Published online Nov 21, 2015. doi: 10.3748/wjg.v21.i43.12234
Peer-review started: May 8, 2015
First decision: August 31, 2015
Revised: September 5, 2015
Accepted: October 23, 2015
Article in press: October 26, 2015
Published online: November 21, 2015
Processing time: 202 Days and 17.8 Hours
Irinotecan hydrochloride is a camptothecin derivative that exerts antitumor activity against a variety of tumors. SN-38 produced in the body by carboxylesterase is the active metabolite of irinotecan. After irinotecan was introduced for the treatment of metastatic colorectal cancer (CRC) at the end of the last century, survival has improved dramatically. Irinotecan is now combined with 5-fluorouracil, oxaliplatin and several molecularly-targeted anticancer drugs, resulting in the extension of overall survival to longer than 30 mo. Severe, occasionally life-threatening toxicity occurs sporadically, even in patients in relatively good condition who have a low risk of chemotherapy-induced toxicity, often causing the failure of irinotecan-based chemotherapy. Clinical pharmacological studies have revealed that such severe toxicity is related to exposure to SN-38 and genetic polymorphisms in UDP-glucuronosyltransferase 1A1 gene. The large inter- and intra-patient variability in systemic exposure to SN-38 is determined not only by genetic factors but also by physiological and environmental factors. This review first summarizes the roles of irinotecan in chemotherapy for metastatic CRC and then discusses the optimal dosing of irinotecan based on the aforementioned factors affecting systemic exposure to SN-38, with the ultimate goal of achieving personalized irinotecan-based chemotherapy.
Core tip: Irinotecan is a key anticancer drug for the treatment of metastatic colorectal cancer. By combining irinotecan with 5-fluorouracil, oxaliplatin, and a molecularly-targeted drug, overall survival of longer than 30 mo has been achieved. Exposure to SN-38, the active metabolite of irinotecan, shows large inter- and intra-patient variability and can cause severe irinotecan-related toxicities. Many studies have recommended the dose reduction of irinotecan for patients with UDP-glucuronosyltransferase 1A1 polymorphisms and liver dysfunction. Surprisingly, dose reduction may be required in patients with severe renal failure, even though irinotecan is predominantly eliminated via the liver.
- Citation: Fujita KI, Kubota Y, Ishida H, Sasaki Y. Irinotecan, a key chemotherapeutic drug for metastatic colorectal cancer. World J Gastroenterol 2015; 21(43): 12234-12248
- URL: https://www.wjgnet.com/1007-9327/full/v21/i43/12234.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i43.12234
Irinotecan hydrochloride is an analogue of camptothecin, an extract from the Chinese tree Camptotheca acuminate, with higher aqueous solubility than camptothecin[1]. Irinotecan was a pro-drug that is metabolically activated in the body to 7-ethyl-10-hydroxycamptothecin (SN-38). Irinotecan has a broad spectrum of antitumor activity both in vitro and in vivo[2] and is associated with more predictable and clinically manageable toxicity than the originally isolated structure. After clinical trials, irinotecan became commercially available in Japan for treatment of lung, cervical and ovarian cancers in 1994. Irinotecan was first approved for the treatment of metastatic colorectal cancer (CRC) refractory to 5-fluorouracil (5-FU) in the United States in 1996, followed by approval in combination with 5-FU/leucovorin (LV) for the first-line treatment of metastatic CRC. A wide variety of clinical trials performed to date have revealed a survival advantage of irinotecan-based regimens in patients with metastatic CRC, making irinotecan hydrochloride one of the key drugs for the treatment of metastatic CRC. Recently, overall survival (OS) longer than 30 mo was achieved in patients with metastatic CRC who received irinotecan-based combination chemotherapy[3].
In early clinical development, the dose-limiting toxicity (DLT) of irinotecan hydrochloride was found to be severe neutropenia and delayed diarrhea. Severe, occasionally life-threatening toxicity occurs sporadically, even in patients with relatively good physical condition who have a low risk of chemotherapy-induced toxicity, who are eligible for enrollment in clinical trials of anticancer drugs[4,5]. Interindividual variability in the pharmacokinetics of SN-38 resulting from glucuronide formation is at least one of the major causes of irinotecan-induced severe toxicity[6-8]. Investigators have thus focused primarily on the polymorphic glucuronidation of SN-38 by UDP-glucuronosyltransferase (UGT) 1A1[9], since UGT1A1 is the enzyme primarily involved in endogenous bilirubin glucuronidation as well as in irinotecan glucuronidation. Such studies have shown that genetic polymorphisms in the UGT1A1 gene, such as UGT1A1*28 and UGT1A1*6 are associated with irinotecan-induced severe toxicities[10-13], resulting in revision of the package inserts in the United States, Japan, and other countries, including a recommendation to use a lower initial dose of irinotecan in patients with UGT1A1*28/*28, UGT1A1*6/*6 or UGT1A1*6/*28 genotype. However, pharmacogenetic factors other than UGT1A1, physiological factors, and environmental factors can also cause the large interindividual variability in SN-38 pharmacokinetics and contribute to irinotecan-induced toxicities. Therefore, clinical pharmacological studies are needed to establish personalized dosing strategy for irinotecan-based chemotherapy.
This review article first introduces the general pharmacokinetics and pharmacodynamics of irinotecan and then describes the development of clinical applications of irinotecan in patients with metastatic CRC. We next discuss the optimal dosing of irinotecan on the basis of factors affecting systemic exposure to irinotecan, such as pharmacogenetic factors, physiological factors, and environmental factors, with the ultimate goal of achieving personalized chemotherapy.
Irinotecan is unique among camptothecin analogs in that it must first be converted by a carboxylesterase (CES) to the active metabolite SN-38[14,15]. SN-38 is the major metabolite believed to be responsible for irinotecan’s biologic effects, including efficacy and toxicity. It is subsequently detoxified, predominantly by UGT1A1 in the liver, to form inactive SN-38 glucuronide (SN-38G). Irinotecan is also metabolized in the liver by CYP3A4/5 to form inactive metabolites. In addition to these drug-metabolizing enzymes, transporters expressed in the liver are imprecated in various aspects of SN-38 pharmacokinetics. A primary active transport system is involved in permeation of SN-38 across canalicular membranes in both humans and rats, and ATP-binding cassette (ABC) transporter, subfamily C, member 2 (ABCC2), ABC transporter, subfamily G, number 2 (ABCG2), and ABC transporter, subfamily B, number 1 (ABCB1) play roles in mediating its biliary excretion[16-18] (http://www.pharmgkb.org/do/serve?objId=PA2001&objCls=Pathway). A portion of SN-38 produced in the liver is thought to be transported to the systemic circulation across sinusoidal membranes by unidentified transporter(s), because SN-38 is detectable in plasma immediately after injection of irinotecan in patients with cancer. SN-38 is a substrate of the organic anion transporting polypeptides (OATPs) 1B1 and OATP1B3, which are localized on sinusoidal membranes in humans[19,20] and participate in the uptake of SN-38 into hepatocytes[21]. The contribution of OATP1B1 to the hepatic uptake of SN-38 was higher than that of OATP1B3[21].
The finding that camptothecin induced single-strand DNA breaks in the presence of topoisomerase I led to identifying this enzyme as a major target for the antitumor effects of camptothecin[22]. Subsequent genetic studies with yeast and mammalian cells revealed that the cellular effects of camptothecin can be attributed entirely to its action on topoisomerase I[1]. The lactone form of camptothecin and all its analogues, including irinotecan, appears to reversibly stabilize the topoisomerase I cleavable complex, resulting in single-strand DNA breaks and inhibition of DNA religation. DNA synthesis is thus blocked in the presence of topoisomerase I inhibitors, leading to irreversible inhibition of DNA synthesis with double-strand DNA breaks. These events induce arrest of the cell cycle in the S-G2 phase and ultimately cause cell death[23]. Because the cytotoxicity of topoisomerase I inhibitors is S-G2 phase specific, prolonged infusion times might theoretically enhance the efficacy of irinotecan[14].
Many other factors can potentially affect the pharmacodynamics of irinotecan. Tyrosyl-DNA phosphodiesterase 1 (TDP1) participates in the repair of strand breaks by removing abortive topoisomerase I and DNA complexes. Thus, a role of TDP1 in counteracting DNA damage induced by camptothecins has been proposed[24]. X-ray repair cross complementing group 1 (XRCC1), a scaffolding protein, plays a critical role in base excision repair pathway by bringing together a complex of DNA repair proteins, including poly (ADP-ribose) polymerase I[25]. Overexpression of XRCC1 leads to camptothecin resistance in cells[26]. Cell cycle division 45-like protein (CDC45L) is responsible for DNA replication. CDC45L was shown to be an important determinant of camptothecin sensitivity[27]. Small ubiquitin-like modifier-1 (SUMO1) has been demonstrated to compete with ubiquitin in conjugation of a protein at the same site. As a result, sumoylation, catalyzed by the sole E2-conjugating enzyme, UBC9, can stabilize the protein by preventing ubiquitin-proteasome-mediated degradation. Topoisomerase I sumoylation may inhibit ubiquitination and degradation of this enzyme, because topoisomerase I is modified by SUMO1 after camptothecin treatment[28].
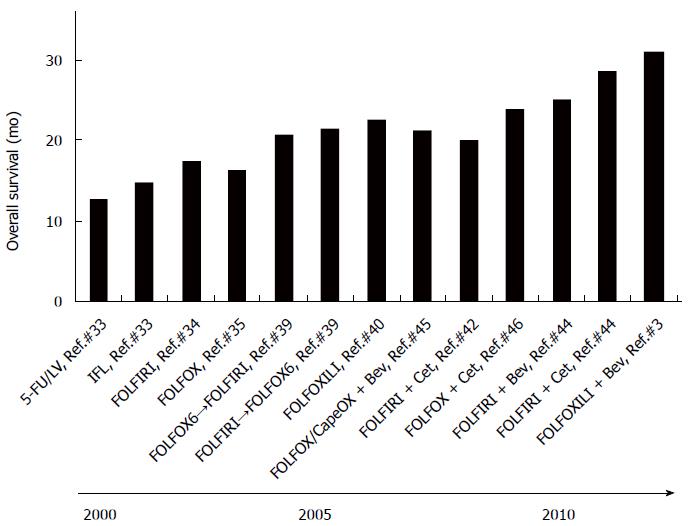
Since the introduction of 5-FU for the treatment of metastatic CRC, the survival had been gradually extended until the end of the last century. However, after the introduction of irinotecan and oxaliplatin, followed by molecularly-targeted anticancer agents such as bevacizumab, cetuximab, and panitumumab, a dramatic extension of survival has been achieved (Figure 1). We introduce the roles of irinotecan in the extension of survival in patients with metastatic CRC below (Figure 1 and Table 1).
| Trials (primary endpoint) | Regimens | Overall survival (OS) (mo) | P value | Progression-free survival (PFS) (mo) | P value | Response rate (RR) | P value | Ref. |
| 0038 (PFS) | 5-FU/LV | 12.6 | 0.040 | 4.3 | 0.004 | 21% | < 0.001 | [33] |
| IFL | 14.8 | 7.0 | 39% | |||||
| V303 (RR) | 5-FU/LV | 14.1 | 0.031 | 4.4 | < 0.001 | 22% | < 0.005 | [34] |
| FOLFIRI | 17.4 | 6.7 | 35% | |||||
| (PFS) | 5-FU/LV | 14.7 | 0.120 | 6.2 | < 0.001 | 22.3% | < 0.001 | [35] |
| FOLFOX | 16.2 | 9.0 | 50.7% | |||||
| V308 (2nd PFS)1 | FOLFIRI→FOLFOX6 | 21.5 | 0.990 | 8.5 | 0.260 | 56% | > 0.05 | [39] |
| FOLFOX6→FOLFIRI | 20.6 | 8.0 | 54% | |||||
| GONO (RR) | FOLFIRI | 16.7 | 0.032 | 6.9 | 0.001 | 41% | < 0.001 | [40] |
| FOLFOXIRI | 22.6 | 9.8 | 66% | |||||
| NO16966 (PFS) | FOLFOX/CapeOX | 19.9 | 0.077 | 8.0 | 0.002 | 49% | 0.310 | [45] |
| FOLFOX/CapeOX + Bev | 21.3 | 9.4 | 47% | |||||
| CRYSTAL (PFS) | FOLFIRI | 18.6 | 0.310 | 8.0 | 0.048 | 38.7% | 0.004 | [42] |
| FOLFIRI + Cet | 19.9 | 8.9 | 46.9% | |||||
| PRIME (PFS) | FOLFOX | 19.7 | 0.072 | 8.0 | 0.020 | 48% | 0.068 | [46] |
| FOLFOX + Pan | 23.9 | 9.6 | 55% | |||||
| FIRE-3 (PFS) | FOLFIRI + Bev | 25.0 | 0.017 | 10.3 | 0.550 | 58% | 0.180 | [44] |
| FOLFIRI + Cet | 28.7 | 10.0 | 62% | |||||
| TRIBE (PFS) | FOLFIRI + Bev | 25.8 | 0.054 | 9.7 | 0.003 | 53.1% | 0.006 | [3] |
| FOLFOXIRI + Bev | 31.0 | 12.1 | 65.1% |
The most popular schedules of irinotecan treatment are 30- or 90-min intravenous infusions of 125 mg/m2 given weekly for 4 of every 6 wk or 350 mg/m2 given every 3 wk. The weekly-times-4 schedule is more popular in North America, and the every-3-wk schedule was developed predominantly in Europe. On the other hand, regimens of 100 mg/m2 every week or 150 mg/m2 every other week have been frequently used in Japan. In comparative clinical studies, none of these regimens was shown to be superior in terms of antitumor efficacy[14,29]. Remarkably, the dose intensity of all applied dosage regimens of irinotecan is approximately 100 mg/m2 per week, which suggests that response does not depend on the specific treatment schedule. Phase II studies consistently obtained response rates (RR) of 10%-35% with single-agent irinotecan in metastatic CRC, independently of the administration schedule. There was also no apparent difference in the median remission duration or median survival time between different treatment schedules[30].
Treatment with irinotecan given by intravenous infusion at a dose of 300 to 350 mg/m2 every 3 wk was compared with best supportive care in a randomized phase III study in patients with metastatic CRC refractory to previous treatment with 5-FU-based chemotherapy. The 1-year survival rate was significantly higher in the irinotecan-treated group (36%) than in the control group (14%)[31]. Another randomized phase III study comparing irinotecan with three different continuous intravenous infusion schedules of 5-FU in patients with previously treated metastatic CRC demonstrated a survival advantage of irinotecan over 5-FU, regardless of the treatment schedule used[32].
5-FU combinations: Based on the results of phase I/II studies showing that irinotecan combined with 5-FU/LV was active in patients with CRC, two randomized phase III studies were performed to compare irinotecan plus 5-FU/LV with 5-FU/LV alone as first-line treatment for metastatic CRC. Saltz et al[33] showed that a weekly-times-4 regimen of 125 mg/m2 irinotecan and 20 mg/m2 LV, followed by an intravenous bolus of 500 mg/m2 5-FU (IFL) yielded a significantly longer OS than conventional low-dose 5-FU/LV (Table 1). Douillard et al[34] investigated the efficacy of an intravenous bolus of 5-FU (400 mg/m2)/LV (200 mg/m2 on days 1 and 2) plus continuous 5-FU infusion (600 mg/m2 for 22 h on days 1 and 2), the so-called LV5FU2 regimen, with or without intravenous irinotecan infusion (180 mg/m2 for 90 min) as first-line treatment for metastatic CRC. Irinotecan combined with infusional 5-FU (FOLFIRI) was well tolerated and increased the RR and prolonged the time to progression and OS (Table 1). The treatment schedules used in both studies were approved by the FDA as first-line chemotherapy for patients with metastatic CRC. However, because of the higher toxicity associated with the IFL regimen[33], it has not been further used in the clinical practice.
In addition to irinotecan, oxaliplatin combined with infusional 5-FU has also extended OS in patients with metastatic CRC. A phase III study has shown that LV5FU2 combined with oxaliplatin (FOLFOX4) is beneficial as first-line therapy in metastatic CRC, prolonging progression-free survival (PFS), but not OS (Table 1), with acceptable tolerability and maintenance of quality of life[35]. To simplify the treatment procedure and improve the quality of life of patients, a simplified LV5FU2 regimen has been combined with irinotecan (FOLFIRI) and with oxaliplatin (FOLFOX6) and evaluated as second-line therapy[36-38]. One phase III study compared two sequences of FOLFIRI and FOLFOX6, i.e., first-line FOLFIRI followed by second-line FOLFOX6, and the reverse order[39]. Previously untreated patients were randomly assigned to receive a 2-h infusion of 200 mg/m2 LV or 400 mg/m2 dL LV followed by a 400 mg/m2 bolus of 5-FU and 2400 to 3000 mg/m2 of 5-FU as a 46-h infusion every 2 wk, either with 180 mg/m2 irinotecan or with 100 mg/m2 oxaliplatin as a 2-h infusion on day 1. On disease progression, irinotecan was replaced by oxaliplatin, or oxaliplatin was replaced by irinotecan. Median OS was almost equivalent in both arms (Table 1). The primary end point of the second PFS in each arm was 14.2 mo and 10.9 mo, respectively.
The Gruppo Oncologico Nord Ovest conducted a phase III study comparing 5-FU, LV, oxaliplatin, and irinotecan [FOLFOXIRI (165 mg/m2 irinotecan, 85 mg/m2 oxaliplatin, 200 mg/m2 LV on day 1, and 3200 mg/m2, 5-FU as a 48-h continuous infusion starting on day 1, every 2 wk)] with infusional 5-FU, LV, and irinotecan (FOLFIRI) as first-line treatment for metastatic CRC[40]. The FOLFOXIRI regimen improved RR, PFS, and OS compared with FOLFIRI (Table 1), with an increased but manageable toxicity in patients with metastatic CRC with favorable prognostic characteristics.
Molecularly targeted-drug combinations: Currently, two promising classes of molecularly-targeted compounds have been introduced for the clinical management of metastatic CRC: epidermal growth factor receptor (EGFR) antagonists and angiogenesis inhibitors[41]. For example, cetuximab, a monoclonal antibody against the extracellular binding domain of EGFR, has single-agent activity against CRC, and augments the effects of irinotecan-based chemotherapy. In the CRYSTAL trial, the efficacy of cetuximab plus FOLFIRI as first-line treatment for metastatic CRC and associations between the mutation status of the KRAS gene in tumors and the clinical response to cetuximab were investigated[42]. The hazard ratio for PFS in the cetuximab-FOLFIRI group as compared with the FOLFIRI group was 0.85. There was no significant difference in OS between the two treatment groups (HR = 0.93). There was a significant interaction between treatment group and KRAS mutation status for tumor response, but not for PFS or OS. The hazard ratio for PFS among patients with wild-type-KRAS tumors was 0.68, in favor of the cetuximab-FOLFIRI group. These results indicate that first-line treatment with cetuximab plus FOLFIRI reduced the risk of progression of metastatic CRC as compared with FOLFIRI alone, although the benefit of cetuximab was limited to patients with KRAS wild-type tumors.
The addition of bevacizumab to irinotecan also led to a statistically significant increase in the RR and a 4.7 mo prolongation of median OS (20.3 mo vs 15.6 mo for IFL and placebo, respectively)[43]. The addition of cetuximab or bevacizumab to FOLFIRI was compared in a phase III study (FIRE-3 trial) in patients with KRAS (exon 2) codon 12/13 wild-type metastatic CRC[44]. Although median PFS was almost equal in both cetuximab and bevacizumab groups (HR = 1.06), median OS in the cetuximab group was significantly longer than that in the bevacizumab group (HR = 0.77) (Table 1). The association with longer OS suggests that FOLFIRI plus cetuximab might be the preferred first-line regimen for patients with KRAS exon 2 wild-type metastatic CRC.
In the case of oxaliplatin, the addition of bevacizumab to first-line FOLFOX4 significantly improved PFS, but not OS and RR in patients with metastatic CRC (Table 1)[45]. Panitumumab, a fully humanized anti-EGFR monoclonal antibody, has been approved as monotherapy for patients with chemotherapy-refractory metastatic CRC because it improved PFS[46]. The efficacy and safety of panitumumab plus FOLFOX4 (panitumumab-FOLFOX4) were compared with those of FOLFOX4 alone as initial treatment for metastatic CRC in the PRIME study[47]. In the patients with wild-type KRAS, panitumumab-FOLFOX4 significantly improved PFS compared with FOLFOX4 (Table 1). A non-significant increase in OS was observed for panitumumab-FOLFOX4 vs FOLFOX4 (Table 1). In the patients with mutant KRAS, PFS and OS were reduced in the panitumumab-FOLFOX4 group vs the FOLFOX4 group. This study demonstrated that panitumumab-FOLFOX4 was well tolerated and significantly improved PFS in patients with wild-type KRAS tumors and emphasized the importance of KRAS testing for patients with metastatic CRC.
