Published online Jun 21, 2015. doi: 10.3748/wjg.v21.i23.7084
Peer-review started: January 29, 2015
First decision: March 26, 2015
Revised: April 9, 2015
Accepted: May 4, 2015
Article in press: May 4, 2015
Published online: June 21, 2015
Processing time: 143 Days and 0.2 Hours
Hepatitis B virus (HBV) infection is a major global health problem. Although current therapies, such as the use of nucleos(t)ide analogs, inhibit HBV replication efficiently, they do not eliminate covalently closed circular DNA (cccDNA), which persists in hepatocyte nuclei. As HBV cccDNA is a viral transcription template, novel therapeutic approaches to directly target HBV cccDNA are necessary to completely eradicate persistent HBV infections. HBV cccDNA levels in HBV-infected human liver cells are extremely low; thus, more reliable and simple measurement methods are needed to correctly monitor their levels during therapeutic treatment. Although reverse transcription-polymerase chain reaction or Southern blot procedures are currently used in research studies, these methods are not completely reliable and are also time-consuming and labor-intensive. Genome editing technologies, such as zinc finger nucleases, transcription activator-like effector nucleases, and the clustered regularly interspaced short palindromic repeats/Cas9 (CRISPR/Cas9) system, which are designed to target specific DNA sequences, represent highly promising potential therapeutic tools. In particular, the CRISPR/Cas9 system is an easily customizable sequence-specific nuclease with high flexibility and may be the most feasible approach to target HBV cccDNA. Further research to develop easier, safer, and more effective protocols should be pursued.
Core tip: Hepatitis B virus (HBV) infection is a global health problem. Because current therapeutic approaches do not eliminate HBV nuclear covalently closed circular DNA (cccDNA), novel therapeutics to target cccDNA are needed to eradicate HBV infection. Genome editing technologies are expected to be promising therapeutic options against HBV cccDNA. In particular, the clustered regularly interspaced short palindromic repeats/Cas9 system is an easily customizable sequence-specific nuclease with great flexibility and may be the most feasible approach to target HBV cccDNA, as further research studies develop more effective protocols.
- Citation: Ohno M, Otsuka M, Kishikawa T, Yoshikawa T, Takata A, Koike K. Novel therapeutic approaches for hepatitis B virus covalently closed circular DNA. World J Gastroenterol 2015; 21(23): 7084-7088
- URL: https://www.wjgnet.com/1007-9327/full/v21/i23/7084.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i23.7084
Hepatitis B virus (HBV) infection is a major global health problem, despite the availability of effective vaccines. Approximately 2 billion people are infected with HBV, and over 350 million patients are chronic HBV carriers[1]. Chronic hepatitis B leads to a risk of liver cirrhosis and hepatocellular carcinoma, and more than 700000 people die from HBV-associated diseases each year[2].
Nucleos(t)ide analogs and interferon (IFN) are the currently available treatments for chronic hepatitis B. Although nucleos(t)ide analogs efficiently inhibit HBV replication, they do not completely eliminate HBV, and stopping treatment can result in viral relapse, which is why patients must be administered nucleos(t)ide analog for life once treatment has begun. Moreover, HBV can develop resistance to drugs over the long term. High-dose INF-α may help degrade nuclear viral DNA[3], but the effect is limited, and the adverse systemic events of high doses are not well tolerated by most patients. Therefore, novel therapeutic options to completely eradicate HBV infection are needed.
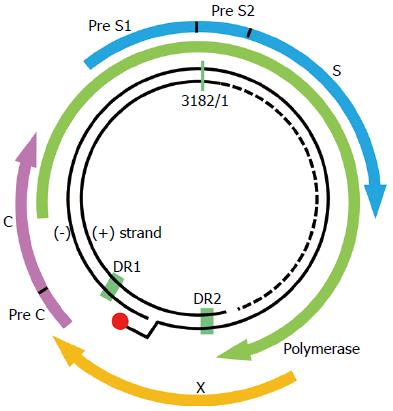
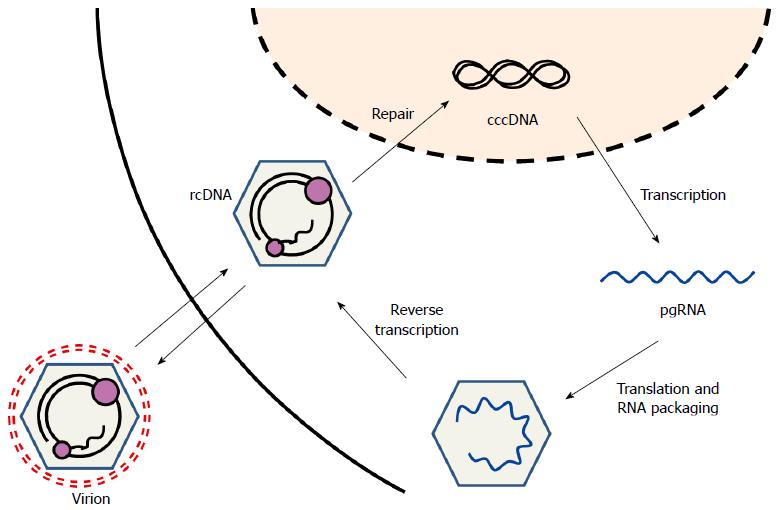
HBV is a DNA virus with a 3.2 kb-long partially double-stranded relaxed circular DNA (rcDNA) as its genome (Figure 1). Covalently closed circular DNA (cccDNA) is formed from the conversion of rcDNA after HBV infection. HBV cccDNA exists persistently in hepatocyte nuclei in an episomal state, where it acts as a viral transcription template. Transcribed RNA, known as pregenomic RNA (pgRNA), serves as mRNA for the viral proteins or serves as a template for the viral genome DNA through reverse transcription. Cytoplasmic pgRNA and the P protein (viral reverse transcriptase) are co-packaged into viral envelope proteins, and rcDNA is produced from the reverse transcription of pgRNA. Nucleocapsids containing rcDNA are released from the host cell as virions or are converted to cccDNA in the nucleus (Figure 2).
Nucleos(t)ide analogs, the primary drugs that are currently used to treat patients with chronic hepatitis B, restrain HBV replication by inhibiting pgRNA reverse transcription into rcDNA. However, they do not eliminate nuclear cccDNA, which is responsible for the persistence of an HBV infection. Therefore, novel therapeutic approaches to target HBV cccDNA are urgently needed.
The cccDNA levels in HBV-infected human liver cells are low, ranging from 1 to 50 copies per hepatocyte[4]. Therefore, stable and simple methods to measure cccDNA are necessary to evaluate HBV infection status and to assess the effects of antiviral treatment. Two methods are mainly used to measure cccDNA levels. Real-time polymerase chain reaction (PCR) amplification with specific primers detects cccDNA. However, PCR amplification can also include noise derived from other co-extracted viral DNA, leading to unreliable results. Southern blotting detects cccDNA directly and can distinguish cccDNA from other viral DNAs via the migration distance in electrophoresis[5,6]. However, Southern blotting is not convenient and is time-consuming. Furthermore, supercoiled cccDNA may be nicked during the DNA extraction procedure, which can change the cccDNA structure and its electrophoretic pattern; thus, the extraction step must be performed carefully. These procedures are frequently utilized in research, but they need to be refined to establish more reliable and convenient protocols.
Immune-based therapies have been applied to eradicate persistent HBV infections. IFN-α exerts immunomodulatory and antiviral effects leading to HBeAg seroconversion or normalization of ALT. However, its effect is limited and associated with many adverse events, such as flu-like symptoms, bone marrow suppression, fatigue, impaired glucose tolerance, and depression. A recent study reported that IFN-α and lymphotoxin(LT)-β receptor activation pathways can degrade cccDNA through deamination by APOBEC3A and 3B, and the LT-β receptor agonist is effective at low doses without toxicity[3]. These results indicate that cytokines or cytokine receptor agonists that can induce cccDNA deamination and degradation may be potential therapeutic candidates against HBV with minimal side effects.
For the complete eradication of HBV, it is necessary to target persistent cccDNA. Genome editing technologies, such as zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), and the clustered regularly interspaced short palindromic repeats/CRISPR associated (CRISPR/Cas) system, have attracted attention as potential therapeutic tools[7-9]. ZFNs and TALENs are engineered chimeric nucleases with sequence-specific binding domains and Fok1 nuclease domains that function as dimers and induce DNA double-strand breaks at specific target sites. These technologies have been used in genetic engineering in various fields, including HBV therapeutics, although certain limitations remain. ZFN specificity can be altered by context-dependent effects caused by interactions among neighboring zinc fingers of the DNA binding domain[10]. TALENs are better than ZFNs in regard to DNA specificity, but the large size of TALENs is a disadvantage, that makes delivery by viral vectors difficult[11]. ZFNs and TALENs have been used to target HBV cccDNA and have yielded some favorable effects[12,13]. However, both of ZFNs and TALENs require a pair of site-specific nucleases for each target, and thus, substantial efforts are needed to produce customized proteins.
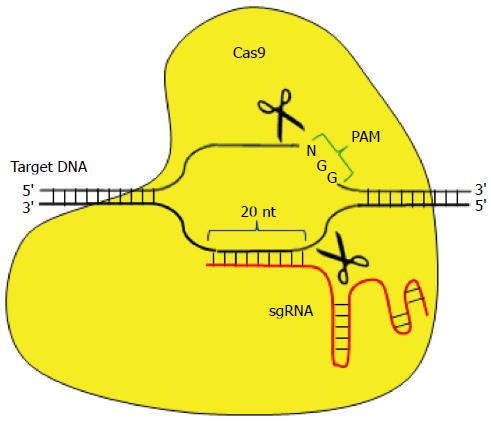
The CRISPR/Cas system has rapidly developed as a novel genome editing tool, which is more easily customizable than ZFNs and TALENs[8]. It is derived from the acquired immune system of bacteria and archaea against invading foreign DNA. The type II CRISPR/Cas system of Streptococcus pyogenes has been utilized for research. It consists of a Cas9 nuclease and a single guide RNA (sgRNA), which leads Cas9 to the target site. A 20-nt complementary sequence to the target DNA is positioned at the 5′ end of the sgRNA, which is adjacent to the 5′ part of a proto spacer adjacent motif (PAM) matching the 5′-NGG sequence. The Cas9/sgRNA complex is guided to the target site via RNA-DNA complementary base pairing, and induces site-specific double-strand breaks (Figure 3). This system can be used flexibly by designing sgRNA to any sequences with the PAM form.
Although the CRISPR/Cas9 system has been simulated using mathematical models as a therapeutic option against HBV cccDNA[14] and is being used to target various sites of cccDNA in some recent reports[15-17], the protocols need to be improved. The efficiency is still limited, as the appropriate methods to deliver the nuclease into target cells have not been established. Delivery methods via viral vectors, plasmid DNA expressing Cas9 and sgRNAs, in vitro transcribed Cas9 mRNA and sgRNAs are currently used[18]. However, the conventional transfection methods of plasmid DNA or mRNA, such as electroporation or cationic lipids, are inefficient, transient, and confined to certain cell lines. Viral vectors, including adenoviral and lentiviral vectors, can more efficiently deliver the nucleases compared to the transfection methods. Up to now, some studies targeting cccDNA via the CRISPR/Cas9 system have been performed using lentiviral vectors[16,17]. Lentiviral vectors enable the constitutive expression of Cas9 and/or sgRNAs. However, these can be integrated to the host genome, which may cause insertional mutagenesis. It is reported that integrase-deficient lentiviral vectors can deliver ZFNs[19], which may also be applicable to the CRISPR/Cas9 system. In the future, the development of improved delivery methods would lead to further progress in this field.
It is also important to select effective and specific target sequences in the viral DNA. It may be more effective to target several sequences in the HBV genome in parallel for better results. In addition, the target sequences are desirable to be well-conserved among virus isolates, and contain non-homologous sequences in the human genomes to minimize toxicity. In the CRISPR/Cas9 system, off-target effects can be observed if the sequence has up to five mismatches to the sgRNA[20]. Because HBV genomic sequences are not highly homologous with the human genome, there are many possible candidate sequences for HBV-specific sgRNAs in the HBV genome. However, these settings of sgRNAs may result in differences in the editing efficiency, which should be precisely verified in the future. After the cleavage of cccDNA, DNA repair can occur with accompanying mutations, which may be harmful to the host. Previous reports have claimed that some specific mutations of the HBV genome are associated with exacerbation of malignancy[21-23]. Thus, it may be better to carefully avoid such sites whose mutations can accelerate carcinogenesis when selecting the targets.
For practical use, there is still plenty room for the improvements to achieve safer and more effective therapeutic options using genome editing technology.
To eradicate HBV infections, we should develop novel therapeutics to target cccDNA, which works as a reservoir of virus within nuclei. Genome editing technologies, which can cleave specific DNA sequences, have recently emerged and are expected to be potential therapeutic utilities. Among them, the CRISPR/Cas9 system may be the most feasible approach for targeting HBV cccDNA. It is necessary to further improve the strategy to maximize the effects and minimize toxicity in the future.
P- Reviewer: Shen T, Tian YP, Zeng Z S- Editor: Qi Y L- Editor: A E- Editor: Liu XM
| 1. | Liaw YF, Chu CM. Hepatitis B virus infection. Lancet. 2009;373:582-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 929] [Cited by in RCA: 993] [Article Influence: 62.1] [Reference Citation Analysis (1)] |
| 2. | Lozano R, Naghavi M, Foreman K, Lim S, Shibuya K, Aboyans V, Abraham J, Adair T, Aggarwal R, Ahn SY. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2095-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9500] [Cited by in RCA: 9576] [Article Influence: 736.6] [Reference Citation Analysis (0)] |
| 3. | Lucifora J, Xia Y, Reisinger F, Zhang K, Stadler D, Cheng X, Sprinzl MF, Koppensteiner H, Makowska Z, Volz T. Specific and nonhepatotoxic degradation of nuclear hepatitis B virus cccDNA. Science. 2014;343:1221-1228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 640] [Cited by in RCA: 739] [Article Influence: 67.2] [Reference Citation Analysis (0)] |
| 4. | Seeger C, Mason WS. Hepatitis B virus biology. Microbiol Mol Biol Rev. 2000;64:51-68. [PubMed] |
| 5. | Cai D, Nie H, Yan R, Guo JT, Block TM, Guo H. A southern blot assay for detection of hepatitis B virus covalently closed circular DNA from cell cultures. Methods Mol Biol. 2013;1030:151-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 118] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 6. | Guo H, Aldrich CE, Saputelli J, Xu C, Mason WS. The insertion domain of the duck hepatitis B virus core protein plays a role in nucleocapsid assembly. Virology. 2006;353:443-450. [PubMed] |
| 7. | Gaj T, Gersbach CA, Barbas CF. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013;31:397-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2477] [Cited by in RCA: 2480] [Article Influence: 206.7] [Reference Citation Analysis (0)] |
| 8. | Sander JD, Joung JK. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat Biotechnol. 2014;32:347-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2175] [Cited by in RCA: 2217] [Article Influence: 201.5] [Reference Citation Analysis (0)] |
| 9. | Schiffer JT, Aubert M, Weber ND, Mintzer E, Stone D, Jerome KR. Targeted DNA mutagenesis for the cure of chronic viral infections. J Virol. 2012;86:8920-8936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 96] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 10. | Maeder ML, Thibodeau-Beganny S, Osiak A, Wright DA, Anthony RM, Eichtinger M, Jiang T, Foley JE, Winfrey RJ, Townsend JA. Rapid “open-source” engineering of customized zinc-finger nucleases for highly efficient gene modification. Mol Cell. 2008;31:294-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 609] [Cited by in RCA: 520] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 11. | Holkers M, Maggio I, Liu J, Janssen JM, Miselli F, Mussolino C, Recchia A, Cathomen T, Gonçalves MA. Differential integrity of TALE nuclease genes following adenoviral and lentiviral vector gene transfer into human cells. Nucleic Acids Res. 2013;41:e63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 216] [Cited by in RCA: 213] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 12. | Cradick TJ, Keck K, Bradshaw S, Jamieson AC, McCaffrey AP. Zinc-finger nucleases as a novel therapeutic strategy for targeting hepatitis B virus DNAs. Mol Ther. 2010;18:947-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 146] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 13. | Bloom K, Ely A, Mussolino C, Cathomen T, Arbuthnot P. Inactivation of hepatitis B virus replication in cultured cells and in vivo with engineered transcription activator-like effector nucleases. Mol Ther. 2013;21:1889-1897. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 169] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 14. | Schiffer JT, Swan DA, Stone D, Jerome KR. Predictors of hepatitis B cure using gene therapy to deliver DNA cleavage enzymes: a mathematical modeling approach. PLoS Comput Biol. 2013;9:e1003131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 15. | Lin SR, Yang HC, Kuo YT, Liu CJ, Yang TY, Sung KC, Lin YY, Wang HY, Wang CC, Shen YC. The CRISPR/Cas9 System Facilitates Clearance of the Intrahepatic HBV Templates In Vivo. Mol Ther Nucleic Acids. 2014;3:e186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 299] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 16. | Seeger C, Sohn JA. Targeting Hepatitis B Virus With CRISPR/Cas9. Mol Ther Nucleic Acids. 2014;3:e216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 227] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 17. | Kennedy EM, Bassit LC, Mueller H, Kornepati AV, Bogerd HP, Nie T, Chatterjee P, Javanbakht H, Schinazi RF, Cullen BR. Suppression of hepatitis B virus DNA accumulation in chronically infected cells using a bacterial CRISPR/Cas RNA-guided DNA endonuclease. Virology. 2015;476:196-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 185] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 18. | Ain QU, Chung JY, Kim YH. Current and future delivery systems for engineered nucleases: ZFN, TALEN and RGEN. J Control Release. 2015;205:120-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 19. | Lombardo A, Genovese P, Beausejour CM, Colleoni S, Lee YL, Kim KA, Ando D, Urnov FD, Galli C, Gregory PD. Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nat Biotechnol. 2007;25:1298-1306. [PubMed] |
| 20. | Fu Y, Foden JA, Khayter C, Maeder ML, Reyon D, Joung JK, Sander JD. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat Biotechnol. 2013;31:822-826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2216] [Cited by in RCA: 2425] [Article Influence: 202.1] [Reference Citation Analysis (0)] |
| 21. | Liu S, Zhang H, Gu C, Yin J, He Y, Xie J, Cao G. Associations between hepatitis B virus mutations and the risk of hepatocellular carcinoma: a meta-analysis. J Natl Cancer Inst. 2009;101:1066-1082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 279] [Cited by in RCA: 328] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 22. | Wu Y, Gan Y, Gao F, Zhao Z, Jin Y, Zhu Y, Sun Z, Wu H, Chen T, Wang J. Novel natural mutations in the hepatitis B virus reverse transcriptase domain associated with hepatocellular carcinoma. PLoS One. 2014;9:e94864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Park YM, Jang JW, Yoo SH, Kim SH, Oh IM, Park SJ, Jang YS, Lee SJ. Combinations of eight key mutations in the X/preC region and genomic activity of hepatitis B virus are associated with hepatocellular carcinoma. J Viral Hepat. 2014;21:171-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |