Published online Oct 21, 2014. doi: 10.3748/wjg.v20.i39.14308
Revised: March 12, 2014
Accepted: June 14, 2014
Published online: October 21, 2014
Processing time: 326 Days and 3.9 Hours
The introduction of laparoscopy in the surgeon’s armamentarium was in fact a “revolution in the history of surgery”. Since this technique involves insufflation of carbon dioxide it produces several pathophysiological changes which have to be understood by the anaesthesiologist who can modify the anaesthesia technique accordingly. Advantages of laparoscopy include reduced pain, small scars and early return to work. Certain complications specific to laparoscopic surgery are due to carboperitoneum and increased intra-abdominal pressure. Venous air embolism, although very rare, can be lethal if not managed promptly. Other complications include subcutaneous emphysema, haemodynamic compromise and arrhythmias. Although associated with minimal postoperative morbidity, postoperative pain, nausea and vomiting can be quite problematic. The limitations of laparoscopy have been overcome by the introduction of robotic surgery. There are important implications for the anaesthesiologist during robotic surgeries which have to be practiced accordingly. Robotic surgery has a learning curve for both the surgeon and the anaesthesiologist. The robot is bulky, and cannot be disengaged after docking. Therefore it is important that the anaesthetized patient remains immobile throughout surgery and anaesthesia is reversed only after the robot has been disengaged at the end of surgery. Advances in laparoscopy and robotic surgery have modified anaesthetic techniques too.
Core tip: Laparoscopy is the gold standard for cholecystectomy. The layman may perceive laparoscopy as “minimally invasive”, but the procedure has profound effects on the physiology of the patient. The surgeon and anaesthesiologist must be well versed with these pathophysiological changes so that the safety of the patient is not compromised. Robotic assisted laparoscopic surgeries are gaining popularity; here the role of the anaesthesiologist is paramount. Anaesthesiologists must fine-tune their anaesthetic techniques to meet the specific requirements of such operations.
- Citation: Sood J. Advancing frontiers in anaesthesiology with laparoscopy. World J Gastroenterol 2014; 20(39): 14308-14314
- URL: https://www.wjgnet.com/1007-9327/full/v20/i39/14308.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i39.14308
After the successful demonstration of anaesthesia using ether on 16th October, 1846 at the Massachusetts General Hospital in Boston, surgery began to progress. This day is celebrated as the “World Anaesthesia Day”. Gradually anaesthetic techniques and apparatus improved, allowing major surgical procedures with improved results.
Major advances in surgery were possible due to refinements in anaesthesia. One such major advancement was the introduction of laparoscopy in the surgical armamentarium, which can be called the laparoscopic “revolution.” The scars were minimal, there was less pain, early discharge from hospital and patient satisfaction was immense. The history of this laparoscopic “revolution” is very intriguing.
The creation of pneumoperitoneum was not a new idea. Insufflation of air into the abdominal cavity for diagnosis and treatment of abdominal tuberculosis was being practiced as early as 1882. Kelling strongly believed in this concept and he wanted not only to inject air into the abdomen, but also observe its effects by using an endoscope[1].
Jacobaeus described the first therapeutic application of laparoscopy in 1912. He was an energetic proponent of laparoscopy and did much to popularize laparothoracoscopy.
All these procedures were being performed under local anaesthesia. In the late 1920s, Albert Decker a surgeon in New York performed coelioscopy for about 10 years before giving it up, because it required general anaesthesia. He remarked that with good anaesthesia and an operating room he preferred to explore the abdomen and at the same time correct the condition properly. He preferred “culdoscopy” over laparoscopy, since it was successfully carried out under local anaesthesia[1].
Raoul Palmer, a French gynecologist who migrated to the United States, performed culdoscopy under local anaesthesia mainly because of technical reasons: the small rooms did not allow enough space for an anaesthesiologist and the anaesthetic equipment. Palmer observed that patients who had submitted to laparoscopic examination under local anaesthesia were unwilling to repeat the experience. It was only in 1952 that Palmer was finally allotted a permanent berth in the operation theatre where he performed laparoscopies under general anaesthesia[1]. In 1959 the first book about “Methods of Endoscopic Examination in Gynecology” was published by Hans Frangenheim.
A key breakthrough in endoscopic technique was the invention of the so called “cold light”.
After 1969, interest in laparoscopy boomed as patients rushed for the new technique. Cohen remarked “I am confident that with the availability of fine optical instruments and safe anaesthesia the technique is here to stay”[1].
Introduction of videoscopic technologies in the 1980s began the era of video guided surgery in which laparoscopic surgical techniques could be viewed on a monitor and could be used for more complicated procedures. This advance in technology proved to be the critical factor that allowed the widespread acceptance of laparoscopy in the surgical armamentarium. In 1987, Philippe Mouret performed the first complete removal of a diseased gall bladder in Lyon, France.
In its nascent stages, laparoscopy was used only for short diagnostic surgery in young patients with no comorbidities; but gradually as the popularity of this technique increased, older and high risk patients were also considered for it.
The present scenario is such that major surgeries including gastrectomy, radical prostatectomy and bariatric surgery are all being performed laparoscopically.
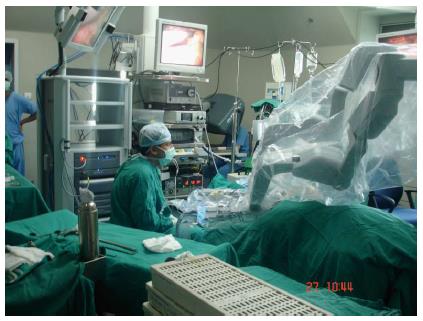
Even after several decades of its popularity the basic drawbacks of laparoscopic surgery still remain: though the monitors show enlarged images of the operating field, it takes time to get accustomed working in a three dimensional space while looking at a flat image; also, the long surgical instruments used and enlarged images obviously magnify natural hand tremors. In an effort to overcome these shortcomings remote instrumentation was envisaged[2]. The idea of combining telerobotic surgery with laparoscopic surgery eventually led to the development of the da Vinci and the Zeus Robotic Surgical Systems in 1999. Robotic surgery was a significant improvement on minimally invasive surgical procedures with several advantages (Figure 1)[3].
The advantages of extremely precise instrument manipulation, reduction in intraoperative blood loss, reduced analgesic requirements and length of stay, and advantages of ergonomics have made robotic assisted laparoscopic surgery a popular option.
In fact, with the spurt in the demand for minimally invasive surgery and further improvements of the existing robotic systems it is possible that in the future robotic assistance will be the norm for all surgical procedures.
New surgical procedures pose new anaesthetic challenges and demand consequent changes in anaesthesia techniques and practice. Robotic assisted laparoscopic surgery requires special anaesthetic considerations.
Although visually minimally invasive to the patient, laparoscopy results in several trespasses to normal homeostasis which are very important for the anaesthesiologist to understand, so that appropriate modifications are made to the anaesthetic technique for an uneventful surgical and anaesthesia outcome. These trespasses are patient positioning, insufflation of exogenous gas and increased intra-abdominal pressure.
Since retraction is limited in laparoscopic surgery, the patient is positioned several times during the surgical procedure using gravity to move viscera away from the operating site for good exposure of the lesion. Exaggerated positions are frequently employed in laparoscopic surgery, especially robotic surgery.
The Trendelenburg position adopted for operative gynaecological surgical procedures may favour haemodynamics but adversely affects the respiratory system. It may also result in an increase in the intracranial and intraocular pressure. The steep Trendelenburg position adopted for robotic assisted laparoscopic surgery (radical prostatectomy) predisposes to acid reflux leading to oral ulceration, conjunctival burns[4], and with prolonged head down position, there may be laryngeal edema and respiratory stridor following extubation of the trachea at the end of the operation[5]. A rare, but dangerous complication, “Well Leg Compartment Syndrome” may develop during prolonged surgery in a steep Trendelenburg position. This is due to a combination of impaired arterial perfusion in the raised lower limbs, compression of venous vessels by lower limb supports and reduced femoral venous drainage due to carboperitoneum[6].
The robotic system does not allow for changes in patient position on the operating table once the robot has been docked. Access to the patient’s airway is sometimes very difficult, since the OR table is rotated 90°-180° away from the anaesthesiologist, as in upper abdominal and thoracic surgeries[3].
