Published online Mar 14, 2015. doi: 10.3748/wjg.v21.i10.2905
Peer-review started: May 9, 2014
First decision: June 10, 2014
Revised: June 27, 2014
Accepted: October 15, 2014
Article in press: October 15, 2014
Published online: March 14, 2015
Processing time: 311 Days and 23.2 Hours
AIM: To study the prevalence of human papillomavirus (HPV) in esophageal carcinoma in Tangshan, China, a high-incidence area.
METHODS: Formalin-fixed, paraffin-embedded tissue specimens from 198 patients who were pathologically diagnosed with esophageal squamous cell carcinoma from 2011 to 2013 were obtained from a pathology department in Tangshan. DNA was extracted from all 198 specimens to detect HPV by polymerase chain reaction (PCR). β-globin PCR was performed to check the quality of the DNA extraction procedure. PCR was performed to detect a wide range of HPV types, and type-specific PCR was performed to detect HPV types 16 and 18. Negative and positive controls were used for HPV 16 and 18 detection.
RESULTS: The DNA extraction method in this study appeared to be more effective than other previously reported methods. After DNA extraction, more than 98% of the tissue specimens had an acceptable result in the DNA qualification test (β-globin PCR). The overall prevalence of HPV in tumor tissues by GP6+/GP5+ PCR was 79.79%, and the prevalence of HPV types 16 and 18 was 40.40% and 47.47%, respectively. PCR demonstrated the presence of HPV, and direct sequencing confirmed the HPV genotypes. All HPV-positive PCR products were checked by DNA sequence analysis using DNAman and compared with the known HPV sequences listed in the Basic Local Alignment Search Tool database to evaluate the HPV types. This analysis confirmed the presence of HPV types 16 and 18.
CONCLUSION: DNA of high-risk HPV types 16 and 18 is present in esophageal tumors, implicating HPV as a possible etiologic factor for esophageal squamous cell carcinoma.
Core tip: China is considered to have a high incidence of esophageal cancer. Many etiologic factors for esophageal cancer have been described; in China, human papillomavirus (HPV) infection could be an important cause of esophageal cancer. Tangshan in Hebei, China has a high incidence of esophageal cancer. However, no large-sample analyses of the prevalence of HPV in this area have been performed. We analyzed the prevalence and types of HPV in 198 esophageal cancer specimens in this area of China. These findings have important significance for analysis of the various causes of local esophageal cancer.
- Citation: Mehryar MM, Li SY, Liu HW, Li F, Zhang F, Zhou YB, Zeng Y, Li JT. Prevalence of human papillomavirus in esophageal carcinoma in Tangshan, China. World J Gastroenterol 2015; 21(10): 2905-2911
- URL: https://www.wjgnet.com/1007-9327/full/v21/i10/2905.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i10.2905
Esophageal cancer is the eighth most common cancer and sixth most common cause of cancer death worldwide[1]. Human papillomavirus (HPV) belongs to a group of nonenveloped, double-stranded DNA viruses, more than 100 genotypes of which have been recognized. Many molecular and epidemiologic studies have confirmed that HPV, especially high-risk HPV types, has an important role in the development of cervical cancer[2,3]. HPV DNA must be integrated into the host cell genome for malignant transformation of the cervical epithelium to occur[4]. Several studies have evaluated the role of HPV in extragenital cancers, but have produced conflicting results regarding the involvement of HPV[5,6].
In 1982, Syrjänen[7] was the first to report the probable involvement of HPV in esophageal squamous cell carcinoma (ESCC). Many researchers subsequently became interested in HPV infection as a possible cause of the development of ESCC. A comprehensive review in 2002 reported that HPV was positive in 22.9% of 1485 ESCC specimens evaluated by in situ hybridization and in 15.2% of 2020 ESCC specimens evaluated by PCR[8]. Esophageal cancer usually begins in the epithelial layer of the esophagus. Approximately 90% of esophageal cancers are squamous cell carcinomas. In some countries, such as the United States, the frequency of ESCC is almost identical to that of adenocarcinoma[9]. Different studies around the world have shown that, for unknown reasons, the incidence of esophageal cancer differs among various countries, as well as among different regions within the same country. According to the World Health Organization, West Africa is considered to be of low risk for ESCC, while China is considered to be of high risk[10]. Among North American countries, Peru and Mexico have the lowest mortality rates for esophageal carcinoma, and Argentina, Chile, and Brazil have the highest rates[11]. Many studies have been performed in different areas of China, but their results are conflicting. Studies on HPV in the Anyang, Shandong, and Gansu areas of China have confirmed the presence of the HPV genome, especially the high-risk types, in esophageal cancer[12]. However, this finding was not supported by another study conducted in Linxian, China[13].
Studies on HPV involvement in esophageal cancer have reported conflicting results, with prevalence rates ranging from 0 to 71%[14]. Variations in the specificity and sensitivity of the evaluation techniques used are a probable cause of these differences. Polymerase chain reaction (PCR) is the most rapid and sensitive method for detecting DNA in any type of sample; however, the use of PCR with a suitable pair of primers is critical[15].
Collection of fresh cancerous tissues on a large scale is difficult. On the other hand, examination of fresh tissues allows samples to be evaluated in a short period of time. Therefore, formalin-fixed, paraffin-embedded (FFPE) archived tissue specimens were used in this study. Various methods are available to examine paraffin-embedded tissues, such as immunohistochemistry or other techniques for extraction and examination of DNA or RNA. Among these different methods, PCR has been confirmed to be a rapid and particularly sensitive method for examining DNA from paraffin-embedded tissues. Adequate storage of samples and the production of high-quality extracted DNA are important factors in obtaining accurate PCR results.
ESCC specimens were obtained from a pathology department in Tangshan from 2011 to 2013. The samples comprised 198 FFPE tissue blocks of histologically confirmed ESCC.
The FFPE tissue blocks were cut into 20-μm sections and dewaxed by incubation for 2 h in pure xylene with remixing of the tube contents every 45 min. The remixing was repeated seven times, and the dewaxed samples were then washed and dehydrated with 100% ethanol six times. They were then dried at room temperature. The deparaffinized samples were digested with 600 μL of lysis buffer [10 mmol/L Tris-HCL (pH 8.0), 0.1 mol/L EDTA, 0.5% SDS, and 20 μg/mL RNase A]. Next, 20 μL of 20 μg/mL proteinase was added. After mixing, the tubes were incubated at 55 °C for 12 h with remixing of the tube contents every 2 h. After complete digestion, DNA was extracted by phenol/chloroform precipitation. This process was performed twice for every sample and then followed by one phenol/chloroform precipitation. The DNA was precipitated with 100% ethanol and incubated at -20 °C overnight. After precipitation of the DNA, the tubes were centrifuged at 13000 rpm for 15 min, rewashed with 75% ethanol, and dried at room temperature. Finally, 80 μL of TE buffer was added to every tube, and tubes were incubated at 4 °C for a few hours to dissolve the precipitated DNA.
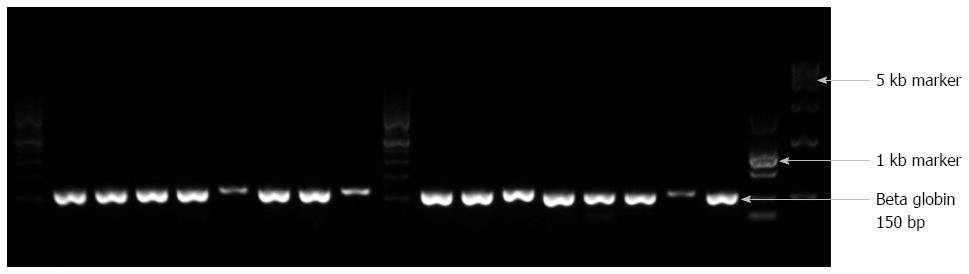
The obtained DNA was amplified for the β-globin gene using a Takara PCR kit (Takara Inc., Otsu, Shiga, Japan) and PC04/GH20 primers to evaluate the performance of the DNA extraction (Table 1). A GP5+/GP6+ primer set was used with a low annealing temperature and small PCR product of 150 bp to detect a wide range of HPV types[16-18]. Two sets of primers were used for specific detection of HPV types 16 and 18. The annealing temperature and PCR products of these two sets are mentioned in Table 1. In each batch, water was used as the negative control and the genome of HPV-positive cervical cells was used as the positive control. The PCR products were checked on a 1.0% agarose gel and visualized by ethidium bromide staining.
| Primer | Sequence, 5’-3’ | Annealing temperature,°C | PCR product, bp |
| PC04 | CAACTTCATCCACGTTCACC | 62 | 150 |
| GH20 | GAAGAGCCAAGGACAGGTAC | ||
| GP5+ | TTTGTTACTGTGGTAGATACTAC | 55 | 150 |
| GP6+ | CTTATACTAAATGTCAAATAAAAAG | ||
| HPV16E6F | CAACAAGACATACATCGACC | 60 | 350 |
| R | CAACAAGACATACATCGACC | ||
| HPV18E6F | CACTTCACTGCAAGACATAGA | 55 | 350 |
| R | GTTGTGAAATCGTCGTTTTTCA |
All PCR-positive products were sequenced and analyzed using the T7 sequence version 2.0 DNA PCR product sequencing kit (Affymetrix, Santa Clara, CA, United States) to identify the HPV types and any sequence variations. The nucleotide sequences were subsequently confirmed using the Basic Local Alignment Search Tool (BLAST).
All PCR processes were performed twice to confirm the presence of the PCR products of interest. The products of the first PCR process were used as the template for the second PCR process. During the second PCR process, the amounts of all PCR mixture materials excluding the 8-μL template were similar to those in the first PCR process. Table 2 summarizes the results of every PCR process of all 198 ESCC specimens examined in this study. All results shown in Table 2 are those of the second PCR process.
The DNA extraction method seemed to be more effective than other previously reported methods. After DNA extraction, all samples were subjected to β-globin PCR to test the quality of the extracted DNA. The DNA quality was good in 190/198 (> 98%) of the ESCC specimens. PCR using the PC04/GH20 primer pair resulted in clearly differentiated DNA fragments of 150 bp (Figure 1).
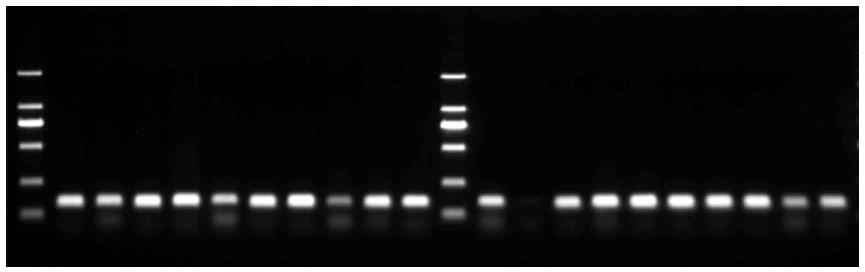
After β-globin PCR, all 198 specimens were examined using the GP5+/GP6+ primer set to visualize the HPV-positive cases on an agarose gel. PCR using the GP5+/GP6+ primer pair resulted in clearly differentiated DNA fragments of 150 bp (Figure 2). In total, 158/198 (79.79%) specimens were positive for HPV as indicated by the presence of the 150-bp PCR product.
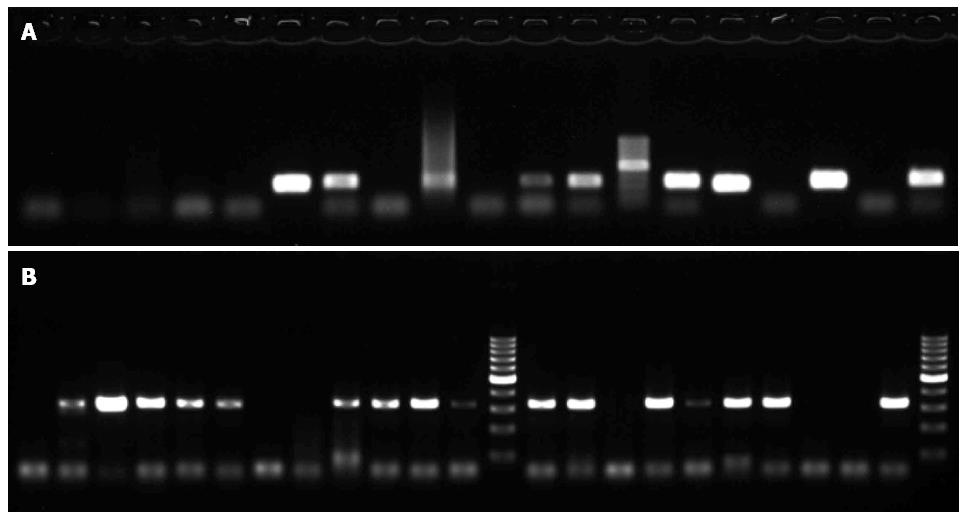
All samples were amplified using the primer sets specific for HPV 16 (JHPV16E6F/JHPV16E6R) and HPV 18 (X18E6F/X18E6R) with the sequences listed in Table 1. These two PCR processes yielded an intense band of the correct size (350 bp) for both HPV types 16 and 18 (Figure 3). Of all 198 specimens, 80 (40.40%) were positive for HPV 16, and 94 (47.47%) were positive for HPV 18 as indicated by the presence of the corresponding PCR products (Table 2).
The DNA sequences of all HPV-positive PCR products were analyzed with DNAman software, and the results were compared with the known HPV sequences in the DNA database using BLAST to identify the various HPV types. The specimens contained DNA of both HPV types 16 and 18. The DNA software analysis results confirmed the presence of HPV DNA with a few mutations in the viral DNA (Figure 4).
ESCC has one of the highest mortality rates worldwide. In some countries with a low prevalence of ESCC, tobacco and alcohol use may be the main causative factors of ESCC. However, in regions with a high incidence of ESCC, including China and northern Iran, only a small portion of ESCC cases are related to smoking or alcohol consumption. Therefore, other risk factors should be considered to help explain the high incidence of ESCC in these areas. Probable factors that could be responsible for this high incidence include a low intake of fruits and vegetables, drinking of hot tea, consumption of opium products and tobacco, Helicobacter pylori infection, contaminated water sources, and genetic susceptibility[19]. Polycyclic aromatic hydrocarbons and N-nitroso compounds are the most important mutagens that cause esophagus cancer[20]. Since the first study by Syrjänen in 1982[7], several studies have been performed in different countries and in different parts of the same country to identify and confirm the involvement of HPV in the development of ESCC. Many studies have shown the presence of the HPV genome in DNA isolated from esophageal cancer tissues; however, their results are conflicting[21-28]. On the other hand, there are differences in the HPV prevalence rates from the same areas. For example, although Chang et al[29] and Lu et al[30] reported a high incidence (36.8%) of HPV infection in patients with ESCC in northern Iran, another study found no HPV infection in 92 patients with ESCC in another Iranian city (Shiraz). Similarly, studies from Australia using the same evaluation method reported different HPV infection rates of 50% and 23% in patients with esophageal cancer[31,32]. Using molecular methods, the majority of these studies have shown the presence of high-risk HPV types in a variable proportion of cases[33-35]. However, HPV has not been identified as an etiologic agent of esophageal cancer, even in highly prevalent regions[13,36].
In the present study, we examined tissue specimens from 198 patients with a pathologic diagnosis of ESCC and found a high prevalence of HPV DNA (approximately 67% of all specimens). These rates are clearly higher than in some previous studies in other regions of the same country (China). In another study, although the evaluation methods, PCR processes, and samples (paraffin-embedded cancerous tissues) were almost identical, the rates in Shandong and Gansu were lower than in Shantou[12]. In the present study, 19.0% of the specimens were HPV-positive in contrast to 77.2% reported in a previous evaluation in Shantou[10,37]. Previous studies performed throughout the world, including China, have shown a high incidence of HPV DNA in almost all samples examined. These results implicate HPV as a suspected causative agent of esophageal cancer in addition to other etiologies (environmental or genetic). Several factors may contribute to the controversial results among different countries or among different regions of the same country: sample preparation (including all processes from intraoperative cutting of the suspected tissue by the surgeon to DNA extraction and analysis for the viral genome), sensitivity of the evaluation method (any item related to analysis of the HPV genome, including instrument sensitivity and the use of high-quality materials during DNA analysis in the PCR procedure), and regional diet habits or other special customs.
In conclusion, a potential role of HPV in the development of ESCC has emerged as evidenced by the HPV-like histologic changes in the mucosa of patients with esophageal cancer and the presence of HPV antigens and HPV DNA in cancerous tissues. Although various types of HPV have been detected by PCR amplification and other methods, clear evidence of the etiologic significance of HPV in ESCC is still lacking. Considering that no studies on the relationship between HPV and esophageal cancer have been performed in Tangshan, China, we hope that the present study promotes further efforts to confirm the etiologic significance of HPV in the development of ESCC.
Human papillomavirus (HPV) was introduced as a possible causative agent in the development of esophageal squamous cell carcinoma (ESCC) in 1982. Many studies have since been performed to prove this relationship; however, the results have been controversial.
Many recent studies have focused on the relationship between many cancers and high-risk HPV infection. In this study, we focused on Tangshan, China, one area characterized by a high incidence of ESCC. HPV may be an important cause of the occurrence of ESCC. The authors anticipate that the results of this study will encourage researchers to launch new studies in other regions of China with a high incidence of ESCC to confirm this relationship. Their findings will also provide new ideas for additional studies on potential vaccines for HPV infection-induced ESCC.
In this study, 79.79% of all ESCC specimens were positive for HPV. Differentially, 40.40% and 47.47% were positive for HPV 16 and 18, respectively. Conflicting results from different regions of China have also been reported, with HPV positive rates ranging from 0 to 71%.
One possible reason for the variability in HPV infection rates is the use of different evaluation methods. In this study, the most sensitive and common method (PCR) was used to evaluate cancerous tissues. The use of other complementary methods to confirm the PCR results is ideal.
More than 150 types of HPV exist, and they may be divided into two groups: high risk and low risk. Both groups can induce growth of abnormal cells, but only the high-risk types, specifically types 16 and 18, lead to malignant transformation.
This study introduced HPV as a probable cause of esophageal carcinoma. However, for confirmation of this causative relationship, other studies using different methods should be performed in other geographic areas. One limitation of this study was the lack of an ideal negative control; normal esophageal tissue is too difficult to obtain. This study can promote research using other methods or further research in other regions to clarify the etiologic significance of HPV in ESCC.
P- Reviewer: Tovey FI S- Editor: Ma YJ L- Editor: Wang TQ E- Editor: Ma S
| 1. | Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893-2917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11128] [Cited by in RCA: 11832] [Article Influence: 845.1] [Reference Citation Analysis (4)] |
| 2. | Muñoz N, Bosch FX, de Sanjosé S, Shah KV. The role of HPV in the etiology of cervical cancer. Mutat Res. 1994;305:293-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 3. | Walboomers JM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV, Snijders PJ, Peto J, Meijer CJ, Muñoz N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189:12-19. [PubMed] |
| 4. | zur Hausen H. Human papillomaviruses in the pathogenesis of anogenital cancer. Virology. 1991;184:9-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 642] [Cited by in RCA: 626] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 5. | Gillison ML, Shah KV. Chapter 9: Role of mucosal human papillomavirus in nongenital cancers. J Natl Cancer Inst Monogr. 2003;57-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 127] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 6. | Snijders PJ, Steenbergen RD, Meijer CJ, Walboomers JM. Role of human papillomaviruses in cancer of the respiratory and upper digestive tract. Clin Dermatol. 1997;15:415-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Syrjänen KJ. Histological changes identical to those of condylomatous lesions found in esophageal squamous cell carcinomas. Arch Geschwulstforsch. 1982;52:283-292. [PubMed] |
| 8. | Syrjänen KJ. HPV infections and oesophageal cancer. J Clin Pathol. 2002;55:721-728. [PubMed] |
| 9. | Noori S, Monabati A, Ghaderi A. The prevalence of human papilloma virus in esophageal squamous cell carcinoma. Iran J Med Sci. 2012;37:126-133. [PubMed] |
| 10. | Stewart BW, Kleihues P, editors . World cancer report. Lyon: IARC press 2003; 223-227. |
| 11. | Bosetti C, Malvezzi M, Chatenoud L, Negri E, Levi F, La Vecchia C. Trends in cancer mortality in the Americas, 1970-2000. Ann Oncol. 2005;16:489-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 97] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 12. | Li T, Lu ZM, Chen KN, Guo M, Xing HP, Mei Q, Yang HH, Lechner JF, Ke Y. Human papillomavirus type 16 is an important infectious factor in the high incidence of esophageal cancer in Anyang area of China. Carcinogenesis. 2001;22:929-934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 72] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Gao GF, Roth MJ, Wei WQ, Abnet CC, Chen F, Lu N, Zhao FH, Li XQ, Wang GQ, Taylor PR. No association between HPV infection and the neoplastic progression of esophageal squamous cell carcinoma: result from a cross-sectional study in a high-risk region of China. Int J Cancer. 2006;119:1354-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Sur M, Cooper K. The role of the human papilloma virus in esophageal cancer. Pathology. 1998;30:348-354. [PubMed] |
| 15. | Van den Brule AJC, Snijders PJF, Meijer CJLM, Walboomers JMM. PCR-based detection of genital HPV genotype: an update and future perspectives. Papillomavirus Report. 1993;4:95-99. |
| 16. | Qu W, Jiang G, Cruz Y, Chang CJ, Ho GY, Klein RS, Burk RD. PCR detection of human papillomavirus: comparison between MY09/MY11 and GP5+/GP6+ primer systems. J Clin Microbiol. 1997;35:1304-1310. [PubMed] |
| 17. | Baay MF, Quint WG, Koudstaal J, Hollema H, Duk JM, Burger MP, Stolz E, Herbrink P. Comprehensive study of several general and type-specific primer pairs for detection of human papillomavirus DNA by PCR in paraffin-embedded cervical carcinomas. J Clin Microbiol. 1996;34:745-747. [PubMed] |
| 18. | Zehbe I, Wilander E. Two consensus primer systems and nested polymerase chain reaction for human papillomavirus detection in cervical biopsies: A study of sensitivity. Hum Pathol. 1996;27:812-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Farhadi M, Tahmasebi Z, Merat S, Kamangar F, Nasrollahzadeh D, Malekzadeh R. Human papillomavirus in squamous cell carcinoma of esophagus in a high-risk population. World J Gastroenterol. 2005;11:1200-1203. [PubMed] |
| 20. | Sadjadi AR, Marjani H, SemnaniSh , Naseri-Mogaddam S. Oesophageal cancer in Iran: a review. Middle East J cancer. 2010;1:5-14. |
| 21. | Sagar PM. Aetiology of cancer of the oesophagus: geographical studies in the footsteps of Marco Polo and beyond. Gut. 1989;30:561-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 22. | Chalasani N, Wo JM, Waring JP. Racial differences in the histology, location, and risk factors of esophageal cancer. J Clin Gastroenterol. 1998;26:11-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Sitas F, Madhoo J, Wessie J. Incidence of histologically diagnosed cancer in south Africa. Johannesburg: South Africa Institute for medical research 1998; . |
| 24. | Loke SL, Ma L, Wang M. Human papillomavirus in oesophageal squamous cell carcinoma. J Clin Pathol. 1990;27:812-815. |
| 25. | West AB, Soloway GN, Lizarraga G, Tyrrell L, Longley JB. Type 73 human papillomavirus in esophageal squamous cell carcinoma: a novel association. Cancer. 1996;77:2440-2444. [PubMed] |
| 26. | Poljak M, Cerar A, Seme K. Human papillomavirus infection in esophageal carcinomas: a study of 121 lesions using multiple broad-spectrum polymerase chain reactions and literature review. Hum Pathol. 1998;29:266-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Miller BA, Davidson M, Myerson D, Icenogle J, Lanier AP, Tan J, Beckmann AM. Human papillomavirus type 16 DNA in esophageal carcinomas from Alaska Natives. Int J Cancer. 1997;71:218-222. [PubMed] |
| 28. | Chang F, Syrjänen S, Shen Q, Wang L, Wang D, Syrjänen K. Human papillomavirus involvement in esophageal precancerous lesions and squamous cell carcinomas as evidenced by microscopy and different DNA techniques. Scand J Gastroenterol. 1992;27:553-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 29. | Chang F, Syrjänen S, Shen Q, Wang L, Syrjänen K. Screening for human papillomavirus infections in esophageal squamous cell carcinomas by in situ hybridization. Cancer. 1993;72:2525-2530. [PubMed] |
| 30. | Lu S, Luo F, Li H. [Detection of human papilloma virus in esophageal squamous cell carcinoma and adjacent tissue specimens in Linxian]. Zhonghua Zhong Liu Zazhi. 1995;17:321-324. [PubMed] |
| 31. | Kulski J, Demeter T, Sterrett GF, Shilkin KB. Human papilloma virus DNA in oesophageal carcinoma. Lancet. 1986;2:683-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 32. | Kulski JK, Demeter T, Mutavdzic S, Sterrett GF, Mitchell KM, Pixley EC. Survey of histologic specimens of human cancer for human papillomavirus types 6/11/16/18 by filter in situ hybridization. Am J Clin Pathol. 1990;94:566-570. [PubMed] |
| 33. | Yao PF, Li GC, Li J, Xia HS, Yang XL, Huang HY, Fu YG, Wang RQ, Wang XY, Sha JW. Evidence of human papilloma virus infection and its epidemiology in esophageal squamous cell carcinoma. World J Gastroenterol. 2006;12:1352-1355. [PubMed] |
| 34. | Matsha T, Erasmus R, Kafuko AB, Mugwanya D, Stepien A, Parker MI. Human papillomavirus associated with oesophageal cancer. J Clin Pathol. 2002;55:587-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 35. | Shuyama K, Castillo A, Aguayo F, Sun Q, Khan N, Koriyama C, Akiba S. Human papillomavirus in high- and low-risk areas of oesophageal squamous cell carcinoma in China. Br J Cancer. 2007;96:1554-1559. [PubMed] |
| 36. | Kamangar F, Qiao YL, Schiller JT, Dawsey SM, Fears T, Sun XD, Abnet CC, Zhao P, Taylor PR, Mark SD. Human papillomavirus serology and the risk of esophageal and gastric cancers: results from a cohort in a high-risk region in China. Int J Cancer. 2006;119:579-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 37. | Zhang QY, Zhang DH, Shen ZY, Xu LY, Li EM, Au WW. Infection and integration of human papillomavirus in esophageal carcinoma. Int J Hyg Environ Health. 2011;214:156-161. [PubMed] |