In a randomized phase III study (TRIBE)[3], the median PFS was 12.1 mo in patients with metastatic CRC who received first-line FOLFOXIRI plus bevacizumab, as compared with 9.7 mo in those who received FOLFIRI plus bevacizumab group (HR = 0.75). OS in the FOLFIRI plus bevacizumab group was slightly but not significantly longer (31.0 mo vs 25.8 mo; HR for death = 0.79). FOLFOXIRI plus bevacizumab improved outcome in patients with metastatic CRC to achieve the OS of longer than 30 mo.
Even after a specific dose is determined for a specific patient population on the bases of the results of clinical trials, this does not necessarily mean that the determined dose will be the optimal dose for each individual patient. The dose determined for a specific population will often be suboptimal for most patients. The large inter-patient and intra-patient variability in systemic exposure to a given drug is a limiting factor in determining the optimal dose, because of non-tumor-related differences in pharmacokinetics among individuals. These differences include pharmacogenetic factors, physiological factors and environmental factors (Table 2). In the case of irinotecan, the relation between systemic exposure to SN-38 and irinotecan-induced severe toxicity has received special attention, because toxicity often necessitates a decrease in planned dose intensity, resulting in the incomplete success of irinotecan treatment.
| Factors | Exposure to SN-38 | Irinotecan-induced toxicity | Dosage recommendation | Ref. |
| Pharmacogenetic factors | ||||
| UGT1A1*6 and *28 | AUC ↑ | Severe neutropenia (diarrhea)↑ | Need to be reduced (prescription information in the US and Japan etc.) | [10-13,53,54,59,60] |
| SLCO1B1*15 | AUC ↑ | Severe neutropenia↑ | No recommendation exists (need to be reduced?) | [62-64,67] |
| Physiological factors | ||||
| Age (elderly patients) | Comparable to younger | Comparable to younger | No need to be modified | [82,87-89] |
| Body size (obesity) | Similar in BSA-based and flat-fixed dosing | Similar in BSA-based and flat-fixed dosing | No need to be modified (flat-fixed dosing) | [48,54,68] |
| Organ dysfunctions | ||||
| Liver | AUC ↑ | Severe neutropenia (diarrhea)↑ | Need to be reduced | [95-97] |
| Kidney | AUC ↑ | Mild to moderate, but prolonged neutropenia | Probably need to be reduced | [101,103] |
| Gender | Lower in female? | Severe hematologic toxicity in female? | No recommendation exists | [8,65,88,112,113] |
| Environmental factors | ||||
| Medication (drug-drug interactions) | No data available | Polypharmacy-related toxicity | No recommendation exists | [114] |
| Life style | ||||
| Smoking | AUC ↓ | Lower toxicity | No recommendation exists | [115] |
UGT1A1: Interindividual variability in the clearance of irinotecan is reported to be approximately 30%, whereas that of SN-38 is much higher (about 80%)[48]. Variability in SN-38 pharmacokinetics resulting from glucuronide formation is at least one of the major causes of irinotecan-induced severe toxicity[7,8].
UGT1A1 is the enzyme primarily responsible for endogenous bilirubin glucuronidation as well as SN-38 glucuronidation[9]. Decreased bilirubin glucuronidation capacity of UGT1A1 is evident in patients with Gilbert’s syndrome, for which the genetic basis has been elucidated. Gilbert’s syndrome is most commonly related to homozygotes of the seven repeat of TA allele (UGT1A1*28) in the proximal promoter region of UGT1A1[49], causing decreased gene expression of UGT1A1[50]. In addition to this promoter-region polymorphism, missense polymorphisms in exon 1 and in the shared exons 2 to 5 have been found. Of particular relevance to East Asian populations, including Japanese, is a mutation in exon 1 (211G>A, G71R), referred to as UGT1A1*6[51]. Homozygotes for this mutation might have decreased catalytic activity by 60%[52]. Many clinical studies have linked UGT1A1*28 and UGT1A1*6 genotypes to irinotecan-induced toxicity, especially severe neutropenia[10-13,53] (Table 2). Frequencies of high-risk patients were nearly 10% in whites (UGT1A1*28/*28)[53,54] as well as Japanese (UGT1A1*28/*28, UGT1A1*6/*6, and UGT1A1*6/*28)[55].
Stewart et al[56] have demonstrated that severe toxicities such as grade 3 and 4 neutropenia and diarrhea did not increase in pediatric patients with the UGT1A1*28/*28 genotype when irinotecan was given according to a low-dose protracted schedule, although such patients tended to have higher area under the plasma concentration-time curve (AUC) of SN-38 and lower SN-38G to SN-38 AUC ratios. A meta-analysis has revealed that the risk of toxicity increased in an irinotecan dose-dependent fashion in patients with the UGT1A1*28/*28 genotype, but not in patients with the UGT1A1*1/*1 or UGT1A1*1/*28 genotype[57], probably because the glucuronidation of SN-38 by UGT1A1 might be saturated in patients harboring two genetic variations, i.e., UGT1A1*6/*6, UGT1A1*28, or UGT1A1*6/*28, when higher doses of irinotecan were given[58]. Subsequent dose escalation studies have demonstrated a higher recommended dose of irinotecan in white cancer patients with UGT1A1*1/*1 and UGT1A1*1/*28 than in those with UGT1A1*28/*28[54,59,60]. These results indicate that the UGT1A1*28 genotype can be used to individualize dosing of irinotecan (Table 2).
Transporters: In addition to drug metabolism, kinetic processes relevant to irinotecan disposition are highly depending on the interplay with drug transport in organs such as the liver. In this context, an area of investigation that remains relatively poorly explored and understood in connection with irinotecan includes hepatocellular uptake transporters such as OATP1B1 and OATP1B3[19-21], and active transport systems involved in permeation of SN-38 across canalicular membranes, including ABCC2 and ABCG2[16-18]. The International Transporter Consortium presented two polymorphisms for which there is compelling evidence supporting their clinical relevance: SLCO1B1 (521T>C, V174A, rs4149056) and ABCG2 (421C>A, Q141K, rs2231142)[61]. Given this important finding, we first summarize the relevance of these two polymorphisms in SLCO1B1 and ABCG2 to pharmacokinetics and clinical outcomes of irinotecan.
Life-threatening toxicities and higher exposure to SN-38 were observed in a Japanese patient with cancer harboring both UGT1A1*6/*28 and SLOC1B1*15/*15[62]. SLCO1B1*15 haplotype consists of G allele at 388A>G and C allele at 521T>C. Significantly higher exposure to SN-38 was observed in Asian cancer patients with SLCO1B1*15[63]. In a phase II study of chemotherapy with irinotecan and cisplatin in Korean patients with non-small cell lung cancer[64], the 521TC or CC and -11187AA genotypes were associated with increased AUC of SN-38. Patients with SLCO1B1*15 showed significantly higher AUC of SN-38 than those harboring haplotypes without 521T>C. Grade 4 neutropenia was associated with the 521TC or CC genotypes, whereas grade 3 diarrhea was associated with 388GG genotype. However, in a comprehensive pharmacogenetics analysis of irinotecan-induced neutropenia and pharmacokinetics[65], there was no apparent relation between SLCO1B1 521T>C and the pharmacokinetics of SN-38 or irinotecan-induced toxicities. Negative results were also observed in a study performed by De Mattia et al[66]. Possible reasons for the discordant results include heterogeneous subjects enrolled in these studies and design of studies, i.e., patients were prospectively enrolled, and the pharmacogenetic analyses were performed retrospectively. In a recent prospective study in patients with advanced cancer who received irinotecan-based regimens, SLCO1B1 521T>C allele was found to be significantly associated with increased SN-38 exposure[67]. However, because exposure to SN-38 was evaluated on the basis of the plasma concentration obtained immediately after the 90-min infusion of irinotecan, the relation between the SLCO1B1 521T>C allele and the AUC or clearance of SN-38, which have been proposed to be related to clinical outcome of irinotecan treatment[1], was unclear. Available evidence thus suggest that the SLCO1B1 521T>C and the related haplotype are involved in SN-38 disposition and predictive marker for severe toxicity of irinotecan (Table 2), although further prospective studies are needed to draw definitive conclusion.
As for the polymorphism ABCG2 421C>A, no positive association with increased exposure to SN-38 or with severe irinotecan-related toxicities was observed[65,66,68-70].
Since SN-38 is a substrate of ABCC2, de Jong et al[71] explored associations of ABCC2 polymorphisms and haplotypes with irinotecan disposition and diarrhea. The haplotype ABCC2*2 was found to be associated with lower irinotecan clearance and with a reduced incidence of severe diarrhea, probably because of reduced hepatobiliary secretion of irinotecan. Han et al[72] and Fujita et al[73] also found that specific polymorphisms in ABCC2 can influence disposition or tumor responses to irinotecan by regulating transporter activity.
Other drug-metabolizing enzymes: Previous in vitro experiments revealed that CES2 is associated with a high-affinity and high-velocity to catalyze irinotecan hydrolysis to form SN-38[74]. However, CES2 expression is high in the intestine and kidney, but low in the liver, whereas CES1 is abundantly expressed in the liver[75]. Although minor genetic variations in CES2 found in Japanese were functionally deficient[76], and some of them were associated with lower irinotecan metabolism in vitro and in vivo[76,77], major CES2 haplotypes (*1b and *1c) did not affect irinotecan pharmacokinetics[77]. Interestingly, a gene-dose effect of functional CES1A genes on SN-38 formation was observed in irinotecan-treated Japanese patients with cancer[78], probably because CES1 is expressed at higher levels in the liver, a major organ for activating of irinotecan, although metabolic intrinsic clearance of CES1 is much lower than that of CES2[74].
Because genetic diversity is observed in the genes encoding CYP3A4 and CYP3A5[79], it has been suggested that genotyping for variants in these genes may be useful for predicting the pharmacokinetic profiles of irinotecan. However, multiple studies have demonstrated that this approach does not lead to significant correlations of CYP3A polymorphisms with irinotecan pharmacokinetics or clinical outcomes[80]. This failure to demonstrate clinically meaningful correlations may be due to the low allele frequency of most CYP3A variant genotypes or may reflect the relatively lower impact of these variants on enzyme activity in vivo[79]. CYP3A4 activity is determined not only by genetic variants, but also by complex regulations at the transcriptional and posttranscriptional levels, physiological factors, and environmental interactions. The role of CYP3A4 and CYP3A5 genotyping in improving treatment with irinotecan remains doubtful.
Age (elderly patients): The elderly population has been increasing in recent years because of the prolongation of average life expectancy. The longer the average life expectancy becomes, the higher is the incidence of cancer. Consequently, the number of elderly patients with cancer is increasing. Although many cancers arise in elderly individuals, elderly patients have been underrepresented in clinical trials designed to establish new anticancer treatment[81], leading to inadequate data to support evidence-based decisions with respect to chemotherapy[82]. Older cancer patients show considerable heterogeneity in their handling of drugs as a result of age-related changes in body composition, including decreased muscle mass, increased adipose tissue, and decreased liver and renal functions. Aging is accompanied by an about 30% decrease in liver volume and an about 40% decrease in hepatic blood flow[83]. Thus, the clearance of drugs with a high hepatic elimination rate, which is limited by blood flow, might decrease in the elderly[84,85]. Age-related decreases in the functions of some drug-metabolizing enzymes have also been identified, but their clinical significance remains uncertain[83,86].
Pharmacokinetic variables such as the maximum concentration and AUC of irinotecan, SN-38, and SN-38G in patients 65 years or older were comparable to the respective values in younger patients (within 3% of difference)[82,87]. A phase II trial was performed to evaluate the antitumor activity and toxicity of irinotecan in patients with metastatic CRC that had recurred or progressed after 5-FU-based chemotherapy[88]. This trial included patients 65 years or older. Elderly patients were twice as likely to develop grade 3 or 4 diarrhea as compared with younger patients when all courses of therapy were evaluated, suggesting that older patients are more sensitive to irinotecan-induced diarrhea than younger patients. However, older age did not significantly predict a higher incidence of first-course diarrhea. In addition, RRs do not depend on age[88]. On the basis of these findings Lichtman et al[82,87] concluded that currently available evidence does not support a specific dose modification of irinotecan in elderly patients. A systematic review also concluded that pharmacokinetic and clinical data suggest that fit elderly patients may tolerate irinotecan as well as younger population. RR and survival achieved in elderly patients who receive irinotecan-based combination chemotherapy appear to be equivalent to those obtained in younger patients. For the subgroup of fit elderly patients, irinotecan may be used similarly to younger patients[89] (Table 2). However, a reduced starting dose of irinotecan has been recommended for patients older than 70 years who have received prior pelvic irradiation or have a poor performance status[29,32].
Body size (obesity): According to the World Health Organization, worldwide obesity has nearly doubled since 1980 and now represents the fifth leading risk factor for global mortality, involved in the deaths of at least 2.8 million adults per each year (http://www.who.int/mediacentre/factsheets/fs311/en/). The proportion of the overweight population is projected to increase over the coming years in many industrialized countries, making obesity a major public health issue[90].
Considerable lines of evidence suggest that the dose intensity of chemotherapy in overweight and obese patients with cancer in actual clinical practice is often lower than the recommended dose intensity[91]; nevertheless, retrospective and prospective clinical data have indicated an association of dose intensity with both clinical efficacy and toxicity. Because of the variability and uncertainty about the appropriate dose regimens of chemotherapy in obese patients, the American Society of Clinical Oncology has issued clinical practice guidelines for appropriate chemotherapy dosing in obese adults with cancer[92]. The guidelines recommend that after considering any comorbidities chemotherapy dosing should be calculated on the basis of body surface area (BSA) using actual weight, rather than an estimate or idealization of weight[92].
However, Mathijssen et al[48] have advocated for many years that BSA is not significantly related to the marked differences among patients in exposure to SN-38. Dosing based on BSA did not reduce clearance variability[93], as compared with an unadjusted dose. Patients who received a flat-fixed irinotecan dose of 600 mg did not show greater interindividual pharmacokinetic variability than a control group who received the registered dose of 350 mg/m2[68]. Because toxicity also did not significantly differ, it was concluded that flat-fixed dosing could safely be used to supplant the BSA-based dosing strategy of irinotecan. A recent study by Innocenti et al[54] also demonstrated that with flat dosing of irinotecan BSA was not a significant predictor of the absolute neutrophil count nadir, a measure of irinotecan-induced myelosuppression. Collectively, flat dosing of irinotecan might be recommended (Table 2). Despite these observations, many prescribers and regulators maintain the erroneous belief that a patient with a larger BSA will always require a higher dose to induce the same drug effects[94]. Therefore, it is unfortunately unlikely that this strategy will be globally abandoned any time soon.
Organ dysfunction: Liver failure - since irinotecan and its metabolites are extensively eliminated via the liver, impaired liver function should be critical to the disposition of irinotecan as well as to the clinical outcomes of irinotecan-based chemotherapy.
In a phase I study, irinotecan was administered by an every-3-wk schedule to patients with varying degrees of liver dysfunction[95]. High bilirubin and alkaline phosphatase levels were associated with an exponentially decreased clearance of irinotecan. Drug toxicity was correlated with the serum bilirubin concentration. Patients who had total bilirubin levels less than 1.5 times the upper limit of normal (ULN) tolerated full-dose therapy (350 mg/m2 every 3 wk). The maximum tolerated dose for patients who had total bilirubin levels 1.5 to 3.0 times the ULN was 200 mg/m2 every 3 wk. Three patients with bilirubin levels higher than 3.0 times the ULN received one cycle of irinotecan at a dose of 100 mg/m2. Although none of three patients had DLT, 2 patients had rapid hepatic tumor progression associated with exacerbation of liver dysfunction and worsening of performance status. Therefore, no dosing recommendations could be made for such patients. The most common DLTs in patients with hyperbilirubinemia were grade 4 febrile neutropenia and diarrhea. A separate phase I study confirmed that irinotecan dose reductions are required in patients with liver impairment[96]. Twelve patients with hyperbilirubinemia (median serum bilirubin, 2.1 mg/dL) were given irinotecan according to an every-3-wk schedule. Three of five patients had DLT at a dose of 145 mg/m2, while none of seven patients had DLT at a dose of 115 mg/m2. Two of the DLTs were neutropenia, and one was exacerbation of liver function. The recommended starting doses and the pharmacokinetics of irinotecan in a weekly schedule were also examined in patients with solid tumors who had impaired liver function, evaluated on the basis of the baseline serum total bilirubin level, and aspartate aminotransferase and alanine aminotransferase[97]. Irinotecan was given as a 90-min intravenous infusion weekly for the first 4 wk of each 6-wk cycle at starting doses ranging from 40 to 75 mg/m2. Hepatic dysfunction reduced irinotecan clearance while increasing relative exposure to SN-38. SN-38 exposures in patients who received doses of 40 to 75 mg/m2 irinotecan were comparable to the level of exposure in patients with normal liver function who received a starting dose of 125 mg/m2. The administered starting doses of irinotecan seemed to be safe for patients with hepatic impairment who received irinotecan according to a weekly schedule. At these starting doses, exposure to SN-38 and the adverse event profile are similar to those in patients with normal liver function, and antitumor activity can be expected. These results indicate that patients with impaired liver function should be received a reduced dose of irinotecan because of increased exposure to SN-38 and an increased risk of irinotecan-induced toxicity (Table 2).
Impaired renal function - Even in cancer patients with severe renal failure, chemotherapeutic agents are given when patient’s life expectancy is most likely to be determined by malignancy, not by renal dysfunction. Such patients are typically given anticancer drugs that are predominantly metabolized in the liver or eliminated into bile (or both), instead of drugs that are mainly excreted renally. Irinotecan is therefore administered to cancer patients with severe renal dysfunction, because it is extensively subjected to hepatic metabolism and excreted into bile. Urinary excretion of SN-38 accounts for less than 1% of the total administered dose of irinotecan[98-100].
In a prospective clinical pharmacological study of irinotecan performed by us, the plasma concentration of SN-38, but not irinotecan or SN-38G, was significantly higher in patients with severe renal failure who had a creatinine clearance (CLcr) of less than 20 mL/min and received hemodialysis than in patients without renal failure (terminal elimination rate constant, 0.0084 h-1vs 0.081 h-1)[101], even though irinotecan is predominantly eliminated by the liver via glucuronidation and biliary excretion. The mean AUC of SN-38 calculated from 0 to 24 h in the patients with severe renal failure was 1.7-fold greater than that in the patients without renal failure (1.31 μmol/L·h vs 0.77 μmol/L·h)[101]. It should be pointed out that all patients with severe renal failure had mild or moderate but prolonged neutropenia even though they were receiving dialysis. The second course of irinotecan was delayed according to the prolonged neutropenia[101]. Because SN-38 concentrations have been reported to be still detectable even 500 h after administration of irinotecan in patients with normal renal function[102], a long period of exposure to relatively high concentrations of SN-38 was postulated to be one of the causes for the prolonged neutropenia in such patients. A previous study has demonstrated that patients with slower CLcr (35-66 mL/min) had a four-fold higher risk of grade 3 or 4 neutropenia, although the pharmacokinetics of irinotecan and its metabolites did not differ from those in patients with normal kidney function[103]. Increased plasma SN-38 concentrations were found only in patients with severe renal failure associated with a CLcr of less than 20 mL/min in our studies[101]. These findings suggest that irinotecan is not necessarily safe in cancer patients with renal failure, even though this anticancer drug is predominantly eliminated via the liver (Table 2).
We have previously investigated potential mechanism(s) for delayed SN-38 elimination and found that SN-38 uptake by human hepatocytes was significantly inhibited by a mixture of organic anion uremic toxins 3-carboxy-4-methyl-5-propyl-2-furanpropionate (CMPF), indoxyl sulfate, hippuric acid, and indole acetate], when the concentrations of these toxins were clinically relevant[21]. CMPF directly inhibited the uptake of SN-38 by human hepatocytes and most potently decreased SN-38 uptake mediated by cDNA-expressed OATP1B1 among the uremic toxins tested. Furthermore, of SLCO1B1 and SLCO1B3 gene expression in hepatocytes was significantly down-regulated by treatment with human uremic plasma. The inhibition of OATP1B1-mediated SN-38 uptake by uremic toxins and the down-regulation of SLCO1B1 gene expression may thus at least partly contribute to the mechanisms responsible for the delayed SN-38 elimination in patients with severe renal dysfunction. Because no differences in the pharmacokinetics of irinotecan or SN-38G were found between patients with and those without severe renal failure, changes in CES or UGT1A1 activity appear unlikely, although further studies are needed.
To determine whether these findings in patients with severe renal failure are generally applicable, we have to examine whether similar findings are obtained for other drugs that are predominantly taken up into liver by OATP1B1. Repaglinide is a nonrenally eliminated drug, which is a substrate of OATP transporter. Therefore, repaglinide metabolism in the liver may be limited by the uptake process of this transporter[104,105]. The AUC of repaglinide in patients with severe renal failure was approximately 3-fold greater than that in patients with normal renal function[106]. A physiologically-based pharmacokinetic (PBPK) model analysis by Zhao et al[107] has shown that an approximately 52% reduction in the OATP1B1-mediated hepatic uptake of repaglinide was required in a virtual population of patients with severe renal impairment to obtain an AUC value comparable to that observed in humans[106]. Our results and the findings of Zhao et al[107] indicate that the increased pharmacokinetic profile of drugs that are predominantly subjected to OATP1B1-mediated uptake into the liver in patients with severe renal failure is caused by reduced uptake capacity of OATP1B1. If the reduction in hepatic uptake by the direct inhibition of OATP1B1 activity with uremic toxins or by suppression of SLCO1B gene expression (or by both) could be quantitatively predicted, PBPK models could potentially be used to calculate appropriate doses for cancer patients with severe renal failure that would produce AUCs similar to those obtained in patients with normal kidney function.
Confirmation of these results may lead to the development of a new concepts for establishing evidence-based treatment strategies for irinotecan as well as other anticancer drugs that are substrates of OATP1B in cancer patients with severe renal dysfunction.
Gender: Because female-predominant expression of CYP3A4 is caused by RXRα-mediated sex-dependent effects of growth hormone on CYP3A expression[108], the more rapid clearance of various drugs in women as compared with men has been reported. AUC ratios of inactive metabolites to irinotecan, an in vivo parameter for CYP3A4 activity, were significantly higher in females than in males[109]. Although, sex-related differences in UGT activity are relatively small and are confined to several UGTs, including UGT2B15[110], systemic exposure to SN-38 was predicted by sex and hepatic function in a population pharmacokinetic analysis[111]. A previous study has also demonstrated that both the maximum plasma concentration and the AUC of irinotecan and SN-38 are lower in women[112], suggesting gender-dependent irinotecan pharmacokinetics (higher clearance in female). Although early studies indicated no significant association of gender with grade 3 or 4 toxicities[8,88], more recent findings suggest that female gender is an independent predictor of severe hematologic toxicity induced by irinotecan[65,113]. However, further confirmation is necessary.
Multiple concomitant medications were significantly associated with severe irinotecan-related toxicity in patients given monotherapy or FOLFIRI[114]. The incidence of severe irinotecan-related toxicities increased in parallel to the number of concomitant medications. Thus, polypharmacy should be effectively managed to decrease the risk of adverse drug reactions in patients with cancer who receive irinotecan-based chemotherapy.
Smoking significantly lowers both the exposure to irinotecan and SN-38, and treatment-induced neutropenia, indicating a potential risk of treatment failure[115]. Modulation of CYP3A and UGT1A1 by ingredients in smoking may partly cause these phenomena, although the underlying mechanism remains poorly understood.
This review discusses the contribution of irinotecan to chemotherapy for metastatic CRC and the optimal dosing to achieve the personalized chemotherapy. Irinotecan became a key anticancer drug because it prolonged OS. By combining irinotecan with 5-FU, oxaliplatin and molecularly-targeted drug, OS longer than 30 mo has been achieved. Exposure to SN-38, an active metabolite of irinotecan, is characterized by large inter- and intra-patient variability and can cause irinotecan-related severe toxicities. A large number of studies have recommended the dose reduction of irinotecan for patients with UGT1A1 polymorphisms and liver dysfunction. Studies by us suggest that the dose of irinotecan should be reduced in patients with severe renal failure, even though irinotecan is predominantly eliminated via the liver.
P- Reviewer: Hammerman A, Tural D S- Editor: Yu J L- Editor: A E- Editor: Liu XM
| 1. | Gerrits CJ, de Jonge MJ, Schellens JH, Stoter G, Verweij J. Topoisomerase I inhibitors: the relevance of prolonged exposure for present clinical development. Br J Cancer. 1997;76:952-962. [PubMed] [Cited in This Article: ] |
| 2. | Kunimoto T, Nitta K, Tanaka T, Uehara N, Baba H, Takeuchi M, Yokokura T, Sawada S, Miyasaka T, Mutai M. Antitumor activity of 7-ethyl-10-[4-(1-piperidino)-1-piperidino]carbonyloxy-camptothec in, a novel water-soluble derivative of camptothecin, against murine tumors. Cancer Res. 1987;47:5944-5947. [PubMed] [Cited in This Article: ] |
| 3. | Loupakis F, Cremolini C, Masi G, Lonardi S, Zagonel V, Salvatore L, Cortesi E, Tomasello G, Ronzoni M, Spadi R. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014;371:1609-1618. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 689] [Cited by in F6Publishing: 738] [Article Influence: 73.8] [Reference Citation Analysis (0)] |
| 4. | Kudoh S, Fujiwara Y, Takada Y, Yamamoto H, Kinoshita A, Ariyoshi Y, Furuse K, Fukuoka M. Phase II study of irinotecan combined with cisplatin in patients with previously untreated small-cell lung cancer. West Japan Lung Cancer Group. J Clin Oncol. 1998;16:1068-1074. [PubMed] [Cited in This Article: ] |
| 5. | Rothenberg ML, Meropol NJ, Poplin EA, Van Cutsem E, Wadler S. Mortality associated with irinotecan plus bolus fluorouracil/leucovorin: summary findings of an independent panel. J Clin Oncol. 2001;19:3801-3807. [PubMed] [Cited in This Article: ] |
| 6. | Kudoh S, Fukuoka M, Masuda N, Yoshikawa A, Kusunoki Y, Matsui K, Negoro S, Takifuji N, Nakagawa K, Hirashima T. Relationship between the pharmacokinetics of irinotecan and diarrhea during combination chemotherapy with cisplatin. Jpn J Cancer Res. 1995;86:406-413. [PubMed] [Cited in This Article: ] |
| 7. | Gupta E, Lestingi TM, Mick R, Ramirez J, Vokes EE, Ratain MJ. Metabolic fate of irinotecan in humans: correlation of glucuronidation with diarrhea. Cancer Res. 1994;54:3723-3725. [PubMed] [Cited in This Article: ] |
| 8. | Gupta E, Mick R, Ramirez J, Wang X, Lestingi TM, Vokes EE, Ratain MJ. Pharmacokinetic and pharmacodynamic evaluation of the topoisomerase inhibitor irinotecan in cancer patients. J Clin Oncol. 1997;15:1502-1510. [PubMed] [Cited in This Article: ] |
| 9. | Guillemette C, Lévesque É, Rouleau M. Pharmacogenomics of human uridine diphospho-glucuronosyltransferases and clinical implications. Clin Pharmacol Ther. 2014;96:324-339. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 102] [Cited by in F6Publishing: 118] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 10. | Ando Y, Saka H, Ando M, Sawa T, Muro K, Ueoka H, Yokoyama A, Saitoh S, Shimokata K, Hasegawa Y. Polymorphisms of UDP-glucuronosyltransferase gene and irinotecan toxicity: a pharmacogenetic analysis. Cancer Res. 2000;60:6921-6926. [PubMed] [Cited in This Article: ] |
| 11. | Han JY, Lim HS, Shin ES, Yoo YK, Park YH, Lee JE, Jang IJ, Lee DH, Lee JS. Comprehensive analysis of UGT1A polymorphisms predictive for pharmacokinetics and treatment outcome in patients with non-small-cell lung cancer treated with irinotecan and cisplatin. J Clin Oncol. 2006;24:2237-2244. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 240] [Cited by in F6Publishing: 235] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 12. | Innocenti F, Undevia SD, Iyer L, Chen PX, Das S, Kocherginsky M, Karrison T, Janisch L, Ramírez J, Rudin CM. Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol. 2004;22:1382-1388. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 767] [Cited by in F6Publishing: 712] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 13. | Minami H, Sai K, Saeki M, Saito Y, Ozawa S, Suzuki K, Kaniwa N, Sawada J, Hamaguchi T, Yamamoto N. Irinotecan pharmacokinetics/pharmacodynamics and UGT1A genetic polymorphisms in Japanese: roles of UGT1A1*6 and *28. Pharmacogenet Genomics. 2007;17:497-504. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 233] [Cited by in F6Publishing: 223] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 14. | Mathijssen RH, van Alphen RJ, Verweij J, Loos WJ, Nooter K, Stoter G, Sparreboom A. Clinical pharmacokinetics and metabolism of irinotecan (CPT-11). Clin Cancer Res. 2001;7:2182-2194. [PubMed] [Cited in This Article: ] |
| 15. | Sparreboom A, Fujita K, Zamboni WC. Topoisomerase I-Targeting Drugs. Cancer Chemotherapy and Biotherapy: Principles and Practice. 5th ed. Philadelphia: Lippincott Williams & Wilkins 2010; 342-355. [Cited in This Article: ] |
| 16. | Chu XY, Kato Y, Sugiyama Y. Multiplicity of biliary excretion mechanisms for irinotecan, CPT-11, and its metabolites in rats. Cancer Res. 1997;57:1934-1938. [PubMed] [Cited in This Article: ] |
| 17. | Chu XY, Kato Y, Ueda K, Suzuki H, Niinuma K, Tyson CA, Weizer V, Dabbs JE, Froehlich R, Green CE. Biliary excretion mechanism of CPT-11 and its metabolites in humans: involvement of primary active transporters. Cancer Res. 1998;58:5137-5143. [PubMed] [Cited in This Article: ] |
| 18. | Nakatomi K, Yoshikawa M, Oka M, Ikegami Y, Hayasaka S, Sano K, Shiozawa K, Kawabata S, Soda H, Ishikawa T. Transport of 7-ethyl-10-hydroxycamptothecin (SN-38) by breast cancer resistance protein ABCG2 in human lung cancer cells. Biochem Biophys Res Commun. 2001;288:827-832. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 139] [Cited by in F6Publishing: 149] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 19. | Nozawa T, Minami H, Sugiura S, Tsuji A, Tamai I. Role of organic anion transporter OATP1B1 (OATP-C) in hepatic uptake of irinotecan and its active metabolite, 7-ethyl-10-hydroxycamptothecin: in vitro evidence and effect of single nucleotide polymorphisms. Drug Metab Dispos. 2005;33:434-439. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 239] [Cited by in F6Publishing: 232] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 20. | Yamaguchi H, Kobayashi M, Okada M, Takeuchi T, Unno M, Abe T, Goto J, Hishinuma T, Mano N. Rapid screening of antineoplastic candidates for the human organic anion transporter OATP1B3 substrates using fluorescent probes. Cancer Lett. 2008;260:163-169. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 47] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 21. | Fujita K, Sugiura T, Okumura H, Umeda S, Nakamichi N, Watanabe Y, Suzuki H, Sunakawa Y, Shimada K, Kawara K. Direct inhibition and down-regulation by uremic plasma components of hepatic uptake transporter for SN-38, an active metabolite of irinotecan, in humans. Pharm Res. 2014;31:204-215. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 43] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 22. | Hsiang YH, Liu LF. Identification of mammalian DNA topoisomerase I as an intracellular target of the anticancer drug camptothecin. Cancer Res. 1988;48:1722-1726. [PubMed] [Cited in This Article: ] |
| 23. | Hsiang YH, Lihou MG, Liu LF. Arrest of replication forks by drug-stabilized topoisomerase I-DNA cleavable complexes as a mechanism of cell killing by camptothecin. Cancer Res. 1989;49:5077-5082. [PubMed] [Cited in This Article: ] |
| 24. | Beretta GL, Cossa G, Gatti L, Zunino F, Perego P. Tyrosyl-DNA phosphodiesterase 1 targeting for modulation of camptothecin-based treatment. Curr Med Chem. 2010;17:1500-1508. [PubMed] [Cited in This Article: ] |
| 25. | Hu Z, Ma H, Lu D, Zhou J, Chen Y, Xu L, Zhu J, Huo X, Qian J, Wei Q. A promoter polymorphism (-77T& gt; C) of DNA repair gene XRCC1 is associated with risk of lung cancer in relation to tobacco smoking. Pharmacogenet Genomics. 2005;15:457-463. [PubMed] [Cited in This Article: ] |
| 26. | Park SY, Lam W, Cheng YC. X-ray repair cross-complementing gene I protein plays an important role in camptothecin resistance. Cancer Res. 2002;62:459-465. [PubMed] [Cited in This Article: ] |
| 27. | Rasheed ZA, Rubin EH. Mechanisms of resistance to topoisomerase I-targeting drugs. Oncogene. 2003;22:7296-7304. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 124] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 28. | Han JY, Lee GK, Yoo SY, Yoon SJ, Cho EY, Kim HT, Lee JS. Association of SUMO1 and UBC9 genotypes with tumor response in non-small-cell lung cancer treated with irinotecan-based chemotherapy. Pharmacogenomics J. 2010;10:86-93. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 29. | Fuchs CS, Moore MR, Harker G, Villa L, Rinaldi D, Hecht JR. Phase III comparison of two irinotecan dosing regimens in second-line therapy of metastatic colorectal cancer. J Clin Oncol. 2003;21:807-814. [PubMed] [Cited in This Article: ] |
| 30. | Vanhoefer U, Harstrick A, Achterrath W, Cao S, Seeber S, Rustum YM. Irinotecan in the treatment of colorectal cancer: clinical overview. J Clin Oncol. 2001;19:1501-1518. [PubMed] [Cited in This Article: ] |
| 31. | Cunningham D, Pyrhönen S, James RD, Punt CJ, Hickish TF, Heikkila R, Johannesen TB, Starkhammar H, Topham CA, Awad L. Randomised trial of irinotecan plus supportive care versus supportive care alone after fluorouracil failure for patients with metastatic colorectal cancer. Lancet. 1998;352:1413-1418. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 946] [Cited by in F6Publishing: 882] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 32. | Rougier P, Van Cutsem E, Bajetta E, Niederle N, Possinger K, Labianca R, Navarro M, Morant R, Bleiberg H, Wils J. Randomised trial of irinotecan versus fluorouracil by continuous infusion after fluorouracil failure in patients with metastatic colorectal cancer. Lancet. 1998;352:1407-1412. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 752] [Cited by in F6Publishing: 714] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 33. | Saltz LB, Cox JV, Blanke C, Rosen LS, Fehrenbacher L, Moore MJ, Maroun JA, Ackland SP, Locker PK, Pirotta N. Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. Irinotecan Study Group. N Engl J Med. 2000;343:905-914. [PubMed] [Cited in This Article: ] |
| 34. | Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000;355:1041-1047. [PubMed] [Cited in This Article: ] |
| 35. | de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18:2938-2947. [PubMed] [Cited in This Article: ] |
| 36. | André T, Louvet C, Maindrault-Goebel F, Couteau C, Mabro M, Lotz JP, Gilles-Amar V, Krulik M, Carola E, Izrael V. CPT-11 (irinotecan) addition to bimonthly, high-dose leucovorin and bolus and continuous-infusion 5-fluorouracil (FOLFIRI) for pretreated metastatic colorectal cancer. GERCOR. Eur J Cancer. 1999;35:1343-1347. [PubMed] [Cited in This Article: ] |
| 37. | Maindrault-Goebel F, de Gramont A, Louvet C, André T, Carola E, Gilles V, Lotz JP, Tournigand C, Mabro M, Molitor JL. Evaluation of oxaliplatin dose intensity in bimonthly leucovorin and 48-hour 5-fluorouracil continuous infusion regimens (FOLFOX) in pretreated metastatic colorectal cancer. Oncology Multidisciplinary Research Group (GERCOR). Ann Oncol. 2000;11:1477-1483. [PubMed] [Cited in This Article: ] |
| 38. | Maindrault-Goebel F, Louvet C, André T, Carola E, Lotz JP, Molitor JL, Garcia ML, Gilles-Amar V, Izrael V, Krulik M. Oxaliplatin added to the simplified bimonthly leucovorin and 5-fluorouracil regimen as second-line therapy for metastatic colorectal cancer (FOLFOX6). GERCOR. Eur J Cancer. 1999;35:1338-1342. [PubMed] [Cited in This Article: ] |
| 39. | Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22:229-237. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2282] [Cited by in F6Publishing: 2165] [Article Influence: 108.3] [Reference Citation Analysis (1)] |
| 40. | Falcone A, Ricci S, Brunetti I, Pfanner E, Allegrini G, Barbara C, Crinò L, Benedetti G, Evangelista W, Fanchini L. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007;25:1670-1676. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 830] [Cited by in F6Publishing: 855] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 41. | Meyerhardt JA, Mayer RJ. Systemic therapy for colorectal cancer. N Engl J Med. 2005;352:476-487. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 859] [Cited by in F6Publishing: 835] [Article Influence: 43.9] [Reference Citation Analysis (0)] |
| 42. | Van Cutsem E, Köhne CH, Hitre E, Zaluski J, Chang Chien CR, Makhson A, D’Haens G, Pintér T, Lim R, Bodoky G. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med. 2009;360:1408-1417. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2901] [Cited by in F6Publishing: 3040] [Article Influence: 202.7] [Reference Citation Analysis (1)] |
| 43. | Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335-2342. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7832] [Cited by in F6Publishing: 7592] [Article Influence: 379.6] [Reference Citation Analysis (1)] |
| 44. | Heinemann V, von Weikersthal LF, Decker T, Kiani A, Vehling-Kaiser U, Al-Batran SE, Heintges T, Lerchenmüller C, Kahl C, Seipelt G. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial. Lancet Oncol. 2014;15:1065-1075. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1117] [Cited by in F6Publishing: 1227] [Article Influence: 122.7] [Reference Citation Analysis (0)] |
| 45. | Saltz LB, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS, Rivera F. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26:2013-2019. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2302] [Cited by in F6Publishing: 2226] [Article Influence: 139.1] [Reference Citation Analysis (0)] |
| 46. | Van Cutsem E, Peeters M, Siena S, Humblet Y, Hendlisz A, Neyns B, Canon JL, Van Laethem JL, Maurel J, Richardson G. Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol. 2007;25:1658-1664. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1444] [Cited by in F6Publishing: 1432] [Article Influence: 84.2] [Reference Citation Analysis (0)] |
| 47. | Douillard JY, Siena S, Cassidy J, Tabernero J, Burkes R, Barugel M, Humblet Y, Bodoky G, Cunningham D, Jassem J. Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J Clin Oncol. 2010;28:4697-4705. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1296] [Cited by in F6Publishing: 1343] [Article Influence: 95.9] [Reference Citation Analysis (0)] |
| 48. | Mathijssen RH, de Jong FA, Loos WJ, van der Bol JM, Verweij J, Sparreboom A. Flat-fixed dosing versus body surface area based dosing of anticancer drugs in adults: does it make a difference? Oncologist. 2007;12:913-923. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 104] [Cited by in F6Publishing: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 49. | Bosma PJ, Chowdhury JR, Bakker C, Gantla S, de Boer A, Oostra BA, Lindhout D, Tytgat GN, Jansen PL, Oude Elferink RP. The genetic basis of the reduced expression of bilirubin UDP-glucuronosyltransferase 1 in Gilbert’s syndrome. N Engl J Med. 1995;333:1171-1175. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1077] [Cited by in F6Publishing: 1094] [Article Influence: 37.7] [Reference Citation Analysis (0)] |
| 50. | Beutler E, Gelbart T, Demina A. Racial variability in the UDP-glucuronosyltransferase 1 (UGT1A1) promoter: a balanced polymorphism for regulation of bilirubin metabolism? Proc Natl Acad Sci USA. 1998;95:8170-8174. [PubMed] [Cited in This Article: ] |
| 51. | Akaba K, Kimura T, Sasaki A, Tanabe S, Wakabayashi T, Hiroi M, Yasumura S, Maki K, Aikawa S, Hayasaka K. Neonatal hyperbilirubinemia and a common mutation of the bilirubin uridine diphosphate-glucuronosyltransferase gene in Japanese. J Hum Genet. 1999;44:22-25. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 64] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 52. | Gagné JF, Montminy V, Belanger P, Journault K, Gaucher G, Guillemette C. Common human UGT1A polymorphisms and the altered metabolism of irinotecan active metabolite 7-ethyl-10-hydroxycamptothecin (SN-38). Mol Pharmacol. 2002;62:608-617. [PubMed] [Cited in This Article: ] |
| 53. | Toffoli G, Cecchin E, Corona G, Russo A, Buonadonna A, D’Andrea M, Pasetto LM, Pessa S, Errante D, De Pangher V. The role of UGT1A1*28 polymorphism in the pharmacodynamics and pharmacokinetics of irinotecan in patients with metastatic colorectal cancer. J Clin Oncol. 2006;24:3061-3068. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 261] [Cited by in F6Publishing: 254] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 54. | Innocenti F, Schilsky RL, Ramírez J, Janisch L, Undevia S, House LK, Das S, Wu K, Turcich M, Marsh R. Dose-finding and pharmacokinetic study to optimize the dosing of irinotecan according to the UGT1A1 genotype of patients with cancer. J Clin Oncol. 2014;32:2328-2334. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 113] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 55. | Akiyama Y, Fujita K, Nagashima F, Yamamoto W, Endo H, Sunakawa Y, Yamashita K, Ishida H, Mizuno K, Araki K. Genetic testing for UGT1A1*28 and *6 in Japanese patients who receive irinotecan chemotherapy. Ann Oncol. 2008;19:2089-2090. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 56. | Stewart CF, Panetta JC, O’Shaughnessy MA, Throm SL, Fraga CH, Owens T, Liu T, Billups C, Rodriguez-Galindo C, Gajjar A. UGT1A1 promoter genotype correlates with SN-38 pharmacokinetics, but not severe toxicity in patients receiving low-dose irinotecan. J Clin Oncol. 2007;25:2594-2600. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 57. | Hoskins JM, Goldberg RM, Qu P, Ibrahim JG, McLeod HL. UGT1A1*28 genotype and irinotecan-induced neutropenia: dose matters. J Natl Cancer Inst. 2007;99:1290-1295. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 364] [Cited by in F6Publishing: 344] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 58. | Ichikawa W, Araki K, Fujita K, Yamamoto W, Endo H, Nagashima F, Tanaka R, Miya T, Kodama K, Sunakawa Y. Re: UGT1A1*28 genotype and irinotecan-induced neutropenia: dose matters. J Natl Cancer Inst. 2008;100:224-225; author reply 225. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 59. | Marcuello E, Páez D, Paré L, Salazar J, Sebio A, del Rio E, Baiget M. A genotype-directed phase I-IV dose-finding study of irinotecan in combination with fluorouracil/leucovorin as first-line treatment in advanced colorectal cancer. Br J Cancer. 2011;105:53-57. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 91] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 60. | Toffoli G, Cecchin E, Gasparini G, D’Andrea M, Azzarello G, Basso U, Mini E, Pessa S, De Mattia E, Lo Re G. Genotype-driven phase I study of irinotecan administered in combination with fluorouracil/leucovorin in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28:866-871. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 130] [Cited by in F6Publishing: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 61. | Giacomini KM, Balimane PV, Cho SK, Eadon M, Edeki T, Hillgren KM, Huang SM, Sugiyama Y, Weitz D, Wen Y. International Transporter Consortium commentary on clinically important transporter polymorphisms. Clin Pharmacol Ther. 2013;94:23-26. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 127] [Cited by in F6Publishing: 128] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 62. | Takane H, Kawamoto K, Sasaki T, Moriki K, Moriki K, Kitano H, Higuchi S, Otsubo K, Ieiri I. Life-threatening toxicities in a patient with UGT1A1*6/*28 and SLCO1B1*15/*15 genotypes after irinotecan-based chemotherapy. Cancer Chemother Pharmacol. 2009;63:1165-1169. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 63. | Xiang X, Jada SR, Li HH, Fan L, Tham LS, Wong CI, Lee SC, Lim R, Zhou QY, Goh BC. Pharmacogenetics of SLCO1B1 gene and the impact of *1b and *15 haplotypes on irinotecan disposition in Asian cancer patients. Pharmacogenet Genomics. 2006;16:683-691. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 80] [Cited by in F6Publishing: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 64. | Han JY, Lim HS, Shin ES, Yoo YK, Park YH, Lee JE, Kim HT, Lee JS. Influence of the organic anion-transporting polypeptide 1B1 (OATP1B1) polymorphisms on irinotecan-pharmacokinetics and clinical outcome of patients with advanced non-small cell lung cancer. Lung Cancer. 2008;59:69-75. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 81] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 65. | Innocenti F, Kroetz DL, Schuetz E, Dolan ME, Ramírez J, Relling M, Chen P, Das S, Rosner GL, Ratain MJ. Comprehensive pharmacogenetic analysis of irinotecan neutropenia and pharmacokinetics. J Clin Oncol. 2009;27:2604-2614. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 197] [Cited by in F6Publishing: 201] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 66. | De Mattia E, Toffoli G, Polesel J, D’Andrea M, Corona G, Zagonel V, Buonadonna A, Dreussi E, Cecchin E. Pharmacogenetics of ABC and SLC transporters in metastatic colorectal cancer patients receiving first-line FOLFIRI treatment. Pharmacogenet Genomics. 2013;23:549-557. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 67. | Teft WA, Welch S, Lenehan J, Parfitt J, Choi YH, Winquist E, Kim RB. OATP1B1 and tumour OATP1B3 modulate exposure, toxicity, and survival after irinotecan-based chemotherapy. Br J Cancer. 2015;112:857-865. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 68. | de Jong FA, Mathijssen RH, Xie R, Verweij J, Sparreboom A. Flat-fixed dosing of irinotecan: influence on pharmacokinetic and pharmacodynamic variability. Clin Cancer Res. 2004;10:4068-4071. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 69. | Jada SR, Lim R, Wong CI, Shu X, Lee SC, Zhou Q, Goh BC, Chowbay B. Role of UGT1A1*6, UGT1A1*28 and ABCG2 c.421C& gt; A polymorphisms in irinotecan-induced neutropenia in Asian cancer patients. Cancer Sci. 2007;98:1461-1467. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 97] [Cited by in F6Publishing: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 70. | Han JY, Lim HS, Park YH, Lee SY, Lee JS. Integrated pharmacogenetic prediction of irinotecan pharmacokinetics and toxicity in patients with advanced non-small cell lung cancer. Lung Cancer. 2009;63:115-120. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 71. | de Jong FA, Scott-Horton TJ, Kroetz DL, McLeod HL, Friberg LE, Mathijssen RH, Verweij J, Marsh S, Sparreboom A. Irinotecan-induced diarrhea: functional significance of the polymorphic ABCC2 transporter protein. Clin Pharmacol Ther. 2007;81:42-49. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 133] [Cited by in F6Publishing: 125] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 72. | Han JY, Lim HS, Yoo YK, Shin ES, Park YH, Lee SY, Lee JE, Lee DH, Kim HT, Lee JS. Associations of ABCB1, ABCC2, and ABCG2 polymorphisms with irinotecan-pharmacokinetics and clinical outcome in patients with advanced non-small cell lung cancer. Cancer. 2007;110:138-147. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 151] [Cited by in F6Publishing: 154] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 73. | Fujita K, Nagashima F, Yamamoto W, Endo H, Sunakawa Y, Yamashita K, Ishida H, Mizuno K, Matsunaga M, Araki K. Association of ATP-binding cassette, sub-family C, number 2 (ABCC2) genotype with pharmacokinetics of irinotecan in Japanese patients with metastatic colorectal cancer treated with irinotecan plus infusional 5-fluorouracil/leucovorin (FOLFIRI). Biol Pharm Bull. 2008;31:2137-2142. [PubMed] [Cited in This Article: ] |
| 74. | Humerickhouse R, Lohrbach K, Li L, Bosron WF, Dolan ME. Characterization of CPT-11 hydrolysis by human liver carboxylesterase isoforms hCE-1 and hCE-2. Cancer Res. 2000;60:1189-1192. [PubMed] [Cited in This Article: ] |
| 75. | Hosokawa M. Structure and catalytic properties of carboxylesterase isozymes involved in metabolic activation of prodrugs. Molecules. 2008;13:412-431. [PubMed] [Cited in This Article: ] |
| 76. | Kubo T, Kim SR, Sai K, Saito Y, Nakajima T, Matsumoto K, Saito H, Shirao K, Yamamoto N, Minami H. Functional characterization of three naturally occurring single nucleotide polymorphisms in the CES2 gene encoding carboxylesterase 2 (HCE-2). Drug Metab Dispos. 2005;33:1482-1487. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 77. | Kim SR, Sai K, Tanaka-Kagawa T, Jinno H, Ozawa S, Kaniwa N, Saito Y, Akasawa A, Matsumoto K, Saito H. Haplotypes and a novel defective allele of CES2 found in a Japanese population. Drug Metab Dispos. 2007;35:1865-1872. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 78. | Sai K, Saito Y, Tatewaki N, Hosokawa M, Kaniwa N, Nishimaki-Mogami T, Naito M, Sawada J, Shirao K, Hamaguchi T. Association of carboxylesterase 1A genotypes with irinotecan pharmacokinetics in Japanese cancer patients. Br J Clin Pharmacol. 2010;70:222-233. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 79. | Werk AN, Cascorbi I. Functional gene variants of CYP3A4. Clin Pharmacol Ther. 2014;96:340-348. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 143] [Cited by in F6Publishing: 172] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 80. | Fujita K, Sparreboom A. Pharmacogenetics of irinotecan disposition and toxicity: a review. Curr Clin Pharmacol. 2010;5:209-217. [PubMed] [Cited in This Article: ] |
| 81. | Minami H, Ohe Y, Niho S, Goto K, Ohmatsu H, Kubota K, Kakinuma R, Nishiwaki Y, Nokihara H, Sekine I. Comparison of pharmacokinetics and pharmacodynamics of docetaxel and Cisplatin in elderly and non-elderly patients: why is toxicity increased in elderly patients? J Clin Oncol. 2004;22:2901-2908. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 75] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 82. | Lichtman SM, Wildiers H, Chatelut E, Steer C, Budman D, Morrison VA, Tranchand B, Shapira I, Aapro M. International Society of Geriatric Oncology Chemotherapy Taskforce: evaluation of chemotherapy in older patients--an analysis of the medical literature. J Clin Oncol. 2007;25:1832-1843. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 175] [Cited by in F6Publishing: 180] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 83. | Hämmerlein A, Derendorf H, Lowenthal DT. Pharmacokinetic and pharmacodynamic changes in the elderly. Clinical implications. Clin Pharmacokinet. 1998;35:49-64. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 198] [Cited by in F6Publishing: 201] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 84. | Bach B, Hansen JM, Kampmann JP, Rasmussen SN, Skovsted L. Disposition of antipyrine and phenytoin correlated with age and liver volume in man. Clin Pharmacokinet. 1981;6:389-396. [PubMed] [Cited in This Article: ] |
| 85. | Durnas C, Loi CM, Cusack BJ. Hepatic drug metabolism and aging. Clin Pharmacokinet. 1990;19:359-389. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 119] [Cited by in F6Publishing: 130] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 86. | Sotaniemi EA, Arranto AJ, Pelkonen O, Pasanen M. Age and cytochrome P450-linked drug metabolism in humans: an analysis of 226 subjects with equal histopathologic conditions. Clin Pharmacol Ther. 1997;61:331-339. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 289] [Cited by in F6Publishing: 259] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 87. | Lichtman SM, Skirvin JA, Vemulapalli S. Pharmacology of antineoplastic agents in older cancer patients. Crit Rev Oncol Hematol. 2003;46:101-114. [PubMed] [Cited in This Article: ] |
| 88. | Rothenberg ML, Cox JV, DeVore RF, Hainsworth JD, Pazdur R, Rivkin SE, Macdonald JS, Geyer CE, Sandbach J, Wolf DL. A multicenter, phase II trial of weekly irinotecan (CPT-11) in patients with previously treated colorectal carcinoma. Cancer. 1999;85:786-795. [PubMed] [Cited in This Article: ] |
| 89. | Sastre J, Puente J, García-Saenz JA, Díaz-Rubio E. Irinotecan in the treatment of elderly patients with advanced colorectal cancer. Crit Rev Oncol Hematol. 2008;68:250-255. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 90. | Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet. 2011;378:815-825. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1835] [Cited by in F6Publishing: 1721] [Article Influence: 132.4] [Reference Citation Analysis (0)] |
| 91. | Lyman GH, Sparreboom A. Chemotherapy dosing in overweight and obese patients with cancer. Nat Rev Clin Oncol. 2013;10:451-459. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 92. | Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM, Morrison VA, Pini TM, Runowicz CD, Rosner GL. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2012;30:1553-1561. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 355] [Cited by in F6Publishing: 353] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 93. | Mathijssen RH, Verweij J, de Jonge MJ, Nooter K, Stoter G, Sparreboom A. Impact of body-size measures on irinotecan clearance: alternative dosing recommendations. J Clin Oncol. 2002;20:81-87. [PubMed] [Cited in This Article: ] |
| 94. | Phelps MA, Sparreboom A. Irinotecan pharmacogenetics: a finished puzzle? J Clin Oncol. 2014;32:2287-2289. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 95. | Raymond E, Boige V, Faivre S, Sanderink GJ, Rixe O, Vernillet L, Jacques C, Gatineau M, Ducreux M, Armand JP. Dosage adjustment and pharmacokinetic profile of irinotecan in cancer patients with hepatic dysfunction. J Clin Oncol. 2002;20:4303-4312. [PubMed] [Cited in This Article: ] |
| 96. | Venook AP, Enders Klein C, Fleming G, Hollis D, Leichman CG, Hohl R, Byrd J, Budman D, Villalona M, Marshall J. A phase I and pharmacokinetic study of irinotecan in patients with hepatic or renal dysfunction or with prior pelvic radiation: CALGB 9863. Ann Oncol. 2003;14:1783-1790. [PubMed] [Cited in This Article: ] |
| 97. | Schaaf LJ, Hammond LA, Tipping SJ, Goldberg RM, Goel R, Kuhn JG, Miller LL, Compton LD, Cisar LA, Elfring GL. Phase 1 and pharmacokinetic study of intravenous irinotecan in refractory solid tumor patients with hepatic dysfunction. Clin Cancer Res. 2006;12:3782-3791. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 98. | de Jong FA, Kitzen JJ, de Bruijn P, Verweij J, Loos WJ. Hepatic transport, metabolism and biliary excretion of irinotecan in a cancer patient with an external bile drain. Cancer Biol Ther. 2006;5:1105-1110. [PubMed] [Cited in This Article: ] |
| 99. | Slatter JG, Schaaf LJ, Sams JP, Feenstra KL, Johnson MG, Bombardt PA, Cathcart KS, Verburg MT, Pearson LK, Compton LD. Pharmacokinetics, metabolism, and excretion of irinotecan (CPT-11) following I.V. infusion of [(14)C]CPT-11 in cancer patients. Drug Metab Dispos. 2000;28:423-433. [PubMed] [Cited in This Article: ] |
| 100. | Sparreboom A, de Jonge MJ, de Bruijn P, Brouwer E, Nooter K, Loos WJ, van Alphen RJ, Mathijssen RH, Stoter G, Verweij J. Irinotecan (CPT-11) metabolism and disposition in cancer patients. Clin Cancer Res. 1998;4:2747-2754. [PubMed] [Cited in This Article: ] |
| 101. | Fujita K, Sunakawa Y, Miwa K, Akiyama Y, Sugiyama M, Kawara K, Ishida H, Yamashita K, Mizuno K, Saji S. Delayed elimination of SN-38 in cancer patients with severe renal failure. Drug Metab Dispos. 2011;39:161-164. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 102. | Kehrer DF, Yamamoto W, Verweij J, de Jonge MJ, de Bruijn P, Sparreboom A. Factors involved in prolongation of the terminal disposition phase of SN-38: clinical and experimental studies. Clin Cancer Res. 2000;6:3451-3458. [PubMed] [Cited in This Article: ] |
| 103. | de Jong FA, van der Bol JM, Mathijssen RH, van Gelder T, Wiemer EA, Sparreboom A, Verweij J. Renal function as a predictor of irinotecan-induced neutropenia. Clin Pharmacol Ther. 2008;84:254-262. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 104. | Kajosaari LI, Niemi M, Neuvonen M, Laitila J, Neuvonen PJ, Backman JT. Cyclosporine markedly raises the plasma concentrations of repaglinide. Clin Pharmacol Ther. 2005;78:388-399. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 164] [Cited by in F6Publishing: 153] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 105. | Kalliokoski A, Backman JT, Kurkinen KJ, Neuvonen PJ, Niemi M. Effects of gemfibrozil and atorvastatin on the pharmacokinetics of repaglinide in relation to SLCO1B1 polymorphism. Clin Pharmacol Ther. 2008;84:488-496. [PubMed] [Cited in This Article: ] |
| 106. | Marbury TC, Ruckle JL, Hatorp V, Andersen MP, Nielsen KK, Huang WC, Strange P. Pharmacokinetics of repaglinide in subjects with renal impairment. Clin Pharmacol Ther. 2000;67:7-15. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 90] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 107. | Zhao P, Vieira Mde L, Grillo JA, Song P, Wu TC, Zheng JH, Arya V, Berglund EG, Atkinson AJ, Sugiyama Y. Evaluation of exposure change of nonrenally eliminated drugs in patients with chronic kidney disease using physiologically based pharmacokinetic modeling and simulation. J Clin Pharmacol. 2012;52:91S-108S. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 108. | Li J, Wan Y, Na S, Liu X, Dong G, Yang Z, Yang J, Yue J. Sex-dependent regulation of hepatic CYP3A by growth hormone: Roles of HNF6, C/EBPα, and RXRα. Biochem Pharmacol. 2015;93:92-103. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 109. | Sai K, Saito Y, Fukushima-Uesaka H, Kurose K, Kaniwa N, Kamatani N, Shirao K, Yamamoto N, Hamaguchi T, Kunitoh H. Impact of CYP3A4 haplotypes on irinotecan pharmacokinetics in Japanese cancer patients. Cancer Chemother Pharmacol. 2008;62:529-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 110. | Court MH. Interindividual variability in hepatic drug glucuronidation: studies into the role of age, sex, enzyme inducers, and genetic polymorphism using the human liver bank as a model system. Drug Metab Rev. 2010;42:209-224. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 154] [Cited by in F6Publishing: 158] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 111. | Klein CE, Gupta E, Reid JM, Atherton PJ, Sloan JA, Pitot HC, Ratain MJ, Kastrissios H. Population pharmacokinetic model for irinotecan and two of its metabolites, SN-38 and SN-38 glucuronide. Clin Pharmacol Ther. 2002;72:638-647. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 56] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 112. | Miya T, Goya T, Fujii H, Ohtsu T, Itoh K, Igarashi T, Minami H, Sasaki Y. Factors affecting the pharmacokinetics of CPT-11: the body mass index, age and sex are independent predictors of pharmacokinetic parameters of CPT-11. Invest New Drugs. 2001;19:61-67. [PubMed] [Cited in This Article: ] |
| 113. | Cecchin E, Innocenti F, D’Andrea M, Corona G, De Mattia E, Biason P, Buonadonna A, Toffoli G. Predictive role of the UGT1A1, UGT1A7, and UGT1A9 genetic variants and their haplotypes on the outcome of metastatic colorectal cancer patients treated with fluorouracil, leucovorin, and irinotecan. J Clin Oncol. 2009;27:2457-2465. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 171] [Cited by in F6Publishing: 161] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 114. | Sasaki T, Fujita K, Sunakawa Y, Ishida H, Yamashita K, Miwa K, Saji S, Kato Y, Sasaki Y. Concomitant polypharmacy is associated with irinotecan-related adverse drug reactions in patients with cancer. Int J Clin Oncol. 2013;18:735-742. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 115. | van der Bol JM, Mathijssen RH, Loos WJ, Friberg LE, van Schaik RH, de Jonge MJ, Planting AS, Verweij J, Sparreboom A, de Jong FA. Cigarette smoking and irinotecan treatment: pharmacokinetic interaction and effects on neutropenia. J Clin Oncol. 2007;25:2719-2726. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 98] [Cited by in F6Publishing: 92] [Article Influence: 5.4] [Reference Citation Analysis (0)] |