Adequate pressure point padding is absolutely imperative since no movement of the patient is possible after docking, and it is difficult to access the patient intraoperatively. A steep Trendelenburg position along with carboperitoneum interferes with effective ventilation. The reverse Trendelenburg position, however, contributes to betterment of diaphragm function but it adversely affects the cardiovascular system, reducing venous return and cardiac output[7]. Gravity induced venous stasis in the lower limbs is another drawback with the steep head up position, and deep vein thrombosis, especially in lengthy procedures, is a real possibility. Deep vein thrombosis prophylaxis is necessary in the population at risk[2]. Thus, to prevent haemodynamic disturbances, it is important to gradually alter the position of the anaesthetized patients[8].
The insufflating gas used for pneumoperitoneum should be non-toxic, inert, colourless, non-inflammable, readily soluble in blood, easily ventilated through the lungs and inexpensive.
Air was the first gas to be used since it was cheap and easily available. Later oxygen was also used for a long time. However both these gases are inflammable and have a low Ostwald’s blood gas solubility coefficient (0.006, 0.013), thus having a potential for air embolism in case of an accidental entry into a blood vessel. In the 1970s, nitrous oxide emerged as the gas preferred by gynaecologists, however it supports combustion if mixed with methane (from the bowel). CO2 is the choice of insufflating gas used for carboperitoneum. It is relatively inert, non-flammable, readily absorbed if there is an accidental entry into a blood vessel (blood gas solubility coefficient 0.48), so it produces less severe air embolism and is easily expired by the lungs[9]. Alternative gases including helium, argon and xenon have been studied. They are inert, and provide a good optical vision, but have a very low blood gas solubility coefficient (0.00018) and so there are very high chances of air embolism if there is accidental entry into a blood vessel; however they are very diffusible. They are very expensive as well[10].
Hypercarbia can occur with carboperitoneum where the rising end tidal CO2 (EtCO2) is due to absorption of CO2 from the peritoneal cavity[11]. Normocarbia can be maintained by a 15%-20% increase in minute ventilation[12]. The minute ventilation increase is achieved by increasing the respiratory rate rather than increasing the tidal volume.
Usually there is a good correlation between the EtCO2 and the arterial carbon dioxide (PaCO2) but where the surgery is prolonged or a preoperative pulmonary pathology is present there is a decreased capacity of CO2 elimination, leading to serum acidosis and hypercarbia[13]. A direct measurement of arterial CO2 by ABG may be required in such instances[14].
Normally intraoperative hemodynamics are altered only when there is a 30% or more increase above the normal carbon dioxide levels. Even a mild hypercapnia results in sympathetic stimulation manifesting as tachycardia, increased cardiac output and systemic resistance[15]. These in turn could lead to coronary ischemia because of a short left ventricular ejection time and a short diastolic filling phase, and result in complications for patients suffering from ischemic heart disease. Reduced left ventricular function and negative inotropic action result from severe hypercapnia[16].
Earlier, CO2 absorption was observed to be greater during extraperitoneal rather than intraperitoneal insufflation[17]. However later studies showed that it was not so, and that absorption of CO2 was not greater in extraperitoneal surgery[18]. Slow mobilization of CO2 from body stores to the blood for removal from the body is responsible for the effects of hypercarbia persisting in the postoperative period. The shoulder pain seen in the postoperative period is thought to be due to residual CO2 forming carbonic acid in the intrapleural area[19].
The insufflation of several litres of CO2 results in an increase in intra-abdominal pressure (IAP). Several pathophysiological changes occur due to carboperitoneum and an increase in IAP which require adjustments in the anaesthesia technique.
The most important reason for hemodynamic instability during laparoscopic procedures is high IAP[20]. During insufflation the peritoneum is stretched causing vagal stimulation which could lead to bradycardia[21].
Gradual insufflation is therefore recommended. The threshold pressure that produces minimal changes in haemodynamics is 12 mmHg. Up to an IAP of 10 mmHg, the filling pressures are maintained and the cardiac output may increase. Beyond an IAP of 15 mmHg, there is a fall in the preload and an increase in the afterload and systemic vascular resistance (SVR). The increase in SVR is manifested as hypertension[22]. The mean arterial pressure rises and the cardiac output falls.
The rise in the IAP and decrease in the cardiac output are directly proportional to each other. The cardiac index may be reduced by as much as 50%, due to the combination of reverse Trendelenburg position, anaesthesia and peritoneal insufflation (increase in IAP)[23].
The hypertensive response should not be corrected with opioids or inhalation agents since they will delay recovery, but should be managed pharmacologically with a β blocker (esmolol or metoprolol) or an α agonist (dexmedetomidine)[24].
The anaesthesiologist must be aware that inadvertent right endobronchial intubation and consequent hypoxemia is a distinct possibility during laparoscopic surgery because of the Trendelenburg position, which with the raised IAP, lifts the diaphragm and carina, while the tracheal tube remains fixed at its proximal end[25].
Therefore the position of the tracheal tube should be confirmed intermittently. A rise in intragastric pressure consequent to the increase in IAP may predispose these patients to gastric regurgitation and aspiration. However this complication is usually not observed since the lower oesophageal sphincter tone which increases more than the intragastric pressure during carboperitoneum, increases the barrier pressure[26,27].
A thorough preanesthetic assessment is mandatory and all comorbidities should be optimized in case the procedure needs to be converted to an open one.
Premedication should include a short acting anxiolytic, an H2 receptor antagonist and an analgesic for pre-emptive analgesia[28].
Thromboprophylaxis is essential in the morbidly obese scheduled for bariatric surgery. Low molecular weight heparin, enoxaparin 0.6 mL s/c, dalteparin 5000 IU s/c or fondaparineux 2.5 mg s/c is given 12 h before surgery. Mechanical thromboprophylaxis with sequential compression devices is mandatory[29-31].
General anaesthesia with tracheal intubation and controlled ventilation is recommended. This provides optimal control of CO2, facilitates surgical access and protects against aspiration of gastric contents.
Traditional volume controlled ventilation ensures an adequate tidal volume, but there is an increased risk of high inflation pressures and barotrauma. This is especially seen in bariatric surgery for the morbidly obese. Use of pressure controlled ventilation minimizes peak pressure and improves oxygenation in such situations. Addition of alveolar recruitment maneuvers and titrated levels of PEEP are recommended, since they improve oxygenation, probably by opening the collapsed alveoli and increasing respiratory compliance, but should be used cautiously since PEEP can further compromise cardiac output[32,33].
The use of laryngeal mask airways is controversial since it does not give protection against aspiration of gastric contents beyond 20 cm H2O intrathoracic pressure. However the ProSeal LMA has been found to be as effective as a tracheal tube for pulmonary ventilation in laparoscopic surgery[34,35]. ProSeal has also been found to increase the “safe apnoea time” during difficult mask ventilation in morbidly obese patients during induction of anaesthesia[36].
Maintenance of anaesthesia with desflurane ensures a quick recovery[37]. Benefits of laparoscopic procedures are particularly important for patients with pre-existing cardio-pulmonary disease[38].
Recommendations for laparoscopic surgery in high risk cardiac patients are that induction of anaesthesia and release of carboperitoneum should be slow, smooth and progressive. Gradual positioning helps to maintain the haemodynamic status of the patient. Increased intrathoracic pressure interferes with interpretation of measured central venous and pulmonary artery pressure, and so should be interpreted accordingly. Normocarbia should be achieved by adjusting the minute volume. Use of vasodilators instead of inhalation anaesthetics counteracts the increase in MAP due to increase in SVR[10].
Pregnant women undergoing laparoscopic surgery present several challenges to the anaesthesiologist. Important concerns are the effect of carboperitoneum and increased intra-abdominal pressure on the uteroplacental blood flow and its overall effect on the well being of the foetus. In addition, space constraint, trocar insertion and surgical manipulation are compounding factors.
The gravid uterus pressing on the diaphragm leads to further decreases in FRC, increases in V/Q mismatch and an increase in the arterio-alveolar gradient. The SAGES guidelines for laparoscopic surgery during pregnancy should be followed[39]. These include placing the patient in left lateral decubitus position, using open Hasson’s technique for gaining access to the abdominal cavity, maintaining the IAP as low as possible and continuously monitoring the maternal EtCO2.
Fast-track laparoscopic bariatric surgery (Figure 2) is practiced in some centres due to advances in anaesthesia and surgical techniques[40]. While propofol is the induction agent of choice, dexmedetomidine (an α2 adrenoreceptor agonist which possesses hypnotic, sedative, anxiolytic, sympatholytic and analgesic properties) decreases intraoperative fentanyl use, antiemetic therapy and length of stay in the post anaesthesia care unit[41].
Target control infusions of remifentanil and propofol, maintenance with BIS titrated desflurane, multimodal analgesia and antiemetic prophylaxis result in a successful and safe “short stay” bariatric surgery programme[42].
Pheochromocytoma excision is a challenge to the surgeon and the anaesthesiologist and laparoscopic excision adds further trespasses of CO2 insufflation and raised IAP. Low IAP (< 10 mmHg) along with slow insufflation of CO2 and gradual positioning of the patient has been found to produce minimal changes in catecholamine levels and haemodynamics and is therefore recommended[43].
Thoracoscopy too has been practised since 1912. Initially these procedures were conducted with local anaesthetic injected at the point of entry, or under intercostal block. This often resulted in hypoxia and hypercarbia due to the pneumothorax[44].
Thoracoscopic surgery is now traditionally done under general anaesthesia and one lung ventilation. As thoracoscopic surgery can lead to capnothorax, slow insufflation of CO2 in the pleural cavity to produce capnothorax should be done since fast insufflation may result in major haemodynamic compromise[3].
Robotic thoracoscopic surgeries, including thymectomy, lobectomy and coronary revascularization procedures are being done regularly in some centres. Similar precautions as in other robotic surgeries should be practised.
The anaesthesiologist has a major role in the success of the robotic programme. To provide safe patient care, the anaesthesiologist has to understand the impact of robotically assisted laparoscopic surgery on anaesthetic management.
In the “docked” position, the robotic arms of the bulky robot have hardly any natural elasticity. It is therefore paramount that the patient remains completely immobile with no movement whatsoever or else tearing can occur at the port sites. A continuous infusion of neuromuscular blocking drug is indispensable[2]. Reversal of neuromuscular block should be done only after the robotic arms have been detached from the patient at the close of surgery[11]. Since the robot is so bulky, an emergency drill for removal of the robotic cart should be practised, in case the need arises.
Mandatory monitoring includes noninvasive blood pressure, pulse oximetry, capnography and ECG. Peak and plateau airway pressure can be measured from the anaesthesia machine. An IBP is indicated in high risk patients undergoing major surgery[6].
CVP is misleading since the raised IAP and airway pressure interfere with pressure based indices of preload. Pulse pressure variation and stroke volume variation may be measured by lithium dilution cardiac output monitor or vigileo monitor[45].
Common complaints in the postoperative period are postoperative nausea, vomiting and pain - mainly abdominal and shoulder[46]. Several mechanisms have been implicated including residual CO2 in the abdominal cavity[19], neuropraxia due to stretching of the phrenic nerve and acidic milieu due to H2CO3.
Multimodal analgesia with Non Steroidal Anti Inflammatory Drugs, short acting opioids, dexmedetomidine and local infiltration of local anaesthetics provide superior pain relief with reduced postoperative nausea and vomiting (PONV) and early discharge[46].
PONV can be quite problematic after laparoscopic surgery. Some of the causative factors implicated are peritoneal insufflation and irritation, bowel manipulation and pelvic surgery[47]. A multimodal technique including propofol, NSAIDs and antiemetics is essential for reducing the incidence of PONV[48].
The anaesthesiologist should be aware of the complications which are likely to occur during laparoscopic procedures so that he can diagnose and manage them as soon as possible[22].
Accidental misplacement of the Veress needle may result in subcutaneous emphysema. It resolves by itself after a couple of hours of mechanical ventilation.
Pneumothorax, pneumomediastinum and pneumopericardium may occur. Treatment is usually conservative[8].
Venous air embolism is the most rare but lethal complication in case of an accidental entry of gas into a blood vessel. It has to be managed on a war footing[22].
Rhabdomyolysis is a potential threat after bariatric surgery. Damage of muscle sarcolemma with leakage of intracellular contents into the circulation leads to renal failure[49]. Severity ranges from mild, elevated muscle enzyme levels to mortality.
As robotic surgery finds new applications, perhaps the foreseeable future will have telerobotic surgery reaching far-flung rural areas. In such situations it will be important and crucial to ensure that the anaesthesiologist who is present in the remote site knows all the aspects of laparoscopic and robotic surgery.
Our daily lives have been affected by mobile technology. In a similar manner, mobile technology may also be implemented in mobile or digital health care, including surgery in the near future.
Natural orifice transluminal endoscopic surgery is an emerging field in which the peritoneal cavity is accessed via a hollow viscous to perform diagnostic and therapeutic procedures. Since these procedures are in an investigational stage, anaesthetic considerations will need to be modified accordingly[50].
Laparoscopy is the gold standard for cholecystectomy. Although laparoscopy is labelled “minimally invasive”, the anaesthesiologist and surgeon must be thoroughly cognizant of the unique important pathophysiological changes that take place during laparoscopic procedures and be able to manage any intraoperative complications that may result from these changes[13]. With robotic assisted laparoscopic surgeries gaining popularity, anaesthesiologists must be aware of the specific requirements of these procedures and adjust their anaesthesia practice accordingly to provide safe patient care.
P- Reviewer: Aly EH, Kim DK, Zheng X S- Editor: Gou SX L- Editor: O’Neill M E- Editor: Liu XM
| 1. | Litynski GS. Highlights in the history of laparoscopy. Frankfurt/Main: Barbara Bernert Verlag 1996; . |
| 2. | Irvine M, Patil V. Anaesthesia for robot-assisted laparoscopic surgery. Contin Educ Anaesth Crit Care Pain. 2009;9:125-129. [RCA] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 3. | Sullivan MJ, Frost EA, Lew MW. Anesthetic care of the patient for robotic surgery. Middle East J Anesthesiol. 2008;19:967-982. [PubMed] |
| 4. | Conacher ID, Soomro NA, Rix D. Anaesthesia for laparoscopic urological surgery. Br J Anaesth. 2004;93:859-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 5. | Phong SV, Koh LK. Anaesthesia for robotic-assisted radical prostatectomy: considerations for laparoscopy in the Trendelenburg position. Anaesth Intensive Care. 2007;35:281-285. [PubMed] |
| 6. | Hayden P, Cowman S. Anaesthesia for laparoscopic surgery. Contin Educ Anaesth Crit Care Pain. 2011;11:177-180. [RCA] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 7. | Marco AP, Yeo CJ, Rock P. Anesthesia for a patient undergoing laparoscopic cholecystectomy. Anesthesiology. 1990;73:1268-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 8. | Sood J, Kumra VP. Anaesthesia for Laparoscopic surgery. Indian J Surg. 2003;65:232-240. |
| 9. | Menes T, Spivak H. Laparoscopy: searching for the proper insufflation gas. Surg Endosc. 2000;14:1050-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 71] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Sood J, Jain AK; Anaesthesia in Laparoscopic Surgery. Jaypee Brothers Medical Publishers, 200. . |
| 11. | Magno R, Medegård A, Bengtsson R, Tronstad SE. Acid-base balance during laparoscopy. The effects of intraperitoneal insufflation of carbon dioxide and nitrous oxide on acid-base balance during controlled ventilation. Acta Obstet Gynecol Scand. 1979;58:81-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Wahba RW, Mamazza J. Ventilatory requirements during laparoscopic cholecystectomy. Can J Anaesth. 1993;40:206-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 56] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 13. | Wittgen CM, Andrus CH, Fitzgerald SD, Baudendistel LJ, Dahms TE, Kaminski DL. Analysis of the hemodynamic and ventilatory effects of laparoscopic cholecystectomy. Arch Surg. 1991;126:997-1000; discussion 1000-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 240] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 14. | Fitzgerald SD, Andrus CH, Baudendistel LJ, Dahms TE, Kaminski DL. Hypercarbia during carbon dioxide pneumoperitoneum. Am J Surg. 1992;163:186-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 84] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 15. | Marshall RL, Jebson PJ, Davie IT, Scott DB. Circulatory effects of carbon dioxide insufflation of the peritoneal cavity for laparoscopy. Br J Anaesth. 1972;44:680-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 80] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Rasmussen JP, Dauchot PJ, DePalma RG, Sorensen B, Regula G, Anton AH, Gravenstein JS. Cardiac function and hypercarbia. Arch Surg. 1978;113:1196-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 80] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 17. | Sumpf E, Crozier TA, Ahrens D, Bräuer A, Neufang T, Braun U. Carbon dioxide absorption during extraperitoneal and transperitoneal endoscopic hernioplasty. Anesth Analg. 2000;91:589-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 18. | Stolzenburg JU, Settman MT, Liatsikos EN (Eds). In: Endoscopic Extraperitoneal Radical Prostatectomy: Laparoscopic and Robot-Assisted Surgery. Chapter 7: Springer 2007; 66-114. |
| 19. | Phelps P, Cakmakkaya OS, Apfel CC, Radke OC. A simple clinical maneuver to reduce laparoscopy-induced shoulder pain: a randomized controlled trial. Obstet Gynecol. 2008;111:1155-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 92] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Dexter SP, Vucevic M, Gibson J, McMahon MJ. Hemodynamic consequences of high- and low-pressure capnoperitoneum during laparoscopic cholecystectomy. Surg Endosc. 1999;13:376-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 108] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 21. | Motew M, Ivankovich AD, Bieniarz J, Albrecht RF, Zahed B, Scommegna A. Cardiovascular effects and acid-base and blood gas changes during laparoscopy. Am J Obstet Gynecol. 1973;115:1002-1012. [PubMed] |
| 22. | Gerges FJ, Kanazi GE, Jabbour-Khoury SI. Anesthesia for laparoscopy: a review. J Clin Anesth. 2006;18:67-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 131] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 23. | Joris JL, Noirot DP, Legrand MJ, Jacquet NJ, Lamy ML. Hemodynamic changes during laparoscopic cholecystectomy. Anesth Analg. 1993;76:1067-1071. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 284] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 24. | Joshi GP. Ambulatory surgery in the adult patient with morbid obesity and/or sleep apnea syndrome. ASA Refresher Courses in Anesthesiology. 2012;40:80-86. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 25. | Wilcox S, Vandam LD. Alas, poor Trendelenburg and his position! A critique of its uses and effectiveness. Anesth Analg. 1988;67:574-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 62] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Heijke SA, Smith G, Key A. The effect of the Trendelenburg position on lower oesophageal sphincter tone. Anaesthesia. 1991;46:185-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Jones MJ, Mitchell RW, Hindocha N. Effect of increased intra-abdominal pressure during laparoscopy on the lower esophageal sphincter. Anesth Analg. 1989;68:63-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 28. | Joris JL. Anaesthesia for laparoscopic surgery. Miller RD (ed) In: Text Book of Anaesthesia. 7th ed. Churchill Livingstone. Elsevier Health Sciences. 2009;2185-2202. |
| 29. | Magee CJ, Barry J, Javed S, Macadam R, Kerrigan D. Extended thromboprophylaxis reduces incidence of postoperative venous thromboembolism in laparoscopic bariatric surgery. Surg Obes Relat Dis. 2010;6:322-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 30. | Zachari E, Sioka E, Tzovaras G, Zacharoulis D. Venous thromboembolism in bariatric surgery. Dr. Cobanoglu U (Ed) In: Intech 2012; 67-74. |
| 31. | Raeder J. Bariatric surgery - Anesthesiologic Concerns. Dr. Huang CK (Ed), In: Intech 2012; 144-146. |
| 32. | Almarakbi WA, Fawzi HM, Alhashemi JA. Effects of four intraoperative ventilatory strategies on respiratory compliance and gas exchange during laparoscopic gastric banding in obese patients. Br J Anaesth. 2009;102:862-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 84] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Talab HF, Zabani IA, Abdelrahman HS, Bukhari WL, Mamoun I, Ashour MA, Sadeq BB, El Sayed SI. Intraoperative ventilatory strategies for prevention of pulmonary atelectasis in obese patients undergoing laparoscopic bariatric surgery. Anesth Analg. 2009;109:1511-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 147] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 34. | Sharma B, Sood J, Sahai C, Kumra VP. Efficacy and Safety Performance of ProSeal™ Laryngeal Mask Airway in Laparoscopic Surgery. Experience of 1000 cases. Indian J Anaesth. 2008;52:288-296. |
| 35. | Lim Y, Goel S, Brimacombe JR. The ProSeal laryngeal mask airway is an effective alternative to laryngoscope-guided tracheal intubation for gynaecological laparoscopy. Anaesth Intensive Care. 2007;35:52-56. [PubMed] |
| 36. | Sinha A, Jayaraman L, Punhani D, Panigrahi B. ProSeal laryngeal mask airway improves oxygenation when used as a conduit prior to laryngoscope guided intubation in bariatric patients. Indian J Anaesth. 2013;57:25-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 37. | McKay RE, Malhotra A, Cakmakkaya OS, Hall KT, McKay WR, Apfel CC. Effect of increased body mass index and anaesthetic duration on recovery of protective airway reflexes after sevoflurane vs desflurane. Br J Anaesth. 2010;104:175-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 38. | Safran D, Sgambati S, Orlando R. Laparoscopy in high-risk cardiac patients. Surg Gynecol Obstet. 1993;176:548-554. [PubMed] |
| 39. | Guidelines for laparoscopic surgery during pregnancy. Surg Endosc. 1998;12:189-190. [PubMed] |
| 40. | Bamgbade OA, Adeogun BO, Abbas K. Fast-track laparoscopic gastric bypass surgery: outcomes and lessons from a bariatric surgery service in the United Kingdom. Obes Surg. 2012;22:398-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 41. | Tufanogullari B, White PF, Peixoto MP, Kianpour D, Lacour T, Griffin J, Skrivanek G, Macaluso A, Shah M, Provost DA. Dexmedetomidine infusion during laparoscopic bariatric surgery: the effect on recovery outcome variables. Anesth Analg. 2008;106:1741-1748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 207] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 42. | Bergland A, Gislason H, Raeder J. Fast-track surgery for bariatric laparoscopic gastric bypass with focus on anaesthesia and peri-operative care. Experience with 500 cases. Acta Anaesthesiol Scand. 2008;52:1394-1399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 62] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 43. | Sood J, Jayaraman L, Kumra VP, Chowbey PK. Laparoscopic approach to pheochromocytoma: is a lower intraabdominal pressure helpful? Anesth Analg. 2006;102:637-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Conacher ID. Anesthesia for thoracoscopic surgery. J Minim Access Surg. 2007;3:127-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 45. | Koliopanos A, Zografos G, Skiathitis S, Stithos D, Voukena V, Karampinis A, Papastratis G. Esophageal Doppler (ODM II) improves intraoperative hemodynamic monitoring during laparoscopic surgery. Surg Laparosc Endosc Percutan Tech. 2005;15:332-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 46. | Michaloliakou C, Chung F, Sharma S. Preoperative multimodal analgesia facilitates recovery after ambulatory laparoscopic cholecystectomy. Anesth Analg. 1996;82:44-51. [PubMed] |
| 47. | Malins AF, Field JM, Nesling PM, Cooper GM. Nausea and vomiting after gynaecological laparoscopy: comparison of premedication with oral ondansetron, metoclopramide and placebo. Br J Anaesth. 1994;72:231-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 48. | Kapur PA. The big “little problem”. Anesth Analg. 1991;73:243-245. [PubMed] |
| 49. | de Menezes Ettinger JE, dos Santos Filho PV, Azaro E, Melo CA, Fahel E, Batista PB. Prevention of rhabdomyolysis in bariatric surgery. Obes Surg. 2005;15:874-879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 36] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 50. | Al-Haddad M, McKenna D, Ko J, Sherman S, Selzer DJ, Mattar SG, Imperiale TF, Rex DK, Nakeeb A, Jeong SM. Deep sedation in natural orifice transluminal endoscopic surgery (NOTES): a comparative study with dogs. Surg Endosc. 2012;26:3163-3173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |