Published online Feb 28, 2014. doi: 10.3748/wjg.v20.i8.2071
Revised: November 15, 2013
Accepted: December 3, 2013
Published online: February 28, 2014
Processing time: 205 Days and 19.6 Hours
AIM: To investigate the mechanisms of chloride intracellular channel 1 (CLIC1) in the metastasis of colon cancer under hypoxia-reoxygenation (H-R) conditions.
METHODS: Fluorescent probes were used to detect reactive oxygen species (ROS) in LOVO cells. Wound healing assay and transwell assay were performed to examine the migration and invasion of LOVO cells. Expression of CLIC1 mRNA and protein, p-ERK, MMP-2 and MMP-9 proteins was analyzed by reverse transcription-polymerase chain reaction and Western blot.
METHODS: H-R treatment increased the intracellular ROS level in LOVO cells. The mRNA and protein expression of CLIC1 was elevated under H-R conditions. Functional inhibition of CLIC1 markedly decreased the H-R-enhanced ROS generation, cell migration, invasion and phosphorylation of ERK in treated LOVO cells. Additionally, the expression of MMP-2 and MMP-9 could be regulated by CLIC1-mediated ROS/ERK pathway.
CONCLUSION: Our results suggest that CLIC1 protein is involved in the metastasis of colon cancer LOVO cells via regulating the ROS/ERK pathway in the H-R process.
Core tip: Hypoxia-reoxygenation (H-R) treatment increases the intracellular reactive oxygen species (ROS) level to activate the MAPK/ERK pathway, resulting in the promotion of migration and invasion in colon cancer LOVO cells. Inhibition of chloride intracellular channel 1 (CLIC1) using specific inhibitor IAA94 can markedly decrease the H-R-enhanced ROS generation, migration, invasion and phosphorylation of ERK. The results presented in the current study suggest that CLIC1 is involved in the metastasis of colon cancer LOVO cells via regulating the ROS/ERK pathway in the H-R process.
- Citation: Wang P, Zeng Y, Liu T, Zhang C, Yu PW, Hao YX, Luo HX, Liu G. Chloride intracellular channel 1 regulates colon cancer cell migration and invasion through ROS/ERK pathway. World J Gastroenterol 2014; 20(8): 2071-2078
- URL: https://www.wjgnet.com/1007-9327/full/v20/i8/2071.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i8.2071
Colorectal cancer is one of the most common malignancies that result in the death of people in the world[1,2]. It is important for patients with colorectal cancer to get early detection and treatment. The 5-year survival rate in patients with colorectal cancer at early stages is higher than 90%; however, it drops to less than 10% in patients with distant metastases[3]. It is well known that the tumor microenvironment can influence the progression and metastasis of various cancer models, including colon cancer. Emerging evidence has suggested that local hypoxia is a common feature in the solid tumor microenvironment. Tumor cells exposed to hypoxic conditions may undergo transformation to a more aggressive phenotype to promote the metastasis of cancer[4].
However, little is known about hypoxia and reoxygenation (H-R) microenvironment that occurs in tumors. Recent discoveries have shown that cancer cells are under the H-R microenvironment attributing to irregular microvascular network and blood flow patterns[5,6]. Permanent or transient limitations in blood perfusion may contribute to the migration and invasion of cancer cells under the H-R process[7,8]. It is known that during the H-R process the cells can produce abundant reactive oxygen species (ROS), leading to the injury of membrane proteins and nucleic acids of organisms and the damage to cells and tissues[9]. However, recent reports indicated that ROS production in cancer cells can function as a secondary signaling molecule, which has been shown to play a role in cell proliferation, apoptosis, differentiation[10], migration and invasion[4] in cancer.
Chloride channel 1 (CLIC1, formerly NCC27), a member of the CLIC family, was first cloned and identified by subtractive cloning in 1997[11]. Recent studies have indicated that CLIC1 is significantly up-regulated in tumor tissues, such as gastric carcinoma and lung carcinoma[12,13]. In addition, CLIC1, proposed as a novel potential prognostic factor, was significantly up-regulated in gastric cancer and strongly correlated with lymph node metastasis[12]. It was presumed that elevated expression of CLIC1 can modulate cell division and anti-apoptosis signaling, resulting in cellular transformation[14,15]. Moreover, CLIC1 may act as a “sensor” and an “effector” of the redox state of the cells caused by oxidative stress[16], and it is known that the initiation and progression of cancer are closely correlated with redox state disequilibrium in cells[10]. CLIC1 also contributes to acquisition of the radioresistant phenotype of laryngeal cancer through regulation of ROS production[17], and ROS up-regulation can result in increasing cell motility and invasiveness of cancer cells[4]. In our previous study, we found that CLIC1 participates in colonic carcinoma metastasis under H-R conditions[18]. However, the molecular mechanisms of CLIC1 in colon cancer metastasis remain unclear.
In this study, our data showed that H-R treatment increased the intracellular ROS level to activate the MAPK/ERK pathway, resulting in the promotion of migration and invasion in colon cancer LOVO cells. The mRNA and protein expression of CLIC1 was elevated under H-R conditions. Functional inhibition of CLIC1 using specific inhibitor IAA94 markedly decreased H-R-enhanced ROS generation, cell migration, invasion and phosphorylation of ERK in treated colon cancer LOVO cells. Additionally, the expression of MMP-2 and MMP-9, two important mediators of cancer metastasis, could be regulated by CLIC1-mediated ROS/ERK pathway. The results presented in the current study suggest that CLIC1 protein is involved in the metastasis of colon cancer LOVO cells via regulating the ROS/ERK pathway in the H-R process.
The human colon cancer cell line LOVO was incubated in Dulbecco’s modified Eagle’s medium (DMEM) plus 10% (v/v) fetal calf serum (FCS) (Hyclone, United States), at 37 °C in a humidified atmosphere of 5% CO2 in air. The generation of H-R conditions was performed as previously described[7,8]. Briefly, cells were cultured in an air-tight hypoxic (5% CO2 and 95% N2) chamber incubator (Thermo Electron, Waltham, MA, United States) for 4 h, rapidly transferred to an incubator with a humidified atmosphere of 5% CO2, and additionally cultured for 20 h. For normoxia (N) control treatment, cells were maintained in a humidified incubator with a 95% air/5% CO2 atmosphere for the same period of time as the H-R groups.
IAA94 was purchased from Sigma and prepared in dimethylsulphoxide. Specific inhibitor of NADPH [diphenyleneiodonium (DPI)] was from Sigma Chemical Co. (St. Louis, MO, United States). Fluorescent probe DCFH-DA, inhibitors of ROS [N-acetylcysteine (NAC)] and MAPK/ERK (PD98059) were purchased from Beyotime Institute of Biotechnology (Nantong, Jiangsu, China). Antibodies against CLIC1, MMP-2, MMP-9, total-ERK and phospho-ERK were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, United States).
LOVO cells were trypsinized and cultured in 96 well plates (1 × 104 cell/well). To determine the effect of specific inhibitors on ROS production, cells were pretreated with DPI (15 μmol/L), NAC (30 mmol/L) or IAA94 (1, 20 and 40 μmol/L) for 1 h before H-R treatment. For DCF-DA ROS measurements, culture medium was replaced with regular culture medium without FCS containing 10 μmol/L of DCF-DA for 30 min. Cells were rinsed with DMEM without FCS, and fluorescence was then measured at 488 nm for excitation and 525 nm for emission with the Fluoroscan Ascent FL fluorimeter (Labsystems, France). All measurements were performed at 37 °C.
Cells were cultured to a confluent monolayer in 6-well plates. A sterile 200 μL pipette tip was used to scratch the cell monolayer to form a wound. For the wound healing assays under H-R conditions, cells were pretreated with DPI (15 μmol/L), NAC (30 mmol/L), PD98059 (50 μmol/L) or IAA94 (1, 20 and 40 μmol/L) for 1 h. Pictures of the wound area were taken at 0 and 24 h at × 100 magnification.
The in vitro invasive ability of LOVO cells was tested by the Boyden chamber invasion assay. Matrigel (BD Biosciences) was diluted with cold filtered distilled water, and added to 8-μm pore size poly-carbonate membrane filters. The cells were trypsinized and seeded to the upper part of Boyden chamber at a density of 3 × 105 cells/mL in 300 μL of serum-free medium. The bottom chamber contained medium with 10% FCS as a chemoattractant. Cells were preloaded with DPI (15 μmol/L), NAC (30 mmol/L), PD98059 (50 μmol/L) or IAA94 (1, 20 and 40 μmol/L) for 1 h before H-R. After the incubation time was complete (6 h hypoxia followed by 18 h reoxygenation or 24 h normoxia), the cells that had invaded to the lower surface of the membrane were fixed with paraformaldehyde, and stained with crystal violet. The cells were counted in five randomly selected fields under a microscope at × 400 magnification.
Total RNA was extracted from cells using the Simply P RNA Extraction kit (Bioer Biotech Co., Latd) according to the manufacturer’s instructions. Total RNA (1 µg) was reverse-transcribed into cDNA using the Reverse Transcript Kit (Cwbio Biotech Co, China), and amplified by polymerase chain reaction (PCR). For PCR, 1/25 of the reverse transcription reaction mixture was amplified using 35 cycles for CLIC1 and 35 cycles for GAPDH. Amplified products were separated by electrophoresis on a 2% agarose gel and photographed. The sequences of the primers (Sagon biotech Co., China) used in the real-time RT-PCR were as follows: CLIC1: 5’-GTATTGACCAGTCTCCCTTCCAGC-3’ (forward) and 5’-GGTCTTTATGAGGAGGTCGTGGG-3’ (reverse); GAPDH: 5’-TCATGAAGTGTGACGTTGACATCCGT-3’ (forward) and 5’-CCTAGAAGCATTTGCGGTGCACGATG-3’ (reverse).
Briefly, proteins were separated on a 10% denaturing polyacrylamide gel and electro-transferred to Immuno-Blot nitrocellulose membranes. The membranes were blocked in TBS-T containing 5% fat-free dry milk and then incubated with a primary antibody overnight at 4 °C, followed by incubation with a horseradish peroxidase-conjugated secondary antibody for 1 h. Primary antibodies against ERK (1:400), p-ERK (1:400), MMP-2 (1:200), MMP-9 (1:200) and GAPDH (1:1000) were used. Proteins were detected using ECL reagents (GE Healthcare, NJ, United States).
All data are expressed as mean ± SD. The data for each condition were subjected to analysis of variance followed by the Student-Newman-Keuls test for comparisons between the means. Differences were considered significant when P < 0.05.
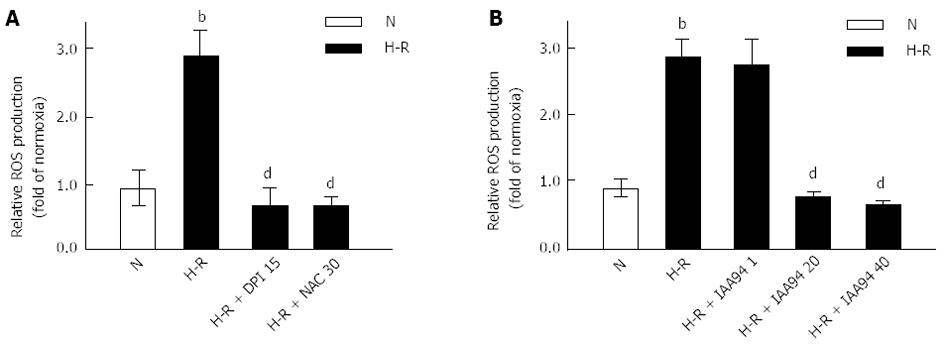
Cells were cultured with DPI (15 μmol/L), NAC (30 mmol/L) or IAA 94 (1, 20 and 40 μmol/L) for 1 h before H-R, and control cells were only incubated in a humidified atmosphere of 95% air/5% CO2. Intracellular ROS were measured by preloading the cells with DCF-DA followed by H-R or normoxia treatment. As shown in Figure 1, the level of intracellular ROS was significantly increased in cells after exposure to H-R compared with normoxia (P < 0.01). Further investigation indicated that the ROS level was significantly abated in cells pretreated with DPI (15 μmol/L) or NAC (30 mmol/L) compared with H-R cells (P < 0.01) (Figure 1A). These data provide evidence that ROS production is involved in the H-R process in colon cancer LOVO cells, which is consistent with the finding of a previous study[8].
To determine whether inhibition of CLIC1 could reduce ROS generation during the H-R treatments, cells were cultured in the presence of CLIC1 blocker IAA94. As shown in Figure 1B, the intracellular ROS level was significantly increased under H-R conditions. However, preloading with 20 or 40 μmol/L of IAA94 significantly decreased the ROS levels in LOVO cells (P < 0.01). These results suggest that CLIC1 can regulate the intracellular ROS production in colon cancer cells in the H-R process.
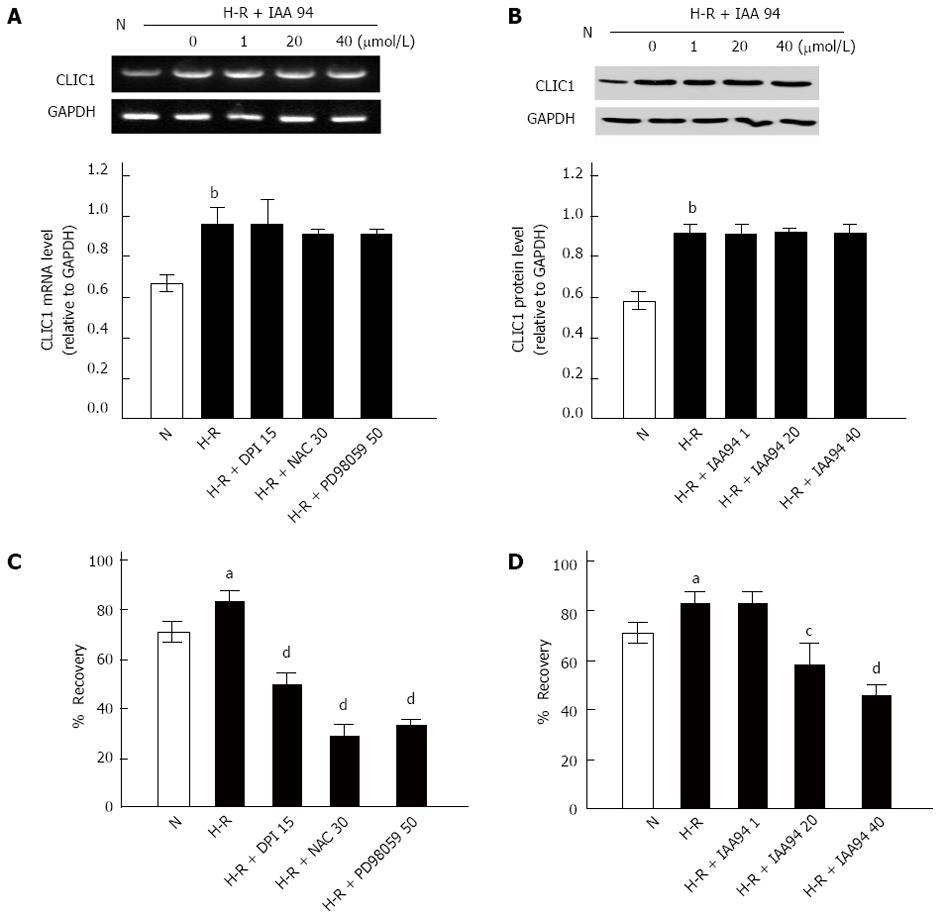
It has been previously documented that CLIC1 may act as a “sensor” and an “effector” of the oxidative stress of the cells[16], and H-R circumstances are closely correlated with metastasis of colon cancer[7]. Therefore, in our present study, the mRNA and protein expression of CLIC1 was determined by RT-PCR and Western blot under H-R conditions, respectively. As shown in Figure 2, CLIC1 mRNA and protein expression was significantly elevated in the H-R group compared with the normoxia control group (P < 0.01). However, we found no significant changes in CLIC1 mRNA or protein expression in LOVO cells treated with IAA94 at different concentrations (1, 20 or 40 μmol/L) (Figure 2). Our results suggest that functional inhibition of CLIC1 can play a role in down-regulating ROS generation in colon cancer.
During cancer metastasis process, tumor cells must firstly undergo several morphological changes so that they could pass through narrow extracellular spaces to metastasize[18]. Recent studies showed that ROS up-regulation could lead to morphological transformation and increase cell motility[19]. The effect of the ROS/ERK pathway on cell motility during the H-R treatments was then assessed using wound-healing assays. As shown in Figure 2C and D, the mobility of LOVO cells was increased after exposure to H-R conditions (P < 0.05). This effect was significantly suppressed by NAC, DPI or PD 98059, respectively (Figure 2C). These results implied that the ROS/ERK pathway could regulate the migration of colon cancer cells under H-R conditions. Furthermore, as shown in Figure 2D, preloading LOVO cells with IAA 94 (20 or 40 μmol/L) could also decrease the cell motility potential for H-R, and the effect was dose-dependent. All these data provide evidence that CLIC1 is involved in the migration of colon cancer cells under H-R conditions.
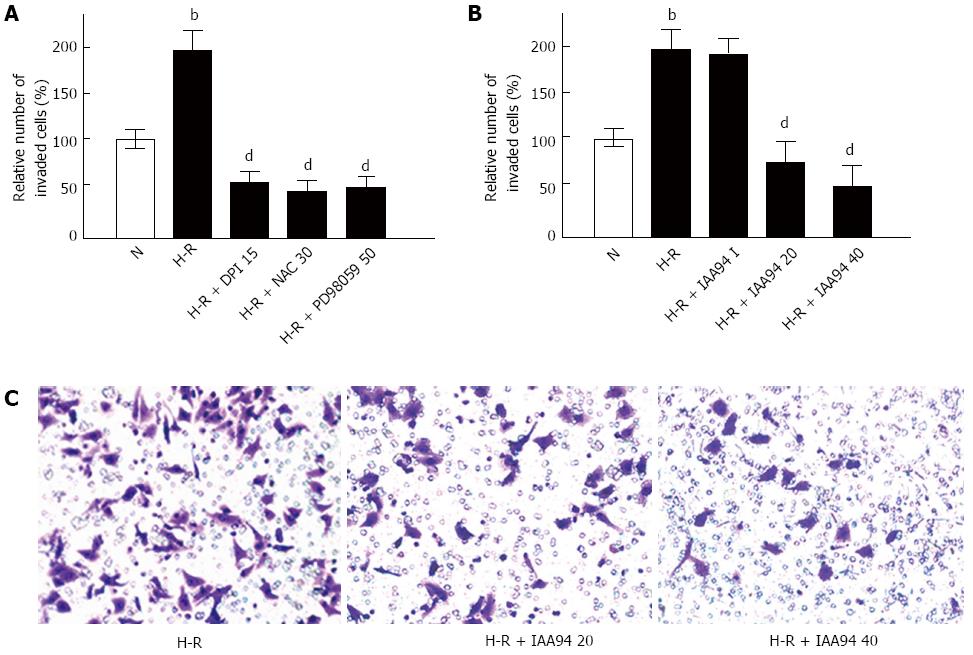
The effect of CLIC1 on colon cancer cell invasiveness was examined by Matrigel invasion assays in vitro. It was found that H-R caused a remarkable increase in the invasiveness of LOVO cells (Figure 3A). However, treatment with DPI (15 μmol/L), NAC (30 mmol/L) or PD 98059 (15 μmol/L) could significantly prevent colonic cancer cells from invading after H-R exposure. As expected, a similar effect of inhibiting CLIC1 on LOVO cells invasion was observed. After pretreatment with 20 or 40 μmol/L of IAA94 for 1 h, the invasiveness of LOVO cells was significantly decreased under H-R conditions (Figure 3B and C). These results suggest that H-R can promote the invasiveness of colon cancer cells, and the effect can be inhibited by CLIC1 under H-R conditions.
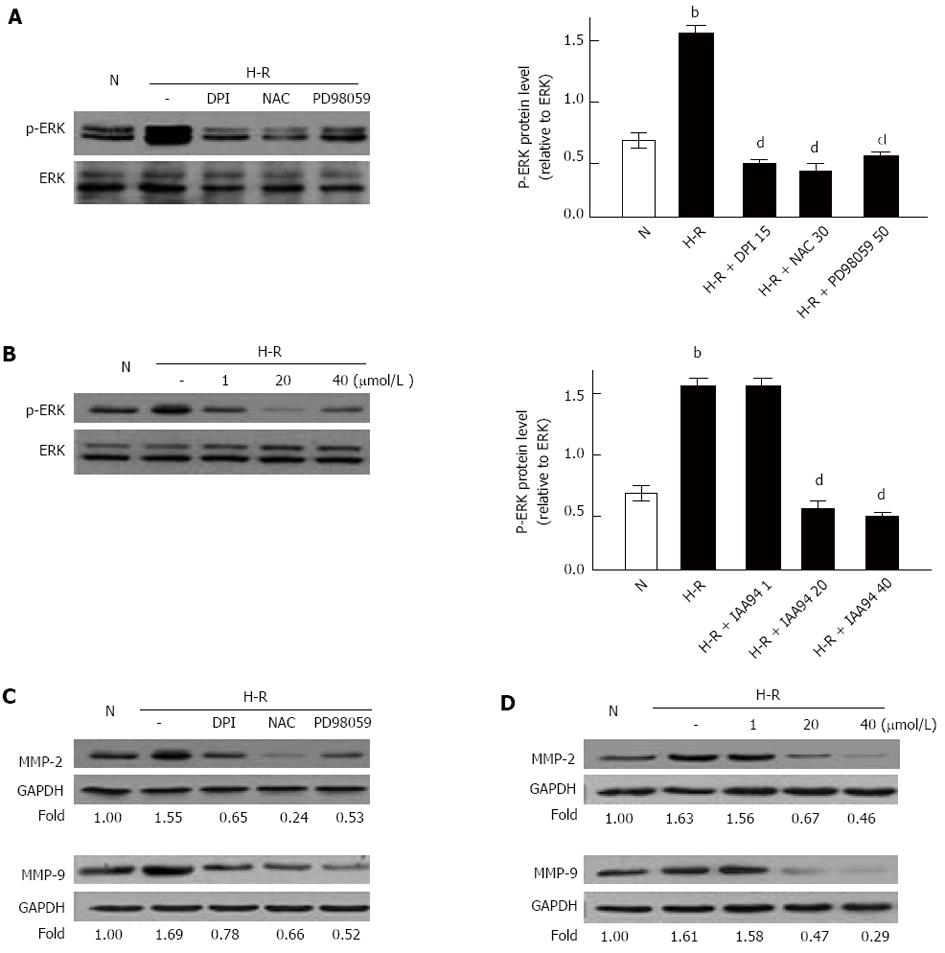
It has been reported that MAPK/ERK and ROS/ERK pathways are involved in the metastasis of cancer[4,20]. However, it is still unknown whether those pathways are correlated with metastasis of colon cancer under H-R conditions. In our present study, the expression of phosphorylated ERK (p-ERK), MMP-2 and MMP-9 was monitored by Western blot analysis in the H-R process. Our results suggested that p-ERK, MMP-2 and MMP-9 proteins were significantly elevated in H-R conditions when compared with the normoxia control (Figure 4). However, p-ERK, MMP-2 and MMP-9 protein levels in H-R conditions were strongly down-regulated with addition of DPI or NAC (Figure 4A and C). These findings implied that ROS could activate the ERK/MMPs pathway in H-R conditions. We also examined the effect of CLIC1 on the ERK/MMPs pathway by Western blot analysis. As shown in Figure 4, pretreatment with IAA 94 (20 or 40 μmol/L) significantly decreased p-ERK protein, MMP-2 and MMP-9 protein levels under H-R conditions in a dose dependent manner. ERK inhibitor PD 98059 could also decrease MMP-2 and MMP-2 protein levels under H-R conditions. Taken together, our findings demonstrated that the ROS/ERK pathway is involved in the metastasis of colonic cancer, and ERK/MMPs pathway is regulated by CLIC1 via regulating ROS production under H-R conditions.
Under physiological circumstances, ROS can be continually eliminated by endosomatic antioxidase, which is dependent on the stable system of cellular redox state. It has been demonstrated that cancer cells are characterized by the defect of redox system development, and persistently elevated intracellular ROS can function as second messengers and are involved in proliferation, differentiation, migration, and invasion of cancer cells[4,10,20]. Many solid tumors possess irregular microvascular network and blood flow patterns, which is the main cause that cancer cells are usually present under H-R conditions[5,6]. In the present study, we confirmed that the ROS level was significantly elevated under the H-R conditions in LOVO cells when compared with the normoxia control. Moreover, ROS production was significantly decreased by pretreatment with DPI or NAC under the H-R conditions. Further investigations by wound healing and invasion assays showed that the metastasis potential of colon cancer cells was significantly increased in response to H-R treatments, and this effect was abrogated by preloading with DPI or NAC. Taken together, our results confirmed that ROS play an important role in regulating the migration and invasion of colon cancer cells under H-R conditions. Our findings also suggested that colon cancer cells under H-R microenvironment can undergo transformation to a more aggressive phenotype to promote cancer metastasis.
Previous studies of CLIC1 are mostly focused on its physiological function, its association with non-tumorous disease, and its ability to act as an ion channel[21]. However, some recent studies suggest that the expression of CLIC1 is up-regulated in tumor tissues. In addition, CLIC1 was significantly up-regulated and correlated with metastasis of tumor cells in gastric cancer[14]. Additionally, a recent study has indicated that CLIC1 is highly expressed in colorectal cancer tissues[17]. However, it is still unknown whether CLIC1 plays a role in the metastasis of colon cancer under H-R conditions. In this study, it was found that CLIC1 mRNA and protein expression was significantly elevated under H-R conditions. Preloading with CLIC1 blocker IAA 94 could not affect the expression of CLIC1, but the ROS level was significantly decreased by IAA 94 treatment, suggesting that functional inhibition of CLIC1 channel activity could reduce the intracellular ROS production in the H-R process. This is supported by previous findings that CLIC1 channel activity is increased in the oxidative environment and inhibition of CLIC1 reduces the ROS production via blocking NADPH oxidization[16], and testified by structural analysis of CLIC1 showing that it can dimerize in the presence of strong oxidizing stress[22,23]. In addition, the migration and invasion of colon cancer cells were obviously decreased in the presence of IAA94 under H-R conditions. Taken together, our results indicated that CLIC1 was involved the metastasis of colon cancer through regulating intracellular ROS levels.
We further explored the molecular signaling pathways that may be involved in H-R conditions. It is known that MAPK/ERK and ROS/ERK pathways can promote the metastasis of cancer. The ROS/ERK pathway, involved in cell migration and activation of matrix metalloproteinases, is activated by cell oxidation[4,20]. In the present study, Western blot analysis showed that the MAPK/ERK pathway was activated in H-R conditions. The effect could be suppressed by DPI, NAC or PD98059 treatment. As expected, the MAPK/ERK pathway was significantly blocked by treatment with IAA94. Taken together, our findings implied that CLIC1 promoted the mobility and invasive capacity of cancer by regulating NADPH-derived ROS via the MAPK/ERK pathway. MMPs can degrade the basement membrane and play main roles in promotion of cancer invasion and metastasis[24]. MMP-2 and MMP-9, members of the MMPs family, are thought as the two important mediators of cancer metastasis in patients with colorectal carcinoma[25]. In the present study, we found that both MMP-2 and MMP-9 proteins were up-regulated in LOVO cells under H-R conditions, and the effect was abated by suppressing the ROS production, MAPK/EER pathway or CLIC1. Our results demonstrated that MMP-2 and MMP-9 played an important role in the invasion of colon cancer in the H-R process.
In summary, our findings provide the evidence that H-R conditions act as a relevant key factor for the promotion of colonic cancer metastasis. CLIC1 is an important candidate protein that may serve as an effective metastasis-associated regulator for colon cancer. However, the accumulating evidence suggests that ROS derived from NADPH oxidase can mediate tumor growth and angiogenesis, which is essential for tumor metastasis[26]. Our findings warrant further investigation to explore some other possible molecular mechanisms of CLIC1 in colon cancer metastasis.
Colorectal cancer is one of the most common malignancies that result in the death of people in the world. The tumor microenvironment hypoxia-reoxygenation (H-R) can influence the progression and metastasis of colon cancer but the molecular basis for such a link has not been well understood.
Chloride intracellular channel 1 (CLIC1) is highly expressed in colorectal cancer, and our previous study has reported that CLIC1 participates in the metastasis of colorectal cancer. However, the precise mechanisms of CLIC1 in the metastasis of colonic cancer under H-R conditions is still unknown. In this study, the authors demonstrated that CLIC1 protein was involved in the metastasis of colon cancer LOVO cells via regulating reactive oxygen species (ROS)/ERK pathway in the H-R process.
Although previous studies have demonstrated that CLIC1 is correlated with tumor metastasis, the precise mechanisms are not well understood. In this study, it was found that tumor H-R microenvironment increased the intracellular ROS levels to activate the MAPK/ERK pathway, resulting in the promotion of migration and invasion in colon cancer LOVO cells. The findings, for the first time, provide the evidence that CLIC1 is involved in the metastasis of colon cancer LOVO cells via regulating ROS/ERK pathway under H-R conditions.
The tumor H-R microenvironment can promote the progression and metastasis of colon cancer. By understanding how CLIC1 is involved in the metastasis of colon cancer in the H-R process, this study may represent a future strategy for the treatment of patients with colon cancer.
CLIC1 can act as a ‘‘sensor’’ and an ‘‘effector’’ during changes in the redox state of the cells caused by oxidative stress via regulating the oxidation of GAPDH because of its similar structure to GAPDH. Non-surprisingly, CLIC1 may have some effects on the production of ROS in the H-R process.
In this study, the authors have assessed the relationship between protein expression of CLIC1, which has previously shown to be increased in tumor metastasis, and intracellular ROS level, which in turn activates the MAPK/ERK pathway. The activation of this pathway resulted in the promotion of migration and invasion of colon cancer LOVO cells. This is a new topic which merits further investigation; on this subject, the study is well written and the results are interesting.
P- Reviewer: Franceschi F S- Editor: Zhai HH L- Editor: Wang TQ E- Editor: Wu HL
| 1. | Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74-108. [PubMed] |
| 2. | Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23762] [Cited by in RCA: 25545] [Article Influence: 1824.6] [Reference Citation Analysis (7)] |
| 3. | O’Connell JB, Maggard MA, Ko CY. Colon cancer survival rates with the new American Joint Committee on Cancer sixth edition staging. J Natl Cancer Inst. 2004;96:1420-1425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1063] [Cited by in RCA: 1162] [Article Influence: 55.3] [Reference Citation Analysis (0)] |
| 4. | Law AY, Wong CK. Stanniocalcin-2 promotes epithelial-mesenchymal transition and invasiveness in hypoxic human ovarian cancer cells. Exp Cell Res. 2010;316:3425-3434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 72] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nat Rev Cancer. 2009;9:239-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2949] [Cited by in RCA: 2767] [Article Influence: 172.9] [Reference Citation Analysis (0)] |
| 6. | Vaupel P, Kallinowski F, Okunieff P. Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res. 1989;49:6449-6465. [PubMed] |
| 7. | Kokura S, Yoshida N, Imamoto E, Ueda M, Ishikawa T, Uchiyama K, Kuchide M, Naito Y, Okanoue T, Yoshikawa T. Anoxia/reoxygenation down-regulates the expression of E-cadherin in human colon cancer cell lines. Cancer Lett. 2004;211:79-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 8. | Binker MG, Binker-Cosen AA, Richards D, Gaisano HY, de Cosen RH, Cosen-Binker LI. Hypoxia-reoxygenation increase invasiveness of PANC-1 cells through Rac1/MMP-2. Biochem Biophys Res Commun. 2010;393:371-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 9. | Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8502] [Cited by in RCA: 8906] [Article Influence: 468.7] [Reference Citation Analysis (0)] |
| 10. | Clerkin JS, Naughton R, Quiney C, Cotter TG. Mechanisms of ROS modulated cell survival during carcinogenesis. Cancer Lett. 2008;266:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 165] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 11. | Valenzuela SM, Martin DK, Por SB, Robbins JM, Warton K, Bootcov MR, Schofield PR, Campbell TJ, Breit SN. Molecular cloning and expression of a chloride ion channel of cell nuclei. J Biol Chem. 1997;272:12575-12582. [PubMed] |
| 12. | Chen CD, Wang CS, Huang YH, Chien KY, Liang Y, Chen WJ, Lin KH. Overexpression of CLIC1 in human gastric carcinoma and its clinicopathological significance. Proteomics. 2007;7:155-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 90] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Wang W, Xu X, Wang W, Shao W, Li L, Yin W, Xiu L, Mo M, Zhao J, He Q. The expression and clinical significance of CLIC1 and HSP27 in lung adenocarcinoma. Tumour Biol. 2011;32:1199-1208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Huang JS, Chao CC, Su TL, Yeh SH, Chen DS, Chen CT, Chen PJ, Jou YS. Diverse cellular transformation capability of overexpressed genes in human hepatocellular carcinoma. Biochem Biophys Res Commun. 2004;315:950-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 125] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 15. | Kang MK, Kang SK. Pharmacologic blockade of chloride channel synergistically enhances apoptosis of chemotherapeutic drug-resistant cancer stem cells. Biochem Biophys Res Commun. 2008;373:539-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Averaimo S, Milton RH, Duchen MR, Mazzanti M. Chloride intracellular channel 1 (CLIC1): Sensor and effector during oxidative stress. FEBS Lett. 2010;584:2076-2084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 90] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Kim JS, Chang JW, Yun HS, Yang KM, Hong EH, Kim DH, Um HD, Lee KH, Lee SJ, Hwang SG. Chloride intracellular channel 1 identified using proteomic analysis plays an important role in the radiosensitivity of HEp-2 cells via reactive oxygen species production. Proteomics. 2010;10:2589-2604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 18. | Wang P, Zhang C, Yu P, Tang B, Liu T, Cui H, Xu J. Regulation of colon cancer cell migration and invasion by CLIC1-mediated RVD. Mol Cell Biochem. 2012;365:313-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | Alexandrova AY, Kopnin PB, Vasiliev JM, Kopnin BP. ROS up-regulation mediates Ras-induced changes of cell morphology and motility. Exp Cell Res. 2006;312:2066-2073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 20. | Kim KH, Cho YS, Park JM, Yoon SO, Kim KW, Chung AS. Pro-MMP-2 activation by the PPARgamma agonist, ciglitazone, induces cell invasion through the generation of ROS and the activation of ERK. FEBS Lett. 2007;581:3303-3310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 21. | Singh H. Two decades with dimorphic Chloride Intracellular Channels (CLICs). FEBS Lett. 2010;584:2112-2121. [PubMed] |
| 22. | Littler DR, Harrop SJ, Fairlie WD, Brown LJ, Pankhurst GJ, Pankhurst S, DeMaere MZ, Campbell TJ, Bauskin AR, Tonini R. The intracellular chloride ion channel protein CLIC1 undergoes a redox-controlled structural transition. J Biol Chem. 2004;279:9298-9305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 181] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 23. | Singh H, Ashley RH. Redox regulation of CLIC1 by cysteine residues associated with the putative channel pore. Biophys J. 2006;90:1628-1638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 24. | Björklund M, Koivunen E. Gelatinase-mediated migration and invasion of cancer cells. Biochim Biophys Acta. 2005;1755:37-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 267] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 25. | Kang JC, Chen JS, Lee CH, Chang JJ, Shieh YS. Intratumoral macrophage counts correlate with tumor progression in colorectal cancer. J Surg Oncol. 2010;102:242-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 117] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 26. | Ushio-Fukai M, Nakamura Y. Reactive oxygen species and angiogenesis: NADPH oxidase as target for cancer therapy. Cancer Lett. 2008;266:37-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 513] [Cited by in RCA: 468] [Article Influence: 27.5] [Reference Citation Analysis (0)] |