin + GEM |
| NCT01373164 | Eli Lilly and Company | LY2157299(TGF-b inhibitor) | First line | II | LY2157299 + GEM | |
| NCT01621243 | Momenta Pharmaceuticals, Inc | M402 (heparan sulfate) | First line | I/II | GEM + nab-paclitaxel ±M402 | |
| NCT01783171 | National Cancer Institute (NCI) | Dinaciclib | First line | I | Dinaciclib + MK-226 |
Angiogenesis pathway: Angiogenesis is crucial for the growth of malignancies. Anti-angiogenic therapies have shown efficacy in renal cell carcinoma, colorectal cancer, lung cancer, glioblastoma, and ovarian epithelial cancers[23]. VEGF is one of the key factors of angiogenesis that promotes tumor growth and metastasis[24]. VEGF is overexpressed in over 90% of patients with pancreatic cancer[25], thus providing justification for VEGF-targeted therapy for pancreatic cancer. In contrast, a randomized phase III trial (CALGB 80303) found no improvements in the survival of patients who were given a combination of bevacizumab, which is a monoclonal antibody to VEGF, and gemcitabine, compared to the results of those treated with gemcitabine and a placebo, despite promising outcomes in phase II[26]. Taken together, efforts to use targeted agents such as sorafenib and axitinib have been unfavorable[27-29]. A phase II trial using TL-118, a novel anti-angiogenic drug, combined with gemcitabine to treat metastatic pancreatic cancer (NCT01509911) is currently ongoing (Table 1). Foretinib was developed as an ATP-binding site competitor to inhibit receptor tyrosine kinases with reported activity against VEGFRs, RON, c-Met, c-KIT, FLT-3, and platelet-derived growth factor receptors (PDGFRs)[30]. Mounting evidence has indicated that foretinib targets multi-additional kinases, resulting in the growth inhibition of tumors. Since hepatocyte growth factor (HGF) and c-MET are frequently overexpressed in pancreatic cancer[31], targeting these pathways has attracted much attention. A recent study concluded that foretinib inhibits tumor growth, angiogenesis, and lymphangiogenesis in xenograft animals by inhibiting not only c-MET but VEGFR-2, VEGFR-3, and TIE-2 signaling as well. These results suggest that simultaneous inhibitory effects to reduce pancreatic tumor growth can be expected from multikinase inhibition[32].
EGFR pathway: EGFR, a transmembrane tyrosine kinase receptor of the ErbB family, plays an important role in tumor cell behavior. Aberrant EGFR activity leads to receptor dimerization and subsequently activates downstream signals, including members of the RAS and PI3K/Akt/mTOR pathways[33]. Overexpression of this growth factor receptor is seen in over 90% of pancreatic cancers[34]. Sustained EGFR activation has been reported in pancreatic cancer cell lines, and EGFR inhibitors have been found to lead to decreased proliferation[35]. Thus, EGFR pathway activation seems to provide a rationale for EGFR-targeted inhibition strategies. However, clinical trials using the anti-EGFR and anti-Erbb2 antibodies have yielded negative results[36,37]. In contrast, a large randomized phase III in which patients with pancreatic cancer were assigned to receive gemcitabine with or without erlotinib[38] reported that the patients who received the combination treatment had a modest but statistically significant improvement in overall median survival (OS) (P = 0.038) and progression-free survival (P = 0.004). The data from a subset analysis of this trial failed to indicate whether the KRAS mutation status or EGFR was a predictive marker for the therapeutic response to erlotinib[39]. Even though the median OS was only prolonged by 2 wk, this trial is remarkable because it is the only one to have shown an improvement in survival outcomes with combination gemcitabine/erlotinib in metastatic pancreatic cancer. On the other hand, the oncogenic benefit of erlotinib should be balanced with its potential complications, some of which have been reported to be fatal[40]. Another EGFR monoclonal antibody, nimotuzumab, achieved survival benefits when added to gemcitabine (8.7 mo vs 6.1 mo) with tolerable toxicity in a recent phase II trial involving patients with locally advanced pancreatic cancer (J Clin Oncol 2013;31:abstr 4009). Clinical trials to evaluate the effects of nimotuzumab combined with gemcitabine are ongoing in patients with the RAS wild type of locally advanced or metastatic pancreatic cancer (NCT 02395016).
IGF1R pathway: IGF1R belongs to the insulin receptor family. IGFR1 signaling is highly expressed in pancreatic cancer, and this activation leads to a signaling cascade that triggers pathways such as ERK and PI3K/Akt/mTOR. It also plays a role in cancer survival and proliferation through RAS-dependent and -independent pathways. Inhibition of IGF1R signaling enhances the cytotoxicity of gemcitabine in pancreatic cancer xenografts[41]. However, the IGF1R inhibitor, AMG-479, and the monoclonal antibody, cixutumumab, failed to provide any survival benefits in a previous study (NCT01231347) (J Clin Oncol 2012;30:abstr 198). Meanwhile, the use of IGF1R in conjunction with ErbB would appear to be a good strategy for overcoming the chemoresistance common in pancreatic cancer. A previous study demonstrated that the simultaneous blockade of IGF1R and EGFR/Her-2 synergistically inhibits the pancreatic tumor growth and completely abolishes the activation of IRS-1, Akt, and MAPK phosphorylation. These results suggest that the combined application of these two inhibitors averts the resistance associated with monotherapy[42].
RAS pathway: The RAS/RAF/MEK/ERK (MAPK) pathway is activated by numerous growth signals via their receptors, including EGFR, and is crucial in mediating uncontrolled growth and survival[43]. As previously discussed, MAPK plays a crucial role in the development of pancreatic cancer. Although trametinib, a MEK inhibitor, is currently approved for the treatment of melanoma, this drug has failed to show survival benefit when combined with gemcitabine in advanced pancreatic cancer (J Clin Oncol 2013;31:abstr 291). A phase II trial of another MEK inhibitor, AZD6244, in combination with erlotinib as a second line treatment of advanced pancreatic cancer is currently underway (NCT01222689).
PI3K/Akt/mTOR pathway: Upon activation by RAS or EGFR, PI3K activates Akt, which subsequently triggers multiple downstream targets such as mTOR, leading to the regulation of many essential cellular processes, including cell growth, metabolism, survival, metastasis, and resistance to chemotherapy[44]. The PI3K-Akt pathway is activated in 59% of patients with pancreatic cancer[45]. Deregulation of this pathway through absent or reduced expression of PTEN (phosphatase and tensin homolog, a natural antagonist of PI3K) is frequently found in pancreatic cancer[46]. A phase II trial is currently testing the combination of an Akt antisense oligonucleotide, RX-0201, with gemcitabine in metastatic pancreatic cancer (NCT01028495). A phase I study of BKM120, a pan-class 1A PI3K inhibitor, with mFOLFOX6 in patients with metastatic pancreatic cancer is also now recruiting (NCT01571024). Everolimus, an mTOR inhibitor, has shown antitumor activities, including the inhibition of cell proliferation, apoptosis, and angiogenesis, and displayed synergistic effects when combined with other anticancer agents[47]. A phase II study was conducted to explore treatment activity of the combination of capecitabine with everolimus in patients with advanced pancreatic cancer. The results revealed a response rate (RR) of 6.5% and an OS of 8.9 mo, suggesting that this combination therapy might enhance the efficacy of capecitabine[48]. Sorafenib, a multikinase inhibitor targeting Raf-1, BRaf, VEGFR1, VEGFR2, VEGFR3, and PDGFRβ, has been confirmed to be efficacious against advanced hepatocellular carcinoma[49]. It acts by inhibiting Raf-1 and BRaf as well as the activities of VEGFR1, VEGFR2, VEGFR3, and PDGFRβ. A phase I/II combination trial of sorafenib and everolimus in advanced pancreatic cancer has been completed, but the results have yet to be released (NCT00981162). Metformin, an antidiabetic drug that has direct metabolic effects through the activation of adenosine monophosphate-activated protein kinases, can inhibit the mTOR pathway by activating the tumor suppressor gene, TSC2. A randomized phase II study exploring the activity and safety of erlotinib and metformin combined with gemcitabine in patients with metastatic pancreatic cancer is also currently underway (NCT01210911).
Src pathway: Src, a family of proto-oncogenic non-receptor protein tyrosine kinases, plays a pivotal role in regulating multiple signal transduction pathways via its interactions with a number of proteins, including receptor tyrosine kinases and G-protein coupled receptors. C-Src is frequently overexpressed and/or aberrantly activated in a number of malignancies including 70% of pancreatic cancers[50]. Dasatinib, a compound related to saracatinib, was examined in a phase II trial in patients with metastatic pancreatic cancer; however, encouraging results were not obtained. A phase II trial to explore the efficacy of dasatinib combined with 5-fluorouracil, leucovorin, and oxaliplatin against metastatic pancreatic cancer is currently recruiting subjects (FOLFOX-D, NCT01652976).
JAK/STAT pathways: Activation of the Janus kinase/signal transducer and transcription (JAK/STAT) pathway has been found in many human cancers[51]. JAKs are a family of cytoplasmic tyrosine kinases, comprised of four members-JAK1, JAK2, JAK3, and Tyk2. JAK activation occurs upon the binding of a ligand to cell surface receptors, which leads to the creation of sites for interaction with proteins that contain phosphotyrosine-binding Src homology 2 (SH2) domains. STATs, a family of downstream transcription factors for JAKs[52], contain a tyrosine residue phosphorylated by JAKs, leading to nuclear translocation. In the nucleus, STATs serve as transcription factors that initiate the transcription of downstream target genes. Abnormalities of the JAK/STAT pathway contribute directly to cellular transformation, increased cell proliferation, apoptosis, and angiogenesis. JAK mutations and STAT activation have been reported in pancreatic cancer[53,54]. In a randomized phase II study of capecitabine plus either ruxolitinib or placebo, patients with metastatic pancreatic cancer demonstrated an improvement in survival (NCT01423604). An early phase clinical trial of ruxolitinib and gemcitabine with or without nab-paclitaxel is also currently underway (NCT01822756).
A small population of pancreatic cancer stem cells (CSCs) has been suggested to be resistant to chemotherapy and radiation therapy. CSCs are believed to be responsible for tumor carcinogenesis, progression, and metastasis in cancers including the pancreatic type[55,56]. Hedgehog, Notch, and Wnt have been shown to play a pivotal role in the development of pancreatic cancer stem cells[57]. Remarkable progress in understanding the involvement of CSCs in pancreatic cancer might highlight these cells as attractive targets for therapy.
Notch signaling: Recent evidence has suggested that Notch signaling is implicated in tumor growth and survival as well as involved in the development and function of many organs[58]. This pathway is thought to sustain a pool of pancreatic progenitor cells at an early stage of pancreatic development, and regulates pancreatic ductal cell differentiation[59]. Notch ligands and receptors have been shown to be highly expressed in pancreatic cancer; they also promote epithelial-mesenchymal transition (EMT) by regulating several transcription factors such as Snail, Slug, and TGFβ[60]. A number of studies have shown that chemotherapy-resistant pancreatic CSCs are related to Notch signaling activation[61]. The ALPINE trial is a phase Ib study exploring the anti-Notch2/3 inhibitor, OMP-59R5, in combination with nab-paclitaxel and gemcitabine in patients with untreated metastatic pancreatic cancer (J Clin Oncol 2014;32:abstr). This study showed good tolerability and positive responses (partial response = 46%, durable complete response = 77%). One appealing target for blocking this pathway is the γ-secretase enzyme, which causes proteolytic cleavage and controls the release of the Notch intracellular domain as well as the production of its active form. Clinical trials are currently underway to investigate the effects of MK0752, a γ-secretase inhibitor, combined with gemcitabine in patients with advanced pancreatic cancer (NCT01098344).
Hedgehog signaling: The Hedgehog (HH) pathway regulates embryogenesis, which is undetectable in normal pancreatic tissue. HH binds to the extracellular receptor and the transcriptional target gene, Patched (Ptch), the latter of which releases the Smoothened seven-transmembrane protein (SMO). This allows SMO to translocate to the cell surface and results in the activation of GLI transcription factors and the consequent induction of HH target genes, including GLI and Ptch1 (Figure 1B). The pathological behavior of HH signaling is well known, and an increased expression has been observed during pancreatic tumorigenesis[62]. It has been found that hedgehog signaling is phosphorylated in earlier pancreatic tumor lesions, and the expression of pathway substrates becomes elevated during the progression to an advanced stage[63]. HH signaling has also been shown to be closely associated with KRAS mutations, which drive the early stages of pancreatic neoplasia[64]. Interestingly, HH signaling in pancreatic cancer is localized to the stromal compartment, and the overexpression of sonic HH in the pancreas is sufficient to initiate precancerous lesions in transgenic mice, which contributes to maintaining the tumor microenvironment[65,66]. Interestingly, the attenuated action of sonic HH has resulted in improved gemcitabine delivery, the depletion of dense stroma, and an enhanced vascularization of the tumors in mouse models[67], suggesting that this pathway could be an appealing target for drug development. GDC-0449, also known as vismodegib, a small-molecule SMO antagonist, inhibits the HH signaling pathway. A pilot study evaluating the effects of GDC-0449 in combination with gemcitabine was performed in patients with metastatic pancreatic adenocarcinoma; however, the joint use of GDC-0449 and gemcitabine was not found to be superior to the sole use of gemcitabine in the treatment of metastatic pancreatic cancer[68]. Similarly, a randomized phase II trial of gemcitabine with or without vismodegib in treating patients with recurrent or metastatic pancreatic cancer also yielded disappointing results (NCT01064622). A phase II randomized study evaluating IPI-926, a small molecule SMO antagonist, in combination with gemcitabine in metastatic pancreatic cancer patients was recently completed, and the publication of its results is highly anticipated (NCT01130142).
Wnt signaling: Evidence that the Wnt signaling pathway plays a pivotal role in the regulation of stem cells has been accumulating[69,70]. Recent studies of Wnt signaling have suggested its roles in tumor biology and the pathogenesis of pancreatic cancer. Based on findings, it is plausible that the dysregulation of Wnt signaling pathway is closely associated with pancreatic cancer chemoresistance[71]. Wnt signaling inhibitors such as LGK974 and PRI-724 are currently under investigation in active phase I clinical studies on advanced solid tumors including pancreatic cancers (NCT01302405 and NCT01351103, respectively). An open-label phase 1b dose-escalation study to elucidate the safety and tolerability of OMP-54 F28 (NCT02050178) and Vantictumab/OMP-18R5 (NCT02005315) when combined with nab-paclitaxel and gemcitabine is currently in progress.
TGFβ: TGF-β is intimately involved in regulating numerous physiological processes, including cellular differentiation, homeostasis, and EMT in pancreatic cancer[72]. A phase II study of gemcitabine in combination with LY2157299, a specific type 1 receptor inhibitor of TGFβ, or a placebo is currently being implemented (NCT01373164).
One of the most important obstacles that impairs the effects of anticancer agents is the extracellular matrix (ECM) and stromal cells[73]. It has been reported that tumor-stroma interactions result in a complicated signaling network that leads to tumor progression in many kinds of solid cancers[74,75]. Pancreatic cancer shows abundant stroma in the tumor microenvironment[76]. Pancreatic cancer is uniquely characterized by a rich tumor stroma that might interfere with the delivery of agents to tumors and induce a complex interplay of intercellular signaling. Stromal depletion strategies such as the degradation of hyaluronic acid could potentially facilitate drug delivery to tumor sites[67]. Currently, there is a randomized phase II trial comparing the treatment effects of PEGPH20, a pegylated formulation of recombinant hyaluronidase, in combination with nab-paclitaxel and gemcitabine comparing the treatment effects of PEGPH20 with the treatment effects of nab-paclitaxel alone in metastatic pancreatic cancer (Halo-109-202, NCT01839487). A partially randomized phase I/II evaluation of modified FOLFIRINOX with or without PEGPH20 in patients with newly diagnosed metastatic pancreatic cancer is also now under testing (S1313, NCT01959139). Heparan sulfate proteoglycans (HSPGs) are complex polysaccharides that regulate several aspects of cancer biology, including tumorigenesis, tumor development, and metastasis[77]. HSPGs have been shown to be associated with the tumor microenvironment by binding to factors that support tumor proliferation. M402, a mimetic of heparan sulfate, blocks the multiple interactions associated with heparan sulfate[77]. A phase II clinical trial evaluating the effects of M402 in conjunction with standard chemotherapy is actively ongoing (NCT01621243).
Breaks in the DNA double-strand are generally repaired by homologous recombination, which is mediated by BRCA1 and BRCA2 proteins that sustain genomic stability and cell death. The poly (ADP-ribose) polymerase pathway takes on the main role in DNA repair when BRCA dysfunction occurs[78]. The PARP proteins undertake roles in a wide range of cellular functions including DNA transcription, DNA damage response, genomic stability maintenance, and cell cycle regulation. Inhibitors of PARP enzymes result in synthetic lethality in cancers with DNA repair failure or homologous repair deficiency. They have also been shown to be clinically effective in cancers with poor DNA repair due to germ-line mutations in BRCA1 and BRCA2, which are estimated to be 5%-7% of patients with pancreatic cancer. A randomized phase II trial of cisplatin plus gemcitabine with or without veliparib, which is a selective PARP inhibitor, in locally advanced or metastatic pancreatic cancer patients is ongoing (NCT01585805). Another PARP inhibitor, Olaparib, at 100 mg b.i.d. with 600 mg/m2 of gemcitabine is tolerated in patients with advanced pancreatic cancer[79]. A randomized multi-center phase I/II trial is currently investigating the effects of irinotecan, cisplatin, and mitomycin C combined with olaparib in patients with advanced pancreatic cancer (NCT01296763). Conversely, a randomized, double blind phase III study of olaparib monotherapy in patients with BRCA1/2 mutated metastatic pancreatic cancer who have not progressed from first line platinum-based chemotherapy is also now underway (NCT02184195).
Despite numerous clinical trials utilizing known targeted agents, the overall advancement made in pancreatic cancer treatment has been relatively modest in comparison to the advancement made in the treatment of other types of tumors. Therefore, the exploration of novel agents targeted at certain signaling pathways is one of the most important undertakings to improve the outcome of patients with lethal pancreatic cancer. In this section, we will focus on the recent experimental studies that may open the door to the development of novel and hopefully more effective strategies for the treatment of pancreatic cancer. A list of novel therapeutic targets and drugs is presented in Table 2.
| Targeted therapeutics | Ref. | Cell lines (cell type) | Main results |
| Multikinase inhibitors | |||
| Foretinib | Chen et al[32] | Panc-1(P) | Foretinib inhibited tumor growth, angiogenesis and lymphangiogenesis in xenograft animals, by inhibiting c-MET but VEGFR-2, VEGFR-3, and TIE-2 signaling |
| SKLB261 | Pan et al[80] | BxPC-3 (P), | Application of SKLB261 resulted in more potent antitumor activities than dasatinib, gemcitabine, or erlotinib in pancreatic cancer xenografts |
| Panc-1 (P), | |||
| AsPC-1 (S), HPAC (P) | |||
| Nintedanib | Awasthi et al[81] | AsPC-1 (S), | A triple angiokinase inhibitor, nintedanib inhibited growth of pancreatic cancer cell lines, with gemcitabine enhancing inhibitory effects |
| BxPC-3 (P), | |||
| Panc-1 (P), | |||
| MIA-PaCa-2 (P), | |||
| Dual inhibition | |||
| Lapatinib and trametinib | Lindberg et al[101] | MAD 08-608, 08-738, 09-366 (P) | Dual anti-EGFR and anti-HER2 therapy significantly enhanced the growth inhibitory effects of the MEK1/2 inhibitor trametinib |
| ZSTK474 and RO5126766 | Van Dort et al[99] | Panc-1 (P) | PI3K inhibitor and the Raf/MEK inhibitor RO5126766 resulted in high in vitro inhibition of both PI3Kand MEK1 also decreased cellular viability in pancreatic cancer cell line |
| NVP-AEW541 and lapatinib | Urtasun et al[42] | NP-9, -18, -29 (P) | Combined treatment with the IGF-IR and EGFR/Her-2 inhibitors synergistically inhibited pancreatic cancer cell growth which is associated with abolishment of Akt, Efk, and IRS-1 activity |
| CP15T, ,15A (p) | |||
| Novel pathways | |||
| PX-478 (HIF-a) | Zhao et al[84] | CFPAC-1 (S), BxPC-3 (P), | Combined treatment with gemcitabine/PX-478 significantly enhanced the anti-tumor effect which is associated with immunogenic cell death |
| Panc-1 (P), | |||
| MIA-PaCa-2 (P) | |||
| SB216763(GSK-3b) | Marchand et al[86] | Panc-1 (P), | Inhibition of GSK-3βn induced apoptosis by mechanism involving JNK-cJUN activation |
| MIA-PaCa-2 (P), BxPC-3 (P) | |||
| Micro RNA | |||
| miR-142-3p | MacKenzie et al[95] | MIA-PaCa-2 (P), Capan-1(S), | A unique HSP70 inhibiting compounds, miR-142-3p regulate triptolide-induced inhibition of pancreatic cancer growth |
| HEK-293 (P), | |||
| S2-013 (P) | |||
| miR-146a | Li et al[96] | AsPC-1 (s), | miR-146a takes significant roles in pancreatic cancer invasion and metastasis but lower expressed in pancreatic cancer compared with normal pancreatic tissue |
| Panc-1 (P) | |||
| miR-494 | Li et al[97] | Colo357 (s), | miR-494, identified to affect levels of FOXM1 in pancreatic cancer cell lines and act as a negative regulator of this transcriptional activator, blocked nuclear translocation β-catenin |
| Panc-1 (P) |
There is growing evidence to suggest that novel tyrosine kinase inhibitors can target multiple different pathways and/or signaling processes that have never been seen before in pancreatic cancer. As previously explained, utilizing the multikinase inhibitor, foretinib, may provide a simultaneous inhibitory effect on pancreatic cancer[32]. SKLB261 is a multikinase inhibitor obtained recently through lead optimization with reported activity against EGFR, Src, and VEGFR2. The application of SKLB261 has resulted in more potent antitumor activities than that of dasatinib, gemcitabine, or erlotinib in pancreatic cancer xenografts as well as more prolonged survival in mice compared with gemcitabine-treated groups[80]. Nintedanib, a triple angiokinase inhibitor that targets the VEGFR1/2/3, FGFR1/2/3, and PDGFRα/β pathways, has been shown to strongly inhibit the growth of pancreatic cancer cell lines in addition to enhancing the inhibitory effects of gemcitabine. Nintedanib also induces apoptosis in pancreatic cancer cells associated with stromal cells, providing a strong rationale for the clinical evaluation of nintedanib combined with conventional cytotoxic agents[81].
Masitinib, a multi-targeted protein tyrosine kinase inhibitor with possible anticancer activity, selectively binds to and inhibits both the wild-type and mutated forms of the stem cell factor receptor (c-Kit; SCFR), PDGFR, and FGFR3. In the recently conducted randomized phase III trial, masitinib combined with gemcitabine prolonged the survival of patients in subgroups defined by an overexpression of acyl-CoA oxidase-1 (ACOX1) in secondary analyses. In the ACOX1 subgroup, the patients treated with masitinib plus gemcitabine showed a median OS of 11.7 mo (95%CI: 8.3-19.9) compared with a median OS of 5.6 mo (95%CI: 3.7-12.9) for the placebo plus gemcitabine treatment-arm[82].
Hypoxia inducible factor-1 (HIF-1) is a principal mediator of cell adaption to hypoxia, and is extensively expressed in 88% of pancreatic tumors[83]. PX-478, an HIF-1 inhibitor, has been shown to promote the anti-cancer effects of gemcitabine, which are closely associated with immunogenic cell death in pancreatic cancer[84]. Recent studies have demonstrated that GSK3β is closely related to pancreatic cancer cell growth, providing a rationale for targeting GSK3β in the treatment of patients with pancreatic cancer[85]. GSK3β inhibition has been shown to induce apoptosis by a mechanism involving JNK-cJUN activation[86]. CXCR4 (CXC chemokine receptor type 4), a G-protein coupled receptor of CXCL12 (SDF-1), has been found to promote GSK3β expression and the invasion ability of pancreatic cancer cells by Akt signaling. This finding suggests that CXCR4 inhibition may open a new therapeutic avenue that will impact the capacity to effectively treat pancreatic cancer patients[87]. Forkhead box M1 (FOXM1) is a transcription factor in the FOX protein superfamily containing a conserved winged helix DNA-binding domain[88]. FOXM1 is an important transcription factor for many genes key to regulating a variety of processes in tumor pathogenesis, such as tumor cell survival, growth, and EMT. Several bodies of evidence have demonstrated that FOXM1 plays a crucial role in pancreatic cancer progression[89]. A recent paper asserted that FOXM1 contributes to the development of pancreatic cancer by enhancing the uPAR gene transcription and subsequently the EMT of cancer[90]. These findings suggest that the deregulation of FOXM1 signaling may be a new attractive strategy in the development of novel therapeutic targets to control pancreatic cancer.
Recently discovered micro RNA (miRNA) are short non-coding RNAs involved in the negative regulation of miRNAs translation. It is important to note that miRNAs mediate a variety of cellular functions and their dysregulation is a crucial event in tumor initiation[91]. The expression of miRNAs in pancreatic cancer cells differs from those in normal pancreatic cells and in patients with chronic pancreatitis. Over 130 miRNAs have been proven to be deregulated in pancreatic cancer[92]. Several of these miRNAs play a role in new therapeutic prospects in the treatment of pancreatic cancer. It has been shown that nanomolar concentrations of antisense miR-21 and miR-221 oligonucleotides significantly repress their targets and reduce the growth of pancreatic cancer cell lines[93,94]. A unique HSP70 inhibiting compound, miR-142-3p, regulates the triptolide, a diterpene triepoxide extract from the Chinese herb Tripterygium wilfordii, -induced inhibition of pancreatic cancer growth[95]. The miR-146a compound plays a significant role in pancreatic cancer invasion and metastasis[96]. Interestingly, miR-146a has been shown to restore the proteolytic activity of pancreatic cancer cells but with less expression in pancreatic cancer than in normal pancreatic tissue. Utilizing isoflavones or 3,3’-diinodolylmethane has been found to increase the expression of miR-146a, which may be a promising approach to blocking the invasion of pancreatic cancer. MicroRNA-494 (miR-494) is known to affect levels of FOXM1 in pancreatic cancer cell lines and act as a negative regulator of this transcriptional activator. It has also been shown to block nuclear translocation of β-catenin, which leads to cell proliferation, migration, and the increase of the sensitivity of pancreatic cancer cells to gemcitabine[97].
Pancreatic cancer illustrates genetic heterogeneity, and the complications in molecular signaling crosstalk cause the failure of existing treatment strategies. Currently, only a few targeted agents, including erlotinib, have yielded a significant survival benefit for patients with pancreatic cancer. To overcome these obstacles, a considerable effort needs to be put into investigating the effective use of these therapies. Another challenge in evaluating novel targeted therapies in pancreatic cancer treatment is to identify underlying pathological features and incorporate them into trials with molecular biomarkers. Analysis of pancreatic cancer pathogenesis with its molecular characteristics may help to identify the biomarker-defined subsets of patients that can be targeted to optimize the therapeutic benefit[98].
The identification of effective targeted combination therapies may be useful for generating enhanced strategies of treating pancreatic cancer. For instance, suppression of the PI3K/Akt /mTOR pathway may result in an escape via the MAPK pathway as well as in diminished effects due to the intensive crosstalk between these pathways. Therefore, a combination of agents that inhibit separate pathways may be crucial for achieving the desired efficacy against tumors. A recent study found that the application of a prototype dual-acting agent designed using the PI3K inhibitor, ZSTK474, and the Raf/MEK inhibitor, RO5126766, resulted in high in vitro inhibition of both PI3K and MEK1 as well as decreased cellular viability in pancreatic cancer cell lines[99]. In a previous study, the combined effect of the cyclin-dependent kinase inhibitor, dinaciclib, and the pan-Akt inhibitor, MK-2206, dramatically inhibited tumor growth and metastasis in eight pancreatic cancer models. Remarkably, several complete responses were induced by this combination treatment. These results suggest that blocking RAF in combination with other effector pathways downstream from KRAS may provide increased efficacy in pancreatic cancer treatment[100]. Notably, preclinical studies predicting the effects of combination therapies with EGFR and other pathway inhibitors in pancreatic cancer xenografts have presented promising results. A recent study asserted that dual anti-EGFR and anti-HER2 therapy significantly enhanced the growth inhibitory effects of the MEK1/2 inhibitor, trametinib, in different patient-derived pancreatic cancer xenografts. This highlights the importance of designing therapeutic interventions that target not only the EGFR-HER2 but also the KRAS pathways to achieve maximal therapeutic efficacy[101].
Selecting drug combinations with novel agents that target not only tumor initiation but also the surrounding stroma may be one such approach. There is growing evidence to suggest that proteins expressed by stromal cells (Cox-2, stromal-derived factor, integrins, secreted protein acidic and rich in cysteine, and HH elements) are related to poor outcomes and the resistance to current therapies[102]. In the meantime, it is also essential to pay attention to the toxicity resulting from the use of targeted agents with conventional chemotherapy or agents targeting other pathways. A thorough understanding of the underlying mechanisms involved in toxicity will be crucial to furthering the development of pancreatic cancer therapies.
Thus far, most clinical trials involving targeted therapies for pancreatic cancer have not yielded successful results. The lack of effectiveness of several targeted agents in pancreatic cancer, despite their success in treating other types of malignancies, suggests that pancreatic cancer has particularly challenging biological characteristics that have yet to be well defined. As discussed in this review, several experimental studies have demonstrated promising new pathogeneses and efficacy in the treatment of pancreatic cancer. Therefore, continuing to develop and investigate treatments that fight pancreatic cancer from as many angles as possible will likely increase the chances of achieving positive outcomes for patients.
P- Reviewer: Talukdar R S- Editor: Ma YJ L- Editor: Filipodia E- Editor: Wang CH
| 1. | Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10-29. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8406] [Cited by in F6Publishing: 8928] [Article Influence: 744.0] [Reference Citation Analysis (0)] |
| 2. | Oettle H, Post S, Neuhaus P, Gellert K, Langrehr J, Ridwelski K, Schramm H, Fahlke J, Zuelke C, Burkart C. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297:267-277. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1779] [Cited by in F6Publishing: 1708] [Article Influence: 100.5] [Reference Citation Analysis (0)] |
| 3. | Borazanci E, Von Hoff DD. Nab-paclitaxel and gemcitabine for the treatment of patients with metastatic pancreatic cancer. Expert Rev Gastroenterol Hepatol. 2014;8:739-747. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 4. | Thota R, Pauff JM, Berlin JD. Treatment of metastatic pancreatic adenocarcinoma: a review. Oncology (Williston Park). 2014;28:70-74. [PubMed] [Cited in This Article: ] |
| 5. | Jones S, Zhang X, Parsons DW, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Kamiyama H, Jimeno A. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science. 2008;321:1801-1806. [PubMed] [Cited in This Article: ] |
| 6. | Rozenblum E, Schutte M, Goggins M, Hahn SA, Panzer S, Zahurak M, Goodman SN, Sohn TA, Hruban RH, Yeo CJ. Tumor-suppressive pathways in pancreatic carcinoma. Cancer Res. 1997;57:1731-1734. [PubMed] [Cited in This Article: ] |
| 7. | Yashiro M, Carethers JM, Laghi L, Saito K, Slezak P, Jaramillo E, Rubio C, Koizumi K, Hirakawa K, Boland CR. Genetic pathways in the evolution of morphologically distinct colorectal neoplasms. Cancer Res. 2001;61:2676-2683. [PubMed] [Cited in This Article: ] |
| 8. | Moskaluk CA, Hruban RH, Kern SE. p16 and K-ras gene mutations in the intraductal precursors of human pancreatic adenocarcinoma. Cancer Res. 1997;57:2140-2143. [PubMed] [Cited in This Article: ] |
| 9. | Hruban RH, van Mansfeld AD, Offerhaus GJ, van Weering DH, Allison DC, Goodman SN, Kensler TW, Bose KK, Cameron JL, Bos JL. K-ras oncogene activation in adenocarcinoma of the human pancreas. A study of 82 carcinomas using a combination of mutant-enriched polymerase chain reaction analysis and allele-specific oligonucleotide hybridization. Am J Pathol. 1993;143:545-554. [PubMed] [Cited in This Article: ] |
| 10. | Schutte M, Hruban RH, Geradts J, Maynard R, Hilgers W, Rabindran SK, Moskaluk CA, Hahn SA, Schwarte-Waldhoff I, Schmiegel W. Abrogation of the Rb/p16 tumor-suppressive pathway in virtually all pancreatic carcinomas. Cancer Res. 1997;57:3126-3130. [PubMed] [Cited in This Article: ] |
| 11. | Muller PA, Vousden KH. p53 mutations in cancer. Nat Cell Biol. 2013;15:2-8. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1319] [Cited by in F6Publishing: 1358] [Article Influence: 123.5] [Reference Citation Analysis (0)] |
| 12. | Iacobuzio-Donahue CA, Song J, Parmiagiani G, Yeo CJ, Hruban RH, Kern SE. Missense mutations of MADH4: characterization of the mutational hot spot and functional consequences in human tumors. Clin Cancer Res. 2004;10:1597-1604. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 80] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 13. | Blackford A, Serrano OK, Wolfgang CL, Parmigiani G, Jones S, Zhang X, Parsons DW, Lin JC, Leary RJ, Eshleman JR. SMAD4 gene mutations are associated with poor prognosis in pancreatic cancer. Clin Cancer Res. 2009;15:4674-4679. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 259] [Cited by in F6Publishing: 282] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 14. | Hezel AF, Kimmelman AC, Stanger BZ, Bardeesy N, Depinho RA. Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev. 2006;20:1218-1249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 803] [Cited by in F6Publishing: 842] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 15. | Calhoun ES, Jones JB, Ashfaq R, Adsay V, Baker SJ, Valentine V, Hempen PM, Hilgers W, Yeo CJ, Hruban RH. BRAF and FBXW7 (CDC4, FBW7, AGO, SEL10) mutations in distinct subsets of pancreatic cancer: potential therapeutic targets. Am J Pathol. 2003;163:1255-1260. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 174] [Cited by in F6Publishing: 170] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 16. | Hingorani SR, Petricoin EF, Maitra A, Rajapakse V, King C, Jacobetz MA, Ross S, Conrads TP, Veenstra TD, Hitt BA. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell. 2003;4:437-450. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1827] [Cited by in F6Publishing: 1813] [Article Influence: 86.3] [Reference Citation Analysis (0)] |
| 17. | Ardito CM, Grüner BM, Takeuchi KK, Lubeseder-Martellato C, Teichmann N, Mazur PK, Delgiorno KE, Carpenter ES, Halbrook CJ, Hall JC. EGF receptor is required for KRAS-induced pancreatic tumorigenesis. Cancer Cell. 2012;22:304-317. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 380] [Cited by in F6Publishing: 403] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 18. | Collins MA, Bednar F, Zhang Y, Brisset JC, Galbán S, Galbán CJ, Rakshit S, Flannagan KS, Adsay NV, Pasca di Magliano M. Oncogenic Kras is required for both the initiation and maintenance of pancreatic cancer in mice. J Clin Invest. 2012;122:639-653. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 500] [Cited by in F6Publishing: 557] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 19. | Cantley LC. The phosphoinositide 3-kinase pathway. Science. 2002;296:1655-1657. [PubMed] [Cited in This Article: ] |
| 20. | Altomare DA, Tanno S, De Rienzo A, Klein-Szanto AJ, Tanno S, Skele KL, Hoffman JP, Testa JR. Frequent activation of AKT2 kinase in human pancreatic carcinomas. J Cell Biochem. 2002;87:470-476. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 114] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 21. | Hirakawa T, Yashiro M, Murata A, Hirata K, Kimura K, Amano R, Yamada N, Nakata B, Hirakawa K. IGF-1 receptor and IGF binding protein-3 might predict prognosis of patients with resectable pancreatic cancer. BMC Cancer. 2013;13:392. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Neesse A, Michl P, Frese KK, Feig C, Cook N, Jacobetz MA, Lolkema MP, Buchholz M, Olive KP, Gress TM. Stromal biology and therapy in pancreatic cancer. Gut. 2011;60:861-868. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 541] [Cited by in F6Publishing: 603] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 23. | Whipple C, Korc M. Targeting angiogenesis in pancreatic cancer: rationale and pitfalls. Langenbecks Arch Surg. 2008;393:901-910. [PubMed] [Cited in This Article: ] |
| 24. | Koch S, Claesson-Welsh L. Signal transduction by vascular endothelial growth factor receptors. Cold Spring Harb Perspect Med. 2012;2:a006502. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 535] [Cited by in F6Publishing: 595] [Article Influence: 54.1] [Reference Citation Analysis (0)] |
| 25. | Seo Y, Baba H, Fukuda T, Takashima M, Sugimachi K. High expression of vascular endothelial growth factor is associated with liver metastasis and a poor prognosis for patients with ductal pancreatic adenocarcinoma. Cancer. 2000;88:2239-2245. [PubMed] [Cited in This Article: ] |
| 26. | Kindler HL, Niedzwiecki D, Hollis D, Sutherland S, Schrag D, Hurwitz H, Innocenti F, Mulcahy MF, O’Reilly E, Wozniak TF. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28:3617-3622. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 647] [Cited by in F6Publishing: 655] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 27. | Cardin DB, Goff L, Li CI, Shyr Y, Winkler C, DeVore R, Schlabach L, Holloway M, McClanahan P, Meyer K. Phase II trial of sorafenib and erlotinib in advanced pancreatic cancer. Cancer Med. 2014;3:572-579. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 28. | Saif MW. Pancreatic cancer: Sorafenib: no effect on efficacy of chemotherapy in pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2014;11:8-9. [PubMed] [Cited in This Article: ] |
| 29. | Ioka T, Okusaka T, Ohkawa S, Boku N, Sawaki A, Fujii Y, Kamei Y, Takahashi S, Namazu K, Umeyama Y. Efficacy and safety of axitinib in combination with gemcitabine in advanced pancreatic cancer: subgroup analyses by region, including Japan, from the global randomized Phase III trial. Jpn J Clin Oncol. 2015;45:439-448. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Kataoka Y, Mukohara T, Tomioka H, Funakoshi Y, Kiyota N, Fujiwara Y, Yashiro M, Hirakawa K, Hirai M, Minami H. Foretinib (GSK1363089), a multi-kinase inhibitor of MET and VEGFRs, inhibits growth of gastric cancer cell lines by blocking inter-receptor tyrosine kinase networks. Invest New Drugs. 2012;30:1352-1360. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 31. | Kiehne K, Herzig KH, Fölsch UR. c-met expression in pancreatic cancer and effects of hepatocyte growth factor on pancreatic cancer cell growth. Pancreas. 1997;15:35-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 47] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 32. | Chen HM, Tsai CH, Hung WC. Foretinib inhibits angiogenesis, lymphangiogenesis and tumor growth of pancreatic cancer in vivo by decreasing VEGFR-2/3 and TIE-2 signaling. Oncotarget. 2015;6:14940-14952. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 33. | Voldborg BR, Damstrup L, Spang-Thomsen M, Poulsen HS. Epidermal growth factor receptor (EGFR) and EGFR mutations, function and possible role in clinical trials. Ann Oncol. 1997;8:1197-1206. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 266] [Cited by in F6Publishing: 298] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 34. | Tobita K, Kijima H, Dowaki S, Kashiwagi H, Ohtani Y, Oida Y, Yamazaki H, Nakamura M, Ueyama Y, Tanaka M. Epidermal growth factor receptor expression in human pancreatic cancer: Significance for liver metastasis. Int J Mol Med. 2003;11:305-309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 35. | Yamanaka Y, Friess H, Kobrin MS, Buchler M, Beger HG, Korc M. Coexpression of epidermal growth factor receptor and ligands in human pancreatic cancer is associated with enhanced tumor aggressiveness. Anticancer Res. 1993;13:565-569. [PubMed] [Cited in This Article: ] |
| 36. | Philip PA, Benedetti J, Corless CL, Wong R, O’Reilly EM, Flynn PJ, Rowland KM, Atkins JN, Mirtsching BC, Rivkin SE. Phase III study comparing gemcitabine plus cetuximab versus gemcitabine in patients with advanced pancreatic adenocarcinoma: Southwest Oncology Group-directed intergroup trial S0205. J Clin Oncol. 2010;28:3605-3610. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 446] [Cited by in F6Publishing: 484] [Article Influence: 34.6] [Reference Citation Analysis (0)] |
| 37. | Safran H, Iannitti D, Ramanathan R, Schwartz JD, Steinhoff M, Nauman C, Hesketh P, Rathore R, Wolff R, Tantravahi U. Herceptin and gemcitabine for metastatic pancreatic cancers that overexpress HER-2/neu. Cancer Invest. 2004;22:706-712. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 123] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 38. | Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, Au HJ, Murawa P, Walde D, Wolff RA. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25:1960-1966. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2835] [Cited by in F6Publishing: 2707] [Article Influence: 159.2] [Reference Citation Analysis (0)] |
| 39. | da Cunha Santos G, Dhani N, Tu D, Chin K, Ludkovski O, Kamel-Reid S, Squire J, Parulekar W, Moore MJ, Tsao MS. Molecular predictors of outcome in a phase 3 study of gemcitabine and erlotinib therapy in patients with advanced pancreatic cancer: National Cancer Institute of Canada Clinical Trials Group Study PA.3. Cancer. 2010;116:5599-5607. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 116] [Cited by in F6Publishing: 134] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 40. | Togashi Y, Hayashi H, Nakagawa K, Nishio K. Clinical utility of erlotinib for the treatment of non-small-cell lung cancer in Japanese patients: current evidence. Drug Des Devel Ther. 2014;8:1037-1046. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 41. | Pollak MN, Schernhammer ES, Hankinson SE. Insulin-like growth factors and neoplasia. Nat Rev Cancer. 2004;4:505-518. [PubMed] [Cited in This Article: ] |
| 42. | Urtasun N, Vidal-Pla A, Pérez-Torras S, Mazo A. Human pancreatic cancer stem cells are sensitive to dual inhibition of IGF-IR and ErbB receptors. BMC Cancer. 2015;15:223. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 43. | De Luca A, Maiello MR, D’Alessio A, Pergameno M, Normanno N. The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin Ther Targets. 2012;16 Suppl 2:S17-S27. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 458] [Cited by in F6Publishing: 520] [Article Influence: 43.3] [Reference Citation Analysis (0)] |
| 44. | Willems L, Tamburini J, Chapuis N, Lacombe C, Mayeux P, Bouscary D. PI3K and mTOR signaling pathways in cancer: new data on targeted therapies. Curr Oncol Rep. 2012;14:129-138. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 132] [Cited by in F6Publishing: 149] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 45. | Schlieman MG, Fahy BN, Ramsamooj R, Beckett L, Bold RJ. Incidence, mechanism and prognostic value of activated AKT in pancreas cancer. Br J Cancer. 2003;89:2110-2115. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 175] [Cited by in F6Publishing: 200] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 46. | Asano T, Yao Y, Zhu J, Li D, Abbruzzese JL, Reddy SA. The PI 3-kinase/Akt signaling pathway is activated due to aberrant Pten expression and targets transcription factors NF-kappaB and c-Myc in pancreatic cancer cells. Oncogene. 2004;23:8571-8580. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 225] [Cited by in F6Publishing: 235] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 47. | Matsuzaki T, Yashiro M, Kaizaki R, Yasuda K, Doi Y, Sawada T, Ohira M, Hirakawa K. Synergistic antiproliferative effect of mTOR inhibitors in combination with 5-fluorouracil in scirrhous gastric cancer. Cancer Sci. 2009;100:2402-2410. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 48. | Kordes S, Klümpen HJ, Weterman MJ, Schellens JH, Richel DJ, Wilmink JW. Phase II study of capecitabine and the oral mTOR inhibitor everolimus in patients with advanced pancreatic cancer. Cancer Chemother Pharmacol. 2015;75:1135-1141. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 41] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 49. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9016] [Cited by in F6Publishing: 9738] [Article Influence: 608.6] [Reference Citation Analysis (1)] |
| 50. | Thomas SM, Brugge JS. Cellular functions regulated by Src family kinases. Annu Rev Cell Dev Biol. 1997;13:513-609. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1956] [Cited by in F6Publishing: 1960] [Article Influence: 75.4] [Reference Citation Analysis (0)] |
| 51. | Jatiani SS, Baker SJ, Silverman LR, Reddy EP. Jak/STAT pathways in cytokine signaling and myeloproliferative disorders: approaches for targeted therapies. Genes Cancer. 2010;1:979-993. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 145] [Cited by in F6Publishing: 155] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 52. | Seidel HM, Lamb P, Rosen J. Pharmaceutical intervention in the JAK/STAT signaling pathway. Oncogene. 2000;19:2645-2656. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 75] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 53. | Müller S, Raulefs S, Bruns P, Afonso-Grunz F, Plötner A, Thermann R, Jäger C, Schlitter AM, Kong B, Regel I. Next-generation sequencing reveals novel differentially regulated mRNAs, lncRNAs, miRNAs, sdRNAs and a piRNA in pancreatic cancer. Mol Cancer. 2015;14:94. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 159] [Cited by in F6Publishing: 194] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 54. | Lili LN, Matyunina LV, Walker LD, Daneker GW, McDonald JF. Evidence for the importance of personalized molecular profiling in pancreatic cancer. Pancreas. 2014;43:198-211. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 55. | Tanase CP, Neagu AI, Necula LG, Mambet C, Enciu AM, Calenic B, Cruceru ML, Albulescu R. Cancer stem cells: involvement in pancreatic cancer pathogenesis and perspectives on cancer therapeutics. World J Gastroenterol. 2014;20:10790-10801. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 37] [Cited by in F6Publishing: 40] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 56. | Rao CV, Mohammed A. New insights into pancreatic cancer stem cells. World J Stem Cells. 2015;7:547-555. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 57. | Castellanos JA, Merchant NB, Nagathihalli NS. Emerging targets in pancreatic cancer: epithelial-mesenchymal transition and cancer stem cells. Onco Targets Ther. 2013;6:1261-1267. [PubMed] [Cited in This Article: ] |
| 58. | Leach SD. Epithelial differentiation in pancreatic development and neoplasia: new niches for nestin and Notch. J Clin Gastroenterol. 2005;39:S78-S82. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 59. | Avila JL, Kissil JL. Notch signaling in pancreatic cancer: oncogene or tumor suppressor? Trends Mol Med. 2013;19:320-327. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 78] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 60. | Ristorcelli E, Lombardo D. Targeting Notch signaling in pancreatic cancer. Expert Opin Ther Targets. 2010;14:541-552. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 61. | Wang Z, Ahmad A, Li Y, Azmi AS, Miele L, Sarkar FH. Targeting notch to eradicate pancreatic cancer stem cells for cancer therapy. Anticancer Res. 2011;31:1105-1113. [PubMed] [Cited in This Article: ] |
| 62. | Chen JK, Taipale J, Young KE, Maiti T, Beachy PA. Small molecule modulation of Smoothened activity. Proc Natl Acad Sci USA. 2002;99:14071-14076. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 815] [Cited by in F6Publishing: 790] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 63. | Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernández-del Castillo C, Yajnik V. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature. 2003;425:851-856. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1151] [Cited by in F6Publishing: 1140] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 64. | Pasca di Magliano M, Sekine S, Ermilov A, Ferris J, Dlugosz AA, Hebrok M. Hedgehog/Ras interactions regulate early stages of pancreatic cancer. Genes Dev. 2006;20:3161-3173. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 234] [Cited by in F6Publishing: 249] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 65. | Bailey JM, Swanson BJ, Hamada T, Eggers JP, Singh PK, Caffery T, Ouellette MM, Hollingsworth MA. Sonic hedgehog promotes desmoplasia in pancreatic cancer. Clin Cancer Res. 2008;14:5995-6004. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 370] [Cited by in F6Publishing: 397] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 66. | Tanaka S. Cancer stem cells as therapeutic targets of hepato-biliary-pancreatic cancers. J Hepatobiliary Pancreat Sci. 2015;22:531-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 67. | Olive KP, Jacobetz MA, Davidson CJ, Gopinathan A, McIntyre D, Honess D, Madhu B, Goldgraben MA, Caldwell ME, Allard D. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457-1461. [PubMed] [Cited in This Article: ] |
| 68. | Kim EJ, Sahai V, Abel EV, Griffith KA, Greenson JK, Takebe N, Khan GN, Blau JL, Craig R, Balis UG. Pilot clinical trial of hedgehog pathway inhibitor GDC-0449 (vismodegib) in combination with gemcitabine in patients with metastatic pancreatic adenocarcinoma. Clin Cancer Res. 2014;20:5937-5945. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 192] [Cited by in F6Publishing: 224] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 69. | Baarsma HA, Königshoff M, Gosens R. The WNT signaling pathway from ligand secretion to gene transcription: molecular mechanisms and pharmacological targets. Pharmacol Ther. 2013;138:66-83. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 119] [Cited by in F6Publishing: 127] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 70. | Espada J, Calvo MB, Díaz-Prado S, Medina V. Wnt signalling and cancer stem cells. Clin Transl Oncol. 2009;11:411-427. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 73] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 71. | Cui J, Jiang W, Wang S, Wang L, Xie K. Role of Wnt/β-catenin signaling in drug resistance of pancreatic cancer. Curr Pharm Des. 2012;18:2464-2471. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 110] [Cited by in F6Publishing: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 72. | Fuxe J, Karlsson MC. TGF-β-induced epithelial-mesenchymal transition: a link between cancer and inflammation. Semin Cancer Biol. 2012;22:455-461. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 145] [Cited by in F6Publishing: 164] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 73. | Oberstein PE, Olive KP. Pancreatic cancer: why is it so hard to treat? Therap Adv Gastroenterol. 2013;6:321-337. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 206] [Cited by in F6Publishing: 217] [Article Influence: 19.7] [Reference Citation Analysis (1)] |
| 74. | Yashiro M, Chung YS, Sowa M. Role of orthotopic fibroblasts in the development of scirrhous gastric carcinoma. Jpn J Cancer Res. 1994;85:883-886. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 75. | Yashiro M, Ikeda K, Tendo M, Ishikawa T, Hirakawa K. Effect of organ-specific fibroblasts on proliferation and differentiation of breast cancer cells. Breast Cancer Res Treat. 2005;90:307-313. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 76. | Rucki AA, Zheng L. Pancreatic cancer stroma: understanding biology leads to new therapeutic strategies. World J Gastroenterol. 2014;20:2237-2246. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 87] [Cited by in F6Publishing: 98] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 77. | Zhou H, Roy S, Cochran E, Zouaoui R, Chu CL, Duffner J, Zhao G, Smith S, Galcheva-Gargova Z, Karlgren J. M402, a novel heparan sulfate mimetic, targets multiple pathways implicated in tumor progression and metastasis. PLoS One. 2011;6:e21106. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 123] [Cited by in F6Publishing: 139] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 78. | Helleday T, Bryant HE, Schultz N. Poly(ADP-ribose) polymerase (PARP-1) in homologous recombination and as a target for cancer therapy. Cell Cycle. 2005;4:1176-1178. [PubMed] [Cited in This Article: ] |
| 79. | Bendell J, O’Reilly EM, Middleton MR, Chau I, Hochster H, Fielding A, Burke W, Burris H. Phase I study of olaparib plus gemcitabine in patients with advanced solid tumours and comparison with gemcitabine alone in patients with locally advanced/metastatic pancreatic cancer. Ann Oncol. 2015;26:804-811. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 80. | Pan Y, Zheng M, Zhong L, Yang J, Zhou S, Qin Y, Xiang R, Chen Y, Yang SY. A preclinical evaluation of SKLB261, a multikinase inhibitor of EGFR/Src/VEGFR2, as a therapeutic agent against pancreatic cancer. Mol Cancer Ther. 2015;14:407-418. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 81. | Awasthi N, Hinz S, Brekken RA, Schwarz MA, Schwarz RE. Nintedanib, a triple angiokinase inhibitor, enhances cytotoxic therapy response in pancreatic cancer. Cancer Lett. 2015;358:59-66. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 82. | Deplanque G, Demarchi M, Hebbar M, Flynn P, Melichar B, Atkins J, Nowara E, Moyé L, Piquemal D, Ritter D. A randomized, placebo-controlled phase III trial of masitinib plus gemcitabine in the treatment of advanced pancreatic cancer. Ann Oncol. 2015;26:1194-1200. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 71] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 83. | Shibaji T, Nagao M, Ikeda N, Kanehiro H, Hisanaga M, Ko S, Fukumoto A, Nakajima Y. Prognostic significance of HIF-1 alpha overexpression in human pancreatic cancer. Anticancer Res. 2003;23:4721-4727. [PubMed] [Cited in This Article: ] |
| 84. | Zhao T, Ren H, Jia L, Chen J, Xin W, Yan F, Li J, Wang X, Gao S, Qian D. Inhibition of HIF-1α by PX-478 enhances the anti-tumor effect of gemcitabine by inducing immunogenic cell death in pancreatic ductal adenocarcinoma. Oncotarget. 2015;6:2250-2262. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 99] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 85. | Miyashita K, Nakada M, Shakoori A, Ishigaki Y, Shimasaki T, Motoo Y, Kawakami K, Minamoto T. An emerging strategy for cancer treatment targeting aberrant glycogen synthase kinase 3 beta. Anticancer Agents Med Chem. 2009;9:1114-1122. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 86. | Marchand B, Tremblay I, Cagnol S, Boucher MJ. Inhibition of glycogen synthase kinase-3 activity triggers an apoptotic response in pancreatic cancer cells through JNK-dependent mechanisms. Carcinogenesis. 2012;33:529-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 87. | Ma S, Li Q, Pan F. CXCR4 promotes GSK3β expression in pancreatic cancer cells via the Akt pathway. Int J Clin Oncol. 2015;20:525-530. [PubMed] [Cited in This Article: ] |
| 88. | Clark KL, Halay ED, Lai E, Burley SK. Co-crystal structure of the HNF-3/fork head DNA-recognition motif resembles histone H5. Nature. 1993;364:412-420. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 957] [Cited by in F6Publishing: 992] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 89. | Wang Z, Banerjee S, Kong D, Li Y, Sarkar FH. Down-regulation of Forkhead Box M1 transcription factor leads to the inhibition of invasion and angiogenesis of pancreatic cancer cells. Cancer Res. 2007;67:8293-8300. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 192] [Cited by in F6Publishing: 209] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 90. | Huang C, Xie D, Cui J, Li Q, Gao Y, Xie K. FOXM1c promotes pancreatic cancer epithelial-to-mesenchymal transition and metastasis via upregulation of expression of the urokinase plasminogen activator system. Clin Cancer Res. 2014;20:1477-1488. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 55] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 91. | Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281-297. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25833] [Cited by in F6Publishing: 27138] [Article Influence: 1356.9] [Reference Citation Analysis (0)] |
| 92. | Rachagani S, Kumar S, Batra SK. MicroRNA in pancreatic cancer: pathological, diagnostic and therapeutic implications. Cancer Lett. 2010;292:8-16. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 93. | Park WG, Yan BM, Schellenberg D, Kim J, Chang DT, Koong A, Patalano C, Van Dam J. EUS-guided gold fiducial insertion for image-guided radiation therapy of pancreatic cancer: 50 successful cases without fluoroscopy. Gastrointest Endosc. 2010;71:513-518. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 106] [Cited by in F6Publishing: 102] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 94. | Hwang JH, Voortman J, Giovannetti E, Steinberg SM, Leon LG, Kim YT, Funel N, Park JK, Kim MA, Kang GH. Identification of microRNA-21 as a biomarker for chemoresistance and clinical outcome following adjuvant therapy in resectable pancreatic cancer. PLoS One. 2010;5:e10630. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 219] [Cited by in F6Publishing: 228] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 95. | MacKenzie TN, Mujumdar N, Banerjee S, Sangwan V, Sarver A, Vickers S, Subramanian S, Saluja AK. Triptolide induces the expression of miR-142-3p: a negative regulator of heat shock protein 70 and pancreatic cancer cell proliferation. Mol Cancer Ther. 2013;12:1266-1275. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 91] [Cited by in F6Publishing: 106] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 96. | Li Y, Vandenboom TG, Wang Z, Kong D, Ali S, Philip PA, Sarkar FH. miR-146a suppresses invasion of pancreatic cancer cells. Cancer Res. 2010;70:1486-1495. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 319] [Cited by in F6Publishing: 330] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 97. | Li L, Li Z, Kong X, Xie D, Jia Z, Jiang W, Cui J, Du Y, Wei D, Huang S. Down-regulation of microRNA-494 via loss of SMAD4 increases FOXM1 and β-catenin signaling in pancreatic ductal adenocarcinoma cells. Gastroenterology. 2014;147:485-97.e18. [PubMed] [Cited in This Article: ] |
| 98. | Kern SE, Shi C, Hruban RH. The complexity of pancreatic ductal cancers and multidimensional strategies for therapeutic targeting. J Pathol. 2011;223:295-306. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 99. | Van Dort ME, Galbán S, Wang H, Sebolt-Leopold J, Whitehead C, Hong H, Rehemtulla A, Ross BD. Dual inhibition of allosteric mitogen-activated protein kinase (MEK) and phosphatidylinositol 3-kinase (PI3K) oncogenic targets with a bifunctional inhibitor. Bioorg Med Chem. 2015;23:1386-1394. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 100. | Hu C, Dadon T, Chenna V, Yabuuchi S, Bannerji R, Booher R, Strack P, Azad N, Nelkin BD, Maitra A. Combined Inhibition of Cyclin-Dependent Kinases (Dinaciclib) and AKT (MK-2206) Blocks Pancreatic Tumor Growth and Metastases in Patient-Derived Xenograft Models. Mol Cancer Ther. 2015;14:1532-1539. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 101. | Lindberg JM, Newhook TE, Adair SJ, Walters DM, Kim AJ, Stelow EB, Parsons JT, Bauer TW. Co-treatment with panitumumab and trastuzumab augments response to the MEK inhibitor trametinib in a patient-derived xenograft model of pancreatic cancer. Neoplasia. 2014;16:562-571. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 102. | Tang SC, Chen YC. Novel therapeutic targets for pancreatic cancer. World J Gastroenterol. 2014;20:10825-10844. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 56] [Cited by in F6Publishing: 60] [Article Influence: 6.0] [Reference Citation Analysis (0)] |