Published online Nov 14, 2014. doi: 10.3748/wjg.v20.i42.15616
Revised: April 7, 2001
Accepted: June 26, 2014
Published online: November 14, 2014
Processing time: 156 Days and 19.6 Hours
Endometriosis is a gynecological condition that presents as endometrial-like tissue outside the uterus and induces a chronic inflammatory reaction. Up to 15% of women in their reproductive period are affected by this condition. Deep endometriosis is defined as endometriosis located more than 5 mm beneath the peritoneal surface. This type of endometriosis is mostly found on the uterosacral ligaments, inside the rectovaginal septum or vagina, in the rectosigmoid area, ovarian fossa, pelvic peritoneum, ureters, and bladder, causing a distortion of the pelvic anatomy. The frequency of bowel endometriosis is unknown, but in cases of bowel infiltration, about 90% are localized on the sigmoid colon or the rectum. Colorectal involvement results in alterations of bowel habits such as constipation, diarrhea, tenesmus, dyschezia, and, rarely, rectal bleeding. Differential diagnosis must be made in case of irritable bowel syndrome, solitary rectal ulcer syndrome, and a rectal tumor. A precise diagnosis about the presence, location, and extent of endometriosis is necessary to plan surgical treatment. Multidisciplinary laparoscopic treatment has become the standard of care. Depending on the size of the lesion and site of involvement, full-thickness disc excision or bowel resection needs to be performed by an experienced colorectal surgeon. Long-term outcomes, following bowel resection for severe endometriosis, regarding pain and recurrence rate are good with a pregnancy rate of 50%.
Core tip: Multidisciplinary laparoscopic treatment of extensive endometriosis with bowel involvement has become the standard of care. Depending on the size of the bowel lesion and site of bowel involvement, full-thickness disc excision or bowel resection needs to be performed by an experienced colorectal surgeon. This narrative review discusses in depth the role of a colorectal surgeon in a multidisciplinary team treating bowel endometriosis. From clinical and diagnostic work-up to surgical treatment for different colorectal localizations of endometriosis, each issue is addressed. Furthermore, postoperative outcomes, regarding symptom control, recurrence, and fertility, are discussed.
- Citation: Wolthuis AM, Meuleman C, Tomassetti C, D’Hooghe T, de Buck van Overstraeten A, D’Hoore A. Bowel endometriosis: Colorectal surgeon’s perspective in a multidisciplinary surgical team. World J Gastroenterol 2014; 20(42): 15616-15623
- URL: https://www.wjgnet.com/1007-9327/full/v20/i42/15616.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i42.15616
Endometriosis is a common health disorder in women, which is defined as the presence of endometrial-like tissue outside the uterus, inducing a chronic inflammatory reaction. The three clinical presentations are peritoneal endometriosis, endometriotic ovarian cysts (endometriomas), and deeply infiltrative endometriosis or deep endometriosis (DE)[1]. DE is defined as endometriosis located more than 5 mm beneath the peritoneal surface. It is mostly found on the uterosacral ligaments, inside the rectovaginal septum or vagina, in the rectosigmoid area, ovarian fossa, pelvic peritoneum, ureters, and bladder. DE can cause a complete distortion of the pelvic anatomy[2]. This review specifically focuses on the role of a colorectal surgeon in the treatment of DE in a multidisciplinary setting. Therefore, the aim of this paper is to underline the point of view of a colorectal surgeon in the multidisciplinary therapy of bowel endometriosis.
Endometriosis occurs during the active reproductive period. It is estimated that up to 15% of all women of reproductive age have endometriosis. About one-third of infertile women suffer from endometriosis[3]. Endometriosis affects the bowel in 3%-37% of all cases[4], and in 90% of these cases the rectum or sigmoid colon are also involved[5,6]. The histopathological diagnosis of endometriosis is usually straightforward. The term “bowel endometriosis” should be used when endometrial-like glands and stroma infiltrate the bowel wall reaching at least the subserous fat tissue or adjacent subserous plexus[7]. In general, the most common presenting clinical features are pelvic pain and infertility. The natural history of the disease has never been well defined due to the asymptomatic nature of the disease in many women. Even women with bowel endometriosis may be asymptomatic. For these patients, the clinical course appears to be benign, and surgical resection is probably not indicated, especially when there is no associated infertility. In women with moderate to severe disease, some degree of intestinal symptoms may be present. Intestinal symptoms due to endometriosis may vary depending on location and menstrual cycle[8]. Symptoms of endometriosis can be non-specific with considerable overlap with other clinical conditions, delaying a diagnosis and treatment. Moreover, physical examination, especially vaginal examination, may be completely normal, which hampers the diagnosis in young females. Chronic pelvic pain, often more severe during menstruation or at ovulation, is the most common symptom associated with endometriosis. Rectal involvement may result in alterations in bowel habits such as constipation, diarrhea, dyschezia, tenesmus, and, rarely, rectal bleeding. Differential diagnosis must be made with irritable bowel syndrome, solitary rectal ulcer syndrome and a rectal tumor[9]. Although colonic endometriosis may be asymptomatic, it can also present as large bowel obstruction. Colonic endometriosis must be differentiated from Crohn’s disease, diverticular disease, adhesions, or neoplasm. Also for small bowel implants secondary to endometriosis, difficulty exists to differentiate this condition from Crohn’s disease, because a similar endoscopic and histologic image can be seen. Intestinal perforation due to endometriosis may occur in the colon[10] and also in an appendix with transmural endometriosis.
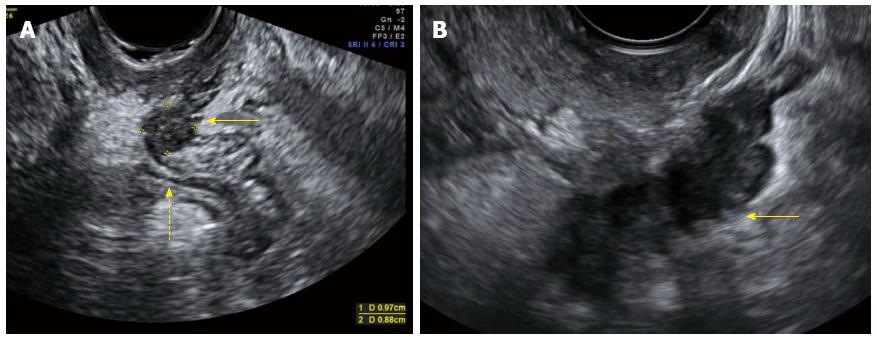
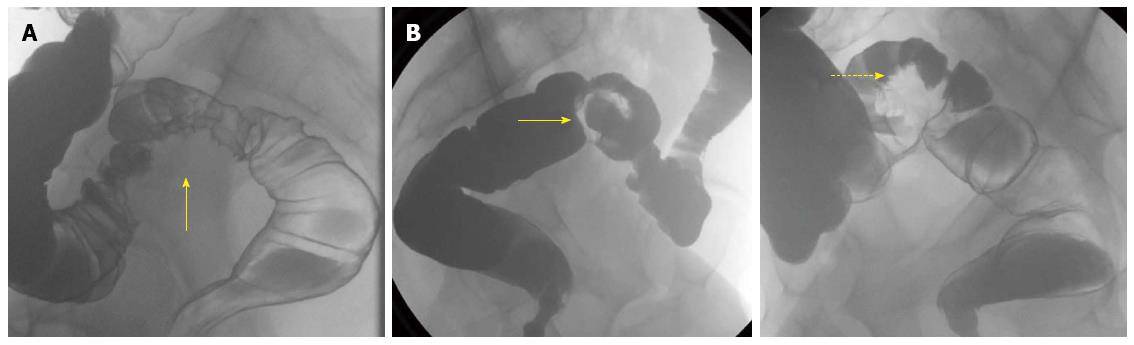
Preoperative work-up is essential in planning a multidisciplinary surgical treatment and is usually done by the gynecologist. Precise diagnosis is necessary with regard to location and extent of bowel endometriosis. For the evaluation of bowel endometriosis, with or without involvement of the rectovaginal septum, transvaginal ultrasonography, barium enema examination, and magnetic resonance imaging (MRI) are the imaging techniques of choice. These technical investigations should aim to: (1) document the extent of the disease; (2) help in planning a multidisciplinary treatment; and (3) counsel patients regarding postoperative complications. Transvaginal ultrasonography is a non-invasive tool, available at the gynecological outpatient clinic (Figure 1). DE can be detected as a heterogeneous, hypo-echoic, and sometimes spiculated mass[11]. According to a recent meta-analysis, bowel endometriosis can be diagnosed by transvaginal ultrasound with pooled estimates of sensitivity and specificity of 91% and 98%, respectively[12]. If bowel endometriosis is suspected, a barium enema examination is performed to investigate the extent of the disease (Figure 2). Deep invasion of the bowel wall appears as an extrinsic mass compressing the bowel lumen in association with fine crenulation of the mucosa in this particular region. Also, bowel strictures at the rectosigmoid junction can be seen. Based on this diagnostic potential, barium enema can guide the colorectal surgeon regarding the level of dissection, although the exact distance to the anal sphincter cannot be measured[13]. MRI can be helpful in diagnosis of multifocal endometriosis as well as in defining the condition’s anatomical location. A contrast enhanced mass or hyperintense foci on T1-weighted or fat-suppression T1-weighted MRIs strongly suspect the presence of hemorrhagic foci or hyperintense cavities secondary to endometriosis. A hypointense nodule can be seen on T2-weighted images with the signal of the tissue mass close to that of pelvic muscles. The sensitivity and specificity of MRI, to detect pelvic endometriosis, is around 90%[14,15]. Rectosigmoidoscopy or colonoscopy are of little value in the diagnosis of bowel endometriosis, because endometriosis is an extrinsic, typically a non-transmural disease.
The abovementioned imaging techniques are required to adequately assess the extent of endometriosis. However, exploratory laparoscopy is the gold standard to diagnose and to uniformly classify endometriosis[4]. Exploratory laparoscopy allows for examination of the uterus along with the uterosacral ligaments, ovaries and ureters, the sigmoid colon, and the upper rectum. The accuracy of exploratory laparoscopy is investigator-dependent and should be performed by an experienced gynecologist in cooperation with a colorectal surgeon. However, the pouch of Douglas cannot always be exposed, and rectovaginal endometriosis can be difficult to diagnose because of its intraperitoneal location[16]. The goal of endometriosis surgery is to obtain good long-term outcomes with regard to pain relief, recurrence rates, and fertility. Therefore, complete excision of all endometrial implants without compromising ovarian function is mandatory. Prevention of postoperative adhesion formation and improvement of quality of life are secondary aims. The association between different types of endometriotic lesions, such as peritoneal endometriosis, endometriomas, or DE, should not influence surgical treatment, as radical removal of endometriosis resulting in functioning pelvic organs is of paramount importance. Multidisciplinary laparoscopic treatment has become the standard of care for treatment of bowel endometriosis in optimization of patients’ outcomes during their reproductive years[17,18]. Non-randomized studies have shown that laparotomy and laparoscopy are equally effective in the treatment of endometriosis-associated pain[19]. Because laparoscopy is usually associated with a better postoperative recovery, shorter hospital stay, and better cosmesis, it is preferred to open surgery. If the relevant experience with laparoscopy is not available, or when DE is suspected or diagnosed, it is recommended to refer the patient to an expert center that offers minimally invasive treatment in a multidisciplinary context[20]. There is a general consensus that symptomatic endometriosis, especially DE, is best treated by a single laparoscopic operation in order to restore pelvic anatomy and to improve pain, quality of life, and fertility[21,22]. Experienced surgical judgment by both gynecological and colorectal surgeons is essential to determine whether a bowel resection is required. In our center, a three-step surgical procedure was performed, and summarized below[23]. In the first step, after placing temporary ureteric stents by the urologist, the gynecological surgeon excises all visible endometriosis laparoscopically with a CO2 laser to restore normal pelvic anatomy. In the second step, the urologist is consulted to evaluate and/or restore integrity of the ureters and the bladder. Finally, the colorectal surgeon evaluates the bowel. The decision to perform a laparoscopic segmental colectomy is taken under the following conditions: (1) large direct full-thickness trauma too extensive to be sutured; (2) extensive lesion to the bowel wall musculature in the absence of full-thickness damage but with impact on functionality; and (3) extensive lateral dissection compromising the bowel wall vascularization and/or innervation. In literature, controversy exists with regard to bowel resection. In our opinion, a patient-tailored approach is of utmost importance regarding the treatment of bowel endometriosis with emphasis on the patient’s symptoms, radical removal of endometriosis, and bowel function. It is difficult to compare published data on outcome of the treatment of bowel endometriosis, because surgical techniques are not standardized. Moreover, bias with regard to selection of patients and surgical procedures is often present in observational studies reporting on the outcome of endometriosis surgery[24]. Large prospective studies with standardized reporting of data will be necessary to evaluate surgical outcome in different centers[24-26].
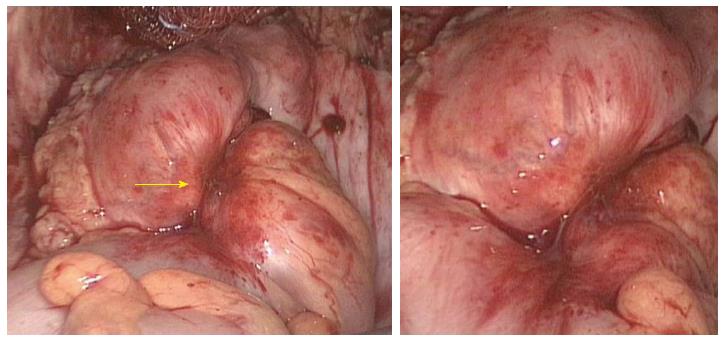
Rectovaginal endometriosis extends from the top of the posterior vaginal wall to the anterior rectal wall and to the uterosacral ligaments laterally. Laparoscopic resection is the treatment of choice for DE involving the rectovaginal septum, and several authors have reported good clinical outcomes with improvement in dysmenorrhea, dyspareunia, and chronic pelvic pain of 60%-92%, 70%-100%, and 60%-93%, respectively[23,24,26-28]. However, to date, the debate with regard to different surgical techniques and the necessity of either a full-thickness disc rectal excision or a restorative resection is still ongoing[24,25]. If there is no rectal wall invasion, laparoscopic resection can be done with few complications and good outcome[29]. In our opinion, it is necessary to prepare the rectum with mechanical bowel preparation in case of rectal wall invasion. Subsequently, probes will be placed into both the uterus and the rectum to improve exposure. Pararectal spaces are opened and a circular incision around the nodule is made. It is dissected free from the uterosacral ligaments safeguarding the ureters. Then, a plane between the rectum and the lesion can be found using CO2 laser dissection. Only after completion of the resection from the rectal wall, the nodule is dissected from the posterior vaginal wall. In case of vaginal wall invasion, the posterior fornix is excised and closely sutured. Superficial rectal lesions can be excised from the bowel using the “shaving” technique, but the integrity of the wall should be carefully assessed and checked with an air leak test[30,31]. An experienced colorectal surgeon should judge on how to best restore rectal wall integrity by weighing two different surgical options, full-thickness disc excision or partial rectal resection with reanastomosis. Moreover, in our center, radical excision of all endometriotic implants with clear margins is equally important in surgical decision-making. The decision to perform full-thickness disc excision or restorative bowel resection is mainly based on precise judgment by an experienced colorectal surgeon with expertise in radical endometriosis surgery. Because there are no guidelines whether or not to perform a resection, the decision is made on a case-by-case basis. Invasion of more than 50% of the bowel circumference, multiple nodules, or nodules larger than 3 cm are indications for a bowel resection[32]. Thus, full-thickness disc excision is limited to the size of the lesion, depth of infiltration, and the location. The procedure fails to be microscopically complete in 40% of women due to active glandular endometrial foci deeply infiltrating the bowel wall[32,33]. For a small nodule, full-thickness disc excision of the anterior rectal wall can be performed safely[32-36]. However, the rectal wall should be well vascularized and innervated allowing a single layer transverse closure with separate sutures. If a restorative rectal resection is performed, care should be taken to preserve optimal bowel function. A side-to-end anastomosis or a colonic J pouch anastomosis is often necessary when part of the rectal ampulla is excised. In our center, a defunctioning loop ileostomy is performed only when there are sutures on the vagina, the rectum, and the ureter/bladder. An omentoplasty to protect the pelvic sutures is not performed routinely. Complications, such as anastomotic leakage or rectovaginal fistula, after bowel resection and anastomosis can occur[37]. Different studies reported anastomotic related complications, such as leakage, pelvic abscess, and rectovaginal fistula. The incidence varied 1%-6%, at a mean rate of 4.7%[38]. In papers reporting on bowel resection for endometriosis, the incidences of anastomotic leakages, abscesses, and rectovaginal fistulae were 1.5%, 0.3%, and 2.7%, respectively[24]. In papers reporting on both bowel resection and full-thickness disc excision, the incidences for abovementioned complications were 0.7%, 0.3%, and 0.7%, respectively[24]. Therefore, bowel resection leads to good operative outcomes with a low and acceptable major complication rate.
In case of bowel endometriosis infiltrating the sigmoid colon, a laparoscopic segmental colectomy may be required in order to obtain complete macroscopic clearance resulting in a good outcome regarding pain, quality of life, and infertility[6,39-41]. In 1991, the first case of laparoscopic colectomy for bowel endometriosis was described[42]. Since then, several groups have reported good results[23,37,43-46]. However, a selective approach towards bowel resection is justified, because endometriosis is a benign condition and operative morbidity should be kept low. On the other hand, for sigmoidal endometriosis, a sigmoid resection or so-called high anterior resection is suggested considering it is technically impossible to perform a discoid excision on the sigmoid colon (Figure 3). A superior rectal artery sparing approach is performed with preservation of autonomic nerves, which has been shown to improve sexual and urological functioning[47]. When neoplasia is a concern, an oncological segmental colectomy with lymphadenectomy should be performed. A randomized clinical trial comparing laparoscopically-assisted and open colorectal resection for endometriosis showed benefits on pain and postoperative complications in the laparoscopic group. Moreover, a laparoscopic approach offered a higher spontaneous pregnancy rate and similar improvement in quality of life compared to open surgery[48]. Therefore, in a tailored approach of segmental colectomy for severe symptomatic endometriosis, access trauma to the abdomen should be minimal to allow fast recovery. We described a technique to extract the specimen transanally, so that a laparotomy can be avoided. This natural orifice specimen extraction colectomy (NOSE-colectomy) has good outcomes in patients requiring a colorectal resection for bowel endometriosis[49]. Recently, a prospective cohort study comparing patients with and without bowel resection after CO2 laser laparoscopic excision, observed good clinical outcomes and low major morbidity; anastomotic leakage was only 1%[23].
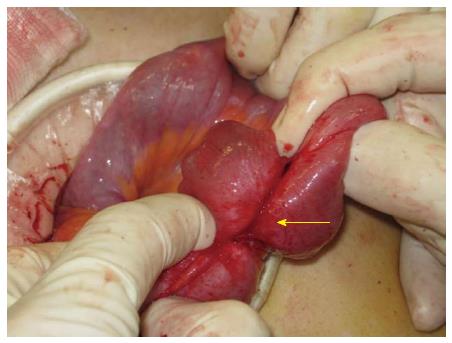
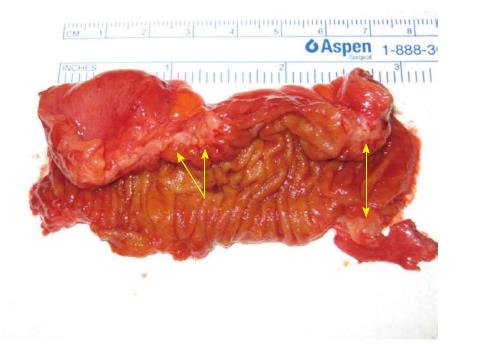
Small bowel or appendiceal involvement is rare and does not always cause symptoms. Usually, non-specific symptoms, such as recurrent pain and abdominal bloating, can accompany this type of endometriosis. However, acute small bowel obstruction due to fibrotic adhesions secondary to endometriosis has been described[50]. Endometriosis can cause acute appendicitis with typical symptoms such as right iliac fossa pain, fever, nausea, and elevated white blood cell count. Appendiceal endometriosis can also act as a lead point leading to ileocecal invagination. Moreover, it can cause obstruction of the appendicular orifice with secondary mucocele formation. Appendiceal endometriosis is treated by appendectomy. The treatment of small bowel endometriosis depends on the size and location of the lesion. Small implants may be treated by wedge excision and sutured closure. If more than 50% of the bowel wall is affected or if the lesion is close to the ileocecal valve, a segmental small bowel resection or ileocecal resection may sometimes be performed (Figures 4 and 5)[51].
Long-term outcome of radical endometriosis surgery evaluates three important parameters: (1) symptom control; (2) recurrence rate; and (3) pregnancy. Due to heterogeneous data reports on pain outcomes, it is difficult to compare results. Differences among reported results are mostly due to considerate variations in pain measurement, method of evaluation, and length of follow-up. Overall, most of the studies show an improvement in pain, gynecological, and intestinal symptoms after bowel resection[24]. Moreover, a recent study showed that radical but fertility sparing surgery, with or without bowel resection, has comparable and good psychological outcomes concerning depression levels, relationship satisfaction, and sexual function[47]. Endometriosis recurrence rate, after bowel resection, has been reported in 4.7%-25% of cases after a follow-up period of more than two years[24]. Meuleman and colleagues reported on three different cohorts of patients and showed a recurrence rate between 3% and 7% after CO2 laser ablation and bowel resection. In a multidisciplinary setting, aiming at radical excision of endometriosis, recurrence rates can be low and results can be consistent over time[23,41,52]. Observed post-operative pregnancy rates after bowel resection vary between 24% and 57%[1,5,17,28,36,41,46,53,54]. A recent prospective cohort study showed similar pregnancy rates comparing a group of patients who had bowel resection to a group of patients without bowel resection, 50% vs 51%, respectively[23]. The cumulative pregnancy rate was 44%, 58%, and 73% after 1, 2, and 3 years, respectively. This is in line with other retrospective studies reporting a cumulative pregnancy rate of 49% (at two years) and 52% (at 29 mo)[41]. More recently, the endometriosis fertility index staging system was validated to predict non-assisted reproductive technology pregnancy after endometriosis surgery. This scoring system is the best available staging tool to counsel patients after a radical surgery about their fertility prognosis and eventual need for fertility treatment[55,56]. Postoperative medical treatment is beyond the scope of this article; nevertheless, important considerations regarding active child wish and secondary prevention should be taken into account, during follow-up of patients who underwent a radical multidisciplinary endometriosis surgery. Although guidelines regarding the role of postoperative hormonal therapy exist, there is insufficient evidence showing significant benefit of hormonal therapy accompanying endometriosis surgery on the outcomes, specifically pain management[20,57]. However, postoperative hormonal therapy could be prescribed for indications, such as contraception or secondary prevention[20,23].
There is still a discussion about the optimal treatment of endometriosis involving the gastrointestinal tract. A standardized preoperative work-up for bowel endometriosis diagnosis is necessary. A multidisciplinary laparoscopic treatment, in an expert center, should be planned in advance to ensure adequate patient counseling. Laparoscopy is the preferred approach, as it is associated with less postoperative pain and faster recovery. Furthermore, fertility outcome is improved with a laparoscopic approach when bowel resection is required. An experienced colorectal surgeon should make a precise judgment on whether or not a full-thickness disc excision or bowel resection is indicated. In our opinion, a patient-tailored approach is crucial and the least invasive radical option should be chosen. In the future, research should focus on long-term outcome improvements of the multidisciplinary treatment for bowel endometriosis with regard to symptoms, quality of life, cosmetic outcome, recurrence of endometriosis, and fertility.
P- Reviewer: Lee WK, Souza CA S- Editor: Wen LL L- Editor: A E- Editor: Ma S
| 1. | Donnez J, Nisolle M, Casanas-Roux F, Bassil S, Anaf V. Rectovaginal septum, endometriosis or adenomyosis: laparoscopic management in a series of 231 patients. Hum Reprod. 1995;10:630-635. [PubMed] |
| 2. | Jenkins S, Olive DL, Haney AF. Endometriosis: pathogenetic implications of the anatomic distribution. Obstet Gynecol. 1986;67:335-338. [PubMed] |
| 3. | D’Hooghe TM, Debrock S, Hill JA, Meuleman C. Endometriosis and subfertility: is the relationship resolved? Semin Reprod Med. 2003;21:243-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 203] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 4. | Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67:817-821. [PubMed] |
| 5. | Bailey HR, Ott MT, Hartendorp P. Aggressive surgical management for advanced colorectal endometriosis. Dis Colon Rectum. 1994;37:747-753. [PubMed] |
| 6. | Campagnacci R, Perretta S, Guerrieri M, Paganini AM, De Sanctis A, Ciavattini A, Lezoche E. Laparoscopic colorectal resection for endometriosis. Surg Endosc. 2005;19:662-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 7. | Chapron C, Fauconnier A, Vieira M, Barakat H, Dousset B, Pansini V, Vacher-Lavenu MC, Dubuisson JB. Anatomical distribution of deeply infiltrating endometriosis: surgical implications and proposition for a classification. Hum Reprod. 2003;18:157-161. [PubMed] |
| 8. | Kaufman LC, Smyrk TC, Levy MJ, Enders FT, Oxentenko AS. Symptomatic intestinal endometriosis requiring surgical resection: clinical presentation and preoperative diagnosis. Am J Gastroenterol. 2011;106:1325-1332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Seaman HE, Ballard KD, Wright JT, de Vries CS. Endometriosis and its coexistence with irritable bowel syndrome and pelvic inflammatory disease: findings from a national case-control study--Part 2. BJOG. 2008;115:1392-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 85] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 10. | Floberg J, Bäckdahl M, Silferswärd C, Thomassen PA. Postpartum perforation of the colon due to endometriosis. Acta Obstet Gynecol Scand. 1984;63:183-184. [PubMed] |
| 11. | Roseau G, Dumontier I, Palazzo L, Chapron C, Dousset B, Chaussade S, Dubuisson JB, Couturier D. Rectosigmoid endometriosis: endoscopic ultrasound features and clinical implications. Endoscopy. 2000;32:525-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 12. | Hudelist G, English J, Thomas AE, Tinelli A, Singer CF, Keckstein J. Diagnostic accuracy of transvaginal ultrasound for non-invasive diagnosis of bowel endometriosis: systematic review and meta-analysis. Ultrasound Obstet Gynecol. 2011;37:257-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 188] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 13. | Anaf V, El Nakadi I, De Moor V, Coppens E, Zalcman M, Noel JC. Anatomic significance of a positive barium enema in deep infiltrating endometriosis of the large bowel. World J Surg. 2009;33:822-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 14. | Bazot M, Darai E, Hourani R, Thomassin I, Cortez A, Uzan S, Buy JN. Deep pelvic endometriosis: MR imaging for diagnosis and prediction of extension of disease. Radiology. 2004;232:379-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 417] [Cited by in RCA: 378] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 15. | Grasso RF, Di Giacomo V, Sedati P, Sizzi O, Florio G, Faiella E, Rossetti A, Del Vescovo R, Zobel BB. Diagnosis of deep infiltrating endometriosis: accuracy of magnetic resonance imaging and transvaginal 3D ultrasonography. Abdom Imaging. 2010;35:716-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 16. | Del Frate C, Girometti R, Pittino M, Del Frate G, Bazzocchi M, Zuiani C. Deep retroperitoneal pelvic endometriosis: MR imaging appearance with laparoscopic correlation. Radiographics. 2006;26:1705-1718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 111] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 17. | Keckstein J, Wiesinger H. Deep endometriosis, including intestinal involvement--the interdisciplinary approach. Minim Invasive Ther Allied Technol. 2005;14:160-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 68] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Kennedy S, Bergqvist A, Chapron C, D’Hooghe T, Dunselman G, Greb R, Hummelshoj L, Prentice A, Saridogan E. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum Reprod. 2005;20:2698-2704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 992] [Cited by in RCA: 1003] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 19. | Crosignani PG, Vercellini P, Biffignandi F, Costantini W, Cortesi I, Imparato E. Laparoscopy versus laparotomy in conservative surgical treatment for severe endometriosis. Fertil Steril. 1996;66:706-711. [PubMed] |
| 20. | Dunselman GA, Vermeulen N, Becker C, Calhaz-Jorge C, D’Hooghe T, De Bie B, Heikinheimo O, Horne AW, Kiesel L, Nap A. ESHRE guideline: management of women with endometriosis. Hum Reprod. 2014;29:400-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1516] [Cited by in RCA: 1349] [Article Influence: 122.6] [Reference Citation Analysis (0)] |
| 21. | Garry R. The effectiveness of laparoscopic excision of endometriosis. Curr Opin Obstet Gynecol. 2004;16:299-303. [PubMed] |
| 22. | Jacobson TZ, Duffy JM, Barlow D, Koninckx PR, Garry R. Laparoscopic surgery for pelvic pain associated with endometriosis. Cochrane Database Syst Rev. 2009;CD001300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 23. | Meuleman C, Tomassetti C, Wolthuis A, Van Cleynenbreugel B, Laenen A, Penninckx F, Vergote I, D’Hoore A, D’Hooghe T. Clinical outcome after radical excision of moderate-severe endometriosis with or without bowel resection and reanastomosis: a prospective cohort study. Ann Surg. 2014;259:522-531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 102] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 24. | Meuleman C, Tomassetti C, D’Hoore A, Van Cleynenbreugel B, Penninckx F, Vergote I, D’Hooghe T. Surgical treatment of deeply infiltrating endometriosis with colorectal involvement. Hum Reprod Update. 2011;17:311-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 248] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 25. | Meuleman C, D’Hoore A, Van Cleynenbreugel B, Tomassetti C, D’Hooghe T. Why we need international agreement on terms and definitions to assess clinical outcome after endometriosis surgery. Hum Reprod. 2011;26:1598-159; author reply 1598-159;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Meuleman C, Tomassetti C, D’Hooghe TM. Clinical outcome after laparoscopic radical excision of endometriosis and laparoscopic segmental bowel resection. Curr Opin Obstet Gynecol. 2012;24:245-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 27. | Angioni S, Peiretti M, Zirone M, Palomba M, Mais V, Gomel V, Melis GB. Laparoscopic excision of posterior vaginal fornix in the treatment of patients with deep endometriosis without rectum involvement: surgical treatment and long-term follow-up. Hum Reprod. 2006;21:1629-1634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 28. | Redwine DB, Wright JT. Laparoscopic treatment of complete obliteration of the cul-de-sac associated with endometriosis: long-term follow-up of en bloc resection. Fertil Steril. 2001;76:358-365. [PubMed] |
| 29. | Chapron C, Jacob S, Dubuisson JB, Vieira M, Liaras E, Fauconnier A. Laparoscopically assisted vaginal management of deep endometriosis infiltrating the rectovaginal septum. Acta Obstet Gynecol Scand. 2001;80:349-354. [PubMed] |
| 30. | Donnez J, Squifflet J. Complications, pregnancy and recurrence in a prospective series of 500 patients operated on by the shaving technique for deep rectovaginal endometriotic nodules. Hum Reprod. 2010;25:1949-1958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 187] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 31. | Healey M, Ang WC, Cheng C. Surgical treatment of endometriosis: a prospective randomized double-blinded trial comparing excision and ablation. Fertil Steril. 2010;94:2536-2540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 81] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 32. | Remorgida V, Ragni N, Ferrero S, Anserini P, Torelli P, Fulcheri E. The involvement of the interstitial Cajal cells and the enteric nervous system in bowel endometriosis. Hum Reprod. 2005;20:264-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 62] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 33. | Roman H, Opris I, Resch B, Tuech JJ, Sabourin JC, Marpeau L. Histopathologic features of endometriotic rectal nodules and the implications for management by rectal nodule excision. Fertil Steril. 2009;92:1250-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 34. | Abrão MS, Podgaec S, Dias JA, Averbach M, Silva LF, Marino de Carvalho F. Endometriosis lesions that compromise the rectum deeper than the inner muscularis layer have more than 40% of the circumference of the rectum affected by the disease. J Minim Invasive Gynecol. 2008;15:280-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 35. | Anaf V, El Nakadi I, Simon P, Van de Stadt J, Fayt I, Simonart T, Noel JC. Preferential infiltration of large bowel endometriosis along the nerves of the colon. Hum Reprod. 2004;19:996-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 99] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 36. | Kavallaris A, Köhler C, Kühne-Heid R, Schneider A. Histopathological extent of rectal invasion by rectovaginal endometriosis. Hum Reprod. 2003;18:1323-1327. [PubMed] |
| 37. | Daraï E, Bazot M, Rouzier R, Houry S, Dubernard G. Outcome of laparoscopic colorectal resection for endometriosis. Curr Opin Obstet Gynecol. 2007;19:308-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 38. | De Cicco C, Corona R, Schonman R, Mailova K, Ussia A, Koninckx P. Bowel resection for deep endometriosis: a systematic review. BJOG. 2011;118:285-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 201] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 39. | Dubernard G, Rouzier R, David-Montefiore E, Bazot M, Darai E. Use of the SF-36 questionnaire to predict quality-of-life improvement after laparoscopic colorectal resection for endometriosis. Hum Reprod. 2008;23:846-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 40. | Kössi J, Setälä M, Mäkinen J, Härkki P, Luostarinen M. Quality of life and sexual function 1 year after laparoscopic rectosigmoid resection for endometriosis. Colorectal Dis. 2013;15:102-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 41. | Meuleman C, D’Hoore A, Van Cleynenbreugel B, Beks N, D’Hooghe T. Outcome after multidisciplinary CO2 laser laparoscopic excision of deep infiltrating colorectal endometriosis. Reprod Biomed Online. 2009;18:282-289. [PubMed] |
| 42. | Redwine DB, Sharpe DR. Laparoscopic segmental resection of the sigmoid colon for endometriosis. J Laparoendosc Surg. 1991;1:217-220. [PubMed] |
| 43. | Duepree HJ, Senagore AJ, Delaney CP, Marcello PW, Brady KM, Falcone T. Laparoscopic resection of deep pelvic endometriosis with rectosigmoid involvement. J Am Coll Surg. 2002;195:754-758. [PubMed] |
| 44. | Mereu L, Ruffo G, Landi S, Barbieri F, Zaccoletti R, Fiaccavento A, Stepniewska A, Pontrelli G, Minelli L. Laparoscopic treatment of deep endometriosis with segmental colorectal resection: short-term morbidity. J Minim Invasive Gynecol. 2007;14:463-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 80] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 45. | Ruffo G, Scopelliti F, Scioscia M, Ceccaroni M, Mainardi P, Minelli L. Laparoscopic colorectal resection for deep infiltrating endometriosis: analysis of 436 cases. Surg Endosc. 2010;24:63-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 120] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 46. | Stepniewska A, Pomini P, Bruni F, Mereu L, Ruffo G, Ceccaroni M, Scioscia M, Guerriero M, Minelli L. Laparoscopic treatment of bowel endometriosis in infertile women. Hum Reprod. 2009;24:1619-1625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 97] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 47. | Van den Broeck U, Meuleman C, Tomassetti C, D’Hoore A, Wolthuis A, Van Cleynenbreugel B, Vergote I, Enzlin P, D’Hooghe T. Effect of laparoscopic surgery for moderate and severe endometriosis on depression, relationship satisfaction and sexual functioning: comparison of patients with and without bowel resection. Hum Reprod. 2013;28:2389-2397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 48. | Daraï E, Dubernard G, Coutant C, Frey C, Rouzier R, Ballester M. Randomized trial of laparoscopically assisted versus open colorectal resection for endometriosis: morbidity, symptoms, quality of life, and fertility. Ann Surg. 2010;251:1018-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 180] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 49. | Wolthuis AM, Meuleman C, Tomassetti C, D’Hooghe T, Fieuws S, Penninckx F, D’Hoore A. Laparoscopic sigmoid resection with transrectal specimen extraction: a novel technique for the treatment of bowel endometriosis. Hum Reprod. 2011;26:1348-1355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 50. | De Ceglie A, Bilardi C, Blanchi S, Picasso M, Di Muzio M, Trimarchi A, Conio M. Acute small bowel obstruction caused by endometriosis: a case report and review of the literature. World J Gastroenterol. 2008;14:3430-3434. [PubMed] |
| 51. | Ruffo G, Stepniewska A, Crippa S, Serboli G, Zardini C, Steinkasserer M, Ceccaroni M, Minelli L, Falconi M. Laparoscopic ileocecal resection for bowel endometriosis. Surg Endosc. 2011;25:1257-1262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 52. | Meuleman C, Tomassetti C, D’Hoore A, Buyens A, Van Cleynenbreugel B, Fieuws S, Penninckx F, Vergote I, D’Hooghe T. Clinical outcome after CO2 laser laparoscopic radical excision of endometriosis with colorectal wall invasion combined with laparoscopic segmental bowel resection and reanastomosis. Hum Reprod. 2011;26:2336-2343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 53. | Ghezzi F, Cromi A, Ciravolo G, Rampinelli F, Braga M, Boni L. A new laparoscopic-transvaginal technique for rectosigmoid resection in patients with endometriosis. Fertil Steril. 2008;90:1964-1968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 54. | Minelli L, Fanfani F, Fagotti A, Ruffo G, Ceccaroni M, Mereu L, Landi S, Pomini P, Scambia G. Laparoscopic colorectal resection for bowel endometriosis: feasibility, complications, and clinical outcome. Arch Surg. 2009;144:234-29; discussion 239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 120] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 55. | Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2010;94:1609-1615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 233] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 56. | Tomassetti C, Geysenbergh B, Meuleman C, Timmerman D, Fieuws S, D’Hooghe T. External validation of the endometriosis fertility index (EFI) staging system for predicting non-ART pregnancy after endometriosis surgery. Hum Reprod. 2013;28:1280-1288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 60] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 57. | Yap C, Furness S, Farquhar C. Pre and post operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev. 2004;CD003678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |